There are three types of cells that are usually involved in skin cancer:
- Squamous cells
- Basal cells
- Melanocytes
What is Skin Cancer?
In the United States, skin cancer is the most commonly diagnosed cancer. It is also one of the most preventable cancers in non-hereditary cases, as a major risk factor is prolonged exposure to ultraviolet (UV) radiation. Symptoms often include a growth or lump on the skin, and may vary.
There are three types of cells that are usually involved in skin cancer:
- Squamous cells
- Basal cells
- Melanocytes
On a case-by-case basis, the particular type of cells in the skin that are affected by cancer is what classifies basal cell carcinoma, squamous cell carcinoma, or melanoma. The most serious type of skin cancer is melanoma, due to its tendency to spread to other organs. Skin cancer is a very treatable disease if detected early and treated by a dermatologist or surgical oncologist. Skin cancer is a subtype, or speciality, inside the general study of dermatology/oncology and carcinogenesis, cancer cell biology, cancer diagnosis, and cancer therapy.
Physician type:
Dermatologist / Surgical oncologist
ANZSRC Categories:
1112 Oncology and Carcinogenesis
RCDC Category: Skin Cancer
Keywords: skin, cancer, squamous, basal, melanoma, melanocytes, resistance, circulating, factor, derived, prognostic, metastasis
Table of Contents
Research Papers
Damage-associated molecular pattern (DAMP) activation in melanoma: investigation of the immunogenic activity of 15-deoxy, Δ12,14 prostamide J2
DOI: 10.18632/oncotarget.27856
Ahmed Elhassanny, Rene Escobedo, Daniel Ladin, Colin Burns and Rukiyah Van Dross _
Metastatic melanoma is the most deadly skin neoplasm in the United States. Outcomes for this lethal disease have improved dramatically due to the use of both targeted and immunostimulatory drugs. Immunogenic cell death (ICD) has emerged as another approach for initiating antitumor immunity. ICD is triggered by tumor cells that display damage-associated molecular patterns (DAMPs). These DAMP molecules recruit and activate dendritic cells (DCs) that present tumor-specific antigens to T cells which eliminate neoplastic cells. Interestingly, the expression of DAMP molecules occurs in an endoplasmic reticulum (ER) stress-dependent manner. We have previously shown that ER stress was required for the cytotoxic activity of the endocannabinoid metabolite, 15-deoxy, Δ12,14 prostamide J2 (15dPMJ2). As such, the current study investigates whether 15dPMJ2 induces DAMP signaling in melanoma. In B16F10 cells, 15dPMJ2 caused a significant increase in the cell surface expression of calreticulin (CRT), the release of ATP and the secretion of high-mobility group box 1 (HMGB1), three molecules that serve as surrogate markers of ICD. 15dPMJ2 also stimulated the cell surface expression of the DAMP molecules, heat shock protein 70 (Hsp70) and Hsp90. In addition, the display of CRT and ATP was increased by 15dPMJ2 to a greater extent in tumorigenic compared to non-tumorigenic melanocytes. Consistent with this finding, the activation of bone marrow-derived DCs was upregulated in co-cultures with 15dPMJ2-treated tumor compared to non-tumor melanocytes. Moreover, 15dPMJ2-mediated DAMP exposure and DC activation required the electrophilic cyclopentenone double bond within the structure of 15dPMJ2 and the ER stress pathway. These results demonstrate that 15dPMJ2 is a tumor-selective inducer of DAMP signaling in melanoma.
Research Papers
Downregulation of SOX2 by inhibition of Usp9X induces apoptosis in melanoma
DOI: 10.18632/oncotarget.27869
Harish Potu, Malathi Kandarpa, Luke F. Peterson, Alison Durham, Nicholas J. Donato and Moshe Talpaz _
Melanoma tumors driven by BRAF mutations often do not respond to BRAF/MEK/ERK pathway inhibitors currently used in treatment. One documented mechanism of resistance is upregulation of SOX2, a transcription factor that is essential for tumor growth and expansion, particularly in melanoma tumors with BRAF mutations. Targeting transcription factors pharmacologically has been elusive for drug developers, limiting treatment options. Here we show that ubiquitin-specific peptidase 9, X-linked (Usp9x), a deubiquitinase (DUB) enzyme controls SOX2 levels in melanoma. Usp9x knockdown in melanoma increased SOX2 ubiquitination, leading to its depletion, and enhanced apoptotic effects of BRAF inhibitor and MEK inhibitors. Primary metastatic melanoma samples demonstrated moderately elevated Usp9x and SOX2 protein expression compared to tumors without metastatic potential. Usp9x knockdown, as well as inhibition with DUB inhibitor, G9, blocked SOX2 expression, suppressed in vitro colony growth, and induced apoptosis of BRAF-mutant melanoma cells. Combined treatment with Usp9x and mutant BRAF inhibitors fully suppressed melanoma growth in vivo. Our data demonstrate a novel mechanism for targeting the transcription factor SOX2, leveraging Usp9x inhibition. Thus, development of DUB inhibitors may add to the limited repertoire of current melanoma treatments.
Reviews
Using drug scheduling to manage adverse events associated with hedgehog pathway inhibitors for basal cell carcinoma
DOI: 10.18632/oncotarget.28145
John T. Lear _, Reinhard Dummer and Alexander Guminski
Basal cell carcinoma (BCC) is the most common malignancy and form of skin cancer worldwide; advanced BCC, either as locally advanced BCC (laBCC) or metastatic BCC (mBCC), can cause substantial tissue invasion and morbidity. Until the recent availability of the hedgehog pathway inhibitors (HHIs) sonidegib and vismodegib, treatment options for advanced BCC were limited. These agents demonstrate efficacy in patients with laBCC and mBCC; however, the adverse events (AEs) associated with these agents can lead to treatment interruption or discontinuation and reduced quality of life, all of which significantly impact long-term adherence to therapy, which might affect clinical outcome. Given that most AEs are class-related effects, switching HHIs does not appear to lead to a significantly different AE profile, underscoring the importance of maintaining patients on their first HHI. Interrupting treatment of sonidegib and vismodegib does not appear to undermine the efficacy of these agents and is therefore a practical option to manage AEs in order to maintain continued treatment and disease control.
Research Papers
Mutually exclusive lymphangiogenesis or perineural infiltration in human skin squamous-cell carcinoma
DOI: 10.18632/oncotarget.27915
Julien Schaller, Hélène Maby-El Hajjami, Sylvie Rusakiewicz, Kalliopi Ioannidou, Nathalie Piazzon, Alexandra Miles, Déla Golshayan, Olivier Gaide, Daniel Hohl, Daniel E. Speiser and Karin Schaeuble _
Although tumor-associated lymphangiogenesis correlates with metastasis and poor prognosis in several cancers, it also supports T cell infiltration into the tumor and predicts favorable outcome to immunotherapy. The role of lymphatic vessels in skin squamous-cell carcinoma (sSCC), the second most common form of skin cancer, remains mostly unknown. Although anti-PD-1 therapy is beneficial for some patients with advanced sSCC, a greater understanding of disease mechanisms is still needed to develop better therapies.
Using quantitative multiplex immunohistochemistry, we analyzed sSCC sections from 36 patients. CD8+ T cell infiltration showed great differences between patients, whereby these cells were mainly excluded from the tumor mass. Similar to our data in melanoma, sSCC with high density of lymphatic endothelial cells showed increased CD8+ T cell density in tumor areas. An entirely new observation is that sSCC with perineural infiltration but without metastasis was characterized by low lymphatic endothelial cell density. Since both, metastasis and perineural infiltration are known to affect tumor progression and patients’ prognosis, it is important to identify the molecular drivers, opening future options for therapeutic targeting. Our data suggest that the mechanisms underlying perineural infiltration may be linked with the biology of lymphatic vessels and thus stroma.
Case Reports
Immunotherapy in Xeroderma Pigmentosum: a case of advanced cutaneous squamous cell carcinoma treated with cemiplimab and a literature review
DOI: 10.18632/oncotarget.27966
Marco Rubatto _, Martina Merli, Gianluca Avallone, Andrea Agostini, Luca Mastorino, Virginia Caliendo, Amelia Barcellini, Viviana Vitolo, Francesca Valvo, Maria Teresa Fierro, Simone Ribero and Pietro Quaglino
Xeroderma Pigmentosum (XP) is a rare genetic disorder with a poor prognosis due to high photosensitivity in affected patients.
Herein, we describe the first case of the use of cemiplimab in a patient with XP, a 19-year-old girl presented with locally advanced squamous cell carcinoma of the right periorbital and nasal region. This treatment has been undertaken after a cycle of proton beam radiotherapy.
Besides, it is reported a description of the few cases in the literature describing the effectiveness of immunotherapy on skin cancers in XP-patients.
This case is in line with those reported, underlining how anti-PD1 monoclonal antibodies may be a promising treatment in this genodermatosis.
