INTRODUCTION
Accurate segregation of chromosomes during mitosis is important to the survival of human cells. Errors in mitosis result in cells with an abnormal number of chromosomes, known as aneuploidy, which can lead to cell-cycle arrest, cell death, or tumorigenesis [1, 2]. Whether aneuploidy is a cause or consequence of cancer remains controversial; aneuploidy has been detected in most solid tumors, including lung cancer [2-4]. Chromosome instability, defined as a high rate of either gain or loss of whole chromosomes and causing aneuploidy, is also one of the hallmarks of human solid tumors [2, 5].
During mitosis, there are various checkpoints to ensure the fidelity of chromosome segregation. Checkpoint with FHA and RING finger domains (CHFR) and spindle assembly are examples of mitotic checkpoints [6, 7]. A number of genes, such as mitotic arrest deficient (MAD) 1–3 and budding uninhibited by benzimidazole (BUB) 1–3, are also related to mitotic checkpoints to guarantee accurate chromosome segregation [8-11]. Defects in mitotic checkpoints can cause chromosome instability, which contributes to tumorigenesis. [12, 13].
Mutations in mitotic checkpoint-related genes such as BUB1 and MAD1L1 have been reported in various human cancers, including lung cancer [14-17]. We hypothesized that mutations in mitotic checkpoint-related genes affect not only tumorigenesis, but also the clinical outcomes of cancers. Therefore, we evaluated the association between potentially functional single nucleotide polymorphisms (SNPs) in mitotic checkpoint-related genes and the clinical outcomes of early-stage of non-small cell lung cancers (NSCLC).
RESULTS
Patient characteristics and clinical predictors
The clinical and pathological characteristics of patients and their association with overall survival (OS) and disease-free survival (DFS) are shown in Table 1. Pathological stage was significantly associated with both OS and DFS in univariate analysis (log-rank P = 9 × 10-13 and 4 × 10-18, respectively). Age, gender, and smoking status were also associated with OS (log-rank P = 0.01, 7 × 10-4 and 8 × 10-4, respectively).
Table 1: Univariate analysis for overall survival and disease-free survival by clinicopathologic features.
Overall survival |
Disease-free survival |
||||||
|---|---|---|---|---|---|---|---|
Variables |
No. of cases |
No. of death |
5Y-OSR |
Log-Rank |
No. of event |
5Y-DFSR |
Log-Rank |
Overall |
766 |
214 (27.9) |
61 |
337 (44.0) |
44 |
||
Age (years) |
|||||||
< 65 |
375 |
94 (25.1) |
66 |
0.01 |
160 (42.7) |
48 |
0.14 |
≥ 65 |
391 |
120 (30.7) |
55 |
177 (45.3) |
40 |
||
Gender |
|||||||
Male |
560 |
177 (31.6) |
58 |
7 × 10-4 |
260 (46.4) |
42 |
0.09 |
Female |
206 |
37 (18.0) |
71 |
77 (37.4) |
53 |
||
Smoking status |
|||||||
Never |
229 |
43 (18.8) |
73 |
8 × 10-4 |
89 (38.9) |
50 |
0.12 |
Ever |
537 |
171 (31.8) |
56 |
248 (46.2) |
42 |
||
Pack-yearsc |
|||||||
< 40 |
250 |
72 (28.8) |
57 |
0.27 |
111 (44.4) |
42 |
0.79 |
≥ 40 |
287 |
99 (34.5) |
55 |
137 (47.7) |
42 |
||
Histological type |
|||||||
SCC |
344 |
108 (31.4) |
59 |
0.30 |
151 (43.9) |
46 |
0.35 |
AC |
405 |
100 (24.7) |
63 |
176 (43.5) |
43 |
||
LCC |
17 |
6 (35.3) |
61 |
10 (58.8) |
39 |
||
Pathologic stage |
|||||||
I |
363 |
57 (15.7) |
76 |
9 × 10-13 |
102 (28.1) |
61 |
4 × 10-18 |
II-IIIA |
403 |
157 (39.0) |
48 |
235 (58.3) |
30 |
||
Adjuvant therapyd |
|||||||
No |
182 |
75 (41.7) |
47 |
0.71 |
104 (57.8) |
35 |
0.51 |
Yes |
221 |
88 (36.8) |
49 |
131 (58.7) |
25 |
||
SCC, squamous cell carcinoma; AC, adenocarcinoma; LCC, large cell carcinoma.
aRow percentage.
bFive year-overall survival rate (5Y-OSR) and 5 year-disease free survival rate (5Y-DFSR), proportion of survival derived from Kaplan-Meier analysis.
cIn ever-smokers.
dIn pathologic stage II + IIIA: 184 cases received paclitaxel- cisplatin chemotherapy, 11 cases received radiotherapy, and 26 cases received chemotherapy and radiotherapy.
Association between SNPs and survival outcomes
The 73 SNPs evaluated and results of multivariate analyses are shown in Supplementary Table 1. Among the 73 SNPs examined, 4 SNPs (rs7897156, rs1059476, rs1895320, and rs1374297) were associated with survival outcomes. BUB3 rs7897156C>T was associated with worse overall survival under a recessive model (CC+CT (88.2%) vs. TT (11.8%), adjusted hazard ratio [aHR] = 1.58, 95% confidence interval [CI] = 1.07–2.33, P = 0.02; Table 2 and Figure 1). Aurora kinase B (AURKB) rs1059476G>A was associated with better overall survival under a recessive model (GG+GA (84.6%) vs. AA (15.4%), aHR = 0.64, 95% CI = 0.41–0.99, P = 0.05; Table 2 and Figure 1). Although significant P values were not reached, the same trends as OS were observed in DFS for BUB3 rs7897156 and AURKB rs1059476 (Table 2). Pituitary tumor-transforming 1 (PTTG1) rs1895320 and RAD21 cohesin complex component (RAD21) rs1374297 were associated with worse DFS (under a recessive model, TT+TC (97.6%) vs. CC (2.4%), aHR = 2.46, 95% CI = 1.43–4.23, P = 0.001 and under a codominant model, CC (34.8%) vs. CG (48.0%) vs. GG (17.2%), aHR = 1.18, 95% CI = 1.01–1.38, P = 0.04, respectively; Table 2 and Figure 1). There was no association between 4 SNPs and EGFR, ALK, and RET (Supplementary Table 2).
Table 2: Overall survival and disease-free survival according to genotypes in patients with non-small lung cancer.
Overall survival |
Disease-free survival |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
Gene/SNP |
Genotypee |
No. of cases (%)a |
No. of deaths (%)b |
5Y-OSR (%)c |
Log-rank P |
HR (95% CI)c |
Pd |
No. of events (%)b |
5Y-DFSR (%)c |
Log-rank P |
HR (95% CI)c |
Pd |
BUB3 |
CC |
325 (43.2) |
88 (27.1) |
61 |
0.08 |
1.00 |
144 (44.3) |
42 |
0.57 |
1.00 |
||
rs7897156 |
CT |
339 (45.0) |
90 (26.6) |
67 |
1.04 (0.77-1.39) |
0.81 |
144 (42.5) |
50 |
0.97 (0.77-1.22) |
0.77 |
||
TT |
89 (11.8) |
31 (34.8) |
38 |
1.61 (1.06-2.44) |
0.03 |
39 (43.8) |
34 |
1.25 (0.87-1.79) |
0.23 |
|||
Dominant |
0.54 |
1.14 (0.86-1.50) |
0.36 |
0.71 |
1.01 (0.81-1.26) |
0.90 |
||||||
Recessive |
0.02 |
1.58 (1.07-2.33) |
0.02 |
0.40 |
1.27 (0.91-1.78) |
0.17 |
||||||
Codominant |
1.20 (0.98-1.47) |
0.08 |
1.07 (0.90-1.26) |
0.47 |
||||||||
AURKB |
GG |
259 (34.4) |
75 (29.0) |
54 |
0.07 |
1.00 |
118 (45.6) |
40 |
0.09 |
1.00 |
||
rs1059476 |
GA |
378 (50.2) |
109 (28.8) |
62 |
0.93 (0.69-1.25) |
0.62 |
169 (44.7) |
45 |
0.89 (0.70-1.12) |
0.32 |
||
AA |
116 (15.4) |
22 (19.0) |
75 |
0.61 (0.38-0.98) |
0.04 |
40 (34.5) |
55 |
0.71 (0.50-1.02) |
0.06 |
|||
Dominant |
0.21 |
0.85 (0.64-1.13) |
0.27 |
0.15 |
0.85 (0.67-1.06) |
0.15 |
||||||
Recessive |
0.03 |
0.64 (0.41-0.99) |
0.05 |
0.05 |
0.76 (0.55-1.07) |
0.11 |
||||||
Codominant |
0.82 (0.67-1.01) |
0.07 |
0.86 (0.73-1.01) |
0.06 |
||||||||
PTTG1 |
TT |
520 (69.8) |
148 (28.5) |
60 |
0.05 |
1.00 |
214 (41.2) |
47 |
0.001 |
1.00 |
||
rs1895320 |
TC |
207 (27.8) |
49 (23.7) |
67 |
0.74 (0.53-1.03) |
0.07 |
93 (44.9) |
44 |
1.01 (0.79-1.29) |
0.94 |
||
CC |
18 (2.4) |
9 (50.0) |
48 |
1.52 (0.77-2.99) |
0.23 |
14 (77.8) |
10 |
2.47 (1.42-4.27) |
0.001 |
|||
Dominant |
0.31 |
0.80 (0.59-1.09) |
0.16 |
0.17 |
1.10 (0.87-1.39) |
0.45 |
||||||
Recessive |
0.05 |
1.65 (0.84-3.23) |
0.14 |
0.0001 |
2.46 (1.43-4.23) |
0.001 |
||||||
Codominant |
0.90 (0.69-1.18) |
0.44 |
1.18 (0.96-1.46) |
0.11 |
||||||||
RAD21 |
CC |
260 (34.8) |
69 (26.5) |
62 |
0.59 |
1.00 |
104 (40.0) |
50 |
0.18 |
1.00 |
||
rs1374297 |
CG |
358 (48.0) |
99 (27.7) |
62 |
1.06 (0.78-1.45) |
0.70 |
157 (43.9) |
44 |
1.12 (0.87-1.43) |
0.39 |
||
GG |
128 (17.2) |
38 (29.7) |
57 |
1.26 (0.85-1.88) |
0.26 |
62 (48.4) |
37 |
1.41 (1.03-1.94) |
0.03 |
|||
Dominant |
0.59 |
1.11 (0.83-1.49) |
0.48 |
0.18 |
1.19 (0.94-1.50) |
0.16 |
||||||
Recessive |
0.32 |
1.22 (0.85-1.73) |
0.28 |
0.10 |
1.32 (1.00-1.75) |
0.05 |
||||||
Codominant |
1.11 (0.91-1.36) |
0.29 |
1.18 (1.01-1.38) |
0.04 |
||||||||
aColumn percentage.
bRow percentage.
cFive year-overall survival rate (5Y-OSR) and 5 year-disease free survival rate (5Y-DFSR), proportion of survival derived from Kaplan-Meier analysis.
dHazard ratios (HRs), 95% confidence intervals (CIs) and corresponding P-values were calculated using multivariate Cox proportional hazard models, adjusted for age, sex, smoking status, tumor histology, pathologic stage and adjuvant therapy.
eGenotype failure: 13 cases for the rs7897156, 13 cases for the rs1059476, 21 cases for the rs1895320, and 20 cases for the rs1374297.
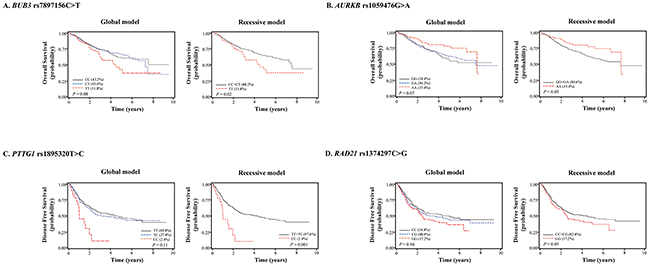
Figure 1: Kaplan-Meier plots of overall survival or disease-free survival according to (A) BUB3 rs7897156C>T, (B) AURKB rs1059476G>A, (C) PTTG1 rs1895320T>C, and (D) RAD21 rs1374297C>G genotypes. P values, multivariate Cox proportional hazard model.
Effect of rs7897156C>T on promoter activity of BUB3
rs7897156C>T is located at the 5′ untranslated region of BUB3 and can change the promoter activity of BUB3. We investigated the effect of rs7897156C>T on the promoter activity of BUB3 using a luciferase assay in the H1299 and A549 NSCLC cell lines. The rs7897156T allele showed significantly higher luciferase activity of the BUB3 promoter compared to the 7897156C allele in both cell lines (P = 0.02 and 0.003, respectively; Figure 2A).
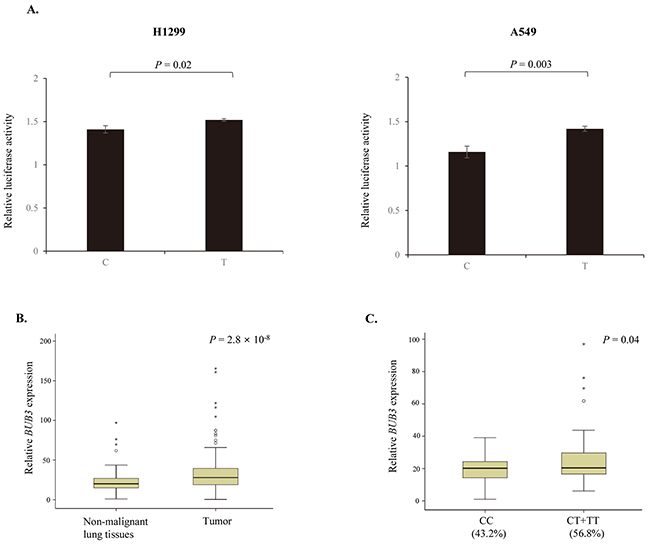
Figure 2: Effect of BUB3 rs7897156C>T. (A) Functional analysis of BUB3 rs7897156C>T by luciferase reporter assay. Transcription activity of rs7897156C>T was measured using a Dual-Luciferase Reporter Assay System in the H1299 and A549 cell lines. Firefly luciferase activity was normalized to Renilla luciferase activity. (B) mRNA expression levels of BUB3 in tumor and non-malignant lung tissues. (C) BUB3 mRNA expression by the rs7897156C>T genotypes in non-malignant lung tissues. The BUB3 mRNA expression levels and association with the rs7897156C>T genotypes were examined in 114 cases with tumor and paired non-malignant lung tissues. The mRNA expression level of BUB3 was normalized to that of the β-actin gene. P values, Student’s t-test.
Effect of rs7897156C>T on BUB3 mRNA expression
Increased BUB3 expression was observed in tumor tissues compared to in paired non-malignant lung tissues (P = 2.8 × 10-8; Figure 2B). To identify the functional effect of BUB3 rs7897156C>T, we evaluated the mRNA expression according to the rs7897156 genotypes. BUB3 mRNA expression was higher for the rs7897156CT or TT genotypes (56.8%) than for the rs7897156CC genotype (43.2%) in non-malignant lung tissues (P = 0.04; Figure 2C).
Functional prediction of AURKB rs1059476G>A
AURKB rs1059476G>A is a non-synonymous SNP. Changing the rs1059476G-to-A results in an amino acid change of threonine to methionine at codon 298. We evaluated whether this amino acid change affects protein function using DelPhi calculation and Pymol (http://pymol.org). As shown in Figure 3, the Thr-to-Met change at codon 298 provides increased hydrophobicity of Aurora kinases B.
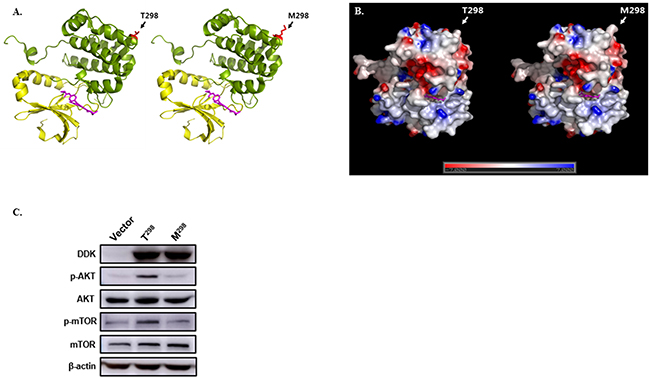
Figure 3: Effect of AURKB rs1059476G>A. (A) Structure model of Aurora kinase (PDB ID: 4AF3) wild-type (T298) and variant type (M298). Kinase domain (yellow) and substrate selective region (green) are shown with kinase inhibitor, VX680 (magenta). Single nucleotide polymorphism residue is highlighted in red. (B) The surface electrostatic potential of wild-type and variant-type was calculated using DelPhi calculation; all figures were constructed using Pymol (http://pymol.org). (C) Western blot of DDK-tagged AURKB wild-type (T298) and variant-type (M298), for phosphorylated AKT (p-AKT), total AKT, phosphorylated mTOR (p-mTOR), and total mTOR, which were transfected into 293T cells. β-actin was used as internal control.
In vitro effect of rs1059476G>A
To assess the functional importance of AURKB variant-type (M298) identified in this study, the effect of the variant-type (M298) on AURKB was investigated in vitro. DDK-tagged wild-type (T298) and variant-type (M298) were transfected into 293T cells. Western blot for the phosphorylation of AKT and mTOR in the variant-type (M298) was significantly lower compared with that in the wild-type (T298) (Figure 3C). Total protein level of AKT and mTOR was not changed. These findings suggest that AURKB variant-type (M298) decreases the phosphorylation of AKT and mTOR.
DISCUSSION
In the present study, we evaluated the effect of 73 SNPs in 25 mitotic-checkpoint related genes on the survival outcomes in patients with surgically resected NSCLC. We found that 4 SNPs (rs7897156, rs1059476, rs1895320, and rs1374297) were associated with OS or DFS.
BUB3 is a mitotic checkpoint protein that is essential for the establishment of correct kinetochore-microtubule attachments [18]. BUB3, in complex with BUB1, delays the onset of anaphase until all mitotic chromosomes are properly attached to the mitotic spindle [8, 19]. Overexpression of BUB3 was reported in several cancers such as breast and gastric cancer [20, 21]. In this study, BUB3 mRNA expression was higher in lung tumor tissues than in non-malignant lung tissues. Up-regulation of mitotic checkpoint gene expression is frequently observed in tumors, but the reason remains unclear. Overexpression of mitotic checkpoint genes may occur because of compensatory mechanisms for other defects in mitotic checkpoint function. Alternatively, overexpression of mitotic checkpoint genes may result in defective mitotic checkpoint function [22].
Associations between the overexpression of mitotic checkpoint genes and poor prognosis have been reported in several cancers [23-27]. In the present study, a higher level of BUB3 expression was observed in lung tumor, and worse OS in patients with the BUB3 rs7897156TT genotype was observed. Our finding is biologically plausible given the putative function of the SNP. In the in vitro luciferase assay, the rs7897156C-to-T change increased the activity of the BUB3 promoter. In addition, the rs7897156CT or TT genotypes showed significantly higher BUB3 mRNA expression than the rs7897156CC genotype. These results suggest that BUB3 rs7897156C>T is a functional SNP. Therefore, the rs7897156C-to-T change may have resulted in overexpression of BUB3 and may affect mitotic checkpoint function to influence the prognosis of patients with NSCLC. To the best of our knowledge, this is the first study to show that BUB3 rs7897156C>T is a functional SNP and can affect prognosis of NSCLC.
The family of Aurora kinases (A, B, and C) act as key regulators of mitotic cell division by regulating the functions of centrosomes, bipolar spindle assembly, and chromosome segregation [28]. Up-regulation of Aurora kinases has been reported in various human cancers, and therapeutic inhibition of these kinases are actively researched as anti-mitotic therapies for cancers [9, 28]. In this study, patients with the AURKB rs1059476AA genotype showed better OS than those with the AURKB rs1059476GA or GG genotypes. The rs1059476G>A is located in exon of AURKB and a non-synonymous SNP. The rs1059476G-to-A change results in the replacement of threonine to methionine at codon 298. The prediction of the three-dimensional model using DelPhi calculation and Pymol program showed increased hydrophobicity of Aurora kinases B by the amino acid change of T298M. Western blot for the phosphorylation of AKT and mTOR was decreased in AURKB the variant-type (M298) compared to AURKB the wild-type (T298). However, the biological mechanism between the AURKB rs1059476G>A and survival outcomes remains unclear. Further studies are needed to clarify the role of AURKB rs1059476G>A in cancer.
In the present study, PTTG1 rs1895320T>C and RAD21 rs1374297C>G were associated with worse DFS. The PTTG1 encodes securin, which inhibits the sister-chromatids from separating until late anaphase [29]. Overexpression of PTTG1 and its relationship with poor prognosis were observed in hepatocellular carcinoma and adrenocortical cancer [27, 30]. Li et al. [31] reported that overexpression of PTTG1 was associated with lymph node and distant metastases in NSCLC and was correlated with poor OS. RAD21 is a component of the cohesion complex and is involved in chromosome segregation during mitosis, DNA repair, and apoptosis [32]. Enhanced RAD21 expression was associated with early relapse and poor prognosis of breast cancer and KRAS mutant colorectal cancer [32, 33]. An association between high expression of RAD21 and cisplatin resistance was reported in NSCLC [34].
In summary, we found that 4 SNPs (rs7897156, rs1059476, rs1895320, and rs1374297) in mitotic-checkpoint related genes were associated with survival outcomes of NSCLC. In addition, functional data suggest that BUB3 rs7897156C>T and AURKB rs1059476G>A are functional SNPs. Additional studies are required to confirm the effect of these SNPs in diverse ethnic groups.
MATERIALS AND METHODS
Study populations
In this study, 766 patients with pathologic stage I, II, and IIIA (micro-invasive N2) NSCLC were enrolled. All patients underwent surgery with curative intent at the Kyungpook National University Hospital, Daegu, Korea (n = 338) or Seoul National University Bundang Hospital, Bundang, Korea (n= 428). Written informed consent was obtain from all patients before surgery. All patients in this study were of Korean ethnicity. This study was approved by the Institutional Review Boards of the Kyungpook National University Hospital and Seoul National University Bundang Hospital.
SNP selection and genotyping
By searching published review articles, 32 mitotic checkpoint-related genes were selected [8-10, 35, 36]. Using HapMap database, 2035 SNPs in 32 mitotic checkpoint-related genes were collected. Among the 2035 SNPs, 968 SNPs were excluded because the minor allele frequency was < 0.05 in the HapMap JPT data. To identify potentially functional SNPs, the FuncPred in the SNP info web server (http://snpinfo.niehs.nih.gov) was used and 116 potentially functional SNPs were selected from the remaining 1067 SNPs. Among these, 43 SNPs were excluded because of linkage disequilibrium (r2 > 0.8), based on HaploView (http://broad.mit.edu/mpg/haploview). A total of 73 SNPs in 25 mitotic checkpoint-related genes were genotyped by the MassARRAY® iPLEX assay (SEQUENOM Inc., San Diego, CA, USA). Approximately 5% of the samples were randomly selected to be genotyped again by a different investigator, by a restriction fragment length polymorphism assay, and the results were 100% concordant.
Promoter-luciferase constructs and luciferase assay
We investigated whether BUB3 rs7897156C>T modulates the activity of the BUB3 gene promoter in a luciferase assay. Promoter fragments of BUB3, including rs7897156, were synthesized by PCR using the following primers: BUB3 forward, 5′-GGGGTACCCCGTAGTTTCCACGCGTCCAGC-3′; BUB3 rev erse, 5′-CCGCTCGAGCGG GAAACGGGATCCCCTGC GAA-3′. The PCR products were cloned into the KpnI/XhoI site of the pGL3-basic plasmid (Promega, Madison, WI, USA). The correct sequences of all clones were verified by DNA sequencing. The NSCLC cell lines H1299 and A549 were transfected with each reported construct and pRL-SV40 vector (Promega), using Effectene transfection reagent (Qiagen, Hilden, Germany). The cells were collected 48 hour after transfection. Luciferase activity was measured using the Dual-Luciferase® Reporter Assay System (Promega) and the results were normalized based on the activity of Renilla luciferase. All experiments were performed in triplicate.
RNA preparation and quantitative reverse transcription-PCR (qRT-PCR)
BUB3 mRNA expression was examined by qRT-PCR. Total RNAs from tumors and paired non-malignant lung tissues (n = 114) were isolated using Trizol (Invitrogen, Carlsbad, USA). Real-time PCR was performed using a LightCycler 480 (Roche Applied Science, Mannheim, Germany) with QuantiFast SYBR Green PCR Master Mix (Qiagen). The real-time PCR primers for BUB3 and β-actin genes involved the following primers: BUB3 forward, 5′-AGTGTTGGTGTGGGACTTAC-3′; BUB3 reverse, 5′-AATACTCAACTGCCACTCGG-3′; β-actin forward, 5′-TTGTTACAGGAAGTCCCTTGCC-3′; β-actin reverse, 5′- ATGCTATCACCTCCCCTGTGT-3′. Each sample was run in duplicate. Relative BUB3 mRNA expression was normalized to that of β-actin expression and then evaluated using the 2- ΔΔCt method [37].
Construction of three-dimensional model for Aurora kinase
To evaluate the potential effect of AURKB rs1059476G>A, which substitutes threonine with methionine at codon 298, the three-dimensional model of the Aurora kinase (PDB ID: 4AF3) SNP variant type was obtained by in silico substitution of Thr298 by Met without altering the backbone geometry, using Pymol program (http://pymol.org). The surface electrostatic potential was calculated using DelPhi calculation and displayed using Pymol.
Construction of expression plasmids for AURKB wild (T298) and variant-type (M298)
To examine the potential effect of wild-type (T298) and variant-type (M298) of AURKB, we constructed wild-type and variant-type plasmids. The AURKB (Myc-DDK-tagged) Human cDNA ORF Clone from Origene Tech (Rockville, MD, USA) was used as a wild-type (T298). The variant-type (M298) construct was created from pCMV6-AURKB-wild-type (T298) using the Quick-change II Site-Directed Mutagenesis kit (Stratagene, Cedar Creek, TX, USA) according to the manufacturer’s protocol. The mutagenesis experiment was amplified using 5’- CCC GCT TCC GTG CCC ATG GGA GCC CAG GAC CTC -3’ (forward) and 5’- GAG GTC CTG GGC TCC CAT GGG CAC GGA AGC GGG -3’ (reverse) primers. Both constructs were verified by sequencing analysis.
Cell culture and transfection
The 293T cells were cultured in Dulbecco’s modified Eagle’s medium supplemented with 10% heat-inactivated fetal bovine serum. Cells were seeded in a six well plate as the cells were approximately 80~90% confluent. At the next day, cells were transfected with pCMV6-AURKB-wild-type (T298), pCMV6-AURKB-variant-type (M298), or pCMV6 empty vector using the Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s protocol. After two days from transfection, cells were harvested.
Western blot
Cell proteins were extracted using M-PER Mammalian Protein Extraction Reagent (Thermo Fisher Scientific, Waltham, MA, USA), supplemented with protease inhibitor and phosphatase inhibitor (Sigma, St. Louis, MO, USA). Western blots were prepared by standard procedures using anti-DDK (Origene Tech, Rockville, MD, USA), anti-actin (Santa Cruz, CA, USA), anti-AKT, anti-phospho-AKT, anti-mTOR, and anti-phospho-mTOR (Cell signaling Technology, Danvers, MA, USA). Immunoreactivity was detected by an Immunobilon Western Chemiluminescent HRP Substrate (Millipore, Billerica, MA, USA).
Statistical analysis
OS was measured from the day of surgery to the day of death or last follow-up. DFS was counted between the day of surgery and recurrence or death from any cause. The Kaplan-Meier test was performed to assess OS and DFS according to genotype. The aHR and 95% CI were calculated using multivariate Cox proportional hazard models, adjusted for age (< 65 years vs. ≥ 65 years), gender (female vs. male), smoking status (never vs. ever), histological type (squamous cell carcinoma vs. adenocarcinoma), pathological stage (stage I vs. stage II or IIIA), and adjuvant therapy (yes vs. no). A P value of less than 0.05 was considered statistically significant. The statistical data were obtained using Statistical Analysis System for Windows, version 9.2 (SAS Institute, Cary, NC, USA).
ACKNOWLEDGMENTS
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (grant number: NRF-2014R1A6A3A01008953), in part by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI14C0402).
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
REFERENCES
1. Storchova Z, Kuffer C. The consequences of tetraploidy and aneuploidy. J Cell Sci. 2008; 121: 3859-3866.
2. Gordon DJ, Resio B, Pellman D. Causes and consequences of aneuploidy in cancer. Nat Rev Genet. 2012; 13: 189-203.
3. Mitelman F, Heim S. Chromosome abnormalities in cancer. Cancer Detect Prev. 1990; 14: 527-537.
4. Sen S. Aneuploidy and cancer. Curr Opin Oncol. 2000; 12: 82-88.
5. Geigl JB, Obenauf AC, Schwarzbraun T, Speicher MR. Defining ‘chromosomal instability’. Trends Genet. 2008; 24: 64-69.
6. Scolnick DM, Halazonetis TD. Chfr defines a mitotic stress checkpoint that delays entry into metaphase. Nature. 2000; 406: 430-435.
7. Lara-Gonzalez P, Westhorpe FG, Taylor SS. The spindle assembly checkpoint. Curr Biol. 2012; 22: R966-980.
8. Kops GJ, Weaver BA, Cleveland DW. On the road to cancer: aneuploidy and the mitotic checkpoint. Nat Rev Cancer. 2005; 5: 773-785.
9. Jackson JR, Patrick DR, Dar MM, Huang PS. Targeted anti-mitotic therapies: can we improve on tubulin agents? Nat Rev Cancer. 2007; 7: 107-117.
10. Baker DJ, Chen J, van Deursen JM. The mitotic checkpoint in cancer and aging: what have mice taught us? Curr Opin Cell Biol. 2005; 17: 583-589.
11. Yuen KW, Montpetit B, Hieter P. The kinetochore and cancer: what’s the connection? Curr Opin Cell Biol. 2005; 17: 576-582.
12. Dobles M, Liberal V, Scott ML, Benezra R, Sorger PK. Chromosome missegregation and apoptosis in mice lacking the mitotic checkpoint protein Mad2. Cell. 2000; 101: 635-645.
13. Michel LS, Liberal V, Chatterjee A, Kirchwegger R, Pasche B, Gerald W, Dobles M, Sorger PK, Murty VV, Benezra R. MAD2 haplo-insufficiency causes premature anaphase and chromosome instability in mammalian cells. Nature. 2001; 409: 355-359.
14. Cahill DP, Lengauer C, Yu J, Riggins GJ, Willson JK, Markowitz SD, Kinzler KW, Vogelstein B. Mutations of mitotic checkpoint genes in human cancers. Nature. 1998; 392: 300-303.
15. Gemma A, Seike M, Seike Y, Uematsu K, Hibino S, Kurimoto F, Yoshimura A, Shibuya M, Harris CC, Kudoh S. Somatic mutation of the hBUB1 mitotic checkpoint gene in primary lung cancer. Genes Chromosomes Cancer. 2000; 29: 213-218.
16. Ohshima K, Haraoka S, Yoshioka S, Hamasaki M, Fujiki T, Suzumiya J, Kawasaki C, Kanda M, Kikuchi M. Mutation analysis of mitotic checkpoint genes (hBUB1 and hBUBR1) and microsatellite instability in adult T-cell leukemia/lymphoma. Cancer Lett. 2000; 158: 141-150.
17. Tsukasaki K, Miller CW, Greenspun E, Eshaghian S, Kawabata H, Fujimoto T, Tomonaga M, Sawyers C, Said JW, Koeffler HP. Mutations in the mitotic check point gene, MAD1L1, in human cancers. Oncogene. 2001; 20: 3301-3305.
18. Logarinho E, Resende T, Torres C, Bousbaa H. The human spindle assembly checkpoint protein Bub3 is required for the establishment of efficient kinetochore-microtubule attachments. Mol Biol Cell. 2008; 19: 1798-1813.
19. Tang Z, Shu H, Oncel D, Chen S, Yu H. Phosphorylation of Cdc20 by Bub1 provides a catalytic mechanism for APC/C inhibition by the spindle checkpoint. Mol Cell. 2004; 16: 387-397.
20. Yuan B, Xu Y, Woo JH, Wang Y, Bae YK, Yoon DS, Wersto RP, Tully E, Wilsbach K, Gabrielson E. Increased expression of mitotic checkpoint genes in breast cancer cells with chromosomal instability. Clin Cancer Res. 2006; 12: 405-410.
21. Grabsch H, Takeno S, Parsons WJ, Pomjanski N, Boecking A, Gabbert HE, Mueller W. Overexpression of the mitotic checkpoint genes BUB1, BUBR1, and BUB3 in gastric cancer--association with tumour cell proliferation. J Pathol. 2003; 200: 16-22.
22. Warren CD, Brady DM, Johnston RC, Hanna JS, Hardwick KG, Spencer FA. Distinct chromosome segregation roles for spindle checkpoint proteins. Mol Biol Cell. 2002; 13: 3029-3041.
23. Rimkus C, Friederichs J, Rosenberg R, Holzmann B, Siewert JR, Janssen KP. Expression of the mitotic checkpoint gene MAD2L2 has prognostic significance in colon cancer. Int J Cancer. 2007; 120: 207-211.
24. Hernando E, Nahle Z, Juan G, Diaz-Rodriguez E, Alaminos M, Hemann M, Michel L, Mittal V, Gerald W, Benezra R, Lowe SW, Cordon-Cardo C. Rb inactivation promotes genomic instability by uncoupling cell cycle progression from mitotic control. Nature. 2004; 430: 797-802.
25. Portella G, Passaro C, Chieffi P. Aurora B: a new prognostic marker and therapeutic target in cancer. Curr Med Chem. 2011; 18: 482-496.
26. Vischioni B, Oudejans JJ, Vos W, Rodriguez JA, Giaccone G. Frequent overexpression of aurora B kinase, a novel drug target, in non-small cell lung carcinoma patients. Mol Cancer Ther. 2006; 5: 2905-2913.
27. Fujii T, Nomoto S, Koshikawa K, Yatabe Y, Teshigawara O, Mori T, Inoue S, Takeda S, Nakao A. Overexpression of pituitary tumor transforming gene 1 in HCC is associated with angiogenesis and poor prognosis. Hepatology. 2006; 43: 1267-1275.
28. Katayama H, Brinkley WR, Sen S. The Aurora kinases: role in cell transformation and tumorigenesis. Cancer Metastasis Rev. 2003; 22: 451-464.
29. Marangos P, Carroll J. Securin regulates entry into M-phase by modulating the stability of cyclin B. Nat Cell Biol. 2008; 10: 445-451.
30. Demeure MJ, Coan KE, Grant CS, Komorowski RA, Stephan E, Sinari S, Mount D, Bussey KJ. PTTG1 overexpression in adrenocortical cancer is associated with poor survival and represents a potential therapeutic target. Surgery. 2013; 154: 1405-1416; discussion 1416.
31. Li H, Yin C, Zhang B, Sun Y, Shi L, Liu N, Liang S, Lu S, Liu Y, Zhang J, Li F, Li W, Liu F et al. PTTG1 promotes migration and invasion of human non-small cell lung cancer cells and is modulated by miR-186. Carcinogenesis. 2013; 34: 2145-2155.
32. Xu H, Yan M, Patra J, Natrajan R, Yan Y, Swagemakers S, Tomaszewski JM, Verschoor S, Millar EK, van der Spek P, Reis-Filho JS, Ramsay RG, O’Toole SA et al. Enhanced RAD21 cohesin expression confers poor prognosis and resistance to chemotherapy in high grade luminal, basal and HER2 breast cancers. Breast Cancer Res. 2011; 13: R9.
33. Deb S, Xu H, Tuynman J, George J, Yan Y, Li J, Ward RL, Mortensen N, Hawkins NJ, McKay MJ, Ramsay RG, Fox SB. RAD21 cohesin overexpression is a prognostic and predictive marker exacerbating poor prognosis in KRAS mutant colorectal carcinomas. Br J Cancer. 2014; 110: 1606-1613.
34. Zhao J, Fu W, Liao H, Dai L, Jiang Z, Pan Y, Huang H, Mo Y, Li S, Yang G, Yin J. The regulatory and predictive functions of miR-17 and miR-92 families on cisplatin resistance of non-small cell lung cancer. BMC Cancer. 2015; 15: 731.
35. Clarke DJ, Gimenez-Abian JF. Checkpoints controlling mitosis. Bioessays. 2000; 22: 351-363.
36. Wang X, Dai W. Shugoshin, a guardian for sister chromatid segregation. Exp Cell Res. 2005; 310: 1-9.
37. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001; 25: 402-408.

