INTRODUCTION
Liver metastasis is the main cause of colon-cancer-related death [1]. In some cases, surgical resection of colon cancer liver metastasis is effective; however, the recurrence rate is extremely high [2]. The efficacy of adjuvant chemotherapy has been limited [3]. Therefore, development of effective treatment for colon cancer metastasis is needed.
The tumor-targeting Salmonella typhimurium A1-R (S. typhimurium A1-R), developed by our laboratory [4], is auxotrophic for Leu—Arg, which prevents it from mounting a continuous infection in normal tissues. S. typhimurium A1-R was effective against primary and metastatic tumors as monotherapy in nude mouse models of major cancers [5], including prostate [6, 7], breast [8–10], lung [11, 12], pancreatic [13–17], ovarian [18, 19] stomach [20], and cervical cancer [21], as well as sarcoma cell lines [22–25] and glioma [5, 26], all of which are highly aggressive tumor models. In addition, S. typhimurium A1-R was effective against patient-derived orthotopic models of pancreatic cancer [27, 28], sarcoma [25, 29–32] and melanoma [33].
In orthotopic mouse models, S. typhimurium A1-R, delivered iv, targeted liver metastases and significantly reduced their growth. The results of this previous study demonstrated the future clinical potential of S. typhimurium A1-R targeting of liver metastasis [34].
Regional chemotherapy of metastasis has resulted in higher levels of active 5-FU metabolites in the liver [35, 36]. Previous studies have demonstrated that 5-FU administered directly into the portal vein adjuvantly may decrease distant metastases [37, 38]. Chang, et al. [39] reported that peri-operative intraportal (iPV) chemotherapy combined with adjuvant chemotherapy was useful to prolong disease-free survival after primary tumor resection and decreased liver metastasis for stage II and III colon-cancer patients without liver metastasis.
Previously, we administered 5-FU, ip, 2 h before hepatic resection of the human colon tumors, with therapy continued postoperatively for 4 consecutive days. We termed this procedure neo-neoadjuvant chemotherapy. Neo-neoadjuvant therapy significantly prolonged animal survival compared with standard preoperative 5-FU neoadjuvant therapy, 5-FU post-operative adjuvant therapy, surgery alone, 5-FU without surgery, or the untreated control. When all animals with neoadjuvant 5-FU treatment had died, 70% of animals with neo-neoadjuvant treatment were still alive. Survival of mice treated with 5-FU without surgery, surgery alone, and adjuvant postoperative chemotherapy, was not significantly different from the untreated control group. Whereas 100% of animals in the control, 90% in the 5-FU alone, 70% in the surgery alone, 60% in the 5-FU adjuvant, and 40% in the neoadjuvant groups had metastases in the lymph nodes draining the liver, only 10% of animals in the neo-neoadjuvant group had metastases [40].
The present study evaluates the efficacy and safety of iPV of S. typhimurium A1-R on colon cancer liver metastasis in a nude-mouse orthotopic model.
RESULTS AND DISCUSSION
iPV injection is more effective for delivery of S. typhimurium A1-R to the liver then iv injection
Two days after injection of S. typhimurium A1-R (iv: 5×107 CFU/100 μl; iPV: 1×104 CFU/100 μl) to mice without liver metastasis, the liver was removed and cultured on Luria-Bertani (LB) agar. The presence of S. typhimurium A1-R was confirmed by bright-field and GFP-expressing colony formation 24 hours after culture (Figure 1A-1D). There was no significant difference in colony formation between iv injection of 5×107 CFU/100 μl and iPV injection of 1×104 CFU/100 μl S. typhiurium A1-R. These results showed that iPV injection was 5×103 times more effective for delivery of S. typhiurium A1-R to the liver than iv injection (Figure 1E).
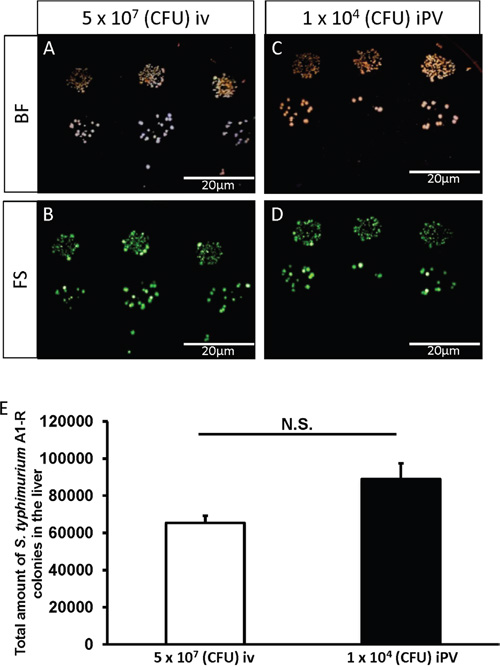
Figure 1: Culture of S. typhimurium A1-R from mouse liver. A-D. Representative images of S. typhimurium A1-R colony formation: Bright field (BF) (A) and fluorescence (FS) (B) after intravenous (iv) injection of S. typhimurium A1-R (5 x 107 CFU). BF (C) and FS (D) after intra-portal-vein (iPV) injection of S. typhimurim A1-R (1 x 104 CFU). The liver was minced and mixed with PBS and was seeded on LB-Agar with serial dilution in triplicate. Fluorescent S. typhimurium A1-R colonies were observed with the OV100 Small Animal Imaging System (Olympus Corp, Tokyo, Japan). E. S. typhimurium A1-R colony number in the liver after iv and iPV injection.
Efficacy of S. typhimurium A1-R iPV injection on liver metastasis
The fluorescent area and total fluorescence intensity of HT29 growing in the liver were measured on day 14 (Figure 2A) after iPV or iv injection of S. typhimurium A1-R. iPV delivery of S. typhiurium A1-R significantly suppressed growth of HT29 in the liver compared to both the untreated-control group and iv treatment group (p<0.01, p<0.05, respectively) (Figure 2B, 2C), with IVP formatting arresting metastatic growth. There was no significant body-weight difference between the groups (Figure 3).
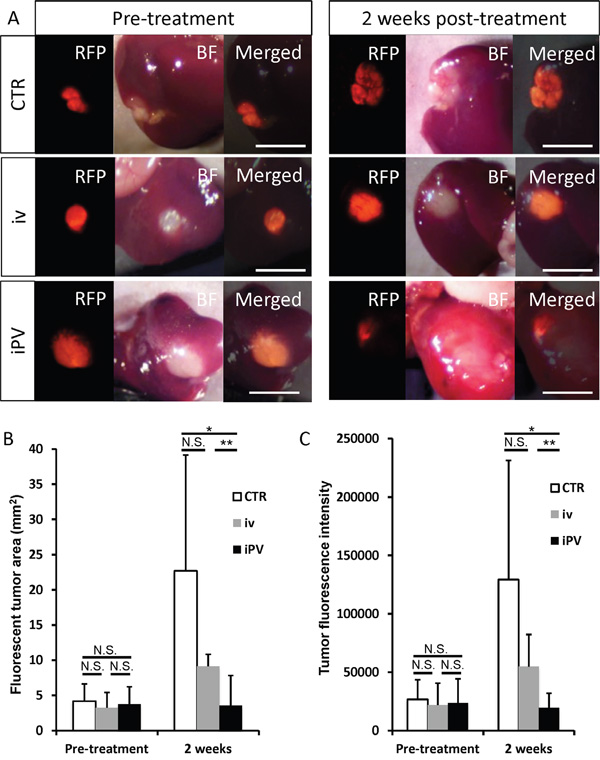
Figure 2: Efficacy of S. typhimurium A1-R on HT29-RFP liver metastasis. A. Pre-treatment and 14 days post-treatment of S. typhimurium A1-R: iv, 5 × 107 CFU/100μ; iPV, 1 × 104 CFU/100 μl. No treatment control (CTR). B-C. Bar graphs show the tumor fluorescent area (mm2) (B) and fluorescence intensity (C) at day 14. Scale bars: 5 mm.
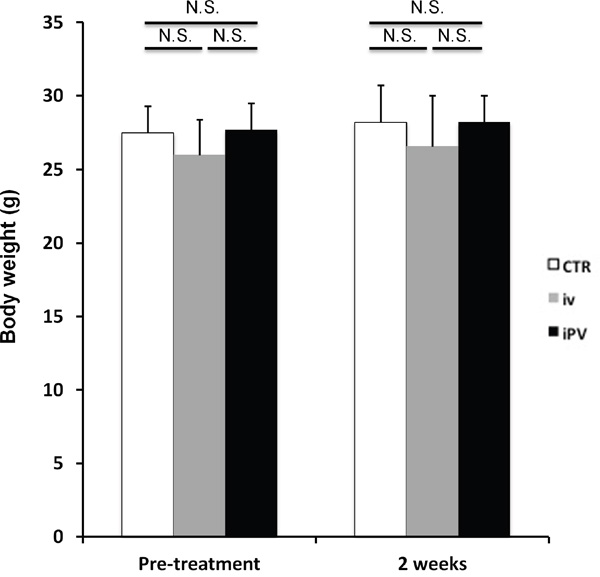
Figure 3: Safety evaluation of S. typhimurium A1-R therapy. Bar graph shows body weight in each group at pre-treatment and on day 14. There were no significant differences between the treated groups and control.
This is the first study to administer S. typhimurium A1-R via the iPV route to the liver directly. Most importantly, the treatment efficacy of iPV injection was significantly better than iv injection on HT29 cells growing in the liver.
Intra-operative therapy could be useful to decrease the risk of recurrence after cancer resection and could increase the surgical cure rate for colon-cancer liver metastasis patients. It has been reported that peri-operative intra-portal chemotherapy combined with adjuvant chemotherapy prolonged disease-free survival after primary tumor resection and decreased liver metastasis for stage II and III colon cancer patients, without liver metastasis [39].
These results are consistent with our previous studies which showed peri-operative “neo-neoadjuvant chemotherapy” was effective against colon-cancer liver metastasis in orthotopic nude mouse models [40].
In the present study, we demonstrated a strong correlation between tumor fluorescence area and fluorescence intensity (p<0.0001) (Figure 4). Fluorescence intensity of tumors could be a readily-measured simple parameter for determination of efficacy.
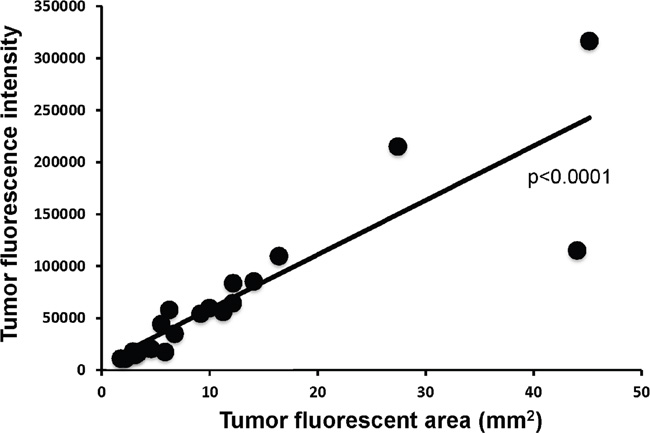
Figure 4: Correlation of tumor fluorescent area in tumor fluorescence intensity. Tumor area significantly correlated with fluorescence intensity (p<0.0001).
Intravital color-coded imaging of bacterial targeting of liver metastasis
Intravital color-coded imaging visualized S. typhimurium GFP targeting the HT29-RFP liver metastasis cells 14 days after iPV injection. The GFP-expressing bacteria can be seen growing in the HT29-RFP cells (Figure 5). The present study develops a new concept of iPV delivery of tumor-targeting S. typhimurium A1-R to treat colon-cancer liver metastasis, the results of which are very promising. Bacterial therapy of cancer, which was first line for sarcoma and other cancers in the early part of the last century [5], is now having an exciting resurgence, including clinical trials [41].
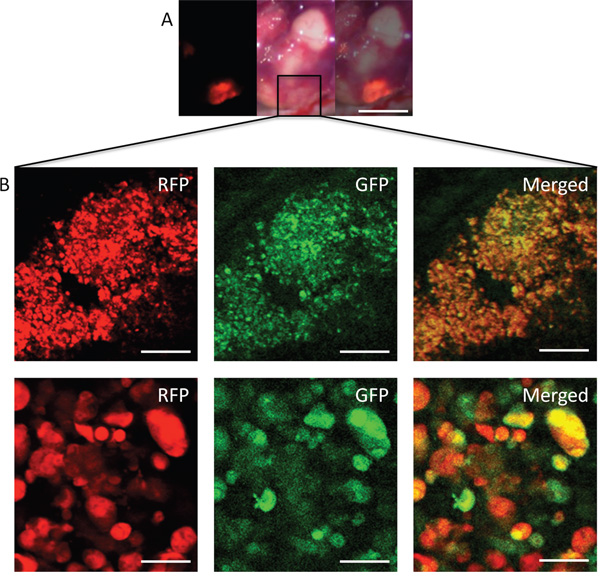
Figure 5: Intravital imaging of S. typhimurium A1-R-GFP targeting HT29-RFP liver metastasis via iPV injection. S. typhimuriumu A1-R-GFP was visualized targeting the HT29-RFP liver metastases at day 14 after iPV injection. A. Liver metastases were visualized with the OV100 by fluorescence (left); brightfield (center) and merge (right). B. Confocal imaging with the FV1000 demonstrated S. typhimuriumu A1-R-GFP targeting the HT29-RFP liver metastasis at the cellular level. Scale bars: (A) 5 mm; (B) Upper panels: 50 μm; Lower panels: 12.5 μm.
Previously developed concepts and strategies of highly selective tumor targeting can take advantage of bacterial targeting of tumors [42–46].
MATERIALS AND METHODS
Cell line
HT29 human colon cancer cells expressing RFP [34, 47–49] were maintained in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA) supplemented with heat-inactivated 10% fetal bovine serum (FBS) (Gemini Biologic Products, Calabasas, CA), 2 mM glutamine, 100 units/ml penicillin, 100 μg/ml streptomycin and 0.25 μg/ml amphotericin B (Life Technologies, Inc., Grand Island, NY). The cells were incubated at 37°C in 5% CO2.
Mice
Athymic nu/nu nude mice (AntiCancer Inc., San Diego, CA), 4–6 weeks old, were used in this study. All mouse surgical procedures and imaging were performed with the animals anesthetized by subcutaneous injection of a ketamine mixture (0.02 ml solution of 20 mg/kg ketamine, 15.2 mg/kg xylazine, and 0.48 mg/kg acepromazine maleate). All animal studies were conducted in accordance with the principles and procedures outlined in the National Institutes of Health Guide for the Care and Use of Animals under Assurance Number A3873-1.
Establishment of liver metastases
HT29-RFP cells were harvested by trypsinization and washed twice with serum-free medium. HT29-RFP cells (5×105 in 50 μl serum-free medium with 50% Matrigel) were injected into the superior and inferior pole of the spleen in nude mice. Three weeks after injection, experimental liver metastases were established.
Surgical orthotopic implantation of liver metastasis
Experimental liver metastases, as described above, were resected and cut into blocks (8 mm3). A single tumor block was orthotopically implanted into the left lobe of the liver in nude mice. Four weeks later, orthotopic liver metastasis were observed at the implanted site by RFP expression.
Preparation of S. typhimurium A1-R
GFP-expressing S. typhimurium A1-R bacteria (AntiCancer Inc.,) were grown overnight on LB medium (Fisher Sci., Hanover Park, IL, USA) and then diluted 1:10 in LB medium. Bacteria were harvested at late-log phase, washed with PBS, and then diluted in PBS [6–8].
iPV injection
The anesthetized mice were placed in a supine position. After disinfecting the skin in the area of surgery, a median abdominal incision was performed followed by mobilization of the duodenum to identify the portal vein. S. typhimurium A1-R (100 μl) was injected into the portal vein using a 31 G needle. After removal of the needle, bleeding was stopped by gently pressing the puncture site with a cotton swab. After injection, the intestine was repositioned and the abdominal wall was closed with non-absorbable sutures.
Targeting of the liver with S. typhimurium A1-R by iPV or iv routes
Nude mice without tumor were injected with S. typhimurium A1-R (iPV: 1×104 CFU; iv: 5×107 CFU). On day 2 after injection, the liver was resected, minced, and mixed with PBS. The minced liver was placed on LB agar to identify S. typhimurium A1-R. Fluorescent S. typhimurium A1-R bacteria were observed with the OV100 Small Animal Imaging System (Olympus Corp., Tokyo, Japan).
Imaging of tumor-targeted bacteria
The OV100 (Olympus) variable-magnification fluorescence imager [50] was used to image colonies of S. typhimurium A1-R from resected tumors. The FV1000 confocal microscope (Olympus) [51] was used to image resected tumors for the presence of S. typhimurium A1-R-GFP.
