Introduction
Gastric cancer is the fourth most common malignancy and the third most common cause of cancer morbidity and mortality according to GLOBOCAN2012[1]. Over the past decade, in-depth understanding of the biological mechanisms of gastric cancer has led to novel diagnostic, predictive, prognostic biomarkers and targeted therapies. Five-year overall survival of gastric cancer patients is still less than 25%, despite improved surgical and adjuvant approaches[2]. For locally advanced gastric cancer (LAGC) patients(stage II or higher, with no evidence of distant metastases, or locally advanced inoperable disease, as evaluated by CT, chest radiography, ultrasonography, or laparoscopy)[3], even comprehensive strategy including R0 resection with extended lymphadenectomy followed by adjuvant chemotherapy does not warrant long-term survival[4]; distant metastases and loco-regional recurrences still account for 40-51% of cases[5][6].
Tangible progress has been made in the area of therapeutics for LAGC. Neoadjuvant chemotherapy (NAC) is currently accepted worldwide as the initial treatment for LAGC, since its ability to facilitate curative surgery (R0 resection) as well as improve survival when combined with adjuvant chemotherapy was approved by two randomized phase III studies(MAGIC trial[3] and ACCORD-07 trial[7][8]). Those results prompted the National Comprehensive Cancer Network (NCCN) to adjust its treatment guidelines, recommending NAC as preferred option for LAGC (category 1 evidence) since the year 2008[9]. Although, a subsequent a phase III trial examined the value of purely NAC in LAGC patients with strict preoperative staging and standardized D2 resection failed to reach a significant survival benefit[10]. Indeed, two meta-analysis studies suggested that NAC could improve R0 resection rate and overall survival(OS), without affecting perioperative morbidity and mortality[11][12].
The underlying principles behind NAC is to increase R0 resection by shrinking /down-staging tumor and eliminating occult metastatic disease as early as possible[13][14]. Additionally, NAC can provide a valuable opportunity to test chemosensitivity in vivo and predict patient’s respond to subsequent adjuvant chemotherapy[13]. Furthermore, NAC in clinical management of locally advanced operable tumor provides a useful platform for investigation and validation of potential predictive biomarkers, which is essentially important for tailoring individualized treatment[15].
However, the major clinical response rate after different NAC is only ranging from 20% to 45%[16]. Inter-individual differences in the response to NAC are currently observed among essentially all available NAC regimens. Such ‘unpredictable’ drug responses are particularly detrimental in the context of NAC. In other words, some patients undergo toxic, expensive, and fruitless chemotherapy in vain. Moreover, it is also possible that patients who are potentially curable by appropriate surgery would have progression of their disease while receiving NAC. Up to now, evidence supporting the idea that non-responder’s prognosis by immediate surgical intervention is scarce. Nevertheless, the possibility of benefiting from upfront surgery in non-responders is low. Potential mechanisms might include, but not limited to, the following: chemotherapy-induced toxicity, chemotherapy-resistant tumor cell selection led to more invasive tumor cells and delayed surgical treatment[17][18]. In fact, patients who progress while on chemotherapy are unlikely to benefit from resection and can be spared radical surgery. The theoretic advantages and disadvantages of NAC are summarized in Figure 1. The long therapy developmental time period for NAC in gastric cancer over the last thirty years partially explains some of the skepticism about this treatment option[19]. Reliable predictive biomarkers for chemosensitivity which can be implemented before or shortly after chemotherapy are urgently needed in the NAC setting. Indeed, prediction of chemosensitivity with high accuracy is currently anticipated to further improve benefit from NAC[20][21].
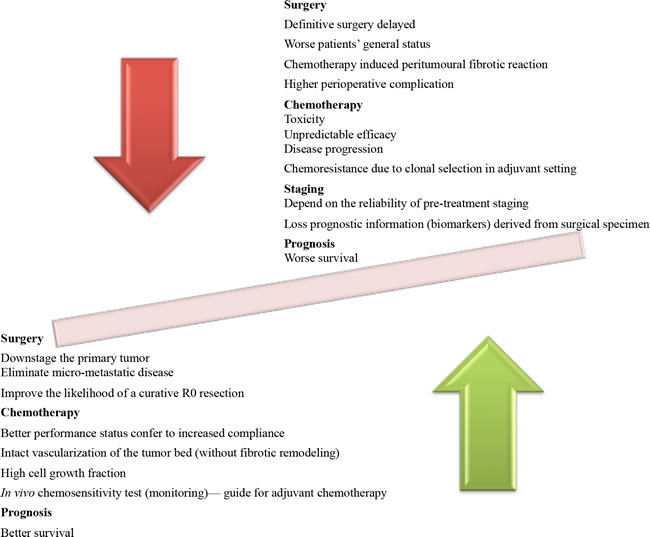
Figure 1: Summarized theoretic benefits and potential risks of neoadjuvant chemotherapy.
The main goal of precision medicine is to predict patient’s response to specific drugs. Identifying non-responders is crucial in avoiding/ or reducing potential harmful NAC. The premise of predicting chemotherapy response is that early prediction and detection of non-responsive tumors can prevent late and incurable disease. Therefore, a well-established strategy that allows predicting histopathological response after receiving NAC is crucial for implementation of NAC in LAGC patients. Unfortunately, standardized, readily accessible predictive assay remains scarce. The response rate to NAC is unmet and seems unpredictable. Although drug resistance in gastric cancer has been extensively explored in postoperative setting (reviewed in [22]). These kinds of results might be transferable to preoperative setting, but it is reasonable to speculate there are inherent difference between them, based on patients’ tumor burden, tumor-host interaction, immune response, clinicopathological properties, and regimens they are administered. Their potential applicability may have to be readdressed in light of neoadjuvant setting. This review will focus on the current status of predictive biomarkers for chemotherapy in gastric cancer limited in neoadjuvant setting, discussing the direct evidence on predictive strategy for chemosensitivity from NAC.
We initially provide a brief overview on the existing histopathological response scoring system and then focus on the current status of biomarker research, including clinicopathological parameters, imaging studies, and molecular biomarkers that have shown promise and possible application in predicting tumor response to NAC in LAGC. Furthermore, future strategies for biomarker development in this field will also be discussed.
Literature Search Criteria
PubMed and MEDLINE were searched for articles in English published before May 2015 using the terms “Gastric cancer”, “Neoadjuvant chemotherapy”, “preoperative chemotherapy”, “predictive biomarker”, “chemosensitivity”, “histopathological assessment”.
Histopathological Assessment
Compared with adjuvant chemotherapy, NAC allows clinician to assess efficacy in relatively more objective and timely manner. Post-NAC tumor regression grade offers an alternative endpoint, which is the current gold standard for discriminating NAC responders from non-responders[15]. The best criterion and endpoint for the effectiveness of a specific anticancer therapy is survival and patient outcome. In line with this, it has been perceived that responders have a significantly better outcome since 1999[23]. Despite the fact that only a limited number of studies presented conflicting result[20][24][25], this conception was further confirmed by several subsequent studies[26][27][28][29]. Taken together, tumor response to NAC could be served as an independent prognostic factor for better prognosis (Figure 2). Most recently, based on data originating from two phase II trials of NAC, another study demonstrated that histopathological criteria (Japan criteria) is a better surrogate endpoint for overall survival than RECIST or Japanese Classification of Gastric Carcinoma (JCGC) criteria [30][31].
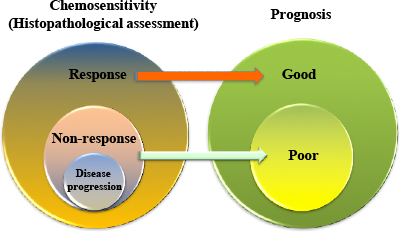
Figure 2: The correlation between histopathological response following neoadjuvant chemotherapy and long-term survival.
Post-operative histopathological response assessment (post hoc analysis) cannot be guide pre- and during the commencing course of NAC (endoscopy re-biopsy is difficult and inaccurate). Based on aforementioned studies, predictive biomarkers for tumor regression grade can provide additional information for individualizing treatment. Moreover, since the well-established correlation between NAC and histopathological regression, NAC is the crucial platform for developing novel predictive biomarkers and can subsequently be used for detailing adjuvant chemotherapy. Finally, taking advantage of NAC platform can facilitate the investigation on the correlation between patients’ clinicopathological background and the effect of NAC. If some of the future additional large scale, prospective clinical studies further re-confirm that histopathological response is a reliable surrogate for overall survival, post-NAC histopathological assessment in gastric cancer could serve as the primary end point and lead to accelerating the regulatory approval of novel agents in this disease.
To date, the Becker[26], Ninomiya[32] and Mandard[33] scoring systems are the most frequently used for evaluating the histological response in gastric cancer treated with NAC (summarized in Table 1). All applied types of histopathology, to various degrees, are correlated with prognosis. These different criteria are considered to have the following two drawbacks: the lack of uniform standards between each other and the existence of observer-dependent bias(relevant observer-related)[13]. Mandard et al.[33] first established the histopathological regression for esophageal cancer post chemoradiotherapy. Subsequently, Becker et al. [26] modified Mandard’s regression score to make it more adaptable to gastric cancer. Specifically, according to Becker’s scoring system, histopathological responders are defined as patients with less than 10% residual tumor cells after NAC[28][34]. While another study proposed that only patients without residual tumor cells(complete tumor regression) could be considered to be histopathological responders[35]. On the contrary, patients with less than 50% residual tumor cells were classified by Shah et al.[36], Liu et al.[37] and Mansour et al.[38] as histopathological responders. Comparisons between studies and use of response criteria in routine practice are hampered by the lack of a universally accepted grading system[39].
Table 1: Histological response criteria following neoadjuvant chemotherapy in gastric cancers
Scoring system |
Category |
Criteria |
Mandard |
TRG 1 |
Absence of residual cancer and fibrosis extending through the layers of esophageal wall |
TRG 2 |
Presence of rare residual cancer cells |
|
TRG 3 |
Increase in number of residual cancer cells, but fibrosis still predominant |
|
TRG 4 |
Showing residual cancer out-growing fibrosis |
|
TRG 5 |
Absence of regressive changes |
|
Japan(Ninomiya) |
Grade 0 |
No change ± neither necrosis nor cellular or structural change can be seen throughout the lesion |
Grade 1a |
Necrosis or disappearance of the tumour is present in less than 1/3 of the whole lesion |
|
Grade 1b |
Necrosis or disappearance of the tumour is present in no more than 2/3 of the whole lesion |
|
Grade 2 |
Moderate change ± necrosis or disappearance of the tumour is present in more than 2/3 of the whole lesion, but viable tumour cells remain |
|
Grade 3 |
Marked change ± the whole lesion falls into necrosis and/or is replaced by fibrosis, with or without granulomatous changes. No viable tumour cells |
|
Becker |
1A |
No residual tumour/tumour bed |
1B |
<10% tumour cells |
|
2 |
10–50% residual tumour/tumour bed |
|
3 |
>50% no signs of neoplastic regression |
Different scoring systems’ reproducibility among various observers and their prognostic value were compared by Mirza et al[39]. Becker’s scoring system was found to be the most reproducible for histological response assessment[39]. It is important to point out that even the Becker’s criteria has high risk of inter-observer variability (κ-scores =0.51). Besides, only Mandard and Becker scores were found to be correlated with 5-year overall survival. Specifically, the 5-year survival rates were 100%(complete or nearly complete histopathological responders) and 35%(non-responders), respectively[39].
The homogenization of the various histopathological response assessment system will facilitate the comparison between different studies. Besides, since all aforementioned scoring systems only take residual tumor cells in the tumor bed into account, potentially neglecting the status of metastatic tumor cells regional to the lymph node. A previous study included involved and metastatic lymph nodes following neoadjuvant radiochemotherapy for esophageal cancer[40]. We suggested involved lymph nodes should also be evaluated in gastric cancer undergo NAC, which might present additional information. This hypothesis needs to be validated.
Table 2: Investigated Clinicopathological variables for neoadjuvant chemotherapy in patients with LAGC
Clinicopathological variable |
Brief summarization |
Reference |
Hemoglobin level |
Hemoglobin level was related to the response |
[41] |
Lymph node metastasis |
The presence of lymph node metastasis was correlated with NAC Chemosensitivity |
[41] |
Tumor size |
Tumor size was independent predictors of tumor regression |
[28] |
Tumor localization |
Tumor localization in the middle third of the stomach was related to the response |
[29] |
Differentiation |
Well tumor differentiation were related to better histopathological response |
[29] |
Laure’s classification |
Intestinal tumor type according to Lauren’s classification accurately predicts histopathological response and prognosis in neoadjuvant treated LAGC |
[29] |
Serum low-density lipoprotein |
Higher low-density lipoprotein is statistically significant with histopathological response in LAGC patients undergoing NAC |
[42] |
Endoscopy(second evaluation) |
Re-biopsies taken after NAC do not help in determining the response, since the biopsy might have been taken from an area of localized complete response |
[15] |
Laparoscopy(re-staging) |
Staging laparoscopy might detect not only pre-existing condition but also disease progression during NAC |
[43] |
Clinicopathological Variables (summarized in Table 3)
Many clinicopathological variables have been correlated with histopathological response. Pre-treatment hemoglobin level and the presence of LN involvement were found to be related with histopathological response in a retrospective study of 119 gastric cancer patients treated with single-agent NAC(S1)[41]. Another study suggested TNM staging, histological type, tumor location, sex and age were correlated with histopathological response[27], however, the tumor regression related factors were not fully investigated in this study. Our previous retrospective study of 108 LAGC patients revealed that both tumor size and tumor differentiation were independent predictive marker for NAC responders, who were with better overall survival[28]. Interestingly, based on our updated database, a hypothesis-generating study found serum low-density lipoprotein measurement is useful in predicting chemosensitivity, higher low-density lipoprotein is statistically significant with histopathological response in LAGC patients undergoing NAC[42]. By analyzing 410 NAC treated LAGC, Lorenzen et al demonstrated that a predictive system, which is comprised of three pre-treatment clinicopathological variables (tumor localization, differentiation, and Lauren’s classification), could predict chemosensitivity and prognosis [29].
Table 3: Investigated image studies for neoadjuvant chemotherapy in patients with LAGC
Modality |
Brief summarization |
Reference |
Endoscopy ultrasound (EUS) Computed tomography (CT) |
The reliability of EUS and CT in predicting NAC response is still controversial. CT volumetry reduction, T and/or N down-staging by EUS and double enhance contrast ultrasound (DCUS) might be promising modalities. |
|
Magnetic Resonance (MR) |
Usefulness of MRI in predicting pathological response after NAC has not been investigated in LAGC, High-b-value diffusion-weighted MR imaging might be promising tool. |
|
Positron emission tomography ( PET ) |
18FDG-PET allow early differentiation of responding and nonresponding tumors during NAC, despite are non-avid 18FDG non-avid patients(intestinal type and nonmucinous tumors) are not suitable for response monitoring using the PET tracer 18FDG. Adding 18FLT-PET to 18FDG-PET might improve early prediction of response to NAC. |
[70] |
Re-biopsies (second evaluation) taken during or after completion of NAC are proposed to be generally inaccurate (unreliable in the prediction of response)[26][43] and useful only in cases that are macroscopically highly suspicious, and can only be evaluated by post-operation histopathological examination. This technique may also be potentially quite risky, and certainly expensive, particularly if it is to be done on several occasions. Moreover, a negative result at histopathology does not prove that there is no tumor growth[15], since the biopsy might have been taken from an area of localized complete response. Moreover, staging laparoscopy has been shown to detect the presence of occult metastases approximately 8% to 26% of patients[44]. Pre and during the course of NAC, staging laparoscopy might detect not only pre-existing condition but also disease progression.
Up to our knowledge, prospective clinical trials focusing on investigation or validation of aforementioned or new predictive clinicopathological markers are urgently needed. These results generated from currently available studies are limited by their retrospective design, small sample size and various histopathological scoring criteria utilized. However, due to their ready availability, incorporation of these clinicopathological factors into approaching individualized treatment might be considered in clinical practice, if they are further revalidated by future prospective, large scale studies.
Image Studies (summarized in Table 3)
Conventional anatomic imaging modalities
Morphologic based imaging modalities were considered to have disadvantage in assessing clinical response for gastric cancer patients. WHO criteria proposed that gastric cancer is not suitable for bi-dimensional evaluation[45]. On contrast, the one-dimensional based Response Evaluation Criteria in Solid Tumor (RECIST) criteria was considered to be applicable for gastric cancer[46]. Combination of endoscopy, computed tomography (CT) scans (with distention protocol) and endoscopy ultrasound (EUS) were implemented in assessing NAC treated patients during treatment or pre-operation. This comprehensive imaging modality was proved to be predictive of tumor regression at experienced centers[23][47][48][49]. Unfortunately, other studies revealed that morphologic imaging techniques, including CT and EUS, failed to accurately identify residual tumorous tissue within chemotherapy-treated areas due to occlusion by chemotherapy-induced fibrosis[50][51]. Be consistent with this, two other studies have also found that those conventional modalities were lacking of reliability for predicting response to NAC in esophageal cancer[52][43], might be due to chemotherapy induced edema and fibrosis. Besides, since the distension degree of the stomach is a determining factor in measuring gastric wall thickness, standardized distension protocol is essential for this procedure. Additionally, the formation of hyaline amorphous scar post chemo-induced tumor cell death and the connective tissue stromal component of the tumor making evaluation of viable tumor cell fraction in a residual mass difficult[53]. Moreover, chemotherapy or radiotherapy induced tumor size decline is considered to be a late event, which greatly limits the application of anatomic based imaging studies in predicting NAC response[53]. Another imaging study, which was based on a randomized phase II study, further re-confirmed these disadvantages of CT. The authors demonstrated that CT re-staging after NAC was inaccurate for gastric patients. In particular, the radiologic T-staging change after NAC could not be integrated into clinical decision-making process [54].
Nevertheless, currently, CT is the most widely utilized imaging modality for assessing response in gastric cancer patients treated with chemotherapy. The standardized protocol and parameters of CT for evaluating tumor response to treatment have been well established for these patients[50]. Preoperative clinical stage evaluation could provide prognostic information for gastric cancer patients[55]. Besides, it was proposed that clinical stage should be incorporated as a stratification factor in RCTs investigating preoperative therapy on patients with gastric cancer[55]. Moreover, low cost and readily accessibility enable CT to serve as the widespread utilized and standardized tools for identifying patients with disease progression during the course of NAC. Compared with measuring tumor diameter changes, volume changes calculated by CT demonstrated a higher correlation with early histopathological tumor regression to 2 weeks of NAC and a higher inter-observer consistency[56]. Moreover, the sensitivity and specificity of predicting histopathological response base on early volume change were 100% and 53%, respectively[56]. Be consistent with this, Lee et al. demonstrated that CT volumetry might be a reliable tool in the predicting tumor regression following NAC in patients with LAGC[57]. CT volumetry predicted pathologic response with surprisingly high accurate, being superior to standardized uptake values (SUV) taken from PET scans[57]. Their study suggested that patients, whose post-NAC volume reduction rate exceeds 35.6%, could be categorized as pathologic responders with 100% sensitivity[57].
EUS is thought to be an unreliable tool for response evaluation, as it is not able to distinguish between esophageal edema, fibrosis, and scarring from residual tumors[15]. In contrast, Guo et al. investigated the clinical feasibilities of EUS in predicting histopathological response after NAC. Patients with T and/or N down-staging(46%) had a relative more favorable pathological response to NAC than patients without T and/or N down-staging(54%)[58]. Be consistent with this, another study also showed that T and/or N down-staging by EUS was correlated with favorable overall survival and recurrence free survival. Taken together, T and/or N down-staging after NAC demonstrated by EUS may be served as a potential predictive marker for a favorable prognosis in patients with LAGC[59]. Recently, Ang et al. demonstrated that double enhance contrast ultrasound (DCUS) may represent an novel modality for more precisely predicting tumor regression[34].
