Oncotarget: MET gene analysis by next-generation sequencing & fluorescence
2021-10-29
Oncotarget published "Comparison of MET gene amplification analysis by next-generation sequencing and fluorescence in situ hybridization" which reported that MET amplifications present a potential therapeutic target in non-small cell lung cancer. Although next-generation sequencing and fluorescence in situ hybridization are conventionally used to assess MET amplifications, there are currently no clinically defined cut-off values for NGS, with FISH still being the gold standard.
NGS allows for DNA- and RNA-based analysis of specific MET gene amplifications, point mutations or rearrangements.
NGS allows for DNA- and RNA-based analysis of specific MET gene amplifications, point mutations or rearrangements.
Dr. Christina Schmitt from The University Hospital Frankfurt said, "The proto-oncogene MET encodes for a receptor tyrosine kinase which is a ubiquitously expressed cell surface receptor consisting of an extracellular alpha-chain disulfide-bonded to a membrane spanning beta-chain."
The binding of the receptor to its ligand, the extracellular hepatocyte growth factor/scatter factor, induces dimerization of the receptor and triggers a conformational change. As a consequence, the activity of MET tyrosine kinase is activated and can lead to effects on cell growth, mortality, survival, invasion and angiogenesis. Dysregulations in the signaling pathway were first investigated in 1990 as a possible cause of lung cancer. In one study, resistance to EGFR inhibitors was found in 21% of lung cancer patients. Each of the affected patients displayed a MET amplification.
For these reasons, MET is considered a potentially targetable oncogenic driver. The tyrosine kinase inhibitors for MET amplifications are currently in the clinical stage of approval and show promising therapeutic results. The detection of MET amplification can be performed by several techniques such as polymerase chain reaction, southern blot, immunohistochemistry and fluorescence in situ hybridization . It is very important to reliably detect MET changes. The standard analytical method for the detection of MET amplifications is FISH.
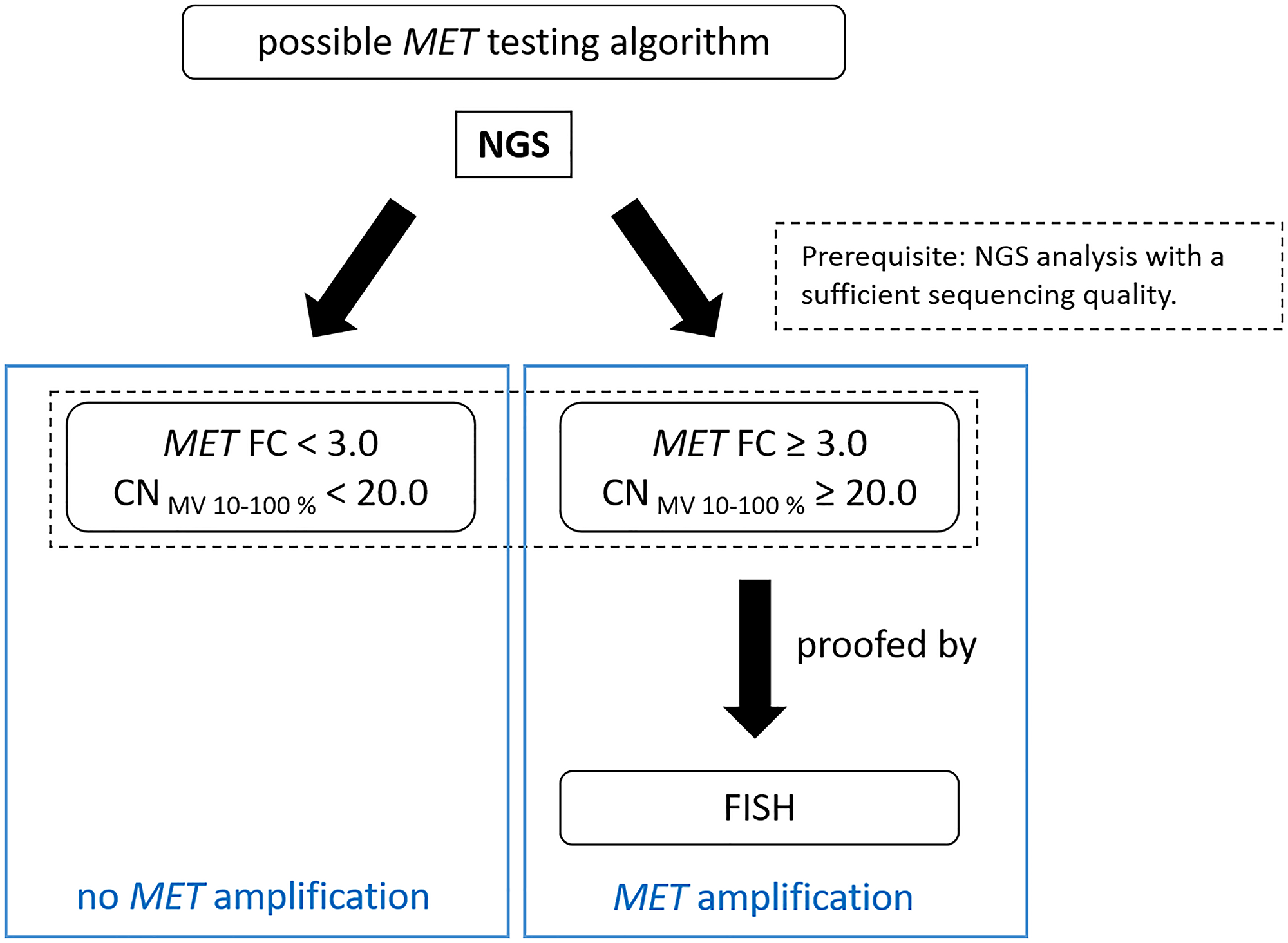
Figure 3: Suggested testing workflow for the detection of MET amplifications by QIAGEN workflow.
The Schmitt Research Team concluded in their Oncotarget Research Output, "In the next step we are going to increase the cohort size, including cases with negative, low-level, intermediate-level and high-level MET amplification. Additionally, copy number arrays (OncoScan Arrays; Thermo Fisher Scientific) as well as another NGS panel (Oncomine Comprehensive v3; Thermo Fisher Scientific) will be used to validate the results. Further clinical data are needed to define clinically relevant MET alterations and to establish a testing algorithm for routine diagnostics."
Sign up for free Altmetric alerts about this article
DOI - https://doi.org/10.18632/oncotarget.28092
Full text - https://www.oncotarget.com/article/28092/text/
Correspondence to - Christina Schmitt - [email protected]
Keywords - next-generation sequencing (NGS), non-small cell lung cancer (NSCLC), fluorescence in situ hybridization (FISH), MET amplification, routine diagnostics
About Oncotarget
Oncotarget is a biweekly, peer-reviewed, open access biomedical journal covering research on all aspects of oncology.
To learn more about Oncotarget, please visit https://www.oncotarget.com or connect with:
SoundCloud - https://soundcloud.com/oncotarget
Facebook - https://www.facebook.com/Oncotarget/
Twitter - https://twitter.com/oncotarget
LinkedIn - https://www.linkedin.com/company/oncotarget
Pinterest - https://www.pinterest.com/oncotarget/
Reddit - https://www.reddit.com/user/Oncotarget/
Oncotarget is published by Impact Journals, LLC please visit https://www.ImpactJournals.com or connect with @ImpactJrnls
Media Contact
[email protected]
18009220957x105
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC




