The Cancer Problem
Cancer remains unsolved. It is a problem that has resisted solution for centuries, in spite of the application of immense bodies of knowledge from diverse fields, including molecular biology, biochemistry, pharmacology and physics [1]. Cancer functions as a complex system that enables an unrelenting adaptability to a large range of environmental changes, including alterations in nutrients, oxygen, pH, temperature, and treatment with interventional agents. New ways of thinking about the cancer problem are still needed.
A still prevalent cancer paradigm from the time of Beale (1860) and Boveri (1902) is that cancer is associated with structural abnormalities, from tissue and cell structure to DNA organization [2-6]. Cancer also manifests many structure-independent functional abnormalities, including dysregulated metabolism, epigenetic alterations, and transcription factor dysfunction [7-11]. Information storage at the level of DNA is another level of disruption in cancer that includes mutations and translocations [12, 13]. In cancer, the transformation from the well-regulated homeostasis of the normal cell to the chaotic, uncoordinated cancer state has been described as a phase transition that involves extensive change in structure, function and information [14]. The cancer transition involves alterations across all scales, including DNA, the cell, and communication among cancer and cancer-supporting cells. Therein lies the complexity of cancer: the myriad of innumerable interactions among the individual molecular agents within the cancer cell, among other cells types in the cancer microenvironment and across the organ systems of the host.
This cancer feature is similar to a problem in physics that is simple in its description, but has withstood exact solution since the time of Newton: the three-body problem [15]. When two bodies, such as the sun and the Earth, interact dynamically through gravitation, their movement in space can be computed with ease. Adding just one additional body, for example the Moon, introduces a level of complexity and nonlinearity to the problem such that even an approximate solution is computed with difficulty. As more bodies are added, the problem difficulty increases in exponential computation time. Therefore, it is not surprising that cancer biology remains at an impasse as it confronts a problem that involves a practicably uncountable number of interacting cell agents. In the three-body or multi-body problems of physics, major simplification is introduced by consideration of symmetry, resulting in new solutions that can be classified based on their symmetry features [16-18]. Analogously, the problem of cancer may be better understood and rendered more tractable by analysis of its symmetry features. The field of biology is filled with underappreciated examples of symmetry and symmetry breaking. Normal cell and tissue function is a result of tightly controlled maintenance of symmetry and concomitant symmetry breaking when required.
Symmetry in physics and biology
Symmetry is an elemental feature of space and time that underlies the geometric and dynamical properties of the observable universe [19, 20]. Symmetry is information: information about that which remains unchanged or, in physics nomenclature, is invariant when an operation on a system is carried out. Geometric symmetries are best known and are observed throughout the natural world. The snowflake is a well-known example; it possesses 6-fold rotational and 12-fold reflection symmetry. In the example of rotational symmetry, the results of an experiment performed in a spaceship far away from gravitational or other fields don’t have to be replicated for every direction the laboratory is oriented. Physical symmetries also extend to the known forces and particles in the universe, such as the existence of the negatively charged electron and the positively charged, but equal mass, positron. The search for the Higgs boson and its recent discovery was the result of employing symmetry principles [21]. At an information level, symmetry permits a more compact system description and simplifies computational problems.
The complement of symmetry is symmetry breaking [22-24]. Broken symmetry can result from explicit or from spontaneous symmetry breaking. Explicit symmetry breaking is the most familiar and occurs, for example, when the side of an apple is cut off or when an egg is dropped on the floor and shatters. Spontaneous symmetry breaking is conceptually more difficult, but occurs throughout the universe with regularity. When a magnet is heated above a certain temperature called the Curie point the magnetization is lost as all the individual magnetic particles assume random orientations in a symmetric pattern. When the temperature is reduced below the Curie point in the spaceship laboratory far from the Earth’s magnetic field the individual particles re-coalesce into a magnet, but the north-south orientation of the magnet is not predictable— this is an example of spontaneous symmetry breaking. Similarly, when water freezes, the axis orientation of the ice crystals is random. In the language of physics, at the exact transition point of instability the lowest energy solution that respects the initial symmetry ceases to be the lowest energy solution and a new, but asymmetric solution becomes the new low energy solution. In biology, an organism utilizes symmetry breaking along well-defined axes for functional diversification on every scale, from molecular assemblies, to subcellular structures, to cell types themselves, to tissue architecture [25]. Normal cell and tissue function is a result of tightly controlled maintenance of symmetry and symmetry breaking when required, for example during development.
In physics, symmetry is most often preserved and therefore forms a basis for the search of fundamental particles and forces. In biology symmetry breaking occurs continuously and indeed, is a condition for life [26, 27]. This symmetry breaking is, however, always incomplete: completely broken symmetry is complete disorder, which could not sustain life. Correspondingly, perfect order and global symmetry would also be incompatible with life. The informational content of a perfectly symmetric system is inadequate for the complex functions of life. Life exists in the intermediate realm between order and disorder. Cancer may be a state of broken symmetry beyond that of the normal homeostasis and the controlled system of sustainable life.
Symmetry breaking
The complexity of any system can be described and quantified by three components: combinatorial, geometric and functional [27]. Each of these components can be characterized by their symmetries, which can then be applied to understanding specific features of cancer. While each component can be described independently, it is important to note that biology makes use of all three together.
Combinatorial symmetry breaking
Combinatorial complexity at the cellular level refers to the number of configurations—genetic or phenotypic—in which cells can be exchanged while maintaining the overall functional invariance of the system [27]. In most normal tissues, cell division results in identical or nearly identical daughter pairs. In cancer, cell division is often an asymmetric process that can be thought of as a series of symmetry breaking events. When occurring over many cell divisions a population of cancer cells displays tumor heterogeneity where a high number of cells have slightly different genetic and phenotypic states. This is one key feature that enables the profound adaptability of cancer. A complete picture of cancer’s combinatorial complexity and symmetry also requires consideration of the cells in the microenvironment, including immune cells, tumor macrophages and many others.
