The exact cause of the onset of cancer in this organ is still unknown. However, known risk factors include cigarette smoking, alcohol consumption, diabetes, genetics/heredity, and chronic pancreatitis. Pancreatitis occurs when enzyme secretions build up in the pancreas and it begins to digest itself. The most common form of pancreatic cancer is an exocrine tumor called pancreatic adenocarcinoma, which arises in the cells that line the pancreatic duct.
Pancreatic cancer has an overall poor prognosis. The general 5-year survival rate for people with pancreatic cancer is 10%. However, if detected very early and treated by an oncologist/surgical oncologist, it is treatable and can even be cured.
What is Pancreatic Cancer?
The pancreas is an important organ in humans, between six and ten inches long, and found behind the lower part of the stomach in the upper abdomen. This spongy organ is responsible for helping the body with digestion and blood sugar regulation by releasing the correct amount of enzymes at the appropriate times.
The exact cause of the onset of cancer in this organ is still unknown. However, known risk factors include cigarette smoking, alcohol consumption, diabetes, genetics/heredity, and chronic pancreatitis. Pancreatitis occurs when enzyme secretions build up in the pancreas and it begins to digest itself. The most common form of pancreatic cancer is an exocrine tumor called pancreatic adenocarcinoma, which arises in the cells that line the pancreatic duct.
Pancreatic cancer has an overall poor prognosis. The general 5-year survival rate for people with pancreatic cancer is 10%. However, if detected very early and treated by an oncologist/surgical oncologist, it is treatable and can even be cured.
Physician type:
Oncologist / Surgical oncologist
ANZSRC Categories:
1112 Oncology and Carcinogenesis
RCDC Category: Pancreatic Cancer
Keywords: pancreas, cancer, pancreatic cancer, prognostic, metastasis, survival, therapeutics
Table of Contents
Research Perspectives
Pancreatic cancer driver mutations are targetable through distant alternative RNA splicing dependencies
DOI: 10.18632/oncotarget.27901
Ryan R. Kawalerski, Steven D. Leach and Luisa F. Escobar-Hoyos _
Pancreatic ductal adenocarcinoma (PDAC), the most common histological subtype of pancreatic cancer, has one of the highest case fatality rates of all known solid malignancies. Over the past decade, several landmark studies have established mutations in KRAS and TP53 as the predominant drivers of PDAC pathogenesis and therapeutic resistance, though treatment options for PDACs and other tumors with these mutations remain extremely limited. Hampered by late tumor discovery and diagnosis, clinicians are often faced with using aggressive and non-specific chemotherapies to treat advanced disease. Clinically meaningful responses to targeted therapy are often limited to the minority of patients with susceptible PDACs, and immunotherapies have routinely encountered roadblocks in effective activation of tumor-infiltrating immune cells. Alternative RNA splicing (ARS) has recently gained traction in the PDAC literature as a field from which we may better understand and treat complex mechanisms of PDAC initiation, progression, and therapeutic resistance. Here, we review PDAC pathogenesis as it relates to fundamental ARS biology, with an extension to implications for PDAC patient clinical management.
Editorial
Mechanisms of resistance to mitochondria-targeted therapy in pancreatic cancer
DOI: 10.18632/oncotarget.27976
Stepana Boukalova _, Zuzana Ezrova and Jiri Neuzil
News
LY6 gene family presents a novel class of potential biomarkers associated with overall survival outcome of pancreatic ductal adenocarcinoma
DOI: 10.18632/oncotarget.27968
Claudia Gravekamp _
Priority Research Papers
An exploratory study of metformin with or without rapamycin as maintenance therapy after induction chemotherapy in patients with metastatic pancreatic adenocarcinoma
DOI: 10.18632/oncotarget.27586
Katherine M. Bever _, Erkut H. Borazanci, Elizabeth A. Thompson, Jennifer N. Durham, Kimberly Pinero, Gayle S. Jameson, Amber Vrana, Meizheng Liu, Cara Wilt, Annie A. Wu, Wei Fu, Hao Wang, Yafu Yin, Jeffrey P. Leal, Ana De Jesus-Acosta, Lei Zheng, Daniel A. Laheru, Daniel D. Von Hoff, Elizabeth M. Jaffee, Jonathan D. Powell and Dung T. Le
Purpose: Metformin combined with the mTOR inhibitor rapamycin showed potential synergistic anti-tumor activity in preclinical studies in pancreatic ductal adenocarcinoma (PDA). This phase 1b study (NCT02048384) was conducted to evaluate the feasibility and activity of metformin +/– rapamycin in the maintenance setting for unselected patients with metastatic PDA (mPDA) treated with chemotherapy.
Materials and Methods: Eligible patients with stable or responding mPDA after ≥ 6 months on chemotherapy were randomized 1:1 to metformin alone (Arm A) or with rapamycin (Arm B), stratified by prior treatment with FOLFIRINOX. Fluorodeoxyglucose (FDG) PET scans and peripheral blood mononuclear cells were obtained for exploratory analyses.
