INTRODUCTION
Lymphoma is one of the first discovered hematologic malignancies derived from lymph nodes or lymph tissues. Occurrence of lymphoma is often correlated with malignant proliferation of lymphocytes in immune responses. In China, Non-Hodgkin lymphoma (NHL) accounts for about 90% of malignant lymphadenoma (ML) and non-malignant lymphadenoma (NL) accounts for 10%. Worldwide, lymphomas developed in 566,000 people in 2012 and caused 305,000 deaths. They make up 3–4% of all cancers, making them as a group the seventh-most common form. In children, they are the third-most common cancer.
Biopsy of lymph node or bone marrow and histopathological analysis can not diagnose it accurately. Identification of novel molecule is crucial for lymphoma treatment and prognosis.
The P2X7 receptor (P2X7R) is expressed on the cell membrane; when activated by ATP, P2X7R forms a cation channel or a non-specific large pore, allowing Na+, K+, Ca2+ or other large hydrophilic molecules to pass through [1]. P2X7R is widely expressed on non-cancer cells, including hematopoietic stem cells, macrophages, mast cells, T-lymphocytes and B-lymphocytes [2], but it is often expressed on cancer cells, such as leukemia [3], prostate cancer [4], neuroblastoma [5] and thyroid papillary carcinoma [6]. Some studies have reported different levels of P2X7R associated with different levels of cancer severity and Zhang found P2X7R expression levels were different in different types of leukemia cells [3]. After one course of standard induction therapy, the survival rate in a high P2X7R expression group was lower than that in either a P2X7R negative group or a low P2X7R expression group. In B-cell chronic lymphocytic leukemia patients, Adinofi found the expression of P2X7R was significantly higher in lymphocytes from patients with the evolutive compared with the indolent variant [7]. These findings further suggest the expression of P2X7R is associated with the severity of cancer.
T complex polypeptide 1 (TCP-1) which acts as a member of the chaperonin assists the synthesis of cytoskeletal proteins, so far it has approved that TCP-1 is over-expressed in varieties of cancers and shows increased expression with advancing stage [8, 9]. As a member of the group II chaperonin family, can assist with the folding of newly synthesized proteins [10], including tubulins and actins [11], cyclin E [12], α-transducin [13] and von Hippel Lindau protein [14], which are all essential for cell functions. There are few reports of the relationship between TCP-1 and tumors; however, TCP-1 has been found to be correlated with the occurrence of liver cancer and colon cancer, and the content of TCP-1 was positively correlated with the severity of liver cancer and colon cancer [8]. Further, it has been reported that TCP-1 interacts with some tumor metastasis-associated proteins, such as tubulins and actins [11].
In our earlier study, in which we altered the expression of P2X7R in P388D1 cells and observed the changes in P388D1 metastatic capability, we found that the expression of P2X7R appeared to be correlated with P388D1 cells metastasis [15]. In this study, proteins from lymph nodes which metastasized by transfected P2X7R shRNA P388D1 cells and control cells were separated by two-dimensional gel electrophoresis (2DE). After that we analyzed the results of two groups by mass spectrum, TCP-1 appeared to have a close relationship with P2X7R in tumor metastasis. This results rouse our great interests and finally let us put TCP-1 into the focus of our study. As mass spectrometry (MS) results shown, TCP-1 expression was down-regulated in the metastatic lymph nodes of P2X7R shRNA DBA/2 mice than control group, indicating there might be correlation between the expression of TCP-1 and P2X7R. We silenced the expression of P2X7R in P388D1 cells with shRNA in vivo and in vitro. It was found that TCP-1 expression was also decreased significantly same as P2X7R. This result supported the indication that TCP-1 was correlated with P2X7R. Interestingly, we also found the positive relation between P2X7R and TCP-1 in human lymphoma tissues. In order to know if TCP-1 mediates the function that P2X7R inhibits the metastasis of murine lymphoid neoplasm cells to lymph nodes, we reduced the expression of TCP-1 in the P388D1, L5178Y and L1210 cell lines by TCP-1 shRNA and finally found that down-regulating TCP-1 could inhibit lymphoma cells metastasis in vivo.
In this study, we have proved that silence P2X7R expression can significantly decreased the expression of TCP-1 both in vivo and in vitro, the correlative expression of P2X7R and TCP-1 was also founded in human lymphoma tissues. In addition, we found that down-regulating TCP-1 can greatly attenuate the metastatic capability of the mouse lymphoma cells. Therefore, these results suggested that TCP-1 as a novel downstream molecular of P2X7R, played a crucial role in the inhibition of lymphoid neoplasm metastasis by silencing P2X7R. We propose that TCP-1 may be a new target for the therapy of lymphoma.
RESULTS
Proteins differentially expressed between the lymph nodes of P2X7R shRNA group and the controls
On the basis of our earlier study, it was concluded that P2X7R has a potential role in tumor dissemination to peripheral lymph nodes, P388D1 cells (6 × 105) transfected with P2X7R shRNA and P2X7R control shRNA were injected into the footpads of 6 mice respectively. The survival rate of mice treated by P2X7R shRNA was significantly longer than that of the control groups. The lymph nodes in the P2X7R shRNA group were much smaller than those in the control group [15]. In order to further study its mechanism, we got the lymph nodes by the same way as before, and the lymph nodes from P2X7R shRNA group and controls were analyzed by 2DE and staining with silver nitrate detected the protein spots in the gels within a pH range of 3 to 10. We selected 8 remarkable spots of differentially expressed protein between P2X7R shRNA group and the controls via statistical analysis of the 2DE gels (Figure 1).
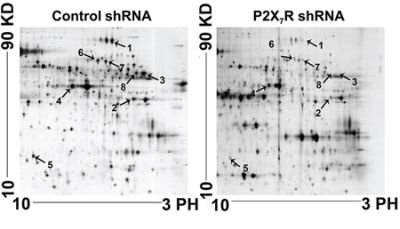
Figure 1: Proteomic analysis of the lymph nodes of P2X7R shRNA groups and control groups using 2-DE gels. The arrows indicate protein spots that differ significantly between the P2X7R shRNA and control group. Relative spot intensity was calculated on the basis of the spot volume using Imagemaster™ 2D Platinum software. Only the spots indicated with arrows were used for identification.
Mass spectrum identification of differentially expressed proteins
The 8 spots were identified by MS/MS analysis and 64 proteins were affirmed from these spots (Supplementary Table S1). Compared to these 64 proteins, we found 8 proteins with higher mass sequence coverage (> 30%): transferrin, an unnamed protein product (gi|74216774), fibrinogen β chain, hypothetical protein LOC433182, purine nucleoside phosphorylase, stress-induced phosphoprotein 1, phosphoglycerate kinase 1 and TCP-1. In contrast with TCP-1 the former 6 proteins were seldom correlated with tumor metastasis, and the phosphoglycerate kinase 1 participating mainly in the angiogenesis is critical for tumor expansion and metastasis [16]. Apart from the 7 proteins, TCP-1 displayed a significant decrease in P2X7R shRNA group. Yokota [8] found TCP-1 was correlated with liver cancer and colon cancer, and the level of TCP-1 was positively related with the severity of the cancer. TCP-1 can interact with a variety of proteins, including cytoskeletal proteins that impact on tumor metastasis [11]. Therefore, we focused on TCP-1 in our study. The mass spectra of TCP-1 in lymph nodes of P2X7R shRNA group and the P2X7R control shRNA group are shown in Figure 2. The MS/MS data supporting the suggestion appeared to be reliable (MASCOT score 461, sequence coverage 34%).
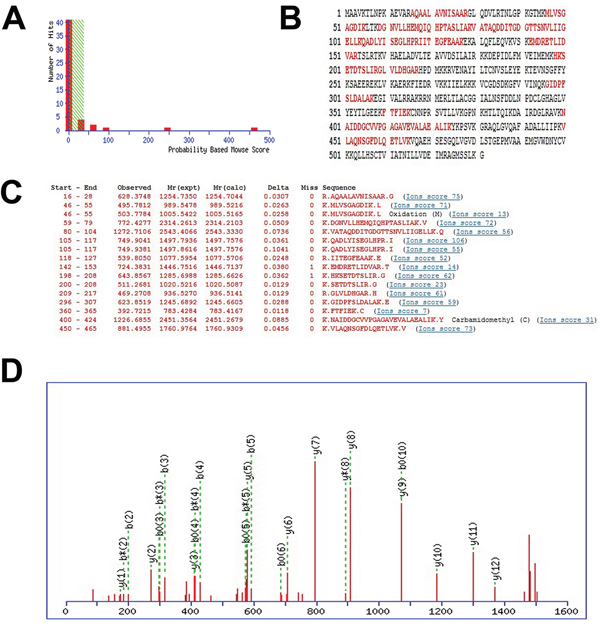
Figure 2: The identification of differentially expressed proteins. A, B, and C. output of the database searching by the MASCOT engine using MS data used in the identification of TCP-1. The matched peptides were shown in bold red. D. MS/MS map marked with b ions and y ions for TCP-1 identification.
Validation of the down-regulation of TCP-1 expression in P2X7R shRNA group in vitro and in vivo
The levels of P2X7R and TCP-1 in P388D1 cells transfected by P2X7R shRNA and P2X7R control shRNA were measured by RT-PCR and immunofluorescence respectively. As it shown, P2X7R shRNA simultaneously decreased the expression of P2X7R and TCP-1 in P388D1 cells compared to the controls (Figure 3A). Immunofluorescence analysis also demonstrated that lower of P2X7R and TCP-1 were expressed in P2X7R shRNA transfected P388D1 cells compared to the controls (Figure 3B). The differential expression of TCP-1 in DBA/2 mice lymph nodes of P2X7R shRNA group and control shRNA group was verified at the protein level by Western blot analysis with TCP-1 specific antibody (Figure 3C), we found TCP-1 expression in lymph nodes of P2X7R shRNA group was decreased significantly. Furthermore, immunohistochemistry also showed that TCP-1 expression was down-regulated in the lymph nodes of P2X7R shRNA group compared to the control shRNA group (Figure 3D). The results confirmed that lower levels of TCP-1 were expressed in the lymph nodes of P2X7R shRNA group than control group. These results further proved that silencing P2X7R can down-regulate the TCP-1 expression. It suggested, at least partly, the close relation between the expression of P2X7R and TCP-1.
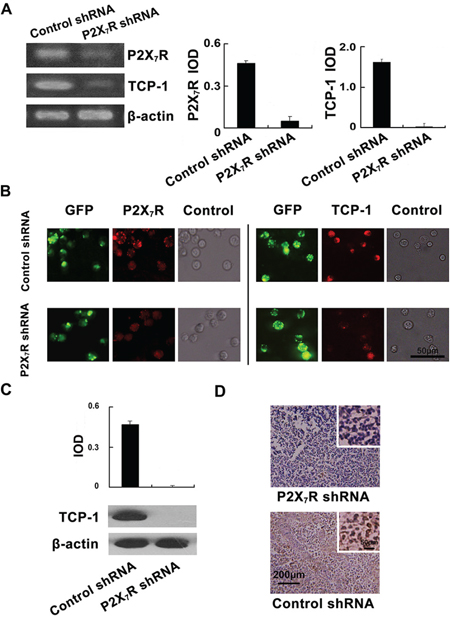
Figure 3: Validation of the down-regulation of TCP-1 expression in P2X7R shRNA group in vitro and in vivo. A. P2X7R expression in P388D1 cells of P2X7R shRNA group and P2X7R control shRNA group was detected by RT-PCR. P2X7R as well as TCP-1 expressed in P388D1 cells in the P2X7R shRNA group was lower than that in the control group (Left). Densitometric analysis of the RT-PCR. Data were obtained using the Gel-Pro32 analyzer software with the values normalized to β-actin levels (Right). B. (Left Panel) P2X7R shRNA transfected cells and control cells were labeled with GFP; P2X7R was stained red with TRITC-labeled P2X7R antibody and located at the cell membrane. The expression of P2X7R in P388D1 cells of P2X7R shRNA group [10.4(±1.5) %, n = 4] was decreased compared to control group [72.6(±1.2) %, n = 4] (p < 0.001); The P388D1 cells in two groups were observed in bright field. (Right Panel) The transfection of P388D1 cells was visualized by GFP; P388D1 cells in two groups were incubated with TCP-1 goat polyclonal antibody and stained with TRITC-labeled secondary antibody. TCP-1 expressed in the cytoplasm of P388D1 cells was stained red. The expression of TCP-1 in P388D1 cells of the P2X7R shRNA group [9.8(±1.19) %, n = 4] were decreased compared to the controls [63.4(±1.2) %, n = 4] (p < 0.001); The P388D1 cells of the two groups were observed in bright field. Error bars represent ±S.D. of the mean. Scale bar represents 50 μm for all the micrographs. C. TCP-1 in the lymph nodes of the P2X7R shRNA group and control group was analyzed by Western blot. The corresponding quantification of the Western blot, values were normalized to β-actin levels. D. Immunohistochemical staining of TCP-1 in mouse lymph nodes of two groups. TCP-1 expressed in the cytoplasm of lymph nodes cells was stained brown.
Analysis of the expression correlation between P2X7R and TCP-1 in lymphoma tissues from patients by immunohistochemistry
In order to know the relationship between P2X7R and TCP-1 in human lymphoma, immunohistochemical analysis about P2X7R and TCP-1 was performed. P2X7R and TCP-1 were observed expressing in both lymphoma and benign lymphadenopathy (Figure 4A). In lymphoma cases, the cases expressed P2X7R and TCP-1 (P2X7R+/TCP-1+) was more than 86%. In all the P2X7R positive cases, more than 96.8% expressed TCP-1. In benign lymphadenopathy, more than 88.2% cases had no expression of P2X7R and TCP-1 (P2X7R-/TCP-1-). In all the P2X7R positive cases, there is 50% expressed TCP-1 (Figure 4B and 4C). Details were shown in Table 1, According to Fisher exact test, the result suggested the positive correlation between P2X7R and TCP-1 in both lymphoma and benign lymphadenopathy of lymph nodes.
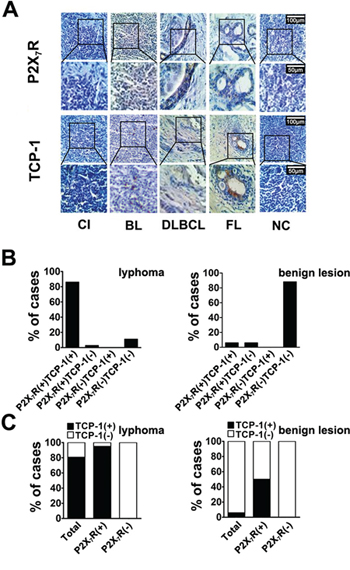
Figure 4: Immunohistochemical analysis of P2X7R and TCP-1 expression in human lymphoma and benign lymphadenopathy. A. P2X7R and TCP-1 in various types of human lymphoma and benign lymphadenopathy was specifically detected by corresponding antibody. Sections came from a case of CI (Chronic inflammation), a case of BL (B cell lymphoma), a case of DLBCL (Diffuse large B cell lymphoma type), a case of FL (Follicular lymphoma) and a case of NC (Negative control). P2X7R and TCP-1 expression in same pathological specimen was observed by microscope, Scale bar represents 100 μm (upper plane) and 50 μm (bottom plane) respectively. P2X7R was stained brown on cell membrane and TCP-1 was shown in cytoplasm with brown. B. the different expression patterns of P2X7R and TCP-1 among all the lymphoma cases (Left); the different expression patterns of P2X7R and TCP-1 among benign lymphadenopathy (Right). C. the distribution of TCP-1 positive and negative cases among all lymphoma cases, P2X7R positive and negative lymphoma cases (Left); the distribution of TCP-1 positive and negative cases among all benign lymphadenopathy cases, P2X7R positive cases and negative benign lymphadenopathy (Right).
Table 1: The correlation of P2X7R and TCP-1 expression in lymphoma and benign lymphadenopathy
Consistent results |
Inconsistent results |
P valuea |
|||
|---|---|---|---|---|---|
− / − |
+ / + |
− / + |
+ / − |
||
Lymphoma P2X7R/TCP-1 |
8 |
62 |
0 |
2 |
P < 0.05 |
benign lymphadenopathy P2X7R/TCP-1 |
15 |
1 |
0 |
1 |
P < 0.05 |
aThe P values were statistically analyzed in Fisher exact.
(+)stands for positive results: (–)stands for negative results.
TCP-1 expressed in murine lymphoma neoplasm cells and could be silenced by TCP-1 shRNA
We detected the expression of TCP-1 in three kinds of murine lymphoma cell lines, P388D1, L5178Y and L1210, RT-PCR and Western blot confirmed that TCP-1 was indeed expressed in these cell lines (Figure 5A). All the cell lines were transfected by two different TCP-1 shRNA and control shRNA respectively. The effect of two TCP-1 specific shRNA was examined by RT-PCR and Western blot analysis (Figure 5B and 5C). Comparing with the control shRNA group, both of TCP-1 shRNA a and TCP-1 shRNA b could reduce the TCP-1 expression in the three cell lines, but TCP-1 shRNA a was more efficient than TCP-1 shRNA b. In TCP-1 shRNA a group. This data shown that the expression of TCP-1 was efficiently down-regulated in P388D1, L5178Y and L1210 cells transfected by TCP-1 shRNA a. Thus, TCP-1 shRNA a was chosen in the subsequent experiments.
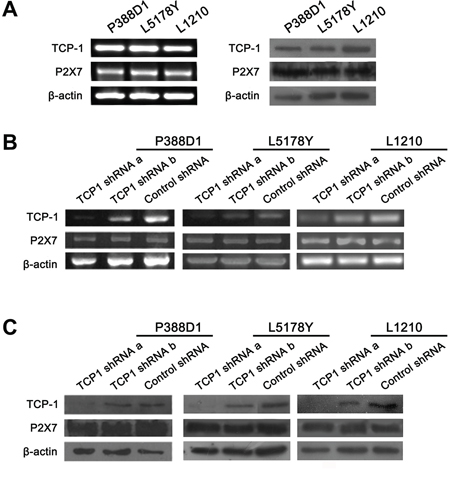
Figure 5: The expression of P2X7R and TCP-1 in different murine lymphoid neoplasm cell lines in TCP-1 shRNA group, control shRNA group and control group in vitro. A. The expression of TCP-1 and P2X7R in P388D1, L5178Y and L1210 cell lines was detected by RT-PCR (Left) and western-blot assay (Right). B. the expression of TCP-1 and P2X7R in P388D1, L51778Y and L1210 cell line in different groups was detected by RT-PCR. C. TCP-1 and P2X7R expression in P388D1, L51778Y and L1210 cell line in different groups was detected by Western-blot assay.β-actin was used as control.
Experimental metastasis in DBA/2 mice
We have verified that TCP-1 shRNA a could silence the expression of TCP-1, and then we want to know whether the reduced expression of TCP-1 could regulate lymphoma progression and metastasis. For P388D1 cell line, 15 female and male inbred strain DBA/2 mice were divided at random into three groups (five mice in each group), TCP-1 shRNA a group, control shRNA group and control group. Each mouse was injected in the footpad with 6 × 105 P388D1 cells. The same process was performed with L1210 cell line. Kaplan-Meier survival curves were constructed to compare the development of two cell lines of three groups. With P388D1 cell line, we found the survival percentage of mice in TCP-1 shRNA a group was conspicuous longer than it in control shRNA group and control group (P < 0.05), With L1210 cell line, the survival percentage of mice in TCP-1 shRNA a group was also longer than control shRNA group and control group (P < 0.01) (Figure 6A). Two cell lines formed tumor in all mice, in the comparison in three groups of each cell lines, we found the same results that lymph nodes of metastasized tumor cells from the mice in TCP-1 shRNA a group were smaller and lighter than those in the control shRNA and control group, no difference was found between control shRNA group and control group (Figure 6B and 6C). It was shown that down-regulated TCP-1 could reduce the metastasis of the tumor greatly. These results indicated that TCP-1 expressed in P388D1 and L1210 cells was necessary in the process that tumor metastasis to lymph nodes. To confirm this hypothesis, immunohistochemistry assay was performed to detected the expression of TCP-1 in the lymph nodes from three groups (Figure 6D). As expected, same results were got with two cell lines that TCP-1 was expressed lower in the TCP-1 shRNA group lymph nodes than other two groups. Based on these results, we concluded that TCP-1 played a crucial role in the inhibition of lymphoid neoplasm metastasis.
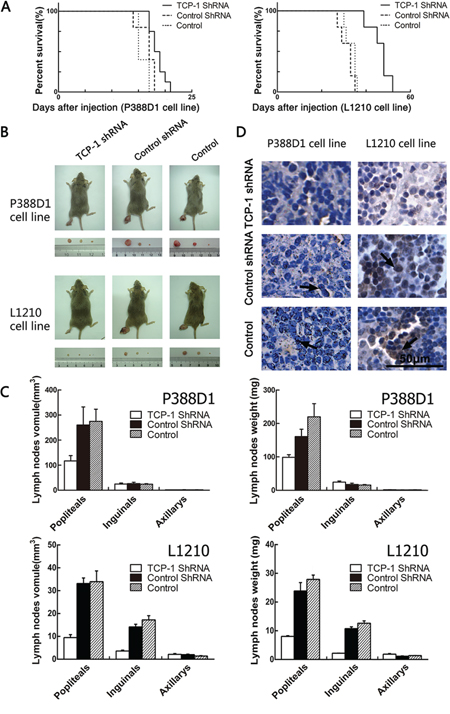
Figure 6: Experimental metastasis in DBA/2 mice with P388D1 and L1210. A. comparison of the survival percentage of the TCP-1 shRNA group, control shRNA group and control group (values were mean ± S.D., n = 5; P < 0.05). B. the lymph nodes got from the mice in TCP-1 shRNA group were significantly smaller than those in control shRNA group and control group. C. Reduced tumor volumes in the popliteal lymph nodes of DBA/2 mice injected with TCP-1 shRNA treatment P388D1 cells, compared with control shRNA treated (P = 0.0016) and non-treated P388D1 cells (P = 0.0016), and reduced tumor weight in the popliteal lymph nodes of DBA/2 mice injected with TCP-1 shRNA treatment P388D1 cells, compared with control shRNA treated (P = 0.0034) and non-treated P388D1 cells (P = 0.0026). Volumes of the popliteal lymph nodes of DBA/2 mice injected with TCP-1 shRNA treatment L1210 cells was significantly decreased compared to control shRNA treated (P = 0.000026) and non-treated L1210 cells (P = 0.0011), Volumes of the inguinal lymph nodes of TCP-1 shRNA treatment L1210 cells group was also significantly decreased compared to control shRNA treated group (P = 0.000029) and non-treated L1210 cells group (P = 0.000079); reduced tumor weight in the popliteal lymph nodes of DBA/2 mice injected with TCP-1 shRNA treatment L1210 cells, compared with control shRNA treated (P = 0.00069) and non-treated L1210 cells (P = 0.00013) and reduced tumor weight in the inguinal lymph nodes of DBA/2 mice injected with TCP-1 shRNA treatment L1210 cells, compared with control shRNA treated (P = 0.00016) and non-treated L1210 cells (P = 0.00022). Each datum point is the mean ± S.D. of five lymph nodes. D. immunohistochemical staining with TCP-1. Tissues came from lymph nodes metastasized P388D1 and L1210 cells respectively. TCP-1 was stained brown in lymph node tissues of three groups, Scale bar represents 50 μm for all the micrographs.
DISCUSSION
Our results showed the metastatic capacity was decreased significantly in the P2X7R-silenced P388D1 cells. We found that the expression of TCP-1 in tumor metastatic lymph nodes was also decreased while P2X7R was silenced. These results supported our suggestion that TCP-1 is involved in lymphoma lymphatic metastasis. We have several suggestions to explain this phenomenon. First, TCP-1 assists the correct folding of the cytoskeletal proteins actin and tubulin [11], which are important components of the filaments and microtubules needed for cell movements. A decreased level of TCP-1 reduces actin and tubulin synthesis, thus reducing the amount of cell motility-related proteins and weakening tumor metastatic potential [17]. Second, studies have shown TCP-1 can influence the synthesis of cell-cycle proteins, such as cyclin E, which has been reported to be highly expressed in many types of cancer cells and to regulate the cell cycle [18, 19]. A decreased level of TCP-1 lowers the content of cyclin E, which reduces the capability of tumor cell division and proliferation and, thus, reduces tumor metastasis. Finally, TCP-1 can interact with other proteins, such as the von Hippel-Lindau (VHL) tumor suppressor protein [14], histone deacetylase [20] and protein phosphatase PP2A regulatory subunit B [21], myosin and transducin. In mammalian cells, the unassembled form of VHL is associated with two different molecular chaperones, Hsp70 and TCP-1 [14, 22]. In vitro translation experiments in TCP-1 immunodepleted extracts indicate that this chaperonin is required for VHL incorporation into von Hippel-Lindau protein (VHL)-elongin BC [22]. More than 70 unidentified polypeptide species associated with TCP-1 in vivo can be immunoprecipitated [10].
In addition, the P2X7R has been shown to mediate ATP-induced cell death in human embryonic kidney cells [23] and human cervical epithelial cells [24], and its downstream signaling is coupled to pro-inflammatory cascades [25]. The P2X7R-mediated apoptosis of mast cells involved the activation of caspases −3 and −8, and the cleavage of caspase-3 substrate PARP [26]. It has been suggested that in the time period between commitment to apoptosis and actual cell death, agonistic stimulation of P2X7R also activates additional signaling pathways, which may lead to cytokine production in case of proteolytic processing and release of IL-1 from LPS-primed macrophages that precedes cell death [27], and activation of various transcription factors like NFAT or NF-κB [28]. Brief stimulation of the P2X7R with ATP results in formation of a non-selective cationic channel that promotes the influx of Ca2+ and the equilibration of the transmembrane sodium and potassium gradients leading to membrane depolarization [29]. Extracellular release of active IL-1β and IL-18 is dependent on ATP-sensitive P2X7R activation [30]. The anti-apoptotic property of the P2X7R has been described in neutrophils of patients with rheumatoid arthritis (RA) [31]. Serum amyloid A (SAA) purified from the plasma of patients with RA, or recombinant SAA, suppressed both spontaneous and α-FAS (CD95) induced-neutrophil apoptosis of human neutrophils in vitro; Oxidized ATP is an unselective P2X7R antagonist and it can attenuate inflammatory responses independent of P2 receptor blockade and inhibit this SAA-mediated anti-apoptotic effect [30]. Nagaoka et al. have also demonstrated an anti-apoptotic action of P2X7R [32]. So this anti-apoptotic action was reduced in the P2X7R-silenced P388D1 cells, it resulted a promotion of apoptosis, thus reduced tumor metastasis. Although no direct association with cancer metastasis has been found, these proteins have close relationships with gene expression, cell signal transduction and the cell cycle; therefore, TCP-1 affecting the synthesis of these proteins is likely to influence tumor metastasis.
The related mechanism of P2X7R and TCP-1 may be very complex, possibly involving signaling pathway, cytokine and other multilevel regulation. There are several signal pathways which may be associated with it. Recent studies found activation of the MAPK pathway in cancer cells can enhance cell invasion and promote peripheral vascular generation, thereby increasing the metastatic potential of cancer [33]. It has been reported that inhibiting P2X7R expression can suppress activation of the MAPK pathway [34, 35]. Inhibition of the MAPK pathway can reduce the synthesis of ternary complex factors (TCFs), which further inhibit expression of the TCP-1 gene [36]. PI3K is also a possible pathway. Activation of the PI3K pathway can promote transformation of normal cells into abnormal calls, and can enhance cancer cell invasion and metastatic potential [37]. AKT is the important intermediate molecule in the PI3K pathway. It has been reported that inhibition of P2X7R expression could inhibit activation of the PI3K pathway and reduce the level of AKT phosphorylation [38]. TCP-1 is the downstream effector of AKT and decreased AKT phosphorylation reduces the level of TCP-1 phosphorylation [39]. But this demonstrated only that inhibition of P2X7R causes a decrease of TCP-1 phosphorylation; whether it changes TCP-1 content is still not known. Besides, caspase as an important apoptosis pathway is closely related with P2X7R. Studies have found in skin neoplasias of mice the activation of P2X7R can activate caspase-9 and caspase-3 so as to activate the mitochondrial - caspase-9 apoptosis pathway [40]; the study of TCP-1 also found that in uterine cancer cells the disruption of TCP-1 activates both intrinsic (caspase-3, caspase-6, caspase-7, and caspase-9) and extrinsic (caspase-2, caspase-8, and caspase-10) caspase-dependent apoptotic pathway [41]. These to some extent manifested the apoptosis pathway is likely to be linked to P2X7R and TCP-1.
MATERIALS AND METHODS
Ethics statement
Investigation has been conducted in accordance with the ethical standards and according to the Declaration of Helsinki and according to national and international guidelines and has been approved by the Research Ethics Committee of Dalian Medical University in agreement with institutional guidelines.
Patients
Between 2003 and 2011, a total of 89 consecutive patients, include 72 lymphoma and 17 benign lymphadenopathy, at the 2nd Affiliated Hospital of Dalian Medical University were involved in the study. The histological types were as follows: in 72 lymphoma, there were B-cell lymphoma (n = 17), T-cell lymphoma (n = 9), diffuse large B-cell lymphoma (n = 28), follicular lymphoma (n = 10), Hodgkin’s lymphoma (n = 6), mantle cell lymphoma (n = 1), small lymphocytic lymphoma (n = 1). 17 benign lymphadenopathy include chronic inflammation (n = 8), reactive hyperplasia (n = 9). Informed consent was obtained from all participants. Clinic pathological characteristics of patients was shown in Table 2. More detail information of patients could be seen in Supplementary Table S2.
Table 2: Clinic pathological characteristics of patients
Characteristic |
Patients |
% |
|---|---|---|
Lymphoma |
72 |
|
Age Median (range) |
56.5 (20–18) |
|
Mean ± SD |
56.29 ± 14.66 |
|
Gender |
||
Male |
42 |
58.3 |
Female |
30 |
41.7 |
Histologic type |
||
B-cell lymphoma |
17 |
23.6 |
T-cell lymphoma |
9 |
12.5 |
Diffuse large B-cell lymphoma |
28 |
38.9 |
Follicular lymphoma |
10 |
13.9 |
Mantle cell lymphoma |
1 |
1.4 |
Small lymphocytic lymphoma |
1 |
1.4 |
Hodgkin’s lymphoma |
6 |
8.3 |
Benign lymphadenopathy |
17 |
|
Age Median (range) |
47 (4–75) |
|
Mean ± SD |
44.65 ± 21.3 |
|
Gender |
||
Male |
8 |
47.1 |
Female |
9 |
52.9 |
Histologic type |
||
Chronic inflammation |
8 |
47.1 |
Reactive hyperplasia |
9 |
52.9 |
Cell culture and mice
Three murine lymphoma cell lines were used in this study, P388D1, L5178Y and L1210 respectively. All the cell lines were purchased from the Institute of Biochemistry and Cell Biology, the Chinese Academy of Sciences (Shanghai, China). P388D1 cells were cultured in RPMI 1640 medium (Sigma) supplemented with 10% FBS and 100 U/ml penicillin/streptomycin (Beyotime) at 37°C in a humidified 5% CO2 atmosphere. L5178Y and L1210 cells were cultured in DMEM medium (Sigma) supplemented with 10% FCS and 100 U/ml penicillin/streptomycin (Beyotime) at 37°C in a humidified 5% CO2 atmosphere. 6–8 weeks old DBA/2 mice were obtained from the specific pathogen free animal center of Dalian Medical University (Dalian, China).
Plasmids and lentivirus
shRNA sequence for knockdown of P2X7R was 5′-UGAGCGAUAAGCUGUACCAUUGAAGAAGUGG UACAGCUUAUCGCUCAUU-3′. The control shRNA sequence for P2X7R was 5′-GTTCTCCGAACGTGTCAC GTCAAGAGATTACGTGACACGTTCGGAGAATT-3′. Two different sequences for silencing TCP-1 were a: 5′-GCTGGAGACATCAAACTTACT-3′; and b: 5′-GCGATGGCACTACATCCAATG-3′. A scrambled sequence having no significant homology with target sequence databases as negative control. It was 5′-TTCTCCGAACGTGTCACGTTTC-3′. Five plasmids contained P2X7R shRNA, P2X7R control shRNA, two kinds of TCP-1 shRNA, and TCP-1 control shRNA respectively and three lentivirus contained two kinds of TCP-1 shRNA, and TCP-1 control shRNA were constructed by Shanghai Gene Pharma Co. Ltd.
Stable transfection of cells
For plasmids transfection, P388D1 and L5178Y cells were plated at 40–50% confluence in 3.5 cm cell culture dishes and kept overnight. TCP-1 shRNA and control shRNA were transfected with Effectene Transfection Reagent (Qiagen, Cat. No. 301425) following the manufacturer’s instructions. For lentivirus infection, the most suitable multiplicity of infection (MOI) and the minimum concentration of puromycin (Sigma, Cat. No. P8833) required to kill uninfected cells were determined by preliminary test. L1210 cells in logarithmic growth phase were seeded in 24 wells culture plates with 5 × 104 cells/well. Since grown to 30% confluence, cells were infected with lentivirus at a MOI of 60 with polybrene (10 μg/mL). After 24 h, the medium containing virus were replaced with fresh complete medium. Stably transfected L1210 cells were selected by adding puromycin (4 μg/mL) in medium. GFP in the successfully transfected cells was verified by fluorescent microscope and the transfection efficiency was measured by the percentage of GFP positive cells. The silence efficiency was evaluated by RT-PCR and western blot analysis.
Animal experiments
The lymph node tissues were obtained as our earlier described [15]. Briefly, transfected murine lymphoma cells (6 × 105) were injected into the footpads of DBA/2 mice in different groups respectively. The mice were sacrificed after several weeks and the lymph nodes were collected and prepared for subsequent experiments.
Two-dimensional gel electrophoresis (2DE) and image analysis
The lymph node tissue sample (0.1 g) from mice injected by P3388D1 treated by P2X7R shRNA and control shRNA was ground into a powder in liquid nitrogen, homogenized in 1 ml of lysis buffer (40 mM Tris-HCl, 7 M urea, 2 M thiourea, 4% Chaps, 1% DTT, 1 mM EDTA) on ice, vortex mixed thoroughly, kept on ice for 1 h then centrifuged at 15,000 g for 1 h at 4°C. The supernatant was collected and placed into an Eppendorf tube. After preparation of the total protein, isoelectric focusing (IEF) was done according to the guide provided with the IPGphor system (Amersham Pharmacia Biotech). The IEF gel strips were equilibrated for 15 min in 10 ml of equilibrium solution (50 mM Tris-HCl, pH 8.8, 30% glycerol, 6 M urea, 2% SDS and a trace of bromophenol blue). After equilibration, separation in the second dimension was done by SDS-PAGE (13% polyacrylamide gel) using the PROTEN II xi Cell system (BIO-RAD) and then the gel was stained with silver nitrate. The protein spots were detected, quantified and matched using ImageMaster 2D Platinum (Amersham Pharmacia Biotech).
In-gel tryptic digestion and protein identification by MS
After 2DE, the sample was subjected to in-gel tryptic digestion and protein identification by MS. Briefly, protein spots of interest were excised and destained. In-gel digestion was done with 0.01 μg/μl trypsin for 15 h at 37°C. The tryptic peptides were extracted from the gel and dried by centrifugal lyophilization. The dried peptide mixture was dissolved in 5% trifluoroacetic acid (TFA). Electrospray ionization (ESI-MS/MS) was carried out with a hybrid quadrupole orthogonal acceleration tandem mass spectrometer (Q-TOF2, Micromass Ltd., Manchester, UK). Glu-Fibrinopeptide was used to calibrate the instrument in the MS/MS mode. MS/MS spectra were transformed using MaxEnt3 (MassLynx, Micromass Ltd.). The database search was finished with the Mascot search engine (http://www.matrixscience.co.uk) using a Mascot MS/MS ion search through National Center for Biotechnology Information nonredundant (NCBInr) databases (date: 20090410, 8201094 sequences). The parameters for searching were enzyme of trypsin; Taxonomy: Mus. (144439 sequences); Max Missed Cleavages: 1; Variable modifications of Acetyl (Protein N-term), Carbamidomethyl (C), Oxidation (M); peptide mass tolerance and fragment mass tolerance were both ± 0.2 Da.
Western blot analysis
The extracted proteins were prepared and separated on 12% SDS-PAGE gel, and then transferred to NC membrane (Amersham Biosciences). The membrane was put into blocking buffer (5% skimmed milk in TBST) for 1 h at room temperature and blotted with primary antibodies, P2X7R rabbit polyclonal antibody (1:100, Santa Cruz Biotechnology, sc-25698), TCP-1 goat polyclonal antibody (1:100, Santa Cruz Biotechnology, sc-13896), in 5% skimmed milk overnight at 4°C. After washing three times for 10 min each, NC membrane was incubated with secondary antibodies, HRP-conjugated goat anti-rabbit IgG (1:5000, ZSGB-BIO), HRP-conjugated rabbit anti-goat IgG (1:5000, ZSGB-BIO), for 45 min and washed three times for 10 min each. Finally, the bands were visualized with ECL chemiluminescence detection (Beyotime). The quantity of protein loaded was verified by staining the same membranes with β-actin antibody (1:200, ZSGB-BIO). β-actin was also used to quantitatively normalize the signal.
Immunohistochemistry (IHC)
Archival formalin-fixed paraffin-embedded sections from the 2nd Affiliated Hospital of Dalian Medical University and sections of mice lymph nodes were analyzed. The sections were deparaffinized in xylene and rehydrated with a series of graded ethanol, after blocking, sections were incubated with primary TCP-1 rabbit polyclonal antibody (1:200, Bioworld technology co., BS5959), followed by HRP-conjugated anti-rabbit antibody (1:1000, ZSGB-BIO) for 30 min at 37°C. The sections were finally stained with 3,3′-diaminobenzidine (DAB) and counterstained with haematoxylin. The sections were examined independently by two pathologists who didn’t know the clinicopathological information.
Reverse transcription polymerase chain reaction
Total RNA was isolated from cells each group using TRIzol® reagent (TaKaRa Biotechnology). The sequence specificity primers was designed to amplify the coding sequence of the mouse P2X7R based on the recently published mouse P2X7R cDNA sequence by polymerase chain reaction (PCR). The primers used to detect P2X7R mRNA were as follows: forward primer, 5′-ATATCCACTTCCCCGGCCAC-3′, reverse primer, 5′-TCGGCAGATGGGACCAG-3′. Cycling parameters were: 94°C, 5 min for 1 cycle; 94°C, 30 sec, 57°C, 1 sec, 72°C, 1 min for 35 cycles, with extension at 72°C for 5 min. The primers used for TCP-1 were forward primer, 5′-CCTGTAAGCCTAGCCCTTTG-3′, reverse primer, 5′-CAGGGAGTTGGCTGGATAAT-3′. Cycling parameters were: 95°C, 5 min for 1 cycle; 95°C, 30 sec, 55°C, 45 sec, 72°C, 1 min for 35 cycles, with extension at 72°C for 10 min. When used as a control, the cDNA of the constitutively expressed β-actin gene was always amplified from the same cDNA preparations using specific primers, forward primer: 5′-GGCTGTATTCCCCTCCATCG-3′, reverse primer: 5′-CCAGTTGGTAACAATGCCATGT-3′. The PCR amplicon were separated in a 1.2% agarose (Pronadisa, Spain) gel containing 0.5 μg/ml ethidium bromides and visualized under UV light. Result was analyzed by Gel-Pro32 analyzer software with the values normalized to β-actin levels.
Immunofluorescence analysis
P388D1 cells transfected with P2X7R shRNA and control shRNA were blocked with 10% fetal bovine serum, respectively incubated with P2X7R rabbit polyclonal antibody and TCP-1 goat polyclonal antibody (Santa Cruz Biotechnology), then incubated with goat anti-rabbit IgG-TRITC and rabbit anti-goat IgG-TRITC. The control shRNA group P388D1 cells was used as control.
Statistical analyses
Statistical analyses were performed with GraphPad Prism 6 software. Student’s t-test, Fisher exact, and Kaplan-Meier test were used as appropriate.
ACKNOWLEDGMENTS AND FUNDING
This work was supported by grants from the Chinese State Key Program in Basic Research (2012CB822103), the Chinese National Science Foundation Projects (81372669, 31270867, and 31470800), the Science and Technology Planning Project of Liao Ning province, China (2012225020), and the Project of Chinese Ministry of Health (W2012RQ23).
CONFLICTS OF INTEREST
There is no Conflicts of Interest.
REFERENCES
1. Kubick C, Schmalzing G, Markwardt F. The effect of anions on the human P2X7 receptor. Biochimica et Biophysica Acta (BBA)-Biomembranes. 2011; 1808:2913–2922.
2. Furber M, Alcaraz L, Bent JE, Beyerbach A, Bowers K, Braddock M, Caffrey MV, Cladingboel D, Collington J, Donald DK. Discovery of potent and selective adamantane-based small-molecule P2X7 receptor antagonists/interleukin-1β inhibitors. Journal of medicinal chemistry. 2007; 50:5882–5885.
3. Zhang X-J, Zheng G-G, Ma X-T, Yang Y-H, Li G, Rao Q, Nie K, Wu K-F. Expression of P2X7 in human hematopoietic cell lines and leukemia patients. Leukemia research. 2004; 28:1313–1322.
4. Qiu Y, Li W-h, Zhang H-q, Liu Y, Tian X-X, Fang W-G. P2X7 Mediates ATP-Driven Invasiveness in Prostate Cancer Cells. PloS one. 2014; 9:e114371.
5. Gutiérrez-Martín Y, Bustillo D, Gómez-Villafuertes R, Sánchez-Nogueiro J, Torregrosa-Hetland C, Binz T, Gutiérrez LM, Miras-Portugal MT, Artalejo AR. P2X7 receptors trigger ATP exocytosis and modify secretory vesicle dynamics in neuroblastoma cells. Journal of Biological Chemistry. 2011; 286:11370–11381.
6. Gu L-Q, Li F-Y, Zhao L, Liu Y, Chu Q, Zang X-X, Liu J-M, Ning G, Zhao Y-J. Association of XIAP and P2X7 receptor expression with lymph node metastasis in papillary thyroid carcinoma. Endocrine. 2010; 38:276–282.
7. Adinolfi E, Melchiorri L, Falzoni S, Chiozzi P, Morelli A, Tieghi A, Cuneo A, Castoldi G, Di Virgilio F, Baricordi OR. P2X7 receptor expression in evolutive and indolent forms of chronic B lymphocytic leukemia. Blood. 2002; 99:706–708.
8. Yokota S-i, Yamamoto Y, Shimizu K, Momoi H, Kamikawa T, Yamaoka Y, Yanagi H, Yura T, Kubota H. Increased expression of cytosolic chaperonin CCT in human hepatocellular and colonic carcinoma. Cell stress & chaperones. 2001; 6:345.
9. Coghlin C, Carpenter B, Dundas S, Lawrie L, Telfer C, Murray G. Characterization and over-expression of chaperonin t-complex proteins in colorectal cancer. The Journal of pathology. 2006; 210:351–357.
10. Yam AY, Xia Y, Lin H-TJ, Burlingame A, Gerstein M, Frydman J. Defining the TRiC/CCT interactome links chaperonin function to stabilization of newly made proteins with complex topologies. Nature structural & molecular biology. 2008; 15:1255–1262.
11. Sternlicht H, Farr GW, Sternlicht ML, Driscoll JK, Willison K, Yaffe MB. The t-complex polypeptide 1 complex is a chaperonin for tubulin and actin in vivo. Proceedings of the National Academy of Sciences. 1993; 90:9422–9426.
12. Won K-A, Schumacher RJ, Farr GW, Horwich AL, Reed SI. Maturation of human cyclin E requires the function of eukaryotic chaperonin CCT. Molecular and cellular biology. 1998; 18:7584–7589.
13. Farr GW, Scharl EC, Schumacher RJ, Sondek S, Horwich AL. Chaperonin-mediated folding in the eukaryotic cytosol proceeds through rounds of release of native and nonnative forms. Cell. 1997; 89:927–937.
14. Hansen WJ, Ohh M, Moslehi J, Kondo K, Kaelin WG, Welch WJ. Diverse effects of mutations in exon II of the von Hippel-Lindau (VHL) tumor suppressor gene on the interaction of pVHL with the cytosolic chaperonin and pVHL-dependent ubiquitin ligase activity. Molecular and cellular biology. 2002; 22:1947–1960.
15. Ren S, Zhang Y, Wang Y, Lui Y, Wei W, Huang X, Mao W, Zuo Y. Targeting P2X7 receptor inhibits the metastasis of murine P388D1 lymphoid neoplasm cells to lymph nodes. Cell biology international. 2010; 34:1205–1211.
16. Lay AJ, Jiang X-M, Kisker O, Flynn E, Underwood A, Condron R, Hogg PJ. Phosphoglycerate kinase acts in tumour angiogenesis as a disulphide reductase. nature. 2000; 408:869–873.
17. Hall A. The cytoskeleton and cancer. Cancer and Metastasis Reviews. 2009; 28:5–14.
18. Lee SH, Lee JK, Jin SM, Lee KC, Sohn JH, Chae SW, Kim DH. Expression of cell-cycle regulators (cyclin D1, cyclin E, p27kip1, p57kip2) in papillary thyroid carcinoma. Otolaryngology—Head and Neck Surgery. 2010; 142:332–337.
