The cover for issue 7 of Oncotarget features Figure 4E, "UXT-KO in prostate cancer cells increased DNA damage accumulation and sensitized cells to cisplatin," by Wang, et al.
Here we analyzed a tissue microarray consisting of normal prostate, benign prostatic hyperplasia, high grade prostatic intraepithelial neoplasia and primary prostate cancer cases for UXT protein expression.
Loss of UXT expression in primary prostate cancer is positively associated with high Gleason grade and poor relapse-free survival.
UXT depletion in prostate cancer cells also increased retroelements expression, including LINE-1 and Alu.
"The androgen receptor pathway plays a vital role in prostate cancer growth."
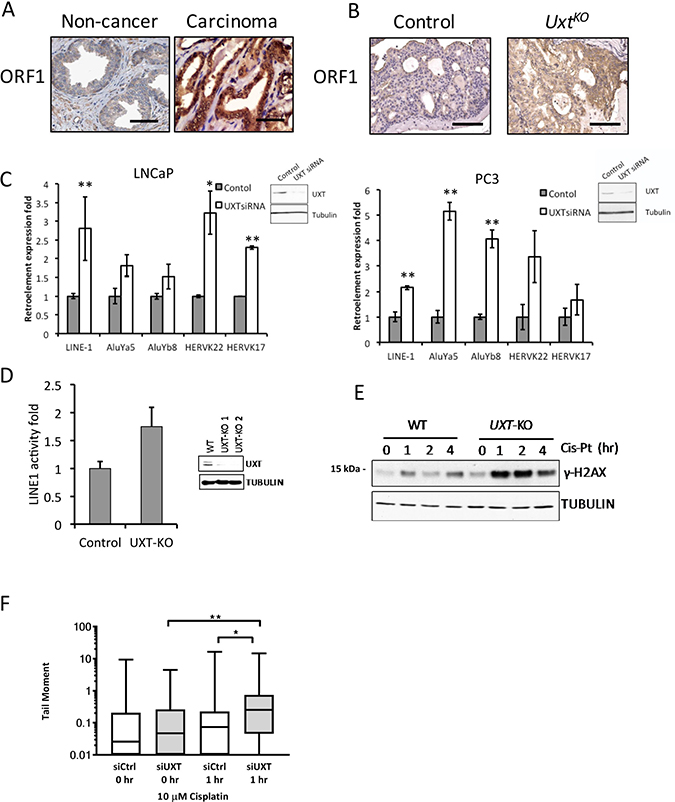
Figure 4: Loss of UXT in prostate promoted LINE-1 retroelement expression and retrotransposition. (A) IHC analysis shows ORF1 staining in human normal prostate (n = 5) and prostate cancer (n = 5). (B) IHC analysis shows ORF1 expression in control and UxtKO mice. Scale bar = 100 μm (C) siRNA-mediated UXT knockdown in human prostate cancer cells induced mRNA expression of Alu, HERVK and LINE-1 retroelements. RNA was harvested from control and siUXT LNCaP and PC3 cells. Retroelement expression was quantified by qRT-PCR. Inset panels show loss of UXT protein in UXT-KO cells. *p < 0.05, **p < 0.01 (D) Retrotransposition assays show that TALEN-mediated UXT-KO induced retrotransposition in C4-2 cells (p = 9 × 10-4). Inset shows dramatically decreased UXT protein expression levels. (E) UXT-KO in prostate cancer cells increased DNA damage accumulation and sensitized cells to cisplatin. Control and UXT-KO C4-2 cells were treated with cisplatin (10 μM) before cell lysates were harvested for western blot. Unrepaired DNA double strand breaks were measured by phospho-γH2AX by western blots. (F) Loss of UXT increases the tail moment in response to cisplatin. C4-2 control (siCtrl) and UXT knockdown (siUXT) cells were treated with cisplatin (10 μM) for 1 hour. The median (line), 25th-75th percentiles (boxes), and minimum and maximum values (whiskers) are shown; *p = 0.0106 and **p = 0.0054.
However, prostate cancer patients receiving androgen deprivation therapy often relapse and develop castration-resistant prostate cancer, which still relies on the AR pathway for growth.
Our previous work identified UXT as a transcription factor that directly binds to the AR N-terminus and modulates AR transcriptional activity in prostate cancer cells.
Depletion of UXT activates DNA damage checkpoint genes, promotes proliferation, and induces cell resistance to anti-androgen treatment in prostate cancer cells.
Depletion of UXT also decreases the protein expression of unconventional prefoldin RBP5 interactor in prostate cells; we have shown that UXT and URI affect each others stability, and in complex together, UXT/URI represses AR mediated transcription and AR occupancy on AR target genes.
We then used prostate-specific Uxt KO transgenic mice and prostate cancer cell culture models to investigate UXT function in Ca P development and progression.
"Interestingly, the prostate hyperplasia in these two types of genetically altered mice has different morphologies with papillary hyperplasia more prevalent in the Pten deletion and cribriform hyperplasia more prevalent in the Uxt deletion.
In addition, the Uxt deletion mice exhibited moderate levels of prostate secretion fluid blockage, indicating that the enlargement of prostate resulted in gland obstruction and disfiguration of the organ.
Second, the accumulation of prostate secretion fluid in the anterior prostate and inflation in these glands may be a direct result of the dorsal/ventral obstruction, leading to a further increase in prostate size.
Altogether our results suggest that loss of UXT in human prostate may promote genome instability in normal prostate epithelial cells and that loss of UXT as an early event in prostate cancer may lead to chromosomal alterations."
Full text - https://doi.org/10.18632/oncotarget.26573
Correspondence to - Susan K. Logan - [email protected]
Keywords - UXT, retroelement, prostate cancer



