Introduction
When treating recurrent breast cancer, therapeutic agents are commonly selected on the basis of hormone receptor (HR) and human epidermal growth factor receptor 2 (HER2) protein expression. These molecular markers are well established as useful predictors of therapeutic effect in recurrent breast cancer treatment, similar to in primary breast cancer. The American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) guidelines recommended that hormone receptors and HER2 are tested in tissue specimens obtained from recurrence or metastases in breast cancer patients [1–4]. This is due to the possibility that the receptor state may change during the progress from primary to recurrent tumor [5–8].
However, the collection of tissue specimens from metastatic or recurrent foci is often difficult. Cytological analysis can be applied to metastatic lesion specimens, such as body cavity fluids and sites where tissue biopsy is difficult. Therefore, cytological diagnosis is useful for pathological diagnosis of cancer metastasis. In addition, applying cytological analysis to receptor examination may provide a useful treatment guide for a selected relapsed patient.
In order to examine the breast cancer receptor using a cytology specimen, it is necessary to prepare multiple slides containing cancer cells. For this, we adopted the cell block (CB) method. There are already some reports of receptor examination using breast cancer cytology specimens, and for hormone receptors, the agreement rate between the cytological and tissue specimen is good [9–13]. For the HER2 test, it is reported that the agreement rate with the tissue specimen is improved by adding the dual in situ hybridization (DISH) assay for the case of HER2 2+ by immunostaining [14–16].
In addition, receptor testing using cytological specimens can be a quick, inexpensive, and less invasive alternative when compared to methods using tissue specimens.
As part of a multi-institutional study by the National Hospital Organization, we conducted a study to examine hormone receptors using cytological specimens from breast cancer metastases.
We adopted the receptor examination method in cytological specimens using CBs unified in multiple institutions, and stable test results were obtained [17]. Furthermore, in order to confirm whether receptor assessment in breast cancer metastatic lesions is useful for determining a breast cancer treatment strategy, we analyzed the treatment course of metastatic breast cancer patients registered for this multi-institutional study.
Results
Registered specimens
During the registration period, the number of registrations in which patient consent was obtained was 62. For these breast cancer metastasis foci, receptor examination using CB was performed. The background of the cases is shown in Table 1.
Table 1: Patients characteristics
| Variable | Number |
|---|---|
| Age (mean ±SD) | 61.7 ± 11.0 |
| Clinical Stage at diagnosis | |
| I | 5 |
| II | 29 |
| III | 13 |
| IV | 10 |
| unknown | 5 |
| Metastatic lesion | |
| Pleural fluid | 44 |
| Ascites | 14 |
| Lymph node | 2 |
| Pericardial fluid | 1 |
| Subcutaneous metastatic nodule on the back | 1 |
| Primary lesion of breast | |
| Solitary | 57 |
| Synchronous multiple lesion | 4 |
| Metachronous multiple lesion | 1 |
| Time of distant metastasis | |
| Metachronous metastasis | 56 |
| Synchronous metastasis | 6 |
All patients were females aged 40 to 80 years (average 61.7 years old, median 63 years old). The types of metastatic lesions were pleural effusion (44 specimen), ascites (14 specimen), lymph node (2 specimen), pericardial effusion (1 specimen), and dorsal subcutaneous mass (1 specimen).
Difference in receptor expression between primary tumor and metastatic lesion
The status of receptor expression in the primary tumor and metastatic lesion was compiled in a paired sample (Table 2).
Table 2: Discordance rate of ER, PR and HER2 expression between primary and metastatic lesion
| Metastatic lesion | Discordance | (95% CI) | |||
|---|---|---|---|---|---|
| ER | positive | negative | |||
| Primary lesion | positive | 35 | 7 | ||
| negative | 3 | 10 | 10/55(18.2%) | (7.9–28.5%) | |
| PR | positive | negative | |||
| Primary lesion | positive | 16 | 17 | ||
| negative | 3 | 19 | 20/55(36.4%) | (23.7–49.1%) | |
| HER2 | positive | negative | |||
| Primary lesion | positive | 3 | 3 | ||
| negative | 1 | 42 | 4/49(8.2%) | (0.1–16.3%) | |
In ER and PR, there was a difference in receptor expression between the primary tumor and metastatic lesion in 10 (18.2% (95% confidence interval (CI): 7.9–28.8%)) and 20 specimens (36.4% (95% CI: 23.7–49.1%)) respectively, of 55 specimens. HER2 protein expression did not agree in 4 out of 49 specimens (8.2% (95% CI: 0.1–16.3%)) between the primary tumor and metastatic lesion.