19. Gou Y, Shi Y, Zhang Y, Nie Y, Wang J, Song J, Jin H, He L, Gao L, Qiao L. Ribosomal protein L6 promotes growth and cell cycle progression through upregulating cyclin E in gastric cancer cells. Biochemical and biophysical research communications. 2010; 393:788–793.
20. Farooq M, Hozzein WN, Elsayed EA, Taha NA, Wadaan M. Identification of histone deacetylase 1 protein complexes in liver cancer cells. Asian Pacific Journal of Cancer Prevention. 2013; 14:915–921.
21. Herzog F, Kahraman A, Boehringer D, Mak R, Bracher A, Walzthoeni T, Leitner A, Beck M, Hartl F-U, Ban N. Structural probing of a protein phosphatase 2A network by chemical cross-linking and mass spectrometry. Science. 2012; 337:1348–1352.
22. Feldman DE, Thulasiraman V, Ferreyra RG, Frydman J. Formation of the VHL-elongin BC tumor suppressor complex is mediated by the chaperonin TRiC. Molecular cell. 1999; 4:1051–1061.
23. Lee S-Y, Jo S, Lee GE, Jeong LS, Kim Y-C, Park C-S. Establishment of an assay for P2X7 receptor-mediated cell death. Molecules and cells. 2006; 22:198–202.
24. Wang Q, Wang L, Feng Y-H, Li X, Zeng R, Gorodeski GI. P2X7 receptor-mediated apoptosis of human cervical epithelial cells. American Journal of Physiology-Cell Physiology. 2004; 287:C1349–C1358.
25. Trubiani O, Horenstein AL, Caciagli F, Caputi S, Malavasi F, Ballerini P. Expression of P2X7 ATP Receptor Mediating the IL8 and CCL20 Release in Human Periodontal Ligament Stem Cells. Journal of cellular biochemistry. 2014; 115:1138–1146.
26. Bulanova E, Budagian V, Orinska Z, Hein M, Petersen F, Thon L, Adam D, Bulfone-Paus S. Extracellular ATP induces cytokine expression and apoptosis through P2X7 receptor in murine mast cells. The Journal of Immunology. 2005; 174:3880–3890.
27. Ferrari D, Chiozzi P, Falzoni S, Dal Susino M, Melchiorri L, Baricordi O, Di Virgilio F. Extracellular ATP triggers IL-1 beta release by activating the purinergic P2Z receptor of human macrophages. The Journal of Immunology. 1997; 159:1451–1458.
28. Ferrari D, Stroh C, Schulze-Osthoff K. P2X7/P2Z purinoreceptor-mediated activation of transcription factor NFAT in microglial cells. Journal of Biological Chemistry. 1999; 274:13205–13210.
29. Kong Q, Wang M, Liao Z, Camden JM, Yu S, Simonyi A, Sun GY, Gonzalez FA, Erb L, Seye CI. P2X7 nucleotide receptors mediate caspase-8/9/3-dependent apoptosis in rat primary cortical neurons. Purinergic signalling. 2005; 1:337–347.
30. Arulkumaran N, Unwin RJ, Tam FW. A potential therapeutic role for P2X7 receptor (P2X7R) antagonists in the treatment of inflammatory diseases. Expert opinion on investigational drugs. 2011; 20:897–915.
31. Christenson K, Björkman L, Tängemo C, Bylund J. Serum amyloid A inhibits apoptosis of human neutrophils via a P2X7-sensitive pathway independent of formyl peptide receptor-like 1. Journal of leukocyte biology. 2008; 83:139–148.
32. Nagaoka I, Tamura H, Hirata M. An antimicrobial cathelicidin peptide, human CAP18/LL-37, suppresses neutrophil apoptosis via the activation of formyl-peptide receptor-like 1 and P2X7. The Journal of Immunology. 2006; 176:3044–3052.
33. Reddy KB, Nabha SM, Atanaskova N. Role of MAP kinase in tumor progression and invasion. Cancer and Metastasis Reviews. 2003; 22:395–403.
34. Wang C-M, Chang Y-Y, Sun SH. Activation of P2X7 purinoceptor-stimulated TGF-β1 mRNA expression involves PKC/MAPK signalling pathway in a rat brain-derived type-2 astrocyte cell line, RBA-2. Cellular signalling. 2003; 15:1129–1137.
35. Gendron F-P, Neary JT, Theiss PM, Sun GY, Gonzalez FA, Weisman GA. Mechanisms of P2X7 receptor-mediated ERK1/2 phosphorylation in human astrocytoma cells. American Journal of Physiology-Cell Physiology. 2003; 284:C571–C581.
36. Yamazaki Y, Kubota H, Nozaki M, Nagata K. Transcriptional Regulation of the Cytosolic Chaperonin θ Subunit Gene, Cctq, by Ets Domain Transcription Factors Elk-1, Sap-1a, and Net in the Absence of Serum Response Factor. Journal of Biological Chemistry. 2003; 278:30642–30651.
37. Wang G, Wang F, Ding W, Wang J, Jing R, Li H, Wang X, Wang Y, Ju S, Wang H. APRIL induces tumorigenesis and metastasis of colorectal cancer cells via activation of the PI3K/Akt pathway. PloS one. 2013; 8:e55298.
38. Jacques-Silva MC, Rodnight R, Lenz G, Liao Z, Kong Q, Tran M, Kang Y, Gonzalez FA, Weisman GA, Neary JT. P2X7 receptors stimulate AKT phosphorylation in astrocytes. British journal of pharmacology. 2004; 141:1106–1117.
39. Abe Y, Yoon S-O, Kubota K, Mendoza MC, Gygi SP, Blenis J. p90 ribosomal S6 kinase and p70 ribosomal S6 kinase link phosphorylation of the eukaryotic chaperonin containing TCP-1 to growth factor, insulin, and nutrient signaling. Journal of Biological Chemistry. 2009; 284:14939–14948.
40. Fu W, McCormick T, Qi X, Luo L, Zhou L, Li X, Wang B-C, Gibbons HE, Abdul-Karim FW, Gorodeski GI. Activation of P2X7-mediated apoptosis inhibits DMBA/TPA-induced formation of skin papillomas and cancer in mice. BMC cancer. 2009; 9:114.
41. Lin Y-F, Tsai W-P, Liu H-G, Liang P-H. Intracellular β-tubulin/chaperonin containing TCP1-β complex serves as a novel chemotherapeutic target against drug-resistant tumors. Cancer research. 2009; 69:6879–6888.

