INTRODUCTION
Liver cancer is one of the most common human malignancies and most primary liver cancers occurring worldwide are hepatocellular carcinoma (HCC) [1]. Surgery is the only potentially curative treatment option for patients who have resectable HCC. Unfortunately, even after surgery, the 5-year overall survival (OS) rate is estimated to 50% and the 5-year recurrence rate exceeds 70% [2, 3] .Thus, it is crucial to explore and identify biomarkers for predicting the prognosis in HCC patients after surgery.
Systemic inflammatory response is increasingly recognized to play decisive roles at different stages of tumor development, including initiation, promotion, malignant conversion, invasion, and metastasis [4]. Previous studies reported the systemic inflammatory response was associated with cancer progression [5]. Recently there has been increasing interest in improving cancer prognostication using inflammatory biomarkers. The neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR), have both been demonstrated to be prognostic markers for patients with various types of tumors [6-14]. Previous studies of hematologic malignancies suggested that an increased LMR indicate a good prognosis [15, 16]. There have been a few reports focusing on the prognostic significance of LMR in patients with solid tumors, including gastric [17], colon [18], bladder [19, 20], renal [21] and lung cancers [22].
Gamma-glutamyltransferase (GGT) plays an important role in the metabolism of glutathione. GGT was investigated as a liver enzyme and a high level of serum GGT has been usually deemed as an alert sign for potential liver disease clinically. Recently, some studies suggested elevated GGT was a promising biomarker for poor OS of HCC patients who underwent hepatic resection [23], radiofrequency-ablation treatment [24] or transcatheter arterial chemoembolization [25].
In this study, we created a systemic inflammation score (SIS) basing on preoperative serum GGT and LMR. It may serve as a better prognostic predictor for clinical outcome in HCC patients after hepatectomy. We conducted this retrospective study in a large cohort of HCC patients undergoing potentially curative resection, attempting to investigate the prognostic value of the systemic inflammatory biomarkers in HCC patients undergoing curative resection.
RESULTS
Clinicopathological characteristics
Clinical and pathologic characteristics of all patients were summarized in Table 1. Of the 271 patients, 213 (78.6%) were males and 58 (21.4%) were females. The median age of the entire cohort was 60 years (range, 27-81 years). The median follow-up was 26 months (range, 5 to 101 months). The mean preoperative GGT was 102.37±146.79 (U/L). The mean platelet, absolute lymphocyte and absolute monocyte counts were 188.36±64.92(×109L-1), 1.61±0.56 (×109L-1) and 0.45±0.20 (×109L-1), respectively. The mean LMR was 4.09±1.99.
The optimal cut-off value for LMR
The optimal cut-off value of LMR was determined by the receiver operating characteristic (ROC) curve analysis for OS. The cut-off value was 4.5 when OS was employed as end-point for LMR, which yielded the largest sensitivity and specificity. LMR was stratified into < 4.5 or ≥4.5 for subsequent analyses. 170 patients (62.7%) and 101 patients (37.3%) were included in the low-LMR group ( < 4.5) and high-LMR group (≥4.5), respectively.
Systemic inflammation score (SIS) evaluation
The continuous variable GGT was stratified into < 50 or ≥50 U/L and the continuous variable LMR was stratified into < 4.5 or ≥4.5 for subsequent analyses. Kaplan-Meier analysis indicated that the high-GGT and low-LMR were both associated with shorter OS (P < 0.001 for both). In order to further discriminate patients with different outcome, we subsequently dichotomized patients into four subgroups basing on serum GGT and LMR levels. In subgroups of either high-GGT or low-LMR, the OS was not different significantly (P = 0.518). Therefore, these two subgroups were combined and SIS was scored as follows: Patients with low-GGT and high-LMR were allocated a score of 0, patients with either high-GGT or low-LMR were allocated a score of 1, and patients with both high-GGT and low-LMR were allocated a score of 2.
Associations of LMR, GGT and SIS with clinicopathological characteristics
The clinicopathological characteristics grouped by LMR, GGT and SIS were summarized in Table 1. In patients with HCC, LMR was associated with TNM stage (P = 0.020). Elevated GGT was associated with male (P = 0.002), alpha fetoprotein(AFP)>20μg/L (P = 0.046), ALT>40U/L (P = 0.001), AST>40U/L (P < 0.001), tumor size >5 cm (P < 0.001), tumor encapsulation (P = 0.018) and TNM stage (P < 0.001). SIS was significantly associated with sex (P = 0.014), albumin (P = 0.049), ALT (P = 0.007), AST (P < 0.001), tumor size (P = 0.001) and TNM stage (P = 0.001).
Table 1: Association of LMR,GGT and SIS with clinicopathological characteristics
Total |
LMR |
GGT |
SIS |
||||||||
N=271 |
<4.5 (n=170) |
≥4.5 (n=101) |
P |
<50 (n=129) |
≥50 (n=142) |
P |
0 (n=58) |
1 (n=114) |
2 (n=99) |
P |
|
Age(year) |
0.157 |
0.909 |
0.602 |
||||||||
≤50 |
106 |
61 |
45 |
50 |
56 |
26 |
43 |
37 |
|||
>50 |
165 |
109 |
56 |
79 |
86 |
32 |
71 |
62 |
|||
Sex |
0.179 |
0.002 |
0.014 |
||||||||
Female |
58 |
32 |
26 |
38 |
20 |
19 |
26 |
13 |
|||
Male |
213 |
138 |
75 |
91 |
122 |
39 |
88 |
86 |
|||
HBsAg |
0.477 |
0.077 |
0.107 |
||||||||
Positive |
218 |
139 |
79 |
98 |
120 |
41 |
95 |
82 |
|||
Negative |
53 |
31 |
22 |
31 |
22 |
17 |
19 |
17 |
|||
AFP |
0.084 |
0.046 |
0.517 |
||||||||
≤20μg/L |
101 |
70 |
31 |
56 |
45 |
20 |
47 |
34 |
|||
>20μg/L |
170 |
100 |
70 |
73 |
97 |
38 |
67 |
65 |
|||
TB |
0.056 |
0.479 |
0.229 |
||||||||
≤17.1umol/L |
265 |
164 |
101 |
127 |
138 |
58 |
112 |
95 |
|||
>17.1umol/L |
6 |
6 |
0 |
2 |
4 |
0 |
2 |
4 |
|||
Albumin |
0.069 |
0.058 |
0.049 |
||||||||
≤40 g/L |
102 |
71 |
31 |
41 |
61 |
15 |
42 |
45 |
|||
>40 g/L |
169 |
99 |
70 |
88 |
81 |
43 |
72 |
54 |
|||
ALT |
0.393 |
0.001 |
0.007 |
||||||||
≤40 U/L |
221 |
136 |
85 |
116 |
105 |
51 |
99 |
71 |
|||
>40 U/L |
50 |
34 |
16 |
13 |
37 |
7 |
15 |
28 |
|||
AST |
0.064 |
<0.001 |
<0.001 |
||||||||
≤40 U/L |
230 |
139 |
91 |
121 |
109 |
54 |
104 |
72 |
|||
>40 U/L |
41 |
31 |
10 |
8 |
33 |
4 |
10 |
27 |
|||
Liver cirrhosis |
0.064 |
0.063 |
0.136 |
||||||||
No |
41 |
31 |
10 |
25 |
16 |
6 |
23 |
12 |
|||
Yes |
230 |
139 |
91 |
104 |
126 |
52 |
91 |
87 |
|||
Tumor size |
0.215 |
<0.001 |
0.001 |
||||||||
≤5 cm |
121 |
71 |
50 |
76 |
45 |
35 |
56 |
30 |
|||
>5 cm |
150 |
99 |
51 |
53 |
97 |
23 |
58 |
69 |
|||
Tumor number |
0.069 |
0.118 |
0.834 |
||||||||
Single |
207 |
136 |
71 |
104 |
103 |
45 |
85 |
77 |
|||
Mutiple |
64 |
34 |
30 |
25 |
39 |
13 |
29 |
22 |
|||
Tumor encapsulation |
0.937 |
0.018 |
0.181 |
||||||||
Complete |
135 |
85 |
50 |
74 |
61 |
31 |
62 |
42 |
|||
None |
136 |
85 |
51 |
55 |
81 |
27 |
52 |
57 |
|||
Vascular invasion |
0.903 |
0.131 |
0.647 |
||||||||
No |
181 |
114 |
67 |
92 |
89 |
41 |
77 |
63 |
|||
Yes |
90 |
56 |
34 |
37 |
53 |
17 |
37 |
36 |
|||
Tumor differentiation |
0.997 |
0.163 |
0.612 |
||||||||
I+II |
169 |
106 |
63 |
86 |
83 |
38 |
73 |
58 |
|||
III+IV |
102 |
64 |
38 |
43 |
59 |
20 |
41 |
41 |
|||
TNM stage |
0.020 |
<0.001 |
0.001 |
||||||||
I |
210 |
124 |
86 |
112 |
98 |
53 |
92 |
65 |
|||
II+III |
61 |
46 |
15 |
17 |
44 |
5 |
22 |
34 |
|||
Abbreviations: AFP=alpha fetoprotein; ALT=alanine aminotransferase; AST=aspartate transaminase; GGT=gamma-glutamyltransferase; HBsAg=hepatitis B surface antigen; LMR= lymphocyte-to-monocyte ratio; SIS=systemic inflammation score; TB= total bilirubin; TNM = tumor–node–metastasis.
Analysis of the prognostic impact of LMR, GGT and SIS
The median OS of the entire cohort was 29.3 months and 5-year OS was 36.6 %. The median time to recurrence (TTR) of the entire cohort was 18.0 months. The relationships between preoperative LMR, GGT, and OS and TTR were shown in Figure 1. An elevated preoperative LMR was significantly associated with increased OS (P < 0.001, Figure 1A) and TTR (P = 0.022, Figure 1B). Whereas an elevated preoperative GGT was significantly associated with inferior OS (P < 0.001, Figure 1C) and TTR (P = 0.005, Figure 1D). In addition, low SIS was significantly associated with increased OS (P < 0.001, Figure 2A) and TTR (P = 0.005, Figure 2B). The 1-, 3-, and 5-year OS rates of patients with SIS = 0 (89.5%, 65.9%, and 52.6%, respectively) were significantly higher than patients with SIS = 1 (76.3%, 52.2%, and 38.5%, respectively) and SIS = 2 (63.6%, 26.8%, and 25.1%, respectively). Moreover, the 1-, 3-, and 5-year cumulative recurrence rates of patients with SIS = 0 were 27.6%, 44.2%, and 52.7%, respectively, which were significantly lower than those of SIS = 1 (44.0%, 61.7%, and 65.4%, respectively) and SIS = 2 (53.3%, 69.5%, and 74.6%, respectively).
