INTRODUCTION
Protein is the fundamental entity in the maintenance of cellular functions [1]. Optimum protein performance is highly dependent on the network of molecular chaperones, which are constituents of protein quality control system [2]. The chaperones function to check and facilitate the refolding of misfolded polypeptides, thus responsible for degradation of ubiquitin-tagged proteins [3].
Carboxyl terminus of heat shock cognate protein 70 (Hsc70)-interacting protein (CHIP), a co-chaperone E3 ligase, was initially identified as a novel tetratricopeptide repeat (TPR)-containing protein in 1999 [4]. It is also known as STIP1 homology and U-box containing protein 1 (STUB1). This gene encodes a novel 34.5-kDa cytoplasmic protein with a deduced amino acid sequence of 303 residues. The expression of CHIP is ubiquitously but varies among tissues. For example, hypermetabolic tissues such as skeletal muscle and heart have high expression. As CHIP is an evolutionarily ancient protein, chip gene shows extensive phylogenetic conservation [4-6]. Human CHIP exhibits 98% amino acid similarity with mouse CHIP and 60% with Drosophila CHIP [1].
As a connecting link between chaperones and ubiquitin proteasomes, CHIP is actively involved in a diverse array of cellular processes such as protein refolding, degradation, protein trafficking, signaling, transcription and apoptosis [7]. Hence, it is not surprising that CHIP regulates abundant proteins and consequently ailments which arise due to abnormalities in those proteins. This review provides the current understanding of CHIP and its binding partners, followed by the diverse roles of CHIP in human disorders, with a focus of cancer.
CHIP structure and function
CHIP comprises triple tandem TPR domains, a U-box domain and a central coiled coil domain (Figure 1) [8]. The 34-amino-acid TPR domains at the amino terminus with an adjacent charged region (amino acid residues 1-197) together form a binding site for heat shock protein (Hsp)/Hsc70 and Hsp90. Notably, the recruitment of Hsp/Hsc70 by CHIP involves a reciprocal allosteric interaction between the TPR and U-box domains [9]. Such an interaction stimulates numerous biochemical reactions and subsequent physiological functions. For instance, CHIP and Hsp90 heterocomplex elicits release of the regulatory cofactor p23, thereby suppressing the affinity and refolding activity of Hsp90 for substrate proteins [5,10,11]. On the other hand, CHIP competes for Hsp70 from Hsp40/Hsp70 complex, which attenuates their ATPase activity and refolding capacity for denatured substrates [4]. Consequently, due to inhibition of accompanying Hsp, CHIP acts as a bridge between chaperones and the proteasome system; that is, CHIP transforms the refolding machinery into the destructive pathway [12].
At the carboxyl terminus, the U-box domain contributes to its ligase activity [13]. Of note, CHIP is the first identified chaperone that possesses intrinsic E3 ubiquitin ligase activity. General E3 ubiquitin ligases contain a Homologous to E6AP Carboxyl Terminus (HECT) or Really Interesting New Gene (RING) domain. U-box domain in CHIP is structurally and functionally similar to the RING domain [13]. The U-box domain can bind to UbcH4/UbcH5 and deliver ubiquitin molecule from E2 to an unfolded substrate protein, whereas CHIP (ΔU) lacking the U-box domain cannot [6]. Between the TPR and U-box domains, the central domain is rich in charged residues, with two possible nuclear localization signals. This charged domain might facilitate TPR-dependent interactions [1].
Although the chaperone functions of CHIP have been well characterized over the past decades, the underling mechanisms of proteasomal degradation remain largely unclear. S5a and HC8 proteasome subunits [5] or Bcl-2-associated athanogene 1 (BAG-1) [14] have been found to facilitate cooperation between CHIP and proteasome. Particularly, BAG-1 binds to proteasome via its carboxy-terminal BAG domain, thus targeting chaperone substrates for degradation [14]. BAG-2, however, inhibits the ubiquitin ligase activity by abrogating the CHIP/E2 interaction [15].

Figure 1: Structure of CHIP, a 34.5-kDa cytoplasmic protein with a deduced amino acid sequence of 303 residue. CHIP consists of triple tandem TPR domains, U-box domain and a central coiled coil domain.
CHIP interaction with functional protein
Interactions between CHIP and heat shock protein
Recent advances in cellular biology and biochemistry have led to widespread acceptance of the concept that CHIP is a bona fide binding partner to diverse proteins, of which Hsp is the major one. Similar to CHIP, Hsp is highly conserved across species and widely distributed among plant and animal cells. Any stress, including heat, hypoxia, as well as cancer, can induce Hsp production. Normally, it functions as chaperone to participate in refolding of vital cellular proteins, which promotes cellular proliferation and inhibits apoptosis. For example, Hsp70 increases the expression of anti-apoptotic protein Bcl-2, further protecting neurons and astrocytes from anoxic conditions [16, 17].
Previous studies have demonstrated that Hsp70-/Hsp90-dependent chaperone machinery is required for CHIP activity. Interestingly, Hsp90 stabilizes associated client proteins, whereas Hsp70 promotes protein degradation by polyubiquitination [18]. Via this mechanism, CHIP ubiquitinates and degrades glucocorticoid receptor (GR), androgen receptor (AR), estrogen receptor (ER), ErbB2, and α-synuclein, only when bound to Hsp [19-21]. Theoretically, all of the substrates of Hsp70 or Hsp90 are potential substrates of CHIP. On the other hand, CHIP can directly bind to specific substrates independent of Hsp. Rosser MF et al. [22] demonstrated that, CHIP bound to certain proteins (such as Smad1, HSF, Raf-1) through intrinsic polypeptide binding sites, indicating CHIP itself is a molecular chaperone upon stress [23]. Interestingly, CHIP plays a dual role in regulation of Hsp70 expression. In the early stage of stress, CHIP increases heat shock factor 1 (HSF1) activity, thereby enhancing Hsp70 expression and inhibiting apoptosis [24]. In the recovery stage, however, CHIP leads to ubiquitination and degradation of the Hsp70-bound denatured protein as well as of Hsp70 [25]. This dual-phase modulation allows for cellular homeostasis.