In a comparison based on four groups of primary/metastatic receptor status (positive/positive, positive/negative, negative/negative, negative/positive), patients who changed from primary negative to metastatic positive ER status had taken a significantly longer time for metastatic foci to appear (Table 3). In addition, many patients with metastatic negative ER and PR from primary positive status received hormonal therapy until registration, either as postoperative adjuvant therapy or treatment after recurrence (Table 4). Similarly, in patients with metastatic negative HER2 from primary positive status, anti-HER2 therapy was administered as a recurrence treatment before registration.
Table 3: Relationship between expression status of ER, PR, HER and the interval until re-examination for metastatic lesion
| Status (primary/metastatic) | Number | Interval (month) | |
|---|---|---|---|
| ER | Negative/negative | 10 | 58.7 |
| Negative/positive | 3 | 187.3* | |
| Positive/positive | 35 | 85.9 | |
| Positive/negative | 7 | 89.1 | |
| PR | Negative/negative | 19 | 67.7 |
| Negative/positive | 3 | 161.7 | |
| Positive/positive | 16 | 78.3 | |
| Positive/negative | 17 | 103.3 | |
| HER2 | Negative/negative | 42 | 87.9 |
| Negative/positive | 1 | 90.0 | |
| Positive/positive | 3 | 35.3 | |
| Positive/negative | 3 | 47.3 |
Table 4: Relationship between expression status of ER, PR, HER and the previous treatment
