INTRODUCTION
Cancer is a substantial public health burden, with an estimated 1.7 million new cancer cases and 0.6 million cancer-related deaths in the United States in 2016 [1]. Although the etiology of carcinogenesis has not yet been fully elucidated, many lines of evidence suggest that cancer is a multifactorial disease caused by intricate interactions between multiple hereditary and environmental factors [2, 3]. Numerous studies have demonstrated that inflammation is critically implicated in the development of some cancers [4, 5]. In view of this, it is plausible that genetic polymorphisms in inflammation-related genes could modify cancer susceptibility [6-8].
Nuclear factor-kappa B (NF-κB) is a pleiotropic transcription factor discovered by Sen and Baltimore in 1986 [9]. In mammals, the NF-κB family consists of five members: c-Rel (Rel), Rel B, p65 (RelA), p50/p105 (NF-κB1), and p52/p100 (NF-κB2) [10]. This group of molecules function as key regulators of a variety of genes implicated in diverse biological events including cell survival, apoptosis, inflammation, differentiation, and autophagy [11, 12]. Recently, high levels of NF-κB have been observed in many cancers, including pancreatic cancer [13], lung cancer [14], colorectal cancer [15], breast cancer [16], melanoma [17], and multiple myeloma [18]. Although various dimeric forms of NF-κB exist, the most common is the p50 and p65/RelA heterodimer, encoded by the NFKB1 and RelA genes, respectively [19].
The human NFKB1 gene, spanning 156-kb, is located on chromosome 4q23-q24 and encodes a 105 kD protein (p105) which is cleaved into an active subunit (p50) [20]. Several variations have been identified in the NFKB1 gene, including rs72696119 (C>G), rs28362491 (-94 ins/del ATTG), rs4648068 (A>G), and rs12509517 (G>C) [21]. Among these, NFKB1 rs28362491, namely the -94insertion/deletion ATTG polymorphism, is potentially functional and the most widely investigated [21]. This modification occurs between two important regulatory elements (activator protein 1 and κB binding site) in the promoter region of the NFKB1 gene. The deletion of four bases (ATTG) reduces or prevents the binding to nuclear proteins and leads to lower transcript levels of the NFKB1 gene, thereby changing mRNA stability and regulating translation efficiency [21, 22].
Numerous case-control studies have assessed the association between the NFKB1 -94ins/delATTG promoter polymorphism and cancer risk, with discrepant results. While some studies indicated an increased risk for some types of cancers [23-25], other studies showed instead a decreased risk, or no association [26, 27]. Several meta-analyses attempted to solve the controversy, but did not yield consistent results [28-33]. To provide a more precise evaluation of such association, we performed a comprehensive, updated meta-analysis. In addition, to minimize random errors and strengthen the robustness of our conclusions, we also performed trial sequential analysis (TSA).
RESULTS
Study characteristics
The study selection process for this meta-analysis is shown in Figure 1, 258 potentially relevant published records were retrieved from PubMed, EMBASE, China National Knowledge infrastructure (CNKI), and WANFANG databases. After screening the titles and reading the abstracts, 69 studies remained and were carefully reviewed. Among these, 23 publications were further excluded: 7 were case-only studies [34-39]; 6 were meta-analyses [28-33]; 5 deviated from Hardy-Weinberg equilibrium (HWE) [40-44]; 3 were duplicated publications [45-47]; 1 was a review [48], and 1 lacked sufficient data to calculate odds ratio (OR) and 95% confidence interval (CI) [49]. Thus, 46 publications were included in the final analysis [23-27, 48, 50-89]. Among these, publications that contained different case groups but used the same controls, or that studied one cancer type in different populations, were considered separate studies.
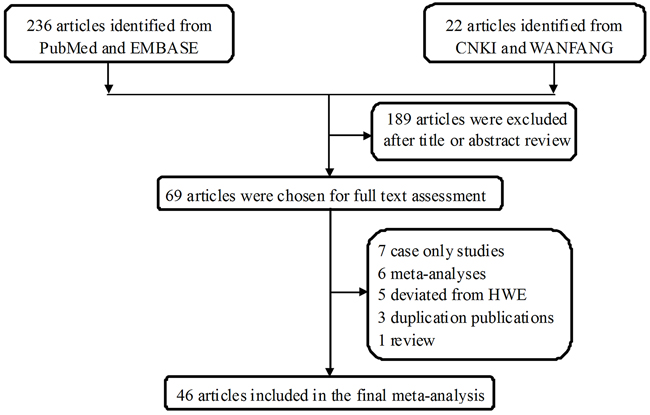
Figure 1: Flowchart of the study inclusion protocol.
After this selection procedure, 50 studies extracted from 46 publications with 18,299 cases and 23,484 controls ultimately entered our final meta-analysis (Table 1). 38 of these studies included Asians subjects, and 12 included Caucasians. Regarding cancer types, 6 studies addressed hepatocellular carcinoma, 5 lung cancer, 4 colorectal cancer, 4 nasopharyngeal carcinoma, 4 prostate cancer, 4 ovarian cancer, 3 bladder cancer, 3 gastric cancer, 3 cervical cancer, 2 oral squamous cell carcinoma, 2 breast cancer, and 10 studies addressed other cancers. Moreover, 40 studies used a population-based design, and 10 were hospital-based. 19 studies had a quality score >9, and the remaining 31 had a quality score ≤9.
Table 1: Characteristics of the studies included in the current meta-analysis
Surname |
Year |
Cancer type |
Country |
Ethnicity |
Control Source |
Genotype method |
Case |
Control |
MAF |
HWE |
Score |
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
II |
ID |
DD |
All |
II |
ID |
DD |
All |
||||||||||
Lin [25] |
2006 |
OSCC |
China |
Asian |
HB |
PCR-PAGE |
59 |
103 |
50 |
212 |
43 |
100 |
58 |
201 |
0.54 |
0.993 |
7 |
Riemann [26] |
2006 |
Colorectal cancer |
Germany |
Caucasian |
HB |
Pyro sequencing |
54 |
58 |
27 |
139 |
118 |
141 |
48 |
307 |
0.39 |
0.586 |
9 |
Riemann [26] |
2006 |
CLL |
Germany |
Caucasian |
HB |
Pyro sequencing |
18 |
41 |
13 |
72 |
118 |
141 |
48 |
307 |
0.39 |
0.586 |
9 |
Riemann [26] |
2006 |
RCC |
Germany |
Caucasian |
HB |
Pyro sequencing |
47 |
76 |
17 |
140 |
118 |
141 |
48 |
307 |
0.39 |
0.586 |
9 |
Bu [48] |
2007 |
Melanoma |
Sweden |
Caucasian |
HB |
PCR-RFLP |
67 |
84 |
34 |
185 |
116 |
255 |
67 |
438 |
0.44 |
0.000 |
10 |
Riemann [83] |
2007 |
Bladder cancer |
Germany |
Caucasian |
HB |
Pyro sequencing |
88 |
124 |
30 |
242 |
118 |
141 |
48 |
307 |
0.39 |
0.586 |
10 |
Lehnerdt [27] |
2008 |
HNSCC |
Germany |
Caucasian |
HB |
Pyro sequencing |
132 |
179 |
53 |
364 |
118 |
141 |
48 |
307 |
0.39 |
0.586 |
8 |
He [82] |
2009 |
HCC |
China |
Asian |
HB |
PCR-RFLP |
83 |
84 |
35 |
202 |
97 |
183 |
124 |
404 |
0.53 |
0.070 |
9 |
He [89] |
2009 |
HCC |
China |
Asian |
HB |
PCR-RFLP |
55 |
65 |
30 |
150 |
70 |
136 |
94 |
300 |
0.54 |
0.130 |
8 |
Lo [24] |
2009 |
Gastric cancer |
China |
Asian |
HB |
PCR |
62 |
89 |
31 |
182 |
20 |
62 |
34 |
116 |
0.56 |
0.361 |
7 |
Zhang [81] |
2009 |
Prostate cancer |
China |
Asian |
HB |
PCR-PAGE |
46 |
57 |
14 |
117 |
44 |
68 |
31 |
143 |
0.45 |
0.624 |
8 |
Zhou [80] |
2009 |
NPC |
China |
Asian |
HB |
PCR-PAGE |
74 |
67 |
22 |
163 |
71 |
90 |
42 |
203 |
0.43 |
0.177 |
7 |
Tang [79] |
2010 |
Bladder cancer |
China |
Asian |
HB |
PCR–PAGE |
89 |
92 |
26 |
207 |
74 |
108 |
46 |
228 |
0.44 |
0.565 |
10 |
Zhou [78] |
2010 |
Cervical cancer |
China |
Asian |
HB |
PCR–PAGE |
108 |
105 |
20 |
233 |
135 |
166 |
64 |
365 |
0.40 |
0.297 |
9 |
Fan [77] |
2011 |
Ovarian cancer |
China |
Asian |
HB |
PCR-CE |
78 |
84 |
17 |
179 |
76 |
103 |
44 |
223 |
0.43 |
0.396 |
8 |
Lin [76] |
2012 |
OSCC |
China |
Asian |
HB |
TaqMan |
116 |
246 |
100 |
462 |
81 |
271 |
168 |
520 |
0.58 |
0.099 |
9 |
Song [92] |
2012 |
Colorectal cancer |
China |
Asian |
HB |
PCR-RFLP |
363 |
500 |
138 |
1001 |
297 |
522 |
186 |
1005 |
0.44 |
0.102 |
14 |
Tang [87] |
2012 |
HCC |
China |
Asian |
HB |
PCR-RFLP |
52 |
84 |
14 |
150 |
57 |
82 |
11 |
150 |
0.35 |
0.011 |
7 |
Ungerback [75] |
2012 |
Colorectal cancer |
Sweden |
Caucasian |
HB |
TaqMan |
114 |
187 |
43 |
344 |
256 |
270 |
96 |
622 |
0.37 |
0.079 |
8 |
Vangsted [74] |
2012 |
Multiple myeloma |
Denmark |
Caucasian |
PB |
TaqMan |
110 |
163 |
55 |
328 |
655 |
778 |
253 |
1686 |
0.38 |
0.303 |
7 |
Arisawa [73] |
2013 |
Gastric cancer |
Japan |
Asian |
PB |
PCR-SSCP |
172 |
239 |
68 |
479 |
342 |
435 |
103 |
880 |
0.36 |
0.046 |
11 |
Cheng [23] |
2013 |
HCC |
China |
Asian |
HB |
TaqMan |
42 |
64 |
29 |
135 |
81 |
271 |
168 |
520 |
0.58 |
0.099 |
7 |
Huang [72] |
2013 |
Lung cancer |
China |
Asian |
PB |
TaqMan |
372 |
459 |
225 |
1056 |
355 |
491 |
210 |
1056 |
0.43 |
0.090 |
10 |
Huang [72] |
2013 |
Lung cancer |
China |
Asian |
PB |
TaqMan |
169 |
230 |
104 |
503 |
189 |
289 |
145 |
623 |
0.46 |
0.092 |
10 |
Huo [71] |
2013 |
Ovarian cancer |
China |
Asian |
HB |
Mass ARRAY |
83 |
82 |
22 |
187 |
71 |
103 |
47 |
221 |
0.45 |
0.399 |
7 |
Kopp [70] |
2013 |
Prostate cancer |
Denmark |
Caucasian |
PB |
RT-PCR |
128 |
152 |
54 |
334 |
109 |
161 |
64 |
334 |
0.43 |
0.741 |
11 |
Li [69] |
2013 |
Bladder cancer |
China |
Asian |
HB |
TaqMan |
189 |
269 |
151 |
609 |
223 |
324 |
93 |
640 |
0.40 |
0.156 |
11 |
Liu [86] |
2013 |
NPC |
China |
Asian |
PB |
TaqMan |
116 |
135 |
49 |
300 |
86 |
143 |
71 |
300 |
0.48 |
0.443 |
12 |
Song [84] |
2013 |
EC |
China |
Asian |
HB |
PCR-RFLP |
42 |
52 |
6 |
100 |
56 |
39 |
5 |
100 |
0.25 |
0.588 |
6 |
Song [85] |
2013 |
Cervical cancer |
China |
Asian |
HB |
PCR-RFLP |
34 |
56 |
10 |
100 |
37 |
55 |
8 |
100 |
0.36 |
0.044 |
5 |
Umar [68] |
2013 |
ESCC |
India |
Asian |
HB |
PCR |
131 |
132 |
27 |
290 |
160 |
129 |
22 |
311 |
0.28 |
0.561 |
10 |
Gao [67] |
2014 |
HCC |
China |
Asian |
PB |
TaqMan |
68 |
102 |
40 |
210 |
171 |
160 |
79 |
410 |
0.39 |
0.000 |
12 |
Hua [66] |
2014 |
Gastric cancer |
China |
Asian |
HB |
Mass ARRAY |
92 |
182 |
127 |
401 |
120 |
230 |
83 |
433 |
0.46 |
0.144 |
9 |
Oltulu [65] |
2014 |
Lung cancer |
Turkey |
Caucasian |
HB |
PCR-RFLP |
35 |
44 |
16 |
95 |
46 |
47 |
6 |
99 |
0.30 |
0.194 |
7 |
Wang [64] |
2014 |
Breast cancer |
China |
Asian |
HB |
PCR-RFLP |
93 |
210 |
171 |
474 |
162 |
216 |
123 |
501 |
0.46 |
0.003 |
9 |
Zhang [63] |
2014 |
HCC |
China |
Asian |
PB |
PCR |
205 |
312 |
107 |
624 |
542 |
790 |
274 |
1606 |
0.42 |
0.631 |
10 |
Chen [62] |
2015 |
Ovarian cancer |
China |
Asian |
HB |
Mass ARRAY |
120 |
195 |
95 |
410 |
85 |
235 |
122 |
442 |
0.54 |
0.136 |
9 |
Cui [61] |
2015 |
Prostate cancer |
China |
Asian |
HB |
PCR-RFLP |
198 |
246 |
99 |
543 |
212 |
355 |
186 |
753 |
0.48 |
0.125 |
10 |
Han [60] |
2015 |
Prostate cancer |
China |
Asian |
PB |
PCR-RFLP |
63 |
339 |
534 |
936 |
38 |
331 |
567 |
936 |
0.78 |
0.230 |
12 |
Kopp [59] |
2015 |
Colorectal cancer |
Denmark |
Caucasian |
PB |
KASP |
320 |
449 |
146 |
915 |
679 |
787 |
253 |
1719 |
0.38 |
0.311 |
11 |
Li [58] |
2015 |
Osteosarcoma |
China |
Asian |
HB |
PCR-RFLP |
60 |
114 |
46 |
220 |
50 |
106 |
66 |
222 |
0.54 |
0.551 |
9 |
Liu [57] |
2015 |
NPC |
China |
Asian |
HB |
TaqMan |
236 |
331 |
117 |
684 |
274 |
438 |
195 |
907 |
0.46 |
0.420 |
9 |
Liu [57] |
2015 |
NPC |
China |
Asian |
HB |
TaqMan |
316 |
438 |
152 |
906 |
336 |
512 |
224 |
1072 |
0.45 |
0.262 |
9 |
Pallavi [56] |
2015 |
Cervical cancer |
Iran |
Asian |
HB |
PCR-RFLP |
98 |
116 |
26 |
240 |
73 |
104 |
113 |
290 |
0.57 |
0.000 |
9 |
Wang [55] |
2015 |
Lung cancer |
China |
Asian |
HB |
PCR-RFLP |
113 |
219 |
89 |
421 |
89 |
205 |
131 |
425 |
0.55 |
0.595 |
10 |
Wang [54] |
2015 |
Thyroid carcinoma |
China |
Asian |
HB |
PCR-PAGE |
106 |
186 |
60 |
352 |
171 |
209 |
79 |
459 |
0.40 |
0.273 |
11 |
Zhang [53] |
2015 |
Lung cancer |
China |
Asian |
HB |
PCR-RFLP |
434 |
252 |
32 |
718 |
352 |
290 |
76 |
718 |
0.31 |
0.162 |
9 |
Eskandari [52] |
2016 |
Breast cancer |
Iran |
Asian |
HB |
AS-PCR |
96 |
122 |
18 |
236 |
62 |
106 |
35 |
203 |
0.43 |
0.368 |
8 |
Lu [51] |
2016 |
Ovarian cancer |
China |
Asian |
HB |
PCR-RFLP |
115 |
351 |
221 |
687 |
95 |
339 |
253 |
687 |
0.61 |
0.271 |
10 |
Rybka [50] |
2016 |
AML |
Poland |
Caucasian |
HB |
PCR |
25 |
30 |
7 |
62 |
43 |
69 |
14 |
126 |
0.38 |
0.079 |
4 |
MAF, minor allele frequency; HWE, Hardy-Weinberg equilibrium; OSCC, oral squamous cell carcinoma; CLL, chronic lymphocytic leukemia; RCC, renal cell carcinoma; HNSCC, head and neck squamous cell carcinoma; HCC, hepatocellular carcinoma; NPC, nasopharyngeal carcinoma; ESCC, esophageal squamous cell carcinoma; EC, endometrial carcinoma; AML, acute myeloid leukemia; PB, population based; HB, hospital based; PCR-PAGE, polymerase chain reaction-polyacrylamide gel electrophoresis; PCR-RFLP, polymerase chain reaction-restriction fragment length polymorphism; PCR-CE, polymerase chain reaction-capillary electrophoresis; PCR-SSCP, polymerase chain reaction-single strand conformation polymorphism; KASP, kompetitive allele specific PCR; AS-PCR, allele-specific polymerase chain reaction.
Meta-analysis results
The main results of this meta-analysis are shown in Table 2 and Figure 2. Overall, the pooled analysis demonstrated a significant, negative association between the NFKB1 -94ins/delATTG polymorphism and overall cancer risk under all five genetic models (described in the Materials and Methods section): DD vs. II: OR = 0.75, 95% CI = 0.64-0.87; ID vs. II: OR = 0.91, 95% CI = 0.83-0.99; DD vs. ID/II: OR = 0.81, 95% CI = 0.71-0.91; ID/DD vs. II: OR = 0.86, 95% CI = 0.78-0.95; and D vs. I: OR = 0.88, 95% CI = 0.81-0.95.
Table 2: Meta-analysis of the association between the NFKB1 -94ins/delATTG (rs28362491) polymorphism and overall cancer risk
Variables |
No. of studies |
Sample size |
Homozygous |
Heterozygous |
Recessive |
Dominant |
Allele |
|||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
DD vs. II |
ID vs. II |
DD vs. ID/II |
ID/DD vs. II |
D vs. I |
||||||||
OR (95% CI) |
Phet |
OR (95% CI) |
Phet |
OR (95% CI) |
Phet |
OR (95% CI) |
Phet |
OR (95% CI) |
Phet |
|||
All |
50 |
18299/ 23484 |
0.75 (0.64-0.87) |
<0.001 |
0.91 (0.83-0.99) |
<0.001 |
0.81 (0.71-0.91) |
<0.001 |
0.86 (0.78-0.95) |
<0.001 |
0.88 (0.81-0.95) |
<0.001 |
Cancer type |
||||||||||||
HCC |
6 |
1471/ 3390 |
0.65 (0.38-1.11) |
<0.001 |
0.82 (0.56-1.19) |
<0.001 |
0.74 (0.54-1.02) |
0.006 |
0.75 (0.50-1.15) |
<0.001 |
0.80 (0.61-1.07) |
<0.001 |
Lung |
5 |
2793/ 2921 |
0.77 (0.48-1.26) |
<0.001 |
0.84 (0.74-0.96) |
0.337 |
0.82 (0.54-1.26) |
<0.001 |
0.83 (0.67-1.03) |
0.012 |
0.88 (0.70-1.09) |
<0.001 |
Colorectal |
4 |
2399/ 3653 |
0.96 (0.65-1.43) |
0.001 |
1.08 (0.79-1.47) |
<0.001 |
0.92 (0.69-1.22) |
0.022 |
1.05 (0.77-1.44) |
<0.001 |
1.01 (0.82-1.24) |
<0.001 |
NPC |
4 |
2053/ 2482 |
0.66 (0.56-0.78) |
0.467 |
0.85 (0.75-0.97) |
0.537 |
0.73 (0.63-0.85) |
0.755 |
0.79 (0.69-0.90) |
0.371 |
0.81 (0.74-0.89) |
0.316 |
Prostate |
4 |
1930/ 2166 |
0.59 (0.48-0.72) |
0.684 |
0.74 (0.62-0.88) |
0.803 |
0.77 (0.65-0.92) |
0.267 |
0.69 (0.59-0.81) |
0.760 |
0.79 (0.72-0.87) |
0.540 |
Ovarian |
4 |
1463/ 1573 |
0.54 (0.40-0.73) |
0.173 |
0.73 (0.61-0.87) |
0.416 |
0.68 (0.52-0.89) |
0.102 |
0.67 (0.56-0.79) |
0.462 |
0.75 (0.65-0.86) |
0.181 |
Bladder |
3 |
1058/ 1175 |
0.93 (0.40-2.21) |
<0.001 |
0.95 (0.74-1.22) |
0.193 |
0.97 (0.43-2.17) |
<0.001 |
0.96 (0.67-1.37) |
0.026 |
0.97 (0.66-1.43) |
<0.001 |
Gastric |
3 |
1062/ 1429 |
0.97 (0.41-2.31) |
<0.001 |
0.88 (0.60-1.30) |
0.032 |
1.11 (0.57-2.15) |
<0.001 |
0.90 (0.54-1.49) |
0.002 |
0.98 (0.65-1.50) |
<0.001 |
Cervical |
3 |
573/ 755 |
0.41 (0.15-1.11) |
0.001 |
0.85 (0.67-1.08) |
0.627 |
0.44 (0.17-1.12) |
0.001 |
0.69 (0.45-1.04) |
0.050 |
0.66 (0.39-1.10) |
<0.001 |
OSCC |
2 |
674/ 721 |
0.49 (0.33-0.72) |
0.222 |
0.67 (0.51-0.88) |
0.570 |
0.63 (0.49-0.81) |
0.308 |
0.60 (0.46-0.77) |
0.377 |
0.70 (0.60-0.82) |
0.300 |
Breast |
2 |
710/ 704 |
0.92 (0.13-6.42) |
<0.001 |
1.13 (0.51-2.54) |
0.002 |
0.85 (0.20-3.61) |
<0.001 |
1.13 (0.38-3.37) |
<0.001 |
1.04 (0.43-2.53) |
<0.001 |
Others |
10 |
2113/ 4263 |
1.07 (0.88-1.31) |
0.281 |
1.16 (0.94-1.42) |
0.006 |
1.00 (0.86-1.16) |
0.483 |
1.14 (0.94-1.38) |
0.008 |
1.06 (0.95-1.19) |
0.062 |
Ethnicity |
||||||||||||
Asians |
38 |
15079/ 18673 |
0.67 (0.55-0.80) |
<0.001 |
0.86 (0.79-0.94) |
<0.001 |
0.75 (0.65-0.86) |
<0.001 |
0.80 (0.72-0.89) |
<0.001 |
0.83 (0.76-0.91) |
<0.001 |
Chinese |
34 |
13834/ 16989 |
0.68 (0.56-0.81) |
<0.001 |
0.84 (0.77-0.93) |
<0.001 |
0.77 (0.67-0.88) |
<0.001 |
0.80 (0.71-0.89) |
<0.001 |
0.84 (0.76-0.91) |
<0.001 |
Caucasians |
12 |
3220/ 6559 |
1.08 (0.92-1.27) |
0.221 |
1.11 (0.94-1.30) |
0.004 |
1.02 (0.88-1.17) |
0.269 |
1.10 (0.95-1.28) |
0.010 |
1.06 (0.98-1.15) |
0.141 |
Source of control |
||||||||||||
HB |
40 |
12614/ 15682 |
0.70 (0.58-0.85) |
<0.001 |
0.88 (0.80-0.98) |
<0.001 |
0.76 (0.65-0.89) |
<0.001 |
0.84 (0.74-0.94) |
<0.001 |
0.85 (0.77-0.94) |
<0.001 |
PB |
10 |
5685/ 9550 |
0.95 (0.79-1.15) |
0.001 |
1.00 (0.86-1.15) |
0.002 |
0.98 (0.88-1.09) |
0.161 |
0.98 (0.84-1.14) |
<0.001 |
0.98 (0.90-1.08) |
<0.001 |
Quality score |
||||||||||||
>9 |
19 |
9894/ 13117 |
0.87 (0.73-1.04) |
<0.001 |
0.93 (0.84-1.04) |
<0.001 |
0.92 (0.80-1.05) |
<0.001 |
0.91 (0.81-1.03) |
<0.001 |
0.94 (0.86-1.02) |
<0.001 |
≤9 |
31 |
8405/ 12115 |
0.68 (0.53-0.86) |
<0.001 |
0.89 (0.79-1.01) |
<0.001 |
0.73 (0.60-0.88) |
<0.001 |
0.83 (0.71-0.96) |
<0.001 |
0.84 (0.75-0.95) |
<0.001 |
Het, heterogeneity; HCC, hepatocellular carcinoma; NPC, nasopharyngeal carcinoma; OSCC, oral squamous cell carcinoma; HB, hospital-based; PB, population-based.
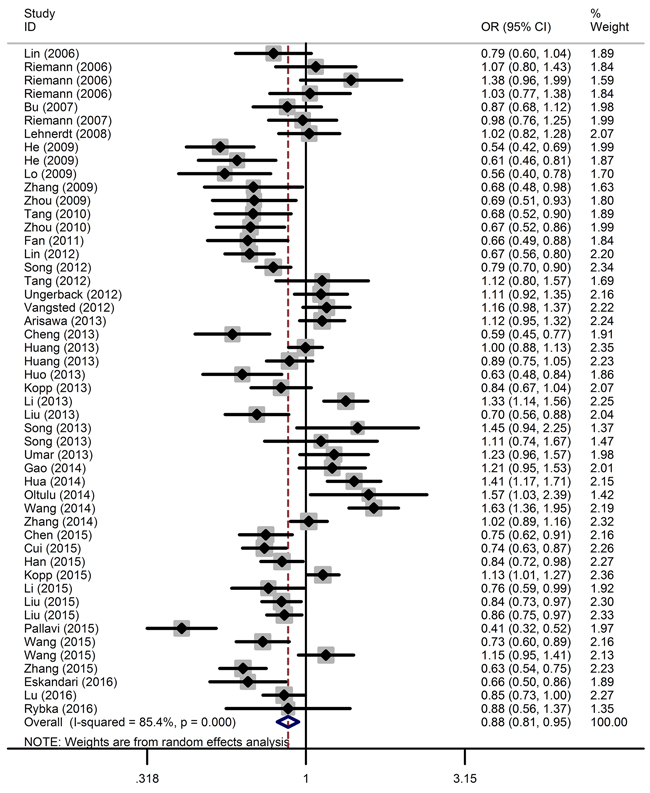
Figure 2: Forest plot of the association between the NFKB1 -94ins/delATTG polymorphism and overall cancer susceptibility in the allele contrast model. The horizontal lines represent the study-specific ORs and 95% CIs, respectively. The diamond represents the pooled OR and corresponding 95% CI.
Stratified analysis by cancer type revealed that the -94ins/delATTG polymorphism significantly decreased lung cancer risk (ID vs. II: OR = 0.84, 95% CI = 0.74-0.96), nasopharyngeal carcinoma risk (DD vs. II: OR = 0.66, 95% CI = 0.56-0.78; ID vs. II: OR = 0.85, 95% CI = 0.75-0.97; DD vs. ID/II: OR = 0.73, 95% CI = 0.63-0.85; ID/DD vs. II: OR = 0.79, 95% CI = 0.69-0.90; D vs. I: OR = 0.81, 95% CI = 0.74-0.89), prostate cancer risk (DD vs. II: OR = 0.59, 95% CI = 0.48-0.72; ID vs. II: OR = 0.74, 95% CI = 0.62-0.88; DD vs. ID/II: OR = 0.77, 95% CI = 0.65-0.92; ID/DD vs. II: OR = 0.69, 95% CI = 0.59-0.81; D vs. I: OR = 0.79, 95% CI = 0.72-0.87), ovarian cancer risk (DD vs. II: OR = 0.54, 95% CI = 0.40-0.73; ID vs. II: OR = 0.73, 95% CI = 0.61-0.87; DD vs. ID/II: OR = 0.68, 95% CI = 0.52-0.89; ID/DD vs. II: OR = 0.67, 95% CI = 0.56-0.79; D vs. I: OR = 0.75, 95% CI = 0.65-0.86), and oral squamous cell carcinoma risk (DD vs. II: OR = 0.49, 95% CI = 0.33-0.72; ID vs. II: OR = 0.67, 95% CI = 0.51-0.88; DD vs. ID/II: OR = 0.63, 95% CI = 0.49-0.81; ID/DD vs. II: OR = 0.60, 95% CI = 0.46-0.77; D vs. I: OR = 0.70, 95% CI = 0.60-0.82). However, no correlation was observed between NFKB1 -94ins/delATTG polymorphism and other types of cancer.
When stratified by population, a significant association between NFKB1 -94ins/delATTG polymorphism and decreased cancer risk among Asians was detected under all genetic models (DD vs. II: OR = 0.67, 95% CI = 0.55-0.80; ID vs. II: OR = 0.86, 95% CI = 0.79-0.94; DD vs. ID/II: OR = 0.75, 95% CI = 0.65-0.86; ID/DD vs. II: OR = 0.80, 95% CI = 0.72-0.89; D vs. I: OR = 0.83, 95% CI = 0.76-0.91). As most of the studies were performed on the Chinese population, we determined the association of NFKB1 -94ins/delATTG polymorphism with cancer risk on Chinese subjects. In this case, the results also showed a protective role against cancer (DD vs. II: OR = 0.68, 95% CI = 0.56-0.81; ID vs. II: OR = 0.84, 95% CI = 0.77-0.93; DD vs. ID/II: OR = 0.77, 95% CI = 0.67-0.88; ID/DD vs. II: OR = 0.80, 95% CI = 0.71-0.89; D vs. I: OR = 0.84, 95% CI = 0.76-0.91). No association was observed, however, for Caucasians.
Upon stratification based on the sources of controls, the NFKB1 -94ins/delATTG polymorphism had a protective role against cancer in hospital-based groups (DD vs. II: OR = 0.70, 95% CI = 0.58-0.85; ID vs. II: OR = 0.88, 95% CI = 0.80-0.98; DD vs. ID/II: OR = 0.76, 95% CI = 0.65-0.89; ID/DD vs. II: OR = 0.84, 95% CI = 0.74-0.94; D vs. I: OR = 0.85, 95% CI = 0.77-0.94).
After stratification by quality score, a significantly decreased cancer risk was observed for studies with quality scores ≤9 (DD vs. II: OR = 0.68, 95% CI = 0.53-0.86; DD vs. ID/II: OR = 0.73, 95% CI = 0.60-0.88; ID/DD vs. II: OR = 0.83, 95% CI = 0.71-0.96; D vs. I: OR = 0.84, 95% CI = 0.75-0.95).
Heterogeneity and sensitivity analysis
Statistically significant between-study heterogeneity was found in the pooled analysis under the five genetic models (P < 0.001). Thus, the random-effect model was applied to calculate the ORs and 95% CIs. Sensitivity analysis using the leave-one-out cross-validation method was conducted to assess the impact of each single study on the overall risk estimates. The omission of each individual study did not have substantial influence on the risk estimates, supporting the credibility and reliability of this meta-analysis (data not shown).
Publication bias
Publication bias was assessed by Begg’s funnel plot and quantitative Egger’s test. The funnel plot showed a symmetrical shape (Figure 3), suggesting no publication bias, a conclusion further supported by the Egger’s test (DD vs. II: P = 0.158; ID vs. II: P = 0.340, DD vs. ID/II: P = 0.157; ID/DD vs. II: P = 0.221; and D vs. I: P = 0.250).
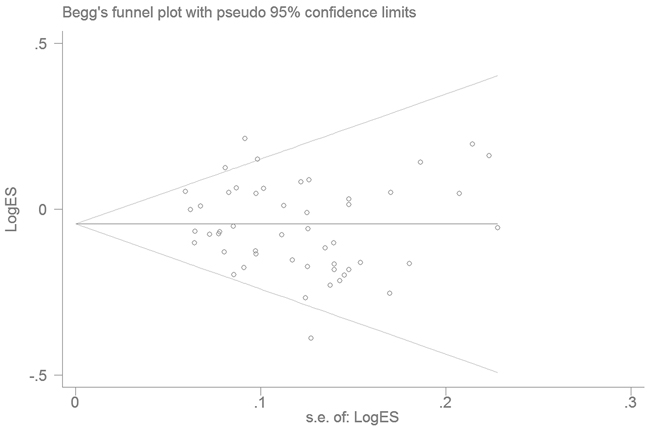
Figure 3: Funnel plot analysis to detect publication bias for NFKB1 -94ins/delATTG polymorphism under the allele contrast model. Each point represents a separate study.
Trial sequential analysis and false-positive report probability (FPRP) analyses
To minimize random errors and strengthen the robustness of our conclusions, we performed TSA (Figure 4). This analysis showed that the cumulative z-curve crossed the trial sequential monitoring boundary before reaching the required information size, suggesting that the cumulative evidence is sufficient and no further evidence is needed to verify the conclusions.
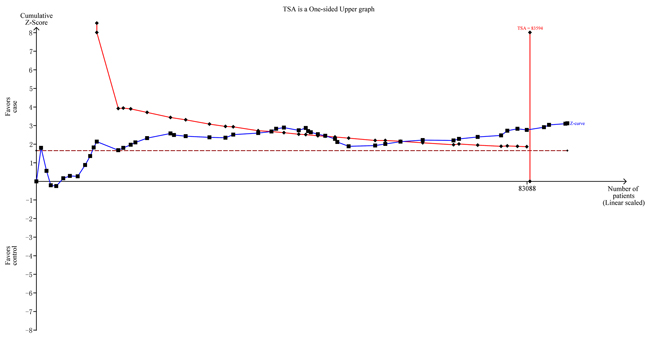
Figure 4: Trial sequential analysis for NFKB1 -94ins/delATTG polymorphism under the allele contrast model.
We finally calculated the FPRP values for all observed significant findings. With the assumption of a prior probability of 0.1, the FPRP values were all <0.20, suggesting that these significant associations were noteworthy (Table 3).
Table 3: False-positive report probability values for associations between the NFKB1 -94ins/delATTG polymorphism and overall cancer risk
Variables |
OR (95% CI) |
P a |
Power b |
Prior Probability |
||||
|---|---|---|---|---|---|---|---|---|
0.25 |
0.1 |
0.01 |
0.001 |
0.0001 |
||||
Homozygous (DD vs. II) |
||||||||
All |
0.75 (0.64-0.87) |
2.45*10-4 |
1.000 |
0.001 |
0.002 |
0.024 |
0.197 |
0.710 |
NPC |
0.66 (0.56-0.78) |
1.99*10-6 |
0.652 |
0.000 |
0.000 |
0.000 |
0.003 |
0.030 |
Prostate |
0.59 (0.48-0.72) |
6.40*10-7 |
0.860 |
0.000 |
0.000 |
0.000 |
0.001 |
0.007 |
Ovarian |
0.54 (0.40-0.73) |
6.62*10-6 |
0.612 |
0.000 |
0.000 |
0.001 |
0.011 |
0.098 |
OSCC |
0.49 (0.33-0.72) |
3.26*10-4 |
0.388 |
0.003 |
0.008 |
0.077 |
0.457 |
0.894 |
Asians |
0.67 (0.55-0.80) |
1.81*10-5 |
1.000 |
0.000 |
0.000 |
0.002 |
0.018 |
0.153 |
Chinese |
0.68 (0.56-0.81) |
3.45*10-5 |
1.000 |
0.000 |
0.000 |
0.003 |
0.033 |
0.257 |
HB |
0.70 (0.58-0.85) |
3.30*10-4 |
1.000 |
0.001 |
0.003 |
0.032 |
0.248 |
0.767 |
QS ≤9 |
0.68 (0.53-0.86) |
1.63*10-3 |
1.000 |
0.005 |
0.014 |
0.139 |
0.620 |
0.942 |
Heterozygous (ID vs. II) |
||||||||
All |
0.91 (0.83-0.99) |
0.024 |
1.000 |
0.066 |
0.175 |
0.700 |
0.959 |
0.996 |
Lung |
0.84 (0.74-0.96) |
7.74*10-3 |
1.000 |
0.023 |
0.065 |
0.434 |
0.885 |
0.987 |
NPC |
0.85 (0.75-0.97) |
0.017 |
1.000 |
0.048 |
0.131 |
0.623 |
0.943 |
0.994 |
Prostate |
0.74 (0.62-0.88) |
7.44*10-4 |
0.998 |
0.002 |
0.007 |
0.069 |
0.427 |
0.882 |
Ovarian |
0.73 (0.61-0.87) |
5.68*10-4 |
0.979 |
0.002 |
0.005 |
0.054 |
0.367 |
0.853 |
OSCC |
0.67 (0.51-0.88) |
3.99*10-3 |
0.800 |
0.015 |
0.043 |
0.331 |
0.833 |
0.980 |
Asians |
0.86 (0.79-0.94) |
7.59*10-4 |
1.000 |
0.002 |
0.007 |
0.070 |
0.431 |
0.884 |
Chinese |
0.84 (0.77-0.93) |
4.59*10-4 |
1.000 |
0.001 |
0.004 |
0.043 |
0.314 |
0.821 |
HB |
0.88 (0.80-0.98) |
0.015 |
1.000 |
0.043 |
0.118 |
0.596 |
0.937 |
0.993 |
Recessive (DD vs. ID/II) |
||||||||
All |
0.81 (0.71-0.91) |
3.22*10-4 |
1.000 |
0.001 |
0.003 |
0.031 |
0.243 |
0.763 |
NPC |
0.73 (0.63-0.85) |
3.26*10-5 |
0.841 |
0.000 |
0.000 |
0.004 |
0.037 |
0.279 |
Prostate |
0.77 (0.65-0.92) |
3.78*10-3 |
0.999 |
0.011 |
0.033 |
0.272 |
0.791 |
0.974 |
Ovarian |
0.68 (0.52-0.89) |
4.62*10-3 |
0.976 |
0.014 |
0.041 |
0.319 |
0.826 |
0.979 |
OSCC |
0.63 (0.49-0.81) |
2.37*10-4 |
0.356 |
0.002 |
0.006 |
0.062 |
0.399 |
0.869 |
Asians |
0.75 (0.65-0.86) |
6.02*10-5 |
1.000 |
0.000 |
0.001 |
0.006 |
0.057 |
0.376 |
Chinese |
0.77 (0.67-0.88) |
1.44*10-4 |
1.000 |
0.000 |
0.001 |
0.014 |
0.126 |
0.591 |
HB |
0.76 (0.65-0.89) |
5.23*10-4 |
1.000 |
0.002 |
0.005 |
0.049 |
0.343 |
0.840 |
QS ≤9 |
0.73 (0.60-0.88) |
1.23*10-3 |
1.000 |
0.004 |
0.011 |
0.109 |
0.551 |
0.925 |
Dominant (ID/DD vs. II) |
||||||||
All |
0.86 (0.78-0.95) |
2.68*10-4 |
1.000 |
0.008 |
0.024 |
0.210 |
0.728 |
0.964 |
NPC |
0.79 (0.69-0.90) |
2.94*10-4 |
0.997 |
0.001 |
0.003 |
0.028 |
0.227 |
0.747 |
Prostate |
0.69 (0.59-0.81) |
8.36*10-6 |
0.796 |
0.000 |
0.000 |
0.001 |
0.010 |
0.095 |
Ovarian |
0.67 (0.56-0.79) |
3.45*10-6 |
0.531 |
0.000 |
0.000 |
0.001 |
0.006 |
0.061 |
OSCC |
0.60 (0.46-0.77) |
9.87*10-5 |
0.186 |
0.002 |
0.005 |
0.050 |
0.347 |
0.842 |
Asians |
0.80 (0.72-0.89) |
9.05*10-5 |
1.000 |
0.000 |
0.001 |
0.009 |
0.083 |
0.475 |
Chinese |
0.80 (0.71-0.89) |
9.39*10-5 |
1.000 |
0.000 |
0.001 |
0.009 |
0.086 |
0.484 |
HB |
0.84 (0.74-0.94) |
2.29*10-3 |
1.000 |
0.007 |
0.020 |
0.185 |
0.696 |
0.958 |
QS ≤9 |
0.83 (0.71-0.96) |
0.013 |
1.000 |
0.038 |
0.105 |
0.563 |
0.929 |
0.992 |
Allele (D vs. I) |
||||||||
All |
0.88 (0.81-0.95) |
8.86*10-4 |
1.000 |
0.003 |
0.008 |
0.081 |
0.469 |
0.899 |
NPC |
0.81 (0.74-0.89) |
9.60*10-6 |
1.000 |
0.000 |
0.000 |
0.001 |
0.009 |
0.088 |
Prostate |
0.79 (0.72-0.87) |
1.03*10-6 |
1.000 |
0.000 |
0.000 |
0.000 |
0.001 |
0.010 |
Ovarian |
0.75 (0.65-0.86) |
3.47*10-5 |
1.000 |
0.000 |
0.000 |
0.003 |
0.033 |
0.257 |
OSCC |
0.70 (0.60-0.82) |
1.11*10-5 |
0.809 |
0.000 |
0.000 |
0.001 |
0.014 |
0.121 |
Asians |
0.83 (0.76-0.91) |
4.78*10-5 |
1.000 |
0.000 |
0.000 |
0.005 |
0.046 |
0.323 |
Chinese |
0.84 (0.76-0.91) |
9.24*10-5 |
1.000 |
0.000 |
0.001 |
0.009 |
0.085 |
0.480 |
HB |
0.85 (0.77-0.94) |
9.63*10-4 |
1.000 |
0.003 |
0.009 |
0.087 |
0.490 |
0.906 |
QS ≤9 |
0.84 (0.75-0.95) |
4.84*10-3 |
1.000 |
0.014 |
0.042 |
0.324 |
0.829 |
0.980 |
CI, confidence interval; OR, odds ratio; NPC, Nasopharyngeal carcinoma; OSCC, oral squamous cell carcinoma; HB, Hospital based; QS, quality score.
a Chi-square test was adopted to calculate the genotype frequency distributions.
b Statistical power was calculated using the number of observations in the subgroup and the OR and P values in this table.
DISCUSSION
In this meta-analysis, we found that the NFKB1 -94ins/delATTG promoter polymorphism was significantly associated with decreased overall cancer risk under the five genetic models. To the best of our knowledge, this is the most comprehensive meta-analysis on this topic by now.
Numerous studies have suggested that polymorphisms in genes encoding inflammatory response factors, such as TNF-alpha -308G>A [6], IL6 -174G>C [90], and NFKBIA -826C>T [91] may contribute to cancer susceptibility. Song et al. [92] reported that the NFKB1 -94ins/delATTG polymorphism analyzed here (rs28362491) increased the risk of colorectal cancer in a Southern Chinese population; this association was also observed in several publications [71]. However, contradictory conclusions were also reported, namely a null association, or decreased cancer susceptibility. To address this controversy, at least six meta-analyses were performed. The first one, performed in 2011 by Zou et al. [32], included only 2,743 cases and 2,195 controls from 11 studies. They did not observe any association between the -94ins/delATTG variant and overall cancer. However, an ethno-specific association was detected by subgroup analysis; the D allele was protective against cancer in Asians, but increased the risk in Caucasians. Afterwards, Wang et al. [33] conducted an updated meta-analysis including 5,196 cases and 6,614 controls from 19 publications. They found that variant homozygotes (DD) had a decreased risk of cancer compared with wild-type homozygotes (II). The association was also found under the dominant genetic model (DD+DI vs. II). In subgroup analysis, a significantly decreased risk was observed in Asians but not in Caucasians. In addition, this susceptibility was cancer-specific, as it was observed for all cancer types examined, except for colorectal cancer. In 2014, four updated meta-analyses were published. Upon revision of 6,494 cases and 9,884 controls from 23 studies, Xu et al. [29] found that the -94ins/delATTG polymorphism was significantly associated with increased cancer risk under all the inheritance models. Stratified analysis by cancer type showed significant associations for ovarian cancer, hepatocellular carcinoma, and oral squamous cell carcinoma, but not for bladder cancer or lung cancer. Ethnicity subgroup analysis indicated that the polymorphism contributed to cancer risk in the Asian, but not the Caucasian, population. Another study by Yang et al. [28], which included 21 reports with 6,127 cases and 9,238 controls, also detected an increased overall cancer risk. Stratified analysis revealed a significant association between the polymorphism and ovarian, oral, and prostate cancers. These findings were also specific to the Asian population. Duan et al. [31] reviewed a total of 25 studies that included 8,750 cancer cases and 9,170 controls. They found that the insertion allele of the -94ins/delATTG polymorphism significantly increased cancer risk, both in overall genetic analysis as well as in Asians. Stratified analysis revealed that the polymorphism was associated with increased risk for oral squamous cell carcinoma and ovarian cancer, but not for colorectal cancer, bladder cancer, or renal cell cancer. In another meta-analysis involving 7,281 cases and 10,039 controls from 25 case-control studies, Nian et al. [30] found that the -94ins/delATTG polymorphism was significantly associated with decreased susceptibility to cancer in overall population under homozygous, recessive, dominant, and allele contrast models. Subgroups analysis based on ethnicity revealed that the polymorphism conferred decreased cancer susceptibility in the Asian population.
Since then, approximately 20 new relevant case-control studies in English and Chinese have emerged, some containing large samples and convincing results. Our study re-evaluated the impact of the NFKB1 -94ins/delATTG polymorphism on cancer risk. In line with some previous meta-analyses, our pooled analysis revealed a significant association with decreased cancer risk under all five genetic models. Conversely, we found that the del allele of the NFKB1 -94ins/delATTG polymorphism conferred a significantly decreased risk of cancer in the pooled analysis. Compared with the ins allele, the ins allele significantly enhances the binding ability to nuclear proteins and increases transcriptional activity, which eventually upregulates p50 (the active NF-κB1 subunit) expression [21]. Given the tumor-promoting role of p50 and NF-κB, it is biologically plausible that the -94del allele confers decreased cancer susceptibility.
In line with previous data, our study detected a significant association between the -94ins/delATTG polymorphism and cancer risk in Asians, but not in Caucasians, under all five genetic models. It is thus likely that the allelic distribution of this polymorphism vary geographically and ethnically, thus leading to the discrepancies in cancer risk. This may indicate that these groups have distinct environmental or genetic cancer co-etiologies. Stratification by cancer type showed that the NFKB1 -94ins/delATTG polymorphism was inversely associated with the risk of lung cancer, nasopharyngeal carcinoma, prostate cancer, ovarian cancer, and oral squamous cell carcinoma, but no association was found for hepatocellular carcinoma, colorectal cancer, bladder cancer, gastric cancer, cervical cancer, breast cancer, or other cancers. This phenomenon may be partly attributed to the inherent heterogeneity of oncogenic progression in different cancer types [93], although the insufficient statistical power caused by the relatively small number of studies on each cancer type may also be a factor.
The credibility of our conclusions is supported by the inclusion of Chinese-language studies, exclusion of publications with controls violating the Hardy-Weinberg equilibrium, and inclusion of subgroup, publication bias, and sensitivity analyses. Among the limitations of our meta-analysis are a significant between-study heterogeneity, detected in some comparisons, which may diminish the strength of our conclusions. The source of this heterogeneity may be ascribed to sample size, genotyping methods, ethnicity, source of controls, as well as the studies’ diverse quality scores. Second, we assessed the association between the NFKB1 -94ins/delATTG polymorphism and cancer risk from a genetic perspective only, by using unadjusted ORs. Multiple potentially influential factors such as life style, environmental exposure, and gene-environment interactions should be considered to obtain a more precise risk estimation. Third, the number of studies in certain subgroup analyses was too small to obtain a reliable association. For instance, only six publications were included for hepatocellular carcinoma, and fewer studies were available for breast cancer and oral squamous cell carcinoma, which restrains further analysis for risk factors. Finally, the meta-analysis is a type of retrospective study with several inherent drawbacks: inconsistent qualities of primary studies, incomplete histological details, misclassified genotypes, different definitions of disease status, and improperly matched sources of controls.
In conclusion, despite these limitations, and in agreement with several previous studies, this meta-analysis draws the robust conclusion that NFKB1 -94ins/delATTG polymorphism is associated with decreased cancer risk, especially in the Asian population. These findings indicate a possible involvement of NFKB1 in the etiology of tumorigenesis, and suggest the potentially relevant therapeutic value of NF-κB modulation in cancer prevention. Further multi-center, well-designed investigations with larger sample sizes that include gene-environment interactions assessment are warranted to confirm our findings.
MATERIALS AND METHODS
Publication search
We performed a comprehensive literature search by using the PubMed and EMBASE databases, without language limitations, up to July 1, 2016. The following search terms were used: “polymorphism or SNP or single nucleotide polymorphism or variant” and “NFKB1/NF-κB1 or nuclear factor kappa B1”, and “tumor or cancer or neoplasm or carcinoma”. We also searched the China National Knowledge Infrastructure (CNKI) and WANFANG databases to obtain additional, relevant studies. Retrieved articles were manually screened to determine eligible studies. When two or more publications containing overlapping data were found, the largest study was included in the final meta-analysis.
Inclusion/exclusion criteria
All articles included in the current analysis met the following criteria: 1) evaluation of the association between NFKB1 -94ins/delATTG polymorphism and cancer risk; 2) case-control studies; 3) sufficient information provided to estimate ORs and 95% CIs; 4) NFKB1 -94ins/delATTG genotype frequency in agreement with HWE in controls. Exclusion criteria were as follows: 1) case-only studies; 2) meta-analysis or reviews; 3) studies that lacked detailed genotyping data; 4) duplicates of previous publications.
Data extraction
Two authors (Z.Z. and W.F.) evaluated all eligible studies independently and extracted the following information: first author’s surname, year of publication, cancer type, country, ethnicity, source of controls, genotyping methods, and genetic distribution of cases and controls. Stratification analyses were conducted by cancer type, ethnicity (Asians, Caucasians), source of control (hospital-based and population-based) and quality score (>9 and ≤9). If a study contained two or more ethnic groups or cancer types, we divided the study accordingly.
Trial sequential analysis
TSA was performed as described by us previously [94]. Briefly, after adopting a level of significance of 5% for type I error and of 30% for type II error, the required information size was calculated, and TSA monitoring boundaries were built.
FPRP analysis
The FPRP values at different prior probability levels for all significant findings were calculated as described by us previously [95]. Briefly, 0.2 was set as FPRP threshold and assigned a prior probability of 0.01 to detect an OR of 0.67 (for protective effects) for an association with genotypes under investigation. A FPRP value <0.2 denoted a noteworthy association.
Statistical methods
Goodness-of-fit χ2 test was used to assess HWE in the control subjects. Departure from HWE was assessed using a P < 0.05 as threshold in each study. The strength of the association between NFKB1 -94ins/delATTG polymorphism and cancer risk was assessed by calculating ORs and corresponding 95% CIs. Five genetic models were adopted: homozygote model (DD, homozygous deletion (del/del) vs. II, homozygous insertion (ins/ins) or wild-type); heterozygote model (ID, heterozygous ins/del vs. II); recessive model (DD vs. ID/II); dominant model (ID/DD vs. II); and allele contrast model (D vs. I). Subgroup and stratification analyses were also performed to test the association by ethnicity, cancer type, source of control and quality score. We performed χ2-based Q-test to assess heterogeneity between study results. The fixed-effects model (Mantel-Haenszel method) was used if the studies were found to be homogeneous (with P > 0.10 for the Q-test). Otherwise, the random-effects model (DerSimonian and Laird method) was adopted to estimate the pooled OR [96-99]. Quality assessment for each study was performed using the quality assessment criteria described previously (Supplementary Table 1) [98]. Sensitivity analysis was carried out by individually removing each study and reanalyzing the pooled risk estimates. Potential publication bias was estimated by Begg’s funnel plot and Egger’s linear regression, where an asymmetric plot and a P value < 0.05, respectively, indicate the presence of publication bias. All the data management and statistical analyses were completed using STATA software (Stata Corporation, College Station, TX; version 11.0). All the P values were two-sided. A P value of < 0.05 was considered statistically significant.
ACKNOWLEDGMENTS
This study was supported by grants from the National Natural Science Foundation of China (Grant No. 81502046), and the Special Financial Grant from the China Postdoctoral Science Foundation (Grant No. 2014T70836).
CONFLICTS OF INTEREST
The authors declare no competing financial interests.
REFERENCES
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016; 66:7-30.
2. Pharoah PD, Dunning AM, Ponder BA, Easton DF. Association studies for finding cancer-susceptibility genetic variants. Nat Rev Cancer. 2004; 4:850-860.
3. Foulkes WD. Inherited susceptibility to common cancers. N Engl J Med. 2008; 359:2143-2153.
4. Baldwin AS, Jr. Series introduction: the transcription factor NF-kappaB and human disease. J Clin Invest. 2001; 107:3-6.
5. Eiro N, Vizoso FJ. Inflammation and cancer. World J Gastrointest Surg. 2012; 4:62-72.
6. He YQ, Zhu JH, Huang SY, Cui Z, He J, Jia WH. The association between the polymorphisms of TNF-alpha and non-Hodgkin lymphoma: a meta-analysis. Tumour Biol. 2014; 35:12509-12517.
7. Zhang T, Xie S, Zhu JH, Li QW, He J, Zeng AP. Association of IL10 -819C>T and -592C>A Polymorphisms with Non-Hodgkin Lymphoma Susceptibility: Evidence from Published Studies. J Cancer. 2015; 6:709-716.
8. Zhang Y, Xia ZG, Zhu JH, Chen MB, Wang TM, Shen WX, He J. Association of Interleukin-10 -3575T>A and -1082A>G polymorphisms with non-Hodgkin lymphoma susceptibility: a comprehensive review and meta-analysis. Mol Genet Genomics. 2015; 290:2063-2073.
9. Sen R, Baltimore D. Multiple nuclear factors interact with the immunoglobulin enhancer sequences. Cell. 1986; 46:705-716.
10. Sethi G, Sung B, Aggarwal BB. Nuclear factor-kappaB activation: from bench to bedside. Exp Biol Med (Maywood). 2008; 233:21-31.
11. Hayden MS, Ghosh S. Signaling to NF-kappaB. Genes Dev. 2004; 18:2195-2224.
12. Baldwin AS, Jr. The NF-kappa B and I kappa B proteins: new discoveries and insights. Annu Rev Immunol. 1996; 14:649-683.
13. Weichert W, Boehm M, Gekeler V, Bahra M, Langrehr J, Neuhaus P, Denkert C, Imre G, Weller C, Hofmann HP, Niesporek S, Jacob J, Dietel M, Scheidereit C, Kristiansen G. High expression of RelA/p65 is associated with activation of nuclear factor-kappaB-dependent signaling in pancreatic cancer and marks a patient population with poor prognosis. Br J Cancer. 2007; 97:523-530.
14. Tew GW, Lorimer EL, Berg TJ, Zhi H, Li R, Williams CL. SmgGDS regulates cell proliferation, migration, and NF-kappaB transcriptional activity in non-small cell lung carcinoma. J Biol Chem. 2008; 283:963-976.
15. Scartozzi M, Bearzi I, Pierantoni C, Mandolesi A, Loupakis F, Zaniboni A, Catalano V, Quadri A, Zorzi F, Berardi R, Biscotti T, Labianca R, Falcone A, Cascinu S. Nuclear factor-kB tumor expression predicts response and survival in irinotecan-refractory metastatic colorectal cancer treated with cetuximab-irinotecan therapy. J Clin Oncol. 2007; 25:3930-3935.
16. Chua HL, Bhat-Nakshatri P, Clare SE, Morimiya A, Badve S, Nakshatri H. NF-kappaB represses E-cadherin expression and enhances epithelial to mesenchymal transition of mammary epithelial cells: potential involvement of ZEB-1 and ZEB-2. Oncogene. 2007; 26:711-724.
17. Yang J, Pan WH, Clawson GA, Richmond A. Systemic targeting inhibitor of kappaB kinase inhibits melanoma tumor growth. Cancer Res. 2007; 67:3127-3134.
18. Annunziata CM, Davis RE, Demchenko Y, Bellamy W, Gabrea A, Zhan F, Lenz G, Hanamura I, Wright G, Xiao W, Dave S, Hurt EM, Tan B, et al. Frequent engagement of the classical and alternative NF-kappaB pathways by diverse genetic abnormalities in multiple myeloma. Cancer Cell. 2007; 12:115-130.
19. Chen F, Castranova V, Shi X, Demers LM. New insights into the role of nuclear factor-kappaB, a ubiquitous transcription factor in the initiation of diseases. Clin Chem. 1999; 45:7-17.
20. Heron E, Deloukas P, van Loon AP. The complete exon-intron structure of the 156-kb human gene NFKB1, which encodes the p105 and p50 proteins of transcription factors NF-kappa B and I kappa B-gamma: implications for NF-kappa B-mediated signal transduction. Genomics. 1995; 30:493-505.
21. Karban AS, Okazaki T, Panhuysen CI, Gallegos T, Potter JJ, Bailey-Wilson JE, Silverberg MS, Duerr RH, Cho JH, Gregersen PK, Wu Y, Achkar JP, Dassopoulos T, et al. Functional annotation of a novel NFKB1 promoter polymorphism that increases risk for ulcerative colitis. Hum Mol Genet. 2004; 13:35-45.
22. Hegazy DM, O’Reilly DA, Yang BM, Hodgkinson AD, Millward BA, Demaine AG. NFkappaB polymorphisms and susceptibility to type 1 diabetes. Genes Immun. 2001; 2:304-308.
23. Cheng CW, Su JL, Lin CW, Su CW, Shih CH, Yang SF, Chien MH. Effects of NFKB1 and NFKBIA gene polymorphisms on hepatocellular carcinoma susceptibility and clinicopathological features. PLoS One. 2013; 8:e56130.
24. Lo SS, Chen JH, Wu CW, Lui WY. Functional polymorphism of NFKB1 promoter may correlate to the susceptibility of gastric cancer in aged patients. Surgery. 2009; 145:280-285.
25. Lin SC, Liu CJ, Yeh WI, Lui MT, Chang KW, Chang CS. Functional polymorphism in NFKB1 promoter is related to the risks of oral squamous cell carcinoma occurring on older male areca (betel) chewers. Cancer Lett. 2006; 243:47-54.
26. Riemann K, Becker L, Struwe H, Nuckel H, Duhrsen U, Alakus H, Winde G, Neuhauser M, Rubben H, Schmitz KJ, Wohlschlaeger J, Schmid KW, Siffert W. No association of the NFKB1 insertion/deletion promoter polymorphism with survival in colorectal and renal cell carcinoma as well as disease progression in B-cell chronic lymphocytic leukemia. Pharmacogenet Genomics. 2006; 16:783-788.
27. Lehnerdt GF, Bankfalvi A, Grehl S, Adamzik M, Lang S, Schmid KW, Siffert W, Riemann K. No association of the NF-kappaB1 -94ins/delATTG promoter polymorphism with relapse-free and overall survival in patients with squamous cell carcinomas of the head and neck region. Int J Immunopathol Pharmacol. 2008; 21:827-832.
28. Yang X, Li P, Tao J, Qin C, Cao Q, Gu J, Deng X, Wang J, Liu X, Wang Z, Wu B, Gu M, Lu Q, Yin C. Association between NFKB1 -94ins/del ATTG Promoter Polymorphism and Cancer Susceptibility: An Updated Meta-Analysis. Int J Genomics. 2014; 2014:612972.
29. Xu L, Huang S, Chen W, Song Z, Cai S. NFKB1 -94 insertion/deletion polymorphism and cancer risk: a meta-analysis. Tumour Biol. 2014; 35:5181-5187.
30. Nian X, Zhang W, Li L, Sun Y, Sun E, Han R. Meta-analysis of studies on the association between the NF-kappaB1-94ins/del ATTG promoter polymorphism and cancer. Tumour Biol. 2014; 35:11921-11931.
31. Duan W, Wang E, Zhang F, Wang T, You X, Qiao B. Association between the NFKB1-94ins/del ATTG polymorphism and cancer risk: an updated meta-analysis. Cancer Invest. 2014; 32:311-320.
32. Zou YF, Yuan FL, Feng XL, Tao JH, Ding N, Pan FM, Wang F. Association between NFKB1 -94ins/delATTG promoter polymorphism and cancer risk: a meta-analysis. Cancer Invest. 2011; 29:78-85.
33. Wang X, Lu P, Xu L, Xu Y, Shi Z, Xu J, Wang Y, Zhang J, Wang X, Cao L, Liu N, Yin Y, You Y. Updated meta-analysis of NFkappaB1 -94ins/Delattg promoter polymorphism and cancer risk based on 19 case-control studies. Asian Pac J Cancer Prev. 2011; 12:2479-2484.
34. Xu WN, Jiang ZJ, Li YH, Xiao HW, Gao Y, Pang Y, Ouyang L, Liu ZH, Zhang LQ, Wang Y, Xiao Y. [Relation of MBL ExonI 54 and NFkappaB1-94ins/del ATTG Polymorphism with Fever during Neutropenia in Patients with Acute Leukaemia after Chemotherapy]. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2015; 23:1258-1264.
35. Varga G, Mikala G, Andrikovics H, Koszarska M, Balassa K, Adam E, Kozma A, Tordai A, Masszi T. NFKB1 -94ins/delATTG polymorphism is a novel prognostic marker in first line-treated multiple myeloma. Br J Haematol. 2015; 168:679-688.
36. Dzhugashvili M, Luengo-Gil G, Garcia T, Gonzalez-Conejero R, Conesa-Zamora P, Escolar PP, Calvo F, Vicente V, Ayala de la Pena F. Role of genetic polymorphisms in NFKB-mediated inflammatory pathways in response to primary chemoradiation therapy for rectal cancer. Int J Radiat Oncol Biol Phys. 2014; 90:595-602.
37. Giachelia M, Voso MT, Tisi MC, Martini M, Bozzoli V, Massini G, D’Alo F, Larocca LM, Leone G, Hohaus S. Interleukin-6 plasma levels are modulated by a polymorphism in the NF-kappaB1 gene and are associated with outcome following rituximab-combined chemotherapy in diffuse large B-cell non-Hodgkin lymphoma. Leuk Lymphoma. 2012; 53:411-416.
38. Vangsted AJ, Klausen TW, Ruminski W, Gimsing P, Andersen NF, Gang AO, Abildgaard N, Knudsen LM, Nielsen JL, Gregersen H, Vogel U. The polymorphism IL-1beta T-31C is associated with a longer overall survival in patients with multiple myeloma undergoing auto-SCT. Bone Marrow Transplant. 2009; 43:539-545.
39. Kim JG, Sohn SK, Chae YS, Moon JH, Kim SN, Kang BW, Kim GC, Lee MH, Jeon SW, Chung HY, Yu W. No association of the NFKB1 insertion/deletion promoter polymorphism with survival in patients with gastric cancer. Jpn J Clin Oncol. 2009; 39:497-501.
40. Escobar GF, Arraes JA, Bakos L, Ashton-Prolla P, Giugliani R, Callegari-Jacques SM, Santos S, Bakos RM. Polymorphisms in CYP19A1 and NFKB1 genes are associated with cutaneous melanoma risk in southern Brazilian patients. Melanoma Res. 2016; 26:348-353.
41. Mohd Suzairi MS, Tan SC, Ahmad Aizat AA, Mohd Aminudin M, Siti Nurfatimah MS, Andee ZD, Ankathil R. The functional -94 insertion/deletion ATTG polymorphism in the promoter region of NFKB1 gene increases the risk of sporadic colorectal cancer. Cancer Epidemiol. 2013; 37:634-638.
42. Cai H, Sun L, Cui L, Cao Q, Qin C, Zhang G, Mao X, Wang M, Zhang Z, Shao P, Yin C. A functional insertion/deletion polymorphism (-94 ins/del ATTG) in the promoter region of the NFKB1 gene is related to the risk of renal cell carcinoma. Urol Int. 2013; 91:206-212.
43. Burnik FS, Yalcin S. NFKB1 -94 insertion/deletion ATTG polymorphism in gastroenteropancreatic neuroendocrine tumors. Chemotherapy. 2009; 55:381-385.
44. Lewander A, Butchi AK, Gao J, He LJ, Lindblom A, Arbman G, Carstensen J, Zhang ZY, Sun XF and Swedish Low-Risk Colorectal Cancer Study G. Polymorphism in the promoter region of the NFKB1 gene increases the risk of sporadic colorectal cancer in Swedish but not in Chinese populations. Scand J Gastroenterol. 2007; 42:1332-1338.
45. Zhang HX, Li K, Zhong HJ. Study on the association between the polymorphism of the promoter of NFKB1 and ovarian epithelial carcinomas. Journal Of Ningxia Medical Unversity. 2013; 35:296-298.
46. Deng XZ, Liao B, Yu XD, Cui S, Jiang G. Association of insertion/deletion (ATTG) polymorphism in the promoter region of NF-κB1 gene with susceptibility to transitional cell carcinoma of bladder. Chongqing Medicine. 2011; 40:1598-1600.
47. Andersen V, Christensen J, Overvad K, Tjonneland A, Vogel U. Polymorphisms in NFkB, PXR, LXR and risk of colorectal cancer in a prospective study of Danes. BMC Cancer. 2010; 10:484.
48. Bu H, Rosdahl I, Sun XF, Zhang H. Importance of polymorphisms in NF-kappaB1 and NF-kappaBIalpha genes for melanoma risk, clinicopathological features and tumor progression in Swedish melanoma patients. J Cancer Res Clin Oncol. 2007; 133:859-866.
49. Yin J, Wang H, Vogel U, Wang C, Hou W, Ma Y. Association and interaction of NFKB1 rs28362491 insertion/deletion ATTG polymorphism and PPP1R13L and CD3EAP related to lung cancer risk in a Chinese population. Tumour Biol. 2016; 37:5467-5473.
50. Rybka J, Gebura K, Wrobel T, Wysoczanska B, Stefanko E, Kuliczkowski K, Bogunia-Kubik K. Variations in genes involved in regulation of the nuclear factor - kappaB pathway and the risk of acute myeloid leukaemia. Int J Immunogenet. 2016; 43:101-106.
51. Lu ZH, Gu XJ, Shi KZ, Li X, Chen DD, Chen L. Association between genetic polymorphisms of inflammatory response genes and the risk of ovarian cancer. J Formos Med Assoc. 2016; 115:31-37.
52. Eskandari-Nasab E, Hashemi M, Ebrahimi M, Amininia S. The functional 4-bp insertion/deletion ATTG polymorphism in the promoter region of NF-KB1 reduces the risk of BC. Cancer Biomark. 2016; 16:109-115.
53. Zhang JW, Chen QS, Zhai JX, Lv PJ, Sun XY. Polymorphisms in NF-kappaB pathway genes & their association with risk of lung cancer in the Chinese population. Pak J Med Sci. 2015; 31:1411-1416.
54. Wang X, Peng H, Liang Y, Sun R, Wei T, Li Z, Gong Y, Gong R, Liu F, Zhang L, Zhu J. A functional insertion/deletion polymorphism in the promoter region of the NFKB1 gene increases the risk of papillary thyroid carcinoma. Genet Test Mol Biomarkers. 2015; 19:167-171.
55. Wang Y, Chen L, Pan L, Xue J, Yu H. The association between NFKB1-94ins/del ATTG polymorphism and non-small cell lung cancer risk in a Chinese Han population. Int J Clin Exp Med. 2015; 8:8153-8157.
56. Pallavi S, Anoop K, Showket H, Alo N, Mausumi B. NFKB1/NFKBIa polymorphisms are associated with the progression of cervical carcinoma in HPV-infected postmenopausal women from rural area. Tumour Biol. 2015; 36:6265-6276.
57. Liu Y, Qiu F, Yang L, Yang R, Yang X, Huang D, Fang W, Zhang L, Jiang Q, Zhang L, Zhou Y, Lu J. Polymorphisms of NFkappaB1 and IkappaBalpha and Their Synergistic Effect on Nasopharyngeal Carcinoma Susceptibility. Biomed Res Int. 2015; 2015:362542.
58. Li X, Zhang C, Qiao W, Zhou X, Sun M. NFKB1 -94ins/del ATTG polymorphism increases osteosarcoma risk in a Chinese Han population. Int J Clin Exp Med. 2015; 8:1420-1423.
59. Kopp TI, Andersen V, Tjonneland A, Vogel U. Polymorphisms in NFKB1 and TLR4 and interaction with dietary and life style factors in relation to colorectal cancer in a Danish prospective case-cohort study. PLoS One. 2015; 10:e0116394.
60. Han X, Zhang JJ, Yao N, Wang G, Mei J, Li B, Li C, Wang ZA. Polymorphisms in NFKB1 and NFKBIA Genes Modulate the Risk of Developing Prostate Cancer among Han Chinese. Med Sci Monit. 2015; 21:1707-1715.
61. Cui X, Yan H, Ou TW, Jia CS, Wang Q, Xu JJ. Genetic Variations in Inflammatory Response Genes and Their Association with the Risk of Prostate Cancer. Biomed Res Int. 2015; 2015:674039.
62. Chen LP, Cai PS, Liang HB. Association of the genetic polymorphisms of NFKB1 with susceptibility to ovarian cancer. Genet Mol Res. 2015; 14:8273-8282.
63. Zhang Q, Ji XW, Hou XM, Lu FM, Du Y, Yin JH, Sun XY, Deng Y, Zhao J, Han X, Yang GS, Zhang HW, Chen XM, Shen HB, Wang HY, Cao GW. Effect of functional nuclear factor-kappaB genetic polymorphisms on hepatitis B virus persistence and their interactions with viral mutations on the risk of hepatocellular carcinoma. Ann Oncol. 2014; 25:2413-2419.
64. Wang Z, Liu QL, Sun W, Yang CJ, Tang L, Zhang X, Zhong XM. Genetic polymorphisms in inflammatory response genes and their associations with breast cancer risk. Croat Med J. 2014; 55:638-646.
65. Oltulu YM, Coskunpinar E, Ozkan G, Aynaci E, Yildiz P, Isbir T, Yaylim I. Investigation of NF-kappaB1 and NF-kappaBIA gene polymorphism in non-small cell lung cancer. Biomed Res Int. 2014; 2014:530381.
66. Hua T, Qinsheng W, Xuxia W, Shuguang Z, Ming Q, Zhenxiong L, Jingjie W. Nuclear factor-kappa B1 is associated with gastric cancer in a Chinese population. Medicine (Baltimore). 2014; 93:e279.
67. Gao J, Xu HL, Gao S, Zhang W, Tan YT, Rothman N, Purdue M, Gao YT, Zheng W, Shu XO, Xiang YB. Genetic polymorphism of NFKB1 and NFKBIA genes and liver cancer risk: a nested case-control study in Shanghai, China. BMJ Open. 2014; 4:e004427.
68. Umar M, Upadhyay R, Kumar S, Ghoshal UC, Mittal B. Association of common polymorphisms in TNFA, NFkB1 and NFKBIA with risk and prognosis of esophageal squamous cell carcinoma. PLoS One. 2013; 8:e81999.
69. Li P, Gu J, Yang X, Cai H, Tao J, Yang X, Lu Q, Wang Z, Yin C, Gu M. Functional promoter -94 ins/del ATTG polymorphism in NFKB1 gene is associated with bladder cancer risk in a Chinese population. PLoS One. 2013; 8:e71604.
70. Kopp TI, Friis S, Christensen J, Tjonneland A, Vogel U. Polymorphisms in genes related to inflammation, NSAID use, and the risk of prostate cancer among Danish men. Cancer Genet. 2013; 206:266-278.
71. Huo ZH, Zhong HJ, Zhu YS, Xing B, Tang H. Roles of functional NFKB1 and beta-TrCP insertion/deletion polymorphisms in mRNA expression and epithelial ovarian cancer susceptibility. Genet Mol Res. 2013; 12:3435-3443.
72. Huang D, Yang L, Liu Y, Zhou Y, Guo Y, Pan M, Wang Y, Tan Y, Zhong H, Hu M, Lu W, Ji W, Wang J, et al. Functional polymorphisms in NFkappaB1/IkappaBalpha predict risks of chronic obstructive pulmonary disease and lung cancer in Chinese. Hum Genet. 2013; 132:451-460.
73. Arisawa T, Tahara T, Shiroeda H, Yamada K, Nomura T, Yamada H, Hayashi R, Matsunaga K, Otsuka T, Nakamura M, Shimasaki T, Toshikuni N, Kawada N, Shibata T. Functional promoter polymorphisms of NFKB1 influence susceptibility to the diffuse type of gastric cancer. Oncol Rep. 2013; 30:3013-3019.
74. Vangsted AJ, Nielsen KR, Klausen TW, Haukaas E, Tjonneland A, Vogel U. A functional polymorphism in the promoter region of the IL1B gene is associated with risk of multiple myeloma. Br J Haematol. 2012; 158:515-518.
75. Ungerback J, Belenki D, Jawad ul-Hassan A, Fredrikson M, Fransen K, Elander N, Verma D, Soderkvist P. Genetic variation and alterations of genes involved in NFkappaB/TNFAIP3- and NLRP3-inflammasome signaling affect susceptibility and outcome of colorectal cancer. Carcinogenesis. 2012; 33:2126-2134.
76. Lin CW, Hsieh YS, Hsin CH, Su CW, Lin CH, Wei LH, Yang SF, Chien MH. Effects of NFKB1 and NFKBIA gene polymorphisms on susceptibility to environmental factors and the clinicopathologic development of oral cancer. PLoS One. 2012; 7:e35078.
77. Fan Y, Yu W, Ye P, Wang H, Wang Z, Meng Q, Duan Y, Liang X, An W. NFKB1 insertion/deletion promoter polymorphism increases the risk of advanced ovarian cancer in a Chinese population. DNA Cell Biol. 2011; 30:241-245.
78. Zhou B, Qie M, Wang Y, Yan L, Zhang Z, Liang A, Wang T, Wang X, Song Y, Zhang L. Relationship between NFKB1 -94 insertion/deletion ATTG polymorphism and susceptibility of cervical squamous cell carcinoma risk. Ann Oncol. 2010; 21:506-511.
79. Tang T, Cui S, Deng X, Gong Z, Jiang G, Wang P, Liao B, Fei Z, Xian S, Zeng D, Li J. Insertion/deletion polymorphism in the promoter region of NFKB1 gene increases susceptibility for superficial bladder cancer in Chinese. DNA Cell Biol. 2010; 29:9-12.
80. Zhou B, Rao L, Li Y, Gao L, Wang Y, Chen Y, Xue H, Song Y, Peng Y, Liao M, Zhang L. A functional insertion/deletion polymorphism in the promoter region of NFKB1 gene increases susceptibility for nasopharyngeal carcinoma. Cancer Lett. 2009; 275:72-76.
81. Zhang P, Wei Q, Li X, Wang K, Zeng H, Bu H, Li H. A functional insertion/deletion polymorphism in the promoter region of the NFKB1 gene increases susceptibility for prostate cancer. Cancer Genet Cytogenet. 2009; 191:73-77.
82. He Y, Zhang H, Yin J, Xie J, Tan X, Liu S, Zhang Q, Li C, Zhao J, Wang H, Cao G. IkappaBalpha gene promoter polymorphisms are associated with hepatocarcinogenesis in patients infected with hepatitis B virus genotype C. Carcinogenesis. 2009; 30:1916-1922.
83. Riemann K, Becker L, Struwe H, Rubben H, Eisenhardt A, Siffert W. Insertion/deletion polymorphism in the promoter of NFKB1 as a potential molecular marker for the risk of recurrence in superficial bladder cancer. Int J Clin Pharmacol Ther. 2007; 45:423-430.
84. Song HL, Li L. Relationship between genetic polymorphism of NF-кB signaling pathway and endometrial carcinoma. Shandong Medical Journal. 2013; 53:12-14.
85. Song HL, Zhang JQ. Genetic polymorphism of NF-κB signaling pathway in patients with cervical cancer. Chinese Journal Of Laboratory Diagnosis. 2013; 17:1251-1253.
86. Liu YH, Yang L, Qiu FM, Lv JC, Ji WD. Study on the association between the polymorphism of the promoter of factor-κB1 gene and nasopharyngeal carcinoma in Guangdong. Modern Preventive Medicine. 2013; 40:2185-2188.
87. Tang GT, Li XP, Liu TQ, Yang JR, Liang ZX. Genetic polymorphism of NF-κB signaling pathway in patients with primary liver cancer. Guangdong Medical Journal. 2012; 08.
88. Song QL, He XX, Yang H, Li J, Chen M, Wang MY, Liu Q, Yu JL, Yao JJ, Liu LF, Sun SZ, Lin JS. Association of a TANK gene polymorphism with outcomes of hepatitis B virus infection in a Chinese Han population. Viral Immunol. 2012; 25:73-78.
89. He YC, Yin JH, Xie JX, Zhang HW, Liu SJ, Chang WJ, Li CZ, Zhang Q, Ma LY, Gao GW. Relationship between polymorphism of NF-κB1 and NF-κBIα and susceptibility to hepatitis B virus-associated hepatocellular carcinoma: a case-control study. Academic Journal Of Second Military Medical University. 2009; 30:349.
90. Lim WY, Chen Y, Ali SM, Chuah KL, Eng P, Leong SS, Lim E, Lim TK, Ng AW, Poh WT, Tee A, Teh M, Salim A, Seow A. Polymorphisms in inflammatory pathway genes, host factors and lung cancer risk in Chinese female never-smokers. Carcinogenesis. 2011; 32:522-529.
91. Tan SC, Suzairi MS, Aizat AA, Aminudin MM, Nurfatimah MS, Bhavaraju VM, Biswal BM, Ankathil R. Gender-specific association of NFKBIA promoter polymorphisms with the risk of sporadic colorectal cancer. Med Oncol. 2013; 30:693.
92. Song S, Chen D, Lu J, Liao J, Luo Y, Yang Z, Fu X, Fan X, Wei Y, Yang L, Wang L, Wang J. NFkappaB1 and NFkappaBIA polymorphisms are associated with increased risk for sporadic colorectal cancer in a southern Chinese population. PLoS One. 2011; 6:e21726.
93. Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA, Jr., Kinzler KW. Cancer genome landscapes. Science. 2013; 339:1546-1558.
94. Xie S, Shan XF, Shang K, Xu H, He J, Cai ZG. Relevance of LIG4 gene polymorphisms with cancer susceptibility: evidence from a meta-analysis. Sci Rep. 2014; 4:6630.
95. He J, Wang MY, Qiu LX, Zhu ML, Shi TY, Zhou XY, Sun MH, Yang YJ, Wang JC, Jin L, Wang YN, Li J, Yu HP, Wei QY. Genetic variations of mTORC1 genes and risk of gastric cancer in an Eastern Chinese population. Mol Carcinog. 2013; 52:E70-79.
96. He J, Shi TY, Zhu ML, Wang MY, Li QX, Wei QY. Associations of Lys939Gln and Ala499Val polymorphisms of the XPC gene with cancer susceptibility: a meta-analysis. Int J Cancer. 2013; 133:1765-1775.
97. He J, Xi B, Ruiter R, Shi TY, Zhu ML, Wang MY, Li QX, Zhou XY, Qiu LX, Wei QY. Association of LEP G2548A and LEPR Q223R polymorphisms with cancer susceptibility: evidence from a meta-analysis. PLoS One. 2013; 8:e75135.
98. He J, Liao XY, Zhu JH, Xue WQ, Shen GP, Huang SY, Chen W, Jia WH. Association of MTHFR C677T and A1298C polymorphisms with non-Hodgkin lymphoma susceptibility: evidence from a meta-analysis. Sci Rep. 2014; 4:6159.
99. He J, Wang F, Zhu JH, Chen W, Cui Z, Jia WH. No association between MTR rs1805087 A > G polymorphism and non-Hodgkin lymphoma susceptibility: evidence from 11 486 subjects. Leuk Lymphoma. 2015; 56:763-767.