Research Papers
A diagnostic autoantibody signature for primary cutaneous melanoma
DOI: 10.18632/oncotarget.25669
Pauline Zaenker _, Johnny Lo, Robert Pearce, Phillip Cantwell, Lester Cowell, Mark Lee, Christopher Quirk, Henry Law, Elin Gray and Mel Ziman
Melanoma is an aggressive form of skin cancer that is curable by surgical excision in the majority of cases, if detected at an early stage. To improve early stage melanoma detection, the development of a highly sensitive diagnostic test is of utmost importance. Here we aimed to identify antibodies to a panel of tumour associated antigens that can differentiate primary melanoma patients and healthy individuals. A total of 245 sera from primary melanoma patients and healthy volunteers were screened against a high-throughput microarray platform containing 1627 functional proteins. Following rigorous statistical analysis, we identified a combination of 10 autoantibody biomarkers that, as a panel, displays a sensitivity of 79%, specificity of 84% and an AUC of 0.828 for primary melanoma detection. This melanoma autoantibody signature may prove valuable for the development of a diagnostic blood test for routine population screening that, when used in conjunction with current melanoma diagnostic techniques, could improve the early diagnosis of this malignancy and ultimately decrease the mortality rate of patients.
Research Papers
Improved therapeutic efficacy of unmodified anti-tumor antibodies by immune checkpoint blockade and kinase targeted therapy in mouse models of melanoma
DOI: 10.18632/oncotarget.27868
Rolando Pérez-Lorenzo, Stephanie O. Erjavec, Angela M. Christiano _ and Raphael Clynes
The use of specific anti-tumor antibodies has transformed the solid cancer therapeutics landscape with the relative successes of therapies such as anti-HER2 in breast cancer, and anti-EGFR in HNSCC and colorectal cancer. However, these therapies result in toxicity and the emergence of resistant tumors. Here, we showed that removing immune suppression and enhancing stimulatory signals increased the anti-tumor activity of unmodified TA99 antibodies (anti-TYRP1) with a significant reduction of growth of solid tumors and lung metastases in mouse models of melanoma. Immune checkpoint blockade enhanced the efficacy of TA99, which was associated with greater CD8+/Foxp3+, NK1.1+ and dendritic cell infiltrates, suggestive of an increased anti-tumor innate and adaptive immune responses. Further, MEK inhibition in melanoma cell lines increased the expression of melanosomal antigens in vitro, and combining TA99 and MEKi in vivo resulted in enhanced tumor control. Moreover, we found an improved therapeutic effect when YUMM tumor-bearing mice were treated with TA99 combined with MEKi and immune checkpoint blockade (anti-PD1 and anti-CTLA4). Our findings suggest that MEKi induced an increased expression of tumor-associated antigens, which in combination with anti-tumor antibodies, generated a robust adaptive anti-tumor response that was sustained by immune checkpoint inhibition therapy. We postulate that combining anti-tumor antibodies with standard-of-care strategies such as immune checkpoint blockade or targeted therapy, will improve therapeutic outcomes in cancer.
Research Papers
Mutation profile of primary subungual melanomas in Caucasians
DOI: 10.18632/oncotarget.27642
Aneta Borkowska _, Anna Szumera-Ciećkiewicz, Mateusz Spałek, Paweł Teterycz, Anna Czarnecka, Artur Kowalik and Piotr Rutkowski
Background: Specific genomic profile of cutaneous melanomas is related to UVR exposure, which exerts biological and therapeutic impact. Subungual melanoma (SUM) is an exceedingly rare disease; therefore, it is not well characterized. SUM pathogenesis is not related to UVR induced DNA damage and expected to differ from other melanoma subtypes. Our study aimed to define the mutation profile of SUM in Caucasians.
Materials and Methods: Next-generation sequencing-based genomic analysis was used to identify frequently mutated loci in 50 cancer-related genes in 31 SUM primary tumors.
Results: The most abundant mutations in SUM were found in KIT – in 13% of cases and NRAS – also in 13%, while BRAF - only in 3% of cases.
Conclusions: Our findings confirmed a high frequency of KIT and NRAS mutations in SUM, as well as a low incidence of BRAF mutations. We reported novel KRAS, CTNNB1, TP53, ERBB2, and SMAD4 mutations in SUM. Our findings provide new insights into the molecular pathogenesis of SUM.
Research Papers
Intralesional rose bengal in melanoma elicits tumor immunity via activation of dendritic cells by the release of high mobility group box 1
Hao Liu, Pasquale Patrick Innamarato, Krithika Kodumudi, Amy Weber, Satoshi Nemoto, John L. Robinson, Georgina Crago, Timothy McCardle, Erica Royster, Amod A. Sarnaik and Shari Pilon-Thomas _
Intralesional (IL) therapy is under investigation to treat dermal and subcutaneous metastatic cancer. Rose Bengal (RB) is a staining agent that was originally used by ophthalmologists and in liver function studies. IL injection of RB has been shown to induce regression of injected and uninjected tumors in murine models and clinical trials. In this study, we have shown a mechanism of tumor-specific immune response induced by IL RB. In melanoma-bearing mice, IL RB induced regression of injected tumor and inhibited the growth of bystander lesions mediated by CD8+ T cells. IL RB resulted in necrosis of tumor cells and the release of High Mobility Group Box 1 (HMGB1), with increased dendritic cell (DC) infiltration into draining lymph nodes and the activation of tumor-specific T cells. Treatment of DC with tumor supernatants increased the ability of DCs to stimulate T cell proliferation, and blockade of HMGB1 in the supernatants suppressed DC activity. Additionally, increased HMGB1 levels were measured in the sera of melanoma patients treated with IL RB. These results support the role of IL RB to activate dendritic cells at the site of tumor necrosis for the induction of a systemic anti-tumor immune response.
Research Papers
Epigenetic reprogramming and aberrant expression of PRAME are associated with increased metastatic risk in Class 1 and Class 2 uveal melanomas
DOI: 10.18632/oncotarget.10962
Matthew G. Field, Michael A. Durante, Christina L. Decatur, Bercin Tarlan, Kristen M. Oelschlager, John F. Stone, Jeffim Kuznetsov, Anne M. Bowcock, Stefan Kurtenbach and J. William Harbour _
Background: We previously identified PRAME as a biomarker for metastatic risk in Class 1 uveal melanomas. In this study, we sought to define a threshold value for positive PRAME expression (PRAME+) in a large dataset, identify factors associated with PRAME expression, evaluate the prognostic value of PRAME in Class 2 uveal melanomas, and determine whether PRAME expression is associated with aberrant hypomethylation of the PRAME promoter.
Results: Among 678 samples analyzed by qPCR, 498 (73.5%) were PRAME- and 180 (26.5%) were PRAME+. Class 1 tumors were more likely to be PRAME-, whereas Class 2 tumors were more likely to be PRAME+ (P < 0.0001). PRAME expression was associated with shorter time to metastasis and melanoma specific mortality in Class 2 tumors (P = 0.01 and P = 0.02, respectively). In Class 1 tumors, PRAME expression was directly associated with SF3B1 mutations (P < 0.0001) and inversely associated with EIF1AX mutations (P = 0.004). PRAME expression was strongly associated with hypomethylation at 12 CpG sites near the PRAME promoter.
Materials and methods: Analyses included PRAME mRNA expression, Class 1 versus Class 2 status, chromosomal copy number, mutation status of BAP1, EIF1AX, GNA11, GNAQ and SF3B1, and genomic DNA methylation status. Analyses were performed on 555 de-identified samples from Castle Biosciences, 123 samples from our center, and 80 samples from the TCGA.
Conclusions: PRAME is aberrantly hypomethylated and activated in Class 1 and Class 2 uveal melanomas and is associated with increased metastatic risk in both classes. Since PRAME has been successfully targeted for immunotherapy, it may prove to be a companion prognostic biomarker.
