Introduction
ER stress is triggered by a disturbance in endoplasmic reticulum homeostasis as a result of Ca2+ depletion, hypoxia, or N-terminal glycosylation dysfunction [1, 2]. The misfolded and unfolded proteins accumulate in the ER during stress, resulting in initiation of the unfolded protein response (UPR) to alleviate the side effects from ER stress and promote cell survival [3-5]. Therefore, the UPR is an evolutionarily conserved adaptive mechanism for increasing cell survival under ER stress conditions [6, 7].
Apoptosis is induced by persistent or excessive ER stress via the activation of the following three ER stress-mediated apoptotic pathways: (1) pro-apoptotic molecular CHOP (C/EBP-homologous protein, growth arrest and DNA damage-inducible gene 153[GADD153] and DNA—damage inducible transcription 3[DDIT3]); (2) phosphorylated c-Jun N-terminal kinase (p-JNK); and (3) cleaved caspase-4 in humans and caspase-12 in rodents [8-16].
The UPR is initiated to relieve the ER load through the following three pathways: (1) PERK (pancreatic ER kinase)/eIF2α (eukaryotic initiation factor 2α)/ATF4 (activating transcription factor 4); (2) IRElα (inositol requiring enzyme 1α)/XBP-1 (X-box-binding protein); and (3) ATF6α (activating transcription factor 6α). It is accompanied by the dislocation of the ER chaperonin glucose-regulated protein 78-kDa (GRP78, also known as Bip) from the ER membrane with PERK, IRElα, and ATF6α; from there, GRP78 enters the ER lumen [8]. Through these three pathways, the ER load is ameliorated by following three methods: (1) a reduction in the entry of newly synthesized proteins into the ER through attenuating protein translation; (2) an increase in the protein-folding capacity by upregulating ER gene expression; and (3) the degradation of misfolded and unfolded proteins through ER-associated degradation (ERAD) and lysosome-mediated autophagy.
The misfolded and unfolded proteins are mainly degraded by ERAD through the ubiquitin-proteasome system (termed ERAD I) [17, 18], though lysosome-mediated autophagy is also triggered when the ERAD is impaired, therefore, lysosome-mediated autophagy has been referred to as the ERAD II pathway [17, 19].
The role of the ER stress and the UPR in several physiological and pathological processes has been previously reviewed. However, the comprehensive role of ER stress and the UPR in stem cells has not been summarized.
Stem cells have been identified in various tissues. These cells correlate with development, tissue renewal and some disease processes. Many stem cells persist throughout the entire adult life of the organism [20]. This observation raises questions about quality maintenance and cellular homeostasis mechanisms. Several papers have highlighted the importance of autophagy in stem cells [20-24] and the activation of autophagy in these cells during self-renewal, pluripotency, differentiation and quiescence [23, 24]. Consistent with autophagy, the UPR is also confirmed as an evolutionarily conserved adaptive mechanism to maintain cell homeostasis through protein synthesis, remolding and degradation, and crosstalk between autophagy and ER stress has been widely revealed in several studies [25]. ER stress mediates autophagy [26], whereas autophagy inhibits ER stress [27]. The relationship between autophagy and ER stress depends on the cell type and conditions. Oxidative stress, mitochondrial dysfunction and ER stress also interact with one another [28-31]. Moreover, the interplay among oxidative stress, mitochondrial dysfunction and autophagy is dependent on cell type [32-33]. Mitochondrial function and oxidative stress are all widely related to stem cells [34-37]. However, it is largely unknown whether ER stress and the UPR interact with mitochondrial dysfunction, oxidative stress and autophagy in stem cells.
Thus, in addition to autophagy, the vital role of ER stress and the UPR in stem cells should be emphasized.
ER stress and the UPR in embryonic stem cells
Embryonic stem cells (ESCs) are derived from blastocyst the inner cell mass (ICM). In vivo, they differentiate into the three primary germ layers: the ectoderm, endoderm and mesoderm [38]. These differentiation processes are regulated by complicated mechanisms.
UPR in embryonic development
The development of a peri-implantation embryo is inhibited by excessive UPR. Following embryo demise, the UPR is induced by ER stress mediated apoptosis, this situation can be reversed by the ER stress inhibitor TUDCA (tauroursodeoxycholic acid) [39-42]. Inhibiting ER stress signaling has yielded beneficial effects on embryo survival and long-term developmental potential [43]. Furthermore, blastocyst formation in vitro during preimplantation embryo development was prevented by UPR [44]. The role of ER stress and the UPR in preimplantation embryonic development and developmental arrest has been reviewed [7, 45]. Additionally, hypoxia supplies a ‘niches’ for embryonic stem and progenitor cells, and low oxygen (O2) regulates embryonic stem and progenitor cell differentiation [46]. Up-regulation of the UPR after hypoxia suggests potential roles for the UPR in embryonic stem and progenitor cells [47]. Heavy proteins loaded on the ER are comprised of metabolic and secreted enzymes, antibodies, serum proteins and extracellular matrix (ECM) components during development in different cell types. In these conditions, cellular homeostasis is restored by the UPR, which is initiated by physiological ER stress [48]. Early embryogenesis in IVP (in vitro-produced) bovine embryos depends on an appropriate balance between autophagy and ER stress [49]. Therefore, the UPR is involved in embryonic development and embryonic stem cell and progenitor cells. However, whether autophagy and ER stress correlate with each other in embryogenesis is unknown.
Embryo development failure is the result of a deficiency in several UPR-regulating genes. Targeted mutation of the mouse GRP94 (glucose regulated protein 94) gene disrupts development and perturbs ER stress and the UPR signaling pathway [50]. A activation of UPR-regulating genes and apoptotic cell death by deletion of ERp44 (endoplasmic reticulum (ER) resident protein 44) in mice and zebrafish causes embryonic lethality at E9.5 to E12.5 and abnormal heart development [51].
As an ER molecular chaperone, GRP78 is essential for embryonic cell growth and pluripotent cell survival. A GRP78 deficiency results in ceased proliferation and a massive increase in apoptosis in the ICM [52]. Ubiquitous and high expression of the UPR regulating genes GRP78 and GRP94 occurs in late embryogenesis, followed by downregulation immediately after birth, indicating the important role of the UPR in embryogenesis [53]. The vital role of the UPR and two glucose regulated proteins (GRPs) GRP78 and GRP94 in regulating homeostasis of organs originating from the endoderm, mesoderm and ectoderm has been reviewed [54].
UPR in differentiation and pluripotency of ESCs
Embryonic stem cells are useful tools for studying the regulatory mechanisms of cell proliferation and differentiation. Induction of the UPR during neuronal differentiations of rBMSCs (rat bone marrow stromal cells) and mESs (mouse embryonic stem cells) as well as neuronal marker NF-L (neurofilament-L) expression by non-cytotoxic concentrations of ER stress inducers strongly suggest the potential role of the UPR in neuronal differentiation [55]. Indeed, ER stress participates in ESC differentiation induced by retinoic acid through ERAD (ER-associated degradation) [56]. ER stress-induced UPR enhances definitive endodermal differentiation and inhibits other germ layer commitments of ESCs in EBs (embryoid bodies) of mouse origin via both the Smad2 and β-catenin signaling pathways [57]. DKK3 (dickkopf homolog 3) regulates ESC-SMC (smooth muscle cell) differentiation by activating ATF6 and promoting myocardin expression [58]. ESC differentiation is ceased by blocking the ER stress-induced UPR, which has been shown in several studies. Although the undifferentiated condition are maintained by the addition of LIF (leukemia inhibitor factor) into the culture medium, the cells have the potential to differentiate toward meso-endoderm lineages in the long-term mESC cultures because of spontaneously secreted vascular endothelial growth factors (VEGFs), which are attributed to hypoxia-inducible factor alpha (HIF1α) and the ER stress induced-UPR. Therefore, blocking ER stress following the inhibition of the VEGF and VEGFR signaling pathways prevents cell differentiation and promotes mESCs self-renewal [59]. Inhibition of ER stress by TUDCA prevented the differentiation of ESCs into definitive endodermal cells even with activin A treatment [57].
The self-renewal of stem cells is maintained by a balance between the positive regulation pathway of promoting growth and the negative regulation pathway that promotes pro-apoptosis [60].
Blanco-Gelaz reported that hESCs must resolve ER stress in culture conditions to survive and maintain pluripotency [61]. ERp44 (endoplasmic reticulum protein 44)-deficient ESCs showed higher [Ca2+] transient amplitudes, elevated ROS, and enhanced sensitivity to Tu (tunicamycin, 5 μg/mL), which induced ER stress mediated apoptosis [51].
In summary, the UPR is involved in the proliferation, differentiation, self-renewal and ER stress mediated apoptosis of embryonic stem cells. Moreover, embryonic demise is caused by UPR dysfunction.
Hematopoietic stem/progenitor cells and the UPR
Hematopoietic stem cells (HSCs) are widely studied because of the relative ease of access to cell populations in the bone marrow and blood for in vitro and in vivo assays. A large number of mature blood cell lineages are produced each day through the proliferation and differentiation of progenitors originating from a small number of quiescent stem cells. The life span of mature blood cells is very limited despite the fact that HSCs exist in the organs throughout life. HSCs are identified and isolated by the positive expression of cell surface protein markers (ATXN1/Sca-1 and KIT/c-Kit) or the negative expression of lineage markers (ITGAM/MAC-1 for mature myeloid cells, CD8 for T cells, and PTPRC/B220 for B cells) on immature cells [20, 21]. HSCs exist in hypoxic microenvironments (niche) in the bone marrow, and the majority of HSCs are in a quiescent state [62].
UPR in the HSCs self-renewal
The blood system is maintained by a pool of HSCs that are long-living due to their capacity for self-renewal. Consequently, cell damage occurs by stress stimuli including reactive oxygen species (ROS), nutrient fluctuation and DNA damage [63, 64]. It is unclear how to prevent the loss of function and clonal persistence of oncogenic mutations that increase the risk of leukemogenesis in stressed HSCs [65, 66]. In the physiological stem cell hierarchy, the UPR has been shown to regulate the self-renewal capacities of HSCs, the importance of the UPR in HSCs has been reviewed by Kharabi Masouleh et al [67]. A gene expression analysis revealed that, PERK is predominately activated in HSCs compared to downstream progenitor populations [68]. The pharmacological PERK inhibitor GSK2606414 causes apoptosis in HSC populations, whereas the progenitor populations are resistant, even at high concentrations [68]. The UPR governs the integrity of the hematopoietic stem-cell pool during stress. Van Galen suggested that the HSC pool maintains clonal integrity by the clearance of individual HSCs after stress to prevent the propagation of damaged stem cells through the UPR to either resolve stress or initiate apoptosis [68]. Indeed, the UPR is induced by ROS [69], and ROS is initiated by ER stress [70].
UPR in HSC death and homeostasis
Elevated ER stress and activation of the UPR are caused by a deficiency in UFBP1 (Ufm1 binding protein 1, also known as DDRGK1, Dashurin and C20orf116), which subsequently leads to the death of hematopoietic stem/progenitor cells. Thus, UFBP1 plays pleiotropic roles in the regulation of hematopoietic cell survival and differentiation via modulating ER homeostasis and erythroid lineage-specific gene expression [71]. A Runx1 deficiency of in hematopoietic stem and progenitor cells (HSPCs) results in slow growth, low biosynthesis, small cell phenotype and markedly reduced ribosome biogenesis (Ribi), which is consistent with lower p53 levels, reduced apoptosis, attenuated UPR, and resistance to genotoxic and ER stress [72]. Cripto regulates hematopoietic stem cells as a hypoxic-niche-related factor through the cell surface receptor GRP78 [73]. Soluble Cripto-1 has been shown to selectively regulate cells expressing high levels of surface GRP78, to activate the PI3K/Akt pathway and to promote stem cell maintenance ex vivo [74]. A proteomics analysis revealed that Cripto-GRP78 binding stimulates glycolytic metabolism-related proteins and results in lower HSC mitochondrial potential [74]. ER stress also correlates with mitochondrial bioenergetics [75, 76]. Cripto-GRP78 is a novel HSC regulatory signaling pathway that primarily works in the hypoxic niche [77]. Acute inducible ablation revealed a role for GRP78 in hematopoietic stem cell survival, lymphogenesis and stress signaling regulation [78]. Dppa5 overexpression robustly increases the reconstitution capacity of HSCs after in vitro culture, and ER stress levels and subsequent apoptotic signals are significantly decreased upon ectopic Dppa5 expression. Conversely, downregulation of Dppa5 expression abolished long-term reconstitution by HSCs because of increased apoptosis caused by elevated ER stress levels [79]. HIF2α protects human hematopoietic stem/progenitors and acute myeloid leukemia cells from apoptosis induced by ER stress [80]. RCAD/Ufl1, an Ufm1 E3 ligase, is essential for embryonic development, HSC survival and erythroid differentiation. The loss of RCAD/Ufl1 blocked autophagic degradation, increased mitochondrial mass and reactive oxygen species, and led to a DNA damage response, p53 activation and enhanced cell death of HSCs [81], accompanied by elevated ER stress and activation of the UPR [81]. ATF4-deficient mice showed fewer hematopoietic progenitor cells in the liver [82]. ATF4 is a downstream target of PERK, which is one pathway that imitates the UPR [7]. In contrast, myeloid cell and eosinophils progenitors selectively activate the endoribonuclease IRE1α and Xbp1 mRNA splicing for the UPR without inducing parallel ER stress signaling pathways [83]. Inherited heterozygous point mutations in the ELANE gene, which encodes neutrophil elastase (NE), activates severe congenital neutropenia (SCN) [84, 85]. Genome editing and differentiation protocol results obtained in studies with both normal and SCN patient-derived induced pluripotent stem cells (iPSCs) suggest that promyelocyte death and differentiation arrest are caused by ELANE point mutations and are associated with NE mislocalization and activation of the UPR/ER stress. This disadvantage is reversed by sivelestat, an NE-specific small-molecule inhibitor, and the UPR/ER stress is ameliorated [86].
