INTRODUCTION
Anemia is a well-known consequence of chronic kidney disease (CKD), and the third National Health and Nutrition Examination Survey has shown that the prevalence of anemia increases in subjects with an estimated glomerular filtration rate (eGFR) of less than 60 mL/min/1.73 m2 [1]. The lower the hematocrit, the greater the risk of developing ESRD [2]. Hypoxia of the tubular cells plays an important role in tubulointerstitial damage associated with CKD, and in the progression of renal failure [3]. Three randomized studies have suggested that anemia correction may slow the progression of CKD [4-6]. Anemia commonly contributes to a poor quality of life, and also significantly reduces patient survival.
Patients with advanced CKD encounter anemia due to insufficient erythropoietin (EPO) production by the peritubular fibroblasts of the diseased kidney [7]. Expression of EPO is controlled by hypoxia-inducible transcription factor (HIF), which is a heterodimeric transcription factor composed of a hypoxia-inducible alpha-subunit, and a constitutively expressed beta-subunit. Indoxyl sulfate (IS), a colon derived uremic solute, has been reported to contribute to the development of uremic symptoms [8, 9]. Recent studies have indicated that serum IS level is a valuable marker for predicting atherosclerosis, cardiovascular disease, renal function decline, and peripheral arterial disease in patients with advanced chronic kidney disease [10-14]. Furthermore, IS may inhibit the HIF system in addition to aggravating the hypoxia of the kidney, by causing functional impairment of the HIF-1-alpha C-terminal transactivation domain [15]. As revealed by a cell and animal study, IS was able to impair oxygen-sensing in EPO-producing cells, suppressing renal EPO messenger RNA expression and the plasma EPO levels [16]. These results showed the toxic effects of IS on EPO synthesis in vitro. However, whether this effect can be seen in humans remains unclear. The aim of our study was to investigate the role of IS in renal anemia in a CKD cohort and animal model. The results of our study reveal the relationship between IS and EPO levels.
RESULTS
Patient characteristics
One hundred and thirteen stable patients with CKD were recruited in the study, and the demographic and clinical characteristics of the patients are given in Table 1 . This study population consisted of 57 males (50.4%) and 56 females (49.6%), with a mean age of 57.1 ± 14.4 years. Fifty-six patients had diabetes mellitus (49.6%), 55 patients had hypertension (48.7%), and 48 patients had cardiovascular disease (42.8%). Among them, 17 patients (15%) were classified as CKD stage-2, 27 patients (23.8%) were classified as CKD stage-3, 41 patients (36.2%) were classified as CKD stage-4, and 28 patients (24.8%) were classified as CKD stage-5. The clinical parameters were as follows: albumin: 4.5 ± 0.5 (g/dL); hematocrit: 26.8 ± 6.0 (%); blood urine nitrogen: 57.3 ± 22.5 (mg/dL); creatinine: 3.1 ± 2.4 (mg/dL); estimated GFR: 20.9 ± 23.7 (ml/min/1.73m2); calcium: 9.1 ±0.8 (mg/dL); phosphate: 4.3 ± 1.1 (mg/dL); c-reactive protein: 0.5 ± 0.3 (mg/dL); ferrous: 64.3 ± 23.7 (μg/dL), ferritin: 196.9 ± 95.2 (ng/mL) and EPO: 12.2 ±11.9 (mIU/ml). The total IS (T-IS) and free IS (F-IS) were 15.8 ± 6.9 and 0.22 ± 0.6 mg/L, respectively.
Table 1: Baseline characteristics of the study patients.
Variables |
All (n=113) |
|---|---|
Age (yr) |
57.1 ± 14.4 |
Male (%) |
50.4 |
Diabetes Mellitus (%) |
49.6 |
Hypertension (%) |
48.7 |
Cardiovascular disease (%) |
42.8 |
SBP (mmHg) |
144.6 ± 16.1 |
DBP (mmHg) |
75.7 ± 10.5 |
CKD stage (%) |
|
2 |
15.2 |
3 |
23.8 |
4 |
36.2 |
5 |
24.8 |
Albumin (g/dL) |
4.5 ± 0.5 |
Hematocrit (%) |
26.8 ± 6.0 |
BUN (mg/dL) |
57.3 ± 22.5 |
Creatinine (mg/dL) |
3.1 ± 2.4 |
eGFR (ml/min/1.73m2) |
20.9 ± 23.7 |
Calcium (mg/dL) |
9.1 ± 0.8 |
Phosphate (mg/dL) |
4.3 ± 1.1 |
CRP (mg/dL) |
0.51 ± 0.32 |
Fe (ug/dL) |
64.3 ± 23.7 |
Ferritin (ng/mL) |
196.9 ± 95.2 |
Free IS (mg/L) |
0.2 ± 0.6 |
Total IS (mg/L) |
15.8 ± 6.9 |
EPO (mIU/ml) |
12.2 ± 11.9 |
PS: values were expressed as mean ± SD unless otherwise defined.
Abbreviation: BUN: blood Urea Nitrogen; eGFR: estimated glomerular filtration rate; CRP: C-reactive protein; Fe: ferrous; IS: indoxyl sulfate.; EPO: erythropoietin.
Correlation between EPO and the clinical variables
The results of the simple and multiple stepwise linear regression analyses for EPO as a dependent variable are shown in Table 2 . On the simple linear regression analyses, the EPO was significantly correlated to hematocrit (B=0.02, P=0.032), free IS (B=-0.50, P=0.021), and total IS (B=-0.03, P=0.041). Because of collinearity between free and total IS, we used hematocrit and free IS in model I, and hematocrit and total IS in model II. After adjusting for other independent parameters, only serum free IS (B=-0.55, p=0.008) was significantly associated with EPO levels on multiple variable analysis (model I). There was also a significantly negative correlation between EPO and total IS (B=-0.04, p=0.013) in model II. Moreover, the agreement between EPO and total IS was analyzed by Pearson’s correlation. EPO levels were significantly correlated with total IS (r=-0.22, p=0.041) (Figure 1A) and free IS (r=-0.25, p=0.021)( Figure 1B ).
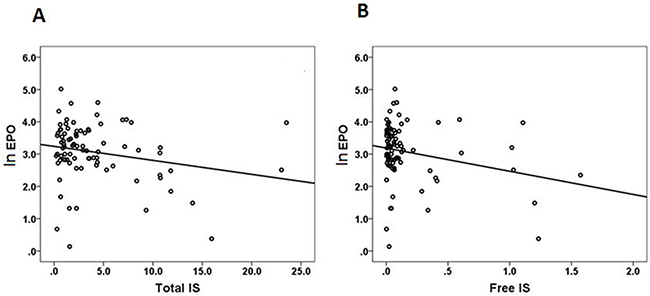
Figure 1: Agreement between EPO and IS analyzed by Pearson’s correlation. (A) ln EPO vs T-IS, r=-0.22, p=0.041 (B) ln EPO vs F-IS, r=-0.25, p=0.021.
Table 2: Simple and multiple stepwise linear regression analyses for EPO as a dependent.
Simple variable analysis |
Multiple variables analysis(Model I) |
Multiple variables analysis(Model II) |
|||||||
|---|---|---|---|---|---|---|---|---|---|
B |
95% C.I. |
p |
B |
95% C.I. |
p |
B |
95% C.I. |
p |
|
Sex(male/female) |
0.05 |
-0.22~0.33 |
0.693 |
||||||
Age(y) |
-0.01 |
-0.01~0.01 |
0.915 |
||||||
DM (yes/no) |
-0.16 |
-0.44~0.12 |
0.252 |
||||||
Hypertension(yes/no) |
0.04 |
-0.23~0.31 |
0.771 |
||||||
CVD(yes/no) |
0.29 |
-0.07~0.65 |
0.114 |
||||||
CKD stages |
-0.10 |
-0.22~0.01 |
0.086 |
||||||
Hematocrit(%) |
0.02 |
0.00~0.05 |
0.032 |
||||||
Albumin (g/dL) |
0.02 |
-0.33~0.38 |
0.906 |
||||||
BUN(mg/dL) |
-0.01 |
-0.01~0.00 |
0.227 |
||||||
Creatinine(mg/dL) |
-0.05 |
-0.12~0.02 |
0.132 |
||||||
eGFR(ml/min/1.73m2) |
0.01 |
0.00~0.01 |
0.409 |
||||||
Sodium (mg/dL) |
0.03 |
-0.02~0.08 |
0.280 |
||||||
Potassium(mg/dL) |
-0.12 |
-0.37~0.13 |
0.335 |
||||||
Calcium(mg/dL) |
0.06 |
-0.14~0.25 |
0.553 |
||||||
Phosphate(mg/dL) |
-0.08 |
-0.24~0.09 |
0.349 |
||||||
CRP(mg/dL) |
0.03 |
-0.50~0.56 |
0.904 |
||||||
Fe(ug/dL) |
-0.01 |
-0.01~0.01 |
0.727 |
||||||
Ferritin(ng/mL) |
-0.01 |
0.00~0.00 |
0.879 |
||||||
Free IS(mg/L) |
-0.50 |
-0.92~-0.08 |
0.021 |
-0.55 |
-0.96~-0.15 |
0.008 |
|||
Total IS(mg/L) |
-0.03 |
-0.06~0.00 |
0.041 |
-0.04 |
-0.06~-0.01 |
0.013 |
|||
Association between IS and EPO expression status in the animal study
All animals were divided into three groups: control-sham, CKD, and CKD + AST-120 groups. The serum total and free IS levels were increased in CKD group (6.73 ± 5.12, 1.10 ± 1.54 mg/L, respectively), and were lowered when treated with AST-120 (4.33 ± 2.70, 0.46 ± 0.39 mg/L, respectively). In Figure 2A , the EPO mRNA expression was inhibited in CKD rats versus the control group (P<0.01), and significantly increased when treated with AST-120 versus the CKD group, measured by quantitative RT-PCR (P<0.05). However, the EPO protein expression analyzed using the western blotting method was also decreased in CKD group, and this inhibition in CKD group was reversible in CKD + AST-120 group (P<0.05) (Figure 2B). These data indirectly demonstrated that IS may be involved in the development of renal anemia.
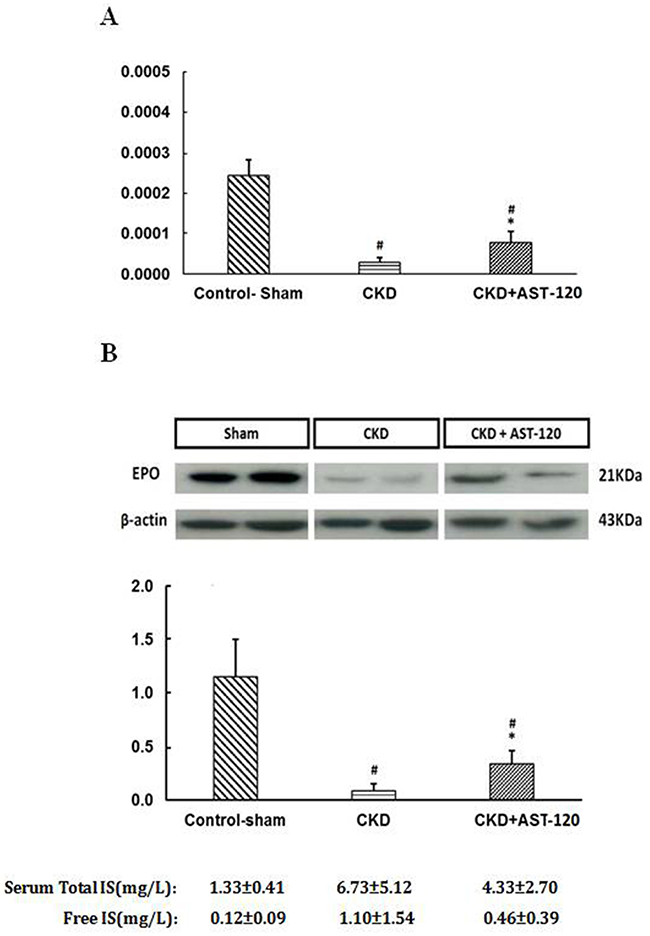
Figure 2: The EPO expression status in an animal model. (A) Real-time quantitative PCR was performed to assess the change of EPO mRNA expression in control-sham, CKD and CKD+AST-120 group. EPO mRNA was suppressed in CKD rat. It was restored in rats with feeding AST-120 by lowering serum IS. (B) Western blot analysis. EPO protein expression was also decreased significantly in CKD rat. AST-120 could reverse the inhibition of EPO expression in CKD. n=5 for each, #, p<0.01 compared to control-sham group, *, p<0.05 compared CKD group.
