INTRODUCTION
Stroke is one important reason of age-related cognitive decline and dementia. As a major cause of adult chronic disease, it is one of the most common causes of death [1, 2]. Stroke is a common higher incidence disease in adult males over the age of 45 which has a strong impact on human health and life. From the data of The World Health Organization, we found that more than 15 million new stroke patients each year which including 5 million fatal or permanent disability cases [3]. More and more studies evidenced genetic predisposition is an important attribution of stroke. However, the identification of genetic molecular mechanisms contributing to stroke is a challenge. Because of ischemic stroke comprises approximately 80% of all stroke cases, and remaining 20% mainly due to primary hemorrhage which induced intracerebral and subarachnoid stroke. The common causes of ischemic stroke include cardiac emboli and small vessel disease that is very critical to research into stroke-susceptibility genes and has taught us attach importance to the classification of patients. In addition, after cardiovascular disease and malignant tumors, acute ischemic stroke has been listed in the third-leading cause of death [4]. The incidence of ischemic stroke could be reduced by control of risk factors, such as hypertension, hypercholesteremia, cigarette smoking, excessive drinking, diabetes mellitus and heart failure. But it only reduces the incidence of ischemic stroke to a certain degree which suggests the existence of other potential risk factors [5, 6].
Identification of novel risk factors which will promote the development of strategies for prevention and treatment of ischemic stroke. From amount of researches, we found various diseases pathophysiological mechanisms, such as atherothrombosis or embolism, can lead to arterial occlusion which is a major cause of stroke. Previous genome-wide association studies (GWAS) identified many genetics variants associated with complex human diseases and have provided unique insights from genetic architecture [7]. Genetic variants associated with risk of ischemic stroke have been revealed by GWAS, these variants often have effects with obvious biological significance [5].
From some studies, we detected the genes encoding the enzymes which metabolize aldehyde dehydrogenase 2 (ALDH2) exists polymorphism association with stroke in the pathogenesis process [8, 9]. ALDH2 gene is located on chromosome 12, which can affect the blood acetaldehyde concentrations. ALDH2 protein is abundantly expressed in normal lung and liver, and is also present in organs that require high mitochondrial content, especially in heart and brain [10]. A previous study has indicated that ALDH2 gene is able to metabolize some short-chain aliphatic aldehydes, as well as polycyclic aldehydes and so forth [11]. In addition, Several previous studies have assessed SNPs in the ALDH2 gene for the association with the risk of diseases, such as coronary artery disease [12], diabetes [13], hypertension [14], lacunar infarcts [15]. Recently, a meta-analysis of GWAS has been found a tight association between ALDH2 genetic variations and pathogenesis of stroke from Asian decedents [16].
Thus, it is possible that locus–locus interactions within the ALDH2 gene may be associated with risk for ischemic stroke. However, the exact genetic basis of susceptibility to ischemic stroke is still not well defined. To further investigate potential relationships between ALDH2 polymorphisms and locus–locus interactions in the etiology of ischemic stroke. We randomly selected six tag candidate SNPs (rs886205, rs2238152, rs441, rs4646778, rs671, and rs7296651) from the SNPs in ALDH2, and designed the corresponding primers used for each SNP in the present study are listed in Table 1 and performed a case-control association analysis in Han Chinese population. Our study provides sufficient evidence for the association between ALDH2 gene polymorphisms and the risk of ischemic stroke.
Table 1: Primers used for this study
SNP_ID |
1st-PCRP |
2nd-PCRP |
UEP-SEQ |
|---|---|---|---|
rs886205 |
ACGTTGGATGTCTCGCTTTTGGGTTTACGG |
ACGTTGGATGCCTTTGACCCCAATGTGAAC |
GGGCGACCCTGACCT |
rs2238152 |
ACGTTGGATGAATCCCACCTTTATTTAAG |
ACGTTGGATGTGTTGTAAAAAGCACCAACC |
CCAACCTCAAAGCCAAA |
rs441 |
ACGTTGGATGAGCCTGGGTGCCAGAGAGA |
ACGTTGGATGCCCTGACAGCATTCACTTAG |
GGTTTTTGTTTGTTTTTTGAG |
rs4646778 |
ACGTTGGATGGTTTTCTGCTATTGGCCCTG |
ACGTTGGATGTATGCAGGCAACAAGACAAC |
GCAACAAGACAACTGGGAAAT |
rs671 |
ACGTTGGATGCCTTTGGTGGCTACAAGATG |
ACGTTGGATGAGGTCCCACACTCACAGTTT |
gaTCCCACACTCACAGTTTTCACTT |
rs7296651 |
ACGTTGGATGGGGCAAGACCCAGATTTGAA |
ACGTTGGATGCACGTGGCCTGTAACTATGA |
gGGCCTGTAACTATGATTTTGATGAA |
RESULTS
Basic characteristics of all the participants were presented in Table 2. All the tested SNPs are in HWE in the control population of this study (Table 3). Association results between ALDH2 tSNP genotypes and the risk of stroke were listed in Table 4. We identified two significant SNP genotypes associated with the increased risk of stroke, one was genotype “AA’’ of rs886205 (OR = 5.99; 95% CI = 1.11–32.23; p = 0.037) and the other was genotype ‘‘AA’’ of rs7296651 (OR = 8.93; 95% CI = 1.31–60.78; p = 0.025). MAF in cases and controls were listed in Table 5. Further model association analyses were performed by logistic tests. Three models – additive, dominant and recessive model – were applied for analyzing the association between polymorphisms and stroke, which was adjusted by the age and gender of the participants. The rs886205 (OR = 6.39; 95% CI = 1.19-34.38; p = 0.03) and rs7296651 (OR = 9.29; 95% CI = 1.37-63.21; p = 0.02) were observed to be associated with stroke risk in recessive model analyses. By haplotype analysis, one block was explored among the ALDH2 SNPs (Figure 1). Block 1 contains rs2238152, rs441, rs4646778, and rs671. We performed the association between the block and ischemic stroke, and found the result is statistically insignificant.
Table 2: Basic characteristics of controls and cases
Variables |
Cases N(%) |
Controls N(%) |
p valve |
|---|---|---|---|
Age (years) |
<0.01a |
||
Mean ± SD |
63.96 ± 11.06 |
50.36 ± 7.79 |
|
Sex |
<0.01b |
||
Male |
325 (66.6) |
195 (38.8) |
|
Female |
163 (33.4) |
308 (61.2) |
a p values were calculated by Student t tests.
b p values were calculated from two-sided Chi-square tests.
Table 3: Basic information of candidate SNPs in ALDH2 and associations with stroke
SNP_ID |
Chromosome |
Position |
HWE p value |
OR (95% CI) |
p value |
|---|---|---|---|---|---|
rs886205 |
12q24.12 |
112204427 |
0.412 |
1.08 (0.83-1.40) |
0.577 |
rs2238152 |
12q24.12 |
112214459 |
0.806 |
1.13 (0.94-1.39) |
0.198 |
rs441 |
12q24.12 |
112228849 |
0.526 |
0.87 (0.71-1.05) |
0.147 |
rs4646778 |
12q24.12 |
112235783 |
0.614 |
1.15 (0.95-1.40) |
0.151 |
rs671 |
12q24.12 |
112241766 |
0.814 |
0.95 (0.74-1.23) |
0.712 |
rs7296651 |
12q24.12 |
112246954 |
0.357 |
1.04 (0.81-1.34) |
0.766 |
OR, odds ratio; CI, confidence interval; SNP, single nucleotide polymorphism.
Table 4: Association between ALDH2 tSNP genotypes and the risk of stroke
SNP_ID |
Genotype |
No. (frequency) |
OR (95% CI) |
p value |
|
|---|---|---|---|---|---|
Case |
Control |
||||
rs886205 |
AA |
10 (2.1) |
4 (0.8) |
5.99 (1.11-32.23) |
0.037 |
AG |
106 (21.8) |
131 (26) |
0.78 (0.53-1.14) |
0.192 |
|
GG |
371 (76.1) |
368 (73.2) |
− |
||
rs2238152 |
TT |
42 (8.6) |
41 (8.3) |
0.75 (0.41-1.36) |
0.34 |
TG |
210 (43.5) |
192 (39) |
1.33 (0.94-1.88) |
0.109 |
|
GG |
231 (47.9) |
259 (52.7) |
− |
||
rs441 |
CC |
46 (9.4) |
44 (8.7) |
0.82 (0.46-1-45) |
0.493 |
CT |
208 (42.6) |
192 (38.2) |
1.33 (0.95-1.88) |
0.1 |
|
TT |
234 (48) |
267 (53.1) |
− |
||
rs4646778 |
AA |
47 (9.6) |
43 (8.6) |
0.83 (0.47-1.48) |
0.533 |
AC |
206 (42.2) |
192 (38.4) |
1.33 (0.94-1.88) |
0.103 |
|
CC |
234 (48.2) |
265 (53) |
− |
||
rs671 |
AA |
11 (2.3) |
8 (1.6) |
1.21 (0.38-3.90) |
0.744 |
AG |
108 (22.1) |
124 (24.7) |
0.83 (0.56-1.23) |
0.359 |
|
GG |
369 (75.6) |
371 (73.7) |
− |
||
rs7296651 |
AA |
10 (2.1) |
3 (0.7) |
8.93 (1.31-60.78) |
0.025 |
AG |
118 (24.9) |
134 (27) |
0.87 (0.60-1.26) |
0.449 |
|
GG |
346 (73) |
359 (72.3) |
− |
||
OR, odd ratio; CI, confidence interval.
Table 5: Association of SNPs with risk of stroke based on logistic tests adjusted by gender and age
SNP_ID |
Minor |
MAF |
MAF |
Additive model |
Dominant model |
Recessive model |
|||
|---|---|---|---|---|---|---|---|---|---|
OR (95% CI) |
p |
OR (95% CI) |
p |
OR (95% CI) |
p |
||||
rs886205 |
A |
0.129 |
0.138 |
0.75 (0.67-1.34) |
0.76 |
0.85 (0.58-1.23) |
0.38 |
6.39 (1.19-34.38) |
0.03* |
rs2238152 |
T |
0.304 |
0.278 |
1.03 (0.80-1.32) |
0.84 |
1.20 (0.86-1.66) |
0.28 |
0.66 (0.37-1.17) |
0.16 |
rs441 |
C |
0.307 |
0.278 |
1.05 (0.82-1.34) |
0.71 |
1.21 (0.88-1.68) |
0.24 |
0.72 (0.41-1.25) |
0.25 |
rs4646778 |
A |
0.307 |
0.278 |
1.05 (0.82-1.35) |
0.68 |
1.22 (0.88-1.28) |
0.24 |
0.74 (0.42-1.28) |
0.28 |
rs671 |
A |
0.133 |
0.139 |
0.91 (0.65-1.26) |
0.56 |
0.86 (0.59-1.25) |
0.43 |
1.27 (0.40-4.05) |
0.69 |
rs7296651 |
C |
0.146 |
0.141 |
1.04 (0.74-1.46) |
0.84 |
0.94 (0.65-1.35) |
0.74 |
9.29 (1.37-63.21) |
0.02* |
MAF, minor allele frequency; OR, odd ratio; CI, confidence interval.
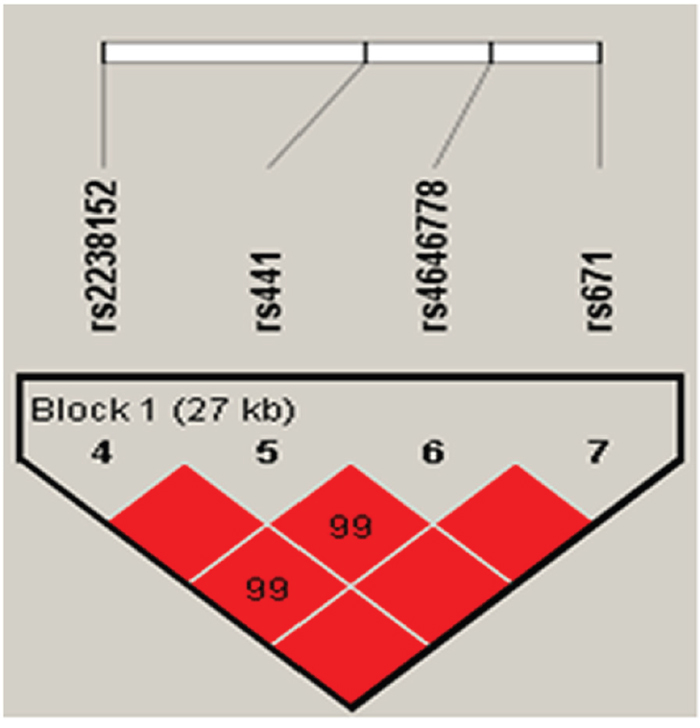
Figure 1: Linkage disequilibrium of polymorphic sites in the ALDH2 gene.
DISCUSSION
It is well known that genetic polymorphisms has an effect on the regulation of gene expression, which contributes to the differences between individuals in the susceptibility to a disease and its severity. Stroke is a heterogeneity disease, and the pathogenesis of stroke showed individualization in human. Stroke tends to run in families, it shows that genetic factors affect the development of the stroke. The Framingham study verified parental stroke by 65 years of age resulted in a 2.79-fold independent increase in the risk of offspring stroke [17]. In the present case–control study, we investigated the associations between the 6 SNPs of ALDH2 and risk of ischemic stroke. To the best of knowledge, we identified rs886205 and rs7296651 in the ALDH2 gene associated with an increased risk of ischemic stroke. In recent years, one research showed that carrying the wild-type allele of the ALDH2 polymorphism increased stroke risk among Korean men, but not in Korean women [18]. ALDH2 may have the same effect on cerebral infarction as on stroke, and cerebral infarction is one of the most common types of ischemic stroke. One previous study in Japanese reported that there was no significant association between the ALDH2 genotype and the presence of lacunar infarction, but the ALDH2*1/*1 genotype was significantly associated with the development lacunar infarcts [16]. Besides, Sun et al. [19] found that ALDH2 can prevent stroke by clearing 4-hydroxy-2-nonenal. In general, these findings indicate that ethnic differences among the ALDH2 gene variants may affect the development of stroke in different populations.
Stroke is a multifactorial disease attributable to genetic, environmental and other factors. Heavy alcohol consumption has been reported to be a positive risk factor for the susceptibility to stroke. Alcoholism is a complex polygenic behavioral disorder, because of complex gene and environment interactions. Current evidence indicates that ALDH2*2 may influence the risk of alcoholism in Koreans [20]. However, the effect of moderate drinking remains controversial. Soo et al. [21] identified that light to moderate alcohol consumption may be associated with a reduced risk of ischemic stroke in a Korean population. Mani et al. [22] suggested that the functional ALDH2 rs886205 polymorphism does not affect risk for risky alcohol consumption in German populations. This finding is consistent with our experimental results. Therefore, alcohol consumption is not a major factor for stroke, heavy alcohol consumption is just one of the main predisposing factor for stroke.
Heavy alcohol consumption also conferred by ALDH2 has been reported to be associated with hypertension in men [23]. ALDH2 is responsible for consumption ethanol in the process of blood pressure. ALDH2 genetic polymorphism can be altered ethanol pharmacokinetic properties, and leading to accumulation of the acetaldehyde following alcohol consumption. ALDH2 variants are considered to be governed by the accumulation of acetaldehyde, acetaldehyde is a major metabolic product of ethanol. The mutant ALDH2*2 gene was associated with an increased risk of hypertension in human, the blood pressure and ALDH2 enzymatic activity may be affected by gene and environment, such as life-style and ethnicity. For example, the main symptoms of alcoholism includes the myocardial hypertrophy, derangement of myofibrillary architecture, interstitial fibrosis, which may lead to a greater risk of cardiovascular anomalies such as stroke, and hypertension [24]. Heavy alcohol consumption exhibits a higher systolic blood pressure and a higher overall prevalence of hypertension compared with light to moderate drinkers. It is estimated that one tenth hypertensive cases may be the result of alcohol abuse. The correlation between alcohol intake and blood pressure appears to be independent of any other pathological variables such as diabetes mellitus, age, coronary heart disease, and cigarette smoking. Furthermore, moderate alcohol consumption displays reduced incidence of atherosclerosis, coronary heart diseases, and stroke. Given the hypertension is an important factor of cardiovascular morbidity, limiting alcohol intake is vital prevent measures for patients with hypertension.
Plenty of clinical and experimental studies have indicated that influence of ALDH2 genotype on blood pressure may be quite complex, depending on the amount of alcohol intake, blood pressure measurement, environmental and genetic factors. Based on these observations, it is plausible that the mutant ALDH2 serves as a beneficial factor against hypertension by altered drinking behavior and as an independent risk factor for hypertension. Ohsawa et al. [25] reported that ALDH2 functioned as a protector against oxidative stress and might influence the onset of hypertension and myocardial infarction. They also reported the rs671 GG genotype should be associated with a lower incidence of hypertension. The ALDH2 SNP rs671 was investigated in alcoholism Mongolians and the results indicated that ALDH2*1 genotype allele frequencies were significantly higher in patients with essential hypertension, in conjunction with a much lower frequency of the ALDH2*2 alleles in patients with essential hypertension. However, we found no differences in the distribution of the rs761 genetic variants in Han population in our study. These differences may be due to ethnic and regional differences, environmental factors, and dietary habits. In addition, ALDH2 decreased Ang II-induced ROS generation, thereby preventing ROS-induced constriction. This study describes that ALDH2 is beneficial in ROS-induced vascular contraction in the AngII hypertensive model. Then ALDH2 mutation is tied with the increased prevalence of hypertension [26]. However, such increased risk has been associative with alcoholism. Heavy alcohol consumption conferred by ALDH2 genotype has been reported to be associated with hypertension in men [26]. Considering the hypertension is an important factor of cardiovascular morbidity and stroke is a most important cause of cardiovascular disease, so it is necessary to control blood pressure which would further prevention of cardiovascular disease.
Our study has several advantages. Our population was enrolled from Hainan Province. The region has a high geographic stability, which could significantly reduce the potential confounding effects of the heterogeneous participants in the study. However, some limitations should be considered. First, the sample size (488 cases and 503 controls) of our study was relatively small. Second, correlations between polymorphisms and histological subtypes were not evaluated in this study. Third, we haven’t collected the data of vascular risk factors and alcohol drinking behavior to further analysis. Finally, the functions of the genetic variants and the mechanisms have not further explored in this study.
MATERIALS AND METHODS
Study population and data collection
For perform the study, we recruited a total of 991 participants (488 cases, 503 controls) from the People’s Hospital of Hainan Province during July 2012 to November 2015, and they are all Haikou and the surrounding areas civilians Stroke is divided into ischemic and Hemorrhage. The 488 ischemic stroke patients (325 males, 163 females; mean age 63.93 ± 11.06 years) all met the World Health Organization’s stratified criteria for ischemic stroke in our study, and further diagnosed with modern computerized tomography investigative modalities, such as computerized tomographic (CT) and magnetic resonance imaging (MRI). Patients only have a history of the previous stroke, head trauma, brain stroke precipitated during surgery or angiography, excluded with a bleeding diathesis, illicit drug use history, or concomitant serious medical illness such as malignancy, live cirrhosis, sepsis, meningoencephalitis, autoimmune disorders, evidence of cerebral vascular malformation or symptoms of a transient ischaemic attack. None of the patients had suffered any treatment before recruitment. All the patients were all recently diagnosed and histologically identified to be ischemic stroke cases. Meantime, we also recruited a random sample of 503 unrelated healthy individuals as controls (195 males, 308 females; mean age 50.36 ± 7.79 years) from the same hospital, and according to standard recruitment and exclusion criteria. All the participants are Chinese Han ethnic, and were informed the purpose and experimental procedures of the study.
Demographic and clinical data
A standardized epidemiological questionnaire including age, gender, smoking status, alcohol consumption, residential region, ethnicity, and family history of stroke, was used to collect demographic and personal data. We obtained clinical information for the patients through consulted with their treating physicians or from reviews of their medical charts. After signing an informed consent form, venous blood samples (5 ml) were obtained from each participant. The ethics Committee of the People’s Hospital of Hainan Province approved the use of blood samples and the protocol of the research.
SNP selection and genotyping
Using the NCBI database, we randomly selected six tag candidate SNPs (rs886205, rs2238152, rs441, rs4646778, rs671, and rs7296651) from the SNPs in ALDH2 gene with minor allele frequencies (MAFs) >5% in the Asian population. Genomic DNA was extracted from the whole blood samples from the 488 cases and the 503 controls using a Blood DNA Extraction Kit (GoldMag Co. Ltd., Xi'an City, China), and DNA concentrations were measured using a NanoDrop 2000 (Thermo Scientific, Waltham, Massachusetts, USA). MassARRAY Assay Design 3.0 Software (Sequenom, San Diego, CA, USA) was used to design the Multiplexed SNP MassEXTEND assay [27], and SNP genotyping was performed using the Sequenom MassARRAY RS1000 (Sequenom Inc., San Diego, CA, USA) system according to the standard protocol. Sequenom Typer 4.0 Software (Sequenom Inc., San Diego, California, USA) was used to manage and analyze the data.
Statistical analysis
We used SPSS version 17.0 statistical software (SPSS Inc., Chicago, IL, United States) and Excel (Microsoft Corp., Redmond, WA, United States) for statistical analyses. The Chi-squared test and the Student’s t-test were used to compare genotype frequencies and allele frequencies between patients and controls. The Hardy–Weinberg equilibrium (HWE) of each SNP was determined by the Chi-squared test, which compared the actual and expected frequencies of the genotypes in the controls. Genetic associations between SNPs and the risk of stroke were tested through various genetic models which including additive, dominant, and recessive models which using SNPStats webpage (http://bioinfo.iconcologia.net). The effects of the polymorphisms on the risk of stroke were reflected as odds ratios (ORs) with 95% confidence intervals (95% CIs) which were calculated by unconditional logistic regression analyses adjusted for age and gender [28]. A linkage disequilibrium (LD) analysis was performed by Haploview v4.2 with genotype data. The pattern of LD was analyzed using D’ and evaluated the haplotypes of the candidate SNPs. All of the statistical tests were two-sided, and p-values < 0.05 were statistically significant.
CONCLUSION
In summary, in this study of a Han Chinese sample, we identified two novel ALDH2 tSNPs associated with ischemic stroke susceptibility which indicates that ALDH2 gene may be provide new insights into the etiology of ischemic stroke. To better understand the relationships between gene polymorphisms and stroke progression, additional genetic risk factors and new candidate genes should be identified and analyzed.
Ethics approval and consent to participate
This research was performed in accordance with the Helsinki Declaration and was approved by the ethics committee of People’s Hospital of Hainan Province (Ethics Committee of Research in Humans). Informed consent for this research was conducted under the approval of the ethics committee of People’s Hospital of Hainan Province.
Abbreviations
ALDH2, aldehyde dehydrogenase 2, SNP, single-nucleotide polymorphisms, CI, confidence interval, GWAS, genome-wide association studies, CT, computerized tomographic, MRI, magnetic resonance imaging, HWE, Hardy–Weinberg equilibrium, LD, linkage disequilibrium.
Author contributions
SS and JH drafted the manuscript. Y Zhang, RX, and MY performed the statistical analyses and LW helped edit the manuscript. YR and Y Zhu performed the genotyping. TJ and YX reviewed all the genotypes. TJ assisted in the study design. TJ and YX conceived the study and co-supervised the work. All the authors have read and approved the final manuscript.
ACKNOWLEDGMENTS
We are grateful to all the patients and individuals in the study who made this work possible. We would also like to thank the clinicians and hospital staffs who contributed to data collection for this study.
CONFLICTS OF INTEREST
The authors declare that they have no competing interests.
FUNDING
This work was supported by the Hainan natural fund (No. 310147) and the National Natural Science Foundation (No. 81360190). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
REFERENCES
1. Donnan G, Fisher M, Macleod M, Davis S. Stroke. Lancet. 2008; 371:1612-1623.
2. Jauch EC, Cucchiara B, Adeoye O, Meurer W, Brice J, Chan YY, Gentile N, Hazinski MF. Part 11: adult stroke: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2010; 122:S818-S828.
3. Marsh JD, Keyrouz SG. Stroke prevention and treatment. J Am Coll Cardiol. 2010; 56:683-691.
4. Poungvarin N. Stroke in the developing world. Lancet. 1998; 352:S19-S22.
5. Traylor M, Farrall M, Holliday EG, Sudlow C, Hopewell JC, Cheng YC, Fornage M, Ikram MA, Malik R, Bevan S, Thorsteinsdottir U, Nalls MA, Longstreth W, et al. Genetic risk factors for ischaemic stroke and its subtypes (the METASTROKE collaboration): a meta-analysis of genome-wide association studies. Lancet Neurol. 2012; 11:951-962.
6. Bevan S, Traylor M, Adib-Samii P, Malik R, Paul NL, Jackson C, Farrall M, Rothwell PM, Sudlow C, Dichgans M, Markus HS. Genetic heritability of ischemic stroke and the contribution of previously reported candidate gene and genomewide associations. Stroke. 2012; 43:3161-3167.
7. Manolio TA, Collins FS, Cox NJ, Goldstein DB, Hindorff LA, Hunter DJ, McCarthy MI, Ramos EM, Cardon LR, Chakravarti A, Cho JH, Guttmacher AE, Kong A, et al. Finding the missing heritability of complex diseases. Nature. 2009; 461:747-753.
8. Guo JM, Liu AJ, Zang P, Dong WZ, Ying L, Wang W, Xu P, Song XR, Cai J, Zhang SQ. ALDH2 protects against stroke by clearing 4-HNE. Cell Res. 2013; 23:915-930.
9. Yao CT, Cheng CA, Wang HK, Chiu SW, Chen YC, Wang MF, Yin SJ, Peng GS. The role of ALDH2 and ADH1B polymorphism in alcohol consumption and stroke in Han Chinese. Hum Genomics. 2011; 5:569-576.
10. Stewart MJ, Malek K, Crabb DW. Distribution of messenger RNAs for aldehyde dehydrogenase 1, aldehyde dehydrogenase 2, and aldehyde dehydrogenase 5 in human tissues. J Invest Med. 1996; 44:42-46.
11. Klyosov AA. Kinetics and specificity of human liver aldehyde dehydrogenases toward aliphatic, aromatic, and fused polycyclic aldehydes. Biochemistry. 1996; 35:4457-4467.
12. Zhao J, You L, Wang DW, Cui W. Impacts of common variants in ALDH2 on coronary artery disease patients. Gene. 2016; 585:104-109.
13. Zhang R, Wang JL, Chen YG. [Progress in the development of diabetes mellitus and its complications: the role of ALDH2]. [Article in Chinese]. Sheng Li Ke Xue Jin Zhan. 2015; 46:229-232.
14. Jia K, Wang H, Dong P. Aldehyde dehydrogenase 2 (ALDH2) Glu504Lys polymorphism is associated with hypertension risk in Asians: a meta-analysis. Int J Clin Exp Med. 2015; 8:10767-10772.
15. Nagasawa H, Wada M, Arawaka S, Kawanami T, Kurita K, Daimon M, Adachi M, Hosoya T, Emi M, Muramatsu M, Kato T. A polymorphism of the aldehyde dehydrogenase 2 gene is a risk factor for multiple lacunar infarcts in Japanese men: the Takahata Study. Eur J Neurol. 2007; 14:428-434.
16. Kato N, Takeuchi F, Tabara Y, Kelly TN, Min JG, Sim X, Wan TT, Chen CH, Zhang Y, Yamamoto K. Meta-analysis of genome-wide association studies identifies common variants associated with blood pressure variation in east Asians. Nat Genet. 2011; 43:531-538.
17. Sudha S, Alexa B, Aleksandra P, Himali JJ, Margaret KH, Stephanie D, Destefano AL, Romero JR, Kase CS, Wolf PA. Parental occurrence of stroke and risk of stroke in their children: the Framingham study. Circulation. 2010; 121:1304-1312.
18. Chol S, Kyubum K, Cho NH, Seonghwan K, Inkyung B. Sex-specific differences in the association of a common aldehyde dehydrogenase 2 gene polymorphism and alcohol consumption with stroke risk in a Korean population: a prospective cohort study. Nutr Res Pract. 2015; 76:418–424.
19. Sun A, Ren J. ALDH2, a novel protector against stroke? Cell Res. 2013; 23:874-875.
20. Kim DJ, Choi IG, Park BL, Lee BC, Ham BJ, Yoon S, Bae JS, Cheong HS, Shin HD. Major genetic components underlying alcoholism in Korean population. Hum Mol Genet. 2008; 17:854-858.
21. Lee SJ, Cho YJ, Kim JG, Ko Y, Hong KS, Park JM, Kang K, Park TH, Park SS, Lee KB. Moderate alcohol intake reduces risk of ischemic stroke in Korea. Neurology. 2015; 85:1950-1956.
22. Nassab MH, Rhein M, Heese P, Glahn A, Frieling H, Linnebank M, Bleich S, Kornhuber J, Heberlein A, Grallert H. No association between the ALDH2 promoter polymorphism rs886205, alcohol dependence, and risky alcohol consumption in a German population. Psychiatr Genet. 2015; 25:41-42.
23. Ruth MP, Alexander P, Nihan K, Alexandre S, Robert R, Cox DR, Hinds DA, Pennacchio LA, Anne TH, Folsom AR. A common allele on chromosome 9 associated with coronary heart disease. Science. 2007; 316:1488-1491.
24. Guo R, Ren J. Alcohol and acetaldehyde in public health: from marvel to menace. Int J Environ Res Public Health. 2010; 7:1285-1301.
25. Ohsawa I, Kamino K, Nagasaka K, Ando F, Niino N, Shimokata H, Ohta S. Genetic deficiency of a mitochondrial aldehyde dehydrogenase increases serum lipid peroxides in community-dwelling females. J Hum Genet. 2003; 48:404-409.
26. Takagi S, Baba S, Iwai N, Fukuda M, Katsuya T, Higaki J, Mannami T, Ogata J, Goto Y, Ogihara T. The aldehyde dehydrogenase 2 gene is a risk factor for hypertension in Japanese but does not alter the sensitivity to pressor effects of alcohol: the Suita study. Hypertens Res. 2001; 24:365-370.
27. Gabriel S, Ziaugra L, Tabbaa D. SNP genotyping using the Sequenom MassARRAY iPLEX platform. Curr Protoc Hum Genet. 2009; Chapter 2:2.12.11-12.12.16.
28. Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D, Maller J, Sklar P. de Bakker PI, Daly MJ, Sham PC. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet. 2007; 81:559-575.

