INTRODUCTION
The prostate cancer progression has been related to many factors such as inflammation [1] and the so-called angiogenic switch that implies enhanced angiogenesis, characterized by high vascular endothelial growth factor (VEGF) and VEGF receptor (VEGFR) levels [2]. Although significance of inflammation in cancer progression is still a debated issue, some evidences suggest pathways directed to connection between inflammation and cancer evolution [3]. Indeed, the proliferative inflammatory atrophy has been proposed as possible precursor of prostatic intraepithelial neoplasm. Since inflammatory microenvironment releases growth factors (such as VEGF) and cytokines, proangiogenic factors may influence the activation of the vascular endothelial cells and signal transduction in these cells [3]. Angiogenesis is an important step in the development of cancer and is necessary for primary tumor growth, invasiveness, and metastasis. This neovessel formation is stimulated by different proangiogenic factors secreted by cells pertaining to the tumor itself and to its microenvironment [4]. Kinase insert domain receptor (KDR or VEGFR-2) is the principal receptor that promotes the pro-angiogenic action of vascular endothelial growth factor and is recognized as the main target of anti-angiogenic therapies.
The extracellular signalling molecule ATP (eATP) has been shown to mediate various biological functions including synaptic neurotransmission, nociception, smooth muscle contraction, and endocrine secretion [5, 6]. eATP acts via specific P2 receptors, ion-gated channels classified into ionotropic (P2X) and metabotropic (P2Y) subtypes. In the last fifteen years, attention has been paid to the complex relationship between purinergic signal and cancer [5, 7]. Among the various ATP-sensitive receptors, the P2X7 receptor (P2X7R) has attracted our attention as potential mediator of the cellular processes described above, especially in prostatic carcinoma, where an increased P2X7R expression has been reported, irrespective of Gleason grade or patient age [8]. Moreover, Ravenna and colleagues have suggested that an up-regulation of P2X7R in bioptic specimens of prostate cancer might trigger the generation of other pro-inflammatory molecules through NF-kB activation [9].
The gene encoding for P2X7R, located at chromosome position 12q24, is highly polymorphic. Several loss-of-function polymorphisms may alter the normal trafficking and activity of the receptor [10, 11]. The VEGFR-2 gene is located on chromosome 4q11-q12. Several single-nucleotide polymorphisms (SNPs) have been described in the VEGFR-2 gene, some of which able to increase/decrease gene expression itself, circulating levels of soluble VEGFR-2 and the VEGF binding efficiency to the receptor [12]. Many authors have pointed out the unlikeliness that just a SNP can predict the prognosis or the therapeutic response to drugs, mainly due to the complexity of the involved biological systems [4, 13, 14]. Therefore, the prognosis of advanced prostate cancer could depend on many factors, likely interacting with a complex genetic background. Indeed, the effect of a SNP on its corresponding gene may be the result of an interaction between the polymorphisms of other functionally linked genes. This phenomenon is defined as epistasis [15, 16]. Gene–gene interactions are an essential part of gene regulation, signal transduction, biochemical networks, and homeostatic, developmental, and physiological pathways. Indeed, the epistasis phenomenon involves DNA sequence variations, biomolecules and their physical interactions giving rise to a phenotype at a particular time point [17]. Therefore, the current approach of correlating the prostate cancer prognosis to a single SNP may be replaced by a genetic analysis of the interaction between SNPs. The aim of this new approach should be to unveil a genetic profile with a reliable prognostic value. Beretta and colleagues have created and validated a methodology called survival dimensionality reduction (SDR), an innovative approach to detect epistasis in presence of right-censored data, to identify a genetic profile with the ability to predict a better survival of patients [18].
Based on these hypotheses, we conducted a retrospective study to assess the ability of SDR methodology to identify a genetic profile of VEGFR-2 and P2X7R polymorphisms associated to the overall survival (OS) in an unselected population of prostate cancer patients.
RESULTS
One-hundred patients with histological diagnosis of prostate adenocarcinoma were enrolled into the study (Table 1). Ninety-three patients were treated with chemotherapy (45 patients received more than 2 lines of chemotherapy) such as docetaxel, estramustine, mitoxantrone, cyclophosphamide, and vinorelbine. Radiotherapy was administered in 6 patients, whereas 95 patients received androgen deprivation therapy soon after the prostate cancer diagnosis or at the time of the metastatic disease. Thirty-four patients underwent surgery.
Table 1: Patients Characteristics (n = 100)
N° |
% |
|||
Age |
||||
Median |
70.5 |
|||
Range |
48–91 |
|||
ECOG Performance Status |
||||
0 |
71 |
71 |
||
1 |
22 |
22 |
||
2 |
5 |
5 |
||
Not available |
2 |
2 |
||
Metastatic Disease at the Diagnosis |
||||
Yes |
44 |
44 |
||
No |
51 |
51 |
||
Not available |
5 |
5 |
||
Sites of Metastatic Disease at the Diagnosis |
||||
Bone and Nodes |
7 |
7 |
||
Bone |
31 |
31 |
||
Nodes |
6 |
6 |
||
Not available |
5 |
5 |
||
Hormonal Manipulations |
||||
Yes |
95 |
95 |
||
No |
5 |
5 |
||
Surgery |
34 |
34 |
||
Radiotherapy |
6 |
6 |
||
Chemotherapy |
||||
Yes |
93 |
93 |
||
No |
5 |
5 |
||
Not available |
2 |
2 |
||
Number of Lines of Chemotherapy |
||||
1 |
24 |
24 |
||
≥ 2 |
67 |
67 |
||
Not available |
2 |
2 |
||
Serum PSA at the Diagnosis |
||||
Median (ng/ml) |
99 |
|||
Range |
2.36–5307 |
|||
Gleason Score at the Diagnosis |
||||
<7 |
12 |
12 |
||
≥7 |
56 |
56 |
||
Not available |
32 |
32 |
||
In Table 2 the associations of clinical and pathological characteristics with OS are reported. The univariate Cox regression analysis confirmed the expected role of the Gleason score, the ECOG PS and the number of metastatic sites in determining the prognosis of this group of patients.
