INTRODUCTION
Laparoscopic surgery is a procedure largely preferred than traditional surgery because of its several advantages including slight hemorrhage, less body injury due to small incisions and shorter recovery time [1]. Nevertheless, this procedure is not free from adverse effects and many of them are still under investigation.
To perform laparoscopic surgery the insufflation of a gas is required to induce pneumoperitoneum that provide enough working space inside the abdomen and a better visibility of the internal organs and tissues. At present, carbon dioxide (CO2) is the most commonly used gas to induce pneumoperitoneum [2]. The usual pressure value (11–14mmHg) of pneumoperitoneum is higher than the normal portal blood pressure of 7–10 mmHg [3]. High pressure can cause reduction in liver blood flow which results in oxidative stress and tissue injury [4, 5]. The deflation restores the normal liver blood circulation, but it results in ischemia-reperfusion injury [6].
Liver ischemia induces oxidative stress and reactive oxygen species (ROS) are generated during reperfusion, resulting in a serious liver injury [7]. In addition to oxidative stress, the mechanisms of ischemia reperfusion injury include cell apoptosis and cytokine/chemokine release [8–10]. Although ischemia reperfusion injury is due to multiple factors, ROS play a key role in inducing this injury [11–13]. Several studies show that hydrogen reduces ROS by exerting an antioxidant action, thus protecting against ischemia-reperfusion injury [14–16]. As a therapeutic medical gas, hydrogen can mitigate many diseases including parkinson’s disease, acute pancreatitis, obstructive jaundice and sepsis [17–20]. Moreover, hydrogen protects against tissue damage induced by ischemia-reperfusion by reducing oxidative stress, cell apoptosis and inflammatory cytokines release [21–24]. However, no studies evaluating the protective effects of hydrogen to reduce liver injury during pneumoperitoneum are available. Therefore, the present study was performed to investigate the protective effects of hydrogen on liver during pneumoperitoneum.
RESULTS
Hypodermic injection of hydrogen gas protected against liver injury induced by pneumoperitoneum
Serum AST and ALT levels increased in P15 and H2 group compared with C group at 2 h and 6 h. However, their levels were significantly reduced in H2 group compared with P15 group (P < 0.05, Figure 1A and 1B). AST and ALT levels in liver tissue showed the same trend (P < 0.05, Figure 1C and 1D).
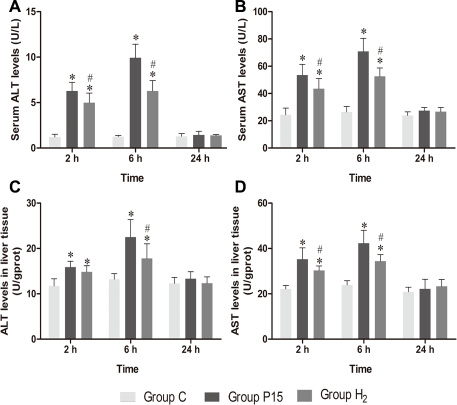
Figure 1: Hypodermic injection of hydrogen gas protected against liver injury induced by pneumoperitoneum. (A, B) The ALT and AST levels in serum. (C, D) The AST and ALT levels in liver tissue. Data are presented as mean ± SD. *p < 0.05 vs C group. #p < 0.05 vs P15 group.
Hypodermic injection of hydrogen gas protected against liver oxidative stress injury induced by pneumoperitoneum
MDA, SOD and GSH concentrations in serum and liver tissue homogenate showed a similar trend among each other. MDA concentration significantly increased in P15 group compared with C group at 2 h and 6 h, while its concentration is significantly reduced in H2 group compared with P15 group (P < 0.05, Figure 2A and 2D). SOD concentration significantly decreased in P15 group compared with C group at 2 h and 6 h, while its concentration significantly increased in H2 group compared with P15 group (P < 0.05, Figure 2B and 2E) at 2 h. GSH concentration significantly decreased in P15 group compared with C group at 6 h, while its concentration significantly increased in H2 group compared with P15 group (P < 0.05, Figure 2C and 2F) at 6 h.
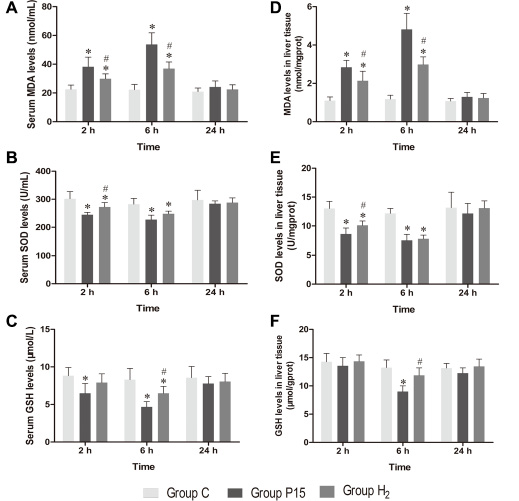
Figure 2: Hypodermic injection of hydrogen gas protected against liver oxidative stress injury induced by pneumoperitoneum. (A, B and C) The MDA, SOD and GSH levels in serum. (D, E and F) The MDA, SOD and GSH levels in liver tissue. Data are presented as mean ± SD. *p < 0.05 vs C group. #p < 0.05 vs P15 group.
Hypodermic injection of hydrogen gas protected against liver histology injury induced by pneumoperitoneum
Examination under the light microscope at 6 h revealed no significant pathological changes in C group (Figure 3A and 3B). Differences were observed in liver histology between P15 group and H2 group. Hepatocytes’ nuclei pyknosis, hepatocytes swelling and vacuoles degeneration were observed in P15 group (Figure 3C and 3D). In contrast, in H2 group, similar changes were found but in lesser degree (Figure 3E and 3F).
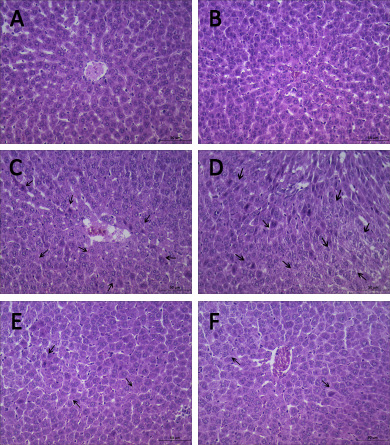
Figure 3: Hypodermic injection of hydrogen gas protected against liver histology injury induced by pneumoperitoneum. Histopathology examination at 6 h. (A, B) C group. (C, D) P15 group. (E, F) H2 group.
Hypodermic injection of hydrogen gas activated Nrf2 signaling pathway
Nrf2 mRNA expression was increased after pneumoperitoneum and hypodermic injection of hydrogen gas resulted in a further significant increase (P < 0.05, Figure 4A) at 2 h and 6 h. HO-1 and NQO1 mRNA expression were also examined using real-time PCR., Their expressions in H2 group remarkably increased compared with P15 group (P < 0.05, Figure 4B and 4C). Real-time PCR results were confirmed by western blot at 6 h (Figure 5).
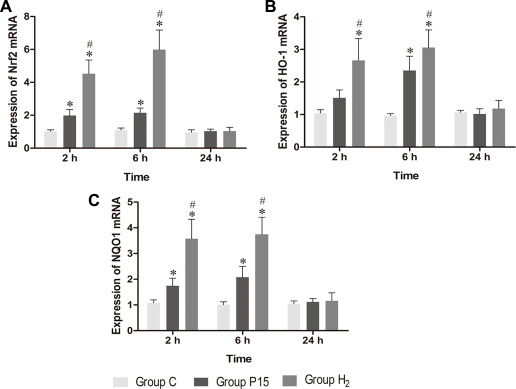
Figure 4: Hypodermic injection of hydrogen gas activated Nrf2 signaling pathway. The Nrf2, HO-1 and NQO1 mRNA expression in liver. Data are presented as mean ± SD. *p < 0.05 vs C group. #p < 0.05 vs P15 group.
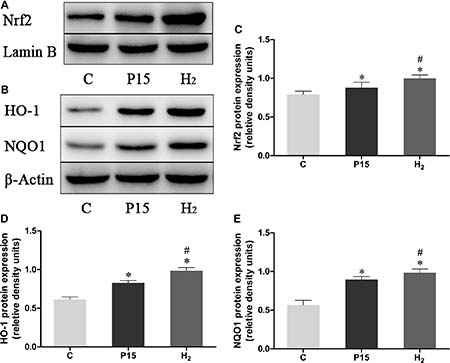
Figure 5: Hypodermic injection with hydrogen gas activated Nrf2 signaling pathway. The Nrf2, HO-1 and NQO1 protein expression in liver at 6 h. Data are presented as mean ± SD. *p < 0.05 vs C group. #p < 0.05 vs P15 group.
Hypodermic injection of hydrogen gas protected against liver injury induced by pneumoperitoneum by reducing apoptosis
Bax/Bcl-2 and Caspase-3 mRNA expression was increased after pneumoperitoneum, while hypodermic injection of hydrogen gas resulted in a significant decrease (P < 0.05, Figure 6) at 2 h and 6 h. Real-time PCR results were confirmed by western blot at 6 h (Figure 7).
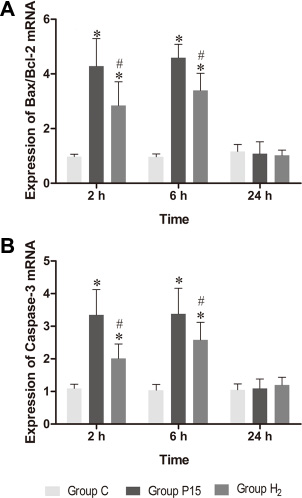
Figure 6: Hypodermic injection of hydrogen gas protected against liver injury induced by pneumoperitoneum by reducing apoptosis. The Bax/Bcl-2 and Caspase-3 mRNA expression in liver. Data are presented as mean ± SD. *p < 0.05 vs C group. #p < 0.05 vs P15 group.
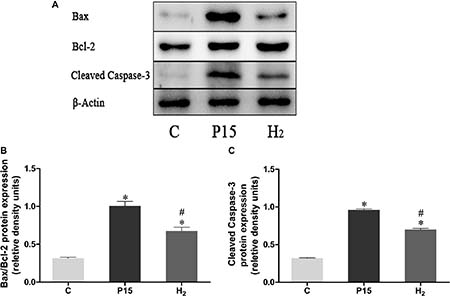
Figure 7: Hypodermic injection of hydrogen gas protected against liver injury induced by pneumoperitoneum by reducing apoptosis. The Bax/Bcl-2 and Cleaved Caspase-3 protein expression in liver at 6 h. Data are presented as mean ± SD. *p < 0.05 vs C group. #p < 0.05 vs P15 group.
Hypodermic injection of hydrogen gas protected against liver injury induced by pneumoperitoneum by reducing inflammatory cytokine production
Real-time PCR was used to measure TNF-α, ICAM-1 and IL-10 mRNA in the liver, while ELISA was used to evaluate protein expression in the liver tissue homogenate. TNF-α and ICAM-1 mRNA levels were significantly decreased in H2 group compared with P15 group (P < 0.05, Figure 8A and 8B) at 6 h. IL-10 mRNA expression was increased after pneumoperitoneum, and hypodermic injection of hydrogen gas resulted in a further significant increase (P < 0.05, Figure 8C) at 2 h and 6 h. Similarly, TNF-α and ICAM-1 proteins in liver tissue homogenate were significantly lower (P < 0.05, Figure 9A and 9B) while IL-10 protein was significantly higher (P < 0.05, Figure 9C) in H2 group compared with P15 group at 6 h.
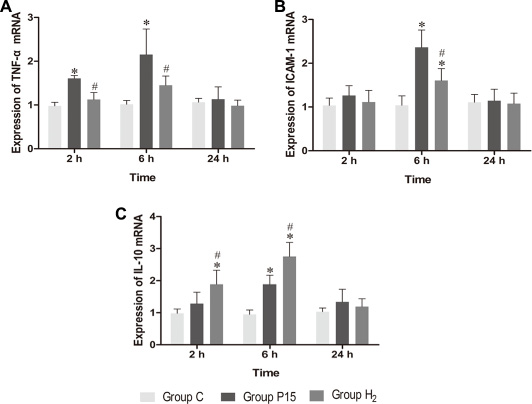
Figure 8: Hypodermic injection of hydrogen gas protected against liver injury induced by pneumoperitoneum by reducing inflammatory cytokine production. The TNF-α, ICAM-1 and IL-10 mRNA expression in liver. Data are presented as mean ± SD. *p < 0.05 vs C group. #p < 0.05 vs P15 group.
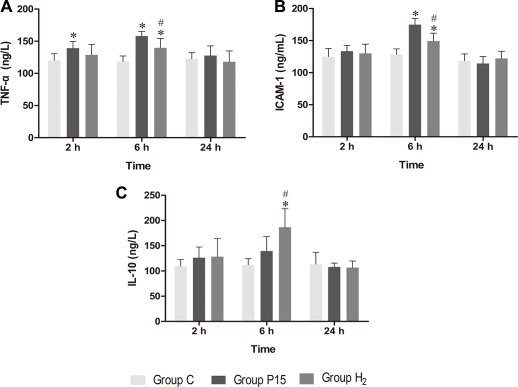
Figure 9: Hypodermic injection of hydrogen gas protected against liver injury induced by pneumoperitoneum by reducing inflammatory cytokine production. The TNF-α, ICAM-1 and IL-10 protein expression in liver by ELISA. Data are presented as mean ± SD. *p < 0.05 vs C group. #p < 0.05 vs P15 group.
DISCUSSION
This study was performed to determine the protective effect of hydrogen subcutaneous injection on CO2 pneumoperitoneum inducing liver injury. Pneumoperitoneum is required in laparoscopic surgery and CO2 is the most commonly used gas during pneumoperitoneum. CO2 insufflation can result in a significant ischemia of splanchnic organs, followed by increased reperfusion injury after deflation [25]. Thus, CO2 pneumoperitoneum can cause negative effects on body health. Splanchnic organs such as liver, kidneys, spleen, intestine, ovaries and testicles are susceptible to pneumoperitoneum adverse effects [6, 26–30]. The liver receives blood supply from the hepatic artery and portal vein and it is one of the most susceptible organs affected by ischemia. It has been reported that 10 mm Hg pneumoperitoneum caused significant decrease in hepatic arterial, portal, and microvascular perfusion [31]. In addition, pneumoperitoneum increase from 10 to 15 mm Hg can decrease liver flow by 39%, causing liver ischemia injury, and deflation after surgery resulting in blood reperfusion results in liver ischemia reperfusion injury [32, 33]. Several studies showed that CO2 pneumoperitoneum affects liver enzyme function and causes tissue injury [34, 35]. Our results showed a similar result. Indeed our results showed that ALT and AST levels were increased in both P15 and H2 group, although their levels in H2 group were lower than in P15 group. The histopathological examination reflecting the degree of liver injury showed that injury in H2 group was less than P15 group. Therefore, hydrogen subcutaneous injection could reduce liver injury during CO2 pneumoperitoneum.
The use of H2 results in many advantages. More than 38 diseases can be relieved by the biological effects of H2 [36]. The methods of introducing H2 into the body can be different and include hydrogen gas inhalation, hydrogen water ingestion, and hydrogen-rich saline injection [36]. It has been demonstrated that the protective effect of hydrogen subcutaneous injection is the same as the protective effect obtained by hydrogen water intraperitoneal injection [37]. Furthermore, subcutaneous injection of hydrogen is more convenient, safer and rapidly absorbed compared with hydrogen gas inhalation and hydrogen-rich saline injection. In our study, the results of the biochemical analysis and the histopathological examination showed that subcutaneous injection of hydrogen reduced liver injury induced by CO2 pneumoperitoneum. This protective effect was associated with reduced oxidative stress, apoptosis and inflammatory cytokine production.
Ischemia reperfusion injury producing excess of ROS, results in the downregulation of some endogenous antioxidant enzymes including SOD [38]. SOD is a metal enzyme with a specific biological catalytic function. Superoxide radical can be catalyzed by SOD and transformed into molecular oxygen or hydrogen peroxide. MDA originates from the decomposition of oxidizes protein and lipid peroxyl radicals, thus its content reflects the degrees of lipid peroxidation [39]. MDA can be a cause of oxidative stress as well as a result. SOD and MDA levels usually estimated anti-oxidative ability of the tissue [40]. GSH is an important endogenous antioxidant [41]. GSH protects tissues from oxidative stress injury through ROS detoxification [42]. Our results showed that hydrogen subcutaneous injection reduced liver oxidative stress by decreasing MDA levels and increasing SOD and GSH levels, suggesting a protective effect of hydrogen against oxidative stress. Nrf2 is a transcription factor which acts against oxidative stress injury by mediating the levels of endogenous antioxidants. Keap1 normally binds to Nrf2 to suppress it and keep it into the cytoplasm. However, Keap-1-Nrf2 complex is dissociated after activation of Nrf2 due to oxidative damage, enabling Nrf2 to translocate into to the nucleus. Here, it binds to the antioxidant response element (ARE) and activates the transcription of antioxidant enzymes, including HO-1 and NQO1 [43–45]. Recent studies have shown that Nrf2 can protect liver against ischemia reperfusion injury [46]. Both HO-1 and NQO1 are key response genes, and their activation can protect against oxidative damage in cells [47]. HO-1 exerts cytoprotection by inhibiting inflammation and apoptosis [48]. It can also degrade heme to CO, iron, and biliverdin, exerting an indirect anti-oxidative effect [49]. NQO1 is a flavoprotein and its flavin co-factor plays a key role in the direct scavenging of superoxide [50]. Therefore, NQO1 may directly exert an antioxidant protection. Our results showed that Nrf2, HO-1 and NQO1 mRNA and protein levels were increased by pneumoperitoneum. After subcutaneous injection of hydrogen, both their mRNA and protein levels were significantly increased suggesting an improved liver protection against ischemia reperfusion injury during CO2 pneumoperitoneum through the increase of the antioxidants level.
Nrf2 also has an anti-inflammatory ability [51]. Inflammatory response can lead to liver injury during ischemia-reperfusion [52]. Inflammatory response is associated with the release of proinflammatory cytokines. TNF-α is a proinflammatory cytokine, and it is the main cytokine acting in the acute-phase response [53]. ICAM-1 is a cell adhesion molecules that can be induced by TNF-α, while IL-10 is an anti-inflammatory cytokine that represses proinflammatory response, thus reducing tissue damage caused by inflammation [54, 55]. Previous studies suggest that elevated intra-abdominal pressure can enhance proinflammatory cytokine expression [56]. Indeed, Kupffer cells are activated at the initial phase of reperfusion and they consequently release both proinflammatory cytokines such as TNF-α and anti-inflammatory cytokines such as IL-10 [57, 58]. Furthermore, ischemia-reperfusion could increase ICAM-1 expression in liver endothelial cells [59]. In our study, IL-10, TNF-α and ICAM-1 expression was increased after pneumoperitoneum. However, hydrogen subcutaneous injection enhanced IL-10 mRNA and protein expression and reduced TNF-α and ICAM-1 mRNA and protein expression, indicating that hydrogen improved inflammation caused by CO2 pneumoperitoneum.
Apoptosis can be stimulated during liver ischemia-reperfusion injury [60]. Indeed, liver ischemia-reperfusion can increase the expression of Bax and cleaved caspase-3 [61]. Also a high CO2 pneumoperitoneum pressures causes an increased apoptosis [62]. Bax and Bcl-2 are important genes in cell apoptosis progress. Bax is a pro-apoptotic protein, while Bcl-2 is an anti-apoptotic protein. Bax can induce caspases activation among other various apoptotic molecules [63]. Cleaved caspase-3 plays an important role in the caspase cascade characterizing the apoptotic pathway. Activated caspase-3 leads to DNA fragmentation and apoptosis [64]. In our study, pneumoperitoneum did not increase Bcl-2 expression, but it increased Bax and cleaved caspase-3 expression, suggesting that pneumoperitoneum induced hepatocyte apoptosis. After hydrogen subcutaneous injection, Bax and cleaved caspase-3 expression was reduced, indicating a decreased hepatocyte apoptosis during CO2 pneumoperitoneum.
Some methods are available to reduce pneumoperitoneum-induced injury such as ischemic-preconditioning, pneumoperitoneum preconditioning, low intra-abdominal pressure, stepwise rising CO2 insufflation, melatonin, theophylline, erythropoietin and dexmedetomidine [65–71]. Some agents are also available to reduce ischemia-reperfusion injury such as epigallocatechin-3-gallate, gastrodin, eupatilin and oxymatrine [38, 72–74]. If H2 can be used in clinical practice it would be convenient because it is cheaper compared with those methods, although its inhalation may be dangerous because of its inflammable and explosive characteristics. Subcutaneous injection of hydrogen was safer and simpler than inhalation. Moreover, H2 does not leave residues compared with other drugs. As a potential antioxidant, H2 has many advantages such as rapid diffusion, no direct elimination of functionally important ROS, no toxicity even at higher concentration [36]. Therefore our study highlighted that subcutaneous injection of hydrogen could exert a protective effect on liver injury during CO2 pneumoperitoneum through reducing oxidative stress, cell apoptosis and inflammatory cytokines release. Hence, subcutaneous injection of hydrogen might be a promising approach against pneumoperitoneum-induced livers injury.
MATERIALS AND METHODS
Animals
Forty-five male Wistar rats, weighing 250–300 g, were housed under an alternating 12-h light/12-h dark cycle and free access to standard rat food and water; the environmental conditions were constant. Food was withheld 12 h prior to anesthesia, but free access to water was allowed. All experimental procedures were performed according to the guidelines approved by the Northeast Agricultural University, Harbin, China.
Surgical technique
Forty-five rats were randomly divided into three groups (n = 15). Rats were anaesthetized at the beginning of the experiment using isoflurane inhalation in a 1.5% mixture of oxygen, delivered using an animal anesthetic respiratory system (Surgivet Co., Ltd, USA). After anesthesia was performed, animals were placed in a supine position, and the abdominal skin was disinfected. Hydrogen gas was provided by a hydrogen generator (Saikesaisi hydrogen energy Co., Ltd, Shandong, China). Pneumoperitoneum was generated in all animals, except the control group, by inserting a 22G I.V. cannula needle. CO2 was insufflated using an automatic device (Olympus Co., Ltd, JPN) at a low rate until the intra-abdominal pressure reached 15 mmHg. Rats in the control group (C group) were subjected only to anesthesia for 90 min. Rats in the pneumoperitoneum group (P15 group) received an abdominal insufflation of CO2 for 90 min at an intra-abdominal pressure of 15 mmHg. Rats in the H2 group received a hypodermic injection of hydrogen gas (0.2 mL/kg) and after 10 min they received an abdominal insufflation of CO2 for 90 min at an intra-abdominal pressure of 15 mm Hg.
Blood and tissue samples collection
Rats were sacrificed by decapitation at 2 h, 6 h and 24 h after deflation. Blood samples were collected and centrifuged at 3500 rpm for 10 min and serums were stored at -20°C until analysis. A part of liver tissue was placed in 10% formalin for histological studies. Another part was homogenized in ice-cold saline and centrifuged at 2500 rpm for 10 min and supernatants were stored at -20°C until analysis. A third part was frozen immediately in liquid nitrogen and stored at −80°C until analysis.
Biochemical analysis
MDA, SOD, GSH, AST and ALT in serums and liver tissue homogenates were measured using the respective detection kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer’s instructions.
ELISA
TNF-α, ICAM-1 and IL-10 in the liver tissue homogenate were determined using the respective ELISA kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) as described in the instructions. Absorbance was measured at 450 nm according to a standard curve and cytokines were expressed as ng/L or ng/mL.
Real-time RT-PCR
Total RNA was extracted from the liver using Trizol reagent (Invitrogen, Carlsbad, USA) as described in the instructions. cDNA synthesis was performed in 1 μg of total RNA using PrimeScript™ RT reagent Kit with gDNA Eraser (Takara, Dailian, China) as described in the instructions. Quantitative real-time PCR was performed in LightCycler 2.0 (Roche Applied Science, Penzberg, Germany) using the SYBR® Premix Ex Taq™ II (Takara, Dailian, China) as described in the instructions. Primers (Table 1) were synthesized by Takara (Dailian, China). Reaction parameter was: 1 cycle of 95°C for 30 s, 45 cycles of 95°C for 5 s, primer-specific annealing temperature for 20 s, and extension at 72°C for 20 s. The relative mRNA expression was analyzed using the 2−ΔΔCt method.
Table 1: Primer sequences
Gene |
Primer Sequence (5′-3′) |
|---|---|
Nrf2 |
GAGACGGCCATGACTGAT (forward) |
GTGAGGGGATCGATGAGTAA (reverse) |
|
HO-1 |
ATCGTGCTCGCATGAAC (forward) |
CAGCTCCTCAAACAGCTCAA (reverse) |
|
NQO1 |
CAGCGGCTCCATGTACT (forward) |
GACCTGGAAGCCACAGAAG (reverse) |
|
Bcl-2 |
GGGATGCCTTTGTGGAACTA (forward) |
CTCACTTGTGGCCCAGGTAT (reverse) |
|
Bax |
TGTTTGCTGATGGCAACTTC (forward) |
GATCAGCTCGGGCACTTTAG (reverse) |
|
Caspase-3 |
ACAGAGCTGGACTGCGGTAT (forward) |
TGCGGTAGAGTAAGCATACAGG (reverse) |
|
TNF-α |
GGCCACCACGCTCTTCTGTC (forward) |
GGGCTACGGGCTTGTCACTC (reverse) |
|
ICAM-1 |
AAACGGGAGATGAATGGT (forward) |
TCTTCCTCTGGCGGTAAT (reverse) |
|
IL-10 |
TGGACAACATACTGCTGACAG (forward) |
GGTAAAACTTGATCATTTCTGACAAG (reverse) |
|
Actb |
GTCGTACCACTGGCATTGTG (forward) |
CTCTCAGCTGTGGTGGTGAA (reverse) |
Western blot
Frozen liver tissues were homogenized in ice-cold RIPA Lysis Buffer (Beyotime Biotechnology, Nanjing, China) or Nuclear and Cytoplasmic Protein Extraction Kit (Beyotime Biotechnology, Nanjing, China) containing PMSF (Beyotime Biotechnology, Nanjing, China) as described in the instructions. The homogenization was centrifuged at 12,000 × g for 20 min at 4°C. The supernatant was collected and protein concentration was determined using the Enhanced BCA Protein Assay Kit (Beyotime Biotechnology, Nanjing, China) as described in the instructions. Protein samples (30 μg) were denatured for 5 min at 100°C in 5× SDS-PAGE Sample Loading Buffer (Beyotime Biotechnology, Nanjing, China). Electrophoresis was performed using SDS-PAGE Gel Quick Preparation Kit (Beyotime Biotechnology, Nanjing, China), 10% acrylamide gels for β-actin, NRF2, Lamin B, and 12% acrylamide gels for HO-1, NQO1, Bcl-2, Bax, and 15% acrylamide gels for Cleaved Caspase-3. Protein were transferred to nitrocellulose membranes using the VE 186 Trans-Blot apparatus (Tanon, Shanghai, China). Next, they were blocked for 2 h with 5 % skim milk at room temperature. Follow by the membranes were incubated with primary antibodies overnight at 4°C. The primary antibodies were used at the following dilutions: anti-β-actin diluted 1:5000 (ABclonal, Wuhan, China), anti-Nrf2 diluted 1:1000 (ABclonal, Wuhan, China), anti-Nrf2 diluted 1:1000 (ABclonal, Wuhan, China), anti-HO-1 and NQO1 diluted 1:25000 (Abcam, MA, USA), anti-Lamin B, Bcl-2, Bax and Cleaved Caspase-3 diluted 1:1000 (Wanlei, Shenyang, China). Membranes were incubated with secondary antibody (dilution 1:5000, Zhongshan Goldenbridge Biotech, Beijing, China) for 2 hour at room temperature. The signals were visualized using the ECL kits (Beyotime Biotechnology, Nanjing, China). The blots were scanned using a Tanon 5200 Imaging System (Tanon Science & Technology Co., Shanghai, China).
Histopathology examination
Livers in 10% formalin were embedded in paraffin, cut into sections and stained with hematoxylin and eosin. All specimens were examined under light microscope.
Statistical analysis
Data are presented as mean ± standard deviation (SD). Statistical analysis was performed using SPSS version 22.0. A value of p < 0.05 was considered statistically significant.
Abbreviations
ALT, Alanine aminotransferase; AST, aspartate aminotransferase; MDA, malondialdehyde; SOD, superoxide dismutase; GSH, glutathione; Nrf2, nuclear factor E2-related factor 2; HO-1, Heme oxygenase-1; NQO1, NAD(P)H quinone dehydrogenase 1; BCL-2, B cell lymphoma/lewkmia-2; Bax, Bcl-2 assaciated x protein; Caspase-3, cysteinyl aspartate specific proteinase 3; TNF-α, tumor necrosis factor α; ICAM-1, intercellular cell adhesion molecule-1; IL-10, interlenkin-10.
Author contributions
MC and JZ designed the study. MC and JZ cared for animals and collected samples. All authors carried out experimental work. MC, LJ and GB collected and analyzed data. MC wrote and edited the manuscript. All authors read and approved the manuscript.
CONFLICTS OF INTEREST
None.
FUNDING
This work was supported by the National Natural Science Foundation of China (31402261), “Young Talents” Project of Northeast Agricultural University (14QC21) and China Postdoctoral Science Foundation (2014M551210).
REFERENCES
1. Nguyen KT, Marsh JW, Tsung A, Steel JJ, Gamblin TC, Geller DA. Comparative benefits of laparoscopic vs open hepatic resection: a critical appraisal. Arch Surg. 2011; 146:348–56. https://doi.org/10.1001/archsurg.2010.248.
2. Kuntz C, Wunsch A, Bodeker C, Bay F, Rosch R, Windeler J, Herfarth C. Effect of pressure and gas type on intraabdominal, subcutaneous, and blood pH in laparoscopy. Surg Endosc. 2000; 14:367–71.
3. Schmandra TC, Kim ZG, Gutt CN. Effect of insufflation gas and intraabdominal pressure on portal venous flow during pneumoperitoneum in the rat. Surg Endosc. 2001; 15:405–8. https://doi.org/10.1007/s004640000331.
4. Windberger UB, Auer R, Keplinger F, Langle F, Heinze G, Schindl M, Losert UM. The role of intra-abdominal pressure on splanchnic and pulmonary hemodynamic and metabolic changes during carbon dioxide pneumoperitoneum. Gastrointest Endosc. 1999; 49:84–91.
5. Sammour T, Mittal A, Loveday BP, Kahokehr A, Phillips AR, Windsor JA, Hill AG. Systematic review of oxidative stress associated with pneumoperitoneum. Br J Surg. 2009; 96:836–50. https://doi.org/10.1002/bjs.6651.
6. Nickkholgh A, Barro-Bejarano M, Liang R, Zorn M, Mehrabi A, Gebhard MM, Buchler MW, Gutt CN, Schemmer P. Signs of reperfusion injury following CO2 pneumoperitoneum: an in vivo microscopy study. Surg Endosc. 2008; 22:122–8. https://doi.org/10.1007/s00464-007-9386-6.
7. Zar HA, Tanigawa K, Kim YM, Lancaster JR Jr. Rat liver postischemic lipid peroxidation and vasoconstriction depend on ischemia time. Free Radic Biol Med. 1998; 25:255–64.
8. Zhai Y, Busuttil RW, Kupiec-Weglinski JW. Liver ischemia and reperfusion injury: new insights into mechanisms of innate-adaptive immune-mediated tissue inflammation. Am J Transplant. 2011; 11:1563–9. https://doi.org/10.1111/j.1600-6143.2011.03579.x.
9. Liu Y, Yang L, Tao K, Vizcaychipi MP, Lloyd DM, Sun X, Irwin MG, Ma D, Yu W. Protective effects of hydrogen enriched saline on liver ischemia reperfusion injury by reducing oxidative stress and HMGB1 release. BMC Gastroenterol. 2014; 14:12. https://doi.org/10.1186/1471-230X-14-12.
10. Parks DA, Granger DN. Ischemia-reperfusion injury: a radical view. Hepatology. 1988; 8:680–2.
11. Drefs M, Thomas MN, Guba M, Angele MK, Werner J, Conrad M, Steib CJ, Holdt LM, Andrassy J, Khandoga A, Rentsch M. Modulation of Glutathione Hemostasis by Inhibition of 12/15-Lipoxygenase Prevents ROS-Mediated Cell Death after Hepatic Ischemia and Reperfusion. Oxid Med Cell Longev. 2017; 2017:8325754. https://doi.org/10.1155/2017/8325754.
12. Chen K, Li JJ, Li SN, Feng J, Liu T, Wang F, Dai WQ, Xia YJ, Lu J, Zhou YQ, Guo CY. 15-Deoxy-Delta12,14-prostaglandin J2 alleviates hepatic ischemia-reperfusion injury in mice via inducing antioxidant response and inhibiting apoptosis and autophagy. Acta Pharmacol Sin. 2017; 38:672–87. https://doi.org/10.1038/aps.2016.108.
13. Paradies G, Petrosillo G, Pistolese M, Di Venosa N, Federici A, Ruggiero FM. Decrease in mitochondrial complex I activity in ischemic/reperfused rat heart: involvement of reactive oxygen species and cardiolipin. Circ Res. 2004; 94:53–9. https://doi.org/10.1161/01.RES.0000109416.56608.64.
14. Ohsawa I, Ishikawa M, Takahashi K, Watanabe M, Nishimaki K, Yamagata K, Katsura K, Katayama Y, Asoh S, Ohta S. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nature Medicine. 2007; 13:688–94. https://doi.org/10.1038/nm1577.
15. Oharazawa H, Igarashi T, Yokota T, Fujii H, Suzuki H, Machide M, Takahashi H, Ohta S, Ohsawa I. Protection of the retina by rapid diffusion of hydrogen: administration of hydrogen-loaded eye drops in retinal ischemia-reperfusion injury. Invest Ophthalmol Vis Sci. 2010; 51:487–92. https://doi.org/10.1167/iovs.09-4089.
16. Fukuda KI, Asoh S, Ishikawa M, Yamamoto Y, Ohsawa I, Ohta S. Inhalation of hydrogen gas suppresses hepatic injury caused by ischemia/reperfusion through reducing oxidative stress. Biochemical and Biophysical Research Communications. 2007; 361:670–4. https://doi.org/10.1016/j.bbrc.2007.07.088.
17. Xie KL, Yu YH, Pei YP, Hou LC, Chen SY, Xiong LZ, Wang GL. Protective Effects of Hydrogen Gas on Murine Polymicrobial Sepsis Via Reducing Oxidative Stress and Hmgb1 Release. Shock. 2010; 34:90–7. https://doi.org/10.1097/SHK.0b013e3181cdc4ae.
18. Fu Y, Ito M, Fujita Y, Ito M, Ichihara M, Masuda A, Suzuki Y, Maesawa S, Kajita Y, Hirayama M, Ohsawa I, Ohta S, Ohno K. Molecular hydrogen is protective against 6-hydroxydopamine-induced nigrostriatal degeneration in a rat model of Parkinson’s disease. Neuroscience Letters. 2009; 453:81–5. https://doi.org/10.1016/j.neulet.2009.02.016.
19. Liu Q, Shen WF, Sun HY, Fan DF, Nakao A, Cai JM, Yan G, Zhou WP, Shen RX, Yang JM, Sun XJ. Hydrogen-rich saline protects against liver injury in rats with obstructive jaundice. Liver International. 2010; 30:958–68. https://doi.org/10.1111/j.1478-3231.2010.02254.x.
20. Chen H, Sun YP, Li Y, Liu WW, Xiang HG, Fan LY, Sun Q, Xu XY, Cai JM, Ruan CP, Su N, Yan RL, Sun XJ, et al. Hydrogen-rich saline ameliorates the severity of L-arginine-induced acute pancreatitis in rats. Biochemical and Biophysical Research Communications. 2010; 393:308–13. https://doi.org/10.1016/j.bbrc.2010.02.005.
21. Fukuda K, Asoh S, Ishikawa M, Yamamoto Y, Ohsawa I, Ohta S. Inhalation of hydrogen gas suppresses hepatic injury caused by ischemia/reperfusion through reducing oxidative stress. Biochem Biophys Res Commun. 2007; 361:670–4. https://doi.org/10.1016/j.bbrc.2007.07.088.
22. Mao YF, Zheng XF, Cai JM, You XM, Deng XM, Zhang JH, Jiang L, Sun XJ. Hydrogen-rich saline reduces lung injury induced by intestinal ischemia/reperfusion in rats. Biochem Biophys Res Commun. 2009; 381:602–5. https://doi.org/10.1016/j.bbrc.2009.02.105.
23. Wang F, Yu G, Liu SY, Li JB, Wang JF, Bo LL, Qian LR, Sun XJ, Deng XM. Hydrogen-rich saline protects against renal ischemia/reperfusion injury in rats. J Surg Res. 2011; 167:e339–44. https://doi.org/10.1016/j.jss.2010.11.005.
24. Huang CS, Kawamura T, Toyoda Y, Nakao A. Recent advances in hydrogen research as a therapeutic medical gas. Free Radic Res. 2010; 44:971–82. https://doi.org/10.3109/10715762.2010.500328.
25. Eleftheriadis E, Kotzampassi K, Botsios D, Tzartinoglou E, Farmakis H, Dadoukis J. Splanchnic ischemia during laparoscopic cholecystectomy. Surg Endosc. 1996; 10:324–6.
26. Xingwei X, Xin G, Peng Z, Tao F, Bowen D, Xiaoming K, Wu J, Ning L, Jieshou L. Low-dose ketamine pretreatment reduces oxidative damage and inflammatory response following CO2 pneumoperitoneum in rats. Clin Invest Med. 2014; 37:E124.
27. Kirsch AJ, Hensle TW, Chang DT, Kayton ML, Olsson CA, Sawczuk IS. Renal effects of CO2 insufflation: oliguria and acute renal dysfunction in a rat pneumoperitoneum model. Urology. 1994; 43:453–9.
28. Arikan Y, Tosun M, Saykol V, Kalkan S, Erdem S. p53 Expression and apoptosis in liver and spleen during CO2 pneumoperitoneum. Langenbecks Arch Surg. 2008; 393:877–82. https://doi.org/10.1007/s00423-007-0254-6.
29. Guven S, Muci E, Unsal MA, Yulug E, Alver A, Kadioglu Duman M, Mentese A. The effects of carbon dioxide pneumoperitoneum on ovarian blood flow, oxidative stress markers, and morphology during laparoscopy: a rabbit model. Fertil Steril. 2010; 93:1327–32. https://doi.org/10.1016/j.fertnstert.2008.10.053.
30. Imamoglu M, Cay A, Unsal MA, Aydin S, Ozdemir O, Karahan C, Sari A, Sarihan H. The effects of increased intraabdominal pressure on testicular blood flow, oxidative stress markers, and morphology. J Pediatr Surg. 2006; 41:1118–24. https://doi.org/10.1016/j.jpedsurg.2006.02.004.
31. Diebel LN, Wilson RF, Dulchavsky SA, Saxe J. Effect of increased intra-abdominal pressure on hepatic arterial, portal venous, and hepatic microcirculatory blood flow. J Trauma. 1992; 33:279–82; discussion 82–3.
32. Schilling MK, Redaelli C, Krahenbuhl L, Signer C, Buchler MW. Splanchnic microcirculatory changes during CO2 laparoscopy. J Am Coll Surg. 1997; 184:378–82.
33. Lee JY, Choi SH. Results of hepatic and renal function tests to different CO2 pneumoperitoneum conditions: An experimental capnoperitoneum study in dogs. Res Vet Sci. 2015; 101:1–5. https://doi.org/10.1016/j.rvsc.2015.04.014.
34. Tan M, Xu FF, Peng JS, Li DM, Chen LH, Lv BJ, Zhao ZX, Huang C, Zheng CX. Changes in the level of serum liver enzymes after laparoscopic surgery. World J Gastroenterol. 2003; 9:364–7.
35. Yoshida M, Ikeda S, Sumitani D, Takakura Y, Yoshimitsu M, Shimomura M, Noma M, Tokunaga M, Okajima M, Ohdan H. Alterations in portal vein blood pH, hepatic functions, and hepatic histology in a porcine carbon dioxide pneumoperitoneum model. Surg Endosc. 2010; 24:1693–700. https://doi.org/10.1007/s00464-009-0831-6.
36. Ohta S. Recent Progress Toward Hydrogen Medicine: Potential of Molecular Hydrogen for Preventive and Therapeutic Applications. Current Pharmaceutical Design. 2011; 17:2241–52. https://doi.org/10.2174/138161211797052664.
37. Chen S, Jiang W. Effect of hydrogen injected subcutaneously on testicular tissues of rats exposed to cigarette smoke. International Journal of Clinical and Experimental Medicine. 2015; 8:5565–70.
38. Jiang G, Liu X, Wang M, Chen H, Chen Z, Qiu T. Oxymatrine ameliorates renal ischemia-reperfusion injury from oxidative stress through Nrf2/HO-1 pathway. Acta Cir Bras. 2015; 30:422–9. https://doi.org/10.1590/S0102-865020150060000008.
39. Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979; 95:351–8.
40. Tartibian B, Maleki BH. The Effects of Honey Supplementation on Seminal Plasma Cytokines, Oxidative Stress Biomarkers, and Antioxidants During 8 Weeks of Intensive Cycling Training. Journal of Andrology. 2012; 33:449–61. https://doi.org/10.2164/jandrol.110.012815.
41. Meister A. Glutathione-ascorbic acid antioxidant system in animals. J Biol Chem. 1994; 269:9397–400.
42. Knight TR, Ho YS, Farhood A, Jaeschke H. Peroxynitrite is a critical mediator of acetaminophen hepatotoxicity in murine livers: protection by glutathione. J Pharmacol Exp Ther. 2002; 303:468–75. https://doi.org/10.1124/jpet.102.038968.
43. Ungvari Z, Bagi Z, Feher A, Recchia FA, Sonntag WE, Pearson K, de Cabo R, Csiszar A. Resveratrol confers endothelial protection via activation of the antioxidant transcription factor Nrf2. Am J Physiol Heart Circ Physiol. 2010; 299:H18–24. https://doi.org/10.1152/ajpheart.00260.2010.
44. Thimmulappa RK, Mai KH, Srisuma S, Kensler TW, Yamamoto M, Biswal S. Identification of Nrf2-regulated genes induced by the chemopreventive agent sulforaphane by oligonucleotide microarray. Cancer Res. 2002; 62:5196–203.
45. Higdon A, Diers AR, Oh JY, Landar A, Darley-Usmar VM. Cell signalling by reactive lipid species: new concepts and molecular mechanisms. Biochem J. 2012; 442:453–64. https://doi.org/10.1042/BJ20111752.
46. Kudoh K, Uchinami H, Yoshioka M, Seki E, Yamamoto Y. Nrf2 activation protects the liver from ischemia/reperfusion injury in mice. Ann Surg. 2014; 260:118–27. https://doi.org/10.1097/SLA.0000000000000287.
47. Patel A, Zhang S, Shrestha AK, Maturu P, Moorthy B, Shivanna B. Omeprazole induces heme oxygenase-1 in fetal human pulmonary microvascular endothelial cells via hydrogen peroxide-independent Nrf2 signaling pathway. Toxicol Appl Pharmacol. 2016; 311:26–33. https://doi.org/10.1016/j.taap.2016.10.002.
48. Morse D, Lin L, Choi AM, Ryter SW. Heme oxygenase-1, a critical arbitrator of cell death pathways in lung injury and disease. Free Radic Biol Med. 2009; 47:1–12. https://doi.org/10.1016/j.freeradbiomed.2009.04.007.
49. Motterlini R, Green CJ, Foresti R. Regulation of heme oxygenase-1 by redox signals involving nitric oxide. Antioxid Redox Signal. 2002; 4:615–24. https://doi.org/10.1089/15230860260220111.
50. Siegel D, Gustafson DL, Dehn DL, Han JY, Boonchoong P, Berliner LJ, Ross D. NAD(P)H:quinone oxidoreductase 1: role as a superoxide scavenger. Mol Pharmacol. 2004; 65:1238–47. https://doi.org/10.1124/mol.65.5.1238.
51. Osburn WO, Yates MS, Dolan PD, Chen S, Liby KT, Sporn MB, Taguchi K, Yamamoto M, Kensler TW. Genetic or pharmacologic amplification of Nrf2 signaling inhibits acute inflammatory liver injury in mice. Toxicological Sciences. 2008; 104:218–27. https://doi.org/10.1093/toxsci/kfn079.
52. Jaeschke H. Mechanisms of Liver Injury. II. Mechanisms of neutrophil-induced liver cell injury during hepatic ischemia-reperfusion and other acute inflammatory conditions. Am J Physiol Gastrointest Liver Physiol. 2006; 290:G1083–8. https://doi.org/10.1152/ajpgi.00568.2005.
53. Grande M, Tucci GF, Adorisio O, Barini A, Rulli F, Neri A, Franchi F, Farinon AM. Systemic acute-phase response after laparoscopic and open cholecystectomy. Surg Endosc. 2002; 16:313–6. https://doi.org/10.1007/s00464-001-9042-5.
54. Lee IT, Liu SW, Chi PL, Lin CC, Hsiao LD, Yang CM. TNF-alpha mediates PKCdelta/JNK1/2/c-Jun-dependent monocyte adhesion via ICAM-1 induction in human retinal pigment epithelial cells. PLoS One. 2015; 10:e0117911. https://doi.org/10.1371/journal.pone.0117911.
55. Ouyang W, Rutz S, Crellin NK, Valdez PA, Hymowitz SG. Regulation and functions of the IL-10 family of cytokines in inflammation and disease. Annu Rev Immunol. 2011; 29:71–109. https://doi.org/10.1146/annurev-immunol-031210-101312.
56. Ozmen MM, Zulfikaroglu B, Col C, Cinel I, Isman FK, Cinel L, Besler TH. Effect of Increased Abdominal Pressure on Cytokines (IL1 beta, IL6, TNF alpha), C-reactive Protein (CRP), Free Radicals (NO, MDA), and Histology. Surgical Laparoscopy Endoscopy & Percutaneous Techniques. 2009; 19:142–7.
57. Cutrn JC, Perrelli MG, Cavalieri B, Peralta C, Rosell Catafau J, Poli G. Microvascular dysfunction induced by reperfusion injury and protective effect of ischemic preconditioning. Free Radic Biol Med. 2002; 33:1200–8.
58. Tsukamoto H. Redox regulation of cytokine expression in Kupffer cells. Antioxid Redox Signal. 2002; 4:741–8. https://doi.org/10.1089/152308602760598882.
59. Farhood A, McGuire GM, Manning AM, Miyasaka M, Smith CW, Jaeschke H. Intercellular adhesion molecule 1 (ICAM-1) expression and its role in neutrophil-induced ischemia-reperfusion injury in rat liver. J Leukoc Biol. 1995; 57:368–74.
60. Selzner M, Rudiger HA, Selzner N, Thomas DW, Sindram D, Clavien PA. Transgenic mice overexpressing human Bcl-2 are resistant to hepatic ischemia and reperfusion. J Hepatol. 2002; 36:218–25.
61. Park MS, Joo SH, Kim BS, Lee JW, Kim YI, Hong MK, Ahn HJ. Remote Preconditioning on Rat Hepatic Ischemia-Reperfusion Injury Downregulated Bax and Cleaved Caspase-3 Expression. Transplant Proc. 2016; 48:1247–50. https://doi.org/10.1016/j.transproceed.2015.12.125.
62. Tosun M, Yucel M, Kucuk A, Sezen S. P53 related apoptosis in kidneys in CO(2) pneumoperitoneum rat model: an immunohistochemical study. Mol Biol Rep. 2014; 41:6391–5. https://doi.org/10.1007/s11033-014-3519-5.
63. Weng C, Li Y, Xu D, Shi Y, Tang H. Specific cleavage of Mcl-1 by caspase-3 in tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis in Jurkat leukemia T cells. J Biol Chem. 2005; 280:10491–500. https://doi.org/10.1074/jbc.M412819200.
64. Thornberry NA, Lazebnik Y. Caspases: enemies within. Science. 1998; 281:1312–6.
65. Isbir T, Yilmaz H, Agachan B, Karaali ZE. Cholesterol ester transfer protein, apolipoprotein E and lipoprotein lipase genotypes in patients with coronary artery disease in the Turkish population. Clinical Genetics. 2003; 64:228–34. https://doi.org/10.1034/j.1399-0004.2003.00137.x.
66. Cevrioglu AS, Yilmaz S, Koken T, Tokyol C, Yilmazer M, Fenkci IV. Comparison of the effects of low intra-abdominal pressure and ischaemic preconditioning on the generation of oxidative stress markers and inflammatory cytokines during laparoscopy in rats. Hum Reprod. 2004; 19:2144–51. https://doi.org/10.1093/humrep/deh380.
67. Ates E, Yilmaz S, Ihtiyar E, Yasar B, Karahuseyinoglu E. Preconditioning-like amelioration of erythropoietin against laparoscopy-induced oxidative injury. Surg Endosc. 2006; 20:815–9. https://doi.org/10.1007/s00464-005-0428-7.
68. Sahin DA, Haliloglu B, Sahin FK, Akbulut G, Fidan H, Koken G, Buyukbas S, Aktepe F, Arikan Y, Dilek ON. Stepwise rising CO2 insufflation as an ischemic preconditioning method. J Laparoendosc Adv Surg Tech A. 2007; 17:723–9. https://doi.org/10.1089/lap.2007.0008.
69. Cay A, Imamoglu M, Unsal MA, Aydin S, Alver A, Akyol A, Sarihan H. Does anti-oxidant prophylaxis with melatonin prevent adverse outcomes related to increased oxidative stress caused by laparoscopy in experimental rat model? J Surg Res. 2006; 135:2–8. https://doi.org/10.1016/j.jss.2005.12.025.
70. Cekic B, Geze S, Ozkan G, Besir A, Sonmez M, Karahan SC, Mentese A. The effect of dexmedetomidine on oxidative stress during pneumoperitoneum. Biomed Res Int. 2014; 2014:760323. https://doi.org/10.1155/2014/760323.
71. Ozturk SA, Ceylan C, Serel TA, Doluoglu OG, Soyupek AS, Guzel A, Ozorak A, Uz E, Savas HB, Baspinar S. Protective effect of theophylline on renal functions in experimental pneumoperitoneum model. Ren Fail. 2015; 37:1044–9. https://doi.org/10.3109/0886022X.2015.1040706.
72. Tak E, Park GC, Kim SH, Jun DY, Lee J, Hwang S, Song GW, Lee SG. Epigallocatechin-3-gallate protects against hepatic ischaemia-reperfusion injury by reducing oxidative stress and apoptotic cell death. J Int Med Res. 2016; 44:1248–62. https://doi.org/10.1177/0300060516662735.
73. Liu B, Li F, Shi J, Yang D, Deng Y, Gong Q. Gastrodin ameliorates subacute phase cerebral ischemiareperfusion injury by inhibiting inflammation and apoptosis in rats. Mol Med Rep. 2016; 14:4144–52. https://doi.org/10.3892/mmr.2016.5785.
74. Lee HM, Jang HJ, Kim SS, Kim HJ, Lee SY, Oh MY, Kwan HC, Jang DS, Eom DW. Protective Effect of Eupatilin Pretreatment Against Hepatic Ischemia-Reperfusion Injury in Mice. Transplant Proc. 2016; 48:1226–33. https://doi.org/10.1016/j.transproceed.2016.01.024.

