INTRODUCTION
The management options for patients with brain metastases have improved significantly over time due to effective methods for earlier detection [1], better brain-directed therapies such as combined modality therapies [2], and improvements in systemic chemotherapy [3, 4]. While a number of clinical trials have assessed the role of specific systemic agents for select populations of patients with brain metastases [5, 6], it remains unclear to what degree targeted systemic agents have affected the brain metastasis population as a whole. One population of particular interest is patients who receive stereotactic radiosurgery (SRS) as these patients are selected to have a limited burden of disease and a longer life expectancy. At this time, it is unclear whether the development of newer systemic agents, such as targeted agents, has improved clinical endpoints after SRS for the brain metastasis population as a whole, though evidence has emerged that certain subpopulations may benefit.
One population for which the use of targeted agents has affected clinical outcomes for brain metastases after SRS is patients with renal cell carcinoma. A recent series has shown that targeted agents not only improve overall survival in patients receiving SRS, but also improve upon the local efficacy of SRS on brain metastases [3]. Over the past decade, the use of targeted agents has proliferated for renal cell carcinoma [7–9], breast cancer [10], lung cancer [11], and melanoma [12] with improvements seen in overall survival in patients with metastatic disease. Given these benefits in metastatic disease, one question that emerges is whether or not targeted agent use affects brain metastasis outcomes.
The goal of the current study was to assess whether the promising interactions seen in the renal cell carcinoma population between targeted agents and SRS also exists with other primary tumors that metastasize to the brain. The current series represents one of the largest single institution series of patients treated with SRS for brain metastases. The study particularly aimed to assess the effect of targeted agent therapy on overall survival, local control, and the likelihood of distant brain failure in patients who received SRS for brain metastases, and whether these effects are primary tumor type or histology-specific.
RESULTS
Patient demographics
We identified 737 patients with brain metastases between January 2000 and December 2013. In total, 248 (33%) received targeted agents ever and 489 (67%) did not. A total of 167 patients (23%) received targeted agents either concurrently or within 30 days of SRS. Of these patients, 38 of 102 (37%) breast cancer, 10 of 40 colorectal cancer (25%), 70 of 364 (19%) lung cancer, 20 of 117 (17%) melanoma and 24 of 68 renal cell cancer patients (35%) received targeted agents within 30 days of SRS. Patients receiving targeted agents within 30 days of SRS had a younger age (median 58 vs 63 years, p = 0.002) and greater disease burden (43% vs. 32% widespread disease, p = 0.02) than those who did not. By the time of our analysis, 632 patients (86%) had died.
Overall survival
Patients who received targeted agents within 30 days of SRS had a significant improvement in overall survival (Figure 1). The median overall survival was 7 months for the non-targeted agent use group and 18 months for the targeted agent group. Overall survival for targeted agent vs. non-targeted agent use groups was 90% vs. 55% at 6 months, 65% vs. 30% at 12 months, and 35% vs. 15% at 24 months (log rank p < 0.0001). There was no difference in neurologic death between patients receiving targeted agents within 30 days of SRS and those who did not (32% vs. 30%, respectively).
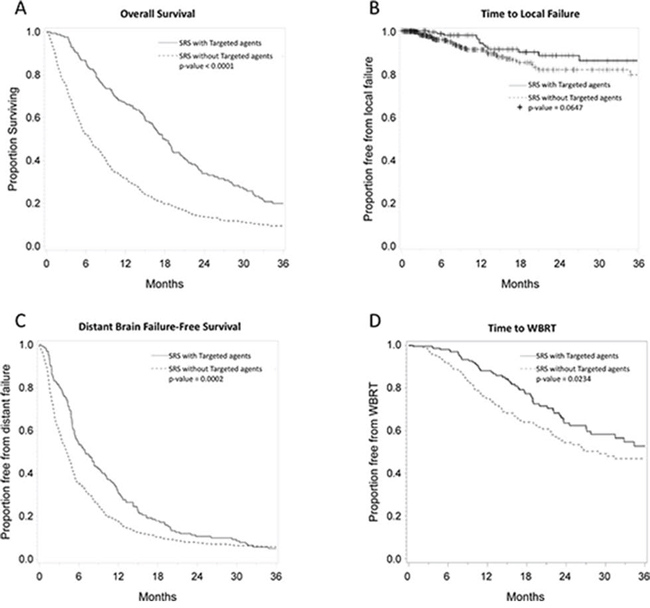
Figure 1: Kaplan Meier plots comparing patients who received targeted agents vs. those who did not in terms of overall survival A. freedom from local failure B. freedom from distant failure C. and freedom from salvage WBRT D.
