INTRODUCTION
Childhood trauma, including a spectrum of sexual, physical and emotional forms of abuse, as well as physical or emotional neglect, is highly prevalent and associated with risk for poor health outcomes in childhood and throughout the life course [1]. In recent years, increasing studies have focused on the long-term consequences of childhood trauma and a growing body of evidence suggests a link between childhood trauma and higher risk for cognitive impairments [2], especially executive dysfunction [3], in later life. It has been demonstrated that exposure to childhood trauma is often associated with poorer performance of executive function in patients with psychiatric disorders [4], individuals with ultra-high risk for psychosis [5], and even healthy school-aged children [6]. Moreover, a previous study has also revealed that childhood trauma may be specifically correlated with the subsequent development of cognitive symptoms of major depression [7]. Nevertheless, to date, the neural underpinnings of this consistently replicated relationship remain poorly understood.
Recently, resting-state functional magnetic resonance imaging (rs-fMRI) studies of samples characterized by a history of childhood trauma enable us to evaluate the neurobiological correlates of adverse early life experiences. In particular, functional neuroimaging studies have suggested that childhood trauma are associated with altered functional connectivity (FC) within- and between intrinsic neural networks [8]. A prominent finding is reduction of FC in the default mode network (DMN) [9, 10], while other key findings include disruptions in emotional processing networks [11], executive network [12], salience network [13], and amygdala-DMN, as well as insula-hippocampus connectivity [14]. In accordance with these findings, our prior study also reported that childhood trauma, in the absence of psychiatric diagnosis, was associated with altered DMN, cerebellum-DMN, and insula-DMN connectivity, along with regional homogeneity (ReHo) changes in the inferior parietal lobule (IPL), superior temporal gyrus (STG), insula, cerebellum, and middle temporal gyrus [15]. Meanwhile, Philip et al. (2013) used ReHo approach based on whole brain analysis and found a correlation of brain regional dysfunction in the IPL and STG with early life stress as well [16], suggesting that childhood trauma not only impacts FC but also brain regional activity.
Disturbances in intrinsic brain function have already been suggested to contribute to cognitive impairments observed in patients with psychiatric disorders [17]. It has been demonstrated that altered DMN connectivity is associated with executive processing deficits in patients with bipolar disorder [18] and Alzheimer’s disease (AD) [19]. Simultaneously, executive deficits in patients with major depressive disorder (MDD) have been repeatedly identified to be impacted by a dysfunction of prefrontal cortical regions [20]. However, such associations in subjects with childhood trauma are not clearly elucidated until now. Interestingly, limited task-related fMRI studies have revealed a number of functionally aberrant cortical and subcortical regions in subjects with childhood trauma during paradigms examining executive functions, such as working memory task [21] and response-inhibition task [22]. This could be interpreted as that brain functional abnormalities might also be implicated in the pathophysiology of childhood trauma related executive impairments.
Based on the above considerations, we present here a study trying to detect the long-term impacts of childhood trauma on executive performance and resting-state brain function in a group of young healthy adults and especially to investigate the associations between these two aspects in such a sample. In this study, the fractional amplitude of low frequency fluctuation (fALFF) method [23] was introduced to assess brain regional spontaneous activity. Then, selecting the identified group differences of fALFF as seed regions, the seed-based FC analysis [24] was used to evaluate interregional temporal connectivity. The fALFF approach is an advanced technique to measure local fluctuations in neuronal activity, rather than generalized neuronal activity, which can provide more specific index of low frequency oscillatory phenomena [25]. While coupled fALFF and FC analyses may allow us to completely evaluate brain intra-regional activity and interregional connectivity.
RESULTS
Demographics and clinical measures
As shown in Table 1, the two groups of subjects did not differ with respect to age, gender, educational level, self-rating anxiety scale (SAS) score, and self-rating depression scale (SDS) score. As we would expect, the two experimental groups differed on levels of Childhood Trauma Questionnaire (CTQ) and its sub-scales except sexual abuse. In maltreated subjects, the most common aspect of childhood trauma experience was emotional neglect (17, 70.8%); a proportion of 62.5% (15) of traumatic subjects experienced at least two forms of childhood trauma exposures.
Table 1: Demographic characteristics of all subjects (n = 48)
CTE group n = 24 means (SD) |
Non-CTE group n = 24 means (SD) |
t/χ2 |
p-values |
|
|---|---|---|---|---|
Age (Years) |
21.5 (3.98) |
21.5 (3.69) |
−0.075 |
0.940 |
Gender (Male/Female) |
9/15 |
9/15 |
0.000 |
1.000 |
Educational level (Years) |
14.0 (1.30) |
14.7 (1.92) |
−1.407 |
0.166 |
SDS score |
36.2 (6.06) |
34.5 (5.30) |
1.014 |
0.316 |
SAS score |
34.0 (4.51) |
32.0 (4.78) |
1.430 |
0.160 |
Mean FD (mm) |
0.11 (0.04) |
0.10 (0.03) |
0.835 |
0.408 |
CTQ score |
||||
Emotional abuse |
9.21 (2.36) |
6.21 (1.22) |
5.539 |
0.000 |
Physical abuse |
7.83 (2.93) |
5.71 (1.33) |
3.324 |
0.002 |
Sexual abuse |
5.46 (0.83) |
5.38 (0.58) |
0.403 |
0.689 |
Emotional neglect |
15.2 (3.28) |
7.38 (2.65) |
9.094 |
0.000 |
Physical neglect |
10.2 (2.72) |
5.63 (0.93) |
7.821 |
0.000 |
Total |
47.9 (6.08) |
30.2 (4.63) |
11.38 |
0.000 |
CTE, n (%) |
||||
Emotional abuse |
2 (8.33) |
|||
Physical abuse |
8 (33.3) |
|||
Sexual abuse |
0 (0) |
|||
Emotional neglect |
17 (70.8) |
|||
Physical neglect |
14 (58.3) |
|||
Multiply Exposures |
15 (62.5) |
|||
Single Exposure |
9 (37.5) |
CTE, Childhood Trauma Exposures; CTQ, Childhood Trauma Questionnaire; FD, Framewise Displacement; SAS, Self-rating Anxiety Scale; SD, Standard Deviation; SDS, Self-rating Depression Scale.
Neuropsychological tests
As compared with the control group, individuals with childhood trauma had more preservative errors and less finished categories in Wisconsin Card Sorting Test (WCST). In Stroop Color Word Test (SCWT), the two groups exhibited significant difference in SCWT-A task. However, performance on Trail-making test (TMT) did not differ between two groups. For more details, please refer to Table 2. Correlation analyses revealed that scores of SCWT-A and completed categories in WCST were negatively (r = −0.289 ~ −0.424, p = 0.003 ~ 0.046), while preservative errors in WCST were positively (r = 0.395 ~ 0.420, p = 0.003 ~ 0.005) associated with CTQ scores (CTQ total, emotional neglect, and physical neglect scores) in the whole sample.
Table 2: Results of neuropsychological assessment of all subjects (n = 48)
CTE group n = 24 means (SD) |
Non-CTE group n = 24 means (SD) |
t |
p-values |
|
|---|---|---|---|---|
WSCT |
||||
TT |
47.5 (7.79) |
46.9 (1.93) |
1.007 |
0.319 |
CT |
27.5 (9.24) |
32.2 (8.46) |
−1.841 |
0.072 |
TE |
20.0 (9.97) |
14.8 (9.36) |
1.881 |
0.066 |
PE |
13.0 (6.63) |
8.71 (6.05) |
2.320 |
0.025 |
RE |
7.04 (5.03) |
6.04 (4.31) |
0.739 |
0.463 |
Categories |
3.50 (1.82) |
4.67 (1.71) |
−2.290 |
0.027 |
SCWT A |
103.4 (16.5) |
113.2 (13.7) |
−2.239 |
0.030 |
SCWT B |
74.8 (14.8) |
79.3 (12.9) |
−1.122 |
0.268 |
SCWT C |
44.0 (10.3) |
47.8 (9.63) |
−1.320 |
0.193 |
SCWT interference |
30.7 (9.75) |
31.4 (9.26) |
−0.258 |
0.798 |
TMT A (s) |
29.9 (7.89) |
31.7 (7.70) |
−0.781 |
0.439 |
TMT B (s) |
63.3 (16.0) |
59.9 (13.5) |
0.792 |
0.433 |
CTE, Childhood Trauma Exposures; CT, Correct Trials; PE, Preservative Errors; RE, Random Errors; SD, Standard Deviation; SWCT, Stroop Color Word Test; TE, Total Errors; TMT, Trail-making Test; TT, Total Trials; WSCT, Wisconsin Card Sorting Test.
fALFF analysis
As compared with subjects without childhood trauma, individuals with adverse experiences in childhood shared altered activity in two DMN regions, increased fALFF in the right precuneus, as well as decreased fALFF in the right STG (see Table 3 and Figure 1). There were positive correlations found between fALFF in the right precuneus and CTQ scores, including CTQ total (x = 6, y = −57, z = 57, cluster = 10, T = 4.00, r = 0.595), emotional neglect (x = 6, y = −57, z = 48, cluster = 7, T = 4.42, r = 0.600), and emotional abuse scores (x = 6, y = -57, z = 57, cluster = 9, T = 4.27, r = 0.571) in the whole sample. However, no association was found between cognitive tests and fALFF changes in the present study.
Table 3: Brain regions showing altered fALFF values in CTE individuals as compared with subjects without CTE
Brain region |
Hemisphere |
Cluster size |
T value |
MNI coordinate |
||
|---|---|---|---|---|---|---|
x |
Y |
z |
||||
Increased |
||||||
Precuneus |
R |
14 |
4.30 |
12 |
−57 |
57 |
Decreased |
||||||
STG |
R |
10 |
−5.22 |
57 |
−6 |
−6 |
(p < 0.05, AlphaSim corrected). CTE, Childhood Trauma Exposures; MNI, Montreal Neurological Institute; STG, Superior Temporal Gyrus.
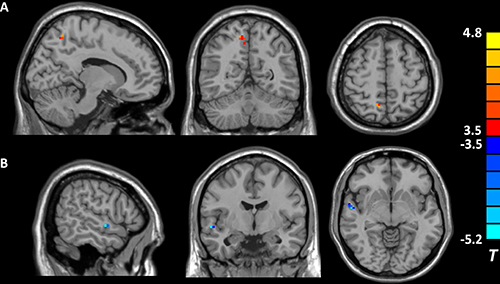
Figure 1. Brain regions showing different fALFF values between individuals with and without childhood trauma (p < 0.05, AlphaSim corrected). Hot and cold colors indicate increased and decreased fALFF in individuals with childhood trauma, respectively, compared with subjects without childhood trauma. (A) the right precuneus; (B) the right superior temporal gyrus.
FC analysis
The fALFF analysis revealed that individuals with childhood trauma showed altered brain activity in two DMN regions. Furthermore, fALFF values in the right precuneus, a core hub of DMN, exhibited significant between-group difference and correlated with CTQ scores in the whole sample. Therefore, the right precuneus (14 voxels) was selected as seed region for subsequent DMN connectivity analysis. Subjects with childhood trauma showed decreased FC in the left ventromedial prefrontal cortex (vmPFC), left orbitofrontal cortex (OFC), and right cerebellum (see Table 4 and Figure 2). Altered right precuneus-based FC was negatively correlated with CTQ scores, including CTQ total, emotional neglect, and physical neglect (r = −0.520 ~ −0.700, p < 0.05, AlphaSim corrected) in the whole sample. In addition, a positive correlation between WCST completed categories and precuneus-based FC in the left vmPFC, as well as negative associations of preservative errors in WCST with precuneus-based FC in the left vmPFC and left OFC were found in the total sample (see Table 5).
Table 4: Brain regions showing reduced functional connectivity with the right precuneus in CTE individuals as compared with subjects without CTE
Brain region |
Hemisphere |
Cluster Size |
T value |
MNI coordinate |
||
|---|---|---|---|---|---|---|
x |
y |
z |
||||
vmPFC |
L |
25 |
−4.29 |
−3 |
48 |
36 |
OFC |
L |
13 |
−3.84 |
−39 |
33 |
−12 |
Cerebellum |
R |
15 |
−4.06 |
33 |
−60 |
−42 |
(p < 0.05, AlphaSim corrected). CTE, Childhood Trauma Exposures; FC, Functional Connectivity; MNI, Montreal Neurological Institute; OFC, Orbitofrontal Cortex; vmPFC, Ventromedial Prefrontal Cortex; PE, Preservative Errors.
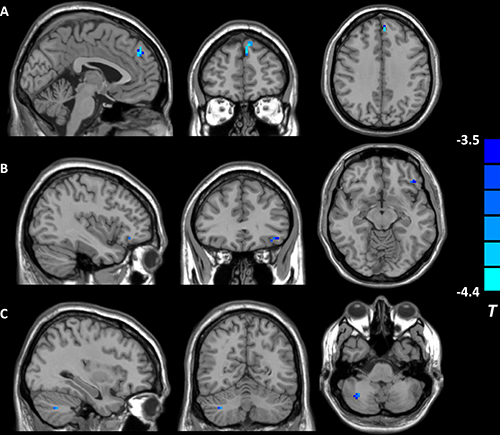
Figure 2: Brain regions showing decreased right precuneus-based functional connectivity in individuals reporting childhood trauma. (p < 0.05, AlphaSim corrected). (A) the left ventromedial prefrontal cortex; (B) the left orbitofrontal cortex; (C) the right cerebellum.
Table 5: Correlation analyses between executive function and the right precuneus-based functional connectivity in the whole sample
Brain region |
Hemisphere |
Cluster Size |
T value |
MNI coordinate |
r |
||
|---|---|---|---|---|---|---|---|
x |
y |
z |
|||||
WSCT PE |
|||||||
OFC |
L |
11 |
−4.02 |
−48 |
24 |
−9 |
−0.518 |
vmPFC |
L |
14 |
−4.16 |
−6 |
45 |
42 |
−0.556 |
WSCT Categories |
|||||||
vmPFC |
L |
14 |
4.63 |
−9 |
45 |
42 |
0.579 |
(p < 0.05, AlphaSim corrected). FC, Functional Connectivity; OFC, Orbitofrontal Cortex; PE, Preservative Errors; vmPFC, Ventromedial Prefrontal Cortex; WSCT, Wisconsin Card Sorting Test.
DISCUSSION
The present study investigated the executive performance and resting-state brain functional changes as measured by fALFF and seed-based FC analyses in young healthy adults with and without early trauma exposures. The current results revealed a significantly damaged executive function and aberrant brain regional activity and interregional FC within the DMN in subjects with childhood trauma, and furthermore, a significant association between executive deficits and diminished DMN connectivity in the whole sample. These outcomes together with previous findings in rodent and nonhuman primate studies [26] might help us understand the neuromechanisms underlying executive dysfunction resulting from chronic early life stress.
Cognitive disturbance has been increasingly recognized as an important aspect of childhood stress, characterized by poor performance on measures of executive function, processing speed, and working memory [27]. The present finding that individuals with childhood trauma show impaired executive function is consistent with the results of previous studies. For instance, Hanson et al. (2013) reported that early neglect experiences were associated with pervasive executive deficits in children raised in institutional settings [28]. And similarly, Nikulina et al. (2013) found that childhood maltreatment, especially childhood neglect, might be an important influence in the development of the executive function deficits of middle adulthood [29]. Moreover, childhood trauma has also been demonstrated to have detrimental effects on performance of neurocognitive tasks which are specially designed to evaluate important aspects of executive domains [30, 31]. In general, all these findings suggest that childhood trauma might adversely impact the development of cognitive systems that support executive functioning.
A second noteworthy finding of the current study was altered fALFF in two DMN regions, the right precuneus and STG, in subjects with childhood trauma. The precuneus, a major posterior node of the DMN, is proved to be involved in theory of mind, self-processing, social cognition, and episodic memory retrieval [32]. While the STG is implicated in receptive and nonverbal auditory and language processing [33], and is also thought to be involved in the processing of emotion [34]. Our data is generally comparable to a recent report in patients with schizophrenia showing an association between childhood trauma and activation of the precuneus when performing a Theory-of-Mind task [35]. The present finding is, at least partially, consistent with prior neuroimaging modalities, which have demonstrated altered spontaneous activity of the precuneus in combat related posttraumatic stress disorder (PTSD) [36] and typhoon survivors [37]. Beyond the precuneus, regional dysfunction in the STG which is assessed by using resting-state ReHo has already been reported in adults with early life stress [16]. Moreover, Hein et al. (2017) performed a quantitative meta-analysis to examine the effects of child maltreatment on brain function and found that the STG was hyperactive in maltreated individuals relative to controls when responding to threatening emotion [38]. Taken together, we speculate that altered fALFF in the precuneus and STG might contribute to specific disturbances in cognitive and emotional processing in individuals with childhood trauma, which may reflect risk for the development of trauma-related disorders [39].
Using a seed-based connectivity approach, the present study also demonstrated diminished resting-state DMN connectivity associated with childhood trauma in subjects even without psychiatric symptoms or medical illness. Our result of decreased precuneus-PFC (including vmPFC and OFC) connectivity among healthy adults reporting histories of childhood trauma is generally in line with past findings. Bluhm et al. (2009) first revealed an association between patients with chronic PTSD related to early life trauma and disrupted DMN connectivity showing decreased FC of posterior cingulate cortex (PCC) with mPFC [10]. Then, in two recent studies, altered connectivity between PCC and mPFC was observed in healthy adults with early life stress [9] and infants with higher interparental conflict since birth [40], respectively, both demonstrating the relationship of childhood trauma with DMN dysfunction. Furthermore, the current finding also provides new evidence to the hypothesis that early life adversity may disrupt the posterior-to-anterior connectivity which is established as part of the normal developmental process [41]. Our study builds on the existing literatures, and suggests that aberrant DMN function at rest may be an independent reflection of early trauma exposure.
Finally, and most importantly, this study confirmed a linear relationship of childhood trauma related executive deficits with disrupted DMN connectivity as revealed by decreased precuneus-vmPFC and precuneus-OFC connectivity, potentially elucidating the neural mechanism underlying executive manifestations of individuals with early trauma. The putative DMN is a task-negative network measured by resting-state fMRI, which includes mainly the medial temporal, frontal, and parietal cortical areas [42]. DMN is suggested to be closely related to higher cognitive functions [43] and this association has been extensively studied in patients with AD. Celebi et al. (2016) found that DMN activity was altered in AD and PCC functional connectivity with other parts of DMN was related to cognitive function scores, such as, memory, attention, and executive function [44]. More interestingly, a recent study in patients with amnestic mild cognitive impairment further demonstrated that accompanied by the ameliorative DMN connectivity, the cognitive performance improved significantly after two-year treatment [45], suggesting a possible relationship between these two aspects. Additionally, it was also shown that among trauma-exposed patients with MDD, higher scores on measures of verbal memory and executive functioning were related to increased connectivity within the DMN [46]. Owing to these findings and ours, it is believed that altered DMN connectivity is involved in disturbances of executive processing.
This study had several limitations. First, the sample size in each group was relatively small which restricted us to compare the different effects of five types of childhood trauma on brain activity. Second, the present FC analysis was limited to precuneus-based DMN connectivity only while ignoring other brain networks that were related to childhood trauma. Third, it was conceivable that childhood trauma had impact on several cognitive domains, however, only executive function was assessed in this study. Finally, this study was cross-sectional designed, which restricted causal analysis. Nevertheless, our novel findings may provide a new insight into exploring the neural underpinnings of childhood trauma. Previous studies have revealed that childhood trauma is associated with biological alterations, such as dysregulation of the hypothalamo-pituitary-adrenal (HPA) axis [47] and inflammation system [48], and interestingly, such disturbances may also contribute to brain abnormalities [49, 50]. In this context, future longitudinal studies containing the above mentioned aspects may be helpful to elucidate the detailed pathway underlying childhood trauma related manifestations. In addition, animal studies should further explore if childhood trauma is causally related to neural differences and subsequently cognitive deficits.
In summary, the present findings suggest that childhood trauma is associated with executive deficits and aberrant DMN functions even in healthy adults. In addition, this study demonstrates that executive dysfunction is related to disrupted DMN connectivity.
MATERIALS AND METHODS
Participants
The study group comprised 48 subjects (male/female, 18/30), ages 18-33 years, including 24 subjects with childhood trauma experiences (CTE group) and 24 age- and gender-matched subjects without childhood trauma exposures (non-CTE group). For assignment to the CTE group, individuals must have had experienced chronic moderate-severe trauma exposures (abuse or/and neglect) before the age of 16. All participants were recruited from a survey that we had carried out to investigate the occurrence of childhood trauma in local communities and universities. Subjects responded with no direct reference to childhood trauma as a key variable in the study. All subjects were thoroughly interviewed by two professional psychiatrists and were free from any current or lifetime history of psychiatric disorders according to Diagnostic and Statistical Manual of Mental Disorders, IV Edition (DSM- IV) criteria, as screened with the Structured Clinical Interview for DSM-IV interview (SCID). The general exclusions were as follows: (1) left handedness, (2) standard scores > 50 on SDS [51] or > 40 on SAS [52], (3) significant medical illness, (4) presence of major sensorimotor handicaps, (5) history of seizures, head trauma, or unconsciousness, (6) intake of any psychotropic medication or hormone, (7) alcohol or substance abuse, (8) women with pregnancy/lactating or in their menstrual period, (9) contraindications to MRI scan, including metallic implants, retractors or braces, and claustrophobia, and (10) inability to keep still during MRI scanning. The demographic data were collected using a self-designed questionnaire from all the participants. This study was approved by the ethic committee of the Second Xiangya Hospital of Central South University. A complete description of the study was provided to every subject, after that written informed consent was obtained from each participant.
Assessment of childhood trauma
The CTQ is a reliable and valid self-reporting questionnaire with 28 items [53]. It can yield five sub-scales which evaluate five aspects of CTE, emotional abuse, emotional neglect, sexual abuse, physical abuse, and physical neglect, respectively. Existence of CTE is determined by cutoff score of each CTQ sub-scale. Subjects who score higher than the threshold of a sub-scale are treated as existence of corresponding CTE. The cutoffs of each sub-scale for moderate-severe exposure are as follows: 1) emotional abuse ≥ 13, 2) emotional neglect ≥ 15, 3) sexual abuse ≥ 8, 4) physical abuse ≥ 10, and 5) physical neglect ≥ 10. The Chinese version of CTQ was introduced in our study. It was translated into Chinese by Zhao et al. (2005) [54], the CTQ has good internal consistency (Cronbach’s alpha) for the CTQ total score (0.77) and the five subscales range from 0.41 to 0.68 in a Chinese sample of 794 individuals. It has been widely used in many Chinese studies although many researchers would like to revise the CTQ and hope its subscales to be more suitable for the Chinese population [55].
Neuropsychological tests
A well-known neuropsychological test battery on executive function was administered to each subject which included WCST [56], SCWT [57], and TMT [58].
1) WCST: Subjects were asked to sort 48 cards on the basis of three possible categories (color, number, and shape). After six consecutive correct responses, subjects were asked to change the sorting principle to another category. The test ended when subjects completed all six categories correctly or used all 48 cards. We evaluated the primary efficacy outcome by using indices of WCST for the performance of executive function that included total trials (TT), total correct (CT), total errors (TE), preservative errors (PE), random errors (RE), and categories completed.
2) SCWT: In part A, the subjects were asked to read randomized color names printed in black type. Then, in part B, the subjects were required to name the color of dots. Finally, in part C, the subjects were instructed to name the ink color of a color word which was not the same as the ink color. All the subjects would have 45 seconds in each part. The performance for each condition was calculated by the finished amounts. The difference in part B relative to part C is called the ‘interference’ effect. SCWT was conducted to measure selective attention/processing speed (SCWT-A and SCWT-B), behavioral inhibition (SCWT-C), and executive function (SCWT interference).
3) TMT: In TMT-A, the subject was required to quickly draw lines to connect consecutively numbered circles. In TMT-B, the subject was asked to alternately combine numbers with letters in ascending order. The task completion is measured in seconds. Part A measured visuo-spatial attention and performance speed, whereas Part B required mental flexibility, ability to shift attention, and strategy.
MRI acquisition
The imaging data were obtained on a 3.0T Philips Achieva scanner at the Second Xiangya Hospital, Central South University. Subjects were asked to lie on the scanner and keep eyes closed. A standard birdcage head coil was used, and the restraining foam pads were placed on two sides of the head to minimize head motion while cotton plug was used with the purpose of diminishing the noise. A total of 180 volumes of echo planar images were obtained axially, the parameters were as follows: repetition time = 3,000 ms; echo time = 30 ms; slices = 36; thickness = 4 mm, no slice gap; field of view = 240 × 240 mm2; resolution = 64 × 64; flip angle = 90°; scan time = 9’09’’.
Data preprocessing
The MRIConvert program (http://lcni.uoregon.edu/~jolinda/MRIConvert/) was used to convert neuroimaging data to Neuroimaging Informatics Technology Initiative data format. All the rs-fMRI data were processed using Data Processing Assistant for Resting-State fMRI Advanced Edition V2.2 (DPARSFA, http://www.restfmri.net/forum/DPARSF). The functional images were conducted for slice acquisition correction and head motion correction. The fMRI data which had less than 1.0 mm of head motion and 1.0° of angular rotation were included. Moreover, the mean framewise displacement (FD) was computed by averaging FDi from every time point for each subject [59]. There was no difference for the mean FD between two groups (t = 0.835, p = 0.408) (Table 1). Then the fMRI images were normalized to the standard Montreal Neurological Institute (MNI) template provided by Statistical Parametric Mapping (SPM) 8 and resample to the 3-mm isotropic voxels. The normalized images were smoothed using a 4-mm full width at half maximum (FWHM) Gaussian kernel. A temporal filter (0.01 Hz < f < 0.08 Hz) were used to reduce the low frequency drift and physiological high frequency respiratory and cardiac noise, and finally, the linear trend was removed.
fALFF and FC calculation
The measurement of fALFF was performed as described previously by Zou et al. (2008) [23]. The time series for each voxel was firstly transformed to the frequency domain using a Fast Fourier Transform (FFT), and then computing the sum of frequencies in the low frequency band (0.01–0.08 Hz). The ALFF measure at each voxel was the averaged square root of the power in the 0.01–0.08 Hz window, normalized by the mean within-brain ALFF value for that subject. This averaged square root was taken as the amplitude of LFF. The fALFF value was the ratio of the power spectrum of low-frequency (0.01–0.08 Hz) to that of the entire frequency range.
A seed-based interregional FC analysis was used in the present study. Areas where between-group fALFF differences correlated with childhood trauma were selected as seeds. The average time series data from seed regions were extracted and FC analysis was performed using the data resulting from preprocessing. The nuisance covariates, including cerebrospinal fluid signals, global mean signals, white matter signals, as well as six head motion parameters were regressed out; then a voxel-wise correlation analysis was conducted between the seed region and the rest of the brain (r score); and finally, Fisher’s r to z transformation was performed to improve the normality of the correlation coefficient.
Statistical analysis
Demographic and clinical data were analyzed by using Statistical Package for the Social Sciences version 16.0 (SPSS Inc., Chicago, IL, USA). Independent two-sample t tests and Chi-square tests (χ2) were respectively used to tests for the continuous variables and categorical variables between two groups. Values are given as mean ± standard deviation. The level of two-tailed statistical significance was set at p < 0.05 for all tests.
Between-group differences of fALFF and FC were analyzed using two-sample t-tests on a voxel-by-voxel basis with SPM8 software. The t map was set at a corrected significance level of p < 0.05 [combined height threshold p < 0.001 (T > 3.28) and cluster ≥ 6]. Threshold correction was performed by using AlphaSim program (parameters were as follows: individual voxel p = 0.001, 1000 simulations, FWHM = 4 mm, with mask) in the REST software, which applied Monte Carlo simulation to calculate the probability of false-positive detection by taking into consideration both the individual voxel probability thresholding and cluster size [60].
Furthermore, to evaluate any correlations of between-group fALFF or FC differences with CTQ and cognitive scores, whole brain multiple regression analyses integrated in SPM basic models were performed at p < 0.05 (AlphaSim corrected). Then, the mean fALFF and FC values of the survived clusters were extracted by using region of interest (ROI) analyses. Correlations were conducted using Pearson’s product moment.
Author contributions
Author Shaojia Lu and Fen Pan designed the study and wrote the first draft of the manuscript. Authors Shaojia Lu, Weijia Gao, Zhaoguo Wei, Dandan Wang, Shaohua Hu, and Manli Huang recruited the sample, finished the clinical assessments, and statistical analyses. Authors Yi Xu and Lingjiang Li also designed the study and had full access to all of the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis. All authors contributed to and have approved the final manuscript.
CONFLICTS OF INTEREST
All authors declare that they have no conflicts of interest.
FUNDING
We sincerely thank the support of funds from the National Natural Science Foundation of China (81601182 to Shaojia Lu), the National Science and Technology Program (2015BAI13B02 to Lingjiang Li), Key Research Project of Zhejiang Province (2015C03040 to Yi Xu), and the Medical Science and Technology Project of Zhejiang Province (2015KYA082 to Shaojia Lu).
REFERENCES
1. Traub F, Boynton-Jarrett R. Modifiable Resilience Factors to Childhood Adversity for Clinical Pediatric Practice. Pediatrics. 2017; 139. https://doi.org/10.1542/peds.2016-2569.
2. Majer M, Nater UM, Lin JM, Capuron L, Reeves WC. Association of childhood trauma with cognitive function in healthy adults: a pilot study. BMC Neurol. 2010; 10:61. https://doi.org/10.1186/1471-2377-10-61.
3. Zou Z, Meng H, Ma Z, Deng W, Du L, Wang H, Chen P, Hu H. Executive functioning deficits and childhood trauma in juvenile violent offenders in China. Psychiatry Res. 2013; 207:218–24. https://doi.org/10.1016/j.psychres.2012.09.013.
4. Aas M, Navari S, Gibbs A, Mondelli V, Fisher HL, Morgan C, Morgan K, MacCabe J, Reichenberg A, Zanelli J, Fearon P, Jones PB, Murray RM, et al. Is there a link between childhood trauma, cognition, and amygdala and hippocampus volume in first-episode psychosis? Schizophr Res. 2012; 137:73–9. https://doi.org/10.1016/j.schres.2012.01.035.
5. Üçok A, Kaya H, Uğurpala C, Çıkrıkçılı U, Ergül C, Yokuşoğlu Ç, Bülbül Ö, Direk N. History of childhood physical trauma is related to cognitive decline in individuals with ultra-high risk for psychosis. Schizophr Res. 2015; 169:199–203. https://doi.org/10.1016/j.schres.2015.08.038.
6. Bucker J, Kapczinski F, Post R, Cereser KM, Szobot C, Yatham LN, Kapczinski NS, Kauer-Sant’Anna M. Cognitive impairment in school-aged children with early trauma. Compr Psychiatry. 2012; 53:758–64. https://doi.org/10.1016/j.comppsych.2011.12.006.
7. Vares EA, Salum GA, Spanemberg L, Caldieraro MA, Souza LH, Borges RP, Fleck MP. Childhood trauma and dimensions of depression: a specific association with the cognitive domain. Rev Bras Psiquiatr. 2015; 38:127–34. https://doi.org/10.1590/1516-4446-2015-1764.
8. Philip NS, Tyrka AR, Albright SE, Sweet LH, Almeida J, Price LH, Carpenter LL. Early life stress predicts thalamic hyperconnectivity: A transdiagnostic study of global connectivity. J Psychiatr Res. 2016; 79:93–100. https://doi.org/10.1016/j.jpsychires.2016.05.003.
9. Philip NS, Sweet LH, Tyrka AR, Price LH, Bloom RF, Carpenter LL. Decreased default network connectivity is associated with early life stress in medication-free healthy adults. Eur Neuropsychopharmacol. 2013; 23:24–32. https://doi.org/10.1016/j.euroneuro.2012.10.008.
10. Bluhm RL, Williamson PC, Osuch EA, Frewen PA, Stevens TK, Boksman K, Neufeld RW, Theberge J, Lanius RA. Alterations in default network connectivity in posttraumatic stress disorder related to early-life trauma. J Psychiatry Neurosci. 2009; 34:187–94.
11. Cisler JM, James GA, Tripathi S, Mletzko T, Heim C, Hu XP, Mayberg HS, Nemeroff CB, Kilts CD. Differential functional connectivity within an emotion regulation neural network among individuals resilient and susceptible to the depressogenic effects of early life stress. Psychol Med. 2013; 43:507–18. https://doi.org/10.1017/S0033291712001390.
12. Philip NS, Valentine TR, Sweet LH, Tyrka AR, Price LH, Carpenter LL. Early life stress impacts dorsolateral prefrontal cortex functional connectivity in healthy adults: informing future studies of antidepressant treatments. J Psychiatr Res. 2014; 52:63–9. https://doi.org/10.1016/j.jpsychires.2014.01.014.
13. Marusak HA, Etkin A, Thomason ME. Disrupted insula-based neural circuit organization and conflict interference in trauma-exposed youth. Neuroimage Clin. 2015; 8:516–25. https://doi.org/10.1016/j.nicl.2015.04.007.
14. van der Werff SJ, Pannekoek JN, Veer IM, van Tol MJ, Aleman A, Veltman DJ, Zitman FG, Rombouts SA, Elzinga BM, van der Wee NJ. Resilience to childhood maltreatment is associated with increased resting-state functional connectivity of the salience network with the lingual gyrus. Child Abuse Negl. 2013; 37:1021–9. https://doi.org/10.1016/j.chiabu.2013.07.008.
15. Lu S, Gao W, Wei Z, Wang D, Hu S, Huang M, Xu Y, Li L. Intrinsic brain abnormalities in young healthy adults with childhood trauma: A resting-state functional magnetic resonance imaging study of regional homogeneity and functional connectivity. Aust N Z J Psychiatry. 2017; 51:614–23. https://doi.org/10.1177/0004867416671415.
16. Philip NS, Kuras YI, Valentine TR, Sweet LH, Tyrka AR, Price LH, Carpenter LL. Regional homogeneity and resting state functional connectivity: associations with exposure to early life stress. Psychiatry Res. 2013; 214:247–53. https://doi.org/10.1016/j.pscychresns.2013.07.013.
17. Huang M, Lu S, Yu L, Li L, Zhang P, Hu J, Zhou W, Hu S, Wei N, Huang J, Weng J, Xu Y. Altered fractional amplitude of low frequency fluctuation associated with cognitive dysfunction in first-episode drug-naive major depressive disorder patients. BMC Psychiatry. 2017; 17:11. https://doi.org/10.1186/s12888-016-1190-1.
18. Nguyen TT, Kovacevic S, Dev SI, Lu K, Liu TT, Eyler LT. Dynamic functional connectivity in bipolar disorder is associated with executive function and processing speed: A preliminary study. Neuropsychology. 2017; 31:73–83. https://doi.org/10.1037/neu0000317.
19. Weiler M, Fukuda A, Massabki LH, Lopes TM, Franco AR, Damasceno BP, Cendes F, Balthazar ML. Default mode, executive function, and language functional connectivity networks are compromised in mild Alzheimer’s disease. Curr Alzheimer Res. 2014; 11:274–82.
20. Wang L, LaBar KS, Smoski M, Rosenthal MZ, Dolcos F, Lynch TR, Krishnan RR, McCarthy G. Prefrontal mechanisms for executive control over emotional distraction are altered in major depression. Psychiatry Res. 2008; 163:143–55. https://doi.org/10.1016/j.pscychresns.2007.10.004.
21. Philip NS, Sweet LH, Tyrka AR, Carpenter SL, Albright SE, Price LH, Carpenter LL. Exposure to childhood trauma is associated with altered n-back activation and performance in healthy adults: implications for a commonly used working memory task. Brain Imaging Behav. 2016; 10:124–35. https://doi.org/10.1007/s11682-015-9373-9.
22. Carrion VG, Garrett A, Menon V, Weems CF, Reiss AL. Posttraumatic stress symptoms and brain function during a response-inhibition task: an fMRI study in youth. Depress Anxiety. 2008; 25:514–26. https://doi.org/10.1002/da.20346.
23. Zou QH, Zhu CZ, Yang Y, Zuo XN, Long XY, Cao QJ, Wang YF, Zang YF. An improved approach to detection of amplitude of low-frequency fluctuation (ALFF) for resting-state fMRI: fractional ALFF. J Neurosci Methods. 2008; 172:137–41. https://doi.org/10.1016/j.jneumeth.2008.04.012.
24. Uddin LQ, Kelly AM, Biswal BB, Castellanos FX, Milham MP. Functional connectivity of default mode network components: correlation, anticorrelation, and causality. Hum Brain Mapp. 2009; 30:625–37. https://doi.org/10.1002/hbm.20531.
25. Liu CH, Ma X, Wu X, Fan TT, Zhang Y, Zhou FC, Li LJ, Li F, Tie CL, Li SF, Zhang D, Zhou Z, Dong J, et al. Resting-state brain activity in major depressive disorder patients and their siblings. J Affect Disord. 2013; 149:299–306. https://doi.org/10.1016/j.jad.2013.02.002.
26. Lupien SJ, McEwen BS, Gunnar MR, Heim C. Effects of stress throughout the lifespan on the brain, behaviour and cognition. Nat Rev Neurosci. 2009; 10:434–45. https://doi.org/10.1038/nrn2639.
27. Saleh A, Potter GG, McQuoid DR, Boyd B, Turner R, MacFall JR, Taylor WD. Effects of early life stress on depression, cognitive performance and brain morphology. Psychol Med. 2017; 47:171–81. https://doi.org/10.1017/S0033291716002403.
28. Hanson JL, Adluru N, Chung MK, Alexander AL, Davidson RJ, Pollak SD. Early neglect is associated with alterations in white matter integrity and cognitive functioning. Child Dev. 2013; 84:1566–78. https://doi.org/10.1111/cdev.12069.
29. Nikulina V, Widom CS. Child maltreatment and executive functioning in middle adulthood: a prospective examination. Neuropsychology. 2013; 27:417–27. https://doi.org/10.1037/a0032811.
30. Marshall DF, Passarotti AM, Ryan KA, Kamali M, Saunders EF, Pester B, McInnis MG, Langenecker SA. Deficient inhibitory control as an outcome of childhood trauma. Psychiatry Res. 2016; 235:7–12. https://doi.org/10.1016/j.psychres.2015.12.013.
31. Savitz JB, van der Merwe L, Stein DJ, Solms M, Ramesar RS. Neuropsychological task performance in bipolar spectrum illness: genetics, alcohol abuse, medication and childhood trauma. Bipolar Disord. 2008; 10:479–94. https://doi.org/10.1111/j.1399-5618.2008.00591.x.
32. Cavanna AE, Trimble MR. The precuneus: a review of its functional anatomy and behavioural correlates. Brain. 2006; 129:564–83. https://doi.org/10.1093/brain/awl004.
33. De Bellis MD, Keshavan MS, Frustaci K, Shifflett H, Iyengar S, Beers SR, Hall J. Superior temporal gyrus volumes in maltreated children and adolescents with PTSD. Biol Psychiatry. 2002; 51:544–52.
34. Radua J, Phillips ML, Russell T, Lawrence N, Marshall N, Kalidindi S, El-Hage W, McDonald C, Giampietro V, Brammer MJ, David AS, Surguladze SA. Neural response to specific components of fearful faces in healthy and schizophrenic adults. Neuroimage. 2010; 49:939–46. https://doi.org/10.1016/j.neuroimage.2009.08.030.
35. Quide Y, Ong XH, Mohnke S, Schnell K, Walter H, Carr VJ, Green MJ. Childhood trauma-related alterations in brain function during a Theory-of-Mind task in schizophrenia. Schizophr Res. 2017 Feb 16. https://doi.org/10.1016/j.schres.2017.02.012. [Epub ahead of print].
36. Yan X, Brown AD, Lazar M, Cressman VL, Henn-Haase C, Neylan TC, Shalev A, Wolkowitz OM, Hamilton SP, Yehuda R, Sodickson DK, Weiner MW, Marmar CR. Spontaneous brain activity in combat related PTSD. Neurosci Lett. 2013; 547:1–5. https://doi.org/10.1016/j.neulet.2013.04.032.
37. Ke J, Chen F, Qi R, Xu Q, Zhong Y, Chen L, Li J, Zhang L, Lu G. Post-traumatic stress influences local and remote functional connectivity: a resting-state functional magnetic resonance imaging study. Brain Imaging Behav. 2016 Oct 8. https://doi.org/10.1007/s11682-016-9622-6. [Epub ahead of print].
38. Hein TC, Monk CS. Research Review: Neural response to threat in children, adolescents, and adults after child maltreatment - a quantitative meta-analysis. J Child Psychol Psychiatry. 2017; 58:222–30. https://doi.org/10.1111/jcpp.12651.
39. Parsons RG, Ressler KJ. Implications of memory modulation for post-traumatic stress and fear disorders. Nat Neurosci. 2013; 16:146–53. https://doi.org/10.1038/nn.3296.
40. Graham AM, Pfeifer JH, Fisher PA, Carpenter S, Fair DA. Early life stress is associated with default system integrity and emotionality during infancy. J Child Psychol Psychiatry. 2015; 56:1212–22. https://doi.org/10.1111/jcpp.12409.
41. Fransson P, Aden U, Blennow M, Lagercrantz H. The functional architecture of the infant brain as revealed by resting-state fMRI. Cereb Cortex. 2011; 21:145–54. https://doi.org/10.1093/cercor/bhq071.
42. Raichle ME. The brain’s default mode network. Annu Rev Neurosci. 2015; 38:433–47. https://doi.org/10.1146/annurev-neuro-071013-014030.
43. Spreng RN, Mar RA, Kim AS. The common neural basis of autobiographical memory, prospection, navigation, theory of mind, and the default mode: a quantitative meta-analysis. J Cogn Neurosci. 2009; 21:489–510. https://doi.org/10.1162/jocn.2008.21029.
44. Celebi O, Uzdogan A, Oguz KK, Has AC, Dolgun A, Cakmakli GY, Akbiyik F, Elibol B, Saka E. Default mode network connectivity is linked to cognitive functioning and CSF Aβ1-42 levels in Alzheimer’s disease. Arch Gerontol Geriatr. 2016; 62:125–32. https://doi.org/10.1016/j.archger.2015.09.010.
45. Zhang JY, Liu Z, Zhang HM, Yang CS, Li H, Li X, Chen KW, Zhang ZJ. A Two-Year Treatment of Amnestic Mild Cognitive Impairment using a Compound Chinese Medicine: A Placebo Controlled Randomized Trial. Sci Rep. 2016; 6: 28982. https://doi.org/10.1038/srep30511.
46. Parlar M, Densmore M, Hall GB, Frewen PA, Lanius RA, McKinnon MC. Relation between patterns of intrinsic network connectivity, cognitive functioning, and symptom presentation in trauma-exposed patients with major depressive disorder. Brain Behav. 2017; 7:e00664. https://doi.org/10.1002/brb3.664.
47. Lu S, Gao W, Huang M, Li L, Xu Y. In search of the HPA axis activity in unipolar depression patients with childhood trauma: Combined cortisol awakening response and dexamethasone suppression test. J Psychiatr Res. 2016; 78:24–30. https://doi.org/10.1016/j.jpsychires.2016.03.009.
48. Lu S, Peng H, Wang L, Vasish S, Zhang Y, Gao W, Wu W, Liao M, Wang M, Tang H, Li W, Li Z, Zhou J, et al. Elevated specific peripheral cytokines found in major depressive disorder patients with childhood trauma exposure: a cytokine antibody array analysis. Compr Psychiatry. 2013; 54:953–61. https://doi.org/10.1016/j.comppsych.2013.03.026.
49. Lu S, Gao W, Wei Z, Wu W, Liao M, Ding Y, Zhang Z, Li L. Reduced cingulate gyrus volume associated with enhanced cortisol awakening response in young healthy adults reporting childhood trauma. PLoS One. 2013; 8:e69350. https://doi.org/10.1371/journal.pone.0069350.
50. Felger JC, Li Z, Haroon E, Woolwine BJ, Jung MY, Hu X, Miller AH. Inflammation is associated with decreased functional connectivity within corticostriatal reward circuitry in depression. Mol Psychiatry. 2016; 21:1358–65. https://doi.org/10.1038/mp.2015.168.
51. Zung WW, Richards CB, Short MJ. Self-rating depression scale in an outpatient clinic. Further validation of the SDS. Arch Gen Psychiatry. 1965; 13:508–15.
52. Zung WW. A rating instrument for anxiety disorders. Psychosomatics. 1971; 12:371–9.
53. Bernstein DP, Stein JA, Newcomb MD, Walker E, Pogge D, Ahluvalia T, Stokes J, Handelsman L, Medrano M, Desmond D, Zule W. Development and validation of a brief screening version of the Childhood Trauma Questionnaire. Child Abuse Negl. 2003; 27:169–90. https://doi.org/10.1016/S0145-2134(02)00541-0.
54. Zhao X, Zhang Y, Li L, Zhou Y, Li H, Yang S. Reliability and validity of the Chinese version of Childhood Trauma Questionnaire. Chinese Journal of Clinical Rehabilitation. Zhongguo Linchuang Kangfu. 2005; 9:105–07.
55. Lu S, Wei Z, Gao W, Wu W, Liao M, Zhang Y, Li W, Li Z, Li L. White matter integrity alterations in young healthy adults reporting childhood trauma: A diffusion tensor imaging study. Aust N Z J Psychiatry. 2013; 47:1183–90. https://doi.org/10.1177/0004867413508454.
56. Monchi O, Petrides M, Petre V, Worsley K, Dagher A. Wisconsin Card Sorting revisited: distinct neural circuits participating in different stages of the task identified by event-related functional magnetic resonance imaging. J Neurosci. 2001; 21:7733–41.
57. Howieson DB, Lezak MD, Loring DW. Orientation and attention. Neuropsychological assessment. Oxford University Press. 2004.
58. Arnett JA, Labovitz SS. Effect of physical layout in performance of the Trail Making Test. Psychol Assess. 1995; 7:220–21. https://doi.org/10.1037/1040-3590.7.2.220.
59. Liao W, Zhang Z, Mantini D, Xu Q, Wang Z, Chen G, Jiao Q, Zang YF, Lu G. Relationship between large-scale functional and structural covariance networks in idiopathic generalized epilepsy. Brain Connect. 2013; 3:240–54. https://doi.org/10.1089/brain.2012.0132.
60. Poline JB, Worsley KJ, Evans AC, Friston KJ. Combining spatial extent and peak intensity to test for activations in functional imaging. Neuroimage. 1997; 5:83–96. https://doi.org/10.1006/nimg.1996.0248.

