Introduction
Skeleton is one of the most common sites for metastasis. A majority of patients may present osteolytic and osteoblastic metastasis, which will result in marked disturbances of bone remodeling that can be lytic and/or blastic. Osteolytic metastasis, a common complication in breast cancer, lung cancer, prostate cancer, or multiple myeloma [1, 2], brings about significant morbidity of intractable pain, spinal cord compression, pathologic fracture, functional impairment and hypercalcemia [3, 4]. In addition, bone micro-metastasis can serve as an independent predictor of poor outcome in patients with tumor even among lymph node-negative patients with primary tumors of less than 2 cm [5]. Early diagnosis of osteolytic metastasis is definitely important for its correct treatment and better prognosis.
Anatomical imaging (e.g. CT and MRI) has been commonly used for the diagnosis of osteolytic lesions. However, the diagnostic sensitivity of CT is relatively low in cases of a mineral content loss of less than 50% at the lesions sites [6]. Meanwhile, MRI could not readily display bone degradation [7]. 99mTc-diphosphonates (99mTc-MDP)-based planar bone scan is considered as the standard technique for the detection of skeletal metastasis as it shows high sensitivity, especially for bone lesions with osteogenesis. As is known to all, the accumulation of 99mTc-MDP in the bone is highly relied on the osteoblast activity [8]. Absence of 99mTc-MDP or high false negative was reported at early period in osteolytic lesions rich in osteoclast, tumor cells and different degree of osteolysis. Therefore, the detection value of 99mTc-MDP imaging in osteolytic lesions was lower than that in the osteoblastic lesions [9]. On this basis, it is urgent to develop appropriate imaging technique(s) for the detection of osteoclasts and adjacent tumor cells in patients with osteolytic metastases in an early and accurate manner.
Malignant interaction between tumor cells and osteoclasts in bone microenvironment plays a pivotal role in the pathogenesis of metastatic bone disease such as osteolytic and/or osteoblastic bone metastases. Tumor cells contributed to the recruitment and activation of osteoclasts, which triggered regional osteolysis and tumor cell proliferation [10]. Tumor cells and osteoclasts are the main component of osteolytic metastatic lesions. Integrin αvβ3, highly expressed in several tumor cells and activated endothelial cells in newly-generated vessels, has been considered as a target for tumor imaging with radiolabeled arginine-glycine-aspartic acid (RGD) peptides and analogues. Furthermore, osteoclast expressing high level of integrin αvβ3 had attracted great attention for positive imaging of osteolytic bone metastasis [11].
99mTc-HYNIC-3PEG4-E[c(RGDfK)2] (99mTc-3P-RGD2) is a cyclic RGD dimmer peptide with high specificity and affinity to integrin αvβ3. To date, 99mTc-3P4-RGD2 scintigraphy has been commonly used for differential diagnosis of solitary pulmonary nodule, lymph node metastasis and treatment response monitoring [12-15]. This retrospective study was designed to investigate the detective and diagnostic value of 99mTc-3P4-RGD2 imaging for osteolytic bone metastasis.
MATERIALS AND METHODS
Patients
This study was approved by the Institute Review Boards of both Nanjing Medical University and Nanjing First Hospital. Written informed consent was obtained from each patient. All reported investigations were conducted in accordance with the Declaration of Helsinki and with our national regulations. Eighty-eight patients were diagnosed with malignant tumors such as primary lung cancer, lung metastases from thyroid cancer, malignant chromaffin-cell tumor, gastric cancer, breast cancer based on the assessment of case history, CT, MRI, SPECT/CT, PET/CT histopathology and 6-12 month follow-up data. Sixty-nine (48.6%) patients presenting pulmonary nodule were suspected with lung cancer and concurrent osteolytic bone metastases. The exclusion criteria were as follows: (i) received treatment before imaging; (ii) pregnant and breastfeeding patients; (iii) those with a history of bone trauma, fracture, and bone inflammation such as tuberculosis within one year; or (iv) those could not accomplish the required examinations because of severe pain or claustrophobia.
Radiopharmaceutical preparation
99mTc-3P-RGD2 was prepared according to our previous description[16, 17]. Briefly, 1 mL Na99mTcO4 solution (1, 110-1, 850 MBq) was added into lyophilized kit formulation containing 20 mg hydrazinonicotinamide-PEG4-E[PEG4-c(RGDfK)]2 (HYNIC-3P-RGD2), 6.5 mg tricine, 5 mg trisodium triphenylphosphine-3, 39, 399-trisulfonate (TPPTS), 38.5 mg disodium succinate hexahydrate, 40 mg mannitol, and 12.7 mg succinic acid. The reaction system was incubated in water at 100°C for 20 min. The radiochemical purity of the product was > 98% by radio-HPLC. Radiopharmaceuticals were then subject to a 0.20mm Milex-LG filter and diluted to 740MBq/ml for clinical study.
Imaging protocol
We evaluated all of the patients using 99mTc-3P-RGD2 imaging and 99mTc-MDP imaging [including both whole body scan (WBS) and SPECT-CT with consistent imaging field] within one week. The imaging was performed on a SPECT-CT equipment (Symbia T6, Simense, Germany) according to manufacturer’s instructions. All patients had urinated completely prior to imaging. Imaging was performed within one week. Anterior and posterior WBS was performed 1h after intravenous injection of 99mTc-3P-RGD2 (750±37.5MBq, in 1.0 ml saline) or 3h after intravenous injection of 99mTc-MDP (750±75MBq, in 1.0 ml saline). WBS imaging parameters included low-energy high-resolution collimators, bed movement speed of 10.0 cm/min, energy window of 20% width and centered on 140keV. SPECT data set was obtained (ZOOM of 1.3, 256 by 256 matrix size, 30s/Frame for 32 frames and 180° each head of the dual head-camera) immediately after WBS using a low-energy high-resolution collimator at 140 keV with a window width of 20%. Then the data were reconstructed with ordered subset expectation maximization (OSEM). CT scan was performed in the same anatomic locations as SPECT with a tube voltage of 130 kV and current intensity at 120 mA/slice with a slice thickness of 3 mm.
Image analysis
Image analysis was performed by 2 nuclear medicine physicians and 2 radiologists blinded to the case history, examination results, and pathologic diagnosis or follow up data. Visual observation and semiquantitative analysis of 99mTc-3P-RGD2 or 99mTc-MDP imaging were performed based on the accumulation of radiotracer at the lesion sites comparing to the contralateral or surrounding normal bone tissues (T/N ratio). The 4-point grade system was also adopted to describe the uptake degree of radiotracers in osteolytic lesions [18]: grade 0, tracer uptake similar to surrounding normal bone structure; grade 1, uptake less (99mTc-MDP) or slightly higher (99mTc-3P-RGD2) than surrounding normal bone structure; grade 2, uptake significantly less (99mTc-MDP) or higher (99mTc-3P-RGD2) than surrounding normal bone structure; grade 3, abnormal aggregation of 99mTc-3P-RGD2 or almost absence of 99mTc-MDP with or without regional accumulation surrounding the lesion or regional aggregation of 99mTc-MDP in confirmed osteolytic lesions. Osteolytic bone metastasis was ascertained according to any of the following criteria: tracer uptake score greater or equal to grade 2 on either 99mTc-3P-RGD2 or 99mTc-MDP imaging; tracer uptake score equals to grade 1 on both 99mTc-3P-RGD2 and 99mTc-MDP imaging especially with bone pain symptom, or increased uptake on 99mTc-MDP imaging but confirmed to be osteolytic on other imaging modalities such as CT (including CT on SPECT-CT imaging), 18F-FDG PET-CT imaging, and MRI.
Verification of osteolytic bone metastases
The final diagnosis of osteolystic bone metastases was based on histopathology, imaging findings (i.e. CT, SPECT/CT, PET/CT, MRI) and clinical follow-up data using specific standards as follows: (a) histopathologically proven; and (b) Besides primary malignancy diagnosis, CT or magnetic resonance imaging (MRI) results indicated obvious bone destruction without osteogenic imaging performance. (c) Increased range and/or lesions of bone destruction during follow-up [19-21]. During the clinical follow-up, multiple imagings were required for the patients, and all patients were followed up at least 6 months. Lesions exhibiting both osteolytic and osteosclerotic changes were verified by either type, depending on the predominant changes [22].
Statistical analysis
Data were expressed as mean ± standard deviation. The number of osteolytic bone metastases detected using 99mTc-3P-RGD2 and 99mTc-MDP was compared. Sensitivity of 99mTc-3P-RGD2 imaging and 99mTc-MDP imaging modality were compared using McNemar test. The lesion detection consistency of the two modalities were compared using the Kappa test. P < 0.05 was considered to be statistically significant.
Results
Patient characteristics and distribution of osteolytic lesions
Sixty-nine cases with malignant tumor were confirmed with 131 osteolytic metastases according to the comprehensive data based on case history, CT, MRI, SPECT/CT, PET/CT examinations, histopathology and 6-12 monthes follow-up. Patients’ characteristics and the number of osteolytic, lymph node metastases were listed in Table 1.
Table 1: Demographic profile and the final histological diagnosis of 69 patients with malignant tumors and distant metastasis
Category |
number of people |
proportion (%) |
number of osteolytic metastasis |
number of lymph node metastasis |
Gender |
||||
Male |
39 |
56.5 |
||
Female |
30 |
43.5 |
||
Tumor type |
||||
Lung cancer |
||||
adenocarcinoma |
27 |
39.1 |
74 |
4 |
Squamous-cell carcinoma |
32 |
46.4 |
40 |
15 |
Thyroid cancer |
3 |
4.3 |
4 |
2 |
Malignant chromaffin-cell tumor |
2 |
2.9 |
5 |
|
breast cancer |
1 |
1.4 |
1 |
0 |
Gastric cancer |
4 |
5.8 |
5 |
1 |
Visual analysis of osteolytic bone metastasis on 99mTc-MDP and 99mTc-3P-RGD2 imaging
The distribution of the 131 osteolytic bone metastases was summarized in Table 2. Among these metastases, 57 were localized in the neck and chest including cervical vertebra (8), thoracic vertebra (11), sternum (7), rib (31), 20 in the lumbar vertebra, 33 in the sacrococcyx and pelvis, and 21 in the appendicular skeleton including limb (14) and shoulder (7).
Based on visual analysis, osteolytic bone metastases were mainly manifested as significant accumulation (hot area, Figure 1A and 1B), occasionally slight higher uptake (Figure 1C) or absence of 99mTc-3P-RGD2 on WBS and SPECT-CT imaging with or without bone destruction on CT (cold area, Figure 1D). For 99mTc-MDP, osteolytic bone metastases were manifested as “cold area” and occasionally as increased uptake (Figure 2 and 3). Single osteolytic metastasis (with a long diameter of 4.1 cm) was manifested as cold region with slight elevation of 99mTc-MDP uptake in the peripheral part, while negative findings were observed on 99mTc-3P-RGD2 WBS for the single osteolytic metastasis.
Table 2: Comparative analysis of 99mTc-3P-RGD2 and 99mTc-MDP whole body scan imaging (WBS) in the detection of osteolytic metastases in axial, appendicular skeleton
Bone area |
number of lesions |
WBS |
p value |
|
99mTc-MDP |
99mTc-3P-RGD2 |
|||
axial skeleton |
110 |
60 |
92 |
<0.001 |
appendicular skeleton |
21 |
17 |
19 |
>0.05 |
total |
131 |
77 |
111 |
<0.001 |
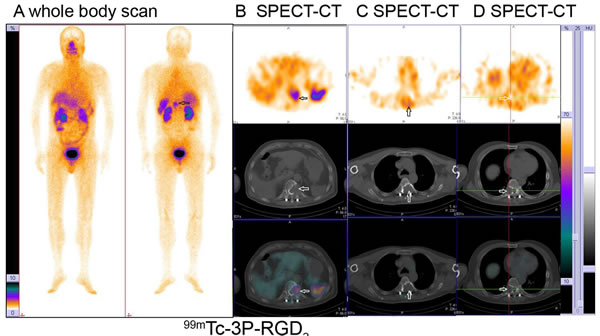
Figure 1: Osteolytic bone metastases from lung cancer displayed as significant accumulation (hot area) (A, B), slight higher uptake (C) or absence (cold area) (D) of 99mTc-3P-RGD2 on whole body planar scan (A) and SPECT-CT (B, C, D) with bone destruction on CT. (see arrows)
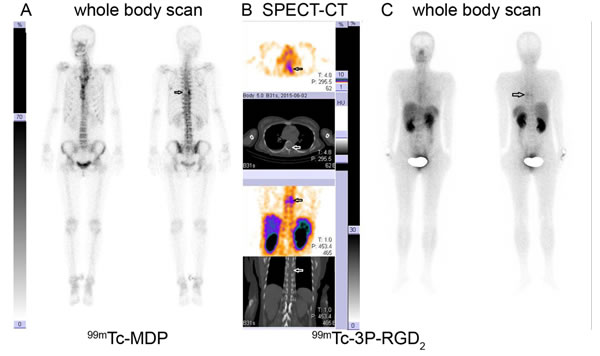
Figure 2: 99mTc-MDP Whole Body Scan (WBS) versus 99mTc-3P-RGD2 WBS and SPECT-CT in a lung cancer patient. 99mTc-MDP WBS demonstrated tracer absence (cold area) in the left side of the 7th thoracic vertebral body A. 99mTc-3P-RGD2 SPECT-CT imaging demonstrates bone destruction and corresponding tracer accumulation in the left side of 7th thoracic vertebral body and adjacent rib with T/N ratio of 6.31 B. 99mTc-3P-RGD2 WBS displayed as “hot region” in the left side of the 7th thoracic vertebral body C. A: 99mTc-MDP WBS; B: 99mTc-3P-RGD2 SPECT-CT; C: 99mTc-3P-RGD2 WBS. (see arrows)
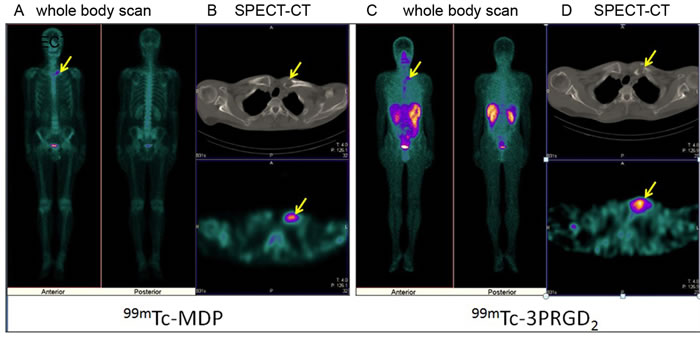
Figure 3: 99mTc-MDP imaging versus 99mTc-3P-RGD2 imaging [Whole Body Scan (WBS) and SPECT-CT) in a patient with lung cancer. Osteolytic bone metastases in the left clavicular head demonstrates as “hot region” on both 99mTc-MDP A., B. and 99mTc-3P-RGD2 imaging C., D. and bone destruction on CT(B,D) . A: 99mTc-MDP WBS; B: 99mTc-MDP SPECT-CT; C: 99mTc-3P-RGD2 WBS; B: 99mTc-3P-RGD2 SPECT-CT. (see arrows).
When scored visually based on whole body scan, 30.5% (40/131) of osteolytic metastatic lesions was graded equally between 99mTc-MDP and 99mTc-3P-RGD2, 57.3% (75/131) was graded higher on 99mTc-3P-RGD2 and 12.2% (16/131) was graded higher on 99mTc-MDP. Comparative analysis of 99mTc-3P-RGD2 and 99mTc-MDP whole body scan imaging (WBS) in the detection of osteolytic metastases was summarized in Table 2. The sensitivity of 99mTc-3P-RGD2 WBS on metastatic lesions was superior to that of 99mTc-MDP WBS, especially in axial skeleton.
The score grade showed obvious change in both imaging modalities after SPECT-CT (Table 3). 99mTc-3P-RGD2 imaging also displayed abnormal uptake in primary tumor and distant metastatic lesions (Figure 4 and 5, Table 1). Regional accumulation of 99mTc-3P-RGD2 contributed to the detection of primary tumor in thyroid cancer (n = 3), breast cancer (n = 1), malignant chromaffin-cell tumor (n = 2), gastric cancer (n = 4), lymph node metastasis (Figure 5). On this basis, the clinical stage of the 69 patient was changed.
Table 3: Visual analysis of 131 osteolytic bone metastasis on 99mTc-3P-RGD2 and 99mTc-MDP WBS imaging and SPECT-CT
grade |
WBS |
WBS+SPECT-CT |
||
99mTc-MDP |
99mTc-3P-RGD2 |
99mTc-MDP |
99mTc-3P-RGD2 |
|
0 |
54 |
20 |
27 |
10 |
1 |
15 |
29 |
27 |
32 |
2 |
28 |
54 |
39 |
61 |
3 |
34 |
28 |
38 |
28 |
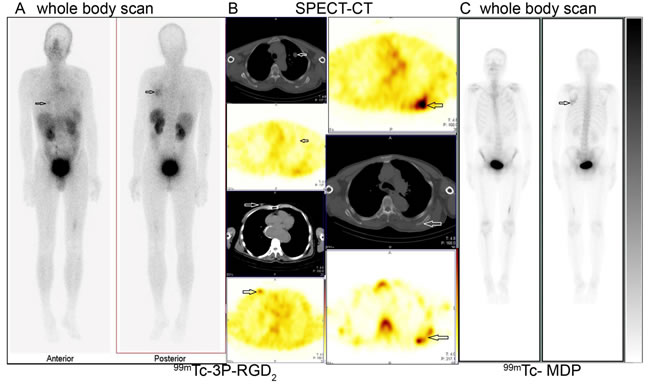
Figure 4: 99mTc-3P-RGD2 imaging promoted the detection of primary tumor. High tracer accumulation of 99mTc-3P-RGD2 in right breast (A. left image and B left bottom two images, T/N=3.7) and left scapula (A: right image and B right upper two images) were demonstrated in one breast cancer patient with pulmonary nodule (B. left upper 2 images, T/N=1.08) in the left lung. The osteolytic bone metastasis in the left scapula displayed internal regional absence and slight elevation of 99mTc-MDP uptake in the surrounding bone tissues (B: right bottom and C). A: 99mTc-3P-RGD2 whole body scan; B: 99mTc-3P-RGD2 SPECT-CT (the right bottom image is 99mTc-MDP SPECT); C. 99mTc-MDP whole body scan. (see arrows).
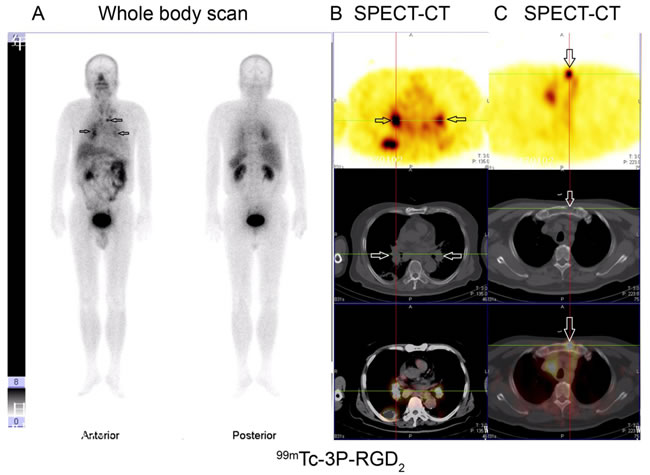
Figure 5: 99mTc-3P-RGD2 imaging of one patient with lung cancer and multi-lymph node metastases (mediastinal lymph node, hilar lymph node and presternal osteolytic). A. 99mTc-3P-RGD2 whole body scan; B. and C. 99mTc-3P-RGD2 SPECT-CT. (see arrows).
Detection value of 99mTc-3P-RGD2 and 99mTc-MDP imaging for osteolytic bone metastases
Semiquantification of tracer uptake in osteolytic bone mestastasis was expressed as T/N. A large variance (0.73-13.5) was noticed in the 99mTc-3P-RGD2 in tracer uptake in osteolytic bone metastases. T/N on 99mTc-3P-RGD2 WBS was significantly lower than that on SPECT-CT (4.93±2.20 vs. 6.80±3.19, t = 15.1, p < 0.01). The positive rate of 99mTc-3P-RGD2 WBS showed significant increase compared to 99mTc-MDP WBS based on T/N analysis (80.9% vs. 46.6%, p < 0.05, Supplemental Table 1). When combining with SPECT-CT, the detective sensitivity of 99mTc-3P-RGD2 imaging increased to 96.2% (126/131). The osteolytic metastases demonstrated that lower or similar 99mTc-3P-RGD2 uptake was more commonly seen in large size lesions with existence of predominant bone destruction, internal necrosis or/and surrounding cyclic hyperostosis/osteosclerosis. T/N of 99mTc-3P-RGD2 in osteolytic metastatic lesions in lung adenocarcinoma patients showed no statitical difference from that in squamous-cell carcinoma (6.84±3.46 vs. 7.33±3.22, t = 0.39, p = 0.71). T/N of 99mTc-3P-RGD2 in osteolytic metastases from primary lung cancer was significantly higher than that from thyroid cancer (7.05±3.01 vs. 4.11±2.67, p = 0.03).
Discussion
99mTc-MDP is widely used to detect metastatic bone lesions via adhering to hydroxyapatite crystal and collagen in bone matrix. It is manifested as a “hot zone” in osteoblastic bone metastases even before changes of anatomical structure. 99mTc-MDP WBS imaging is superior for the detection of osteoblastic metastases, however, its efficiency in the detection of osteolytic lesions is hampered due to overlaping from normal bone tissues. Osteolytic bone metastasis, characterized by the activation of osteoclasts and the resulting bone resorption, is visualized as a “cold area” (negative imaging) due to absence of 99mTc-MDP uptake. However, osteolytic metastatic lesions, consisting of a great number of tumor cells and osteoclasts expressing high level of integrin αvβ3 and αvβ5 [23], are visualized as a “hot zone” (positive imaging) as it can bind to 99mTc-3P-RGD2. This was the main mechanism of 99mTc-3P-RGD2 imaging for the detection of osteolytic bone metastases with high T/N ratio. Our study showed that 99mTc-3P-RGD2 WBS was more effective than 99mTc-MDP WBS in the detection of osteolytic bone metastases, which may be related to the following aspects: Firstly, “hot area” was more easy to be visualized than “code area” on WBS. Secondly, distribution of 99mTc-3P-RGD2 in normal bone structures was extremely low, which made the osteolytic bone metastases much easier to be detected with low background. Thirdly, overlaping of normal bone structures contributed to low grade for the lesions in vetebrea, sternum, sacrococcyx and pelvis, which finally induced misdiagnosis by 99mTc-MDP WBS [24].
Clinically, SPECT-CT was performed in presence of abnormality or highly suspicious on WBS images. For the highly suspected patients, it is difficult for a patient to accept the whole body SPECT-CT each time during his first imaging or during regular follow-up. The detection of suspicious osteolytic bone metastases on WBS is of great importance as it serves as a gate for selective SPECT-CT imaging that will facilitate lesion detection, boundary delineation for semiquantitive analysis and accurate diagnosis [25, 26]. The detection value of osteolytic bone metastases increased by about 15% and several patients with score grade increased on both imaging modalities after combining with SPECT-CT. SPECT-CT is vital for 99mTc-3P-RGD2 imaging due to absence of normal skeleton appearance for lesion localization. Furthermore, moderate-to-intense 99mTc-3P-RGD2 accumulation in visceral organs might make the mild-uptake lesions undetectable in lower thoracic, upper lumber vertebra and sacrococcyx [27]. The diagnostic sensitivity of 99mTc-3P-RGD2 imaging for osteolytic bone metastases was significantly higher than that of 99mTc-MDP imaging in our study. Also, its sensitivity was slightly lower than 18F-Alfatide II PET-CT imaging [18]. This may be related to the higher spatial resolution of PET and the large database. Besides detection of osteolytic bone metastases, 99mTc-3P-RGD2 imaging was also useful for the differential diagnosis of primary tumor and even its staging [14, 28]. In our study, primary lung cancer, thyroid cancer, malignant chromaffin-cell tumor and gastric cancer were highly positive on 99mTc-3P-RGD2 imaging [29]. Thus, 99mTc-3P-RGD2 imaging was superior to 99mTc-MDP in the detection of osteolytic bone metastases, unknown primary tumor, distant metastases and clinical staging. T/N of 99mTc-3P-RGD2 in osteolytic metastasis may be partially influenced by integrin αvβ3 expression level on primary tumor cells [30]. In future, further studies involving more cases of different malignant tumors are needed.
In metastatic bone tissues, 99mTc-MDP imaging reflects bone metabolism and osteogenesis, while 99mTc-3P-RGD2 imaging reflects the existence of integrin αvβ3–overexpressing tumor cells, osteoclast, angiogenesis [23, 31]. Osteolytic bone metastases usually demonstrated as increased accumulation of 99mTc-3P-RGD2 imaging and low uptake of 99mTc-MDP. However, “hot area” or “cold area” can be visualized on both imaging modalities at the same lesion in this study. The process of osteolytic bone metastases formation is triggered by the interaction among tumor cells, bone marrow environment and bone cells (vicious cycle) [32]. The tissue component changes during the “vicious cycle” until bone destruction and structure absence. “Hot area” on both imaging modalities is possibly due to increased activation of osteolysis and osteogenesis at early stage, while “cold area” is possibly associated with bone structure resorption, destruction, internal necrosis at late stage of osteolytic bone metastasis. The CT information on SPECT-CT is important to distinguish malignant osteolytic bone destruction from benign ones when they demonstrate as “cold area” on both 99mTc-3P-RGD2 and 99mTc-MDP imaging. Thus, these two imaging modalities are potential to play complementary roles in reflecting pathophysiological status of osteolytic lesions. In future, further studies are needed to develop targeted therapies for the management of osteolytic metastasis using 177Lu and 90Y labeled RGD peptide.
Conclusions
Using integrin αvβ3 highly expressed in the main component of osteolytic bone metastases (e.g. tumor cells and osteoclasts) as a target, 99mTc-3P-RGD2 imaging demonstrated mainly as “hot region” on whole body imaging with low background.
99mTc-3P-RGD2 imaging showed significantly higher detective rate than 99mTc-MDP imaging based on visual and semiquantitive analysis. Such technique is potentially applicable for the detection of some unknown primary tumors and distant metastases.
ACKNOWLEDGMENTS
We are grateful to Prof. Shuang Liu for his help in RGD dimer kit donation and Wei Gu, Muhong Guo for clinical data collection and follow-up.
This study was financially supported by grants from National Natural Science Foundation of China projects (81301247), the Natural Science Foundation of Jiangsu Province (BK20130082), Jiangsu Provincial Medical Youth Talent (QNRC2016075) and the Nanjing Outstanding Youth Fund (JQX14009).
Conflicts of interest
There is no conflict of interest.
REFERENCES
1. Ell B, Kang Y. SnapShot: Bone Metastasis. Cell. 2012; 151:690-690 e691. doi: 10.1016/j.cell.2012.10.005.
2. Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014; 64:9-29. doi: 10.3322/caac.21208.
3. Kakonen SM, Mundy GR. Mechanisms of osteolytic bone metastases in breast carcinoma. Cancer. 2003; 97:834-839. doi: 10.1002/cncr.11132.
4. Kuchuk M, Kuchuk I, Sabri E, Hutton B, Clemons M, Wheatley-Price P. The incidence and clinical impact of bone metastases in non-small cell lung cancer. Lung Cancer. 2015; 89:197-202. doi: 10.1016/j.lungcan.2015.04.007.
5. Braun S, Vogl FD, Naume B, Janni W, Osborne MP, Coombes RC, Schlimok G, Diel IJ, Gerber B, Gebauer G, Pierga JY, Marth C, Oruzio D, et al. A pooled analysis of bone marrow micrometastasis in breast cancer. N Engl J Med. 2005; 353:793-802. doi: 10.1056/NEJMoa050434.
6. Eustace S, Tello R, DeCarvalho V, Carey J, Wroblicka JT, Melhem ER Yucel EK. A comparison of whole-body turboSTIR MR imaging and planar 99mTc-methylene diphosphonate scintigraphy in the examination of patients with suspected skeletal metastases. AJR Am J Roentgenol. 1997; 169:1655-1661. doi: 10.2214/AJR.11.8351.
7. Mouchess ML, Sohara Y, Nelson MD Jr, DeCLerck YA, Moats RA. Multimodal imaging analysis of tumor progression and bone resorption in a murine cancer model. J Comput Assist Tomogr. 2006; 30:525-534. doi: 10.1097/00004728-200605000-00030.
8. Peterson JJ, Kransdorf MJ, O’Connor MI. Diagnosis of occult bone metastases: positron emission tomography. Clin Orthop Relat Res. 2003; 415:S120-128. doi: 10.1097/01.blo.0000093051.96273.7c.
9. Roodman GD. Mechanisms of bone lesions in multiple myeloma and lymphoma. Cancer. 1997; 80:1557-1563.
10. Mundy GR. Metastasis to bone: causes, consequences and therapeutic opportunities. Nat Rev Cancer. 2002; 2:584-593. doi: 10.1038/nrc867.
11. Boissy P, Machuca I, Pfaff M, Ficheux D, Jurdic P. Aggregation of mononucleated precursors triggers cell surface expression of alphavbeta3 integrin, essential to formation of osteoclast-like multinucleated cells. J Cell Sci. 1998; 111:2563-2574.
12. Chen B, Zhao G, Ma Q, Ji B, Ji T, Xin H, Gao S. (99m)Tc-3P-RGD2 SPECT to monitor early response to bevacizumab therapy in patients with advanced non-small cell lung cancer. Int J Clin Exp Pathol. 2015; 8:16064-16072.
13. Jin X, Liang N, Wang M, Meng Y, Jia B, Shi X, Li S, Luo J, Luo Y, Cui Q, Zheng K, Liu Z, Shi J, et al. Integrin Imaging with 99mTc-3PRGD2 SPECT/CT Shows High Specificity in the Diagnosis of Lymph Node Metastasis from Non-Small Cell Lung Cancer. Radiology. 2016; 281:958-966. doi: 10.1148/radiol.2016150813.
14. Ma Q, Ji B, Jia B, Gao S, Ji T, Wang X, Han Z, Zhao G. Differential diagnosis of solitary pulmonary nodules using (9)(9)mTc-3P(4)-RGD(2) scintigraphy. Eur J Nucl Med Mol Imaging. 2011; 38:2145-2152. doi: 10.1007/s00259-011-1901-2.
15. Zhu Z, Miao W, Li Q, Dai H, Ma Q, Wang F, Yang A, Jia B, Jing X, Liu S, Shi J, Liu Z, Zhao Z, et al. 99mTc-3PRGD2 for integrin receptor imaging of lung cancer: a multicenter study. J Nucl Med. 2012; 53:716-722. doi: 10.2967/jnumed.111.098988.
16. Jia B, Liu Z, Zhu Z, Shi J, Jin X, Zhao H, Li F, Liu S, Wang F. Blood clearance kinetics, biodistribution, and radiation dosimetry of a kit-formulated integrin alphavbeta3-selective radiotracer 99mTc-3PRGD 2 in non-human primates. Mol Imaging Biol. 2011; 13:730-736. doi: 10.1007/s11307-010-0385-y.
17. Shao G, Zhou Y, Wang F, Liu S. Monitoring glioma growth and tumor necrosis with the U-SPECT-II/CT scanner by targeting integrin alphavbeta3. Mol Imaging. 2013; 12:39-48.
18. Mi B, Yu C, Pan D, Yang M, Wan W, Niu G, Chen X. Pilot Prospective Evaluation of (18)F-Alfatide II for Detection of Skeletal Metastases. Theranostics. 2015; 5:1115-1121. doi: 10.7150/thno.12938.
19. Even-Sapir E, Metser U, Mishani E, Lievshitz G, Lerman H, Leibovitch I. The detection of bone metastases in patients with high-risk prostate cancer: 99mTc-MDP Planar bone scintigraphy, single- and multi-field-of-view SPECT, 18F-fluoride PET, and 18F-fluoride PET/CT. J Nucl Med. 2006; 47:287-297.
20. Hahn S, Heusner T, Kummel S, Koninger A, Nagarajah J, Muller S, Boy C, Forsting M, Bockisch A, Antoch G, Stahl A. Comparison of FDG-PET/CT and bone scintigraphy for detection of bone metastases in breast cancer. Acta Radiol. 2011; 52:1009-1014. doi: 10.1258/ar.2011.100507.
21. Kim MR, Roh JL, Kim JS, Choi SH, Nam SY, Kim SY. 18F-fluorodeoxyglucose-positron emission tomography and bone scintigraphy for detecting bone metastases in patients with malignancies of the upper aerodigestive tract. Oral Oncol. 2008; 44:148-152. doi: 10.1016/j.oraloncology.2007.01.011.
22. Uchida K, Nakajima H, Miyazaki T, Tsuchida T, Hirai T, Sugita D, Watanabe S, Takeura N, Yoshida A, Okazawa H, Baba H. (18)F-FDG PET/CT for Diagnosis of Osteosclerotic and Osteolytic Vertebral Metastatic Lesions: Comparison with Bone Scintigraphy. Asian Spine J. 2013; 7:96-103. doi: 10.4184/asj.2013.7.2.96.
23. Sprague JE, Kitaura H, Zou W, Ye Y, Achilefu S, Weilbaecher KN, Teitelbaum SL, Anderson CJ. Noninvasive imaging of osteoclasts in parathyroid hormone-induced osteolysis using a 64Cu-labeled RGD peptide. J Nucl Med. 2007; 48:311-318.
24. Ito S, Kato K, Ikeda M, Iwano S, Makino N, Tadokoro M, Abe S, Nakano S, Nishino M, Ishigaki T, Naganawa S. Comparison of 18F-FDG PET and bone scintigraphy in detection of bone metastases of thyroid cancer. J Nucl Med. 2007; 48:889-895. doi: 10.2967/jnumed.106.039479.
25. McLoughlin LC, O’Kelly F, O’Brien C, Sheikh M, Feeney J, Torreggiani W, Thornhill JA. The improved accuracy of planar bone scintigraphy by adding single photon emission computed tomography (SPECT-CT) to detect skeletal metastases from prostate cancer. Ir J Med Sci. 2016; 185:101-105. doi: 10.1007/s11845-014-1228-7.
26. Zhang Y, Shi H, Gu Y, Xiu Y, Li B, Zhu W, Chen S, Yu H. Differential diagnostic value of single-photon emission computed tomography/spiral computed tomography with Tc-99m-methylene diphosphonate in patients with spinal lesions. Nucl Med Commun. 2011; 32:1194-1200. doi: 10.1097/MNM.0b013e32834bd82e.
27. Miao W, Zheng S, Dai H, Wang F, Jin X, Zhu Z, Jia B. Comparison of 99mTc-3PRGD2 integrin receptor imaging with 99mTc-MDP bone scan in diagnosis of bone metastasis in patients with lung cancer: a multicenter study. PLoS One. 2014; 9:e111221. doi: 10.1371/journal.pone.0111221.
28. Beer AJ, Lorenzen S, Metz S, Herrmann K, Watzlowik P, Wester HJ, Peschel C, Lordick F, Schwaiger M. Comparison of integrin alphaVbeta3 expression and glucose metabolism in primary and metastatic lesions in cancer patients: a PET study using 18F-galacto-RGD and 18F-FDG. J Nucl Med. 2008; 49:22-29. doi: 10.2967/jnumed.107.045864.
29. Zhao D, Jin X, Li F, Liang J, Lin Y. Integrin alphavbeta3 imaging of radioactive iodine-refractory thyroid cancer using 99mTc-3PRGD2. J Nucl Med. 2012; 53:1872-1877. doi: 10.2967/jnumed.112.107821.
30. Cheng W, Wu Z, Liang S, Fu H, Wu S, Tang Y, Ye Z, Wang H. Comparison of 18F-AIF-NOTA-PRGD2 and 18F-FDG uptake in lymph node metastasis of differentiated thyroid cancer. PLoS One. 2014; 9:e100521. doi: 10.1371/journal.pone.0100521.
31. Sutherland M, Gordon A, Shnyder SD, Patterson LH, Sheldrake HM. RGD-Binding Integrins in Prostate Cancer: Expression Patterns and Therapeutic Prospects against Bone Metastasis. Cancers (Basel). 2012; 4:1106-1145. doi: 10.3390/cancers4041106.
32. Chirgwin JM, Guise TA. Molecular mechanisms of tumor-bone interactions in osteolytic metastases. Crit Rev Eukaryot Gene Expr. 2000; 10:159-178.

