Introduction
Heme oxygenases catalyze the degradation of heme into biliverdin, carbon monoxide (CO) and ferric iron [1-5] (Figure 1A). Heme functions as the prosthetic group in hemoproteins, e.g., nitric oxide synthase, cyclooxygenases, soluble guanylate cyclase, cytochrome P450, peroxidase, and catalase and since HO is the sole physiological pathway of heme degradation. It consequently plays a critical role in the regulation of cellular heme-dependent enzyme levels [6-10] (Figure 1B). To date, two HO isoforms have been shown to be catalytically active in heme degradation and each is encoded by a different gene [2, 11]. HO-1 is expressed at low levels under basal conditions and it is induced by polyphenols [12-18], statins [19], metals [20-23] and a variety of stimuli such as inflammation, oxidative stress, hyperoxia, hypoxia and trauma [24-30]. Such upregulation represents an intrinsic defense mechanism to maintain cellular homeostasis and enhance cell survival [31-34]. In particular, HO-1 is considered to play a major role as an essential survival factor, protecting against chemotherapy-induced reactive oxygen species (ROS) increase [27, 35-39]. Most of the studies so far were directed at evaluating the protective effect of these enzymes because of their ability to generate antioxidant and antiapoptotic molecules such as CO and bilirubin [40-47]. In contrast, HO-2 is responsible for the most HO constitutive activity [48-51]. Interestingly, recent evidence suggests that HO may possess other important physiological functions, which are not related to its enzymatic activity and for which we would like to introduce for the first time the term “non canonical functions” (Figure 2). In particular, we discuss three potential signaling mechanisms that we refer to as the non-canonical functions of the HO isoforms: protein-protein interaction, intracellular compartmentalization, and extracellular secretion. The aim of the present review is to describe each of this mechanism and the aspects warranting additional studies in order to unravel all the functions of the HO system.
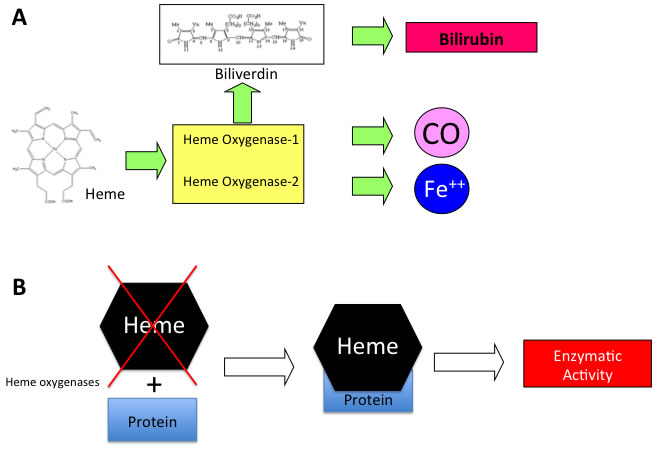
Figure 1: A. Schematic representation of enzymatic reaction catalyzed by HO isoforms and B. mechanism of heme dependent protein regulation.
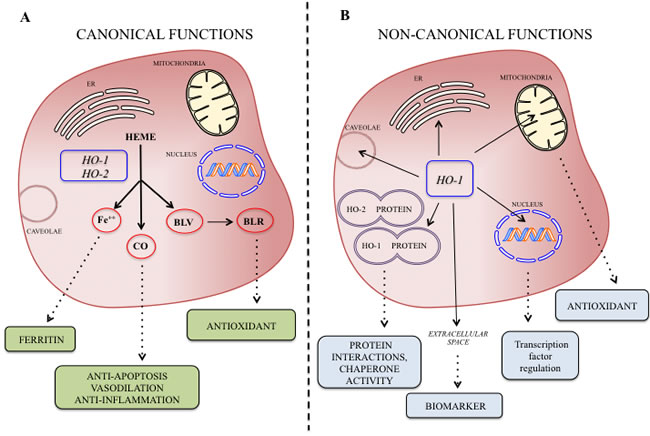
Figure 2: A. Canonical and non-canonical B. functions of the HO isoforms at a glance and possible biological significance of their byproducts.
Protein-Protein Interaction
The first paper reporting the possibility that HO isoforms may form protein-protein interaction was suggested in the elegant description of HO-1 purification where a 68-kDa protein was identified [4]. It is now known that the molecular mass of HO-1 is 32 and HO-2 36 kDa. Therefore, it is conceivable that a complex of the two proteins was initially isolated under partially denaturing conditions. On the basis of this simple observation, Weng Y. et al [52] showed an interaction between HO-2 and HO-2 and demonstrated the effect of the HO-1/HO-2 protein complex on HO activity. The authors elegantly concluded that this interaction serves to limit HO activity in certain tissues where the two co-enzymes co-localize and such negative regulation of HO activity may be important to ensure a cytoprotective range of HO expression. However this intriguing evidence and possible non-canonical functions of HO isoforms remained unexplored for several years until when our research group was able to demonstrate the presence of HO-1 isoform in the extracellular compartment (i.e. human milk) [53]. The presence of the protein in such biological fluid was not particularly surprising given the apocrine nature of the mammary gland. However, since the enzymatic activity was undetectable in human milk, we were wondering on the possible biological significance of such protein in the extracellular compartment. To this regard, basing on the HO-1 amino acidic sequence homologies with Hsp70, we identified CD91 as a possible interactor of HO-1 in the extracellular space. To this regard, PatchDock molecular docking algorithm and FireDock analysis showed that the charged and polar residues observed on HO-1 are Glu63, Tyr78, Glu81, Glu82, His84, Lys86, Glu90, Gln91 and Gln102. The charged and polar residues observed on CD91 in the region are Arg571, Thr576, Thr536, Arg553, Trp556 and Ser565. Hydrogen bond interactions were observed between Glu90 and Gly571, between Glu63 and Thr576, and between Gln102 and Thr536. Furthermore, hydrophobic-hydrophobic interactions were observed between Tyr55, Val59 (HO-1) and Val535 (CD91). Salt bridges were also observed between Lys177 (HO-1) and Glu332 (CD91) outside of this region. Our successive studies also showed the possibility to form protein-protein interaction also for the HO-2 isoform. In particular, this observation derived again from a casual observation of HO-2 -/- animals exhibiting a metabolic syndrome phenotype and reduced circulating levels of adiponectin [54]. Our data showed that pharmacological upregulation of HO-1 rescued such phenotype and restored adiponectin levels. This adipokyne is formed in the endoplasmic reticulum and requires specific chaperone activity in order to maturate and be secreted in the extracellular space in its active form [55]. Given the endoplasmic reticulum localization of HO-2 and the possibility that it may form protein-protein interaction with other proteins we decided to test the hypothesis that HO-2 may serve as a chaperone for physiological secretion of adiponectin from the endoplasmic reticulum. In particular, our in silico analysis showed two hypothetical symmetrical binding regions making contact with two different regions of adiponectin were identified. Multiple structural motifs appear to be involved in both the recognition and binding process between HO-2 and adiponectin. In particular, an extended and structured area of 13 amino acids (a.a. 235-247) in HO-2 seems to interact with a specific sequence of adiponectin (a.a. 238-246). Interestingly one single Arginine residue on the HO was found in the two hypothetical contact regions. These results were further validated in vivo by using the Bacteriomatch two-hybrid system.
Similar results were also obtained for HO-2 isoform by Spencer AL et al. [56] by Fluorescence Resonance Energy Transfer. In particular, the authors showed that this protein may form a protein-protein interaction with cytochrome P450 reductase leading to the formation of a dynamic ensemble of complex(es) that precede formation of the productive electron transfer complex. Finally, Williams SE et al showed that HO-2 is part of the calcium-sensitive potassium (BK) channels complex and enhances channel activity in normoxia [57]. In particular, the authors showed that inhibition of BK channels by hypoxia was dependent on HO-2 expression and was augmented by HO-2 stimulation.
Intracellular compartmenta-lization of heme oxygenases
As above mentioned the HO isoforms were identified and localized in the endoplasmic reticulum where they exert their enzymatic function. However, later studies were devoted at investigating the subcellular compartmentalization of these two isoforms [58]. As far as concerning the HO-2 isoform, no studies so far identified the presence of this enzyme in other cellular compartment rather than the endoplasmic reticulum. On the contrary, HO-1 isoform was found to be compartmentalized (i.e. nuclei, mitochondria, caveolae) intracellularly under various experimental conditions and different cell types. The most studied and fascinating aspects of the non-canonical functions of HO-1 are related to nuclear translocation. Such possibility is substantiated by bioinformatic analysis demonstrating the nuclear import sequence into the amino acidic sequence of HO-1 (Figure 3A-3D). Successive studies demonstrated nuclear translocation under various experimental conditions. In particular, Suttner DM et al. [59] showed that HO-1 migrated into the nuclear compartment following oxygen toxicity in lung cells and such translocation may account for the regulation of cytoprotective pathways. Similarly, we also demonstrated that excitotoxic injury leads to a significant increase of HO-1 protein expression in primary astroglial cell cultures and a concomitant nuclear translocation of this protein [60]. However, at that time we were not able to identify any possible role of nuclear HO-1 and we proposed only that nuclear HO-1 may still possess the ability to bind heme and may therefore serve as a regulator of heme dependent transcription factor [61]. Since then, several other reports evaluated the presence of HO-1 into the nuclear compartment. In particular, Lin Q et al [62] elegantly showed that HO-1 translocate into the nuclear compartment under hypoxic condition and this is associated with increased activation of antioxidant responsive promoter and activation of transcription factors such as AP-1 and NFkB which are also known transcription factors of HO-1 itself [63-65] (Figure 4). These results were also confirmed by the same group in a transgenic animal model of lung injury following hyperoxia [66] in which the authors showed that nuclear translocation overexpression inhibits repair from hyperoxic lung injury by inhibiting DNA repair, which may predispose the lung to later malignant transformation. Consistently with these data, our docking analysis showed that HO-1 might interact with p65 subunit of NFkB (Figure 5). According to simulations p65 interacting surface seems not to involve DNA binding domains suggesting that the inhibitory control may be exerted by allosteric control. Successive reports showed that nuclear localization of HO-1 was associated with cancer stadiation or chemoresistance. As far as concern clinical stadiation of cancer, Gandini NA et al [67] showed that nuclear HO-1 increases with tumor progression in a mouse model of squamous cell carcinoma and in human head and neck squamous cell carcinoma. Interestingly, the same authors showed that no association of HO-1 nuclear localization with glioblastoma patients survival was detected [68] thus suggesting that nuclear translocation of this protein occurs only under certain specific pathological conditions. Consistently with these results Wegiel B et al. [69] also showed that nuclear HO-1 exhibits reduced enzymatic activity and correlates with poorer prognosis in prostate cancer. As far as concern chemoresistance our research group showed that nuclear translocation confers resistance to imatinib in chronic myeloid leukemia cells [70]. Interestingly, we showed that inhibition of nuclear HO-1 translocation by E64d, a cysteine protease inhibitor, restores sensitivity to imatinib, whereas HO byproducts CO or bilirubin had no effects. Similarly, we have recently shown that HO-1 nuclear translocation is also associated to chemoresistance to Bortezomib, a proteasome inhibitor, in various multiple myeloma cell lines [71]. Interestingly, we showed that nuclear translocation of HO-1 was associated to genetic instability thus suggesting that other functions, beside interaction with nuclear transcription factors, could be associated with nuclear HO-1 (Figure 5). Therefore, it becomes of great importance to identify the precise mechanisms underlying HO-1 cleavage allowing its possible nuclear localization in order to overcome chemoresistance. To this regard, the signal peptide peptidase (SPP) catalyzes the intramembrane cleavage of HO-1 allowing nuclear translocation and promoting cancer cell proliferation and invasion independently from its enzymatic activity [72].

Figure 3: A. 3D structure of HO-1 (Structural Biology Knowledgebase: 3TGM, UniProtKB AC: P09601). In red the predicted monopartite Nuclear Localization Signal (NLS) and in yellow the predicted bipartite NLS. Molecular graphics and analyses were performed with the ChimeraX package; B. Amino acid sequence of HO-1 (GenBank: CAG30391.1); C. Prediction of monopartite NLSs specific of HMOX1 (cNLS Mapper tool); D. Prediction of bipartite NLSs of HMOX1 (cNLS Mapper tool).
Other subcellular localizations have been described for HO-1 and may be related to its non-canonical functions. In particular, Converso DP et al. [73] demonstrated for the first time the localization of HO-1 protein in mitochondria suggesting its important biological roles in regulating mitochondrial heme protein turnover and in protecting against conditions such as hypoxia, neurodegenerative diseases, or sepsis, in which substantially increased mitochondrial nitric oxide and oxidant production have been implicated. Similar results were obtained by Slebos DJ et al. [74] showing that HO-1 localized to mitochondria in a primary culture of human small airway epithelial cells following cigarette smoke extract exposure. Interestingly, the authors showed that such translocation was accompanied by a significant increase of the HO mitochondrial activity. These results were confirmed by Bindu S et al. [75] showing that mitochondrial translocation of HO-1 also resulted in time-dependent inhibition of apoptosis during gastric mucosal injury following indomethacin treatment. The mitochondrial significance of HO-1 was further supported by the interesting observations of Bolisetty S et al [76], which were able to target specifically renal epithelial cell mitochondria with HO-1 protein. In these set of experiments the authors showed that specific mitochondrially targeted HO-1 under acute pathological conditions may have beneficial effects, but the selective advantage of long-term expression is constrained by a negative impact on the synthesis of heme-containing mitochondrial proteins. This latter observation was further confirmed by Bansal L et al [77] showing that cells expressing mitochondria targeted HO-1 exhibited higher ROS production leading also to increased autophagy and reduction of cytochrome c oxidase activity. Finally, HO-1 was also demonstrated in caveolae, the small flask-shaped and detergent insoluble invaginations in plasma membrane and are implicated to function in the vesicular transport processes and the transduction of receptor generated signals. To this regard Jung NH et al. [78] showed that HO-1 is localized in caveolae of mouse mesangial cells where it may co-localize with important proteins such as caveolin-1 and caveolin-2. In addition, Wang XM et al showed that such translocation is dependent on p38MAPK and it may regulate the interaction between caveolin-1 and Toll Like Receptor-4 [79].
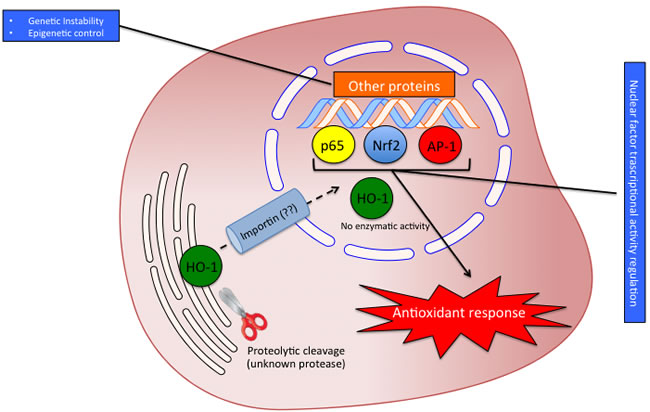
Figure 4: Possible significance of HO-1 nuclear translocation. The proteolytic cleavage allows the translocation of HO-1 into the nucleus probably by interaction with importin. In the nucleus HO-1 loses enzyme activity and regulates the transcriptional factors activity interacting with p65, AP-1 and Nrf2.
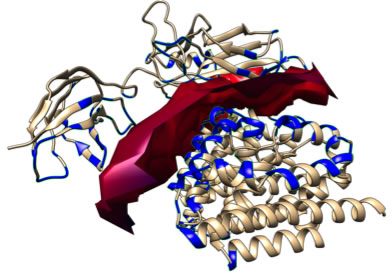
Figure 5: Docking simulation for p65/HO-1 interaction. According to the predicted molecular complex structure, p65 (upper structure) and HO-1 (lower structure) binding is defined by and extended surface of molecular complementarity. Interaction surface is represented by solid red area while residues involved in protein contacts are represented in blue. The local estimated energy of the binding is -31.65 kcal/mol.
The heme oxygenases in the extracellular space
Several reports evaluated so far the presence of HO-1 in extracellular compartments and biological fluids thus suggesting that HO-1 may serve as a possible biomarker of disease [80, 81] or may play additional roles in the extracellular space as a receptor ligand [82]. Interestingly, no reports reported the presence of HO-2 in such compartments thus suggesting that the presence of HO-1 is not the results of cell necrosis and passive release in extracellular compartments but could be related to a still unknown specific mechanism of secretion. In the following sections we will therefore evaluate the significance only of HO-1 in various biological fluids.
HO-1 in plasma and serum
Most of the reports evaluating the presence of HO-1 in extracellular space refer to serum or plasma. In particular, Eide IP et al. [83] showed that serum HO-1 levels were significantly higher among pre-eclampsia patients compared to controls supporting the role of oxidative stress and excessive maternal inflammatory response in the pathogenesis of pre-eclampsia. Other clinical studies reported the increase of serum HO-1 under other pathological conditions such as Alzheimer disease. In particular, Mueller C et al [84] showed that serum HO-1 among other proteins is increased in Alzheimer’s disease and such levels correlated to cognition impairment grade. Similarly plasma HO-1 is increased in patients resuscitated from out-of-hospital cardiac arrest [85] or suffering from peripheral artery disease [80]. These results were further confirmed in an animal model of lung injury induced by Ischemia/reperfusion [86] in which the authors showed increased serum HO-1 during the 3h observation period.
As far as concern the possible release mechanism(s) of HO-1 in serum or plasma, no data are so far available. However, it is possible that HO-1 requires proteolytic cleavage in order to be secreted. To this regard, Zager RA et al. [87] showed that plasma HO-1 is increased in patients suffering acute kidney injury and interestingly the western blot showed a 16Kda band rather than the canonical 32Kda of the full-length protein. Taken all together, these data suggest that HO-1 is increased in plasma or serum in those all conditions in which oxidative stress is increased. Therefore, even though the release of HO-1 in the extracellular space may be of some biological importance, its possible use as a potential biomarker for a particular disease could be limited by its specificity. Furthermore, the evidence that liraglutide treatment resulted in a significant reduction of plasma HO-1 levels in type-2 diabetes mellitus patients supports the idea that extracellular HO-1 should be considered as an active secretory mechanism. Furthermore, the hypothesis that plasma HO-1 is the results of an active mechanism of secretion and not the consequence of cell necrosis is supported by previous work showing that HO-1 is increased in patients with acute myocardial infarction independently of cell necrosis biomarkers (i.e troponin and creatine phosphokinase) [25].
HO-1 in cerebrospinal fluid
Cerebrospinal fluid is often used for the evaluating central nervous system specific markers and as a diagnostic routine (i.e. multiple sclerosis) [88]. To this regard, the biological significance of HO-1 in such biological fluid could be of particular interest. To this regard, Schipper HM et al [89] showed that HO-1 protein is decreased of cerebrospinal fluid of Alzheimer’s disease patients. On the contrary, HO-1 is increased in cerebrospinal fluid from infants and children after severe traumatic injury [90-92]. Similarly, a successive study also showed that HO-1 protein is increased in cerebrospinal fluid in patients with Fisher Grade III aneurysmal subarachnoid hemorrhage and this may serve also as an effective outcome indicator in patients with Fisher Grade III aneurysmal subarachnoid hemorrhage [93].
Conclusions and future perspectives
Taken all together, the above-mentioned studies suggest that the HO system may possess important biological functions beyond its enzymatic activity. In the present review, we reported what we called the non-canonical function of the heme oxygenases. Three different effects should be included in such class of functions: protein-protein interaction, subcellular compartmentalization and secretion into the extracellular space. However, several issues are still open and warrant future studies. In particular the most important questions needing to be addressed are related to the mechanism(s) underlying proteolytic cleavage of HO-1 allowing the protein to be mobilized from the endoplasmic reticulum. Another important aspect requiring further investigation regards the extracellular release of the protein. As previously discussed, it was reported that the plasma HO-1 is cleaved to a 16 KDa fragment. However, no mechanisms are reported and even hypothesized regarding on how HO-1 is exported in the extracellular compartment. In particular, future studies should be directed in evaluating the presence of HO-1 in exosomes or cellular microvescicles (Figure 6).
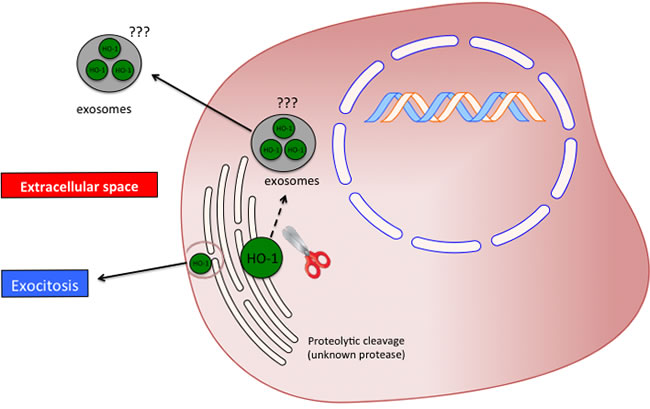
Figure 6: Possible significance and release mechanism of HO-1 in the extracellular space.
Conflicts of Interest
All the Authors declare to have no conflict of interest.
Funding
This work was supported by a grant from the University of Catania (FIR 2013-2015) (Catania, Italy) and from “Associazione Mani Amiche” Onlus, Catania (Italy).
References
1. Eisenstein RS, Garcia-Mayol D, Pettingell W and Munro HN. Regulation of ferritin and heme oxygenase synthesis in rat fibroblasts by different forms of iron. Proceedings of the National Academy of Sciences of the United States of America. 1991; 88:688-692.
2. Shibahara S, Yoshizawa M, Suzuki H, Takeda K, Meguro K and Endo K. Functional analysis of cDNAs for two types of human heme oxygenase and evidence for their separate regulation. J Biochem. 1993; 113:214-218.
3. Tenhunen R, Marver HS and Schmid R. The enzymatic conversion of heme to bilirubin by microsomal heme oxygenase. Proceedings of the National Academy of Sciences of the United States of America. 1968; 61:748-755.
4. Maines MD, Ibrahim NG and Kappas A. Solubilization and partial purification of heme oxygenase from rat liver. The Journal of biological chemistry. 1977; 252:5900-5903.
5. Shibahara S. The heme oxygenase dilemma in cellular homeostasis: new insights for the feedback regulation of heme catabolism. Tohoku J Exp Med. 2003; 200:167-186.
6. Li Volti G, Sorrenti V, Murabito P, Galvano F, Veroux M, Gullo A, Acquaviva R, Stacchiotti A, Bonomini F, Vanella L and Di Giacomo C. Pharmacological induction of heme oxygenase-1 inhibits iNOS and oxidative stress in renal ischemia-reperfusion injury. Transplant Proc. 2007; 39:2986-2991.
7. Li Volti G, Seta F, Schwartzman ML, Nasjletti A and Abraham NG. Heme oxygenase attenuates angiotensin II-mediated increase in cyclooxygenase-2 activity in human femoral endothelial cells. Hypertension. 2003; 41:715-719.
8. Abraham NG, Quan S, Mieyal PA, Yang L, Burke-Wolin T, Mingone CJ, Goodman AI, Nasjletti A and Wolin MS. Modulation of cGMP by human HO-1 retrovirus gene transfer in pulmonary microvessel endothelial cells. American journal of physiology Lung cellular and molecular physiology. 2002; 283:L1117-1124.
9. Sacerdoti D, Escalante B, Abraham NG, McGiff JC, Levere RD and Schwartzman ML. Treatment with tin prevents the development of hypertension in spontaneously hypertensive rats. Science. 1989; 243:388-390.
10. Turkseven S, Kruger A, Mingone CJ, Kaminski P, Inaba M, Rodella LF, Ikehara S, Wolin MS and Abraham NG. Antioxidant mechanism of heme oxygenase-1 involves an increase in superoxide dismutase and catalase in experimental diabetes. Am J Physiol Heart Circ Physiol. 2005; 289:H701-707.
11. McCoubrey WK, Jr., Ewing JF and Maines MD. Human heme oxygenase-2: characterization and expression of a full-length cDNA and evidence suggesting that the two HO-2 transcripts may differ by choice of polyadenylation signal. Arch Biochem Biophys. 1992; 295:13-20.
12. Barbagallo I, Galvano F, Frigiola A, Cappello F, Riccioni G, Murabito P, D’Orazio N, Torella M, Gazzolo D and Li Volti G. Potential therapeutic effects of natural heme oxygenase-1 inducers in cardiovascular diseases. Antioxidants & redox signaling. 2013; 18:507-521.
13. Li Volti G, Sacerdoti D, Di Giacomo C, Barcellona ML, Scacco A, Murabito P, Biondi A, Basile F, Gazzolo D, Abella R, Frigiola A and Galvano F. Natural heme oxygenase-1 inducers in hepatobiliary function. World journal of gastroenterology. 2008; 14:6122-6132.
14. Acquaviva R, Campisi A, Raciti G, Avola R, Barcellona ML, Vanella L and Li Volti G. Propofol inhibits caspase-3 in astroglial cells: role of heme oxygenase-1. Curr Neurovasc Res. 2005; 2:141-148.
15. Marrazzo G, Bosco P, La Delia F, Scapagnini G, Di Giacomo C, Malaguarnera M, Galvano F, Nicolosi A and Li Volti G. Neuroprotective effect of silibinin in diabetic mice. Neurosci Lett. 2011; 504:252-256.
16. Vanella L, Tibullo D, Godos J, Pluchinotta FR, Di Giacomo C, Sorrenti V, Acquaviva R, Russo A, Li Volti G and Barbagallo I. Caffeic Acid Phenethyl Ester Regulates PPAR’s Levels in Stem Cells-Derived Adipocytes. PPAR Res. 2016; 2016:7359521.
17. Li Volti G, Musumeci T, Pignatello R, Murabito P, Barbagallo I, Carbone C, Gullo A and Puglisi G. Antioxidant potential of different melatonin-loaded nanomedicines in an experimental model of sepsis. Exp Biol Med (Maywood). 2012; 237:670-677.
18. Dore S. Unique properties of polyphenol stilbenes in the brain: more than direct antioxidant actions; gene/protein regulatory activity. Neurosignals. 2005; 14:61-70.
19. Jang HJ, Hong EM, Kim M, Kim JH, Jang J, Park SW, Byun HW, Koh DH, Choi MH, Kae SH and Lee J. Simvastatin induces heme oxygenase-1 via NF-E2-related factor 2 (Nrf2) activation through ERK and PI3K/Akt pathway in colon cancer. Oncotarget. 2016; doi: 10.18632/oncotarget.10078.
20. Li Volti G, Zappala A, Leggio GM, Mazzola C, Drago F, La Delia F, Serapide MF, Pellitteri R, Giannone I, Spatuzza M, Cicirata V and Cicirata F. Tin chloride enhances parvalbumin-positive interneuron survival by modulating heme metabolism in a model of cerebral ischemia. Neurosci Lett. 2011; 492:33-38.
21. Maines MD and Kappas A. Metals as regulators of heme metabolism. Science. 1977; 198:1215-1221.
22. Sunderman FW, Jr. Metal induction of heme oxygenase. Annals of the New York Academy of Sciences. 1987; 514:65-80.
23. Alam J, Cai J and Smith A. Isolation and characterization of the mouse heme oxygenase-1 gene. Distal 5’ sequences are required for induction by heme or heavy metals. The Journal of biological chemistry. 1994; 269:1001-1009.
24. Kushida T, LiVolti G, Goodman AI and Abraham NG. TNF-alpha-mediated cell death is attenuated by retrovirus delivery of human heme oxygenase-1 gene into human microvessel endothelial cells. Transplant Proc. 2002; 34:2973-2978.
25. Novo G, Cappello F, Rizzo M, Fazio G, Zambuto S, Tortorici E, Gammazza AM, Corrao S, Zummo G, De Macario EC, Macario AJ, Assennato P, Novo S and Li Volti G. Hsp60 and heme oxygenase-1 (Hsp32) in acute myocardial infarction. Translational research. 2011; 157:285-292.
26. Sacerdoti D, Colombrita C, Ghattas MH, Ismaeil EF, Scapagnini G, Bolognesi M, Li Volti G and Abraham NG. Heme oxygenase-1 transduction in endothelial cells causes downregulation of monocyte chemoattractant protein-1 and of genes involved in inflammation and growth. Cell Mol Biol (Noisy-le-grand). 2005; 51:363-370.
27. Salerno L, Pittala V, Romeo G, Modica MN, Siracusa MA, Di Giacomo C, Acquaviva R, Barbagallo I, Tibullo D and Sorrenti V. Evaluation of novel aryloxyalkyl derivatives of imidazole and 1,2,4-triazole as heme oxygenase-1 (HO-1) inhibitors and their antitumor properties. Bioorg Med Chem. 2013; 21:5145-5153.
28. Barbagallo I, Nicolosi A, Calabrese G, David S, Cimino S, Madonia M and Cappello F. The role of the heme oxygenase system in the metabolic syndrome. Curr Pharm Des. 2014; 20:4970-4974.
29. Sacerdoti D, Olszanecki R, Li Volti G, Colombrita C, Scapagnini G and Abraham NG. Heme oxygenase overexpression attenuates glucose-mediated oxidative stress in quiescent cell phase: linking heme to hyperglycemia complications. Curr Neurovasc Res. 2005; 2:103-111.
30. Otterbein L, Sylvester SL and Choi AM. Hemoglobin provides protection against lethal endotoxemia in rats: the role of heme oxygenase-1. American journal of respiratory cell and molecular biology. 1995; 13:595-601.
31. Ryter SW and Choi AM. Heme oxygenase-1/carbon monoxide: from metabolism to molecular therapy. American journal of respiratory cell and molecular biology. 2009; 41:251-260.
32. Soares MP and Bach FH. Heme oxygenase-1: from biology to therapeutic potential. Trends Mol Med. 2009; 15:50-58.
33. Bach FH. Heme oxygenase-1: a therapeutic amplification funnel. FASEB J. 2005; 19:1216-1219.
34. Seo GS, Jiang WY, Chi JH, Jin H, Park WC, Sohn DH, Park PH and Lee SH. Heme oxygenase-1 promotes tumor progression and metastasis of colorectal carcinoma cells by inhibiting antitumor immunity. Oncotarget. 2015; 6:19792-19806. doi: 10.18632/oncotarget.4075.
35. Goswami B, Rajappa M, Sharma M and Sharma A. Inflammation: its role and interplay in the development of cancer, with special focus on gynecological malignancies. Int J Gynecol Cancer. 2008; 18:591-599.
36. Abe M. Guest editorial: understanding the pathogenesis and the evolving treatment paradigm for multiple myeloma in the era of novel agents. Int J Hematol. 2011; 94:307-309.
37. Meister S, Schubert U, Neubert K, Herrmann K, Burger R, Gramatzki M, Hahn S, Schreiber S, Wilhelm S, Herrmann M, Jack HM and Voll RE. Extensive immunoglobulin production sensitizes myeloma cells for proteasome inhibition. Cancer Res. 2007; 67:1783-1792.
38. Teicher BA, Ara G, Herbst R, Palombella VJ and Adams J. The proteasome inhibitor PS-341 in cancer therapy. Clin Cancer Res. 1999; 5:2638-2645.
39. Richardson PG, Baz R, Wang M, Jakubowiak AJ, Laubach JP, Harvey RD, Talpaz M, Berg D, Liu G, Yu J, Gupta N, Di Bacco A, Hui AM and Lonial S. Phase 1 study of twice-weekly ixazomib, an oral proteasome inhibitor, in relapsed/refractory multiple myeloma patients. Blood. 2014; 124:1038-1046.
40. Barbagallo I, Marrazzo G, Frigiola A, Zappala A and Li Volti G. Role of carbon monoxide in vascular diseases. Curr Pharm Biotechnol. 2012; 13:787-796.
41. Salomone F, Li Volti G, Rosso C, Grosso G and Bugianesi E. Unconjugated bilirubin, a potent endogenous antioxidant, is decreased in patients with non-alcoholic steatohepatitis and advanced fibrosis. J Gastroenterol Hepatol. 2013; 28:1202-1208.
42. Stocker R, Yamamoto Y, McDonagh AF, Glazer AN and Ames BN. Bilirubin is an antioxidant of possible physiological importance. Science. 1987; 235:1043-1046.
43. Li Volti G, Sacerdoti D, Sangras B, Vanella A, Mezentsev A, Scapagnini G, Falck JR and Abraham NG. Carbon monoxide signaling in promoting angiogenesis in human microvessel endothelial cells. Antioxidants & redox signaling. 2005; 7:704-710.
44. Bach FH. Carbon monoxide: from the origin of life to molecular medicine. Trends Mol Med. 2006; 12:348-350.
45. Motterlini R and Otterbein LE. The therapeutic potential of carbon monoxide. Nat Rev Drug Discov. 2010; 9:728-743.
46. Ryter SW and Otterbein LE. Carbon monoxide in biology and medicine. Bioessays. 2004; 26:270-280.
47. Nemeth Z, Csizmadia E, Vikstrom L, Li M, Bisht K, Feizi A, Otterbein S, Zuckerbraun B, Costa DB, Pandolfi PP, Fillinger J, Dome B, Otterbein LE and Wegiel B. Alterations of tumor microenvironment by carbon monoxide impedes lung cancer growth. Oncotarget. 2016; 7:23919-32. doi: 10.18632/oncotarget.8081.
48. Munoz-Sanchez J and Chanez-Cardenas ME. A review on hemeoxygenase-2: focus on cellular protection and oxygen response. Oxid Med Cell Longev. 2014; 2014:604981.
49. Parfenova H and Leffler CW. Cerebroprotective functions of HO-2. Curr Pharm Des. 2008; 14:443-453.
50. Kemp PJ. Hemeoxygenase-2 as an O2 sensor in K+ channel-dependent chemotransduction. Biochem Biophys Res Commun. 2005; 338:648-652.
51. Marrazzo G, Bellner L, Halilovic A, Li Volti G, Drago F, Dunn MW and Schwartzman ML. The role of neutrophils in corneal wound healing in HO-2 null mice. PloS one. 2011; 6:e21180.
52. Weng YH, Yang G, Weiss S and Dennery PA. Interaction between heme oxygenase-1 and -2 proteins. The Journal of biological chemistry. 2003; 278:50999-51005.
53. Li Volti G, Galvano F, Frigiola A, Guccione S, Di Giacomo C, Forte S, Tringali G, Caruso M, Adekoya OA and Gazzolo D. Potential immunoregulatory role of heme oxygenase-1 in human milk: a combined biochemical and molecular modeling approach. J Nutr Biochem. 2010; 21:865-871.
54. Vanella L, Li Volti G, Guccione S, Rappazzo G, Salvo E, Pappalardo M, Forte S, Schwartzman ML and Abraham NG. Heme oxygenase-2/adiponectin protein-protein interaction in metabolic syndrome. Biochem Biophys Res Commun. 2013; 432:606-611.
55. Wang ZV, Schraw TD, Kim JY, Khan T, Rajala MW, Follenzi A and Scherer PE. Secretion of the adipocyte-specific secretory protein adiponectin critically depends on thiol-mediated protein retention. Mol Cell Biol. 2007; 27:3716-3731.
56. Spencer AL, Bagai I, Becker DF, Zuiderweg ER and Ragsdale SW. Protein/protein interactions in the mammalian heme degradation pathway: heme oxygenase-2, cytochrome P450 reductase, and biliverdin reductase. The Journal of biological chemistry. 2014; 289:29836-29858.
57. Williams SE, Wootton P, Mason HS, Bould J, Iles DE, Riccardi D, Peers C and Kemp PJ. Hemoxygenase-2 is an oxygen sensor for a calcium-sensitive potassium channel. Science. 2004; 306:2093-2097.
58. Dunn LL, Midwinter RG, Ni J, Hamid HA, Parish CR and Stocker R. New insights into intracellular locations and functions of heme oxygenase-1. Antioxidants & redox signaling. 2014; 20:1723-1742.
59. Suttner DM, Sridhar K, Lee CS, Tomura T, Hansen TN and Dennery PA. Protective effects of transient HO-1 overexpression on susceptibility to oxygen toxicity in lung cells. Am J Physiol. 1999; 276:L443-451.
60. Li Volti G, Ientile R, Abraham NG, Vanella A, Cannavo G, Mazza F, Curro M, Raciti G, Avola R and Campisi A. Immunocytochemical localization and expression of heme oxygenase-1 in primary astroglial cell cultures during differentiation: effect of glutamate. Biochem Biophys Res Commun. 2004; 315:517-524.
61. Hon T, Hach A, Tamalis D, Zhu Y and Zhang L. The yeast heme-responsive transcriptional activator Hap1 is a preexisting dimer in the absence of heme. The Journal of biological chemistry. 1999; 274:22770-22774.
62. Lin Q, Weis S, Yang G, Weng YH, Helston R, Rish K, Smith A, Bordner J, Polte T, Gaunitz F and Dennery PA. Heme oxygenase-1 protein localizes to the nucleus and activates transcription factors important in oxidative stress. The Journal of biological chemistry. 2007; 282:20621-20633.
63. Lavrovsky Y, Schwartzman ML and Abraham NG. Novel regulatory sites of the human heme oxygenase-1 promoter region. Biochem Biophys Res Commun. 1993; 196:336-341.
64. Lavrovsky Y, Schwartzman ML, Levere RD, Kappas A and Abraham NG. Identification of binding sites for transcription factors NF-kappa B and AP-2 in the promoter region of the human heme oxygenase 1 gene. Proceedings of the National Academy of Sciences of the United States of America. 1994; 91:5987-5991.
65. Alam J and Den Z. Distal AP-1 binding sites mediate basal level enhancement and TPA induction of the mouse heme oxygenase-1 gene. The Journal of biological chemistry. 1992; 267:21894-21900.
66. Namba F, Go H, Murphy JA, La P, Yang G, Sengupta S, Fernando AP, Yohannes M, Biswas C, Wehrli SL and Dennery PA. Expression level and subcellular localization of heme oxygenase-1 modulates its cytoprotective properties in response to lung injury: a mouse model. PloS one. 2014; 9:e90936.
67. Gandini NA, Fermento ME, Salomon DG, Blasco J, Patel V, Gutkind JS, Molinolo AA, Facchinetti MM and Curino AC. Nuclear localization of heme oxygenase-1 is associated with tumor progression of head and neck squamous cell carcinomas. Exp Mol Pathol. 2012; 93:237-245.
68. Gandini NA, Fermento ME, Salomon DG, Obiol DJ, Andres NC, Zenklusen JC, Arevalo J, Blasco J, Lopez Romero A, Facchinetti MM and Curino AC. Heme oxygenase-1 expression in human gliomas and its correlation with poor prognosis in patients with astrocytoma. Tumour Biol. 2013.
69. Wegiel B, Gallo D, Csizmadia E, Harris C, Belcher J, Vercellotti GM, Penacho N, Seth P, Sukhatme V, Ahmed A, Pandolfi PP, Helczynski L, Bjartell A, Persson JL and Otterbein LE. Carbon monoxide expedites metabolic exhaustion to inhibit tumor growth. Cancer Res. 2013; 73:7009-7021.
70. Tibullo D, Barbagallo I, Giallongo C, La Cava P, Parrinello N, Vanella L, Stagno F, Palumbo GA, Li Volti G and Di Raimondo F. Nuclear translocation of heme oxygenase-1 confers resistance to imatinib in chronic myeloid leukemia cells. Curr Pharm Des. 2013; 19:2765-2770.
71. Tibullo D, Barbagallo I, Giallongo C, Vanella L, Conticello C, Romano A, Saccone S, Godos J, Di Raimondo F and Li Volti G. Heme oxygenase-1 nuclear translocation regulates bortezomibinduced cytotoxicity and mediates genomic instability in myeloma cells. Oncotarget. 2016; 7:28868-80. doi: 10.18632/oncotarget.7563.
72. Hsu FF, Yeh CT, Sun YJ, Chiang MT, Lan WM, Li FA, Lee WH and Chau LY. Signal peptide peptidase-mediated nuclear localization of heme oxygenase-1 promotes cancer cell proliferation and invasion independent of its enzymatic activity. Oncogene. 2015; 34:2360-2370.
73. Converso DP, Taille C, Carreras MC, Jaitovich A, Poderoso JJ and Boczkowski J. HO-1 is located in liver mitochondria and modulates mitochondrial heme content and metabolism. FASEB J. 2006; 20:1236-1238.
74. Slebos DJ, Ryter SW, van der Toorn M, Liu F, Guo F, Baty CJ, Karlsson JM, Watkins SC, Kim HP, Wang X, Lee JS, Postma DS, Kauffman HF and Choi AM. Mitochondrial localization and function of heme oxygenase-1 in cigarette smoke-induced cell death. American journal of respiratory cell and molecular biology. 2007; 36:409-417.
75. Bindu S, Pal C, Dey S, Goyal M, Alam A, Iqbal MS, Dutta S, Sarkar S, Kumar R, Maity P and Bandyopadhyay U. Translocation of heme oxygenase-1 to mitochondria is a novel cytoprotective mechanism against non-steroidal anti-inflammatory drug-induced mitochondrial oxidative stress, apoptosis, and gastric mucosal injury. The Journal of biological chemistry. 2011; 286:39387-39402.
76. Bolisetty S, Traylor A, Zarjou A, Johnson MS, Benavides GA, Ricart K, Boddu R, Moore RD, Landar A, Barnes S, Darley-Usmar V and Agarwal A. Mitochondria-targeted heme oxygenase-1 decreases oxidative stress in renal epithelial cells. Am J Physiol Renal Physiol. 2013; 305:F255-264.
77. Bansal S, Biswas G and Avadhani NG. Mitochondria-targeted heme oxygenase-1 induces oxidative stress and mitochondrial dysfunction in macrophages, kidney fibroblasts and in chronic alcohol hepatotoxicity. Redox Biol. 2014; 2:273-283.
78. Jung NH, Kim HP, Kim BR, Cha SH, Kim GA, Ha H, Na YE and Cha YN. Evidence for heme oxygenase-1 association with caveolin-1 and -2 in mouse mesangial cells. IUBMB Life. 2003; 55:525-532.
79. Wang XM, Kim HP, Nakahira K, Ryter SW and Choi AM. The heme oxygenase-1/carbon monoxide pathway suppresses TLR4 signaling by regulating the interaction of TLR4 with caveolin-1. Journal of immunology. 2009; 182:3809-3818.
80. Signorelli SS, Li Volsi G, Pitruzzella A, Fiore V, Mangiafico M, Vanella L, Parenti R, Rizzo M and Li Volti G. Circulating miR-130a, miR-27b, and miR-210 in Patients With Peripheral Artery Disease and Their Potential Relationship With Oxidative Stress: A Pilot Study. Angiology. 2016.
81. Serpero LD, Bellissima V, Colivicchi M, Sabatini M, Frigiola A, Ricotti A, Ghiglione V, Strozzi MC, Li Volti G, Galvano F and Gazzolo D. Next generation biomarkers for brain injury. The journal of maternal-fetal & neonatal medicine. 2013; 26 Suppl 2:44-49.
82. Serpero LD, Frigiola A and Gazzolo D. Human milk and formulae: neurotrophic and new biological factors. Early Hum Dev. 2012; 88 Suppl 1:S9-12.
83. Eide IP, Isaksen CV, Salvesen KA, Langaas M, Schonberg SA and Austgulen R. Decidual expression and maternal serum levels of heme oxygenase 1 are increased in pre-eclampsia. Acta Obstet Gynecol Scand. 2008; 87:272-279.
84. Mueller C, Zhou W, Vanmeter A, Heiby M, Magaki S, Ross MM, Espina V, Schrag M, Dickson C, Liotta LA and Kirsch WM. The heme degradation pathway is a promising serum biomarker source for the early detection of Alzheimer’s disease. J Alzheimers Dis. 2010; 19:1081-1091.
85. Siren J, Vaahersalo J, Skrifvars M, Pettila V, Tiainen M, Tikkanen I, Lakkisto P and Group FS. Plasma Heme Oxygenase-1 in Patients Resuscitated from out-of-Hospital Cardiac Arrest. Shock. 2016; 45:320-325.
86. Park YY. Ischemia/reperfusion Lung Injury Increases Serum Ferritin and Heme Oxygenase-1 in Rats. Korean J Physiol Pharmacol. 2009; 13:181-187.
87. Zager RA, Johnson AC and Becker K. Plasma and urinary heme oxygenase-1 in AKI. Journal of the American Society of Nephrology. 2012; 23:1048-1057.
88. Smolinska A, Blanchet L, Buydens LM and Wijmenga SS. NMR and pattern recognition methods in metabolomics: from data acquisition to biomarker discovery: a review. Anal Chim Acta. 2012; 750:82-97.
89. Schipper HM, Chertkow H, Mehindate K, Frankel D, Melmed C and Bergman H. Evaluation of heme oxygenase-1 as a systemic biological marker of sporadic AD. Neurology. 2000; 54:1297-1304.
90. Cousar JL, Lai Y, Marco CD, Bayir H, Adelson PD, Janesko-Feldman KL, Kochanek PM and Clark RS. Heme oxygenase 1 in cerebrospinal fluid from infants and children after severe traumatic brain injury. Dev Neurosci. 2006; 28:342-347.
91. Murabito P, Giarratano A, Astuto M, Raineri M and Li Volti G. The role of heme oxygenase and Hsp70 in trauma. EuroMediterranean Biomedical Journal. 2015; 10:120-125.
92. Aronowski J and Zhao X. Molecular pathophysiology of cerebral hemorrhage: secondary brain injury. Stroke. 2011; 42:1781-1786.
93. Wang KC, Tang SC, Lee JE, Lai DM, Huang SJ, Hsieh ST, Jeng JS and Tu YK. Prognostic value of intrathecal heme oxygenase-1 concentration in patients with Fisher Grade III aneurysmal subarachnoid hemorrhage. J Neurosurg. 2014; 121:1388-1393.