Patterns of failure
Freedom from local failure for targeted agent vs. non-targeted agent use groups was 99% vs. 96% at 6 months, 94% vs. 91% at 12 months, and 88% vs. 84% at 24 months (log rank p = 0.06) (Figure 1). Distant brain failure-free survival for targeted agent vs. non-targeted agent use groups was 55% vs. 35% at 6 months, 32% vs. 18% at 12 months, and 10% vs. 8% at 24 months (log rank p = 0.0001). Time to salvage WBRT was 98% vs. 92% at 6 months, 88% vs. 77% at 12 months, and 64% vs. 54% at 24 months (log rank p = 0.03).
Primary tumor-specific outcomes
Figures 2, 3, and 4 depict primary tumor-specific rates of overall survival, freedom from local failure, and distant brain failure-free survival for patients receiving targeted agents vs those that did not. For breast cancer brain metastases, patients receiving targeted agents experienced improved overall survival (median 24 months vs. 9 months, p < 0.0001), freedom from local failure (p < 0.01), and distant brain failure-free survival (median 10 vs. 5 months, p = 0.002). For brain metastases from non-small cell lung cancer, Kaplan Meier analysis revealed that patients receiving targeted agents experienced improved overall survival (median 13 vs. 7 months, p = 0.01) and freedom from distant brain failure-free survival (median 7 vs. 5 months, p = 0.048). For renal cell cancer brain metastases, patients receiving targeted agents experienced improved survival (21 vs. 6 months, p = 0.016), freedom from local failure (p = 0.04), but no significant improvement in distant brain failure-free survival rate (5 months for each group). For melanoma brain metastases, patients receiving targeted agents experienced improved survival (18 vs. 5 months, p = 0.009), but no significant improvement in rate of local failure (p = 0.1) or distant failure rate (4 months for each group). For brain metastases from colorectal cancers, patients receiving targeted agents did not experience any statistically significant differences in overall survival, local failure, or distant failure rates.
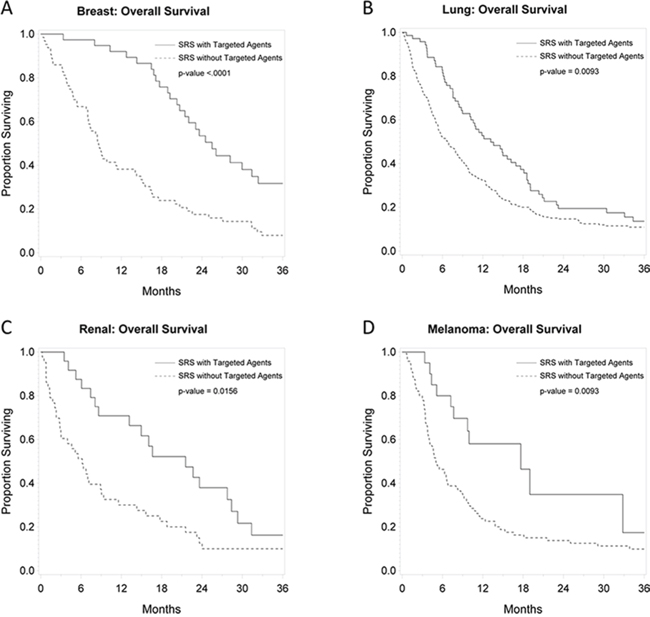
Figure 2: Primary tumor-specific Kaplan Meier plots for overall survival comparing patients who received targeted agents vs. those who did not receive targeted agents concurrently or soon after SRS for breast cancer A. lung cancer B. renal cell cancer C. and melanoma D.
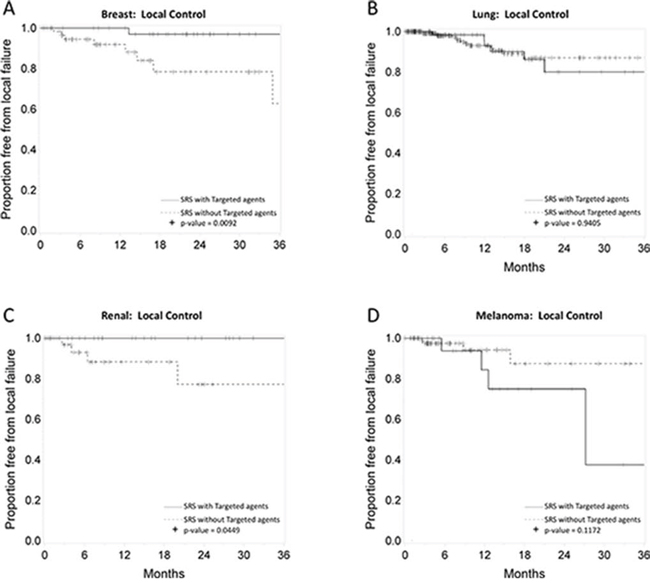
Figure 3: Primary tumor-specific Kaplan Meier plots for freedom from local failure comparing patients who received targeted agents vs. those who did not receive targeted agents concurrently or soon after SRS for breast cancer A. lung cancer B. renal cell cancer C. and melanoma D.