Research Papers
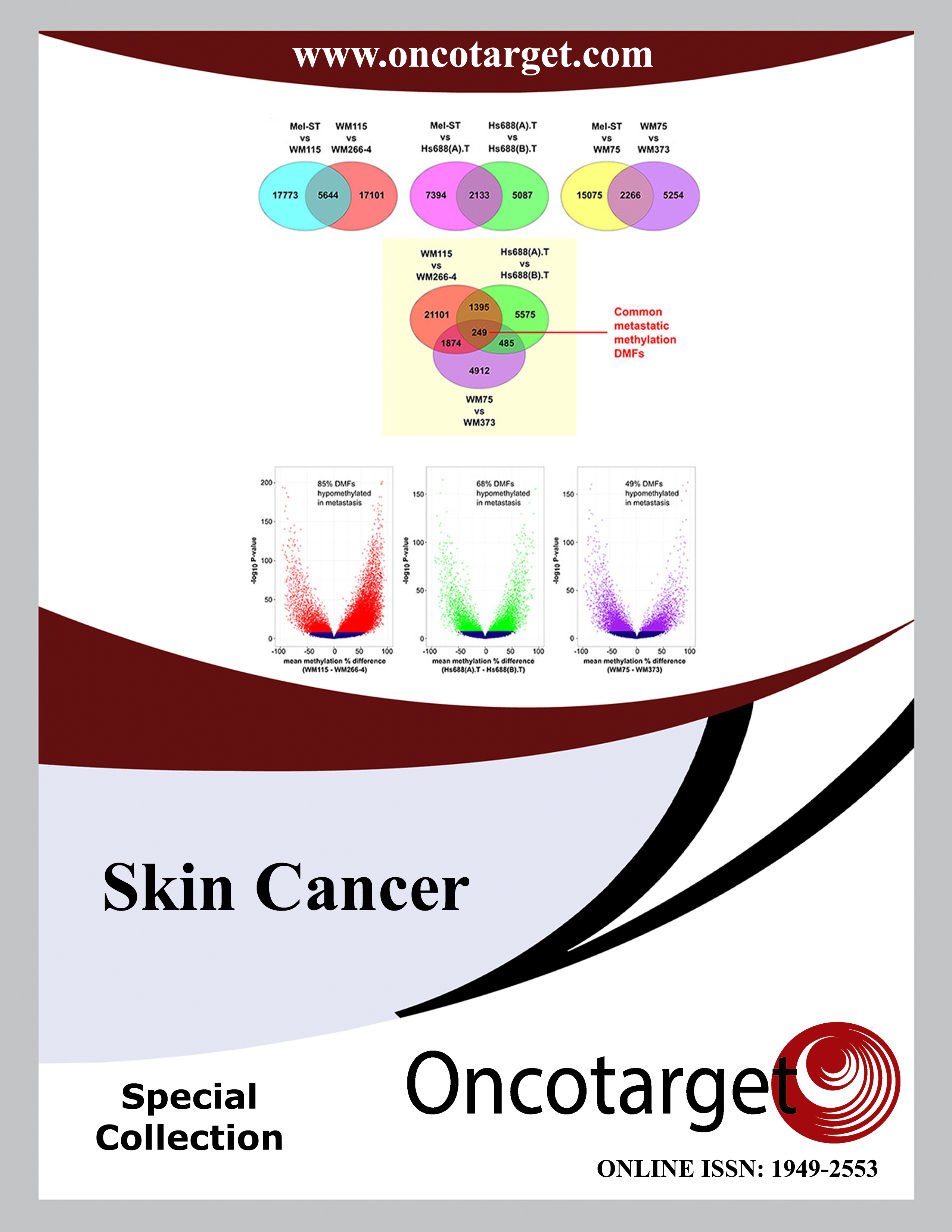
Genome-wide methylation sequencing of paired primary and metastatic cell lines identifies common DNA methylation changes and a role for EBF3 as a candidate epigenetic driver of melanoma metastasis
DOI: 10.18632/oncotarget.14042
Aniruddha Chatterjee, Peter A Stockwell, Antonio Ahn, Euan J Rodger, Anna L Leichter and Michael R Eccles _
Epigenetic alterations are increasingly implicated in metastasis, whereas very few genetic mutations have been identified as authentic drivers of cancer metastasis. Yet, to date, few studies have identified metastasis-related epigenetic drivers, in part because a framework for identifying driver epigenetic changes in metastasis has not been established. Using reduced representation bisulfite sequencing (RRBS), we mapped genome-wide DNA methylation patterns in three cutaneous primary and metastatic melanoma cell line pairs to identify metastasis-related epigenetic drivers. Globally, metastatic melanoma cell lines were hypomethylated compared to the matched primary melanoma cell lines. Using whole genome RRBS we identified 75 shared (10 hyper- and 65 hypomethylated) differentially methylated fragments (DMFs), which were associated with 68 genes showing significant methylation differences. One gene, Early B Cell Factor 3 (EBF3), exhibited promoter hypermethylation in metastatic cell lines, and was validated with bisulfite sequencing and in two publicly available independent melanoma cohorts (n = 40 and 458 melanomas, respectively). We found that hypermethylation of the EBF3 promoter was associated with increased EBF3 mRNA levels in metastatic melanomas and subsequent inhibition of DNA methylation reduced EBF3 expression. RNAi-mediated knockdown of EBF3 mRNA levels decreased proliferation, migration and invasion in primary and metastatic melanoma cell lines. Overall, we have identified numerous epigenetic changes characterising metastatic melanoma cell lines, including EBF3-induced aggressive phenotypic behaviour with elevated EBF3 expression in metastatic melanoma, suggesting that EBF3 promoter hypermethylation may be a candidate epigenetic driver of metastasis.