Combinatorial complexity is quantified as:
Kc = log(N!/ni!) for N cells of i types with ni of each type (! denoting the factorial operation) [27]. In normal tissues, there is a relatively small number of cell types within a given organ, each with similar function for a given class (e.g., epithelial, lymphoid, vascular). In cancer, new cell types emerge within the cancer mass, manifested as tumor cell heterogeneity (TCH). In order to appreciate the immense combinatorial complexity of biological systems, consider 1 gram of tissue with 109 cells and only three different cell types. In this case Kc = 4.8 x108 (since Kc is the logarithm of the combinations, the actual number of combinations is on the order of 10 followed by 100 million zeros). As the tissue volume and number of cancer and cancer-supporting cell types increases, global cancer combinatorial complexity increases further to astronomical levels. As with the three-body problem in physics, the problem can benefit by consideration and quantification of system symmetries and symmetry breaking. It is possible that combinatorial complexity mathematics could be applied as a measure of genomic TCH, leading to a new way to monitor how a tumor cell population is evolving and adapting over time. For example, what level of intervention is needed to effectively disrupt the cancer complexity and does combinatorial complexity increase or decrease after treatment with a therapeutic agent?
How does the cancer state of broken symmetry and a combinatorial complexity beyond that of normal tissues originate and evolve? A key hallmark of cancer is its ability to metastasize. It is thought that a key step in metastasis is the transformation of epithelial cells to mesenchymal cells [28]. Whereas epithelial cells may undergo malignant change and then grow within a tumor, these cells do not readily spread to distant sites in the organism. In contrast, when an epithelial cell is transformed to a mesenchymal cell the potential for cell spread outside the tissue of origin increases greatly. This is known as epithelial-to-mesenchymal transition or EMT. Mesenchymal cells have the physical configuration and other cellular machinery designed for movement. Known transcription factors and other cellular constituents are now recognized as key for maintaining a cell in the epithelial state, transformation to the mesenchymal state or conversion back to the epithelial cell type [28-30]. But what causes the molecular changes that then lead to cell symmetry breaking?
This question has recently been examined by the study of transcription factor and miRNA levels that together operate as a molecular switch to determine phenotypic cell fate. In this model, the transcription factors (e.g. ZEB, SLUG, and TWIST) and miRNA’s (e.g., miR-200 and miR-34) interact as auto-catalytic and inhibitory network components in a symmetry breaking decision network [31-33]. Small perturbations in one or more components can then cause a cell fate phase transition. High miR-200 and miR-34, low ZEB and SNAIL define the epithelial phenotype (E) and low miR-200 and miR-34, high ZEB and SNAIL result in the mesenchymal phenotype (M). Recent results suggest that intermediate levels of these cell constituents can result in a metastable E-M or partial EMT phenotype that, under specific environmental conditions or spontaneous stochastic cell fluctuations, can break symmetry into an E or M cell (Figure 1) [31, 34]. Of clinical significance is the observation that the symmetric hybrid E-M cell type in various cancers correlate with increased aggressiveness and metastatic characteristics [31, 34]. The hybrid E-M cells also are more prone to exist in the circulatory system as bound clusters of cells, which is thought to promote survival in the blood stream and eventual seeding of distant tissues [34]. Thus, symmetry retention or symmetry breaking of the E-M hybrid cell is likely a key factor in metastasis. Understanding of the underlying molecular events in stabilizing and transforming the E, M and E-M cell subtypes could lead to a better understanding of the metastatic process and permit control of these processes in cancer patients.
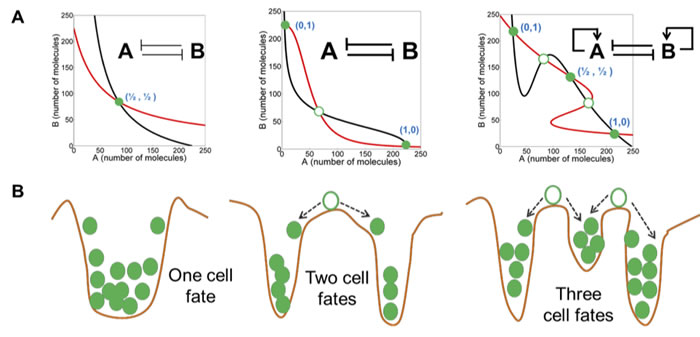
Figure 1: Symmetry breaking in mutually inhibitory feedback loops (from Jolly et al). Different levels of mutual inhibition and self-activation between two molecules, A and B, results in symmetry breaking from the configuration of equal numbers of A and B to bistability and tristability. In the tristable configuration, the intermediate state could represent a hybrid cell state. Red and black curves describe nullclines for A and B, and their intersections are the steady states. Green filled circles represent stable steady states, and green hollow circles show unstable steady states. For more information, see Jolly et al, 2015.
Tissue cells that lead to the creation of tumors and continually repopulate them with new cancer cells are known as Cancer Stem Cells (CSCs). Importantly, CSCs possess the traits to evade cancer therapeutics and to lie dormant for long time periods until they begin a process of high growth rate and a high evolution potential that gives rise to lethal phenotypic variation. Recent studies indicate that the E-M hybrid cell state is more likely to gain stemness properties [34, 35] and therefore to readily switch between invasive and proliferative modes to enhance survivability and correspondingly, lethality to the host. The search is now on to identify cell markers of stemness and E-M hybridicity. Network and molecular switch symmetry and symmetry breaking considerations could contribute to this effort by determining the conditions to break the E-M hybrid cell symmetry toward the E state, which has the least metastatic potential.
Geometric symmetry breaking
Geometric symmetry breaking requires consideration of two modes: conventional geometric structures and fractal structures. Conventional symmetry breaking is routinely observed by pathologists in the daily diagnosis and characterization of cancer cells using light microscopy . Indeed, abnormal cell and nuclear shape is one of the most reliable diagnostic criteria for cancer and is closely related to prognosis [36-38]. The malignant potential of almost all cancers is based on the grading of abnormal nuclear structure by pathologists. Fractal structure in cancer is less well characterized, but is increasingly investigated as a diagnostic and prognostic indicator [39-42]. The loss of geometric self-similarity in cancer can occur at different spatial scales, from the structure of the plasma membrane to that of chromatin.