DISCUSSION
The novel finding of the present study is that total IS has been significantly associated with the EPO levels in a CKD cohort. Moreover, the inhibition of EPO expression by IS has also been indirectly confirmed on the animal model. These findings further support the role of IS in the regulation and development of renal anemia in CKD patients.
The main cause of renal anemia in advanced CKD is a deficient production of EPO. According to the study reported by Bernhardt, et al., the pharmacological activation of hypoxia-inducible transcription factors (HIFs) in dialysis patients increases the EPO production, suggesting that the desensitization of the oxygen-sensing mechanism is responsible for the development of renal anemia [17]. The HIF is a master transcriptional regulator of the adaptive responses against hypoxia, and regulates the expression of EPO, angiogenic factors, glycolytic enzymes, glucose transporters, anti-oxidative enzymes, and other protective mechanisms [18].
IS is protein-bound uremic toxin derived from the fermentation of tryptophan by the intestinal flora. Evidence suggests that the kidney, liver function and gut determine the serum IS levels [12, 19, 20]. Moreover, IS may be involved in the regulation of renal anemia. Chiang et al. provided evidence that links IS to the impairment of EPO production through a reduced nuclear accumulation of HIFs following hypoxia in HepG2 cells [16]. The finding that the administration of indole leads to the suppression of renal EPO mRNA expression and decreases plasma EPO levels, is also confirmed on the animal model. Another report suggest that IS could suppress EPO expression in an HIF-independent manner, by inducing endoplasmic reticulum stress and thereby contributing to the progression of cellular damage in tubular epithelial cells [21]. Moreover, IS also suppresses the EPO receptor Akt signaling pathway in the extra-hematopoietic cells, leading to EPO resistance in the human umbilical vein endothelial cells [22]. Another in vitro study has reported that IS stimulates suicidal erythrocyte death (eryptosis), an effect in large part due to extracellular Ca2+ entry with subsequent stimulation of cell shrinkage and cell membrane scrambling [23].
These basic studies suggest that IS plays a role in the development of renal anemia.
However, there is little direct evidence to demonstrate the relationship between IS levels and the EPO concentration in humans. One previous clinical study has revealed that in hemodialysis patients the IS levels are increased and the patients require a higher dosage of EPO [24]. Another randomized, fixed dose, controlled study has reported that in advanced CKD patients the adjuvant administration of AST-120 (an active charcoal that can effectively absorb IS) is associated with an improvement in the renal function and hemoglobin levels, versus the use of the EPO stimulating agent alone [25]. This suggests that using AST-120 improves renal anemia not only by reducing IS, but also by improving the EPO production. These two studies have provided indirect evidence linking the correlation of IS and EPO levels.
To our knowledge, our study is the first to demonstrate the relationship between IS and the EPO concentration in humans, after adjusting for other confounding variables. Our results provide a direct evidence of the association of IS and the EPO levels in CKD patients. Moreover, our animal model also confirmed that lowering the uremic toxin can increase the EPO expression in CKD rat when treated with AST-120. It indirectly reflects that IS may play an inhibitory role in the EPO production. The inadequate synthesis of EPO can also be reversed by reducing IS with AST-120.
One study published by Palm, et al. has reported that IS dysregulates the oxygen metabolism and enhances the oxygen consumption in the tubular cells, thus aggravating tubule interstitial hypoxia, which can be reversed by AST-120 in the remnant kidney [26]. This suggests that AST-120 may help in preserving more renal EPO-producing cells, enhancing the EPO activity, and ameliorating the EPO resistance, by absorbing IS. These evidences reinforce the association between the uremic status and inadequate synthesis of EPO. Thus, these findings might have positive impact on the future CKD management. In addition to adequate dose of EPO injection, how to lower serum IS is also an essential step to synergistically improve renal anemia in patients with advanced CKD.
There are some limitations of our study. First, the number of patients is small, as only patients from one regional hospital were included in the study. Second, the association between IS and the nuclear fraction of HIF-1-alpha after AST-120 administration was not confirmed in this study. Third, our study is a cross-sectional design, so we cannot answer the questions about the definite mechanism and interactions between IS and the EPO levels. Fourth, we did not measure p-cresyl sulfate (PCS), another protein-bound uremic solute in this study. Although, both IS and PCS exhibit a similar effect on clinical outcomes [27–29], there are some differences between them. There is also little evidence from in vitro studies indicating that PCS dysregulates HIFs. It is unclear if PCS is also involved in the regulation of renal anemia. Thus, further prospective investigations are required to clarify this intriguing question.
In conclusion, our study reveals that the serum EPO levels are significantly and negatively associated with serum IS in CKD patients. Our animal study has also indirectly demonstrated the association between IS and the EPO expression. These findings further support the idea that IS is a key factor in renal anemia and the regulation of the EPO production in CKD.
MATERIALS AND METHODS
Subjects
This study recruited 113 patients with stable CKD (stage 2, 3, 4 and 5) from our institution’s outpatient department. Patients with acute infection, liver cirrhosis, malignancy, or those who were younger than 18 years of age were excluded from the study. The etiology of CKD among the patients included type-2 diabetic nephropathy (49.6%), and chronic glomerular nephritis (50.4%). The study included human and animal research, and was performed according to the principles of the Declaration of Helsinki, and approved by the ethics committee of the Mackay Memorial Hospital in Taiwan. All patients gave written Informed consent before the study was initiated.
Laboratory assessment
After enrollment, fasting blood samples were obtained from all patients with CKD. The estimated glomerular filtration rate (eGFR) was calculated using the Modification of Diet in Renal Disease equation as follows: 175 x (Scr)-1.154 x (Age)-0.203 x (0.742 if female). The following data were collected: blood urea nitrogen (BUN)(mg/dL), creatinine (mg/dL), hemoglobin (g/dL), hematocrit (%), calcium (Ca), (mg/dL), phosphate (mg/dL), albumin (g/dL), IS (mg/L) and EPO (μIU/mL). EPO was measured with a human ELISA Kit (R&D systems, USA). The bromocresol green method was used to determine the serum albumin levels.
Measurement of IS
Serum IS levels were analyzed using LC-MS/MS (4000 QTRAP, USA). Briefly, serum samples were prepared and deproteinized by heat denaturation. The free concentrations of IS were measured in serum ultrafiltrates, which were obtained using Microcon YM-30 separators (Millipore, Billerica, MA, USA), followed by the same sample preparation and analysis methods used for analyzing the serum levels of IS: High Performance Liquid Chromatography (HPLC) was performed at room temperature using a dC18 column (3.0 × 50 mm, Atlantis, Waters). The buffers used were (A) 0.1% formic acid and (B) 1 mM NH4OAc plus 0.1% formic acid in 100% acetonitrile. The flow rate was 0.6 mL/min with a 3.5-min gradient cycling from 90% A/10% B to 10% A/90% B. Under these conditions, IS was eluted at 2.48 minutes. Standard curves for IS were set at 1, 5, 10, 50, 250, 500 and 1,000 μg/L, and correlated with the serum samples, with average r2 values of 0.996 ± 0.003. These samples were diluted if the IS concentration exceeded a standard curve. Quantitative results were obtained and calculated in terms of their concentrations (mg/L). The sensitivity of this assay was 1 μg/L for IS.
Animals
CKD rat (5/6 nephrectomy)
Six-week-old male Sprague-Dawley rats (weight 160–180 g) were purchased from Lasco Taiwan Inc. At 8 weeks of age (weight 210–230 g), the rats were anesthetized with Zoletil 50/Xylazine (6 mg/1.3 mg/kg body weight), and the upper and lower thirds of the right kidney were removed. One week after the first operation, the left kidney was removed, leaving approximately 1/6 of the total renal mass. Sham operated rats were used as controls. The animals had free access to tap water and standard chow. Two weeks after the 5/6 nephrectomy, the rats were randomized to receive either AST-120 (5/6-nephrectomy + AST-120) or no treatment (5/6-nephrectomy) for 4-weeks. The AST-120 (Kremezin, Kureha Pharmaceuticals, Tokyo, Japan) was administrated post-operatively in the chow at 8% w/w. The remnant kidneys were removed after perfusion at the end of experiment, for EPO gene and protein expression studies. All animal were divided into three groups: control-sham, CKD, and CKD + AST120 groups.
The local committee for care and use of laboratory animals at the Mackay Memorial Hospital approved this study. In this study, 3 of 18 rats died after surgery, while the other rats survived until the end of study. All experimental procedures of animal were performed according to Institutional Animal Care and Use Committee (IACUC) policy. All rats were monitored every day and were treated with meperidine (0.3-0.5 mg/100g body weight for pain control (if need) based on the physiological parameters as following: 1. Avoidance, vocalization and aggressiveness (mainly if the animal cannot escape). 2. Spontaneous activities are reduced. The animal is isolated from the social group. 3. Altered gait. 4. Hunched posture. 5. Pilo-erection. 6. Reduced grooming; dark-red stain around the eyes and at nostrils. 7. Reduced appetite and subsequent weight loss. 8. Increased respiration rate. 9. Failure to explore cage when disturbed. We followed IACUC policy by using CO2 Euthanasia as the method of sacrifice at the end of study.
Quantitative reverse transcriptase-polymerase chain reaction (RT-PCR)
Total RNA was extracted from the renal cortex trimmed from fresh frozen renal tissue using the Trizol Reagent (Life Technologies), as per the manufacturer recommended protocol. The renal cortex (150 mg) was homogenized with 1 mL Trizol, and the RNA-containing aqueous phase was obtained by centrifugation. Following the precipitation of RNA by centrifugation with 0.5 volume isopropanol and purification with 75% ethanol, the extracted total RNA was dissolved in 80 μL nuclease-free deionized distilled water, and stored at -20°C.The concentration and purity of the RNA were determined by using the Nanodrop Spectrophotometer (Thermo Scientific). First-strand cDNA was synthesized from the template RNA (1 μg) using Transcriptor First Strand cDNA Synthesis Kit (Roche), according to the manufacturer recommended protocol. A real-time PCR was performed using a 2 μL template in a 20 μL reaction, containing 1 μM of each primer and 10 μL of Light Cycler Fast Start Universal SYBR Green Master (ROX) (Roche).
The following Lightcycler®96 conditions were used: Pre-incubation at 95°C for 10 minutes, followed by 40 cycles with 2-step amplification at 95°C for 10 seconds, 60°C for 30 seconds, melting at 95°C for 10 seconds, 65°C for 60 seconds, 97°C for 1 second, and cooling at 37°C for 30 seconds (Roche). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a housekeeping gene, to normalize the gene expression data.
The primer sequences were as follows: TACGTAGCCTCACTTCACTGCTT and GCAGAA AGTATCCGCTGTGAGTGTTC. The PCR was conducted in triplicate for each sample.
Western blotting analysis
For determining the EPO protein, 30 μg nuclear extract protein was loaded per lane.
These protein samples were separated by electrophoresis on an 8% SDS-polyacrylamide gel, followed by an electro-transfer to polyvinylidene difluoride membranes (GE Healthcare Bio-Sciences, Little Chalfont, UK). The transferred membranes were blocked with 5% fat-free skim milk in tris-buffered saline, with 0.01% Tween 20 for 60 minutes at room temperature. The membrane was incubated with rabbit polyclonal anti-EPO antibody (diluted 1:1000 in PBS/5% skim milk; Novus. Biologicals), overnight at 4°C. Horseradish peroxidase (HRP) conjugated anti-rabbit IgG (Promega) was then used as the secondary antibody. The immunoreactive protein was visualized by using the chemiluminescent protocol (ECL, GE HealthcareBio-Sciences). Anti-actin rabbit polyclonal antibody (1:1000; Sigma-Aldrich) was used for the calibration. We employed beta-actin as an internal control.
Statistical analysis
The human demographic data were expressed as the mean ± SD. Independent clinical variables were ln transformed, if not normally distributed. The agreement between EPO and IS were analyzed by Pearson’s correlation. To assess the influence of the tested parameters, the simple and multiple stepwise linear regression analyses (we put all significant variables in a univariate linear regression analysis) were used to evaluate EPO as a dependent variable. In animal studies, data were expressed as mean ± SEM. One-way ANOVA with Tukey’s post hoc test was used to compare the differences between the control and study groups in the animal model. A P value of less than 0.05 was considered to be statistically significant. All statistical analyses were conducted using the SPSS ver. 21.0 software program (IBM, Armonk, New York, USA).
ACKNOWLEDGEMENTS AND FUNDING
We would like to thank all the patients who were involved in this study. This study was supported in part by Mackay Memorial Hospital (MMH-106-31).
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
REFERENCES
1. Nangaku M, Mimura I, Yamaguchi J, Higashijima Y, Wada T, Tanaka T. Role of uremic toxins in erythropoiesis-stimulating agent resistance in chronic kidney disease and dialysis patients. J Ren Nutr. 2015; 25: 160-163.
2. Iseki K, Ikemiya Y, Iseki C, Takishita S. Haematocrit and the risk of developing end-stage renal disease. Nephrol Dial Transplant. 2003; 18: 899-905.
3. Rossert J, Froissart M. Role of anemia in progression of chronic kidney disease. Semin Nephrol. 2006; 26: 283-289.
4. Roth D, Smith RD, Schulman G, Steinman TI, Hatch FE, Rudnick MR, Sloand JA, Freedman BI, Williams WW Jr, Shadur CA, Benz RL, Teehan BP, Revicki DA, et al. Effects of recombinant human erythropoietin on renal function in chronic renal failure predialysis patients. Am J Kidney Dis. 1994; 24: 777-784.
5. Kuriyama S, Tomonari H, Yoshida H, Hashimoto T, Kawaguchi Y, Sakai O. Reversal of anemia by erythropoietin therapy retards the progression of chronic renal failure, especially in nondiabetic patients. Nephron. 1997; 77: 176-185.
6. Gouva C, Nikolopoulos P, Ioannidis JP, Siamopoulos KC. Treating anemia early in renal failure patients slows the decline of renal function: a randomized controlled trial. Kidney Int. 2004; 66: 753-760.
7. Kohagura K, Tomiyama N, Kinjo K, Takishita S, Iseki K. Prevalence of anemia according to stage of chronic kidney disease in a large screening cohort of Japanese. Clin Exp Nephrol. 2009; 13: 614-620.
8. Vanholder R, Schepers E, Pletinck A, Nagler EV, Glorieux G. The uremic toxicity of indoxyl sulfate and p-cresyl sulfate: a systematic review. J Am Soc Nephrol. 2014; 25: 1897-1907.
9. Niwa T. Uremic toxicity of indoxyl sulfate. Nagoya J Med Sci. 2010; 72: 1-11.
10. Lin CJ, Chen HH, Pan CF, Chuang CK, Wang TJ, Sun FJ, Wu CJ. p-Cresylsulfate and indoxyl sulfate level at different stages of chronic kidney disease. J Clin Lab Anal. 2011; 25: 191-197.
11. Lin CJ, Liu HL, Pan CF, Chuang CK, Jayakumar T, Wang TJ, Chen HH, Wu CJ. Indoxyl sulfate predicts cardiovascular disease and renal function deterioration in advanced chronic kidney disease. Arch Med Res. 2012; 43: 451-456.
12. Barreto FC, Barreto DV, Liabeuf S, Meert N, Glorieux G, Temmar M, Choukroun G, Vanholder R, Massy ZA; European Uremic Toxin Work Group (EUTox). Serum indoxyl sulfate is associated with vascular disease and mortality in chronic kidney disease patients. Clin J Am Soc Nephrol. 2009; 4: 1551-1558.
13. Taki K, Tsuruta Y, Niwa T. Indoxyl sulfate and atherosclerotic risk factors in hemodialysis patients. Am J Nephrol. 2007; 27: 30-35.
14. Lin CJ, Pan CF, Liu HL, Chuang CK, Jayakumar T, Wang TJ, Chen HH, Wu CJ. The role of protein-bound uremic toxins on peripheral artery disease and vascular access failure in patients on hemodialysis. Atherosclerosis. 2012; 225: 173-179.
15. Tanaka T, Yamaguchi J, Higashijima Y, Nangaku M. Indoxyl sulfate signals for rapid mRNA stabilization of Cbp/p300-interacting transactivator with Glu/Asp-rich carboxy-terminal domain 2 (CITED2) and suppresses the expression of hypoxia-inducible genes in experimental CKD and uremia. FASEB J. 2013; 27: 4059-4075.
16. Chiang CK, Tanaka T, Inagi R, Fujita T, Nangaku M. Indoxyl sulfate, a representative uremic toxin, suppresses erythropoietin production in a HIF-dependent manner. Lab Invest. 2011; 91: 1564-1571.
17. Bernhardt WM, Wiesener MS, Scigalla P, Chou J, Schmieder RE, Gunzler V, Eckardt KU. Inhibition of prolyl hydroxylases increases erythropoietin production in ESRD. J Am Soc Nephrol. 2010; 21: 2151-2156.
18. Krock BL, Skuli N, Simon MC. Hypoxia-induced angiogenesis: good and evil. Genes Cancer. 2011; 2:1117-33. https://doi.org/10.1177/1947601911423654.
19. Lin CJ, Liou TC, Pan CF, Wu PC, Sun FJ, Liu HL, Chen HH, Wu CJ. The Role of Liver in Determining Serum Colon-Derived Uremic Solutes. PLoS One. 2015; 10: e0134590.
20. Zeng YQ, Dai Z, Lu F, Lu Z, Liu X, Chen C, Qu P, Li D, Hua Z, Qu Y, Zou C. Emodin via colonic irrigation modulates gut microbiota and reduces uremic toxins in rats with chronic kidney disease. Oncotarget. 2016; 7: 17468-17478. https://doi.org/10.18632/oncotarget.8160.
21. Chiang CK, Nangaku M, Tanaka T, Iwawaki T, Inagi R. Endoplasmic reticulum stress signal impairs erythropoietin production: a role for ATF4. Am J Physiol Cell Physiol. 2013; 304: C342-353.
22. Adelibieke Y, Shimizu H, Saito S, Mironova R, Niwa T. Indoxyl sulfate counteracts endothelial effects of erythropoietin through suppression of Akt phosphorylation. Circ J. 2013; 77: 1326-1336.
23. Ahmed MS, Abed M, Voelkl J, Lang F. Triggering of suicidal erythrocyte death by uremic toxin indoxyl sulfate. BMC Nephrol. 2013; 14: 244.
24. Kato A, Odamaki M, Hishida A. Association between blood indoxyl sulfate and carbonyl stress marker in hemodialysis patients. Clin Nephrol. 2003; 60: 161-167.
25. Wu IW, Hsu KH, Sun CY, Tsai CJ, Wu MS, Lee CC. Oral adsorbent AST-120 potentiates the effect of erythropoietin-stimulating agents on Stage 5 chronic kidney disease patients: a randomized crossover study. Nephrol Dial Transplant. 2014; 29: 1719-1727.
26. Palm F, Nangaku M, Fasching A, Tanaka T, Nordquist L, Hansell P, Kawakami T, Nishijima F, Fujita T. Uremia induces abnormal oxygen consumption in tubules and aggravates chronic hypoxia of the kidney via oxidative stress. Am J Physiol Renal Physiol. 2010; 299: F380-386.
27. Bammens B, Evenepoel P, Keuleers H, Verbeke K, Vanrenterghem Y. Free serum concentrations of the protein-bound retention solute p-cresol predict mortality in hemodialysis patients. Kidney Int. 2006; 69: 1081-1087.
28. Meijers BK, De Loor H, Bammens B, Verbeke K, Vanrenterghem Y, Evenepoel P. P-Cresyl sulfate and indoxyl sulfate in hemodialysis patients. Clin J Am Soc Nephrol. 2009; 4: 1932-1938.
29. Lin CJ, Wu CJ, Pan CF, Chen YC, Sun FJ, Chen HH. Serum protein-bound uraemic toxins and clinical outcomes in haemodialysis patients. Nephrol Dial Transplant. 2010; 25: 3693-700.