Research Papers
HDAC inhibitors enhance the immunotherapy response of melanoma cells
DOI: 10.18632/oncotarget.17950
Laurence Booth, Jane L. Roberts, Andrew Poklepovic, John Kirkwood and Paul Dent _
We focused on the ability of the pan-histone deacetylase (HDAC) inhibitors AR42 and sodium valproate to alter the immunogenicity of melanoma cells. Treatment of melanoma cells with HDAC inhibitors rapidly reduced the expression of multiple HDAC proteins as well as the levels of PD-L1, PD-L2 and ODC, and increased expression of MHCA. In a cell-specific fashion, melanoma isolates released the immunogenic protein HMGB1 into the extracellular environment. Very similar data were obtained in ovarian and H&NSCC PDX isolates, and in established tumor cell lines from the lung and kidney. Knock down of HDAC1, HDAC3, HDAC8 and HDAC10, but not HDAC6, recapitulated the effects of the HDAC inhibitors on the immunotherapy biomarkers. Using B16 mouse melanoma cells we discovered that pre-treatment with AR42 or sodium valproate enhanced the anti-tumor efficacy of an anti-PD-1 antibody and of an anti-CTLA4 antibody. In the B16 model, both AR42 and sodium valproate enhanced the anti-tumor efficacy of the multi-kinase inhibitor pazopanib. In plasma from animals exposed to [HDAC inhibitor + anti-PD-1], but not [HDAC inhibitor + anti-CTLA4], the levels of CCL2, CCL5, CXCL9 and CXCL2 were increased. The cytokine data from HDAC inhibitor plus anti-PD-1 exposed tumors correlated with increased activated T cell, M1 macrophage, neutrophil and NK cell infiltration. Collectively, our data support the use of pan-HDAC inhibitors in combination with kinase inhibitors or with checkpoint inhibitor antibodies as novel melanoma therapeutic strategies.
Research Papers
Capturing the biological impact of CDKN2A and MC1R genes as an early predisposing event in melanoma and non melanoma skin cancer.
María José Escamez, Francisco Garcia-Garcia, Gemma Tell-Marti, Angels Fabra, Lucía Martínez-Santamaría, Celia Badenas, Paula Aguilera, Marta Pevida, Joaquín Dopazo, Marcela del Río and Susana Puig _
Germline mutations in CDKN2A and/or red hair color variants in MC1R genes are associated with an increased susceptibility to develop cutaneous melanoma or non melanoma skin cancer.
We studied the impact of the CDKN2A germinal mutation p.G101W and MC1R variants on gene expression and transcription profiles associated with skin cancer. To this end we set-up primary skin cell co-cultures from siblings of melanoma prone-families that were later analyzed using the expression array approach.
As a result, we found that 1535 transcripts were deregulated in CDKN2A mutated cells, with over-expression of immunity-related genes (HLA-DPB1, CLEC2B, IFI44, IFI44L, IFI27, IFIT1, IFIT2, SP110 and IFNK) and down-regulation of genes playing a role in the Notch signaling pathway. 3570 transcripts were deregulated in MC1R variant carriers. In particular, genes related to oxidative stress and DNA damage pathways were up-regulated as well as genes associated with neurodegenerative diseases such as Parkinson’s, Alzheimer and Huntington.
Finally, we observed that the expression signatures indentified in phenotypically normal cells carrying CDKN2A mutations or MC1R variants are maintained in skin cancer tumors (melanoma and squamous cell carcinoma). These results indicate that transcriptome deregulation represents an early event critical for skin cancer development.
Clinical Research Papers
Evaluation of real-world treatment outcomes in patients with distant metastatic Merkel cell carcinoma following second-line chemotherapy in Europe
DOI: 10.18632/oncotarget.19218
Jürgen C. Becker _, Eva Lorenz, Selma Ugurel, Thomas K. Eigentler, Felix Kiecker, Claudia Pföhler, Ivonne Kellner, Friedegund Meier, Katharina Kähler, Peter Mohr, Carola Berking, Gabriele Haas, Christoph Helwig, Dina Oksen, Dirk Schadendorf, Lisa Mahnke and Murtuza Bharmal
Background and aims: Merkel cell carcinoma (MCC) is a rare, aggressive skin cancer; few treatments exist for patients with advanced disease. Once tumors metastasize to distant sites, patients generally receive chemotherapy, but response duration and progression-free survival (PFS) are typically short. Few studies have assessed the efficacy of second-line chemotherapy for metastatic MCC. Here, we studied outcomes in patients who received ≥ 2 lines of chemotherapy for metastatic MCC.
Materials and Methods: Patients in an MCC-specific registry diagnosed with stage IV MCC between November 1, 2004, and September 15, 2015, and treated with second-line or later chemotherapy were analyzed retrospectively. Patient records, including baseline characteristics, immunocompetent status, and responses to prior chemotherapy, were evaluated. Patients meeting eligibility criteria were followed through December 31, 2015.