Results: 22 subjects (11 per arm) received treatment per protocol. Median PFS/OS were 3.5 and 13.2 months respectively, with 2 year OS rate of 37%; there were no differences between arms. No responses were observed by RECIST; however, decreases in FDG avidity and/or CA19-9 were observed in several long-term survivors. Treatment related adverse events of Grade ≥ 3 occurred in 0% vs 27% of patients in Arm A vs B and were asymptomatic hematologic or electrolyte abnormalities that were not clinically significant. Improved survival was associated with low baseline neutrophil: lymphocyte ratio, baseline lack of assessable disease by PET, and greater expansion of dendritic cells following treatment.
Conclusions: Metformin +/– rapamycin maintenance for mPDA was well-tolerated and several patients achieved stable disease associated with exceptionally long survival. Further prospective studies are needed to clarify the role of these agents in the maintenance setting and to enhance patient selection for such approaches.
Research Perspectives
Targeting desmoplasia in pancreatic cancer as an essential first step to effective therapy
DOI: 10.18632/oncotarget.27745
Nancy D. Ebelt, Vic Zamloot and Edwin R. Manuel _
Pancreatic cancer is considered one of the most lethal cancers in the US. It contributes to an estimated 47,000 deaths annually and is predicted to surpass prostate, breast and colorectal cancers as the leading cause of cancer-related death. Although major advancements in cancer treatment have improved outcomes for many cancer types, survival rate for pancreatic cancer has not improved in nearly four decades despite tremendous effort. One attribute of pancreatic cancer that is considered a major barrier to effective treatment is the formation of fibrotic tissue around tumor cells known as desmoplasia. A number of promising approaches have been developed to deplete fibrotic components in pancreatic tumors to enhance drug delivery, some of which have been tested in clinical trials of advanced, unresectable pancreatic cancer. Here, we discuss previous efforts, shortcomings and new considerations for developing more effective agents to eliminate desmoplasia.
Research Papers
Mebendazole disrupts stromal desmoplasia and tumorigenesis in two models of pancreatic cancer
DOI: 10.18632/oncotarget.28014
Tara Williamson, Michelle Carvalho de Abreu, Dimitri G. Trembath, Cory Brayton, Byunghak Kang, Thais Biude Mendes, Paulo Pimentel de Assumpção, Janete M. Cerutti and Gregory J. Riggins _
The five-year survival rate for metastatic pancreatic cancer is currently only 3%, which increases to 13% with local invasion only and to 39% with localized disease at diagnosis. Here we evaluated repurposed mebendazole, an approved anthelminthic drug, to determine how mebendazole might work at the different stages of pancreatic cancer formation and progression. We asked if mebendazole could prevent initiation of pancreatic intraepithelial neoplasia precursor lesions, interfere with stromal desmoplasia, or suppress tumor growth and liver metastasis. In both the KrasLSL.G12D/+; Pdx1-Cre (KC) mouse model of caerulein-induced inflammatory pancreatitis and the KrasLSL.G12D/+; Tp53R172H/+; Pdx1-Cre (KPC) mouse model of advanced pancreatic cancer, mebendazole significantly reduced pancreas weight, dysplasia and intraepithelial neoplasia formation, compared to controls. Mebendazole significantly reduced trichrome-positive fibrotic connective tissue and α-SMA-positive activated pancreatic stellate cells that heralds fibrogenesis. In the aggressive KPC model, mebendazole significantly suppressed pancreatic tumor growth, both as an early and late intervention. Mebendazole reduced the overall incidence of pancreatic cancer and severity of liver metastasis in KPC mice. Using early models of pancreatic cancer, treatment with mebendazole resulted in less inflammation, decreased dysplasia, with the later stage model additionally showing a decreased tumor burden, less advanced tumors, and a reduction of metastasis. We conclude that mebendazole should be investigated further as a component of adjuvant therapy to slow progression and prevent metastasis, and well as for primary prevention in the highest risk patients.
Research Papers
Scent test using Caenorhabditis elegans to screen for early-stage pancreatic cancer
DOI: 10.18632/oncotarget.28035
Ayumu Asai, Masamitsu Konno, Miyuki Ozaki, Koichi Kawamoto, Ryota Chijimatsu, Nobuaki Kondo, Takaaki Hirotsu and Hideshi Ishii _
Although early detection and diagnosis are indispensable for improving the prognosis of patients with pancreatic cancer, both have yet to be achieved. Except for pancreatic cancer, other cancers have already been screened through scent tests using animals or microorganisms, including Caenorhabditis elegans. While such a method may greatly improve the prognosis of pancreatic cancer, no studies have investigated the same, mainly given the difficulty of collecting suitable samples from patients with early-stage pancreatic cancer. In this study, we organized a nationwide study group comprising high-volume centers throughout Japan to collect patients with very-early-stage pancreatic cancer (stage 0 or IA). We initially performed an open-label study involving 83 cases (stage 0–IV), with subsequent results showing significant differences after surgical removal in stage 0–IA (×10 dilution: p < 0.001; ×100 dilution: p < 0.001). Thereafter, a blinded study on 28 cases (11 patients with stage 0 or IA disease and 17 healthy volunteers) was conducted by comparing very-early-stage pancreatic cancer patients with healthy volunteers to determine whether C. elegans could detect the scent of cancer for the diagnosis of early-stage pancreatic cancer. Preoperative urine samples had a significantly higher chemotaxis index compared to postoperative samples in patients with pancreatic cancer [×10 dilution: p < 0.001, area under the receiver operating characteristic curve (AUC) = 0.845; ×100 dilution: p < 0.001, AUC = 0.820] and healthy volunteers (×10 dilution: p = 0.034; ×100 dilution: p = 0.088). Moreover, using the changes in preoperative and postoperative chemotaxis index, this method had a higher sensitivity for detecting early pancreatic cancer compared to existing diagnostic markers. The clinical application C. elegans for the early diagnosis of cancer can certainly be expected in the near future.
Research Papers
Perioperative changes in the plasma metabolome of patients receiving general anesthesia for pancreatic cancer surgery
DOI: 10.18632/oncotarget.27956
Johanna Mock-Ohnesorge _, Andreas Mock, Thilo Hackert, Stefan Fröhling, Judith Schenz, Gernot Poschet, Dirk Jäger, Markus W. Büchler, Florian Uhle and Markus A. Weigand
Background: Modern anesthesia strives to offer personalized concepts to meet the patient’s individual needs in sight of clinical outcome. Still, little is known about the impact of anesthesia on the plasma metabolome, although many metabolites have been shown to modulate the function of various immune cells, making it particularly interesting in the context of oncological surgery. In this study longitudinal dynamics in the plasma metabolome during general anesthesia in patients undergoing pancreatic surgery were analyzed.
Materials and Methods: Prospective, observational study with 10 patients diagnosed with pancreatic (pre-) malignancy and subjected to elective resection surgery under general anesthesia. Plasma metabolites (n = 630) were quantified at eight consecutive perioperative timepoints using mass spectrometry-based targeted metabolomics.
Results: 39 metabolites significantly changed during the perioperative period. Tryptophan concentrations decreased by 45% with the maximum decrease after anesthesia induction (p = 6.24E-07), while taurine synthesis increased (p = 1.46E-04). Triacylglycerides and lysophosphatidylcholines were significantly reduced with increased liberation of free monounsaturated fatty acids (p = 0.03). Carnitine levels decreased significantly (p = 9.30E-04).
Conclusions: The major finding of this study was perioperative tryptophan depletion and increased taurine synthesis. Both are essential for immune cell function and are therefore of significant interest for perioperative management. Further studies are needed to identify influencing anesthetic factors.
Research Papers
A comprehensive analysis of clinical trials in pancreatic cancer: what is coming down the pike?
DOI: 10.18632/oncotarget.27727
Erryk S. Katayama, Jonathan J. Hue, David L. Bajor, Lee M. Ocuin, John B. Ammori, Jeffrey M. Hardacre and Jordan M. Winter _
Objective: Pancreatic cancer is the most aggressive common cancer and is desperately in need of novel therapies. Unlike many other common cancers, there have been no new paradigm-changing therapies in the past 40 years beyond multi-agent chemotherapy. In this study, we perform the first comprehensive analysis of the current clinical trial landscape in pancreatic cancer to better understand the pipeline of new therapies.
Materials and Methods: We queried
Results: As of May 18, 2019, there were 430 total active therapeutic interventional trials testing 590 interventions. The vast minority of trials (n = 37, 8.6%) are in phase III testing. 189 (31%) interventions are immunotherapies, 69 (11%) target cell signaling pathways, 154 (26%) target cell cycle or DNA biology, and 35 (6%) target metabolic pathways. Of the late phase trials, only 14 are currently testing novel interventions. Rather, 23 phase III trials examine new ways to deliver existing FDA-approved drugs, procedures, or pain management.
Conclusions: A large number of novel therapeutic strategies are currently under investigation. They include a broad range of therapies targeting diverse biologic processes. However, only a small number of novel therapies are in late-stage testing, suggesting that future progress is likely several years away, and dependent on the success of early-stage trials.
Research Papers
A microRNA-based signature predicts local-regional failure and overall survival after pancreatic cancer resection
DOI: 10.18632/oncotarget.27496
Adam R. Wolfe, Patrick Wald, Amy Webb, Nikhil Sebastian, Steve Walston, Ryan Robb, Wei Chen, Marall Vedaie, Mary Dillhoff, Wendy L. Frankel, Wooil Kwon, Jin-Young Jang and Terence M. Williams _
Resectable pancreatic adenocarcinoma (PC) is generally managed with surgery followed by chemotherapy, but the role of postoperative chemoradiation (pCRT) is controversial. We sought to identify a microRNA (miRNA) expression profile associated with higher risk for local-regional recurrence (LRR), which might help identify patients that may benefit from pCRT. Total RNA was isolated from viable tumor from 88 patients who underwent PC resection with or without chemotherapy, but did not receive radiation. Digital miRNA expression profiling was performed and risk scores were calculated based on the expression levels of the four most significantly correlated miRNAs, and dichotomized about the median to detect correlations between risk group, LRR and overall survival (OS). Two cohorts from The Cancer Genome Atlas (TCGA) and Seoul National University (SNU) were used for validation. Patients with high-risk scores had significantly worse LRR (p = 0.001) and worse OS (p = 0.034). Two-year OS rates for the high- and low-risk groups were 27.7% and 52.2%, respectively. On multivariable analysis, the risk score remained significantly associated with LRR (p = 0.018). When validated on TCGA data, a high-risk score was associated with worse OS on univariate (p = 0.03) and multivariable analysis (p = 0.017). When validated on the SNU cohort, a high-risk score was likewise associated with worse OS (p = 0.042). We have developed a 4-miRNA molecular signature that is associated with risk of LRR and OS after PC resection and validated on two separate cohorts. This signature has the potential to select patients most likely to benefit from pCRT, and should be tested further.
Research Papers
Dynamic changes during the treatment of pancreatic cancer
DOI: 10.18632/oncotarget.24483
Robert A. Wolff, Andrea Wang-Gillam, Hector Alvarez, Hervé Tiriac, Dannielle Engle, Shurong Hou, Abigail F. Groff, Anthony San Lucas, Vincent Bernard, Kelvin Allenson, Jonathan Castillo, Dong Kim, Feven Mulu, Jonathan Huang, Bret Stephens, Ignacio I. Wistuba, Matthew Katz, Gauri Varadhachary, YoungKyu Park, James Hicks, Arul Chinnaiyan, Louis Scampavia, Timothy Spicer, Chiara Gerhardinger, Anirban Maitra, David Tuveson, John Rinn, Gregory Lizee, Cassian Yee and Arnold J. Levine _
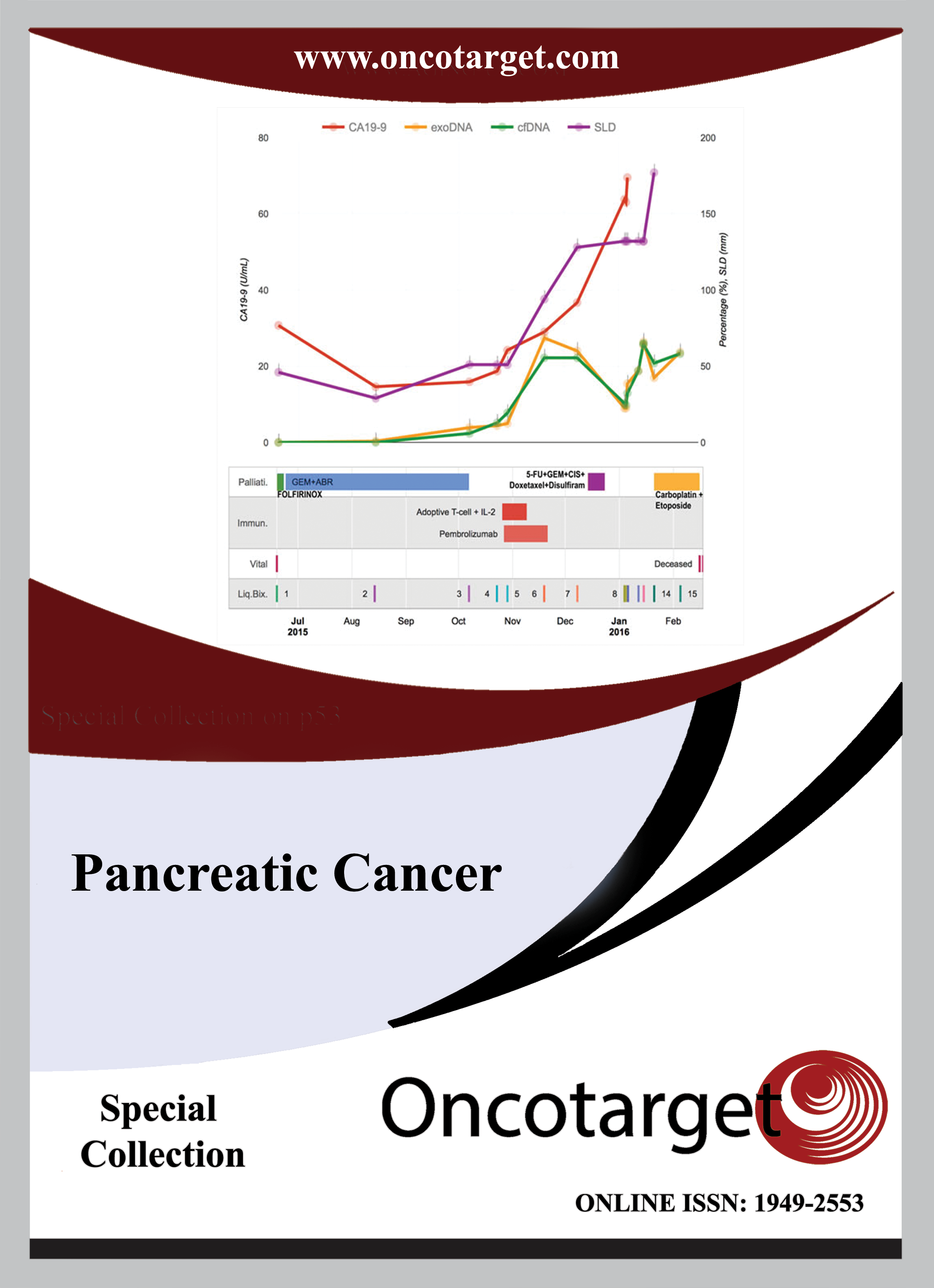
This manuscript follows a single patient with pancreatic adenocarcinoma for a five year period, detailing the clinical record, pathology, the dynamic evolution of molecular and cellular alterations as well as the responses to treatments with chemotherapies, targeted therapies and immunotherapies. DNA and RNA samples from biopsies and blood identified a dynamic set of changes in allelic imbalances and copy number variations in response to therapies. Organoid cultures established from biopsies over time were employed for extensive drug testing to determine if this approach was feasible for treatments. When an unusual drug response was detected, an extensive RNA sequencing analysis was employed to establish novel mechanisms of action of this drug. Organoid cell cultures were employed to identify possible antigens associated with the tumor and the patient’s T-cells were expanded against one of these antigens. Similar and identical T-cell receptor sequences were observed in the initial biopsy and the expanded T-cell population. Immunotherapy treatment failed to shrink the tumor, which had undergone an epithelial to mesenchymal transition prior to therapy. A warm autopsy of the metastatic lung tumor permitted an extensive analysis of tumor heterogeneity over five years of treatment and surgery. This detailed analysis of the clinical descriptions, imaging, pathology, molecular and cellular evolution of the tumors, treatments, and responses to chemotherapy, targeted therapies, and immunotherapies, as well as attempts at the development of personalized medical treatments for a single patient should provide a valuable guide to future directions in cancer treatment.
Reviews
Prognostic and predictive factors in pancreatic cancer
DOI: 10.18632/oncotarget.27518
Emanuela Dell’Aquila, Claudia Angela Maria Fulgenzi, Alessandro Minelli, Fabrizio Citarella _, Marco Stellato, Francesco Pantano, Marco Russano, Maria Concetta Cursano, Andrea Napolitano, Tea Zeppola, Bruno Vincenzi, Giuseppe Tonini and Daniele Santini
Pancreatic cancer is one of the leading causes of cancer death worldwide. Its high mortality rate has remained unchanged for years. Radiotherapy and surgery are considered standard treatments in early and locally advanced stages. Chemotherapy is the only option for metastatic patients. Two treatment regimens, i. e. the association of 5-fluorouracil- irinotecan-oxaliplatin (FOLFIRINOX) and the association of nab-paclitaxel with gemcitabine, have been shown to improve outcomes for metastatic pancreatic adenocarcinoma patients. However, there are not standardized predictive biomarkers able to identify patients who benefit most from treatments. CA19-9 is the most studied prognostic biomarker, its predictive role remains unclear. Other clinical, histological and molecular biomarkers are emerging in prognostic and predictive settings. The aim of this review is to provide an overview of prognostic and predictive markers used in clinical practice and to explore the most promising fields of research in terms of treatment selection and tailored therapy in pancreatic cancer.
Research Papers
Genome-wide DNA methylation analysis reveals molecular subtypes of pancreatic cancer
DOI: 10.18632/oncotarget.15993
Nitish Kumar Mishra and Chittibabu Guda _
Pancreatic cancer (PC) is the fourth leading cause of cancer deaths in the United States with a five-year patient survival rate of only 6%. Early detection and treatment of this disease is hampered due to lack of reliable diagnostic and prognostic markers. Recent studies have shown that dynamic changes in the global DNA methylation and gene expression patterns play key roles in the PC development; hence, provide valuable insights for better understanding the initiation and progression of PC. In the current study, we used DNA methylation, gene expression, copy number, mutational and clinical data from pancreatic patients. We independently investigated the DNA methylation and differential gene expression profiles between normal and tumor samples and correlated methylation levels with gene expression patterns. We observed a total of ~23-thousand differentially methylated CpG sites (Δβ≥0.1) between normal and tumor samples, where majority of the CpG sites are hypermethylated in PC, and this phenomenon is more prominent in the 5’UTRs and promoter regions compared to the gene bodies. Differential methylation is observed in genes associated with the homeobox domain, cell division and differentiation, cytoskeleton, epigenetic regulation and development, pancreatic development and pancreatic signaling and pancreatic cancer core signaling pathways. Correlation analysis suggests that methylation in the promoter region and 5’UTR has mostly negative correlations with gene expression while gene body and 3’UTR associated methylation has positive correlations. Regulatory element analysis suggests that HOX cluster and histone core proteins are upstream regulators of hypomethylation, while SMAD4, STAT4, STAT5B and zinc finger proteins (ZNF) are upstream regulators of hypermethylation. Non-negative matrix factorization (NMF) clustering of differentially methylated sites generated three clusters in PCs suggesting the existence of distinct molecular subtypes. Cluster 1 and cluster 2 showed samples enriched with clinical phenotypes like neoplasm histological grade and pathologic T-stage T3, respectively, while cluster 3 showed the enrichment of samples with neoplasm histological grade G1. To the best of our knowledge, this is the first genome-scale methylome analysis of PC data from TCGA. Our clustering analysis provides a strong basis for future work on the molecular subtyping of epigenetic regulation in pancreatic cancer.
Priority Research Papers
Anti-pancreatic cancer activity of ONC212 involves the unfolded protein response (UPR) and is reduced by IGF1-R and GRP78/BIP
DOI: 10.18632/oncotarget.20819
Avital Lev _, Amriti R. Lulla, Jessica Wagner, Marie D. Ralff, Joshua B. Kiehl, Yan Zhou, Cyril H. Benes, Varun V. Prabhu, Wolfgang Oster, Igor Astsaturov, David T. Dicker and Wafik S. El-Deiry
Pancreatic cancer is chemo-resistant and metastasizes early with an overall five-year survival of ~8.2%. First-in-class imipridone ONC201 is a small molecule in clinical trials with anti-cancer activity. ONC212, a fluorinated-ONC201 analogue, shows preclinical efficacy in melanoma and hepatocellular-cancer models. We investigated efficacy of ONC201 and ONC212 against pancreatic cancer cell lines (N=16 including 9 PDX-cell lines). We demonstrate ONC212 efficacy in 4 in-vivo models including ONC201-resistant tumors. ONC212 is active in pancreatic cancer as single agent or in combination with 5-fluorouracil, irinotecan, oxaliplatin or RTK inhibitor crizotinib. Based on upregulation of pro-survival IGF1-R in some tumors, we found an active combination of ONC212 with inhibitor AG1024, including in vivo. We show a rationale for targeting pancreatic cancer using ONC212 combined with targeting the unfolded-protein response and ER chaperones such as GRP78/BIP. Our results lay the foundation to test imipridones, anti-cancer agents, in pancreatic cancer, that is refractory to most drugs.
Reviews
Germline mutations in pancreatic cancer and potential new therapeutic options
DOI: 10.18632/oncotarget.17291
Rille Pihlak, Juan W. Valle and Mairéad G. McNamara _
Due to short-lived treatment responses in unresectable disease, pancreatic ductal adenocarcinoma (PDAC) continues to be one of the deadliest cancers. There is availability of new information about germline and sporadic mutations in the deoxyribonucleic acid (DNA) damage repair pathway in PDAC in recent decades and the expectation is that novel targeted therapies will thus be developed. A variety of germline mutations (BRCA2, BRCA1, PALB2, CDKN2A, ATM, TP53 and mismatch repair genes MLH1, MSH2, MSH6) have been reported in these patients with the highest prevalence being BRCA1/2. Positive results have been reported with the use of targeted therapies, particularly poly (ADP-ribose) polymerase inhibitors in BRCA-mutated ovarian and breast cancers, and their use is currently being investigated in germline-mutated pancreatic cancer. The aim of this review is to provide an outline of germline DNA damage repair mutations in pancreatic cancer and their effect on the incidence, outcomes and responses to different therapeutic options.
Research Papers
The anti-mesothelin monoclonal antibody amatuximab enhances the anti-tumor effect of gemcitabine against mesothelin-high expressing pancreatic cancer cells in a peritoneal metastasis mouse model
DOI: 10.18632/oncotarget.26117
Tatsuzo Mizukami _, Hirofumi Kamachi, Yuki Fujii, Fumihiko Matsuzawa, Takahiro Einama, Futoshi Kawamata, Nozomi Kobayashi, Yutaka Hatanaka and Akinobu Taketomi
Pancreatic cancer often has a very poor prognosis, even after complete resection. The recurrence of hepatic and peritoneal metastases is an important prognostic factor; therefore, the development of improved adjuvant therapy is urgently required. Mesothelin is a cell surface glycoprotein whose expression is restricted to a variety of cancer types, including pancreatic cancer. This expression pattern makes mesothelin an attractive target for cancer therapy, and several agents targeting mesothelin are currently in clinical trials. Here, we used the chimerized high-affinity anti-mesothelin monoclonal antibody amatuximab to investigate its effect on peritoneal metastasis. We used the AsPC-1 pancreatic cancer cell line engineered to express Gaussia luciferase (Gluc), (AsPC-1-Gluc) for in vivo experiments. Results showed that while amatuximab was not directly cytotoxic on an AsPC-1-Gluc tumor cells in a peritoneal metastasis model, it prevented the formation of tumor growth. In combination therapy with gemcitabine, amatuximab exhibited synergistic killing. Our results suggest that blockade of mesothelin by amatuximab may be a useful strategy for preventing the peritoneal dissemination of pancreatic cancer under an adjuvant setting.
Research Papers
The role of circulating miRNAs and CA19-9 in pancreatic cancer diagnosis
DOI: 10.18632/oncotarget.28038
Nivaldo Faria Vieira, Luciano Neder Serafini, Paulo Cezar Novais, Fermino Sanches Lizarte Neto, Mucio Luiz de Assis Cirino, Rafael Kemp, José Celso Ardengh, Fabiano Pinto Saggioro, Alberto Facury Gaspar, Ajith Kumar Sankarankutty, Jorge Resende Lopes Júnior, Daniela Pretti da Cunha Tirapelli and José Sebastião dos Santos _
Diagnosis and treatment of pancreatic ductal adenocarcinoma (PA) remains a challenge in clinical practice. The aim of this study was to assess the role of microRNAs (miRNAs-21, -23a, -100, -107, -181c, -210) in plasma and tissue as possible biomarkers in the diagnosis of PA. Samples of plasma (PAp-n = 13), pancreatic tumors (PAt-n = 18), peritumoral regions (PPT-n = 9) were collected from patients during the surgical procedure. The control group consisted of samples from patients submitted to pancreatic surgery for trauma or cadaveric organs (PC-n = 7) and healthy volunteers donated blood (PCp-n = 6). The expression profile of microRNAs was measured in all groups using RT-PCR, serum CA19-9 levels were determined in PA and PC. In tissue samples, there was a difference in the expression of miRNAs-21, -210 (p < 0.05) across the PAt, PC and PPT groups. The PAp showed overexpression of miRNAs-181c, -210 (p < 0.05) when compared to PCp. The combination of miRNAs-21, -210 tissue expression and serum CA19-9 showed 100% accuracy in the diagnosis of PA, as well as miR-181c expression in the plasma (PApxPCp). The expression of microRNAs in plasma proved to be a promising tool for a noninvasive detection test for PA, as well as further studies will evaluate the utility of microRNAs expression as biomarkers for prognostic and response to therapy in PA.
Clinical Research Papers
RNAi therapy targeting KRAS in combination with chemotherapy for locally advanced pancreatic cancer patients
Talia Golan _, Elina Zorde Khvalevsky, Ayala Hubert, Rachel Malka Gabai, Naama Hen, Amiel Segal, Abraham Domb, Gil Harari, Eliel Ben David, Stephen Raskin, Yuri Goldes, Eran Goldin, Rami Eliakim, Maor Lahav, Yael Kopleman, Alain Dancour, Amotz Shemi and Eithan Galun
Purpose: The miniature biodegradable implant siG12D-LODER™ was inserted into a tumor and released a siRNA drug against KRAS(G12D) along four months. This novel siRNA based drug was studied, in combination with chemotherapy, as targeted therapy for Locally Advanced Pancreatic Cancer (LAPC).
Methods: An open-label Phase 1/2a study in the first-line setting of patients with non-operable LAPC was initiated. In this study patients were assigned to receive a single dose of siG12D-LODERs, in three escalating dose cohorts (0.025mg, 0.75mg and 3.0mg). Gemcitabine was given on a weekly basis, following the siG12D-LODERTM insertion, until disease progression. The recommended dose was further examined with modified FOLFIRINOX. The follow up period was eight weeks and survival until death.
Results: Fifteen patients with LAPC were enrolled. Among the 15 treated patients, the most frequent adverse events observed were grade 1or 2 in severity (89%); five patients experienced serious adverse events (SAEs). In 12 patients analyzed by CT scans, none showed tumor progression, the majority (10/12) demonstrated stable disease and two showed partial response. Decrease in tumor marker CA19-9 was observed in 70% (7/10) of patients. Median overall survival was 15.12 months; 18 month survival was 38.5%.
Conclusions: The combination of siG12D-LODER™ and chemotherapy is well tolerated, safe and demonstrated a potential efficacy in patients with LAPC. NCT01188785
Research Papers
Aspirin counteracts cancer stem cell features, desmoplasia and gemcitabine resistance in pancreatic cancer
Yiyao Zhang, Li Liu, Pei Fan, Nathalie Bauer, Jury Gladkich, Eduard Ryschich, Alexandr V. Bazhin, Nathalia A. Giese, Oliver Strobel, Thilo Hackert, Ulf Hinz, Wolfgang Gross, Franco Fortunato and Ingrid Herr _
Pancreatic ductal adenocarcinoma (PDA) is characterized by an extremely poor prognosis. An inflammatory microenvironment triggers the pronounced desmoplasia, the selection of cancer stem-like cells (CSCs) and therapy resistance. The anti-inflammatory drug aspirin is suggested to lower the risk for PDA and to improve the treatment, although available results are conflicting and the effect of aspirin to CSC characteristics and desmoplasia in PDA has not yet been investigated. We characterized the influence of aspirin on CSC features, stromal reactions and gemcitabine resistance. Four established and 3 primary PDA cell lines, non-malignant cells, 3 patient tumor-derived CSC-enriched spheroidal cultures and tissues from patients who did or did not receive aspirin before surgery were analyzed using MTT assays, flow cytometry, colony and spheroid formation assays, Western blot analysis, antibody protein arrays, electrophoretic mobility shift assays (EMSAs), immunohistochemistry and in vivo xenotransplantation. Aspirin significantly induced apoptosis and reduced the viability, self-renewal potential, and expression of proteins involved in inflammation and stem cell signaling. Aspirin also reduced the growth and invasion of tumors in vivo, and it significantly prolonged the survival of mice with orthotopic pancreatic xenografts in combination with gemcitabine. This was associated with a decreased expression of markers for progression, inflammation and desmoplasia. These findings were confirmed in tissue samples obtained from patients who had or had not taken aspirin before surgery. Importantly, aspirin sensitized cells that were resistant to gemcitabine and thereby enhanced the therapeutic efficacy. Aspirin showed no obvious toxic effects on normal cells, chick embryos or mice. These results highlight aspirin as an effective, inexpensive and well-tolerated co-treatment to target inflammation, desmoplasia and CSC features PDA.
Research Papers
The effects of curcumin (diferuloylmethane) on body composition of patients with advanced pancreatic cancer
Henrique A. Parsons _, Vickie E. Baracos, David S. Hong, James Abbruzzese, Eduardo Bruera and Razelle Kurzrock
Background: Curcumin is a natural product that is often explored by patients with cancer. Weight loss due to fat and muscle depletion is a hallmark of pancreatic cancer and is associated with worse outcomes. Studies of curcumin’s effects on muscularity show conflicting results in animal models.
