Introduction
Cardiovascular disease (CVD) represents a major cause of morbidity and mortality worldwide [1]. Despite continuous advances in current clinical therapies, CVD is still considered to be a major global epidemic in need of innovative therapeutics [2]. In light of this, extraordinary efforts have been devoted to the study of novel cellular mechanisms characterizing cardiac physiology and pathology. Recently, cardiac repair has attracted significant interest as understanding its underlying mechanism may provide new avenues for fatal CVD.
Unlike amphibians and fish, the majority of cardiomyocytes in the mammalian heart lose the capacity to proliferate after birth [3, 4]. Recently, mounting evidence suggests that developing hearts have a strong growth and regenerative capacity arising from either differentiation of cardiac progenitor cells (CPCs) or proliferation of cardiomyocytes [5-7]. Of note, nearly half of cardiomyocytes are replaced during a whole human lifespan [3]. In the normal mouse heart, cardiomyocyte turnover occurs mainly through activation of resident cardiomyocytes at a rate of ~1.3-4%/year [8]. After myocardial injury, the rate of cardiomyocyte renewal may increase significantly, especially in the infarct border zone [9]. However, during a significant myocardial injury, a large number of cardiomyocytes are lost and may lead to heart failure, as the cardiac regenerative response is often insufficient to replace the lost cardiomyocytes. This ultimately leads to cardiac dysfunction, poor contractile function and massive fibrosis [10]. Cardiac repair is extremely limited due to the weak capacity of the adult mammalian heart to regenerate. Therefore, developing new therapeutic strategies which could enhance cardiomyocyte regenerative capacity is of significant clinical importance.
Non-coding RNAs (ncRNAs) are functional RNA molecules without protein coding functions [11]. ncRNAs can be divided into small (<200 nt) non-coding RNAs and longer RNAs (>200 nt). Small ncRNAs include microRNAs (miRs), transfer RNAs, and small nucleolar RNAs. Longer ncRNAs include ribosomal RNAs, natural antisense transcripts and other long non-coding RNAs (lncRNAs) [12]. Recently, the function of many of the newly identified ncRNAs has been validated, including the identification of miRs and lncRNAs involved in cardiac regeneration and repair [13].
Cardiac regeneration
Certain fish and amphibians retain a strong capacity for cardiac regeneration throughout their whole life [14-16]. For example, the zebrafish can fully regenerate its heart after surgical amputation of the cardiac apex and can tolerate injury of up to 20% of the ventricular mass [17, 18]. Using a Cre/loxp system to trace the lineage of cardiomyocytes in the adult fish, studies demonstrate that newly-formed cardiomyocytes are derived from the division of differentiated cardiomyocytes through increased expression of polo-like kinase 1 (plk1) [19]. Although mammalian hearts lack the robust regenerative capacity as observed in the zebrafish, postnatal mammalian hearts also experience a degree of cardiomyocyte renewal in physiological or pathological conditions [20,21]. To detect the origin of mammalian cardiomyocyte renewal, a study combining two lineage tracing approaches, genetic fate-mapping with isotope labeling and multi-isotope imaging mass spectrometry, reported murine cardiomyocyte genesis occurs at a very low rate and mainly derives from the differentiation of pre-existing cardiomyocytes in both the normal ageing process and in myocardial injury. Interestingly, the rate of cardiomyocyte renewal is significantly increased adjacent to areas of myocardial injury [22].
In addition to division of pre-existing cardiomyocytes, progenitor/stem cells also contribute to cardiomyocyte renewal [8, 23-25]. A study using genetic fate mapping in conditional green fluorescent protein (GFP)-labeled transgenic mice (cardiomyocytes are GFP+ and stem or precursor cells are GFP-) revealed that during normal ageing, the percentage of GFP+ cardiomyocytes remained unchanged. This finding indicates cardiomyocyte turnover occurs mainly through differentiation of resident cardiomyocytes, found to be at a rate of ~1.3-4%/year [8]. However, in injured hearts, especially myocardial infarction, the number of GFP- cardiomyocytes increased and the percentage of GFP+ cardiomyocytes decreased. This suggests that stem or precursor cells replace injured cardiomyocytes at a significant rate [26].
Despite these observations, cardiac regeneration capacity is still limited due to the extremely low rate of cardiomyocyte production in the adult heart. Thus, it is of great clinical importance to understand the cellular and molecular mechanisms underlying cardiac regeneration. Overall, there are three strategies to induce cardiac regeneration in the adult heart: (1) transplant exogenous progenitor/stem cells to damaged myocardium, (2) promote resident progenitor/stem cells to differentiate into mature cardiomyocytes, and (3) enhance the proliferation of pre-existing cardiomyocytes. For strategies 1 and 2, multiple studies have used adult stem cells, pluripotent stem cells (iPSCs), or cellular reprogramming to protect the injured heart [7, 20, 27, 28]. For example, in a GFP transgenic mouse model of myocardial injury, cell therapy with bone marrow-derived c-kit+ cells diluted the GFP+ cardiomyocyte pool and ultimately improved cardiac function, suggesting that there is transdifferentiation or cell fusion of exogenous c-kit+ cells to cardiomyocytes with resulting improved functionality [29]. Other studies indicate that heart failure (HF)-derived bone marrow multipotent mesenchymal stromal cells (BM-MMSCs) demonstrate an early decrease of proliferative capacity, they also upregulate genes that control regeneration in addition to fibrosis. However, low density seeding in combination with moderate hypoxia results in improved regeneration and expansion of BM-MMSCs as well as prevention of lost replication potential, thus (HF)-derived BM-MMSCs can also be applied to cell therapy by changing culture condition [30]. For strategy 3, enhancing the endogenous signaling pathway of cardiomyocyte regeneration is also of significant importance. For example, growth factor neuregulin1 (NRG1) and its tyrosine kinase receptor (ErbB4) are reported to regulate cardiac regeneration by inducing cardiomyocyte proliferation [31]. Genetically activating ErbB4 and pharmacologically injecting NRG1 in adult mice induces cardiomyocyte cell-cycle activity to promotes cardiac regeneration and protects against myocardial injury [32]. Physiological exercise is another way to control cardiac regeneration, cardiomyocyte size, and apoptosis by regulating IGF-1-PI3k-Akt or other signaling pathways [33, 34]. Despite extensive studies, the progress in cardiac regeneration has been slow.