Results: Of 29 patients with metastatic MCC and immunocompetent status who had received ≥ 2 lines of chemotherapy, 3 achieved a partial response, for an objective response rate (ORR) of 10.3% (95% CI, 2.2–27.4). In the overall population including patients with immunocompetent and immunocompromised status (n = 34), the ORR was 8.8% (95% CI, 1.9–23.7). The median duration of response was 1.9 months (range, 1.3–2.1 months; 95% CI, 1.3–2.1). In the immunocompetent population, median PFS and overall survival were 3.0 months (95% CI, 2.5–6.0) and 5.3 months (95% CI, 4.3–6.0), respectively.
Conclusions: The low response rates and limited durability confirm previous reports of the ineffectiveness of second-line or later chemotherapy in patients with metastatic MCC and provide a benchmark for assessing clinical benefit of new treatments.
Research Papers: Immunology
BRAF inhibitors stimulate inflammasome activation and interleukin 1 beta production in dendritic cells
DOI: 10.18632/oncotarget.25511
Eva Hajek, Franziska Krebs, Rebekka Bent, Katharina Haas, Antje Bast, Ivo Steinmetz, Andrea Tuettenberg, Stephan Grabbe _ and Matthias Bros
Melanoma is the most dangerous form of skin cancer with a growing incidence over the last decades. Fourty percent of all melanomas harbor a mutation in the signaling adaptor BRAF (V600E) that results in ERK hyperactivity as an oncogenic driver. In these cases, treatment with the BRAFV600E inhibitors Vemurafenib (VEM) or Dabrafenib (DAB) coapplied with the MEK1/2 inhibitors Cobimetinib (COB) or Trametinib (TRA) can result in long-term suppression of tumor growth. Besides direct suppression of ERK activity, these inhibitors have been reported to also modulate tumor immune responses, and exert pro-inflammatory side effects such as fever and rash in some patients. Here we asked for potential effects of BRAFV600E inhibitors on dendritic cells (DC) which are essential for the induction of adaptive anti-tumor responses. Both splenic and bone marrow-derived (BM) mouse dendritic cells (DC) up-regulated costimulator expression (CD80, CD86) in response to DAB but not VEM treatment. Moreover, DAB and to lesser extent VEM enhanced IL-1β (interleukin 1 beta) release by splenic DC, and by LPS-stimulated BMDC. We demonstrate that DAB and VEM activated the NLRC4/Caspase-1 inflammasome. At high concentration, DAB also induced inflammasome activation independent of Caspase-1. TRA and COB elevated MHCII expression on BMDC, and modulated the LPS-induced cytokine pattern. Immunomodulatory activity of DAB and VEM was also observed in human monocyte-derived DC, and DAB induced IL-1β in human primary DC. Altogether, our study shows that BRAFV600E inhibitors upregulate IL-1β release by mouse and human DC which may affect the DC-mediated course of anti-tumor immune responses.
Meta-Analysis
Statin use and non-melanoma skin cancer risk: a meta-analysis of randomized controlled trials and observational studies
DOI: 10.18632/oncotarget.20034
Keming Yang, Andrew Marley, Huilin Tang, Yiqing Song, Jean Y. Tang and Jiali Han _
Background: Existing evidence of the association between statin use and non-melanoma skin cancer (NMSC) risk has been inconsistent.
Objective: To maximize statistical power to synthesize prospective evidence on this relationship.
Materials and Methods: PubMed, EMBASE, Web of Science, Cochrane Central Register of Controlled Trials, and ClinicalTrial.gov were systematically searched up to December 11, 2016. A random-effects meta-analysis was conducted to calculate summary estimates.
Results: Our meta-analysis of 14 randomized controlled trials (RCTs) including 63,157 subjects showed no significant association between statin use and NMSC risk (RR = 1.09, 95%CI = 0.85–1.39). However, meta-analysis of four observational studies including 1,528,215 participants showed significantly increased risk of NMSC among statin users compared to non-users (RR = 1.11, 95%CI = 1.02–1.22). Furthermore, ever using lipophilic statins (RR = 1.14, 95%CI = 1.04–1.24) or lower-potency statins (RR = 1.14, 95%CI = 1.03–1.26), as well as usage of any statin longer than one year (RR = 1.14, 95%CI = 1.09–1.18) were significantly associated with increased NMSC risk based on observational studies.