Mitochondrial UPR and HSC aging
The mitochondrial UPR is triggered by mitochondrial dysfunction or the accumulation of unfolded proteins within mitochondria, it promotes cell survival along with the repair and recovery of defective mitochondria [87]. The degeneration of mitochondrial quality and activity contributes to aging, including cellular senescence, chronic inflammation, and the age-dependent decline in stem cell activity [88].
Reduced quiescence in HSCs is caused by SIRT7 inactivation, accompanied by increases in the mitochondrial UPR (UPRmt) and compromised regenerative HSC capacity. SIRT7 up-regulation improved the regenerative capacity of aged HSCs via decreasing the UPRmt pathway [34]. HSCs repress mitochondrial biogenesis and OxPhos (oxidative phosphorylation) via the UPRmt signaling pathway to coordinate the metabolism required for stem cell maintenance [34].
In summary, the UPR maintains HSC self-renewal, while the ER stress mediated apoptotic pathway initiates HSC deaths. Whether HSC differentiation and proliferation are regulated by the UPR is unknown and must be explored in the future.
Neuronal stem cells and the UPR
Recent studies have suggested that the ER stress induced UPR pathway is involved in neurodegenerative diseases, and that ER stress is a potential therapeutic target for preventing these diseases [89-92]. In addition to being involved in the pathology of neurological diseases, the UPR also plays vital roles in neural differentiation during brain development [53, 93-95]. All of these results emphasize the importance of ER stress and the UPR on neurogenesis.
UPR in NSCs differentiation
ER stress causes aberrant neuronal differentiation of NSCs followed by neurite growth inhibition [96]. Induction of the UPR during rBMSC (rat bone marrow mesenchymal stem cell) and mESC (mouse embryonic stem cell) neuronal differentiation as well as NF-L (neurofilament) expression by ER stress inducers strongly suggest a potential role for the UPR in neuronal differentiation [54]. Human pluripotent stem cell (hCN)-derived cortical neurons (CNs) under mild-to-moderate hypothermia (28-32°C) induce cold-shock protein expression and mild ER stress, which subsequently activates the UPR. Blocking a principal UPR pathway mitigates the protective effect of cooling against oxidative stress, while pre-cooling neurons abrogates toxic injury produced by the ER stressor tunicamycin. Therefore, hypothermia is a potent neuroprotective mechanism that upregulates the adaptive chaperone-driven pathways of the UPR and even maintains ER-hormesis [97].
UPR in NSCs self-renewal
In addition to resulting in neuronal cell death through calcium toxicity and apoptotic pathways, ER stress also triggers a series of adaptive responses including the UPR, autophagy, the expression of pro-survival proteins and the enhancement of ER self-repair abilities, leading to less ischemic brain damage [98].
Inhibition or down-regulation of DOT1L in neural stem cells (NSCs) in the cerebral cortex impairs NSC proliferation and survival, induces genes that are activated during the UPR in the endoplasmic reticulum (ER), and results in the loss of H3K79me2 at the ATF4- and CHOP-promoters, which appears to mark a point-of-no-return that activates the death program in NSCs [99]. Activation of the galanin receptor (GalR)3 increases NSC viability; this protective effect occurs because of decreased apoptosis and CHOP levels [100]. ER stress, mitochondrial dysfunction and calpain/JNK activation are involved in oligodendrocyte precursor cell death by unconjugated bilirubin [101]. Human cytomegalovirus induces apoptosis in neural stem/progenitor cells derived from induced pluripotent stem cells by generating mitochondrial dysfunction and ER stress [102]. FTD (frontotemporal dementia) neurons differentiated from induced pluripotent stem cells (iPSCs) from individuals with FTD-associated MAPT mutations show activation of the UPR; a transcriptome analysis of these cells exhibited disease-associated gene expression profiles [103]. Upregulation of GRP78 and GRP94 suggests that the USH2A variant (Arg4192His) in retinitis pigmentosa patients causes the disease through protein misfolding and ER stress [104]. These results indicate that the transcriptome is potentially reprogrammed by the UPR.
Therefore, the UPR is involved in neuron cell differentiation and self-renewal, and ER stress dysfunction initiates apoptosis.
Esophageal epithelium precursor / stem cells and the UPR
The self-renewal of most epithelial tissues is attributed to the presence of stem cells, and the existence of esophageal epithelium precursor/stem cells has been demonstrated by several papers [105-109]. Esophageal epithelial stem cells demonstrate several unusual properties, and their identification may facilitate studies on esophageal carcinogenesis [106]. Controlling differentiation, proliferation and self-renewal is highly important to epithelium precursor/stem cells.
UPR in the differentiation of esophageal epithelium stem/precursor cells
UPR induced epithelial differentiation in precursor cells in the esophageal epitheliummay serve as a quality control mechanism that protects against the development of esophageal cancer [110]. The expression of the highest and lowest levels of intestinal stem cell (ISC)-related genes in the CD44hiGRP78−/lo and CD44loGRP78+ cell populations reveals that the combination of GRP78 and CD44 can largely distinguish ISCs from progenitor cells [111]. Rapid loss of intestinal epithelial stem cells was caused by the initiation of the UPR by ER stress [112]. IRE1a acts as a novel mediator in regulating osteogenic differentiation through negative feedback with BMP2 [113].
UPR in the self-renewal of esophageal epithelium stem/precursor cells
Intestinal stem cell proliferation is controlled by the integration of UPRER (the unfolded protein response of the endoplasmic reticulum) and the oxidative stress signaling pathway [114]. MSC proliferation is inhibited by treatment with low-intensity and intermittent negative pressure, accompanied by triggering ER stress-associated cellular apoptosis, which further enhances the osteogenesis activity and induces differentiation to osteoblasts [115]. The UPRER is a critical regulator of intestinal stem cell (ISC) quiescence in Drosophila melanogaster. Wang’s report found that ISCs require activation of the UPRER for regenerative responses, though a tissue-wide increase in ER stress triggers ISC hyperproliferation and epithelial dysplasia in aging animals [114]. ADAR1 (adenosine deaminase acting on RNA 1) plays vital roles in survival, maintenance of intestinal stem cells and intestinal homoeostasis by suppressing ER stress and interferon (IFN) signaling [116]. The UPR transcription factor Xbp1 suppresses intestinal tumorigenesis by directly acting on intestinal stem cells [117]. PERK controls Drosophila lifespan by promoting ISCs proliferation in response to ER stress [118].
Hence, the UPR plays a vital role in esophageal epithelium stem/precursor cells processes, including differentiation, proliferation, self-renewal and quiescence.
Cancer stem cells and the UPR
Cancer stem cells (CSCs) are a small subset of cancer cells with the indefinite potential of self-renewal and the capability to drive tumorigenesis [119]. These cells are believed to be responsible for tumor initiation, progression, metastasis, chemotherapy and radiation resistance, and tumor relapse after therapy [120, 121]. Therefore, targeting CSCs is crucial for the effective treatment of cancer [122].
UPR in CSC differentiation
Colon cancer stem cells (colon-CSCs) are more resistant to conventional chemotherapy than differentiated cancer cells [123-127]. The ER is a vital organelle, and that plays an important role in regulating the homeostasis of normal intestinal stem cells [128]. ER-stress-induced activation of the UPR forces colon-CSCs to differentiate [128], and the rapid loss of intestinal epithelial stemness results in their enhanced sensitivity to chemotherapy in vitro and in vivo [112]. Induced activation of the UPR may be used to increase the sensitivity of colon-CSCs to the effects of conventional chemotherapy [128]. Thus, the UPR plays an important role in the regulation of intestinal epithelial stem cell differentiation [128]. At the intestinal level, 17-allylamino-demethoxygeldanamycin (17AAG), which is an inhibitor of the stress protein HSP90, promotes splicing of the transcription factor X-box binding protein 1 (XBP1), accompanied by a decrease in intestinal damage and an increase in Lgr5+ stem cells [129]. Activation of the UPR in healthy intestines by genetic deletion of the ER chaperone GRP78 results in a loss of stem cells due to differentiation and a quick repopulation of healthy stem cells that were not recombined [112]. GRP78 as a pluripotent stemness regulator is up-regulated in oral cancer patients with an areca nut-chewing habit [130]. GRP78 may serve as a molecular target that can be further developed for the eradication of refractory HNC (head-neck cancer) with a stemness phenotype [131].
The antitumor effect of shikonin is enhanced by inhibiting endoplasmic reticulum stress via the JNK/c-Jun pathway in human glioblastoma stem cells [132]. Because SEL1L is an UPR and ER-associated degradation protein, downregulation of SEL1L sensitizes glioma stem cells to the cytotoxic effects of valproic acid [133]. In vivo RNAi screening for BMI1 targets suggests that the TGF-β/BMP-ER stress pathways are key regulators of neural- and malignant glioma-stem cell homeostasis [134]. Tumor cells that secrete GRP78 induce the differentiation of bone marrow mesenchymal stem cells to cancer-associated fibroblasts [135]. The metastases are caused by disseminated tumor cells (DTCs), which colonize secondary organs. Breast cancer DTC cell lines with stem/progenitor cell cancer phenotypes (CD44(high)/CD24(low)) show high expression of the UPR proteins GRP78 and GRP94. These observations provide novel insights for curing breast cancer by controlling the UPR [136].
UPR in CSC self-renewal
High expression of Abcg1 in mouse and human LG-GSCs (low-grade glioma stem cells) is critical for protecting mouse LG-GSCs from ER-stress-induced apoptosis [137]. The major ER chaperone GRP78 is in a dynamic equilibrium between folding proteins and ER transmembrane receptors. Activation of the UPR is initiated by an increased load of folding proteins and accompanied by the dislocation of GRP78 away from the transmembrane receptors [128]. Cell surface levels of GRP78 are high in stem cells and tumor cells. The elevated Grp78 levels in human tumors are closely related with the promotion of tumor growth, malignancy and therapy resistance [138-140]. The dietary compound isoliquiritigenin (ISL) has synergistic effects with chemotherapeutic drugs for inhibiting breast cancer stem cell proliferation and colony formation, inhibiting self-renewal and multidifferentiation abilities, and targeting GRP78 via the β-catenin/ABCG2 pathway [141]. Ovarian cancer stem-like cells (ID8) increase the expression of plasma membrane GRP78 ((mem)GRP78), which increases self-renewing abilities. The down-regulation or inhibition of the GRP78 protein by antibodies from the carboxy-terminal domain of GRP78 reduces the self-renewal ability of ovarian cancer cells [142]. The mechanisms that allow residual multiple myeloma (MM) cells to persist after bortezomib (Bz) treatment remain unclear. A recent study suggested that GRP78 is expressed in MM quiescent cells. The expression of GRP78 was downregulated when a specific SubAB bacterial toxin was used to kill Bz-surviving MM cells. A high number of GRP78(high)/CD138+ MM cells are associated with progressive disease [143].
Pterostilbene (PT) treatment suppresses self-renewal and the irradiation-resistant abilities of GSCs (glioma stem cells) via negatively modulating the GRP78 signaling pathway. Subsequently, GSC development is suppressed [144]. GRP78 is required for cancer stem-like cell subpopulation in the MCF-7 breast cancer cell line that are resistant to IR (radiotherapy); knockdown of this gene augments the effects of IR, whereas GRP78 overexpression increases the radiation resistance of the subpopulation to IR [145]. BFA (Brefeldin A) effectively reduces the survival of suspension Colo 205 cells (IC50 = ~15 ng/mL) by inducing apoptosis and inhibiting the clonogenic activity of Colo 205 CSCs (cancer stem cells) in a tumor sphere formation assay and soft agar colony formation assay in the same nanogram per milliliter range. At such low concentrations, BFA effectively induced the UPR, which was indicated by the increased in the mRNA expression of UPR-related genes, such as GRP78, XBP1 and CHOP [146].
Overexpression of the REIC/Dkk-3 gene showed a potent selective therapeutic effect on various human cancers by triggering ER stress and the UPR [147].
Therefore, the importance of the UPR is demonstrated through its role in cancer stem cell self-renewal, differentiation, and apoptosis, which is dependent on the cell types. The roles of the UPR in cancer stem cells will provide a novel target for cancer therapy.
Mammary stem cells and the UPR
Mammary stem cells (MaSM) constitute a quiescent and self-renewing population isolated from fetal and adult mammary epithelial cells [148]. They are capable of developing into differentiated ductal, alveolar and myoepithelial cells [149].
The behavior of isolated stem cells from fetal and adult mammary epithelial cells is regulated by CRIPTO and its cell-surface receptor GRP78; CRIPTO inhibition promotes stem cell differentiation and reduces self-renewal when cultured ex vivo. In contrast, CRIPTO treatment maintains the stem cell phenotype and increases the mammary reconstitution capacity. Mammary gland reconstitution potential is ceased by the deletion of GRP78 from adult mammary epithelial cells. These results suggest that CRIPTO/GRP78 correlate with the fetal and adult mammary stem cell behavior ex vivo [150]. Furthermore, CRIPTO-1 activates branching morphogenesis and the epithelial-mesenchymal transition in the mammary gland both in vitro and in vivo, and together with the cofactor GRP78, it is critical for the maintenance of mammary stem cells ex vivo [151]. Knockout of GRP78 in mammary epithelial stem/progenitor cells fails to regenerate the mammary glands [152]. IGF-1 is involved in mammary gland development via GRP78 regulation [153].
In summary, the UPR is involved in the control of self-renewal, differentiation and multipotency in MaSCs.
UPR is involved in the differentiation of adipose-derived stem cells
Adipose-derived stem cells (ASCs) are a population of multipotent stem cells that are isolated from the adipose tissue. They are capable of differentiating into various types of terminally differentiated cells, including but not limited to adipocytes, osteocytes and chondrocytes [154-158].
Tauroursodeoxycholic acid (TUDCA) treatment of hASCs significantly decreases the representative UPR regulating gene GRP78, the adipogenic markers peroxisome proliferator-activated receptor gamma (PPARγ) and glycerol-3-phosphate dehydrogenase 1 (GPDH), and lipid accumulation. Moreover, it decreases adipogenic differentiation from hASCs and significantly decreases in vivo adipogenic tissue formation and ER stress [159]. ER stress signaling reduces adipocyte differentiation and attenuates adipogenesis in mice by upregulating phosphorylated eIF2α and its downstream target CHOP but not IRE1α. ER stress inhibits adipocyte differentiation in 3T3-L1 cells [160]. In contrast, impaired eIF2α phosphorylation enhances adipocyte differentiation in mice MEFs (embryonic fibroblasts) [160]. Mice that are deficient for CHOP (CHOP-/-) gain more fat mass than wild-type mice on high-fat diets. Additionally, CHOP deletion in genetically obese Leprdb/db (leptin receptor) mice increased body fat mass without altering adipocyte size. In contrast to the eIF2α-CHOP pathway, the activation or deletion of IRE1a (also known as Ern1) did not alter adipocyte differentiation in 3T3-L1 cells [160]. ER stress is involved in human adipocytes through the regulation of inflammation and adiponectin levels [161].
Thus, the UPR is involved in the differentiation of adipose-derived stem cells.
UPR is involved in the differentiation and self-renewal of MSCs
Mesenchymal stem cells (MSCs) with self-renewal potential and multipotency can be isolated from various adult and fetal tissues. MSCs have the capacity to proliferate and differentiate into a variety of cell lineages such as osteoblasts, chondrocytes, adipocytes, fibroblasts, cardiomyocytes, etc. [162].
Palmitate induces apoptosis in hUC-MSCs (human umbilical cord-derived mesenchymal stem cells), which might be attributed to ER stress [163]. ER stress contributes to arsenic trioxide-induced intrinsic apoptosis in human umbilical and bone marrow mesenchymal stem cells; moreover, arsenic trioxide significantly reduces the viability of HUMSCs and HMSC-bm (bone marrow) in a concentration- and time-dependent manner [164]. LPA (lysophosphatidic acid) treatment decreases ER stress-mediated apoptosis in hypoxia and serum deprivation-stimulated mesenchymal stem cells [165]. Therefore, ER stress-associated apoptosis is involved in mesenchymal stem cell apoptosis [166-168]. GRP78 regulates ER stress-mediated apoptosis in cartilage development during the course of chondrocyte differentiation; GRP78 overexpression inhibits ER stress-mediated apoptosis, while GRP78 knockdown via siRNA activates the ER stress-specific caspase cascade in developing chondrocyte tissues from C3H10T1/2 cells [169]. Osteoblast proliferation and differentiation are impaired in Arl6ip5 knocked-down and deficient primary osteoblasts; it also induced ER stress and enhanced ER stress-mediated apoptosis. CHOP is involved in the regulation of apoptosis and differentiation in Arl6ip5 knocked-down osteoblasts [170] from the stromal/osteoblast cell line (UAMS-32). The cysteine-rich ER stress inducible factor with EGF-like domains 2 (Creld2) is an important mediator of BMP9-regulated osteogenic differentiation in mesenchymal stem cells [171].
IRE1a negatively regulates BMP2-induced osteoblast differentiation, and this IRE1a inhibition effect depends on the GEP (granulin-epithelin precursor) growth factor in pluripotent mesenchymal C2C12 cells [172]. Chondrocyte differentiation is controlled by IRE1α based on its enzymatic activity [173]. ATF6 positively regulates chondrocyte hypertrophy and endochondral bone formation through activating Runx2-mediated hypertrophic chondrocyte differentiation in C3H10T1/2 cells [174]. The chronic exposure of BMSCs (mesenchymal stem cells in bone marrow) to alcohol induces adipogenesis and disrupts or inhibits osteogenesis accompanied by inducing ER stress, ATF4/CHOP and activation of TNF-α signaling by ATF4/CHOP; the knockdown of either ATF4 and CHOP or TNF-α with siRNAs is able to reverse ethanol-induced adipogenesis [175].
Hence, the differentiation and self-renewal of MSCs is regulated by the UPR.
Cardiac stem cells and UPR
Cardiac stem cells have the capacity of self-renewal and can differentiate into multiple cell types [176, 177]; they are identified by the following markers: c-kit, Sca-1, and Isl1 [178].
PARM-1 (prostatic androgen repressed message-1) is an endoplasmic reticulum (ER) molecule that is involved in ER stress-induced apoptosis in cardiomyocytes.
PARM-1 plays an important role in the cardiomyogenic differentiation of P19CL6 cells by regulating the BMP/Smad signaling pathways, demonstrating a novel role for PARM-1 in the cardiomyogenic differentiation of stem cells [179]. This result hints at the potential role of the UPR in cardiac stem cells, but the accurate function of the UPR must be explored in the future.
UPR and iPSCs
Induced pluripotent stem cells (iPSCs) are self-renewable and can differentiate into different types of adult cells [180]. iPSCs are established through nuclear reprogramming by four key reprogramming gene factors (POU5F1(OCT4), SOX2, KLF4, and c-MYC) [181]; they are called the OKSM or Yamanaka factors [182]. The four factors have been thought to mainly modify gene expression profiles and epigenetic markings. Whether the UPR is involved in installing iPSCs remains unclear. A number of studies have revealed that the UPR is associated with reprogramming. Cell growth arrest and death are triggered by Gln (glutamine) and Glc (glucose) depletion, accompanied with dramatic gene expression reprogramming and distinguishable ER stress and the UPR [182]. Thus, the importance of the UPR in controlling transcriptional and translational reprogramming is reviewed [183]. Furthermore, c-myc translation and expression are regulated by ER stress inducers [184]. The UPR is activated during normal epidermal KC (keratinocyte) differentiation and induces C/EBPbeta, KLF4, and ABCA12 mRNAs [185]. Compared to protected sites, endothelia in athero-susceptible regions shows activation of ER stress and the UPR; moreover, there is evidence of altered expression of pro-inflammatory nuclear factor kappa B (NFκB) and oxidant/antioxidant pathways and low expression of major protective factors, notably endothelial nitric oxide synthase and Kruppel-like factors KLF2 and KLF4 [186]. Similar to ESCs, iPSCs show a reduction in mitochondrial mass and ROS generation [187, 188]. The adaptive UPR response affects mitochondrial function [189-191]. Additionally, ER stress and oxidative stress crosstalk with one another [192, 193]. These results suggest the potential role of the UPR in iPSCs.
Concluding remarks
Stem cell self-renewal, pluripotency, differentiation and quiescence have been studied in the past decades, but knowledge of the role of ER stress and the UPR in stem cells is in its infancy. Stem cells are particularly vulnerable to stress. The UPR and ER stress have not yet been extensively studied in these cells; however, the data implicate pivotal roles for ER stress and the UPR in stem cells (Figure 1). A number of ER stress and UPR regulated genes are involved in stem cells (Table 1). Activation of the ER stress and the UPR is not only a mechanism for the eliminating of stem cells that encounter insults during development. Instead, activation of ER stress and the UPR might also be beneficial for directing stem cells into differentiation or for maintaining a proliferative status [96, 194, 195, 196]. Thus, the UPR must be tightly regulated and adapted to various cellular needs to balance cell fate decisions ranging from differentiation to cell death [99]. However, the role of ER stress and the UPR is dependent on the cell type. Therefore, (i) the functions of ER stress and UPR in the different stem cells should be explored further. (ii) The potential for controlling cell differentiation by regulating ER stress and the UPR should also be explored, particularly that of cancer stem cells. The latter could potentially provide novel insights for cancer therapy. (iii) Finally, the crosstalk between ER stress and autophagy, oxidative stress, and mitochondrial dysfunction should be explored to supply novel insights for controlling stem cell differentiation, proliferation, self-renewal and homeostasis maintenance.
Table 1: List of ER stress and UPR modulators in stem cell
Modulator |
Functions |
|
Reference |
ADAR1 |
ADAR1 is essential for intestinal homeostasis and stem cell maintenance and survival through suppressing endoplasmic reticulum (ER) stress |
[116] |
|
ASK1 |
ASK1 knockdown in C17.2 neural stem cells diminished high glucose- or tunicamycin-induced IRE1α activation, reveals that ASK1 plays a causal role in diabetes- induced ER stress and apoptosis |
[198] |
|
ATF6 |
ATF6 upregulates XBP1s and inhibits ER stress-mediated apoptosis in osteoarthritis cartilage |
[199] |
|
ATF6 |
ATF6 positively regulates chondrocyte hypertrophy and endochondral bone formation through activating Runx2-mediated hypertrophic chondrocyte differentiation |
[174] |
|
Bmi1 |
In vivo RNAi screen for BMI1 targets reveals TGF-β/BMP-ER stress pathways as key regulators of neural- and malignant glioma-stem cell homeostasis |
[134] |
|
BMP-2 |
BMP-2 stimulates differentiation of myoblasts into osteoblasts through the PERK-eIF2α-ATF4 pathway, additionally stimulates Tmem119 and Tmem119 increases ATF4 |
[197] |
|
C/EBPβ |
Mechanical strain downregulates C/EBPβ in MSC as well as ameliorates Endoplasmic Reticulum Stress |
[168] |
|
Creld2 |
Endoplasmic reticulum (ER) stress inducible factor cysteine-rich with EGF-like domains 2 (Creld2) is an important mediator of BMP9-regulated osteogenic differentiation of mesenchymal stem cells |
[171] |
|
DOT1L |
The lysine methyltransferase DOT1L/KMT4 promotes proliferation and protects cortical neural stem cells from activation of ATF4-DDIT3-mediated ER stress In Vitro |
[99] |
|
Dppa5 |
Dppa5 promotes hematopoietic stem cell activity by decreases endoplasmic reticulum stress |
[79] |
|
ELANE |
Promyelocyte death and differentiation arrest are caused by ELANE point mutations, and is related with NE mislocalization and activation of the unfolded protein response/ER stress |
[86] |
|
ERp44 |
Deficiency of ERp44 in mouse and zebrafish results in significant embryonic lethality, abnormal heart development, altered Ca(2+) dynamics, reactive oxygen species generation, activated ER stress gene profiles, and apoptotic cell death |
[51] |
|
GalR3 |
GalR3 activation promotes adult neural stem cell survival due to decreased apoptosis and CHOP levels |
[100] |
|
Grp78 |
GRP78 is essential for the growth of embryonic cell and the survival of pluripotent cell |
[52] |
|
GRP78 |
GRP78 plays a pleiotropic role in BM cells and contributes to HSC survival and the maintenance of the lymphoid lineage through UPR signaling pathways |
[78] |
|
HSP90 |
Inhibition of the stress protein HSP90, promotes the splicing of the transcription factor X-box binding protein 1 (XBP1), accompanied with a decreasing intestinal damage and an increasing Lgr5+ stem cells |
[129] |
|
IRE1a |
IRE1a negatively regulates BMP2-induced osteoblast differentiation through UPR and ER stress pathway |
[173] |
|
PERK |
PERK promotes intestinal stem cell proliferation in response to ER stress and limits drosophila lifespan |
[118] |
|
RCAD/Ufl1 |
RCAD/Ufl1 is essential for hematopoietic stem cell function and murine hematopoiesis associated with ER stress and UPR signaling pathway |
[81] |
|
Runx1 |
Runx1-deficient HSPCs leads to lower p53 levels, reduced apoptosis, an attenuated unfolded protein response, and accordingly are resistant to genotoxic and ER stress |
[72] |
|
SEL1L |
Downregulation of SEL1L synergy enhances VPA cytotoxic effects to GSCs proliferation and self-renewal properties through UPR and ER stress |
[133] |
|
SMN1 |
Inhibition of ER stress improves survival of SMA patient-derived induced pluripotent stem cells produced motor neurons |
[200] |
|
TAU |
FTD neurons shows an activation of the unfolded protein response, links to mutant TAU protein |
[103] |
|
UFBP1 |
UFBP1 deficiency leads to elevated ER stress and initiation of unfolded protein response (UPR), following causes cell death of hematopoietic stem/progenitor cells |
[71] |
|
Wfs1 |
Wfs1 might protect cells during neural differentiation via regulating UPR response in adult brain |
[53] |
|
XBP1 |
The transcription factor XBP1 is selectively required for eosinophil differentiation without inducing parallel endoplasmic reticulum (ER) stress signaling pathways |
[83] |
|
XBP1 |
ER stress transcription factor Xbp1 directs intestinal stem cells and suppresses intestinal tumorigenesis through restraint of a pathway that involves an Ire1α- and Stat3-mediated regenerative response of the epithelium |
[117] |
|
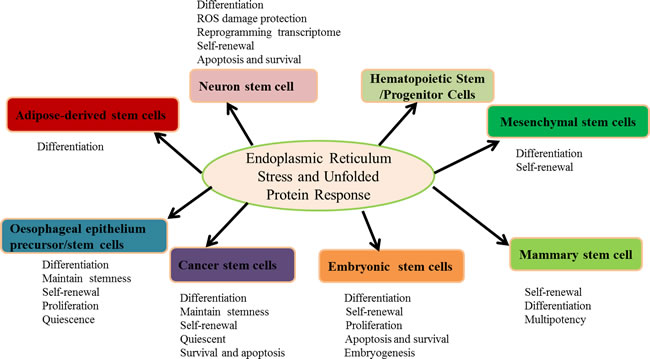
Figure 1: The roles of ER stress and the UPR in stem cells. The activation of ER stress and the UPR are involved in differentiation, self-renewal, proliferation, survival and apoptosis to maintain multipotency. Furthermore, ER stress and the UPR govern the stem cell pool integrity and initiate adaptive responses to the hypoxic niche, regulate mitochondrial potential, and reprogram the transcriptome.
Acknowledgments
This work was partially supported by the Ningxia higher school science and technology research project (NGY2015093), the Natural Science Foundation of China (81560259), and the CUHK One-off Funding for Joint Lab/Research Collaboration, Projects 3132970 and 3132973.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
References
1. Kaufman RJ. Stress signaling from the lumen of the endoplasmic reticulum: coordination of gene transcriptional and translational controls. Genes Dev. 1999; 13:1211-1233.
2. Koumenis C. ER stress, hypoxia tolerance and tumor progression. Curr Mol Med. 2006; 6:55-69.
3. Kaufman RJ. Orchestrating the unfolded protein response in health and disease. J Clin Invest. 2002; 110:1389-1398.
4. Xu C, Bailly-Maitre B, Reed JC. Endoplasmic reticulum stress: cell life and death decisions. J Clin Invest 2005; 115:2656-2664.
5. Yang ES, Bae JY, Kim TH, Kim YS, Suk K, Bae YC. Involvement of endoplasmic reticulum stress response in orofacial inflammatory pain. Exp Neurobiol. 2014; 23:372-380.
6. Claudio Hetz. Protein Misfolding Disorders: A Trip into the ER. www.benthamscience.com/ebooks/9781608050130
7. Zhang JY, Lee KS, Kim JS, Song BS, Jin DI, Koo DB, Yu K. Functional characterization of the ER stress induced X-box-binding protein-1(Xbp-1) in the porcine system. BMC Mol Biol. 2011; 12:25.
8. Yang Y, Pei X, Jin Y, Wang Y, Zhang C. The roles of endoplasmic reticulum stress response in female mammalian reproduction. Cell Tissue Res. 2016; 363: 589-597.
9. Oh J, Riek AE, Weng S, Petty M, Kim D, Colonna M, Cella M, Bernal-Mizrachi C. Endoplasmic reticulum stress controls M2 macrophage differentiation and foam cell formation. J Biol Chem. 2012; 287:11629-11641.
10. Sheveleva EV, Landowski TH, Samulitis BK, Bartholomeusz G, Powis G, Dorr RT. Imexon induces an oxidative endoplasmic reticulum stress response in pancreatic cancer cells. Mol Cancer Res. 2012; 10:392-400.
11. Tersey SA, Nishiki Y, Templin AT, Cabrera SM, Stull ND, Colvin SC. Islet -cell endoplasmic reticulum stress precedes the onset of type 1 diabetes in the nonobese diabetic mouse model. Diabetes. 2012; 61:818-827.
12. Hitomi J, Katayama T, Eguchi Y, Kudo T, Taniguchi M, Koyama Y, Manabe T, Yamagishi S, Bando Y, Imaizumi K, Tsujimoto Y, Tohyama M. Involvement of caspase-4 in endoplasmic reticulum stress-induced apoptosis and Abeta-induced cell death. J Cell Biol. 2014; 165:347-356.
13. Li B, Yi P, Zhang B, Xu C, Liu Q, Pi Z, Xu X, Chevet E, Liu J. Differences in endoplasmic reticulum stress signalling kinetics determine cell survival outcome through activation of MKP-1. Cell Signal. 2011; 23:35-45.
14. Siman R, Flood DG, Thinakaran G, Neumar RW. Endoplasmic reticulum stress induced cysteine protease activation in cortical neurons: effect of an Alzheimer’s disease-linked presenilin-1 knock-in mutation. J Biol Chem. 2001; 276: 44736-44743.
15. Szegezdi E, Logue SE, Gorman AM, Samali A. Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep. 2006; 7:880-885.
16. Urano F, Wang X, Bertolotti A, Zhang Y, Chung P, Harding HP, Ron D. Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1. Science. 2000; 287:664-666.
17. Fujita E, Kouroku Y, Isoai A, Kumagai H, Misutani A, Matsuda C, Hayashi YK, Momoi T. Two endoplasmic reticulum-associated degradation (ERAD) systems for the novel variant of the mutant dysferlin: ubiquitin/proteasome ERAD(I) and autophagy/lysosome ERAD(II). Hum Mol Genet. 2007; 16:618-629.
18. Nakatsukasa K, Kamura T, Brodsky JL. Recent technical developments in the study of ER-associated degradation. Curr Opin Cell Biol. 2014; 29:82-91.
19. Hetz C, Thielen P, Matus S, Nassif M, Court F, Kiffin R, Martinez G, Cuervo AM, Brown RH, Glimcher LH. XBP-1 deficiency in the nervous system protects against amyotrophic lateral sclerosis by increasing autophagy. Genes Dev. 2009; 23:2294-2306.
