Introduction
Nectins are immunoglobulin-like cell adhesion molecules and regulate besides cell adhesion various cellular functions, such as cell motility, proliferation, polarization, survival and differentiation [1–3] and appear to have an immunomodulatory function [4, 5]. Four nectins have been described. Nectin-4 has been shown to be a successful therapy target in solid tumors including bladder cancer. Challita-Eid et al. conducted an immunohistochemical analysis of Nectin-4 expression in 2,394 patient specimens from different tumor entities. They observed the highest expression levels overall of nectin-4 in bladder (60%) and breast cancer (53%), in ovarian, head and neck and esophageal tumors the staining intensity was significantly lower [6]. In a study with patients with bladder cancer after treatment with platinum and anti programmed death 1/ programmed death ligand 1 therapy Nectin-4 expression was detected in all patient samples tested [7].
Enfortumab verdotin (EV), a Nectin-4-targeted antibody conjugated with the microtubule disrupting agent monomethyl auristatin E (MMAE), has demonstrated excellent response rates in locally advanced bladder cancer [8, 6, 9]. Approval of EV by the U.S. Food and Drug Administration (FDA) in a platinum and immune checkpoint inhibition refractory setting of bladder cancer was based on findings, which demonstrated objective response rates of 44% [10]. In 2020 EV combined with pembrolizumab was accepted by FDA as a firstline treatment for cisplatin-ineligible patients with locally advanced or metastatic urothelial carcinoma [11].
Although the vast majority of head and neck carcinomas are squamous cell carcinomas (HNSCC), they share some morphological and molecular homology with urothelial carcinoma, especially in their mutation profile [12]. HNSCC has a high mortality rate of 40% to 50% [13]. Major risk factors are tobacco use, alcohol consumption and infection with oncogenic human papillomavirus (HPV) [14]. Principal treatments are surgical resection of the primary tumor and neck dissection followed by a risk adapted adjuvant radiation or/ and platinum based chemotherapy or primary definitive chemoradiation [15]. So far cetuximab, a monoclonal antibody targeting epidermal growth factor receptor (EGFR) that has led to an increase of survival compared to conventional platinum based chemotherapy is the only successful targeted therapy approach in this disease [16]. Latest additions to treatment options have been agents targeting immune checkpoint receptors such as program death 1: they are currently approved for cisplatin refractory recurrent and or metastatic HNSCC [17–19]. However, only a minority of HNSCC patients show a clinical response to anti-PD-1 therapy. In a small cohort about a third of patients treated with anti-PD-L1/PD-1 agents showed hyperprogression, which correlated with shorter survival [20].
Therefore, further research to identify new and additional targets in HNSCC is urgently needed. In this study, we aimed to determine the rate of Nectin-4 positivity in a contemporary cohort of HNSCC and to correlate these findings with clinico-pathological parameters.
Results
Patient characteristics
All patients were diagnosed with HNSCC at the Institute of Pathology of the University Hospital Bonn between 2004 and 2014. 16.6% were female, 83.4% were male. Mean patient age at diagnosis was 64.4 years (± 9.6 years SD). The majority of the patients presented with a pT2 tumor stage (40%), followed by pT3 (25%). About a half of the patients had lymph node metastases (48.5%). Most tumors had an intermediate differentiation (G2 56.5%, Table 1).