Conclusions: Evidence from observational studies supported an association between statin use and increased NMSC risk. This finding should be interpreted with caution due to modest number of included studies, possible between-study heterogeneity and inherent limitations of observational studies.
Research Papers
The PP4R1 sub-unit of protein phosphatase PP4 is essential for inhibition of NF-κB by merkel polyomavirus small tumour antigen
DOI: 10.18632/oncotarget.15836
Hussein Abdul-Sada, Marietta Müller, Rajni Mehta, Rachel Toth, J. Simon C. Arthur, Adrian Whitehouse and Andrew Macdonald _
Merkel cell carcinoma (MCC) is a highly aggressive skin cancer with a high metastatic potential. The majority of MCC cases are caused by the Merkel cell polyomavirus (MCPyV), through expression of the virus-encoded tumour antigens. Whilst mechanisms attributing tumour antigen expression to transformation are being uncovered, little is known of the mechanisms by which MCPyV persists in the host. We previously identified the MCPyV small T antigen (tAg) as a novel inhibitor of nuclear factor kappa B (NF-kB) signalling and a modulator of the host anti-viral response. Here we demonstrate that regulation of NF-kB activation involves a previously undocumented interaction between tAg and regulatory sub-unit 1 of protein phosphatase 4 (PP4R1). Formation of a complex with PP4R1 and PP4c is required to bridge MCPyV tAg to the NEMO adaptor protein, allowing deactivation of the NF-kB pathway. Mutations in MCPyV tAg that fail to interact with components of this complex, or siRNA depletion of PP4R1, prevents tAg-mediated inhibition of NF-kB and pro-inflammatory cytokine production. Comparison of tAg binding partners from other human polyomavirus demonstrates that interactions with NEMO and PP4R1 are unique to MCPyV. Collectively, these data identify PP4R1 as a novel target for virus subversion of the host anti-viral response.
Research Papers
Dissecting the role of RNA modification regulatory proteins in melanoma
DOI: 10.18632/oncotarget.26959
Parmanand Malvi, Biao Wang, Shreni Shah and Romi Gupta
Melanoma is the deadliest form of skin cancer. Despite recent advances in medicine and the development of new treatments for melanoma, cures remain elusive as acquired resistance to both targeted and immunotherapies are becoming common. Therefore, more studies are conducted to dissect underlying molecular mechanisms that drive melanoma growth in order to provide better therapeutic option. Here, employing a comprehensive and unbiased analysis of different RNA modification regulatory proteins using various publicly available databases we identify the most relevant RNA modifying proteins that plays crucial role in melanoma development. Our study started with the analysis of various genetic alterations (amplifications, mutations/deletion) as well as RNA overexpression of these RNA modification regulatory proteins in The Cancer Genome Atlas melanoma database. We then analyzed their expression in The Human Protein Atlas data. The result of analysis revealed that only a subset of RNA modification regulatory proteins are overexpressed in >75% of melanoma patient cases as compared to normal skin. However, when examined in Oncomine dataset we found only two genes (METTL4 and DNMT3A) were significantly overexpressed in melanoma samples versus normal skin samples and matched with the results of The Human Protein Atlas data. Therefore, we functionally validated METTL4 and DNMT3A using shRNA-mediated knockdown and found that their knockdown in melanoma cells led to melanoma cells growth inhibition. Collectively, in this study, we investigated the epitranscriptomic landscape of melanoma using various publicly available database and identified DNMT3A and METTL4 as the most relevant potential regulators of melanoma growth.
Reviews
Management of intracranial melanomas in the era of precision medicine
DOI: 10.18632/oncotarget.19223
Grace J. Young, Wenya Linda Bi, Winona W. Wu, Tanner M. Johanns, Gavin P. Dunn and Ian F. Dunn _
Melanoma is the most lethal of skin cancers, in part because of its proclivity for rapid and distant metastasis. It is also potentially the most neurotropic cancer in terms of probability of CNS metastasis from the primary lesion. Despite surgical resection and radiotherapy, prognosis remains guarded for patients with brain metastases. Over the past five years, a new domain of personalized therapy has emerged for advanced melanoma patients with the introduction of BRAF and other MAP kinase pathway inhibitors, immunotherapy, and combinatory therapeutic strategies. By targeting critical cellular signaling pathways and unleashing the adaptive immune response against tumor antigens, a subset of melanoma patients have demonstrated remarkable responses to these treatments. Over time, acquired resistance to these modalities inexorably develops, providing new challenges to overcome. We review the rapidly evolving terrain for intracranial melanoma treatment, address likely and potential mechanisms of resistance, as well as evaluate promising future therapeutic approaches currently under clinical investigation.
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC