INTRODUCTION
Cervical cancer (CC) is one of the most common malignancies among women worldwide, particularly in developing countries [1].CC accounts for 9% of the total new cancer cases and 8% of the total cancer deaths among women [2]. Despite preventive strategies and innovative treatments, it is estimated that by the year 2020, there will be 609,270 new CC cases and 317,727 deaths [1]. Although experimental and epidemiological evidence indicates that infection with high-risk human papillomavirus (hrHPV) is the main CC etiologic agent, it is not sufficient to cause the malignancy [3]. Rather, CC results from interactions of various factors, including the HPV infection, environmental, behavioral, and genetic factors [4, 5].
Pathogenesis of CC is a multistep process that results from the accumulation of several genomic alterations, and is characterized by unrestricted proliferation, invasion and metastasis [6]. MicroRNAs (miRNAs) are small (18–25 nucleotides) non-coding RNAs that modulate post-transcriptional mRNA expression. Since miRNAs regulate expression of genes involved in cell proliferation, differentiation and apoptosis, they can function as potential oncogenes or tumor suppressors [7–11]. We have previously found that chromosome mutations and the change of single nucleotide polymorphisms (SNPs) are important factors that induce malignant transformation of cervical epithelial cells [12].
In this case-control study, we have investigated the relationship between the SNPs in miRNA genes and the risk of CC, and performed a comprehensive association analysis in China Xinjiang Uygur population.
RESULTS
A total of 247 CC patients and 285 healthy subjects were enrolled in our study. Detailed information about the SNPs selected is presented in Table 1. As a risk factor, the minor allele of each SNP was compared with the wild-type allele. All of the tested SNPs were in agreement with the HWE in the control population of this study (p > 0.05) except for rs8756 (p = 0.040) and rs11175982 (p = 0.024); therefore, they were excluded from the analysis. Comparing the differences in frequency distributions of alleles between cases and controls by χ2 test, we found there is a correlation between two loci (rs300574, SPRY1, OR = 1.312, 95% CI: 1.034–1.677, p = 0.026; rs1042725, HMGA2, OR = 1.309, 95% CI: 1.009–1.699, p = 0.043) and increased CC development under allele model. On the contrary, the T allele of rs3744935 (BCL2, OR = 0.450, 95% CI: 0.214–0.947, p = 0.031) was found to be a protective factor (Table 1). Besides, the other loci under allele model had not been found to be associated with the disease. We also performed a Bonferroni correction and determined that none of the SNPs showed statistically significant associations with CC risk.
Table 1: Basic information of SNPs in this study
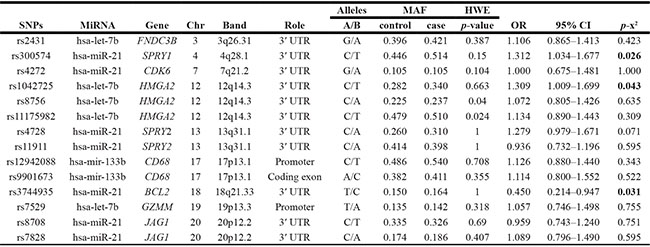
SNPs: Single nucleotide polymorphisms; MiRNA: microRNA; Chr: chromosome; MAF: Minor allele frequency; HWE: Hardy-Weinberg equilibrium; OR: Odds ratio; CI: Confidence interval.
P-value was calculated by Pearson’s χ2 test.
P value < 0.05 indicates statistical significance.
Further model analysis was conducted by unconditional logistic regression analysis and only the SNPs associated with CC were included (Table 2). We found that rs300574 was associated with CC under codominant model (OR = 1.79, 95% CI: 1.10–2.91, p = 0.004), recessive model (OR = 1.97, 95% CI: 1.31–2.97, p = 0.001), over-dominant model (OR = 0.66, 95% CI: 0.47–0.93, p = 0.018) and log-additive model (OR = 1.31, 95% CI: 1.03–1.67, p = 0.026). We also noticed that rs1042725 was associated with increased CC risk in dominant model (OR = 1.50, 95% CI: 1.06–2.11, p = 0.021) and log-additive model (OR = 1.34, 95% CI: 1.02–1.76, p = 0.035). Besides, rs3744935 actually decreased the risk of CC under codominant model (OR = 0.44, 95% CI: 0.21–0.93, p = 0.026). In addition, no positive results were observed after Bonferroni correction.
Table 2: Unconditional logistic regression analysis of the association between the single-nucleotide polymorphisms and CC risk
SNPs |
Model |
Genotype |
Controls (n %) |
Cases (n %) |
OR (95% CI) |
P-value |
AIC |
BIC |
|---|---|---|---|---|---|---|---|---|
rs300574 |
Codominant |
T/T |
81 (28.4%) |
66 (26.7%) |
1 |
|||
T/C |
154 (54%) |
108 (43.7%) |
0.86 (0.57–1.29) |
|||||
C/C |
50 (17.5%) |
73 (29.6%) |
1.79 (1.10–2.91) |
0.004 |
729.5 |
742.4 |
||
Dominant |
T/T |
81 (28.4%) |
66 (26.7%) |
1 |
||||
T/C-C/C |
204 (71.6%) |
181 (73.3%) |
1.09 (0.74–1.59) |
0.660 |
738.6 |
747.2 |
||
Recessive |
T/T-T/C |
235 (82.5%) |
174 (70.5%) |
1 |
||||
C/C |
50 (17.5%) |
73 (29.6%) |
1.97 (1.31–2.97) |
0.001 |
728.1 |
736.6 |
||
Over-dominant |
T/T-C/C |
131 (46%) |
139 (56.3%) |
1 |
||||
T/C |
154 (54%) |
108 (43.7%) |
0.66 (0.47–0.93) |
0.018 |
733.2 |
741.7 |
||
Log-additive |
--- |
--- |
--- |
1.31 (1.03–1.67) |
0.026 |
733.9 |
742.4 |
|
rs1042725 |
Codominant |
T/T |
145 (50.9%) |
101 (40.9%) |
1 |
|||
C/T |
119 (41.8%) |
124 (50.2%) |
1.50 (1.05–2.14) |
0.070 |
735.5 |
748.3 |
||
C/C |
21 (7.4%) |
22 (8.9%) |
1.50 (0.79–2.88) |
|||||
Dominant |
T/T |
145 (50.9%) |
101 (40.9%) |
1 |
||||
C/T-C/C |
140 (49.1%) |
146 (59.1%) |
1.50 (1.06–2.11) |
0.021 |
733.5 |
742 |
||
Recessive |
T/T-C/T |
264 (92.6%) |
225 (91.1%) |
1 |
||||
C/C |
21 (7.4%) |
22 (8.9%) |
1.23 (0.66–2.29) |
0.520 |
738.4 |
746.9 |
||
Over-dominant |
T/T-C/C |
166 (58.2%) |
123 (49.8%) |
1 |
||||
C/T |
119 (41.8%) |
124 (50.2%) |
1.41 (1.00–1.98) |
0.051 |
735 |
743.5 |
||
--- |
--- |
--- |
1.34 (1.02–1.76) |
0.035 |
734.4 |
742.9 |
||
rs3744935 |
Codominant |
C/C |
260 (91.2%) |
237 (96%) |
1 |
|||
T/C |
25 (8.8%) |
10 (4%) |
0.44 (0.21–0.93) |
0.026 |
733.8 |
742.4 |
||
T/T |
0 |
0 |
--- |
--- |
--- |
--- |
SNPs: Single nucleotide polymorphisms; OR: Odds ratio; CI: Confidence interval.
P-value was calculated by Pearson’s χ2 test.
P-value < 0.05 indicates statistical significance.
The association between SNPs and different CC patient clinical subtypes was also analyzed only positive results (Table 3). From the results, we found that rs300574 (OR = 1.404, 95% CI: 1.060–1.860, p = 0.018) and rs1042725 (OR = 1.403, 95% CI: 1.040–1.892, p = 0.026)were associated with clinical stage I/IICC patients. Rs4728 (OR = 1.473, 95% CI: 1.029–2.110, p = 0.034) was associated with an increased CC risk in clinical stage III/IVcases. In addition, rs1042725 (OR = 3.266, 95% CI: 1.196–8.918, p = 0.015) was associated with adenomatous carcinoma patients. Rs300574 (OR = 1.330, 95% CI: 1.042–1.698, p = 0.022) and rs3744935 (OR=0.371, 95% CI: 0.166–0.831, p = 0.012)were associated with squamous carcinoma patients.
Table 3: The association between SNPs and different clinical subtypes of CC patients
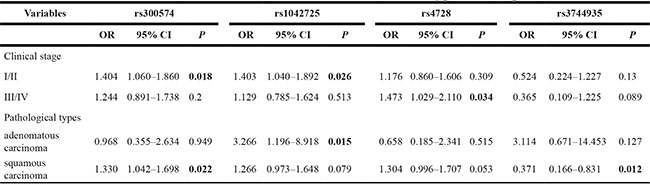
SNPs: Single nucleotide polymorphisms; OR: Odds ratio; CI: Confidence interval.
P-value was calculated by Pearson’s χ2 test.
P-value <0.05 indicates statistical significance.
DISCUSSION
The goal of this study was to explore the correlation of SNPs with the susceptibility to CC in Xinjiang Uygur population. We have identified four SNPs: rs300574 (SPRY1), rs3744935 (BCL2), rs1042725 (HMGA2), and rs4728 (SPRY2) that are associated with CC risk in Xinjiang Uygur population.
The minor allele “C” of rs300574 in SPRY1 gene was associated with an increased risk of CC based on the analytic results of the allele, codominant, recessive and log-additive model, but an opposite result was found in the over-dominant model. The minor allele “C” of rs1042725 in HMGA2 gene was associated with an increased risk of CC under the allele, dominant and log-additive model. HMGA2 rs1042725 has been reported to contribute to height variability in European population [13], and US Caucasian and Chinese populations [14], but not in Korean [15] and Japanese population [16] To our knowledge, this is the first study that reports the association between rs1042725 in HMGA2 gene and cancer. In addition, in clinical stage III/IVCC patients, rs4728 in SPRY2 gene was associated with a decreased risk. Finally, we also found that the minor allele “T” of rs3744935 in BCL2 gene was associated with CC under allele and codominant model. The above two loci have not been reported previously.
Recent studies have revealed that miRNA deregulation correlates with various human cancers and is involved in the initiation and progression of human tumors [17]. Since the first miRNA lin-4 was discovered in Caenorhabditis elegans, miRNA-dependent gene regulation has been widely investigated [18, 19]. As miRNAs can inhibit mRNA translation or induce mRNA degradation, thus regulating a wide range of biological processes including cell proliferation, differentiation and apoptosis abnormal miRNA expression is a common feature of human cancers.
Homo sapiens miR-21 (hsa-miR-21) is one of the first miRNAs detected in the human genome and is the major oncogene up-regulated in many types of human cancer including glioblastoma multiforme [20], breast [21], lung [22], esophageal gastrointestinal [23], hepatocellular [24], cholangiocarcinoma [25], pancreatic [26], ovarian [27], bladder [28], NK-cell lymphoma [29], laryngeal carcinoma [30] and tongue squamous cell carcinoma [31]. Aldaz et al. found that by direct 3′ -UTR binding, miR-21 up-regulation decreases SPRY1 expression, thus contributing to cancer development [32]. Thus, we speculate that the mechanism by which miRNA gene SPRY1 increases the risk of CC development might be similar to the study by Aldaz et al. SPRY2 has also been reported to promote apoptosis of cancer cells which is associated with activation of the phosphatase and tensin homolog deleted on chromosome 10 (PTEN) pathway and the blockade of Ras-Raf-Erk signaling [33]. In addition, it was suggested high BCL2 expression were associated with unfavourable prognostic in diffuse large B-cell lymphoma [34]. Sung Han Kim et al. found that BCL2 gene might play distinctive roles in cisplatin resistance in bladder cancer [35].
Hsa-let-7b, a member of hsa-let-7 family of tumor suppressor miRNAs, possesses a high homology to 3′-UTR of transcripts encoding for proteins involved in proliferation, differentiation and cell death [36]. HMGA2 (High Mobility Group AT-2 hook) belongs to HMG (High Mobility Group) family of proteins, and is an essential component of the enhanceosome, which drives DNA to the transcriptional complexes [37, 38]. HMGA2 expression correlates with metastases and reduced survival, and is increased in several malignancies, such as lung, prostate, colon, pancreatic, gastric and breast cancer [39–43]. In addition, it has been reported that hsa-let-7b is able to regulate targets HMGA2 and the absence of hsa-let-7b has been linked to high levels of HMGA2 [39]. Di Fazio et al. found that HMGA2 expression was controlled by tumor suppressor miRNA hsa-let-7b after inhibition of deacetylases in liver cancer cell lines [44]. Therefore, it seems plausible that downregulation of hsa-let-7b leads to increased levels of HMGA2, which further contributes to the generation of liver cancer. Though the association between hsa-let-7b, HMGA2 and CC development has not been reported previously, we hypothesize that guessed the functional mechanism might be similar to that in liver cancer.
We found no statistically significant association between SNPs and the risk of CC using Bonferroni correction in our statistical analysis. This may be due to the relatively small sample size, the selection criteria for SNPs (minor allele frequency [MAF] > 5%), and the weakness of Bonferroni correction itself (the interpretation of a finding depends on the number of other tests performed). Future studies should confirm our conclusions using a larger sample size, other population groups, consider patients’ age, as well as other factors, such as smoking, bacterial and viral infections, and social status.
In summary, we have identified novel associations between four SNPs, rs300574 (SPRY1), rs3744935 (BCL2), rs1042725 (HMGA2) and rs4728 (SPRY2) with CC risk in Xinjiang Uygur population. This may provide new strategies for CC screening and identify new genes and mechanisms of CC pathogenesis.
MATERIALS AND METHODS
Study participants
In this case-control study, a total of 247 patients with invasive cervical cancer and 285 healthy women were recruited at People’s Hospital of Xinjiang Uyghur Autonomous Region from January 2014 to June 2016. The included patients were recently diagnosed by cervical biopsy and histopathologically confirmed as primary CC. We excluded the patients with other cancers who underwent radiotherapy or chemotherapy. The controls who had an annual health check were recruited from the health checkup center of the same hospitals. All the controls were matched with the cases, and all of them had no history of cancer.
Tumors were staged according to International Federation of Gynecology and Obstetrics (FIGO) classification. The factors that could influence the mutation rate were minimized. All participants enlisted were women at least 18 years old with good mental condition and no blood relationship going back three generations. Besides, both cohorts belong to the same ethnically homogenous population (Xinjiang Uygur population).
Informed consents were obtained from all participants and the study protocols were approved by the institutional review board of People’s Hospital of Xinjiang Uyghur Autonomous Region.
SNP selection and genotyping
Validated SNPs, associated with other cancers published in previous studies, were selected with a minor allele frequency (MAF) > 5% in the HapMap Asian population [45–53]. Venous blood samples (5 ml) were collected from each patient during laboratory examination. For patients, blood was collected prior to radiation or chemotherapy. DNA was extracted from whole blood samples using the Gold Mag-Mini Whole Blood Genomic DNA Purification Kit (GoldMag Ltd., Xi’an, China) and stored at−80°C after centrifugation. DNA concentration was evaluated by spectrometry (DU530 UV/VIS spectrophotometer, Beckman Instruments, Fullerton, CA, USA). Sequenom MassARRAY Assay Design 3.0 software (Sequenom, Inc, San Diego, CA, USA) was used to design multiplexed SNP Mass EXTEND assay. The SNP genotypes were performed by a Sequenom MassARRAY RS1000 (Sequenom, Inc) according to the standard protocol recommended by the manufacturer. The Sequenom Typer 4.0 Software™ (Sequenom, Inc) was used to perform data management and analyses. All primers were made by Sangon (Shanghai, China), their sequences are available upon request. The corresponding primers used for each SNP in our study are listed in Table 4. As a result, fourteen SNPs were selected including: rs2431, rs300574, rs4272, rs1042725, rs8756, rs11175982, rs4728, rs11911, rs12942088, rs9901673, rs3744935, rs7529, rs8708 and rs7828. The SNPs genetic information included in this study is shown in Table 1.
Table 4: Primers used for this study
SNP_ID |
1st-PCRP |
2nd-PCRP |
UEP_SEQ |
|---|---|---|---|
rs2431 |
ACGTTGGATGTTTTAGGT CAGTCTTGTTCC |
ACGTTGGATGATGCACTAG ATGCTCTTATC |
CAGAAGATTAATAATACT ACAGATG |
rs300574 |
ACGTTGGATGGGGTCAGGG TAAACCATCAT |
ACGTTGGATGCAAAAAACA GCCACAACTTG |
AAAAAACAGCCACAACT TGAAAGCTA |
rs4272 |
ACGTTGGATGGATAAAA CACCTAGATACCC |
ACGTTGGATGCTAAGCCCCC AAATAAGCTG |
AAATAAGCTGCATGCATT TGTAACA |
rs1042725 |
ACGTTGGATGGCATTGC TTGTGTATAGCAG |
ACGTTGGATGCCCCACTA CTCAATACTACC |
ACTACCTCTGAATGTTACAA |
rs8756 |
ACGTTGGATGCTCCTGCAA ACATACCTAAC |
ACGTTGGATGTTCTTTGC TGTTGTTGGTCG |
gGTTGTTGGTCGCAGCTA |
rs11175982 |
ACGTTGGATGCCTGCTCTT TGTTACAGTTC |
ACGTTGGATGATGACTTG TGAGTGTCTCCC |
ACTTGTGAGTGTCTCCCT CCTTGATCT |
rs4728 |
ACGTTGGATGGTGGTTCA GTATTATGTACG |
ACGTTGGATGAATTGTGTG AACTTGGAAGC |
TTGGAAGCACACCAATCT |
rs11911 |
ACGTTGGATGGTTGGTTG CATATGTGTAT |
ACGTTGGATGCCCTAGGA ACTTTGAAAAACC |
ACAGTAATGAGGATTTTTTCT |
rs12942088 |
ACGTTGGATGTCTAGCCAC CTCTCTCTTAG |
ACGTTGGATGACCCATGT GACACTGTTGAC |
ACTGTTGACTTTGCC CTGA |
rs9901673 |
ACGTTGGATGCATGGCGGT GGAGTACAATG |
ACGTTGGATGCGTCTAGTG GTAGCAATGAG |
AAGGGAAGGAGGTT ACTTAC |
rs3744935 |
ACGTTGGATGCCTCTACA GTGATACATGTC |
ACGTTGGATGTGTGGCTG GGCCTGTCACC |
aGCCCTCCAGGTAGGCCC |
rs7529 |
ACGTTGGATGTGCCTGT CAGCTCCAGGTC |
ACGTTGGATGCAAAGCAGG GAAAAGAGTCG |
CGGCCACGGTGAATCCGT CCACTC |
rs8708 |
ACGTTGGATGGAGCTTAC GTAGTTCTACCG |
ACGTTGGATGTACCCCGATT ACCAGAACAG |
AGGGTGGCAGAAGCACA |
rs7828 |
ACGTTGGATGGTGGTTCA GTATTATGTACG |
ACGTTGGATGAATTGTGTGAA CTTGGAAGC |
TTGGAAGCACACCAATCT |
Statistical analysis
All statistical analyses were conducted by SPSS version 17.0 statistical package (SPSS, Chicago, IL, USA) and Microsoft Excel. Pearson’s χ2 test was used to compare the distribution of categorical variables and Student’s t-test was used for continuous variables. Hardy–Weinberg equilibrium (HWE) of each SNP was tested by an exact test to compare the expected frequency of controls. Allele and genotype frequencies for each SNP of CC patients and control subjects were compared by χ2 test. Odds ratios (ORs) and 95% confidence intervals (CIs) were tested by unconditional logistic regression analysis. We used SNP analysis (http://pngu.mgh.harvard.edu/Purcell/plink/), website software to test the associations between certain SNPs and the risk of CC in five models (Codominant, Dominant, Recessive, Over-dominant and Log-additive model). For all results, p values presented in this study are two-sided and p < 0.05 was considered to represent statistically significant.
Abbreviations
SNP: single nucleotide polymorphism; CC: cervical cancer; MAF: minor allele frequencies; HWE: Hardy-Weinberg Equilibrium; OR: odds ratio; CI: confidence interval.
ACKNOWLEDGMENTS AND FUNDING
This work was supported by The Natural Science Foundation of Xinjiang Uygur autonomous region (No. 2014211A061). The authors are also grateful to all participants in the study. We thank the clinicians and hospital staff who contributed to the sample and data collection for this study.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
REFERENCES
1. Vargas-Torres SL, Portari EA, Silva AL, Klumb EM, da Rocha Guillobel HC, de Camargo MJ, Santos-Reboucas CB, Russomano FB, Macedo JM. Roles of CDKN1A gene polymorphisms (rs1801270 and rs1059234) in the development of cervical neoplasia. Tumour biology. 2016.
2. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. Cancer J Clin. 2015; 65:87–108.
3. Wang Q, Zhang C, Walayat S, Chen HW, Wang Y. Association between cytokine gene polymorphisms and cervical cancer in a Chinese population. European journal of obstetrics, gynecology, and reproductive biology. 2011; 158:330–333.
4. de Freitas AC, Gurgel AP, Chagas BS, Coimbra EC and do Amaral CM. Susceptibility to cervical cancer: an overview. Gynecologic oncology. 2012; 126:304–311.
5. Munoz N, Castellsague X, de Gonzalez AB, Gissmann L. Chapter 1: HPV in the etiology of human cancer. Vaccine. 2006; 24:1–10.
6. Berezikov E, Guryev V, van de Belt J, Wienholds E, Plasterk RH, Cuppen E. Phylogenetic shadowing and computational identification of human microRNA genes. Cell. 2005; 120:21–24.
7. Macfarlane LA, Murphy PR. MicroRNA: Biogenesis, Function and Role in Cancer. Current genomics. 2010; 11:537–561.
8. Tan GS, Garchow BG, Liu X, Metzler D, Kiriakidou M. Clarifying mammalian RISC assembly in vitro. BMC molecular biology. 2011; 12:19.
9. Ventura A, Jacks T. MicroRNAs and cancer: short RNAs go a long way. Cell. 2009; 136:586–591.
10. Visone R, Croce CM. MiRNAs and cancer. The American journal of pathology. 2009; 174:1131–1138.
11. Ruan K, Fang X, Ouyang G. MicroRNAs: novel regulators in the hallmarks of human cancer. Cancer letters. 2009; 285:116–126.
12. Lee JW, Choi CH, Choi JJ, Park YA, Kim SJ, Hwang SY, Kim WY, Kim TJ, Lee JH, Kim BG, Bae DS. Altered MicroRNA expression in cervical carcinomas. Clinical cancer research. 2008; 14:2535–2542.
13. Bouatia-Naji N, Marchand M, Cavalcanti-Proenca C, Daghmoun S, Durand E, Tichet J, Marre M, Balkau B, Froguel P, Levy-Marchal C. Smallness for gestational age interacts with high mobility group A2 gene genetic variation to modulate height. European journal of endocrinology. 2009; 160:557–560.
14. Yang TL, Guo Y, Zhang LS, Tian Q, Yan H, Guo YF, Deng HW. HMGA2 is confirmed to be associated with human adult height. Annals of human genetics. 2010; 74:11–16.
15. Kim JJ, Lee HI, Park T, Kim K, Lee JE, Cho NH, Shin C, Cho YS, Lee JY, Han BG, Yoo HW, Lee JK. Identification of 15 loci influencing height in a Korean population. Journal of human genetics. 2010; 55:27–31.
16. Takeshita H, Fujihara J, Soejima M, Koda Y, Kimura-Kataoka K, Ono R, Yuasa I, Iida R, Ueki M, Nagao M, Yasuda T. Confirmation that SNPs in the high mobility group-A2 gene (HMGA2) are associated with adult height in the Japanese population; wide-ranging population survey of height-related SNPs in HMGA2. Electrophoresis. 2011; 32:1844–1851.
17. Asangani IA, Rasheed SA, Nikolova DA, Leupold JH, Colburn NH, Post S, Allgayer H. MicroRNA-21 (miR-21) post-transcriptionally downregulates tumor suppressor Pdcd4 and stimulates invasion, intravasation and metastasis in colorectal cancer. Oncogene. 2008; 27:2128–2136.
18. Lee RC, Feinbaum RL, Ambros V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell. 1993; 75:843–854.
19. Wightman B, Ha I, Ruvkun G. Posttranscriptional regulation of the heterochronic gene lin-14 by lin-4 mediates temporal pattern formation in C. elegans. Cell. 1993; 75:855–862.
20. Chan JA, Krichevsky AM, Kosik KS. MicroRNA-21 is an antiapoptotic factor in human glioblastoma cells. Cancer research. 2005; 65:6029–6033.
21. Iorio MV, Ferracin M, Liu CG, Veronese A, Spizzo R, Sabbioni S, Magri E, Pedriali M, Fabbri M, Campiglio M, Menard S, Palazzo JP, Rosenberg A, et al. MicroRNA gene expression deregulation in human breast cancer. Cancer research. 2005; 65:7065–7070.
22. Seike M, Goto A, Okano T, Bowman ED, Schetter AJ, Horikawa I, Mathe EA, Jen J, Yang P, Sugimura H, Gemma A, Kudoh S, Croce CM, Harris CC. MiR-21 is an EGFR-regulated anti-apoptotic factor in lung cancer in never-smokers. Proceedings of the National Academy of Sciences of the United States of America. 2009; 106:12085–12090.
23. Hiyoshi Y, Kamohara H, Karashima R, Sato N, Imamura Y, Nagai Y, Yoshida N, Toyama E, Hayashi N, Watanabe M, Baba H. MicroRNA-21 regulates the proliferation and invasion in esophageal squamous cell carcinoma. Clinical cancer research. 2009; 15:1915–1922.
24. Meng F, Henson R, Wehbe-Janek H, Ghoshal K, Jacob ST, Patel T. MicroRNA-21 regulates expression of the PTEN tumor suppressor gene in human hepatocellular cancer. Gastroenterology. 2007; 133:647–658.
25. Meng F, Henson R, Lang M, Wehbe H, Maheshwari S, Mendell JT, Jiang J, Schmittgen TD, Patel T. Involvement of human micro-RNA in growth and response to chemotherapy in human cholangiocarcinoma cell lines. Gastroenterology. 2006; 130:2113–2129.
26. Moriyama T, Ohuchida K, Mizumoto K, Yu J, Sato N, Nabae T, Takahata S, Toma H, Nagai E, Tanaka M. MicroRNA-21 modulates biological functions of pancreatic cancer cells including their proliferation, invasion, and chemoresistance. Molecular cancer therapeutics. 2009; 8:1067–1074.
27. Iorio MV, Visone R, Di Leva G, Donati V, Petrocca F, Casalini P, Taccioli C, Volinia S, Liu CG, Alder H, Calin GA, Menard S, Croce CM. MicroRNA signatures in human ovarian cancer. Cancer research. 2007; 67:8699–8707.
28. Catto JW, Miah S, Owen HC, Bryant H, Myers K, Dudziec E, Larre S, Milo M, Rehman I, Rosario DJ, Di Martino E, Knowles MA, Meuth M, et al. Distinct microRNA alterations characterize high- and low-grade bladder cancer. Cancer research. 2009; 69:8472–8481.
29. Yamanaka Y, Tagawa H, Takahashi N, Watanabe A, Guo YM, Iwamoto K, Yamashita J, Saitoh H, Kameoka Y, Shimizu N, Ichinohasama R, Sawada K. Aberrant overexpression of microRNAs activate AKT signaling via down-regulation of tumor suppressors in natural killer-cell lymphoma/leukemia. Blood. 2009; 114:3265–3275.
30. Liu M, Wu H, Liu T, Li Y, Wang F, Wan H, Li X, Tang H. Regulation of the cell cycle gene, BTG2, by miR-21 in human laryngeal carcinoma. Cell research. 2009; 19:828–837.
31. Li J, Huang H, Sun L, Yang M, Pan C, Chen W, Wu D, Lin Z, Zeng C, Yao Y, Zhang P, Song E. MiR-21 indicates poor prognosis in tongue squamous cell carcinomas as an apoptosis inhibitor. Clinical cancer research. 2009; 15:3998–4008.
32. Aldaz B, Sagardoy A, Nogueira L, Guruceaga E, Grande L, Huse JT, Aznar MA, Diez-Valle R, Tejada-Solis S, Alonso MM, Fernandez-Luna JL, Martinez-Climent JA, Malumbres R. Involvement of miRNAs in the differentiation of human glioblastoma multiforme stem-like cells. PloS one. 2013; 8:e77098.
33. Feng YH, Wu CL, Shiau AL, Lee JC, Chang JG, Lu PJ, Tung CL, Feng LY, Huang WT, Tsao CJ. MicroRNA-21-mediated regulation of Sprouty2 protein expression enhances the cytotoxic effect of 5-fluorouracil and metformin in colon cancer cells. International journal of molecular medicine. 2012; 29:920–926.
34. Brito AB, Delamain MT, de Oliveira C, Fanelli MF, Soares FA, de Souza CA, Vassallo J, Lima CS. Association of BAX G(-248)A, BCL2 C(-717)A polymorphisms with outcome in diffuse large B-cell lymphoma patients. British journal of haematology. 2016.
35. Kim SH, Ho JN, Jin H, Lee SC, Lee SE, Hong SK, Lee JW, Lee ES, Byun SS. Upregulated expression of BCL2, MCM7, and CCNE1 indicate cisplatin-resistance in the set of two human bladder cancer cell lines: T24 cisplatin sensitive and T24R2 cisplatin resistant bladder cancer cell lines. Investigative and clinical urology. 2016; 57:63–72.
36. Boyerinas B, Park SM, Hau A, Murmann AE, Peter ME. The role of let-7 in cell differentiation and cancer. Endocrine-related cancer. 2010; 17:F19–36.
37. Joynt S, Morillo V, Leng F. Binding the mammalian high mobility group protein AT-hook 2 to AT-rich deoxyoligonucleotides: enthalpy-entropy compensation. Biophysical journal. 2009; 96:4144–4152.
38. Winter N, Nimzyk R, Bosche C, Meyer A, Bullerdiek J. Chromatin immunoprecipitation to analyze DNA binding sites of HMGA2. PloS one. 2011; 6:e18837.
39. Liu Q, Lv GD, Qin X, Gen YH, Zheng ST, Liu T, Lu XM. Role of microRNA let-7 and effect to HMGA2 in esophageal squamous cell carcinoma. Molecular biology reports. 2012; 39:1239–1246.
40. Fusco A, Fedele M. Roles of HMGA proteins in cancer. Nature reviews Cancer. 2007; 7:899–910.
41. Rahman MM, Qian ZR, Wang EL, Sultana R, Kudo E, Nakasono M, Hayashi T, Kakiuchi S, Sano T. Frequent overexpression of HMGA1 and 2 in gastroenteropancreatic neuroendocrine tumours and its relationship to let-7 downregulation. British journal of cancer. 2009; 100:501–510.
42. Huang ML, Chen CC, Chang LC. Gene expressions of HMGI-C, HMGI(Y) are associated with stage and metastasis in colorectal cancer. International journal of colorectal disease. 2009; 24:1281–1286.
43. Motoyama K, Inoue H, Nakamura Y, Uetake H, Sugihara K, Mori M. Clinical significance of high mobility group A2 in human gastric cancer and its relationship to let-7 microRNA family. Clinical cancer research. 2008; 14:2334–2340.
44. Di Fazio P, Montalbano R, Neureiter D, Alinger B, Schmidt A, Merkel AL, Quint K, Ocker M. Downregulation of HMGA2 by the pan-deacetylase inhibitor panobinostat is dependent on hsa-let-7b expression in liver cancer cell lines. Experimental cell research. 2012; 318:1832–1843.
45. Abu-Amero KK, Helwa I, Al-Muammar A, Strickland S, Hauser MA, Allingham RR, Liu Y. Case-control association between CCT-associated variants and keratoconus in a Saudi Arabian population. Journal of negative results in biomedicine. 2015; 14:10.
46. Chen C, Wang HJ, Yang LY, Jia XB, Xu P, Chen J, Liu Y. [Expression of MiR-130a in Serum Samples of Patients with Epithelial Ovarian Cancer and Its Association with Platinum Resistance]. [Article in Chinese]. Sichuan da xue xue bao Yi xue ban. 2016; 47:60–63.
47. Zhuo HC, Song YF, Ye J, Lai GX, Liu DL. MicroRNA-154 functions as a tumor suppressor and directly targets HMGA2 in human non-small cell lung cancer. Genetics and molecular research. 2016; 15.
48. Masoumi-Moghaddam S, Amini A, Wei AQ, Robertson G, Morris DL. Sprouty2 protein in prediction of post-treatment ascites in epithelial ovarian cancer treated with adjuvant carbotaxol chemotherapy. American journal of cancer research. 2015; 5:2498–2507.
49. Gutierrez-Hurtado IA, Puebla-Perez AM, Delgado-Saucedo JI, Figuera LE, Zuniga-Gonzalez GM, Gomez-Mariscal K, Ronquillo-Carreon CA, Gallegos-Arreola MP. Association between TNF-alpha-308G > A and -238G > A gene polymorphisms and TNF-alpha serum levels in Mexican colorectal cancer patients. Genetics and molecular research. 2016; 15.
50. Alves CL, Elias D, Lyng MB, Bak M, Kirkegaard T, Lykkesfeldt AE, Ditzel HJ. High CDK6 protects cells from fulvestrant-mediated apoptosis and is a predictor of resistance to fulvestrant in estrogen receptor-positive metastatic breast cancer. Clinical cancer research. 2016.
51. Kim EK, Yoon SO, Jung WY, Lee H, Kang Y, Jang YJ, Hong SW, Choi SH, Yang WI. Implications of NOVA1 suppression within the microenvironment of gastric cancer: association with immune cell dysregulation. Gastric cancer. 2016.