Table 1: Clinico-pathological characteristics and Nectin-4 expression
| Bonn HNSCC cohort | ||||||
|---|---|---|---|---|---|---|
| Total | Nectin-4 negative | Low Nectin-4 | Medium Nectin-4 | High Nectin-4 | p-value* | |
| All HNSCC cases | n = 159 | 22 (13.8%) | 85 (53.5%) | 43 (27.0%) | 9 (5.7%) | |
| Patients with clinical data | n = 151 | |||||
| Normal tissue | n = 28 | 1 (3.6%) | 8 (28.6%) | 19 (67.9%) | 0 (0%) | |
| Gender | ||||||
| Male | 126 (83.4%) | 13 | 66 | 38 | 9 | 0.0101 |
| Female | 25 (16.6%) | 5 | 17 | 3 | 9 | |
| Age [mean, standard deviation] | 64.4 [9.6] | 65.27 [11.84] | 64.16 [9.60] | 64.45 [10.10] | 64.17 [5.19] | |
| Localization | ||||||
| Oral cavity | 8 (7.8%) | 1 | 5 | 0 | 2 | 0.9922 |
| Oropharynx | 59 (57.8%) | 8 | 29 | 18 | 4 | |
| Hypopharynx | 7 (6.8%) | 0 | 5 | 2 | 0 | |
| Larynx | 28 (27.2%) | 2 | 17 | 9 | 0 | |
| Unknown | 49 | |||||
| Tobacco consumption | ||||||
| Non-smokers | 6 (12.5%) | 0 | 0 | 4 | 2 | 0.0011 |
| Smokers | 42 (87.5%) | 2 | 27 | 13 | 0 | |
| Unknown | 111 | |||||
| Alcohol consumption | ||||||
| No alcohol | 2 (4.5%) | 0 | 2 | 0 | 0 | 0.0672 |
| Occasional | 28 (63.6%) | 1 | 14 | 12 | 1 | |
| Frequent | 14 (31.8%) | 1 | 9 | 3 | 1 | |
| Unknown | 115 | |||||
| HPV status (p16) | ||||||
| p16 negative | 117 (73.6%) | 19 | 67 | 26 | 5 | 0.0051 |
| p16 positive | 42 (26.4%) | 3 | 18 | 17 | 4 | |
| T-stage | ||||||
| pTis | 1 (0.7%) | 0 | 1 | 0 | 0 | 0.4442 |
| pT1 | 26 (18.6%) | 2 | 17 | 5 | 2 | |
| pT2 | 56 (40.0%) | 7 | 27 | 19 | 3 | |
| pT3 | 35 (25.0%) | 6 | 22 | 5 | 2 | |
| pT4 | 22 (15.7%) | 2 | 11 | 8 | 1 | |
| Unknown | 19 | |||||
| Lymph node involvement | ||||||
| pN0 | 54 (41.9%) | 9 | 29 | 14 | 2 | 0.5802 |
| pN1 | 19 (14.7%) | 1 | 10 | 8 | 0 | |
| pN2 | 53 (41.1%) | 7 | 28 | 13 | 5 | |
| pN3 | 3 (2.3%) | 0 | 3 | 0 | 0 | |
| Unknown | 29 | |||||
| Distant metastases | ||||||
| M0 | 70 (98.6%) | 7 | 42 | 18 | 3 | 0.6951 |
| M1 | 1 (1.4%) | 0 | 1 | 0 | 0 | |
| Unknown | 88 | |||||
| Grading | ||||||
| G1 | 3 (2.2%) | 0 | 1 | 2 | 0 | 0.5452 |
| G2 | 78 (56.5%) | 8 | 46 | 18 | 6 | |
| G3 | 57 (41.3%) | 8 | 29 | 17 | 3 | |
| Unknown | 21 | |||||
Expression patterns of Nectin-4 in HNSCC
Immunohistochemistry of Nectin-4 showed a cytoplasmic and membranous staining pattern in benign and malignant tissues.
Normal tissue was available of 28 patients, which showed low or medium expression of Nectin-4 in most cases (96.4%). In normal mucosa, Nectin-4 was mainly expressed in keratinocytes, with emphasis on basal layers, in which epidermal cells with proliferative ability reside. In HNSCC, 13.8% of cases were Nectin-4 negative, 53.5% showed a weak expression, 27.0% showed moderate levels and 5.7% show a high expression of Nectin-4 (Figure 1).
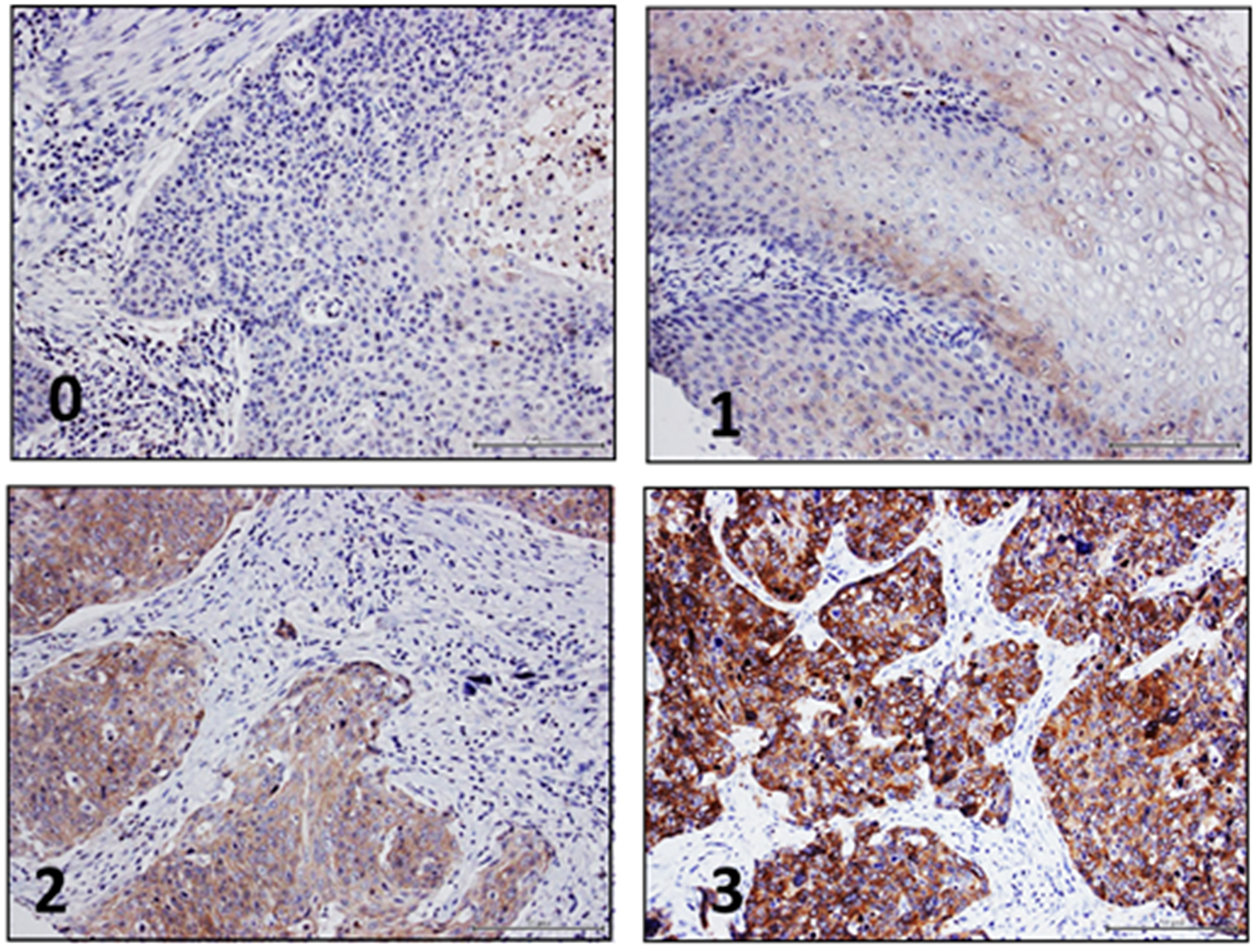
Figure 1: Nectin-4 immunohistochemistry in HNSCC. Representative images of HNSCC demonstrating negative (0), low (1), medium (2) and high (3) Nectin-4 protein levels.
Association between Nectin-4 expression and clinico-pathological parameters
In total, Nectin-4 protein expression was quantified in 159 tissue samples by immunohistochemistry (IHC). Clinical information including follow up data was available for 151 patients (Table 1).
Explorative statistics showed no associations of Nectin-4 expression with tumor stage, grade, occurrence of lymph node metastases, distant metastases, tumor localization, or patient age. Tumors of female patients and non-smokers showed significant higher rates of Nectin-4 expression than male patients or smokers (p = 0.010, p = 0.001, Table 1). Furthermore, p16 positive HNSCC also showed significantly higher Nectin-4 expression (p = 0.005, Table 1) and Nectin-4 expression was correlated with positive p16- status (p = 0.004, correlation coefficient 0.225).
A prognostic potential of Nectin-4 expression in HNSCC was indicated by Kaplan-Meier estimates (Figure 2): Nectin-4 positive tumors showed a significantly better survival (log rank test p = 0.006). In a multivariable Cox regression model Nectin-4 expression was also a significant marker for survival when adjusting for tumor stage, lymph node metastases and p16 (p-value model p = 0.006, Hazard rate Nectin-4 0.418, regression coefficient −0.969, p = 0.041; Table 2).
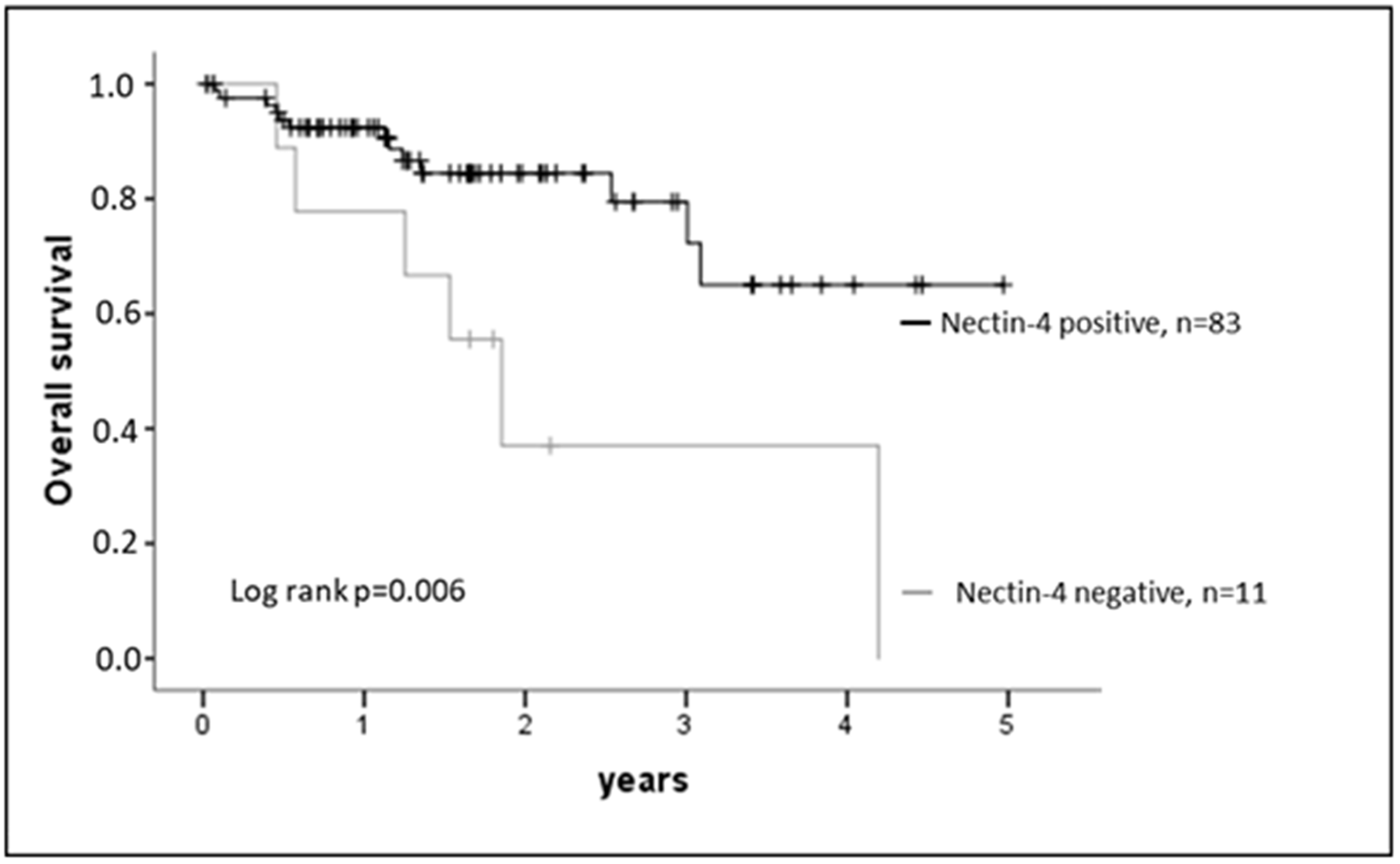
Figure 2: Nectin-4 expression and outcome. Nectin-4 expression correlates with overall survival of HNSCC patients. Log rank test p = 0.006.
Table 2: Multivariate cox regression model (inclusion)
| Hazard rate [95% confidence intervals] | Regression coefficient | p-value | p-value model | |
|---|---|---|---|---|
| pT | 1.898 [1.119–3.219] | 0.701 | 0.008 | 0.006 |
| pN | 1.623 [0.989–2.666] | 0.391 | 0.053 | |
| p16 | 0.521 [0.128–2.129] | −0.521 | 0.461 | |
| Nectin-4 | 0.418 [0.166–1.055] | −0.969 | 0.041 |
DISCUSSION
Squamous cell carcinomas of the head and neck region are among the ten most commonly diagnosed malignancies in men in the Western world (Cancer Statistics, 2021). Still, the survival rate for HNSCC has improved only modestly over the past three decades [21] and new therapy options are needed. Nectin-4 is the target molecule of an antibody-drug conjugate (Enfortumab vedotin, EV) that has been FDA-approved for the treatment of metastatic urothelial carcinoma following progression on a platinum-containing chemotherapy and immune checkpoint blockade. Response rates to EV in this tumor type are encouraging (Rosenberg, 2019). This may be explained by the high expression rates of Nectin-4 in urothelial carcinomas. The positive clinical responses seen to EV therapy in eligible urothelial carcinoma cases encourage to identify further fields of application in other tumors.
Nectins are members of the immunoglobulin superfamily and play a role in E-cadherin-based cell adhesion of epithelia [22, 23].
Nectin-4 is relative widely expressed in solid tumors. In an immunohistochemistry based screening study, Chalitta-Eid et al. found positivity of Nectin-4 in bladder cancer (83%), and other solid cancers including HNSCC, which was positive in 59% of cases [6]. We found a slightly higher rate of Nectin-4 positivity, with only 13.7% negative cases, but this difference may be ascribed to different sensitivities of the immunohistochemistry assays. In our cohort Nectin-4 was mostly expressed at low or medium levels, which matches previous data [6].
To our knowledge we are the first to analyze Nectin-4 protein expression levels in HNSCC in correlation with clinico-pathological data. We did not observe an association of Nectin-4 expression with tumor stage, lymph node involvement or grading. This matches observations in other tumor entities (e.g., gastric cancer, ovarian cancer) [24, 25]. In gastric cancer high Nectin-4 expression was associated with higher tumor stage, lymph node metastases and poorer grading [26]. In breast cancer some studies link Nectin-4 expression with lower tumor stage and less lymph node involvement [27].
In this study, significant associations of Nectin-4 expression with gender, smoking and p16 status could be demonstrated. Higher Nectin-4 expression was found in the group of p16/ HPV positive HNSCC and non-smokers. This is of interest, as the mutational profile of HPV positive and negative HPV HNSCC differs significantly [28, 29]. HPV positive HNSCC show a better therapeutic response and survival [30, 31].
Several studies have linked Nectin-4 expression to disease progression. A poorer prognosis of patients with Nectin-4 positive tumors has been reported for bladder cancer [6] and esophageal cancer [32], whereas the data for breast cancer appears to be inconclusive so far [33, 27]. In this study, Nectin-4 positive HNSCC cases showed longer survival times. One may assume that this may be due to the positive, but weak correlation of Nectin-4 with p16, as p16 positive tumors have clinically a better prognosis. However, the multivariable analysis including p16 as a prognostic variable confirms an independent prognostic value of Nectin-4, which ought to be validated in larger cohorts.
The better prognosis of Nectin-4 positive HNSCC matches results of Tanaka et al.: In in vitro studies of cutaneous squamous cell carcinoma silencing of Nectin-4 lead to increased cell migration and inhibited proliferation [3]. In other tumor entities like ovarian cancer in vitro studies also suggest a link between Nectin-4 and proliferation and migration, but the exact effects were partly contradictory [34]. Another possible explanation for the better prognosis of Nectin-4 positive HNSCC could be due to the immune modulatory function of Nectin-4. Nectins are ligands of the inhibitory receptor T-cell immunoreceptor with Ig and ITIM domains (TIGIT), a inhibitory checkpoint receptor, which is expressed on most natural killer cells cells and multiple T cell subsets, including CD8+ tumor infiltrating lymphocytes (TILs) [4, 5]. Further studies are needed to evaluate the function of Nectin-4 in HNSCC.
In summary, this study confirms a high positivity rate of HNSCC for Nectin-4, which supports to explore its value as a target for EV therapy. It needs to be clarified in further studies, which cases qualify for this type of experimental therapy, as this is not without side effects [35].
Materials and Methods
Patient population and tumor specimens
Evaluation of Nectin-4 protein expression was performed on tissue microarrays (TMA) constructed from a cohort of 159 clinically annotated HNSCC patients treated surgically with curative or palliative intent between 2011 and 2014 at the University Hospital Bonn as described previously [36]. The study was approved by the institutional review board of the University of Bonn (#148/11) and the ethic committee of the University Hospital Bonn.
Immunohistochemistry
Immunohistochemical staining was conducted as described previously [36]. Nectin-4 antibody was used at a dilution of 1:1,000 (clone EPR15613-68, dilution 1:1,000, Abcam, Cambridge, UK). All supplementary reagents used for Nectin-4 staining were obtained Ventana Medical Systems. All tissue sections were counterstained with Mayer´s hematoxylin (Merck, Darmstadt, Germany). CS and JFL assessed Nectin-4 protein expression independently. Staining intensity was classified into four categories: no expression (0), low expression (1), medium expression (2) and high expression (3) (Figure 1).
Statistics
Statistical analyses were carried out using the SPSS 25 software package (IBM, Armonk, NY). Comparisons between variable clinical pathological groups were made using the Mann–Whitney test and the Kruskal–Wallis test. Correlation analyses between variable groups were determined by Spearman’s rank correlation coefficient. Survival time was estimated by Kaplan-Meier analyses and compared among patient subsets using log-rank test, multivariate regression model was performed using Cox regression. All statistical tests were two-sided. P-values under 0.05 were considered significant.
ACKNOWLEDGMENTS
The authors thank Susanne Steiner and Carsten Golletz (Institute of Pathology, University Hospital Bonn) for their excellent technical assistance.
CONFLICTS OF INTEREST
The authors have no conflicts of interest to declare.
Ethical statement and consent
The study was approved by the institutional review board of the University of Bonn (#148/11) and the ethic committee of the University Hospital Bonn.
FUNDING
This research did not receive grants from any funding agency in the public, commercial or not-for-profit sectors.
References
1. Rikitake Y, Mandai K, Takai Y. The role of nectins in different types of cell-cell adhesion. J Cell Sci. 2012; 125:3713–22. https://doi.org/10.1242/jcs.099572. [PubMed].
2. Takai Y, Miyoshi J, Ikeda W, Ogita H. Nectins and nectin-like molecules: roles in contact inhibition of cell movement and proliferation. Nat Rev Mol Cell Biol. 2008; 9:603–15. https://doi.org/10.1038/nrm2457. [PubMed].
3. Tanaka Y, Murata M, Oda Y, Furue M, Ito T. Nectin Cell Adhesion Molecule 4 (NECTIN4) Expression in Cutaneous Squamous Cell Carcinoma: A New Therapeutic Target? Biomedicines. 2021; 9:355. https://doi.org/10.3390/biomedicines9040355. [PubMed].
4. Reches A, Ophir Y, Stein N, Kol I, Isaacson B, Charpak Amikam Y, Elnekave A, Tsukerman P, Kucan Brlic P, Lenac T, Seliger B, Jonjic S, Mandelboim O. Nectin4 is a novel TIGIT ligand which combines checkpoint inhibition and tumor specificity. J Immunother Cancer. 2020; 8:e000266. https://doi.org/10.1136/jitc-2019-000266. [PubMed].
5. Yeo J, Ko M, Lee DH, Park Y, Jin HS. TIGIT/CD226 Axis Regulates Anti-Tumor Immunity. Pharmaceuticals (Basel). 2021; 14:200. https://doi.org/10.3390/ph14030200. [PubMed].
6. Challita-Eid PM, Satpayev D, Yang P, An Z, Morrison K, Shostak Y, Raitano A, Nadell R, Liu W, Lortie DR, Capo L, Verlinsky A, Leavitt M, et al. Enfortumab Vedotin Antibody-Drug Conjugate Targeting Nectin-4 Is a Highly Potent Therapeutic Agent in Multiple Preclinical Cancer Models. Cancer Res. 2016; 76:3003–13. https://doi.org/10.1158/0008-5472.CAN-15-1313. [PubMed].
7. Rosenberg JE, O’Donnell PH, Balar AV, McGregor BA, Heath EI, Yu EY, Galsky MD, Hahn NM, Gartner EM, Pinelli JM, Liang SY, Melhem-Bertrandt A, Petrylak DP. Pivotal Trial of Enfortumab Vedotin in Urothelial Carcinoma After Platinum and Anti-Programmed Death 1/Programmed Death Ligand 1 Therapy. J Clin Oncol. 2019; 37:2592–600. https://doi.org/10.1200/JCO.19.01140. [PubMed].
8. Alt M, Stecca C, Tobin S, Jiang DM, Sridhar SS. Enfortumab Vedotin in urothelial cancer. Ther Adv Urol. 2020; 12:1–10. https://doi.org/10.1177/1756287220980192. [PubMed].
9. Hanna KS. Clinical Overview of Enfortumab Vedotin in the Management of Locally Advanced or Metastatic Urothelial Carcinoma. Drugs. 2020; 80:1–7. https://doi.org/10.1007/s40265-019-01241-7. [PubMed].
10. Rosenberg J, Sridhar SS, Zhang J, Smith D, Ruether D, Flaig TW, Baranda J, Lang J, Plimack ER, Sangha R, Heath EI, Merchan J, Quinn DI, et al. EV-101: A Phase I Study of Single-Agent Enfortumab Vedotin in Patients With Nectin-4-Positive Solid Tumors, Including Metastatic Urothelial Carcinoma. J Clin Oncol. 2020; 38:1041–49. https://doi.org/10.1200/JCO.19.02044. [PubMed].
11. Heath EI, Rosenberg JE. The biology and rationale of targeting nectin-4 in urothelial carcinoma. Nat Rev Urol. 2021; 18:93–103. https://doi.org/10.1038/s41585-020-00394-5. [PubMed].
12. Yoshihara K, Wang Q, Torres-Garcia W, Zheng S, Vegesna R, Kim H, Verhaak RG. The landscape and therapeutic relevance of cancer-associated transcript fusions. Oncogene. 2015; 34:4845–54. https://doi.org/10.1038/onc.2014.406. [PubMed].