Efficacy of S. typhimurium A1-R on liver metastasis
Four weeks after orthotopic implantation of HT- 29-RFP to the liver, 21 mice were randomized into 3 groups: untreated control group (n = 7); S. typhimurium A1-R iv treatment group (S. typhimurium A1-R, 5 × 107 CFU/body, iv, n = 7); and S. typhimurium A1-R iPV treatment group (S. typhimurium A1-R, 1 × 104 CFU/body, iPV, n = 7). The left lobe of the liver with metastasis was exposed before (at day 1) and after treatment (at day 15) for imaging with the OV100. The tumor fluorescent area and total fluorescence intensity were analyzed with UVP software (UVP, Upland, CA). Treatment efficacy in each mouse was evaluated by fluorescent area and total fluorescence intensity at day 15 compared to at the beginning of treatment. Liver metastasis in the S. typhimurium A1-R iPV treatment group was also imaged with the FV1000 confocal microscope at day 15 to observe S. typhimurium A1-R-GFP targeting the RFP-expressing HT29 liver metastasis at the cellular level.
Statistical analysis
JMP version 11.0 was used for all statistical analyses. Significant differences for continuous variables were determined using the Mann-Whitney U test. Bar graphs expressed average values, and error bar showed SE. A probability value of P ≤ 0.05 was considered statistically significant.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
DEDICATION
This paper is dedicated to the memory of A.R. Moossa, M.D. and Sun Lee, M.D.
REFERENCES
1. Rothbarth J, van de Velde CJ. Treatment of liver metastases of colorectal cancer. Ann Oncol. 2005; 16 Suppl 2:ii144-9.
2. de Jong MC, Pulitano C, Ribero D, Strub J, Mentha G, Schulick RD, Choti MA, Aldrighetti L, Capussotti L, Pawlik TM. Rates and patterns of recurrence following curative intent surgery for colorectal liver metastasis: an international multi-institutional analysis of 1669 patients. Ann Surg. 2009; 250:440-448.
3. Zhang W, Song T. The progress in adjuvant therapy after curative resection of liver metastasis from colorectal cancer. Drug Discov Ther. 2014; 8:194-200.
4. Zhang Y, Zhang N, Zhao M, Hoffman RM. Comparison of the selective targeting efficacy of Salmonella typhimurium A1-R and VNP20009 on the Lewis lung carcinoma in nude mice. Oncotarget. 2015; 6:14625-14631. doi: 10.18632/oncotarget.3342.
5. Hoffman RM, editor. Bacterial Therapy of Cancer: Methods and Protocols. Methods in Molecular Biology 1409. Walker, John M., series ed. Humana Press (Springer Science+Business Media New York), 2016.
6. Zhao M, Yang M, Li XM, Jiang P, Baranov E, Li S, Xu M, Penman S, Hoffman RM. Tumor-targeting bacterial therapy with amino acid auxotrophs of GFP-expressing Salmonella typhimurium. Proc Natl Acad Sci USA. 2005; 102:755-760.
7. Zhao M, Geller J, Ma H, Yang M, Penman S, Hoffman RM. Monotherapy with a tumor-targeting mutant of Salmonella typhimurium cures orthotopic metastatic mouse models of human prostate cancer. Proc Natl Acad Sci USA. 2007; 104:10170-10174.
8. Zhao M, Yang M, Ma H, Li X, Tan X, Li S, Yang Z, Hoffman RM. Targeted therapy with a Salmonella typhimurium leucine-arginine auxotroph cures orthotopic human breast tumors in nude mice. Cancer Res. 2006; 66:7647-7652.
9. Zhang Y, Tome Y, Suetsugu A, Zhang L, Zhang N, Hoffman RM, Zhao M. Determination of the optimal route of administration of Salmonella typhimurium A1-R to target breast cancer in nude mice. Anticancer Res. 2012; 32:2501-2508.
10. Zhang Y, Miwa S, Zhang N, Hoffman RM, Zhao M. Tumor-targeting Salmonella typhimurium A1-R arrests growth of breast-cancer brain metastasis. Oncotarget. 2015; 6:2615-2622. doi: 10.18632/oncotarget.2811.
11. Uchugonova A, Zhao M, Zhang Y, Weinigel M, König K, Hoffman RM. Cancer-cell killing by engineered Salmonella imaged by multiphoton tomography in live mice. Anticancer Res. 2012; 32:4331-4337.
12. Liu F, Zhang L, Hoffman RM, Zhao M. Vessel destruction by tumor-targeting Salmonella typhimurium A1-R is enhanced by high tumor vascularity. Cell Cycle. 2010; 9:4518-4524.
13. Nagakura C, Hayashi K, Zhao M, Yamauchi K, Yamamoto N, Tsuchiya H, Tomita K , Bouvet M, Hoffman RM. Efficacy of a genetically-modified Salmonella typhimurium in an orthotopic human pancreatic cancer in nude mice. Anticancer Res. 2009; 29:1873-1878.
14. Yam C, Zhao M, Hayashi K, Ma H, Kishimoto H, McElroy M, Bouvet M, Hoffman RM. Monotherapy with a tumor-targeting mutant of S. typhimurium inhibits liver metastasis in a mouse model of pancreatic cancer. J Surg Res. 2010; 164:248-255.
15. Hiroshima Y, Zhao M, Zhang Y, Maawy A, Hassanein MK, Uehara F, Miwa S, Yano S, Momiyama M, Suetsugu A, Chishima T, Tanaka K, Bouvet M, et al. Comparison of efficacy of Salmonella typhimurium A1-R and chemotherapy on stem-like and non-stem human pancreatic cancer cells. Cell Cycle. 2013; 12:2774-2780.
16. Hiroshima Y, Zhao M, Maawy A, Zhang Y, Katz MH, Fleming JB, Uehara F, Miwa S , Yano S, Momiyama M, Suetsugu A, Chishima T, Tanaka K, et al. Efficacy of Salmonella typhimurium A1-R versus chemotherapy on a pancreatic cancer patient-derived orthotopic xenograft (PDOX). J Cell Biochem. 2014; 115:1254-1261.
17. Hiroshima Y, Zhang Y, Murakami T, Maawy AA, Miwa S, Yamamoto M, Yano S, Sato S, Momiyama M, Mori R, Matsuyama R, Chishima T, Tanaka K, et al. Efficacy of tumor-targeting Salmonella typhimurium A1-R in combination with anti-angiogenesis therapy on a pancreatic cancer patient-derived orthotopic xenograph (PDOX) and cell line mouse models. Oncotarget. 2014; 5:12346-12357. doi: 10.18632/oncotarget.2641.
18. Matsumoto Y, Miwa S, Zhang Y, Hiroshima Y, Yano S, Uehara F, Yamamoto M, Toneri M, Bouvet M, Matsubara H, Hoffman RM, Zhao M. Efficacy of tumor-targeting Salmonella typhimurium A1-R on nude mouse models of metastatic and disseminated human ovarian cancer. J Cell Biochem. 2014; 115:1996-2003.
19. Matsumoto Y, Miwa S, Zhang Y, Zhao M, Yano S, Uehara F, Yamamoto M, Hiroshima Y, Toneri M, Bouvet M, Matsubara H, Tsuchiya H, Hoffman RM. Intraperitoneal administration of tumor-targeting Salmonella typhimurium A1-R inhibits disseminated human ovarian cancer and extends survival in nude mice. Oncotarget. 2015; 6:11369-11377. doi: 10.18632/oncotarget.3607.
20. Yano S, Zhang Y, Zhao M, Hiroshima Y, Miwa S, Uehara F, Kishimoto H, Tazawa H, Bouvet M, Fujiwara T, and Hoffman RM. Tumor-targeting Salmonella typhimurium A1-R decoys quiescent cancer cells to cycle as visualized by FUCCI imaging and become sensitive to chemotherapy. Cell Cycle. 2014; 13:3958-3963.
21. Hiroshima Y, Zhang Y, Zhao M, Zhang N, Murakami T, Maawy A, Mii S, Uehara F, Yamamoto M, Miwa S, Yano S, Momiyama M, Mori R, et al. Tumor-targeting Salmonella typhimurium A1-R in combination with Trastuzumab eradicates HER-2-positive cervical cancer cells in patient-derived mouse models. PLoS One. 2015; 10:e0120358.
22. Hayashi K, Zhao M, Yamauchi K, Yamamoto N, Tsuchiya H, Tomita K, Hoffman RM. Cancer metastasis directly eradicated by targeted therapy with a modified Salmonella typhimurium. J Cell Biochem. 2009; 106:992-998.
23. Hayashi K, Zhao M, Yamauchi K, Yamamoto N, Tsuchiya H, Tomita K, Kishimoto H, Bouvet M, Hoffman RM. Systemic targeting of primary bone tumor and lung metastasis of high-grade osteosarcoma in nude mice with a tumor-selective strain of Salmonella typhimurium. Cell Cycle. 2009; 8:870-875.
24. Miwa S, Zhang Y, Baek K-E, Uehara F, Yano S, Yamamoto M, Hiroshima Y, Matsumoto Y, Kimura H, Hayashi K, Yamamoto N, Bouvet M, Tsuchiya H, et al. Inhibition of spontaneous and experimental lung metastasis of soft-tissue sarcoma by tumor-targeting Salmonella typhimurium A1-R. Oncotarget. 2014; 5:12849-12861. doi: 10.18632/oncotarget.2561.
25. Murakami T, DeLong J, Eilber FC, Zhao M, Zhang Y, Zhang N, Singh A, Russell T, Deng S, Reynoso J, Quan C, Hiroshima Y, Matsuyama R, et al. Tumor-targeting Salmonella typhimurium A1-R in combination with doxorubicin eradicate soft tissue sarcoma in a patient-derived orthotopic xenograft PDOX model. Oncotarget. 2016; 7:12783-12790. doi: 10.18632/oncotarget.7226.
26. Momiyama M, Zhao M, Kimura H, Tran B, Chishima T, Bouvet M, Endo I, Hoffman RM. Inhibition and eradication of human glioma with tumor-targeting Salmonella typhimurium in an orthotopic nude-mouse model. Cell Cycle. 2012; 11:628-632.
27. Hiroshima Y, Zhao M, Maawy A, Zhang Y, Katz MH, Fleming JB, Uehara F, Miwa S, Yano S, Momiyama M, Suetsugu A, Chishima T, Tanaka K, et al. Efficacy of Salmonella typhimurium A1-R versus chemotherapy on a pancreatic cancer patient-derived orthotopic xenograft (PDOX). J Cell Biochem. 2014; 115:1254-1261.
28. Hiroshima Y, Zhang Y, Murakami T, Maawy AA, Miwa S, Yamamoto M, Yano S, Sato S, Momiyama M, Mori R, Matsuyama R, Chishima T, Tanaka K, et al. Efficacy of tumor-targeting Salmonella typhimurium A1-R in combination with anti-angiogenesis therapy on a pancreatic cancer patient-derived orthotopic xenograph (PDOX) and cell line mouse models. Oncotarget. 2014; 5:12346-12357. doi: 10.18632/oncotarget.2641.
29. Hiroshima Y, Zhao M, Zhang Y, Zhang N, Maawy A, Murakami T, Mii S, Uehara F, Yamamoto M, Miwa S, Yano S, Momiyama M, Mori R, et al. Tumor-targeting Salmonella typhimurium A1-R arrests a chemo-resistant patient soft-tissue sarcoma in nude mice. PLoS One. 2015; 10:e0134324.
30. Kiyuna T, Murakami T, Tome Y, Kawaguchi K, Igarashi K, Zhang Y, Zhao M, Li Y, Bouvet M, Kanaya F, Singh A, Dry S, Eilber FC, et al. High efficacy of tumor-targeting Salmonella typhimurium A1-R on a doxorubicin- and dactolisib-resistant follicular dendritic-cell sarcoma in a patient-derived orthotopic xenograft PDOX nude mouse model. Oncotarget. 2016; 7:33046-33054. doi: 10.18632/oncotarget.8848.
31. Murakami T, Singh AS, Kiyuna T, Dry SM, Li Y, James AW, Igarashi K, Kawaguchi K, DeLong JC, Zhang Y, Hiroshima Y, Russell T, Eckardt MA, et al. Effective molecular targeting of CDK4/6 and IGF-1R in a rare FUS-ERG fusion CDKN2A-deletion doxorubicin-resistant Ewing’s sarcoma in a patient-derived orthotopic xenograft (PDOX) nude-mouse model. Oncotarget. 2016; 7:47556-64. doi: 10.18632/oncotarget.9879.
32. Kiyuna T, Murakami T, Tome Y, Igarashi K, Kawaguchi K, Russell T, Eckhardt MA, Crompton J, Singh A, Bernthal N, Bukata S, Federman N, Kanaya F, et al. Labeling the stroma of a patient-derived orthotopic xenograft (PDOX) mouse models of undifferentiated pleomorphic soft-tissue sarcoma with red fluorescent protein for rpaid non-invasive drug screening. J Cell Biochem, Epub ahead of print.
33. Yamamoto M, Zhao M, Hiroshima Y, Zhang Y, Shurell E, Eilber FC, Bouvet M, Noda M, Hoffman RM. Efficacy of tumor-targeting Salmonella typhimurium A1-R on a melanoma patient-derived orthotopic xenograft (PDOX) nude-mouse model. PLoS One. 2016; 11:e0160882.
34. Murakami T, Hiroshima Y, Zhao M, Zhang Y, Chishima T, Tanaka K, Bouvet M, Endo I, Hoffman RM. Adjuvant treatment with tumor-targeting Salmonella typhimurium A1-R reduces recurrence and increases survival after liver metastasis resection in an orthotopic nude mouse model. Oncotarget. 2015; 6:41856-41862. doi: 10.18632/oncotarget.6170.
35. Morales F, Bell M, McDonald GO, Cole WH. The prophylactic treatment of cancer at the time of operation. Ann Surg. 1957; 146:588-593.
36. Taylor I, Machin D, Mullee M, Trotter G, Cooke T, West C. A randomized controlled trial of adjuvant portal vein cytotoxic perfusion in colorectal cancer. Br J Surg. 1985; 72:359-363.
37. Long-term results of single course of adjuvant intraportal chemotherapy for colorectal cancer. Swiss Group for Clinical Cancer Research (SAKK). Lancet. 1995; 345:349-353.
38. Wolmark N, Rockette H, Wickerham DL, Fisher B, Redmond C, Fisher ER, Potvin M, Davies RJ, Jones J, Robidoux A, Wexler M, Gordon P, Cruz AB, Jr, et al. Adjuvant therapy of Dukes' A B and C adenocarcinoma of the colon with portal-vein fluorouracil hepatic infusion: preliminary results of National Surgical Adjuvant Breast and Bowel Project Protocol C-02. J Clin Oncol. 1990; 8:1466-1475.
39. Chang W, Wei Y, Ren L, Zhong Y, Yu Y, Chen J, Zhu D, Ye L, Qin C, Zhao N, Niu W, Qin X, Xu J. Randomized controlled trial of intraportal chemotherapy combined with adjuvant chemotherapy (mFOLFOX6) for stage II and III colon cancer. Ann Surg. 2016; 263:434-439.
40. Rashidi B, An Z, Sun F-X, Moossa AR, Hoffman RM. Antimetastatic intraoperative chemotherapy of human colon tumors in the livers of nude mice. Clinical Cancer Res. 2000; 6:2464-2468.
41. Roberts NJ, Zhang L, Janku F, Collins A, Bai RY, Staedtke V, Rusk AW, Tung D, Miller M, Roix J, Khanna KV, Murthy R, Benjamin RS, et al. Intratumoral injection of Clostridium novyi-NT spores induces antitumor responses. Sci Transl Med. 2014; 6:249ra111.
42. Blagosklonny MV. Matching targets for selective cancer therapy. Drug Discov Today. 2003; 8:1104-1107.
43. Blagosklonny MV. Teratogens as anti-cancer drugs. Cell Cycle. 2005; 4:1518-1521.
44. Blagosklonny MV. Treatment with inhibitors of caspases, that are substrates of drug transporters, selectively permits chemotherapy-induced apoptosis in multidrug-resistant cells but protects normal cells. Leukemia. 2001; 15:936-941.
45. Blagosklonny MV. Target for cancer therapy: proliferating cells or stem cells. Leukemia 2006;20:385-391.
46. Apontes P, Leontieva OV, Demidenko ZN, Li F, Blagosklonny MV. Exploring long term protection of normal human fibroblasts and epithelial cells from chemotherapy in cell culture. Oncotarget. 2011; 2:222-233. doi: 10.18632/oncotarget.248.
47. Hoffman RM, Yang M. Subcellular imaging in the live mouse. Nature Protocols. 2006;1:775-782.
48. Hoffman RM, Yang M. Color-coded fluorescence imaging of tumor-host interactions. Nature Protocols. 2006; 1:928-935.
49. Hoffman RM, Yang M. Whole-body imaging with fluorescent proteins. Nature Protocols. 2006; 1:1429-1438.
50. Yamauchi K, Yang M, Jiang P, Xu M, Yamamoto N, Tsuchiya H, Tomita K, Moossa AR, Bouvet M, Hoffman RM. Development of real-time subcellular dynamic multicolor imaging of cancer-cell trafficking in live mice with a variable-magnification whole-mouse imaging system. Cancer Res. 2006; 66:4208-4214.
51. Uchugonova A Duong J Zhang N König K Hoffman RM. The bulge area is the origin of nestin-expressing pluripotent stem cells of the hair follicle. J Cell Biochem. 2011; 112:2046-2050.

