INTRODUCTION
High-risk neuroblastoma (HR-NB) has long included MYCN-amplified stage 2 or 3 disease [1]. Because this entity is found in <10% of NB patients [1], the literature on this subset is limited [2-15], with the largest reported group-wide studies including 12-to-32 patients [4-6, 8-14]. Only one report covers this subset exclusively [9]. Other reports use definitions of MYCN amplification that are no longer accepted (MYCN copy number ≥3 [8, 10, 12] or various threshold values [13]). Analysis is further hampered because several reports, [4-8, 10-12, 16, 17] in presenting outcome data, group these patients with MYCN-amplified stage 1, MYCN-amplified stage 4S, and/or MYCN-non-amplified stage 3 (which we reported has an excellent outcome when treated by surgery alone at diagnosis [18]). Further, therapy for low-stage MYCN-amplified NB has varied widely from surgery alone [13] to myeloablative therapy with autologous stem-cell transplantation (ASCT) [9, 11, 13, 16]; details of treatment were not provided in the largest cohort of stage 3 reported to date [15].
With conventional chemotherapy alone, MYCN-amplified stage 2/3 patients typically relapsed in the primary as well as in distant sites [2-4, 7-9, 14]. ASCT plus local radiation therapy (RT) improved outcome in a small series (n=12), [9] but ASCT did not improve event-free survival (EFS) or overall survival (OS) of either MYCN-amplified stage 2 patients (n=39) collected by the International NB Risk Group (INRG) [13] or stage 3 patients (n=72) deemed high risk (including MYCN-non-amplified disease) in a national study [14]. Other reports concerning ASCT ± RT do not provide outcome data on MYCN-amplified stage 2/3 [11, 16].
Anti-GD2 antibodies such as dinutuximab and 3F8 are active against HR-NB [19]. Although no study compares efficacy, it is reasonable to assume approximately comparable anti-NB activity of different anti-GD2 antibodies. Immunotherapy using these agents recently became standard for HR-NB. However, reports showing a benefit do not include [20], or do not provide details on [21], stage 2/3 patients. We reported on stage 4 HR-NB treated at Memorial Sloan Kettering (MSK) with 3F8 alone [22] or plus granulocyte-macrophage colony-stimulating factor (GM-CSF) [23]. We now present results with MYCN-amplified stage 2/3.
Since 1990, the MSK treatment program for HR-NB has included dose-intensive chemotherapy [24, 25] and tumor resection [26] for induction, followed by consolidation using 3F8 [23] and local RT [27]. We added ASCT and isotretinoin for consolidation in 2000 after those treatments were reported as beneficial, [28] but we discontinued ASCT (though not isotretinoin) in 2003 because published ASCT studies [29–31] showed no survival advantage compared to the earlier MSK non-ASCT programs that used 3F8 without cytokines [22, 23]. We found no significant difference with or without ASCT in EFS or OS of stage 4 HR-NB patients in CR consolidated with 3F8/GM-CSF+isotretinoin plus local RT [23, 32]. We now report excellent outcome of MYCN-amplified stage 2/3, with or without ASCT.
RESULTS
Patient characteristics
The 20 patients (male:female, 11:9) had features typical of MYCN-amplified stage 2 (n=2) or stage 3 (n=18) HR-NB, including young age (10-75 [median 25] months), abdominal site, predominance of unfavorable histology, and elevated serum levels of lactate dehydrogenase at diagnosis (Table 1).
Table 1: Clinical and biological features
Patient #/sex/age at Dx/stage/site |
LDH (U/L) |
Ferritin (ng/mL) |
ALK |
Histology |
Pathology at 2nd-look surgery |
Outcome (time from Dx) |
|---|---|---|---|---|---|---|
A. Patients without consolidative therapy |
||||||
Complete remission with induction but no subsequent consolidation |
||||||
1/M/32m/3/RP |
5300 |
74 |
F1174L |
… |
(-) |
PD (abd, bones) at 18m. Dod at 36m |
PD with induction |
||||||
2/M/19m/3/R Adrenal |
… |
169 |
Wild type |
UH |
(+) |
PD (abd) at 5m. Dod at 6.5m |
B. Non-ASCT patients with 1st remission consolidated by 3F8/GM-CSF, radiotherapy, and isotretinoin |
||||||
Patients in continuing CR |
||||||
3/F/10m/3/L Adrenal |
3958 |
197 |
… |
UH |
(+) |
CR at 162m |
4/M/54m/3/RP |
1637 |
111 |
Wild type |
UH |
(-) |
CR at 100m |
5/M/25m/3/L Adrenal |
1510 |
185 |
Wild type |
UH |
(+) |
CR at 99m |
6/F/37m/2/L Adrenal |
2789 |
52 |
… |
UH |
… |
CR at 92m |
7/M/18m/3/L Adrenal |
2051 |
175 |
Wild type |
FH |
(+) |
CR at 84m |
8/F/59m/3/L Adrenal |
1953 |
461 |
Wild type |
UH |
(+) |
CR at 61m |
9/M/48m/2/L Adrenal |
401 |
… |
Wild type |
UH |
(+) |
CR at 43m |
10/F/25m/3/RP |
2102 |
… |
R1275L |
UH |
(-) |
CR at 34ma |
11/M/14m/3/R Adrenal |
968 |
… |
Wild type |
UH |
(+) |
CR at 28mb |
12/M/75m/3/R Adrenal |
4955 |
… |
Wild type |
UH |
(+) |
CR at 26mb |
13/M/15m/3/R Adrenal |
2619 |
166 |
Wild type |
UH |
(+) |
CR at 24mb |
Patients who relapsed |
||||||
14/M/31m/3/L Adrenal |
2000 |
… |
Wild Type |
UH |
(-) |
PD (pelvis) at 59m. 2nd CR 94m from relapse |
15/F/47m/3/L Adrenal |
624 |
… |
F1174L |
UH |
(-) |
PD (thorax) at 10m. 2nd CR 34m from relapse |
16/M/13m/3/L Adrenal |
872 |
448 |
Wild Type |
FH |
(+) |
PD (abd) at 10m. Dod at 13m |
C. ASCT patients with 1st remission consolidated by 3F8/GM-CSF, radiotherapy, and isotretinoin |
||||||
17/F/51m/3/RP |
600 |
… |
… |
UH |
(+) |
CR at 206m |
18/F/22m/3/L Adrenal |
8189 |
… |
… |
… |
(+) |
CR at 202m |
19/F/18m/3/RP |
2642 |
125 |
… |
FH |
(+) |
CR at 195m |
20/F/19m/3/RP |
613 |
154 |
Wild type |
UH |
(+) |
CR at 145m |
abd, abdomen; ASCT, autologous stem-cell transplantation; Dod, died of disease; Dx, diagnosis; F, female; FH, favorable histology; LDH, lactate dehydrogenase; M, male; PD, progressive disease; RP, retroperitoneal; UH, unfavorable histology.
a Started difluoromethylornithine (DFMO)33 after completing 3F8/GM-CSF+isotretinoin.
b Started vaccine34 after completing 3F8/GM-CSF+isotretinoin.
Overview of outcome of the entire cohort
Of the 20 patients (Table 1), 19 (95%) achieved CR/VGPR with induction while one patient (#2) had PD with induction and died of PD at 6.5 months post-diagnosis. One CR patient (#1) received no consolidation (social reasons), had a widespread relapse documented at 18 months, and, despite aggressive salvage therapy, died of PD at 36 months post-diagnosis. The other 18 CR/VGPR patients received consolidation with 3F8/GM-CSF+isotretinoin+RT, including four with and 14 without prior ASCT. Minimal residual disease (MRD) was negative before 3F8/GM-CSF in 17 patients and positive in only one patient (#20), and was negative in all 18 patients after 2 cycles of this immunotherapy. The number of patients with either favorable histology (n=3) or an ALK mutation (n=3) was too small to assess for possible prognostic significance (Table 1). At 5 years from diagnosis, the entire cohort of 20 patients had EFS 72% ± standard error 11% and OS was 84% ± 9% (Figure 1).
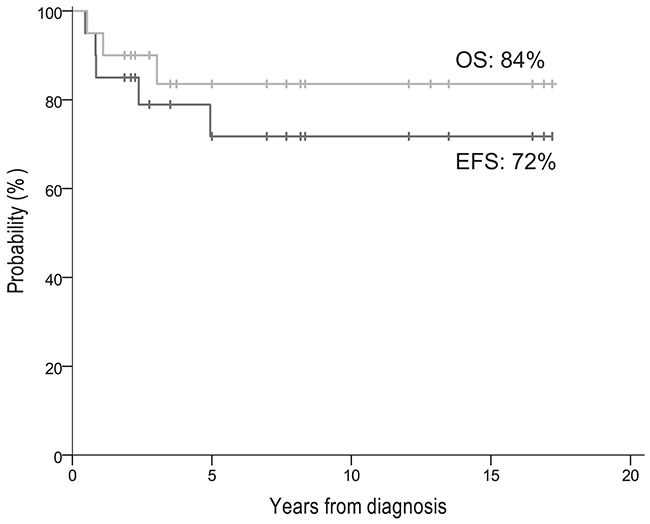
Figure 1: EFS and OS of all 20 patients.
Five patients had pathologic CR at 2nd-look surgery (Table 1) including the sole CR patient (#1) who did not receive consolidation and eventually relapsed (see above), and four non-ASCT patients, two of whom relapsed (patients #14 and #15). With thoraco-abdominal explorations, gross total resections were achieved at 2nd-look surgery in all but one patient (#20; ~80% resected, to avoid injury to portal vein and pancreatic ducts). Surgical complications included nephrectomy (patient #5), cholecystectomy for gall bladder obstruction (patient #13), hypertension from renal vasculature insufficiency (patient #14), and recurrent pleural effusions (patient #18). No unexpected chemotherapy-related complications occurred.
Outcome of patients consolidated with 3F8/GM-CSF+isotretinoin
At five years from the start of 3F8/GM-CSF, the EFS and OS rates of the 18 patients whose CR was consolidated with 3F8/GM-CSF+isotretinoin+RT were, respectively, 81% ± 10% and 94% ± 5% (Figure 2A). All four ASCT patients remain in CR at 145-206 (median 199) months from diagnosis, including the only patient (#20) with MRD detected in bone marrow (BM) pre-3F8/GM-CSF (MRD was negative when re-assessed after cycle 2 of 3F8/GM-CSF).
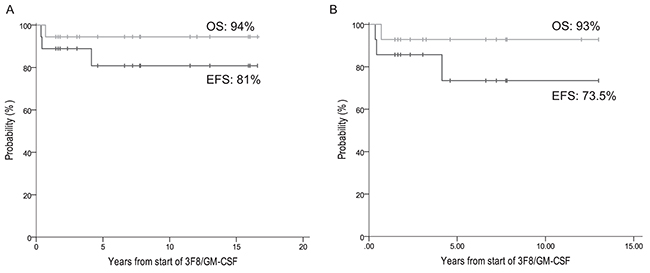
Figure 2: Patients in complete remission treated with 3F8/GM-CSF+isotretinoin. EFS and OS of: (A) all 18 patients, and (B) the 14 non-ASCT patients.
Eleven of the 14 non-ASCT patients remain relapse-free at 24-162 (median 60) months from diagnosis and 19-156 (median 55) months from the start of 3F8/GM-CSF, including four who received additional consolidative adjuvant (experimental and unproven) therapy with DFMO [33] (n=1) or a vaccine [34] (n=3). Two of the three patients who relapsed – one early at 10 months and one late at 59 months from diagnosis, both only in soft tissue outside the RT fields – have achieved prolonged ongoing continuous 2nd CRs (95+ months for patient #14, 36+ months for patient #15). Their successful salvage therapy included previously described 2nd-line chemotherapy regimens, [35-37] resection of relapsed tumor, and local RT (21 cGy [26]). They also received repeat treatment with 3F8/GM-CSF+isotretinoin to consolidate their 2nd CRs, as described [38], plus additional immunotherapy using a vaccine [34]. The other relapse was early, at 10 months, in soft tissue within the RT field, followed three months later by death from PD (patient #16). Overall, this cohort of 14 non-ASCT patients had EFS 73.5% ± 14% and OS 93% ± 7% at 5 years from the start of 3F8/GM-CSF (Figure 2).
As previously noted [23], 3F8/GM-CSF had manageable toxicities – hence, treatment was outpatient for the non-ASCT and the ASCT patients. 3F8 caused grade 1-2 generalized pain and urticaria but no unexpected toxicities [23, 32]. The only grade 3 toxicity of 3F8/GM-CSF was transient hypertension in the 1st cycle of one patient (#13). Common side-effects of isotretinoin were grade 1-2 dry skin and cheilitis.
DISCUSSION
This report is the first to focus on MYCN-amplified localized NB in the era that began when anti-GD2 immunotherapy became standard of care for HR-NB. This period coincided with the routine use of dose-intensive induction and consolidation with local RT and isotretinoin for HR-NB. Regarding MYCN-amplified stage 2/3, our experience shows that 1) dose-intensive chemotherapy and surgery can achieve a high CR rate, and 2) excellent long-term survival is associated with consolidation using anti-GD2 antibody, isotretinoin, and RT. The 5-year EFS/OS rates of 72%/84% (Figure 1) might have been even better had one patient (#1 in Table 1) received consolidation after achieving CR with induction. This child’s relapse, despite a complete pathologic response documented at 2nd-look surgery, underscores the importance of consolidative measures.
In past reports on HR-NB [23, 32], we assessed multiple prognostic markers in univariate and multivariate analyses, but in the current study the low number of events precluded the utility of prognostic markers. Nevertheless, it would appear that for this subset of patients there is the same lack of survival advantage with ASCT as seen in our entire cohort of HR-NB patients [23, 32]. Of note, since 1988, our definition of HR-NB has not included MYCN-non-amplified localized (including stage 3) disease which we have managed upfront with surgery alone [18].
Four of the long-term survivors received DFMO or vaccine (patients #10-13 in Table 1) after completing antibody treatment. These investigational therapies are of unproven benefit for HR-NB; it cannot be certain whether their use improved outcome. If efficacy is ultimately confirmed with more data in ongoing formal clinical trials, then either or both might be incorporated into the standard of care for HR-NB.
The MSK results with MYCN-amplified stage 2/3 are comparable to the 6-year EFS/OS rates of 83%/83% in the series of 12 similar patients whose consolidation included ASCT+RT, though not anti-GD2 antibody or isotretinoin, as presented in the only other report focused specifically on this subset of patients [9]. These EFS/OS rates in turn are better than those in other reports with data on MYCN-amplified stage 2/3 treated with conventional chemotherapy ± RT, including 5-year EFS/OS 32%/36% (n=22) [4] and 6-year OS 25% (n=20) [9] in early French studies, and 1/12 (8%) patients surviving in the Italian experience [8].
Inferior results compared to the current MSK report and the French ASCT+RT experience [9] were also seen in the only two published studies with data on ASCT in high-risk stage 2 or 3. Thus, the CCG-3891 trial which involved randomizations of ASCT and isotretinoin, but not routine use of local RT, showed 5-year EFS/OS rates of 25%/25% for MYCN-amplified stage 3 (n=24); neither ASCT nor isotretinoin had a significant impact on outcome for the entire stage 3 cohort (n=72), which included MYCN-non-amplified disease [14]. In that report, the “overall poor prognosis” led the authors to note that “[f]urther studies are warranted to determine if myeloablative consolidation followed by 13-cis-RA maintenance therapy statistically significantly improves outcome.” Similarly, ASCT did not improve survival in the INRG cohort of MYCN-amplified stage 2 NB (n=39, including nine ASCT), with 5-year EFS/OS of 57%/67% [13].
Reports on more recent large studies of HR-NB do not include patients with MYCN-amplified stage 2/3 [20, 39], have few such patients (n=6 [40], n=4 [41]), or do not separate out the results in this subset [11, 16]. Patients underwent ASCT but local RT was routine in only one of those reports [16] and anti-GD2 therapy was not used [40-42] or was received by a minority of patients [11, 16, 20]. For the large cohorts of HR-NB patients in those studies, [11, 16, 20, 39-41] the 5-year EFS rates of 24%-to-47% and the 5-year OS rates of 26%-to-60% were inferior to the outcome of the MYCN-amplified stage 2/3 patients in our series and in the report using ASCT+RT [9]. One interpretation of these differences is that in the contemporary era of dose-intensive induction plus aggressive consolidation including local RT, MYCN-amplified localized NB has a better prognosis than metastatic HR-NB.
The substantial number of non-ASCT patients in the current report and past reports [23, 32] makes the MSK experience unique because ASCT has been a major component of HR-NB treatment programs since 2000 [42]. Hence, the MSK database offers an opportunity not otherwise available to reassess whether ASCT (which is not standard for other extracranial solid tumors [43, 44]) should be routine for HR-NB. Compelling reasons for revisiting this issue include the recent update of the landmark CCG-3891 trial [27] which showed no OS advantage with ASCT [17], and a recent meta-analysis which found that ASCT for HR-NB did not improve OS [45].
An additional consideration supporting a reeval-uation of ASCT for HR-NB is that the only randomized ASCT studies – dating from 1982-1985 [46], 1991-1996 [14, 17, 28], and 1997-2002 [11] – have limited contemporary relevance because of the low dose-intensity of the induction regimens and the absence or irregular use of local RT and anti-GD2 antibody. In fact, local control of soft tissue NB is excellent with dose-intensive chemotherapy, resection, and RT [26, 27], and eradication of even histologically-evident chemoresistant NB in BM is reliably achieved with anti-GD2 antibodies [47]. Other drawbacks of the randomized studies are that the control arm in one received no therapy [46] and in another received only oral cyclophosphamide [11], and the cytoreduction ranged from single agent (melphalan)48 to multiple agents plus total body irradiation, [14, 17, 28] which is no longer routinely used for NB.
The three randomized studies also preceded modern improvements in salvage therapy and in the early detection of recurrent NB [48] – i.e., interventions that could favorably impact OS. In this regard, two non-ASCT patients (#14 and #15 in Table 1) are in continuous 2nd CR and off all therapy 36+ and 95+ months, respectively, from relapse. These welcome outcomes undermine the long-held view that HR-NB relapse is an ultimately lethal event [49]. This view is why EFS of HR-NB patients has been accepted as the most meaningful measure of treatment efficacy. For curability of HR-NB, however, long-term OS may now supersede EFS endpoints as recent developments offer hope that the equivalence between relapse and lethality may no longer hold true. Thus, as in our two patients, close monitoring [48] can detect localized relapses [23], which might be controlled by surgery and/or focal RT, supplemented by systemic therapies that are non-cross-resistant with prior treatments. Examples include chemotherapy regimens [37, 50] and novel agents [33, 51]. Consolidation of 2nd CR in our two patients included retreatment with 3F8/GM-CSF+isotretinoin, as previously reported [38]. They also received a vaccine used with oral β-glucan; this immunotherapy has shown promise in consolidating 2nd CR [34].
In conclusion, a cautious interpretation of the MSK experience is that ASCT may not be warranted when local RT, anti-GD2 antibodies, and isotretinoin are used for consolidation after dose-intensive induction chemotherapy. This possibility is supported by a critical review of ASCT for HR-NB reaching back 30 years [45], the loss of long-term survival advantage with ASCT for stage 3 and 4 in a major randomized study [17], and the absence of a benefit with ASCT for localized HR-NB [13, 14]. A definitive confirmation that ASCT does not improve outcome would require a prospective randomized trial. Discontinuing ASCT for HR-NB would be consistent with the general consensus among pediatric oncologists that this highly toxic treatment is no longer recommended for all other extracranial pediatric solid tumors [43, 44].
MATERIALS AND METHODS
This report covers all MYCN-amplified stage 2/3 patients who received induction [24, 25] at MSK during the era of immunotherapy with 3F8/GM-CSF (2000-2015) [23, 32]. Stage and MYCN amplification were defined by international criteria [52, 53]. ASCT post-induction was standard through 2003 [16, 54]. Consolidation included local RT (21 Gy) [27] applied between the 1st and 2nd cycles of 3F8/GM-CSF, with 3F8 at 100 mg/m2/cycle, as described [23, 32]. Isotretinoin was taken orally (x6 courses, as described27) between cycles of 3F8/GM-CSF, beginning after the 2nd cycle [23, 32]. Informed written consents for all treatments were obtained according to institutional review board rules.
Extent-of-disease evaluations included 123I-metaiodobenzylguanidine (MIBG) scan and computed tomography or magnetic resonance imaging of chest-abdomen-pelvis every 3 months. BM aspirates and biopsies obtained from bilateral posterior and anterior iliac crests were studied by histology every 3-6 months. Disease status was defined by the International NB Response Criteria [52], modified to incorporate 123I-MIBG findings. CR: no evidence of NB, including normal 123I-MIBG scan and BM(-) by histology. VGPR, primary mass reduced by ≥90%, and no evidence of distant disease in soft tissue, bones, or BM; and PD: new lesion or >25% increase in an existing lesion.
Quantitative reverse transcription-polymerase chain reaction was used, as described [23], to assess MRD in BM before the initiation of immunotherapy and then after the 2nd cycle of 3F8/GM-CSF.
EFS and OS were estimated using Kaplan-Meier analyses, calculated from diagnosis or from the start of 3F8/GM-CSF. Events were defined as relapse, secondary neoplasm, or death. OS was defined as time to death or last follow-up.
Abbreviations
HR-NB, high-risk neuroblastoma; ASCT, autologous stem cell transplant; EFS, event free survival; RT, radiation therapy; OS, overall survival; CR/VGPR, complete remission/very good partial remission; MSK, Memorial Sloan Kettering Cancer Center; GM-CSF, granulocyte-macrophage colony-stimulating factor; BM, bone marrow; MRD, minimal residual disease.
Author contributions
BHK MPL, SM, SLW, EMB, SSR, KK, KY, IYC, NKVC critically reviewed, revised, and approved the final manuscript. BHK, NKVC, SM, EMB, SSR, KK, KY, IYC and NKVC analyzed and interpreted data. BHK, MPL, SM, SLW, EMB, SSR, KK, KY, IYC, NKVC collected and assembled data. BK, NKVC conceived and designed the study.
ACKNOWLEDGMENTS
We wish to thank Joe Olechnowicz for editorial assistance.
CONFLICTS OF INTEREST
Antibody 3F8 was licensed to Ymabs Inc. by Memorial Sloan Kettering Cancer Center (MSK). MSK and NKC have financial interest in Ymabs.
FUNDING
This work was supported in part by the Core Grant (P30 CA008748) and by grants from the National Institutes of Health (CA106450), Bethesda, MD; the Robert Steel Foundation, New York, NY; Katie’s Find A Cure Fund, New York, NY; and the Arnold J. Jacobs Pediatric Cancer Fund, New York, NY.
REFERENCES
1. Cohn SL, Pearson AD, London WB, Monclair T, Ambros PF, Brodeur GM, Faldum A, Hero B, Iehara T, Machin D, Mosseri V, Simon T, Garaventa A, et al. The International Neuroblastoma Risk Group (INRG) classification system: an INRG Task Force report. J Clin Oncol. 2009; 27:289-297.
2. West DC, Shamberger RC, Macklis RM, Kozakewich HP, Wayne AS, Kreissman SG, Korf BR, Lavally B, Grier HE. Stage III neuroblastoma over 1 year of age at diagnosis: improved survival with intensive multimodality therapy including multiple alkylating agents. J Clin Oncol. 1993; 11:84-90.
3. De Bernardi B, Conte M, Mancini A, Donfrancesco A, Alvisi P, Toma P, Casale F, Cordero di Montezemolo L, Cornelli PE, Carli M. Localized resectable neuroblastoma: results of the second study of the Italian Cooperative Group for Neuroblastoma. J Clin Oncol. 1995; 13:884-893.
4. Rubie H, Hartmann O, Michon J, Frappaz D, Coze C, Chastagner P, Baranzelli MC, Plantaz D, Avet-Loiseau H, Benard J, Delattre O, Favrot M, Peyroulet MC. N-Myc gene amplification is a major prognostic factor in localized neuroblastoma: results of the French NBL 90 study. Neuroblastoma Study Group of the Societe Francaise d’Oncologie Pediatrique. J Clin Oncol. 1997; 15:1171-1182.
5. Matthay KK, Perez C, Seeger RC, Brodeur GM, Shimada H, Atkinson JB, Black CT, Gerbing R, Haase GM, Stram DO, Swift P, Lukens JN. Successful treatment of stage III neuroblastoma based on prospective biologic staging: a Children’s Cancer Group study. J Clin Oncol. 1998; 16:1256-1264.
6. Kaneko M, Tsuchida Y, Uchino J, Takeda T, Iwafuchi M, Ohnuma N, Mugishima H, Yokoyama J, Nishihira H, Nakada K, Sasaki S, Sawada T, Kawa K, et al. Treatment results of advanced neuroblastoma with the first Japanese study group protocol. Study Group of Japan for Treatment of Advanced Neuroblastoma. J Pediatr Hematol Oncol. 1999; 21:190-197.
7. Perez CA, Matthay KK, Atkinson JB, Seeger RC, Shimada H, Haase GM, Stram DO, Gerbing RB, Lukens JN. Biologic variables in the outcome of stages I and II neuroblastoma treated with surgery as primary therapy: a children’s cancer group study. J Clin Oncol. 2000; 18:18-26.
8. Garaventa A, Boni L, Lo Piccolo MS, Tonini GP, Gambini C, Mancini A, Tonegatti L, Carli M, di Montezemolo LC, Di Cataldo A, Casale F, Mazzocco K, Cecchetto G, et al. Localized unresectable neuroblastoma: results of treatment based on clinical prognostic factors. Ann Oncol. 2002; 13:956-964.
9. Laprie A, Michon J, Hartmann O, Munzer C, Leclair MD, Coze C, Valteau-Couanet D, Plantaz D, Carrie C, Habrand JL, Bergeron C, Chastagner P, Defachelles AS, et al. High-dose chemotherapy followed by locoregional irradiation improves the outcome of patients with international neuroblastoma staging system Stage II and III neuroblastoma with MYCN amplification. Cancer. 2004; 101:1081-1089.
10. Bagatell R, Rumcheva P, London WB, Cohn SL, Look AT, Brodeur GM, Frantz C, Joshi V, Thorner P, Rao PV, Castleberry R, Bowman LC. Outcomes of children with intermediate-risk neuroblastoma after treatment stratified by MYCN status and tumor cell ploidy. J Clin Oncol. 2005; 23:8819-8827.
11. Berthold F, Boos J, Burdach S, Erttmann R, Henze G, Hermann J, Klingebiel T, Kremens B, Schilling FH, Schrappe M, Simon T, Hero B. Myeloablative megatherapy with autologous stem-cell rescue versus oral maintenance chemotherapy as consolidation treatment in patients with high-risk neuroblastoma: a randomised controlled trial. Lancet Oncol. 2005; 6:649-658.
12. Schneiderman J, London WB, Brodeur GM, Castleberry RP, Look AT, Cohn SL. Clinical significance of MYCN amplification and ploidy in favorable-stage neuroblastoma: a report from the Children’s Oncology Group. J Clin Oncol. 2008; 26:913-918.
13. Bagatell R, Beck-Popovic M, London WB, Zhang Y, Pearson AD, Matthay KK, Monclair T, Ambros PF, Cohn SL; International Neuroblastoma Risk Group. Significance of MYCN amplification in international neuroblastoma staging system stage 1 and 2 neuroblastoma: a report from the International Neuroblastoma Risk Group database. J Clin Oncol. 2009; 27:365-370.
14. Park JR, Villablanca JG, London WB, Gerbing RB, Haas-Kogan D, Adkins ES, Attiyeh EF, Maris JM, Seeger RC, Reynolds CP, Matthay KK; Children’s Oncology Group. Outcome of high-risk stage 3 neuroblastoma with myeloablative therapy and 13-cis-retinoic acid: a report from the Children’s Oncology Group. Pediatr Blood Cancer. 2009; 52:44-50.
15. Meany HJ, London WB, Ambros PF, Matthay KK, Monclair T, Simon T, Garaventa A, Berthold F, Nakagawara A, Cohn SL, Pearson AD, Park JR. Significance of clinical and biologic features in Stage 3 neuroblastoma: a report from the International Neuroblastoma Risk Group project. Pediatr Blood Cancer. 2014; 61:1932-1939.
16. Kreissman SG, Seeger RC, Matthay KK, London WB, Sposto R, Grupp SA, Haas-Kogan DA, Laquaglia MP, Yu AL, Diller L, Buxton A, Park JR, Cohn SL, et al. Purged versus non-purged peripheral blood stem-cell transplantation for high-risk neuroblastoma (COG A3973): a randomised phase 3 trial. Lancet Oncol. 2013; 14:999-1008.
17. Matthay KK, Reynolds CP, Seeger RC, Shimada H, Adkins ES, Haas-Kogan D, Gerbing RB, London WB, Villablanca JG. Long-term results for children with high-risk neuroblastoma treated on a randomized trial of myeloablative therapy followed by 13-cis-retinoic acid: a children’s oncology group study. J Clin Oncol. 2009; 27:1007-1013.
18. Modak S, Kushner BH, LaQuaglia MP, Kramer K, Cheung NK. Management and outcome of stage 3 neuroblastoma. Eur J Cancer. 2009; 45:90-98.
19. Mora J. Dinutuximab for the treatment of pediatric patients with high-risk neuroblastoma. Expert Rev Clin Pharmacol. 2016; 9:647-653.
20. Simon T, Hero B, Faldum A, Handgretinger R, Schrappe M, Klingebiel T, Berthold F. Long term outcome of high-risk neuroblastoma patients after immunotherapy with antibody ch14.18 or oral metronomic chemotherapy. BMC Cancer. 2011; 11:21.
21. Yu AL, Gilman AL, Ozkaynak MF, London WB, Kreissman SG, Chen HX, Smith M, Anderson B, Villablanca JG, Matthay KK, Shimada H, Grupp SA, Seeger R, et al. Anti-GD2 antibody with GM-CSF, interleukin-2, and isotretinoin for neuroblastoma. N Engl J Med. 2010; 363:1324-1334.
22. Cheung NK, Kushner BH, Cheung IY, Kramer K, Canete A, Gerald W, Bonilla MA, Finn R, Yeh SJ, Larson SM. Anti-G(D2) antibody treatment of minimal residual stage 4 neuroblastoma diagnosed at more than 1 year of age. J Clin Oncol. 1998; 16:3053-3060.
23. Cheung NK, Cheung IY, Kushner BH, Ostrovnaya I, Chamberlain E, Kramer K, Modak S. Murine anti-GD2 monoclonal antibody 3F8 combined with granulocyte-macrophage colony-stimulating factor and 13-cis-retinoic acid in high-risk patients with stage 4 neuroblastoma in first remission. J Clin Oncol. 2012; 30:3264-3270.
24. Kushner BH, Kramer K, LaQuaglia MP, Modak S, Yataghene K, Cheung NK. Reduction from seven to five cycles of intensive induction chemotherapy in children with high-risk neuroblastoma. J Clin Oncol. 2004; 22:4888-4892.
25. Park JR, Scott JR, Stewart CF, London WB, Naranjo A, Santana VM, Shaw PJ, Cohn SL, Matthay KK. Pilot induction regimen incorporating pharmacokinetically guided topotecan for treatment of newly diagnosed high-risk neuroblastoma: a Children’s Oncology Group study. J Clin Oncol. 2011; 29:4351-4357.
26. La Quaglia MP, Kushner BH, Su W, Heller G, Kramer K, Abramson S, Rosen N, Wolden S, Cheung NK. The impact of gross total resection on local control and survival in high-risk neuroblastoma. J Pediatr Surg. 2004; 39:412-417; discussion 412-417.
27. Kushner BH, Wolden S, LaQuaglia MP, Kramer K, Verbel D, Heller G, Cheung NK. Hyperfractionated low-dose radiotherapy for high-risk neuroblastoma after intensive chemotherapy and surgery. J Clin Oncol. 2001; 19:2821-2828.
28. Matthay KK, Villablanca JG, Seeger RC, Stram DO, Harris RE, Ramsay NK, Swift P, Shimada H, Black CT, Brodeur GM, Gerbing RB, Reynolds CP. Treatment of high-risk neuroblastoma with intensive chemotherapy, radiotherapy, autologous bone marrow transplantation, and 13-cis-retinoic acid. Children’s Cancer Group. N Engl J Med. 1999; 341:1165-1173.
29. Castel V, Canete A, Navarro S, Garcia-Miguel P, Melero C, Acha T, Navajas A, Badal MD. Outcome of high-risk neuroblastoma using a dose intensity approach: improvement in initial but not in long-term results. Med Pediatr Oncol. 2001; 37:537-542.
30. Kaneko M, Tsuchida Y, Mugishima H, Ohnuma N, Yamamoto K, Kawa K, Iwafuchi M, Sawada T, Suita S. Intensified chemotherapy increases the survival rates in patients with stage 4 neuroblastoma with MYCN amplification. J Pediatr Hematol Oncol. 2002; 24:613-621.
31. De Bernardi B, Nicolas B, Boni L, Indolfi P, Carli M, Cordero Di Montezemolo L, Donfrancesco A, Pession A, Provenzi M, di Cataldo A, Rizzo A, Tonini GP, Dallorso S, et al. Disseminated neuroblastoma in children older than one year at diagnosis: comparable results with three consecutive high-dose protocols adopted by the Italian Co-Operative Group for Neuroblastoma. J Clin Oncol. 2003; 21:1592-1601.
32. Kushner BH, Ostrovnaya I, Cheung IY, Kuk D, Modak S, Kramer K, Roberts SS, Basu EM, Yataghene K, Cheung NK. Lack of survival advantage with autologous stem-cell transplantation in high-risk neuroblastoma consolidated by anti-GD2 immunotherapy and isotretinoin. Oncotarget. 2016; 7:4155-4166. https://doi.org/10.18632/oncotarget.6393.
33. Saulnier Sholler GL, Gerner EW, Bergendahl G, MacArthur RB, VanderWerff A, Ashikaga T, Bond JP, Ferguson W, Roberts W, Wada RK, Eslin D, Kraveka JM, Kaplan J, et al. A phase I trial of DFMO targeting polyamine addiction in patients with relapsed/refractory neuroblastoma. PLoS One. 2015; 10:e0127246.
34. Kushner BH, Cheung IY, Modak S, Kramer K, Ragupathi G, Cheung NK. Phase I trial of a bivalent gangliosides vaccine in combination with beta-glucan for high-risk neuroblastoma in second or later remission. Clin Cancer Res. 2014; 20:1375-1382.
35. Kushner BH, Kramer K, Modak S, Cheung NK. Irinotecan plus temozolomide for relapsed or refractory neuroblastoma. J Clin Oncol. 2006; 24:5271-5276.
36. Kushner BH, Kramer K, Modak S, Qin LX, Cheung NK. Differential impact of high-dose cyclophosphamide, topotecan, and vincristine in clinical subsets of patients with chemoresistant neuroblastoma. Cancer. 2010; 116:3054-3060.
37. Kushner BH, Modak S, Kramer K, Basu EM, Roberts SS, Cheung NK. Ifosfamide, carboplatin, and etoposide for neuroblastoma: a high-dose salvage regimen and review of the literature. Cancer. 2013; 119:665-671.
38. Kushner BH, Ostrovnaya I, Cheung IY, Kuk D, Kramer K, Modak S, Yataghene K, Cheung NK. Prolonged progression-free survival after consolidating second or later remissions of neuroblastoma with Anti-GD2 immunotherapy and isotretinoin: a prospective Phase II study. Oncoimmunology. 2015; 4:e1016704.
39. Pearson AD, Pinkerton CR, Lewis IJ, Imeson J, Ellershaw C, Machin D; European Neuroblastoma Study Group; Children’s Cancer and Leukaemia Group (CCLG formerly United Kingdom Children’s Cancer Study Group). High-dose rapid and standard induction chemotherapy for patients aged over 1 year with stage 4 neuroblastoma: a randomised trial. Lancet Oncol. 2008; 9:247-256.
40. George RE, Li S, Medeiros-Nancarrow C, Neuberg D, Marcus K, Shamberger RC, Pulsipher M, Grupp SA, Diller L. High-risk neuroblastoma treated with tandem autologous peripheral-blood stem cell-supported transplantation: long-term survival update. J Clin Oncol. 2006; 24:2891-2896.
41. Seif AE, Naranjo A, Baker DL, Bunin NJ, Kletzel M, Kretschmar CS, Maris JM, McGrady PW, von Allmen D, Cohn SL, London WB, Park JR, Diller LR, Grupp SA. A pilot study of tandem high-dose chemotherapy with stem cell rescue as consolidation for high-risk neuroblastoma: Children’s Oncology Group study ANBL00P1. Bone Marrow Transplant. 2013; 48:947-952.
42. Grupp SA, Asgharzadeh S, Yanik GA. Neuroblastoma: issues in transplantation. Biol Blood Marrow Transplant. 2012; 18:S92-S100.
43. Kletzel M, Hewlett B. Pediatric transplantation: results in solid tumors. Curr Hematol Rep. 2005; 4:260-269.
44. Ratko TA, Belinson SE, Brown HM, Noorani HZ, Chopra RD, Marbella A, Samson DJ, Bonnell CJ, Ziegler KM, Aronson N. (2012). Hematopoietic Stem-Cell Transplantation in the Pediatric Population. (Rockville (MD)).
45. Yalcin B, Kremer LC, Caron HN, van Dalen EC. High-dose chemotherapy and autologous haematopoietic stem cell rescue for children with high-risk neuroblastoma. Cochrane Database Syst Rev. 2013; 8:CD006301.
46. Pritchard J, Cotterill SJ, Germond SM, Imeson J, de Kraker J, Jones DR. High dose melphalan in the treatment of advanced neuroblastoma: results of a randomised trial (ENSG-1) by the European Neuroblastoma Study Group. Pediatr Blood Cancer. 2005; 44:348-357.
47. Cheung NK, Cheung IY, Kramer K, Modak S, Kuk D, Pandit-Taskar N, Chamberlain E, Ostrovnaya I, Kushner BH. Key role for myeloid cells: phase II results of anti-G(D2) antibody 3F8 plus granulocyte-macrophage colony-stimulating factor for chemoresistant osteomedullary neuroblastoma. Int J Cancer. 2014; 135:2199-2205.
48. Kushner BH, Kramer K, Modak S, Cheung NK. Sensitivity of surveillance studies for detecting asymptomatic and unsuspected relapse of high-risk neuroblastoma. J Clin Oncol. 2009; 27:1041-1046.
49. Cole KA, Maris JM. New strategies in refractory and recurrent neuroblastoma: translational opportunities to impact patient outcome. Clin Cancer Res. 2012; 18:2423-2428.
50. Kushner BH, Kramer K, Modak S, Cheung NK. High-dose carboplatin-irinotecan-temozolomide: treatment option for neuroblastoma resistant to topotecan. Pediatr Blood Cancer. 2011; 56:403-408.
51. Mosse YP, Lim MS, Voss SD, Wilner K, Ruffner K, Laliberte J, Rolland D, Balis FM, Maris JM, Weigel BJ, Ingle AM, Ahern C, Adamson PC, Blaney SM. Safety and activity of crizotinib for paediatric patients with refractory solid tumours or anaplastic large-cell lymphoma: a Children’s Oncology Group phase 1 consortium study. Lancet Oncol. 2013; 14:472-480.
52. Brodeur GM, Pritchard J, Berthold F, Carlsen NL, Castel V, Castelberry RP, De Bernardi B, Evans AE, Favrot M, Hedborg F, Kaneko M, Kemshead J, Lampert F, et al. Revisions of the international criteria for neuroblastoma diagnosis, staging, and response to treatment. J Clin Oncol. 1993; 11:1466-1477.
53. Ambros PF, Ambros IM, Brodeur GM, Haber M, Khan J, Nakagawara A, Schleiermacher G, Speleman F, Spitz R, London WB, Cohn SL, Pearson AD, Maris JM. International consensus for neuroblastoma molecular diagnostics: report from the International Neuroblastoma Risk Group (INRG) Biology Committee. Br J Cancer. 2009; 100:1471-1482.
54. Kushner BH, Kramer K, Modak S, Kernan NA, Reich LM, Danis K, Cheung NK. Topotecan, thiotepa, and carboplatin for neuroblastoma: failure to prevent relapse in the central nervous system. Bone Marrow Transplant. 2006; 37:271-276.