13. Moskovitz J, Moy J, Ferris RL. Immunotherapy for Head and Neck Squamous Cell Carcinoma. Curr Oncol Rep. 2018; 20:22. https://doi.org/10.1007/s11912-018-0654-5. [PubMed].
14. Argiris A, Karamouzis MV, Raben D, Ferris RL. Head and neck cancer. Lancet. 2008; 371:1695–709. https://doi.org/10.1016/S0140-6736(08)60728-X. [PubMed].
15. Kao HF, Lou PJ. Immune checkpoint inhibitors for head and neck squamous cell carcinoma: Current landscape and future directions. Head Neck. 2019 (Suppl 1); 41:4–18. https://doi.org/10.1002/hed.25930. [PubMed].
16. Vermorken JB, Mesia R, Rivera F, Remenar E, Kawecki A, Rottey S, Erfan J, Zabolotnyy D, Kienzer HR, Cupissol D, Peyrade F, Benasso M, Vynnychenko I, et al. Platinum-based chemotherapy plus cetuximab in head and neck cancer. N Engl J Med. 2008; 359:1116–27. https://doi.org/10.1056/NEJMoa0802656. [PubMed].
17. Cohen EEW, Bell RB, Bifulco CB, Burtness B, Gillison ML, Harrington KJ, Le QT, Lee NY, Leidner R, Lewis RL, Licitra L, Mehanna H, Mell LK, et al. The Society for Immunotherapy of Cancer consensus statement on immunotherapy for the treatment of squamous cell carcinoma of the head and neck (HNSCC). J Immunother Cancer. 2019; 7:184. https://doi.org/10.1186/s40425-019-0662-5. [PubMed].
18. Ferris RL, Blumenschein G Jr, Fayette J, Guigay J, Colevas AD, Licitra L, Harrington K, Kasper S, Vokes EE, Even C, Worden F, Saba NF, Iglesias Docampo LC, et al. Nivolumab for Recurrent Squamous-Cell Carcinoma of the Head and Neck. N Engl J Med. 2016; 375:1856–67. https://doi.org/10.1056/NEJMoa1602252. [PubMed].
19. Patel JJ, Levy DA, Nguyen SA, Knochelmann HM, Day TA. Impact of PD-L1 expression and human papillomavirus status in anti-PD1/PDL1 immunotherapy for head and neck squamous cell carcinoma-Systematic review and meta-analysis. Head Neck. 2020; 42:774–86. https://doi.org/10.1002/hed.26036. [PubMed].
20. Saâda-Bouzid E, Defaucheux C, Karabajakian A, Coloma VP, Servois V, Paoletti X, Even C, Fayette J, Guigay J, Loirat D, Peyrade F, Alt M, Gal J, Le Tourneau C. Hyperprogression during anti-PD-1/PD-L1 therapy in patients with recurrent and/or metastatic head and neck squamous cell carcinoma. Ann Oncol. 2017; 28:1605–11. https://doi.org/10.1093/annonc/mdx178. [PubMed].
21. Johnson DE, Burtness B, Leemans CR, Lui VWY, Bauman JE, Grandis JR. Head and neck squamous cell carcinoma. Nat Rev Dis Primers. 2020; 6:92. https://doi.org/10.1038/s41572-020-00224-3. [PubMed].
22. Fabre S, Reymond N, Cocchi F, Menotti L, Dubreuil P, Campadelli-Fiume G, Lopez M. Prominent role of the Ig-like V domain in trans-interactions of nectins. Nectin3 and nectin 4 bind to the predicted C-C’-C”-D beta-strands of the nectin1 V domain. J Biol Chem. 2002; 277:27006–13. https://doi.org/10.1074/jbc.M203228200. [PubMed].
23. Reymond N, Fabre S, Lecocq E, Adelaïde J, Dubreuil P, Lopez M. Nectin4/PRR4, a new afadin-associated member of the nectin family that trans-interacts with nectin1/PRR1 through V domain interaction. J Biol Chem. 2001; 276:43205–15. https://doi.org/10.1074/jbc.M103810200. [PubMed].
24. Derycke MS, Pambuccian SE, Gilks CB, Kalloger SE, Ghidouche A, Lopez M, Bliss RL, Geller MA, Argenta PA, Harrington KM, Skubitz AP. Nectin 4 overexpression in ovarian cancer tissues and serum: potential role as a serum biomarker. Am J Clin Pathol. 2010; 134:835–45. https://doi.org/10.1309/AJCPGXK0FR4MHIHB. [PubMed].
25. Tomiyama E, Fujita K, Rodriguez Pena MDC, Taheri D, Banno E, Kato T, Hatano K, Kawashima A, Ujike T, Uemura M, Takao T, Yamaguchi S, Fushimi H, et al. Expression of Nectin-4 and PD-L1 in Upper Tract Urothelial Carcinoma. Int J Mol Sci. 2020; 21:5390. https://doi.org/10.3390/ijms21155390. [PubMed].
26. Zhang Y, Zhang J, Shen Q, Yin W, Huang H, Liu Y, Ni Q. High expression of Nectin-4 is associated with unfavorable prognosis in gastric cancer. Oncol Lett. 2018; 15:8789–95. https://doi.org/10.3892/ol.2018.8365. [PubMed].
27. Zeindler J, Soysal SD, Piscuoglio S, Ng CKY, Mechera R, Isaak A, Weber WP, Muenst S, Kurzeder C. Nectin-4 Expression Is an Independent Prognostic Biomarker and Associated With Better Survival in Triple-Negative Breast Cancer. Front Med (Lausanne). 2019; 6:200. https://doi.org/10.3389/fmed.2019.00200. [PubMed].
28. Leemans CR, Snijders PJF, Brakenhoff RH. The molecular landscape of head and neck cancer. Nat Rev Cancer. 2018; 18:269–82. https://doi.org/10.1038/nrc.2018.11. [PubMed].
29. Seiwert TY, Zuo Z, Keck MK, Khattri A, Pedamallu CS, Stricker T, Brown C, Pugh TJ, Stojanov P, Cho J, Lawrence MS, Getz G, Brägelmann J, et al. Integrative and comparative genomic analysis of HPV-positive and HPV-negative head and neck squamous cell carcinomas. Clin Cancer Res. 2015; 21:632–41. https://doi.org/10.1158/1078-0432.CCR-13-3310. [PubMed].
30. Fakhry C, Westra WH, Li S, Cmelak A, Ridge JA, Pinto H, Forastiere A, Gillison ML. Improved survival of patients with human papillomavirus-positive head and neck squamous cell carcinoma in a prospective clinical trial. J Natl Cancer Inst. 2008; 100:261–69. https://doi.org/10.1093/jnci/djn011. [PubMed].
31. Yakin M, Seo B, Hussaini H, Rich A, Hunter K. Human papillomavirus and oral and oropharyngeal carcinoma: the essentials. Aust Dent J. 2019; 64:11–18. https://doi.org/10.1111/adj.12652. [PubMed].
32. Lin X, Hu H, Pan Y, Gao S. The Prognostic Role of Expression of Nectin-4 in Esophageal Cancer. Med Sci Monit. 2019; 25:10089–94. https://doi.org/10.12659/MSM.918288. [PubMed].
33. M-Rabet M, Cabaud O, Josselin E, Finetti P, Castellano R, Farina A, Agavnian-Couquiaud E, Saviane G, Collette Y, Viens P, Gonçalves A, Ginestier C, Charafe-Jauffret E, et al. Nectin-4: a new prognostic biomarker for efficient therapeutic targeting of primary and metastatic triple-negative breast cancer. Ann Oncol. 2017; 28:769–76. https://doi.org/10.1093/annonc/mdw678. [PubMed].
34. Boylan KL, Buchanan PC, Manion RD, Shukla DM, Braumberger K, Bruggemeyer C, Skubitz AP. The expression of Nectin-4 on the surface of ovarian cancer cells alters their ability to adhere, migrate, aggregate, and proliferate. Oncotarget. 2017; 8:9717–38. https://doi.org/10.18632/oncotarget.14206. [PubMed].
35. Viscuse PV, Marques-Piubelli ML, Heberton MM, Parra ER, Shah AY, Siefker-Radtke A, Gao J, Goswami S, Ivan D, Curry JL, Campbell MT. Case Report: Enfortumab Vedotin for Metastatic Urothelial Carcinoma: A Case Series on the Clinical and Histopathologic Spectrum of Adverse Cutaneous Reactions From Fatal Stevens-Johnson Syndrome/Toxic Epidermal Necrolysis to Dermal Hypersensitivity Reaction. Front Oncol. 2021; 11:621591. https://doi.org/10.3389/fonc.2021.621591. [PubMed].
36. Müller T, Braun M, Dietrich D, Aktekin S, Höft S, Kristiansen G, Göke F, Schröck A, Brägelmann J, Held SAE, Bootz F, Brossart P. PD-L1: a novel prognostic biomarker in head and neck squamous cell carcinoma. Oncotarget. 2017; 8:52889–900. https://doi.org/10.18632/oncotarget.17547. [PubMed].