Table 2: Association between clinical and pathological characteristics with overall survival in the whole study cohort
Variables |
HR |
p |
95%CI |
Age |
0.99 |
0.88 |
0.96 – 1.03 |
ECOG PS |
1.89 |
0.009 |
1.17 – 3.06 |
PSA at the diagnosis |
1.018 |
0.366 |
0.98 – 1.06 |
Gleason Score |
3.59 |
0.019 |
1.23 – 10.44 |
MTS sites at the diagnosis |
1.51 |
0.061 |
0.98 – 2.32 |
Therapy |
1.39 |
0.184 |
0.85 – 2.27 |
ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; CI, confidence interval; MTS, metastases; PSA, prostate-specific antigen. ECOG PS and MTS were analysed as continuous variable. Gleason Score represents the risk difference between patients with <7 (reference) and ≥7. Therapy represents patients who received chemotherapy (none, 1 and ≥ 2 lines) and hormonal treatment between diagnosis and metastasis period.
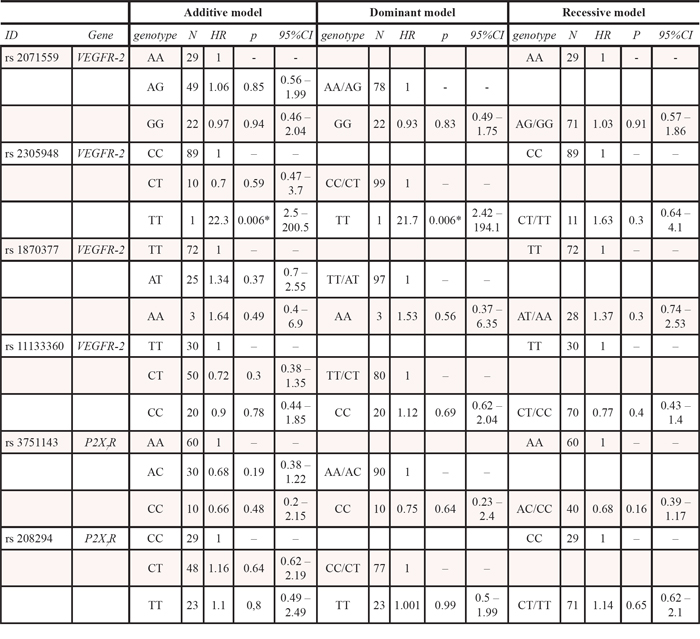
Details about genotypes and allele frequencies of all SNPs in the studied population are reported in Table 3; all the SNPs were in HWE. None of the single SNPs showed a statistically significant association in univariate Cox regression analysis, either under an additive, dominant or recessive model as illustrated in Table 4, with the only exception of VEGFR-2 rs2305948 that reaches the significance threshold in the dominant model. However, this statistically significant association is certainly a false-positive result due to the large confidence interval and the occurrence of just one TT case. Furthermore, the haplotype analysis of the two genetic regions did not reveal any significant association in univariate Cox regression model and Kaplan Meier curves (Supplementary Figure 1).
Table 3: Polymorphisms, genotypes, allele frequencies and Hardy-Weinberg Equilibrium (HWE)
ID |
Gene |
TaqMan SNPgenotyping assays |
genotype |
N |
allele |
N |
% |
HWE p-value |
rs2071559 |
VEGFR-2 |
C__15869271_10 |
AA |
29 |
A |
107 |
0.54 |
0.8435 |
AG |
49 |
G |
93 |
0.46 |
||||
GG |
22 |
|||||||
rs2305948 |
VEGFR-2 |
C__22271999_20 |
CC |
89 |
C |
188 |
0.94 |
0.2956 |
CT |
10 |
T |
12 |
0.06 |
||||
TT |
1 |
|||||||
rs1870377 |
VEGFR-2 |
C__11895315_20 |
TT |
72 |
T |
169 |
0.84 |
0.7 |
AT |
25 |
A |
31 |
0.16 |
||||
AA |
3 |
|||||||
rs11133360 |
VEGFR-2 |
C__26111278_10 |
TT |
30 |
T |
110 |
0.55 |
1 |
CT |
50 |
C |
90 |
0.45 |
||||
CC |
20 |
|||||||
rs3751143 |
P2X7R |
C__27495274_10 |
AA |
60 |
A |
150 |
0.75 |
0.0589 |
AC |
30 |
C |
50 |
0.25 |
||||
CC |
10 |
|||||||
rs208294 |
P2X7R |
C___3019032_1_ |
CC |
29 |
C |
106 |
0.53 |
0.6926 |
CT |
48 |
T |
94 |
0.47 |
||||
TT |
23 |
Table 4: Association between each polymorphism and overall survival (univariate Cox regression model) A p value < 0.0083 was defined as statistically significant (Bonferroni’s correction). *, the statistically significant association is a false-positive result due to the large confidence interval and the occurrence of only one TT case
The SDR analysis revealed a genetic interaction profile, consisting of the combination between specific VEGFR-2 (rs2071559, rs11133360) and P2X7R (rs3751143, rs208294) genotypes, significantly correlated with OS. Table SDR model (Table 5) shows the full analysis conducted by the SDR algorithm in our datasets. Cross-validation prevented over-fitting as the minimum test-IBS (0.1689) was observed for the interaction model, which was therefore chosen as the best epistatic model (i.e. the 4-way SDR model). This model was highly significant at the 0.05 threshold after 1000-fold permutation testing (p < 0.001). In particular, two genetic profiles were identified in patients, as reported in the genetic model table (Table 6). The favorable genetic profile was associated with a greater OS benefit whereas the unfavorable one with a lower OS in the enrolled patients, as shown in Figure 1. Indeed, after a median follow-up of 74+ months (range 14.6 - 231.7+ months), median OS of the enrolled patients was 98.9 months (Figure 1A; 95% CI, 77.38–117.36). Notably, the median OS for the favorable genetic profile was 126 months (Figure 1B; 95% CI, 115.94–152.96 months) vs. the 65.65 months of the unfavorable genetic profile (95%CI, 52.95–76.53 months; p < 0.0001, log-rank test; Figure 1B). Moreover, the Supplementary Figure 2 also shows the median OS of favorable and unfavorable profiles analyzed by the 3-way SDR analysis. The Cox proportional hazards model, performed to assess the adjusted hazard ratio for the OS of the favorable genetic profile, showed a value of 0.29 (95%CI, 0.15–0.56; p = 0.0001; Table 7). Supplementary Table 1 shows the same multivariate Cox regression model including also the variables “therapy” and “metastases sites at the diagnosis” that resulted not significant at the univariate analysis, confirming the lack of influence, in our model, of these two variables on OS. Of note, the probability of an estimated 2-year survival rate was 98% (95% CI, 87–99) in the favorable genetic profile and 92% (95% CI, 78–97) in the unfavorable genetic profile; the estimated 3-year survival was 96% (95% CI, 85–99) and 82% (95% CI, 66–91), respectively. Supplementary Table 2 shows the patient’s characteristics of both favorable and unfavorable groups. No differences were noted with the exception of the number of metastases sites at the diagnosis. Anyway, this variable was used in multivariate Cox regression to obtain an adjusted model (see Supplementary Table 1). However, this analysis confirms that the MTS sites at diagnosis, at least in our model, did not influence significantly the difference in OS between the two groups.
