INTRODUCTION
Non-small-cell lung cancer (NSCLC) is common and accounts for up to 85% of lung cancers [1]. Most patients with NSCLC are diagnosed at an advanced stage, which means that many of these patients lose the opportunity for definitive surgical resection, for which the 5-year survival rate is < 15% [2]. Despite considerable progress in treatment that has been achieved in the last several decades, advanced NSCLC still remains a challenging malignant tumor that is unable to be cured in the majority of patients [3].
Docetaxel (Taxotere), a semi-synthetic taxoid derived from the rare pacific yew tree Taxus Baccata, has demonstrated significant antitumor activity. It is one of the most active single agents in both previously untreated patients and in those who have relapsed or progressed following cisplatin-based chemotherapy [4, 5]. Docetaxel was defined as a new chemotherapy agent according to the American Society of Clinical Oncology (ASCO)[6]. The stabilization of microtubules by docetaxel results in the inhibition of mitotic cell division between metaphase and anaphase, which leads to the initiation of apoptosis. Previous research has shown that single-agent docetaxel at doses of 60, 75 or 100 mg/m2 administered once every 3 weeks could lead to objective response rates of approximately 30% in untreated patients with advanced NSCLC [7, 8].
Cisplatin-based doublets are recommended for the adjuvant or neoadjuvant treatment of potentially operable NSCLC and as a first-line therapy for advanced or metastatic NSCLC [9]. Extensive clinical phase II trials in the first-line setting recorded response rates of 32% to 52% and a survival (median, 8 to 12 months) of 33% to 48% [10, 11]. Kubota et al reported that the docetaxel plus cisplatin (DP) regimen was associated with marked improvements in overall survival rates and in quality of life (QOL), compared with the vindesine plus cisplatin regimen. The use of the DP regimen resulted in greater clinical benefits in patients with previously untreated stage IV NSCLC [12]. In addition, the DP regimen was reported to be an effective and well-tolerated regimen in chemo-naive patients with advanced NSCLC [13, 14]. However, for elderly patients or patients with reduced performance status, cisplatin-based protocols are often too toxic and should only be used with caution [15]. Aging is associated with deterioration of renal and liver function, decreased bone marrow reserves and the presence of comorbid illnesses. Moreover, docetaxel monotherapy was reported to be not inferior to DP, with less toxicity and better tolerability in patients with advanced NSCLC [16, 17].
Several RCTs(randomized clinical trials) were performed to evaluate the activity and toxicity of the DP combination as a first-line treatment of chemotherapy-naive patients with metastatic or unresectable locally advanced NSCLC [18–20]. However, the results varied considerably, and the toxic effects of combination therapy such as grade 3-4 neutropenia, myelosuppression, nausea and vomiting were more common after this therapy compared with others. In addition, published studies that have compared platinum-based combinations with the corresponding non-platinum monotherapies demonstrated a higher response rate and higher overall survival rates in the combination arms [21]. The main arguments against the use of chemotherapy in NSCLC are the marginal (if any) improvements in survival and response as well as the occurrence of severe and even unacceptable toxicity profiles.
Accordingly, in this paper, we conducted a meta-analysis to compare the clinical profile of docetaxel monotherapy with that of DP combination chemotherapy in terms of response rate, overall survival and toxicity in patients with NSCLC.
METHODS
Literature search
The following electronic databases were independently and extensively searched by two investigators from their inception through May 2016: EMBASE, Medline, the Cochrane Library, Web of Science, the China National Knowledge Internet (CNKI), and the Wan-fang database. The search strategy was based on a combination of three concepts adjusted to each database, when necessary. Concept one included all of the terms for docetaxel, and Concept two included the terms for cisplatin; Concept three included all of the terms for non-small-cell lung cancer. We only accepted one set of data on the same topic. The bibliographies of the included studies and dissertations were also searched for additional publications. All of the eligible studies were identified by two independent authors (AL, ZJW), and any disagreements were settled by consensus or consultation with a third author (HD).
Study selection
In order to be included in this analysis, the trials had to fulfill the following inclusion criteria: 1) contain patients with histologically proven NSCLC; 2) previous surgery was allowed if it had been completed at least 4 weeks before inclusion; 3) prior radiotherapy, except for that intended for the primary lesion, was permitted if it had been completed at least 2 weeks before inclusion; 3) randomized controlled clinical trials; 4) docetaxel monotherapy and DP doublet regimens were compared as chemotherapy regimens without the addition of confounding agents or interventions; 5) both groups could receive some foundation therapy or supportive care such as granulocyte colony-stimulating factor (G-CSF), antiemetic treatment with ondansetron and dexamethasone. The exclusion criteria were as follows: 1) an evaluation of the activity, efficacy and toxicity of docetaxel versus DP was absent in the studies; 2) agents were used as pre- or post-operative adjuvant chemotherapy; 3) the study featured comparisons of other types of chemotherapy regimens; 4) Early studies published as a series of articles from the same institution or author that contained significant overlapping data were excluded for fear of multiple publication bias; 5) case reports, editorials, experimental studies, conference articles and other studies that failed to provide detailed results were excluded.
Data extraction
After duplicates were removed and the study selection process was completed, the titles and abstracts were scanned by two independent investigators (AL, ZJW) according to predefined selection criteria, and potentially relevant RCTs were selected. The relevant data were extracted by adopting a predetermined standardized procedure, which involved the first authors, year of publication, country, demographic characteristics of the participants, and the treatment regimen for each group. All data were verified for internal consistency, and controversies were settled by consensus or discussion with a third author. Whenever possible, the first authors were contacted to obtain and clarify the relevant data, when appropriate, as specified by the standardized protocol.
Quality assessment
The methodological quality of the included trials was assessed by the Cochrane Collaboration’s tool [22]. This tool focuses on the internal validity of the trial and on the assessment of the risk of possible bias in different phases of the trial. The following items were assessed: random sequence generation, allocation concealment, blinding of participants, personnel and outcome assessment, incomplete outcome measures, selective outcome reporting and other types of bias. Each item was classified according to the risk of bias; high, low, and unclear risk are represented as High (H), Low (L) and Unclear (U), respectively. All of the eligible studies were identified by two independent authors (AL, ZJW), and any disagreements were settled by consensus or discussion with a third author (HST).
Outcome assessment
The overall survival (OS) was defined as the time that elapsed from random assignment until death from any cause and was censored at the last follow-up date. Response evaluation was performed according to the Response Evaluation Criteria In Solid Tumors [23]. The overall response rate was defined as the sum of the partial response rate (PR) and the complete response rate (CR). One-year survival rates and overall response rates (CR plus PR) were primary outcomes in the meta-analysis. Symptom scores of quality of life (QOL) were evaluated according to the Functional Assessment of Cancer Therapy-Lung (FACT-L), which consists of a seven-item disease-specific subscale [24]. Toxicity profiles were reported according to the World Health Organization (WHO) criteria or the Cooperative Group Common Toxicity Criteria [25]. According to the National Cancer Institute Common Terminology Criteria (NCI-CTC) for Adverse Events (version 3.0), grade 3 or 4 toxic effects included anemia, leucopenia, neutropenia, febrile neutropenia, thrombocytopenia, nausea/vomiting, neurotoxicity, nephrotoxicity, diarrhea, and toxic death. QOL and grade 3 or 4 toxic profiles were secondary outcomes in the meta-analysis.
Statistical analysis
The Cochrane Collaboration’s Review Manager Software (RevMan Version 5.2, The Cochrane Collaboration, Copenhagen, 2014) was used to perform the meta-analyses. The overall effect size of each regimen was calculated as a weighted average of the inverse variance and 95 % confidence intervals (95 % CI) for study-specific estimates. The statistical heterogeneity among the individual studies was evaluated based on Cochrane’s Q test and the I2 index, which express, as a percentage, the proportion of variability of the results due to heterogeneity as opposed to sampling error [26]. The presence of considerable heterogeneity was confirmed if I2 was > 75 % and if P < 0.10 [27]. A variance-based fixed effect model was applied to calculate the pooled effect. Where considerable heterogeneity was reported, the summary effects of the regimens were pooled using a random-effects model [28]. If appropriate, the heterogeneity was identified and explained using a subgroup analysis or sensitivity analysis [27]. A P-value less than 0.05 was considered significant for all statistical tests.
RESULTS
Search results
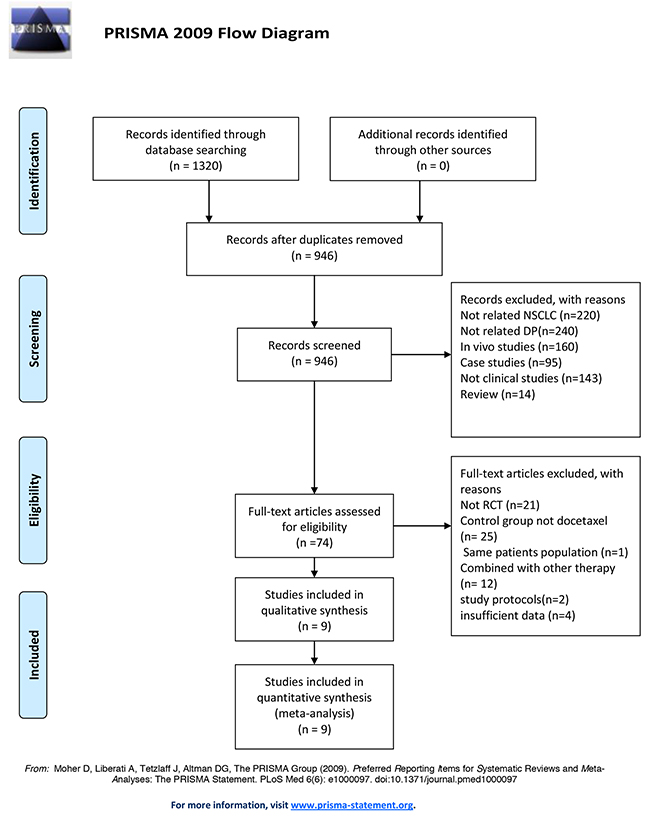
Table 1 contains a flowchart that describes the process by which we screened and selected trials. A total of 1320 relevant reports were initially retrieved from the electronic databases. After the removal of duplicates and after the titles and abstracts were screened, 74 publications met the inclusion criteria, and the full text for all 74 articles was available. Among these articles, 21 articles were excluded because they contained studies that were not RCTs; 12 articles were excluded because the included patients were not treated with a single chemotherapeutic agent, and intervention was preoperative or postoperative adjuvant chemotherapy combined with surgery; 25 articles were excluded because of inappropriate treatment comparisons; 2 articles were excluded because they were study protocols; and 1 article was excluded because it contained the same patient population as another study; 4 articles were excluded because of insufficient information for the current meta-analysis. In addition, a manual search of relevant references did not identify additional studies. Consequently, 9 trials that included patients with advanced NSCLC were ultimately eligible for inclusion in this meta-analysis.
Table 1: Flow diagram of the studies
Characteristics of the trials
Detailed baseline characteristics of the patients included in the nine trials are listed in Table 2. In all, 1257 patients were randomized to receive the docetaxel monotherapy regimen (623 patients) or the DP combination doublet regimen (634 patients). Two trials [29, 30] were performed in Japan, one trial [16] was performed in Greece, and six trials [31–36] were performed in China. Two trials [16, 30] were randomized phase III trials. Five studies reported the permission of the ethics and scientific committees of the participating centers [16, 29–31, 33]. Two trials were registered in the Japan Clinical Oncology Group (JCOG, the former name of the West Japan Oncology Group [WJOG]) Data Center [29, 30]. One trial was conducted by the Lung Cancer Working Group of the Hellenic Oncology Research Group and was a prospective and multicenter trial [16]. None were placebo-controlled, double-blind trials. The patients in five trials [16, 29–31, 33] provided written informed consent before they underwent any study-related procedure. Survival data were well reported and were available in four studies [16, 29, 30, 34]. Response to therapy was reported in all nine studies. QOL assessment and symptom scores were evaluated in three trials [16, 29, 30]. The appropriate sample capacity was calculated before the trials were conducted in two studies [29, 30]. G-CSF support for the DP combination group was given in one trial [16]. Standard antiemetic treatment together with standard pre- and post-medication with dexamethasone or a 5-HT3 antagonist was given to patients in both groups in seven trials in cases of allergic reactions and docetaxel-associated fluid retention syndrome [16, 29, 31, 33–36]. The patients in the studies were middle-aged and elderly. The number of treatment cycles ranged from 1 to 18 in the docetaxel arm and from 1 to 9 in the DP arm. Patient baseline assessments, which consist of complete medical history and physical examination, were performed before the initiation of therapy, and measurable lesions were monitored throughout the trials [16, 29, 30, 33]. Three trials reported major reasons for dose reductions, treatment discontinuation or treatment termination [16, 29, 30]. Additional data were collected on the epidermal growth factor receptor (EGFR) mutation status (exon 19 deletion or L858R point mutation) and post-study treatments; an ad hoc analysis was performed in one study [30].
Table 2: Summary of characteristics in studies included
Study(year) |
country |
Demographics |
Intervention |
|||||
|---|---|---|---|---|---|---|---|---|
Number |
Age(years) |
Male/Female |
ECOG PS |
stage |
D group |
DP group |
||
Tsukada (2015) |
Japan |
D: 63 |
D: 76(70-88) |
D:49/14 |
0-1 |
IIIA/IIIB,IV |
One cycle: D 25 mg/m2 infused over 60 min on days 1, 8 and 15; repeated every 4 weeks |
One cycle: D 20 mg/m2 infused over 60 min plus P 25 mg/m2 infused over 15–20 min on days 1, 8 and 15. repeated every 4 weeks |
Georgoulias (2004) |
Greece |
D:152 |
D: 63(41-77) |
D:137/15 |
0-2 |
IIIB, IV |
One cycle: D 100 mg/m2 in a 1-hour intravenous infusion; repeated every 3 weeks |
One cycle: D 100 mg/m2 over a 1-hour intravenous infusion on day 1 and P 80 mg/m2 on day 2, G-CSF 150 µg/m2 subcutaneously from days 3 to 9 |
Abe (2015) |
Japan |
D:137 |
D: 76(70-87) |
D:95/42 |
0-1 |
III, IV |
One cycle: D 60 mg/m2 infused over 60 minutes on day 1;repeated every 3 weeks |
One cycle: D 20mg/m2 infused over 60 minutes plus P 25mg/m2 infused over 15 to 20 minutes on days 1, 8, and 15 every 4 weeks |
Guo (2015) |
China |
D: 45 |
D: 64.9(63-75) |
D:28/17 |
Nr |
Nr |
One cycle: D 35 mg/m2 infused over 60 min on days 1, 8 and 15; repeated every 4 weeks |
One cycle: D 35 mg/m2 infused over 60 min on days 1, 8 and 15 plus P 40mg infused over 60 min, on days 1, 2 and 3; repeated every 4 weeks |
Liu (2010) |
China |
D: 40 |
Nr |
Nr |
Nr |
III, IV, |
One cycle: D 70 mg/m2 infused over 60 min on days 1, 8; repeated every 3 weeks |
One cycle: D 35 mg/m2 on days 1, 8 plus P 25 mg/m2, on days 1, 2 and 3; repeated every 3 weeks |
Zeng* (2013) |
China |
D: 42 |
D: 54.8±5.4 |
D:22/20 |
Nr |
IIb, III, IV |
One cycle: D 75 mg/m2 on day 1; repeated every 3 weeks |
One cycle: D 75 mg/m2 on day 1 plus P 75 mg/m2, on day 1; repeated every 3 weeks |
Zhang* (2015) |
China |
D: 68 |
D: 63.6±10.5 DP: 62.1 ±11.2 |
D:35 /33 |
Nr |
III, IV, |
One cycle: D 75 mg/m2 on days 1, 8; repeated every 4 weeks |
One cycle: D 75 mg/m2 on day 1 plus P 20mg, on days from 1 to 5; repeated every 4 weeks |
Jing (2014) |
China |
D: 36 |
Nr |
D:24 /12 |
Nr |
IIb, III, IV |
One cycle: D 75-85 mg/m2 infused over 60 min on day 1; repeated every 3 weeks |
One cycle: D 75 mg/m2 on day 1 plus P 25 mg/m2, on days from 1 to 3; repeated every 3 weeks |
Wang (2011) |
China |
D: 40 |
D: 58 (37-70) |
D:31 /9 |
0-2 |
IIIB, IV |
One cycle: D 75 mg/m2 on day 1; repeated every 3weeks |
One cycle: D 75 mg/m2 on day 1 plus P 80 mg/m2 divided into 3 days; repeated every 3 weeks |
Abbreviations: D, docetaxel; DP, docetaxel/cisplatin; G-CSF, granulocyte colony-stimulating factor; ECOG PS, Eastern Cooperative Oncology Group performance status. Age was presented as mdian (range).
*Age was presented as mean ± SD.
Risk of bias assessment
According to the Cochrane Collaboration recommendation, randomization methods were reported in four trials [16, 30–32]. Randomization to each arm was accomplished by stratification according to age, PS, and stage of the disease [16]; institution, disease stage (III v IV or recurrence), and age [30]. The baseline characteristics of the patients were generally well balanced between the treatment arms in each trial. Otherwise, allocation concealments and comprehensive methodological processes, as well as the blinding of participants and personnel, were not reported. Sufficient details of withdrawals and dropouts were described in all 4 studies. Two studies used the intention-to-treat approach in the handling of data [16, 30]. In the majority of the studies, whether enrollment of the participants was actually consecutive or not was unclear, and a selection bias could be completely excluded. The details of the risk of bias are illustrated in Supplementary Figure 1.
Response
Data on the objective response rates were available in all nine trials. The meta-analysis demonstrated that the combination chemotherapy regimens of DP had a higher overall response rate (CR plus PR) compared with the docetaxel regimen (RR = 0.70; 95 % CI, 0.61 to 0.80; P < 0.00001). Additionally, no considerable heterogeneity was found (χ2 = 12.73, I2 = 37 %, P = 0.12) (Figure 1). In the case of G-CSF, a sensitivity analysis revealed that the overall response rate was also in favor of the combination chemotherapy regimen of DP (RR = 0.73; 95 % CI, 0.63 to 0.84; P < 0.0001; χ2 = 11.13, I2 = 37 %, P = 0.13). CR and PR were reported in detail in eight trials [16, 29, 31–36]. In terms of PR, the results were consistent with the overall response rate, as the RR estimates for eight trials favored the combination chemotherapy regimens (RR = 0.71, 95 % CI = 0.59 to 0.86, P = 0.0004; heterogeneity: χ2 = 7.16, P = 0.41, I2 = 2 %) (Supplementary Figure 2). Moreover, the DP regimen yielded superior CR rates compared with docetaxel monotherapy (RR = 0.64, 95 % CI = 0.45 to 0.92, P = 0.01; heterogeneity: χ2 = 4.15, P = 0.53, I2 = 0 %) (Supplementary Figure 3). The overall number of patients who achieved a CR was less than the number who achieved a PR. Particularly, no patients achieved a CR in either of the two arms in two trials [33, 35].
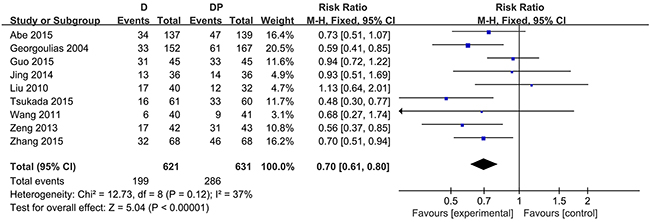
Figure 1: Forest plot for overall response.
Survival
The one-year survival rate was stated in four trials [16, 29, 30, 34]. No statistically significant difference was observed between the docetaxel monotherapy group and the DP group in terms of the one-year survival rate (RR = 1.04; 95 % CI, 0.90 to 1.19; P = 0.62). Substantial heterogeneity was found among the trial estimates (χ2 = 10.72, P = 0.01), and the I2 index indicated that 72 % of the variability across trials was due to heterogeneity rather than chance (Figure 2). Three trials [16, 29, 30] revealed no significant difference between the two groups with regard to the 1-year survival rate, yet one trial [29] showed that the OS was considerably worse in the docetaxel group (hazard ratio for DP over docetaxel, 0.23; 95 % CI, 0.09 to 0.62) for patients 70 – 74 years of age. One trial reported a higher 1-year survival rate in the DP group. In the case of G-CSF, age (≦75), and performance status (≦ 2) in one trial conducted by Georgoulias, the sensitivity analysis revealed that patients in the DP group did not seem to experience an increased survival at 1 year (RR = 1.01, 95 % CI, 0.65 to 1.56, P = 0.98; χ2 = 10.18, P = 0.006, I2 = 80 %).
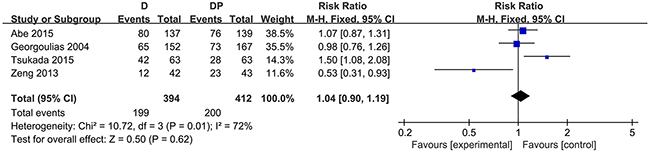
Figure 2: Forest plot for one-year survival rate.
Toxicity
Toxicity profile results with respect to the frequency of NCI-CTC grade 3 – 4 side effects were available for all trials. The number of trials with data available for anemia, thrombocytopenia, nausea/vomiting, nephrotoxicity, hyponatremia and treatment-related deaths was 7, 7, 6, 3, 2 and 4, respectively. The reporting of side effects was heterogeneous among trials. Patients who were treated with the DP regimen were more likely to experience anemia (RR = 0.34, 95 % CI = 0.19 to 0.61, P = 0.0002), thrombocytopenia (RR = 0.27, 95 % CI = 0.12 to 0.59, P = 0.001), and nausea/vomiting (RR = 0.43, 95 % CI = 0.24 to 0.75, P < 0.003) compared with patients who were treated with docetaxel. Moreover, the DP regimen led to more frequent grade 3 or 4 nephrotoxicity (RR = 0.06, 95 % CI = 0.01 to 0.45, P = 0.006), hyponatremia (RR = 0.47, 95 % CI = 0.25 to 0.88, P = 0.02), and treatment-related deaths (RR = 0.19, 95 % CI = 0.04 to 0.86, P = 0.03), and this difference was statistically significant. Almost all treatment-related deaths occurred in the DP treatment group with the exception of one patient who died of febrile neutropenia in the docetaxel group in one trial [16]. In addition, two trials [29, 30] demonstrated that the primary cause of treatment-related deaths was pneumonitis, while the trial [16] performed by Georgoulias showed that the causes of causes were diverse and were primarily febrile neutropenia, acute renal failure, febrile diarrhea and vomiting, grade 3 thrombocytopenia, pulmonary infection, anemia and non-neutropenic infection.
The risk of grade 3 or 4 neurotoxicity was comparable between the two modalities (RR = 0.33, 95 % CI = 0.09 to 1.18, P = 0.09). Four studies assessed the occurrence of neutropenia. Considerable heterogeneity existed in the morbidity associated with neutropenia among the 4 studies (χ2 = 75.60, P < 0.00001, I2 = 96 %), and the random effects model was applied to perform the data analysis. Otherwise, no statistically significant difference in the morbidity associated with neutropenia was observed (RR =0.89, 95 % CI = 0.17 to 4.69, P = 0.89) (Figure 3). Considering the role of G-CSF, a sensitivity analysis was performed and found no apparent significant difference in the incidence of grade 3 or 4 neutropenia between the two arms (RR = 0.85, 95% CI = 0.05 to 13.65, P = 0.91; χ2 = 50.81, P < 0.00001, I2 = 96 %). The results were also consistent in the morbidity associated with febrile neutropenia (RR = 1.68, 95 % CI 0.08 to 35.47, p = 0.74; χ2 = 12.12, P = 0.002, I2 = 83 %). Data on leucopenia were available in six trials. Similarly, pooled data showed that no statistically significant difference existed with respect to leucopenia (RR = 1.04, 95% CI = 0.24 to 4.54, P = 0.96; χ2 = 47.31, P < 0.00001, I2 = 89 %) (Figure 4).
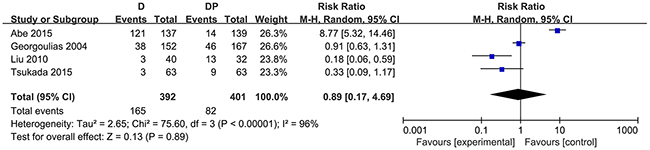
Figure 3: Forest plot for neutropenia.
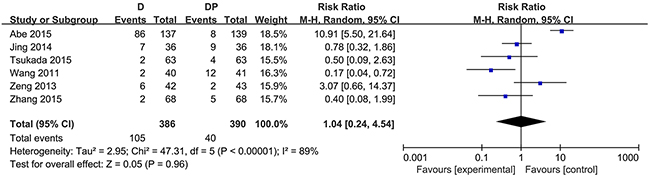
Figure 4: Forest plot for leucopenia.
Diarrhea, fatigue and pneumonitis were evaluated separately in 5, 3, and 2 trials, respectively. Overall, the meta-analysis did not reveal that patients in the DP chemotherapy group experienced a greater incidence rate of the above-mentioned side effects compared with the docetaxel group. Mucositis was reported in two trials [16, 36]. Pooled data revealed a higher incidence rate of mucositis in patients who received the DP regimen (RR = 0.39, 95 % CI 0.22 to 0.70, P = 0.001; χ2 = 1.90, P = 0.17, I2 = 47 %). A summary of hematological and non-hematological events is presented in Table 3.
Table 3: Summary of toxicity meta-analyses comparing D and DP regimens
Analyses |
No. of trials |
P-value for homogeneity |
Model |
RR (95%CI) |
P-value |
|---|---|---|---|---|---|
Anemia |
7 |
0.10 |
F |
0.34(0.19-0.61) |
0.0002 |
Neutropenia |
4 |
<0.00001 |
R |
0.89(0.17 -4.69) |
0.89 |
Febrile neutropenia |
3 |
0.002 |
R |
1.68 (0.08-35.47) |
0.74 |
Thrombocytopenia |
7 |
0.25 |
F |
0.27(0.12-0.59) |
0.001 |
Nausea/vomiting |
6 |
0.02 |
F |
0.43(0.24-0.75) |
0.003 |
Neurotoxicity |
2 |
- |
F |
0.33(0.09-1.18) |
0.09 |
Nephrotoxicity |
3 |
0.52 |
F |
0.06 (0.01-0.45) |
0.006 |
Treatment-related deaths |
4 |
0.88 |
F |
0.19(0.04-0.86) |
0.03 |
Diarrhea |
5 |
0.05 |
F |
0.60(0.32-1.13) |
0.11 |
Hyponatremia |
2 |
0.24 |
F |
0.47(0.25-0.88) |
0.02 |
leukopenia |
6 |
<0.00001 |
R |
1.04 (0.24 -4.54) |
0.96 |
Mucositis |
2 |
0.17 |
F |
0.39 (0.22-0.70) |
0.001 |
Fatigue |
3 |
0.31 |
F |
0.68 (0.28-1.63) |
0.38 |
Pneumonitis |
2 |
0.06 |
F |
3.43(0.85-13.88) |
0.08 |
Abbreviations: RR, risk ratio; R, random effects model; F, fixed effects mode; CI, confidence interval.
Quality of life
Three trials [16, 29, 30] included a QOL assessment. As the data were not available for quantitative synthesis, we incorporated a review here. Symptom score questionnaire responses were assessed after each treatment cycle until the end of the third cycle in one trial [16, 30] and were assessed at baseline, at the end of the third cycle, and at the end of chemotherapy (EOC) in one trial [16]. The compliance for QOL assessment was considered generally high. All three trials reported the numbers of patients with missing data for the QOL assessment. Two trials reported the reason for withdrawal from the QOL assessment during the treatment course. One trial [29] reported least square mean scores at baseline and at 8 weeks and found no significant difference between the groups (two-sided, P = 0.564, ANCOVA with the baseline score as a covariate). Additionally, one trial [16] reported no significant differences in QOL assessments at the end of the third cycle and at the EOC between the two arms. The trial conducted by Abe [30] reported that the mean total score remained near its baseline value in the docetaxel arm after the third cycle of therapy, while the mean total score gradually declined in the DP arm, and a significant difference was observed between the score at baseline and the score at the end of cycle 3 (P < 0.1).
DISCUSSION
Overall, many clinical trials have confirmed the activity, efficacy and toxicity of the DP combination in patients with advanced NSCLC [37–39]. It was reported that combination chemotherapy led to a nearly two-fold increase in the response rate and a modest improvement in the one-year survival rate, with increased toxicity profiles compared with single-agent treatment in patients with NSCLC [40]. In fact, the results of this meta-analysis supported, to some extent, the above findings. The present meta-analysis showed that the DP combination chemotherapy regimens had a higher overall response rate (PR plus CR) than the docetaxel regimen despite the observation that most patients achieved a partial response. However, a survival advantage was not found in the DP combination arm after an analysis of the 1-year survival rate. It appears that toxic effects, including anemia, nausea/vomiting, thrombocytopenia, hyponatremia, mucositis, nephrotoxicity and treatment-related deaths, are more likely to result from DP doublet regimens. The occurrence of toxic effects was in accordance with that in previous studies. According to our analyses, no significant difference in the occurrence of other side effects was established between these two arms, as only a slight difference was noted [21, 40, 41].
Cisplatin-based doublet regimens were associated with a higher response rate (PR plus CR) and lower occurrence of stable disease compared with non-platinum-based doublet regimens [42]. On the contrary, this suggested that an increased survival at 1 year might be the result of a better response to cisplatin. In advanced NSCLC, an improvement in survival without severe treatment-related toxicity is a major challenge in the management of patients with this cancer type. The results of this meta-analysis were consistent with those of the trial conducted by Georgoulias in 2004 [16]. A higher response rate was achieved in patients who received the DC regimen but without improvement in the one-year survival rate. Grade 3/4 nausea/vomiting (P = 0.0001), nephroroxicity (P = 0.006), neurotoxicity (P = 0.017) and diarrhea (P = 0.007) were significantly more common in the DC group. As an alternative, docetaxel could be a reasonable option for patients who cannot tolerate cisplatin in terms of QOL, especially for elderly patients with NSCLC with poor PS. Nevertheless, even though cisplatin-based doublet regimens resulted in a higher incidence rate of toxic effects, the comprehensive appraisal seems to still favor cisplatin-based regimens. The QOL profile comprises the overall performance of activity, efficacy and toxicity of chemotherapy regimens, and in turn, may be associated with the mode of treatment delivery. A higher incidence of side effects was the main cause of the decline in the QOL score. The declining QOL score was responsible for treatment discontinuation and a low progression-free survival (PFS). Otherwise, the lack of significant difference in the QOL between the two groups in three trials seemingly conflicted with the greater toxicity profiles in the DP arms. Belani performed an analysis on an elderly subgroup in the TAX 326 trial, and found that similar activity was achieved with first-line DP chemotherapy in younger and elderly patients with advanced NSCLC. Although slightly more toxicity occurred in older patients compared with younger patients, elderly patients tolerated the DP regime well [43]. However, elderly patients were sufficiently represented in the above clinical trial, which means that it may be unreasonable to extrapolate that result to the general elderly population.
The DP regimen has been studied as an initial therapy for advanced, metastatic and recurrent NSCLC. The acknowledged and recommended total dose/cycle for the two drugs is 75 mg/m2 administered on a single day. Certainly, many variations exist in dose management [19]. Mitsudomi reported that the DP regimen was administered as docetaxel 60 mg/m2 plus cisplatin 80 mg/m2 on day 1 every 21 days in an open label, randomized phase 3 trial that compared gefitinib versus DP for NSCLC [44]. It was reported that when docetaxel was given weekly rather than once every 3 weeks, the risk of neutropenia was reduced, while the antitumor activity appeared to be maintained [45]. In the present meta-analysis, the DP regimens of nine trials were diverse. DP regimens were administered as D 60 mg/m2 plus P 75 mg/m2 in one cycle in two trials [29, 30] and as D 100 mg/m2 plus P 80 mg/m2 in one trial [16]. Out of six Chinese studies, both drugs were administered as 75 mg/m2 on a single day in only one study [34]. Dosing schedules, especially dosing intervals and dosing sequences, might have different effects on toxicity and antitumor effects. Myelosuppression and nephrotoxicity caused by cisplatin were dose-limiting factors, and severe nephrotoxicity was potentially fatal [46]. The reduction of adverse effects and the improvement of antitumor effects as much as possible formed the basic principle of the trials. Using a C57BL/6N Lewis lung carcinoma mouse model, Kodama [46] found that a sequential D-P regimen in which cisplatin was administered 12 h after docetaxel, not only inhibited tumor growth to a great extent but also significantly reduced treatment-related deaths and renal toxicity compared with the DP regimen that was simultaneously used. This corresponded to the results of an in-vitro study [47]. It was reported that the enhanced cytotoxicity of a sequential D-P regimen was attributed to the accumulation of intracellular Pt–glutathione complexes, as docetaxel appeared to suppress the up-regulation of multidrug resistance-associated protein-1 (MRP-1) induced by cisplatin exposure [48]. Otherwise, docetaxel is active in patients who are refractory or resistant to cisplatin, and produces responses that range from 18 % to 25 %; this implies the lack of crossover in the mechanisms of action between docetaxel and cisplatin [49].
Dosage reductions or therapy adjustments need to be implemented after the occurrence of grade 3 or 4 toxicities, but not for grade 1 or 2 toxicities. Therefore, the data on grades 3 or 4 toxicities were quantitatively synthesized in the meta-analysis. Hematologic adverse events were the most common major toxicities because of myelosuppression. Accepted practice guidelines suggested that prophylactic use of colony stimulating factors (CSFs) could lead to a greater than 20 % reduction in the incidence of febrile neutropenia when given with antineoplastic regimens [50]. The use of CSFs was considered for regimens with an incidence of febrile neutropenia between 10 % and 20 %, but was not recommended when the incidence was less than 10 % [50]. With regard to grade 3/4 neutropenia or febrile neutropenia, no significant difference was found between the two groups, which might be attributed to the prophylactic use of CSF in the DP arm in that trial [16].
Efficacy may not be the only factor that might affect a physician’s decision with regard to the choice of chemotherapy regimen for NSCLC. The principal goals of treatment should be palliation, an acceptable quality of life and prolonged survival. Consequently, clinicians should carefully define the best anticancer drug, schedule of administration, and treatment strategy, depending on potential toxicity and the individual patient’s wishes. Some new therapies have been explored in recent years. It has been suggested that matrix metalloproteinase (MMP) inhibitors could significantly reduce vascular density around lung tumors and that the tumor growth inhibition rate could reach 88 % [36]. Thalidomide has anti-angiogenesis effects and has insignificant toxicity. Preliminary data have confirmed the feasibility of thalidomide use for advanced NSCLC. Docetaxel is not soluble in water, which reduces local dosage and its clinical effect. Currently, research that focuses on new formulations of docetaxel has become a hot topic. The new formulations can improve water solubility, reduce adverse reactions, and improve the utilization rate. Researchers have found that docetaxel packed with liposomes in an oil phase or a microemulsion system might result in satisfactory clinical effects [31].
The limitations of this systematic review involve the uniformity of the administration program and the small size of the included RCTs. Given the special features of lung cancer, imbalances in baseline prognostic factors and post-protocol treatment, such as epidermal growth factor receptor (EGFR) mutations, pathological type and comorbid illnesses, may be responsible for unexpectedly large differences in some measures between treatment arms. More patients with non-squamous histology were included in the DP arm than in the docetaxel arm in the trial conducted by Tsukada [29]. Active EGFR mutations are often observed in female patients or in patients with adenocarcinoma and have been reported as a favorable prognostic factor in patients with NSCLC [51, 52]. It was reported in two phase III studies that patients with EGFR gene mutations had increased survival and response rates when treated with docetaxel or gefitinib, compared with patients with wild-type EGFR [53, 54]. Additionally, we used the Cochrane Collaboration’s tool to assess the risk of bias in order to evaluate the methodological quality of the included trials; we also conducted a sensitivity analysis accordingly. Trials with a high or unclear risk of bias could lower the quality of evidence in our results [55]. The reports of the toxicity profiles were very heterogeneous, the causes of which are diverse. Consequently, caution should be taken when estimates of the meta-analysis are interpreted.
CONCLUSION
The DP regimens led to a higher overall response rate in comparison with docetaxel regimens. In addition, unlike the promising survival and favorable toxicity profile seen in many other studies with the DP regimen [56, 57], a survival advantage was not observed with the DP regimen. Moreover, DP doublet regimens seemed to be associated with higher toxicity, including anemia, nausea/vomiting, thrombocytopenia, hyponatremia, mucositis and nephrotoxicity and led to more treatment-related deaths. Although the interpretation of the study results was limited, we believe that to a certain extent, our analyses may provide valuable information for physicians who need to decide the best treatment strategy among all the possible regimens for patients with NSCLC. However, more powered studies with much larger sample sizes are advocated in order to obtain a more concrete conclusion.
Abbreviations
Non-small-cell lung cancer (NSCLC), docetaxel plus cisplatin (DP), China National Knowledge Internet (CNKI), quality of life (QOL), overall survival (OS), partial response(PR), complete response(CR), epidermal growth factor receptor (EGFR), matrix metalloproteinase (MMP), progression-free survival (PFS), colony stimulating factors (CSFs), multidrug resistance-associated protein-1 (MRP-1), randomized clinical trials (RCTs)
CONFLICTS OF INTEREST
The authors declare that they have no competing interests.
GRANT SUPPORT
This work was supported by State Key Program of National Natural Science Foundation of China (81330042), Special Program for Sino-Russian Joint Research Sponsored by the Ministry of Science and Technology, China (2014DFR31210) and Key Program Sponsored by the Tianjin Science and Technology Committee, China (13RCGFSY19000, 14ZCZDSY00044).
REFERENCES
1. Parkin DM, Bray FI, Devesa SS. Cancer burden in the year 2000. The global picture. Eur J Cancer. 2001; 37:S4–66.
2. Anon. SEER cancer survival statistics: 5-year survival rates for non-small cell lung and bronchus cancer. Bethesda, MD: National Cancer Institute, 2003, Table XV-9; available on-line: http://surveillance.cancer.gov/statistics/types/survival.html.
3. NSCLC Meta-Analyses Collaborative Group. Chemotherapy in addition to supportive care improves survival in advanced non-small-cell lung cancer: a systematic review and meta-analysis of individual patient data from 16 randomized controlled trials. J Clin Oncol. 2008; 26:4617–25.
4. Guéritte-Voegelein F, Guénard D, Lavelle F, Le Goff MT, Mangatal L, Potier P. Relationships between the structure of taxol analogues and their antimitotic activity. J Med Chem. 1991; 34:992–98.
5. Fossella FV, DeVore R, Kerr RN, Crawford J, Natale RR, Dunphy F, Kalman L, Miller V, Lee JS, Moore M, Gandara D, Karp D, Vokes E, et al. Randomized phase III trial of docetaxel versus vinorelbine or ifosfamide in patients with advanced non-small-cell lung cancer previously treated with platinum-containing chemotherapy regimens. The TAX 320 Non-Small Cell Lung Cancer Study Group. J Clin Oncol. 2000; 18:2354–62.
6. Lynch TJ Jr. Review of two phase III randomized trials of single-agent docetaxel in previously treated advanced non-small cell lung cancer. Semin Oncol. 2001; 28:5–9.
7. Fossella FV, Lee JS, Shin DM, Calayag M, Huber M, Perez-Soler R, Murphy WK, Lippman S, Benner S, Glisson B. Phase II study of docetaxel for advanced or metastatic platinum-refractory non-small-cell lung cancer. J Clin Oncol. 1995; 13:645–51.
8. Francis PA, Rigas JR, Kris MG, Pisters KM, Orazem JP, Woolley KJ, Heelan RT. Phase II trial of docetaxel in patients with stage III and IV non-small-cell lung cancer. J Clin Oncol. 1994; 12:1232–37.
9. NCCN Clinical Practice Guidelines in Oncology – Non-Small Cell Lung Cancer. V.2.2013. National Comprehensive Cancer NetworkWeb site. http://www.nccn.org/professionals/physician_gls/pdf/myeloid_growth.pdf. Accessed April 4, 2013.
10. Belani C, Lynch T. Docetaxel (Taxotere) in combination with platinums in patients with non-small cell lung cancer: trial data and implications for clinical management. Semin Oncol. 2001; 28:10–14.
11. Belani CP, Bonomi P, Dobbs TW, DeVore RF, Ettinger DS, Jett J, Luketich JD, Cohen LJ, Johnson DH. Docetaxel and cisplatin in patients with advanced non small-cell lung cancer (NSCLC): a multicenter phase II trial. Clin Lung Cancer. 1999; 1:144–50.
12. Kubota K, Watanabe K, Kunitoh H, Noda K, Ichinose Y, Katakami N, Sugiura T, Kawahara M, Yokoyama A, Yokota S, Yoneda S, Matsui K, Kudo S, et al, and Japanese Taxotere Lung Cancer Study Group. Phase III randomized trial of docetaxel plus cisplatin versus vindesine plus cisplatin in patients with stage IV non-small-cell lung cancer: the Japanese Taxotere Lung Cancer Study Group. J Clin Oncol. 2004; 22:254–61.
13. Kaya AO, Buyukberber S, Dane F, Isikdogan A, Ustaalioglu BO, Coskun U, Yumuk PF, Dogu GG, Ozdemir NY, Sevinc A, Gumus M, Ozkan M, Yildiz R, et al, and Anatolian Society of Medical Oncology. Cisplatin plus docetaxel combination in the first-line treatment of metastatic non-small cell lung cancer. Tumori. 2010; 96:400–04.
14. Tartarone A, Romano G, Iodice G, Capobianco A, Coccaro M, Bochicchio A, Di Leo P, Ardito R, Di Renzo N. Cisplatin and weekly docetaxel as first-line therapy in patients with advanced non-small cell lung cancer a phase II study. Tumori. 2005; 91:131–34.
15. Luedke DW, Einhorn L, Omura GA, Sarma PR, Bartolucci AA, Birch R, Greco FA. Randomized comparison of two combination regimens versus minimal chemotherapy in nonsmall-cell lung cancer: a Southeastern Cancer Study Group Trial. J Clin Oncol. 1990; 8:886–91.
16. Georgoulias V, Ardavanis A, Agelidou A, Agelidou M, Chandrinos V, Tsaroucha E, Toumbis M, Kouroussis C, Syrigos K, Polyzos A, Samaras N, Papakotoulas P, Christofilakis C, et al. Docetaxel versus docetaxel plus cisplatin as front-line treatment of patients with advanced non-small-cell lung cancer: a randomized, multicenter phase III trial. J Clin Oncol. 2004; 22:2602–09.
17. Delbaldo C, Michiels S, Syz N, Soria JC, Le Chevalier T, Pignon JP. Benefits of adding a drug to a single-agent or a 2-agent chemotherapy regimen in advanced non-small-cell lung cancer: a meta-analysis. JAMA. 2004; 292:470–84.
18. Le Chevalier T, Monnier A, Douillard JY, Ruffie P, Sun XS, Belli L, Ibrahim N, Bougon N, Bérille J. Docetaxel (Taxotere) plus cisplatin: an active and well-tolerated combination in patients with advanced non-small cell lung cancer. Eur J Cancer. 1998; 34:2032–36.
19. Firvida JL, Amenedo M, Rodríguez R, González A, Salgado M, Ramos M, Losada G. Docetaxel plus fractionated cisplatin is a safe and active schedule as first-line treatment of patients with advanced non-small cell lung cancer: results of a phase II study. Invest New Drugs. 2004; 22:481–87.
20. Kuroki S, Iwanaga K, Kato O, Takahashi K, Haruta Y, Soejima Y, Koyanagi K, Furukawa T, Soejima Y, Nagata M, Naitoh K, Aoki Y, Hayashi S. Efficacy of docetaxel (TXT) combined with cisplatin (CDDP) in non-small cell lung cancer. [Article in Japanese] Gan To Kagaku Ryoho. 2003; 30:371–75.
21. Sederholm C. Gemcitabine (G) compared with gemcitabine plus carboplatin (GC) in advanced non-small cell lung cancer (NSCLC): a phase III study by the Swedish Lung Cancer Study Group (SLUSG). Proc Am Soc Clin Oncol 2002; 21:291a.
22. Higgins JP, Green S, eds. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane-handbook.org (Accessed June, 1 2015).
23. Duffaud F, Therasse P. New guidelines to evaluate the response to treatment in solid tumors. [Article in French] Bull Cancer. 2000; 87:881–86.
24. Cella DF, Bonomi AE, Lloyd SR, Tulsky DS, Kaplan E, Bonomi P. Reliability and validity of the Functional Assessment of Cancer Therapy-Lung (FACT-L) quality of life instrument. Lung Cancer. 1995; 12:199–220.
25. Miller AB, Hoogstraten B, Staquet M, Winkler A. Reporting results of cancer treatment. Cancer. 1981; 47:207–14.
26. Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002; 21:1539–58.
27. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–60.
28. DerSimonian R, Laird N. Meta-analysis in clinical trials revisited. Contemp Clin Trials. 2015; 45:139-145. https://doi.org/10.1016/j.cct.2015.09.002.
29. Tsukada H, Yokoyama A, Goto K, Shinkai T, Harada M, Ando M, Shibata T, Ohe Y, Tamura T, Saijo N, Lung Cancer Study Group of the Japan Clinical Oncology Group (JCOG). Randomized controlled trial comparing docetaxel-cisplatin combination with weekly docetaxel alone in elderly patients with advanced non-small-cell lung cancer: Japan Clinical Oncology Group (JCOG) 0207. Jpn J Clin Oncol. 2015; 45:88–95.
30. Abe T, Takeda K, Ohe Y, Kudoh S, Ichinose Y, Okamoto H, Yamamoto N, Yoshioka H, Minato K, Sawa T, Iwamoto Y, Saka H, Mizusawa J, et al. Randomized phase III trial comparing weekly docetaxel plus cisplatin versus docetaxel monotherapy every 3 weeks in elderly patients with advanced non-small-cell lung cancer: the intergroup trial JCOG0803/WJOG4307L. J Clin Oncol. 2015; 33:575–81.
31. Zhang Lianguo, Chang Yi. Clinical efficacy of docetaxel combined with cis-platinum in treatment of non-small cell lung cancer. Int J Respir. 2015; 10:728-731.
32. Liang G, Su K, Niu J, Niu X. Effective and safety study of docetaxel single agent chemotherapy and combined with platinum based first-line in the treatment of elderly patients with advanced non small cell lung cancer. Chinese Community Doctors. 2015; 31:38–39.
33. Jing X. Efficacy of docetaxei combined wim cisplatin in the treatment of patients with non-small cell lung cancer. J Shanxi Med Univ. 2014; 45:740–42.
34. Zeng G, Zeng W. Clinical efficacy study of docetaxei combined wim cisplatin in the treatment of patients with non-small cell lung cancer. PJCCPVD. 2013; 21:36–37.
35. Wang ZY, Hou M, Wang HY. Efficacy observation of single-agent docetaxel versus docetaxel plus cisplatin as second-line treatment for advanced non-small cell lung cancer. Mod Prev Med. 2011; 38:1754–56.
36. Liu Congmin, Li Jinghua. Docetaxel combined with cisplatin in the treatment of elderly patients with advanced non-small cell lung cancer. Chinese Journal of Practical Medicine. 2010; 37:65,68.
37. Kim MJ, Kim SH, Kang JH, Kim HG, Cho YJ, Jeong YY, Kim HC, Lee JD, Hwang YS, Kim MG, Choi JY, Lee GW. Phase II trial of biweekly chemotherapy with docetaxel and cisplatin in high-risk patients with unresectable non-small cell lung cancer. Chemotherapy. 2013; 59:159–66.
38. Binder D, Hackenthal M, Graseck L, Schweisfurth H, Schäper C, Krüll M, Temmesfeld-Wollbrück B, Suttorp N, Beinert T, Hellriegel KP. A phase II study of weekly docetaxel-cisplatin as first-line treatment for advanced non-small cell lung cancer. J Thorac Oncol. 2009; 4:1144–47.
39. Park SH, Choi SJ, Kyung SY, An CH, Lee SP, Park JW, Jeong SH, Cho EK, Shin DB, Hoon Lee J. Randomized phase II trial of two different schedules of docetaxel plus cisplatin as first-line therapy in advanced nonsmall cell lung cancer. Cancer. 2007; 109:732–40.
40. Lilenbaum RC, Langenberg P, Dickersin K. Single agent versus combination chemotherapy in patients with advanced nonsmall cell lung carcinoma: a meta-analysis of response, toxicity, and survival. Cancer. 1998; 82:116–26.
41. Lilenbaum RC, Herndon J, List M. Single-agent (SA) versus combination chemotherapy (CC) in advanced non-small cell lung cancer (NSCLC): a CALGB randomized trial of efficacy, quality of life (QOL), and cost-effectiveness. Proc Am Soc Clin Oncol 2002; 21:1a.
42. Tsunoda T, Koizumi T, Hayasaka M, Hirai K, Koyama S, Takabayashi Y, Fujimoto K, Kubo K. Phase II study of weekly docetaxel combined with cisplatin in patients with advanced non-small-cell lung cancer. Cancer Chemother Pharmacol. 2004; 54:173–77.
43. Belani CP, Fossella F. Elderly subgroup analysis of a randomized phase III study of docetaxel plus platinum combinations versus vinorelbine plus cisplatin for first-line treatment of advanced nonsmall cell lung carcinoma (TAX 326). Cancer. 2005; 104:2766–74.
44. Mitsudomi T, Morita S, Yatabe Y, Negoro S, Okamoto I, Tsurutani J, Seto T, Satouchi M, Tada H, Hirashima T, Asami K, Katakami N, Takada M, et al, and West Japan Oncology Group. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomised phase 3 trial. Lancet Oncol. 2010; 11:121–28.
45. Miller VA, Kris MG. Docetaxel (Taxotere) as a single agent and in combination chemotherapy for the treatment of patients with advanced non-small cell lung cancer. Semin Oncol. 2000; 27:3–10.
46. Kodama A, To H, Kinoshita T, Ieiri I, Higuchi S. Influence of dosing schedules on toxicity and antitumour effects of combined cisplatin and docetaxel treatment in mice. J Pharm Pharmacol. 2009; 61:615–21.
47. Wang H. Combined effect of docetaxel and cisplatin for non-small cell lung cancer cell lines in vitro. Nagoya J Med Sci. 2000; 63:129–37.
48. Maeda S, Sugiura T, Saikawa Y, Kubota T, Otani Y, Kumai K, Kitajima M. Docetaxel enhances the cytotoxicity of cisplatin to gastric cancer cells by modification of intracellular platinum metabolism. Cancer Sci. 2004; 95:679–84.
49. Le Chevalier T, Bérille J, Zalcberg JR, Millward MJ, Monnier A, Douillard JY, McKeage MJ, James R, Soulas F, Loret C, Bougon N, Bizzari JP. Overview of docetaxel (Taxotere)/cisplatin combination in non-small cell lung cancer. Semin Oncol. 1999; 26:13–18.
50. Smith TJ, Khatcheressian J, Lyman GH, Ozer H, Armitage JO, Balducci L, Bennett CL, Cantor SB, Crawford J, Cross SJ, Demetri G, Desch CE, Pizzo PA, et al. 2006 update of recommendations for the use of white blood cell growth factors: an evidence-based clinical practice guideline. J Clin Oncol. 2006; 24:3187–205.
51. Yoshida K, Yatabe Y, Park J, Ogawa S, Park JY, Shimizu J, Horio Y, Matsuo K, Mitsudomi T, Hida T. Clinical outcomes of advanced non-small cell lung cancer patients screened for epidermal growth factor receptor gene mutations. J Cancer Res Clin Oncol. 2010; 136:527–35.
52. Zhuo ML, Wu MN, Zhao J, Song SW, Bai H, Wang SH, Yang L, An TT, Wang X, Duan JC, Wang YY, Guo QZ, Liu XY, et al. Epidermal growth factor receptor genotype in plasma DNA and outcome of chemotherapy in the Chinese patients with advanced non-small cell lung cancer. Chin Med J (Engl). 2011; 124:3510–14.
53. Douillard JY, Shepherd FA, Hirsh V, Mok T, Socinski MA, Gervais R, Liao ML, Bischoff H, Reck M, Sellers MV, Watkins CL, Speake G, Armour AA, Kim ES. Molecular predictors of outcome with gefitinib and docetaxel in previously treated non-small-cell lung cancer: data from the randomized phase III INTEREST trial. J Clin Oncol. 2010; 28:744–52.
54. Maruyama R, Nishiwaki Y, Tamura T, Yamamoto N, Tsuboi M, Nakagawa K, Shinkai T, Negoro S, Imamura F, Eguchi K, Takeda K, Inoue A, Tomii K, et al. Phase III study, V-15-32, of gefitinib versus docetaxel in previously treated Japanese patients with non-small-cell lung cancer. J Clin Oncol. 2008; 26:4244–52.
55. Balshem H, Helfand M, Schünemann HJ, Oxman AD, Kunz R, Brozek J, Vist GE, Falck-Ytter Y, Meerpohl J, Norris S, Guyatt GH. GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol. 2011; 64:401–06.
56. Ohe Y, Niho S, Kakinuma R, Kubota K, Ohmatsu H, Goto K, Nokihara H, Kunitoh H, Saijo N, Aono H, Watanabe K, Tango M, Yokoyama A, Nishiwaki Y. A phase II study of cisplatin and docetaxel administered as three consecutive weekly infusions for advanced non-small-cell lung cancer in elderly patients. Ann Oncol. 2004; 15:45–50.
57. Niho S, Ohe Y, Kakinuma R, Kubota K, Matsumoto T, Ohmatsu H, Goto K, Nishiwaki Y. Phase II study of docetaxel and cisplatin administered as three consecutive weekly infusions for advanced non-small cell lung cancer. Lung Cancer. 2002; 35:209–14.