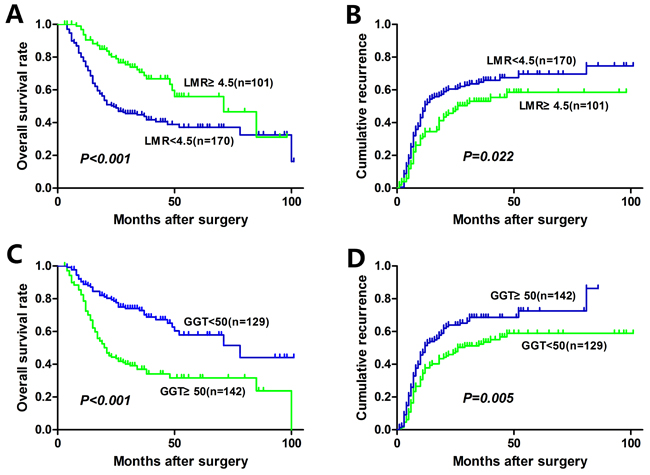
Figure 1: Kaplan-Meier analyses for overall survival and cumulative recurrence rate of HCC patients based on preoperative LMR and GGT.
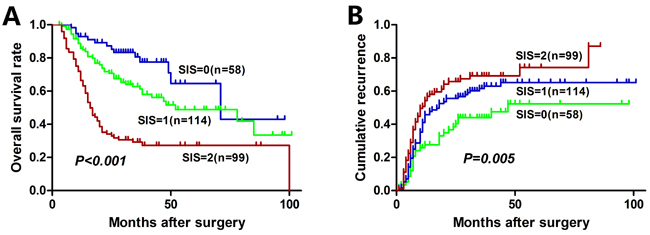
Figure 2: Kaplan-Meier analyses for overall survival and cumulative recurrence rate of HCC patients based on SIS.
Evaluation of the prognostic factors for OS and TTR using the Cox proportional hazard model
The results of univariate and multivariate Cox regression analyses of the factors related to OS and TTR were summarize in Table 2. Univariate analysis indicated that ALT, AST, tumor size, tumor encapsulation, vascular invasion, TNM stage, absolute lymphocyte counts, absolute monocyte counts, LMR, GGT and SIS were significant prognostic factors for OS, and tumor number, tumor encapsulation, vascular invasion, TNM stage, LMR, GGT and SIS were significant prognostic factors for TTR. Two multivariate models were performed separately, considering that SIS is constructed based on GGT and LMR. Multivariate analysis indicated that ALT, tumor encapsulation, vascular invasion, TNM stage, LMR, GGT and SIS were independent prognostic factors for OS, and tumor number, tumor encapsulation, vascular invasion, TNM stage, LMR and SIS were independent prognostic factors for TTR.
Table 2: Univariate and multivariate Cox proportional hazards regression analysis for OS and TTR
OS |
TTR |
|||
HR(95%CI) |
P |
HR(95%CI) |
P |
|
Univariate analysis |
||||
Age ,year(>50y vs. ≤50) |
1.110(0.775-1.589) |
0.569 |
1.072(0.777-1.480) |
0.672 |
Sex(male vs. female) |
1.413(0.884-2.260) |
0.148 |
1.305(0.866-1.965) |
0.203 |
HBsAg(negative vs. positive) |
0.853(0.546-1.331) |
0.483 |
1.071(0.726-1.580) |
0.729 |
AFP, μg/L(>20 vs. ≤20) |
1.008(0.702-1.449) |
0.964 |
1.094(0.786-1.523) |
0.593 |
TB, umol/L(>17.1 vs. ≤17.1) |
1.290(0.457-3.642) |
0.631 |
0.491(0.121-1.982) |
0.318 |
Albumin, g/L(>40 vs. ≤40) |
0.772(0.538-1.107) |
0.159 |
0.875(0.632-1.212) |
0.421 |
ALT, U/L(>40 vs. ≤40) |
1.713(1.139-2.576) |
0.010 |
1.041(0.687-1.577) |
0.852 |
AST, U/L(>40 vs. ≤40) |
2.156(1.417-3.281) |
<0.001 |
1.465(0.959-2.237) |
0.077 |
Liver cirrhosis(yes vs. no) |
1.316(0.788-2.198) |
0.294 |
1.412(0.873-2.284) |
0.159 |
Tumor size,cm(>5 vs. ≤5) |
1.786(1.239-2.574) |
0.002 |
1.366(0.992-1.881) |
0.056 |
Tumor number (multiple vs. single) |
1.278(0.846-1.929) |
0.243 |
1.934(1.365-2.740) |
<0.001 |
Tumor encapsulation (complete vs. none) |
1.973(1.379-2.822) |
<0.001 |
1.511(1.101-2.075) |
0.011 |
Vascular invasion(yes vs. no) |
2.174(1.530-3.090) |
<0.001 |
1.707(1.237-2.356) |
0.001 |
Tumor differentiation(III+IV vs I+II) |
1.079(0.753-1.548) |
0.678 |
1.283(0.931-1.770) |
0.128 |
TNM stage (II+III vs. I) |
2.252(1.525-3.328) |
<0.001 |
1.880(1.311-2.697) |
0.001 |
Absolute lymphocyte counts1 |
0.679(0.493-0.934) |
0.017 |
0.866(0.652-1.150) |
0.321 |
Absolute monocyte counts1 |
2.599(1.083-6.232) |
0.032 |
1.931(0.876-4.257) |
0.103 |
Absolute platelet counts1 |
1.001(0.999-0.004) |
0.297 |
1.001(0.998-1.003) |
0.636 |
LMR (≥4.5 vs. <4.5) |
0.450(0.301-0.673) |
<0.001 |
0.680(0.487-0.949) |
0.023 |
GGT,U/L (≥50 vs. <50) |
2.619(1.805-3.801) |
<0.001 |
1.562(1.136-2.147) |
0.006 |
SIS |
||||
0 |
Reference |
Reference |
||
1 |
1.913(1.051-3.481) |
0.034 |
1.544(0.987-2.416) |
0.057 |
2 |
4.695(2.631-8.378) |
<0.001 |
2.124(1.348-3.347) |
0.001 |
Multivariate analysis 2 |
||||
ALT, U/L(>40 vs. ≤40) |
1.995(1.100-3.618) |
0.023 |
NA |
|
AST, U/L(>40 vs. ≤40) |
0.899(0.501-1.615) |
0.899 |
NA |
|
Tumor size,cm(>5 vs. ≤5) |
0.972(0.601-1.572) |
0.909 |
NA |
|
Tumor number (multiple vs. single) |
NA |
2.139(1.495-3.061) |
<0.001 |
|
Tumor encapsulation (complete vs. none) |
1.952(1.341-2.843) |
<0.001 |
1.547(1.122-2.132) |
0.008 |
Vascular invasion(yes vs. no) |
2.026(1.329-3.089) |
0.001 |
1.684(1.216-2.332) |
0.002 |
TNM stage (II+III vs. I) |
1.770(1.150-2.726) |
0.010 |
1.589(1.097-2.303) |
0.014 |
Absolute lymphocyte counts |
0.807(0.534-1.222) |
0.311 |
NA |
|
Absolute monocyte counts |
0.600(0.178-2.024) |
0.600 |
NA |
|
LMR (≥4.5 vs. <4.5) |
0.467(0.273-0.800) |
0.006 |
0.614(0.433-0.870) |
0.006 |
GGT,U/L (≥50 vs. <50) |
1.963(1.293-2.981) |
0.002 |
1.228(0.886-1.702) |
0.217 |
Multivariate analysis 3 |
||||
ALT, U/L(>40 vs. ≤40) |
1.963(1.085-3.552) |
0.026 |
NA |
|
AST, U/L(>40 vs. ≤40) |
0.885(0.494-1.586) |
0.885 |
NA |
|
Tumor size,cm(>5 vs. ≤5) |
0.956(0.598-1.528) |
0.850 |
NA |
|
Tumor number (multiple vs. single) |
NA |
2.055(1.444-2.923) |
<0.001 |
|
Tumor encapsulation (complete vs. none) |
1.960(1.347-2.851) |
<0.001 |
1.514(1.100-2.083) |
0.011 |
Vascular invasion(yes vs. no) |
2.056(1.348-3.136) |
0.001 |
1.657(1.197-2.293) |
0.002 |
TNM stage (II+III vs. I) |
1.764(1.146-2.716) |
0.010 |
1.585(1.094-2.295) |
0.015 |
Absolute lymphocyte counts |
0.791(0.542-1.155) |
0.225 |
NA |
|
Absolute monocyte counts |
0.611(0.192-1.949) |
0.405 |
NA |
|
SIS |
||||
0 |
Reference |
Reference |
||
1 |
1.680(0.900-3.137) |
0.103 |
1.460(0.930-2.291) |
0.100 |
2 |
3.784(1.865-7.680) |
<0.001 |
1.996(1.249-3.190) |
0.004 |
Abbreviations: AFP=alpha fetoprotein; ALT=alanine aminotransferase; AST=aspartate transaminase; CI=confidence interval; GGT=gamma-glutamyltransferase; HBsAg=hepatitis B surface antigen; HR=hazard ratio; LMR= lymphocyte-to-monocyte ratio; NA=not adopted; OS= overall survival; SIS=systemic inflammation score; TB= total bilirubin; TNM stage= tumor–node–metastasis stage; TTR= time to recurrence.
1Analysed as a continuous variable.