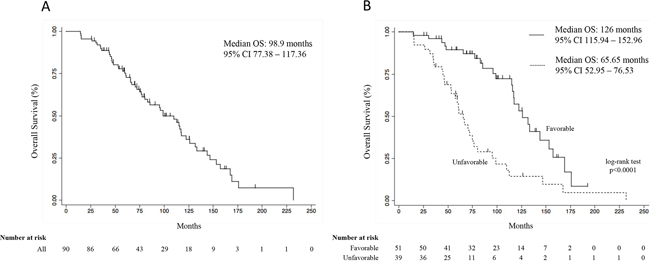
Figure 1: Overall survival curve of all patients (A), and overall survival curves according to the favorable and unfavorable genetic profiles (B). Survival curves were calculated by the Kaplan Meier method. CI, confidence interval.
Table 5: Survival dimensionality reduction (SDR) model for the survival analysis of dataset
IBS |
||||
n-way |
SNPs (genes) in each dimension |
Training |
Testing |
p |
1 |
rs3751143 (P2X7R) |
0.1876 |
0.1877 |
|
2 |
rs1870377 (VEGFR-2), rs3751143 (P2X7R) |
0.1846 |
0.1852 |
|
3 |
rs11133360 (VEGFR-2), rs3751143 (P2X7R), rs208294 (P2X7R) |
0.1741 |
0.1784 |
<0.001 |
4 |
rs2071559 (VEGFR-2), rs11133360 (VEGFR-2), rs3751143 (P2X7R), rs208294 (P2X7R) |
0.1638 |
0.1689 |
<0.001 |
Selection of the best combination of attributes by the SDR method. The model with the minimum testing Integrated Brier Score (IBS) value in the cross-validated testing sets is indicated in boldface type. P values associated with the log-rank test statistic calculated by the 1000-fold permutation test. SNP, single nucleotide polymorphism.
Table 6: Results of the genetic interaction analysis to translate the genotype combinations of the VEGFR-2 (rs2071559, rs11133360) and P2X7R (rs3751143, rs208294) genotypes into favorable or unfavorable genetic profiles for overall survival
Favorable genetic profile |
Unfavorable genetic profile |
||||||
rs2071559 |
rs11133360 |
rs3751143 |
rs208294 |
rs2071559 |
rs11133360 |
rs3751143 |
rs208294 |
AA |
CC |
AA |
CC |
AA |
CC |
AA |
CT |
AA |
CC |
AA |
TT |
AA |
CC |
AC |
CT |
AA |
CC |
CC |
CT |
AA |
CT |
AA |
TT |
AA |
CT |
AA |
CC |
AA |
CT |
AC |
CC |
AA |
CT |
AA |
CT |
AG |
CC |
AA |
CC |
AA |
CT |
AC |
CT |
AG |
CC |
AA |
CT |
AA |
CT |
AC |
TT |
AG |
CT |
AA |
CT |
AA |
TT |
AA |
CC |
AG |
CT |
AC |
TT |
AA |
TT |
AC |
CT |
AG |
TT |
AA |
CC |
AA |
TT |
AC |
TT |
AG |
TT |
AC |
CT |
AG |
CC |
AC |
CT |
AG |
TT |
AC |
TT |
AG |
CC |
AC |
TT |
GG |
CC |
CC |
TT |
AG |
CT |
AA |
CC |
GG |
CT |
AA |
TT |
AG |
CT |
AA |
TT |
GG |
CT |
AC |
CC |
AG |
CT |
AC |
CC |
GG |
TT |
AA |
CC |
AG |
CT |
AC |
CT |
GG |
TT |
AA |
CT |
AG |
TT |
AA |
CT |
||||
AG |
TT |
CC |
CT |
||||
AG |
TT |
CC |
TT |
||||
GG |
CC |
AC |
CT |
||||
GG |
CT |
AA |
CC |
||||
GG |
CT |
AA |
CT |
||||
GG |
CT |
CC |
CC |
||||
GG |
CT |
CC |
TT |
||||
GG |
TT |
AC |
CT |
||||
Table 7: Multivariate Cox regression model
Variables |
HR |
p |
95%CI |
Age |
0.93 |
0.008 |
0.88 – 0.98 |
ECOG PS |
1.40 |
0.34 |
0.7 – 2.8 |
Gleason Score |
5.18 |
0.005 |
1.65 – 16.2 |
Genetic profile |
|||
Unfavorable |
1 |
– |
– |
Favorable |
0.29 |
0.0001 |
0.15 – 0.56 |
ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; CI, confidence interval; ECOG PS was analysed as continuous variable. Gleason Score represents the risk difference between patients with <7 (reference) and ≥7.