| Status (primary/metastatic) | Adjuvant | For metastasis | |
|---|---|---|---|
| Endocrine treatment | Endocrine treatment | ||
| ER | Negative/negative | 0/10(0%) | 0/10(0%) |
| Negative/positive | 1/3(33.3%) | 2/3(66.7%) | |
| Positive/positive | 24/35(68.6%) | 25/35(71.4%) | |
| Positive/negative | 7/7(100%) | 5/7(71.4%) | |
| PR | Negative/negative | 6/19(31.6%) | 6/19(31.6%) |
| Negative/positive | 1/3(33.3%) | 3/3(100%) | |
| Positive/positive | 11/16(68.7%) | 9/16(56.3%) | |
| Positive/negative | 14/17(82.4%) | 13/17(76.4%) | |
| Trastuzumab | Trastuzumab | ||
| HER2 | Negative/negative | 2/42(4.8%) | 2/42(4.8%) |
| Negative/positive | 0/1(0%) | 0/1(0%) | |
| Positive/positive | 1/3(33.3%) | 1/3(33.3%) | |
| Positive/negative | 0/3(0%) | 2/3(66.7%) |
Outcomes of treatment based on receptor status of metastatic lesion
Among ER-positive cases in metastatic lesions, the treatment duration was significantly longer in patients who received endocrine therapy as a post-registration treatment than in those who received chemotherapy by the Log-Rank test (Figure 1).
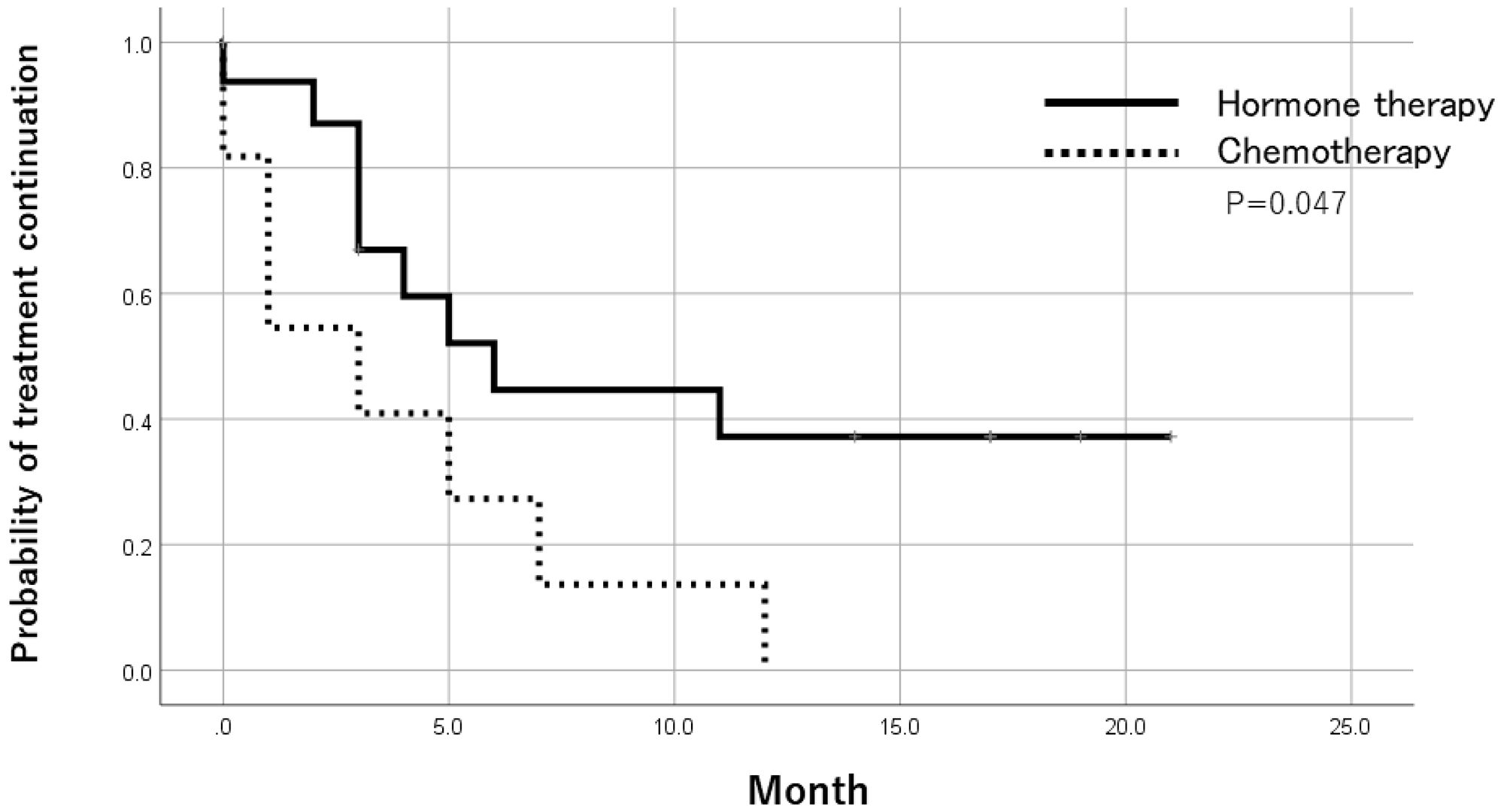
Figure 1: Time to treatment failure after trial registration by treatments in ER-positive patients. In ER-positive cases of metastatic lesions, the treatment period of patients receiving endocrine therapy was significantly longer than patients receiving chemotherapy as a post-registration treatment.
Receptor status in metastatic lesion and prognosis after registration
Patients with positive ER status in metastatic lesions had a significantly better prognosis than ER-negative cases after registration by the Log-Rank test. There was no significant difference in prognosis based on PR and HER expression (Figure 2). In addition, when the prognosis was compared between pleural metastasis, peritoneal metastasis, and others, the prognosis was significantly worse in cases registered as peritoneal metastasis by the Log-Rank test (Figure 3). Table 5 shows the result of Cox multivariate analysis including these factors. The ER status of the metastatic lesion and the metastatic site were independent prognostic factors.
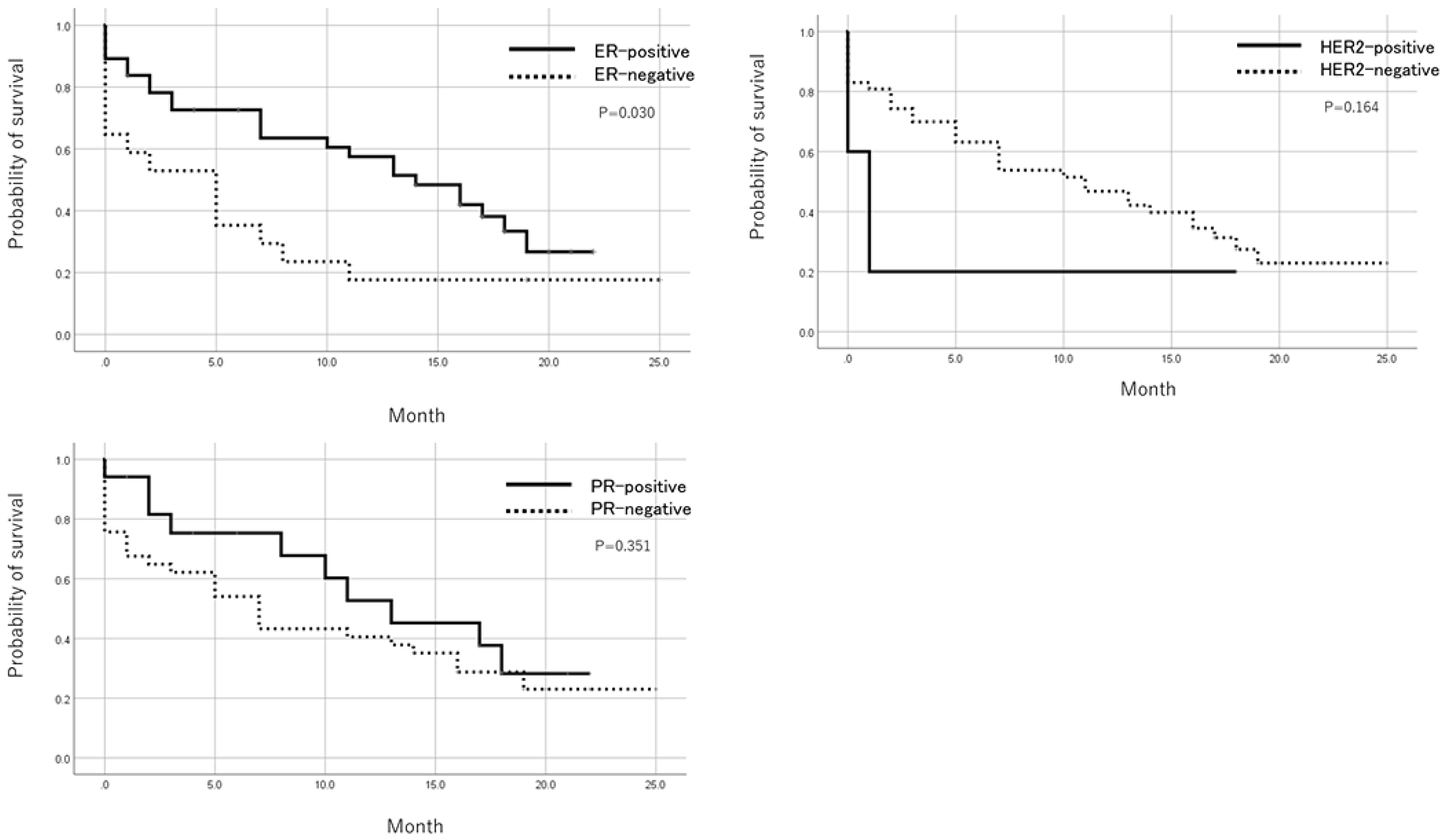
Figure 2: Prognosis after trial registration by ER, PR, and HER2 status. Patients with a positive ER status in metastatic foci showed a significantly better prognosis after registration than ER negative cases. There was no significant difference in prognosis based on PR and HER2 expression.
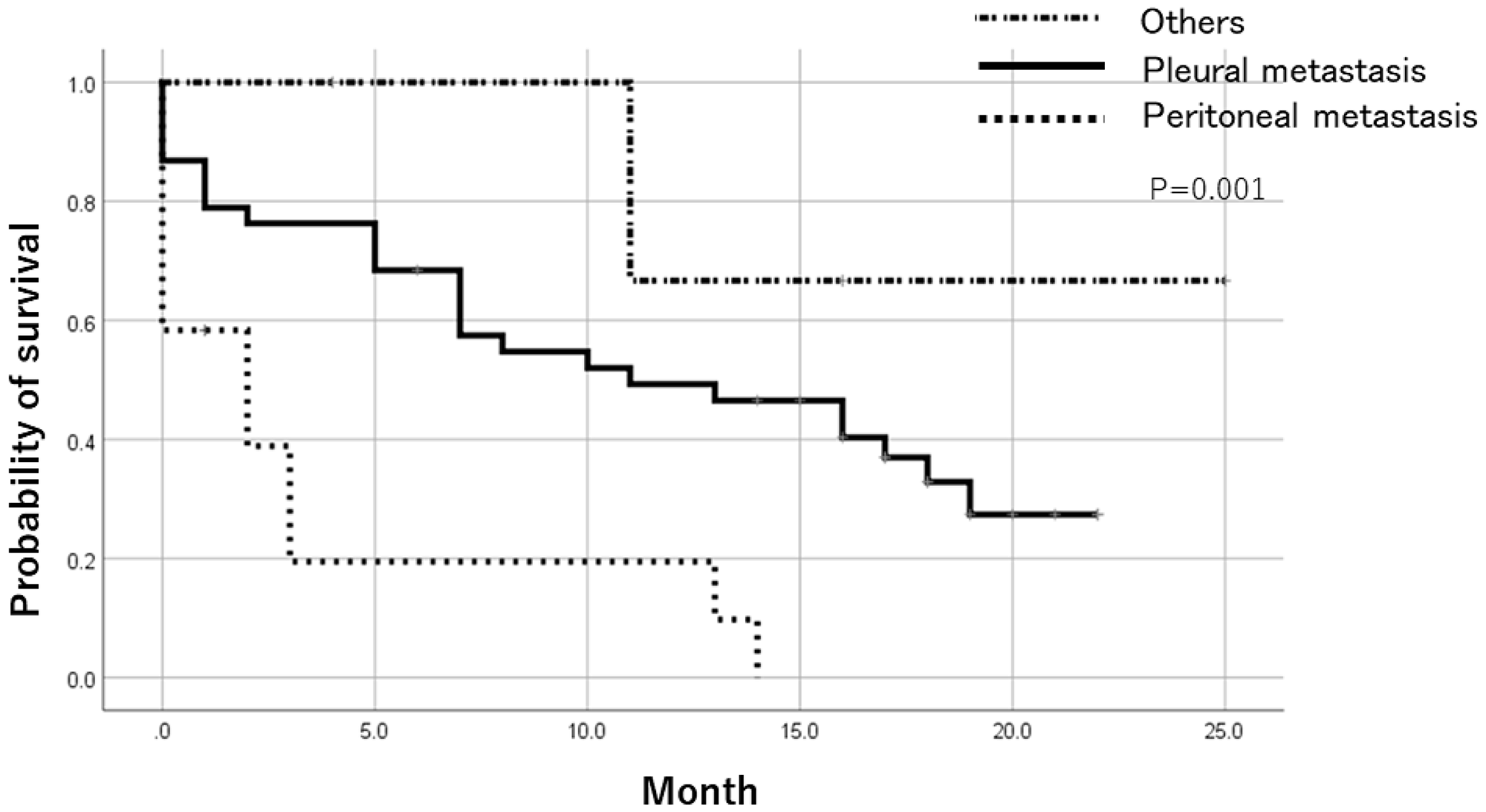
Figure 3: Prognosis after trial registration by metastatic lesion. Comparing the prognosis based on the registered metastatic sites, the prognosis was significantly worse in cases registered as peritoneal metastasis.
Table 5: Multivariate analysis of factors affecting prognosis after registration
| Factor | Hazard ratio | 95% CI | P-value |
|---|---|---|---|
| ER | 0.327 | 0.158–0.678 | 0.003 |
| Metastatic lesion | |||
| Pleural fluid | 0.217 | 0.096–0.488 | <0.001 |
| Others | 0.055 | 0.006–0.465 | 0.008 |
DISCUSSION
The receptor examination method for metastatic breast cancer using CB was easy to introduce. In institutions conducting receptor examination of tissue specimens in daily practice, the procedure is easy and cost-effective, so there are few obstacles to its introduction. Pleural effusion (44 samples) followed by ascites (14 samples) were the most common samples in this study. By examining the receptor status on cytological specimens using CBs, treatment guides were obtained for metastatic lesions that had so far been difficult to evaluate.
In the examination of tissue specimens, it is reported that the discordance rate of receptor status in the primary tumor and metastatic lesion is 12–30% in ER, 18–42% in PR, and 5–16% in HER2 [18–21]. In our study, the discordance rate was 18.2% (95% CI: 7.9–28.8%) in ER, 36.4% (95% CI: 23.7–49.1%) in PR and 8.2% (95% CI: 0.1–16.3%) in HER2, which was comparable with the reported rate. The fact that the discordance rate was similar between the cytological and tissue specimens indicates the reliability of the receptor testing in the cytological specimens.
Although direct comparison of cytology specimens and tissue specimens from breast cancer metastatic foci will prove the reliability of cytology cell block in receptor examination, it is not easy due to the difficulty of sampling metastatic lesions. Vohra examined receptor and HER2 expression in 134 breast cancer patients, compared cytology cell block and tissue samples, and showed high agreement rates, but only 18 metastatic lesions were included [22]. Because most of our target specimens were cytological preparations from liquid samples, direct comparison with tissue samples at the metastatic site was difficult.
Focusing on changes in hormone receptor expression, it was necessary to review treatment strategies. This is because ER did not coincide with the primary tumor at 18.2% of the recurrent foci. As metastatic negative change of HRs from primary positive status may occur when receiving multiple endocrine treatments as adjuvant or recurrent therapy, it is better to reconfirm the receptor status in the metastatic lesion. The discordance rate in PR was greater, with nearly half of the cases with primary positive tumors converted negative status in metastatic lesions. PR expression is controlled by ER, and PR status is most susceptible to the influence of previous endocrine treatments [23]. Additionally, there was a positive change of ER in the metastatic site from primary negative status in a few cases, in which a long time had passed until the metastatic lesion had appeared. In general, hormone-dependent breast cancer progression is slow, and in some patients, metastasis appears over a long period of time. Assuming the heterogeneity of the primary tumor, when a small number of ER-positive clones metastasize over a long period of time, it may deemed an ER-positive conversion in metastatic lesions. In patients with a long course to metastasis, it is therefore recommended to reexamine the receptor status, considering positive change of the hormone receptor.
In metastatic ER-positive cases, the endocrine therapy treatment period after registration was significantly longer compared with that of chemotherapy. Many ER-positive patients who choose chemotherapy have more advanced disease and cannot be easily compared. In the metastatic ER-positive cases, it seems preferable to give priority to endocrine treatment when the general condition is not bad.
Furthermore, metastatic ER-positive cases have better prognosis than ER-negative cases, and it is necessary to prepare a long-term treatment plan. In a clinical setting, patients with ascites due to breast cancer metastasis often progress rapidly after metastasis. Patients with peritoneal metastasis had poor prognosis in this study, consistent with actual clinical experience. Depending on the metastatic site, it is necessary to modify the treatment policy.
Reasons for receptor examination in metastatic lesions include cases in which the primary tumor’s receptor status is unknown or there are multiple primary lesions, and thus receptor testing in metastatic lesions is inevitable. In this study, although the receptor status of the primary tumor was known, there were many patients who wanted to review the treatment strategy according to the receptor status of the metastatic lesion. In cases where the administered treatment was ineffective, a metastatic lesion receptor test was used to monitor the treatment in detail. Receptors and HER2 protein expression are often more difficult to determine in cytological specimens than tissue specimens. However, it is considered an alternative examination method in cases where biopsy cannot be easily performed. Consequently, receptor examination in cytological specimens is useful, as it provides a treatment guide in metastatic breast cancer.
Materials and Methods
Nine institutions belonging to the National Hospital Organization (NHO), (Hokkaido Cancer Center, Sendai Medical Center, Tokyo Medical Center, Nagoya Medical Center, Osaka National Hospital, Kure Medical Center, Nagasaki Medical Center, Kyushu Cancer Center, and the Shikoku Cancer Center), participated in the study.
The study plan was approved by the National Hospital Organization Central Ethical Review Board. The registration period was from April 1, 2015 to March 31, 2016. Patients diagnosed with breast cancer metastasis, pathological diagnosis by cytological specimens, and further referred for receptor examination were targeted. Patients signed the informed consent form and accepted the registration. In each institution, the examination of HR and HER2 protein was performed on cytology specimens obtained from breast metastases.
Preparation of CB and staining of receptor
All participating facilities prepared CB using the same methodology. First, specimens collected from metastatic sites were fixed in 10% buffered formalin for 6 to 48 hours. Thereafter, the tube containing the sample was centrifuged at 3000 rpm for 5 minutes to remove formalin. After this, 0.5 mL of 1% sodium alginate was added, and the tube was centrifuged again at 3000 rpm for 5 minutes, after which 0.5 mL of 1 M calcium chloride was added. The gel pellets formed were embedded in paraffin to make paraffin CB. CB was then prepared in the same way for the tissue specimens.
Sections prepared from CB were stained with hematoxylin-eosin (HE) and underwent immunohistochemistry (IHC). Each facility used an autostainer for IHC staining. The HER2 DISH assay was performed using Ventana Bench Mark in the Shikoku Cancer Center. Evaluation of HR and HER2 immunostaining was performed at each institution, and evaluation of DISH was performed at the Shikoku Cancer Center.
Evaluation of HR expression
Staining for HR was evaluated as positive or negative according to the following criteria: cases where there was any nuclear staining of tumor cells were determined to be positive, and cases where there was no nuclear staining were deemed negative. The positive rate of stained tumor cells was not considered for evaluation. For cytological specimens containing certain non-neoplastic cells, it is difficult to estimate the proportion of stained cells in tumor cells.
Evaluation of HER2 protein expression
We scored staining results of the HER2 protein for 0, 1+, 2+, or 3+, according to the following criteria: 3+, strong staining of the cell membranes of tumor cells; 2 +, intermediate staining of the cell membranes of tumor cells; 1+, weakly incomplete cell membrane staining of tumor cells; 0, no staining. Again, the percentage of stained tumor cells was not considered for scoring. The expression status of the HER2 protein was classified as negative in samples scoring 0 or 1+, undetermined in the sample with a score of 2+, and positive in samples with a score of 3+. When the result of HER2 immunostaining was 2+, DISH was performed.
Evaluation of results of HER2 DISH assay
The INFORM HER2/neu double in situ hybridization DNA probe cocktail assay was used for slides prepared from CB. The DISH assay was performed according to the manufacturer’s recommended protocol for tissue specimens. To avoid subjective bias, HER2/neu (black) and chromosome enumeration of probes 17; a CEP17 (red) ratio were manually counted by two investigators under a light microscope for each sample. At least 20 cells were counted. When the HER2/CEP17 signal number ratio was 2.0 or more, or the signal number ratio was less than 2.0 but the average number of HER2 signals per cell was 6.0 or more, it was deemed amplified.
Discordance rate between the primary tumor and metastatic lesion
We compared the expression of HR and HER2 protein on pairs of samples whose expression status was confirmed in both the primary tumor and metastatic lesion, and calculated the concordance and discordance rate of expression. In addition, the expression of the primary/metastatic receptor status was divided into 4 groups (positive/positive, positive/negative, negative/negative, negative/positive). We compared the period and the details of previous treatment until re-examination of metastatic lesions.
Therapeutic effect and prognosis based on receptor expression of metastatic lesion
For patients with ER-positive metastatic lesions, the duration of treatment after registration was compared between endocrine therapy and chemotherapy. We also compared the prognosis after enrollment based on receptor expression in metastatic lesions. In addition, Cox multivariate analysis was performed on prognosis after registration, including age and metastatic ER, PR, HER2 expression and metastatic site.
Statistical analysis
SPSS Ver 25 was used for statistical analysis. For the comparison of the mean value among 4 groups, analysis of variance was used. Treatment continuation rate and survival rate were calculated by the Kaplan-Mayer method, and the difference was examined by the Log-Rank test. Multivariate prognostic analysis was performed using the Cox proportional hazards model. Significant differences were assessed at a significance level of 5%.
Abbreviations
HR: hormone receptor; HER2: human epidermal growth factor receptor 2; DISH: dual in situ hybridization; ER: estrogen receptor; PR: progesterone receptor; HE: hematoxylin-eosin; IHC: immunohistochemistry; CB: cell block; CI: confidence interval.
ACKNOWLEDGMENTS
We would like to thank the surgeons and clinical laboratory technicians of each facility for cooperation on case registration and specimen preparation. Also, I would like to thank Ms. Chiaki Terasaka of Shikoku Cancer Center for supporting data input.
CONFLICTS OF INTEREST
None of the authors have any conflicts of interest.
FUNDING
This study was supported by clinical research grants from the National Hospital Organization (H26-NHO Cancer General- 02 and H29-NHO Cancer General- 02).
References
1. Hammond ME, Hayes DF, Dowsett M, Allred DC, Hagerty KL, Badve S, Fitzgibbons PL, Francis G, Goldstein NS, Hayes M, Hicks DG, Lester S, Love R, et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. Arch Pathol Lab Med. 2010; 134:907–22. [PubMed].
2. Hammond ME, Hayes DF, Dowsett M, Allred DC, Hagerty KL, Badve S, Fitzgibbons PL, Francis G, Goldstein NS, Hayes M, Hicks DG, Lester S, Love R, et al. American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol. 2010; 28:2784–2795. https://doi.org/10.1200/JCO.2009.25.6529. [PubMed].
3. Wolff AC, Hammond ME, Hicks DG, Dowsett M, McShane LM, Allison KH, Allred DC, Bartlett JM, Bilous M, Fitzgibbons P, Hanna W, Jenkins RB, Mangu PB, et al. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol. 2013; 31:3997–4013. https://doi.org/10.1200/JCO.2013.50.9984. [PubMed].
4. Wolff AC, Hammond ME, Hicks DG, Dowsett M, McShane LM, Allison KH, Allred DC, Bartlett JM, Bilous M, Fitzgibbons P, Hanna W, Jenkins RB, Mangu PB, et al. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. Arch Pathol Lab Med. 2014; 138:241–256. https://doi.org/10.5858/arpa.2013-0953-SA. [PubMed].
5. Aurilio G, Disalvatore D, Pruneri G, Bagnardi V, Viale G, Curigliano G, Adamoli L, Munzone E, Sciandivasci A, De Vita F, Goldhirsch A, Nolè F. A meta-analysis of oestrogen receptor, progesterone receptor and human epidermal growth factor receptor 2 discordance between primary breast cancer and metastases. Eur J Cancer. 2014; 50:277–289. https://doi.org/10.1016/j.ejca.2013.10.004. [PubMed].
6. Broom RJ, Tang PA, Simmons C, Bordeleau L, Mulligan AM, O’Malley FP, Miller N, Andrulis IL, Brenner DM, Clemons MJ. Changes in estrogen receptor, progesterone receptor and Her-2/neu status with time: discordance rates between primary and metastatic breast cancer. Anticancer Res. 2009; 29:1557–1562. [PubMed].
7. Niikura N, Liu J, Hayashi N, Mittendorf EA, Gong Y, Palla SL, Tokuda Y, Gonzalez-Angulo AM, Hortobagyi GN, Ueno NT. Loss of human epidermal growth factor receptor 2 (HER2) expression in metastatic sites of HER2-overexpressing primary breast tumors. J Clin Oncol. 2012; 30:593–599. https://doi.org/10.1200/JCO.2010.33.8889. [PubMed].
8. Yeung C, Hilton J, Clemons M, Mazzarello S, Hutton B, Haggar F, Addison CL, Kuchuk I, Zhu X, Gelmon K, Arnaout A. Estrogen, progesterone, and HER2/neu receptor discordance between primary and metastatic breast tumours-a review. Cancer Metastasis Rev. 2016; 35:427–437. https://doi.org/10.1007/s10555-016-9631-3. [PubMed].
9. Williams SL, Birdsong GG, Cohen C, Siddiqui MT. Immunohistochemical detection of estrogen and progesterone receptor and HER2 expression in breast carcinomas: comparison of cell block and tissue block preparations. Int J Clin Exp Pathol. 2009; 2:476–480. [PubMed].
10. Hanley KZ, Siddiqui MT, Lawson D, Cohen C, Nassar A. Evaluation of new monoclonal antibodies in detection of estrogen receptor, progesterone receptor, and Her2 protein expression in breast carcinoma cell block sections using conventional microscopy and quantitative image analysis. Diagn Cytopathol. 2009; 37:251–257. https://doi.org/10.1002/dc.20989. [PubMed].
11. Shabaik A, Lin G, Peterson M, Hasteh F, Tipps A, Datnow B, Weidner N. Reliability of Her2/neu, estrogen receptor, and progesterone receptor testing by immunohistochemistry on cell block of FNA and serous effusions from patients with primary and metastatic breast carcinoma. Diagn Cytopathol. 2011; 39:328–332. https://doi.org/10.1002/dc.21389. [PubMed].
12. Kumar SK, Gupta N, Rajwanshi A, Joshi K, Singh G. Immunochemistry for oestrogen receptor, progesterone receptor and HER2 on cell blocks in primary breast carcinoma. Cytopathology. 2012; 23:181–186. https://doi.org/10.1111/j.1365-2303.2011.00853.x. [PubMed].
13. Bueno Angela SP, Viero RM, Soares CT. Fine needle aspirate cell blocks are reliable for detection of hormone receptors and HER-2 by immunohistochemistry in breast carcinoma. Cytopathology. 2013; 24:26–32. https://doi.org/10.1111/j.1365-2303.2011.00934.x. [PubMed].
14. Nishimura R, Okamoto N, Satou M, Kojima K, Tanaka S. HER 2 immunohistochemistry for breast cancer cell blocks can be used in the same way as that used for histological specimens. Diagn Cytopathol. 2016; 44:274–279. https://doi.org/10.1002/dc.23433. [PubMed].
15. Nishimura R, Okamoto N, Satou M, Kojima K, Tanaka S, Yamashita N. Bright-field HER2 dual in situ hybridization (DISH) assay on breast cancer cell blocks: a comparative study with histological sections. Breast Cancer. 2016; 23:917–921. https://doi.org/10.1007/s12282-015-0664-1. [PubMed].
16. Kinsella MD, Birdsong GG, Siddiqui MT, Cohen C, Hanley KZ. Immunohistochemical detection of estrogen receptor, progesterone receptor and human epidermal growth factor receptor 2 in formalin-fixed breast carcinoma cell block preparations: correlation of results to corresponding tissue block (needle core and excision) samples. Diagn Cytopathol. 2013; 41:192–198. https://doi.org/10.1002/dc.21815. [PubMed].
17. Nishimura R, Murata Y, Mori K, Yamashiro K, Kuraoka K, Ichihara S, Taguchi K, Suzuki H, Ito M, Yamashita N. Evaluation of the HER2 and Hormone Receptor Status in Metastatic Breast Cancer Using Cell Blocks: A Multi-Institutional Study. Acta Cytol. 2018; 62:288–294. https://doi.org/10.1159/000488382. [PubMed].
18. Hou Y, Shen R, Chaudhary S, Gao F, Li Z. Correlation of Expression of Breast Biomarkers in Primary and Metastatic Breast Carcinomas: A Single-Institution Experience. Acta Cytol. 2016; 60:481–489. https://doi.org/10.1159/000449400. [PubMed].
19. Lower EE, Khan S, Kennedy D, Baughman RP. Discordance of the estrogen receptor and HER-2/neu in breast cancer from primary lesion to first and second metastatic site. Breast Cancer (Dove Med Press). 2017; 9:515–520. https://doi.org/10.2147/BCTT.S137709. [PubMed].
20. Meng X, Song S, Jiang ZF, Sun B, Wang T, Zhang S, Wu S. Receptor conversion in metastatic breast cancer: a prognosticator of survival. Oncotarget. 2016; 7:71887–71903. https://doi.org/10.18632/oncotarget.12114. [PubMed].
21. Monaco SE, Wu Y, Teot LA, Cai G. Assessment of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) status in the fine needle aspirates of metastatic breast carcinomas. Diagn Cytopathol. 2013; 41:308–315. https://doi.org/10.1002/dc.21841. [PubMed].
22. Vohra P, Buelow B, Chen YY, Serrano M, Vohra MS, Berry A, Ljung BM. Estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 expression in breast cancer FNA cell blocks and paired histologic specimens: A large retrospective study. Cancer Cytopathol. 2016; 124:828–835. https://doi.org/10.1002/cncy.21745. [PubMed].
23. Schrijver WAME, Schuurman K, van Rossum A, Peeters T, Ter Hoeve N, Zwart W, van Diest PJ, Moelans CB; Dutch Distant Breast Cancer Metastases Consortium. Loss of steroid hormone receptors is common in malignant pleural and peritoneal effusions of breast cancer patients treated with endocrine therapy. Oncotarget. 2017; 8:55550–55561. https://doi.org/10.18632/oncotarget.15548. [PubMed].