The regenerative rate in the human heart is significantly lower than in mice or rats. Recently, a study reports that the number of cardiomyocytes remains constant during the whole human life span. Cardiomyocyte turnover decreases with age and is <1% in the human adult heart [35]. Thus, there is an urgent need for new therapeutic strategies to enhance cardiomyocyte regenerative capacity.
Non-coding RNA in cardiac regeneration (Figure 1)
Currently, emerging evidence indicates that non-coding RNAs (ncRNAs) are responsible for specialized biological processes during cardiac development, disease and ultimately repair, such as transcriptional regulation, post-transcriptional gene control, epigenetic control, and nuclear genome organization [35-37]. In this context, incorporating ncRNAs within cardiac gene regulatory networks may provide a new opportunity for therapeutic intervention via regenerative medicine in the heart, especially miRs or lncRNAs. In light of current regenerative strategies, the regulatory effects of ncRNAs can be categorized as follows: cardiac proliferation, cardiac differentiation, cardiac survival and cardiac reprogramming.
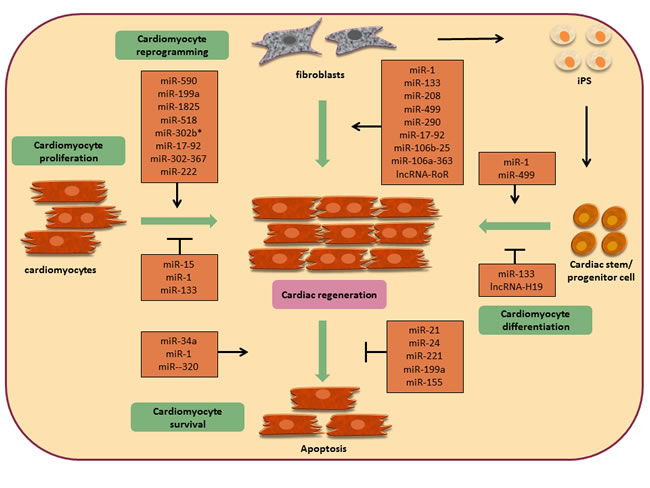
Figure 1: Non-coding RNAs in cardiac regeneration.
Cardiomyocyte proliferation
In a high-throughput functional screen for human miRNAs that enhance neonatal cardiomyocyte proliferation, 204 miRNAs were identified that increased proliferation over 2-fold compared to control [38]. Among these, miR-590 and miR-199a were studied in further detail and shown to activate the cell cycle and promote cardiomyocyte proliferation both in vitro and in vivo. Interestingly, during myocardial infarction, these two miRNAs not only stimulate cardiac regeneration, but also preserve cardiac function and decrease cardiac fibrosis [38]. Other miRNAs, such as miR-1825, miR-518, miR-302b*, have all been screened and validated by EdU, ki67, and phospho-histone H3 staining in rat or mouse cardiomyocytes, but the specific effects of these miRNAs on the heart require further study.
The miR-17-92 cluster has also been identified as a critical regulator of cardiac proliferation [39]. Transgenic overexpression of the miR-17-92 cluster in the heart induces cardiomyocyte proliferation in embryonic, postnatal, and even adult hearts by inhibiting phosphatase and tensin homolog (PTEN). Importantly, overexpression of the miR-17-92 cluster reverses cardiac dysfunction and cardiac fibrosis after myocardial infarction [39]. Another microRNA cluster, miR302-367, also promotes cardiomyocyte proliferation during developmental and adult stages [40]. Transient overexpression of miR302-367 leads to increased cardiomyocyte proliferation, decreased cardiac fibrosis and improved cardiac function after injury by targeting the Hippo signaling pathway.
As demonstrated previously, exercise induces physiological cardiac growth and protects against myocardial injury [41-43]. miR-222, upregulated in two distinct models of exercise (running and swimming), is necessary for exercise-induced cardiac proliferation due to its role in inhibiting cyclin-dependent kinase inhibitor 1B (p27), homeodomain-interacting protein kinase 1 (HIPK1) and homeobox containing 1 (HMBOX1) [44]. Overexpression of miR-222 in the heart confers resistance to cardiac remodeling and dysfunction after ischemia/reperfusion injury, while inhibition of miR-222 blocks cardiac growth in response to exercise [44]. This study links a specific miRNA with exercise as well as ischemia and holds great promise for targeting cardiomyocyte proliferation.
In addition to miRNAs that enhance cardiomyocyte proliferation, there are also miRNAs that suppress cardiac regeneration. The miR-15 family, regulated in the mouse heart at 1 and 10 days of age, suppresses numerous cell cycle genes and mediates postnatal cell cycle arrest [45]. Importantly, the miR-15 family modulates neonatal heart regeneration by repressing postnatal cardiomyocyte proliferation, thus leading to postnatal loss of cardiac regenerative capacity [46]. Inhibition of the miR-15 family from the early postnatal to adult stages increases cardiomyocyte proliferation and improves left ventricular function after myocardial infarction [46, 47].
miR-1 and miR-133 are both expressed in cardiac and skeletal muscle and regulated by the transcription factors MyoD, myocyte enhancer factor-2 (Mef2) and serum response factor (SRF) [48]. In vivo overexpression of miR-1 in the mouse during development inhibits cardiomyocyte proliferation by suppressing the transcription factor heart and neural crest derivatives-expressed protein 2 (Hand2) [48-50]. Deletion of miR-133a (involving both miR-133a-1 and miR-133a-2) results in up-regulation of smooth muscle genes in the heart and aberrant cardiomyocyte proliferation as detected by enhanced phospho-histone H3 staining [51, 52]. Overexpression of miR-133a results in diminished cardiomyocyte proliferation, which is consistent with the conclusion that miR-133a represses cardiac proliferation [53].
It is well known that lncRNA can control proliferation and apoptosis in various cell types; however, knowledge regarding its role in controlling cardiomyocyte proliferation remains limited. Therefore, researching the effects of lncRNA on cardiomyocyte proliferation may attract significant interest in the future. Importantly, studies show that enhanced cardiomyocyte proliferation is associated with protection against myocardial injury, suggesting that non-coding RNAs may be therapeutic targets for heart regeneration and cardiac repair.
Cardiomyocyte differentiation
miRNAs are reported to play important roles in the emergence of cardiac progenitors that trans-differentiate to cardiomyocytes as well as maintenance of the differentiated state in cardiomyocytes [54, 55]. miR-1 is a critical regulator of muscle cell proliferation and differentiation [56]. Overexpression of miR-1 in the developing heart can lead to growth retardation at embryonic day 13.5, thinner ventricular walls and ultimately heart failure results due to decreased cardiomyocyte proliferation by suppressing Hand2 [49]. Deletion of miR-1 (miR-1-2) leads to numerous defects in cardiogenesis, cardiac conduction and the cell cycle [48]. Thus, gain-of-function and loss-of-function studies demonstrate a key role of miR-1 in inhibiting cell proliferation and promoting differentiation. In vitro overexpression of miR-1 upregulates the cardiac differentiation genes nk2 homeobox 5 (NKX2.5) and β-MHC during mouse and human embryonic stem cell (ESC) differentiation [57, 58]. Importantly, transplantation of mouse ESCs overexpressing miR-1 to the border zone of myocardial infarction protects against ischemia-induced apoptosis by activating the PTEN-AKT signaling pathway [59]. These studies confirm miR-1 promotes cardiomyocyte differentiation and reprogramming. miR-1 and miR-133 are transcribed together as they are polycistronically clustered on the same chromosome. However, unlike miR-1 [51], miR-133 appears to inhibit cardiac differentiation. Overexpression of miR-133 suppresses cardiac markers in mouse and human ESCs, and in the absence of miR-133, myocytes proliferate partly through activation of SRF and G1/S-specific cyclin-D2 (CyclinD2) [51, 56], highlighting the inhibitory role of miR-133 in both cardiac proliferation and differentiation.
miR-499, enriched in adult progenitor cells in the heart, is also strongly associated with cardiac differentiation. Similar to miR-1, overexpression of miR-499 reduces cardiac progenitor cell proliferation and promotes the formation of beating embryoid bodies of mouse ESCs by up-regulating GATA4 and Mlc-2v [58]. In human cardiomyocyte progenitor cells (hCPCs), cardiac differentiation is greatly enhanced by increasing spontaneous beating areas of differentiated cells after transfecting with miR-499. In addition, cardiomyocyte-specific genes such as cardiac troponin T, and Mef2 are also up-regulated [50]. In vivo transplantation of human cardiac stem cells (hCSCs) transfected with miR-499 to the border zone of ischemic hearts promotes transformation of hCSCs to mature cardiomyocytes [60]. Overall, these in vitro and in vivo studies demonstrate the role of miR-499 in promoting cardiac differentiation.
A long non-coding RNA, H19, has been reported to negatively regulate body weight and cell proliferation [61]. Recently, down-regulation of H19 has been shown to promote differentiation of parthenogenetic embryonic stem cells (p-ESCs) to cardiomyocytes with strong heart-like beating, supporting the notion that reduction of H19 improves p-ESCs lineage determination to mesoderm cells such as cardiomyocytes [62, 63].
Together, these studies show that ncRNAs help balance cardiomyocyte differentiation with proliferation. These findings may be translated clinically by using ncRNAs to induce transferred stem cells or resident cardiac progenitor cells to differentiate into mature cardiomyocytes and replace damaged myocardium [16].
Cardiomyocyte survival
Although there have been great advances in stem cell therapy for cardiac injury, the low survival rate of transplanted cells in injured myocardium remains a major problem [64]. Therefore, the development of novel treatment strategies to boost cell survival is of significant importance.
miR-21 is decreased within the infarcted area in myocardial infarction (MI), indicating a protective effect of miR-21 on cardiac survival [65]. In vitro gain-of-function and loss-of-function studies of miR-21 confirm that it protects against ischemia-induced cardiomyocytes apoptosis by inhibiting programmed cell death protein 4 (PDCD4) and activator protein 1 (AP-1). Overexpression of miR-21 by adenovirus (Ad-miR-21) in rats decreases cardiac fibrosis by 29% at 24 hours, reduces ventricular dimension at 2 weeks and decreases cell apoptosis in the infarct or border area after acute myocardial infarction (MI) [65, 66]. These studies indicate that miR-21 promotes cardiac survival and apoptosis. miR-24 shares similar functions with miR-21, reducing cardiac apoptosis and increasing cell number by 53% through the repression of BH3-domain-containing protein (Bim). In a mouse MI model, overexpression of miR-24 is protective against cardiac apoptosis as partially mediated by Bim [67]. Interestingly, a study demonstrated that CPCs transfected with a cocktail of 3 microRNAs (miR-21, miR-24 and miR-221) exhibit higher viability compared to untreated CPCs when challenged with serum free medium. In a MI model, intramyocardial injection of CPCs transfected with miRNAs decreases cardiac dysfunction by regulating Bim [68]. In summary, the cocktail of microRNAs (miR-21, miR-24, and miR-221) can greatly boost the survival of transplanted cardiac progenitor cells, thus improving cardiac function.
In a hypoxia preconditioning environment, miR-199a is acutely downregulated in cardiomyocytes through up-regulation of hypoxia-inducible factor (Hif)-1 [69, 70]. Overexpression of miR-199a during hypoxia reduces apoptosis mediated by Hif-1 inhibition and its stabilization of p53, whereas inhibition of miR-199a recapitulates the hypoxic environment by up-regulating Hif-1 and Sirtuin 1(Sirt1). These data indicate that miR-199a plays a critical role in regulating the hypoxia-related pathway and decreases apoptosis in the setting of hypoxic damage. Another miRNA, miR-155, also prevents necrotic cell death in human cardiomyocyte progenitors by 40±2.3% by repressing receptor interacting protein 1 (RIP1) [71].
In addition to miRNAs that promote cardiomyocyte/ progenitor cell survival, there are also several miRNAs that exert the opposite effect on cardiomyocytes. miR-34a is induced in the ageing heart, a state that is a major risk factor for cardiovascular disease and may contribute to worse outcomes in patients with MI [72]. In vivo overexpression of miR-34a contributes to cardiac dysfunction in an age-dependent manner. However, silencing or genetic deletion of miR-34a decreases telomere shortening, DNA damage response and cardiomyocyte death induced by ageing. Inhibition of miR-34a can also reduce cardiac fibrosis and improve cardiac function in mice with MI by activating serine/threonine-protein phosphatase 1 regulatory subunit 10 (PNUTS). miR-1, which plays an important role in cardiomyocyte proliferation and differentiation, also plays a key role in regulating cardiac survival [73]. miR-1 transgenic mice have more severe cardiac ischemia/reperfusion (IR) injury as measured by increased LDH, CK levels, cleaved-caspase3 expression and cardiac infarct areas. In contrast, downregulation of miR-1 has an opposite effect. Studies reveal that miR-1 inhibits protein kinase C epsilon (PKC) and heat shock protein 60 (HSP60) in cardiac injury [73]. miR-320 is significantly increased in hearts with I/R injury both in vivo and in vitro [74]. In turn, gain-of-function of miR-320 results in increased infarction size and apoptosis in hearts with I/R injury both in vivo and ex vivo. Conversely, administration of antagomir-320 reduces cell apoptosis and infarction size relative to controls. Heat-shock protein 20 (Hsp20), a well-known cardioprotective protein, is a candidate target for miR-320. Downregulation of miR-29 protects the heart against IR injury by decreasing the expression of pro-apoptotic molecular Bax and increasing anti-apoptotic molecular Bcl2 [75].
In summary, these studies indicate that manipulating miRNA levels may be a novel therapeutic strategy for cardiovascular disease mediated by injury-induced apoptosis.
Cardiomyocyte reprogramming
Although there have been great advance in cardiac regenerative treatment, repopulation of the injured myocardium with new, functional cardiomyocytes remains a daunting challenge. Recently, landmark reports have raised the possibility of utilizing cellular reprogramming for cardiac regeneration [76-80].
Recently, a study identified a combination of miRNAs (miR-1, miR-133, miR-208 and miR-499) capable of reprogramming fibroblasts to cardiomyocyte-like cells in vitro, an effect which is enhanced 10-fold after the administration of janus kinase (JAK) inhibitor [79]. The myocyte-like cells possess morphological and functional properties of mature cardiomyocytes. Importantly, administration of miRNAs to injured myocardium in vivo results in the direct reprogramming of cardiac fibroblasts to cardiomyocytes, further confirmed by genetic tracing analysis using Fsp1Cre-mice. Enhanced cardiac reprogramming also decreased cardiac fibrosis and improved cardiac function, as indicated by fractional shortening, ejection fraction and other measures of ventricular function [81].
iPS cells were firstly established by overexpressing the four reprogramming factors Oct3/4, Sox2, Klf4, and c-Myc in fibroblasts [82, 83]. The miR-290 family is highly expressed in ESCs and significantly increases the efficiency of reprogramming in mouse embryonic fibroblasts (MEFs). Conversely, antagonizing miR-290 inhibits reprogramming. Within the miR-290 family, miR-284 enhances the efficiency to 75% of that achieved by the three reprogramming factors (Oct3/4, Sox2, and Klf4) alone [84]. Members of the miR-302 family share a similar sequence with miR-290 and also increase the programming efficiency of fibroblasts to mature cardiomyocytes [85-87]. Reportedly, three miR clusters, miR-17-92, miR-106b-25, and the miR-106a-363, are highly expressed when reprogramming fibroblasts to cardiomyocytes. Upregulation of the miR-106b-25 cluster members miR-93 and miR-106b increase cell reprogramming of iPS in combination with three (Oct3/4, Sox2, and Klf4) or four (Oct3/4, Sox2, Klf4, and c-Myc) reprogramming factors [88].
Recently, lncRNA-ST8SIA3 (also named lncRNA-ROR, regulator of reprogramming), is upregulated during iPS generation and enhances the reprogramming of fibroblasts to iPSs or cardiomyocytes [89]. LncRNAs also regulate the pluripotent state in mouse ESCs as evidenced by genome-wide mapping studies [90]. Transcription factors such as Oct4 and Nanog bind promoters of lncRNA. To further elucidate the biological functions of lncRNA expressed in ESCs, loss-of-function studies have been performed. By using a shRNAi high-throughput screen, one study identified 26 lncRNAs required for maintaining the pluripotent state of ESCs; notably, inhibition of these 26 lncRNAs led to loss of pluripotency in ESCs [91]. Therefore, lncRNA provides a new approach in the regulation of reprogramming pluripotent stem cells or fibroblasts to cardiomyocytes.
Overall, recent studies have established that fibroblasts or iPSs can be reprogrammed into mature cardiomyocytes by miRNAs or lncRNA in combination with lineage-significant reprogramming factors, an approach which holds tremendous potential for cardiac regeneration and repair.
Conclusions
The adult mammalian heart has a limited capacity to regenerate during ageing or myocardial injury. As a consequence, the endogenous renewal rate is insufficient to recover cardiomyocyte loss and to retain cardiac function under physiological or pathological conditions [92]. Recently, many newly identified ncRNAs have been found to regulate cardiac regeneration. Therefore, it is important to understand the detailed function and molecular mechanism of non-coding RNAs implicated in cardiac renewal and cardiac repair. In this review, we elucidate several ncRNAs participating in different aspects of cardiac regeneration: cardiac proliferation, differentiation, survival and reprogramming. Various studies have provided convincing evidence that miRNAs or lncRNAs may improve cell therapy or endogenous cardiac repair. However, the development of ncRNA therapy still remains challenging.
miRNA/lncRNA-based treatments is a promising emerging field in medicine [93]. One miRNA can control hundreds of genes, and in turn, one gene may be regulated by several miRNAs. Therefore, a major problem is understanding the regulatory networks between miRNAs and mRNAs. In addition, the underlying mechanisms of miRNAs in modulating cardiac regeneration need to be further explored. In addition, miRNAs and lncRNAs are distributed in several tissues and different cell types, and administration of miRNA chemical compound may lead to undesired side effects in other tissues and cells. Thus, cell type or tissue-specific strategies may be needed in future studies. Several miRNAs or lncRNAs are reported to enhance cardiac reprogramming from fibroblast or iPS to mature cardiomyocyte. However, the maturity and heterogeneity of stem cell or fibroblast derived cardiomyocytes and the low survival and retention of delivered cells remain therapeutic concerns.
Although many challenges remain, non-coding RNAs hold tremendous promise for therapeutic cardiac regeneration and repair.
Financial Support
This work was supported by the grants from National Natural Science Foundation of China (81570362 and 81200169 to JJ Xiao, 81370332 and 81170201 to XL Li, 81400647 to Y Bei), the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD20102013 to XL Li), Innovation Program of Shanghai Municipal Education Commission (13YZ014 to JJ Xiao), Foundation for University Young Teachers by Shanghai Municipal Education Commission (year 2012, to JJ Xiao), Innovation fund from Shanghai University (sdcx2012038 to JJ Xiao), and Program for the integration of production, teaching and research for University Teachers supported by Shanghai Municipal Education Commission (year 2014, to JJ Xiao). Dr XL Li is an Associate Fellow at the Collaborative Innovation Center For Cardiovascular Disease Translational Medicine.
Conflicts of interest
The authors declare there are no conflicts of interest.
References
1. Pagidipati NJ and Gaziano TA. Estimating deaths from cardiovascular disease: A review of global methodologies of mortality measurement. Circulation. 2013;127:749-56.
2. Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, de Ferranti S, Despres JP, Fullerton HJ, Howard VJ, Huffman MD, Judd SE, Kissela BM, Lackland DT, Lichtman JH, Lisabeth LD, Liu S, Mackey RH, Matchar DB, McGuire DK, Mohler ER, 3rd, Moy CS, Muntner P, Mussolino ME, Nasir K, Neumar RW, Nichol G, Palaniappan L, Pandey DK, Reeves MJ, Rodriguez CJ, Sorlie PD, Stein J, Towfighi A, Turan TN, Virani SS, Willey JZ, Woo D, Yeh RW, Turner MB, American Heart Association Statistics C and Stroke Statistics S. Heart disease and stroke statistics—2015 update: a report from the American Heart Association. Circulation. 2015;131:e29-322.
3. Bergmann O, Bhardwaj RD, Bernard S, Zdunek S, Barnabe-Heider F, Walsh S, Zupicich J, Alkass K, Buchholz BA, Druid H, Jovinge S and Frisen J. Evidence for cardiomyocyte renewal in humans. Science. 2009;324:98-102.
4. Maillet M, van Berlo JH and Molkentin JD. Molecular basis of physiological heart growth: fundamental concepts and new players. Nat Rev Mol Cell Biol. 2013; 14: 38-48.
5. Walsh S, Ponten A, Fleischmann BK and Jovinge S. Cardiomyocyte cell cycle control and growth estimation in vivo—an analysis based on cardiomyocyte nuclei. Cardiovasc Res. 2010;86:365-73.
6. van Berlo JH and Molkentin JD. An emerging consensus on cardiac regeneration. Nat Med. 2014;20:1386-93.
7. Tao LC, Bei YH, Zhang HF, Xiao JJ, Li XL. Exercise for the heart: signaling pathways. Oncotarget 2015 accepted.
8. Malliaras K, Zhang Y, Seinfeld J, Galang G, Tseliou E, Cheng K, Sun B, Aminzadeh M and Marban E. Cardiomyocyte proliferation and progenitor cell recruitment underlie therapeutic regeneration after myocardial infarction in the adult mouse heart. EMBO Mol Med. 2013;5:191-209.
9. Koitabashi N and Kass DA. Reverse remodeling in heart failure—mechanisms and therapeutic opportunities. Nat Rev Cardiol. 2012;9:147-57.
10. Srivastava D and Heidersbach AJ. Small solutions to big problems: microRNAs for cardiac regeneration. Cardiovasc Res. 2013;112:1412-4.
11. Sun L, Zhang Y, Zhang Y, Gu Y, Xuan L, Liu S, Zhao X, Wang N, Huang L, Huang Y, Zhang Y, Ren L, Wang Z, Lu Y and Yang B. Expression profile of long non-coding RNAs in a mouse model of cardiac hypertrophy. Int J Cardiol. 2014;177:73-5.
12. Duggirala A, Delogu F, Angelini TG, Smith T, Caputo M, Rajakaruna C and Emanueli C. Non coding RNAs in aortic aneurysmal disease. Front Genet. 2015; 6: 125.
13. Seeger FH, Zeiher AM and Dimmeler S. MicroRNAs in stem cell function and regenerative therapy of the heart. Arterioscler Thromb Vasc Biol. 2013; 33: 1739-46.
14. Itou J, Oishi I, Kawakami H, Glass TJ, Richter J, Johnson A, Lund TC and Kawakami Y. Migration of cardiomyocytes is essential for heart regeneration in zebrafish. Development. 2012;139:4133-42.
15. Lepilina A, Coon AN, Kikuchi K, Holdway JE, Roberts RW, Burns CG and Poss KD. A dynamic epicardial injury response supports progenitor cell activity during zebrafish heart regeneration. Cell. 2006;127:607-19.
16. Lien CL, Schebesta M, Makino S, Weber GJ and Keating MT. Gene expression analysis of zebrafish heart regeneration. PLoS Biol. 2006;4:e260.
17. Kikuchi K, Holdway JE, Werdich AA, Anderson RM, Fang Y, Egnaczyk GF, Evans T, Macrae CA, Stainier DY and Poss KD. Primary contribution to zebrafish heart regeneration by gata4(+) cardiomyocytes. Nature. 2010;464:601-5.
18. Poss KD, Wilson LG and Keating MT. Heart regeneration in zebrafish. Science. 2002;298:2188-90.
19. Jopling C, Sleep E, Raya M, Marti M, Raya A and Izpisua Belmonte JC. Zebrafish heart regeneration occurs by cardiomyocyte dedifferentiation and proliferation. Nature. 2010;464:606-9.
20. Lien CL, Harrison MR, Tuan TL and Starnes VA. Heart repair and regeneration: recent insights from zebrafish studies. Wound Repair Regen. 2012;20:638-46.
21. Haubner BJ, Adamowicz-Brice M, Khadayate S, Tiefenthaler V, Metzler B, Aitman T, Penninger JM. Complete cardiac regeneration in a mouse model of myocardial infarction. Aging (Albany NY). 2012 Dec;4(12):966-77.
22. Senyo SE, Steinhauser ML, Pizzimenti CL, Yang VK, Cai L, Wang M, Wu TD, Guerquin-Kern JL, Lechene CP and Lee RT. Mammalian heart renewal by pre-existing cardiomyocytes. Nature. 2013;493:433-6.
23. Shiba Y, Fernandes S, Zhu WZ, Filice D, Muskheli V, Kim J, Palpant NJ, Gantz J, Moyes KW, Reinecke H, Van Biber B, Dardas T, Mignone JL, Izawa A, Hanna R, Viswanathan M, Gold JD, Kotlikoff MI, Sarvazyan N, Kay MW, Murry CE and Laflamme MA. Human ES-cell-derived cardiomyocytes electrically couple and suppress arrhythmias in injured hearts. Nature. 2012;489:322-5.
24. Makkar RR, Smith RR, Cheng K, Malliaras K, Thomson LE, Berman D, Czer LS, Marban L, Mendizabal A, Johnston PV, Russell SD, Schuleri KH, Lardo AC, Gerstenblith G and Marban E. Intracoronary cardiosphere-derived cells for heart regeneration after myocardial infarction (CADUCEUS): a prospective, randomised phase 1 trial. Lancet. 2012;379:895-904.
25. Domian IJ, Chiravuri M, van der Meer P, Feinberg AW, Shi X, Shao Y, Wu SM, Parker KK and Chien KR. Generation of functional ventricular heart muscle from mouse ventricular progenitor cells. Science. 2009;326:426-9.
26. Hsieh PC, Segers VF, Davis ME, MacGillivray C, Gannon J, Molkentin JD, Robbins J and Lee RT. Evidence from a genetic fate-mapping study that stem cells refresh adult mammalian cardiomyocytes after injury. Nat Med. 2007;13:970-4.
27. Addis RC and Epstein JA. Induced regeneration—the progress and promise of direct reprogramming for heart repair. Nat Med. 2013;19:829-36.
28. Kumar D, Kamp TJ and LeWinter MM. Embryonic stem cells: differentiation into cardiomyocytes and potential for heart repair and regeneration. Coron Artery Dis. 2005;16:111-6.
29. Loffredo FS, Steinhauser ML, Gannon J and Lee RT. Bone marrow-derived cell therapy stimulates endogenous cardiomyocyte progenitors and promotes cardiac repair. Cell Stem Cell. 2011;8:389-98.
30. Dmitrieva RI, Revittser AV, Klukina MA, Sviryaev YV, Korostovtseva LS, Kostareva AA, Zaritskey AY, Shlyakhto EV. Functional properties of bone marrow derived multipotent mesenchymal stromal cells are altered in heart failure patients, and could be corrected by adjustment of expansion strategies. Aging (Albany NY). 2015 Jan;7:14-25.
31. Bersell K, Arab S, Haring B and Kuhn B. Neuregulin1/ErbB4 signaling induces cardiomyocyte proliferation and repair of heart injury. Cell. 2009;138:257-70.
32. Waring CD, Vicinanza C, Papalamprou A, Smith AJ, Purushothaman S, Goldspink DF, Nadal-Ginard B, Torella D and Ellison GM. The adult heart responds to increased workload with physiologic hypertrophy, cardiac stem cell activation, and new myocyte formation. Eur Heart J. 2014;35:2722-31.
33. Calvert JW, Condit ME, Aragon JP, Nicholson CK, Moody BF, Hood RL, Sindler AL, Gundewar S, Seals DR, Barouch LA and Lefer DJ. Exercise protects against myocardial ischemia-reperfusion injury via stimulation of beta(3)-adrenergic receptors and increased nitric oxide signaling: role of nitrite and nitrosothiols. Circ Res. 2011;108:1448-58.
34. Bergmann O, Zdunek S, Felker A, Salehpour M, Alkass K, Bernard S, Sjostrom SL, Szewczykowska M, Jackowska T, Dos Remedios C, Malm T, Andra M, Jashari R, Nyengaard JR, Possnert G, Jovinge S, Druid H and Frisen J. Dynamics of Cell Generation and Turnover in the Human Heart. Cell. 2015;161:1566-75.
35. Batista PJ and Chang HY. Long noncoding RNAs: cellular address codes in development and disease. Cell. 2013;152:1298-307.
36. Mercer TR and Mattick JS. Structure and function of long noncoding RNAs in epigenetic regulation. Nat Struct Mol Biol. 2013;20:300-7.
37. Ounzain S, Crippa S and Pedrazzini T. Small and long non-coding RNAs in cardiac homeostasis and regeneration. Biochim Biophys Acta. 2013;1833:923-33.
38. Eulalio A, Mano M, Dal Ferro M, Zentilin L, Sinagra G, Zacchigna S and Giacca M. Functional screening identifies miRNAs inducing cardiac regeneration. Nature. 2012;492:376-81.
39. Chen J, Huang ZP, Seok HY, Ding J, Kataoka M, Zhang Z, Hu X, Wang G, Lin Z, Wang S, Pu WT, Liao R and Wang DZ. mir-17-92 cluster is required for and sufficient to induce cardiomyocyte proliferation in postnatal and adult hearts. Circ Res. 2013;112:1557-66.
40. Tian Y, Liu Y, Wang T, Zhou N, Kong J, Chen L, Snitow M, Morley M, Li D, Petrenko N, Zhou S, Lu M, Gao E, Koch WJ, Stewart KM and Morrisey EE. A microRNA-Hippo pathway that promotes cardiomyocyte proliferation and cardiac regeneration in mice. Sci Transl Med. 2015;7:279ra38.
41. Bostrom P, Mann N, Wu J, Quintero PA, Plovie ER, Panakova D, Gupta RK, Xiao C, MacRae CA, Rosenzweig A and Spiegelman BM. C/EBPbeta controls exercise-induced cardiac growth and protects against pathological cardiac remodeling. Cell. 2010;143:1072-83.
42. Jin H, Yang R, Li W, Lu H, Ryan AM, Ogasawara AK, Van Peborgh J and Paoni NF. Effects of exercise training on cardiac function, gene expression, and apoptosis in rats. Am J Physiol Heart Circ Physiol. 2000;279:H2994-3002.
43. Golbidi S and Laher I. Molecular mechanisms in exercise-induced cardioprotection. Cardiol Res Pract. 2011;2011:972807.
44. Liu X, Xiao J, Zhu H, Wei X, Platt C, Damilano F, Xiao C, Bezzerides V, Bostrom P, Che L, Zhang C, Spiegelman BM and Rosenzweig A. miR-222 Is Necessary for Exercise-Induced Cardiac Growth and Protects against Pathological Cardiac Remodeling. Cell Metab. 2015;21:584-95.
45. Porrello ER, Johnson BA, Aurora AB, Simpson E, Nam YJ, Matkovich SJ, Dorn GW, 2nd, van Rooij E and Olson EN. MiR-15 family regulates postnatal mitotic arrest of cardiomyocytes. Circ Res. 2011;109:670-9.
46. Porrello ER, Mahmoud AI, Simpson E, Johnson BA, Grinsfelder D, Canseco D, Mammen PP, Rothermel BA, Olson EN and Sadek HA. Regulation of neonatal and adult mammalian heart regeneration by the miR-15 family. Proc Natl Acad Sci U S A. 2013;110:187-92.
47. Hullinger TG, Montgomery RL, Seto AG, Dickinson BA, Semus HM, Lynch JM, Dalby CM, Robinson K, Stack C, Latimer PA, Hare JM, Olson EN and van Rooij E. Inhibition of miR-15 protects against cardiac ischemic injury. Circ Res. 2012; 110:71-81.
48. Zhao Y, Ransom JF, Li A, Vedantham V, von Drehle M, Muth AN, Tsuchihashi T, McManus MT, Schwartz RJ and Srivastava D. Dysregulation of cardiogenesis, cardiac conduction, and cell cycle in mice lacking miRNA-1-2. Cell. 2007;129:303-17.
49. Zhao Y, Samal E and Srivastava D. Serum response factor regulates a muscle-specific microRNA that targets Hand2 during cardiogenesis. Nature. 2005;436:214-20.
50. Sluijter JP, van Mil A, van Vliet P, Metz CH, Liu J, Doevendans PA and Goumans MJ. MicroRNA-1 and -499 regulate differentiation and proliferation in human-derived cardiomyocyte progenitor cells. Arterioscler Thromb Vasc Biol. 2010;30:859-68.
51. Liu N, Bezprozvannaya S, Williams AH, Qi X, Richardson JA, Bassel-Duby R and Olson EN. microRNA-133a regulates cardiomyocyte proliferation and suppresses smooth muscle gene expression in the heart. Genes Dev. 2008;22:3242-54.
52. Liu N, Williams AH, Kim Y, McAnally J, Bezprozvannaya S, Sutherland LB, Richardson JA, Bassel-Duby R and Olson EN. An intragenic MEF2-dependent enhancer directs muscle-specific expression of microRNAs 1 and 133. Proc Natl Acad Sci U S A. 2007;104:20844-9.
53. Yin VP, Lepilina A, Smith A and Poss KD. Regulation of zebrafish heart regeneration by miR-133. Dev Biol. 2012;365:319-27.
54. Small EM, Frost RJ and Olson EN. MicroRNAs add a new dimension to cardiovascular disease. Circulation. 2010;121:1022-32.
55. van Rooij E, Sutherland LB, Qi X, Richardson JA, Hill J and Olson EN. Control of stress-dependent cardiac growth and gene expression by a microRNA. Science. 2007;316:575-9.
56. Chen JF, Mandel EM, Thomson JM, Wu Q, Callis TE, Hammond SM, Conlon FL and Wang DZ. The role of microRNA-1 and microRNA-133 in skeletal muscle proliferation and differentiation. Nat Genet. 2006;38:228-33.
57. Ivey KN, Muth A, Arnold J, King FW, Yeh RF, Fish JE, Hsiao EC, Schwartz RJ, Conklin BR, Bernstein HS and Srivastava D. MicroRNA regulation of cell lineages in mouse and human embryonic stem cells. Cell stem cell. 2008;2:219-29.
58. Wilson KD, Hu S, Venkatasubrahmanyam S, Fu JD, Sun N, Abilez OJ, Baugh JJ, Jia F, Ghosh Z, Li RA, Butte AJ and Wu JC. Dynamic microRNA expression programs during cardiac differentiation of human embryonic stem cells: role for miR-499. Circ Cardiovasc Genet. 2010;3:426-35.
59. Glass C and Singla DK. MicroRNA-1 transfected embryonic stem cells enhance cardiac myocyte differentiation and inhibit apoptosis by modulating the PTEN/Akt pathway in the infarcted heart. Am J Physiol Heart Circ Physiol. 2011;301:H2038-49.
60. Hosoda T, Zheng H, Cabral-da-Silva M, Sanada F, Ide-Iwata N, Ogorek B, Ferreira-Martins J, Arranto C, D’Amario D, del Monte F, Urbanek K, D’Alessandro DA, Michler RE, Anversa P, Rota M, Kajstura J and Leri A. Human cardiac stem cell differentiation is regulated by a mircrine mechanism. Circulation. 2011;123:1287-96.
61. Gabory A, Ripoche MA, Le Digarcher A, Watrin F, Ziyyat A, Forne T, Jammes H, Ainscough JF, Surani MA, Journot L and Dandolo L. H19 acts as a trans regulator of the imprinted gene network controlling growth in mice. Development. 2009;136:3413-21.
62. Ragina NP, Schlosser K, Knott JG, Senagore PK, Swiatek PJ, Chang EA, Fakhouri WD, Schutte BC, Kiupel M and Cibelli JB. Downregulation of H19 improves the differentiation potential of mouse parthenogenetic embryonic stem cells. Stem Cells Dev. 2012;21:1134-44.
63. Yin Y, Wang H, Liu K, Wang F, Ye X, Liu M, Xiang R, Liu N and Liu L. Knockdown of H19 enhances differentiation capacity to epidermis of parthenogenetic embryonic stem cells. Curr Mol Med. 2014;14:737-48.
64. Laflamme MA, Chen KY, Naumova AV, Muskheli V, Fugate JA, Dupras SK, Reinecke H, Xu C, Hassanipour M, Police S, O’Sullivan C, Collins L, Chen Y, Minami E, Gill EA, Ueno S, Yuan C, Gold J and Murry CE. Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts. Nat Biotechnol. 2007;25:1015-24.
65. Dong S, Cheng Y, Yang J, Li J, Liu X, Wang X, Wang D, Krall TJ, Delphin ES and Zhang C. MicroRNA expression signature and the role of microRNA-21 in the early phase of acute myocardial infarction. J Biol Chem. 2009;284:29514-25.
66. Yan M, Chen C, Gong W, Yin Z, Zhou L, Chaugai S and Wang DW. miR-21-3p regulates cardiac hypertrophic response by targeting histone deacetylase-8. Circ Res. 2015;105:340-52.
67. Qian L, Van Laake LW, Huang Y, Liu S, Wendland MF and Srivastava D. miR-24 inhibits apoptosis and represses Bim in mouse cardiomyocytes. The Journal of experimental medicine. 2011;208:549-60.
68. Hu S, Huang M, Nguyen PK, Gong Y, Li Z, Jia F, Lan F, Liu J, Nag D, Robbins RC and Wu JC. Novel microRNA prosurvival cocktail for improving engraftment and function of cardiac progenitor cell transplantation. Circulation. 2011;124:S27-34.
69. Rane S, He M, Sayed D, Vashistha H, Malhotra A, Sadoshima J, Vatner DE, Vatner SF and Abdellatif M. Downregulation of miR-199a derepresses hypoxia-inducible factor-1alpha and Sirtuin 1 and recapitulates hypoxia preconditioning in cardiac myocytes. Circulation research. 2009;104:879-86.
70. Salloum FN, Yin C and Kukreja RC. Role of microRNAs in cardiac preconditioning. J Cardiovasc Pharmacol. 2010;56:581-8.
71. Liu J, van Mil A, Vrijsen K, Zhao J, Gao L, Metz CH, Goumans MJ, Doevendans PA and Sluijter JP. MicroRNA-155 prevents necrotic cell death in human cardiomyocyte progenitor cells via targeting RIP1. J Cell Mol Med. 2011;15:1474-82.
72. Boon RA, Iekushi K, Lechner S, Seeger T, Fischer A, Heydt S, Kaluza D, Treguer K, Carmona G, Bonauer A, Horrevoets AJ, Didier N, Girmatsion Z, Biliczki P, Ehrlich JR, Katus HA, Muller OJ, Potente M, Zeiher AM, Hermeking H and Dimmeler S. MicroRNA-34a regulates cardiac ageing and function. Nature. 2013;495:107-10.
73. Pan Z, Sun X, Ren J, Li X, Gao X, Lu C, Zhang Y, Sun H, Wang Y, Wang H, Wang J, Xie L, Lu Y and Yang B. miR-1 exacerbates cardiac ischemia-reperfusion injury in mouse models. PloS one. 2012;7:e50515.
74. Ren XP, Wu J, Wang X, Sartor MA, Qian J, Jones K, Nicolaou P, Pritchard TJ and Fan GC. MicroRNA-320 is involved in the regulation of cardiac ischemia/reperfusion injury by targeting heat-shock protein 20. Circulation. 2009;119:2357-66.
75. Ye Y, Hu Z, Lin Y, Zhang C and Perez-Polo JR. Downregulation of microRNA-29 by antisense inhibitors and a PPAR-gamma agonist protects against myocardial ischaemia-reperfusion injury. Cardiovasc Res. 2010;87:535-44.
76. Ieda M, Fu JD, Delgado-Olguin P, Vedantham V, Hayashi Y, Bruneau BG and Srivastava D. Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell. 2010;142:375-86.
77. Qian L, Huang Y, Spencer CI, Foley A, Vedantham V, Liu L, Conway SJ, Fu JD and Srivastava D. In vivo reprogramming of murine cardiac fibroblasts into induced cardiomyocytes. Nature. 2012;485:593-8.
78. Song K, Nam YJ, Luo X, Qi X, Tan W, Huang GN, Acharya A, Smith CL, Tallquist MD, Neilson EG, Hill JA, Bassel-Duby R and Olson EN. Heart repair by reprogramming non-myocytes with cardiac transcription factors. Nature. 2012;485:599-604.
79. Jayawardena TM, Egemnazarov B, Finch EA, Zhang L, Payne JA, Pandya K, Zhang Z, Rosenberg P, Mirotsou M and Dzau VJ. MicroRNA-mediated in vitro and in vivo direct reprogramming of cardiac fibroblasts to cardiomyocytes. Circ Res. 2012;110:1465-73.
80. Bichsel C, Neeld D, Hamazaki T, Chang LJ, Yang LJ, Terada N and Jin S. Direct reprogramming of fibroblasts to myocytes via bacterial injection of MyoD protein. Cell Reprogram. 2013;15:117-25.
81. Jayawardena TM, Finch EA, Zhang L, Zhang H, Hodgkinson CP, Pratt RE, Rosenberg PB, Mirotsou M and Dzau VJ. MicroRNA induced cardiac reprogramming in vivo: evidence for mature cardiac myocytes and improved cardiac function. Circ Res. 2015;116:418-24.
82. Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K and Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861-72.
83. Buganim Y, Itskovich E, Hu YC, Cheng AW, Ganz K, Sarkar S, Fu D, Welstead GG, Page DC and Jaenisch R. Direct reprogramming of fibroblasts into embryonic Sertoli-like cells by defined factors. Cell stem cell. 2012;11:373-86.
84. Martinez NJ and Gregory RI. MicroRNA gene regulatory pathways in the establishment and maintenance of ESC identity. Cell stem cell. 2010;7:31-5.
85. Sinkkonen L, Hugenschmidt T, Berninger P, Gaidatzis D, Mohn F, Artus-Revel CG, Zavolan M, Svoboda P and Filipowicz W. MicroRNAs control de novo DNA methylation through regulation of transcriptional repressors in mouse embryonic stem cells. Nat Struct Mol Biol. 2008;15:259-67.
86. Card DA, Hebbar PB, Li L, Trotter KW, Komatsu Y, Mishina Y and Archer TK. Oct4/Sox2-regulated miR-302 targets cyclin D1 in human embryonic stem cells. Mol Cell Biol. 2008;28:6426-38.
87. Subramanyam D, Lamouille S, Judson RL, Liu JY, Bucay N, Derynck R and Blelloch R. Multiple targets of miR-302 and miR-372 promote reprogramming of human fibroblasts to induced pluripotent stem cells. Nat Biotechnol. 2011;29:443-8.
88. Li Z, Yang CS, Nakashima K and Rana TM. Small RNA-mediated regulation of iPS cell generation. EMBO J. 2011;30:823-34.
89. Loewer S, Cabili MN, Guttman M, Loh YH, Thomas K, Park IH, Garber M, Curran M, Onder T, Agarwal S, Manos PD, Datta S, Lander ES, Schlaeger TM, Daley GQ and Rinn JL. Large intergenic non-coding RNA-RoR modulates reprogramming of human induced pluripotent stem cells. Nat Genet. 2010;42:1113-7.
90. Sheik Mohamed J, Gaughwin PM, Lim B, Robson P and Lipovich L. Conserved long noncoding RNAs transcriptionally regulated by Oct4 and Nanog modulate pluripotency in mouse embryonic stem cells. Rna. 2010;16:324-37.
91. Guttman M, Donaghey J, Carey BW, Garber M, Grenier JK, Munson G, Young G, Lucas AB, Ach R, Bruhn L, Yang X, Amit I, Meissner A, Regev A, Rinn JL, Root DE and Lander ES. lincRNAs act in the circuitry controlling pluripotency and differentiation. Nature. 2011;477:295-300.
92. Mahmoud AI, Kocabas F, Muralidhar SA, Kimura W, Koura AS, Thet S, Porrello ER and Sadek HA. Meis1 regulates postnatal cardiomyocyte cell cycle arrest. Nature. 2013;497:249-53.
93. Uchida S and Dimmeler S. Long noncoding RNAs in cardiovascular diseases. Circ Res. 2015;116:737-50.