DISCUSSION
Prostate carcinoma is still a leading cause of morbidity and mortality in developed countries, and the expected number of cancer cases in the next 15 years will increase by > 20% [19]. Localized cancers are usually treated with radical prostatectomy or radiation, while for more advanced cancers, either recurred or metastasized, the gold standard treatment is androgen ablation therapy, followed by the administration of chemotherapeutic drugs (e.g. docetaxel) [20].
The identification of biomarkers able to identify patients with the best prognosis is urgently needed to better define the proper therapeutic approach in the management of this disease. In the present analysis, we have applied the SDR methodology for identifying a genetic profile consisting of the combination between specific VEGFR-2 (rs2071559, rs11133360) and P2X7R (rs3751143, rs208294) genotypes associated with a greater OS as compared to that observed in the whole study population, in an unselected population of patients with prostate cancer. Among several loss-of-function SNPs of the P2X7R gene that may alter the normal trafficking [21] and activity of the receptor, the rs3751143 polymorphism has been associated to the familial form of chronic lymphocytic leukemia [22] and to the follicular variant of papillary thyroid cancer [23]. On the other hand, the polymorphism rs208294, leading the substitution of histidine for tyrosine at position 155 (H155Y) localized in P2X7R ectodomain, causes a gain of function of the receptor and it has been linked with human chronic lymphocytic leukemia lymphocytes [24]. Tumor formed by K562 cells transfected for a new mutation (A559-to-G substitution) showed a proliferative advantage and higher expression of VEGF and MCP1 [25], and haplotypes of the P2X7R containing the rs1718119 polymorphism have been linked to an enhanced IL-1β secretion [10]. The association between the VEGFR-2 SNPs and susceptibility or prognosis in several different types of cancers, such as renal cell carcinoma, colorectal cancer, hepatocellular carcinoma and glioblastoma, has been studied [26–29]. The rs2071559 G > A SNP has been reported to alter the structure of the binding site in VEGFR-2 gene promoter region for transcriptional factor E2F, with a consequent decrease of receptor expression, whereas the exonic polymorphisms rs1870377 and rs2305948 (in exon 11 and exon 7, respectively) resulted in a significant reduction of the VEGF binding efficiency to VEGFR-2 [12]. Moreover, Glubb and colleagues found that the rs1870377 T > A increases VEGFR-2 protein phosphorylation, thus resulting in an increased microvessel density in lung cancer tumor tissues [30].
In our study we firstly associated each individual SNP with OS, but no association between each polymorphism and OS was found with the only exception of VEGFR-2 rs2305948, a false-positive result due to the large confidence interval and the occurrence of just one TT case. As demonstrated by the epistasis phenomenon, the effects of a single SNP can disseminate through numerous gene–gene interactions, determining multiple phenotypes; thus, we think that the current approach of correlating the prognosis to just one SNP should be reconsidered and replaced by a genetic analysis of the interaction among SNPs. For these reasons, we decided to apply the more complex SDR methodology to investigate the interaction between germline SNPs, in order to identify a possible genetic profile associated with the greater OS probability in this unselected population of patients carrying prostate cancer (Table 1 and 5). The analyses conducted in this population revealed a genetic interaction profile, consisting of the combination between specific VEGFR-2 (rs2071559, rs11133360) and P2X7R (rs3751143, rs208294) genotypes, associated with OS. Particularly, two genetic profiles were identified in patients, as reported in Table 6. The first column was associated with a greater OS benefit and the second one with a lower OS, respectively. Because of the connections within biological pathways, the effects of a single mutation can extend through thousands of gene–gene interactions, resulting in multiple phenotypes [31]. In our study we demonstrated, through the SDR methodology, a never previously shown statistically significant interaction between P2X7R and VEGFR-2 gene SNPs that potentially relates to prognosis of prostate cancer patients.
