INTRODUCTION
Alternatively spliced proteins are particularly relevant in oncology since they have been linked to cancer progression and drug resistance [1, 2]. They may provide selective drug targets, or serve as a marker set for cancer diagnosis as well. For example, the tumor suppressor gene p53 is subject to alternative splicing and p53 splice variants are frequently expressed in primary ovarian cancers [3]. The p53δ, encoding a C-terminally truncated protein, was demonstrated to be associated with impaired response to primary platinum-based chemotherapy and might serve as an adverse prognostic marker for recurrence free and overall survival in ovarian cancers [3]. A splicing variant of Merlin, Δ2-4Merlin, promotes tumor metastasis by interfering with the tumor suppression role of wild type Merlin [4]. There is substantial evidence that primary metabolism is altered in cancer cells, and the pyruvate kinase M1 and M2 splicing isoforms control the balance between aerobic and anaerobic glycolysis during tumor progression [5, 6]. These observations emphasized the importance of investigation of alternative splicing genes in cancer for improving targeted therapy.
Affymetrix Exon1.0 array detects gene expression at single exon level. This facilitates the identification of alternative splicing isoform of certain genes as well as the gene fusions. Our previous study has analyzed the Exon1.0 array from 76 Chinese lung adenocarcinomas and identified CCDC6-RET fusion as novel oncogenic driver [7]. Certainly, except for the gene fusion, detection of alternative splicing is another outcome of this dataset.
BMX (bone marrow tyrosine kinase gene in chromosome X), which encodes a non-receptor tyrosine kinase belonging to BTK (Bruton’s tyrosine kinase) family. BMX has been shown to play a pivotal role in the regulation of various cellular processes including proliferation, differentiation, transformation, apoptosis, and cell motility. Previous study described BMX as a direct substrate for caspases and the resulting truncated molecule contains an intact SH2 domain and kinase domain which has an enhanced kinase activity [8]. BMX acts upstream of RhoA and activates RhoA by releasing GDI from the RhoA-GDI complex through the interaction between the PH domain of BMX and RhoA [9]. BMX directly associates with Pak1 via its N-terminal pleckstrin homology domain and also phosphorylates Pak1 on tyrosine residues [10]. Study has also shown that BMX interacts with p53 in response to DNA damage and that such interaction leads to bidirectional inhibition of the activities of both proteins in LNCaP human prostate carcinoma cells [11]. Studies also illustrated some of the upstream activator for BMX. For example, BMX activity is modulated by FAK through an interaction between the PH domain of BMX and the FERM domain of FAK and the activation of BMX by FAK promotes cell migration [12]. In addition, BMX can be induced by growth factors, cytokines [13], the extracellular matrix, and possibly by hormones [14]. More importantly, BMX mediates various signaling pathways including STAT signaling pathway [15, 16], PI-3K signaling pathways [17–19], and GPCR signaling pathway [20].
BMX expression is altered in a number of different cancers, including those of the breast and prostate [10, 21–23], suggesting BMX may play roles in cancers. For example, BMX expression level is up-regulated in hormone-resistant prostate cancer and positively correlated with tyrosine phosphorylation of AR conditions. Overexpression of BMX in androgen-sensitive LNCaP cells promotes tumor growth while knocking down BMX expression in hormone-insensitive prostate cancer cells inhibits tumor growth under androgen-depleted conditions [24].
Here we describe the discovery of a novel spliced variant of BMX, designated as BMXΔN, which results from the skipping exon 1 to exon 8 in BMX gene. BMXΔN is strongly associated with EGFR mutation in clinical samples. Moreover, this isoform promotes lung cancer cell growth, migration, and neoplastic transformation.
RESULTS
Identification of a novel BMX skipping isoform in lung adenocarcinoma
Through bioinformatics analyses of Exon1.0 array data from Chinese lung adenocarcinoma and 5′ RACE, we identified a novel BMX skipping variant (Figure 1A, 1B). We called this novel BMX isoform, BMXΔN, which lacked the N-terminal sequence from exon 1 to exon 8 (Figure 1C). We further found that BMXΔN was absent in all the 14 paired non-cancerous lung tissues. Representative reverse transcription-PCR analysis showed that BMXΔN was detectable in lung adenocarcinomas but not in paired non-cancerous lung samples (Figure 1D). Then, we expanded the study of BMXΔN in a cohort with 174 adenocarcinoma samples and identified a total of 21 lung adenocarcinomas harboring this isoform (12%, 21/174) (Figure 1E).
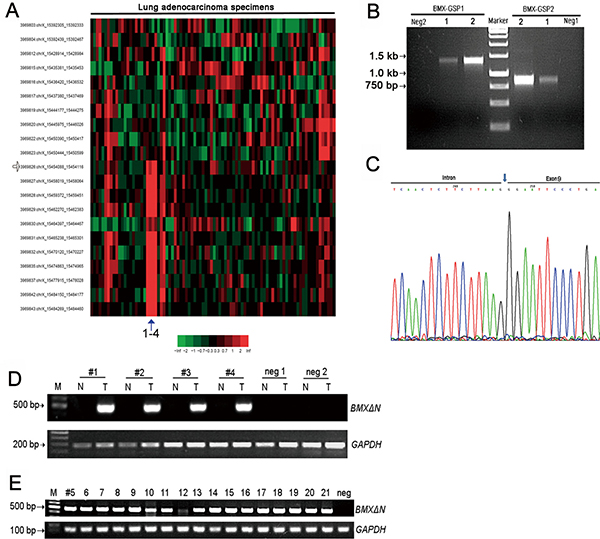
Figure 1: Identification of a novel BMX skipping isoform in human lung adenocarcinomas. (A) Exon array analyses of 78 lung adenocarcinoma samples and 10 paired non-cancerous lung samples have identified BMX abnormal splicing in lung adenocarcinoma sample 1 to 4. The potential break point was indicated by the arrow. (B) 5′ RACE analyses of the lung adenocarcinoma sample1 and sample 2 using two specific BMX primers showed the sharp PCR bands (> 750 bp and > 1300 bp), which is different from the predicted wild type BMX band (about 695 bp and 1177 bp from primer location to breakpoint). (C) Sequencing result confirmed the BMX abnormal splicing in lung adenocarcinoma sample 1and sample 2. The sequencing result showed the detailed N-terminal sequence of BMX lacking exon 1 to exon 8 but retaining part of intron 8. (D) The representative data showed that BMXΔN existed in lung adenocarcinomas but not in paired non-cancerous lung samples and control samples (negative 1 and negative 2). (E) Specific RT-PCR showed the detection of BMXΔN in another 17 lung adenocarcinomas identified from 174 lung adenocarcinomas.
Detection of BMXΔN translation start codon
The sequence of the BMXΔN gene contains four putative start codons (ATG1-ATG4). We detected at which ATG codon BMXΔN translation initiates. We constructed a series of plasmids with different ATGs and then transfected the plasmid into HEK-293T cells (Figure 2A). Western blot analysis of total protein from HEK-293T cells showed that BMXΔN was translated from plasmid carrying ATG3 (Figure 2B), indicating that the ATG located in exon 13 is the start codon for BMXΔN.
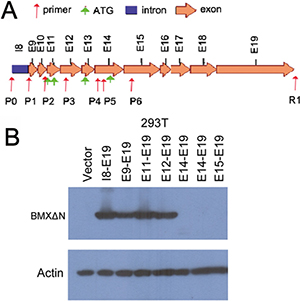
Figure 2: Detection of BMXΔN translation start codon. (A) Schematic representation of the predicted start codon of BMXΔN. The positions of exons are indicated by arrows. The positions of 7 forward primers (P0 to P6) and 1 reverse primer (R1) for PCR are also indicated below the corresponding intron or exons. (B) Immunoblot analysis of BMXΔN and actin in HEK-293T cells. Cell lysates of HEK-293T cells transfected with the indicated cDNAs in expression vectors were analyzed.
The relationship between BMXΔN expression and EGFR mutation
We further analyzed the relationship between BMXΔN expression and clinicopathological features in human lung adenocarcinomas (Tables 1 and 2). BMXΔN expression was not significantly correlated with age, gender, pathological stage (Table 2) and metastasis (Table 1). However, we found that BMXΔN was tightly associated with EGFR mutation (p = 0.002). Indeed, 20 out of 21 samples harbor EGFR mutation (Table 2).
Table 1: Correlation of BMXΔN expression with patients’ clinicopathological variables in 146 cases of adenocarcinomas
Characteristics |
Number of cases |
BMXΔN expression |
P-value |
|
|---|---|---|---|---|
Yes |
No |
|||
Age (years) |
||||
> = 50 |
117 |
12(10.3%) |
105(89.7%) |
|
< 50 |
29 |
7(24.1%) |
22(75.9%) |
0.063 |
Gender |
||||
Male |
40 |
6(15%) |
34(85%) |
|
Female |
106 |
13(12.3%) |
93(87.7%) |
0.661 |
T Classification |
||||
T1–2 |
131 |
18(13.7%) |
113(86.3%) |
|
T3–4 |
15 |
1(6.7%) |
14(93.3%) |
0.441 |
LN metastasis |
||||
Negative |
90 |
12(13.3%) |
78(86.7%) |
|
Positive |
56 |
7(12.5%) |
49(87.5%) |
0.884 |
Clinical Stage |
||||
I–II |
98 |
12(12.2%) |
86(87.8%) |
|
III–IV |
48 |
7(14.6%) |
41(85.4%) |
0.693 |
EGFR mutation |
||||
Wild type |
44 |
0(0%) |
44(100%) |
|
Mutation |
102 |
19(18.6%) |
83(81.4%) |
0.002 |
Only patients with detailed pathological data were compared in statistical analysis.
Table 2: The clinical relevance of BMXΔN expression
Sample |
Gender |
Age (years) |
Pathological stage |
EGFR mutation |
|---|---|---|---|---|
1 |
M |
60 |
IIIb |
L858R |
2 |
M |
63 |
Ia |
L858R |
3 |
F |
59 |
IIa |
L858R |
4 |
F |
54 |
Ib |
L858R |
5 |
M |
46 |
Ia |
746ELREA deletion |
6 |
F |
48 |
Ia |
746ELREA deletion |
7 |
F |
68 |
IIIa |
L858R |
8 |
M |
57 |
IIIa |
L858R |
9 |
M |
46 |
IIa |
746ELREATS = > V 753P = > Q |
10 |
F |
46 |
IIIa |
L858R |
11 |
F |
58 |
Ib |
747LREA deletion |
12 |
M |
46 |
IV |
L858R |
13 |
F |
76 |
Ia |
746ELREA deletion |
14 |
F |
51 |
Ib |
746ELREA deletion |
15 |
M |
76 |
Ia |
L858R |
16 |
F |
52 |
Ia |
L858R |
17 |
F |
39 |
IIIa |
746ELREA deletion |
18 |
F |
47 |
Ia |
L858R |
19 |
N/A |
N/A |
Ia |
No (HER2 mutation) |
20 |
F |
55 |
Ia |
L858R |
21 |
F |
48 |
IIIa |
746ELREA deletion |
P-value |
0.661 |
0.063 |
0.693 |
0.002** |
Abbreviations: F, female; M, male; N/A, not available.
Low expression of BMX in lung adenocarcinomas
On the basis of previous studies showing three transcript variants of BMX, a pair of primers (F8/R12) was designed encompassing exons 8 to 12 of the BMX open reading frame for detection of wild type BMX and other two variants. Another pair of primers (F16/R17) was also designed encompassing exons 16 and 17 of the BMX open reading frame to detect all BMX isoforms including BMXΔN. Using F16/R17 primers to observe the levels of BMX mRNA in lung adenocarcinomas and adjacent non-tumour specimens by quantitative PCR, we found that there were no significant differences in expression. However, the transcript of BMX was different between BMXΔN positive lung adenocarcinomas and adjacent non-tumour specimens (Figure 3). Interestingly, when we use F8/R12 primers to detect the levels of BMX mRNA in BMXΔN positive lung adenocarcinomas, we found very low expression of BMX or even no expression of BMX in these lung adenocarcinomas (data not shown). These findings indicate that BMXΔN is the dominant isoform in these specimens. Because wild type BMX functions as an oncogene in prostate cancer [24], we decided to explore the role of BMXΔN in our study of lung carcinogenesis.
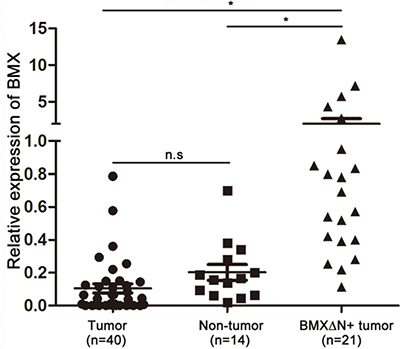
Figure 3: Expression of the BMX gene in lung adenocarcinomas. Detection of wild type BMX and BMXΔN in non-tumour adjacent tissues, lung adenocarcinomas without BMXΔN and with BMXΔN tissues by primers designed from the BMX exon 16 and 17. n.s, not significant; *P < 0.05, based on the student’s t-test.
Effect of BMXΔN on NSCLC cell growth
To investigate the role of BMXΔN in cell growth, we performed cell proliferation assay on A549, CRL-5872, and PC9 cells (Figure 4). Expression of BMXΔN efficiently promoted cell growth in A549 (Figure 4B). The wide type BMX elevated cell growth as well. To examine whether BMXΔN is involved in cell transformation, we performed soft agar colony formation assays on these cells. Figure 4C shows that more colonies were formed in BMXΔN expressing cells compared with control. Similar growth promotion effect and transformation activity were also observed in CRL-5872 cells (Figure 4F–4H). Although BMXΔN did not increase cell proliferation in PC9 cells (Supplementary Figure 1), it induced colony formation in soft agar (Figure 4J, 4K). These data suggested that BMXΔN promoted lung cancer cell growth in vitro.
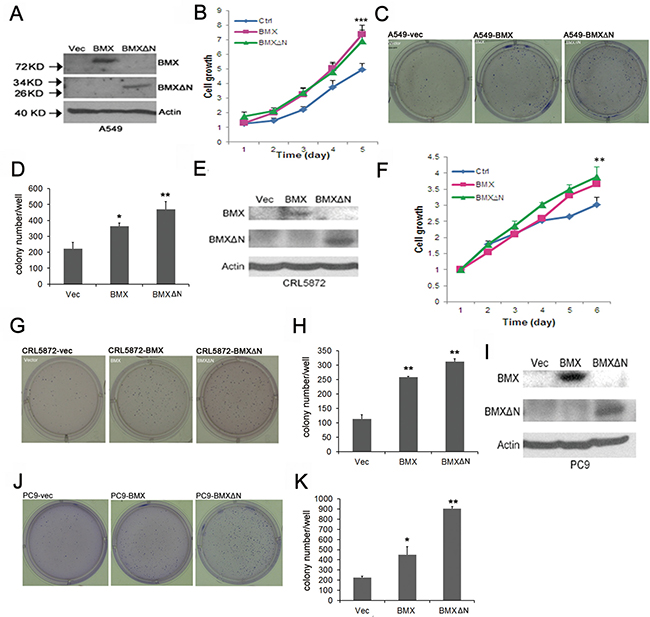
Figure 4: BMXΔN expression promotes cell growth and transformation. (A, E and I) Immunoblot analyses of BMX and BMXΔN expression in A549, CRL-5872 and PC9 cells after enforced expression of BMX or BMXΔN. (B and F) MTT assay of cell viability in A549 and CRL-5872 cells after enforced expression of BMX or BMXΔN. Statistical analysis was performed using Student’s t test (P value is **P < 0.01, ***P < 0.001). (C, D, G, H, J and K) Soft Agar assay of A549, CRL-5872 and PC9 cells after enforced expression of BMX or BMXΔN. Representative images of colony formation are shown in (C, G, J) and quantitation of colonies is shown in (D, H, K). Statistical analysis was performed using Student’s t test (P value is *P < 0.05, **P < 0.01).
BMXΔN facilitates tumor cell migration and enables cell transformation
We further studied the role of BMXΔN in lung cancer cell migration (Figure 5A, 5B). The wound-healing assay showed BMXΔN transfected PC9 cells obtained quicker closure of the scratched “wound” compared with control cells (Figure 5A). Migration was also examined using transwell assays where the cells were incubated in serum-free DMEM medium in the upper compartment and allowed to migrate towards the lower compartment containing 15% FBS. The result showed that enforced BMXΔN expression greatly increased the migration ability of A549 cells (Figure 5B). To evaluate the transformation capacity of BMXΔN, we introduced BMXΔN and mutant EGFR into Ba/F3 cells. BMXΔN-transfected Ba/F3 cells showed accelerated growth rate compared with mock transfectants, whereas no difference in growth was observed between the BMXΔN- and EGFR L858R-transfected cells. These results indicate that BMXΔN was capable of transforming Ba/F3 cells in vitro (Figure 5C). The above findings demonstrated an important role of BMXΔN in lung cell carcinogenesis.
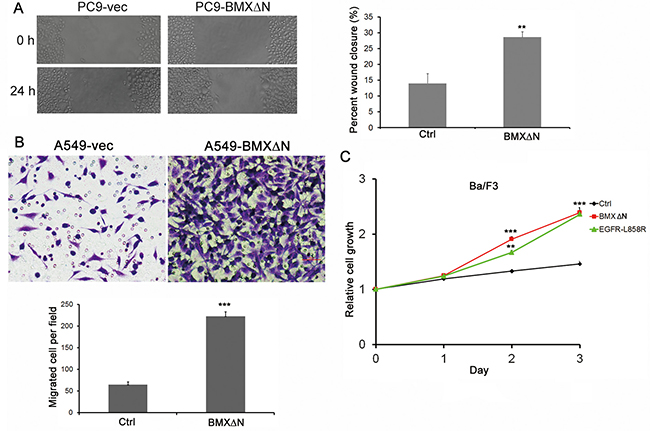
Figure 5: BMXΔN promotes cell migration and Ba/F3 transformation. (A) Wound healing assay of PC9 cells with or without BMXΔN expression. Representative images of cell migration are shown at 0 h and 24 h. (B) BMXΔN promoted A549 cell migration in transwell assay. Statistics analyses were shown below. (C) The effect of BMXΔN and EGFR-L858R on Ba/F3 cell transformation was examined at time indicated. Statistical analysis was performed using Student’s t test (P value is **P < 0.01, ***P < 0.001).
BMXΔN activated ERK in lung cancer cells
Previous studies have shown that BMX expression could activate several signaling pathways, including PI3-AKT pathway and STAT pathway. We tested if these pathways are also involved in BMXΔN function in lung cancer. We detected the phosphorylation of STAT3, ERK, AKT and FAK in lung cancer cells expressing either wild type BMX or BMXΔN and control cells followed by EGF stimulation. As shown in Figure 6, the much higher level of expression of phosphorylated ERK could be detected in BMXΔN transfected cells, suggesting MAPK pathway might contribute to the role of BMXΔN in lung cancer. Our data indicated that other tested pathways were not affected by BMXΔN expression in A549 cells.
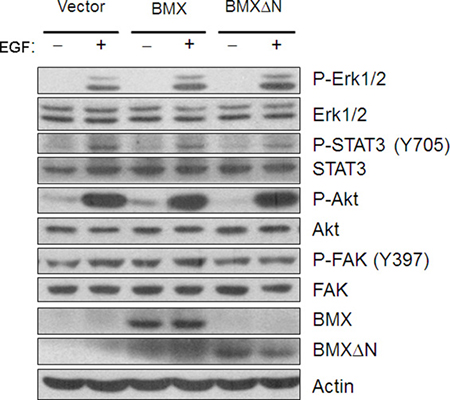
Figure 6: BMXΔN expression activates ERK in lung cancer cells. Immunoblot analysis is performed to detect BMX/BMXΔN protein expression and phosphorylation of ERK, STAT3, AKT and FAK signaling pathway in A549 cells with or without BMX/BMXΔN expression. Cells were treated with EGF (50ng/ml) for two hours before extraction of total cellular protein.
DISCUSSION
The discovery of alternative splicing variants in cancers has been paid much attention recently and their detection potentially increases with the use of innovative approaches. We here identified a BMX skipping isoform through the analyses of Exon1.0 array profiling of human lung adenocarcinoma samples in combination with RACE method. BMXΔN lacks the N-terminal sequence from exon 1 to exon 8. BMXΔN only expresses in lung cancer but not in paired non-cancerous tissues. Through screening a large collection of NSCLC patient samples, we identified a total of 21 lung adenocarcinomas expressing BMXΔN (12%, 21/174). Interestingly, BMXΔN is strongly associated with EGFR mutation in our study. However, the clinical relevance is not known yet.
A few studies have correlated BMX function with tumor growth, metastasis or poor prognosis in cancer. Overexpression of BMX in androgen-sensitive LNCaP cells promotes tumor growth while knocking down BMX expression in hormone-insensitive prostate cancer cells inhibits tumor growth under androgen-depleted conditions [21]. BMX is up-regulated in bladder cancer and predicts poor prognosis in patients with cystectomy [25]. A study has shown that BMX could maintain self-renewal and tumorigenic potential of glioblastoma stem cells by activating STAT3 [26]. Here, we present evidence that BMX was not up-regulated in lung adenocarcinomas. It might not contribute to lung carcinogenesis despite knowing it could promote cell proliferation of NSCLC. Importantly, our results reveal a previously unknown splicing skipping form of BMX. The studies suggested that BMXΔN might play roles in lung tumorigenicity, with expression of BMXΔN promoting cell growth, cell migration, and cell transformation. Future studies are necessary to clarify the mechanism by which BMXΔN activates ERK1/2.
Collectively, this study discovered a novel BMX skipping with crucial function in lung cancer cells. Future studies into this novel BMX variant might provide a better understanding of lung tumorigenesis and clinical implication for therapeutics.
MATERIALS AND METHODS
Specimen collection
The study was approved by the ethics review board at Fudan University Shanghai Cancer Center, Shanghai, China. 174 cases of lung adenocarcinomas with paired pathological normal lungs were collected consecutively with written informed consents from all patients. Fresh surgical specimens were snap-frozen and stored in liquid nitrogen upon resection until use. The pathology of each tumor sample was determined by pathologists. All these specimens were with a minimum of 70% of tumor cellularity, and all patients did not receive neoadjuvant chemotherapy. The status of EGFR mutations and other drive mutations in these specimens was determined as previously described [27, 28]. The correlation of BMXΔN expression and patients’ clinical characteristics were illustrated in 146 lung adenocarcinoma samples, a subset of 174 cases, containing 19 BMXΔN positive samples (Table 1).
Cell culture, DNA constructs and plasmid transfection
A549, CRL-5872 and PC9 cells were purchased from the ATCC. Cells were cultured in DMEM, supplemented with 8% Fetal Bovine Serum (FBS), 100 μg/ml streptomycin and 100 U/ml penicillin, at 37ºC in 5% CO2 incubator. A lentiviral construct expressing wild type BMX or BMXΔN were generated by cloning a DNA fragment corresponding to BMX full length or BMX residues 384-675 (NP-001712.1) into the NheI and NotI sites of pCDH-CMV-copGFP vector (SBI). Viral particles were produced in HEK-293T cells co-transfected with pCDH constructs and packaging plasmids pCMV-VSVG/delta8.2 (System Biosciences) in DMEM media. The progeny viruses released from HEK-293T cells were filtered, collected and used to infect cells.
Gene functional assays
For cell proliferation assay, cells were seeded in 96-well plates at a density of 3×103 cells per well, and cell growth rate was assessed with the 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) kit (Roche Diagnostics). The MTT assays in each cell line repeated three times, respectively. For soft agar colony formation assay, 8×103 cells were seeded in 6-well plates, and after three weeks of culture cell colonies were counted by crystal violet staining. The results are expressed as the mean ± SD of three independent experiments. For wound-healing assay, PC9 cells were cultured on a 12-well plate and maintained in DMEM. At 80% to 90% confluence, the cells were starved for 12 hours cultured in DMEM without FBS. A 10 μl pipette tip was used to create a linear scratch. The cells were then washed with PBS to remove floating cellular debris and fed for an additional 24 hours with full DMEM. Migration photos were captured immediately after scratching and at 24 hours after scratching by a digital camera. Cell migration was also assessed using 12-well transwell chambers (Corning Costar) with a pore size of 8 μm. A549 cells (1 × 105) were seeded in serum-free medium in the upper chamber and incubated at 37 °C for 24 h. Afterward, the cells remained in the upper chamber were carefully removed with a cotton swab, whereas the cells having traversed to reverse face of the membrane were fixed with 5% acetic acid and stained with 0.4% crystal violet. Three random fields were counted at x20 magnification. The results represent the average of samples from three independent experiments. Oncogenic transformation assay was performed in Ba/F3 cells. Ba/F3 cells were infected with lentivirus containing a control vector or a BMXΔN/EGFR-L858R plasmid. Infected cells were incubated with IL-3 (0.5 pg/mL) to support Ba/F3 marginal growth for approximately 72 hours. 3000 Ba/F3 cells per well were plated in quadruplicate in 96-well plates and cultivated for 3 days without IL3. Cell viability was measured daily. The experiments were repeated independently three times.
RT-PCR and quantitative real-time PCR
Total RNA was extracted from tissues with TRIzol® Reagent (Invitrogen) and reverse transcribed into cDNA using the Superscript III Kit (Invitrogen) according to the manufacturer’s instruction. The cDNAs were used as templates in PCR with BMXΔN gene-specific primers (forward primer, 5′-AGGGTGGGATTT GATATTGCATGG-3′ and reverse primer, 5′-CCAGGGA CACAGAGTCGGGGA-3′). The human glyceraldehyde-3-phosphate dehydrogenase gene (forward primer, 5′-GCGACACCCACTCCTCCACCTTT-3′; and reverse primer, 5′-TGCTGTAGCCAAATTCGTTGTCATA-3′) was used as an internal control in PCR amplification. The amplification reactions were performed using AmpliTaq Gold DNA polymerase (Applied Biosystems). The PCR program for detection of BMXΔN and GAPDH is: 95°C 5 minutes; 95°C 15 seconds, 58°C 30 seconds, 72°C 30 seconds, 35 cycles for BMXΔN and 32 cycles for GAPDH; 72°C 7 minute.
Real-time PCR was performed on an Applied Biosystems 7900HT cycler using SYBR Green Master Mix (SA Biosciences). The primers that were used for amplification of wild type BMX (NM_203281.2) and another two variants (NM_001721.6, NM_001320866.1) as follows: 5′-CAGTAACCAAAAAGAAAGAAATG-3′ and 5′-TGTGTTGATGATAATGAATAAGC-3′. The primers that were used for amplification of both wild type BMX and BMXΔN as follows: 5′-CTGCTCGTAAC TGCTTGGTGG-3′ and 5′-CTGACTTGCTGCTGTATT TGA-3′. The primers for housekeeping gene GAPDH were 5′- GCGACACCCACTCCTCCACCTTT-3′ (forward) and 5′- TGCTGTAGCCAAATTCGTTGTCATA-3′ (reverse); the primers for ACTIN were 5′-CTTAGTTGCGTT ACACCCTTTCT-3′ (forward) and 5′-TGCTGTCA CCTTCACCGTTC-3′ (reverse). The following conditions were used for PCR: i) initial denaturation step at 95°C for 8 min; and ii) 40 cycles at 95°C for 15 sec and 58°C for 45 sec. Relative quantity of BMX expression was calculated by the 2-ΔΔCt method standardized to GAPDH or ACTIN expression in corresponding tissues.
Exon array analysis and 5′ RACE
Exon array analysis and 5′ RACE were performed as previously described [7]. Briefly, Affymetrix Human Exon 1.0 microarray was used and the Robust Multichip Average method was applied to perform background correction, normalization and exon-level probe set summarization. 5′ RACE-PCR was performed using SMARTerTM RACE cDNA Amplification Kit from Clontech Laboratories Inc (Mountain View, CA) according to the manufacturer’s instructions. In brief, 1 μg RNA extracted from lung cancer patient tissues was reverse transcribed using primers 5′-RACE CDS primer A and SMARTer II A Oligonucleotide supplied by SMARTerTM RACE cDNA Amplification Kit. PCR was performed with BMX gene specific primer 5′-GGTTCAAGTCCTTTTC CGTGACTCCTCA-3′/5′-CCCGAAGTGGTTCAATGGA AGACAGGA-3′ in conjunction with RACE universal primer A mix (UPM): 5′-CTAATACGACTCACTATA GGGCAAGCAGTGGTATCAACGCAGAGT-3′ and 5′-CT AATACGACTCACTATAGGGC-3′. PCR products were purified for direct sequencing as well as cloning into pGEM-T vector (Promega, Madison, WI) for sequencing.
Western blot
Western blot analyses were performed according to the standard protocol. The following antibodies were used: pEGFR (Y1068, #2236), EGFR (#2232), pErk1/2 (#9101), Erk1/2 (#9102), pAKT (T308, #9257; S473, #9271), AKT (#2920), pSTAT3 (#9138), STAT3 (#9139), pFAK (#8556) and FAK (#13009), all from Cell Signaling Technologies; BMX (C-17, sc-8874) from Santa Cruz and ß-actin (A1978) from Sigma.
Statistical analysis
The statistical analysis was conducted in SPSS 16.0 (SPSS Inc, Chicago, IL, USA). Pearson’s chi-squared test was used on categorical variables. Two group comparisons were analyzed by the two-tailed Student’s t test. A p value less than 0.05 was considered statistically significant.
Abbreviations
BMX: Bone marrow tyrosine kinase gene in chromosome X BTK: Bruton’s tyrosine kinase FBS: Fetal bovine serum MTT: 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide NSCLC: non-small cell lung cancer
Authors’ contributions
YW and JX designed and performed the experiments, interpreted the data and discussed the manuscript. ZF and FL analyzed and interpreted the Exon array data to identify alternative spliced genes, DL, ZW and YF participated in performing the experiments, JZ reorganized clinical information, HC provided clinical information and samples, HJ designed the experiments, interpreted the data and wrote the manuscript. HL designed and performed the experiments, interpreted the data and wrote the manuscript. All authors read and approved the final manuscript.
ACKNOWLEDGMENTS
Not applicable.
CONFLICTS OF INTEREST
The authors have no conflicts of interests to disclose.
FUNDING
This work was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB19000000), the National Natural Science Foundation of China (81372509, 81325015, 81430066, 31621003, 81402276, 81402371, 81401898, 81402498, 81101583, 31370747), Science and Technology Commission of Shanghai Municipality (15XD1504000), the Innovation Project of Shandong Academy of Medical Sciences, the Excellent Innovation Team of Shandong Academy of Medical Sciences, Shanghai Institutes for Biological Sciences (2014KIP304), the Project for Laureate of Taishan Scholar (ts201511075), and the SA-SIBS scholarship program (HL and HJ).
REFERENCES
1. Dvinge H, Kim E, Abdel-Wahab O, Bradley RK. RNA splicing factors as oncoproteins and tumour suppressors. Nat Rev Cancer. 2016; 16:413–430.
2. Zhang J, Manley JL. Misregulation of pre-mRNA alternative splicing in cancer. Cancer Discov. 2013; 3:1228–1237.
3. Hofstetter G, Berger A, Fiegl H, Slade N, Zoric A, Holzer B, Schuster E, Mobus VJ, Reimer D, Daxenbichler G, Marth C, Zeimet AG, Concin N, et al. Alternative splicing of p53 and p73: the novel p53 splice variant p53delta is an independent prognostic marker in ovarian cancer. Oncogene. 2010; 29:1997–2004.
4. Luo ZL, Cheng SQ, Shi J, Zhang HL, Zhang CZ, Chen HY, Qiu BJ, Tang L, Hu CL, Wang HY, Li Z. A splicing variant of Merlin promotes metastasis in hepatocellular carcinoma. Nat Commun. 2015; 6:8457.
5. Christofk HR, Vander Heiden MG, Harris MH, Ramanathan A, Gerszten RE, Wei R, Fleming MD, Schreiber SL, Cantley LC. The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature. 2008; 452:230–233.
6. Mazurek S. Pyruvate kinase type M2: a key regulator of the metabolic budget system in tumor cells. Int J Biochem Cell Biol. 2011; 43:969–980.
7. Li F, Feng Y, Fang R, Fang Z, Xia J, Han X, Liu XY, Chen H, Liu H, Ji H. Identification of RET gene fusion by exon array analyses in “pan-negative” lung cancer from never smokers. Cell Res. 2012; 22:928–931.
8. Wu YM, Huang CL, Kung HJ, Huang CY. Proteolytic activation of ETK/Bmx tyrosine kinase by caspases. J Biol Chem. 2001; 276:17672–17678.
9. Kim O, Yang J, Qiu Y. Selective activation of small GTPase RhoA by tyrosine kinase Etk through its pleckstrin homology domain. J Biol Chem. 2002; 277:30066–30071.
10. Bagheri-Yarmand R, Mandal M, Taludker AH, Wang RA, Vadlamudi RK, Kung HJ, Kumar R. Etk/Bmx tyrosine kinase activates Pak1 and regulates tumorigenicity of breast cancer cells. J Biol Chem. 2001; 276:29403–29409.
11. Jiang T, Guo Z, Dai B, Kang M, Ann DK, Kung HJ, Qiu Y. Bi-directional regulation between tyrosine kinase Etk/BMX and tumor suppressor p53 in response to DNA damage. J Biol Chem. 2004; 279:50181–50189.
12. Chen R, Kim O, Li M, Xiong X, Guan JL, Kung HJ, Chen H, Shimizu Y, Qiu Y. Regulation of the PH-domain-containing tyrosine kinase Etk by focal adhesion kinase through the FERM domain. Nat Cell Biol. 2001; 3:439–444.
13. Pan S, An P, Zhang R, He X, Yin G, Min W. Etk/Bmx as a tumor necrosis factor receptor type 2-specific kinase: role in endothelial cell migration and angiogenesis. Mol Cell Biol. 2002; 22:7512–7523.
14. Qiu Y, Kung HJ. Signaling network of the Btk family kinases. Oncogene. 2000; 19:5651–5661.
15. Saharinen P, Ekman N, Sarvas K, Parker P, Alitalo K, Silvennoinen O. The Bmx tyrosine kinase induces activation of the Stat signaling pathway, which is specifically inhibited by protein kinase Cdelta. Blood. 1997; 90:4341–4353.
16. Tsai YT, Su YH, Fang SS, Huang TN, Qiu Y, Jou YS, Shih HM, Kung HJ, Chen RH. Etk, a Btk family tyrosine kinase, mediates cellular transformation by linking Src to STAT3 activation. Mol Cell Biol. 2000; 20:2043–2054.
17. Qiu Y, Robinson D, Pretlow TG, Kung HJ. Etk/Bmx, a tyrosine kinase with a pleckstrin-homology domain, is an effector of phosphatidylinositol 3’-kinase and is involved in interleukin 6-induced neuroendocrine differentiation of prostate cancer cells. Proc Natil Acad Sci USA. 1998; 95:3644–3649.
18. Ekman N, Arighi E, Rajantie I, Saharinen P, Ristimaki A, Silvennoinen O, Alitalo K. The Bmx tyrosine kinase is activated by IL-3 and G-CSF in a PI-3K dependent manner. Oncogene. 2000; 19:4151–4158.
19. Chau CH, Chen KY, Deng HT, Kim KJ, Hosoya K, Terasaki T, Shih HM, Ann DK. Coordinating Etk/Bmx activation and VEGF upregulation to promote cell survival and proliferation. Oncogene. 2002; 21:8817–8829.
20. Cohen I, Maoz M, Turm H, Grisaru-Granovsky S, Maly B, Uziely B, Weiss E, Abramovitch R, Gross E, Barzilay O, Qiu Y, Bar-Shavit R. Etk/Bmx regulates proteinase-activated-receptor1 (PAR1) in breast cancer invasion: signaling partners, hierarchy and physiological significance. PloS One. 2010; 5:e11135.
21. Chen KY, Huang LM, Kung HJ, Ann DK, Shih HM. The role of tyrosine kinase Etk/Bmx in EGF-induced apoptosis of MDA-MB-468 breast cancer cells. Oncogene. 2004; 23:1854–1862.
22. Jiang X, Borgesi RA, McKnight NC, Kaur R, Carpenter CL, Balk SP. Activation of nonreceptor tyrosine kinase Bmx/Etk mediated by phosphoinositide 3-kinase, epidermal growth factor receptor, and ErbB3 in prostate cancer cells. J Biol Chem. 2007; 282:32689–32698.
23. Dai B, Kim O, Xie Y, Guo Z, Xu K, Wang B, Kong X, Melamed J, Chen H, Bieberich CJ, Borowsky AD, Kung HJ, Wei G, et al. Tyrosine kinase Etk/BMX is up-regulated in human prostate cancer and its overexpression induces prostate intraepithelial neoplasia in mouse. Cancer Res. 2006; 66:8058–8064.
24. Dai B, Chen H, Guo S, Yang X, Linn DE, Sun F, Li W, Guo Z, Xu K, Kim O, Kong X, Melamed J, Qiu S, et al. Compensatory upregulation of tyrosine kinase Etk/BMX in response to androgen deprivation promotes castration-resistant growth of prostate cancer cells. Cancer Res. 2010; 70:5587–5596.
25. Guo S, Sun F, Guo Z, Li W, Alfano A, Chen H, Magyar CE, Huang J, Chai TC, Qiu S, Qiu Y. Tyrosine kinase ETK/BMX is up-regulated in bladder cancer and predicts poor prognosis in patients with cystectomy. PloS One. 2011; 6:e17778.
26. Guryanova OA, Wu Q, Cheng L, Lathia JD, Huang Z, Yang J, MacSwords J, Eyler CE, McLendon RE, Heddleston JM, Shou W, Hambardzumyan D, Lee J, et al. Nonreceptor tyrosine kinase BMX maintains self-renewal and tumorigenic potential of glioblastoma stem cells by activating STAT3. Cancer Cell. 2011; 19:498–511.
27. Gao B, Sun Y, Zhang J, Ren Y, Fang R, Han X, Shen L, Liu XY, Pao W, Chen H, Ji H. Spectrum of LKB1, EGFR, and KRAS mutations in chinese lung adenocarcinomas. J Thorac Oncol. 2010; 5:1130–1135.
28. Fang R, Zheng C, Sun Y, Han X, Gao B, Li C, Liu H, Wong KK, Liu XY, Chen H, Ji H. Integrative genomic analysis reveals a high frequency of LKB1 genetic alteration in Chinese lung adenocarcinomas. J Thorac Oncol. 2014; 9:254–258.