Interaction between CHIP and membrane receptors
Her2/ErbB2. Her2/ErbB2, a member of the epidermal growth factor receptor (EGFR) family, is a transmembrane protein tyrosine kinase. As an oncogene, phosphorylated Her2 triggers activation of various signaling pathways involved in cell proliferation and tumorigenesis. Her2 is overexpressed in 30% of breast cancer and correlates with disease recurrence and prognosis [26]. In the presence of Hsp90, CHIP ubiquitylates Her2, decreasing its endogenous expression. Moreover, CHIP regulates Hsp90 inhibitor geldanamycin (GA) to promote Her2 degradation [27].
c-Met. The tyrosine kinase receptor c-Met (also known as hepatocyte growth factor receptor or HGFR) is overexpressed in a variety of human cancers [28]. Given that c-Met activates cellular growth, migration and invasion, its aberrant expression correlates with poor patient prognosis. In the case of lung cancer, CHIP overexpression destabilizes endogenous c-Met and inhibits cancer growth and invasion, whereas CHIP knockdown increases c-Met expression, suggesting a critical effect of CHIP in c-Met degradation [29].
Hormone receptors. Glucocorticoid receptor (GR), a member of a large superfamily of nuclear receptor, modulates genes controlling development, metabolism, and immune response. CHIP inhibits GR-mediated signal transduction via dissociation of GR from Hsp90. Besides, CHIP promotes ubiquitination of GR in collaboration with Ubc13 [30]. In the setting of androgen receptor (AR), CHIP interacts with the androgen receptor NH2-terminal conserved motif in a phosphorylated specific site [30], which triggers ubiquitination and subsequent degradation [31]. Since AR plays an essential role in the onset and progression of prostate cancer, AR degradation leads to mitotic arrest in prostate cancer cells treated with low dose 2-methoxyestradiol (2-ME, an endogenous estrogen metabolite) [32]. Interestingly, the degradation is considered to be ubiquitin-dependent, mediated by CHIP. Furthermore, CHIP attenuates the monomeric mutant AR in spinal and bulbar muscular atrophy (SBMA) more effectively than does wild type AR, indicating that mutant AR or other substrates are more sensitive to CHIP [33]. Estrogen receptor α (ERα) is another substrate of Hsp90. CHIP induces ERα degradation via direct ubiquitination [34] or the histone deacetylase inhibitor suberoylanilide hydroxamic acid (SAHA) [35], thereby blocking survival signaling in breast cancer cells.
Interaction between CHIP and signaling proteins
PI3K/AKT. The PI3K/AKT pathway is one of the most important signaling pathways in the regulation of cellular growth. Aberrant activation contributes to tumor growth and angiogenesis in diverse tumors [36]. CHIP forms a complex with Hsp90 and AKT. Once this triple complex is assembled, the ubiquitination and degradation of phosphorylated AKT occurs, thereby shutting down the over-active machinery and maintaining cellular pathway homeostasis [37]. Hsp70/CHIP complex is a novel E3 ligase for the p85 subunit of phosphatidylinositol 3-kinase (PI3K) which is responsible for p85 ubiquitination and degradation. Phosphatases, in addition to kinases, are another category of CHIP targets. Phosphatase and tensin homolog (PTEN), the first identified tumor suppressor possessing phosphatase activity, dephosphorylates the target molecule phosphatidylinositol 3,4,5-trisphosphate (PI(3,4,5)P3) downstream of (PI3K to generate PI (4, 5)P2. Thus, PTEN blocks the activation of downstream AKT. In prostatic carcinoma cells, transient binding to the TPR domain of CHIP leads to PTEN ubiquitination and subsequent degradation by the proteasome system [38]. Furthermore, CHIP promotes NF-κB ubiquitination and degradation in gastric cancer cells. NF-κB protein, via activation in the downstream cascade of the PI3K/AKT pathway, is involved in versatile expression of interleukins and chemokines. Additionally, CHIP allows NF-κB to enter the nucleus, where it functions as a transcription factor and suppresses the expression of its target genes (such as IL-8, MMP-2, VEGF). In this way, CHIP inhibits the invasive and metastatic capacities as well as the angiogenic ability of gastric cancer [39].
MAPK. Mitogen-activated protein kinases (MAPKs) comprise a family of cellular serine/threonine protein kinases that control embryogenesis, cell differentiation, proliferation and death [40]. Apoptosis signal-regulating kinase 1 (ASK1), a member of MAP3K family, plays an important role in the pathogenesis of neurodegeneration, cardiovascular diseases and tumorigenesis [41]. Under oxidative stress or inflammatory factor-mediated activation, phosphorylated ASK1 activates the downstream c-Jun N-terminal kinase (JNK) and p38 MAPK pathways, thereby inducing cellular apoptosis. The C-terminus of Hsp70 has affinity for the acceptor domain at the N-terminal TPR of ASK1. Through cooperation with CHIP, Hsp70 promotes the ubiquitin-dependent proteasomal degradation of ASK1 and inhibits TNF-alpha-induced cell apoptosis [42]. In Addition, CHIP triggers translocation of the ASK1 partner death domain-associated protein (Daxx) into the nucleus, further initiating an anti-apoptotic response [43].
p53. P53 is the first characterized and most popular tumor suppressor gene in almost all cancer types. DNA-bound p53 recruits DNA repair proteins and induces mitotic arrest from G1 to S phase. Hence, p53 acts as a guard of the genome against cancerogenesis. Cellular accumulation of mutant p53, on the other hand, induces tumor formation and transformation [44]. MDM2, the main ubiquitin ligase of p53, plays a pivotal role in stimulating p53 turnover via ubiquitin-dependent proteasomal degradation [45]. Interestingly, in normal conditions, CHIP degrades wild-type p53 protein alone, but only targets and ubiquitinates mutant p53 in the presence of Hsp70 or Hsp90 [46]. Upon heat stress, however, CHIP directly interacts with wild-type p53 as a chaperone and inhibits its denaturation. Together with Hsp90 inhibitors, CHIP preferentially binds to misfolded p53 and restores its DNA binding activity [47]. Collectively, CHIP might serve as a chaperone of wild-type p53. It maintains its conformation under physiological condition and rescues mutant p53 into its native, folded state under stress. Furthermore, CHIP assists in degradation of p53 isoform 133p53α degradation, which orchestrates p53-mediated cellular senescence [48]. As a consequence, CHIP functions as a critical modulator with multifaceted effects on the expression and function of p53.
TGF-β. The transforming growth factor-β (TGF-β) pathway regulates cell differentiation, apoptosis and migration by conducting signaling into the nucleus and inducing gene expression through the Smad [49] or Daxx signaling pathway [50]. CHIP with TPR domain binds to the C-terminus of Smad1/5, leading to Smad1/5 ubiquitination and degradation. The phosphorylated SXS motif at the C-terminus of Smad1 enhances its binding affinity for CHIP. Hsp70 and Hsp90 play opposing effects of regulating TGF-β signaling via CHIP-mediated Smad3 ubiquitination and degradation [51]. Additionally, Daxx, an important apoptosis-associated factor, synergizes with the Axin/HIPK2/p53 complex to induce programmed cell death [52]. The stress-induced association of CHIP with Daxx forms a special insoluble fragment and avoids degradation by the proteasome. At the meantime, it inhibits stress-induced p53-dependent apoptosis by interfering with interaction between Daxx and homeodomain-interacting protein kinase 2 (HIPK2). Once the stress is eliminated, Daxx transforms to the soluble functional compartment [53].
TLR. Toll-like receptors (TLRs), expressed mainly on antigen-presenting cells (APCs), is pivotal in bridging innate and adaptive immunity. By recognizing structurally conserved pathogen components termed pathogen-associated molecular patterns (PAMPs) and/or endogenous damage-associated molecular patterns (DAMPs), TLRs trigger cellular signaling pathways and immune responses. In the TLR-induced signaling cascade, CHIP interacts with and polyubiquitinates downstream Src (non-receptor tyrosine kinase) and PKCζ, thereby facilitating LPS/CpG-induced Src and PKC recruitment and activation [54]. The activated kinases further induce the nuclear translocation of NF-κB and IRF3/7, leading to release of cytokines such as IL-6 and IFN-β.
Role of CHIP in human diseases
Given its functions as a quality control E3 ligase, CHIP is pivotal in regulating myriad of proteins and pathologies with abnormal protein expression. Accumulating evidence demonstrates the importance of CHIP in cancer, neurological disorders, cardiac disease and so forth.
CHIP and cancer
Cancer remains the most frequently studied disease in CHIP biology due to aberrant expression of diverse protein in the progression of cancer [55]. Notably, since controversy regarding the oncogenic or tumor suppressive effect of CHIP emerged over the past several years, CHIP has been considered as a double-edged sword in modulation of tumorigenesis (Figure 2).
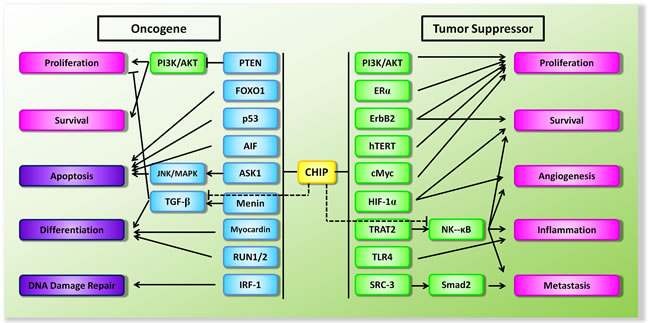
Figure 2: Schematic diagram of the dual functions of CHIP in the context of tumorigenesis. Proteins in square are either oncogenes in green or tumor suppressor genes in blue. Arrow (->) indicates promotion and the symbol (–I) indicates suppression.
CHIP as a tumor suppressor
Since Kajiro et al. [56] demonstrated the tumor suppressive function of CHIP as a hallmark, a series of subsequent studies have elucidated the critical roles of CHIP in proliferation, tumorigenesis and invasion in several malignancies. In the pioneering study, Kajiro et al. observed tumor growth, metastasis and malignancy were inversely correlated with CHIP levels in a nude mouse xenograft human breast tumor model [56]. Conversely, CHIP knockdown (shCHIP) in cancer cells readily resulted in tumor development and a metastatic phenotype in mice. The suppression of tumorigenesis might result from the inhibition of breast cancer stem cells [57]. Likewise, in human breast cancer specimens, CHIP mRNA expression was negatively associated with the malignant grade (TNM stage) and overall patient survival [58]. Collectively, CHIP might be a potentially favorable prognostic marker for breast cancer.
Recently, CHIP has been documented as a tumor suppressor in a variety of settings, with emerging evidence of myriad protein targeted by CHIP (Table 1). Our previous work demonstrated that CHIP ubiquitinated EGFR for proteasome-mediated degradation in pancreatic cancer cells and suppressed EGFR downstream signaling pathways [59]. Consistent with the findings in breast cancer, decreased CHIP expression in pancreatic cancer tissues correlated with poor prognosis and shortened overall survival of patients. In accordance with our observation, a negative correlation has been identified between CHIP expression and tumor malignancy (e.g. TNM stage and lymph node metastasis) in human gastric cancer [39, 60]. Unlike targeting EGFR in pancreatic carcinoma, CHIP bound to NF-κB/p65 and triggered its ubiquitination and proteasomal degradation, which further inhibits NF-κB signaling and IL-8-induced angiogenesis. Therefore, decreased CHIP expression was associated with tumor metastasis, and served as an independent prognostic factor for patients with gastric cancer [39]. The suppressive role is further confirmed in colorectal cancer, with evidence that CHIP overexpression led to decreased expression of NF-κB-targeted oncogenes [61]. Chronic myeloid leukemia (CML) is a non-solid neoplasm that is genetically characterized by the BCR-ABL fusion protein. Not surprisingly, CHIP interacts with the immature fused protein and induces its ubiquitin-dependent degradation, thereby hindering its proliferative ability [62]. In the setting of prostate cancer, AR is a target of CHIP-mediated degradation [32]. As discussed above, the destruction of AR blocks mitosis and further limits tumor proliferation. These findings shed light on the possibility of targeting E3 ligases such as CHIP as a novel strategy to attenuate AR expression and limit prostate cancer.
Thus far, a variety of oncogenic substrates have been identified, including ErbB2, eIF5A, SRC-3, pAkt, c-Myc and hypoxia-inducible factor 1α (HIF-1α), in different types of cancer [27, 56, 63-67]. Notably, K63 ubiquitination mediated by CHIP is necessary for HIF-1α degradation by chaperone-mediated autophagy [68]. These observations indicate a profound and versatile protein network regulated by CHIP in the control of tumorigenesis, and thus CHIP might be an upstream target for therapeutic intervention.
CHIP as an oncogene
In contrast to investigations mentioned above, a number of publications have reported CHIP to target tumor suppressor genes (TSGs), acting as an oncogene (Table 1). Li et al. [69] observed CHIP with the function of ubiquitination and degradation of Forkhead box (Fox) containing transcription factor O1 (FoxO1) in response to tumor necrosis factor-α (TNF-α) stimulation. Given that FoxO1 is responsible for apoptosis, CHIP assists in survival and proliferation of smooth muscle cells following TNF-α induction. Consistently, in prostate cancer cells [38], expression of PTEN correlated inversely with CHIP. Furthermore, CHIP has also been implicated in the modulation of other TSG proteins, including apoptosis-related p53, apoptosis-inducing factor (AIF), and interferon regulatory factor 1 (IRF-1) [46, 70, 71].
Interestingly, Liang et al. [72] reported higher CHIP expression assessed by immunohistochemistry among patients with shorter gallbladder cancer specific survival. This observation suggests a correlation between CHIP expression and poor prognosis in gallbladder carcinoma (GBC). CHIP levels, however, were not associated with other common clinicopathological elements, such as TNM stage. Likewise, significantly high level of CHIP was found in 163 metastatic lymph nodes (MLNs) of esophageal squamous cell carcinoma (ESCC), whereas the primary tumor of ESCC shared comparable level with normal epithelium [73]. Additionally, relatively lower expression of CHIP in MLNs correlated with better survival. Hence, CHIP possibly serves as an independent prognostic factor in ESCC.
The studies mentioned above were limited by their relatively small clinical sample size and retrospective design; these limitations could in part explain the varied results. Despite all the variations, CHIP obviously regulates various aspects of tumors in an intricate and exquisite manner (Figure 2). Apparently, further clarification is of great necessity to elucidate its pathogenic mechanisms in human malignancy.
Table 1: Clinial studies/Human cancer cell studies regarding CHIP and cancer
Cancer Type |
CHIP expression in human cancer |
Clinical Functions |
Cellular Functions |
Target/Substrate |
Reference |
Breast Ca. |
↓ |
(+)Malignancy |
Suppression of tumour growth and metastasis |
SRC-3 |
Kajiro et al(2009) [56] |
Breast Ca. |
↓ |
(-) TNM Stage, (+)OS |
- |
- |
Patani et al(2010) [58] |
Gastric Ca. |
↓ |
(-) TNM Stage, LN Metastasis |
- |
- |
Gan et al(2012) [60] |
Gastric Ca. |
↓ |
(-) Metastasis |
Inhibition of blood vessel formation and growth of xenografts in nude mice |
NF-kB/p65 |
Wang S et al(2013) [39] |
Pancreatic Ca. |
↓ |
(+)Prognosis, (+)OS |
Suppression of cell proliferation,anchor-independent growth, invasion and migration |
EGFR |
Wang T et al(2014) [59] |
Colorectal Ca. |
↓ |
- |
Inhibition of tumor growth, migration and invasion |
NF-kB |
Wang Y et al (2014) [61] |
Colorectal Ca. |
↓ |
- |
- |
eIF5A |
Shang et al (2014) [63] |
Prostate Ca. |
- |
- |
Mitotic arrest |
AR |
Sarkar(2014) [32] |
Gallbladder Ca. |
↑ |
(-)Prognosis, (-)OS |
- |
- |
Liang et al(2013) [72] |
Esophageal Ca. |
↑ |
(+)Metastatic LNs |
- |
- |
Wen et al(2013) [73] |
Prostate Ca. |
↑ |
- |
Regulation of PTEN-dependent transcription |
PTEN |
Ahmed et al.(2012) [38] |
(+): Positive Correlation, (-): Negative Correlation, Ca.: Cancer, OS: Overall Survival, LN:Lymph Node, ↑:up-regulation, ↓:down-regulation
CHIP and other diseases
CHIP and neurological disorders
CHIP has been extensively linked to diverse neurological diseases, largely due to its capacity to remove aberrant disease-causing proteins (Figure 3). In particular, CHIP plays a vital role in the pathogenesis of neurodegenerative diseases (such as Parkinson’s disease and Alzheimer’s disease) that involve accumulation of toxic protein aggregates with aging.
Parkinson’s disease. Parkinson’s disease (PD) is characterized by preferential loss of dopaminergic neurons in the substantia nigra and the accumulation of Lewy bodies (LB), containing α-Synuclein protein [74-75]. CHIP co-localizes with α-synuclein and inhibits its aggregation via proteasomal degradation and suppression of oligomerization [76, 77]. In addition, Parkin, a RING-finger type E3 ligase, is responsible for the proper disposal of multiple substrates involved in PD, including mutated a-synuclein, glycosylated synuclein-22, Parkin-associated endothelin-like receptor (Pael-R), synphilin, cell division control-related protein and a polyglutamine-expanded ataxin-3 mutant [19, 78, 79]. Interestingly, the bifunctional co-chaperone CHIP aids in the dissociation of Hsp70 from the Pael-R/Parkin complex, and this displacement is necessary for Parkin to resume E3 activity toward Pael-R [80].
Alzheimer’s disease. Alzheimer’s disease (AD) is characterized by accumulation of misfolded insoluble β-amyloid and hyperphosphorylated Tau protein [81]. As a chaeperone, CHIP prevents normal β-amyloid precursor protein (βAPP) from degradation. On the other hand, CHIP, as a ligase, cooperates with Hsp and promotes βAPP degradation in an ubiquitin-dependent manner [82]. In this way, the progression of AD is hindered due to less accumulation of β-amyloidin neurons. In the setting of Tau protein, it serves to stabilize microtubules in neurons, but the phosphorylated form is prone to form toxic neurofibrillary tangles [83]. With the functions of eliminating unwanted protein aggregation, CHIP together with Hsp70 pushes p-Tau towards proteasomal degradation by targeting the microtubule-binding repeat region of hyperphosphorylated tau [84-86].
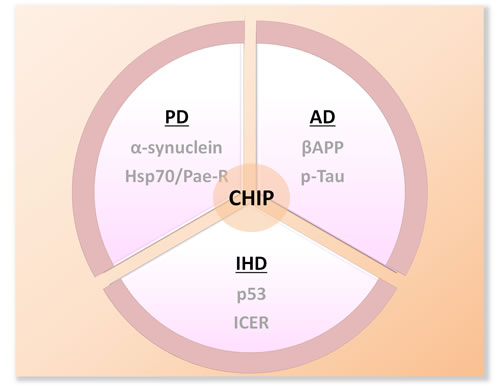
Figure 3: Schematic diagram of the substrates of CHIP in several well-studied neruodegenerative disorders. PD:Parkinson’s Disease; AD: Alzheimer’s Disease; IHD: Ischemic Heart Disease; Hsp70: heat shock protein 70; Pael-R:Parkin-associated endothelin-like receptor; βAPP:β-amyloid precursor protein; p-Tau: phosphorylated Tau; ICER: inducible cAMP early repressor.
CHIP and cardiac diseases
Apoptosis plays a critical role in cardiac remodeling in the setting of ischemic heart injury (Figure 3) [87]. Targeted ablation of p53 suppresses apoptosis and concomitant cardiac rupture after myocardial infarction (MI) in mice [89]. In the screening of heart cDNA pools, CHIP has been identified as an endogenous p53 antagonist in the heart [89]. CHIP decreased p53 level via ubiquitination and proteasomal degradation. Furthermore, Woo et al., [90] found that the cardioprotective function of CHIP-ERK5 (extracellular signal-regulated kinase 5) interaction through destabilization of inducible cAMP early repressor (ICER). In contrast, dissociation of the complex via phosphorylation at S496 of ERK5 results to reversal of cardioprotective phenotype [91].
CONCLUSION
The chaperone and ubiquitin-proteasome systems are the two most important mechanisms that work cooperatively to maintain protein quality control and cellular homeostasis. As a linker between these two systems, CHIP plays a pivotal role in a diverse number of cellular processes involving protein refolding, degradation, trafficking, apoptosis, inflammation and morphogenesis in both physiological and pathological settings. Increasing evidence suggests that CHIP is critical in cancers, neurological disorders, and cardiac diseases, among others [7,55]. Despite some discrepancies, CHIP generally exerts a cyto-protective effect by eliminating detrimental forms of several proteins.
However, many questions remain to be answered, including the underlying mechanism of target selection and the types of polyubiquitin chains as well as the manner by which signal recognition and the transfer of substrates occur in the proteasome system. Further explorations into the regulation and functional aspects of CHIP will provide deeper and more fundamental insights into the biological roles of CHIP and its therapeutic potential for various diseases.
CONFLICTS OF INTEREST
This study was supported by grants from the Research Special Fund for Public Welfare Industry of Health (No. 201202007), the National Science & Technology Pillar Program during the Twelfth Five-year Plan Period (No. 2014BAI09B11), the National Natural Science Foundation of China (No. 81472327), the Fundamental Research Funds for the Central Universities and the PUMC Youth Fund (No. 3332015004). The authors declare that they have no competing interests.
References
1. Schubert U, Antón LC, Gibbs J, Norbury CC, Yewdell JW, Bennink JR. Rapid degradation of a large fraction of newly synthesized proteins by proteasomes. Nature. 2000; 404: 770-774.
2. Wickner S, Maurizi MR, Gottesman S. Posttranslational quality control: folding, refolding, and degrading proteins. Science. 1999; 286: 1888-1893.
3. Broadley SA, Hartl FU. The role of molecular chaperones in human misfolding diseases. FEBS Lett. 2009; 583: 2647-2653.
4. Ballinger CA, Connell P, Wu Y, Hu Z, Thompson LJ, Yin LY, Patterson C. Identification of CHIP, a novel tetratricopeptide repeat-containing protein that interacts with heat shock proteins and negatively regulates chaperone functions. Mol Cell Biol. 1999; 19: 4535-4545.
5. Connell P, Ballinger CA, Jiang J, Wu Y, Thompson LJ, Höhfeld J, Patterson C. The co-chaperone CHIP regulates protein triage decisions mediated by heat-shock proteins. Nat Cell Biol. 2001; 3: 93-96.
6. Jiang J, Ballinger CA, Wu Y, Dai Q, Cyr DM, Höhfeld J, Patterson C. CHIP is a U-box-dependent E3 ubiquitin ligase: identification of Hsc70 as a target for ubiquitylation. J Biol Chem. 2001; 276: 42938-42944.
7. Paul I, Ghosh MK. A CHIPotle in physiology and disease. Int J Biochem Cell Biol. 2015; 58: 37-52.
8. Zhang M, Windheim M, Roe SM, Peggie M, Cohen P, Prodromou C, Pearl LH. Chaperoned ubiquitylation--crystal structures of the CHIP U box E3 ubiquitin ligase and a CHIP-Ubc13-Uev1a complex. Mol Cell. 2005; 20: 525-538.
9. Matsumura Y, Sakai J, Skach WR. Endoplasmic reticulum protein quality control is determined by cooperative interactions between Hsp/c70 protein and the CHIP E3 ligase. J Biol Chem. 2013; 288: 31069-31079.
10. Young JC, Moarefi I, Hartl FU. Hsp90: a specialized but essential protein-folding tool. J Cell Biol. 2001; 154: 267-273.
11. Minami Y, Hohfeld J, Ohtsuka K, Hartl FU. Regulation of the heat-shock protein 70 reaction cycle by the mammalian DnaJ homolog, Hsp40. J Biol Chem. 1996; 271: 19617-19624.
12. McDonough H, Patterson C. CHIP: a link between the chaperone and proteasome systems. Cell Stress Chaperones. 2003; 8: 303-308 (2003).
13. Cyr DM, Hohfeld J, Patterson C. Protein quality control: U-box-containing E3 ubiquitin ligases join the fold. Trends Biochem Sci. 2002; 27: 368-375 (2002).
14. Demand J, Alberti S, Patterson C, Hohfeld J. Cooperation of a ubiquitin domain protein and an E3 ubiquitin ligase during chaperone/proteasome coupling. Curr Biol. 2001; 11: 1569-1577.
15. Arndt V, Daniel C, Nastainczyk W, Alberti S, Hohfeld J. BAG-2 acts as an inhibitor of the chaperone-associated ubiquitin ligase CHIP. Mol Biol Cell. 2005; 16: 5891-5900.
16. Yenari MA, Liu J, Zheng Z, Vexler ZS, Lee JE, Giffard RG. Antiapoptotic and anti-inflammatory mechanisms of heat-shock protein protection. Ann N Y Acad Sci. 2005; 1053: 74-83.
17. Yang S, Qu S, Perez-Tores M, Sawai A, Rosen N, Solit DB, Arteaga CL. Association with Hsp90 inhibits Cbl-mediated down-regulation of mutant epidermal growth factor receptors. Cancer Res. 2006; 66: 6990-6997.
18. Wang X, DeFranco DB. Alternative effects of the ubiquitin-proteasome pathway on glucocorticoid receptor down-regulation and transactivation are mediated by CHIP, an E3 ligase. Mol Endocrinol. 2005; 19: 1474-1482.
19. Shin Y, Klucken J, Patterson C, Hyman BT, McLean PJ. The co-chaperone carboxyl terminus of Hsp70-interacting protein (CHIP) mediates alpha-synuclein degradation decisions between proteasomal and lysosomal pathways. J Biol Chem. 2005; 280: 23727-23734.
20. Rees I, Lee S, Kim H, Tsai FT. The E3 ubiquitin ligase CHIP binds the androgen receptor in a phosphorylation-dependent manner. Biochim Biophys Acta. 2006; 1764: 1073-1079.
21. Fan M, Park A, Nephew KP. CHIP (carboxyl terminus of Hsc70-interacting protein) promotes basal and geldanamycin-induced degradation of estrogen receptor-alpha. Mol Endocrinol. 2005; 19: 2901-2914 (2005).
22. Rosser MF, Washburn E, Muchowski PJ, Patterson C, Cyr DM. Chaperone functions of the E3 ubiquitin ligase CHIP. J Biol Chem. 2007; 282: 22267-22277.
23. Murata S, Minami Y, Minami M, Chiba T, Tanaka K. CHIP is a chaperone-dependent E3 ligase that ubiquitylates unfolded protein. EMBO Rep. 2001; 2: 1133-1138 (2001).
24. Dai Q, Zhang C, Wu Y, McDonough H, Whaley RA, Godfrey V, Li HH, Madamanchi N, Xu W, Neckers L, Cyr D, Patterson C. CHIP activates HSF1 and confers protection against apoptosis and cellular stress. EMBO J. 2003; 22: 5446-5458 (2003).
25. Qian SB, McDonough H, Boellmann F, Cyr DM, Patterson C. CHIP-mediated stress recovery by sequential ubiquitination of substrates and Hsp70. Nature. 2006; 440: 551-555.
26. Tan M, Yu D. Molecular mechanisms of erbB2-mediated breast cancer chemoresistance. Adv Exp Med Biol. 2007; 608: 119-129.
27. Xu W, Marcu M, Yuan X, Mimnaugh E, Patterson C, Neckers L. Chaperone-dependent E3 ubiquitin ligase CHIP mediates a degradative pathway for c-ErbB2/Neu. Proc Natl Acad Sci U S A. 2002; 99: 12847-12852.
28. Birchmeier C, Gherardi E. Developmental roles of HGF/SF and its receptor, the c-Met tyrosine kinase. Trends Cell Biol. 1998; 8: 404-410.
29. Jang KW, Lee JE, Kim SY, Kang MW, Na MH, Lee CS, Song KS, Lim SP. The C-terminus of Hsp70-interacting protein promotes Met receptor degradation. J Thorac Oncol. 2011; 6: 679-687.
30. Slotman JA, da Silva Almeida AC, Hassink GC, van de Ven RH, van Kerkhof P, Kuiken HJ, Strous GJ. Ubc13 and COOH terminus of Hsp70-interacting protein (CHIP) are required for growth hormone receptor endocytosis. J Biol Chem. 2012; 287: 15533-15543.
31. He B, Bai S, Hnat AT, Kalman RI, Minges JT, Patterson C, Wilson EM. An androgen receptor NH2-terminal conserved motif interacts with the COOH terminus of the Hsp70-interacting protein (CHIP). J Biol Chem. 2004; 279: 30643-30653.
32. Sarkar S, Brautigan DL, Parsons SJ, Larner JM. Androgen receptor degradation by the E3 ligase CHIP modulates mitotic arrest in prostate cancer cells. Oncogene. 2014; 33: 26-33.
33. Adachi H, Waza M, Tokui K, Katsuno M, Minamiyama M, Tanaka F, Doyu M, Sobue G. CHIP overexpression reduces mutant androgen receptor protein and ameliorates phenotypes of the spinal and bulbar muscular atrophy transgenic mouse model. J Neurosci. 2007; 27: 5115-5126.
34. Tateishi Y, Kawabe Y, Chiba T, Murata S, Ichikawa K, Murayama A, Tanaka K, Baba T, Kato S, Yanagisawa J. Ligand-dependent switching of ubiquitin-proteasome pathways for estrogen receptor. EMBO J. 2004; 23: 4813-4823.
35. Yi X, Wei W, Wang SY, Du ZY, Xu YJ, Yu XD. Histone deacetylase inhibitor SAHA induces ERalpha degradation in breast cancer MCF-7 cells by CHIP-mediated ubiquitin pathway and inhibits survival signaling. Biochem Pharmacol. 2008; 75: 1697-1705.
36. Jiang BH, Liu LZ. PI3K/PTEN signaling in tumorigenesis and angiogenesis. Biochim Biophys Acta. 2008; 1784: 150-158.
37. Su CH, Wang CY, Lan KH, Li CP, Chao Y, Lin HC, Lee SD, Lee WP. Akt phosphorylation at Thr308 and Ser473 is required for CHIP-mediated ubiquitination of the kinase. Cell Signal. 2011; 23: 1824-1830.
38. Ahmed SF, Deb S, Paul I, Chatterjee A, Mandal T, Chatterjee U, Ghosh MK. The chaperone-assisted E3 ligase C terminus of Hsc70-interacting protein (CHIP) targets PTEN for proteasomal degradation. J Biol Chem. 2012; 287: 15996-16006.
39. Wang S, Wu X, Zhang J, Chen Y, Xu J, Xia X, He S, Qiang F, Li A, Shu Y, Røe OD, Li G, Zhou JW. CHIP functions as a novel suppressor of tumour angiogenesis with prognostic significance in human gastric cancer. Gut. 2013; 62: 496-508.
40. Pearson G, Robinson F, Beers Gibson T, Xu BE, Karandikar M, Berman K, Cobb MH. Mitogen-activated protein (MAP) kinase pathways: regulation and physiological functions. Endocr Rev. 2001; 22: 153-183.
41. Assaife-Lopes N, Wengert M, de Sa Pinheiro AA, Leao-Ferreira LR, Caruso-Neves C. Inhibition of renal Na+-ATPase activity by inosine is mediated by A1 receptor-induced inhibition of the cAMP signaling pathway. Arch Biochem Biophys. 2009; 489: 76-81.
42. Gao Y, Han C, Huang H, Xin Y, Xu Y, Luo L, Yin Z. eHeat shock protein 70 together with its co-chaperone CHIP inhibits TNF-alpha induced apoptosis by promoting proteasomal degradation of apoptosis signal-regulating kinase1. Apoptosis. 2010; 15: 822-833.
43. Hwang JR, Zhang C, Patterson C. C-terminus of heat shock protein 70-interacting protein facilitates degradation of apoptosis signal-regulating kinase 1 and inhibits apoptosis signal-regulating kinase 1-dependent apoptosis. Cell Stress Chaperones. 2005; 10: 147-156.
44. Sonoyama T, Sakai A, Mita Y, Yasuda Y, Kawamoto H, Yagi T, Yoshioka M, Mimura T, Nakachi K, Ouchida M, Yamamoto K, Shimizu K. TP53 codon 72 polymorphism is associated with pancreatic cancer risk in males, smokers and drinkers. Mol Med Rep. 2011; 4: 489-495.
45. Haines DS, Landers JE, Engle LJ, George DL. Physical and functional interaction between wild-type p53 and mdm2 proteins. Mol Cell Biol. 1994; 14: 1171-1178.
46. Esser C, Scheffner M, Hohfeld J. The chaperone-associated ubiquitin ligase CHIP is able to target p53 for proteasomal degradation. J Biol Chem. 2005; 280: 27443-27448.
47. Tripathi V, Ali A, Bhat R, Pati U. CHIP chaperones wild type p53 tumor suppressor protein. J Biol Chem. 2007; 282: 28441-28454.
48. Horikawa I, Fujita K, Jenkins LM, Hiyoshi Y, Mondal AM, Vojtesek B, Lane DP, Appella E, Harris CC. Autophagic degradation of the inhibitory p53 isoform Delta133p53alpha as a regulatory mechanism for p53-mediated senescence. Nat Commun. 2014; 5: 4706.
49. Massaous J, Hata A. TGF-beta signalling through the Smad pathway. Trends Cell Biol. 1997; 7: 187-192.
50. Perlman R, Schiemann WP, Brooks MW, Lodish HF, Weinberg RA. TGF-beta-induced apoptosis is mediated by the adapter protein Daxx that facilitates JNK activation. Nat Cell Biol. 2001; 3: 708-714.
51. Shang Y, Xu X, Duan X, Guo J, Wang Y, Ren F, He D, Chang Z. Hsp70 and Hsp90 oppositely regulate TGF-beta signaling through CHIP/Stub1. Biochem Biophys Res Commun. 2014; 446: 387-392.
52. Li Q, Wang X, Wu X, Rui Y, Liu W, Wang J, Wang X, Liou YC, Ye Z, Lin SC. Daxx cooperates with the Axin/HIPK2/p53 complex to induce cell death. Cancer Res. 2007; 67: 66-74.
53. McDonough H, Charles PC, Hilliard EG, Qian SB, Min JN, Portbury A, Cyr DM, Patterson C. eStress-dependent Daxx-CHIP interaction suppresses the p53 apoptotic program. J Biol Chem. 2009; 284: 20649-20659.
54. Yang M, Wang C, Zhu X, Tang S, Shi L, Cao X, Chen T. eE3 ubiquitin ligase CHIP facilitates Toll-like receptor signaling by recruiting and polyubiquitinating Src and atypical PKC{zeta}. J Exp Med. 2011; 208: 2099-2112.
55. Sun C, Li HL, Shi ML, Liu QH, Bai J, Zheng JN. Diverse roles of C-terminal Hsp70-interacting protein (CHIP) in tumorigenesis. J Cancer Res Clin Oncol. 2014; 140: 189-197.
56. Kajiro M, Hirota R, Nakajima Y, Kawanowa K, So-ma K, Ito I, Yamaguchi Y, Ohie SH, Kobayashi Y, Seino Y, Kawano M, Kawabe Y, Takei H, et al. The ubiquitin ligase CHIP acts as an upstream regulator of oncogenic pathways. Nat Cell Biol. 2009; 11: 312-319.
57. Tsuchiya M, Nakajima Y, Hirata N, Morishita T, Kishimoto H, Kanda Y, Kimura K. Ubiquitin ligase CHIP suppresses cancer stem cell properties in a population of breast cancer cells. Biochem Biophys Res Commun. 2014; 452: 928-932.
58. Patani N, Jiang W, Newbold R, Mokbel K. Prognostic implications of carboxyl-terminus of Hsc70 interacting protein and lysyl-oxidase expression in human breast cancer. J Carcinog. 2010; 9: 9.
59. Wang T, Yang J, Xu J, Li J, Cao Z, Zhou L, You L, Shu H, Lu Z, Li H, Li M, Zhang T, Zhao Y. CHIP is a novel tumor suppressor in pancreatic cancer through targeting EGFR. Oncotarget. 2014; 5: 1969-1986. doi: 10.18632/oncotarget.1890.
60. Gan L, Liu DB, Lu HF, Long GX, Mei Q, Hu GY, Qiu H, Hu GQ. Decreased expression of the carboxyl terminus of heat shock cognate 70 interacting protein in human gastric cancer and its clinical significance. Oncol Rep. 2012; 28: 1392-1398.
61. Wang Y, Ren F, Wang Y, Feng Y, Wang D, Jia B, Qiu Y, Wang S, Yu J, Sung JJ, Xu J, Zeps N, Chang Z. CHIP/Stub1 functions as a tumor suppressor and represses NF-kappaB-mediated signaling in colorectal cancer. Carcinogenesis. 2014; 35: 983-991.
62. Tsukahara F, Maru Y. Bag1 directly routes immature BCR-ABL for proteasomal degradation. Blood. 2010; 116: 3582-3592.
63. Shang Y, Zhao X, Tian B, Wang Y, Ren F, Jia B, Zhai Y, Chen W, He D, Chang Z. CHIP/Stub1 interacts with eIF5A and mediates its degradation. Cell Signal. 2014; 26: 1098-1104.
64. Su CH, Lan KH, Li CP, Chao Y, Lin HC, Lee SD, Lee WP. ePhosphorylation accelerates geldanamycin-induced Akt degradation. Arch Biochem Biophys. 2013; 536: 6-11.
65. Paul I, Ahmed SF, Bhowmik A, Deb S, Ghosh MK. The ubiquitin ligase CHIP regulates c-Myc stability and transcriptional activity. Oncogene. 2013; 32: 1284-1295.
66. Bento CF, Fernandes R, Ramalho J, Marques C, Shang F, Taylor A, Pereira P. The chaperone-dependent ubiquitin ligase CHIP targets HIF-1alpha for degradation in the presence of methylglyoxal. PloS One. 2010; 5: e15062.
67. Ferreira JV, Fôfo H, Bejarano E, Bento CF, Ramalho JS, Girão H, Pereira P. STUB1/CHIP is required for HIF1A degradation by chaperone-mediated autophagy. Autophagy. 2013; 9: 1349-1366.
68. Ferreira JV, Soares AR, Ramalho JS, Pereira P, Girao H. K63 linked ubiquitin chain formation is a signal for HIF1A degradation by chaperone-mediated autophagy. Sci Rep. 2015; 5: 10210.
69. Li F, Xie P, Fan Y, Zhang H, Zheng L, Gu D, Patterson C, Li H. C terminus of Hsc70-interacting protein promotes smooth muscle cell proliferation and survival through ubiquitin-mediated degradation of FoxO1. J Biol Chem. 2009; 284: 20090-20098.
70. Oh KH, Yang SW, Park JM, Seol JH, Iemura S, Natsume T, Murata S, Tanaka K, Jeon YJ, Chung CH. Control of AIF-mediated cell death by antagonistic functions of CHIP ubiquitin E3 ligase and USP2 deubiquitinating enzyme. Cell Death Differ. 2011; 18: 1326-1336.
71. Narayan V, Pion E, Landre V, Muller P, Ball KL. Docking-dependent ubiquitination of the interferon regulatory factor-1 tumor suppressor protein by the ubiquitin ligase CHIP. J Biol Chem. 2011; 286: 607-619.
72. Liang ZL, Kim M, Huang SM, Lee HJ, Kim JM. Expression of carboxyl terminus of Hsp70-interacting protein (CHIP) indicates poor prognosis in human gallbladder carcinoma. Oncol Lett. 2013; 5: 813-818.
73. Wen J, Luo KJ, Hu Y, Yang H, Fu JH. Metastatic lymph node CHIP expression is a potential prognostic marker for resected esophageal squamous cell carcinoma patients. Ann Surg Oncol. 2013; 20: 1668-1675.
74. Moore DJ, West AB, Dawson VL, Dawson TM. Molecular pathophysiology of Parkinson’s disease. Annu Rev Neurosci. 2005; 28: 57-87.
75. Goedert M. Alpha-synuclein and neurodegenerative diseases. Nat Rev Neurosci. 2001; 2: 492-501.
76. Tetzlaff JE, Putcha P, Outeiro TF, Ivanov A, Berezovska O, Hyman BT, McLean PJ. CHIP targets toxic alpha-Synuclein oligomers for degradation. J Biol Chem. 2008; 283: 17962-17968.
77. Kalia LV, Kalia SK, Chau H, Lozano AM, Hyman BT, McLean PJ. Ubiquitinylation of alpha-synuclein by carboxyl terminus Hsp70-interacting protein (CHIP) is regulated by Bcl-2-associated athanogene 5 (BAG5). PloS One. 2011; 6: e14695.
78. Imai Y, Soda M, Takahashi, R. Parkin suppresses unfolded protein stress-induced cell death through its E3 ubiquitin-protein ligase activity. J Biol Chem. 2000; 275: 35661-35664.
79. Imai Y, Soda M, Inoue H, Hattori N, Mizuno Y, Takahashi R. An unfolded putative transmembrane polypeptide, which can lead to endoplasmic reticulum stress, is a substrate of Parkin. Cell. 2001; 105: 891-902.
80. Imai Y, Soda M, Hatakeyama S, Akagi T, Hashikawa T, Nakayama KI, Takahashi R. CHIP is associated with Parkin, a gene responsible for familial Parkinson’s disease, and enhances its ubiquitin ligase activity. Mol Cell. 2002; 10: 55-67.
81. Hashimoto M, Rockenstein E, Crews L, Masliah E. Role of protein aggregation in mitochondrial dysfunction and neurodegeneration in Alzheimer’s and Parkinson’s diseases. Neuromolecular Med. 2003; 4: 21-36.
82. Kumar P, Ambasta RK, Veereshwarayya V, Rosen KM, Kosik KS, Band H, Mestril R, Patterson C, Querfurth HW. CHIP and HSPs interact with beta-APP in a proteasome-dependent manner and influence Abeta metabolism. Hum Mol Genet. 2007; 16: 848-864.
83. Wang JZ, Xia YY, Grundke-Iqbal I, Iqbal K. Abnormal hyperphosphorylation of tau: sites, regulation, and molecular mechanism of neurofibrillary degeneration. J Alzheimers Dis. 2013; 33 Suppl 1: S123-139.
84. Shimura H, Schwartz D, Gygi SP, Kosik KS. CHIP-Hsc70 complex ubiquitinates phosphorylated tau and enhances cell survival. J Biol Chem. 2004; 279: 4869-4876.
85. Hatakeyama S, Matsumoto M, Kamura T, Murayama M, Chui DH, Planel E, Takahashi R, Nakayama KI, Takashima A. U-box protein carboxyl terminus of Hsc70-interacting protein (CHIP) mediates poly-ubiquitylation preferentially on four-repeat Tau and is involved in neurodegeneration of tauopathy. J Neurochem. 2004; 91: 299-307.
86. Kojima Y, Miyoshi H, Clevers HC, Oshima M, Aoki M, Taketo MM. Suppression of tubulin polymerization by the LKB1-microtubule-associated protein/microtubule affinity-regulating kinase signaling. J Biol Chem. 2007; 282: 23532-23540.
87. Crow MT. Revisiting p53 and its effectors in ischemic heart injury. Cardiovascular research. 2006; 70(3):401-403.
88. Matsusaka H, Ide T, Matsushima S, Ikeuchi M, Kubota T, Sunagawa K, Kinugawa S and Tsutsui H. Targeted deletion of p53 prevents cardiac rupture after myocardial infarction in mice. Cardiovascular research. 2006; 70(3):457-465.
89. Naito AT, Okada S, Minamino T, Iwanaga K, Liu ML, Sumida T, Nomura S, Sahara N, Mizoroki T, Takashima A, Akazawa H, Nagai T, Shiojima I and Komuro I. Promotion of CHIP-mediated p53 degradation protects the heart from ischemic injury. Circulation research. 2010; 106(11):1692-1702.
90. Woo CH, Le NT, Shishido T, Chang E, Lee H, Heo KS, Mickelsen DM, Lu Y, McClain C, Spangenberg T, Yan C, Molina CA, Yang J, Patterson C and Abe J. Novel role of C terminus of Hsc70-interacting protein (CHIP) ubiquitin ligase on inhibiting cardiac apoptosis and dysfunction via regulating ERK5-mediated degradation of inducible cAMP early repressor. FASEB journal. 2010; 24(12):4917-4928.
91. Yang K, Zhang TP, Tian C, Jia LX, Du J and Li HH. Carboxyl terminus of heat shock protein 70-interacting protein inhibits angiotensin II-induced cardiac remodeling. American journal of hypertension. 2012; 25(9):994-1001.