Genetic information impacts phenotype through a hierarchy of proteins that are involved in biological processes ranging from transcription to physiological homeostasis. Physical interactions among proteins and other biomolecules and their impact on phenotype are the main components of biological epistasis [32]. Biological epistasis occurs at the level of the single patient and involves DNA sequence variations, biomolecules and their physical interactions determining a particular phenotype for that single individual. Instead, statistical epistasis is a population event that is made possible by interindividual variability in genotypes, biomolecules and in their physical interactions [32]. Thus, the differences in biological epistasis among individuals in a population give rise to statistical epistasis [32]. Evidently, making both hypotheses or conclusions about biological function and causation from statistical epistasis results in patients will always balk in a challenge, unless relevant molecular information has been also acquired [33]. However, SDR or multifactor dimensionality reduction (MDR) methodology [34] has been applied to detect gene-gene interactions for several clinical phenotypes, and it may provide means to find new hypotheses for further testing about epistatic interactions in genetic data also in prostate cancer. As an example, the interaction between VEGF 2482T and VEGFR IVS6 + 54 loci suggests that the inheritance of VEGF and VEGFR IVS6 + 54 sequence variants may jointly modify prostate cancer susceptibility through their influence on angiogenesis [35]. Whilst it is difficult to explain the biological meaning of our statistical epistatic data, it should be noted that several data support a possible link between P2X7R and VEGFR-2 in tumor physiopathology. Although at a first glance the two genes, and, consequently, the two receptors have completely different activities and signaling pathways, it is not surprising that P2X7R and VEGFR-2 share more than a simple biological connection. In fact, it has been demonstrated that the modulation of angiogenesis exerted by eATP through endothelial P2Y receptors can stimulate angiogenic properties such as endothelial cell migration and vascular permeability [36, 37]; moreover activated P2Y2 receptors can transactivate VEGFR-2, suggesting a direct link between extracellular nucleotide regulation and growth factor signaling [38]. Interestingly, Wei and colleagues [39] demonstrated that the chronic exposure of C6 glioma cells to 2′,3′-(benzoyl-4-benzoyl)-ATP (BzATP), a selective P2X7R agonist, enhanced the expression of pro-inflammatory and angiogenic factors, including IL-8 and VEGF, suggesting a link between the receptor activation, inflammation, increased angiogenesis and tumor cell migration. Furthermore, P2X7R-expressing tumors are characterized by increased proliferation, reduced apoptosis, and a more developed vascular network that control tumors and by elevated secreted amounts of VEGF [40]. The growth and neoangiogenesis of P2X7R-expressing tumors was blocked by intratumoral injection of the VEGF-blocking antibody bevacizumab, pharmacologic P2X7R blockade, or P2X7R silencing in vivo [40]. Recently, Amoroso and colleagues showed that P2X7R down-modulation caused a reduction in HIF1alpha levels and VEGF secretion with a decreased vessel formation in a neuroblastoma model [41]. Based on these premises, it is conceivable to hypothesize that, in patients carrying the more unfavorable genetic profile, the pro-inflammatory activity and the tumor angiogenesis is sustained by the gene-gene interaction of both receptors, with a graduality due to the various combinations of genotypes, resulting in a worse prognosis. Such angiogenic phenotype could be due to an increase of the VEGF production attributable to an enhanced P2X7R activity, which may in turn activate an upregulated VEGFR-2. Unfortunately, the phenotypes corresponding to the VEGFR-2 rs11133360 genotypes are still unknown [42]. Therefore, it might be plausible that the genetic background may be responsible, in part, for the prognosis in these metastatic prostate cancer patients. Conversely, in patients with a more favorable genetic profile, the microenvironment conditions due to the different genotype combinations may result in a reduction of the VEGF production and in the presence of fewer VEGFR-2, or of its lower activity, on tumor endothelial cells which are not capable to proliferate, migrate or survive.
Thus, despite the objective difficulties to demonstrate a biological mechanism for the statistically demonstrated gene-gene interaction, our original findings may provide a new reliable prognostic value and suggest previously unknown hypotheses for further testing also biological epistatic interactions. Although this study presents some strengths as the long observational follow-up of the patients, lasting almost 20 years, or the extreme novelty of the approach, however, final considerations are restricted by the exploratory nature of its retrospective design, the limited sample size with the potential risk of an over-estimation of the obtained results. Indeed, these results should be only considered as “hypothesis generating” and only a well-designed prospective clinical trial may eventually confirm the suggested innovative role of the genetic interaction profile between specific VEGFR-2 (rs2071559, rs11133360) and P2X7R (rs3751143, rs208294) genotypes analyzed by SDR. However, understanding the reasons why the singular genotypes of both VEGFR-2 rs2071559 and rs11133360 or P2X7R rs3751143 and rs208294 were not associated to the greater benefit in terms of OS in our study, in contrast to what reported when combined in the interaction analysis, remains a challenge.
The identification of good versus poor prognosis prostate cancer patients through non-invasive and reliable means is a largely unmet clinical need. The possible clinical implication of identifying a favorable and an unfavorable group through SDR analysis could reside on this characteristic. In fact, we could hypothesize that poor prognosis patients may benefit from novel treatment strategies from the very beginning, and perhaps should be considered earlier for clinical trials with innovative drugs rather than waiting until the failure of standard therapies. On the other hand, prostate cancer patients with overall good prognosis may opt for a less aggressive treatment among the available standard therapeutic options.
In conclusion, the SDR methodology has been applied in these unselected prostate cancer patients to investigate the role of an interaction between VEGFR-2 and P2X7R gene polymorphisms in identifying a genetic profile associated with the greater probability of OS. The results have confirmed the relevance of SDR analyses, as already described by other authors, suggesting a more rational approach when SNPs are investigated as possible predictors of prognosis.
MATERIALS AND METHODS
Study population
The present multicenter, retrospective, genetic study was approved by the local ethics committee. Patients were informed of the investigational nature of the study and provided their written informed consent for collecting genetic data. One-hundred patients with age ≥ 18 years and histological diagnosis of prostate adenocarcinoma were enrolled (Table 1), and they were assessed for the present genetic study. Other inclusion criteria were PSA ≥ 10 ng/mL; Eastern Cooperative Oncology Group performance status (ECOG PS) of ≤ 2; adequate bone marrow function (leukocytes, ≥ 3000/mL; neutrophil count, ≥ 1500/mL; hemoglobin level, ≥ 10 g/dL; platelets, ≥ 100,000/mL); adequate liver function (total serum bilirubin level, < 1.5 mg/dl; aspartate aminotransferase and alanine aminotransferase, < 3-fold upper limit of normal); adequate renal function (serum creatinine level, < 1.5 mg/dL), and life expectancy ≥ 3 months. Exclusion criteria at baseline were acute cardiovascular disease (uncontrolled hypertension and arrhythmia; myocardial infarction within 2 years before enrolment; unstable angina; New York Heart Association, grade II or greater congestive heart failure), active infections, high risk of thromboembolic events without prophylactic treatments, untreated hemorrhagic gastric disease, or presence of brain metastases. Sites of metastatic disease were evaluated by radiological exams (radiography, X-ray computed tomography) and ultrasonography (Table 1).
At the enrollment, clinical evaluations included: patient’s medical history; a physical exam with an assessment of weight, vital signs, and ECOG PS; electrocardiogram plus cardiovascular examinations; complete blood count; complete serum biochemistry (i.e., serum creatinine, fasting glucose, sodium, potassium, calcium, lactate dehydrogenase, aspartate aminotransferase, alanine aminotransferase, γGT, ALP, total bilirubin, PT, aPTT, fibrinogen, and D-dimer), PSA serum levels.
