Introduction
Basal cell and squamous cell (keratinocyte-derived) skin cancers are extremely common. It is estimated that the incidence is 3.3–5.4 million patients each year in the U.S [1]. Due to their frequency, these cancers are not reportable in the NCI Surveillance, Epidemiology, and End Results (SEER) cancer registry. The incidence of keratinocyte-derived skin cancers has increased steadily over decades [1].
Cutaneous squamous cell carcinoma (CSCC) represents approximately 20% of keratinocyte-derived skin cancers with an estimated 3–7% of CSCC patients developing recurrent invasive, regionally metastatic, or distant metastatic disease [2]. Precise data concerning the incidence of keratinocyte-derived skin cancers, as well as the number of patients with more advanced stage disease and their outcome are not readily available. Karia estimated that in 2012 between 5604 to 12,572 CSCC patients developed nodal metastasis (~4% of estimated CSCC patients), resulting in 3932 to 8791 deaths (~1.5% mortality) [3].
In general, CSCC is predominantly a disease of elderly, fair skinned, heavily sun-exposed Caucasians. Known risk factors for aggressive CSCC behavior include tumor-related factors, such as head and neck primary sites, indistinct infiltrative borders of the lesion, rapid growth, tumor diameter >2 cm, invasion to >2.0 mm depth, and perineural extension [2]. Host factors that are thought to predict more aggressive behavior include immunosuppression (e.g., organ transplantation, co-morbid conditions such as HIV, indolent lymphomas and CLL) and tumor recurrence after previous surgery or radiotherapy [4]. A number of staging systems have been defined to try to identify high risk individuals for more aggressive therapy and close monitoring [5]. These include the American Joint Commission for Cancer (AJCC), Union for International Cancer Control (UICC) and the Brigham and Women’s Hospital staging systems [6, 7]. In the Brigham and Women’s staging system, T2b or T3 tumors appear to be high-risk and have a greater than 20% risk of lymph node involvement [8, 9].
Treatment options for laCSCC remain poorly defined. In the past, after failure of the initial resection, radiotherapy with or without added chemotherapy was frequently utilized [2, 10, 11]. Since the average advanced CSCC patient is elderly, often with significant comorbid medical conditions, aggressive chemotherapy proved to be poorly tolerated.
The epidermal growth factor receptor (EGFR) is overexpressed in virtually all squamous cell skin cancers [12]. As single agents, EGFR inhibitor monoclonal antibodies cetuximab and panitumumab have shown modest efficacy in phase 2 clinical trials in patients with metastatic cutaneous squamous-cell carcinoma, but durable responses have been uncommon [13–16]. There is, however, an extensive literature concerning treatment squamous cell carcinoma of the head and neck (SCCHN) showing that the EGFR directed monoclonal antibody cetuximab plus radiotherapy has significant activity in this EGFR-driven cancer.
Since there was no established standard therapy for laCSCC patients in 2014, we began treating patients with bulky unresectable tumors with cetuximab and concurrent radiotherapy due to their potential synergy. This was reasonable because most of our patients had primary tumors involving head and neck primary sites. It should be noted, that while synergy of cetuximab plus radiotherapy has been demonstrated in SCCHN, formal evaluation of additive or synergistic benefit in CSCC has not yet been established versus radiotherapy alone. More recently, PD-1 directed monoclonal antibodies such as cemiplimab and pembrolizumab have received FDA approval for treatment of recurrent, unresectable, or metastatic CSCC [17, 18]. Unfortunately, even today, many patients are not candidates for treatment with PD-1 antibodies, due to organ transplants and underlying autoimmune conditions [19]. Thus, additional treatment options are needed.
We performed a retrospectively review of treatment outcome and toxicity in our patients who received concurrent cetuximab and radiotherapy to show an additional potentially effective treatment option for patients with laCSCC. The goal is also to provide data to inform the design of potential prospective clinical trials.
Results
Eighteen eligible patients were identified. Patient characteristics are shown (Table 1). Of the 18 patients identified, 16 were male (88.8%) and 2 were female (11.1%). The median age of patients was 76 ± 11 years (SD) with an age range of 47–92 years. Seventeen patients were Caucasian and 1 was African American.
Table 1: Patient characteristics and treatment
| UPN | Age | Sex | Race | Primary Site | B&W Stage | Comorbidities | Doses cetuximab | Cetuximab duration (months) | Fx size (cGy) | # Fx | Total RT Dose (cGy) | Elapsed time (days) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 60 | M | C | Cheek | T2b, N0 | Lung transplant | 7 | 1.4 | 200 | 25 | 5000* | 32 |
| 2 | 71 | M | C | Pre-auricular skin | T2b, N0 | Gout, arthritis, hypothyroid | 10 | 2.6 | 200 | 33 | 6600* | 64 |
| 3 | 91 | M | C | Neck | III | Polio, arthritis, HTN, CAD | 10 | 2.8 | 35 | 5000 1600 Boost | 54 | |
| 4 | 73 | M | C | Scalp and parotid | III | COPD, DM, CAD, lung nodules, Afib, HTN | 9 | 1.9 | 200 | 30 | 5000* 1000 Boost | 41 |
| 5 | 78 | F | C | Scalp and neck | IVA | Dementia, CKD, Hypothyroidism | 17 | 4.4 | 200 | 25 | 5000* | 35 |
| 6 | 87 | M | C | Scalp | T2b, N0 | None | 11 | 2.6 | 200 | 30 | 6000* | 47 |
| 7 | 70 | M | C | Scalp | III | Lung transplant | 6 | 1.8 | 200 | 21 | 6000 4200 total scalp XRT | 28 |
| 8 | 73 | M | C | Rt Ankle Rt Groin | III | Melanoma, UC | 6 | 1.4 | 200 | 25 | 5000 Rt* ankle, 3800 Rt* Groin | 35 |
| 9 | 67 | M | C | Scalp to nodes | III | None | 7 | 1.7 | 200 | 35 | 7000 Parotid | 58 |
| 10 | 82 | M | C | Cheek | T2b, N0 | Hypothyroidism, BPH | 6 | 1.2 | 200 | 30 | 6000* | 42 |
| 11 | 79 | M | C | Scalp | T3, N0 | COPD, CAD, HTN, Arthritis | 5 | 0.9 | 200 | 21 | 4200 | 28 |
| 12 | 92 | F | C | Eyebrow | T3, N0 | Arthritis, DM, CAD, HTN, Breast Cancer | 7 | 1.4 | 180 | 25 | 4500 | 34 |
| 13 | 76 | M | C | Scalp | T2b, N0 | NHL, DVT, gout | 18 | 15.1 | 200 | 25 25 8 25 | 5000 Scalp* 5000 Neck 1600 Boost 5000 Palliative | 35 36 12 40 |
| 14 | 70 | M | C | Ear | T2b, N0 | Renal transplant, HTN, DM, CHF | 4 | 0.8 | 200 | 25 | 5000 R Ear 6600 Temporal bone | 35 |
| 15 | 76 | M | C | Neck node | III | Asthma | 9 | 12.6 | 200 | 35 | 5000 Neck 2000 Boost | 51 |
| 16 | 74 | M | C | Scalp | IVA | Heart transplant, bronchitis, arthritis, CKD | 9 | 24.2 | 250 | 21 15 | 5250* 3000 | 28 21 |
| 17 | 85 | M | C | Scalp | T2b, N0 | HTN, CAD, Afib, TIA | 9 | 2.3 | 200 | 32 | 6400* | 57 |
| 18 | 47 | M | AA | Legs | T2b, N0 | Gout | 6 | 1.2 | 250 | 24 | 6000* | 32 |
The median number of cetuximab doses was 8.0 ± 3.7 (SD). The range was 4–18. The median cumulative dose of cetuximab was 4750 ± 2872 mg. The median duration of cetuximab therapy was 1.9 ± 6.2 months and ended with completion of radiotherapy.
The response rates for concurrent therapy with cetuximab plus radiation therapy included 10 patients (55.5%) who achieved an objective complete response (CR), 4 (27.7%) had a partial response at the treated site (Table 2). Thus, the objective response rate (CR + PR) was 83.2%. As an example of a typical response is shown, (Figure 1) demonstrating dramatic remission of bulky scalp tumors, including in-transit metastases, following concurrent treatment. This patient achieved a durable complete response after treatment, which continues at over 2-year follow-up.
Table 2: Treatment outcome
| UPN | Cetuxumab toxicity | RT toxicity | OR | PFS (months) | OS (months) | Current status | Cause of death |
|---|---|---|---|---|---|---|---|
| 1 | Acneiform Rash | Dermatitis and mucositis | PD | 1.7 | 25.2 | D-2nd primary | NSCLC |
| 2 | Acneiform Rash | Dermatitis | CR | 17.2 | 17.2 | A-NED | – |
| 3 | Acneiform Rash | Mucositis, moist desquamation | CR | 19.7 | 19.7 | A-NED | – |
| 4 | Acneiform Rash | Dermatitis | PR | 6.2 | 12.2 | D-other | COPD |
| 5 | None | Dermatitis | CR | 17.9 | 17.9 | A-NED | – |
| 6 | Fatigue, acneiform rash | Moist desquamation skin necrosis | CR | 18.2 | 33.4 | A-NED | – |
| 7 | Acneiform Rash | Dermatitis, mucositis. | PR | 8.2 | 8.2 | D-2nd primary | NSCLC |
| 8 | Acneiform Rash | Moist desquamation | PR | 20.0 | 62.5 | DOD | – |
| 9 | None | Moist desquamation with necrosis, fatigue, mucositis | PR | 3.8 | 3.8 | D-other | Suicide |
| 10 | Acneiform Rash | Mucositis. Auditory canal inflammation | CR | 60.4 | 60.4 | A-NED | – |
| 11 | Acneiform Rash | None | PD | 0.9 | 3.5 | DOD | Disease progression |
| 12 | Acneiform Rash | None | CR | 5.6 | 5.6 | D-2nd primary | Second primary |
| 13 | None | Dermatitis and mucositis | CR | 60.7 | 60.7 | D-2nd primary | Disease progression, second primary |
| 14 | Acneiform rash and dry eyes | Moist desquamation | PD | 5.9 | 44.8 | D-other | Disease progression |
| 15 | None | None | CR | 69.9 | 69.9 | A-NED | |
| 16 | SOB | dermatitis, photophobia | SD | 21.6 | 45.8 | DOD | disease progression |
| 17 | acneiform rash | Moist desquamation | CR | 14.4 | 14.4 | A-NED | – |
| 18 | None | dermatitis | CR | 60.3 | 60.3 | A-NED | – |
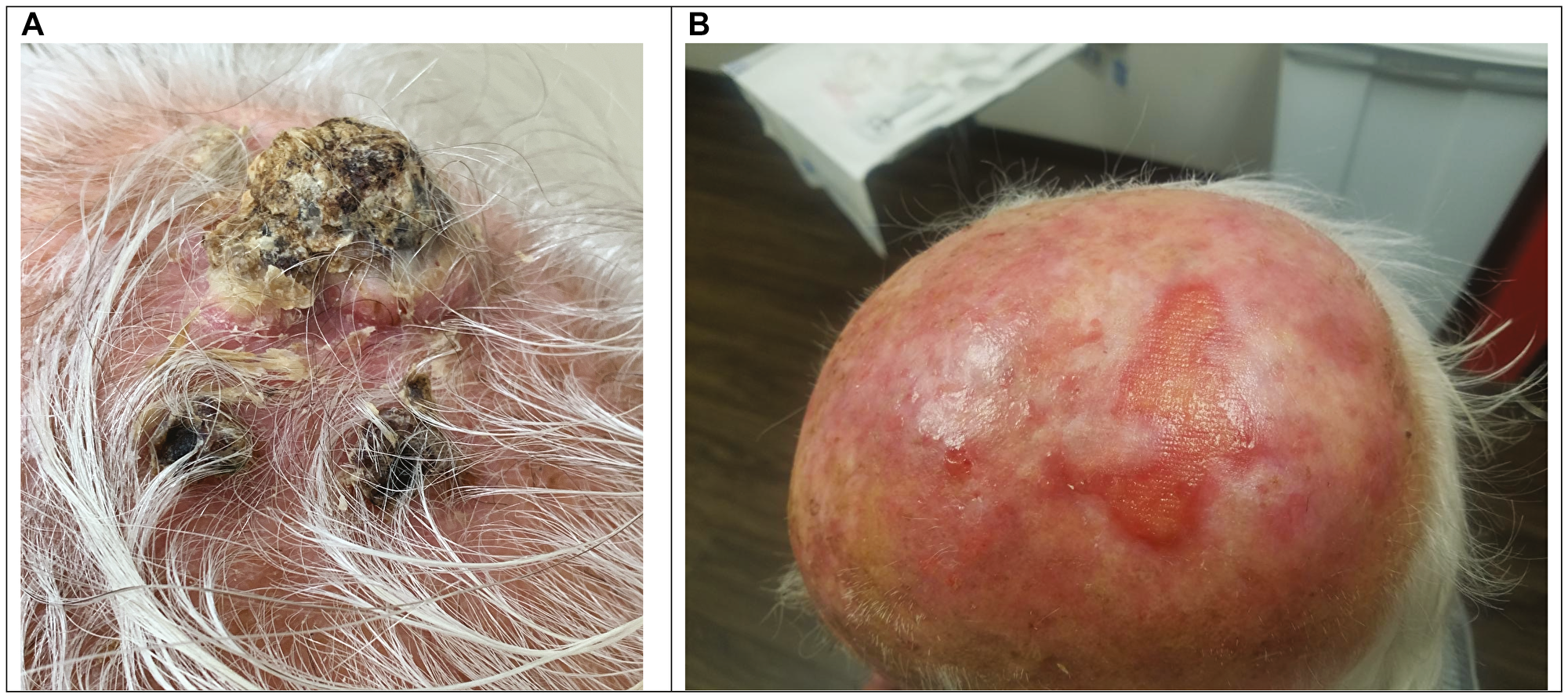
Figure 1: Example of treatment response. Patient 17 Pre-treatment scalp photograph (A). Patient 17 scalp photograph at 14 months post-treatment with Cetuximab/RT (B).
Median progression free survival was 21.6 months at a median follow-up of 18 months (range of 0.9–70 months) (Table 2). The apparent drop-off in the PFS graph after 21 months is due to the diminishing number of patients at risk for progression. Only one patient eventually relapsed within the treatment site (5.5%), 2 relapsed with marginal recurrences at the edge of the irradiated field (11.1%), 3 progressed in regional lymph nodes or in-transit metastases (16.6%). With longer follow-up, 2 patients developed distant metastases (11.1%) and 3 patients developed aggressive 2nd primary neoplasms (16.6%) (one of these also had a concurrent regional recurrence of CSCC). Progression-free survival was 61% at 1 year and 40% at 2 years. (Figure 2).
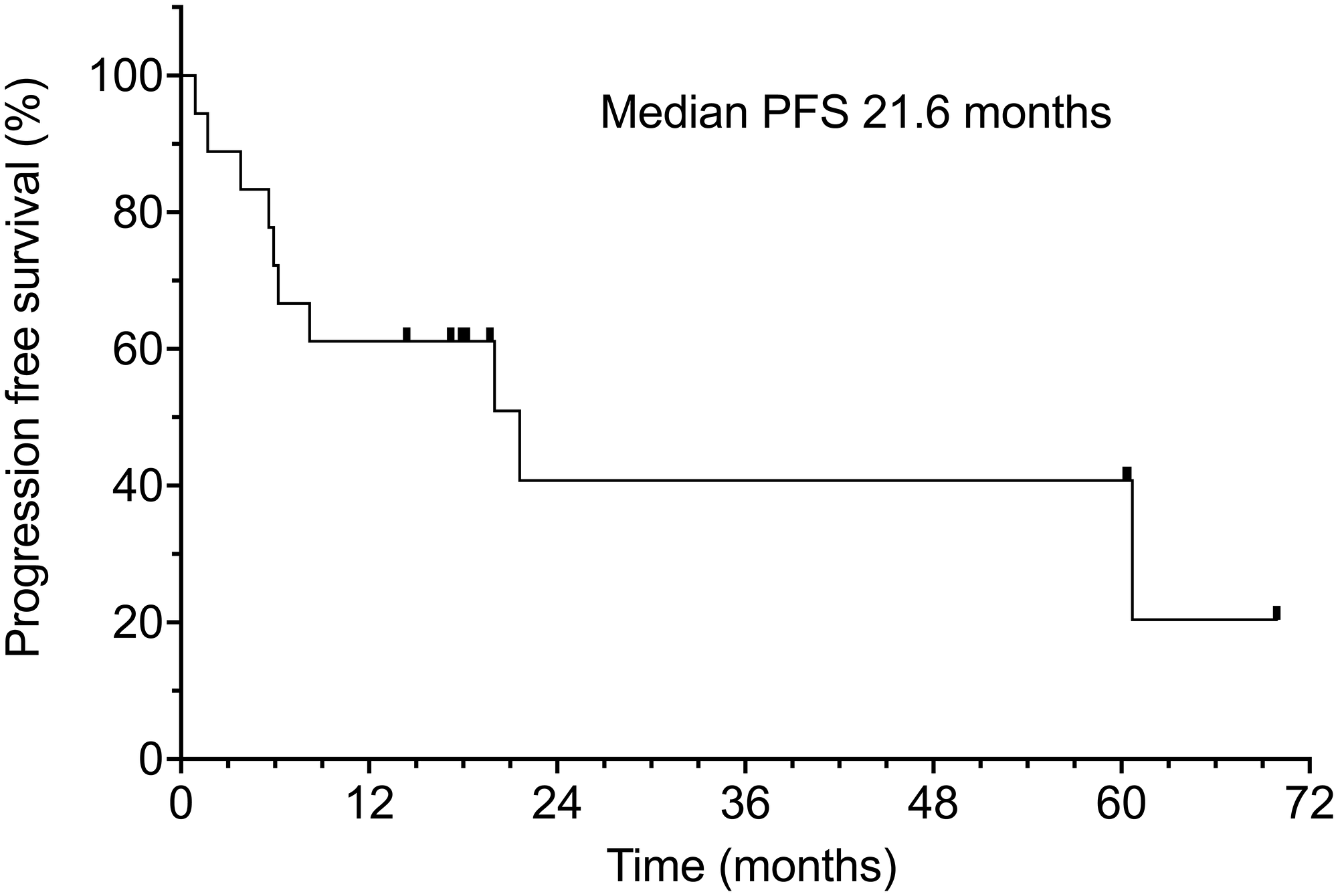
Figure 2: Progression-free survival of all 18 patients. Censored patients are indicated by hash marks.
Of the 18 patients in our study, 8 (44.4%) have died and 10 (55.5%) remain alive. Median overall survival was 60.7% months (range 3.5–62.5 months) (Figure 3). It should be noted that causes of death included comorbid age-related conditions in 3 patients (16.7%), 1 of whom also had concurrent progression of metastatic CSCC (5.5%), and 3 others developed aggressive secondary neoplasms (16.7%).
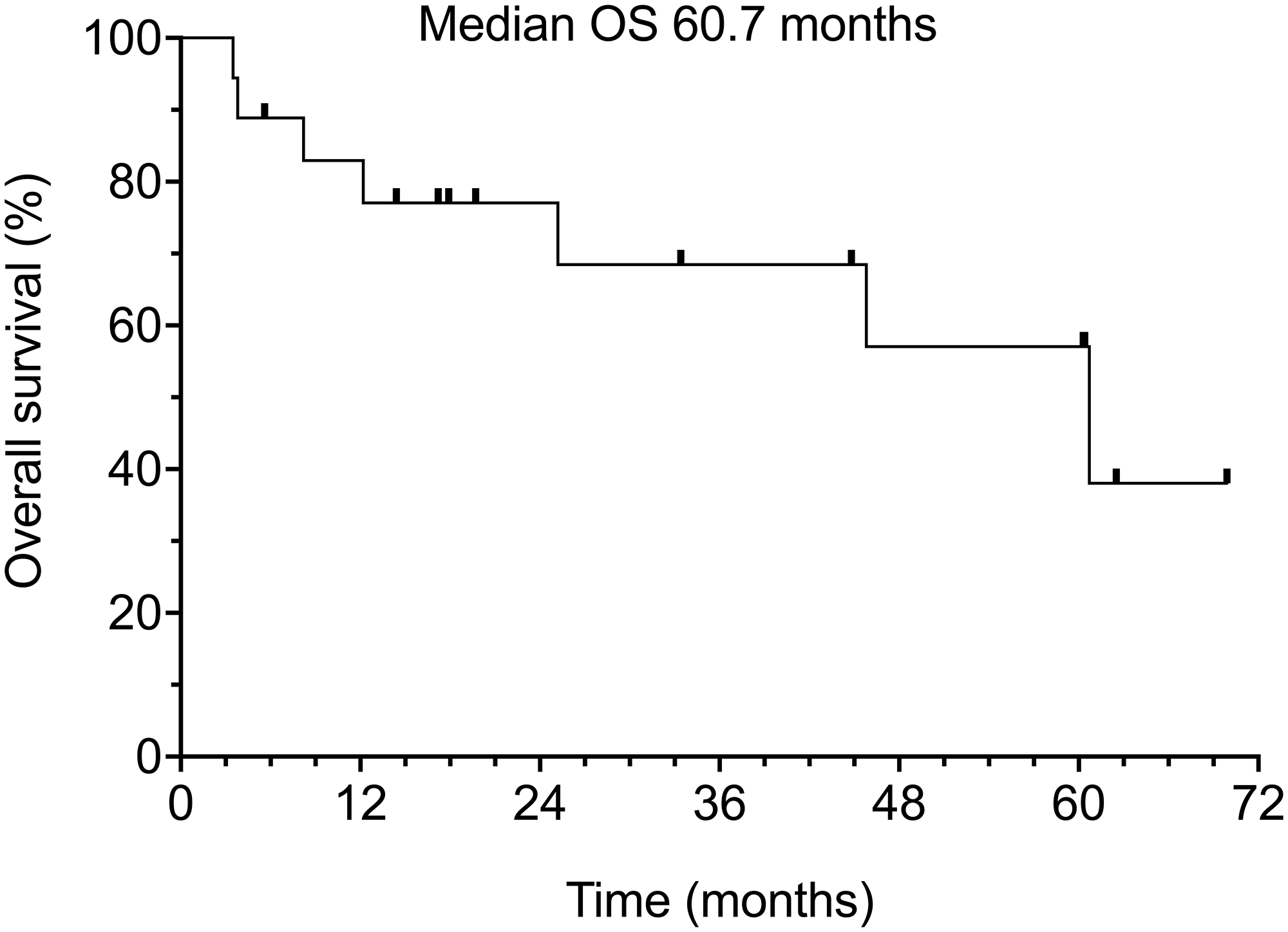
Figure 3: Overall survival of all 18 patients.
Cetuximab treatment was well tolerated. A total of 5 patients experienced no significant cetuximab-related side effects (27.8%). A total of 12 patients (66.7%) developed mild acneiform skin rash controllable with oral and topical antibiotics, or fatigue (reaching at most Grade 1 or 2 toxicity). Radiotherapy produced typical side effects (skin erythema, moist desquamation, and oral mucositis). Three patients had no apparent side effects from radiotherapy (16.7%). Eight patients (44.4%) experienced mild radiation skin changes at the site of radiotherapy. More severe radiation toxicity was seen in other patients, with moist desquamation or other more severe skin toxicity (33.3%), oral cavity mucositis (33.3%), auditory canal inflammation (1 patient). In several patients there were overlapping toxicities. Delayed wound healing at the prior tumor site also was observed after regression of bulky tumors, although this was not precisely quantified. Seven patients required a brief treatment break lasting 1–2 weeks due to radiation dermatitis, moist desquamation, oral pain, and mucositis. Patient number 16 was the only patient to have therapy interrupted more than once for a total of 4 treatment breaks.
Sixteen patients were able to complete planned treatment with minimal side effects. 2 patients (Number 5 and 7) had their treatments discontinued early due to increasing skin toxicity. Despite early discontinuation of therapy in patient 5, after resolution of radiation-induced skin changes, she was found to have achieved a complete response. Patient number 7 developed a second primary cancer, non-Hodgkin’s lymphoma (NHL). This patient was able to restart Erbitux and radiotherapy after being treated with chemotherapy and achieving remission of her NHL. Only 1 patient was ever hospitalized for toxicity. This patient developed syncope due to dehydration while undergoing treatment but was able to continue regimen after recovery (Grade 3 toxicity).
DISCUSSION
The majority of CSCC are small and localized, and thus are easily managed by dermatologists and surgeons, including MOHS microsurgical approaches. High-risk CSCC are usually managed with aggressive surgical resection including peripheral and deep margin assessment [1]. After resection of high-risk patients, adjuvant radiotherapy is frequently added, based on data from small case series. Unfortunately, no larger or randomized studies of adjuvant radiotherapy have been performed in CSCC, to date. There have been additional recent advances in treatment options for laCSCC such as the PD-1 antibodies pembrolizumab and cemiplimab [17, 18]. Additional treatment options are needed, especially for patients who have contraindications to checkpoint inhibitor treatment, such as those with solid organ transplants and autoimmune disease.
EGFR expression is present in normal keratinocytes and is expressed at increasingly higher levels in most patients with laCSCC and in lymph node metastases from CSCC [12, 20–23]. In keratinocyte cultures, epidermal growth factor (EGF) stimulates cell proliferation and suppresses markers of terminal differentiation [24]. EGF stimulated proliferation can be blocked by EGFR antibodies and EGFR tyrosine kinase inhibitors [24]. In vitro and murine models have shown that EGF activation of malignant epithelial cells induces signal transducer and activator of transcription 3 (STAT-3) activation, which drives carcinogenesis [25, 26]. EGFR blockade abrogates this response [25]. Additional preclinical research has shown that EGFR blockade may inhibit telomerase activity in CSCC and thus suppress tumor cell survival [27]. Thus, inhibition of the EGFR signaling pathway seemed to be an attractive treatment option in laCSCC.
Cetuximab is an IgG1 monoclonal antibody, which binds with high affinity to human EGFR. This antibody blocks binding of EGF and other ligands to the EGFR. Cetuximab also induces internalization and downregulation of EGFR and induces enhanced antitumor immune responses via antibody dependent cell-mediated cytotoxicity [28]. In vitro exposure of squamous cell carcinoma (SCC) cell lines derived from head and neck cancer patients to cetuximab inhibited proliferation in a time and dose-dependent manner [29]. Since cetuximab causes tumor cells to accumulate in G1 of the cell cycle, radiosensitivity is enhanced [29].
Numerous studies have evaluated cetuximab plus RT in SCCHN. Bonner et al. conducted a randomized trial comparing RT alone with RT plus cetuximab, in the treatment of stage III or IV nonmetastatic, measurable SCC of the oropharynx, hypopharynx, or larynx [30, 31]. The median duration of locoregional control (primary endpoint) was 24.4 months among patients treated with cetuximab plus RT and 14.9 months among those given RT alone. RT plus cetuximab significantly prolonged progression-free survival and overall survival. The 5-year OS rate was 45.6% in the cetuximab plus RT group and 36.4% in the RT alone group [30, 31].
Despite similarities in histology, there has been only limited testing of EGFR antibodies such as cetuximab and panitumumab in CSCC. Published clinical trials of cetuximab and panitumumab in recurrent or refractory CSCC have shown only limited activity as monotherapy [13–16]. Durable responses or remissions have proven elusive [13–16]. The oral 1st generation EGFR inhibitor Erlotinib has demonstrated virtually no clinical activity in CSCC [32].
There is only limited data published concerning the effectiveness of combining cetuximab with radiotherapy in laCSCC. Joseph et al. published 8 patients with laCSCC treated with cetuximab plus concurrent radiotherapy [33]. At 25 months follow-up, five patients remained in a complete remission. One patient relapsed after a partial response. Two patients died (one of due to progression of disease, the other of an unrelated cause). Treatment in this group of patients proved well tolerated, with most toxicities ≤ grade 2, and no toxicities of grade 4/5 reported. Preneau reported a series of patients treated with cetuximab in combination with either radiotherapy, carboplatin or as monotherapy [16]. The response rate in cetuximab and radiation treated patients (n = 5) was reported as 80% versus 33% for cetuximab alone. The PFS was a disappointing 1.6 months and overall survival only 3 months for the cetuximab plus radiotherapy patients [16]. Samstein and colleagues reported 12 laCSCC patients with concurrent cetuximab and radiotherapy [34]. Complete and partial response was noted in 36% and 27% (response rate, 64%). Median progression-free and overall survival were only 6.4 and 8.0 months, respectively. Grades 3–4 adverse events were noted in 83% of patients; 67% required hospital admission for adverse events. A total of 51% had longer-term disease control with a short median follow-up (7 months). Lu and Lien published a series of patients receiving either cisplatin and RT (15 patients) or cetuximab plus RT (8 patients) for laCSCC. With 2-year median follow-up, both PFS (50 vs. 30%) and OS (73 vs. 40%) appeared to favor the cetuximab/RT group [35].
The activity of cetuximab therapy in conjunction with radiotherapy in SCCHN encouraged us to use this approach for treatment of laCSCC. This was particularly attractive, as most of our patients had primary tumors originating in head and neck sites. We report 19 patients with laCSCC who had progressed after prior surgery, but who had not received prior systemic therapy or radiation therapy. These patients were treated with a consistent treatment approach combining cetuximab with concurrent radiotherapy. We found an objective response rate of 83.2% with 55.5% complete responses and 27.7% partial responses. The median progression-free survival was 21.6 months, with a median follow-up of 18 months. A total of 60% of patients were progression free at 1 year. Median overall survival was 60.7%.
Our data demonstrate that cetuximab plus radiotherapy represents an active treatment option for laCSCC, with manageable toxicity. This treatment option can be considered in patients with laCSCC, including those with contraindications that preclude checkpoint inhibitor therapy, such as organ transplants and autoimmune disease. The overall frequency of complete responses and duration of responses is high enough to be of interest for potential comparison to checkpoint-inhibitor based therapy. Our current study is intended to be hypothesis generating, with a goal of providing preliminary data to supporting development of comparative clinical trials. Potential limitations of our data include a relatively small number of treated patients, the retrospective nature of the analysis and 6-year span of patient accrual. Challenges for future treatment refinements included a small number of patients who failed either at the treatment site or at the margins of the planned radiation. The latter may suggest expansion of planned treatment volumes. Additional challenges include patients who developed regional and distant metastases from CSCC or aggressive second neoplasms.
Materials and Methods
Patient population
A retrospective review of medical records was performed of patients uniformly treated with cetuximab by a single physician (WS) at a single institution. These patients were treated over a 6-year period between 2014–2020. Patients with locoregionally advanced or recurrent cutaneous squamous cell skin cancer (CSCC) treated with cetuximab and concomitant radiotherapy were identified by a search of a HIPAA-compliant electronic medical record system, IKnowMed (McKesson, Inc., Houston TX, USA). Clinical data were extracted from the chart into a spreadsheet and de-identified. The following information was extracted from the record: Gender, age, date of diagnosis, primary site, and stage at diagnosis using UICC 8th edition [6]. Characteristics of cetuximab treatment were recorded, including number of doses and cumulative cetuximab dose administered, start and end dates for cetuximab treatment and any cetuximab-associated toxicity. Data related to radiotherapy were also extracted including start and end date of radiotherapy, elapsed time (days), fraction size, cumulative radiotherapy dose (Gy) and any associated toxicities. Best response, date of relapse and progression sites were identified. Both PFS and OS were calculated from cetuximab start date, cause of death was also recorded if patient was deceased. This study design was deemed exempt from full IRB review by the Western IRB chair. The data cut-off date for data analysis was 12/31/20.
Exclusions included patients with squamous cell carcinoma of the head and neck (SCCHN) with oral, pharyngeal, or laryngeal primary sites. Patients with recurrent or metastatic CSCC after prior radiotherapy or other prior systemic treatments or those who were not treated with concurrent cetuximab plus radiotherapy were excluded.
Cetuximab treatment
The initial loading dose of cetuximab was 400 mg/m² IV infused over 2 hrs. Subsequent doses of 250 mg/m² IV were infused over 60 min each week, starting prior to the initiation of radiotherapy and continuing throughout the entire period of radiation therapy. All patients were seen weekly for close monitoring and lab monitoring during concurrent therapy. Patients were supported with hydration and magnesium replacement as required. Side effects of therapy were treated with symptomatic measures such as topical and oral antibiotics for the frequent cetuximab-induced rash, oral lidocaine for mucositis.
All patients underwent referral and evaluation for concomitant radiation therapy. The treatment consisted of electron beam fields designed to completely encompass superficial skin lesions in most patients, including a 1–2 cm margin. The dose ranged from 5000–6600 cGy at 200–250 cGy per fraction. Field reduction usually after 5000 cGy. If the regional lymph nodes were involved, or the skin tumor invaded facial structures, 3D conformal radiation therapy or IMRT was used with 6MeV photons, bolus used as needed to bring the dose to the surface. For nodal disease, a boost to carry the involved nodes to a total of 6600–7000 cGy was used.
Data analysis
Extracted information was accessed and recorded in a de-identified manner into a password-protected Excel spreadsheet (Microsoft, Redmond WA) for analysis and calculation of descriptive statistics. Progression-free and overall survival were evaluated via Kaplan-Meier analysis [36]. Toxicity was graded using the CTCAE 1.1 criteria [37].
Abbreviations
AJCC: American Joint Commission for Cancer; CR: complete remission; CSCC: cutaneous squamous cell carcinoma; EGF: epidermal growth factor; EGFR: epidermal growth factor receptor; laCSCC: locally advanced cutaneous squamous cell carcinoma; NHL: non-Hodgkin lymphoma; PFS: progression-free survival; PR: partial response; SCCHN: squamous cell carcinoma of the head and neck; UICC: Union for International Cancer Control.
Data availability
The data underlying this article will be shared upon reasonable request to corresponding author.
ACKNOWLEDGMENTS
We appreciate the patients, families, clinic staff, and referring physicians who made this case series possible. The helpful editorial suggestions of Suzanne Samlowski, M. Arch. are also appreciated.
CONFLICTS OF INTEREST
Authors have no conflicts of interest to declare.
Ethical statement
This study design was deemed exempt from full IRB review by the Western IRB chair.
References
1. Nehal KS, Bichakjian CK. Update on Keratinocyte Carcinomas. N Engl J Med. 2018; 379:363–74. https://doi.org/10.1056/NEJMra1708701. [PubMed].
2. Que SKT, Zwald FO, Schmults CD. Cutaneous squamous cell carcinoma: Management of advanced and high-stage tumors. J Am Acad Dermatol. 2018; 78:249–61. https://doi.org/10.1016/j.jaad.2017.08.058. [PubMed].
3. Karia PS, Han J, Schmults CD. Cutaneous squamous cell carcinoma: estimated incidence of disease, nodal metastasis, and deaths from disease in the United States, 2012. J Am Acad Dermatol. 2013; 68:957–66. https://doi.org/10.1016/j.jaad.2012.11.037. [PubMed].
4. Que SKT, Zwald FO, Schmults CD. Cutaneous squamous cell carcinoma: Incidence, risk factors, diagnosis, and staging. J Am Acad Dermatol. 2018; 78:237–47. https://doi.org/10.1016/j.jaad.2017.08.059. [PubMed].
5. Ruiz ES, Karia PS, Besaw R, Schmults CD. Performance of the American Joint Committee on Cancer Staging Manual, 8th Edition vs the Brigham and Women’s Hospital Tumor Classification System for Cutaneous Squamous Cell Carcinoma. JAMA Dermatol. 2019; 155:819–25. https://doi.org/10.1001/jamadermatol.2019.0032. [PubMed].
6. Keohane SG, Proby CM, Newlands C, Motley RJ, Nasr I, Mohd Mustapa MF, Slater DN, and British Association of Dermatologists (Squamous and Basal Cell Carcinoma Guideline Development Groups), and Royal College of Pathologists (Skin Cancer Lead). The new 8th edition of TNM staging and its implications for skin cancer: a review by the British Association of Dermatologists and the Royal College of Pathologists, U.K. Br J Dermatol. 2018; 179:824–28. https://doi.org/10.1111/bjd.16892. [PubMed].
7. Gonzalez JL, Cunningham K, Silverman R, Madan E, Nguyen BM. Comparison of the American Joint Committee on Cancer Seventh Edition and Brigham and Women’s Hospital Cutaneous Squamous Cell Carcinoma Tumor Staging in Immunosuppressed Patients. Dermatol Surg. 2017; 43:784–91. https://doi.org/10.1097/DSS.0000000000001038. [PubMed].
8. Karia PS, Jambusaria-Pahlajani A, Harrington DP, Murphy GF, Qureshi AA, Schmults CD. Evaluation of American Joint Committee on Cancer, International Union Against Cancer, and Brigham and Women’s Hospital tumor staging for cutaneous squamous cell carcinoma. J Clin Oncol. 2014; 32:327–34. https://doi.org/10.1200/JCO.2012.48.5326. [PubMed].
9. Schmitt AR, Brewer JD, Bordeaux JS, Baum CL. Staging for cutaneous squamous cell carcinoma as a predictor of sentinel lymph node biopsy results: meta-analysis of American Joint Committee on Cancer criteria and a proposed alternative system. JAMA Dermatol. 2014; 150:19–24. https://doi.org/10.1001/jamadermatol.2013.6675. [PubMed].
10. Cranmer LD, Engelhardt C, Morgan SS. Treatment of unresectable and metastatic cutaneous squamous cell carcinoma. Oncologist. 2010; 15:1320–28. https://doi.org/10.1634/theoncologist.2009-0210. [PubMed].
11. Tanvetyanon T, Padhya T, McCaffrey J, Kish JA, Deconti RC, Trotti A, Rao NG. Postoperative concurrent chemotherapy and radiotherapy for high-risk cutaneous squamous cell carcinoma of the head and neck. Head Neck. 2015; 37:840–45. https://doi.org/10.1002/hed.23684. [PubMed].
12. Maubec E, Duvillard P, Velasco V, Crickx B, Avril MF. Immunohistochemical analysis of EGFR and HER-2 in patients with metastatic squamous cell carcinoma of the skin. Anticancer Res. 2005; 25:1205–10. [PubMed].
13. Maubec E, Petrow P, Scheer-Senyarich I, Duvillard P, Lacroix L, Gelly J, Certain A, Duval X, Crickx B, Buffard V, Basset-Seguin N, Saez P, Duval-Modeste AB, et al. Phase II study of cetuximab as first-line single-drug therapy in patients with unresectable squamous cell carcinoma of the skin. J Clin Oncol. 2011; 29:3419–26. https://doi.org/10.1200/JCO.2010.34.1735. [PubMed].
14. Foote MC, McGrath M, Guminski A, Hughes BGM, Meakin J, Thomson D, Zarate D, Simpson F, Porceddu SV. Phase II study of single-agent panitumumab in patients with incurable cutaneous squamous cell carcinoma. Ann Oncol. 2014; 25:2047–52. https://doi.org/10.1093/annonc/mdu368. [PubMed].
15. Montaudié H, Viotti J, Combemale P, Dutriaux C, Dupin N, Robert C, Mortier L, Kaphan R, Duval-Modeste AB, Dalle S, De Quatrebarbes J, Stefan A, Brunet-Possenti F, et al. Cetuximab is efficient and safe in patients with advanced cutaneous squamous cell carcinoma: a retrospective, multicentre study. Oncotarget. 2020; 11:378–85. https://doi.org/10.18632/oncotarget.27434. [PubMed].
16. Preneau S, Rio E, Brocard A, Peuvrel L, Nguyen JM, Quéreux G, Dreno B. Efficacy of cetuximab in the treatment of squamous cell carcinoma. J Dermatolog Treat. 2014; 25:424–27. https://doi.org/10.3109/09546634.2012.751481. [PubMed].
17. Rischin D, Migden MR, Lim AM, Schmults CD, Khushalani NI, Hughes BGM, Schadendorf D, Dunn LA, Hernandez-Aya L, Chang ALS, Modi B, Hauschild A, Ulrich C, et al. Phase 2 study of cemiplimab in patients with metastatic cutaneous squamous cell carcinoma: primary analysis of fixed-dosing, long-term outcome of weight-based dosing. J Immunother Cancer. 2020; 8:e000775. https://doi.org/10.1136/jitc-2020-000775. [PubMed].
18. Maubec E, Boubaya M, Petrow P, Beylot-Barry M, Basset-Seguin N, Deschamps L, Grob JJ, Dréno B, Scheer-Senyarich I, Bloch-Queyrat C, Leccia MT, Stefan A, Saiag P, et al. Phase II Study of Pembrolizumab As First-Line, Single-Drug Therapy for Patients With Unresectable Cutaneous Squamous Cell Carcinomas. J Clin Oncol. 2020; 38:3051–61. https://doi.org/10.1200/JCO.19.03357. [PubMed].
