INTRODUCTION
Integration of magnetic resonance imaging (MRI) and 18F-fluorodesoxyglucose positron emission tomography (18F-FDG-PET) into one diagnostic system has been reported as high beneficial for investigation of different pelvic malignancies [1, 2]. It has been shown that PET/MRI demonstrated a high resolution morphological information based on MRI combined with metabolic data derived from the PET component [1, 3–6]. Furthermore, Queiroz et al. reported that PET/MRI accuracy was statistically superior to PET/CT for primary tumor delineation, especially in cases with cervical and endometrial cancer [2]. In addition, the authors postulated that PET/MRI may be the preferred imaging modality for staging cervical and endometrial tumors [2].
MRI can be completed by diffusion weighted imaging (DWI), which provides additional information regarding tumor texture, such as cellularity and proliferation potential [7, 8]. According to the literature, combination of apparent diffusion coefficient (ADC) as product of DWI and standardized uptake value (SUV) has been established as a useful tool in detection and staging of different pelvic tumors [9-11]. For example, previous reports suggested that DWI and SUV can predict T-and N-stage of cervical cancer [3]. Furthermore, both parameters have been reported to be useful for treatment monitoring, as well as the prediction of clinical outcome [7]. Finally, some authors observed significant correlations between SUV and ADC values in uterine cervical cancer and postulated that DWI and 18F-FDG-PET might play a complementary role for the clinical assessment of this malignancy [8, 11].
These findings may base on possible associations between PET, DWI and histopathological parameters in several malignancies. Some previous studies indicated that ADC and SUV reflect different aspects of tumor biology [8, 12]. For example, in head and neck cancer it has been shown that SUV and ADC correlated with different histopathological findings and, therefore, can be used as complementary biological markers [12].
We hypothesized that in uterine cervical cancer PET and DWI are also associated with histopathology and can predict biological features of tumors and tumor behavior. This is very important because to the fact that uterine cervical cancer is one of the most frequent malignancies diagnosed in women with high recurrence and 5-year mortality rates. To the best of our knowledge, no previous studies investigated this question. Therefore, the purpose of this study was to analyze possible associations between ADC and SUV values and different histopathological parameters in uterine cervical cancer.
RESULTS
The clinical characteristics of the involved patients are shown in Table 1. In most cases (n = 18, 85.7%) squamous cell carcinoma was diagnosed. The grade of cell differentiation was well (G1) in one patient (4.8%), moderate (G2) in 12 cases (57.1%), and poor (G3) in 8 (38.1%). Most frequently T stages 2b and 4a were diagnosed (Table 1). Seven patients (33.3%) were staged as N0, 13 (61.9%) as N1, and one (4.8%) as N2. Furthermore, distant metastases were found in 8 (38.1%) patients (Table 1).
Table 1: Clinical data of the investigated patients
Case |
Age |
T stage |
N Stage |
M Stage |
|---|---|---|---|---|
1 |
63 |
2b |
1 |
0 |
2 |
76 |
4a |
0 |
0 |
3 |
65 |
2b |
0 |
0 |
4 |
63 |
4a |
1 |
1 |
5 |
34 |
2b |
1 |
0 |
6 |
57 |
4a |
1 |
1 |
7 |
77 |
2b |
1 |
1 |
8 |
50 |
1b |
0 |
0 |
9 |
53 |
2b |
0 |
0 |
10 |
32 |
4a |
1 |
0 |
11 |
32 |
2b |
0 |
0 |
12 |
54 |
3a |
2 |
0 |
13 |
57 |
2a |
1 |
1 |
14 |
79 |
4b |
1 |
0 |
15 |
52 |
4b |
0 |
0 |
16 |
37 |
2b |
1 |
1 |
17 |
72 |
4a |
0 |
0 |
18 |
46 |
2b |
1 |
1 |
19 |
71 |
4 |
1 |
1 |
20 |
50 |
2b |
1 |
1 |
21 |
61 |
4a |
1 |
0 |
The mean, median, and standard deviation values for all analyzed DWI and PET parameters are summarized in Table 2. All of them showed wide variations. There were no significant correlations between different DWI and PET parameters (Table 3).
Table 2: DWI and PET parameters of cervical cancer
Parameters |
M ± SD |
Median |
Range |
|---|---|---|---|
SUVmax |
21.57 ± 10.84 |
17.8 |
9.24 – 56.20 |
SUVmax |
11.60 ± 6.47 |
9.73 |
1.7 – 32 |
rSUVmax |
2.08 ± 0.95 |
1.8 |
1.51 – 5.44 |
ADCmean, × 10-3 mm2s-1 |
0.86 ± 0.13 |
0.82 |
0.64 – 1.18 |
ADCmin, ×10-3 mm2s-1 |
0.58 ± 0.16 |
0.56 |
0.37 – 0.95 |
ADCmax, ×10-3 mm2s-1 |
1.24 ± 0.25 |
1.20 |
0.77 – 2.04 |
rADCmin |
0.67 ± 0.12 |
0.68 |
0.5 – 0.85 |
SUVmax/ ADCmin |
40.75 ± 24.85 |
33.58 |
9.73 – 112.40 |
SUVmax/ ADCmean |
25.42 ± 11.91 |
22.25 |
7.83 – 62.44 |
Table 3: Correlations between DWI and SUV parameters
Parameters |
SUVmax |
SUVmean |
rSUVmax |
|---|---|---|---|
ADCmean |
p = 0.13 |
p = 0.02 |
p = 0.26 |
P = 0.58 |
P = 0.94 |
P = 0.26 |
|
ADCmin |
p = –0.13 |
p = –0.19 |
p = 0.002 |
P = 0.59 |
P = 0.42 |
P = 0.99 |
|
ADCmax |
p = 0.09 |
p = 0.12 |
p = 0.23 |
P = 0.71 |
P = 0.62 |
P = 0.33 |
|
rADCmin |
p = –0.14 |
p = –0.19 |
p = –0.12 |
P = 0.54 |
P = 0.39 |
P = 0.62 |
A comparison analysis of the identified PET and DWI parameters between the tumor grades and stages identified the following results. There were no significant differences in SUV and DWI values between different tumor grades (Table 4). Also the PET and DWI parameters did not differ significantly between T2 and T4 tumor stages (Table 5A). ADCmin and rADCmin were statistically significant lower in N positive tumors (p = 0.017 and 0.03, respectively) (Table 5B, Figure 1). Furthermore, ADCmin tended to be lower in M positive tumors (p = 0.08) (Table 5C). Additionally, the combined parameter SUVmax/ADCmin had a tendency to be higher in distant metastasized cancers (p = 0.09).
Table 4: Comparison of PET and DWI values between different tumor grades
Parameters |
G2 |
G3 |
ANOVA |
|---|---|---|---|
SUVmax |
20.26 ± 12.15 |
20.86 ± 5.89 |
0.13 |
SUVmean |
11.20 ± 7.37 |
10.83 ± 4.05 |
0.23 |
rSUVmax |
2.08 ± 1.06 |
2.11 ± 0.90 |
0,98 |
ADCmin |
0.56 ± 0.17 |
0.62 ± 0.15 |
0.69 |
ADCmean |
0.85 ± 0.13 |
0.86 ± 0.14 |
0.74 |
ADCmax |
1.20.25 ± 1.25 |
1.26 ± 0.37 |
0.34 |
rADCmin/ ADCmean |
0.65 ± 0.12 |
0.72 ± 0.13 |
0,34 |
SUVmax /ADCmin |
39.30 ± 25.18 |
38.06 ± 23.26 |
0.29 |
SUVmax/ADCmean |
24.10 ± 13.33 |
24.97 ± 8.31 |
0.26 |
Table 5A: Comparison of PET and DWI values between different tumor T stages
Parameters |
T2Mean |
T4Mean |
ANOVA |
|---|---|---|---|
SUVmax |
20.02 ± 6.64 |
25.34 ± 14.21 |
0.30 |
SUVmean |
11.33 ± 3.82 |
13.28 ± 8.48 |
0.52 |
rSUVmax |
1.78 ± 0.14 |
2.09 ± 0.84 |
0.400 |
ADCmin |
0.59 ± 0.17 |
0.55 ± 0.08 |
0.61 |
ADCmean |
0.85 ± 0.11 |
0.85 ± 0.12 |
0.99 |
ADCmax |
1.47 ± 0.91 |
1.2 ± 0.23 |
0.40 |
rADCmin |
0.68 ± 0.15 |
0.66 ± 0.08 |
0.55 |
SUVmax /ADCmin |
37.92 ± 21.33 |
47.31 ± 29.75 |
0.43 |
SUVmax/ADCmean |
23.87 ± 8.21 |
29.36 ± 14.63 |
0.30 |
Table 5B: Comparison of PET and DWI values between different tumor N stages
Parameters |
N0 |
N1/2 |
p values |
|---|---|---|---|
SUVmax |
20.72 ± 11.37 |
22.0 ± 10.98 |
0.81 |
SUVmean |
10.90 ± 6.69 |
11.95 ± 6.58 |
0.73 |
rSUVmax |
2.33 ± 1.37 |
1.96 ± 0.69 |
0.39 |
ADCmin |
0.69 ± 0.15 |
0.52 ± 0.13 |
0.017 |
ADCmean |
0.92 ± 0.17 |
0.83 ± 0.09 |
0.13 |
ADCmax |
1.23 ± 0.28 |
1.39 ± 0.77 |
0.62 |
rADCmin |
0.76 ± 0.10 |
0.63 ± 0.12 |
0.03 |
SUVmax /ADCmin |
33.13 ± 23.23 |
44.56 ± 25.58 |
0.33 |
SUVmax/ADCmean |
23.39 ± 12.53 |
26.43 ± 11.93 |
0.59 |
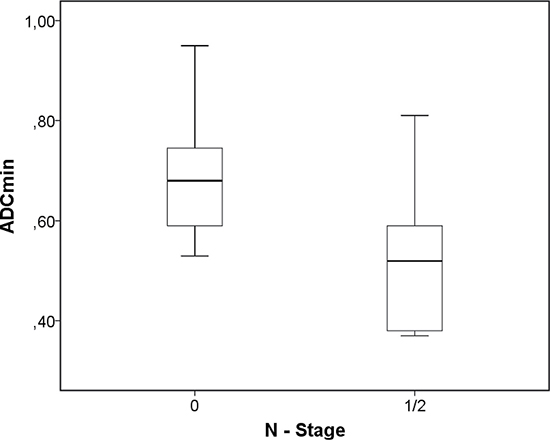
Figure 1: Associations between DWI and tumor stage. Comparison of ADCmin values between N negative and N positive tumors (p = 0.017).
Table 5C: Comparison of PET and DWI values between different tumor M stages
Parameters |
M0 |
M1 |
p values |
|---|---|---|---|
SUVmax |
19.53 ± 8.25 |
24.89 ± 14.10 |
0.28 |
SUVmean |
10.06 ± 5.07 |
14.10 ± 8.00 |
0.17 |
rSUVmax |
2.28 ± 1.18 |
1.76 ± 0.13 |
0.45 |
ADCmin |
0.63 ± 0.13 |
0.50 ± 0.13 |
0.08 |
ADCmean |
0.89 ± 0.14 |
0.81 ± 0.09 |
0.20 |
ADCmax |
1.43 ± 0.81 |
1.18 ± 0.12 |
0.41 |
rADCmin |
0.70 ± 0.11 |
0.64 ± 0.14 |
0.34 |
SUVmax/ADCmin |
33.64 ± 16.87 |
52.30 ± 32.10 |
0.09 |
SUVmax/ADCmean |
22.50 ± 9.02 |
30.15 ± 14.98 |
0.16 |
The results of histopathological analysis are shown in Table 6. Histopathological findings varied with a wide spectrum in the patients.
Table 6: Estimated histopathological parameters of cervical cancer
Parameters |
M ± SD |
Median |
Range |
|---|---|---|---|
Cell count |
1780 ± 334 |
1795 |
1290–2515 |
Ki 67, % |
48.38 ± 18.42 |
49 |
22–76 |
Total nucleic area, μm² |
113665 ± 32709 |
109711 |
58914–181174 |
Average nucleic area, μm² |
63.72 ± 13.66 |
59.87 |
38.21–94.43 |
Epithel area (%) |
30.57 ± 16.55 |
30 |
2–60 |
Stroma area (%) |
69.43 ± 16.55 |
70 |
40–98 |
Furthermore, histopathological findings were correlated with PET and DWI parameters (Table 7A–7C). KI 67 correlated statistically significant with SUVmax (r = 0.59, p = 0.005), and SUVmean (0.45, p = 0.04) (Figure 2A). SUVmax showed strong correlations with epithelial area (r = 0.71, p = 0.001) and stromal areas (r = –0.71, p = 0.001) (Figure 2B). Also SUVmean correlated with epithelial area (r = 0.45, p = 0.04) and stromal areas (r = –0.45, p = 0.04) (Table 7A). In addition, ADCmin correlated inversely with KI 67 (r = –0.48, p = 0.03) (Table 7B, Figure 3).
Table 7A: Correlations between PET and histopathological parameters
Parameters |
Cell count |
Ki 67 |
Total nucleic area |
Average nucleic area |
Epithelial area |
Stromal area |
|---|---|---|---|---|---|---|
SUVmax |
p = 0.24 |
p = 0.59 |
p = 0.29 |
p = 0.16 |
p = 0.71 |
p = –0.71 |
P = 0.29 |
p = 0.005 |
P = 0.19 |
P = 0.48 |
p = 0.001 |
p = 0.001 |
|
SUVmean |
p = 0.03 |
p = 0.45 |
p = 0.13 |
p = 0.17 |
p = 0.45 |
p = –0.45 |
P = 0.88 |
P = 0.04 |
P = 0.58 |
P = 0.45 |
P = 0.04 |
P = 0.04 |
|
rSUVmax |
p = 0.11 |
p = –0.09 |
p = –0.05 |
p = –0.24 |
p = 0.003 |
p = –0.003 |
P = 0.65 |
P = 0.69 |
P = 0.83 |
P = 0.29 |
P = 0.99 |
P = 0.99 |
Table 7B: Correlations between DWI and histopathological parameters
Parameters |
Cell count |
Ki 67 |
Total nucleic area |
Average nucleic area |
Epithelial area |
Stromal area |
|---|---|---|---|---|---|---|
ADCmean |
p = 0.01 |
p = –0.34 |
p = –0.19 |
p = –0.29 |
p = 0.31 |
p = 0.31 |
P = 0.96 |
P = 0.14 |
P = 0.39 |
P = 0.21 |
P = 0.17 |
P = 0.17 |
|
ADCmin |
p = –0.07 |
p = –0.48 |
p = –0.11 |
p = –0.07 |
p = 0.07 |
p = 0.07 |
P = 0.77 |
p = 0.03 |
P = 0.65 |
P = 0.76 |
P = 0.77 |
P = 0.77 |
|
ADCmax |
p = 0.09 |
p = –0.24 |
p = –0–16 |
p = –0.30 |
p = 0.08 |
p = -0.08 |
P = 0.69 |
P = 0.29 |
P = 0.50 |
P = 0.18 |
P = 0.72 |
P = 0.72 |
|
rADCmin |
p = –0.17 |
p = –0.35 |
p = 0.03 |
p = 0.22 |
p = -0,88 |
p = 0,09 |
P = 0.46 |
P = 0.12 |
P = 0.88 |
P = 0.34 |
P = 0,70 |
P = 0,70 |
Table 7C: Correlations between combined PET/DWI and histopathological parameters
Parameters |
Cell count |
Ki 67 |
Total nucleic area |
Average nucleic area |
Epithelial area |
Stromal area |
|---|---|---|---|---|---|---|
SUVmax /ADCmin |
p = 0.149 |
p = 0.71 |
p = 0.27 |
p = 0.19 |
p = 0.27 |
p = 0.27 |
P = 0.52 |
p ≤ 0.001 |
P = 0.23 |
P = 0.39 |
P = 0.24 |
P = 0.24 |
|
SUVmax/ADCmean |
p = 0.20 |
p = 0.75 |
p = 0.41 |
p = 0.35 |
p = 0.49 |
p = –0.49 |
P = 0.39 |
p ≤ 0.001 |
P = 0.07 |
P = 0.12 |
p = 0.03 |
p = 0.03 |
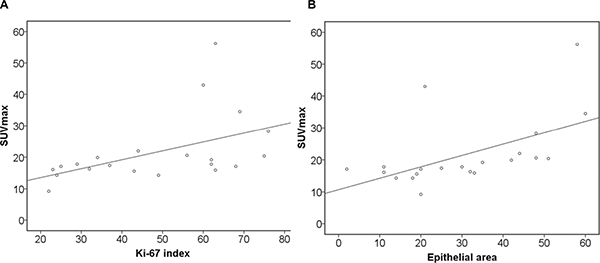
Figure 2: Associations between glucose metabolism and histopathological parameters. (A) Correlation between SUVmax and KI 67 (r = 0.59, p = 0.005). (B) Correlation between SUVmax and epithelial area (r = 0.71, p = 0.001).
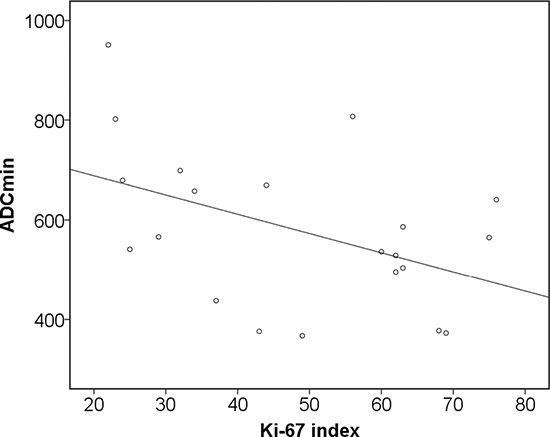
Figure 3: Associations between DWI and histopathology. Correlation between ADCmin and KI 67 (r = −0.48, p = 0.03).
Significant correlations were also observed between KI 67 and the calculated combined parameters SUVmax/ADCmin (r = 0.71, p = 0.001) and SUVmax/ADCmean (0.75, p = 0.001) (Figure 4A and 4B). SUVmax/ADCmean showed moderate correlations with epithelial (r = 0.49, p = 0.03) and stromal areas (r = –0.49, p = 0.03). Finally, SUVmax/ADCmean tended to correlate with total nucleic area (r = 0.41, p = 0.07) (Table 7C).
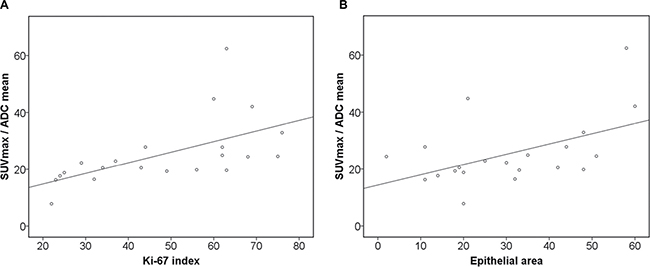
Figure 4: Associations between combined PET/DWI parameters and histopathology. (A) Correlation between SUVmax/ADCmean and KI 67 (r = 0.75, p = 0.001). (B) Correlation between SUVmax/ADCmean and epithelial area (r = 0.49, p = 0.03)
DISCUSSION
The present study identified significant associations between tumor stage, histopathological findings and parameters from simultaneous PET/MRI in uterine cervical cancer.
According to the literature, PET and DWI are independent imaging modalities, which reflect different clinical and histological features in several tumors [8, 12]. For example, Preda et al. reported that both SUV and ADC values can be used as prognosis factors in patients with head and neck cancer [9]. Other authors confirmed these results [10].
In contrast to other malignancies, there were only few studies regarding PET and DWI findings in uterine cervical cancer. Previously, some authors identified significant associations between PET and DWI parameters in uterine cervical cancer [3, 8]. For instance, Brandmaier et al. found significant inverse correlations between SUVmax and ADCmin (r = –0.532, p = 0.05) and between SUVmean and ADCmin (r = –0.403, p = 0.03). in primary tumors [8]. Furthermore, the identified correlations were stronger in recurrent tumors: SUVmax and ADCmin (r = –0.747, p = 0.002) and SUVmean and ADCmin (r = –0.773, p = 0.001) [8]. Also Grueneisen et al. studied correlations between SUVmax and ADCmin in primary and recurrent cervical cancer [3]. It has been shown, however, that the parameters correlated well in primary tumors and associated primary lymph node metastases, but not in recurrent cancer lesions [3]. In contrast to these reports, in the study of Ho et al. no significant correlations between ADCmin as well ADCmean and SUVmax or SUVmean were identified [11]. Nevertheless, the authors calculated two new indexes, namely rADCmin as a ratio ADCmin/ADCmean and rSUVmax as a ratio SUVmax/SUVmean [11]. It has been shown that both parameters correlated well together in adenocarcinomas and adenosquamous tumors but not in squamous carcinomas [11].
In our study, no significant correlations between PET and ADC parameters were found. In addition, also the calculated indexes rADCmin and and rSUVmax did not correlate together. This finding is in agreement with the results of Ho et al. [11] because of the fact that our patients had predominantly squamous cell carcinomas.
Some previous reports indicated that PET and DWI parameters can be used as predictor of tumor stage and grading. So Micco et al. observed significant correlations between ADCmean, SUVmax, metabolic tumor volume, total lesion glycolysis and occurrence of lymph node metastasis [10]. Grueneisen et al. found that ADCmin was statistically significant lower in 2b-4 T stage tumors in comparison to T1-2a carcinomas [3]. Furthermore, ADCmin had a tendency to be lower in tumors with nodal metastases [3]. Other authors confirmed these findings [10, 13].
In the study of Husby et al, ADC was associated with deep myometrial invasion in cervical cancer [14]. It has been shown that invasive tumors had significantly lower mean tumor ADC values in comparison to tumors without myometrial invasion [14]. Similar results were reported for cervical cancer with parametral invasion [15].
Moreover, according to the literature, ADC can also distinguish different histological tumor types of cervical cancer [16]. For example, Liu et al. reported that mean ADC value and minimum ADC value of squamous cell carcinoma were significantly lower than that of adenocarcinoma [16]. However, Grueneisen did not found significant differences in SUV as well ADC values between squamous cell carcinomas and adenocarcinomas [3].
Previous reports indicated that SUV and ADC can be used to distinguish well or moderately differentiated carcinomas and poorly differentiated cervical tumors [3].
For example, it has been shown that G3 tumors had statistically significant lower ADCmin values and higher SUVmax and SUVmean values [3]. Micco et al, however, could not identify significant differences in SUV and ADC values between several tumor grades [10].
In the present study, also an analysis of PET and DWI parameters in dependency on tumor stage and grading was performed. No significant associations between different DWI and PET parameters as well calculated indexes in moderately and poorly differentiated tumors were identified. This finding indicates that tumor grading does not influence PET and DWI in uterine cervical cancer. However, we found significant associations between PET and DWI parameters and different tumor stages. Firstly, ADCmin and rADCmin were statistically significant lower in tumors with nodal metastases. Secondly, ADCmin tended to be lower (p = 0.082) in tumors with distant metastases (M stage). Interestingly, also SUVmax/ADCmin had a tendency to be higher (p = 0.095) in M positive carcinomas. To the best of our knowledge, associations between imaging and M stage have not been reported previously in uterine cervical cancer. These findings have a high clinical relevance and suggests that ADCmin as well SUVmax/ADCmin may be used as M-stage markers. No significant differences were identified between the tumors in dependency to T stage. It may be explained to the fact that, in contrast to previous reports, our patients had predominantly 2b and 4 stages, i.e. advanced tumors. Overall, our results confirmed the hypothesis of some previous studies that PET and DWI parameters can be used as additional predictors for tumor stage.
Presumably, the identified associations base on associations between PET and DWI parameters and histopathological features. In contrast to other tumors, such as head and neck cancers [12], breast carcinoma [17] or lung cancer [18] there were no reports regarding possible correlations between PET/MRI and histopathological findings in uterine cervical cancer.
In our study, a complex analysis of relationships between PET, DWI, and histopathology was performed. Thereby several significant correlations between the investigated parameters were identified. Firstly, ADCmin and SUVmax as well SUVmean correlated significantly with KI 67. Therefore, these parameters can be used to assess proliferation potential in cervical cancer. Secondly, PET parameters did not reflect cell count of the investigated tumors. However, the present study demonstrated well correlations between PET parameters and tumor architecture, in particular epithelial and stromal areas. This finding is very interestingly and may explain missing correlations between SUV fractions and cellularity in uterine cervical cancer and similar results of a previous analysis regarding squamous cell carcinomas in the head and neck region [12]. In fact, each tumor consists on tumor cells and stroma. Only tumor cells have high metabolic activity and influence PET parameters. Therefore, SUVmax and SUVmean reflect tumor cell count/area, but not overall cell count/area.
Thirdly, in our study, no significant correlations between different ADC values and cell count were detected. This finding is difficult to explain. According to the literature, in most reported malignancies, different ADC values correlated significantly with cell count [19–22]. It has been shown that especially ADCmin reflected tumor cellularity [19, 20, 23]. However, there were several lesions, in which also no significant correlations between DWI parameters and cell count were found [29]. For example, Wu et al. did not find any correlations between ADC values and tissue cellularity in different lymphomas [24]. It may be related to the fact that not only cellularity but other histopathological features such as architectural structure [25], extracellulary matrix [20] or nucleic areas [18, 19, 25] may play a role here.
Our study showed that the calculated combined parameters SUVmax/ADCmean and SUVmax/ADCmin demonstrated significant associations with KI 67, epithelial and stromal areas. Moreover, the correlations KI 67 vs the combined parameters were stronger than those vs ADCmin, SUVmax or SUVmean. Therefore, these combined parameters can better predict proliferation potential of uterine cervical cancer.
Furthermore, the ratio SUVmax/ADCmean tended to correlate with total nucleic area. Nuclear size was reported to be a prognostic indicator in several malignancies [26]. It has also been shown that lesions with large nucleic areas had a worse prognosis [26]. The phenomena identified in our study confirmed the assumption that PET and DWI parameters complement one another and they can be combined together.
The present study is limited to the relatively small number of patients. Clearly, further investigations with more patients are needed to confirm the identified associations between clinical, imaging and histopathological parameters.
In conclusion, our results quantitatively demonstrated significant correlations between PET and DWI parameters and different histopathological features in uterine cervical cancer. N positive tumors showed statistically significant lower ADCmin and rADCmin values. Both SUV values, ADCmin, as well combined parameters SUVmax/ADCmin and SUVmax/ADCmean correlated statistically significant with KI 67 and can be used to estimate the proliferation potential of tumors. Finally, both SUV values correlated strong with epithelial area of tumor and, therefore, reflected metabolic active areas but not overall tumor cell count.
MATERIALS AND METHODS
This prospective study was approved by the institutional review board (Ethic Committee of the Medical Faculty, University of Leipzig) and all patients gave written informed consent.
Patients
Overall, 21 patients (mean age, 56.2 ± 14.5 years; median age, 57 years; range, 32-79 years) with histologically proven primary uterine cervical cancer were involved into the study (Table 1).
Whole-body PET/MRI
All 21 patients underwent a whole body simultaneous18F-FDG PET/MRI (Magnetom Biograph mMR - Biograph, Siemens Health Care Sector, Erlangen, Germany) which was performed from the upper thigh to the skull with 5 minutes per bed position. PET images were reconstructed using the iterative ordered subset expectation maximization algorithm with 3 iterations and 21 subsets, a Gaussian filter with 4 mm full width at half maximum (FWHM), and a 256 x 256 image matrix. Attenuation correction of the PET data was performed using a four-tissue (fat, soft tissue, air, background) model attenuation map, which was generated from a Dixon-Vibe MR sequence as previously described by Martinez-Möller et al. 2009 [27].
Radiotracer administration was performed intravenously after a fasting period of at least 6 hours with a body weight-adapted dose of 18F-FDG (4 MBq/kg, range: 152 – 442 MBq, mean±std: 285±70 MBq). PET/MRI image acquisition started on average 122 minutes after 18F-FDG application. Due to radiotracer elimination via the urinary tract, which may influence evaluation of pelvic PET images, all patients received a bladder catheter prior to PET/MRI examination.
Pelvic MRI
Additionally, pelvic MRI was obtained in all cases. For pelvic MRI the following sequences were applied: a transverse T2 turbo spin echo (TSE) sequence, a sagittal T2 TSE sequence, a transverse T1 TSE sequence, a transverse fat saturated T1 TSE after intravenous application of contrast medium (0.1 mmol/kg body weight Gadobutrol, Bayer Healthcare, Germany), a sagittal post contrast T1 TSE, and a transverse diffusion-weighted echo-planar imaging (EPI) sequence by using two b-values: b0 and b1000 s/mm2.
Table 8 provides detailed information for all sequences of the pelvic study protocol.
Table 8: Sequences used in the study
Sequences |
TR, ms |
TE, ms |
Flip angle |
Slice thickness |
|---|---|---|---|---|
Transverse T2 TSE |
5590 |
105 |
140 |
7 |
Sagitta l T2 TSE |
4110 |
131 |
150 |
3 |
Transverse T1 TSE |
1310 |
12 |
140 |
7 |
Transverse post contrast fat saturated T1 TSE |
912 |
12 |
140 |
7 |
sagittal post contrast T1 TSE |
593 |
12 |
140 |
5 |
EPI 2d DWI |
4900 |
105 |
90 |
3 |
Image analysis
Imaging data were analyzed with dedicated viewing software (syngo.via, Siemens Health Care, Erlangen Germany). This was performed by two board certified physicians, a radiologist with 7 years’ experience in gynecological imaging and a nuclear medicine specialist with 8 years’ experience in oncological hybrid imaging.
For PET-imaging, focal lesions with glucose uptake greater than the surrounding tissue were considered suspicious for malignancy. A volume of interest (VOI) was drawn around these lesions using the VOI-isocontour function of the software with a threshold of 40% of SUVmax (Figure 5A and 5B). Mean and maximum standardized uptake values were noted (SUVmean and SUVmax). Furthermore, in every case a relative SUVmax as a ratio SUVmax/SUVmean was calculated as reported previously [11, 28].
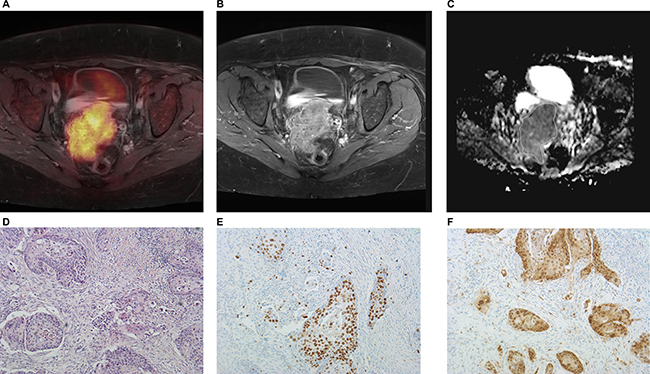
Figure 5: Imaging and histopathological findings in a patient with T4a N0 M0 uterine cervical cancer. (A) fused 18F-FDG-PET/T1 weighted TSE MR image of the lesion, SUVmax = 16.3, SUVmean = 8.68. (B) Postcontrast T1 weighted TSE showing a large lesion in the uterine cervix. (C) ADC map of the tumor. The ADC values (× 10−3 mm2s−1) of the lesion are as follows: ADCmin = 0.70, ADCmean = 0.99, and ADCmax = 1.33. (D–F). Histopathological images. H&E image (D): cell count is 1971, total nucleic area = 108240 μm², average nucleic area = 54.97 μm². (E) Immunohistochemical stain (MIB-1 monoclonal antibody). Ki 67 index is 32%. (F) Immunohistochemical stain (p16 antibody). Epithelial area is 32% and stromal area is 68%.
Only pelvic MR investigations were analyzed in the study. Detected primary cervical cancers were analyzed with cognitive fusion of T2 weighted images and DWI images. ADC maps were automatically generated by the scanner software. For calculation of diffusion parameters of the tumor, the borders of tumor tissue were marked freehand with a polygonal region of interest (ROI) in each slice on the ADC maps (Figure 5C). In all tumors minimal ADC values (ADCmin), mean ADC values (ADCmean), and maximal ADC values (ADCmax) were estimated. Additionally, a relative ADCmin as a ratio ADCmin/ ADCmean was calculated [11].
Furthermore, in each case combined parameters PET and DWI were calculated as follows: SUVmax divided by ADCmin (SUVmax/ADCmin) and SUVmax divided by ADCmean (SUVmax/ADCmean) [12, 29].
Histopathological analysis
All histopathological images were analyzed by one pathologist (10 years of experience).
In all cases the diagnosis was confirmed histopathologically by tumor biopsy. The biopsy specimens were deparaffinized, rehydrated and cut into 5 μm slices. Furthermore, the histological slices were stained by hematoxilin and eosin (H&E), MIB 1 monoclonal antibody (DakoCytomation, Denmark), and p16 (Cintec Histology, Roche, Germany) according to previous descriptions [30].
All histopathological images were digitalized with a research microscope Jenalumar and camera Diagnostic instruments 4.2 (Zeiss, Jena, Germany) and saved as uncompressed Tagged Image File Format (TIFF). The digitalized images were analyzed by using ImageJ software 1.48v (National Institutes of Health Image program) with a Windows operating system [31]. All images were converted to a black and white binary image by setting the image threshold as reported previously [23, 31]. The threshold selected image parts were further analyzed using the Analyze Particles tool [23]. The following histopathological parameters were estimated: cell count, proliferation index KI 67, total nucleic area, average nucleic area, and epithelial/stromal areas of tumors. Cell count was estimated as a number of all nuclei on H&E stained images (Figure 5D). Proliferation index KI 67 (%) was calculated as percentage of stained nuclei on MIB 1 monoclonal antibody stained images (Figure 5E). Thereby the areas with the highest number of positive tumor nuclei were selected. Total nucleic area (μm²) was given as area of stained nuclei on H&E stained images. Furthermore, also an average nucleic area (μm²) as a total nucleic area divided by number of nuclei was calculated. In addition, stained area on p16-stained images divided by total area of image x 100% (in every case, the total area of image was 0.16 mm2) was estimated. Because to the fact that only epithelial cells can be stained by p16 [30], the stained area was acquired as epithelial area of tumor (Figure 5F). Finally, stromal area of tumor (%) as a nonstained area on p16-stained images divided by total area of image x 100% was also calculated.
In every case, 2 five high power fields (0.16 mm2 per field) with a magnification of x400 were analysed.
Statistical analysis
Statistical analysis was performed using IBM SPSS 20™ (SPSS Inc., Chicago, IL, USA). Collected data were evaluated by means of descriptive statistics (absolute and relative frequencies). All measurements were non-normally distributed according to Kolmogorov-Smirnov-test. Spearman’s correlation coefficient was used to analyze associations between ADC and SUV values and histological parameters. P values ≤ 0.05 were considered as statistically significant.
CONFLICTS OF INTEREST
None.
REFERENCES
1. Sun H, Xin J, Zhang S, Guo Q, Lu Y, Zhai W, Zhao L, Peng W, Wang B. Anatomical and functional volume concordance between FDG PET, and T2 and diffusion-weighted MRI for cervical cancer: a hybrid PET/MR study. Eur J Nucl Med Mol Imaging. 2014; 41:898–905.
2. Queiroz MA, Kubik-Huch RA, Hauser N, Freiwald-Chilla B, von Schulthess G, Froehlich JM, Veit-Haibach P. PET/MRI and PET/CT in advanced gynaecological tumours: initial experience and comparison. Eur Radiol. 2015; 25:2222–30.
3. Grueneisen J, Schaarschmidt BM, Heubner M, Aktas B, Kinner S, Forsting M, Lauenstein T, Ruhlmann V, Umutlu L. Integrated PET/MRI for whole-body staging of patients with primary cervical cancer: preliminary results. Eur J Nucl Med Mol Imaging. 2015; 42:1814–24.
4. Wetter A, Lipponer C, Nensa F, Beiderwellen K, Olbricht T, Rübben H, Bockisch A, Schlosser T, Heusner TA, Lauenstein TC. Simultaneous 18F choline positron emission tomography/magnetic resonance imaging of the prostate: initial results. Invest Radiol. 2013; 48:256–62.
5. Pace L, Nicolai E, Luongo A, Aiello M, Catalano OA, Soricelli A, Salvatore M. Comparison of whole-body PET/CT and PET/MRI in breast cancer patients: lesion detection and quantitation of 18Fdeoxyglucose uptake in lesions and in normal organ tissues. Eur J Radiol. 2014; 83:289–96.
6. Grueneisen J, Beiderwellen K, Heusch P, Buderath P, Aktas B, Gratz M, Forsting M, Lauenstein T, Ruhlmann V, Umutlu L. Correlation of standardized uptake value and apparent diffusion coefficient in integrated whole-body PET/MRI of primary and recurrent cervical cancer. PLoS One. 2014; 9:e96751.
7. Nakamura K, Joja I, Kodama J, Hongo A, Hiramatsu Y. Measurement of SUVmax plus ADCmin of the primary tumour is a predictor of prognosis in patients with cervical cancer. Eur J Nucl Med Mol Imaging. 2012; 39:283–90.
8. Brandmaier P, Purz S, Bremicker K, Höckel M, Barthel H, Kluge R, Kahn T, Sabri O, Stumpp P. Simultaneous [18F]FDG-PET/MRI: Correlation of Apparent Diffusion Coefficient (ADC) and Standardized Uptake Value (SUV) in Primary and Recurrent Cervical Cancer. PLoS One. 2015; 10:e0141684.
9. Preda L, Conte G, Bonello L, Giannitto C, Travaini LL, Raimondi S, Summers PE, Mohssen A, Alterio D, Cossu Rocca M, Grana C, Ruju F, Bellomi M. Combining standardized uptake value of FDG-PET and apparent diffusion coefficient of DW-MRI improves risk stratification in head and neck squamous cell carcinoma. Eur Radiol. 2016 Mar 10. PMID: 26965504.
10. Miccò M, Vargas HA, Burger IA, Kollmeier MA, Goldman DA, Park KJ, Abu-Rustum NR, Hricak H, Sala E. Combined pre-treatment MRI and 18F-FDG PET/CT parameters as prognostic biomarkers in patients with cervical cancer. Eur J Radiol. 2014; 83:1169–76.
11. Ho KC, Lin G, Wang JJ, Lai CH, Chang CJ, Yen TC. Correlation of apparent diffusion coefficients measured by 3T diffusion-weighted MRI and SUV from FDG PET/CT in primary cervical cancer. Eur J Nucl Med Mol Imaging. 2009; 36:200–8.
12. Surov A, Stumpp P, Meyer HJ, Gawlitza M, Hoehn AK, Boehm A, Sabri O, Kahn T, Purz S. Simultaneous 18F-FDG-PET/MRI: Associations between diffusion, glucose metabolism and histopathological parameters in patients with head and neck squamous cell carcinoma. Oral Oncol. 2016; 58:14–20.
13. Crivellaro C, Signorelli M, Guerra L, De Ponti E, Buda A, Dolci C, Pirovano C, Todde S, Fruscio R, Messa C. 18F-FDG PET/CT can predict nodal metastases but not recurrence in early stage uterine cervical cancer. Gynecol Oncol. 2012; 127:131–5.
14. Husby JA, Reitan BC, Biermann M, Trovik J, Bjørge L, Magnussen IJ, Salvesen ØO, Salvesen HB, Haldorsen IS. Metabolic Tumor Volume on 18F-FDG PET/CT Improves Preoperative Identification of High-Risk Endometrial Carcinoma Patients. J Nucl Med. 2015; 56:1191–8.
15. Park JJ, Kim CK, Park SY, Park BK. Parametrial invasion in cervical cancer: fused T2-weighted imaging and high-b-value diffusion-weighted imaging with background body signal suppression at 3 T. Radiology. 2015; 274:734-41.
16. Liu Y, Ye Z, Sun H, Bai R. Clinical Application of Diffusion-Weighted Magnetic Resonance Imaging in Uterine Cervical Cancer. Int J Gynecol Cancer. 2015; 25:1073–8.
17. Choi BB, Kim SH, Kang BJ, Lee JH, Song BJ, Jeong SH, Yim HW. Diffusion-weighted imaging and FDG PET/CT: predicting the prognoses with apparent diffusion coefficient values and maximum standardized uptake values in patients with invasive ductal carcinoma. World J Surg Oncol. 2012; 10:126.
18. Liu LP, Zhang XX, Cui LB, Li J, Yang JL, Yang HN, Zhang Y, Zhou Y, Tang X, Qi S, Fang Y, Zhang J, Yin H. Preliminary comparison of diffusion-weighted MRI and PET/CT in predicting histological type and malignancy of lung cancer. Clin Respir J. 2015; doi:10.1111/crj.12316.
19. Surov A, Gottschling S, Mawrin C, Prell J, Spielmann RP, Wienke A, Fiedler E. Diffusion weighted imaging in meningioma: prediction of tumor grade and association with histopathological parameters. Transl Oncol. 2015; 8:517–23.
20. Fornasa F. Diffusion-weighted Magnetic Resonance Imaging: What Makes Water Run Fast or Slow? J Clin Imaging Sci. 2011; 1:27.
21. Chen L, Liu M, Bao J, Xia Y, Zhang J, Zhang L, Huang X, Wang J. The correlation between apparent diffusion coefficient and tumor cellularity in patients: a meta-analysis. PLoS One. 2013; 8:e79008.
22. Langer DL, van der Kwast TH, Evans AJ, Plotkin A, Trachtenberg J, Wilson BC, Haider MA. Prostate tissue composition and MR measurements: investigating the relationships between ADC, T2, K(trans), v(e), and corresponding histologic features. Radiology. 2010; 255:485-94.
23. Surov A, Caysa H, Wienke A, Spielmann RP, Fiedler E. Correlation between different ADC fractions, cell count, Ki-67, total nucleic areas and average nucleic areas in meningothelial meningiomas. Anticancer Res. 2015; 35:6841-6.
24. Wu X, Pertovaara H, Dastidar P, Vornanen M, Paavolainen L, Marjomäki V, Järvenpää R, Eskola H, Kellokumpu-Lehtinen PL. ADC measurements in diffuse large B-cell lymphoma and follicular lymphoma: a DWI and cellularity study. Eur J Radiol. 2013; 82:e158–64.
25. Driessen JP, Caldas-Magalhaes J, Janssen LM, Pameijer FA, Kooij N, Terhaard CH, Grolman W, Philippens ME. Diffusion-weighted MR Imaging in Laryngeal and Hypopharyngeal Carcinoma: Association between Apparent Diffusion Coefficient and Histologic Findings. Radiology. 2014; 272:456–63.
26. Abdalla F, Boder J, Markus R, Hashmi H, Buhmeida A, Collan Y. Correlation of nuclear morphometry of breast cancer in histological sections with clinicopathological features and prognosis. Anticancer Res. 2009; 29:1771-6.
27. Martinez-Möller A, Souvatzoglou M, Delso G, Bundschuh RA, Chefd’hotel C, Ziegler SI, Navab N, Schwaiger M, Nekolla SG. Tissue classification as a potential approach for attenuation correction in whole-body PET/MRI: evaluation with PET/CT data. J Nucl Med. 2009; 50:520–6.
28. Meyer M. Standardized uptake values of 18F-FDG uptake by lung neoplasms: a meta-analysis of 437 cases. J Nucl Med. 2007; 48:352P.
29. Baba S, Isoda T, Maruoka Y, Kitamura Y, Sasaki M, Yoshida T, Honda H. Diagnostic and prognostic value of pretreatment SUV in 18F-FDG/PET in breast cancer: comparison with apparent diffusion coefficient from diffusion-weighted MR imaging. J Nucl Med. 2014; 55:736–42.
30. Liang J, Mittal KR, Wei JJ, Yee H, Chiriboga L, Shukla P. Utility of p16INK4a, CEA, Ki67, P53 and ER/PR in the differential diagnosis of benign, premalignant, and malignant glandular lesions of the uterine cervix and their relationship with Silverberg scoring system for endocervical glandular lesions. Int J Gynecol Pathol. 2007; 26:71–5.
31. Aoyagi T, Shuto K, Okazumi S, Hayano K, Satoh A, Saitoh H, Shimada H, Nabeya Y, Kazama T, Matsubara H. Apparent diffusion coefficient correlation with oesophageal tumour stroma and angiogenesis. Eur Radiol. 2012; 22:1172–7.