2 Analysis including LMR and GGT (omitting SIS).
3 Analysis including SIS (omitting LMR and GGT).
Comparation of predictive ability of SIS and other inflammatory parameters
Predictive ability of the SIS was compared with other inflammatory parameters(GGT, LMR, NLR and PLR) by ROC curves (Figure 3). The discrimination ability was compared by the area under the receiver operating characteristic curve(AUC) for OS (Table 3). The AUC for the SIS was 0.682 (95% CI, 0.618-0.746), which was the strongest factor among inflammatory parameters (GGT, LMR, NLR and PLR) for predicting survival in patients with HCC.
Table 3: Comparation of predictive ability of SIS and other inflammatory parameters
AUC(95%CI) |
P |
|
SIS |
0.682(0.618-0.746) |
<0.001 |
GGT |
0.637(0.570-0.703) |
<0.001 |
LMR |
0.614(0.547-0.681) |
0.001 |
NLR |
0.602(0.535-0.670) |
0.004 |
PLR |
0.558(0.490-0.627) |
0.097 |
Abbreviations: AUC=area under the receiver operating characteristic curve; CI=confidence interval; GGT=gamma-glutamyltransferase; LMR=lymphocyte-to-monocyte ratio; NLR=neutrophil-to-lymphocyte ratio; PLR=platelet-to-lymphocyte ratio; SIS=systemic inflammation score.
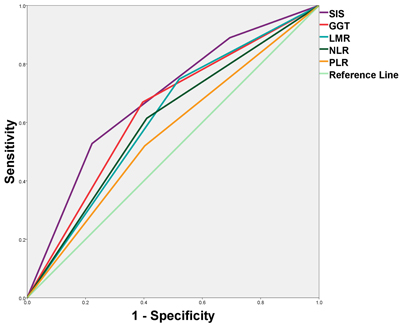
Figure 3: Predictive ability of the SIS was compared with other inflammatory parameters by ROC curves.
DISCUSSION
Links between cancer and inflammation were first described in the nineteenth century. Nowadays, increasing evidence indicating systemic inflammatory response plays an important role in cancer progression [5]. Markers based on systemic inflammation, such as the NLR and PLR, have been reported to be useful in predicting the outcome of cancer patients [6-13]. In the present study, a novel systemic inflammation score (SIS) was constructed based on preoperative serum GGT and LMR. The data indicated GGT, LMR and SIS were independent predictors of survival and recurrence for patients with HCC after hepatectomy. Our results revealed that an elevated preoperative LMR was significantly associated with increased OS and TTR, whereas an elevated preoperative GGT were significantly associated with inferior OS and TTR. The exact reason for the association of elevated preoperative GGT or low LMR with poor prognosis in malignant tumor patients remains largely unclear.
Firstly, our study identified GGT as a prognostic marker for patients with HCC after hepatectomy. GGT is a key enzyme that plays an important role in the metabolism of glutathione, and it is also correlated with tumorigenesis [26]. GGT may induced DNA instability and subsequent oncogenesis, leading to the death of normal liver cells or the loss of normal liver function [27]. A series of studies have suggested that serum GGT was a marker of oxidative stress [28]. The pro-oxidant activity of GGT may contribute to the persistent oxidative stress described in cancer and modulate processes involved in tumor progression [29]. As a consequence, recent studies suggested elevated GGT was a promising biomarker for poor OS of HCC patients who underwent hepatic resection [23], radiofrequency-ablation treatment [24] or transcatheter arterial chemoembolization [25]. The molecular mechanisms of the association between GGT and poor prognosis of HCC need further study.
The second part of the study was successful in defining the utility of the LMR as a prognostic indicator in HCC patients after hepatectomy. Lymphocytes can exert an anti-tumor effect by inhibiting tumor cell proliferation and migration [5, 30]. As a consequence, a low lymphocyte count might result in a weak antitumor reactions and could predict a poor clinical outcome [31]. On the other hand, monocytes are a type of white blood cells that can further differentiate into a range of tissue macrophages and dendritic cells [32]. Monocytes were reported to promote tumorigenesis through local immune suppression [33]. In addition, monocytes can differentiate into tumor associated macrophages (TAMs), which mostly promote tumor growth and may be obligatory for angiogenesis, invasion, and metastasis [34]. Macrophage could also promote the growth, migration and metastasis of tumor cells by releasing some soluble factors [5,35]. Previous studies indicated elevated macrophage content was associated with poor clinical outcome [36-38]. Hence, an elevated absolute monocyte count may predict poor prognosis in tumor patients. LMR, a combination of lymphocytes and monocytes, may represent a balance in host immunity against malignancy has enhanced prognostic value. Previous studies of hematologic malignancies suggested that an increased LMR promised a good prognosis [15, 16]. More recently, some reports indicated the prognostic significance of LMR in patients with solid tumors, including gastric [17], colon [18], bladder [19, 20], renal [21] and lung cancers [22].
Our results indicated the predictive ability of GGT and LMR is stronger than other inflammatory parameters (NLR and PLR). Therefore, we combine these two prognostic markers to construct SIS, assuming that SIS might have a combined predictive effect of GGT and LMR. SIS is a convenient biomaker because serum GGT and complete blood count are routinely measured before surgery in our clinical practice. In the future, basic research may provide an understanding of its molecular mechanisms that may become potential therapeutic targets.
We assessed the association of LMR, GGT and SIS with clinicopathological characteristics. It is worth mentioning that GGT were significantly associated with male, ALT and AST in our study, which was in line with previous studies. Previous studies has demonstrated a sex difference in GGT level [39, 40] and it has been suggested that the lower level of GGT for women is likely to be of a physiological nature. Moreover, fatty liver occurred more frequently in men than women and the distributions of concentrations of liver enzymes differ between men and women [41]. Recommended cutoffs of abnormal liver enzymes were significantly higher for men than women [42].
The present study had several limitations that require discussion. Firstly, the present study was a retrospective design with single-center and missing variables or selection bias are possible because of its retrospective nature. Secondly, peripheral blood cell counts were performed only once, which might cause bias. In addition, C-reactive protein (CRP) was not gathered in our analyses because it was not routinely measured in our clinical practice. Large-scale prospective studies are warranted to substantiate and validate our results.
In conclusion, our results demonstrated low preoperative LMR and high preoperative GGT were associated with a poor prognosis in HCC patients after hepatectomy. SIS, constructed based on preoperative GGT and LMR, is an easily measured and novel prognostic marker that was significantly correlated with OS and TTR. Our results confirmed that the SIS qualifies as a novel prognostic predictor of HCC patients after curative resection.
MATERIALS AND METHODS
Patients
A total of 271 patients with HCC who underwent curative hepatic resection at the Liver Cancer Institute of Zhongshan Hospital (Fudan University, Shanghai, China) between 2008 and 2011 were enrolled after informed consent. Patients who underwent preoperative therapy, such as transarterial chemoembolization, radiofrequency ablation or percutaneous ethanol injection, were excluded from this study. Ethical approval was obtained from the research ethics committee of Zhongshan Hospital, and written informed consent was obtained from each patient.
Follow-up
The patient follow-up and postoperative treatment were administrated as described previously according to our established guidelines [43]. All patients were regularly screened for recurrence through monitoring of serum AFP, abdomen ultrasonography, and chest x-ray every 1 to 6 months according to the postoperative time. For patients with test results suggestive of recurrence, computed tomography and/or magnetic resonance imaging were used to verify the recurrence. TTR was defined as the interval between the date of surgery and the first recurrence, or from the date of surgery to the date of last follow-up patients without recurrence. OS was defined as the interval between surgery and death, or the interval between surgery and the last observation for surviving patients. Patients who were still alive or recurrence-free were censored at the last follow-up date.
Statistical analysis
Data are expressed as the mean ± standard deviation. ROC curve analysis was applied to determine the optimal cut-off level for LMR as predictor of OS. Prediction accuracy was evaluated with area under the ROC curve. The associations of LMR, GGT and SIS with clinicopathological characteristics were examined using the χ2 test or Fisher exact test. The Cox proportional hazards regression model was applied to perform univariate and multivariate analyses, and those variables that achieved statistical significance in the univariate analysis were entered into the multivariable analysis. The Kaplan-Meier method with log-rank test was used to compare survival curves. All statistical analyses were performed using the Statistical Package for Social Sciences version 19.0 (SPSS Inc, Chicago, IL). A two-sided P-value of < 0.05 was considered statistically significant in all tests.
Abbreviations
AFP = alpha fetoprotein; ALT = alanine aminotransferase; AST = aspartate transaminase; AUC = area under the receiver operating characteristic curve; CI = confidence interval; GGT = gamma-glutamyltransferase; HBsAg = hepatitis B surface antigen; HCC = hepatocellular carcinoma; LMR = lymphocyte-to-monocyte ratio; NLR = neutrophil-to-lymphocyte ratio; OS = overall survival; PLR = platelet-to-lymphocyte ratio; ROC = Receiver operating characteristic; SIS = systemic inflammation score; TB = total bilirubin; TNM = tumor-node-metastasis; TTR = time to recurrence.
Author contributions
Shiming Shi and Qing Chen contributed equally to this work. Jian He and Zhi Dai designed and supervised the research; Shiming Shi and Qing Chen collected the data, and drafted the manuscript; Dan Yin and Xuedong Li performed the follow-up; Shiming Shi and Luxi Ye analyzed the data.
ACKNOWLEDGMENTS
The statistical methods of this study were reviewed by Xinping Zhao from Department of Health Statistics and Social Medicine, School of Public Health, Fudan University, Shanghai.
CONFLICTS OF INTEREST
We declare that we have no conflict of interest.
FUNDING
This work was supported by Natural Science Foundation of Shanghai (No.13ZR1406300) and National Natural Science Fund of China (No. 81472218; No.81672330).
References
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87-108.
2. Bruix J, Sherman M. Management of hepatocellular carcinoma: an update. Hepatology. 2011; 53:1020-1022.
3. Grazi GL, Ercolani G, Pierangeli F, Del GM, Cescon M, Cavallari A, Mazziotti A. Improved results of liver resection for hepatocellular carcinoma on cirrhosis give the procedure added value. Ann Surg. 2001; 234:71-78.
4. Grivennikov SI, Greten FR, Karin M. Immunity, Inflammation, and Cancer. Cell. 2010; 140:883-899.
5. Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008; 454:436-444.
6. Gomez D, Farid S, Malik HZ, Young AL, Toogood GJ, Lodge JP, Prasad KR. Preoperative neutrophil-to-lymphocyte ratio as a prognostic predictor after curative resection for hepatocellular carcinoma. World J Surg. 2008; 32:1757-1762.
7. Cho H, Hur HW, Kim SW, Kim SH, Kim JH, Kim YT, Lee K. Pre-treatment neutrophil to lymphocyte ratio is elevated in epithelial ovarian cancer and predicts survival after treatment. Cancer Immunol Immunother. 2009; 58:15-23.
8. Idowu OK, Ding Q, Taktak AF, Chandrasekar CR, Yin Q. Clinical implication of pretreatment neutrophil to lymphocyte ratio in soft tissue sarcoma. Biomarkers. 2012; 17:539-544.
9. Kwon HC, Kim SH, Oh SY, Lee S, Lee JH, Choi HJ, Park KJ, Roh MS, Kim SG, Kim HJ, Lee JH. Clinical significance of preoperative neutrophil-lymphocyte versus platelet-lymphocyte ratio in patients with operable colorectal cancer. Biomarkers. 2012; 17:216-222.
10. Raungkaewmanee S, Tangjitgamol S, Manusirivithaya S, Srijaipracharoen S, Thavaramara T. Platelet to lymphocyte ratio as a prognostic factor for epithelial ovarian cancer. J Gynecol Oncol. 2012; 23:265-273.
11. Xue P, Kanai M, Mori Y, Nishimura T, Uza N, Kodama Y, Kawaguchi Y, Takaori K, Matsumoto S, Uemoto S, Chiba T. Neutrophil-to-lymphocyte ratio for predicting palliative chemotherapy outcomes in advanced pancreatic cancer patients. Cancer Med. 2014; 3:406-415.
12. Chen Q, Dai Z, Yin D, Yang LX, Wang Z, Xiao YS, Fan J, Zhou J. Negative impact of preoperative platelet-lymphocyte ratio on outcome after hepatic resection for intrahepatic cholangiocarcinoma. Medicine (Baltimore). 2015; 94:e574.
13. Chen Q, Yang LX, Li XD, Yin D, Shi SM, Chen EB, Yu L, Zhou ZJ, Zhou SL, Shi YH, Fan J, Zhou J, Dai Z. The elevated preoperative neutrophil-to-lymphocyte ratio predicts poor prognosis in intrahepatic cholangiocarcinoma patients undergoing hepatectomy. Tumour Biol. 2015; 36:5283-5289.
14. Lan H, Zhou L, Chi D, Zhou Q, Tang X, Zhu D, Yue J, Liu B. Preoperative platelet to lymphocyte and neutrophil to lymphocyte ratios are independent prognostic factors for patients undergoing lung cancer radical surgery: A single institutional cohort study. Oncotarget. 2017; 8:35301-10. https://doi.org/10.18632/oncotarget.13312.
15. Li ZM, Huang JJ, Xia Y, Sun J, Huang Y, Wang Y, Zhu YJ, Li YJ, Zhao W, Wei WX, Lin TY, Huang HQ, Jiang WQ. Blood lymphocyte-to-monocyte ratio identifies high-risk patients in diffuse large B-cell lymphoma treated with R-CHOP. Plos One. 2012; 7:e41658.
16. Porrata LF, Ristow K, Colgan JP, Habermann TM, Witzig TE, Inwards DJ, Ansell SM, Micallef IN, Johnston PB, Nowakowski GS, Thompson C, Markovic SN. Peripheral blood lymphocyte/monocyte ratio at diagnosis and survival in classical Hodgkin’s lymphoma. Haematologica. 2012; 97:262-269.
17. Zhou X, Du Y, Xu J, Huang Z, Qiu T, Wang X, Qian J, Zhu W, Liu P. The preoperative lymphocyte to monocyte ratio predicts clinical outcomes in patients with stage II/III gastric cancer. Tumour Biol. 2014; 35:11659-11666.
18. Shibutani M, Maeda K, Nagahara H, Ohtani H, Sakurai K, Yamazoe S, Kimura K, Toyokawa T, Amano R, Tanaka H, Muguruma K, Hirakawa K. Prognostic significance of the lymphocyte-to-monocyte ratio in patients with metastatic colorectal cancer. World J Gastroenterol. 2015; 21:9966-9973.
19. Temraz S, Mukherji D, Farhat ZA, Nasr R, Charafeddine M, Shahait M, Wehbe MR, Ghaida RA, Gheida IA, Shamseddine A. Preoperative lymphocyte-to-monocyte ratio predicts clinical outcome in patients undergoing radical cystectomy for transitional cell carcinoma of the bladder: a retrospective analysis. Bmc Urol. 2014; 14:76.
20. Zhang GM, Zhu Y, Luo L, Wan FN, Zhu YP, Sun LJ, Ye DW. Preoperative lymphocyte-monocyte and platelet-lymphocyte ratios as predictors of overall survival in patients with bladder cancer undergoing radical cystectomy. Tumour Biol. 2015; 36:8537-43.
21. Gu L, Ma X, Wang L, Li H, Chen L, Li X, Zhang Y, Xie Y, Zhang X. Prognostic value of a systemic inflammatory response index in metastatic renal cell carcinoma and construction of a predictive model. Oncotarget. 2017; 8:52094-103. https://doi.org/10.18632/oncotarget.106260.
22. Hu P, Shen H, Wang G, Zhang P, Liu Q, Du J. Prognostic significance of systemic inflammation-based lymphocyte- monocyte ratio in patients with lung cancer: based on a large cohort study. Plos One. 2014; 9:e108062.
23. Fu S, Guo Z, Li S, Kuang M, Hu W, Hua Y, He X, Peng B. Prognostic value of preoperative serum gamma-glutamyltranspeptidase in patients with hepatocellular carcinoma after hepatectomy. Tumour Biol. 2015; 37:3433-40.
24. Ma H, Zhang L, Tang B, Wang Y, Chen R, Zhang B, Chen Y, Ge N, Wang Y, Gan Y, Ye S, Ren Z. gamma-Glutamyltranspeptidase is a prognostic marker of survival and recurrence in radiofrequency-ablation treatment of hepatocellular carcinoma. Ann Surg Oncol. 2014; 21:3084-3089.
25. Zhang JB, Chen Y, Zhang B, Xie X, Zhang L, Ge N, Ren Z, Ye SL. Prognostic significance of serum gamma-glutamyl transferase in patients with intermediate hepatocellular carcinoma treated with transcatheter arterial chemoembolization. Eur J Gastroenterol Hepatol. 2011; 23:787-793.
26. Ikeda Y, Taniguchi N. Gene expression of gamma-glutamyltranspeptidase. Methods Enzymol. 2005; 401:408-425.
27. Wang Z, Song P, Xia J, Inagaki Y, Tang W, Kokudo N. Can gamma-glutamyl transferase levels contribute to a better prognosis for patients with hepatocellular carcinoma? Drug Discov Ther. 2014; 8:134-138.
28. Lim JS, Yang JH, Chun BY, Kam S, Jacobs DJ, Lee DH. Is serum gamma-glutamyltransferase inversely associated with serum antioxidants as a marker of oxidative stress? Free Radic Biol Med. 2004; 37:1018-1023.
29. Corti A, Franzini M, Paolicchi A, Pompella A. Gamma-glutamyltransferase of cancer cells at the crossroads of tumor progression, drug resistance and drug targeting. Anticancer Res. 2010; 30:1169-1181.
30. Rosenberg SA. Progress in human tumour immunology and immunotherapy. Nature. 2001; 411:380-384.
31. Hoffmann TK, Dworacki G, Tsukihiro T, Meidenbauer N, Gooding W, Johnson JT, Whiteside TL. Spontaneous apoptosis of circulating T lymphocytes in patients with head and neck cancer and its clinical importance. Clin Cancer Res. 2002; 8:2553-2562.
32. Auffray C, Sieweke MH, Geissmann F. Blood monocytes: development, heterogeneity, and relationship with dendritic cells. Annu Rev Immunol. 2009; 27:669-692.
33. Gabrilovich DI, Nagaraj S. Myeloid-derived suppressor cells as regulators of the immune system. Annu Rev Immuno. 2009; 9:162-174.
34. Condeelis J, Pollard JW. Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell. 2006; 124:263-266.
35. Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002; 420:860-867.
36. Balkwill F. Cancer and the chemokine network. Nat Rev Cancer. 2004; 4:540-550.
37. Tsutsui S, Yasuda K, Suzuki K, Tahara K, Higashi H, Era S. Macrophage infiltration and its prognostic implications in breast cancer: the relationship with VEGF expression and microvessel density. Oncol Rep. 2005; 14:425-431.
38. Balkwill F, Charles KA, Mantovani A. Smoldering and polarized inflammation in the initiation and promotion of malignant disease. Cancer Cell. 2005; 7:211-217.
39. Nilssen O, Forde OH, Brenn T. The Tromso Study. Distribution and population determinants of gamma-glutamyltransferase. Am J Epidemio. 1990; 132:318-326.
40. Pintus F, Mascia P. Distribution and population determinants of gamma-glutamyltransferase in a random sample of Sardinian inhabitants. ‘ATS-SARDEGNA’ Research Group. Eur J Epidemiol. 1996; 12:71-76.
41. Ford ES, Schulze MB, Bergmann MM, Thamer C, Joost HG, Boeing H. Liver enzymes and incident diabetes: findings from the European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam Study. Diabetes Care. 2008; 31:1138-1143.
42. Ruhl CE, Everhart JE. Elevated serum alanine aminotransferase and gamma-glutamyltransferase and mortality in the United States population. Gastroenterology. 2009; 136:477-485.
43. Gao Q, Qiu SJ, Fan J, Zhou J, Wang XY, Xiao YS, Xu Y, Li YW, Tang ZY. Intratumoral balance of regulatory and cytotoxic T cells is associated with prognosis of hepatocellular carcinoma after resection. J Clin Oncol. 2007; 25:2586-2593.