Geometric symmetry breaking can be utilized to characterize the molecular and biochemical processes that determine cell shape and how they are disrupted in cancer. A fundamental feature of cytoplasmic and nuclear composition is their viscoelastic composition. The viscous and elastic properties of the cell create a structure similar to a soft glass with power-law mechanical properties [43, 44]. The elastic components include the proteins actin and myosin and microtubules represent rigid structures. The presence of elastic components that can create tension together with the rigid microtubules provides for a tensegrity structure of the cell that maintains normal cell shape and is profoundly disrupted in cancer [43, 45, 46]. These structural elements have been collectively termed the tissue matrix (TM) system that is comprised of the extracellular matrix, the membrane matrix, the cytoskeleton, and the nuclear matrix. Conventional tensegrity structures from macro structures in buildings to cell-cell interactions to proteins to DNA consist of struts and cables under tension that can transmit mechanochemical information [43, 44, 47].
A basic property of tensegrity structures is stability [48-51]. Tensegrity stability is reflected in the structural behavior as the result of geometric deformation due to an external load. If the structure returns to its self-equilibrium configuration when the external load is released, then it is stable. Tensegrity structures possess symmetry properties in virtue of their geometric structure. This symmetry results in high stability to deforming forces and efficient information transfer that helps maintain homeostasis. In cancer, this stability is greatly diminished, as cancer evolves to increasingly malignant forms [52-55]. The extracellular cell matrix also possesses tensegrity properties, which, when disrupted, may also contribute to degraded information transfer from the environment and increase metastatic potential [43].
Geometric complexity and symmetry can also be applied to the self-similarity of fractal structures, including down to the spatial level of DNA [41, 56, 57]. Fractals have a repeating geometrical description at any scale or spatial resolution. That is, there is no privileged spatial level of description, i.e., scale invariance exists. Conventional geometric structures obey integer scaling laws, for example the area of a square scaling as the power of 2 of the edge length. Fractal structures, however, display fractional scaling, such as the Koch curve whose length scales as the 4/3 power for each iteration [58] (Figure 2)
In fractal systems power laws describe the frequency of an occurrence x, as: f(x) = x-n. In this description, large events have a significantly greater likelihood of occurrence compared to, for example, when frequencies are distributed according to a normal distribution (i.e., “‘fat tails” exist). Thus, large-deviation phenomena in cancer may be related to fractal properties and power law behavior.
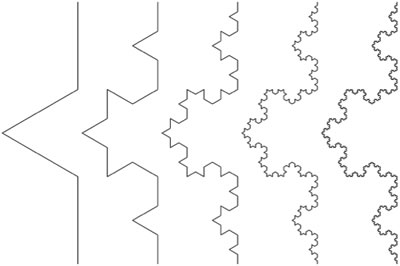
Figure 2 : The Koch curve. The Koch curve is created by dividing each line segment into thirds and replacing the middle segment with an equilateral triangle. Each iteration of the Koch curve produces a curve that is self-similar to the previous ones. It can be readily shown that the total length scales the power law relationship (4/3)n for n iterations and thus the length approaches infinity in the limit. The fractal dimension of a Koch curve is defined as log4/log3 = 1.2619. The Koch curve is continuous, but not differentiable, i.e., it has no tangent at any point.
Fractal structures have been examined from the level of chromatin to the cell membrane to the lung and other organs [39-42, 59]. At the DNA level, the fractal structure of sequences can be examined in a ‘chaos game’ or in ‘DNA walks’, in which DNA sequences are examined to identify long-range correlations (as exist in physical phase transitions) in nucleotide sequences and disruptions represented by duplications, repeats and translocations [40]. In the chaos game a random walk over the DNA sequence space generates a fractal pattern. Using the 4 DNA base dimensionality, an information space pattern arises similar to the hyperspace and hypercycles of Eigen [60]. These analyses, then, are able to detect global gene structure and long-range pattern disruption in cancer that would otherwise remain opaque without considerations of self-similarity and power law dependencies. Thus, long-range correlations and symmetry breaking in gene structure could, in turn, affect integrated cell function and homeostasis as reflected in the loss of top-down and bottom-up global function. Fractal patterns also exist in functional networks, as addressed below [61].
Symmetry and symmetry breaking in the cancer cell have not yet been fully investigated, but this represents a fertile area for additional work in order to understand features of cancer cell shape and chemomechanical information transfer disruptions at a molecular level. In particular, it would be highly desirable to better understand the boundary limits of symmetry disruption in the progression of normal cell function to cancer to cell death. This could permit interventions to either further push the cancer state toward death or back toward normal homeostasis.
Functional symmetry breaking
In functional complexity and symmetry breaking, the interactions among individual cellular constituents is examined [27]. These interactions form networks, such as protein-protein or gene-gene interaction networks. Individual networks don’t function in isolation, but interact with each other and build up a global system network that permits life. In cancer, the network is degraded and homeostasis is lost. Bioinformatics seeks to elucidate regularities and control points in biological networks that are lost in cancer, but as the previous combinatorial complexity examples demonstrate, the problem becomes even more computationally daunting when functional interactions are considered. Symmetry analysis can again contribute to clarifying this component of the cancer problem.
The functional cell structure can be described by a network of interconnected individual molecular agents, which in turn can be analyzed by graph theory [62-64]. For example, in social networks, graphs show all of the connections between individuals. Graphs possess embedded symmetries that are important for stability and homeostasis [64-68]. In graph theory, the agents are the vertices and the connections are the edges of the graph (Figure 3). Graph theory entails a complex and exceedingly powerful mathematical framework that can identify symmetry and other features not readily discernable when the graph size reaches hundreds or thousands of independent agents. Two key graph theory concepts are graph symmetry and graph complexity, which are closely related, but provide different graph informational descriptions [62, 69-75]. Graph symmetry and symmetry breaking can characterize and explain changes in network functionality, complexity and information transfer. It is a foundational feature of a graph.
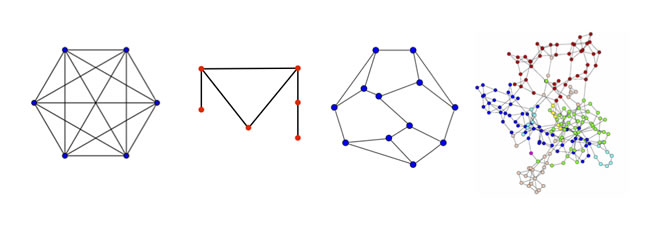
Figure 3: Graphs of differing symmetry. Graph 1 is a complete graph with all nodes connected; it has 6 rotation and 12 reflection symmetries. Graph 2 is the smallest asymmetric graph. Graph 3 is the smallest asymmetric graph with each node possessing 3 edges (degree 3); it is known as the Frucht graph. Graph 4 is a complex graph, such as might exist for interacting proteins, for which any imbedded symmetries are difficult to discern by inspection. Software programs, such as nauty and SAUCY2, can compute the graph automorphisms.
Graph symmetry is illustrated in Figure 3. The hexagonal graph with all vertices connected (a complete graph) is highly symmetric and include 6 rotations, 6 reflections along lines connecting opposite vertices and 6 reflections along lines connecting the midpoints of each edge. Any one or a combination of these symmetry operations maintains the network. In the second graph, there is no rotation, reflection or other change that maintains graph invariance; this graph is the simplest asymmetric planar graph. The third graph increases in complexity, but one can by careful inspection determine that the graph is asymmetric. This is the Frucht graph, which is the smallest asymmetric graph with each vertex having exactly 3 edges. In the fourth graph, the complexity is greatly increased compared to the three other graphs. A protein-protein interaction network might have this appearance. It is difficult to determine by inspection whether this graph has any symmetries.
In graph theory, a graph symmetry is termed a graph automorphism. The collection of all the graph automorphisms is the automorphism group or Aut(G). Measurement of Aut(G) and other graph properties lies on the cutting edge of computation theory and is a currently active area of research in the solution of very difficult problems [76-78]. Fortunately, new algorithms have made the calculation of Aut(G) much more practicable for large networks [79, 80].
The graph Aut(G) characterizes the information content of a functional network by counting all the symmetries. Yet, asymmetric components of a network can also store information and therefore parallel information approaches have been used for network analysis, including Shannon information, Kolmogorov complexity and Gibbs free energy [69, 71, 72, 74, 75, 81]. The Kolmogorov complexity (K(G)) measure is the size of the smallest computer program needed compute or construct the graph [69, 71, 82, 83]. A fully symmetric graph has a large Aut(G), low K(G) and low information. A completely asymmetric network has Aut(G) = 1 (a graph is symmetric with itself) and a K(G) that is a function of the actual size and arrangement of the individual agents, as is the information processing capacity of the network. Network symmetry and complexity measures are approximately reciprocal, but complexity measures also capture the information contained in asymmetric graph.
In recent years, the K(G) and Aut(G) analyses have been applied to a number of biological networks [64, 69-71, 81-88]. One advantage of Kolmogorov complexity over a purely symmetry description using Aut(G) is that it better captures all the graph structure by measuring all of the non-randomness. For a given graph Aut(G) variations in K(G) can be observed. Typically, there is an optimal number of edges for a given vertex number (V(G)) number to maximize K(G), and hence the information processing capacity of a network. For 50 nodes , K(G) is maximum at about 600 edges [83]. Related to this, is the phenomenon of phase transitions in complexity space when an abrupt increase in K(G) occurs at a threshold level of node connnetedness (i.e., the average node degree). This concept is similar to physical phase transitions where long-range interactions and symmetry breaking occur. When few connections exist at each node, K(G)~logV(G), but when the number of edges, E, grows, an abrupt increase in K(G) emerges at edge probability ~logV(G)/V(G) [69]. Is this the point where the phase transition from normal to cancer occurs or where a cancer suddenly develops resistance? Notably, the Aut(G) of protein interactions in different human cancers has been shown to be related to the 5-year survival rate [70]. Average symmetry may be sufficient for some of these analyses and would simplify the computational difficulty [89]. Further investigation could encompass the measurement of Aut(G) and subgroup symmetries in cancer and in the corresponding normal tissue in order to identify the precise location in protein, gene, or other networks where abnormal broken symmetries exist. To our knowledge, such experimental analysis has not yet been carried out. The identified sites could then be used as leverage points to attack and destroy the cancer network with molecular therapies or more challengingly, to restore the lost symmetries.
Biological networks must be stabilizable and controllable in order to store and transmit information necessary for survival of the organism. Survival entails a response to environmental changes, which in turn requires modification or fine-tuning of the cell or organism’s functional network over time. Homeostasis in dynamic, open systems far from equilibrium must exist at the interface between high order, maximum stability and a state of disordered chaos. As the size or number of nodes of a network (the graph order) increases the fraction of controllable systems decreases, thus permitting only a small percentage of complex systems to manifest homeostasis in response to external perturbations [90-93]. A second important concept from the theory of large networks is that a single leader-follower model is insufficient for controllability in complex systems [90, 92]. Thus, a multi-level consideration of top-down and bottom-up functionality is necessary for understanding an integrated system, such as would be required for recovering the controllability of cancer cells that existed prior to the neoplastic transformation. Cellular homeostasis has been recently examined with regard to singularities and stability against coordinate changes that preserve network structure [94]. This article advances our understanding of cellular homeostasis, but does not address symmetry, automorphism groups, or subgroup broken symmetries that could be related to attack tolerance when homeostasis is degraded; this could be a direction for future research.
Network symmetry considerations can contribute to understanding large network controllability and homeostasis. In recent years, the theory and application of network control principles has shown that network symmetry is closely related to controllability and stabilizability in response to external perturbations, decisions, and signals [91-93, 95]. Thus, the network Aut(G) could be a major determinant of controllability. In cancer, knowledge of broken symmetries that have a large effect on loss of homeostasis and creation of instabilities could provide new insights into tumor heterogeneity and metastatic spread. Early recognition of asymmetries could direct specific therapeutic interventions that could repair or reverse neoplastic processes and restore homeostasis or achieve a new homeostasis.
Another concept related to network symmetry is network attack tolerance [67, 96-102]. Attack tolerance is the network’s resilience to random or intentional deletion of nodes or interference with the connection of nodes (deletion of graph edges). Well-known examples are the WWW, power grids, and transportation networks. Among the network properties that determine robustness to attack, symmetry is a key factor [67, 96, 102]. As before, this is the case for the network symmetries defined by Aut(G), as well as the subgroup symmetries [62, 66, 67]. Scale-free symmetries that many real-world graphs possess also play a role in network resilience [61, 96, 97, 103] (Figure 4).
The relationship of network symmetry to cancer could take several different forms. For cancer origin, attack by environmental factors, such as carcinogens, with successful network disruption and conversion of homeostasis to instability could be investigated and better understood by application of symmetry principles, including my measuring the Aut(G) change after experimental application of carcinogens. In cancer treatment, understanding resistance to drugs or radiation could be enhanced though knowledge of attack vulnerability at specific points in the network, perhaps in the subgraphs that are asymmetric and have diminished attack tolerance. These new concepts will require improved elucidation of biologic network graph structure and improved analytics to detect imbedded symmetry groups.
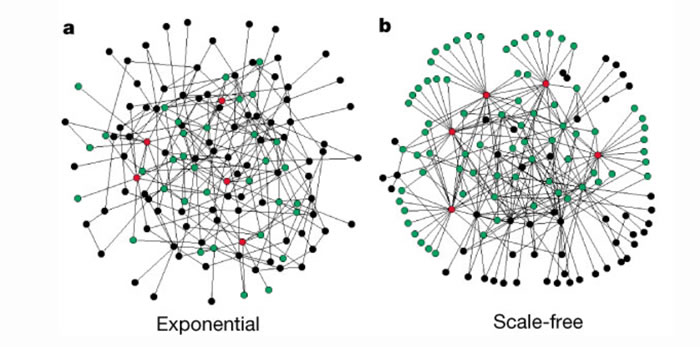
Figure 4: Visual illustration of the difference between an exponential and a scale-free network (from Albert et al). a, The exponential network is homogeneous: most nodes have approximately the same number of links. b, The scale-free network is inhomogeneous: the majority of the nodes have one or two links but a few nodes have a large number of links, guaranteeing that the system is fully connected. Red, the five nodes with the highest number of links; green, their first neighbours. Although in the exponential network only 27% of the nodes are reached by the five most connected nodes, in the scale-free network more than 60% are reached, demonstrating the importance of the connected nodes in the scale-free network Both networks contain 130 nodes and 215 links ((k)=3.3). For additional information see Albert et al, Nature 2000.
Summary and future research directions
Symmetry and symmetry breaking concepts can define the parameters of cancer as a complex adaptive system. Defining symmetry in the physical sciences has been critical to the investigation of the structure of particles to the forces that define the universe. Biology is replete with symmetry and symmetry breaking events that are essential for life and evolution. In cancer, a further symmetry breaking occurs that disrupts the normal cell homeostasis to unleash a virulent and uncontrolled new life form that is often incompatible with host survival.
The three modes of symmetry breaking most commonly function in an integrated manner. For example, functional processes may interact with geometric changes in cancer and indeed, it may be that functional changes lead to the cytoskeletal tensegrity structural changes, each with its own symmetry group. Further examination of the three described modes of symmetry breaking as they apply to core features of cancer, origin, proliferation, metastasis and resistance, will be required. As these are defined for cancer cells to explain genomic and phenotypic diversity and plasticity, the observations and rules can be extended to include the tumor microenvironment and cancer at the tissue level. For example, can measures of system and molecular broken symmetry provide the needed information to determine sites with decreased attack tolerance for treatment? Could identification of broken cell symmetries permit repair in cancer in order to reestablish the lost homeostasis? Communication between the cancer and remote organs might also be better understood through functional symmetry considerations, as related to network stability, interference and attack tolerance, for example, by examining signaling networks from the cancer microenvironment to the metastatic niche of remote organs. Cancer destroys the host network at a system level and therefore, the cancer itself must be understood at its system level. Essential in these considerations is to not merely describe something as possessing a symmetry or broken symmetry, but to search for the molecular or system origin of the property and demonstrate how it can, in principle, be leveraged for the benefit of cancer patients.
Acknowledgments
We offer a tribute and gratitude to the late Dr. Eshel Ben-Jacob who taught and inspired us to think deeply about the connections between physics and biology. We thank Ms. Carrie Price for her incalculable assistance with the literature citations.
Conflicts of Interest
There is no conflict of interest.
Funding
This work was in-part funded by NIH grant nos. P01CA093900, U01 CA196390, and U54CA163214 to K.J.P.
Memoriam
Don Coffey died on November 9, 2017 at 85. He was a tremendous inspiration and driving force for this article and a beloved friend. A remembrance for Don and the lasting contributions he made to cancer research and the careers of so many is included in this Issue.
References
1. Weinberg RA. Coming full circle-from endless complexity to simplicity and back again. Cell. 2014; 157:267–71. https://doi.org/10.1016/j.cell.2014.03.004
2. Beale LS. Examination of sputum from a case of cancer of the pharynx and the adjacent parts. Arch Med. 1860; 2:44–46.
3. Boveri T. Über mehrpolige mitosen als mittel zur analyse des zellkerns. Neu Folge; 1902. p. 35.
4. Boveri T. Concerning the Origin of Malignant Tumours. [Article in German] Journal of Cell Science. 2008; 121: 1-84.
5. Pienta KJ, Partin AW, Coffey DS. Cancer as a disease of DNA organization and dynamic cell structure. Cancer Res. 1989; 49:2525–32.
6. Zink D, Fischer AH, Nickerson JA. Nuclear structure in cancer cells. Nat Rev Cancer. 2004; 4:677–87. https://doi.org/10.1038/nrc1430
7. Bhagwat AS, Vakoc CR. Targeting Transcription Factors in Cancer. Trends Cancer. 2015; 1:53–65. https://doi.org/10.1016/j.trecan.2015.07.001
8. DeBerardinis RJ, Chandel NS. Fundamentals of cancer metabolism. Sci Adv. 2016; 2:e1600200. https://doi.org/10.1126/sciadv.1600200
9. Feinberg AP, Koldobskiy MA, Göndör A. Epigenetic modulators, modifiers and mediators in cancer aetiology and progression. Nat Rev Genet. 2016; 17:284–99. https://doi.org/10.1038/nrg.2016.13
10. Keibler MA, Wasylenko TM, Kelleher JK, Iliopoulos O, Vander Heiden MG, Stephanopoulos G. Metabolic requirements for cancer cell proliferation. Cancer Metab. 2016; 4:16. https://doi.org/10.1186/s40170-016-0156-6
11. Lee TI, Young RA. Transcriptional regulation and its misregulation in disease. Cell. 2013; 152:1237–51. https://doi.org/10.1016/j.cell.2013.02.014
12. Andor N, Maley CC, Ji HP. Genomic Instability in Cancer: Teetering on the Limit of Tolerance. Cancer Res. 2017; 77:2179–85. https://doi.org/10.1158/0008-5472.CAN-16-1553
13. Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA Jr, Kinzler KW. Cancer genome landscapes. Science. 2013; 339:1546–58. https://doi.org/10.1126/science.1235122
14. Davies PC, Demetrius L, Tuszynski JA. Cancer as a dynamical phase transition. Theor Biol Med Model. 2011; 8:30. https://doi.org/10.1186/1742-4682-8-30
15. Chenciner A. Three body problem. Scholarpedia; 2011. p. 2.
16. Cartwright J. Physicists discover a whopping 13 new solutions to three-body problem. Science. 2013.
17. Šuvakov M, Dmitrašinović V. Three classes of newtonian three-body planar periodic orbits. Phys Rev Lett. 2013; 110:114301. https://doi.org/10.1103/PhysRevLett.110.114301
18. Šuvakov M, Dmitrašinović V. Three-Body Gallery. 2013.http://three-body.ipb.ac.rs/ref.php
19. Weyl H. Symmetry. Princeton University: Princeton University Press; 1952. https://doi.org/10.1515/9781400874347.
20. Feynman R. Symmetry in Physical Laws. The Feynman Lectures on Physics; 1963.
21. Maldacena J. The symmetry and simplicity of the laws of physics and the Higgs boson. Eur J Phys. 2016; 37:015802. https://doi.org/10.1088/0143-0807/37/1/015802.
22. Brading K, Castellani E. Symmetry and Symmetry Breaking. The Stanford Encyclopedia of Philosophy; 2013.
23. Collier J. Information originates in symmetry breaking. Symmetry. Cult Sc. 1996; 7:247–56.
24. Strocchi F. Symmetry breaking. Berlin: Springer; 2010.
25. Li R, Bowerman B. Symmetry Breaking in Biology. Cold Spring Harb Perspect Biol. 2010; 2: a003475. https://doi.org/10.1101/cshperspect.a003475.
26. Longo G, Montévil M. From physics to biology by extending criticality and symmetry breakings. Perspectives on Organisms. Springer; 2014. pp. 161–85.
27. Longo G, Montévil M, Springer-Verlag Gmb H. Perspectives on Organisms Biological time. Symmetries and Singularities; 2016.
28. Nieto MA, Huang RY, Jackson RA, Thiery JP. EMT: 2016. Cell. 2016; 166:21–45. https://doi.org/10.1016/j.cell.2016.06.028
29. Roca H, Hernandez J, Weidner S, McEachin RC, Fuller D, Sud S, Schumann T, Wilkinson JE, Zaslavsky A, Li H, Maher CA, Daignault-Newton S, Healy PN, Pienta KJ. Transcription factors OVOL1 and OVOL2 induce the mesenchymal to epithelial transition in human cancer. PLoS One. 2013; 8:e76773. https://doi.org/10.1371/journal.pone.0076773
30. Tania M, Khan MA, Fu J. Epithelial to mesenchymal transition inducing transcription factors and metastatic cancer. Tumour Biol. 2014; 35:7335–42. https://doi.org/10.1007/s13277-014-2163-y
31. Jolly MK, Boareto M, Huang B, Jia D, Lu M, Ben-Jacob E, Onuchic JN, Levine H. Implications of the Hybrid Epithelial/Mesenchymal Phenotype in Metastasis. Front Oncol. 2015; 5:155.
32. Lu M, Jolly MK, Levine H, Onuchic JN, Ben-Jacob E. MicroRNA-based regulation of epithelial-hybrid-mesenchymal fate determination. Proc Natl Acad Sci USA. 2013; 110:18144–49. https://doi.org/10.1073/pnas.1318192110
33. Lu M, Jolly MK, Onuchic J, Ben-Jacob E. Toward decoding the principles of cancer metastasis circuits. Cancer Res. 2014; 74:4574–87. https://doi.org/10.1158/0008-5472.CAN-13-3367
34. Jolly MK, Tripathi SC, Jia D, Mooney SM, Celiktas M, Hanash SM, Mani SA, Pienta KJ, Ben-Jacob E, Levine H. Stability of the hybrid epithelial/mesenchymal phenotype. Oncotarget. 2016; 7:27067–84. https://doi.org/10.18632/oncotarget.8166
35. Jolly MK, Jia D, Boareto M, Mani SA, Pienta KJ, Ben-Jacob E, Levine H. Coupling the modules of EMT and stemness: A tunable ‘stemness window’ model. Oncotarget. 2015; 6:25161–74. https://doi.org/10.18632/oncotarget.4629
36. Sailem HZ, Bakal C. Identification of clinically predictive metagenes that encode components of a network coupling cell shape to transcription by image-omics. Genome Res. 2017; 27:196–207. https://doi.org/10.1101/gr.202028.115
37. Faguet GB. A brief history of cancer: age-old milestones underlying our current knowledge database. Int J Cancer. 2015; 136:2022–36. https://doi.org/10.1002/ijc.29134
38. Lyons SM, Alizadeh E, Mannheimer J, Schuamberg K, Castle J, Schroder B, Turk P, Thamm D, Prasad A. Changes in cell shape are correlated with metastatic potential in murine and human osteosarcomas. Biol Open. 2016; 5:289–99. https://doi.org/10.1242/bio.013409
39. Chan A, Tuszynski JA. Automatic prediction of tumour malignancy in breast cancer with fractal dimension. R Soc Open Sci. 2016; 3:160558. https://doi.org/10.1098/rsos.160558
40. Lennon FE, Cianci GC, Cipriani NA, Hensing TA, Zhang HJ, Chen CT, Murgu SD, Vokes EE, Vannier MW, Salgia R. Lung cancer-a fractal viewpoint. Nat Rev Clin Oncol. 2015; 12:664–75. https://doi.org/10.1038/nrclinonc.2015.108
41. Metze K. Fractal dimension of chromatin: potential molecular diagnostic applications for cancer prognosis. Expert Rev Mol Diagn. 2013; 13:719–35. https://doi.org/10.1586/14737159.2013.828889
42. Sokolov I. Fractals: a possible new path to diagnose and cure cancer? Future Medicine; 2015.
43. Ingber DE, Wang N, Stamenović D. Tensegrity, cellular biophysics, and the mechanics of living systems. Rep Prog Phys. 2014; 77:046603. https://doi.org/10.1088/0034-4885/77/4/046603
44. Schiffhauer ES, Robinson DN. Mechanochemical Signaling Directs Cell-Shape Change. Biophys J. 2017; 112:207–14. https://doi.org/10.1016/j.bpj.2016.12.015
45. Tadeo I, Berbegall AP, Escudero LM, Álvaro T, Noguera R. Biotensegrity of the extracellular matrix: physiology, dynamic mechanical balance, and implications in oncology and mechanotherapy. Front Oncol. 2014; 4:39. https://doi.org/10.3389/fonc.2014.00039.
46. Pienta KJ, Coffey DS. Cellular harmonic information transfer through a tissue tensegrity-matrix system. Med Hypotheses. 1991; 34:88-95.
47. Goehring NW, Grill SW. Cell polarity: mechanochemical patterning. Trends Cell Biol. 2013; 23:72–80. https://doi.org/10.1016/j.tcb.2012.10.009
48. Zhang JY, Guest SD, Ohsaki M. Symmetric prismatic tensegrity structures: part I. Configuration and stability. Int J Solids Struct. 2009; 46:1–14. https://doi.org/10.1016/j.ijsolstr.2008.08.032.
49. Zhang JY, Ohsaki M. Tensetrity Structures: Form, Stability, and Symmetry. Springer. 2015.
50. Hargittai I. Symmetry : unifying human understanding 1 (1989). 1 (1989). New York: Pergamon Press; 1989.[u.a.].
51. Motro R. Structural morphology of tensegrity systems. Meccanica. 2011; 46:27–40. https://doi.org/10.1007/s11012-010-9379-8.
52. DuFort CC, Paszek MJ, Weaver VM. Balancing forces: architectural control of mechanotransduction. Nat Rev Mol Cell Biol. 2011; 12:308–19. https://doi.org/10.1038/nrm3112
53. Paszek MJ, Zahir N, Johnson KR, Lakins JN, Rozenberg GI, Gefen A, Reinhart-King CA, Margulies SS, Dembo M, Boettiger D, Hammer DA, Weaver VM. Tensional homeostasis and the malignant phenotype. Cancer Cell. 2005; 8:241–54. https://doi.org/10.1016/j.ccr.2005.08.010
54. Rubashkin MG, Cassereau L, Bainer R, DuFort CC, Yui Y, Ou G, Paszek MJ, Davidson MW, Chen YY, Weaver VM. Force engages vinculin and promotes tumor progression by enhancing PI3K activation of phosphatidylinositol (3,4,5)-triphosphate. Cancer Res. 2014; 74:4597–611. https://doi.org/10.1158/0008-5472.CAN-13-3698
55. Majeski HE, Yang J. The 2016 John J. Abel Award Lecture: Targeting the Mechanical Microenvironment in Cancer. Mol Pharmacol. 2016; 90:744–54. https://doi.org/10.1124/mol.116.106765
56. Gea L. Fractals in Biology and Medicine. Birkhäuser Verlag; 2005.
57. Mandelbrot B. The Fractal Geometry of Nature. W. H. Freeman and Company; 1982.
58. Cederberg JN. (2001) Chaos to Symmetry: An Introduction to Fractal Geometry. In: A Course in Modern Geometries. Undergraduate Texts in Mathematics. Springer, New York, NY.
59. Dinicola S, D’Anselmi F, Pasqualato A, Proietti S, Lisi E, Cucina A, Bizzarri M. A systems biology approach to cancer: fractals, attractors, and nonlinear dynamics. OMICS. 2011; 15:93–104. https://doi.org/10.1089/omi.2010.0091
60. Eigen M. From strange simplicity to complex familiarity a treatise on matter, information, life and thought. Oxford: Oxford Univ. Press; 2014.
61. Kim JS, Goh KI, Salvi G, Oh E, Kahng B, Kim D. Fractality in complex networks: critical and supercritical skeletons. Phys Rev E Stat Nonlin Soft Matter Phys. 2007; 75:016110. https://doi.org/10.1103/PhysRevE.75.016110
62. Dehmer M, Emmert-Streib F. Quantitative graph theory : mathematical foundations and applications. Boca Raton, London, New York: CRC Press; 2015.
63. Lauwerier HA. (2014). Applied Graph Theory. Elsevier Science.
64. Rietman EA, Karp RL, Tuszynski JA. Review and application of group theory to molecular systems biology. Theor Biol Med Model. 2011; 8:21. https://doi.org/10.1186/1742-4682-8-21
65. Garlaschelli D, Ruzzenenti F, Basosi R. Complex networks and symmetry I: A review. Symmetry (Basel). 2010; 2:1683–709. https://doi.org/10.3390/sym2031683.
66. MacArthur BD, Sanchez-Garcia RJ, Anderson JW. On automorphism groups of networks. Discrete Appl Math. 2008; 156:3525-3531.
67. MacArthur BD, Sánchez-García RJ, Anderson JW. Symmetry in complex networks. Discrete Appl Math. 2008; 156:3525–31. https://doi.org/10.1016/j.dam.2008.04.008.
68. Rodriguez L. Automorphism Groups of Simple Graphs. 2014.
69. Zenil H, Kiani NA, Tegnér J. Methods of information theory and algorithmic complexity for network biology. Seminars in cell & developmental biology. Elsevier; 2016. pp. 32–43.
70. Hinow P, Rietman EA, Tuszynski JA. Algebraic and topological indices of molecular pathway networks in human cancers. Math Biosci Eng. 2015.12;1289-302.
71. Rietman EA, Platig J, Tuszynski JA, Lakka Klement G. Thermodynamic measures of cancer: gibbs free energy and entropy of protein-protein interactions. J Biol Phys. 2016; 42:339–50. https://doi.org/10.1007/s10867-016-9410-y
72. de Badyn MH, Chapman A, Mesbahi M. Network entropy: A system-theoretic perspective. Decision and Control (CDC), 2015 IEEE 54th Annual Conference on: IEEE). 2015:5512-7.https://doi.org/10.1109/CDC.2015.7403083.
73. Zimand M. Symmetry of information: a closer look. Computation, Physics and Beyond. Springer; 2012. pp. 241–6.
74. Mousavian Z, Kavousi K, Masoudi-Nejad A. Information theory in systems biology. Part I: Gene regulatory and metabolic networks. Seminars in cell & developmental biology. Elsevier; 2016. pp. 3–13.
75. Mousavian Z, Díaz J, Masoudi-Nejad A. Information theory in systems biology. Part II: protein-protein interaction and signaling networks. Semin Cell Dev Biol. 2016; 51:14–23. https://doi.org/10.1016/j.semcdb.2015.12.006
76. Babai L. (2016) Graph Isomorphism in Quasipolynomial Time. Cambridge, MA, USA, pp 684-697.
77. Yaveroğlu ON, Malod-Dognin N, Davis D, Levnajic Z, Janjic V, Karapandza R, Stojmirovic A, Pržulj N. Revealing the hidden language of complex networks. Sci Rep. 2014; 4:4547. https://doi.org/10.1038/srep04547
78. Fortnow L. The Golden Ticket: P, NP, and the Search for the Impossible. Princeton University Press; 2013. https://doi.org/10.1515/9781400846610.
79. Weisstein E. Graph Automorphism. MathWorld; 2008.
80. Saucy3: Fast Symmetry Discovery in Graphs. 2012.
81. Rietman EA, Scott JG, Tuszynski JA, Klement GL. Personalized anticancer therapy selection using molecular landscape topology and thermodynamics. Oncotarget. 2017; 8:18735-18745. https://doi.org/10.18632/oncotarget.12932
82. Zenil H. (2017) Algorithmic Data Analytics, Small Data Matters and Correlation versus Causation. In: Pietsch W, Wernecke J, Ott M. (eds) Berechenbarkeit der Welt. Springer VS, Wiesbaden.
83. Zenil H, Soler-Toscano F, Dingle K, Louis AA. Correlation of automorphism group size and topological properties with program-size complexity evaluations of graphs and complex networks. Physica A. 2014; 404:341–58. https://doi.org/10.1016/j.physa.2014.02.060.
84. Breitkreutz D, Hlatky L, Rietman E, Tuszynski JA. Molecular signaling network complexity is correlated with cancer patient survivability. Proc Natl Acad Sci USA. 2012; 109:9209–12. https://doi.org/10.1073/pnas.1201416109
85. Rietman EA, Colt JZ, Tuszynski JA. Interactomes, manufacturomes and relational biology: analogies between systems biology and manufacturing systems. Theor Biol Med Model. 2011; 8:19. https://doi.org/10.1186/1742-4682-8-19
86. Rietman EA, Friesen DE, Hahnfeldt P, Gatenby R, Hlatky L, Tuszynski JA. An integrated multidisciplinary model describing initiation of cancer and the Warburg hypothesis. Theor Biol Med Model. 2013; 10:39. https://doi.org/10.1186/1742-4682-10-39
87. Kiani NA, Zenil H, Olczak J, Tegnér J. Evaluating network inference methods in terms of their ability to preserve the topology and complexity of genetic networks. Seminars in cell & developmental biology. Elsevier; 2016. pp. 44–52.
88. Zenil H, Kiani NA, Tegnér J. Quantifying loss of information in network-based dimensionality reduction techniques. J Complex Netw. 2016; 4:342–62. https://doi.org/10.1093/comnet/cnv025.
89. Alamino RC. Measuring complexity through average symmetry. J Phys A Math Theor. 2015; 48:275101. https://doi.org/10.1088/1751-8113/48/27/275101.
90. Chapman A. Semi-Autonomous Networks Effective Control of Networked Systems through Protocols, Design, and Modeling. Cham: Springer International Publishing; 2015.
91. Chapman A, Mesbahi M. (2015) State Controllability, output controllability and stabilizability of networks: a symmetry perspective. In: IEEE 54th annual conference on decision and control (CDC) Osaka Japan, 4776–4781. https://doi.org/10.1109/CDC.2015.7402964.
92. Rahmani A, Ji M, Mesbahi M, Egerstedt M. Controllability of multi-agent systems from a graph-theoretic perspective. SIAM J Contr Optim. 2009; 48:162–86. https://doi.org/10.1137/060674909.
93. Chapman A. Controllability and Observability of Cartesian Product Networks. Semi-Autonomous Networks. Springer; 2015.
94. Golubitsky M, Stewart I. Homeostasis, singularities, and networks. J Math Biol. 2017; 74:387–407. https://doi.org/10.1007/s00285-016-1024-2
95. Moreno A, Mossio M. (2015) Biological autonomy: a philosophical and theoretical enquiry. Springer, Dordrecht, The Netherlands.
96. Albert R, Jeong H, Barabási AL. Error and attack tolerance of complex networks. Nature. 2000; 406:378–82. https://doi.org/10.1038/35019019
97. Bak JH. Error and Attack Tolerance of Scale-Free Networks: Effects of Geometry. Korea Advanced Institute of Science and Technology; 2010.
98. Crucitti P, Latora V, Marchiori M, Rapisarda A. Error and attack tolerance of complex networks. Physica A. 2004; 340:388–94. https://doi.org/10.1016/j.physa.2004.04.031.
99. Matta J, Borwey J, Ercal G. Comparative resilience notions and vertex attack tolerance of scale-free networks. CoRR, abs/1404.0103, 2014.
100. Schieber TA, Carpi L, Frery AC, Rosso OA, Pardalos PM, Ravetti MG. Information theory perspective on network robustness. Phys Lett A. 2016; 380:359–64. https://doi.org/10.1016/j.physleta.2015.10.055.
101. Nie T, Guo Z, Zhao K, Lu ZM. The dynamic correlation between degree and betweenness of complex network under attack. Physica A. 2016; 457:129–37. https://doi.org/10.1016/j.physa.2016.03.075.
102. Manzano M, Sahneh F, Scoglio C, Calle E, Marzo JL. Robustness surfaces of complex networks. Sci Rep. 2014; 4:6133. https://doi.org/10.1038/srep06133
103. Cavagna A, Cimarelli A, Giardina I, Parisi G, Santagati R, Stefanini F, Viale M. Scale-free correlations in starling flocks. Proc Natl Acad Sci USA. 2010; 107:11865–70. https://doi.org/10.1073/pnas.1005766107

