Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has led to more severe outcomes of patients afflicted with cancer [1]. Cancer development has been a controversial area as a potential long-term effect of SARS-CoV-2 infection [2]. Previous studies have shown that SARS-CoV-2 proteins could increase breast and lung cancer cell proliferation, migration and invasion [3, 4]. There is a need to investigate and better understand whether SARS-CoV-2 may be involved in any way with cancer signaling pathways, molecular mechanisms of cancer development or has effects on therapy.
SARS-CoV-2 spike protein is a key mediator for virus infection of host cells though its two subunits: S1, which binds to human angiotensin-converting enzyme 2 (ACE2), and S2, which mediates a membrane fusion process [5]. The S2 subunit contains multiple domains that facilitate protein-protein interaction. Therefore, the S2 subunit is a factor to explore functional effects of SARS-CoV-2 spike protein in host cells after virus entry.
An in-silico analysis using HADDOCK 2.2 software previously suggested that p53 and BRCA1/2 may interact with the heptic repeat-2 region of the S2 subunit through C-terminal domain [6]. DNA damage or therapy-induced tumor suppressor p53 protein transcriptionally activates genes leading to multiple effects preserving genome integrity, altering metabolism, immune response, cell cycle, DNA repair, cell growth and cell apoptosis to prevent or eliminate transformed cells [7]. Loss of p53 function increases the incidence of carcinogen-induced tumorigenesis and drives chemo-resistance [8]. SARS-CoV-2 infection has been found to alter p53 stabilization. The previous studies have shown that SARS-CoV-2 spike in particular plays a role to stabilize and activate p53 by mediating cell-cell fusion or induction of ROS in host cells during SARS-CoV-2 virus infection [9, 10]. In response to cellular stress, activated p53 regulates specific gene expression, including MDM2 (E3 ligase). MDM2, in turn, binds to p53 and triggers p53 ubiquitination and proteasomal degradation [11], while interruption of MDM2-p53 interaction leads to p53 stabilization. Thus, a putative interaction between SARS-CoV-2 spike, p53 and p53 related signaling pathways following SARS-CoV-2 infection could impact cellular homeostasis, tumorigenic pathways, and/or response to cancer therapeutics.
In this study, we performed cell-based assays to examine the effect of SARS-CoV-2 on p53 activation in cancer cells and demonstrate that SARS-CoV-2 spike interrupts the MDM2-p53 interaction in cancer cells and alters p53 signaling in cancer cells upon chemotherapy including blunted activation of p53 targets involved in growth arrest and apoptosis.
Results
SARS-CoV2-spike overexpression shows reduced p53 interaction with MDM2 in cancer cells
To investigate the interaction between SARS-CoV-2 spike, p53, and MDM2 proteins in cancer cells, we performed an immunoprecipitation assay. The pcDNA3.1-SARS2-spike plasmid was introduced to overexpress SARS-CoV-2 spike protein containing the S2 subunit in cancer cells [12]. The plasmids p-CMV-Neo-Bam-p53wt, pcDNA3.1-SARS2-spike and p-EGFP-MDM2 were co-transfected into p53-knockout U2OS (U2OS-p53KO) cancer cells using lipofectamine.
The immunoprecipitation assay showed that MDM2 protein bound with p53 in the cells while cells with SARS-CoV-2 spike overexpression displayed reduced amounts of MDM2 bound with p53 when compared to the pcDNA3.1 transfection control (Figure 1A). These results suggest that SARS-CoV-2 spike overexpression can alter p53 binding with MDM2 in cancer cells.
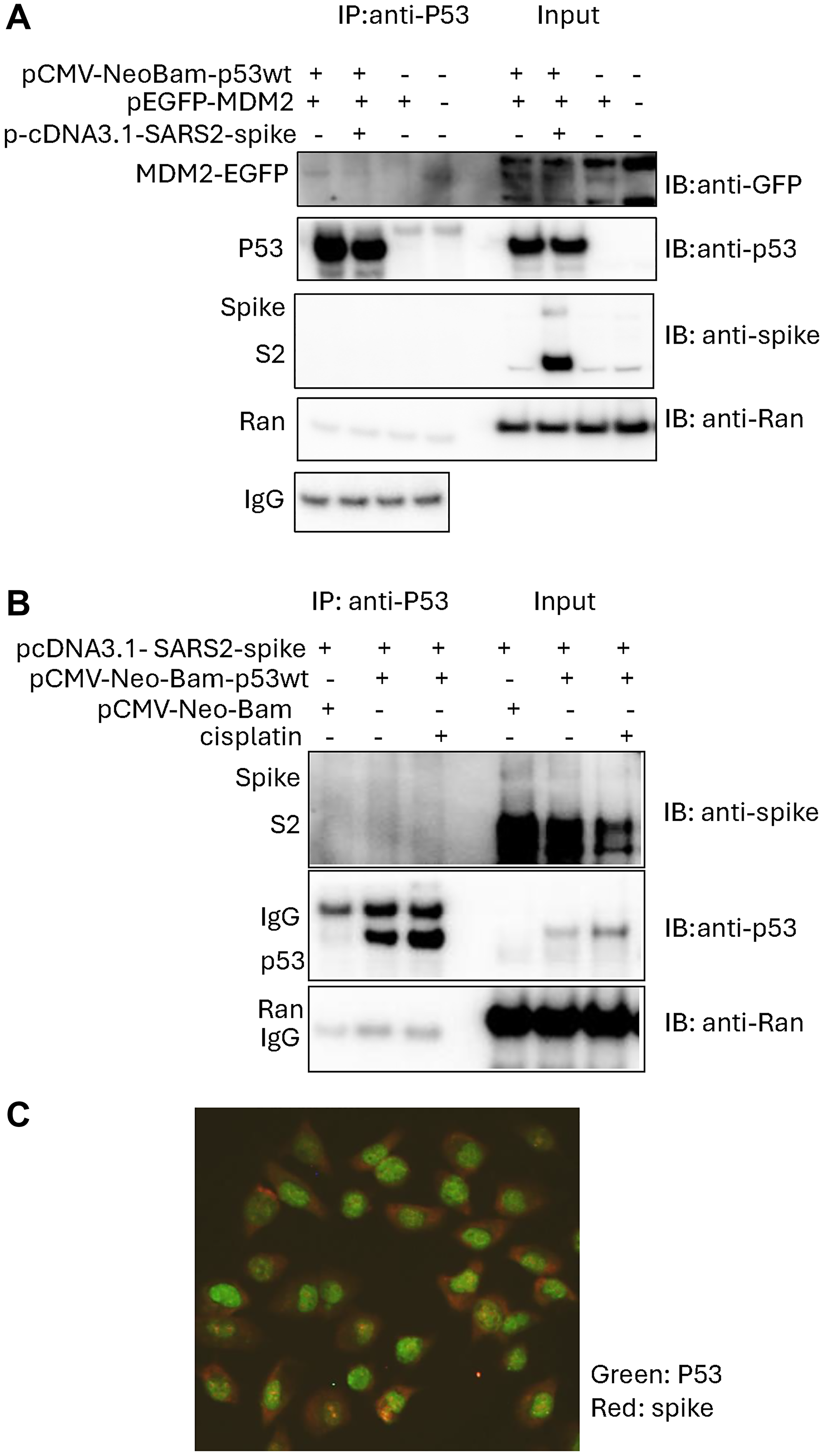
Figure 1: Reduced interaction between p53 and MDM2 following SARS-CoV-2 spike protein overexpression in cancer cells.
(A) Immunoprecipitation (IP) assay in U2OS-p53KO cancer cells. Cells were transiently transfected with the plasmids as indicated. The related empty vectors as controls were included. IP was performed with anti-p53 (DO-1) and IB with anti-p53 (FL393). (B) Immunoprecipitation in p53 wild-type breast cancer cells. MCF7 cancer cells were treated with cisplatin for 4 hours. IP was performed with anti-p53 (DO-1). (C) Immunofluorescence imaging the cellular locations of the SARS-CoV-2 spike and p53. H460 cancer cells were treated with 3 μM of cisplatin for 24 hours.
However, SARS-CoV-2 spike S2 subunit was not observed to bind with p53 protein in the immunoprecipitation assay (Figure 1A), nor did it have any detectable impact when p53 was activated by treatment with cisplatin, a DNA damaging agent that causes interstrand crosslinks (Figure 1B).
Our observations from lack of co-immunoprecipitation between p53 and the SARS-CoV-2 spike S2 protein subunit are consistent with different cellular locations of SARS-CoV-2 spike and p53 in the cancer cells treated with cisplatin (Figure 1C). The immunofluorescence imaging showed that the majority of p53 was localized in the nuclei, while the majority of SARS-CoV-2 spike was localized in the cytoplasm in H460 cells treated with cisplatin (Figure 1C). These results do not demonstrate SARS-CoV-2-spike S2 subunit protein binding to wild-type p53 in cancer cells either in the absence or presence of cisplatin treatment. As this is a protein subunit, and some small amount of nuclear staining is observed, we cannot exclude that spike subunits can gain access to the nucleus or that intact spike could do so as well.
SARS-CoV2 spike attenuates p53 transcriptional activity in cancer cells
To investigate the effect of the SARS-CoV2 spike on p53 signaling in cancer cells further, we conducted a PG13-luciferase (PG13-Luc) reporter assay. PG13-luc contains 13 copies of the p53-binding consensus sequence, and the production of PG13-luc was confirmed by using bioluminescence in cells [13]. The p53-null HCT116 and U2OS-p53KO cancer cells were transiently transfected with PG13-luc together with p-CMV-Neo-Bam-p53wt and pcDNA3.1-SARS2-spike for 20 hours. The cells with pcDNA3.1-SARS2-spike transfection showed reduction of the p53 responsive bioluminescence, as compared to the pcDNA3.1 transfection control (Figure 2A). Further treatment with nutlin-3a, an MDM2 inhibitor which activates p53 signaling, was ineffective at rescuing the reduction of the p53 responsive bioluminescence of PG13-Luc (Figure 2B).
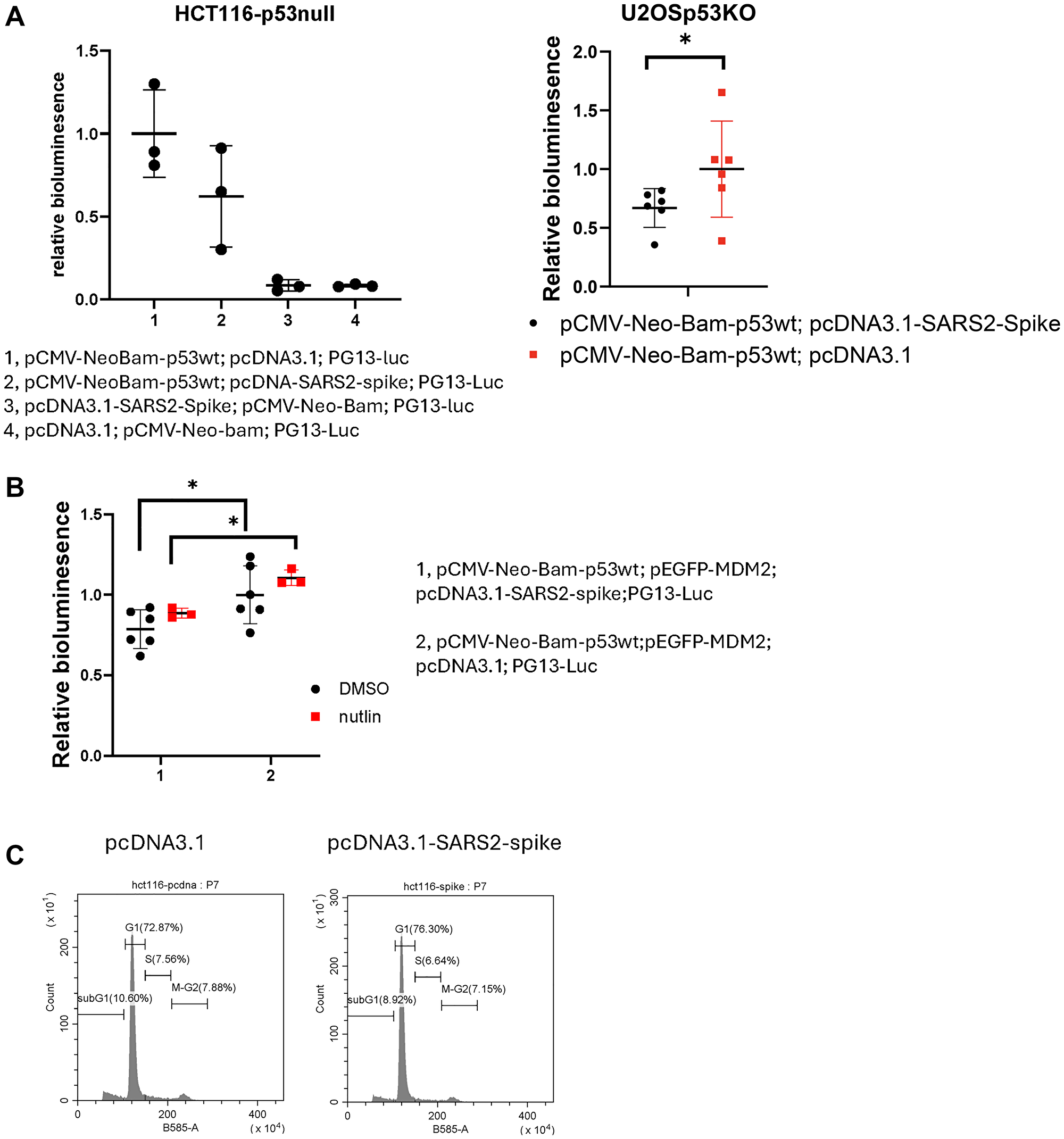
Figure 2: SARS-CoV-2 spike suppresses p53-mediated transcriptional activity in cancer cells.
(A) PG13-luc reporter assay in cancer cells. HCT116 p53-null cells and U2OS-p53KO were transiently transfected with p-CMV-Neo-Bam-p53wt and PG13-luc together with the plasmids as indicated for 20 hours. (B) PG13-luc reporter assay. U2OS-p53KO cells were transfected with the plasmids as indicated, followed with 10 μM of nutlin-3a treatment for 4 hours. (C) Cell cycle profiling in HCT116 cells transiently transfected with the plasmids as indicated for 72 hours.
The suppressive effect of spike on p53 responsive bioluminescence was also detected in a variety of p53 wild-type cancer cell lines by either transient transfection or stable expression of PG-13Luc (Figure 3A, 3B). These results suggest that SARS-CoV-2 spike protein attenuates p53 transcriptional activity. In a preliminary experiment following up on the inhibition of p53 activity by SARS-CoV-2 spike protein, no cell cycle arrest was detected at G1, S or G2-M phases in cancer cells transfected with pcDNA3.1-SARS2-spike, as compared to the pcDNA3.1 transfection control (Figure 2C).
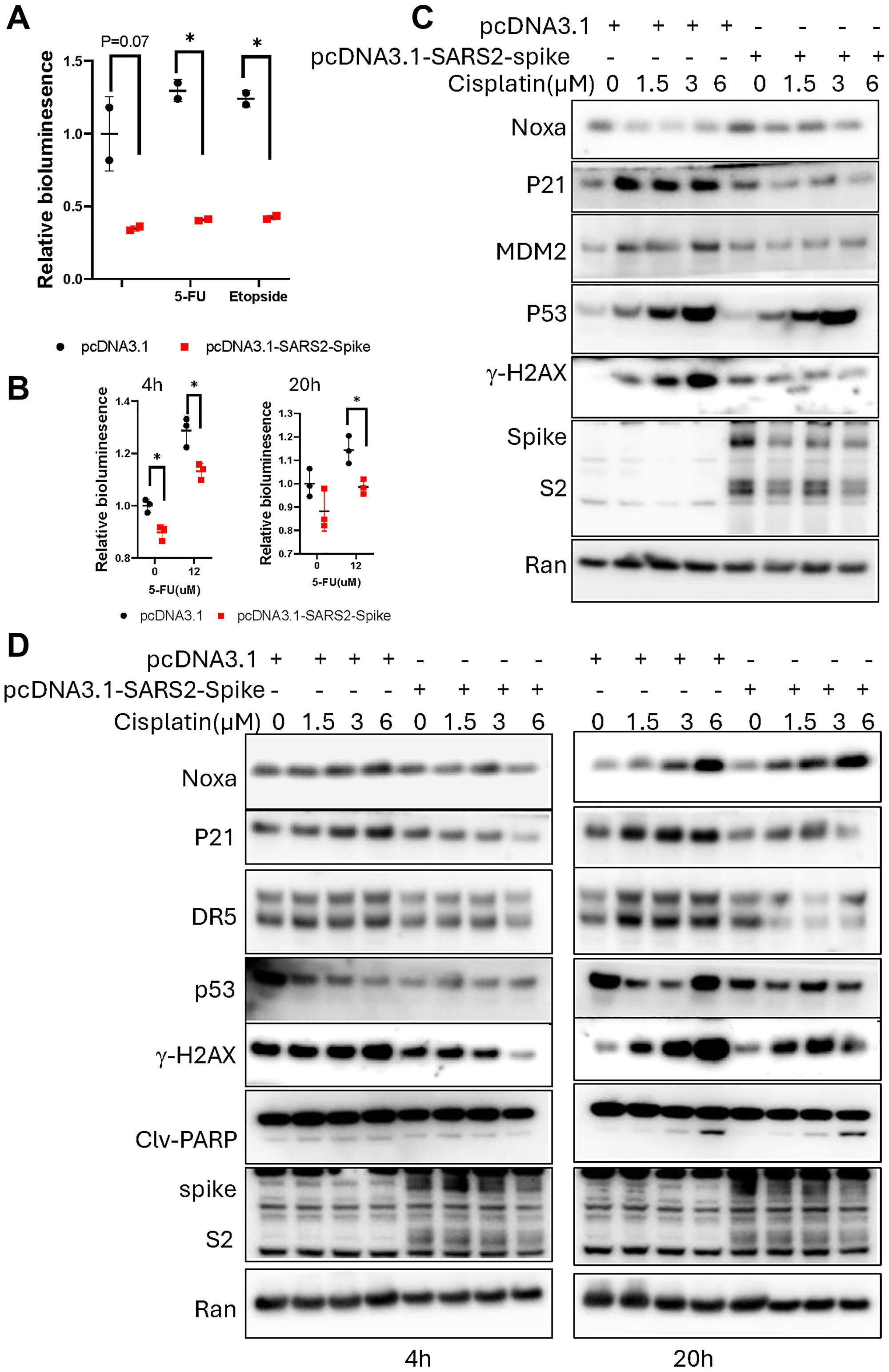
Figure 3: SARS-CoV-2 spike abrogates chemotherapy-induced p53-mediated activation of p21(WAF1), TRAIL Death Receptor DR5 and MDM2 in cancer cells.
(A) PG13-luc reporter assay in cancer cells. Breast cancer MCF7 cells were transiently transfected with pcDNA3.1-SARS2-spike and PG13-luc, followed by exposure to 100 μM of etoposide or 200 μM of 5-FU for 20 hours. (B) PG13-luc reporter assay in HCT116-PG13-luc stable cells. HCT116-PG13-Luc cells were transiently transfected with the pcDNA-SARS2-spike, followed by 12 μM 5-FU treatment for 4 and 20 hours. (C) Protein analysis of p53 targets in MCF7 cells. The cells were transiently transfected with pcDNA3.1-SARS2-spike, followed by cisplatin treatment for 20 hours. (D) Protein analysis of the p53 targets in H460 cells. The cells were transiently transfected with pcDNA3.1-SARS2-spike, followed by cisplatin treatment for 4 and 20 hours.
SARS-CoV-2 spike reduces p53 upregulation of p21(WAF1) and TRAIL Death Receptor DR5 proteins as well as γ-H2AX levels after cisplatin treatment in cancer cells
We further investigated if the SARS-CoV-2 spike protein can inhibit chemotherapy-induced p53 transcriptional activity in cancer cells. p53 wild-type breast cancer cells MCF7 and lung cancer cells H460 were transiently transfected with the pcDNA3.1-SARS2-spike and PG13-luc, followed by etoposide or 5-Fluorouracil (5-FU) treatment. The PG13-luciferase reporter assay showed a reduction of the p53-responsive bioluminescence in the cells transfected with pcDNA3.1-SARS2-spike, as compared to the pcDNA3.1 transfection control (Figure 3A). A similar reduction of p53-responsive transcription-mediated bioluminescence was also observed in p53 wild-type HCT116-PG13-luc cells which have been stably transduced with PG13-luc (Figure 3B). These results suggest that SARS-CoV-2 spike reduces chemotherapy-induced p53 transcriptional activity in cancer cells.
We further examined endogenous p53 targets at the protein level in cancer cells upon cisplatin treatment. Consistent with the reduction of the p53 responsive bioluminescence, a decrease or delay in the p53 transcriptional targets, p21, TRAIL Death Receptor DR5 and MDM2 at the protein level was detected in cancer cells transfected with the pcDNA-SARS2-spike, as compared to the pcDNA3.1 transfection at different post-treatment time points (Figure 3C, 3D). These results suggest that SARS-CoV-2-spike alters chemotherapy-induced p53 signaling in cancer cells of pathways involved in growth arrest and cell death.
p53 is involved in DNA damage response as well as repair [14]. We examined the DNA damage repair response by analyzing γ-H2AX level in cancer cells. The cisplatin treatment caused an increase of γ-H2AX at the protein level in the cancer cells (Figure 3C, 3D), indicating DNA damage repair response in p53 wild-type cancer cells. The levels of the γ-H2AX were reduced in the cohort of the cells transfected with pcDNA3.1-SARS2-spike, as compared to the pcDNA3.1 transfection control (Figure 3C, 3D). These results suggest that the SARS-CoV-2 spike causes an altered DNA damage sensing and repair response in cancer cells.
We further examined the effect of SARS-CoV-2 spike protein on cell growth and death in cancer cells upon chemotherapy treatment. H460 lung cancer cells were transiently transfected with pcDNA3.1-SARS2-spike, followed by cisplatin treatment for 40 hours. The cisplatin treatment increased the PARP cleavage, a death marker, in the H460 cells with pcDNA3.1 transfection at the tested doses (Figure 4A). A similar PARP cleavage was also observed in H460 cells transfected with pcDNA3.1-SARS2-spike. An increase in cell viability was observed in H460 cells transfected with pcDNA-SARS-CoV-2 spike (Figure 4B) at the tested doses of cisplatin, as compared to pcDNA-3.1 transfection.
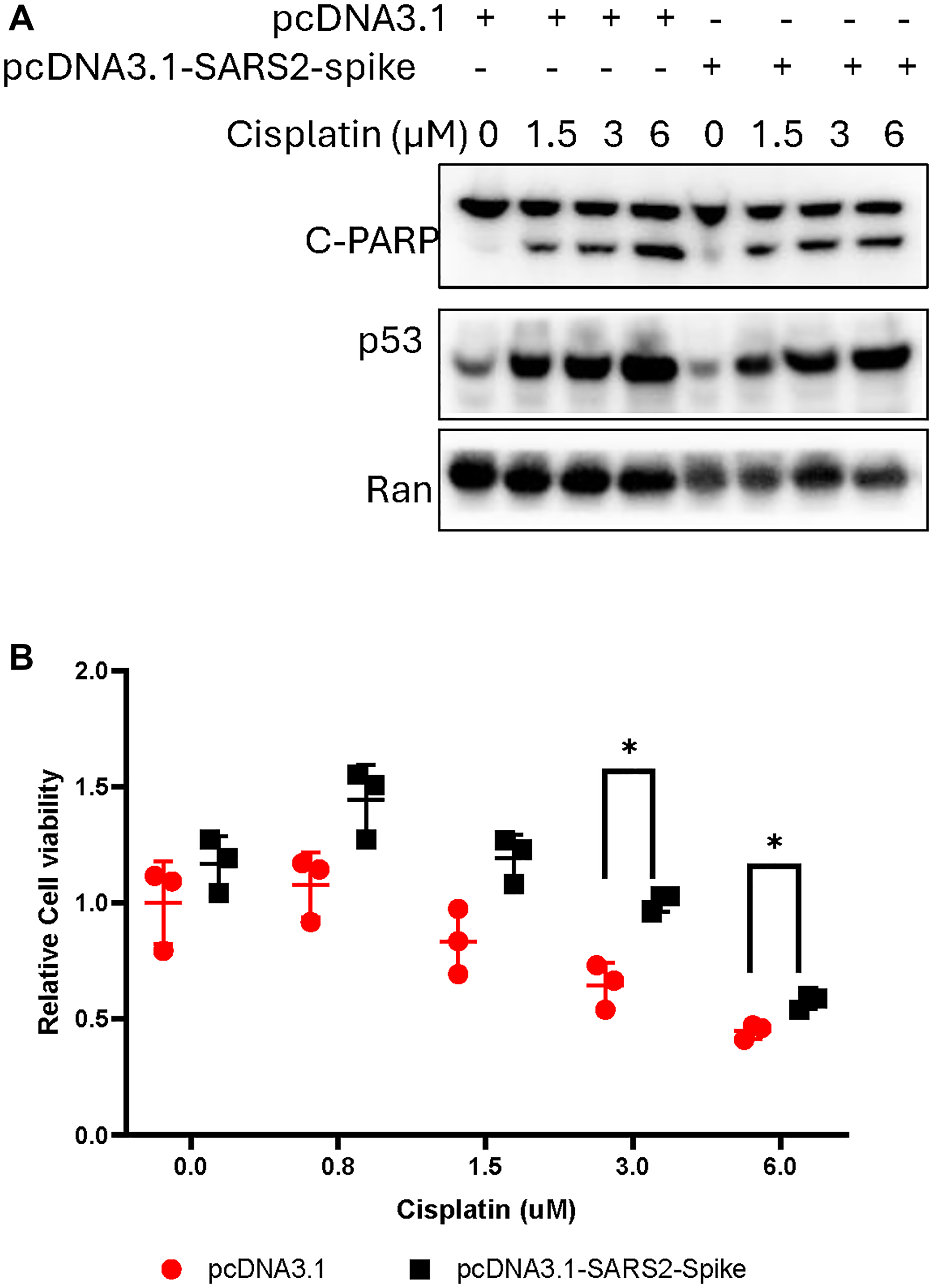
Figure 4: The effect of SARS-CoV-2 spike on cell growth and death.
The cancer cells were transiently transfected with pcDNA-SARS2-spike, followed by cisplatin treatment. (A) Protein analysis of PARP cleavage in H460 cancer cells. The cells were transfected with pcDNA-SARS2-spike, followed with cisplatin treatment for 40 hours. (B) Cell viability assay. H460 cells were transfected with pcDNA-SARS2-spike, followed with cisplatin treatment for 72 hours.
DISCUSSION
The SARS-CoV-2 spike protein plays a key role in SARS-CoV-2 invasion of human cells through spike binding to receptor angiotensin-converting enzyme 2 (ACE2) on the human host cell surface. We show here that spike can alter p53 transcriptional activity in wild-type p53-expressing cancer cells based on reduction of the p53-responsive reporter activity and a decrease in selected p53 targets such as p21(WAF1) or TRAIL Death Receptor DR5 at the protein level.
Our findings differ from previous reports that have shown that SARS-CoV-2 spike stabilized p53 and activated p53 [9, 10]. In the previous studies, the p53 activation and stabilization was caused by the spike-ACE2 mediated cell-cell fusion and an increase in ROS in cancer or normal cells [9, 10].
The reasons for the differences are unclear although the Zhang et al. [10] paper used retinal pigment epithelial cells that were treated with actual recombinant spike protein rather than transfection of spike-expressing plasmid. Lee et al. [9] studied fusogenicity and syncytia formation in SARS-CoV-2 infected cultures and found increased p53 and p21 proteins and a p53/p21-dependent senescence phenotype as well as altered chromatin accessibility states. It is somewhat odd however in the experiment in A549ACE2 TP53ko cells (Figure 5B in Reference 9) both p53 and p21 completely disappear. While the lack of p53 expression is explained by the p53 knockout, the lack of p21 (CDKN1A; WAF1) protein expression in the TP53ko A549ACE2 cells is surprising given that the protein is regulated by numerous p53-independent pathways. It remains unclear whether differences in experimental systems, cell lines, method of exposure to spike explain the differences or whether other factors are involved.
To study the effect of spike on p53 activity as would occur after virus entry into host cells, we transiently transfected SARS-CoV-2 spike into cancer cells thereby avoiding possible effects of a virus infection or cell-cell fusion on p53 signaling. Our findings agree with previous results showing that spike could not stabilize p53 in low ACE2 cells that are unable to undergo cell-cell fusion [9].
The p53 protein is considered as a “genome guardian” by arresting the cell cycle to repair DNA damage or causing cell death in the presence of unrepaired persistent damage and stress [14]. In our preliminary experiments reported here, the SARS-CoV-2 spike-induced inactivation of p53 was correlated to an apparent reduction of expression of DNA damage response protein γ-H2AX after cisplatin exposure and a reduced cell cycle checkpoint response in the cancer cells (Figure 3). Whether these changes are a consequence from the suppressive effect of SARS-CoV-2 spike on p53 signaling in cancer cells needs to be further investigated.
SARS-CoV-2 spike has been found to regulate multiple signaling pathways [15]. The post-translational modifications in p53 including phosphorylation and/or acetylation generally result in activation of p53 through different signaling pathways [16]. It remains unclear whether SARS-CoV-2 spike protein subunits alter p53 binding to the DNA promoters of the targets, or whether SARS-CoV-2 spike inhibits p53 transcriptional activity through post-translational modifications in p53 or other alterations in proteins that complex with p53.
We found that SARS-CoV-2 spike interrupts p53-MDM2 interaction in cancer cells in the absence of exposure to DNA damaging agents. These results were observed by an immunoprecipitation assay using exogenously overexpressed p53 and MDM2 in cancer cells. The effect of SARS-CoV-2 spike on endogenous p53 binding to MDM2 needs to be investigated in more detail in the future. Nevertheless, our observation provides a possible molecular mechanism by which SARS-CoV-2 spike mediates p53 stabilization caused by cell-cell fusion.
We did not observe SARS-CoV-2 spike S2 binding with p53 in cancer cells using immunoprecipitation assay as previously predicted through an in-silico study showing that the S2 subunit of SARS-CoV-2 spike may interact with p53 [6]. In our study, we performed an immunoprecipitation assay in two cancer cell lines transiently transfected with the plasmids carrying SARS-CoV-2 spike. The cell conditions in this study are not the same as SARS-CoV-2 infections that cause syncytia formation. SARS-CoV-2 causes severe disease in multiple organs, and severe effects on cell function that might confer specific post-translational modifications (PTMs). Specific PTMs on spike might modulate host factor binding [17]. It is possible that spike protein with some potential PTMs might interact with p53 under some cellular conditions that were not simulated in our experiments. We note and have no current understanding of the increased γ-H2AX observed on western blot at basal conditions with spike S2 in MCF7 in absence of cisplatin treatment (Figure 3C). This increased basal γ-H2AX expression in spike-expressing cells was not observed in H460 cells at two different time-points (Figure 3D). Future studies will need to determine the veracity of the MCF7 result and its meaning if correct.
While spike appeared to attenuate the induction of p21(WAF1), TRAIL Death Receptor DR5 and MDM2 after DNA damage, there was less of an apparent effect on pro-apoptotic Noxa in our preliminary studies. However, under the experimental conditions, we did not observe more PARP cleavage at the time points evaluated. With cisplatin treatment there was greater cell viability when spike was overexpressed. Additional work needs to determine long-term effects on cell viability, effects on cell survival proteins in chemotherapy-treated spike S2 or SARS-CoV-2 infected cells, in addition to more detailed studies of the DNA damage and repair effects of spike S2. For the latter studies, host cell reactivation assays could be performed with reporters treated with cisplatin either in a tube before transfection or following transfection of human cells with or without spike.
We have not conducted in vivo experiments and some of our experiments lack additional controls such as in flow analysis or by looking at kinetics of cell cycle checkpoint regulation. We have not evaluated normal cells such as airway, muscle, immune, brain or intestinal cells. Cycling vs. quiescent cells are also important to investigate for potential differential effects of spike or other SARS-CoV-2 proteins. We have not investigated immune cell interactions such as NK or T-cells in our experiments where spike protein was overexpressed in culture. These would all be reasonable early future directions.
Our results have implications for the biological effects of spike subunits in human cells whether spike is present due to primary COVID-19 infection or due to mRNA vaccines where its expression is used to promote anti-viral immunity. A perturbed p53 pathway is concerning but also complicated in sorting out since cellular transformation and cancer are a multi-step process that evolves over time. Further detailed studies can more fully characterize the effects of spike, as well as structural determinants within the protein for interaction between the DNA damage sensing and response pathways as well as the p53 tumor suppressing pathway. With respect to the p53 pathway, further studies are needed to unravel how less MDM2 is bound to p53 in the presence of spike and the mechanisms underlying reduced p21(WAF1), TRAIL Death Receptor DR5 as well as MDM2 under conditions where there is less degradation of p53 due to reduced interaction with MDM2.
P21(WAF1) and TRAIL Death Receptor DR5 levels often go up in stress and through many pathways but here we see lack of induction. Why the p53 response is blunted in the presence of spike sub-units remains an open question. The effect of spike on γ-H2AX can be interpreted in a different way too although one would not expect either less damage or accelerated DNA repair with cisplatin. Our interpretation is reduced DNA damage sensing and response after cisplatin exposure in the presence of spike proteins subunits but that remains to be further unraveled. One can investigate the ordered events in the DNA damage sensing and response in different cellular backgrounds including repair-deficient cells such as BRCA mutation or other cancer susceptibility states (ATM, mismatch repair, Fanconi, PTEN, Wnt/beta-catenin/APC, Rb, etc.). Future studies can investigate effects of spike on other cancer pathways, oncogenic or tumor suppressive as well as at a broader range of therapeutic efficacies in the presence of spike or other SARS-CoV-2 encoded proteins. As there is already a history of various viruses associated with human cancer including hepatitis viruses HBV/HCV, EBV, HPV, and potentially SV40, SARS-CoV-2 is a candidate that should be further investigated.
Our experiments highlight an issue that should have earlier consideration more widely in vaccine development. This is about testing numerous natural-variants as well as synthetic-variants of proteins such as spike to maximize anti-viral immunity while minimizing suppression of innate host pathways that respond to invading viruses or their gene products. Such work can be done using recombinant DNA technologies and standard molecular and cellular biology techniques with proteins such as spike or other viral antigens in isolation, in simpler screening systems, and away from viral infection that may have risks from gain-of-function research (or confounding effects due to the stress of viral infection or cell fusion that occurs with SARS-CoV-2). The pathways include the host innate and adaptive immune response as well as host proteins that may be targeted for inactivation including the protective p53 tumor suppressor protein. Manufacturers should be expected to provide such evidence in their development process and this should be publicly available in the future.
In the current manuscript we show preliminary evidence limited to viral protein spike from SARS-CoV-2 impacting on p53 function by inhibiting its transcriptional activation of key genes that mediate its functions in tumor suppression. This occurs in the presence of chemotherapy in spike-expressing cells and associates with greater tumor cell survival. More efforts should be directed at studying those effects as well as the effects of other SARS-CoV-2 viral proteins on host DNA damage sensing, response and repair mechanisms. Such directions are relevant in the context of viral infection as well as mRNA vaccines in the case of spike including for patients with cancer who may be receiving cytotoxic or other cancer treatments. It is clear that the vaccines reduce risk of severe disease among vulnerable elderly individuals or those with co-morbidities, and are strongly recommended by public health officials. It would be helpful to know more about some other risks including long-term risks from foreign antigens that may target host tumor suppressive pathways or that may impact on the efficacy of chemotherapy. Such information would be helpful to patients and physicians as they weigh the risks and benefits of certain medical interventions.
In summary, we identified the SARS-CoV-2 spike as a COVID-19 virus factor that interrupts p53 binding to MDM2 in cancer cells and demonstrated the suppressive effect of SARS-CoV-2 spike on p53 signaling in cancer cells. Correlated to the inhibition of p53 signaling, the short-term expression of spike caused an altered DNA damage response through altered levels of γ-H2AX after DNA damage in cells, altered sensing in the damage response to cisplatin Importantly, the p53-dependent DNA damage induction of growth arrest and apoptotic targets p21(WAF1) and TRAIL Death Receptor DR5 was significantly attenuated under different experimental conditions with spike and this was associated with greater cell viability in the presence of spike and chemotherapy treatment. As loss of p53 function is a known driver of cancer development and confers chemo-resistance, our study provides insight into cellular mechanisms by which SARS-CoV-2 spike may be involved in reducing barriers to tumorigenesis during and post SARS-CoV-2 infections.
Our findings have implications for the natural history of prolonged or repeated SARS-CoV-2 infection as well as design of anti-COVID-19 vaccines that are administered repeatedly as booster shots. Further studies are needed to unravel and clarify issues raised to minimize various risks.
Materials and Methods
Cell culture
Human cell lines used in this study include human lung cancer cells H460 (ATCC), breast cancer cells MCF7 (ATCC), colorectal cancer cells HCT116 (p53 wild-type or p53-null) and sarcoma cells U2OS with p53-knockout (U2OS-P53KO). HCT116 and U2OS-P53KO cells were cultured in McCoy’s 5A (modified) medium, while H460 cells were cultured in RPMI-1640 medium and MCF7 cells were cultured in Eagle’s Minimum Essential Medium. All cell line media were supplemented with 10% FBS and 1% Penicillin-Streptomycin. The cell lines were authenticated and tested to ensure the cultures were free of mycoplasma infection. H460 (lung cancer), MCF7 (breast cancer), HCT116 (colon cancer), and U2OS (osteosarcoma) are the most commonly studied wild-type p53-expressing human tumor cell lines across tumor types.
Plasmid transfection
The plasmids pcDNA3.1-SARS2-spike (#145032) and p-CMV-Neo-Bam-p53wt (#16434) were obtained from Addgene. The plasmids were transiently transfected with lipofectamine 2000 (Life Technologies, catalog no. 11668-027) into cancer cells as stated in the figures.
Luciferase reporter assay
Cancer cells were transfected with an equal amount of each plasmid as indicated in the figures. The PG13-luciferase reporter expression in cancer cells was examined based on bioluminescence using the IVIS imaging system (PerkinElmer, Hopkin, MA, USA) at different time points as indicated in the figures.
Western blot analysis
Cells were seeded with the same density on culture plates and were lysed in loading buffer (Sigma-Aldrich, St. Louis, MO, USA). Equal amounts of cell lysates were electrophoresed through 4–12% SDS-PAGE, then transferred to PVDF membranes. The transferred PVDF membranes were blocked with 5% skim milk at room temperature, then incubated with primary antibodies incubated in a blocking buffer at 4°C overnight. Antibody binding was detected on PVDF with appropriate HRP-conjugated secondary antibodies by a Syngene PXi imaging system (Syngene). Anti-sars-spike antibodies (NB100-56578) were purchased from NOVUS Biologicals, and anti-p53 (DO-1, catalog no. sc-126), anti-MDM2 (SMP14, catalog no. sc-965), and anti-p53 (FL-393) antibodies were purchased from Santa Cruz Biotechnology. Anti-p21 (Ab-1, catalog no. OP64) and anti-Noxa (catalog no. OP180) antibodies were purchased from EMD Millipore. Anti-cleaved PARP (catalog no. 9546) and γ-H2AX (catalog no. 2577) antibodies were purchased from Cell Signaling Technology.
Immunoprecipitation
Cell lysates were incubated with 2 μg of anti-p53 antibody (DO-1) overnight at 4°C; then, they were mixed with 50 μL of nProtein A Sepharose 4 Fast Flow (Cytiva, catalog no. 17528001) for 3 hours at 4°C and washed with lysis buffer three times. The immunoprecipitated proteins were eluted from the nProtein A-Sepharose beads by boiling with 2× sample buffer (Invitrogen, catalog no. NP0007) and subjected to SDS-PAGE.
CellTiter-Glo luminescent cell viability assay
Cell viability was measured by CellTiter-Glo bioluminescence (Promega, Catalog No. G7572), and analyzed using an IVIS imager.
Statistical analysis
The statistical significance of differences between pairs was determined using Student’s t-tests with GraphPad Prism. The minimal level of significance was P < 0.05.
ACKNOWLEDGMENTS
W.S.E-D. is an American Cancer Society Research Professor and is supported by the Mencoff Family University Professorship at Brown University.
CONFLICTS OF INTEREST
W.S.E-D. is a co-founder of Oncoceutics, Inc., a subsidiary of Chimerix, p53-Therapeutics, Inc. and SMURF-Therapeutics, Inc. Dr. El-Deiry has disclosed his relationships and potential conflicts of interest to his academic institution/employer and is fully compliant with NIH and institutional policy that is managing these potential conflicts of interest.
FUNDING
This work began early during the COVID pandemic when it was supported by a Brown University pilot grant. Subsequent experiments were made possible by discretionary start-up funds to Dr. El-Deiry at Brown University.
References
1. Robilotti EV, Babady NE, Mead PA, Rolling T, Perez-Johnston R, Bernardes M, Bogler Y, Caldararo M, Figueroa CJ, Glickman MS, Joanow A, Kaltsas A, Lee YJ, et al. Determinants of Severity in Cancer Patients with COVID-19 Illness. medRxiv. 2020. https://doi.org/10.1101/2020.05.04.20086322.
2. Jahankhani K, Ahangari F, Adcock IM, Mortaz E. Possible cancer-causing capacity of COVID-19: Is SARS-CoV-2 an oncogenic agent? Biochimie. 2023; 213:130–38. https://doi.org/10.1016/j.biochi.2023.05.014. [PubMed].
3. Kim MJ, Kim JY, Shin JH, Son J, Kang Y, Jeong SK, Kim DH, Kim KH, Chun E, Lee KY. The SARS-CoV-2 spike protein induces lung cancer migration and invasion in a TLR2-dependent manner. Cancer Commun (Lond). 2024; 44:273–77. https://doi.org/10.1002/cac2.12485. [PubMed].
4. Nguyen HT, Kawahara M, Vuong CK, Fukushige M, Yamashita T, Ohneda O. SARS-CoV-2 M Protein Facilitates Malignant Transformation of Breast Cancer Cells. Front Oncol. 2022; 12:923467. https://doi.org/10.3389/fonc.2022.923467. [PubMed].
5. Xia S, Liu M, Wang C, Xu W, Lan Q, Feng S, Qi F, Bao L, Du L, Liu S, Qin C, Sun F, Shi Z, et al. Inhibition of SARS-CoV-2 (previously 2019-nCoV) infection by a highly potent pan-coronavirus fusion inhibitor targeting its spike protein that harbors a high capacity to mediate membrane fusion. Cell Res. 2020; 30:343–55. https://doi.org/10.1038/s41422-020-0305-x. [PubMed].
6. Singh N, Bharara Singh A. S2 subunit of SARS-nCoV-2 interacts with tumor suppressor protein p53 and BRCA: an in silico study. Transl Oncol. 2020; 13:100814. https://doi.org/10.1016/j.tranon.2020.100814. [PubMed].
7. Kastenhuber ER, Lowe SW. Putting p53 in Context. Cell. 2017; 170:1062–78. https://doi.org/10.1016/j.cell.2017.08.028. [PubMed].
8. Zhang S, Carlsen L, Hernandez Borrero L, Seyhan AA, Tian X, El-Deiry WS. Advanced Strategies for Therapeutic Targeting of Wild-Type and Mutant p53 in Cancer. Biomolecules. 2022; 12:548. https://doi.org/10.3390/biom12040548. [PubMed].
9. Lee JD, Menasche BL, Mavrikaki M, Uyemura MM, Hong SM, Kozlova N, Wei J, Alfajaro MM, Filler RB, Müller A, Saxena T, Posey RR, Cheung P, et al. Differences in syncytia formation by SARS-CoV-2 variants modify host chromatin accessibility and cellular senescence via TP53. Cell Rep. 2023; 42:113478. https://doi.org/10.1016/j.celrep.2023.113478. [PubMed].
10. Zhang Y, Peng X, Xue M, Liu J, Shang G, Jiang M, Chen D, Liu B, Wang Y, Jia X, Xu J, Zhang F, Hu Y. SARS-COV-2 spike protein promotes RPE cell senescence via the ROS/P53/P21 pathway. Biogerontology. 2023; 24:813–27. https://doi.org/10.1007/s10522-023-10019-0. [PubMed].
11. Chène P. Inhibiting the p53-MDM2 interaction: an important target for cancer therapy. Nat Rev Cancer. 2003; 3:102–9. https://doi.org/10.1038/nrc991. [PubMed].
12. Shang J, Ye G, Shi K, Wan Y, Luo C, Aihara H, Geng Q, Auerbach A, Li F. Structural basis of receptor recognition by SARS-CoV-2. Nature. 2020; 581:221–24. https://doi.org/10.1038/s41586-020-2179-y. [PubMed].
13. el-Deiry WS, Tokino T, Velculescu VE, Levy DB, Parsons R, Trent JM, Lin D, Mercer WE, Kinzler KW, Vogelstein B. WAF1, a potential mediator of p53 tumor suppression. Cell. 1993; 75:817–25. https://doi.org/10.1016/0092-8674(93)90500-p. [PubMed].
14. Williams AB, Schumacher B. p53 in the DNA-Damage-Repair Process. Cold Spring Harb Perspect Med. 2016; 6:a026070. https://doi.org/10.1101/cshperspect.a026070. [PubMed].
15. Suzuki YJ, Gychka SG. SARS-CoV-2 Spike Protein Elicits Cell Signaling in Human Host Cells: Implications for Possible Consequences of COVID-19 Vaccines. Vaccines (Basel). 2021; 9:36. https://doi.org/10.3390/vaccines9010036. [PubMed].
16. Dai C, Gu W. p53 post-translational modification: deregulated in tumorigenesis. Trends Mol Med. 2010; 16:528–36. https://doi.org/10.1016/j.molmed.2010.09.002. [PubMed].
17. Liang B, Zhu Y, Shi W, Ni C, Tan B, Tang S. SARS-CoV-2 Spike Protein Post-Translational Modification Landscape and Its Impact on Protein Structure and Function via Computational Prediction. Research (Wash D C). 2023; 6:0078. https://doi.org/10.34133/research.0078. [PubMed].