These promising primary studies are still in their infancy, further validation are urgently needed. Nevertheless, they pave the way to utilizing readily available conventional imaging tools in predicting chemosensitivity from NAC. Implementation of strategies like ultrasound and CT could be especially beneficial for areas with limited resources.
MRI may also be useful in the prediction of pathological response following NAC in gastric cancer as it can effectively distinguish residual tumor from fibrosis or scar tissue. Recent studies have already demonstrated that MRI could accurately identify residual tumour in breast cancer patients underwent NAC[60]. Moreover, high-b-value diffusion-weighted MR imaging could detect relatively small effects (cell membranes permeability changes, cell swelling, and early cell lysis) in early stages of treatment[61]. Utilizing C26 colon cancer cell bearing mice treated with doxorubicin, Roth et al showed the potential application of this novel MR imaging for predicting treatment efficacy in an early stage[62]. Up to our knowledge, the usefulness of MRI in predicting pathological response after NAC has not been investigated in gastric cancer and warrants further study.
Functional imaging (Metabolic Imaging studies)
Novel molecules based imaging technologies are being investigated to delineate the complexity, diversity and in vivo behavior of cancers while providing clinicians with new tools to tailor personalized treatment decisions[63]. Functional imaging may offer significant advantages in predicting histopathological response over conventional modality by identifying chemo induced alterations that precede a decline in tumor size[64]. Wahl et al. suggested using morphologic imaging techniques alone under WHO, RECIST, and RECIST 1.1 criteria has significant disadvantages and that both qualitative and quantitative approaches have been used to assess response from PET results[65]. Additionally, they proposed a new criteria, namely as PET Response Criteria in Solid Tumors (PERCIST), which could be served as an initial point for application in clinical trials and in structured quantitative clinical reporting[65].
The potential for PET scanning in investigating chemosensitivity is immense[66]. Fuorine-18 fluorodeoxy-glucose (18FDG), a well-established radiopharmaceutical for gauging exogenous glucose metabolism in vivo, is the most frequently used positron-emitting tracer for cancer imaging. Due to its incomplete intracellular degradation in cancer cells, 18FDG specifically accumulates in most malignant tumors, including gastric cancer[67]. The use of PET with 18FDG enables the visualization and quantification of responding areas in the tumor. Thus, the detection of 18FDG accumulation in residual tumor area by PET might predict histopathological response following NAC.
Indeed, PET can also monitor chemotherapeutic effects in gastric tumors by measuring changes in blood flow, metabolism, regional chemical composition, and absorption[68]. PET has already been utilized in predicting NAC efficacy by focusing on the molecular of characteristics cancer cells rather than anatomical properties alone.
Indeed, PET imaging was investigated on its ability to distinguish responding from nonresponding tumors in the early treatment stage of NAC for adenocarcinomas of the esophagogastric junction (AEG)[49]. In line with this study, 18FDG-PET was also proposed as a predictive marker in the MUNICON study, which proved the clinical feasibility of a PET-directed therapy optimization in AEG via early evaluation on tumor’s metabolic change[69].
As early as 2003, Ott et al. prospectively evaluated the predictive value of 18FDG-PET for subsequent histopathological response in LAGC patients treated with NAC[48]. 18FDG-PET imaging accurately predicted histopathological response in 10 (77%) of 13 responders and 19 (86%) of 22 nonresponders through quantitative measurement of changes 18FDG uptake in relative early course of NAC(two weeks after treatment)[48]. Be consistent with this, Ott et al. also demonstrated that decrease of 18FDG uptake post-NAC(35% decrease as cut-off value) was correlated with tumor regression(less than 10% had residual tumors), which could predict the two year survival of the patients[70]. Similar results with different cutoff value was reported by another study. Shah et al. demonstrated that responders (less than 50% having residual tumors) were those with 45% decrease after 35 days post-NAC[36]. It has been proposed that defined cutoff values, standardized test methodology, and homogenized histopathological regression criteria should be established and validated before 18FDG-PET are clinically utilized[70].
Nevertheless, it is worthy to note that the utility of 18FDG-PET may be limited in evaluating gastric cancer of intestinal type and nonmucinous tumors[13][71]. Ott et al. showed that almost 40% of gastric cancers cannot be analyzed by 18FDG-PET due to insufficient contrast[13]. 18FDG-PET is not ideal tool for monitoring or predicting response in those 18FDG non-avid patients[70], whose overall survival were as poor as metabolic non-responders[13].
Improvement in accuracy might be achieved by implementing PET tracers other than 18FDG. Herrmann et al. showed that proliferative marker 18F-Fluorothymidine (18FLT) might be a potential feasible PET tracer[72]. 18FLT PET was consider to be superior than 18FDG PET in 18FDG non-avid patients[72]. The addition of 18FLT-PET to 18FDG-PET could improve early response prediction[70][73]. Moreover, Ott suggested 18FLT-PET might provide additional information on tumor proliferation status[73].
Due to unsatisfied sensitivity, a single PET scan has not been recommended for predicting post-NAC tumor regression in the upper gastrointestinal tract[74]. Serial PET scans, however, have limitations in terms of cost, availability and doses of radionuclide exposure.
Taken together, functional imaging studies are promising modality in individualizing treatment by characterizing tumor biology. However, there are still several issues that need to be resolved. Due to their technical complexity and high cost might be the biggest obstacle of their widespread implementation. Besides, novel tracers and potential specific molecular pathways should be developed and delineated for metabolic imaging.
Molecular Testing(summarized in Table 4)
The promising molecular testing has become a critical component in the management of cancer patients[75][76]. In the era of precision medicine, predictive and prognostic biomarkers which can guiding clinical decision-making is in the center of current and future study. Great progresses have been achieved in clinically feasibility of molecular biomarkers. HER2 amplification status in breast and gastric cancer cells, BRAF mutation in colon cancers, KRAS and EGFR mutation in lung cancer have been widely implemented in clinical setting and improved patients’ prognosis[77].
Potential genetic and epigenetic alterations which are involved in chemotherapeutic agents metabolism were thought be implicated in patients’ responsiveness to chemotherapy[78]. Predictive biomarkers are of particularly significance for selecting NAC for individual patient, and the identification of molecular biomarkers which could predict histopathological response is crucial for the future use of NAC. Up to our knowledge, no clinically reliable predictive molecular biomarkers are currently available for personalizing LAGC patients’ NAC treatment. The investigated molecular biomarkers that have proven to be of predictive value for histopathological response are discussed below.
Table 4: Investigated molecular markers for neoadjuvant chemotherapy in patients with LAGC
Predictive molecular markers |
Brief summarization |
Reference |
Glutathione-S-transferase (GST) |
Overexpression of GST showed a significantly better sensitivity to cisplatin-based NAC |
[81] |
Dihydropyrimidine dehydrogenase (DPD) |
Significant correlation between DPD expression and histopathological response |
[83] |
TP and/or GADD45A |
High expression values of TP and/or GADD45A were exclusively found in nonresponding patients |
[83] |
Damage DNA binding protein complex subunit 2 (DDB2) |
DDB2- and/or ERCC1-high phenotype was significantly correlated with nonresponding patients |
[84] |
Excision repair cross-complementing 1 (ERCC1) |
ERCC1 nuclear expression correlated with lack of histopathological response |
[85] |
Fractional allelic loss (FAL) |
High FAL value) was shown to define a subset of gastric cancer patients who were more likely to benefit from cisplatin-based NAC |
[86] |
Let-7i |
Low levels of let-7i were significantly correlated with poor histopathological response after NAC |
[37] |
Lin28 |
Higher expression of Lin28 was found in nonresponding patients after NAC |
[90] |
Death-associated protein-3 (DAP-3) |
DAP-3 correlated with NAC effectiveness and prognosis of gastric cancer patients following |
[91] |
Genes involved in drug metabolism
In addition to taxane and anthracycline, the most frequently used drugs for NAC of LAGC are 5-Fu/ cisplatin-based. Accumulating studies have shown that 5-Fu or cisplatin metabolism involving pathways may influence patient outcomes following a 5-Fu/ cisplatin-based polychemotherapy.
DNA polymorphisms in the thymidylate synthase (TS) and 5,10-methylene-tetrahydrofolate reductase (MTHFR) genes, which are involved in the 5-FU pathway, were investigated by Ott et al. for their predictive value for histopathological response in LAGC treated with 5-FU based NAC[79]. Unfortunately, authors failed to find the statistically significant association between the TS or MTHFR genotypes and chemosensitivity[79].
Glutathione-S-transferase (GST) enzymes superfamily, which are important in metabolism (detoxification) of chemotherapy agents such as platin derivates[80]. GST-pi expression level of gastric cancer cell lines was proved to be correlated with chemosensitivity to cisplatin[81]. Ott et al. assessed GST polymorphisms as predictive markers for cisplatin-based NAC in LAGC[80]. Unfortunately, these was no significant correlation between the investigated GST polymorphisms or their combinations and chemosensitivity, showing GST polymorphisms could not distinguish response from nonresponse to NAC[80].
Excision repair cross-complementing 1 (ERCC1) is a key enzyme in the nucleotide excision repair (NER) pathway, and its expression was proposed as an predictive biomarker of the prognosis of advanced gastric cancer patients treated with platinum-based chemotherapy[82]. Napieralski et al. investigated whether seven therapy-related genes have the ability to predict the efficacy of 5-Fu/cisplatin-based NAC in LAGC patients[83]. The expressions of the 5-FU-related genes TS, Dihydropyrimidine dehydrogenase (DPD), Thymidine phosphorylase (TP) and of the cisplatin-related genes ERCC1, ERCC4, Ku autoantigen 80 (KU80), and Growth arrest and DNA-Damage-Inducible alpha (GADD45A) were tested by quantitative real-time PCR. Patients with higher TP and/or GADD45A values were exclusively found in nonresponding subgroup (p = 0.002). Significant correlation between DPD expression and histopathological response was also demonstrated, underlining the predictive value of DPD in 5-FU treated LAGC[83].
Damage DNA binding protein complex subunit 2 (DDB2) is the initial damage recognition molecule during nucleotide excision repair. Loss of DDB2 repair function contributes to cancer susceptibility and cellular sensitivity to DNA damage[23]. The potential correlation between the efficacy of DCS (docetaxel, cisplatin, and S-1) therapy and DDB2 and/or ERCC1 expression level of pretreated tumor tissues was examined[84]. The DDB2- and/or ERCC1-high phenotype was observed in 13 lesions (100%) of the nonresponders and in 7 lesions (25.9%) of the responders (p < 0.0001)[84]. Additionally, Fareed et al. revealed that ERCC1 nuclear expression correlated with histopathological non-response (p = 0.006) in gastro-oesophageal cancer patients who received platinum-based NAC[85].
Other molecular markers
A statistically significant association of fractional allelic loss (FAL) with chemosensitivity was found, with a high FAL correlating with better therapy response in gastric cancer patients treated with cisplatin-based NAC[86]. Meanwhile, authors showed that p53 mutation status was not an ideal predictive marker for NAC benefit[86].
Utilizing MethyLight technology, Napieralski et al. investigated the regional hypermethylation status of six tumor-related genes (promoter region), namely, MGMT, LOX, p16, E-cadherin, 14-3-3sigma and HPP1, for associations with therapy response and clinicopathologic features of 61 neoadjuvant-treated (5-FU/cisplatin-based) gastric cancer[78]. Their study demonstrated that a concordant methylation of more than three genes classified subgroups of gastric cancer with distinct biological and genetic characteristics. Methylation did not show a statistically significant correlation with response to cisplatin/5-fluorouracil-based NAC[78].
MicroRNA let-7i is a well established molecular invovled in chemo-resistance [87][88]. Liu et al. demonstrated that let-7i in pre-treatment tumor tissue might be a predictive marker for chemoensitivity in LAGC patients, wih low levels of let-7i correlating with poor histopathological response after NAC[37]. A double negative feedback loop between lin28 and let-7 has been intensively investigated. Manipulating the Lin28/let-7 pathway could provide novel therapeutic opportunities for treatment of cancer[89]. Consistent with these observation, our previous study revealed that Lin28 expression was significantly inverse associated with histopathological response, with higher expression of Lin28 was found in nonresponding patients[90].
Moreover, Death-associated protein-3 (DAP-3) was also found to be a useful predictive biomarker for predicting response to NAC in gastric cancer patients treated[91].
Limitations of current research strategy
Though marvelous progression has been made in predicting chemosensitivity of NAC in gastric cancer, some common limitations of recent studies still impeded further development. Results of these studies may be difficult to implement in clinical settings due to uncontrolled confounding variables in observational studies or rigid protocols in randomized trials. These points must be clearly addressed in future studies in order to facilitate discovery of predictive biomarkers of NAC response in LAGC. Specific limitations include but not limited:
Firstly, there is limited accuracy of current pre-treatment gastric cancer staging system. The disease progression after NAC for LAGC is not necessarily a late event of cancer progression when commencing NAC but more likely a pre-existing condition (for example staging laparoscopy can detect the presence of occult metastases) that was not appreciated before treatment.
Secondly, preliminary results, though promising, are based on relatively small sample sizes. There is an urgent need for large-scale, well-designed randomized controlled trials to aid our understanding of clinicopathological variables in predicting chemosensitivity to NAC. Since it is well accepted that the genetic background (Western vs Asia) influence gastric cancer patients’ response to treatment, coming results generated from different populations should be interpreted with caution. To our knowledge, none of the aforementioned molecular biomarkers has been prospectively validated, and most studies, while well designed, have used small sample sizes. Consequently, measurement of molecular markers are still neither validated nor readily available in clinical settings.
Thirdly, substantial heterogeneity exits between the currently available studies and an optimal duration and regimen of neoadjuvant chemotherapy have not yet been established. Uniformity for the definition of LAGC is required to ensure the improved management of patients. To elaborate, there are no uniform inclusion criteria for tumor location(adenocarcinomas of esophagogastric junction and the lower third of the esophagus were included in some critical studies), tumor stage, surgical procedures, NAC regimen (including variable drug, dosage and course), and time point of response evaluation, even inhomogeneous histopathological assessment criteria. Lack of homogeneity makes comparison between studies difficult. Moreover, it is doubtful whether the results of studies with these faults can clearly determine whether or not one or another type of modality predicts histopathological response.
Fourthly, most of the molecular biomarkers are still in their infancy. Specific mechanisms should be characterized thoroughly and standardized readily accessible assays for these promising biomarkers should be developed. Clinical validation are urgently needed.
Finally, only one or two molecular biomarkers are investigated in most of the current studies. There is limited understanding of the molecular interactive functional networks[92].
Future perspectives
Using predictive markers will hopefully eliminate unnecessary and potential harmful NAC. Identifying patient who is at high risk of loco-regional recurrence and distant metastasis, and targeting critical molecular or pathways will provide more opportunities to cure disease with lower toxicity[93]. On the other end of the spectrum, patients with molecular biomarkers suggesting low risk for local recurrence and/or distant metastasis, and those who are potentially curable but display markers indicating high possibility of resistance to NAC, could undergo upfront surgery[93].
The heterogeneity of gastric cancer is both the obstacle and opportunity for developing predictive and prognostic biomarkers. Different gastric cancer may need different types of treatment. As genomic and epigenomic studies evolve, further sub-classification of gastric cancer into new molecular entities is expected to facilitate treatment decision-making.
Promising and available break-through biomarkers which have been investigated in other cancers and may also be applicable in predicting response to NAC treatment of gastric cancer will also be addressed here. The integration of established clinicopathological indexes, imaging study and molecular testing with state-of-the-art molecular profiling will result in precise prediction of chemosensitivity from NAC in gastric cancer.
Molecular subtype
Molecular classification of breast cancer have identified 3 distinct subclasses of breast cancer[94][95]. The molecular sub-types of breast cancer exhibits consistent prognostic significance and renders the development of therapeutic strategies[94][95].
Since the Lauren system of classifying gastric cancer was developed in 1965, the disease has been defined according to histologic features as either intestinal or diffuse. Can genetic data about gastric tumors provide additional information to inform therapy? Recently Shah et al. utilized GeneChip and bioinformatics to analyze cDNA expression of a gastric cancer specimen. Their study categorized gastric cancer into three different molecular types: proximal nondiffuse, diffuse, and distal nondiffuse gastric cancer. This sub-classification has implications for improving our understanding of unique molecular drivers of each gastric cancer type, aiding in the identification of novel predictive, therapeutic, prognostic biomarkers for each gastric cancer type[96]. Additionally, another group demonstrated that based on gene expression patterns, gastric cancers could be classified into 3 subtypes (proliferative, metabolic and mesenchymal)[97].The subgroups exhibit distinguished molecular/genetic features and different response to therapy; this information might be utilized to individualize treatment approaches for gastric cancer patients[97]. Interestingly, the namely “metabolic” subtype are tend to be more sensitive to 5-Fu contained chemotherapy[97]. Meanwhile, the mesenchymal subtype might be more sensitive to inhibitors targeting the PI3K/AKT/mTOR pathway[97]. More recently, based on TCGA (The Cancer Genome Atlas) project, another study reported a comprehensive molecular characterization of 295 primary gastric cancer patients[98][98]. Their results sub-classify gastric cancer into four intrinsic molecular subtypes: EBV-infected tumors; MSI tumors; genomically stable tumors; and chromosomally unstable tumors[98]. More importantly, authors suggested that this sub-classification system might have the potential to guide targeted therapy for distinct subtypes of gastric cancer patients[98].
Gene-expression profiling based on high throughput sequence can be used to classify gastric cancers into different subtypes, which have differences in molecular and genetic features. These differences may also correlate with response to specific treatments. Albeit, whether this kind of promising molecular subclassification of gastric could implicit in predicting chemosensitivity from NAC in LAGC is still unknown. It is rationale to propose that comprehensive molecular characterization of gastric cancer patients could be served as a promising approach and potentially lead to more-effective, personalized therapy.
Next generation sequencing
Genomic, epigenomic, transcriptomic sequencing data generated from Next Generation Sequencing (NGS) might be implemented to classify molecular which are involved in patients’ responses to NAC. Besides, further reduction in costs and improvements in technology will make full classification of cancer genomes clinically feasible[99][100]. With this kind of trend, this revolutionary approach will be available in routine clinical use and change the way we treat patients in the not too distant future. Optimizing treatment pre and/or during NAC for individual patients can be realized by deeper understanding of the genomics of gastric cancer. Incorporation of this platform into clinical studies and eventually standards of routine clinical care should aid in tailoring individualized strategy.
Moreover, other high throughput technologies, including pharmacogenomic, proteomic and metabolomic, might also shed lights on the future of cancer care. These data has high possibility to document the uniqueness of tumors in regard to treatment response[101]. Using ever faster, ever-cheaper sequencing methods and heavy-duty bioinformatics will enlarge the catalogue of molecular marker changes associated with tumor and treatment response.
Evaluation of these genomic and epigenomic alterations only offers a snapshot of the cancer at a specific time point. As the tumor evolves, more alteration will occur and the heterogeneity of the tumor will increase[102]. Understanding the genomic and epigenetic alterations of each tumor in a dynamic way might have the promise of solving this challenge.
Circulating tumor cells and circulating tumor DNA
The potential of circulating tumor cells (CTCs) in the early and non-invasive monitoring of response to therapy has become clear in the past few years[103][104]. The latest approaches to CTC capture and molecular profiling, including next-generation sequencing, mutation analysis, proteomic profiling, single-cell analysis and mutational heterogeneity analysis might further enhance its use.
Most recently, Dawson et al. showed that compared with CA 15-3 or circulating tumor cells, circulating tumor DNA(ctDNA) levels change of serially collected plasma specimens in metastatic breast cancer patients demonstrated a more closely association with tumor burden changes. Among the measures utilized, ctDNA was proved to be the earliest to predict tumor response to treatment in metastatic breast cancer patients [105]. This proof-of-concept analysis showed that circulating tumor DNA is an informative, inherently specific, and highly sensitive biomarker of metastatic breast cancer. These findings allow physicians to continually adjust treatment strategies as the tumor progresses. Quickly identification of the detailed characteristics of the tumor, which is helpful to select the effective treatment to target the evolved tumor cells[105].
Although this is only a preliminary report of a small group of patients and requires confirmation by others, this study provides a provocative view of the future of cancer monitoring.
Conclusion
In patients with LAGC, precisely Predictive biomarkers can identify tumors that are more likely to respond to specific targeted treatments, and they allow us to avoid ineffective options. The inability to select similarly for or against chemotherapy use, coupled with the toxic effects, costs, and inconvenience of chemotherapy, has been a growing source of concern.
predicting the response to NAC remains lacking reliable tools. The unmet histopathological response rate from NAC highlights the need to explore integrated clinicopathological parameters, imaging studies and molecular markers (Table.2-4) that might identify patients with a high probability of response in order to avoid unnecessary and ineffective treatment in patients unlikely to respond. In any case, the ultimate decision to administer NAC would require a biomarker or combination of biomarkers with high specificity and a high positive predictive value.
The immediate future will be focused on integrating these new strategies in light of characterizing of the potential molecular mechanisms of gastric cancer and identifying optimal treatment choices for a given individual with the ongoing quest of identifying validated predictive biomarkers. Further investigation and validation of the existing promising candidate biomarkers are urgent guaranteed. Meanwhile, there is also an urgent need to continue exploration on novel molecular markers. We propose a combined and integrated approach, which is consisted of clinical, imaging, and molecular biomarkers (Figure 3). Large, well-designed prospective trials are desirable to pinpoint key potential candidate biomarkers to facilitate clinician’s strategy based on a patient’s unique profile. These predictive biomarkers, like signposts, are needed to guide a clinician’s treatment decision for LAGC patients. These decisions should not feel like gambles but should be reasoned choices, grounded in personalized insight and an understanding of existing scientific knowledge. The current ‘blunderbuss’ approach will be replaced by precise one.
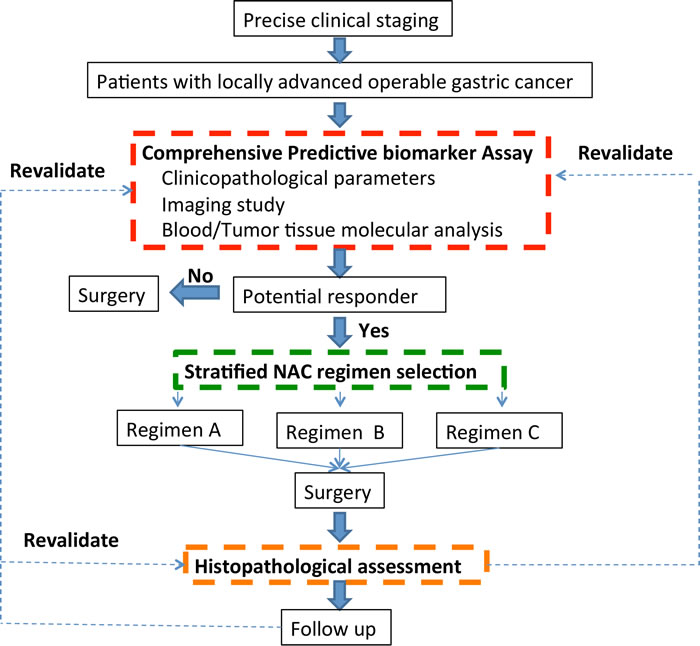
Figure 3: A proposed dynamic model for predicting response from neoadjuvant chemotherapy.
Acknowledgments
We thank Yongxia Chen for critical reading of the manuscript. We also thank reviewers for their critical reading and informative advice during the revision process. We apologize to all researchers whose relevant contributions were not cited due to space limitations.
Conflicts of interest
No potential conflicts of interest were disclosed. None of the contents of this manuscript has been previously published or is under consideration elsewhere. All the authors read and approved the final version of the manuscript prior to submission.
Funding
The Work in the authors’ laboratories is supported by National Natural Science Foundation of China (No. 81602471 and No. 81672729) and by grant from Sub project of China National Program on Key Basic Research Project (973 Program) (No. 2014CB744505).
References
1. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. International Journal of Cancer. 2015; 136: E359–86. doi: 10.1002/ijc.29210.
2. Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin . 2012; 62: 10–29. doi: 10.3322/caac.20138.
3. Cunningham D, Allum WH, Stenning SP, Thompson JN, Van de Velde CJH, Nicolson M, Scarffe JH, Lofts FJ, Falk SJ, Iveson TJ, Smith DB, Langley RE, Verma M, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. The New England journal of medicine. 2006; 355: 11–20. doi: 10.1056/NEJMoa055531.
4. Yoo CH, Noh SH, Shin DW, Choi SH, Min JS. Recurrence following curative resection for gastric carcinoma. The British journal of surgery. 2000; 87: 236–42. doi: 10.1046/j.1365-2168.2000.01360.x.
5. D’Angelica M, Gonen M, Brennan MF, Turnbull AD, Bains M, Karpeh MS. Patterns of initial recurrence in completely resected gastric adenocarcinoma. Annals of surgery. 2004; 240: 808–16. Available from http://www.ncbi.nlm.nih.gov/pubmed/15492562
6. Marrelli D, De Stefano A, de Manzoni G, Morgagni P, Di Leo A, Roviello F. Prediction of recurrence after radical surgery for gastric cancer: a scoring system obtained from a prospective multicenter study. Annals of surgery. 2005; 241: 247–55. Available from http://www.ncbi.nlm.nih.gov/pubmed/15650634
7. Boige V, Pignon J, Saint-Aubert B, Lasser P, Conroy T, Bouché O, Segol P, Bedenne L, Rougier P, Ychou M. Final results of a randomized trial comparing preoperative 5-fluorouracil (F)/cisplatin (P) to surgery alone in adenocarcinoma of stomach and lower esophagus (ASLE): FNLCC ACCORD07-FFCD 9703 trial (Meeting Abstract). Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2007; 25: 4510. Available from http://meeting.ascopubs.org/cgi/content/abstract/25/18_suppl/4510
8. Ychou M, Boige V, Pignon JP, Conroy T, Bouché O, Lebreton G, Ducourtieux M, Bedenne L, Fabre JM, Saint-Aubert B, Genève J, Lasser P, Rougier P. Perioperative Chemotherapy Compared With Surgery Alone for Resectable Gastroesophageal Adenocarcinoma : A FNCLCC and FFCD Multicenter Phase III Trial. Journal of clinical oncology. 2011; 29: 1715–21. doi: 10.1200/JCO.2010.33.0597.
9. National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: gastric cancer Version 2.2013. : Available from:http://www.nccn.org/professionals/p.
10. Schuhmacher C, Gretschel S, Lordick F, Reichardt P, Hohenberger W, Eisenberger CF, Haag C, Mauer ME, Hasan B, Welch J, Ott K, Hoelscher A, Schneider PM, et al. Neoadjuvant chemotherapy compared with surgery alone for locally advanced cancer of the stomach and cardia: European Organisation for Research and Treatment of Cancer randomized trial 40954. Journal of clinical oncology. 2010; 28: 5210–8. doi: 10.1200/JCO.2009.26.6114.
11. Ge L, Wang HJ, Yin D, Lei C, Zhu JF, Cai XH, Zhang GQ. Effectiveness of 5-flurouracil-based neoadjuvant chemotherapy in locally-advanced gastric/gastroesophageal cancer: A meta-analysis. World journal of gastroenterology : WJG. 2012; 18: 7384–93. doi: 10.3748/wjg.v18.i48.7384.
12. Li H, Zhu F, Cao Y, Zhai L, Lin T. Meta-analyses of randomized trials assessing the effect of neoadjuvant chemotherapy in locally advanced gastric cancer. Journal of clinical oncology. 2010; 28: suppl; abstr 4042.
13. Ott K, Lordick F, Herrmann K, Krause BJ, Schuhmacher C, Siewert JR. The new credo: induction chemotherapy in locally advanced gastric cancer: consequences for surgical strategies. Gastric Cancer. 2008; 11: 1–9. doi: 10.1007/s10120-007-0448-1.
14. Cho H, Nakamura J, Asaumi Y, Yabusaki H, Sakon M, Takasu N, Kobayashi T, Aoki T, Shiraishi O, Kishimoto H, Nunobe S, Yanagisawa S, Suda T, et al. Long-term Survival Outcomes of Advanced Gastric Cancer Patients Who Achieved a Pathological Complete Response with Neoadjuvant Chemotherapy: A Systematic Review of the Literature. Annals of Surgical Oncology. 2014; 22: 787–92. doi: 10.1245/s10434-014-4084-9.
15. Brücher BLDM, Swisher SG, Königsrainer A, Zieker D, Hartmann J, Stein H, Kitagawa Y, Law S, Ajani JA. Response to preoperative therapy in upper gastrointestinal cancers. Annals of surgical oncology. 2009; 16: 878–86. doi: 10.1245/s10434-009-0315-x.
16. Ott K, Lordick F, Blank S, Büchler M. Gastric cancer: surgery in 2011. Langenbeck’s archives of surgery / Deutsche Gesellschaft für Chirurgie. 2011; 396: 743–58. doi: 10.1007/s00423-010-0738-7.
17. Kelsen DP, Ginsberg R, Pajak TF, Sheahan DG, Gunderson L, Mortimer J, Estes N, Haller DG, Ajani J, Kocha W, Minsky BD, Roth JA. Chemotherapy followed by surgery compared with surgery alone for localized esophageal cancer. The New England journal of medicine. 1998; 339: 1979–84. doi: 10.1056/NEJM199812313392704.
18. Nooter K, Kok T, Bosman FT, van Wingerden KE, Stoter G. Expression of the multidrug resistance protein (MRP) in squamous cell carcinoma of the oesophagus and response to pre-operative chemotherapy. European journal of cancer. 1998; 34: 81–6. Available from http://www.ncbi.nlm.nih.gov/pubmed/9624242
19. D’Ugo D, Rausei S, Biondi A, Persiani R. Preoperative treatment and surgery in gastric cancer: friends or foes? The lancet oncology. Elsevier Ltd; 2009; 10: 191–5. doi: 10.1016/S1470-2045(09)70021-X.
20. Persiani R, Rausei S, Biondi A, D’Ugo D. Perioperative chemotherapy for gastric cancer: how should we measure the efficacy? Annals of surgical oncology. 2009; 16: 1077–9. doi: 10.1245/s10434-008-0310-7.
21. Fatourou E, Macheras a, Misiakos EP, Liakakos T. Perioperative chemotherapy for gastric cancer. Annals of surgical oncology. 2009; 16: 226–7. doi: 10.1245/s10434-008-0117-6.
22. Zhang D, Fan D. New insights into the mechanisms of gastric cancer multidrug resistance and future perspectives. Future oncology (London, England). 2010; 6: 527–37. doi: 10.2217/fon.10.21.
23. Lowy AM, Mansfield PF, Leach SD, Pazdur R, Dumas P, Ajani JA. Response to neoadjuvant chemotherapy best predicts survival after curative resection of gastric cancer. Annals of surgery. 1999; 229: 303–8. doi: 10.1097/00000658-199903000-00001.
24. Persiani R, D’Ugo D, Rausei S, Sermoneta D, Barone C, Pozzo C, Ricci R, La Torre G, Picciocchi A. Prognostic indicators in locally advanced gastric cancer (LAGC) treated with preoperative chemotherapy and D2-gastrectomy. Journal of surgical oncology. 2005; 89: 227-36-8. doi: 10.1002/jso.20207.
25. D’Ugo D, Persiani R, Rausei S, Biondi A, Vigorita V, Boccia S, Ricci R. Response to neoadjuvant chemotherapy and effects of tumor regression in gastric cancer. European Journal of Surgical Oncology. 2006; 32: 1105–9. doi: 10.1016/j.ejso.2006.07.009.
26. Becker K, Mueller JD, Schulmacher C, Ott K, Fink U, Busch R, Böttcher K, Siewert JR, Höfler H. Histomorphology and grading of regression in gastric carcinoma treated with neoadjuvant chemotherapy. Cancer. 2003; 98: 1521–30. doi: 10.1002/cncr.11660.
27. Ajani JA, Mansfield PF, Crane CH, Wu TT, Lunagomez S, Lynch PM, Janjan N, Feig B, Faust J, Yao JC, Nivers R, Morris J, Pisters PW. Paclitaxel-based chemoradiotherapy in localized gastric carcinoma: degree of pathologic response and not clinical parameters dictated patient outcome. Journal of clinical oncology. 2005; 23: 1237–44. doi: 10.1200/JCO.2005.01.305.
28. Wang LB, Teng RY, Jiang ZN, Hu WX, Dong MJ, Yuan XM, Chen WJ, Jin M, Shen JG. Clinicopathologic variables predicting tumor response to neoadjuvant chemotherapy in patients with locally advanced gastric cancer. Journal of Surgical Oncology. 2012; 105: 293–6. doi: 10.1002/jso.22085.
29. Lorenzen S, Blank S, Lordick F, Siewert JR, Ott K. Prediction of response and prognosis by a score including only pretherapeutic parameters in 410 neoadjuvant treated gastric cancer patients. Annals of surgical oncology. 2012; 19: 2119–27. doi: 10.1245/s10434-012-2254-1.
30. Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma--2nd English edition--response assessment of chemotherapy and radiotherapy for gastric carcinoma: clinical criteria. Gastric cancer. 2001; 4: 1–8.
31. Kurokawa Y, Shibata T, Sasako M. Validity of response assessment criteria in neoadjuvant chemotherapy for gastric cancer (JCOG0507-A). Gastric Cancer. 2013; 17: 1–8. doi: 10.1007/s10120-013-0294-2.
32. Ninomiya Y, Yanagisawa A, Kato Y, Kitagawa T, Ishihara S, Nakajima T. Histological indications of a favorable prognosis with far-advanced gastric carcinomas after preoperative chemotherapy. Journal of cancer research and clinical oncology. 1999; 125: 699–706. Available from http://www.ncbi.nlm.nih.gov/pubmed/10592104
33. Mandard AM, Dalibard F, Mandard JC, Marnay J, Henry-Amar M, Petiot JF, Roussel A, Jacob JH, Segol P, Samama G, Ollivier JM, Bonvalot S, Gignoux M. Pathologic assessment of tumor regression after preoperative chemoradiotherapy of esophageal carcinoma. Clinicopathologic correlations. Cancer. 1994; 73: 2680–6. doi: 10.1002/1097-0142(19940601)73:11<2680::AID-CNCR2820731105>3.0.CO;2-C
34. Ang J, Hu L, Huang PT, Wu JX, Huang LN, Cao CH, Zheng YX, Chen L. Contrast-enhanced ultrasonography assessment of gastric cancer response to neoadjuvant chemotherapy. World journal of gastroenterology : WJG. 2012; 18: 7026–32. doi: 10.3748/wjg.v18.i47.7026.
35. Swisher SG, Hofstetter W, Wu TT, Correa AM, Ajani JA, Komaki RR, Chirieac L, Hunt KK, Liao Z, Phan A, Rice DC, Vaporciyan AA, Walsh GL, et al. Proposed revision of the esophageal cancer staging system to accommodate pathologic response (pP) following preoperative chemoradiation (CRT). Annals of surgery. 2005; 241: 810-7-20. Available from http://www.ncbi.nlm.nih.gov/pubmed/15849517
36. Shah M, Yeung H, Coit D, Trocola R, Ilson D, Randazzo J, Tang L, Brennan M, Divgi C, Kelsen DP. A phase II study of preoperative chemotherapy with irinotecan(CPT) and cisplatin(CIS) for gastric cancer(NCI 5917): FDG-PET/CT predicts patient outcome. J Clin Oncol (Meeting Abstracts). 2007; 25: 4502. Available from http://meeting.ascopubs.org/cgi/content/abstract/25/18_suppl/4502
37. Liu K, Qian T, Tang L, Wang J, Yang H, Ren J. Decreased expression of microRNA let-7i and its association with chemotherapeutic response in human gastric cancer. World journal of surgical oncology. 2012; 10: 225. doi: 10.1186/1477-7819-10-225.
38. Mansour JC, Tang L, Shah M, Bentrem D, Klimstra DS, Gonen M, Kelsen DP, Brennan MF, Coit DG. Does graded histologic response after neoadjuvant chemotherapy predict survival for completely resected gastric cancer? Annals of surgical oncology. 2007; 14: 3412–8. doi: 10.1245/s10434-007-9574-6.
39. Mirza A, Naveed A, Hayes S, Formela L, Welch I, West CM, Pritchard S. Assessment of Histopathological Response in Gastric and Gastro-Oesophageal Junction Adenocarcinoma following Neoadjuvant Chemotherapy: Which Scoring System to Use? ISRN Pathology. 2012; 2012: 1–8. doi: 10.5402/2012/519351.
40. Schneider PM, Baldus SE, Metzger R, Kocher M, Bongartz R, Bollschweiler E, Schaefer H, Thiele J, Dienes HP, Mueller RP, Hoelscher AH. Histomorphologic tumor regression and lymph node metastases determine prognosis following neoadjuvant radiochemotherapy for esophageal cancer: implications for response classification. Annals of surgery. 2005; 242: 684–92. Available from http://www.ncbi.nlm.nih.gov/pubmed/16244542
41. Yonemori K, Shimada Y, Goto A, Ura T, Arai T, Hamaguchi T, Muro K, Yamada Y, Shirao K. Retrospective analysis of clinical results and predictors of response in chemo-naive patients with advanced gastric cancer treated with S-1, an oral fluoropyrimidine derivative, as single-agent chemotherapy. Gastric Cancer. 2004; 7: 204–10. doi: 10.1007/s10120-004-0294-3.
42. Zhou J, Guo JF, Teng R, Wang QC, Wang J, Wei Q, Li ZD, Shen J, Wang LB. New utility of an old marker: serum low-density lipoprotein predicts histopathological response of neoadjuvant chemotherapy in locally advanced gastric cancer. OncoTargets and therapy. 2016; 9: 5041–7. doi: 10.2147/OTT.S97061.
43. Brown WA, Thomas J, Gotley D, Burmeister BH, Lim KH, Martin I, Walpole ET, Thomson DB, Harvey JA, Smithers BM. Use of oesophagogastroscopy to assess the response of oesophageal carcinoma to neoadjuvant therapy. The British journal of surgery. 2004; 91: 199–204. doi: 10.1002/bjs.4411.
44. Blackshaw GRJC, Barry JD, Edwards P, Allison MC, Thomas G V, Lewis WG. Laparoscopy significantly improves the perceived preoperative stage of gastric cancer. Gastric cancer. 2003; 6: 225–9. doi: 10.1007/s10120-003-0257-0.
45. Miller AB, Hoogstraten B, Staquet M, Winkler A. Reporting results of cancer treatment. Cancer. 1981; 47: 207–14. Available from http://www.ncbi.nlm.nih.gov/pubmed/7459811
46. Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, Gwyther SG. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. Journal of the National Cancer Institute. 2000; 92: 205–16. Available from http://www.ncbi.nlm.nih.gov/pubmed/10655437
47. Ott K, Sendler A, Becker K, Dittler HJ, Helmberger H, Busch R, Kollmannsberger C, Siewert JR, Fink U. Neoadjuvant chemotherapy with cisplatin, 5-FU, and leucovorin (PLF) in locally advanced gastric cancer: a prospective phase II study. Gastric cancer : official journal of the International Gastric Cancer Association and the Japanese Gastric Cancer Association. 2003; 6: 159–67. doi: 10.1007/s10120-003-0245-4.