19. Migden MR, Rischin D, Schmults CD, Guminski A, Hauschild A, Lewis KD, Chung CH, Hernandez-Aya L, Lim AM, Chang ALS, Rabinowits G, Thai AA, Dunn LA, et al. PD-1 Blockade with Cemiplimab in Advanced Cutaneous Squamous-Cell Carcinoma. N Engl J Med. 2018; 379:341–51. https://doi.org/10.1056/NEJMoa1805131. [PubMed].
20. Nanney LB, Magid M, Stoscheck CM, King LE Jr. Comparison of epidermal growth factor binding and receptor distribution in normal human epidermis and epidermal appendages. J Invest Dermatol. 1984; 83:385–93. https://doi.org/10.1111/1523-1747.ep12264708. [PubMed].
21. Groves RW, Allen MH, MacDonald DM. Abnormal expression of epidermal growth factor receptor in cutaneous epithelial tumours. J Cutan Pathol. 1992; 19:66–72. https://doi.org/10.1111/j.1600-0560.1992.tb01561.x. [PubMed].
22. Krähn G, Leiter U, Kaskel P, Udart M, Utikal J, Bezold G, Peter RU. Coexpression patterns of EGFR, HER2, HER3 and HER4 in non-melanoma skin cancer. Eur J Cancer. 2001; 37:251–59. https://doi.org/10.1016/s0959-8049(00)00364-6. [PubMed].
23. Shimizu T, Izumi H, Oga A, Furumoto H, Murakami T, Ofuji R, Muto M, Sasaki K. Epidermal growth factor receptor overexpression and genetic aberrations in metastatic squamous-cell carcinoma of the skin. Dermatology. 2001; 202:203–6. https://doi.org/10.1159/000051637. [PubMed].
24. Peus D, Hamacher L, Pittelkow MR. EGF-receptor tyrosine kinase inhibition induces keratinocyte growth arrest and terminal differentiation. J Invest Dermatol. 1997; 109:751–56. https://doi.org/10.1111/1523-1747.ep12340759. [PubMed].
25. Lewis DA, Hurwitz SA, Spandau DF. UVB-induced apoptosis in normal human keratinocytes: role of the erbB receptor family. Exp Cell Res. 2003; 284:316–27. https://doi.org/10.1016/s0014-4827(02)00043-5. [PubMed].
26. Bito T, Sumita N, Ashida M, Budiyanto A, Ueda M, Ichihashi M, Tokura Y, Nishigori C. Inhibition of Epidermal Growth Factor Receptor and PI3K/Akt Signaling Suppresses Cell Proliferation and Survival through Regulation of Stat3 Activation in Human Cutaneous Squamous Cell Carcinoma. J Skin Cancer. 2011; 2011:874571. https://doi.org/10.1155/2011/874571. [PubMed].
27. Ashida M, Bito T, Budiyanto A, Ichihashi M, Ueda M. Involvement of EGF receptor activation in the induction of cyclooxygenase-2 in HaCaT keratinocytes after UVB. Exp Dermatol. 2003; 12:445–52. https://doi.org/10.1034/j.1600-0625.2003.00101.x. [PubMed].
28. Specenier P, Vermorken JB. Cetuximab in the treatment of squamous cell carcinoma of the head and neck. Expert Rev Anticancer Ther. 2011; 11:511–24. https://doi.org/10.1586/era.11.20. [PubMed].
29. Huang SM, Bock JM, Harari PM. Epidermal growth factor receptor blockade with C225 modulates proliferation, apoptosis, and radiosensitivity in squamous cell carcinomas of the head and neck. Cancer Res. 1999; 59:1935–40. [PubMed].
30. Bonner JA, Harari PM, Giralt J, Azarnia N, Shin DM, Cohen RB, Jones CU, Sur R, Raben D, Jassem J, Ove R, Kies MS, Baselga J, et al. Radiotherapy plus cetuximab for squamous-cell carcinoma of the head and neck. N Engl J Med. 2006; 354:567–78. https://doi.org/10.1056/NEJMoa053422. [PubMed].
31. Bonner JA, Harari PM, Giralt J, Cohen RB, Jones CU, Sur RK, Raben D, Baselga J, Spencer SA, Zhu J, Youssoufian H, Rowinsky EK, Ang KK. Radiotherapy plus cetuximab for locoregionally advanced head and neck cancer: 5-year survival data from a phase 3 randomised trial, and relation between cetuximab-induced rash and survival. Lancet Oncol. 2010; 11:21–28. https://doi.org/10.1016/S1470-2045(09)70311-0. [PubMed].
32. Gold KA, Kies MS, William WN Jr, Johnson FM, Lee JJ, Glisson BS. Erlotinib in the treatment of recurrent or metastatic cutaneous squamous cell carcinoma: A single-arm phase 2 clinical trial. Cancer. 2018; 124:2169–73. https://doi.org/10.1002/cncr.31346. [PubMed].
33. Joseph K, Alkaabi K, Warkentin H, Ghosh S, Jha N, Smylie M, Walker J. Cetuximab-radiotherapy combination in the management of locally advanced cutaneous squamous cell carcinoma. J Med Imaging Radiat Oncol. 2019; 63:257–63. https://doi.org/10.1111/1754-9485.12842. [PubMed].
34. Samstein RM, Ho AL, Lee NY, Barker CA. Locally advanced and unresectable cutaneous squamous cell carcinoma: outcomes of concurrent cetuximab and radiotherapy. J Skin Cancer. 2014; 2014:284582. https://doi.org/10.1155/2014/284582. [PubMed].
35. Lu SM, Lien WW. Concurrent Radiotherapy With Cetuximab or Platinum-based Chemotherapy for Locally Advanced Cutaneous Squamous Cell Carcinoma of the Head and Neck. Am J Clin Oncol. 2018; 41:95–99. https://doi.org/10.1097/COC.0000000000000228. [PubMed].
36. Stalpers LJA, De Meere JMM, Kaplan EL. [The mortality in Amsterdam from 1554 to 2021]. Ned Tijdschr Geneeskd. 2021; 165:D5962. [PubMed].
37. U.S. Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) v4.0. NIH, National Cancer Institute. 2010. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm#ctc_40.