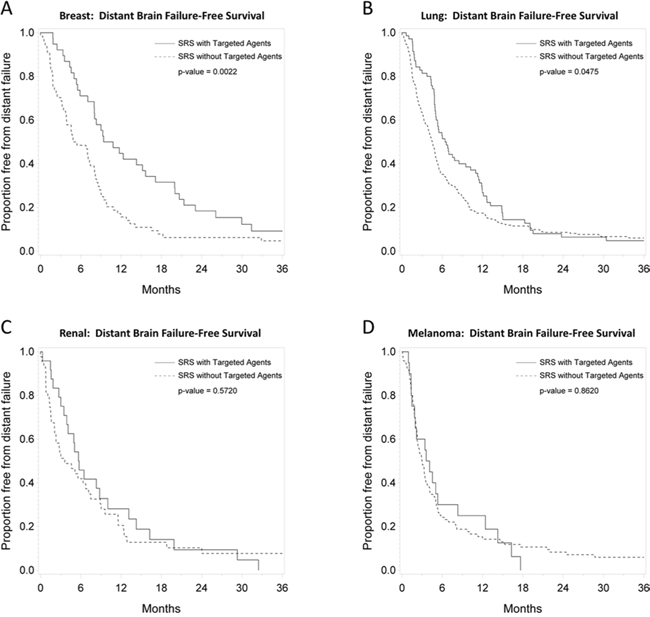
Figure 4: Primary tumor-specific Kaplan Meier plots for distant failure-free survival comparing patients who received targeted agents vs. those who did not receive targeted agents concurrently or soon after SRS for breast cancer A. lung cancer B. renal cell cancer C. and melanoma D.
Multivariate analysis of all-cause mortality
Results of multivariate analysis are shown in Table 1. Post-SRS targeted agent use was associated with significantly decreased hazard for all-cause mortality (p < 0.0001; HR 0.6; 95% CI 0.5–0.7). Increasing numbers of intracranial metastases relative to a solitary metastasis was associated with significantly increased hazard for all-cause mortality. This hazard increase was significant when comparing a solitary metastases with three (p = 0.01; HR 1.4; 95% CI 1.1–1.8), and four or more metastases (p < 0.001; HR 1.6; 95% CI 1.2–2.2). Oligometastatic disease relative to no evidence of disease, widespread disease relative to no evidence of disease, and progressive disease relative to stable disease were all associated with a significantly increased hazard for all-cause mortality (p = 0.04, HR 1.3, 95% CI 1.0–1.7; p = 0.005, HR 1.5, 95% CI 1.1–2.0; p < 0.0001, HR 1.7, 95% CI 1.4–2.0).
Table 1: Multivariate cox proportional hazards model for overall survival a
Covariate |
HR |
95% CI |
P |
Age, 10 yr increase |
1.1 |
1.0, 1.2 |
0.0592 |
GK Treatment year (1 year increase after the year 2000) |
1.0 |
1.0, 1.0 |
0.8778 |
Gender: Women vs. men |
0.9 |
0.8, 1.1 |
0.2291 |
DS-GPA, 1 unit increase |
0.9 |
0.8, 1.0 |
0.0712 |
Number of courses of chemo |
|||
1 vs. 0 |
1.2 |
1.0, 1.6 |
0.1094 |
2 vs. 0 |
1.0 |
0.7, 1.5 |
0.9016 |
3 vs. 0 |
1.1 |
0.8, 1.7 |
0.5055 |
4+ vs. 0 |
1.4 |
0.9, 2.2 |
0.0956 |
Number of intracranial metastasis |
|||
2 vs. 1 |
1.2 |
1.0, 1.5 |
0.0621 |
3 vs. 1 |
1.4 |
1.1, 1.8 |
0.0144 |
4+ vs. 1 |
1.6 |
1.2, 2.2 |
0.0009 |
Disease Burden |
|||
Oligometastatic vs. none |
1.3 |
1.0, 1.7 |
0.0461 |
Widespread vs. none |
1.5 |
1.1, 2.0 |
0.0052 |
Unknown vs. none |
1.0 |
0.7, 1.6 |
0.9190 |
Systemic Disease |
|||
Progressive vs. stable |
1.7 |
1.4, 2.0 |
< 0.0001 |
Unknown vs. stable |
1.2 |
0.9, 1.6 |
0.3151 |
Symptoms: yes vs. no |
1.7 |
1.4, 2.1 |
< 0.0001 |
Neurosurgery: yes vs. no |
0.6 |
0.5, 0.7 |
< 0.0001 |
Targeted agent: yes vs. no |
0.6 |
0.5, 0.7 |
< 0.0001 |
Abbreviations: HR, Hazard ratio; GK, Gamma Knife; DS-GPA, disease-specific Graded Prognostic Assessment; WBRT, Whole Brain Radiotherapy; SRS, Stereotactic Radiosurgery.