48. Ott K, Fink U, Becker K, Stahl A, Dittler HJ, Busch R, Stein H, Lordick F, Link T, Schwaiger M, Siewert JR, Weber WA. Prediction of response to preoperative chemotherapy in gastric carcinoma by metabolic imaging: results of a prospective trial. Journal of clinical oncology. 2003; 21: 4604–10. doi: 10.1200/JCO.2003.06.574.
49. Weber WA, Ott K, Becker K, Dittler HJ, Helmberger H, Avril NE, Meisetschläger G, Busch R, Siewert JR, Schwaiger M, Fink U. Prediction of response to preoperative chemotherapy in adenocarcinomas of the esophagogastric junction by metabolic imaging. Journal of clinical oncology. 2001; 19: 3058–65. Available fromhttp://www.ncbi.nlm.nih.gov/pubmed/11408502
50. Afaq A, Akin O. Imaging assessment of tumor response: past, present and future. Future oncology (London, England). 2011; 7: 669–77. doi: 10.2217/fon.11.38.
51. Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). European journal of cancer (Oxford, England : 1990). 2009; 45: 228–47. doi: 10.1016/j.ejca.2008.10.026.
52. Mallery S, DeCamp M, Bueno R, Mentzer SJ, Sugarbaker DJ, Swanson SJ, Van Dam J. Pretreatment staging by endoscopic ultrasonography does not predict complete response to neoadjuvant chemoradiation in patients with esophageal carcinoma. Cancer. 1999; 86: 764–9. Available from http://www.ncbi.nlm.nih.gov/pubmed/10463973
53. Bain GH, Petty RD. Predicting response to treatment in gastroesophageal junction adenocarcinomas: combining clinical, imaging, and molecular biomarkers. The oncologist. 2010; 15: 270–84. doi: 10.1634/theoncologist.2009-0293.
54. Yoshikawa T, Tanabe K, Nishikawa K, Ito Y, Matsui T, Kimura Y, Hasegawa S, Aoyama T, Hayashi T, Morita S, Miyashita Y, Tsuburaya A, Sakamoto J. Accuracy of CT Staging of Locally Advanced Gastric Cancer after Neoadjuvant Chemotherapy: Cohort Evaluation within a Randomized Phase II Study. Annals of surgical oncology. 2014; 21 Suppl 3: 385–9. doi: 10.1245/s10434-014-3615-8.
55. Park SR, Kim MJ, Ryu KW, Lee JH, Lee JS, Nam BH, Choi IJ, Kim YW. Prognostic value of preoperative clinical staging assessed by computed tomography in resectable gastric cancer patients: a viewpoint in the era of preoperative treatment. Annals of surgery. 2010; 251: 428–35. doi: 10.1097/SLA.0b013e3181ca69a7.
56. Beer AJ, Wieder HA, Lordick F, Ott K, Fischer M, Becker K, Stollfuss J, Rummeny EJ. Adenocarcinomas of esophagogastric junction: multi-detector row CT to evaluate early response to neoadjuvant chemotherapy. Radiology. 2006; 239: 472–80. doi: 10.1148/radiol.2391050043.
57. Lee SM, Kim SH, Lee JM, Im SA, Bang YJ, Kim WH, Kim MA, Yang HK, Lee HJ, Kang WJ, Han JK, Choi BI. Usefulness of CT volumetry for primary gastric lesions in predicting pathologic response to neoadjuvant chemotherapy in advanced gastric cancer. Abdominal imaging. 2009; 34: 430–40. doi: 10.1007/s00261-008-9420-8.
58. Guo T, Yao F, Yang AM, Li XY, Zhong DR, Wu DS, Wu X, Lu XH. Endoscopic ultrasound in restaging and predicting pathological response for advanced gastric cancer patients after neoadjuvant chemotherapy. Asia-Pacific journal of clinical oncology. 2012; 1–5. doi: 10.1111/ajco.12045.
59. Park SR, Lee JHJS, Kim CG, Kim HK, Kook MC, Kim YW, Ryu KW, Bae JM, Choi IJ. Endoscopic ultrasound and computed tomography in restaging and predicting prognosis after neoadjuvant chemotherapy in patients with locally advanced gastric cancer. Cancer. 2008; 112: 2368–76. doi: 10.1002/cncr.23483.
60. Belli P, Costantini M, Malaspina C, Magistrelli A, Latorre G, Bonomo L. MRI accuracy in residual disease evaluation in breast cancer patients treated with neoadjuvant chemotherapy. Clinical radiology. 2006; 61: 946–53. doi: 10.1016/j.crad.2006.07.004.
61. Van Zijl PC, Moonen CT, Faustino P, Pekar J, Kaplan O, Cohen JS. Complete separation of intracellular and extracellular information in NMR spectra of perfused cells by diffusion-weighted spectroscopy. Proceedings of the National Academy of Sciences of the United States of America. 1991; 88: 3228–32. Available from http://www.ncbi.nlm.nih.gov/pubmed/2014244
62. Roth Y, Tichler T, Kostenich G, Ruiz-Cabello J, Maier SE, Cohen JS, Orenstein A, Mardor Y. High-b-value diffusion-weighted MR imaging for pretreatment prediction and early monitoring of tumor response to therapy in mice. Radiology. 2004; 232: 685–92. doi: 10.1148/radiol.2322030778.
63. Weissleder R, Pittet MJ. Imaging in the era of molecular oncology. Nature. 2008; 452: 580–9. doi: 10.1038/nature06917.
64. Mankoff DA, Eary JF, Link JM, Muzi M, Rajendran JG, Spence AM, Krohn KA. Tumor-specific positron emission tomography imaging in patients: [18F] fluorodeoxyglucose and beyond. Clinical cancer research. 2007; 13: 3460–9. doi: 10.1158/1078-0432.CCR-07-0074.
65. Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: Evolving Considerations for PET response criteria in solid tumors. Journal of nuclear medicine. 2009; 50 Suppl 1: 122S–50S. doi: 10.2967/jnumed.108.057307.
66. West CML, Jones T, Price P. The potential of positron-emission tomography to study anticancer-drug resistance. Nature reviews Cancer. 2004; 4: 457–69. doi: 10.1038/nrc1368.
67. De Potter T, Flamen P, Van Cutsem E, Penninckx F, Filez L, Bormans G, Maes A, Mortelmans L. Whole-body PET with FDG for the diagnosis of recurrent gastric cancer. European journal of nuclear medicine and molecular imaging. 2002; 29: 525–9. doi: 10.1007/s00259-001-0743-8.
68. Marcus CD, Ladam-Marcus V, Cucu C, Bouché O, Lucas L, Hoeffel C. Imaging techniques to evaluate the response to treatment in oncology: current standards and perspectives. Critical reviews in oncology/hematology. 2009; 72: 217–38. doi: 10.1016/j.critrevonc.2008.07.012.
69. Lordick F, Ott K, Krause BJ, Weber WA, Becker K, Stein HJ, Lorenzen S, Schuster T, Wieder H, Herrmann K, Bredenkamp R, Höfler H, Fink U, et al. PET to assess early metabolic response and to guide treatment of adenocarcinoma of the oesophagogastric junction: the MUNICON phase II trial. The lancet oncology. 2007; 8: 797–805. doi: 10.1016/S1470-2045(07)70244-9.
70. Ott K, Herrmann K, Krause BJ, Lordick F. The Value of PET Imaging in Patients with Localized Gastroesophageal Cancer. Gastrointestinal cancer research. 2008; 2: 287–94. Available from http://www.ncbi.nlm.nih.gov/pubmed/19259277
71. Dehdashti F, Siegel B a. Neoplasms of the esophagus and stomach. Seminars in nuclear medicine. 2004; 34: 198–208. doi: 10.1053/j.semnuclmed.2004.03.005.
72. Herrmann K, Ott K, Buck AK, Lordick F, Wilhelm D, Souvatzoglou M, Becker K, Schuster T, Wester HJ, Siewert JR, Schwaiger M, Krause BJ. Imaging gastric cancer with PET and the radiotracers 18F-FLT and 18F-FDG: a comparative analysis. Journal of nuclear medicine. 2007; 48: 1945–50. doi: 10.2967/jnumed.107.044867.
73. Kim SK, Kang KW, Lee JS, Kim HK, Chang HJ, Choi JY, Lee JH, Ryu KW, Kim YW, Bae JM. Assessment of lymph node metastases using 18F-FDG PET in patients with advanced gastric cancer. European journal of nuclear medicine and molecular imaging. 2006; 33: 148–55. doi: 10.1007/s00259-005-1887-8.
74. Downey RJ, Akhurst T, Ilson D, Ginsberg R, Bains MS, Gonen M, Koong H, Gollub M, Minsky BD, Zakowski M, Turnbull A, Larson SM, Rusch V. Whole body 18FDG-PET and the response of esophageal cancer to induction therapy: results of a prospective trial. Journal of clinical oncology. 2003; 21: 428–32. Available fromhttp://www.ncbi.nlm.nih.gov/pubmed/12560430
75. Baudhuin LM, Donato LJ, Uphoff TS. How novel molecular diagnostic technologies and biomarkers are revolutionizing genetic testing and patient care. Expert review of molecular diagnostics. 2012; 12: 25–37. doi: 10.1586/erm.11.85.
76. Roukos DH. Novel clinico-genome network modeling for revolutionizing genotype-phenotype-based personalized cancer care. Expert review of molecular diagnostics. 2010; 10: 33–48. doi: 10.1586/erm.09.69.