SNP selection
The SNPs included in our study (Table 3) were selected on the basis of three main considerations: (i) thus far no genetic interaction analysis has been reported between the chosen SNPs and prognosis of prostate cancer, and possible gene-gene interactions could determine previously undescribed statistical epistatic effects; (ii) some of the SNPs have been significantly associated with risk or prognosis in other tumour types [23, 26, 43–47]; (iii) some SNPs have been associated with a modulation of the gene expression, or with impaired activity of the receptors [10–12, 48]. Although for some of chosen SNPs the phenotypic effects are still undefined or controversial, we decided to include them in our research because of the possibility that gene-gene interaction could produce an unexpected statistical epistatic effect.
Genotyping analyses
Blood samples (3 ml) were collected in EDTA tubes and stored at -80°C. P2X7R and VEGFR-2 genes and polymorphisms of Table 3 were chosen for the present analyses. Germline DNA extraction was performed using QIAamp DNA Blood Mini Kit (Qiagen, Valencia, CA, USA). Allelic discrimination of genes was performed using an ABI PRISM 7900 SDS (Applied Biosystems, Carlsbad, CA, USA) and with validated TaqMan® SNP genotyping assays (Table 3; Applied Biosystems). PCR reactions were carried out according to the manufacturer’s protocol. Genotyping was not performed until an adequate number of events (>80% on study population) was reported in terms of OS.
Statistical analysis
All polymorphisms were analyzed for deviation from the Hardy-Weinberg Equilibrium (HWE), through comparison between observed allelic distributions with those expected from the HWE by on χ2 test. Linkage disequilibrium (LD) between markers in VEGFR-2 (n = 4) and P2X7R (n = 2) was analysed using Haploview software package [49]. The association between each individual polymorphism and the most relevant clinical-pathological characteristics with overall survival (OS) was tested using a Cox proportional hazards model. OS were calculated from the date of the diagnosis to the date of death/lost to follow-up.
PHASE software v 2.1 [50] was used to perform haplotype analysis for VEGFR-2 and P2X7R regions separately. Only common haplotypes with a frequency ≥ 5% were included in COX survival analysis. Most common haplotype was used as the reference and were provided hazard ratios (HR) for a given haplotype relative to reference haplotype.
The Survival Dimensionality Reduction (SDR) methodology was applied (using version 2.0a of SDR software available on http://sourceforge.net/projects/sdrproject/) to detect non-linear gene-gene interactions in presence of right-censored data in identifying biomarkers of prognosis in OS [18]. The best combination of SDR model was chosen based on the lowest testing Integrated Brier Score (IBS) value. Log-rank test statistic calculated by the 100-fold permutation test was used to evaluate association and model efficiency.
The genotype combination with the OS benefit and improvement was used for further analyses. The difference in OS between favorable genetic profiles and the unfavorable genetic profiles were assessed with the log-rank test and the Kaplan-Meier method to evaluate survival curves. A Cox proportional hazards model, with the possible genetic profiles and the clinical and pathological patient characteristics individually correlated with the OS, was used to calculate the adjusted hazards ratio (HR) and the 95% confidence interval (95% CI). In these analyses we used Bonferroni’s correction and the p value < 0.0083 (0.05/6 SNPs = 0.0083) was accepted as statistically significant. The Kaplan–Meier and Cox proportional hazards analyses were performed using the STATA package version 11.0 (StataCorp).
ACKNOWLEDGMENTS
We would like to thanks all the patients and their families, and the nurses for their technical assistance.
FUNDING
The present study has been funded by the “Bando Ricercatori 2011” of the University of Pisa.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.
REFERENCES
1. Sfanos KS, Isaacs WB, De Marzo AM. Infections and inflammation in prostate cancer. Am J Clin Exp Urol. 2013; 1:3–11.
2. Stifter S, Dordevic G. Prostate cancer and new insights in angiogenesis. Front Oncol. 2014; 4:243.
3. Sfanos KS, Hempel HA, De Marzo AM. The role of inflammation in prostate cancer. Adv Exp Med Biol. 2014; 816:153–181.
4. Carmeliet P, Jain RK. Molecular mechanisms and clinical applications of angiogenesis. Nature. 2011; 473:298–307.
5. Antonioli L, Blandizzi C, Pacher P, Hasko G. Immunity, inflammation and cancer: a leading role for adenosine. Nat Rev Cancer. 2013; 13:842–857.
6. Santini E, Cuccato S, Madec S, Chimenti D, Ferrannini E, Solini A. Extracellular adenosine 5′-triphosphate modulates insulin secretion via functionally active purinergic receptors of X and Y subtype. Endocrinology. 2009; 150:2596–2602.
7. Roger S, Jelassi B, Couillin I, Pelegrin P, Besson P, Jiang L. Understanding the roles of the P2X7 receptor in solid tumour progression and therapeutic perspectives. Biochim Biophys Acta. 2014; pii:S0005-2736(14)00364-2, doi:10.1016/j.bbamn.2014.10.029.
8. Slater M, Danieletto S, Gidley-Baird A, Teh LC, Barden JA. Early prostate cancer detected using expression of non-functional cytolytic P2X7 receptors. Histopathology. 2004; 44:206–215.
9. Ravenna L, Sale P, Di Vito M, Russo A, Salvatori L, Tafani M, Mari E, Sentinelli S, Petrangeli E, Gallucci M, Di Silverio F, Russo MA. Up-regulation of the inflammatory-reparative phenotype in human prostate carcinoma. Prostate. 2009; 69:1245–1255.
10. Stokes L, Fuller SJ, Sluyter R, Skarratt KK, Gu BJ, Wiley JS. Two haplotypes of the P2X(7) receptor containing the Ala-348 to Thr polymorphism exhibit a gain-of-function effect and enhanced interleukin-1beta secretion. FASEB J. 2010; 24:2916–2927.