aCox model stratified by WBRT (yes/no) and repeat SRS (yes/no)
DISCUSSION
Targeted agents represent a broad class of systemic therapies that inhibit cancer cells by specifically blocking molecular pathways that lead to tumor growth [16]. Because of the specificity of targeted agents and the multiple pathways of carcinogenesis across tumor types, these agents are generally specific to a tumor subtype and/or histological subtype. In the current study, the use of targeted systemic therapy demonstrated a survival advantage in patients with brain metastases across multiple primary tumor types. While a survival advantage is expected, the improvement in distant brain failure-free survival, time to WBRT and particularly in local control has wide clinical implications for the management of brain metastases.
Development of new metastases after SRS and the subsequent salvage treatments can significantly raise the cost of managing a patient with brain metastasis [17]. This is particularly true in patients that require early WBRT since the major advantage of SRS is the ability to avoid WBRT-related toxicty [18]. Several efforts are currently underway to improve patient selection for SRS by attempting to predict which patients will suffer rapid development of new metastases and require early WBRT [19]. Given the results of the current study, it would appear that patients receiving systemic targeted agents are not only less likely to require WBRT, but are also more likely to survive long enough to benefit from the cognitive toxicity-sparing effects of SRS. As such, practitioners may be able to use this factor as part of the selection criteria for patients to receive SRS instead of WBRT.
Morbidity and death from local failure after SRS is an important endpoint. Several series have suggested that some subgroups of brain metastases benefit from concurrent or post-SRS systemic therapy by improving local control following SRS [3, 20]. The current series confirms these previous findings across multiple primary cancers including lung cancer, breast cancer, melanoma, and renal cell cancer, the first time such a finding has been made over multiple histologies. A longstanding oncologic dogma has been that most systemic agents, whether targeted or cytotoxic, do not cross the blood brain barrier at a concentration high enough to lead to a sufficient response for brain metastases. Based on the current study, it appears that systemically administered targeted agents can improve local control of SRS when administered concurrently or soon after SRS. This benefit in local control is analogous to how concurrent chemotherapy increases local control of radiotherapy for patients with such cancers as head and neck cancer [21] or cervical cancer [22] where no blood brain barrier is involved. The possible implications of these findings are that when local control is suboptimal, such as in the case of large metastatic brain tumors, there may be a benefit to post-SRS targeted therapy, even in the absence of active extracranial disease.
There are several mechanisms by which the combination of SRS and targeted agents lead to improved local control in spite of questionable blood brain barrier penetration. First of all, it is thought that one of the reasons that SRS has improved efficacy over fractionated radiation is that it also targets the tumor vasculature [23]. Such targeting may disrupt the blood brain barrier so that drug can penetrate. This theory could explain the several negative prospective studies using targeted agents either as monotherapy [6] or in conjunction with conventionally fractionated whole brain radiotherapy [24]. Another hypothesis is that the increased anti-cancer activity of the combination of SRS and targeted agents may be due to the highly-specific targeting of driver mutations that may not require a high concentration to cause radiosensitization. Finally, a proportion of brain metastases may have pre-existing disruption of the blood brain barrier caused by the cancer so that penetration of targeted agents into brain metastases is possible [25]. Our data indeed showed that while the rate of local failure was significantly improved by the combination of SRS and targeted agents in most cancers, the distant failure rate did not improve universally. Our observations may partly support these hypotheses.
From the current study, it appears that the improvements in brain metastasis outcomes brought about by targeted agents are dependent upon the primary cancer subtype. Breast cancer and renal cell cancer appear to have the greatest benefits in survival compared to other primary cancer subtypes. Median overall survival in each of these populations was greater than 20 months. With regards to local brain failure, only breast cancers and renal cell cancers experienced statistically significant improvements. These primary cancer-specific outcomes imply that clinical studies for the use of targeted agents may require the molecular classification of the tumor type. With molecular classification, specific populations that benefit may then be identified. Another question for future trials will be the question of whether maintenance systemic therapy, given the improvement in distant brain failure-free survival seen across multiple primary cancer subgroups, in the absence of active extracranial disease, may be a worthwhile treatment.
There are several limitations to the current study. While it is among the largest single institution datasets for brain metastases treated with SRS, its retrospective nature limits its interpretation to hypothesis-generation. There is a possibility for patient selection bias as patients with improved performance status may be more likely to receive targeted systemic therapy. Conversely, patients receiving targeted agents in the present study actually had a greater extracranial disease burden (43% vs. 32% widespread disease, p = 0.02) compared to those not receiving targeted agents. The heterogeneity of targeted agents and molecular targets across multiple tumor subtypes did not provide this dataset with sufficient power to stratify the analysis by specific targeted agents. In spite of its limitations, the results of the current study have wide implications for future prospective trials and the use of targeted agents in patients with brain metastases in order to help improve the therapeutic ratio of SRS, and possibly prevent the development of new brain metastases.
MATERIALS AND METHODS
Data source and acquisition
The study cohort was derived from the Wake Forest Gamma Knife database. This database included patients seen between January 2000 and December 2013 (737 patients) who underwent upfront SRS without whole brain radiation treatment (WBRT) for brain metastases. Patients who had previously received WBRT for brain metastases were excluded from the database since this treatment affects outcomes being measured in the study. Patients with brain metastases from sarcoma, ovarian cancer and head and neck cancers were also excluded because these patients represented such a small minority that the numbers were insufficient yield meaningful statistical conclusions. This study was approved by the Wake Forest School of Medicine Institutional Review Board and patient characteristics and treatment outcomes were determined using patients’ electronic medical records. Patient pre-treatment characteristics are shown in Table 2.
Table 2: Baseline characteristics
No. of Patients (%) |
|||
Characteristic |
SRS without Targeted Agents N = 570 |
SRS with Targeted Agents N = 167 |
P |
Age at treatment |
|||
Median (Min, Max) |
63.0 (5.0, 91.0) |
58.0 (21.0, 87.0) |
0.002* |
Gender |
0.17 |
||
Women |
256 (44.9%) |
85 (50.9%) |
|
Men |
314 (55.1%) |
82 (49.1%) |
|
Primary Site of Brain Metastasis |
0.0002 |
||
Lung |
294 (51.6%) |
70 (41.9%) |
|
Breast |
64 (11.2%) |
38 (22.8%) |
|
Renal/RCC |
44 (7.7%) |
24 (14.4%) |
|
Melanoma |
97 (17.0%) |
20 (12.0%) |
|
Colon |
30 (5.3%) |
10 (6.0%) |
|
Esophagus |
15 (2.6%) |
2 (1.2%) |
|
Other |
26 (4.6%) |
3 (1.8%) |
|
Histology |
< 0.0001 |
||
Adenocarcinoma |
213 (37.4%) |
62 (37.1%) |
|
Squamous cell |
54 (9.5%) |
8 (4.8%) |
|
Adenosquamous |
5 (0.9%) |
0 (0.0%) |
|
Large cell NE |
7 (1.2%) |
0 (0.0%) |
|
Non-small cell lung NOS |
47 (8.2%) |
9 (5.4%) |
|
Her2-positive |
18 (3.2%) |
28 (16.8%) |
|
Her2-negative |
38 (6.7%) |
10 (6.0%) |
|
Breast other |
7 (1.2%) |
0 (0.0%) |
|
RCC |
44 (7.7%) |
24 (14.4%) |
|
Melanoma |
97 (17.0%) |
20 (12.0%) |
|
Other |
40 (7.0%) |
6 (3.6%) |
|
Number of Brain Metastases |
0.23 |
||
1 |
299 (52.5%) |
74 (44.3%) |
|
2 |
129 (22.6%) |
40 (24.0%) |
|
3 |
70 (12.3%) |
24 (14.4%) |
|
4 + |
72 (12.6%) |
29 (17.4%) |
|
Disease Burdena |
0.02 |
||
None |
98 (17.2%) |
31 (18.6%) |
|
Oligometastatic |
251 (44.0%) |
54 (32.3%) |
|
Widespread |
180 (31.6%) |
72 (43.1%) |
|
Unknown |
41 (7.2%) |
10 (6.0%) |
|
Extracranial Disease |
0.16 |
||
Stable |
298 (52.4%) |
101 (60.5%) |
|
Progressive |
210 (36.9%) |
53 (31.7%) |
|
Unknown |
61 (10.7%) |
13 (7.8%) |
|
DS-GPA mean (SD) |
2.0 (0.9) |
2.0 (0.9) |
0.92 |
Number of courses of chemo |
< 0.00001 |
||
0 |
442 (77.5%) |
104 (62.2%) |
|
1 |
86 (15.1%) |
14 (8.3%) |
|
2 |
20 (3.5%) |
17 (10.2%) |
|
3 |
13 (2.3%) |
17 (10.2%) |
|
4 + |
9 (1.6%) |
15 (8.9%) |
|
Margin Dose |
|||
Median (IQR) |
18.0 (17.0, 22.0) |
20.0 (18.0, 21.0) |
*0.2067 |
SRS Treatment Date mean |
Feb 2008 |
May 2009 |
< 0.0001 |
Abbreviations: IQR, Interquartile range; RCC, Renal Cell Carcinoma; Large Cell NE, Large cell neuroendocrine; NOS, not otherwise specified; DS-GPA, Disease Specific Graded Prognostic Assessment.
aDisease burden was defined as none, unknown, oligometastatic (≤ 5 extracranial metastases), or widespread (≥ 5 extracranial metastases).
Patient factors including age, histology, disease-specific Graded Prognostic Assessment (ds-GPA), status of extracranial disease, and number of prior lines of systemic therapy were all determined from the electronic medical records. The ds-GPA class was defined as previously reported by Sperduto et al. [13]. The status of extracranial was categorized as “none”, “stable” or “progressive”. The extent of extracranial disease was characterized as none, oligometastatic, or widespread. Oligometastatic disease was defined as ≤ 5 non-brain metastases without diffuse involvement of any one organ. Widespread metastatic disease included patients with > 5 metastases or diffuse distant organ involvement.
Endpoint definitions
Patients were followed clinically and with MRI at 4–8 weeks after primary radiosurgery. If there were no sign of treatment failure at this interval, clinical evaluation and MRI were conducted approximately every 3 months. Local failure was defined as tumor recurrence within the prior radiosurgical treatment volume. Local failure was determined via surgical pathology or imaging evidence of a 25% increase in area of enhancement on an axial MRI slice along with increased perfusion on perfusion-weighted imaging. Local failures were treated with surgical resection, whole brain irradiation, or observation depending on patient health status, the status of extracranial cancer, and physician discretion. Distant brain failure was defined as a new metastasis on follow-up imaging found outside the initial radiosurgical treatment volume. Distant brain failures were generally treated with further SRS, and WBRT was generally reserved for 5 + total brain metastases over time or short-interval distant failures. Neurological death was defined in the same manner as Patchell et al. [14].
Radiosurgical technique
Patients were treated with Leksell Model B, C, or Perfexion units (Elekta AB). Prior to radiosurgery, patients underwent a high-resolution MRI of the brain. Treatment planning was performed using the Leksell GammaPlan Treatment Planning System (Elekta AB). A median dose of 20 Gy prescribed to the 50% isodose line was prescribed. Prescription dose was determined based on the guidelines previously published by Shaw et al. [15].
Use of targeted agents
Targeted agents were generally used at the discretion of the treating medical oncologist. A targeted agent was defined as a systemic drug that inhibits a specific pathway(s) known to drive cancer growth. Selection of targeted agents by the medical oncologists was based on standard treatment algorithms specific to the cancer type of each patient. For example, agents targeting the human epidermal growth factor receptor (HER2), del 19, or L858R activating mutations in EGFR and the anaplastic lymphoma kinase pathway were used in these specific subgroups of breast and non-small cell lung cancer, respectively. BRAF inhibitors were utilized in melanoma as well as ipilumumab (Yervoy), which targets CTLA-4, a protein receptor that downregulates the immune system. Targeted agents utilized for renal cell carcinoma included tyrosine kinase inhibitors, mTOR inhibitors, or bevacizumab (Avastin). Wild type colorectal cancer patients often received agents that targeted EGFR such as cetuximab (Erbitux). Cytotoxic chemotherapeutic agents, those that more indiscriminately kill rapidly dividing cells, were not considered targeted agents in this study. Examples of cytotoxic chemotherapy include doxorubicin (Adriamycin) for breast cancer, pemetrexed (Alimta) for lung cancer, temozolomide (Temodar) for melanoma and capecitabine (Xeloda) for colorectal cancer.
Statistics
Descriptive statistics were generated for the sample (n = 737) by targeted agent status. Patients were assigned to the cohort receiving targeted agents if they received a targeted agent either concurrently with SRS or within 1 month after completion of SRS. Differences between targeted agent status were determined using chi-squared tests for categorical characteristics and Kruskal-Wallis tests for age and margin dose due to the skewed distributions of these continuous measures. Kaplan-Meier plots and log-rank tests were used to compare the targeted agent status for overall survival, time to local failure, time to distant failure, and time to WBRT. These plots and tests were done on the whole sample as well as by primary site. A Cox proportional hazards model was created for the overall survival outcome using the predictors. From this model, hazard ratios, 95 percent confidence intervals were estimated. An alpha level of 0.05 was used to determine significance for all tests. All analyses were conducted using SAS 9.4 (SAS Institute, Cary, NC).
CONCLUSIONS
Targeted agent use after SRS appears to significantly improve overall survival, local control, and the likelihood of distant brain failure. If these findings are prospectively validated, they would potentially provide new indications for targeted agent use in the setting of brain metastases receiving SRS.
ACKNOWLEDGMENTS
None.
FUNDING
This research was supported by the following grant: P30 CA012197-40. There are no financial disclosures from any authors.
REFERENCES
1. Loganathan AG, Chan MD, Alphonse N, Peiffer AM, Johnson AJ, McMullen KP, Urbanic JJ, Saconn PA, Bourland JD, Munley MT, Shaw EG, Tatter SB, Ellis TL. Clinical outcomes of brain metastases treated with Gamma Knife radiosurgery with 3.0 T versus 1.5 T MRI-based treatment planning: have we finally optimised detection of occult brain metastases? J Med Imaging Radiat Oncol. 2012; 56:554–560.
2. Jensen CA, Chan MD, McCoy TP, Bourland JD, deGuzman AF, Ellis TL, Ekstrand KE, McMullen KP, Munley MT, Shaw EG, Urbanic JJ, Tatter SB. Cavity-directed radiosurgery as adjuvant therapy after resection of a brain metastasis. J Neurosurg. 2011; 114:1585–1591.
3. Cochran DC, Chan MD, Aklilu M, Lovato JF, Alphonse NK, Bourland JD, Urbanic JJ, McMullen KP, Shaw EG, Tatter SB, Ellis TL. The effect of targeted agents on outcomes in patients with brain metastases from renal cell carcinoma treated with Gamma Knife surgery. J Neurosurg. 2012; 116:978–983.
4. Vern-Gross TZ, Lawrence JA, Case LD, McMullen KP, Bourland JD, Metheny-Barlow LJ, Ellis TL, Tatter SB, Shaw EG, Urbanic JJ, Chan MD. Breast cancer subtype affects patterns of failure of brain metastases after treatment with stereotactic radiosurgery. J Neurooncol. 2012; 110:381–388.
5. Lee SM, Lewanski CR, Counsell N, Ottensmeier C, Bates A, Patel N, Wadsworth C, Ngai Y, Hackshaw A, Faivre-Finn C. Randomized trial of erlotinib plus whole-brain radiotherapy for NSCLC patients with multiple brain metastases. J Natl Cancer Inst. 2014; 106.
6. Lin NU, Carey LA, Liu MC, Younger J, Come SE, Ewend M, Harris GJ, Bullitt E, Van den Abbeele AD, Henson JW, Li X, Gelman R, Burstein HJ, Kasparian E, Kirsch DG, Crawford A, et al. Phase II trial of lapatinib for brain metastases in patients with human epidermal growth factor receptor 2-positive breast cancer. J Clin Oncol. 2008; 26:1993–1999.
7. Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M, Negrier S, Chevreau C, Solska E, Desai AA, Rolland F, Demkow T, Hutson TE, Gore M, Freeman S, Schwartz B, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med. 2007; 356:125–134.
8. Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Rixe O, Oudard S, Negrier S, Szczylik C, Kim ST, Chen I, Bycott PW, Baum CM, Figlin RA. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007; 356:115–124.
9. Hudes G, Carducci M, Tomczak P, Dutcher J, Figlin R, Kapoor A, Staroslawska E, Sosman J, McDermott D, Bodrogi I, Kovacevic Z, Lesovoy V, Schmidt-Wolf IG, Barbarash O, Gokmen E, O’Toole T, et al. Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med. 2007; 356:2271–2281.
10. Park YH, Park MJ, Ji SH, Yi SY, Lim DH, Nam DH, Lee JI, Park W, Choi DH, Huh SJ, Ahn JS, Kang WK, Park K, Im YH. Trastuzumab treatment improves brain metastasis outcomes through control and durable prolongation of systemic extracranial disease in HER2-overexpressing breast cancer patients. Br J Cancer. 2009; 100:894–900.
11. Sekine A, Satoh H, Iwasawa T, Tamura K, Hayashihara K, Saito T, Kato T, Arai M, Okudela K, Ohashi K, Ogura T. Prognostic factors for brain metastases from non-small cell lung cancer with EGFR mutation: influence of stable extracranial disease and erlotinib therapy. Med Oncol. 2014; 31:228.
12. Chapman PB, Hauschild A, Robert C, Haanen JB, Ascierto P, Larkin J, Dummer R, Garbe C, Testori A, Maio M, Hogg D, Lorigan P, Lebbe C, Jouary T, Schadendorf D, Ribas A, et al. Improved Survival with Vemurafenib in Melanoma with BRAF V600E Mutation. N Engl J Med. 2012; 364:2507–2516.