77. Febbo PG, Ladanyi M, Aldape KD, De Marzo AM, Hammond ME, Hayes DF, Iafrate AJ, Kelley RK, Marcucci G, Ogino S, Pao W, Sgroi DC, Birkeland ML. NCCN Task Force report: Evaluating the clinical utility of tumor markers in oncology. Journal of the National Comprehensive Cancer Network. 2011; 9 Suppl 5: S1–32; quiz S33. Available from http://www.ncbi.nlm.nih.gov/pubmed/22138009
78. Napieralski R, Ott K, Kremer M, Becker K, Boulesteix AL, Lordick F, Siewert JR, Höfler H, Keller G. Methylation of tumor-related genes in neoadjuvant-treated gastric cancer: relation to therapy response and clinicopathologic and molecular features. Clinical Cancer Research. 2007; 13: 5095–102. doi: 10.1158/1078-0432.CCR-07-0241.
79. Ott K, Vogelsang H, Marton N, Becker K, Lordick F, Kobl M, Schuhmacher C, Novotny A, Mueller J, Fink U, Ulm K, Siewert JR, Höfler H, et al. The thymidylate synthase tandem repeat promoter polymorphism: A predictor for tumor-related survival in neoadjuvant treated locally advanced gastric cancer. International journal of cancer. 2006; 119: 2885–94. doi: 10.1002/ijc.22235.
80. Ott K, Lordick F, Becker K, Ulm K, Siewert J, Höfler H, Keller G. Glutathione-S-transferase P1, T1 and M1 genetic polymorphisms in neoadjuvant-treated locally advanced gastric cancer: GSTM1-present genotype is associated with better prognosis in completely resected patients. International journal of colorectal disease. 2008; 23: 773–82. doi: 10.1007/s00384-008-0490-4.
81. Okuyama T, Maehara Y, Endo K, Baba H, Adachi Y, Kuwano M, Sugimachi K. Expression of glutathione S-transferase-pi and sensitivity of human gastric cancer cells to cisplatin. Cancer. 1994; 74: 1230–6. Available from http://www.ncbi.nlm.nih.gov/pubmed/8055443
82. Kwon HC, Roh MS, Oh SY, Kim SH, Kim MC, Kim JS, Kim HJ. Prognostic value of expression of ERCC1, thymidylate synthase, and glutathione S-transferase P1 for 5-fluorouracil/oxaliplatin chemotherapy in advanced gastric cancer. Annals of oncology : official journal of the European Society for Medical Oncology / ESMO. 2007; 18: 504–9. doi: 10.1093/annonc/mdl430.
83. Napieralski R, Ott K, Kremer M, Specht K, Vogelsang H, Becker K, Müller M, Lordick F, Fink U, Rüdiger Siewert J, Höfler H, Keller G. Combined GADD45A and thymidine phosphorylase expression levels predict response and survival of neoadjuvant-treated gastric cancer patients. Clinical cancer research. 2005; 11: 3025–31. doi: 10.1158/1078-0432.CCR-04-1605.
84. Hirakawa M, Sato Y, Ohnuma H, Takayama T, Sagawa T, Nobuoka T, Harada K, Miyamoto H, Sato Y, Takahashi Y, Katsuki S, Hirayama M, Takahashi M, et al. A phase II study of neoadjuvant combination chemotherapy with docetaxel, cisplatin, and S-1 for locally advanced resectable gastric cancer: nucleotide excision repair (NER) as potential chemoresistance marker. Cancer chemotherapy and pharmacology. 2013; 71: 789–97. doi: 10.1007/s00280-013-2073-5.
85. Fareed KR, Al-Attar A, Soomro IN, Kaye P V, Patel J, Lobo DN, Parsons SL, Madhusudan S. Tumour regression and ERCC1 nuclear protein expression predict clinical outcome in patients with gastro-oesophageal cancer treated with neoadjuvant chemotherapy. British journal of cancer. 2010; 102: 1600–7. doi: 10.1038/sj.bjc.6605686.
86. Ott K, Vogelsang H, Mueller J, Becker K, Müller M, Fink U, Siewert JR, Höfler H, Keller G. Chromosomal instability rather than p53 mutation is associated with response to neoadjuvant cisplatin-based chemotherapy in gastric carcinoma. Clinical cancer research. 2003; 9: 2307–15. Available fromhttp://www.ncbi.nlm.nih.gov/pubmed/12796400
87. Yang N, Kaur S, Volinia S, Greshock J, Lassus H, Hasegawa K, Liang S, Leminen A, Deng S, Smith L, Johnstone CN, Chen XM, Liu CG, et al. MicroRNA microarray identifies Let-7i as a novel biomarker and therapeutic target in human epithelial ovarian cancer. Cancer research. 2008; 68: 10307–14. doi: 10.1158/0008-5472.CAN-08-1954.
88. Blower PE, Chung JH, Verducci JS, Lin S, Park JK, Dai Z, Liu CG, Schmittgen TD, Reinhold WC, Croce CM, Weinstein JN, Sadee W. MicroRNAs modulate the chemosensitivity of tumor cells. Molecular cancer therapeutics. 2008; 7: 1–9. doi: 10.1158/1535-7163.MCT-07-0573.
89. Thornton JE, Gregory RI. How does Lin28 let-7 control development and disease? Trends in Cell Biology. Trends in Cell Biology; 2012; 22: 474–82. doi: 10.1016/j.tcb.2012.06.001.
90. Teng RY, Zhou JC, Jiang ZN, Xu CYCP, Li ZD, Wang QC, Xu CYCP, Guo JF, Shen JG, Wang LB. The relationship between Lin28 and the chemotherapy response of gastric cancer. OncoTargets and Therapy. 2013; 6: 1341–5. doi: 10.2147/OTT.S45705.
91. Jia Y, Ye L, Ji K, Zhang L, Hargest R, Ji J, Jiang WG. Death-associated protein-3, DAP-3, correlates with preoperative chemotherapy effectiveness and prognosis of gastric cancer patients following perioperative chemotherapy and radical gastrectomy. British journal of cancer. 2013; doi: 10.1038/bjc.2013.712.
92. Yaffe MB. The scientific drunk and the lamppost: massive sequencing efforts in cancer discovery and treatment. Science signaling. 2013; 6: pe13. doi: 10.1126/scisignal.2003684.
93. Hofstetter WL. Preoperative Chemoradiation in an Era of Suboptimal Clinical Staging. JAMA Surgery. 2015. doi: 10.1001/jamasurg.2015.4047.
94. Perou CM, Sørlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA, Fluge O, Pergamenschikov A, Williams C, et al. Molecular portraits of human breast tumours. Nature. 2000; 406: 747–52. doi: 10.1038/35021093.
95. Sørlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, Hastie T, Eisen MB, van de Rijn M, Jeffrey SS, Thorsen T, Quist H, Matese JC, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proceedings of the National Academy of Sciences of the United States of America. 2001; 98: 10869–74. doi: 10.1073/pnas.191367098.
96. Shah M a, Khanin R, Tang L, Janjigian YY, Klimstra DS, Gerdes H, Kelsen DP. Molecular classification of gastric cancer: a new paradigm. Clinical cancer research. 2011; 17: 2693–701. doi: 10.1158/1078-0432.CCR-10-2203.
97. Lei Z, Tan IB, Das K, Deng N, Zouridis H, Pattison S, Chua C, Feng Z, Guan YK, Ooi CH, Ivanova T, Zhang S, Lee M, et al. Identification of Molecular Subtypes of Gastric Cancer With Different Responses to PI3-Kinase Inhibitors and 5-Fluorouracil. Gastroenterology. Elsevier, Inc; 2013; 145: 554–65. doi: 10.1053/j.gastro.2013.05.010.
98. Bass AJ, Thorsson V, Shmulevich I, Reynolds SM, Miller M, Bernard B, Hinoue T, Laird PW, Curtis C, Shen H, Weisenberger DJ, Schultz N, Shen R, et al. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. Nature Publishing Group; 2014; 513: 202–9. doi: 10.1038/nature13480.
99. Jones SJ, Laskin J, Li YY, Griffith OL, An J, Bilenky M, Butterfield YS, Cezard T, Chuah E, Corbett R, Fejes AP, Griffith M, Yee J, et al. Evolution of an adenocarcinoma in response to selection by targeted kinase inhibitors. Genome biology. 2010; 11: R82. doi: 10.1186/gb-2010-11-8-r82.
100. Roychowdhury S, Iyer MK, Robinson DR, Lonigro RJ, Wu YM, Cao X, Kalyana-Sundaram S, Sam L, Balbin OA, Quist MJ, Barrette T, Everett J, Siddiqui J, et al. Personalized oncology through integrative high-throughput sequencing: a pilot study. Science translational medicine. 2011; 3: 111ra121. doi: 10.1126/scitranslmed.3003161.
101. Ogino S, Goel A. Molecular classification and correlates in colorectal cancer. The Journal of molecular diagnostics : JMD. 2008; 10: 13–27. doi: 10.2353/jmoldx.2008.070082.
102. Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA, Kinzler KW. Cancer genome landscapes. Science (New York, NY). 2013; 339: 1546–58. doi: 10.1126/science.1235122.
103. Matsusaka S, Chìn K, Ogura M, Suenaga M, Shinozaki E, Mishima Y, Terui Y, Mizunuma N, Hatake K. Circulating tumor cells as a surrogate marker for determining response to chemotherapy in patients with advanced gastric cancer. Cancer science. 2010; 101: 1067–71. doi: 10.1111/j.1349-7006.2010.01492.x.
104. Cohen SJ, Punt CJA, Iannotti N, Saidman BH, Sabbath KD, Gabrail NY, Picus J, Morse M, Mitchell E, Miller MC, Doyle G V, Tissing H, Terstappen LWMM, et al. Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. Journal of clinical oncology. 2008; 26: 3213–21. doi: 10.1200/JCO.2007.15.8923.
105. Dawson SJ, Tsui DWY, Murtaza M, Biggs H, Rueda OM, Chin SF, Dunning MJ, Gale D, Forshew T, Mahler-Araujo B, Rajan S, Humphray S, Becq J, et al. Analysis of Circulating Tumor DNA to Monitor Metastatic Breast Cancer. New England Journal of Medicine. 2013; 368: 1199–209. doi: 10.1056/NEJMoa1213261.

