INTRODUCTION
Stereotactic radiotherapy (SRT), including Cyberknife SRT, is used to treat patients with primary and metastatic brain tumors [1–3], whose most common late side effect is brain radiation necrosis[4–6]. Until now, research on tolerated dose of hypofractionated SRT for brain tumors is limited, especially those about the influencing and predicting factors of brain radiation necrosis post Cyberknife radiosurgery. Here we aimed to address this issue by a retrospective analysis and a reference dose for brain tumor patients undergoing Cyberknife therapy was established. Our results also provided advice on how to reduce the risk of brain radiation necrosis.
RESULTS
Occurrence of brain radiation necrosis
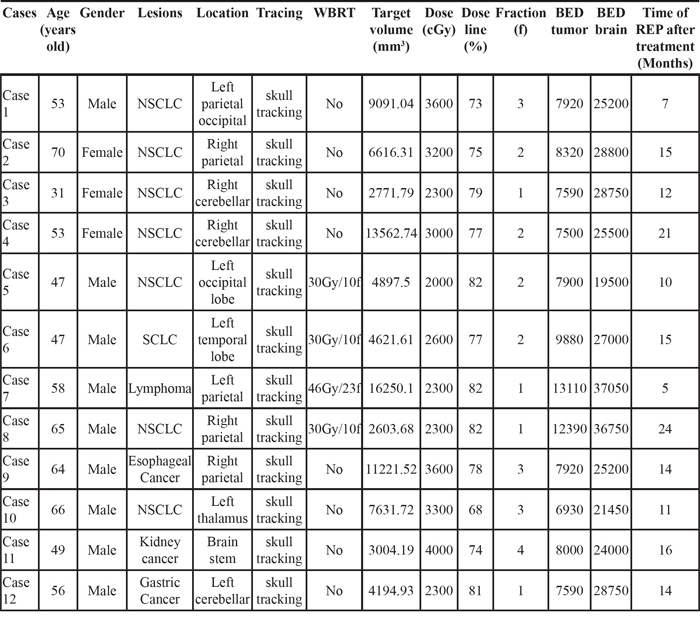
Brain necrosis occurred in 12 participants (9 males and 3 females) aged 31-70 (median: 54.5). A total of 12 targets were found representing with necrosis with a rate of 11.54%. Among these 12 cases, 1 had primary brain lymphoma, 1 brain metastases from stomach cancer, 1 brain metastases from kidney cancer, 1 brain metastases from small cell lung cancer, 1 brain metastases from esophagus cancer, and 7 brain metastases from non-small cell lung cancer. Four patients had received whole brain radiotherapy, including 3 were prescribed a dose of 30 Gy/10f, and 1 a dose of 46 Gy/23f. For all the targets treated with Cyberknife, the median treatment volume was 5756.91 mm3 (2603.68-16250.10 mm3), the median radiation dose 2800 cGy (2000-4000 cGy), the median isodose line 77.5% (68-82%), the median number of fractions 2 (1-4), and the median biologically equivalent prescription dose 7920 cGy (6930-13110 cGy; for those who had received whole brain radiotherapy, the value of BED was obtained by summarizing the BEDs in these two treatments). After Cyberknife therapy it took a median of 14 months (5-24 months) for brain radiation necrosis to develop. See Table 1 for details.
Table 1: Characteristics of the cases of radiation necrosis
Factors associating with brain radiation necrosis
The results of the logistic regression showed that combination with whole brain radiotherapy, fractions, and BED were significantly associated with development of brain radiation necrosis in the model, and thus able to contribute to the development. See Table 2 for details.
Table 2: Logistic regression analysis for radiation necrosis
Factors |
Regression coefficient |
Wald value |
RR value |
95%CI |
P value |
|---|---|---|---|---|---|
Female vs. male |
-0.127 |
0.01428 |
0.881 |
0.109-7.085 |
0.905 |
Age |
-0.0177 |
0.239 |
0.982 |
0.915-1.055 |
0.624 |
Diabetes |
0.792 |
0.327 |
2.207 |
0.146-33.282 |
0.567 |
Cardio-cerebrovascular disease |
-5.178 |
0.372 |
0.00564 |
0.000-94696.156 |
0.542 |
Without or with WBRT |
-15.764 |
4.025 |
0.0000000142 |
0.000-0.695 |
0.0448 |
Target volume |
0.00000118 |
0.030 |
1.000 |
0.999-1.001 |
0.862 |
Dose |
0.0169 |
0.000316 |
0.983 |
0.152-6.342 |
0.985 |
Dose line |
-0.0463 |
0.189 |
0.955 |
0.775-1.177 |
0.664 |
Fraction |
-4.075 |
4.521 |
0.0170 |
0.000397-0.727 |
0.00335 |
BED |
0.00881 |
5.311 |
1.009 |
1.001-1.016 |
0.0212 |
Predictor of brain radiation necrosis
MedCalc software was used to create ROC curves, and factors included BED, combination with whole brain radiation, and fractions (Figure 1). The areas under the ROC curves were 0.892±0.0335, 0.650±0.0717, and 0.712±0.0637 respectively, suggesting that BED had more predictive value for brain radiation necrosis than combination with whole brain radiation and fractions. Moreover, the ROC curve analysis showed the BED threshold was 7410 cGy for brain radiation necrosis, and the sensitivity and specificity were well balanced (Table 3). For the sake of clinical practicability, we further calculated the prescription dose in 1-5 fractions on the basis of the threshold values, and recommended dosages to avoid brain radiation necrosis for clinical reference was shown in Table 4.
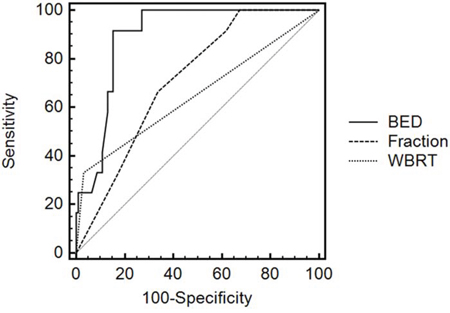
Figure 1: Roc curves of risk factors.
Table 3: Multiple factors in ROC curve analysis
AUC |
95%CI |
z statistic |
P value (Area=0.5) |
Associated criterion |
Sensitivity |
Specificity |
|
|---|---|---|---|---|---|---|---|
With or without WBRT(0:no/1:yes) |
0.650±0.0717 |
0.551 -0.741 |
2.098 |
0.0359 |
>0 |
33.33 |
96.74 |
Fraction(f) |
0.712±0.0637 |
0.615 -0.797 |
3.335 |
0.0009 |
<=2 |
66.67 |
66.30 |
BED(cGy) |
0.892±0.0335 |
0.816 -0.945 |
11.710 |
<0.0001 |
>7410 |
91.67 |
84.78 |
Table 4: Recommended dosages for avoiding brain radiation necrosis in clinical practice
Fractions |
Dose (Gy/F) |
|---|---|
1F |
22.68 |
2F |
14.89 |
3F |
11.49 |
4F |
9.44 |
5F |
8.16 |
DISCUSSION
Brain radiation necrosis is caused by white matter injury characterized by loss of oligodendrocytes, along with demyelination (Glial Injury Hypothesis) [14, 15] or vasogenic edema (Vascular Injury Hypothesis) [16–18]. Radiation damage to glial cells and vascular endothelial cells can lead to various late effects in the brain, and the extent of the damage depends on the biological dose. This study showed that BED, combination with whole brain radiotherapy, and fractions would contribute to brain radiation necrosis following Cyberknife therapy. BED was higher in patients who had received whole brain radiotherapy as a result of adding together the doses of two treatment regimens. Brain belongs to late-responding tissues, and are found to have low values of α/β ratio, and are more susceptible to a single, high dose of radiation. If the total dose keeps unchanged, fewer fractions will lead to a higher BED, thus increasing the risk of brain necrosis. The ROC curve analysis further confirmed the founding from the logistic regression analysis.
Varying doses are always adopted during Cyberknife therapy for various patients with different treatment plans and treatment volumes. For patients with systemic metastases, palliative care with low-dose radiation was usually given to obtain symptom relief; whereas for patients with a single brain metastasis, a higher dose might have been prescribed. Furthermore, tumours with larger volume usually result in palliative treatment. This study created a good chance for research on radiation tolerance of brain tissue with the various prescription doses, and results showed that the treatment volume did not appear to affect the development of brain radiation necrosis.
The rate of brain radiation necrosis was 11.54%, consistent with previous research [19, 20]. Furthermore, we analyzed the factors affecting and predicting brain radiation necrosis, established the threshold value for brain radiation necrosis and given a recommended prescription dose [21, 22]. More importantly, the study fills a gap in the research on the tolerated dose of hypofractionated SRT for brain tumours, and are also clinically important in terms of avoiding the occurrence of brain radiation necrosis following Cyberknife SRT.
In conclusion, by analyzing the brain necrosis cases, we explored various factors that may affect radiation necrosis in the brain, and the tolerated dose of hypofractionated radiotherapy for brain tumours. A reference dose level has been established to reduce the toxicity of Cyberknife therapy. We believe this study has paved the way for future research that will produce more substantial evidence to prevent brain radiation necrosis following Cyberknife SRT and improve treatment of brain cancer using Cyberknife.
MATERIALS AND METHODS
Patient information
The study was carried out in accordance with the institutional ethical guidelines and the use of patient information was approved by the Medical Ethics Committee of Tianjin Medical University Cancer institute and Hospital. Every patient involved in the study was asked to sign a piece of written informed consent which has been approved by the ethics committee of Tianjin Medical University Cancer institute and Hospital. The study was conducted according to the principles expressed in the Declaration of Helsinki. A total of 94 patients (mean age 51.5, range 6-85; 58 males and 36 females) with primary or metastatic brain tumours who had been treated with Cyberknife between September 2006 and October 2011 were collected and analyzed retrospectively. The inclusion criteria included primary or metastatic brain tumours, usage of Cyberknife SRT to treat brain tumours, follow-up of at least two-year, diagnosis of radiation necrosis confirmed by imaging or pathological examination. There were 104 targets, including 81 targets in 81 patients with brain metastases and 23 targets in 13 patients with primary brain tumours. Five patients (7 targets) received whole brain radiotherapy before Cyberknife SRT, among whom 4 (6 targets) were given a dose of 30 Gy/10f; and 1 (1target) 46 Gy/23f. Skull based tracking was used, together with a 60-87% isodose line (median: 79%), a dose of 1200-4500 cGy (median: 2550 cGy) and a BED of 2380-13110 cGy (median: 5130cGy) in 1-8 fractions (median: 3). BED=nd × (1+d/10); n: fraction, d: the dose of one fraction, α/β=10. See Table 5 for details.
Table 5: Clinical characteristics of patients
Characteristics |
Values |
|---|---|
Number of cases (n) |
94 |
Number of targets (n) |
104 |
Gender |
|
Male |
58 |
Female |
36 |
Mean age in years (range) |
51.5 (6-85) |
Targets |
|
Primary |
23 |
Metastatic |
81 |
Combined with WBRT(Targets) |
|
Yes |
7 |
No |
97 |
Mean treatment volume in mm3 (range) |
7805.78 (136.21-92760.70) |
Mean dose line in percentage (range) |
79 (60-87) |
Mean dose in cGy (range) |
2550.00 (1200-4500) |
Mean fraction in f (range) |
3 (1-8) |
Mean BED-tumor in cGy (range) |
5130 (2380-13110) |
Radiation encephalopathy (targets) |
|
Yes |
12 |
No |
92 |
Diagnosis of brain radiation necrosis
Comprehensive imaging is the most realistic and most frequently used method in the diagnosis of brain radiation necrosis [7–9]. While pathological examination, although known as the golden standard, was unachievable due to the following reasons. Firstly, many of the brain tumors treated with Cyberknife are located near the skull base or in the important functional areas, resulting in impossibility of surgical resection or stereotactic puncture. Secondly, patients with history of Cyberknife treatment, especially those with multiple lesions, usually rejected a puncture because it is the last choice in clinical practice to perform puncture biopsy of every single lesion. Furthermore, even a stereotactic puncture may not be able to completely display the pathological characteristics of the involved tissue.
Therefore comprehensive imaging is the most realistic and frequently used method in the diagnosis of brain radiation necrosis[7–9], and we chose to make the diagnosis upon patients’ medical history, signs and symptoms, along with results of various imaging approaches such as MRI, nuclear magnetic resonance spectroscopy, and PET-CT [10–13] in this study. MRI scan and resonance spectroscopy were conducted first, and PET-CT further introduced if the diagnosis cannot be confirmed. Briefly, most brain necrosis showed irregular shape in MRI with hypointense on T1WI and hyperintense on T2WI. Moreover, liquefaction necrosis often represented with lower signal intensity on T1WI and higher signal intensity on T2WI. After administration of Gd-DTPA, irregularly enhanced signal without enhanced nodular was obtained in the lesions center, while a large area of edema belt in T1 and T2 signal without enhancement around the lesions. In MRS, Cho, Cr and NAA levels were reduced, and NAA/Cho and NAA/Cr radio decreased. In PET, the metabolic rate of brain radiation necrosis was lower than that of normal brain tissue, resulting in decreased uptake of FDG and defected radioactive imaging in the corresponding region. The diagnosis was all ultimately determined by 3 independent investigators. If a patient had severe symptoms but also indications for surgery, the lesion could be resected and the diagnosis confirmed by histology. In this study, there were 12 patients with brain necrosis, with 1 confirmed by pathology and 11 by imaging.
Follow-up and statistical methods
MRI assessment of brain lesions should be conducted regularly 2 months after the Cyberknife radiosurgery; patients should be re-examined once every 3 months within the following one year, and then re-examined if necessary for a maximum of 6 months. If symptoms of intracranial lesions occurred, re-examination should be done immediately. All statistical analyses were executed using SPSS 17.0 software. The level of significance was defined as P < 0.05. Logistic regression was performed to explore associations between brain radiation necrosis and factors including diabetes, cardio-cerebrovascular disease, age, gender, combination with whole brain radiation, prescribed dosage, fractions, isodose line, and BED. MedCalc software was used in ROC curve analysis which included all the statistically significant factors in the logistic regression model, and the threshold values of the factors were estimated.
ACKNOWLEDGMENTS
This research was funded by National Natural Science Foundation of China (81301925).
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
REFERENCES
1. Jiang B, Veeravagu A, Lee M, Harsh GR, Lieberson RE, Bhatti I, Soltys SG, Gibbs IC, Adler JR, Chang SD. Management of intracranial and extracranial chordomas with CyberKnife stereotactic radiosurgery. J Clin Neurosci. 2012; 19:1101–6.
2. Olson AC, Wegner RE, Rwigema JC, Heron DE, Burton SA, Mintz AH. Clinical outcomes of reirradiation of brain metastases from small cell lung cancer with Cyberknife stereotactic radiosurgery. J Cancer Res Ther. 2012; 8:411–6.
3. Sio TT, Jang S, Lee SW, Curran B, Pyakuryal AP, Sternick ES. Comparing Gamma Knife and CyberKnife in patients with brain metastases. J Appl Clin Med Phys. 2014; 15:4095.
4. Conti A, Pontoriero A, Arpa D, Siragusa C, Tomasello C, Romanelli P, Cardali S, Granata F, De Renzis C, Tomasello F. Efficacy and toxicity of CyberKnife re-irradiation and ‟dose dense” temozolomide for recurrent gliomas. Acta Neurochir (Wien). 2012; 154:203–9.
5. Milker-Zabel S, Zabel A, Thilmann C, Zuna I, Hoess A, Wannenmacher M, Debus J. Results of three-dimensional stereotactically-guided radiotherapy in recurrent medulloblastoma. J Neurooncol. 2002; 60:227–33.
6. Kim YZ, Kim DY, Yoo H, Yang HS, Shin SH, Hong EK, Cho KH, Lee SH. Radiation-induced necrosis deteriorating neurological symptoms and mimicking progression of brain metastasis after stereotactic-guided radiotherapy. Cancer Res Treat. 2007; 39:16–21.
7. Na A, Haghigi N, Drummond KJ. Cerebral radiation necrosis. Asia Pac J Clin Oncol. 2014; 10:11–21.
8. Stockham AL, Ahluwalia M, Reddy CA, Suh JH, Kumar A, Vogelbaum MA, Barnett GH, Murphy ES, Chao ST. Results of a questionnaire regarding practice patterns for the diagnosis and treatment of intracranial radiation necrosis after SRS. J Neurooncol. 2013; 115:469–75.