13. Sperduto PW, Kased N, Roberge D, Xu Z, Shanley R, Luo X, Sneed PK, Chao ST, Weil RJ, Suh J, Bhatt A, Jensen AW, Brown PD, Shih HA, Kirkpatrick J, Gaspar LE, et al. Summary report on the graded prognostic assessment: an accurate and facile diagnosis-specific tool to estimate survival for patients with brain metastases. J Clin Oncol. 2012; 30:419–425.
14. Patchell RA, Tibbs PA, Regine WF, Dempsey RJ, Mohiuddin M, Kryscio RJ, Markesbery WR, Foon KA, Young B. Postoperative radiotherapy in the treatment of single metastases to the brain: a randomized trial. JAMA. 1998; 280:1485–1489.
15. Shaw E, Scott C, Souhami L, Dinapoli R, Kline R, Loeffler J, Farnan N. Single dose radiosurgical treatment of recurrent previously irradiated primary brain tumors and brain metastases: final report of RTOG protocol 90–05. Int J Radiat Oncol Biol Phys. 2000; 47:291–298.
16. Sledge GW, Jr. What is targeted therapy? J Clin Oncol. 2005; 23:1614–1615.
17. Lester SC, Taksler GB, Kuremsky JG, Lucas JT, Jr., Ayala-Peacock DN, Randolph DM, 2nd Bourland JD, Laxton AW, Tatter SB, Chan MD. Clinical and economic outcomes of patients with brain metastases based on symptoms: an argument for routine brain screening of those treated with upfront radiosurgery. Cancer. 2014; 120:433–441.
18. Greene-Schloesser D, Robbins ME, Peiffer AM, Shaw EG, Wheeler KT, Chan MD. Radiation-induced brain injury: A review. Front Oncol. 2012; 2:73.
19. Ayala-Peacock DN, Peiffer AM, Lucas JT, Isom S, Kuremsky JG, Urbanic JJ, Bourland JD, Laxton AW, Tatter SB, Shaw EG, Chan MD. A nomogram for predicting distant brain failure in patients treated with gamma knife stereotactic radiosurgery without whole brain radiotherapy. Neuro Oncol. 2014; 16:1283–1288.
20. Harris S, Chan MD, Lovato JF, Ellis TL, Tatter SB, Bourland JD, Munley MT, deGuzman AF, Shaw EG, Urbanic JJ, McMullen KP. Gamma knife stereotactic radiosurgery as salvage therapy after failure of whole-brain radiotherapy in patients with small-cell lung cancer. Int J Radiat Oncol Biol Phys. 2012; 83:e53–59.
21. Cooper JS, Pajak TF, Forastiere AA, Jacobs J, Campbell BH, Saxman SB, Kish JA, Kim HE, Cmelak AJ, Rotman M, Machtay M, Ensley JF, Chao KS, Schultz CJ, Lee N, Fu KK. Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck. N Engl J Med. 2004; 350:1937–1944.
22. Morris M, Eifel PJ, Lu J, Grigsby PW, Levenback C, Stevens RE, Rotman M, Gershenson DM, Mutch DG. Pelvic radiation with concurrent chemotherapy compared with pelvic and para-aortic radiation for high-risk cervical cancer. N Engl J Med. 1999; 340:1137–1143.
23. Truman JP, Garcia-Barros M, Kaag M, Hambardzumyan D, Stancevic B, Chan M, Fuks Z, Kolesnick R, Haimovitz-Friedman A. Endothelial membrane remodeling is obligate for anti-angiogenic radiosensitization during tumor radiosurgery. PLoS One. 2010; 5.
24. Pesce GA, Klingbiel D, Ribi K, Zouhair A, von Moos R, Schlaeppi M, Caspar CB, Fischer N, Anchisi S, Peters S, Cathomas R, Bernhard J, Kotrubczik NM, D’Addario G, Pilop C, Weber DC, et al. Outcome, quality of life and cognitive function of patients with brain metastases from non-small cell lung cancer treated with whole brain radiotherapy combined with gefitinib or temozolomide. A randomised phase II trial of the Swiss Group for Clinical Cancer Research (SAKK 70/03). Eur J Cancer. 2012; 48:377–384.
25. Yonemori K, Tsuta K, Ono M, Shimizu C, Hirakawa A, Hasegawa T, Hatanaka Y, Narita Y, Shibui S, Fujiwara Y. Disruption of the blood brain barrier by brain metastases of triple-negative and basal-type breast cancer but not HER2/neu-positive breast cancer. Cancer. 2010; 116:302–308.