Methods and results: Retrospective matched 1:2 case-control study to evaluate the effects of curcumin on body composition (determined by computerized tomography) of 66 patients with advanced pancreatic cancer (22 treated,44 controls). Average age (SEM) was 63(1.8) years, 30/66(45%) women, median number of prior therapies was 2, median (IQR) time from advanced pancreatic cancer diagnosis to baseline image was 7(2-13.5) months (p>0.2, all variables). All patients lost weight (3.3% and 1.3%, treated vs. control, p=0.13). Treated patients lost more muscle (median [IQR] percent change -4.8[-9.1,-0.1] vs. -0.05%[-4.2, 2.6] in controls,p<0.001) and fat (median [IQR] percent change -6.8%[-15,-0.6] vs. -4.0%[-7.6, 1.3] in controls,p=0.04). Subcutaneous fat was more affected in the treated patients. Sarcopenic patients treated with curcumin(n=15) had survival of 169(115-223) days vs. 299(229-369) sarcopenic controls(p=0.024). No survival difference was found amongst non-sarcopenic patients.
Conclusions: Patients with advanced pancreatic cancer treated with curcumin showed significantly greater loss of subcutaneous fat and muscle than matched untreated controls.
Research Papers
A screen for inducers of bHLH activity identifies pitavastatin as a regulator of p21, Rb phosphorylation and E2F target gene expression in pancreatic cancer
DOI: 10.18632/oncotarget.18587
Nicholas Villarino, Lia Signaevskaia, Jaco van Niekerk, Rachel Medal, Heejung Kim, Reyhaneh Lahmy, Kathleen Scully, Anthony Pinkerton, SangWun Kim, Andrew Lowy and Pamela Itkin-Ansari _
The average survival for patients with Pancreatic Ductal Adenocarcinoma (PDA) is merely 6 months, underscoring the need for new therapeutic approaches. During PDA progression, pancreatic acinar cells lose activity of the ClassI/II bHLH factors that regulate quiescence. We previously found that promoting transcriptional activity of the Class I bHLH factor E47 in highly aggressive PDA cells induced stable growth arrest in vitro and in vivo. To translate these findings for clinical utility, we developed a high throughput screening platform to identify small molecule inducers of Class I/II bHLH activity. A screen of 4,375 known drugs identified 70 bHLH activators. Prominent among the hits were members of the statin class of HMG-CoA reductase inhibitors, cholesterol lowering drugs that are also being evaluated in cancer. Studies with pitavastatin in primary patient derived tumor cells and established PDA lines, revealed dose dependent growth inhibition. At the molecular level, pitavastatin induced expression of the cyclin dependent kinase (CDK) inhibitor p21 in a cholesterol independent manner, blocked repressive phosphorylation of the Retinoblastoma tumor suppressor protein at CDK targeted sites, and reduced expression of E2F target genes required for progression through the G1/S boundary. Together, the data provide new insight into mechanisms by which statins constrain proliferation in cancer and establish the effectiveness of a novel screening platform to identify small molecules of clinical relevance in pancreatic cancer.
Research Papers
Association of Fusobacterium species in pancreatic cancer tissues with molecular features and prognosis
Kei Mitsuhashi _, Katsuhiko Nosho, Yasutaka Sukawa, Yasutaka Matsunaga, Miki Ito, Hiroyoshi Kurihara, Shinichi Kanno, Hisayoshi Igarashi, Takafumi Naito, Yasushi Adachi, Mami Tachibana, Tokuma Tanuma, Hiroyuki Maguchi, Toshiya Shinohara, Tadashi Hasegawa, Masafumi Imamura, Yasutoshi Kimura, Koichi Hirata, Reo Maruyama, Hiromu Suzuki, Kohzoh Imai, Hiroyuki Yamamoto and Yasuhisa Shinomura
Recently, bacterial infection causing periodontal disease has attracted considerable attention as a risk factor for pancreatic cancer. Fusobacterium species is an oral bacterial group of the human microbiome. Some evidence suggests that Fusobacterium species promote colorectal cancer development; however, no previous studies have reported the association between Fusobacterium species and pancreatic cancer. Therefore, we examined whether Fusobacterium species exist in pancreatic cancer tissue. Using a database of 283 patients with pancreatic ductal adenocarcinoma (PDAC), we tested cancer tissue specimens for Fusobacterium species. We also tested the specimens for KRAS, NRAS, BRAF and PIK3CA mutations and measured microRNA-21 and microRNA-31. In addition, we assessed epigenetic alterations, including CpG island methylator phenotype (CIMP). Our data showed an 8.8% detection rate of Fusobacterium species in pancreatic cancers; however, tumor Fusobacterium status was not associated with any clinical and molecular features. In contrast, in multivariate Cox regression analysis, compared with the Fusobacterium species-negative group, we observed significantly higher cancer-specific mortality rates in the positive group (p = 0.023). In conclusion, Fusobacterium species were detected in pancreatic cancer tissue. Tumor Fusobacterium species status is independently associated with a worse prognosis of pancreatic cancer, suggesting that Fusobacterium species may be a prognostic biomarker of pancreatic cancer.
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC