INTRODUCTION
Colorectal cancer (CRC) is among the leading causes of cancer-related deaths worldwide [1, 2]. The median age of CRC patients was around 69 years old [3]. Decision-making with regards to treatment for older CRC patients may be complicated by certain age-related physiological changes, impaired functional status, limited social support, ability to tolerate treatment toxicity, and presence of comorbidities [4–7]. Many clinical trials have set 75 years as their upper age limit for study enrollment, excluding the oldest old subset [8]. Evidence for the treatment in older adults were derived primarily from individual trial or pooled subgroup analyses [9, 10] [11, 12] and large population-based studies [13, 14]. Because these studies only evaluated the effects of chemotherapy on CRC, it remains unknown whether patients over 75 years of age would benefit from surgery or radiation therapy. Moreover, little is known about clinicopathologic features and prognostic factors specific for this older and frail subset of CRC patients.
To address these questions, we analyzed the clinical data of CRC patients in the Surveillance, Epidemiology, and End Results (SEER) database. Herein, our analysis unveiled clinical-pathological factors of both younger and older CRC patients. We have further examined prognostic factors for the oldest old subset.
RESULTS
Patient baseline characteristics
A total of 293,616 patients with colorectal cancer, adenocarcinoma type, were identified from the SEER database. Their median age was 68 years. Among these patients, 100,719 (34.3 %) were 75 years or older and 192,897 (65.7%) were younger than 75. Table 1 shows the basic characteristics of these two group of patients.
Table 1: Basic characteristics between younger and older patients
Age<75 |
Age≥75 |
P value |
|
|---|---|---|---|
Gender |
|||
Female |
84,989 (44.06) |
55,597 (55.2) |
|
Male |
107,908 (55.94) |
45,122 (44.8) |
<0.001 |
Age |
|||
(Mean ± SD) |
59.78±10.10 |
81.95±5.00 |
<0.001 |
AJCC 6th TNM stage |
|||
I |
50,354 (26.10) |
27,716 (27.52) |
|
II |
47,324 (24.53) |
32,582 (32.35) |
|
III |
55,333 (28.69) |
25,532 (25.35) |
|
IV |
39,886 (20.68) |
14,889 (14.78) |
<0.001 |
AJCC 6th T stage |
|||
T0 |
242 (0.13) |
117 (0.12) |
|
T1 |
37,325 (19.35) |
16,801 (16.68) |
|
T2 |
25,903 (13.43) |
15,722 (15.61) |
|
T3 |
94,723 (49.11) |
51,414 (51.05) |
|
T4 |
26,291 (13.63) |
12,755 (12.66) |
|
TX |
8,413 (4.36) |
3,910 (3.88) |
<0.001 |
AJCC 6th N stage |
|||
N0 |
108,192 (56.09) |
65,016 (64.55) |
|
N1 |
49,003 (25.40) |
21,359 (21.21) |
|
N2 |
30,394 (15.76) |
11,801 (11.72) |
|
NX |
5,308 (2.75) |
2,543 (2.52) |
<0.001 |
AJCC 6th M stage |
|||
M0 |
153,011 (79.32) |
85,830 (85.22) |
|
M1 |
39,886 (20.68) |
14,889 (14.78) |
<0.001 |
Histology |
|||
Adenocarcinoma |
175,304 (90.88) |
89,796 (89.15) |
|
Mucinous |
15,390 (7.98) |
9,935 (9.86) |
|
Signet ring cell |
2,203 (1.14) |
988 (0.98) |
<0.001 |
Location |
|||
RSCC |
57,291 (29.70) |
45,584 (45.26) |
|
LSCC |
76,463 (39.64) |
29,534 (29.32) |
|
Rectal cancer |
42,952 (22.27) |
14,457 (14.35) |
|
Others |
16,191 (8.39) |
11,144 (11.06) |
<0.001 |
Race |
|||
Caucasian |
148,882 (77.18) |
85,964 (85.35) |
|
African |
25,659 (13.30) |
7,845 (7.79) |
|
American |
|||
Asian |
14,675 (7.61) |
6,041 (6.00) |
|
Others |
3,681 (1.91) |
869 (0.86) |
<0.001 |
Grade |
|||
Well |
16,070 (8.33) |
8,361 (8.30) |
|
Moderate |
127,468 (66.08) |
65,147 (64.68) |
|
Poorly |
29,027 (15.05) |
17,625 (17.5) |
|
Undifferentiated |
3,106 (1.61) |
1,870 (1.86) |
|
Unknown |
17,226 (8.93) |
7,716 (7.66) |
<0.001 |
Surgery |
|||
Yes |
173,503 (89.95) |
90,026 (89.38) |
|
No |
19,175 (9.94) |
10,581 (10.51) |
|
Unknown |
219 (0.11) |
112 (0.11) |
<0.001 |
Radiation |
|||
Yes |
33,375 (17.30) |
7,535 (7.48) |
|
No |
157,700 (81.75) |
92,411 (91.75) |
|
Unknown |
1,822 (0.94) |
773 (0.77) |
<0.001 |
Tumor size |
|||
Mean±SD (mm) |
45.79±35.29 |
46.15±33.60 |
<0.001 |
Diagnosis |
|||
Microscopically confirmed |
192,692 (99.89) |
100,508 (99.79) |
|
Not microscopically confirmed |
152 (0.08) |
189 (0.19) |
|
Unknown |
53 (0.03) |
22 (0.02) |
<0.001 |
Married status |
|||
Married |
114,575 (59.40) |
44,901 (44.58) |
|
Unmarried |
69,453 (36.01) |
51,228 (50.86) |
|
Unknown |
8,869 (4.60) |
4,590 (4.56) |
<0.001 |
Insurance status |
|||
Insured |
126,038 (65.34) |
65,862 (65.39) |
|
Uninsured |
6,341 (3.29) |
240 (0.24) |
|
Unknown |
60,518 (31.37) |
34,617 (34.37) |
<0.001 |
Abbreviation: SD: Standard deviation; AJCC: American Joint Committee on Cancer TNM: Tumor-Node-Metastasis; RSCC: right-sided colon cancer; LSCC: left-sided colon cancer.
In comparison to the young, the older group had a significantly higher proportion of female (55.2% vs. 44.1%, P < 0.001). For the stage of Tumor-Node-Metastasis (TNM), the older patients had a higher percentage of stage I and II diseases. Signet-ring cell carcinoma were less common in the older group, while poorly differentiated and undifferentiated adenocarcinoma were more common in older patients. The older patients had more tumors within the right-sided colon, more Caucasians, and more who were unmarried. In terms of treatment for CRC, significantly fewer patients in the older group received surgery or radiotherapy.
Survival analysis
The 5-year cause-specific survival (CSS) for the entire study population was 55.71% [95% confidence interval (CI): 55.50%-55.91%]. There were 55,436 deaths (55.04 %) in the older group and 63,247 (32.79%) in the young group. Consistently, the 5-year CSS was significantly higher in the younger patients than in the older patients, 63.14% vs. 41.98%, P<0.001 (Figure 1). The 5-year other cause of survival rate was 91.83% and 69.23% for the younger and older patients, respectively, P<0.001.
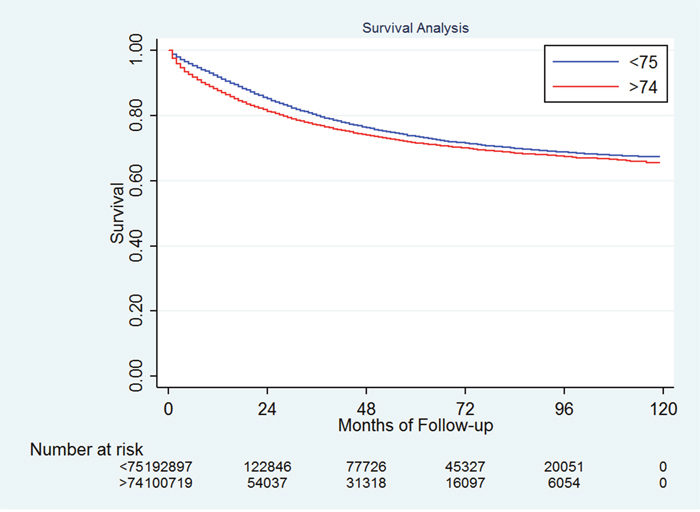
Figure 1: Survival difference between the younger and older patients.
We then analyzed the prognostic factors for patients 75 years old and over (Table 2). Not surprisingly, the stage of TNM was significantly correlated with CSS – the 5-year CSS were 55.78%, 50.66%, 37.44%, and 4.57% for patients with stage I, II, III, and IV, respectively, P<0.001. As for the histology subtypes, patients with signet ring cells had a worse 5-year CSS compared to those with mucinous adenocarcinoma or other adenocarcinoma (25.63% vs. 40.91% vs. 42.28%, P<0.001). When we analyzed the 5-year CSS in patients with different tumor grades, we found that the disease-specific survival worsened as the grade progressed from “well” to “un-differentiated” (49.85%, 45.03%, 35.05%, and 33.25% for well-, moderately, poorly, and un-differentiated tumors, respectively, P<0.001).
Table 2: Survival analysis in older patients
5yr-CSS |
95% CI |
P value |
|
|---|---|---|---|
Gender |
|||
Male |
39.34% |
38.82%-39.87% |
|
Female |
44.12% |
43.64%-44.60% |
<0.001 |
Marry status |
|||
Married |
45.62% |
45.09%-46.16% |
|
Unmarried |
38.57% |
38.08%-39.05% |
|
Unknown |
44.73% |
42.99%-46.46% |
<0.001 |
Insurance status |
|||
Insured |
41.98% |
41.48%-42.47% |
|
Uninsured |
45.83% |
37.47%-53.78% |
|
Unknown |
41.94% |
41.41%-42.46% |
0.5046 |
Ethnicity |
|||
Caucasian |
42.24% |
41.85%-42.62% |
|
African American |
34.83% |
33.59%-36.06% |
|
Asian |
47.35% |
45.84%-48.83% |
|
Others |
44.68% |
40.66%-48.62% |
<0.001 |
Site |
|||
LSCC |
42.39% |
41.74%-43.03% |
|
RSCC |
44.43% |
43.90%-44.96% |
|
Rectum |
36.30% |
35.38%-37.21% |
<0.001 |
AJCC 6th TNM stage |
|||
I |
55.78% |
55.10%-56.46% |
|
II |
50.66% |
50.02%-51.30% |
|
III |
37.44% |
36.74%-38.14% |
|
IV |
4.57% |
4.18%-5.00% |
<0.001 |
Grade |
|||
Well differentiated |
49.85% |
48.62%-51.07% |
|
Moderately differentiated |
45.03% |
44.58%-45.47% |
|
Poorly differentiated |
35.05% |
34.25%-35.84% |
|
Undifferentiated |
33.25% |
30.55%-35.97% |
|
Unknown |
25.53% |
24.42%-26.65% |
<0.001 |
Histology |
|||
Other adenocarcinoma |
42.28% |
41.90%-42.66% |
|
Mucinous adenocarcinoma |
40.91% |
39.80%-42.00% |
|
Signet ring cell |
25.63% |
22.55%-28.80% |
<0.001 |
Size |
|||
<=40mm |
48.52% |
47.97%-49.07% |
|
>40mm |
37.05% |
36.59%-37.50% |
<0.001 |
Surgery |
|||
Yes |
73.0% |
72.7%-73.2% |
|
No |
9.3% |
8.6%-10.1% |
|
Unknown |
26.8% |
19.8%-34.3% |
<0.001 |
Radiation |
|||
No |
46.0% |
44.0%-48.0% |
|
Yes |
69.8% |
69.5%-70.1% |
|
Unknown |
62.5% |
59.1%-65.8% |
<0.001 |
Abbreviation: CSS: Cause specific survival; CI: Confidence interval; AJCC: American Joint Committee on Cancer TNM: Tumor-Node-Metastasis; RSCC: right-sided colon cancer; LSCC: left-sided colon cancer.
Among the older CRC patients, about 89% underwent surgery for CRC and 7.48% received radiation therapy (78.3% of patients who received radiotherapy were rectal adenocarcinoma patients). The older patients who have underwent surgery had a significantly better prognosis in comparison to those who have not.
Moreover, females had a better prognosis compared to the males. The other factors that affect prognosis includes marital status, ethnicity, location of primary tumor, and tumor size. In contrast, whether the patients were covered by insurance or not had no impact on the disease-specific survival.
Multivariate analysis
Variables showing a trend for association with survival (P < 0.05) were selected in the cox proportional hazards model. Gender, married status, ethnicity, location, TNM stage, histologic subtypes, grade, tumor size, radiation as well as surgery were all independent prognostic factors in the multivariable analysis (Table 3).
Table 3: Multivariate analysis of survival in older patients
Hazard ratio |
Standard error |
P value |
95% confidence interval |
|
|---|---|---|---|---|
Sex |
||||
Male |
Reference |
|||
Female |
0.89 |
0.005 |
<0.001 |
0.86-0.91 |
Ethnicity |
||||
Caucasian |
Reference |
|||
Asian |
0.83 |
0.011 |
<0.001 |
0.81-0.86 |
African-American |
1.02 |
0.009 |
0.012 |
1.01-1.04 |
Others |
0.87 |
0.02 |
<0.001 |
0.83-0.92 |
Married status |
||||
Married |
Reference |
|||
Unmarried |
1.44 |
0.009 |
<0.001 |
1.42-1.46 |
Unknown |
1.12 |
0.018 |
<0.001 |
1.08-1.15 |
Site |
||||
RSCC |
Reference |
|||
LSCC |
0.82 |
0.006 |
<0.001 |
0.81-0.83 |
Rectum |
0.86 |
0.009 |
<0.001 |
0.84-0.87 |
AJCC 6th TNM stage |
||||
I |
Reference |
|||
II |
1.29 |
0.013 |
<0.001 |
1.27-1.32 |
III |
1.72 |
0.017 |
<0.001 |
1.69-1.76 |
IV |
5.23 |
0.054 |
<0.001 |
5.12-5.33 |
Histology subtype |
||||
Adenocarcinoma |
Reference |
|||
Mucinous adenocarcinoma |
1.13 |
0.012 |
<0.001 |
1.11-1.15 |
Signet ring cell |
1.42 |
0.034 |
<0.001 |
1.36-1.49 |
Grade |
||||
Well differentiated |
Reference |
|||
Moderate differentiated |
1.09 |
0.014 |
<0.001 |
1.07-1.12 |
Poorly differentiated |
1.46 |
0.021 |
<0.001 |
1.42-1.50 |
Undifferentiated |
1.61 |
0.041 |
<0.001 |
1.54-1.69 |
Unknown |
1.16 |
0.019 |
<0.001 |
1.13-1.20 |
Surgery |
||||
No |
Reference |
|||
Yes |
0.38 |
0.004 |
<0.001 |
0.37-0.39 |
Unknown |
0.71 |
0.028 |
<0.001 |
0.66-0.77 |
Radiation |
||||
Yes |
Reference |
|||
No |
1.25 |
0.014 |
<0.001 |
1.22-1.28 |
Unknown |
1.30 |
0.042 |
<0.001 |
1.22-1.39 |
Size |
||||
<=40mm |
Reference |
|||
>40mm |
1.09 |
0.007 |
<0.001 |
1.08-1.10 |
Abbreviation: RSCC: right-sided colon cancer; LSCC: left-sided colon cancer; AJCC: American Joint Committee on Cancer TNM: Tumor-Node-Metastasis.
Who benefited from surgery?
To better understand the role and benefit of surgery in treatment of patients with CRC who were over 75 years of age patients, we analyzed these patients according to their disease stage and age. We first stratified the patients into 5 age groups – 75-79, 80-84, 85-89, 90-94, and >94 years old. Surgery did not bring survival benefit to the patients who were older than 94 years old and with stage III diseases. As for the rest of the patients, those who received surgery for CRC had a significantly improved 5-year CSS as compared to those did not undergo such surgery (Table 4). Moreover, for the patients with the same stage of TNM, the prognosis for CRC worsened with increasing age.
Table 4: The 5-year CSS for older patients with or without surgery
Surgery |
No surgery |
P value |
|
|---|---|---|---|
Stage I |
|||
75-79 years old |
70.00% (68.95%-71.01%) |
28.38% (23.92%-32.98%) |
<0.001 |
80-84 years old |
58.98% (57.75%-60.19%) |
15.99% (12.93%-19.35%) |
<0.001 |
85-89 years old |
47.41% (45.73%-49.06%) |
9.91% (7.35%-12.92%) |
<0.001 |
90-94 years old |
36.83% (33.76%-39.90%) |
1.80% (0.59%-4.32%) |
<0.001 |
>94 years old |
22.91% (16.53%-29.93%) |
2.06% (0.2%-8.85%) |
<0.001 |
Stage II |
|||
75-79 years old |
61.73% (60.66%-62.78%) |
17.31% (11.83%-23.67%) |
<0.001 |
80-84 years old |
54.07% (52.95%-55.18%) |
13.18% (9.18%-17.92%) |
<0.001 |
85-89 years old |
42.97% (41.57%-44.36%) |
8.35% (4.47%-13.75%) |
<0.001 |
90-94 years old |
28.66% (26.46%-30.90%) |
8.45% (3.71%-15.67%) |
<0.001 |
>94 years old |
21.07% (16.64%-25.87%) |
0 |
0.0006 |
Stage III |
|||
75-79 years old |
47.81% (46.64%-48.98%) |
9.15% (4.25%-16.35%) |
<0.001 |
80-84 years old |
37.81% (36.60%-39.02%) |
9.56% (4.81%-16.26%) |
<0.001 |
85-89 years old |
27.94% (26.46%-29.44%) |
5.00% (1.49%-11.86%) |
<0.001 |
90-94 years old |
18.75% (16.54%-21.07%) |
8.62% (2.75%-18.81%) |
<0.001 |
>94 years old |
11.99% (7.99%-16.87%) |
15.41% (1.12%-45.95%) |
0.2678 |
Abbreviation: CSS: cause specific survival.
DISCUSSION
Colorectal cancer is a highly prevalent malignancy in older adults worldwide [15–17]. With rapid growth of older adults in numbers, particular the oldest old subset aged 75 years and over, the number of older patients with CRC and those who seek treatment will increase rapidly. However, many clinical trials set an upper age limit of 75 years for study enrollment. Even in trials with no such age limit, they only enrolled a small number of patients with advanced age. These limited number of patients are unlikely representative of the general geriatric population with CRC. To address this important issue, we took the advantage of the existing data in the SEER, a composite population-based cancer registry covering several discrete geographic regions. We found that about one third of the CRC patients were of age 75 or over. We then further analyzed their clinicopathologic features as well as prognostic factors.
Firstly, we identified a number of unique clinicopathologic features in this oldest old subset. In comparison to the young group, these older patients were more likely single or widowed. They had larger and more poorly differentiated tumor and their tumor was more likely located in the ascending colon. On the other hand, these older patients had less metastasis of primary tumor and less signet ring cell carcinoma. Nonetheless, the 5-year CSS was lower in the older patients (P < 0.05). Two previous studies reported age as an independent negative prognostic factor in stage I-IV colon cancers [3, 18] in which “older patients” were defined differently; one study used median age (69 years) as the cutoff value [3] while the other used 40 years of age as the cutoff value [18]. This study, for the first time, reports 5-year CSS in patients with CRC who were 75 years old and over, which was nearly 42%.
Much remains to be learned about the prognostic factors for this oldest subset of elderly patients with CRC. In this study, we identified that gender, marital status, ethnicity, the stage of TNM, grade, tumor histologic subtype, tumor size, surgical intervention and radiotherapy were all independent prognostic factors for these older patients. These prognostic factors were not much different from other age groups [3, 14, 19].
As this oldest subset of elderly patients are more often challenged by age-related physiological changes, impaired functional status, limited social support, decreased ability to tolerate treatment toxicity, and presence of comorbidities, it is unclear if they would benefit from cancer treatments similar to the younger patients. Here, we report that surgical intervention provided survival benefit for most patients in this age group except for those who were both over 94 years of age and had stage III disease. Radiation therapy also offered survival benefits for patients with rectal cancer in this age group. We focused our analysis on patients with rectal cancer because they accounted for over 80% of those receiving radiation therapy. Consistent with these results, Pérez Domínguez L et al. found that age did not affect the prognosis after colon cancer resection but was associated with more postoperative morbidity and mortality [22].
This study has limitations. For example, information about chemotherapy was not available in the SEER database. Thus, potential survival benefit of chemotherapy for the oldest old subset of patients with CRC could not be determined in this study, However, subgroup and pooled analyses o phase III clinical trials suggested that the relatively fit older patients with CRC who met the traditional clinical trial inclusion criteria were likely to experience survival benefits from combination Oxaliplatin as the first line therapy similar to the younger patients [20, 21], supporting the hypothesis that the oldest old subset patient would also benefit from first line chemotherapy. Another limitation is that no data were available in the SEER database on comorbidities which are prevalent in the oldest old subset patients and known to influence prognosis and treatment decision-making for these patients. Finally, given that this is a retrospective cohort study, there is potential bias of nonrandomized data between the focused oldest old subset and younger patients with CRC. Despite these limitations, our study has identified specific clinicopathologic features and prognostic factors of the elderly patients with CRC who are 75 years and older. These data will help guide treatment decision-making for this specific and rapid growing group of elderly patients with CRC. In summary, our findings, for the first time showed that the oldest old subset of elderly CRC patients not only had a relatively poor prognosis compared to their younger counterparts but also could potentially benefit from surgery and radiotherapy.
MATERIALS AND METHODS
Database
The Surveillance, Epidemiology, and End Results (SEER) database is the largest publicly available cancer dataset. It is a composite population-based cancer registry. The SEER research data include cancer incidence and prevalence as well as patient demographics as tabulated by age, sex, race/ethnicity, and year of cancer diagnosis. The specific dataset used for this study was the SEER Program (http://www.seer.cancer.gov) Research Data (1973-2013).
Outcome variables
The anatomic sites of the left colon, right colon, and rectum were categorized according to the International Classification of Diseases for Oncology, third edition (ICD-0-3) topography codes. Right-sided colon cancers were identified by using the following SEER cancer site codes: cecum (ICD-0-3 code C18.0), ascending colon (Code C18.2), hepatic flexure (Code C18.3), and transverse colon (Code C18.4); left-sided colon cancers were identified by using the following codes: splenic flexure (Code C18.5), descending colon (code C18.6), sigmoid colon (code C18.7), and rectosigmoid (code C19.9); rectal cancer was identified by using the code C20.9.
In this study, only adenocarcinoma patients were enrolled (SEER histology codes: signet ring cell, 8490; mucinous adenocarcinoma, 8480 and 8481; other adenocarcinoma: 8140 to 8147, 8210 to 8211, 8220 to 8221, and 8260 to 8263).
For the insurance status, individuals in the groups “Any Medicaid”, “Insured” and “Insured/No specifics” were clustered together as “Insured group”.
Patient population
The study population was derived from the SEER cancer registry. Individuals identified as colorectal adenocarcinoma patients from 2004 to 2013, inclusive, were eligible for our study. Patients were excluded if they (1) had more than one primary cancer and the CRC was not the first to appear, and/or (2) had unknown cause(s) of death or unknown survival months. All the patients included in the study had active follow up and a survival of over 1 month.
Age, sex, tumor stage according to the American Joint Committee on Cancer (AJCC) Cancer Staging Manual (6th edition, 2004), tumor histological subtype, tumor grade, insurance status, marital status, time of disease diagnosis, survival time, and CSS were extracted from the SEER database.
Statistical methods
The patients’ demographic characteristics and tumor characteristics are summarized using descriptive statistics (Table or Figure). Comparisons of specific categorical variables between the older and younger patients were performed using the Chi squared test, and continuous variables were compared using the Student’s t-test. The primary endpoint of this study was the 5-year CSS, as calculated from the date of diagnosis to the date of cancer-specific death. Deaths attributed to CRC were treated as events, and deaths from other causes were treated as censored observations. Survival function estimation and comparison between older and younger patients were performed using Kaplan–Meier estimates and log-rank test, respectively. The multivariate Cox proportional hazard model was used to evaluate the hazard ratio (HR) and the 95 % CI for all the known prognostic factors for CRC. All statistical analyses were performed using the Intercooled Stata 13.0 (Stata Corporation, College Station, TX). Statistical significance was set at two-sided P < 0.05.
Abbreviations
AJCC: American Joint Committee on Cancer; CI: confidence interval; CRC: Colorectal cancer; CSS: cause-specific survival; HR: hazard ratio; ICD-O: International Classification of Diseases for Oncology; SEER: Surveillance, Epidemiology, and End Results; TNM: Tumor-Node-Metastasis.
Author contributions
Conceptualization: Mingfang Zhao, Haiyan Zhang. Methodology: Hans Liu, Xin Meng, Jun Yu. Formal analysis: Qi Wang, Qiao Zhou, Yanqing Tang. Writing: Mingfang Zhao, Sean X. Leng, and Haiyan Zhang. Supervision: Haiyan Zhang. Project administration: Mingfang Zhao, Hans Liu. Funding acquisition: Haiyan Zhang, Xin Meng.
ACKNOWLEDGMENTS
We would like to thank the staff of the National Cancer Institute and their colleagues across the United States, and Information Management Services, Inc., all who have been involved with the SEER Program.
CONFLICTS OF INTEREST
All authors declared that there is no conflicts of interest.
FUNDING
This work was supported by the National Natural Science Foundation of China (Grant number: 81301838, 81271292) and funding from the Irma and Paul Milstein Medical Asian American Partnership (MMAAP) Foundation Program for Senior Health fellowship award, supported by the MMAAP Foundation (http://www.mmaapf.org) to Dr. Haiyan Zhang.
REFERENCES
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015; 65: 5-29.
2. Chen W, Zheng R, Zeng H, Zhang S. The updated incidences and mortalities of major cancers in China, 2011. Chin J Cancer. 2015; 34: 502-507.
3. Lin J, Qiu M, Xu R, Dobs AS. Comparison of survival and clinicopathologic features in colorectal cancer among African American, Caucasian, and Chinese patients treated in the United States: Results from the surveillance epidemiology and end results (SEER) database. Oncotarget. 2015; 6: 33935-33943. https://doi.org/10.18632/oncotarget.5223.
4. Moth EB, Vardy J, Blinman P. Decision-making in geriatric oncology: systemic treatment considerations for older adults with colon cancer. Expert Rev Gastroenterol Hepatol. 2016; 10: 1321-1340.
5. Razenberg LG, van Erning FN, Pruijt HF, Ten Tije AJ, van Riel JM, Creemers GJ, Lemmens VE. The impact of age on first-line systemic therapy in patients with metachronous metastases from colorectal cancer. J Geriatr Oncol. 2017; 8:37-43.
6. Haller DG, O'Connell MJ, Cartwright TH, Twelves CJ, McKenna EF, Sun W, Saif MW, Lee S, Yothers G, Schmoll HJ. Impact of age and medical comorbidity on adjuvant treatment outcomes for stage III colon cancer: a pooled analysis of individual patient data from four randomized, controlled trials. Ann Oncol. 2015; 26: 715-724.
7. Seymour MT, Thompson LC, Wasan HS, Middleton G, Brewster AE, Shepherd SF, O'Mahony MS, Maughan TS, Parmar M, Langley RE. Chemotherapy options in elderly and frail patients with metastatic colorectal cancer (MRC FOCUS2): an open-label, randomised factorial trial. Lancet. 2011; 377: 1749-1759.
8. Andre T, Boni C, Navarro M, Tabernero J, Hickish T, Topham C, Bonetti A, Clingan P, Bridgewater J, Rivera F, de Gramont A. Improved overall survival with oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment in stage II or III colon cancer in the MOSAIC trial. J Clin Oncol. 2009; 27: 3109-3116.
9. Adjuvant chemotherapy with oxaliplatin, in combination with fluorouracil plus leucovorin prolongs disease-free survival, but causes more adverse events in people with stage II or III colon cancer Abstracted from: Andre T, Boni C, Mounedji-Boudiaf L, et al. Multicenter international study of oxaliplatin/5-fluorouracil/leucovorin in the adjuvant treatment of colon cancer (MOSAIC) investigators. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med 2004;350:2343-51. Cancer Treat Rev. 2004; 30: 711-713.
10. McCleary NJ, Meyerhardt JA, Green E, Yothers G, de Gramont A, Van Cutsem E, O'Connell M, Twelves CJ, Saltz LB, Haller DG, Sargent DJ. Impact of age on the efficacy of newer adjuvant therapies in patients with stage II/III colon cancer: findings from the ACCENT database. J Clin Oncol. 2013; 31: 2600-2606.
11. Yothers G, O'Connell MJ, Allegra CJ, Kuebler JP, Colangelo LH, Petrelli NJ, Wolmark N. Oxaliplatin as adjuvant therapy for colon cancer: updated results of NSABP C-07 trial, including survival and subset analyses. J Clin Oncol. 2011; 29: 3768-3774.
12. Twelves C, Scheithauer W, McKendrick J, Seitz JF, Van Hazel G, Wong A, Diaz-Rubio E, Gilberg F, Cassidy J. Capecitabine versus 5-fluorouracil/folinic acid as adjuvant therapy for stage III colon cancer: final results from the X-ACT trial with analysis by age and preliminary evidence of a pharmacodynamic marker of efficacy. Ann Oncol. 2012; 23: 1190-1197.
13. Sanoff HK, Carpenter WR, Sturmer T, Goldberg RM, Martin CF, Fine JP, McCleary NJ, Meyerhardt JA, Niland J, Kahn KL, Schymura MJ, Schrag D. Effect of adjuvant chemotherapy on survival of patients with stage III colon cancer diagnosed after age 75 years. J Clin Oncol. 2012; 30: 2624-2634.
14. Zuckerman IH, Rapp T, Onukwugha E, Davidoff A, Choti MA, Gardner J, Seal B, Mullins CD. Effect of age on survival benefit of adjuvant chemotherapy in elderly patients with Stage III colon cancer. J Am Geriatr Soc. 2009; 57: 1403-1410.
15. Garcia-Albeniz X, Hsu J, Bretthauer M, Hernan MA. Effectiveness of Screening Colonoscopy to Prevent Colorectal Cancer Among Medicare Beneficiaries Aged 70 to 79 Years: A Prospective Observational Study. Ann Intern Med. 2016.
16. Leshno A, Moshkowitz M, David M, Galazan L, Neugut AI, Arber N, Santo E. Prevalence of colorectal neoplasms in young, average risk individuals: A turning tide between East and West. World J Gastroenterol. 2016; 22: 7365-7372.
17. Razenberg LG, Creemers GJ, Beerepoot LV, Vos AH, van de Wouw AJ, Maas HA, Lemmens VE. Age-related systemic treatment and survival of patients with metachronous metastases from colorectal cancer. Acta Oncol. 2016; 55: 1443-1449.
18. Jiang Z, Wang X, Tan X, Fan Z. Effect of Age on Survival Outcome in Operated and Non-Operated Patients with Colon Cancer: A Population-Based Study. PLoS One. 2016; 11: e0147383.
19. Nitsche U, Friess H, Agha A, Angele M, Eckel R, Heitland W, Jauch KW, Krenz D, Nussler NC, Rau HG, Ruppert R, Schubert-Fritschle G, Wilhelm D, et al. Prognosis of mucinous and signet-ring cell colorectal cancer in a population-based cohort. J Cancer Res Clin Oncol. 2016; 142: 2357-2366.
20. Sastre J, Aranda E, Massuti B, Tabernero J, Chaves M, Abad A, Carrato A, Reina JJ, Queralt B, Gomez-Espana A, Gonzalez-Flores E, Rivera F, Losa F, et al. Elderly patients with advanced colorectal cancer derive similar benefit without excessive toxicity after first-line chemotherapy with oxaliplatin-based combinations: comparative outcomes from the 03-TTD-01 phase III study. Crit Rev Oncol Hematol. 2009; 70: 134-144.
21. Arkenau HT, Graeven U, Kubicka S, Grothey A, Englisch-Fritz C, Kretzschmar A, Greil R, Freier W, Seufferlein T, Hinke A, Schmoll HJ, Schmiegel W, Porschen R. Oxaliplatin in combination with 5-fluorouracil/leucovorin or capecitabine in elderly patients with metastatic colorectal cancer. Clin Colorectal Cancer. 2008; 7: 60-64.
22. Perez Dominguez L, Caceres Alvarado N, Toscano Novella A, Casal Nunez JE. Results of colon cancer surgery in patients over 75 years old. ANZ J Surg. 2016. Aug 26. doi: 10.1111/ans.13666. [Epub ahead of print]