11. Gu BJ, Sluyter R, Skarratt KK, Shemon AN, Dao-Ung LP, Fuller SJ, Barden JA, Clarke AL, Petrou S, Wiley JS. An Arg307 to Gln polymorphism within the ATP-binding site causes loss of function of the human P2X7 receptor. J Biol Chem. 2004; 279:31287–31295.
12. Wang Y, Zheng Y, Zhang W, Yu H, Lou K, Zhang Y, Qin Q, Zhao B, Yang Y, Hui R. Polymorphisms of KDR gene are associated with coronary heart disease. J Am Coll Cardiol. 2007; 50:760–767.
13. Moore JH, Boczko EM, Summar ML. Connecting the dots between genes, biochemistry, and disease susceptibility: systems biology modeling in human genetics. Mol Genet Metab. 2005; 84:104–111.
14. Potente M, Gerhardt H, Carmeliet P. Basic and therapeutic aspects of angiogenesis. Cell. 2011; 146:873–887.
15. Wilke RA, Reif DM, Moore JH. Combinatorial pharmacogenetics. Nat Rev Drug Discov. 2005; 4:911–918.
16. Moore JH. A global view of epistasis. Nat Genet. 2005; 37:13–14.
17. Moore JH. The ubiquitous nature of epistasis in determining susceptibility to common human diseases. Hum Hered. 2003; 56:73–82.
18. Beretta L, Santaniello A, van Riel PL, Coenen MJ, Scorza R. Survival dimensionality reduction (SDR): development and clinical application of an innovative approach to detect epistasis in presence of right-censored data. BMC Bioinformatics. 2010; 11:416.
19. Weir HK, Thompson TD, Soman A, Moller B, Leadbetter S. The past, present, and future of cancer incidence in the United States: 1975 through 2020. Cancer. 2015; 121:1827–1837.
20. Leibowitz-Amit R, Joshua AM. The changing landscape in metastatic castration-resistant prostate cancer. Curr Opin Support Palliat Care. 2013; 7:243–248.
21. Wiley JS, Dao-Ung LP, Li C, Shemon AN, Gu BJ, Smart ML, Fuller SJ, Barden JA, Petrou S, Sluyter R. An Ile-568 to Asn polymorphism prevents normal trafficking and function of the human P2X7 receptor. J Biol Chem. 2003; 278:17108–17113.
22. Wiley JS, Dao-Ung LP, Gu BJ, Sluyter R, Shemon AN, Li C, Taper J, Gallo J, Manoharan A. A loss-of-function polymorphic mutation in the cytolytic P2X7 receptor gene and chronic lymphocytic leukaemia: a molecular study. Lancet. 2002; 359:1114–1119.
23. Dardano A, Falzoni S, Caraccio N, Polini A, Tognini S, Solini A, Berti P, Di Virgilio F, Monzani F. 1513A>C polymorphism in the P2X7 receptor gene in patients with papillary thyroid cancer: correlation with histological variants and clinical parameters. J Clin Endocrinol Metab. 2009; 94:695–698.
24. Cabrini G, Falzoni S, Forchap SL, Pellegatti P, Balboni A, Agostini P, Cuneo A, Castoldi G, Baricordi OR, Di Virgilio F. A His-155 to Tyr polymorphism confers gain-of-function to the human P2X7 receptor of human leukemic lymphocytes. J Immunol. 2005; 175:82–89.
25. Chong JH, Zheng GG, Ma YY, Zhang HY, Nie K, Lin YM, Wu KF. The hyposensitive N187D P2X7 mutant promotes malignant progression in nude mice. J Biol Chem. 2010; 285:36179–36187.
26. Scartozzi M, Faloppi L, Svegliati Baroni G, Loretelli C, Piscaglia F, Iavarone M, Toniutto P, Fava G, De Minicis S, Mandolesi A, Bianconi M, Giampieri R, Granito A, Facchetti F, Bitetto D, Marinelli S, et al. VEGF and VEGFR genotyping in the prediction of clinical outcome for HCC patients receiving sorafenib: the ALICE-1 study. Int J Cancer. 2014; 135:1247–1256.
27. Scartozzi M, Bianconi M, Faloppi L, Loretelli C, Bittoni A, Del Prete M, Giampieri R, Maccaroni E, Nicoletti S, Burattini L, Minardi D, Muzzonigro G, Montironi R, Cascinu S. VEGF and VEGFR polymorphisms affect clinical outcome in advanced renal cell carcinoma patients receiving first-line sunitinib. Br J Cancer. 2013; 108:1126–1132.
28. Sjostrom S, Wibom C, Andersson U, Brannstrom T, Broholm H, Johansen C, Collatz-Laier H, Liu Y, Bondy M, Henriksson R, Melin B. Genetic variations in VEGF and VEGFR2 and glioblastoma outcome. J Neurooncol. 2011; 104:523–527.
29. Jang MJ, Jeon YJ, Kim JW, Cho YK, Lee SK, Hwang SG, Oh D, Kim NK. Association of VEGF and KDR single nucleotide polymorphisms with colorectal cancer susceptibility in Koreans. Mol Carcinog. 2013; 52 1:E60–69.
30. Glubb DM, Cerri E, Giese A, Zhang W, Mirza O, Thompson EE, Chen P, Das S, Jassem J, Rzyman W, Lingen MW, Salgia R, Hirsch FR, Dziadziuszko R, Ballmer-Hofer K, Innocenti F. Novel functional germline variants in the VEGF receptor 2 gene and their effect on gene expression and microvessel density in lung cancer. Clin Cancer Res. 2011; 17:5257–5267.
31. Tyler AL, Asselbergs FW, Williams SM, Moore JH. Shadows of complexity: what biological networks reveal about epistasis and pleiotropy. Bioessays. 2009; 31:220–227.
32. Moore JH, Williams SM. Traversing the conceptual divide between biological and statistical epistasis: systems biology and a more modern synthesis. Bioessays. 2005; 27:637–646.
33. Edwards TL, Lewis K, Velez DR, Dudek S, Ritchie MD. Exploring the performance of Multifactor Dimensionality Reduction in large scale SNP studies and in the presence of genetic heterogeneity among epistatic disease models. Hum Hered. 2009; 67:183–192.
34. Moore JH, Gilbert JC, Tsai CT, Chiang FT, Holden T, Barney N, White BC. A flexible computational framework for detecting, characterizing, and interpreting statistical patterns of epistasis in genetic studies of human disease susceptibility. J Theor Biol. 2006; 241:252–261.
35. VanCleave TT, Moore JH, Benford ML, Brock GN, Kalbfleisch T, Baumgartner RN, Lillard JW Jr, Kittles RA, Kidd LC. Interaction among variant vascular endothelial growth factor (VEGF) and its receptor in relation to prostate cancer risk. Prostate. 2010; 70:341–352.
36. Kaczmarek E, Erb L, Koziak K, Jarzyna R, Wink MR, Guckelberger O, Blusztajn JK, Trinkaus-Randall V, Weisman GA, Robson SC. Modulation of endothelial cell migration by extracellular nucleotides: involvement of focal adhesion kinase and phosphatidylinositol 3-kinase-mediated pathways. Thromb Haemost. 2005; 93:735–742.
37. Tanaka N, Nejime N, Kagota S, Kubota Y, Yudo K, Nakamura K, Kunitomo M, Takahashi K, Hashimoto M, Shinozuka K. ATP participates in the regulation of microvessel permeability. J Pharm Pharmacol. 2006; 58:481–487.
38. Seye CI, Yu N, Gonzalez FA, Erb L, Weisman GA. The P2Y2 nucleotide receptor mediates vascular cell adhesion molecule-1 expression through interaction with VEGF receptor-2 (KDR/Flk-1). J Biol Chem. 2004; 279:35679–35686.
39. Wei W, Ryu JK, Choi HB, McLarnon JG. Expression and function of the P2X(7) receptor in rat C6 glioma cells. Cancer Lett. 2008; 260:79–87.
40. Adinolfi E, Raffaghello L, Giuliani AL, Cavazzini L, Capece M, Chiozzi P, Bianchi G, Kroemer G, Pistoia V, Di Virgilio F. Expression of P2X7 receptor increases in vivo tumor growth. Cancer Res. 2012; 72:2957–2969.
41. Amoroso F, Capece M, Rotondo A, Cangelosi D, Ferracin M, Franceschini A, Raffaghello L, Pistoia V, Varesio L, Adinolfi E. The P2X7 receptor is a key modulator of the PI3K/GSK3beta/VEGF signaling network: evidence in experimental neuroblastoma. Oncogene. 2015; doi: 10.1038/onc.2014.444.
42. Lambrechts D, Delmar P, Miles DW, Leighl N, Saltz L, Escudier B, Van Cutsem E, Scherer SJ, Carmeliet P, de Haas S. Single nucleotide polymorphism analysis and outcome in advanced-stage cancer patients treated with bevacizumab. Eur J Cancer. 2011; 47:s173.
43. Allegrini G, Coltelli L, Orlandi P, Fontana A, Camerini A, Ferro A, Cazzaniga M, Casadei V, Lucchesi S, Bona E, Di Lieto M, Pazzagli I, Villa F, Amoroso D, Scalese M, Arrighi G, et al. Pharmacogenetic interaction analysis of VEGFR-2 and IL-8 polymorphisms in advanced breast cancer patients treated with paclitaxel and bevacizumab. Pharmacogenomics. 2014; 15:1985–1999.
44. Estevez-Garcia P, Castano A, Martin AC, Lopez-Rios F, Iglesias J, Munoz-Galvan S, Lopez-Calderero I, Molina-Pinelo S, Pastor MD, Carnero A, Paz-Ares L, Garcia-Carbonero R. PDGFRalpha/beta and VEGFR2 polymorphisms in colorectal cancer: incidence and implications in clinical outcome. BMC Cancer. 2012; 12:514.
45. Zheng YB, Zhan MX, Zhao W, Liu B, Huang JW, He X, Fu SR, Zhao Y, Li Y, Hu BS, Lu LG. The relationship of kinase insert domain receptor gene polymorphisms and clinical outcome in advanced hepatocellular carcinoma patients treated with sorafenib. Med Oncol. 2014; 31:209.
46. Dao-Ung LP, Fuller SJ, Sluyter R, SkarRatt KK, Thunberg U, Tobin G, Byth K, Ban M, Rosenquist R, Stewart GJ, Wiley JS. Association of the 1513C polymorphism in the P2X7 gene with familial forms of chronic lymphocytic leukaemia. Br J Haematol. 2004; 125:815–817.
47. Solini A, Cuccato S, Ferrari D, Santini E, Gulinelli S, Callegari MG, Dardano A, Faviana P, Madec S, Di Virgilio F, Monzani F. Increased P2X7 receptor expression and function in thyroid papillary cancer: a new potential marker of the disease?. Endocrinology. 2008; 149:389–396.
48. Ghiadoni L, Rossi C, Duranti E, Santini E, Bruno RM, Salvati A, Taddei S, Solini A. P2X7 receptor polymorphisms do not influence endothelial function and vascular tone in neo-diagnosed, treatment-naive essential hypertensive patients. J Hypertens. 2013; 31:2362–2369.
49. Barrett JC, Fry B, Maller J, Daly MJ. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics. 2005; 21:263–265.
50. Stephens M, Scheet P. Accounting for decay of linkage disequilibrium in haplotype inference and missing-data imputation. Am J Hum Genet. 2005; 76:449–462.