9. Kickingereder P, Dorn F, Blau T, Schmidt M, Kocher M, Galldiks N, Ruge MI. Differentiation of local tumor recurrence from radiation-induced changes after stereotactic radiosurgery for treatment of brain metastasis: case report and review of the literature. Radiat Oncol. 2013; 8:52.
10. Reddy K, Westerly D, Chen C. MRI patterns of T1 enhancing radiation necrosis versus tumour recurrence in high-grade gliomas. J Med Imaging Radiat Oncol. 2013; 57:349–55.
11. Kang TW, Kim ST, Byun HS, Jeon P, Kim K, Kim H, Lee JI. Morphological and functional MRI, MRS, perfusion and diffusion changes after radiosurgery of brain metastasis. Eur J Radiol. 2009; 72:370–80.
12. Chernov MF, Hayashi M, Izawa M, Usukura M, Yoshida S, Ono Y, Muragaki Y, Kubo O, Hori T, Takakura K. Multivoxel proton MRS for differentiation of radiation-induced necrosis and tumor recurrence after gamma knife radiosurgery for brain metastases. Brain Tumor Pathol. 2006; 23:19–27.
13. Takenaka S, Asano Y, Shinoda J, Nomura Y, Yonezawa S, Miwa K, Yano H, Iwama T Comparison of 11C-Methionine, 11C-Choline, and 18F-Fluorodeoxyglucose-PET for Distinguishing Glioma Recurrence from Radiation Necrosis. Neurol Med Chir (Tokyo). 2014; 54:280–9.
14. Siu A, Wind JJ, Iorgulescu JB, Chan TA, Yamada Y, Sherman JH. Radiation necrosis following treatment of high grade glioma—a review of the literature and current understanding. Acta Neurochir (Wien). 2012; 154:191–201.
15. Pearlstein RD, Higuchi Y, Moldovan M, Johnson K, Fukuda S, Gridley DS, Crapo JD, Warner DS, Slater JM. Metalloporphyrin antioxidants ameliorate normal tissue radiation damage in rat brain. Int J Radiat Biol. 2010; 86:145–63.
16. Coderre JA, Morris GM, Micca PL, Hopewell JW, Verhagen I, Kleiboer BJ, van der Kogel AJ. Late effects of radiation on the central nervous system: role of vascular endothelial damage and glial stem cell survival. Radiat Res. 2006; 166:495–503.
17. Nordal RA, Nagy A, Pintilie M, Wong CS. Hypoxia and hypoxia-inducible factor-1 target genes in central nervous system radiation injury: a role for vascular endothelial growth factor. Clin Cancer Res. 2004; 10:3342–53.
18. Ye J, Rong X, Xiang Y, Xing Y, Tang Y. A study of radiation-induced cerebral vascular injury in nasopharyngeal carcinoma patients withradiation-induced temporal lobe necrosis. PLoS One. 2012; 7:e42890.
19. Lee AW, Ng WT, Hung WM, Choi CW, Tung R, Ling YH, Cheng PT, Yau TK, Chang AT, Leung SK, Lee MC, Bentzen SM. Major late toxicities after conformal radiotherapy for nasopharyngeal carcinoma-patient- and treatment-related risk factors. Int J Radiat Oncol Biol Phys. 2009; 73:1121–8.
20. Soussain C, Ricard D, Fike JR, Mazeron JJ, Psimaras D, Delattre JY. CNS complications of radiotherapy and chemotherapy. Lancet. 2009; 374:1639–51.
21. Bhansali A, Banerjee AK, Chanda A, Singh P, Sharma SC, Mathuriya SN, Dash RJ. Radiation-induced brain disorders in patients with pituitary tumours. Australas Radiol. 2004; 48:339–46.
22. Miyata T, Toho T, Nonoguchi N, Furuse M, Kuwabara H, Yoritsune E, Kawabata S, Kuroiwa T, Miyatake SI. The roles of platelet-derived growth factors and their receptors in brain radiation necrosis. Radiat Oncol. 2014; 9:51.

