INTRODUCTION
Capecitabine is an oral fluoropyrimidine that delivers 5-fluorouracil (5-FU) to the tumor [1]. Both alone and in combination with other chemotherapeutic and biological agents, capecitabine is increasingly used in adjuvant and metastatic settings because it is easier to administer and has a more favorable toxicity profile than 5-FU [1]. Since their discovery in 1957, fluoropyrimidines have been the mainstay of treatment of colorectal cancer (CRC), a major cause of morbidity in developed countries [2]. 5-FU acts by inhibiting thymidylate synthase (TYMS) and incorporating drug metabolites into DNA and RNA, thus blocking DNA synthesis [3].
Capecitabine-treated patients commonly experience severe, even fatal, adverse drug reactions (ADRs) at some point during their treatment. These reactions often lead to dose reductions, delays in administration, and discontinuation of treatment [4, 5]. Although capecitabine is a prodrug of 5-FU, its toxicity profile is significantly different. While hematologic toxicity is most often associated with 5-FU, side effects such as diarrhea, nausea, vomiting, and hand-foot syndrome (HFS) are more commonly associated with capecitabine [6].
The toxicity of chemotherapeutic drugs is affected by factors such as age, performance status, organ dysfunction, and the presence of other co-morbidities. Interindividual genetic variability can also play an important role [7]. Many genes, nucleotides, antigens, and enzymes are known to be involved in the metabolism and efficacy of the drugs used in treatment of CRC. In addition, single-nucleotide polymorphisms (SNPs) lead to various outcomes in clinical practice [8]. Pharmacogenetics evaluates the effect of genetic variations on the individual response to and tolerability of therapy. Predicting the individual risk of toxicity for a particular drug could improve the quality of care. High-risk patients could be candidates for lower doses or alternative drugs in order to avoid toxicity [7]. Numerous gene polymorphisms have been associated with capecitabine-induced toxicity. For instance, DPYD variants have been extensively studied, and dosing guidelines have been suggested [9, 10] (http://www.pharmgkb.org/gene/PA166109594). However, the low frequency of toxicity-related alleles and the relatively frequent occurrence of severe ADRs to capecitabine indicate that other factors are involved in the risk of ADR. Laboratory tests have been designed for commercial or research purposes to predict the risk of fluoropyrimidine-induced toxicity. Nonetheless, they have all proven to be insufficiently accurate, thus stressing the need for new markers [11].
Various polymorphisms in CDA, ABCB1, MTHFR, and TYMS have been associated with capecitabine-induced ADRs, although findings are controversial and the evidence poor [7, 11-22]. The relationship between some of these genes and the development of toxicity to capecitabine is not clear. For instance, a meta-analysis describing an ENOSF1 SNP in linkage disequilibrium with TYMS variants identified ENOSF1 as a putative causal genetic variant for capecitabine-related toxicity [11]. However, the authors suggest that this finding needs to be confirmed in new cohorts.
We performed a prospective/retrospective study of a cohort of CRC patients treated with capecitabine-containing regimens in order to evaluate possible associations between severe ADRs to capecitabine and genomic variations in CDA, ABCB1, ENOSF1, TYMS, and MTHFR.
RESULTS
A total of 239 capecitabine-treated patients were selected for the study. The baseline characteristics of the study population are shown in Table 1. The median age at diagnosis was 67 years (range, 30 to 88 years). Sex distribution was nearly homogeneous (54% men and 46% women). Patients had predominantly colon carcinoma (71.1%) and a good performance status (0-2, 98.7%), and 127 patients (53.1%) had metastatic disease. Combination regimens were more frequent than capecitabine monotherapy (74.5 vs 25.5%). Over half of the patients received oxaliplatin as part of a combination regimen (60.3%). Other concomitantly administered drugs included irinotecan and monoclonal antibodies (bevacizumab, cetuximab, and panitumumab).
Delay in administration, dose reduction, or withdrawal of the drug due to toxicity was common (70.7%) (Table 1). In clinical practice, a moderate HFS equal to 1 is often followed by a change in the treatment settings; therefore, we evaluated moderate-severe HFS (grade >1, 22.6%) and severe HFS (grade >2, 6.3%). Other frequently observed ADRs included severe diarrhea (grade >2, 10.9%) and severe hematological toxicity (grade >2, 7.1%).
Nine polymorphisms in five genes were genotyped for the 239 patients who fulfilled the inclusion criteria. No significant deviations from Hardy-Weinberg Equilibrium were detected, except for the SNP rs1045642 in ABCB1 (P=0.01). Therefore, analyses of ABCB1 were performed using haplotype *1 (rs1128503 C, rs2032582 G, and rs1045642 C). Univariate analysis revealed significant associations between multiple severe ADRs and the polymorphisms CDA rs2072671, TYMs rs45445694 and rs34489327, ENOSF1 rs2612091, and ABCB1*1 (Table 2). MTHFR rs1801133 and rs1801131 showed no significant associations in this preliminary analysis and were therefore ruled out for subsequent testing. This analysis enabled us to identify putative risk alleles or variants.
Table 1: Patient characteristics
Characteristic |
N (percentage) |
Age |
|
Median age at diagnosis (range) |
67 (30-88) |
Sex |
|
Male |
129 (54%) |
Female |
110 (46%) |
Hospital |
|
H. Doce de Octubre |
99(41.4%) |
H. Gregorio Marañón |
140 (58.6%) |
Performance status |
|
≤2 |
236 (98.7%) |
>2 |
2 (1.3%) |
Tumor stage |
|
I-II |
30 (12.6%) |
III |
82 (34.3%) |
IV |
127 (53.1%) |
Type of cancer |
|
Colon |
170 (71.1%) |
Rectum |
69 (28.9%) |
Treatment setting |
|
Adjuvant |
112 (46.8%) |
Metastatic |
127 (53.1%) |
Number of cycles |
|
Median (range) |
8 (1-58) |
Regimen |
|
Monotherapy |
61 (25.5%) |
Combination |
178 (74.5%) |
Concomitant drug |
|
Oxaliplatin |
144 (60.3%) |
Irinotecan |
28 (11.7%) |
Antibodies |
53 (22.1%) |
Adverse reactions* |
|
Reduction/Delay/withdrawal treatment |
169 (70.7%) |
Nausea/Vomiting > 2 |
9 (3.8%) |
Diarrhea > 2 |
26 (10.9%) |
Hand-foot syndrome >2 |
15 (6.3%) |
Hand-foot syndrome >1 |
54 (22.6%) |
Hematological toxicity > 2 |
17 (7.1%) |
Mucositis > 2 |
4 (1.7%) |
Anorexia > 2 |
4 (1.7%) |
*Adverse reaction graded according to National Cancer Institute Common Terminology Criteria for Adverse Events v4.0.
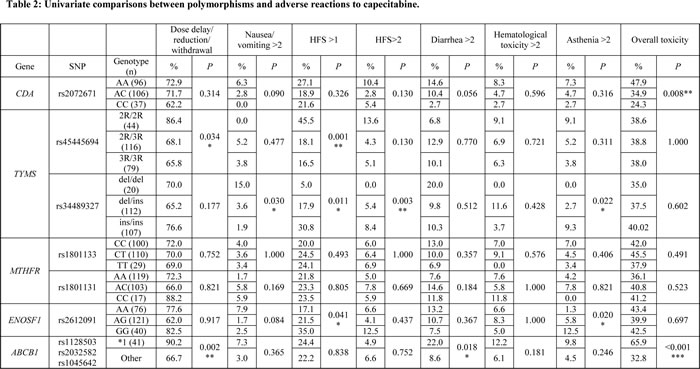
Given the inherent low statistical power of this kind of analysis and in order to obtain more robust significant associations, we performed a binary logistic regression analysis for those genotypes that initially rendered a statistically significant result. In the multivariate analysis, the P value was adjusted for sex, tumor stage, and hospital.
CDA rs2072671 was associated with overall toxicity (any ADR classed as grade 3 or higher). Homozygous AA patients presented a higher risk of overall toxicity than AC or CC patients (OR, 1.84; 95% CI, 1.06-3; P=0.029) (Table 3). A tendency toward HFS >2 was also observed for this SNP, although it was not statistically significant.
The ABCB1 CGC haplotype for the SNPs rs1128503, rs2032582, and rs1045642 (ABCB1*1), respectively, was associated with the following delays in starting chemotherapy (any cycle), dose reduction, or withdrawal of capecitabine; severe diarrhea; and severe overall toxicity (Table 3). The association between ABCB1*1 and severe overall toxicity was particularly relevant (OR, 4.06; 95% CI, 1.97-8.38; P<0.001).
TYMS 2R (rs45445694) was significantly associated with moderate-severe HFS (grade >1) and severe HFS (grade >2) (Table 3). The TYMS 6ins allele was also associated with a higher risk of HFS >1 (P=0.011) and HFS >2 (P=0.003) in the univariate analysis (Table 2), although these associations disappeared after adjusting for sex, hospital, and type of cancer (Table 3).
Homozygous AA individuals harboring ENOSF1 rs2612091 showed a higher risk of HFS >1 in the univariate analysis (P=0.041). This association remained statistically significant in the adjusted analysis (OR, 2.28; 95% CI, 1.10-4.76; P=0.027).
Given that CDA rs2072671 and ABCB1 haplotypes (rs2072671, rs1128503, rs2032582, and rs1045642) were both significantly associated with severe overall toxicity, a CDA-ABCB1 score was calculated based on the number of risk alleles (from 0 to 8). A CDA-ABCB1 score >5 predicted overall toxicity with a positive predictive value of 54.05%, negative predictive value of 68.48%, sensitivity of 43.47%, and specificity of 76.87%.
Table 3: Analysis by logistic regression of previous significant associations
OR |
CI |
Padj |
|
ABCB1 (Ref: no ABCB1*1) |
|||
Delay/reduction/withdrawal |
4.49 |
1.53-13.19 |
0.006** |
Diarrhea >2 |
3.16 |
1.28-7.79 |
0.012* |
Hand-foot syndrome >1 |
1.11 |
0.50-2.46 |
0.798 |
Hand-foot syndrome >2 |
0.72 |
0.15-3.37 |
0.673 |
Hematological toxicity >2 |
2.16 |
0.71-6.56 |
0.173 |
Asthenia >2 |
2.46 |
0.69-8.80 |
0.165 |
Overall toxicity |
4.06 |
1.97-8.38 |
<0.001*** |
CDA rs2072671 (Ref: AC/CC) |
|||
Delay/reduction/withdrawal |
1.25 |
0.69-2.25 |
0.460 |
Diarrhea >2 |
1.83 |
0.79-4.24 |
0.157 |
Hand-foot syndrome >1 |
1.56 |
0.83-2.94 |
0.163 |
Hand-foot syndrome >2 |
2.89 |
0.93-8.98 |
0.066 |
Hematological toxicity >2 |
1.38 |
0.50-3.80 |
0.531 |
Asthenia >2 |
1.40 |
0.44-4.49 |
0.566 |
Overall toxicity |
1.84 |
1.06-3.18 |
0.029* |
TYMS rs45445694 (Ref: 2R-3R /3R-3R) |
|||
Delay/reduction/withdrawal |
3.07 |
1.23-7.70 |
0.016* |
Diarrhea >2 |
0.54 |
1.15-1.90 |
0.336 |
Hand-foot syndrome >1 |
3.78 |
1.86-7.76 |
<0.001*** |
Hand-foot syndrome >2 |
3.63 |
1.18-11.22 |
0.025* |
Hematological toxicity >2 |
1.40 |
0.43-4.56 |
0.576 |
Asthenia >2 |
2.14 |
0.60-7-60 |
0.341 |
Overall toxicity |
0.97 |
0.49-1.93 |
0.937 |
TYMS rs34489327 (Ref: del-ins/ins-ins) |
|||
Delay/reduction/withdrawal |
0.99 |
0.36-2.71 |
0.981 |
Diarrhea >2 |
2.24 |
0.68-7.37 |
0.186 |
Hand-foot syndrome >1 |
0.16 |
0.02.1.23 |
0.078 |
Hand-foot syndrome >2 |
0.58 |
0.27-NA |
0.628 |
Hematological toxicity >2 |
0.43 |
0.37-NA |
0.429 |
Asthenia >2 |
2.13 |
0.60-7.60 |
0.241 |
Overall toxicity |
0.92 |
0.35-2.42 |
0.862 |
ENOSF1 rs2612091 (Ref: GA/AA) |
|||
Delay/Reduction/withdrawal |
2.21 |
0.92-5.27 |
0.074 |
Diarrhea >2 |
0.60 |
0.17-2.12 |
0.431 |
Hand-foot syndrome >1 |
2.28 |
1.10-4.76 |
0.027* |
Hand-foot syndrome >2 |
2.53 |
0.80-8.02 |
0.114 |
Hematological toxicity >2 |
0.62 |
0.14-2.84 |
0.541 |
Asthenia >2 |
3.15 |
0.94-10.57 |
0.063 |
Overall toxicity |
0.91 |
0.45-1.82 |
0.789 |
Adjusted (Padj) P values were calculated using binary logistic regression; *P < 0.05; ** P < 0.01; ***P <0.001; Ref: Reference.
DISCUSSION
Cancer cells are the target of chemotherapy. Adverse effects to cancer therapy result from damage to healthy cells. Selective targeting of cancer cells has been investigated to decrease the side effects of chemotherapy [23, 24]. However, to date, no valid approaches have been implemented in clinical practice. Identification and validation of genetic biomarkers with the aim of preventing severe ADRs to chemotherapeutic drugs could prove crucial in helping oncologists to select the best treatment for their patients. Since capecitabine is one of the most commonly used drugs in the treatment of CRC, a specific tool that would make it possible to predict the risk of toxicity for individual patients would be very useful. All the polymorphisms analyzed in this study have been associated with toxicity in the literature. However, current evidence is insufficient to apply those results in clinical practice. In this study, we demonstrated that functional genetic variants in TYMS, ENOSF1, and ABCB1 were associated with severe toxicity.
The genetic polymorphisms rs34489327 and rs45445694 in TYMS have been studied extensively to determine the patient’s response to fluoropyrimidine-based chemotherapy, although fewer studies have focused on their association with toxicity. In this cohort of capecitabine treated CRC patients we found a correlation between the TYMS polymorphism rs45445694 and HFS (grade >1 and grade >2). This result confirms previous findings, thus suggesting a clear association that could increase the level of evidence for using these biomarkers in clinical practice [11, 25-27].
ENOSF1 was recently proposed as a regulator of TYMS expression via antisense mechanisms because the sequences of both genes are complementary [11]. In addition, an association was found between ENOSF1 rs2612091 and capecitabine-related severe toxicity, mainly HFS. Therefore, a more relevant role has been suggested for ENOSF1—as opposed to TYMS—in capecitabine-related toxicity. We observed an increased risk of HFS grade >1 in patients carrying the G variant in homozygosis in ENOSF1 rs2612091. However, this association did not remain statistically significant when HFS grade >2 was analyzed. The results in our cohort suggest that TYMS rs45445694 (OR, 3.63) is a better predictor of severe HFS than ENOSF1 rs2612091 (OR, 2.53).
We did not find any significant associations between capecitabine-related toxicity and the MTHFR polymorphisms 677C>T and 1298A>C. It has been suggested that the effects of MTHFR are masked in cases of high serum levels of active folate [28]. Although capecitabine is not combined with leucovorin, greater folate intake has been related to a Mediterranean diet, and higher folate levels may explain the lack of effect of the MTHFR polymorphisms 677C>T and 1298A>C [29].
We also found that CDA 79A>C was linked to overall toxicity and observed a trend toward severe diarrhea. CDA plays an important role in the conversion of capecitabine to 5-FU, and variants in this gene have been related to capecitabine-induced toxicity [11, 22, 30]. CDA 79A>C has been associated with CDA activity and with toxicity to cytarabine, which is also metabolized by CDA [31, 32]. In contrast to our findings, those of other studies did not show a connection between this SNP and toxicity induced by capecitabine [11, 22] and fluoropyrimidines [33]. The variables considered responsible for these discrepant results include differences in study design, sample size, criteria for establishing a cut-off for severe ADRs, as well as variations in concomitant medication [34].
ABCB1 is an ATP-dependent drug efflux transporter for a huge variety of substrates [35]. Although capecitabine has not been clearly identified as one of them, ABCB1 expression has been associated with resistance to 5-FU in modified cell lines [36]. The authors showed associations between the haplotype ABCB1*1 and an increased probability of reducing the dose of capecitabine, delaying initiation of therapy, or withdrawing the drug altogether, as well as of diarrhea and overall toxicity. Nevertheless, we were not able to reproduce the association with HFS previously obtained in a smaller cohort [20].
A score covering several polymorphisms located in at least two genes has been designed to predict adverse reactions to fluoropyrimidines. Afzal et al used polymorphisms in TYMS and MTHFR to predict gastrointestinal toxicity in 5-FU-treated patients [19], while Rosmarin et al developed a test to predict overall toxicity with polymorphisms in TYMS and DPYD in capecitabine-treated patients [11]. Jennings et al suggested a predictive test for early adverse events by analyzing TYMP and DPYD variants as a signature (sensitivity of 45.5% and a positive predictive value of 39.9%) [33]. Our results for predicting overall toxicity using the CDA-ABCB1 score are similar to those obtained in the studies, although they do not take into consideration the main factor contributing to toxicity in fluoropyrimidine-based treatments, namely, DPYD variants. All other authors have included DPYD variants in their scores. In our preliminary analyses, we genotyped three SNPs in DPYD, namely, rs67376798, rs55886062, and rs3918290. However, we found only two patients who were heterozygous for rs67376798, and they did not experience any relevant toxicity. Given the low frequency of DPYD variants and our limited sample size, we decided not to include DPYD variants in our score. We suggest that using CDA and ABCB1 polymorphisms could improve the performance of existing predictors for capecitabine-induced toxicity. Alternatively, the novel strategy of using gene-specific scores for different ADRs could help to improve the prediction of a specific ADR. Identifying patients with a very high risk for developing ADRs could be useful when investigating new strategies, such as decreasing chemotherapy dosage in order to reduce toxicity without decreasing efficacy.
In summary, polymorphisms in TYMS, CDA, ENOSF1, and ABCB1 are associated with ADRs to capecitabine-based chemotherapy in CRC patients. Polymorphisms in CDA and ABCB1 should be added to current models to predict overall toxicity. Therefore, genotyping of these variants could help with the implementation of pharmacogenetics in clinical practice.
METHODS
Patients
Samples from patients were provided by the oncology departments of two tertiary teaching hospitals (Hospital General Universitario Gregorio Marañón and Hospital Universitario Doce de Octubre) and by HGM BioBank, which is a member of ReTBioH. Samples were processed following current procedures and frozen immediately on reception.
The inclusion criteria were diagnosis of CRC (any stage), a previous capecitabine-containing regimen in any line of treatment, and age over 18 years. The exclusion criteria were as follows: noncompliance with any of the inclusion criteria, kidney or liver damage prior to treatment, and less than 2 months of treatment, unless higher-grade severe ADRs occurred (grade ≥2 for HFS and grade≥3 for the rest).
The demographic and clinical data collected included sex, age, colon or rectal cancer, disease stage, World Health Organization Performance Status score, treatment setting (adjuvant or metastatic), and other drugs received as part of the chemotherapy regimen. Capecitabine-related adverse reactions including nausea and vomiting, diarrhea, mucositis, hematological toxicity, anorexia, asthenia, and HFS were recorded and graded according to the Common Terminology Criteria for Adverse Events v4.0 (CTCAE) of the National Cancer Institute. All patients were genotyped for polymorphisms in the following genes: MTHFR, TYMS, CDA, ABCB1, and ENOSF1. Associations between the polymorphisms and the ADRs were analyzed. This study was conducted in accordance with the Helsinki Declaration and was approved by the ethics committees of Hospital General Universitario Gregorio Marañón (CEIC-A1) and Hospital Universitario Doce de Octubre (CEIC-A11). Informed consent was obtained from all patients.
DNA isolation
Genomic DNA was isolated from 200 µl of whole blood using the High Pure PCR template preparation kit (Roche Applied Sciences, Penzberg, Germany). The DNA concentration was measured using a NanoDrop spectrophotometer (ThermoScientific, Waltham, Massachusetts, USA).
Genotype analysis
The polymorphisms rs1801133 (MTHFR C677T), rs1801131 (MTHFR A1298C), rs1128503 (ABCB1 1236C>T), rs2032582 (ABCB1 2677G>T/A), rs1045642 (ABCB1 3435C>T), and rs2072671 (CDA 79C/T) were genotyped using SNaPshot, as previously described [20]. The oligonucleotides used in the multiplex PCR reaction were as follows: rs1801133-F TCA CAA AGC GGA AGA AGT, rs1801133-R GCC TCT CCT GAC TGT ATC, rs1801131-F CTT TGT GAC CAT TCC GGT T, rs1801131-R TTT GGG GAG CTG AAG GAC TA, rs1128503-F CTC GAC TCA CCA CAC CAA TG, rs1128503-R TAT CCT GTG TCT GTG AAT TGC C, rs2032582-F TAG TTT GAC TCA CCT TCC CGG, rs2032582-R GGC TAT AGG TTC CAG GCT TG, rs1045642-F CAT GCT CCC AGG CTG TTT AT, rs1045642-R GTA ACT TGG CAG TTT CAG TG, rs2072671-F CTG AAG CCT GAG TGT GTC CA, and rs2072671-R CCA TCC AAC TTC CTT CCT CA. The oligonucleotides used in the SNaPshot reactions were as follows: rs1801133SNAP AGA ATG TGT CAG CCT CAA AGA AAA AGC TGC GTG ATG ATG AAA TCG, rs1801131SNAP TCC GGT TTG GTT CTC CCG AGA GGT AAA GAA CAA AGA CTT CAA AGA CAC TT, rs1128503SNAP GCC CAC TCT GCA CCT TCA GGT TCA G, rs2032582SNAP GAC AAG CAC TGA AAG ATA AGA AAG AAC TAG AAG GT, rs1045642SNAP TGA CTC GAT GAA GGC ATG TAT GTT GGC CTC CTT TGC TGC CCT CAC, and rs2072671SNAP TTT TTC CTG AGT GTG TCC AGC AGC TGC TGG TTT GCT CCA AGG AGG CCA AG.
The SNP rs34489327 (TYMS 6del) was genotyped using PCR amplification length polymorphism analysis with the primers 5′ 6-FAM- CTCAAATCTGAGGGAGCTGAG 3′ and 5′ GCAGAACACTTCTTTATTATAG 3′ and capillary electrophoresis. Genomic DNA (20 ng) was used as a PCR template under the following conditions: 95°C for 5 min, 35 cycles of 95°C for 30 s, 60°C for 40 s, 72°C for 90 s, and a final extension of 72°C for 5 min. PCR products were purified with ExoSapIt [20]. Purified PCR product (1 µL) was loaded onto an ABI Prism 3100 Genetic Analyzer and analyzed using PeakScan v1.0 (Life Technologies, Carlsbad, California, USA).
The SNP 45445694 in TYMS was analyzed by PCR amplification of the region containing it and analysis of the length of the amplification products. The oligonucleotides used in the PCR were 5′ GTGGCTCCTGCGTTTCCCCC 3′ and 5′ GCTCCGAGCCGGCCACAGGCA 3′. PCR parameters were the same as for TYMS 6del. The amplified product was purified using the High Pure PCR product purification kit and subsequently digested with the restriction enzyme HaeIII (both from Roche Applied Science, Penzberg, Germany). For this purpose, 8 µL of the amplification product was incubated with 1 µL of HaeIII and 1 µL of restriction buffer at 37°C for 1 h. After this period, the enzyme was inactivated by heating at 65°C for 15 min. Finally, the amplification products and the products resulting from the digestion were analyzed using the 2100 Bioanalyzer DNA1000 and the reagents kit (Agilent Technologies, Santa Clara, California, USA). Thus, the amplification of the region containing two repeats of 28 bp corresponds to a 214-bp band while 3 repeats would correspond to a 242-bp band.
ENOSF1 rs2612091 was genotyped using a TaqMan probe in a StepOne Plus Real Time PCR system (Life Technologies, Carlsbad, California, USA). Allele discrimination was performed using StepOne software v2.3.
Hardy-Weinberg equilibrium was analyzed to detect deviations in genotype frequency [37].
Statistical analysis
All data were analyzed using the Statistical Package for the Social Sciences v.15 (SPSS, Inc). A linear-by-linear association chi-squared test was used to study the univariate associations between polymorphisms and the grade of adverse events. A P value <0.05 was considered statistically significant. The odds ratio (OR) and 95% confidence intervals (CI) were reported in the multivariate logistic regression models for associations that were statistically significant in the univariate analysis. For ABCB1 variants, we performed a haplotype-based test. The variables analyzed in the models included genotype, sex, tumor stage, and hospital.
Based on the results of the multivariate analysis, a score test was designed based on the number of risk alleles for overall toxicity in ABCB1 and CDA. Negative and positive predictive values, specificity, and sensitivity were calculated.
ACKNOWLEDGEMENTS
We would like to thank Silvia Caballero for technical support. We are particularly grateful to the study patients for their participation, to the Medical Oncology Departments, and HGM BioBank integrated in RETICS, which is a member of ReTBioH. We also acknowledge the collaborating Centres for their generous gifts of the clinical samples used in this study.
Funding
The study was supported by grants from Instituto de Salud Carlos III (PI12/00056) and “Programa Miguel Servet II MSII13/00008” awarded to Luis A. López-Fernández, and supported with FEDER funds from European Union. The HGM BioBank, which is a member of ReTBioH, is supported by Instituto de Salud Carlos III, Spanish Science and Innovation Ministry (Grant nº PT13/0010/0028), and Fundación para la investigación y prevención del SIDA en España (FIPSE).
Conflict of interest statement
The authors declare that they have no conflicts of interest.
REFERENCES
1 Hirsch BR, Zafar SY. Capecitabine in the management of colorectal cancer. Cancer Manag Res 2011; 3: 79–89.
2 Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 2010; 127: 2893–2917.
3 Li LS, Morales JC, Veigl M, Sedwick D, Greer S, Meyers M, Wagner M, Fishel R, Boothman DA. DNA mismatch repair (MMR)-dependent 5-fluorouracil cytotoxicity and the potential for new therapeutic targets. Br J Pharmacol 2009; 158: 679–92.
4 Shastry BS. Pharmacogenetics and the concept of individualized medicine. Pharmacogenomics J 2006; 6: 16–21.
5 Saif MW, Katirtzoglou NA, Syrigos KN. Capecitabine: an overview of the side effects and their management. Anticancer Drugs 2008; 19: 447–464.
6 Kadoyama K, Miki I, Tamura T, Brown JB, Sakaeda T, Okuno Y. Adverse event profiles of 5-fluorouracil and capecitabine: data mining of the public version of the FDA Adverse Event Reporting System, AERS, and reproducibility of clinical observations. Int J Med Sci 2012; 9: 33–9.
7 Braun MS, Richman SD, Thompson L, Daly CL, Meade AM, Adlard JW, Allan JM, Parmar MKB, Quirke P, Seymour MT. Association of molecular markers with toxicity outcomes in a randomized trial of chemotherapy for advanced colorectal cancer: the FOCUS trial. J Clin Oncol 2009; 27: 5519–28.
8 Strimpakos a S, Syrigos KN, Saif MW. Pharmacogenetics and biomarkers in colorectal cancer. Pharmacogenomics J 2009; 9: 147–60.
9 Caudle KE, Thorn CF, Klein TE, Swen JJ, McLeod HL, Diasio RB, Schwab M. Clinical Pharmacogenetics Implementation Consortium guidelines for dihydropyrimidine dehydrogenase genotype and fluoropyrimidine dosing. Clin Pharmacol Ther 2013; 94: 640–5.
10 Offer SM, Fossum CC, Wegner NJ, Stuflesser AJ, Butterfield GL, Diasio RB. Comparative functional analysis of DPYD variants of potential clinical relevance to dihydropyrimidine dehydrogenase activity. Cancer Res 2014; 74: 2545–54.
11 Rosmarin D, Palles C, Church D, Domingo E, Jones A, Johnstone E, Wang H, Love S, Julier P, Scudder C, Nicholson G, Gonzalez-Neira A, Martin M et al. Genetic markers of toxicity from capecitabine and other fluorouracil-based regimens: investigation in the QUASAR2 study, systematic review, and meta-analysis. J Clin Oncol 2014; 32: 1031–9.
12 Tsunoda A, Nakao K, Watanabe M, Matsui N, Ooyama A, Kusano M. Associations of various gene polymorphisms with toxicity in colorectal cancer patients receiving oral uracil and tegafur plus leucovorin: A prospective study. Ann Oncol 2011; 22: 355–361.
13 Boige V, Mendiboure J, Pignon J-P, Loriot M-A, Castaing M, Barrois M, Malka D, Trégouët D-A, Bouché O, Le Corre D, Miran I, Mulot C, Ducreux M et al. Pharmacogenetic assessment of toxicity and outcome in patients with metastatic colorectal cancer treated with LV5FU2, FOLFOX, and FOLFIRI: FFCD 2000-05. J Clin Oncol 2010; 28: 2556–2564.
14 McLeod HL, Sargent DJ, Marsh S, Green EM, King CR, Fuchs CS, Ramanathan RK, Williamson SK, Findlay BP, Thibodeau SN, Grothey A, Morton RF, Goldberg RM. Pharmacogenetic predictors of adverse events and response to chemotherapy in metastatic colorectal cancer: results from North American Gastrointestinal Intergroup Trial N9741. J Clin Oncol 2010; 28: 3227–3233.
15 Etienne-Grimaldi MC, Milano G, Maindrault-Gœbel F, Chibaudel B, Formento JL, Francoual M, Lledo G, André T, Mabro M, Mineur L, Flesch M, Carola E, De Gramont A. Methylenetetrahydrofolate reductase (MTHFR) gene polymorphisms and FOLFOX response in colorectal cancer patients. Br J Clin Pharmacol 2010; 69: 58–66.
16 Ruzzo a, Graziano F, Loupakis F, Santini D, Catalano V, Bisonni R, Ficarelli R, Fontana a, Andreoni F, Falcone a, Canestrari E, Tonini G, Mari D et al. Pharmacogenetic profiling in patients with advanced colorectal cancer treated with first-line FOLFIRI chemotherapy. Pharmacogenomics J 2008; 8: 278–88.
17 Thomas F, Motsinger-Reif A, Hoskins JM, Dvorak a, Roy S, Alyasiri a, Myerson RJ, Fleshman JW, Tan BR, McLeod HL. Methylenetetrahydrofolate reductase genetic polymorphisms and toxicity to 5-FU-based chemoradiation in rectal cancer. Br J Cancer 2011; 105: 1654–62.
18 Kristensen MH, Pedersen PL, Melsen G V, Ellehauge J, Mejer J. Variants in the dihydropyrimidine dehydrogenase, methylenetetrahydrofolate reductase and thymidylate synthase genes predict early toxicity of 5-fluorouracil in colorectal cancer patients. J Int Med Res 2010; 38: 870–83.
19 Afzal S, Gusella M, Vainer B, Vogel UB, Andersen JT, Broedbaek K, Petersen M, Jimenez-Solem E, Bertolaso L, Barile C, Padrini R, Pasini F, Jensen SA et al. Combinations of polymorphisms in genes involved in the 5-Fluorouracil metabolism pathway are associated with gastrointestinal toxicity in chemotherapy-treated colorectal cancer patients. Clin Cancer Res 2011; 17: 3822–3829.
20 Gonzalez-Haba E, García MI, Cortejoso L, López-Lillo C, Barrueco N, García-Alfonso P, Alvarez S, Jiménez JL, Martín ML, Muñóz-Fernández M a, Sanjurjo M, López-Fernández L a. ABCB1 gene polymorphisms are associated with adverse reactions in fluoropyrimidine-treated colorectal cancer patients. Pharmacogenomics 2010; 11: 1715–23.
21 Cortejoso L, García MI, García-Alfonso P, Grávalos C, Robles L, González-Haba E, Sanjurjo M, López-Fernández L a. [Validation of genetic polymorphisms associated to the toxicity of chemotherapy in colorectal cancer patients]. Farm Hosp 2014; 38: 283–90.
22 Caronia D, Martin M, Sastre J, de la Torre J, García-Sáenz JA, Alonso MR, Moreno LT, Pita G, Díaz-Rubio E, Benítez J, González-Neira A. A polymorphism in the cytidine deaminase promoter predicts severe capecitabine-induced hand-foot syndrome. Clin Cancer Res 2011; 17: 2006–13.
23 Blagosklonny M V, Darzynkiewicz Z. Cyclotherapy: protection of normal cells and unshielding of cancer cells. Cell Cycle; 1: 375–82.
24 Blagosklonny M V. How cancer could be cured by 2015. Cell Cycle 2005; 4: 269–78.
25 Shahrokni A, Rajebi MR, Saif MW. Toxicity and efficacy of 5-fluorouracil and capecitabine in a patient with TYMS gene polymorphism: A challenge or a dilemma? Clin Colorectal Cancer 2009; 8: 231–4.
26 Jennings BA, Kwok CS, Willis G, Matthews V, Wawruch P, Loke YK. Functional polymorphisms of folate metabolism and response to chemotherapy for colorectal cancer, a systematic review and meta-analysis. Pharmacogenet Genomics 2012; 22: 290–304.
27 Meulendijks D, Dewit L, Tomasoa NB, van Tinteren H, Beijnen JH, Schellens JHM, Cats A. Chemoradiotherapy with capecitabine for locally advanced anal carcinoma: an alternative treatment option. Br J Cancer 2014. doi:10.1038/bjc.2014.467.
28 Van Huis-Tanja LH, Gelderblom H, Punt CJA, Guchelaar H-J. MTHFR polymorphisms and capecitabine-induced toxicity in patients with metastatic colorectal cancer. Pharmacogenet Genomics 2013; 23: 208–18.
29 Monteagudo C, Mariscal-Arcas M, Palacin A, Lopez M, Lorenzo ML, Olea-Serrano F. Estimation of dietary folic acid intake in three generations of females in Southern Spain. Appetite 2013; 67: 114–8.
30 Loganayagam A, Arenas Hernandez M, Corrigan A, Fairbanks L, Lewis CM, Harper P, Maisey N, Ross P, Sanderson JD, Marinaki AM. Pharmacogenetic variants in the DPYD, TYMS, CDA and MTHFR genes are clinically significant predictors of fluoropyrimidine toxicity. Br J Cancer 2013; 108: 2505–15.
31 Carpi FM, Vincenzetti S, Ubaldi J, Pucciarelli S, Polzonetti V, Micozzi D, Mignini F. CDA gene polymorphisms and enzyme activity : genotype – phenotype relationship in an Italian – Caucasian population R esearch A rticle. 2013; : 769–781.
32 Abraham A, Varatharajan S, Abbas S, Zhang W, Shaji R V, Ahmed R, Abraham A, George B, Srivastava A, Chandy M, Mathews V, Balasubramanian P. Cytidine deaminase genetic variants influence RNA expression and cytarabine cytotoxicity in acute myeloid leukemia. Pharmacogenomics 2012; 13: 269–82.
33 Jennings BA, Loke YK, Skinner J, Keane M, Chu GS, Turner R, Epurescu D, Barrett A, Willis G. Evaluating predictive pharmacogenetic signatures of adverse events in colorectal cancer patients treated with fluoropyrimidines. PLoS One 2013; 8: e78053.
34 Cortejoso L, López-Fernández L a. Pharmacogenetic markers of toxicity for chemotherapy in colorectal cancer patients. Pharmacogenomics 2012; 13: 1173–91.
35 Nieto Montesinos R, Béduneau A, Pellequer Y, Lamprecht A. Delivery of P-glycoprotein substrates using chemosensitizers and nanotechnology for selective and efficient therapeutic outcomes. J Control Release 2012; 161: 50–61.
36 Chockalingam S, Ghosh SS. Amelioration of cancer stem cells in macrophage colony stimulating factor-expressing U87MG-human glioblastoma upon 5-fluorouracil therapy. PLoS One 2013; 8: e83877.
37 Rodriguez S, Gaunt TR, Day INM. Hardy-Weinberg equilibrium testing of biological ascertainment for Mendelian randomization studies. Am J Epidemiol 2009; 169: 505–14.

