Oncotarget: Efficacy of osimertinib against EGFRvIII+ glioblastoma
2020-06-09
Volume 11, Issue 22 of @Oncotarget reported that the present study examined the effects of osimertinib on EGFRv III+ Glioblastoma models, both in vitro and in vivo.
Therefore, a panel of six Glioblastoma stem cells expressing EGFRv III+ was evaluated.
The EGFRv III+ GSC differed in the expression of EGFRv III and other key genes.
The GSC line D317, which expresses high levels of EGFRv III and has robust tyrosine kinase activity, was selected for assessing osimertinibs efficacy.
Herein, the authors report that osimertinib inhibits the constitutive activity of EGFRv III tyrosine kinase with high potency
Dr. Madan M. Kwatra from The Departments of Anesthesiology, Pharmacology and Cancer Biology as well as The Duke Cancer Institute at Duke University Medical Center in Durham, North Carolina said, "Glioblastoma (GBM) is the most common and deadliest primary brain tumor. It carries a median survival of 16 months for newly-diagnosed patients whose treatment aligns with the current standard of care consisting of maximal safe resection followed by radiation and chemotherapy."
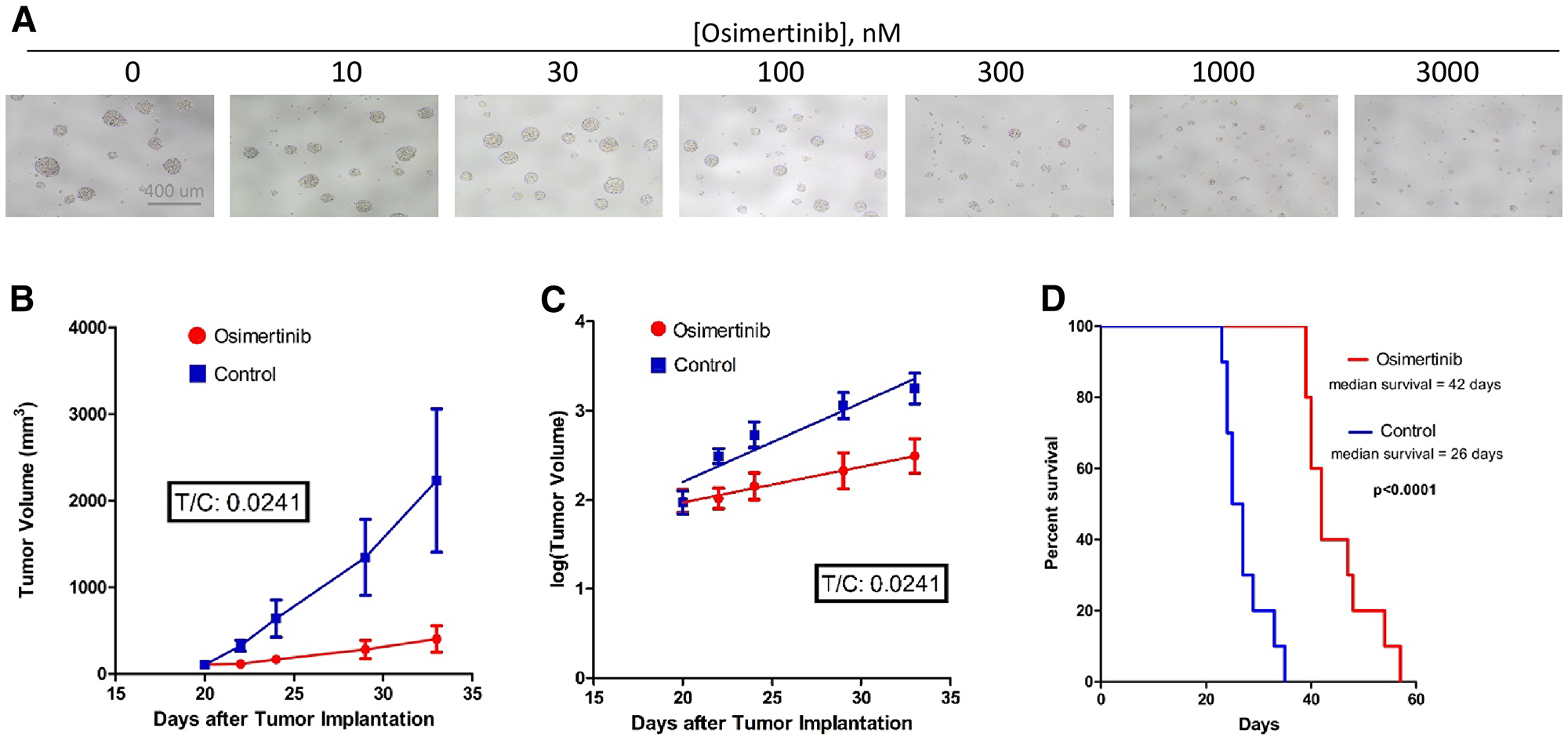
Figure 3: Osimertinib inhibits EGFRvIII+ GBM growth in cell culture as well as in xenograft models. (A) Osimertinib inhibits neurosphere formation and growth of EGFRvIII+ GSC D317. Freshly dissociated GSC D317 at a density of 2000-5000 cells/well in 200 ul media were plated in 96-well plates, and the cells were treated with different concentrations of osimertinib. After 5–7 days of treatment, cells were visualized under the microscope and photographed. (B) Tumor volume vs. time plot in control and treated mice. Athymic mice (n = 20) were injected with100-300K D317 cells subcutaneously, and treatment started once the tumors reached a size of 100–200 mm3 in diameter. Control animals (n = 10) received the vehicle, 0.5% HPMC (hydroxypropyl methyl cellulose), and treated animals (n = 10) received 25 mg/kg osimertinib, given by oral gavage once a day. Animals were sacrificed once tumor size reached 2000 mm3 in diameter. (C) Analysis of the tumor growth data shown in 3B using the rate-based T/C method. A rate-based T/C value below 0.4 indicates the treatment had a significant effect on tumor growth. (D) A group of athymic mice (n = 18) were injected with D317 cells intracranially. Treatment started 7 days after implantation. Control animals (n = 9) were treated with vehicle (0.5% HPC), while treated animals (n = 9) were given 25 mg/kg osimertinib by oral gavage twice a day. Animals were sacrificed when neurological symptoms appeared, including signs of motor disturbances and/or imbalance, decreased food intake, and/or signs of lethargy.
At least four major molecular forms have been identified:
- wild-type EGFR with gene amplification;
- EGFR with a large deletion in the extracellular domain;
- EGFR with kinase domain duplication, and
- EGFR fused with SEPT-14.
There are two central reasons why these agents may have been ineffective in treating Glioblastoma patients:
- All four FDA-approved EGFR-TKIs that have been tested so far in GBM patients do not cross the blood-brain barrier effectively; and
- The extreme molecular and functional heterogeneity of EGFR, as mentioned above, could not be taken into account at the time these clinical trials were performed because this information was not yet understood.
Also, while it spares the wild-type EGFR it inhibits EGFRv III tyrosine kinase with high potency.
The ability of osimertinib to inhibit EGFRv III tyrosine kinase with high potency makes it an attractive candidate to target EGFRv III, a driver of Glioblastoma growth that is present in over 20% of Glioblastoma patients.
"The ability of osimertinib to inhibit EGFRv III tyrosine kinase with high potency makes it an attractive candidate to target EGFRv III"
Herein, they demonstrate that osimertinib inhibits the growth of EGFRv III+ Glioblastoma expressing high EGFRv III tyrosine kinase activity.
The Kwatra Research Team concluded in their Oncotarget Research Paper, "our study establishes that osimertinib penetrates the blood-brain barrier effectively and inhibits the growth of EGFRvIII+ Glioblastomas with higher EGFRvIII tyrosine kinase activity. Additional preclinical studies are needed to define the molecular signatures of EGFRvIII+ Glioblastomas that may make these tumors sensitive to osimertinib."
Sign up for free Altmetric alerts about this article
DOI - https://doi.org/10.18632/oncotarget.27599
Full text - https://www.oncotarget.com/article/27599/text/
Correspondence to - Madan M. Kwatra - [email protected]
Keywords - EGFRvIII, tyrosine kinase, glioblastoma stem cells, osimertinib, xenografts
About Oncotarget
Oncotarget is a biweekly, peer-reviewed, open access biomedical journal covering research on all aspects of oncology.
To learn more about Oncotarget, please visit https://www.oncotarget.com or connect with:
SoundCloud - https://soundcloud.com/oncotarget
Facebook - https://www.facebook.com/Oncotarget/
Twitter - https://twitter.com/oncotarget
LinkedIn - https://www.linkedin.com/company/oncotarget
Pinterest - https://www.pinterest.com/oncotarget/
Reddit - https://www.reddit.com/user/Oncotarget/
Oncotarget is published by Impact Journals, LLC please visit http://www.ImpactJournals.com or connect with @ImpactJrnls
Media Contact
[email protected]
18009220957x105
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC




