INTRODUCTION
With a global incidence of about 1.4 million individuals and 693,900 deaths reported in 2012, colorectal cancer (CRC) is the third most common type of cancer and the fourth most common cause of cancer deaths worldwide [1, 2]. The conventional treatment strategies for CRC include surgery, neoadjuvant radiotherapy (rectal cancer patients), and adjuvant chemotherapy (stage III/IV and high-risk stage II colon cancer patients) [3]. Unfortunately, about 50% of the patients with CRC are diagnosed only at late stages, thereby significantly reducing the availability of different treatment options. Despite the strong inheritance factor related to it, CRC is commonly sporadic and proceeds slowly over 10 years, hence correct diagnosis of the disease-stage becomes crucial for prognosis. The 5-year relative survival ranges from greater than 90% in patients with early-stage localized disease (Stages I and II) to only slightly greater than 13% in patients with late-stage CRC [4]. Nowadays, targeted therapy is one of the principal modes of cancer treatment, which has had varying degrees of success owing to the diverse range of resistance mechanisms [5, 6]. Within a few months into treatment, it is common to see changes in the tumor and development of therapeutic resistance [7].
Therefore, screening and early detection of CRC remain clinical dilemmas and are critically important for raising the chances of a more suitable treatment leading to better long-term survival. Several approaches involving colonoscopy, sigmoidoscopy, fecal occult blood test (FOBT) [2, 8], serum biomarkers such as carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA19-9) [9], together with Magnetic Resonance Imaging (MRI) and Computed Tomography (CT) [10] are commonly used to screen or diagnose CRC. However, the use of CEA or CA19-9 is limited by its low sensitivity and specificity [11]. Because of the inherent characteristics of CT or MRI, some early tumor dissemination or micro-metastases may be missed in tumor detection, so is the case with colonoscopy with a higher risk of complications. Although colonoscopy is still the most effective method to diagnose CRC, this approach has poor patient compliance and might not provide real-time monitoring of tumor progression and therapeutic response.
Therefore, it is necessary to identify ideal bio-markers that can be used for the early diagnosis, detection of recurrence, and monitoring of metastasis for CRC. Using body fluid samples, liquid biopsies may be the ideal approach for the detection of CTCs or for the products of primary or metastatic tumors. The advent of liquid biopsy as an alternate, easy, quick, convenient, and minimally invasive method has shown tremendous potential to help clinicians achieve early diagnosis of cancer and guide decision-making during the course of treatment. It is expected to be an informative or easily accessible tool to provide comprehensive information of tumors beyond the invasive tissue biopsies. The main approaches to liquid biopsies include the detection of CTCs [12], analysis of ctDNA or RNA [13, 14], and the capture of exosomes that are secreted by tumors [15] (Figure 1).
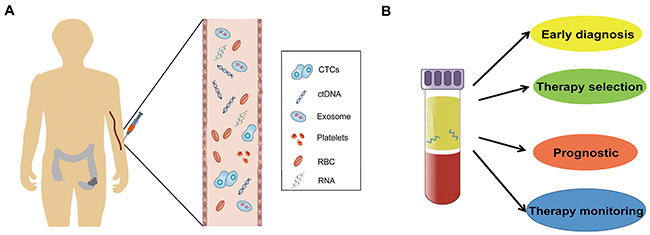
Figure 1: Circulating biomarkers in CRC patient (A) and the clinical application of liquid biopsy (B).
This review summarizes and discusses the current clinical and biological significance of CTCs, ctDNA, and exosomes in CRC. Additionally, the review covers significant advances and limitations of liquid biopsy in the clinical applications, discusses the hurdles that need to be resolved, and provides potential directions for the detection and therapy of CRC.
ORIGINS, MERITS, AND DEMERITS OF CTCS/ CTDNA/ EXOSOMES
Over the past few decades, many procedures have been developed for liquid biopsy ranging from the use of CTCs and ctDNA or RNA to that of exosomes and platelets [16, 17] detection, requiring time to judge and improve their effects. These biomarkers have several advantages over traditional tissue biopsy and their shortcomings, if any, need to solve (Table 1). With a large number of methods available for the detection of circulating biomarkers, there has not yet been a consensus on both the ideal technical method and clinical application.
Table 1: Merits and demerits of CTCs /ctDNA/exosomes
Merits |
Demerits |
|
|---|---|---|
CTCs |
Identified morphologically and molecular characterization |
Low-input amounts and isolate rare cells with limited capture techniques |
Allows immuno-labeling based approaches |
Methodological limitations(sensitivity and specificity) and standardization |
|
Allow functional in vitro/in vivo assays |
Heterogeneity of the CTC populations |
|
ctDNA |
Quick-renewed |
Discrimination of ctDNA from normal cfDNA |
Short half-life |
Extremely low levels of ctDNA |
|
More sensitive for detection of tumor status |
No functional assays |
|
Identified molecular characterization |
Methodological limitations(sensitivity and specificity) and standardization |
|
Exosomes |
Inherent stability, maintain the integrity of contents |
Isolation and purification of exosomes |
With concentrations of ≥109 vesicles/mL |
The enrichment of specific markers within the exosomes |
|
Abundant contents |
Methodological limitations(sensitivity and specificity) and standardization |
|
As vectors for anti-tumor therapy of gene or drug delivery |
Circulating tumor cells
CTCs are circulating tumor cells that are shed into the bloodstream from tumors and have the potential to cause metastatic lesions [18, 19]. The CTC pool in cancer patients may include not only epithelial tumor cells, but also epithelial–mesenchymal transition (EMT) tumor cells, hybrid (epithelial/EMT+) tumor cells, irreversible EMT+ tumor cells, and circulating tumor stem cells (CTSCs) [20]. They can be single or clustered together in circulation, or form metastases. CTCs can be separated from normal blood cells by physico-chemical characteristics or cell surface molecules. As real-time and noninvasive surrogates, the isolation and detection of CTCs is one of the most promising methods to help patients with CRC obtain early diagnosis and accurately predict metastasis or recurrence of malignancies. The most widely used CTC enumeration platform, CellSearch™ System (Veridex LLC, NJ, USA), has been approved for clinical use by the Food and Drug Administration (FDA) in the United States to monitor patients with metastatic colorectal, prostate, and breast cancers [21-25]. In order to study the functional attributes of CTCs, Aceto et al. captured and cultured the CTCs and implanted them into mouse and proved that rare CTC clusters, shed from tumor in situ, contributed to the metastasis through plakoglobin-dependent intercellular adhesion [26].
Although the underlying advantages of CTCs make it a promising tool to help monitor the dynamic course of disease, there are some potential limitations in the existing detection techniques [27-29] and from the heterogeneity of the CTCs. Methods that rely on the physical properties of CTCs for their capture, typically achieve high capture efficiency which is greater than 80%; however, not all CTCs are larger than nucleated blood cells, for example, the tumor cells undergoing apoptosis or those in EMT may be smaller in size [29]. Methods that capture positive biomarkers of CTCs are flawed too, causing the actual CTC load to be underestimated and CTC populations that are highly relevant to the disease progression to be missed [30]. Epithelial cell adhesion molecule (EpCAM) expression was approximately 10-fold lower on CTCs compared to on primary and metastatic tissues, suggesting that it is dependent on the local microenvironment and is down-regulated on CTCs [31]. This may be the reason why CTCs were undetected by the CellSearch™ System in a significant proportion of patients with CRC [32]. Although the CellSearch™ is the only FDA-approved technology applied for CTC enrichment, it cannot capture non-epithelial CTCs, such as the CTCs that have undergone EMT, resulting in a missed detection. Moreover, RNA can be extracted from CTCs and detected for guiding treatment decision. The real-time reverse transcription polymerase chain reaction (RT-PCR) assay targeting mRNA may have the advantage of improved sensitivity, compared to cell-based assays [33, 34]. Until date, no ideal surface marker from CTCs covers all the stages or types of cancer.
Circulating tumor DNA
Circulating free DNA (cfDNA) is a natural phenomenon and is thought to originate from the apoptosis and necrosis of normal and tumor cells, and secretion of tumor cells has also been suggested as a potential source [28, 35]. The fragments of ctDNA showed an enrichment of 166 bp, clearly corresponding to the size of DNA wrapped around the nucleosomes [36, 37]. The mean half-life for circulating fetal DNA has been found to be 16.3 min (range 4–30 min) and they are eliminated within 2 hours [38]. Similarly, ctDNA also has a short half-life and the rapid turnover time allow clinicians to monitor the dynamic changes in tumor within a matter of only few hours rather than weeks [39]. Therefore, ctDNA is a prospective material in cancer research with vast amounts of information. Traditional detection methods such as Sanger sequencing and quantitative PCR can only test higher concentrations of ctDNA due to its low sensitivity. The emergence of BEAMing (beads, emulsion, amplification and magnetics) and digital PCR (dPCR) allows researchers to detect as low as 0.01% ctDNA with known anomalous sequence in circulation [6]. Fortunately, the next generation sequencing (NGS) technology addresses this issue, which can monitor all possible cancer mutations and identify rare mutation variants. Additionally, benign tumors and nonneoplastic conditions do not generally give rise to ctDNA, and therefore may not influence the detection [40].
Many studies have demonstrated that the level of cfDNA is substantially higher in patients with cancer than that in healthy individuals or patients with benign diseases, and it seems to increase with tumor stage [41, 42]. ctDNA analysis of Epstein–Barr virus (EBV) has been certified for the early screening and detection of nasopharyngeal carcinoma (NPC) in patients without any former indications [43, 44]. Although quantification of ctDNA may not be informative enough for tumor diagnosis by itself, a decrease or increase in its level after therapy may be a prognostic factor for residual disease and recurrence [43]. The first prospective study using colonoscopies as the reference standard to assess the accuracy of circulating methylated SEPT9 DNA (mSEPT9) for detecting CRC in a screening population was conducted by Church et al. [45]. Results from 53 CRC cases and 1457 subjects without CRC yielded a standardized sensitivity of 48.2% and specificity of 91.5% [45]. Although early detection and screening strategies based on ctDNA are promising, it can only provide limited information for cancer owing to the related technical challenges and other obstacles [46]. Therefore, the use of ctDNA detection as a CRC screening tool requires further exploration. Nevertheless, the detection of somatic mutations in ctDNA can offer the potential for better diagnostic accuracy and guiding treatment decisions [47].
Exosomes
Exosomes, which are well known for intercellular communication, are small membrane vesicles (30–100 nm) released from diverse cell types under both normal and pathological conditions, that can horizontally transfer functional biomolecules (i.e., DNA, RNA, proteins, and lipids) to recipient cells [48-50]. The exosomal membrane reflects the cell plasma membrane in several ways and can therefore be immuno-isolated utilizing significantly enriched proteins on the membrane surface, e.g., A33 affinity-isolation of CRC cell line exosomes [49]. It may not only serve as an important regulatory mechanism during cancer development and progression, such as promoting adhesion, triggering signaling pathways and inflammatory responses, or immune escape, but also play significant role in the diagnosis, treatment assessment, and prognosis of tumor. Besides, exosomes can be directly used as vectors for cancer intervention through gene or drug delivery.
Previous studies by Peinado and colleagues have demonstrated that exosomes played vital roles in vascular leakiness, inflammation, and bone-marrow progenitor cell recruitment during pre-metastatic niche formation and metastasis itself [51]. Subsequently, data have indicated that integrin expression profiles of plasma exosomes, isolated from cancer patients, could be used as prognostic factors to predict sites of future metastasis [52]. Indeed, exosomes perform a variety of extracellular functions that involve interactions with the cellular microenvironment, such as immunological mediation, cell recruitment, and horizontal transfer of genetic material [53]. Interestingly, with concentrations as high as or above 109 vesicles/mL in blood, the number of exosomes secreted by tumor cells correlates to their malignant behavior [50, 53]. Therefore, to examine whether exosomal DNA (exoDNA) could be utilized as a surrogate for tumor tissues or cells in the detection of tumor-specific genetic mutations just as it is with ctDNA, researchers have tested the exoDNA isolated from various cancer cell lines to detect the BRAF (V600E) mutation and anti-epidermal growth factor receptor (EGFR) mutation, using allele-specific polymerase chain reaction (AS-PCR). The findings demonstrated that exoDNA could reflect the mutational status of the parental cell lines [54]. However, some confounding factors, such as HIV-1 particles, may decrease the accuracy and reliability of the detected results in the analysis of exosomes. Evidently, a relationship between exosomes and tumor is like that of a seed and its plant, and therefore, exosomes can be expected to serve as useful biomarkers owing to their remarkable stability in fluids.
CIRCULATING TUMOR CELLS, CIRCULATING TUMOR DNA, AND EXOSOMES AS BIOMARKERS IN COLORECTAL CANCER
Having discussed the advantages and disadvantages of CTCs, ctDNA, and exosomes in different aspects, it is clear that these hold promise for researchers attempting to monitor tumor-specific changes during the course of cancer. This may also help clinicians to carry out noninvasive real-time assessment in various clinical settings, including diagnosis, therapy monitoring, and prognosis (Table 2).
Table 2: Application value of CTCs/ctDNA/exosomes in CRC
Material |
Biomarkers |
Potential clinical utility |
Methods |
References |
|---|---|---|---|---|
CTCs |
CK-19, EpCAM |
Prognostic |
CK19-Epispot and CellSearch™ |
[58] |
EpCAM |
Predictive and prognostic |
CellSearch™ |
||
CEA, CK19, and CK20 |
Prognostic |
RT-PCR |
[33] |
|
CEA/CK/CD133 |
Prognostic |
RT-PCR |
[34] |
|
EpCAM, CKs; VIM, TWIST1, AKT2, SNAI1 |
Prognostic |
CanPatrolTM |
[60] |
|
CD133, EpCAM, CD26, CD44v6 |
Prognostic(functional research) |
Drug sensitivity analysis of CTC lines |
[62] |
|
KRAS, BRAF, and PI3KCA |
Prognostic |
Label-free Vortex technology |
[28] |
|
EpCAM |
Screening high-risk stage II CRC patients |
CellSearch™ |
[66] |
|
ctDNA |
Methylated SEPT9 |
Screening and early detection |
Duplicate real-time PCRs |
[45] |
APC, KRAS, TP53, and PI3KCA |
Prognostic and therapy monitoring |
BEAMing, real-time PCR |
[39] |
|
A modest panel of 15 genes |
Predictive |
Safe-SeqS |
[67] |
|
APC, KRAS, TP53 |
Predictive |
Safe-SeqS |
[68] |
|
Methylated WIF1 and NPY |
Prognostic |
dPCR |
[69] |
|
Methylated ALU83, ALU244, OSMR, and SFRP1 |
Diagnostic and prognostic |
ALU-based real-time PCR, methylation-specific real-time PCR |
[70] |
|
KRAS |
Therapy selection and monitoring |
BEAMing, real-time PCR, and NGSeq, |
[71] |
|
KRAS |
Therapy selection and monitoring |
BEAMing and real-time PCR |
[7] |
|
KRAS, BRAF |
Therapy selection and monitoring |
Real-time PCR |
[72] |
|
KRAS, NRAS, MET, ERBB2, FLT3, EGFR, and MAP2K1 |
Therapy selection and monitoring |
dPCR and NGS |
[74] |
|
KRAS, BRAF |
Therapy selection and monitoring |
ARMS-qPCR |
[75] |
|
Exosomes |
miRNAs |
Diagnostic |
miRNA microarray analysis and qRT-PCR |
[79] |
miRNAs |
Prognostic |
Microarray and CGH analysis and qRT-PCR |
[80] |
|
Proteins |
Diagnostic |
Mass spectrometry and western blotting |
[81] |
|
Proteins |
Prognostic (function research) |
Proteomics measurements and western blotting |
[82] |
|
Microvesicles (Fas ligand and TNF) |
Prognostic and therapeutic(function research) |
Flow cytometry, western blotting, and immunoelectron microscopy |
[89] |
Clinical applications of CTCs in colorectal cancer
A milestone in CTC research (CellSearch™) in patients with metastatic breast cancer (MBC) was reported in The New England Journal of Medicine in 2004 [55]. The researchers discovered that five or more CTCs/7.5 mL were detected in 49% (87/177) of the patients. Another study on patients with MBC treated with standard therapy found that 46.9% (911/1944) of the patients had a CTC count of ≥5 CTCs/7.5 mL at baseline [56]. These patients with decreased progression-free survival (PFS) and overall survival (OS) were compared with patients with less than five CTCs per 7.5 mL at baseline. These data confirmed the number of CTCs before treatment is an independent predictor of PFS and OS in MBC patients. However, the level of CTCs was lower in peripheral blood of colon cancer patients compared to that in breast or prostate cancer patients, making it more difficult to detect CTCs in colon cancer [57].
Deneve et al. demonstrated that the CTC counts were significantly higher in mesenteric blood than in the peripheral blood, which meant that the liver acts as a filter for CTCs, and the follow-up analysis showed that localized colon-cancer patients with high CTC counts have an unfavorable outcome (n =60) [58]. By contrast, fewer CTCs meant a longer median PFS and OS. Therefore, collection of mesenteric blood may improve the detection ratio of CTCs. Similar observations were made in other cancer types, including prostate and breast cancers [55, 59]. In a published study Bork et al. showed for the first time that preoperative CTC detection by the standardized CellSearch™ System is a strong and independent prognostic factor for disease progression and survival in patients with non-metastatic CRC, although results show a significantly lower detection rate of CTC in patients with non-metastatic CRC [60]. Another prospective, multicenter study evaluated CTCs in 430 patients with metastatic CRC (mCRC) at baseline and after first-, second-, or third-line therapy by using a CellSearch™ System [22]. Patients with unfavorable (≥3 CTCs/7.5 mL) compared with favorable (<3 CTCs/7.5 mL) baseline CTCs had shorter median PFS and OS. They demonstrate that CTCs can serve as both prognostic and predictive factors for patients with mCRC, and that the baseline levels of three or more CTCs /7.5 mL and follow-up CTC levels are strong independent prognostic factors for inferior PFS and OS [22]. A meta-analysis suggested that for patients with CRC, CTC mRNA detection, targeting CEA, cytokeratin (CK) 19, and CK20, using RT-PCR could serve as a prognostic indicator and a mode of CRC staging [33]. Crucially, a study demonstrated that the detection of CEA/CK/CD133 mRNA in the circulating blood of patients with Dukes’ stage B and C CRC who require adjuvant chemotherapy is a useful tool for determining which patients are at high risk for recurrence and poor prognosis [34]. Thus, CTC could serve as a much earlier prognostic and predictive factor than standard anatomical or functional imaging studies to monitor tumor burden in real-time.
Recently, a large cohort of 1203 patients was evaluated for the expression of EMT markers in CTCs by using CanPatrolTM CTC technique. Biophenotypic (epithelial/mesenchymal) CTCs as well as mesenchymal CTCs positively correlated with both clinical stage and lymph node metastasis and distant metastasis, suggesting that CTCs displaying a mesenchymal phenotype would denote a more aggressive and metastatic potential [61]. Their findings suggest that the combination of epithelial (EpCAM, CKs) and mesenchymal (VIM, TWIST1, AKT2, and SNAI1) markers in CTCs analysis may offer valuable aid for tumor staging and metastasis evaluation in patients with CRC, which may be superior to CellSearch™ System [61].
Cancer stem cells are capable of seeding, recirculation, and evolution in metastatic clones, which causes clinical progression of the disease [19]. The first permanent CRC cell line, named CTC-MCC-41 and obtained from the CTCs of a patient with colon cancer, was established by Laure et al. in 2015 [62]. Genome, transcriptome, proteome, and secretome level analyses and functional studies showed that CTC-MCC-41 cells derived from the bone marrow based on osteoprotegerin expression, showed an intermediate epithelial/mesenchymal phenotype with stem cell–like characteristics and could induce in vitro angiogenesis and tumors in immune-deficient mice [62]. By establishing CTC lines from the blood of patients with mCRC, another study also demonstrated that patient-derived colorectal CTCs own all the functional attributes of CTSCs [63]. The cytotoxicity assay confirmed the potential application of this model to predict individual patient-drug response. Strikingly, the CTCs cultured model was simple and took less than a month from blood collection to drug testing [63]. Therefore, the detection, in vitro culture, and molecular characterization of CTCs should divulge the prognosis of mCRC as well as monitor the drug response, in addition to early detection of disease progression with new metastasis.
National Comprehensive Cancer Network (NCCN) highly recommends that patients with mCRC should be tested for the RAS or BRAF mutation status in primary or metastatic tumors. The mutation status of KRAS, BRAF, and PI3KCA could affect the treatment response to EGFR monoclonal antibodies or small molecule inhibitors and have treatment-independent prognostic value [28, 64, 65]. Fabbri et al. demonstrated, for the first time, the feasibility of analyzing pure CTCs at the molecular level and avoiding lymphocyte contamination using a DEPArray, a dielectrophoresis-based platform, as well as a KRAS discordance between CTCs and primary tissue cancer after 100% pure cell recovery and sequencing [66]. The unexpected CTC-primary tissue discordance may be due to the presence of intratumor heterogeneity and multiple metastatic clones, which are disseminated very early during disease progression and remain dormant for years [66]. In a recent study including 23 matched CTC and ctDNA samples, KRAS, BRAF, and PIK3CA hotspot mutations were analyzed by a label-free platform. Researchers found a concordance of 78.2% for KRAS, 73.9% for BRAF, and 91.3% for PIK3CA mutations [28]. In general, complementary assessment of both CTCs and ctDNA should be more superior to assess dynamic tumor profiles.
Thus, in primary or non-metastatic CRC, the existence of CTCs might indicate poor prognosis; while in advanced or metastatic CRC, there should be a positive correlation between the level of CTCs with the tumor progression and poor outcomes; CTCs can guide treatment decisions and assess treatment responses during the course of therapy; the molecular analysis of CTCs may predict drug resistance and the selection of anticancer drugs [61]. Gazzaniga et al. evaluated the CTCs in 37 high-risk patients with stages II-III CRC after primary tumor resection and before the start of adjuvant therapy. Results showed 87.5% (7/8) CTC-positive patients had N1–2 disease and stage III CRC, whereas only one patient who experienced the progression of disease had a high risk stage II disease [67]. The data suggest that the detection of CTCs might help screen high-risk stage II CRC candidates for adjuvant chemotherapy, after enumerating CTCs with the CellSearch system [67]. Whether CTCs detection can differentiate patients with N+ disease and some of the patients with N+ disease can skip chemotherapy need to be further explored. Although the detection of CTCs as a biomarker of tumor has been well accepted, strategies involving the use of CTCs to completely guide the treatment decisions are in progress. Remarkably, the sensitivity and specificity of the CTC detection methods could pose as an obstacle that needs to be urgently resolved.
Clinical applications of ctDNA in colorectal cancer
Diehl et al. applied BEAMing to quantify ctDNA in CRC patients undergoing multimodality therapy. Compared to CEA, the levels of ctDNA were more impressive in the prediction of recurrence (P=0.03), which certified that fluctuations of ctDNA could be used to monitor the course of therapy in patients with mCRC undergoing surgery or chemotherapy [39]. In another study involving 53 patients with mCRC receiving standard first-line chemotherapy, the concordance between ctDNA (Safe-SeqS) and tumor tissue was found to be 92.3%. In general, ctDNA appeared to be an early biomarker to infer the tumor burden of patients with CRC during first-line chemotherapy and predicts an earlier therapeutic reaction than radiographic approaches [68]. In addition, a prospective cohort study of 230 patients with resected stage II colon cancer demonstrated that the detection of ctDNA after resection provides direct evidence of the residual disease and identifies patients at very high risk of recurrence, thus being superior to other clinico-pathological measures [69]. These findings establish the rationale for measuring ctDNA for using as a monitoring tool for recurrence and guiding clinical decisions.
Until date, several studies have focused on the detection of methylated DNA in patients with CRC. Garrigou and colleagues reported that hypermethylation of WIF1 (WNT inhibitory factor 1) and NPY (neuropeptide Y) was significantly higher in the tumor tissue compared to that in normal tissue and these methylated ctDNA (Met ctDNA) were detectable throughout the tumor progression, with their fraction being correlated to the tumor stage [70]. Therefore, MetctDNA could be a promising surrogate marker for tumor follow-up in patients with CRC, which means that WIF1 and NPY could instead be tumor-specific mutations. Recently, Bedin et al. found that the adenoma and methylated cfDNA in patients with CRC, showed a higher quantity of ALU83 and ALU244 [71]. In this study, OSMR and SFRP1 methylation of cfDNA was also significantly higher in advanced CRC compared to that in the adenoma and control samples. This combination can improve the diagnostic efficiency and prognosis for CRC.
Misale et al. demonstrated, for the first time, that KRAS mutations are frequent drivers of acquired resistance to cetuximab in CRC, and indicated that the emergence of KRAS mutant clones can be detected, non-invasively, months prior to radiographic progression [72]. They also suggested early initiation of an MEK inhibitor as a rational strategy for delaying or reversing drug resistance [72]. The same result was reported by Diaz et al. [7]. They also found that each relatively large metastatic lesion was expected to contain a subclone comprising hundreds or thousands of cells with one of ~42 mutations conferring resistance to the antibody, making resistance a fait accompli; the time to recurrence is simply the interval required for the subclone to repopulate the lesion [7]. The first blinded prospective multicenter study compared the mutation status of KRAS and BRAF in CRC tumor tissue, using routine gold-standard methods, and in ctDNA, using a quantitative PCR-based method. cfDNA analysis showed 100% specificity and sensitivity for the BRAF V600E mutation and 98% specificity and 92% sensitivity with a concordance value of 96% for the KRAS mutation, compared with a tumor-tissue analysis [73]. To conclude, the cfDNA analysis may advantageously replace tumor-section analysis and expand the scope of the management of personalized cancer care [73].
As a result of clonal evolution and selection [74], the ctDNA profiles of CRC patients who benefit from multiple challenging with anti-EGFR antibodies, exhibit pulsatile levels of mutant KRAS. Results revealed that the CRC genome adapts dynamically intermittent drug schedules and provided a molecular explanation for the efficacy of rechallenge therapies based on EGFR blockade [75]. Because of the clinical significance of the KRAS gene, KRAS wild type status in the primary tumor is a prerequisite for treatment modality of patients with mCRC with targeted therapy apart from the best supportive care alone [64, 73]. The previous study including 108 patients with mCRC monitored the number of mutant KRAS or BRAF alleles in the plasma at baseline and before each cycle of the third-line treatment with cetuximab and irinotecan. cfDNA and KRAS levels was found to decrease from baseline to cycle 3 and increased in progress (P = 0.08), while the loss of mutations was associated to the benefit of treatment, whereas the appearance of mutations during therapy may be correlated to acquired resistance in primary wild-type disease [76]. The appearance of KRAS mutation in wild-type tumors indicated a shift for the poor progression, while some patients with CRC had tumors that contained mutation but wild-type KRAS in plasma proved to have responded better to the therapy [76].
Most published studies of ctDNA have evaluated a single type of tumor mutation; however, numerous cancers have been reported with several undefined mutations. Further, the detection of a known anomalous gene could not completely reflect the heterogeneity of the tumor or replace the gold standard for tissue biopsy. A study explored the feasibility of Ion Torrent PGM (IonPGM) targeted NGS on cfDNA of mCRC patients undergoing colorectal liver metastasectomy [6]. The panel covered 21 most prevalent and relevant genes in CRC, known until date. Results showed that using NGS with IonPGM to detect cfDNA is feasible; however, it had limited sensitivity in the detection of all somatic mutations present in the tumor, especially in the case of unknown mutations. More importantly, true somatic mutations were present in normal-appearing tumor adjacent tissue, which implies that occult and potential lesions lead to recurrence. A study concluded that exome-wide analysis of ctDNA could complement the current invasive biopsy method to identify mutations related to acquired drug-resistance in advanced cancers [77].
Although increasing the sequencing depth of NGS could make the results more accurate, such an approach will be more time-consuming and costly, which is an obstacle for practical clinical application. However, whole genome sequencing with an appropriate depth of cfDNA could be available for clinical purposes, which can remedy the restrictions of dPCR. Because of the 0.01% error rate of DNA polymerases in the process of amplification, PCR or NGS-based methods that depend on amplification are limited. The newer sequencing technologies such as nanopore sequencing do not require nucleotides, polymerases, or ligases and have the potential of generating very long read-lengths (>10,000–50,000 nt), which might be a more competitive and portable technology for clinical applications [78, 79].
Clinical applications of exosomes in colorectal cancer
Lately, exosomes have greatly attracted researchers’ attention as potential biomarkers of cancer. A study, performed at Kanazawa University on microarray-based profiling of exosomal microRNAs (miRNAs) in sera from patients with primary CRC, healthy controls and cancer cell lines, validated CRC-associated exosomal miRNAs in an independent sample set using quantitative real-time RT-PCR [80]. They found that the serum exosomal levels of seven miRNAs (let-7a, miR-1229, miR-1246, miR-150, miR-21, miR-223, and miR-23a) were significantly higher in patients with primary CRC even in the early stage than that in healthy controls, which was found to decrease significantly after surgical resection. Colon-cancer cell lines secreted significantly higher levels of miRNAs than the normal colon-derived cell line in vitro [80]. To summarize, specific exosomal miRNA can reflect pathological changes in CRC and are therefore promising biomarkers for non-invasive diagnosis of CRC. Similarly, in order to identify specific miRNAs in exosomes as a prognostic factor of recurrence in CRC, Matsumura and colleagues explored miRNA expression profiles and copy number aberrations with microarray and array comparative genomic hybridization (CGH) analysis to confirm exosomal miRNAs in two serum sample sets by qRT-PCR [81]. The study found that the level of exosomal miR-17-92a cluster in serum was associated with the recurrence of CRC. Exosomal miR-19a was found to have significantly increased in patients with CRC compared to in healthy individuals, with gene amplification, and the patients with CRC with high exosomal miR-19a expression showed poorer prognoses than the low expression group (P < .001), which might be a promising prognostic biomarker for recurrence in patients with CRC [81].
Recently, Yanyu Chen et al. presented a quantitative proteomics analysis of purified exosomes from the serum of patients with CRC and normal volunteers, and identified 918 proteins with an overlap of 725 Gene IDs in the Exocarta proteins list [82]. There were 36 proteins that were up-regulated while 22 proteins were found to be down-regulated in the serum-purified exosomes (SPEs) of patients with CRC by bioinformatics analysis. They revealed that the differently expressed proteins of SPEs from patients with CRC are vital for tumor invasiveness and play putative roles in pre-metastatic niche establishment, but exert minimal influence on putative alterations in tumor survival or proliferation [82]. Accordingly, this research might give impetus to clarify the patho-physiological functions of exosomes and the development of CRC diagnostics and therapeutics. Another study revealed that the tumor suppressor gene TP53 changes the tumor microenvironment and promotes canceration via the endosomal sorting complex required for transport (ESCRT)-dependent exosome secretory machinery [83]. A comprehensive proteomic analysis identified that the expression of hepatocyte growth factor-regulated tyrosine kinase substrate (HGS) increases concomitantly with CRC tumorigenesis and is an independent poor prognostic factor, which may present itself as a novel biomarker and target for therapeutic interventions in CRC [83].
The delivery of exosomes to recipient cells could induce cell migration, inflammation, immune responses, angiogenesis, invasion, pre-metastatic niche formation, and metastasis [51-53, 84, 85]. Previous studies have shown that exosomes from mutant KRAS CRC cells can be transferred to wild-type cells to induce cell growth and migration [86, 87]. To test whether exosomal RNAs change the gene expression of recipient cells and whether mutant KRAS regulates the composition of the secreted miRNAs, researchers compared small RNAs of cells and matched exosomes from isogenic CRC cell lines differing only in the KRAS status [84]. Results showed that the KRAS status prominently affects the miRNA profile in cells and their corresponding exosomes, and cellular trafficking of miRNAs is sensitive to neutral sphingomyelinase (nSMase) inhibition in mutant KRAS cells as well as that the transfer of miRNAs between cells can functionally alter gene expression in recipient cells [84]. Similarly, researchers found that circular RNAs (circRNAs) were significantly down-regulated in DLD-1 and DKO-1 and HCT116 cell lines containing KRAS mutations, which indicated a widespread effect of mutant KRAS on circRNA abundance [88]. CircRNAs were more abundant in exosomes than in cells, which suggested that circRNAs may be promising biomarkers of CRC. Besides, CRC exosomes can induce morphological and functional changes in colonic mesenchymal stromal cells (MSCs), which may favor tumor growth and progression [89]. Exosomes can interact with target cells through specific receptor-ligand binding. Studies have shown that microvesicles from CRC released Fas ligand and tumor necrosis factor (TNF) to induce T-cell apoptosis, which can serve as a prognostic factor and can be targeted for novel antitumor therapies with regard to CRC [90].
As miRNAs may find applications in molecular therapies for the treatment of CRC [91], exosomes can be used as vectors for cancer intervention through gene or drug delivery. Dai et al. conducted a phase I clinical trial of the ascites-derived exosomes (Aex) in combination with the granulocyte-macrophage colony-stimulating factor (GM-CSF) in the immunotherapy of patients with CRC [92]. This treatment strategy could induce a beneficial, tumor-specific antitumor cytotoxic T lymphocyte (CTL) response, which may be an alternative choice in immunotherapy of advanced CRC [92]. Curiously, Zhao et al. provided evidence that cancer-associated fibroblast derived exosomes (CDEs) contain complete metabolites, including amino acids, lipids, and TCA-cycle intermediates. Cancer cells utilize central carbon metabolism of CDEs to promote tumor growth under nutrient deprivation or nutrient-stressed conditions, which may develop as a novel therapeutic concept for CRC [93]. Moreover, inhibiting the release of tumor exosomes or blocking the tumor exosome functions may also be a possible option to interfere with cancer. ExoDx™ Lung (ALK) from Exosome Diagnostics Company has been approved by the FDA in the United States and has become the world’s first noninvasive test using exosomal RNA-based liquid biopsy. As one of the promising diagnostic and therapeutic tools for CRC in future, research on the mechanism of exosomes will lay the foundation for a better clinical application.
CONCLUSIONS AND PERSPECTIVES
Tumorigenesis is the result of multiple factors implicated in the destruction of balance between oncogenes and anti-oncogenes. With approaches such as that of liquid biopsy, which are rapid, convenient, and minimally invasive, cancers can be detected from body fluid samples repeatedly, bypassing the need of tumor tissue biopsy while allowing clinicians to monitor the response to therapies and recurrence in real-time.
Although the early detection strategies and the guidance for decision-making during therapies based on liquid biopsy are promising, there have several drawbacks that need to be overcome before they can be applied clinically: 1) The surge in various detection technologies to identify CTCs, ctDNA, or exosomes, urgently calls for the implementation of standard guidelines, internal quality control (IQC) and external quality assessment (EQA). 2) Researchers need to find universal signatures from CTCs that cover any stage or type of cancer, thereby improving the sensitivity and specificity of the detection methods. 3) Large prospective clinical trials involving multicenter studies are needed to validate the clinical significance for detection and prognosis. 4) Complementary assessment of both CTCs and ctDNA will be more superior for the noninvasive diagnosis and prognosis monitoring of CRC. 5) There is an imposing need to explain the inconsistent results between liquid biopsy results and imaging examinations or tissue biopsies. Thus, liquid biopsies could help clinicians make better treatment-related decisions, provided that the above mentioned hurdles are overcome.
Fortunately, investigators have found that platelets might play a role in tumorigenesis and progression. Ratajczak and colleagues demonstrated that microparticles secreted by platelets induce angiogenesis and metastasis in both lung and breast cancer [16, 17]. Some researchers recently studied the total extracellular small RNA profiles from plasma, saliva, and urine of healthy subjects, which might contribute to the detection of health, disease, and injury [94]. Moreover, the occurrence of 8-Hydroxy-2’-deoxyguanosine (8-OHdG) and 5-Aminolevulinic Acid (5-ALA) or other such molecules in urine may potentially be set up as new screening or recurrence prognostic markers for CRC [95, 96, 97]. These could provide other promising directions for the detection of CRC.
In summary, owing to its significant benefits, liquid biopsy can be used as a feasible detection method for a variety of solid tumors and clinical diseases. Despite the existence of several challenges, we believe that the development of ideal detection methods is in progress. Our group has designed and conducted the first nationwide EQA of NGS-based targeted sequencing by laboratories in China in 2015 [98]. In addition, we are striving for the standardization of the detection of ctDNA by EQA. Undoubtedly, liquid biopsy will gain a foothold gradually and will be universally applied for clinical practice, after further optimization and improvement of technologies.
Abbreviations
Colorectal cancer (CRC); circulating tumor cells (CTCs); circulating tumor DNA (ctDNA); fecal occult blood test (FOBT); carcinoembryonic antigen (CEA); carbohydrate antigen 19-9 (CA19-9); Magnetic Resonance Imaging(MRI); Computed Tomography (CT); epithelial–mesenchymal transition (EMT); circulating tumor stem cells (CTSCs); Food and Drug Administration (FDA); epithelial cell adhesion molecule(EpCAM); reverse transcription polymerase chain reaction (RT-PCR); circulating free DNA (cfDNA); BEAMing (beads, emulsion, amplification and magnetics); digital PCR(dPCR); next generation sequencing (NGS); Epstein-Barr virus (EBV); nasopharyngeal carcinoma (NPC); methylated SEPT9 DNA (mSEPT9); exosomal DNA(exoDNA); allele-specific polymerase chain reaction (AS-PCR); metastatic breast cancer (MBC); progression-free survival (PFS); overall survival (OS); metastatic CRC (mCRC); cytokeratins (CKs); National Comprehensive Cancer Network (NCCN); epidermal growth factor receptor (EGFR); WNT inhibitory factor 1 (WIF1); neuropeptide Y(NPY); methylated ctDNA (MetctDNA); Ion Torrent PGM (IonPGM); microRNAs (miRNAs); comparative genomic hybridization (CGH); serum-purified exosomes (SPEs); endosomal sorting complex required for transport (ESCRT); hepatocyte growth factor-regulated tyrosine kinase substrate (HGS); neutral sphingomyelinase (nSMase); circular RNAs (circRNAs); mesenchymal stromal cells (MSCs); tumor necrosis factor (TNF); ascites-derived exosomes (Aex); granulocyte-macrophage colony-stimulating factor (GM-CSF); cytotoxic T lymphocyte (CTL); cancer-associated fibroblast derived exosomes (CDEs); internal quality control(IQC); external quality assessment(EQA); 8-Hydroxy-2’-deoxyguanosine (8-OHdG); 5-Aminolevulinic Acid (5-ALA).
ACKNOWLEDGMENTS
This work was supported by a grant from the Special Fund for Health Scientific Research in the Public Interest from National Population and Family Planning Commission of the People’s Republic China (No. 201402018). It was not supported by any private or public company or organization.
CONFLICTS OF INTEREST
The authors declare no conflict of interests.
REFERENCES
1. Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010; 127:2893-917.
2. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87-108.
3. Brenner H, Kloor M, Pox CP. Colorectal cancer. Lancet. 2014; 383:1490-502.
4. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015; 65:5-29.
5. Holohan C, Van Schaeybroeck S, Longley DB, Johnston PG. Cancer drug resistance: an evolving paradigm. Nat Rev Cancer. 2013; 13:714-26.
6. Beije N, Helmijr JC, Weerts MJ, Beaufort CM, Wiggin M, Marziali A, Verhoef C, Sleijfer S, Jansen MP, Martens JW. Somatic mutation detection using various targeted detection assays in paired samples of circulating tumor DNA, primary tumor and metastases from patients undergoing resection of colorectal liver metastases. Mol Oncol. 2016 Oct 10. doi: 10.1016/j.molonc.2016.10.001. [Epub ahead of print].
7. Diaz LA Jr, Williams RT, Wu J, Kinde I, Hecht JR, Berlin J, Allen B, Bozic I, Reiter JG, Nowak MA, Kinzler KW, Oliner KS, Vogelstein B. The molecular evolution of acquired resistance to targeted EGFR blockade in colorectal cancers. Nature. 2012; 486:537-40.
8. Simon K. Colorectal cancer development and advances in screening. Clin Interv Aging. 2016; 11:967-76.
9. Vukobrat-Bijedic Z, Husic-Selimovic A, Sofic A, Bijedic N, Bjelogrlic I, Gogov B, Mehmedovic A. Cancer Antigens (CEA and CA 19-9) as Markers of Advanced Stage of Colorectal Carcinoma. Med Arch. 2013; 67:397-401.
10. Sun L. Colonography by CT, MRI and PET/CT combined with conventional colonoscopy in colorectal cancer screening and staging. World J Gastroenterol. 2008; 14:853-63.
11. Ludwig JA, Weinstein JN. Biomarkers in cancer staging, prognosis and treatment selection. Nat Rev Cancer. 2005; 5:845-56.
12. Lohr JG, Adalsteinsson VA, Cibulskis K, Choudhury AD, Rosenberg M, Cruz-Gordillo P, Francis JM, Zhang CZ, Shalek AK, Satija R, Trombetta JJ, Lu D, Tallapragada N, et al. Whole-exome sequencing of circulating tumor cells provides a window into metastatic prostate cancer. Nat Biotechnol. 2014; 32:479-84.
13. Spellman PT, Gray JW. Detecting cancer by monitoring circulating tumor DNA. Nat Med. 2014; 20:474-5.
14. Miller MA, Oudin MJ, Sullivan RJ, Wang SJ, Meyer AS, Im H, Frederick DT, Tadros J, Griffith LG, Lee H, Weissleder R, Flaherty KT, Gertler FB, et al. Reduced Proteolytic Shedding of Receptor Tyrosine Kinases is a Post-Translational Mechanism of Kinase Inhibitor Resistance. Cancer Discov. 2016; 6:382-99.
15. Im H, Shao H, Park YI, Peterson VM, Castro CM, Weissleder R, Lee H. Label-free detection and molecular profiling of exosomes with a nano-plasmonic sensor. Nat Biotechnol. 2014; 32:490-5.
16. Janowska-Wieczorek A, Wysoczynski M, Kijowski J, Marquez-Curtis L, Machalinski B, Ratajczak J, Ratajczak MZ. Microvesicles derived from activated platelets induce metastasis and angiogenesis in lung cancer. Int J Cancer. 2005; 113:752-60.
17. Janowska-Wieczorek A, Marquez-Curtis LA, Wysoczynski M, Ratajczak MZ. Enhancing effect of platelet-derived microvesicles on the invasive potential of breast cancer cells. Transfusion. 2006; 46:1199-209.
18. Masuda T, Hayashi N, Iguchi T, Ito S, Eguchi H, Mimori K. Clinical and biological significance of circulating tumor cells in cancer. Mol Oncol. 2016; 10:408-17.
19. Cristofanilli M. Circulating tumour cells: telling the truth about metastasis. Lancet Oncol. 2014; 15:365-6.
20. Hardingham JE, Grover P, Winter M, Hewett PJ, Price TJ, Thierry B. Detection and Clinical Significance of Circulating Tumor Cells in Colorectal Cancer--20 Years of Progress. Mol Med. 2015; 21:S25-31.
21. Cohen SJ, Alpaugh RK, Gross S, O’Hara SM, Smirnov DA, Terstappen LW, Allard WJ, Bilbee M, Cheng JD, Hoffman JP, Lewis NL, Pellegrino A, Rogatko A, et al. Isolation and characterization of circulating tumor cells in patients with metastatic colorectal cancer. Clin Colorectal Canc. 2006; 6:125-32.
22. Cohen SJ, Punt CJ, Iannotti N, Saidman BH, Sabbath KD, Gabrail NY, Picus J, Morse M, Mitchell E, Miller MC, Doyle GV, Tissing H, Terstappen LW, et al. Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. J Clin Oncol. 2008; 26:3213-21.
23. Cohen SJ, Punt CJ, Iannotti N, Saidman BH, Sabbath KD, Gabrail NY, Picus J, Morse MA, Mitchell E, Miller MC, Doyle GV, Tissing H, Terstappen LW, et al. Prognostic significance of circulating tumor cells in patients with metastatic colorectal cancer. Ann oncol. 2009; 20:1223-9.
24. Miller MC, Doyle GV, Terstappen LW. Significance of Circulating Tumor Cells Detected by the CellSearch System in Patients with Metastatic Breast Colorectal and Prostate Cancer. J Oncol. 2010; 2010:617421.
25. Sastre J, Maestro ML, Gomez-Espana A, Rivera F, Valladares M, Massuti B, Benavides M, Gallen M, Marcuello E, Abad A, Arrivi A, Fernandez-Martos C, Gonzalez E, et al. Circulating tumor cell count is a prognostic factor in metastatic colorectal cancer patients receiving first-line chemotherapy plus bevacizumab: a Spanish Cooperative Group for the Treatment of Digestive Tumors study. Oncologist. 2012; 17:947-55.
26. Aceto N, Bardia A, Miyamoto DT, Donaldson MC, Wittner BS, Spencer JA, Yu M, Pely A, Engstrom A, Zhu H, Brannigan BW, Kapur R, Stott SL, et al. Circulating tumor cell clusters are oligoclonal precursors of breast cancer metastasis. Cell. 2014; 158:1110-22.
27. Sarioglu AF, Aceto N, Kojic N, Donaldson MC, Zeinali M, Hamza B, Engstrom A, Zhu H, Sundaresan TK, Miyamoto DT, Luo X, Bardia A, Wittner BS, et al. A microfluidic device for label-free, physical capture of circulating tumor cell clusters. Nat Methods. 2015; 12:685-91.
28. Kidess-Sigal E, Liu HE, Triboulet MM, Che J, Ramani VC, Visser BC, Poultsides GA, Longacre TA, Marziali A, Vysotskaia V, Wiggin M, Heirich K, Hanft V, et al. Enumeration and targeted analysis of KRAS, BRAF and PIK3CA mutations in CTCs captured by alabel-free platform: Comparison to ctDNA and tissue in metastatic colorectal cancer. Oncotarget. 2016; 7:85349-85364. doi: 10.18632/oncotarget.13350.
29. Harouaka R, Kang Z, Zheng SY, Cao L. Circulating tumor cells: advances in isolation and analysis, and challenges for clinical applications. Pharmacol Ther. 2014; 141:209-21.
30. Grover PK, Cummins AG, Price TJ, Roberts-Thomson IC, Hardingham JE. Circulating tumour cells: the evolving concept and the inadequacy of their enrichment by EpCAM-based methodology for basic and clinical cancer research. Ann Oncol. 2014; 25:1506-16.
31. Rao C, Chianese D, Doyle G, Miller M, Russell T, Sanders R, Terstappen L. Expression of epithelial cell adhesion molecule in carcinoma cells present in blood and primary and metastatic tumors. Int J Oncol. 2005; 27:49-57.
32. Gervasoni A, Sandri MT, Nascimbeni R, Zorzino L, Cassatella MC, Baglioni L, Panigara S, Gervasi M, Di Lorenzo D, Parolini O. Comparison of three distinct methods for the detection of circulating tumor cells in colorectal cancer patients. Oncol Rep. 2011; 25:1669-703.
33. Katsuno H, Zacharakis E, Aziz O, Rao C, Deeba S, Paraskeva P, Ziprin P, Athanasiou T, Darzi A. Does the presence of circulating tumor cells in the venous drainage of curative colorectal cancer resections determine prognosis? A meta-analysis. Ann Surg Oncol. 2008; 15:3083-91.
34. Iinuma H, Watanabe T, Mimori K, Adachi M, Hayashi N, Tamura J, Matsuda K, Fukushima R, Okinaga K, Sasako M, Mori M. Clinical significance of circulating tumor cells, including cancer stem-like cells, in peripheral blood for recurrence and prognosis in patients with Dukes’ stage B and C colorectal cancer. J Clin Oncol. 2011; 29:1547-55.
35. Jahr S, Hentze H, Englisch S, Hardt D, Fackelmayer FO, Hesch RD, Knippers R. DNA Fragments in the Blood Plasma of Cancer Patients: Quantitations and Evidence for Their Origin from Apoptotic and Necrotic Cells. Cancer Res. 2001; 61:1659-65.
36. Underhill HR, Kitzman JO, Hellwig S, Welker NC, Daza R, Baker DN, Gligorich KM, Rostomily RC, Bronner MP, Shendure J. Fragment Length of Circulating Tumor DNA. PLoS Genet. 2016; 12:e1006162.
37. Heitzer E, Ulz P, Geigl JB. Circulating tumor DNA as a liquid biopsy for cancer. Clin Chem. 2015; 61:112-23.
38. Lo YM, Zhang J, Leung TN, Lau TK, Chang AM, Hjelm NM. Rapid clearance of fetal DNA from maternal plasma. Am J Hum Genet. 1999; 64:218-24.
39. Diehl F, Schmidt K, Choti MA, Romans K, Goodman S, Li M, Thornton K, Agrawal N, Sokoll L, Szabo SA, Kinzler KW, Vogelstein B, Diaz LA Jr. Circulating mutant DNA to assess tumor dynamics. Nat Med. 2008; 14:985-90.
40. Diehl F, Li M, Dressman D, He Y, Shen D, Szabo S, Diaz LA Jr, Goodman SN, David KA, Juhl H, Kinzler KW, Vogelstein B. Detection and quantification of mutations in the plasma of patients with colorectal tumors. Proc Natl Acad Sci U S A. 2005; 102:16368-73.
41. Sozzi G, Conte D, Leon M, Ciricione R, Roz L, Ratcliffe C, Roz E, Cirenei N, Bellomi M, Pelosi G, Pierotti MA, Pastorino U. Quantification of free circulating DNA as a diagnostic marker in lung cancer. J Clin Oncol. 2003; 21:3902-8.
42. Frattini M, Gallino G, Signoroni S, Balestra D, Lusa L, Battaglia L, Sozzi G, Bertario L, Leo E, Pilotti S, Pierotti MA. Quantitative and qualitative characterization of plasma DNA identifies primary and recurrent colorectal cancer. Cancer Lett. 2008; 263:170-81.
43. To EW, Chan KC, Leung SF, Chan LY, To KF, Chan AT, Johnson PJ, Lo YM. Rapid Clearance of Plasma Epstein-Barr Virus DNA After Surgical Treatment of Nasopharyngeal Carcinoma. Clin Cancer Res. 2003; 9:3254-9.
44. Chan KC, Hung EC, Woo JK, Chan PK, Leung SF, Lai FP, Cheng AS, Yeung SW, Chan YW, Tsui TK, Kwok JS, King AD, Chan AT, et al. Early detection of nasopharyngeal carcinoma by plasma Epstein-Barr virus DNA analysis in a surveillance program. Cancer. 2013; 119:1838-44.
45. Church TR, Wandell M, Lofton-Day C, Mongin SJ, Burger M, Payne SR, Castanos-Velez E, Blumenstein BA, Rosch T, Osborn N, Snover D, Day RW, Ransohoff DF, et al. Prospective evaluation of methylated SEPT9 in plasma for detection of asymptomatic colorectal cancer. Gut. 2014; 63:317-25.
46. Bettegowda C, Sausen M, Leary RJ, Kinde I, Wang Y, Agrawal N, Bartlett BR, Wang H, Luber B, Alani RM, Antonarakis ES, Azad NS, Bardelli A, et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci Transl Med. 2014; 6:224ra224.
47. Siravegna G, Marsoni S, Siena S, Bardelli A. Integrating liquid biopsies into the management of cancer. Nat Rev Clin Oncol. 2017 Mar 2. doi: 10.1038/nrclinonc.2017.14. [Epub ahead of print].
48. Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013; 200:373-83.
49. Mathivanan S, Lim JW, Tauro BJ, Ji H, Moritz RL, Simpson RJ. Proteomics Analysis of A33 Immunoaffinity-purified Exosomes Released from the Human Colon Tumor Cell Line LIM1215 Reveals a Tissue-specific Protein Signature. Mol Cell Proteomics. 2010; 9:197-208.
50. He M, Zeng Y. Microfluidic Exosome Analysis toward Liquid Biopsy for Cancer. J Lab Autom. 2016; 21:599-608.
51. Peinado H, Alečković M, Lavotshkin S, Matei I, Costa-Silva B, Moreno-Bueno G, Hergueta-Redondo M, Williams C, García-Santos G, Ghajar CM, Nitadori-Hoshino A, Hoffman C, Badal K, et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat Med. 2012; 18:883-91.
52. Hoshino A, Costa-Silva B, Shen TL, Rodrigues G, Hashimoto A, Tesic Mark M, Molina H, Kohsaka S, Di Giannatale A, Ceder S, Singh S, Williams C, Soplop N, et al. Tumour exosome integrins determine organotropic metastasis. Nature. 2015; 527:329-35.
53. Ratajczak J, Wysoczynski M, Hayek F, Janowska-Wieczorek A, Ratajczak MZ. Membrane-derived microvesicles: important and underappreciated mediators of cell-to-cell communication. Leukemia. 2006; 20:1487-95.
54. Thakur BK, Zhang H, Becker A, Matei I, Huang Y, Costa-Silva B, Zheng Y, Hoshino A, Brazier H, Xiang J, Williams C, Rodriguez-Barrueco R, Silva JM, et al. Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell Res. 2014; 24:766-9.
55. Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Miller MC, Reuben JM, Doyle GV, Allard WJ, Terstappen LM, Hayes DF. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med. 2004; 351:781-91.
56. Bidard FC, Peeters DJ, Fehm T, Nolé F, Gisbert-Criado R, Mavroudis D, Grisanti S, Generali D, Garcia-Saenz JA, Stebbing J, Caldas C, Gazzaniga P, Manso L, et al. Clinical validity of circulating tumour cells in patients with metastatic breast cancer: a pooled analysis of individual patient data. Lancet Oncol. 2014; 15:406-14.
57. Alix-Panabières C, Bartkowiak K, Pantel K. Functional studies on circulating and disseminated tumor cells in carcinoma patients. Mol Oncol. 2016; 10:443-9.
58. Deneve E, Riethdorf S, Ramos J, Nocca D, Coffy A, Daures JP, Maudelonde T, Fabre JM, Pantel K, Alix-Panabieres C. Capture of Viable Circulating Tumor Cells in the Liver of Colorectal Cancer Patients. Clin Chem. 2013; 59:1384-92.
59. Ligthart ST, Coumans FA, Bidard FC, Simkens LH, Punt CJ, de Groot MR, Attard G, de Bono JS, Perga JY, Terstappen LW. Circulating Tumor Cells Count and Morphological Features in Breast, Colorectal and Prostate Cancer. PLoS One. 2013; 8:e67148.
60. Bork U, Rahbari NN, Scholch S, Reissfelder C, Kahlert C, Buchler MW, Weitz J, Koch M. Circulating tumour cells and outcome in non-metastatic colorectal cancer: a prospective study. Br J Cancer. 2015; 112:1306-13.
61. Zhao R, Cai Z, Li S, Cheng Y, Gao H, Liu F, Wu S, Liu S, Dong Y, Zheng L, Zhang W, Wu X, Yao X. Expression and clinical relevance of epithelial and mesenchymal markers in circulating tumor cells from colorectal cancer. Oncotarget. 2017; 8:9293-9302. doi: 10.18632/oncotarget.14065.
62. Cayrefourcq L, Mazard T, Joosse S, Solassol J, Ramos J, Assenat E, Schumacher U, Costes V, Maudelonde T, Pantel K, Alix-Panabieres C. Establishment and characterization of a cell line from human circulating colon cancer cells. Cancer Res. 2015; 75:892-901.
63. Grillet F, Bayet E, Villeronce O, Zappia L, Lagerqvist EL, Lunke S, Charafe-Jauffret E, Pham K, Molck C, Rolland N, Bourgaux JF, Prudhomme M, Philippe C, et al. Circulating tumour cells from patients with colorectal cancer have cancer stem cell hallmarks in ex vivo culture. Gut. 2016 Jul 25. doi: 10.1136/gutjnl-2016-311447. [Epub ahead of print].
64. Karapetis CS, Khambata-Ford S, Jonker DJ, O’Callaghan CJ, Tu D, Tebbutt NC, Simes RJ, Chalchal H, Shapiro JD, Robitaille S, Price TJ, Shepherd L, Au HJ, et al. K-ras Mutations and Benefit from Cetuximab in Advanced Colorectal Cancer. N Engl J Med. 2008; 359:1757-65.
65. Kosmidou V, Oikonomou E, Vlassi M, Avlonitis S, Katseli A, Tsipras I, Mourtzoukou D, Kontogeorgos G, Zografos G, Pintzas A. Tumor heterogeneity revealed by KRAS, BRAF, and PIK3CA pyrosequencing: KRAS and PIK3CA intratumor mutation profile differences and their therapeutic implications. Hum Mutat. 2014; 35:329-40.
66. Fabbri F, Carloni S, Zoli W, Ulivi P, Gallerani G, Fici P, Chiadini E, Passardi A, Frassineti GL, Ragazzini A, Amadori D. Detection and recovery of circulating colon cancer cells using a dielectrophoresis-based device: KRAS mutation status in pure CTCs. Cancer Lett. 2013; 335:225-31.
67. Gazzaniga P, Gianni W, Raimondi C, Gradilone A, Lo Russo G, Longo F, Gandini O, Tomao S, Frati L. Circulating tumor cells in high-risk nonmetastatic colorectal cancer. Tumour Biol. 2013; 34:2507-9.
68. Tie J, Kinde I, Wang Y, Wong HL, Roebert J, Christie M, Tacey M, Wong R, Singh M, Karapetis CS, Desai J, Tran B, Strausberg RL, et al. Circulating tumor DNA as an early marker of therapeutic response in patients with metastatic colorectal cancer. Ann Oncol. 2015; 26:1715-22.
69. Tie J, Wang Y, Tomasetti C, Li L, Springer S, Kinde I, Silliman N, Tacey M, Wong HL, Christie M, Kosmider S, Skinner I, Wong R, et al. Circulating tumor DNA analysis detects minimal residual disease and predicts recurrence in patients with stage II colon cancer. Sci Transl Med. 2016; 8:346ra92.
70. Garrigou S, Perkins G, Garlan F, Normand C, Didelot A, Le Corre D, Peyvandi S, Mulot C, Niarra R, Aucouturier P, Chatellier G, Nizard P, Perez-Toralla K, et al. A Study of Hypermethylated Circulating Tumor DNA as a Universal Colorectal Cancer Biomarker. Clin Chem. 2016; 62:1129-39.
71. Bedin C, Enzo MV, Del Bianco P, Pucciarelli S, Nitti D, Agostini M. Diagnostic and prognostic role of cell-free DNA testing for colorectal cancer patients. Int J Cancer. 2017; 140:1888-1898.
72. Misale S, Yaeger R, Hobor S, Scala E, Janakiraman M, Liska D, Valtorta E, Schiavo R, Buscarino M, Siravegna G, Bencardino K, Cercek A, Chen CT, et al. Emergence of KRAS mutations and acquired resistance to anti-EGFR therapy in colorectal cancer. Nature. 2012; 486:532-6.
73. Thierry AR, Mouliere F, El Messaoudi S, Mollevi C, Lopez-Crapez E, Rolet F, Gillet B, Gongora C, Dechelotte P, Robert B, Del Rio M, Lamy PJ, Bibeau F, et al. Clinical validation of the detection of KRAS and BRAF mutations from circulating tumor DNA. Nat Med. 2014; 20:430-5.
74. Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA, Kinzler KW. Cancer Genome Landscapes. Science. 2013; 339:1546-58.
75. Siravegna G, Mussolin B, Buscarino M, Corti G, Cassingena A, Crisafulli G, Ponzetti A, Cremolini C, Amatu A, Lauricella C, Lamba S, Hobor S, Avallone A, et al. Clonal evolution and resistance to EGFR blockade in the blood of colorectal cancer patients. Nat Med. 2015; 21:795-801.
76. Spindler KL, Pallisgaard N, Andersen RF, Jakobsen A. Changes in mutational status during third-line treatment for metastatic colorectal cancer--results of consecutive measurement of cell free DNA, KRAS and BRAF in the plasma. Int J Cancer. 2014; 135:2215-22.
77. Murtaza M, Dawson SJ, Tsui DW, Gale D, Forshew T, Piskorz AM, Parkinson C, Chin SF, Kingsbury Z, Wong AS, Marass F, Humphray S, Hadfield J, et al. Non-invasive analysis of acquired resistance to cancer therapy by sequencing of plasma DNA. Nature. 2013; 497:108-12.
78. Jain M, Olsen HE, Paten B, Akeson M. The Oxford Nanopore MinION: delivery of nanopore sequencing to the genomics community. Genome Biol. 2016; 17:239.
79. Branton D, Deamer DW, Marziali A, Bayley H, Benner SA, Butler T, Di Ventra M, Garaj S, Hibbs A, Huang X, Jovanovich SB, Krstic PS, Lindsay S, et al. The potential and challenges of nanopore sequencing. Nat Biotechnol. 2008; 26:1146-53.
80. Ogata-Kawata H, Izumiya M, Kurioka D, Honma Y, Yamada Y, Furuta K, Gunji T, Ohta H, Okamoto H, Sonoda H,Watanabe M, Nakagama H, Yokota J, et al. Circulating Exosomal microRNAs as Biomarkers of Colon Cancer. PLoS ONE. 2014; 9:e92921.
81. Matsumura T, Sugimachi K, Iinuma H, Takahashi Y, Kurashige J, Sawada G, Ueda M, Uchi R, Ueo H, Takano Y, Shinden Y, Eguchi H, Yamamoto H, et al. Exosomal microRNA in serum is a novel biomarker of recurrence in human colorectal cancer. Br J Cancer. 2015; 113:275-81.
82. Chen Y, Xie Y, Xu L, Zhan S, Xiao Y, Gao Y, Wu B, Ge W. Protein content and functional characteristics of serum-purified exosomes from patients with colorectal cancer revealed by quantitative proteomics. Int J Cancer. 2017; 140:900-913.
83. Sun Y, Zheng W, Guo Z, Ju Q, Zhu L, Gao J, Zhou L, Liu F, Xu Y, Zhan Q, Zhou Z, Sun W, Zhao X. A novel TP53 pathway influences the HGS-mediated exosome formation in colorectal cancer. Sci Rep. 2016; 6:28083.
84. Cha DJ, Franklin JL, Dou Y, Liu Q, Higginbotham JN, Demory Beckler M, Weaver AM, Vickers K, Prasad N, Levy S, Zhang B, Coffey RJ, Patton JG. KRAS-dependent sorting of miRNA to exosomes. Elife. 2015; 4:e07197.
85. Wang X, Ding X, Nan L, Wang Y, Wang J, Yan Z, Zhang W, Sun J, Zhu W, Ni B, Dong S, Yu L. Investigation of the roles of exosomes in colorectal cancer liver metastasis. Oncol Rep. 2015; 33:2445-53.
86. Higginbotham JN, Demory Beckler M, Gephart JD, Franklin JL, Bogatcheva G, Kremers GJ, Piston DW, Ayers GD, McConnell RE, Tyska MJ, Coffey RJ. Amphiregulin exosomes increase cancer cell invasion. Curr Biol. 2011; 21:779-86.
87. Demory Beckler M, Higginbotham JN, Franklin JL, Ham AJ, Halvey PJ, Imasuen IE, Whitwell C, Li M, Liebler DC, Coffey RJ. Proteomic Analysis of Exosomes from Mutant KRAS Colon Cancer Cells Identifies Intercellular Transfer of Mutant KRAS. Mol Cell Proteomics. 2013; 12:343-55.
88. Dou Y, Cha DJ, Franklin JL, Higginbotham JN, Jeppesen DK, Weaver AM, Prasad N, Levy S, Coffey RJ, Patton JG, Zhang B. Circular RNAs are down-regulated in KRAS mutant colon cancer cells and can be transferred to exosomes. Sci Rep. 2016; 6:37982.
89. Lugini L, Valtieri M, Federici C, Cecchetti S, Meschini S, Condello M, Signore M, Fais S. Exosomes from human colorectal cancer induce a tumor-like behavior in colonic mesenchymal stromal cells. Oncotarget. 2016; 7:50086-50098. doi: 10.18632/oncotarget.10574.
90. Huber V, Fais S, Iero M, Lugini L, Canese P, Squarcina P, Zaccheddu A, Colone M, Arancia G, Gentile M, Seregni E, Valenti R, Ballabio G, et al. Human Colorectal Cancer Cells Induce T-Cell Death Through Release of Proapoptotic Microvesicles: Role in Immune Escape. Gastroenterology. 2005; 128:1796-804.
91. Li Z, Rana TM. Therapeutic targeting of microRNAs: current status and future challenges. Nat Rev Drug Discov. 2014; 13:622-38.
92. Dai S, Wei D, Wu Z, Zhou X, Wei X, Huang H, Li G. Phase I Clinical Trial of Autologous Ascites-derived Exosomes Combined With GM-CSF for Colorectal Cancer. Mol Ther. 2008; 16:782-90.
93. Zhao H, Yang L, Baddour J, Achreja A, Bernard V, Moss T, Marini JC, Tudawe T, Seviour EG, San Lucas FA, Alvarez H, Gupta S, Maiti SN, et al. Tumor microenvironment derived exosomes pleiotropically modulate cancer cell metabolism. Elife. 2016; 5:e10250.
94. Yeri A, Courtright A, Reiman R, Carlson E, Beecroft T, Janss A, Siniard A, Richholt R, Balak C, Rozowsky J, Kitchen R, Hutchins E, Winarta J, et al. Total Extracellular Small RNA Profiles from Plasma, Saliva, and Urine of Healthy Subjects. Sci Rep. 2017; 7:44061.
95. Feng B, Yue F, Zheng MH. Urinary Markers in Colorectal Cancer. Adv Clin Chem. 2009; 47:45-57.
96. Guo C, Li X, Wang R, Yu J, Ye M, Mao L, Zhang S, Zheng S. Association between Oxidative DNA Damage and Risk of Colorectal Cancer: Sensitive Determination of Urinary 8-Hydroxy-2’-deoxyguanosine by UPLC-MS/MS Analysis. Sci Rep. 2016; 6:32581.
97. Kamada Y, Murayama Y, Ota U, Takahashi K, Arita T, Kosuga T, Konishi H, Morimura R, Komatsu S, Shiozaki A, Kuriu Y, Ikoma H, Nakanishi M, et al. Urinary 5-Aminolevulinic Acid Concentrations as a Potential Tumor Marker for Colorectal Cancer Screening and Recurrence. Anticancer Res. 2016; 36:2445-50.
98. Zhang R, Ding J, Han Y, Yi L, Xie J, Yang X, Fan G, Wang G, Hao M, Zhang D, Zhang K, Lin G, Li J. The reliable assurance of detecting somatic mutations in cancer-related genes by next-generation sequencing: the results of external quality assessment in China. Oncotarget. 2016; 7:58500-58515. doi: 10.18632/oncotarget.11306.