20. Guan JL, Simon AK, Prescott M, Menendez JA, Liu F, Wang F, Wang C, Wolvetang E, Vazquez-Martin A, Zhang J. Autophagy in stem cells. Autophagy. 2013; 9:830-849.
21. Rodolfo C, Di Bartolomeo S, Cecconi F. Autophagy in stem and progenitor cells. Cell Mol Life Sci. 2016; 73:475-496.
22. Wang S, Xia P, Rehm M, Fan Z (2015) Autophagy and cell reprogramming. Cell Mol Life Sci. 2016; 72:1699-1713.
23. Phadwal K, Watson AS, Simon AK. Tightrope act: autophagy in stem cell renewal, differentiation, proliferation, and aging. Cell Mol Life Sci. 2013; 70:89-103.
24. Vessoni AT, Muotri AR, Okamoto OK. Autophagy in stem cell maintenance and differentiation. Stem Cells Dev. 2012; 21:513-520.
25. Antonucci L, Fagman JB, Kim JY, Todoric J, Gukovsky I, Mackey M, Ellisman MH, Karin M. Basal autophagy maintains pancreatic acinar cell homeostasis and protein synthesis and prevents ER stress. Proc Natl Acad Sci U S A. 2015; 112:E6166-6174.
26. Bhardwaj M, Paul S, Jakhar R, Kang SC. Potential role of vitexin in alleviating heat stress-induced cytotoxicity: Regulatory effect of Hsp90 on ER stress-mediated autophagy. Life Sci. 2015; 142:36-48.
27. Jung TW, Hong HC, Hwang HJ, Yoo HJ, Baik SH, Choi KM. C1q/TNF-Related Protein 9 (CTRP9) attenuates hepatic steatosis via the autophagy-mediated inhibition of endoplasmic reticulum stress. Mol Cell Endocrinol. 2015; 417:131-140.
28. Yuzefovych LV, Musiyenko SI, Wilson GL, Rachek LI. Mitochondrial DNA damage and dysfunction, and oxidative stress are associated with endoplasmic reticulum stress, protein degradation and apoptosis in high fat diet-induced insulin resistance mice. PLoS One. 2013; 8:e54059.
29. Chou CT, Lin HT, Hwang PA, Wang ST, Hsieh CH, Hwang DF. Taurine resumed neuronal differentiation in arsenite-treated N2a cells through reducing oxidative stress, endoplasmic reticulum stress, and mitochondrial dysfunction. Amino Acids. 2015; 47:735-744.
30. Park Y. Oxidative stress, mitochondrial dysfunction and endoplasmic reticulum stress. Bio Design. 2014; 1:1-20.
31. Malhotra JD, Kaufman RJ. Endoplasmic reticulum stress and oxidative stress: a vicious cycle or a double-edged sword? Antioxid Redox Signal. 2007; 9:2277-2293.
32. Lee J, Giordano S, Zhang J. Autophagy, mitochondria and oxidative stress: cross-talk and redox signalling. Biochem J. 2012; 441:523-540.
33. Lemasters JJ. Selective mitochondrial autophagy, or mitophagy, as a targeted defense against oxidative stress, mitochondrial dysfunction, and aging. Rejuvenation Res. 2005; 8:3-5.
34. Mohrin M, Shin J, Liu Y, Brown K, Luo H, Xi Y, Haynes CM, Chen D. Stem cell aging. A mitochondrial UPR-mediated metabolic checkpoint regulates hematopoietic stem cell aging. Science. 2015; 347:1374-1377.
35. Velarde MC, Demaria M, Melov S, Campisi J. Pleiotropic age-dependent effects of mitochondrial dysfunction on epidermal stem cells. Proc Natl Acad Sci U S A. 2015; 112:10407-10412.
36. Lages YM, Nascimento JM, Lemos GA, Galina A, Castilho LR, Rehen SK. Low oxygen alters mitochondrial function and response to oxidative stress in human neural progenitor cells. Peer J. 2015; 3:e1486.
37. Mohrin M, Chen D. The mitochondrial metabolic checkpoint and aging of hematopoietic stem cells. Curr Opin Hematol. 2016; In press.
38. Vazin T, Freed WJ. Human embryonic stem cells: derivation, culture, and differentiation: a review. Restor Neurol Neurosci. 2010; 28:589-603.
39. Kim JS, Song BS, Lee KS, Kim DH, Kim SU, Choo YK, Chang KT, Koo DB. Tauroursodeoxycholic acid enhances the pre-implantation embryo development by reducing apoptosis in pigs. Reprod Domest Anim. 2012; 47:791-798.
40. Zhang JY, Diao YF, Kim HR, Jin DI. Inhibition of endoplasmic reticulum stress improves mouse embryo development. PLoS One. 2012; 7:e40433.
41. Zhang JY, Diao YF, Oqani RK, Han RX, Jin DI. Effect of endoplasmic reticulum stress on porcine oocyte maturation and parthenogenetic embryonic development in vitro. Biol Reprod. 2012; 86:128.
42. Lin T, Diao YF, Kang JW, Lee JE, Kim DK, Jin DI. Tauroursodeoxycholic acid improves the implantation and live-birth rates of mouse embryos. Reprod Biol. 2015; 15:101-105.
43. Latham KE. Endoplasmic reticulum stress signaling in mammalian oocytes and embryos: life in balance. Int Rev Cell Mol Biol. 2015; 316:227-265.
44. Basar M, Bozkurt I, Guzeloglu-Kayisli O, Sozen B, Tekmen I, Schatz F, Arici A, Lockwood CJ, Kayisli UA. Unfolded protein response prevents blastocyst formation during preimplantation embryo development in vitro. Fertil Steril. 2014; 102:1777-1784.
45. Michalak M, Gye MC. Endoplasmic reticulum stress in periimplantation embryos. Clin Exp Reprod Med. 2015; 42:1-7.
46. Simon MC, Keith B. The role of oxygen availability in embryonic development and stem cell function. Nat Rev Mol Cell Biol. 2008; 9:285-296.
47. Tagliavacca L, Caretti A, Bianciardi P, Samaja M. In vivo up-regulation of the unfolded protein response after hypoxia. Biochim Biophys Acta. 2012; 1820:900-906.
48. Tsang KY, Chan D, Bateman JF, Cheah KS. In vivo cellular adaptation to ER stress: survival strategies with double-edged consequences. J Cell Sci. 2010; 123:2145-2154.
49. Song BS, Yoon SB, Kim JS, Sim BW, Kim YH, Cha JJ, Choi SA, Min HK, Lee Y, Huh JW, Lee SR, Kim SH, Koo DB, Choo YK, Kim HM, Kim SU, Chang KT. Induction of autophagy promotes preattachment development of bovine embryos by reducing endoplasmic reticulum stress. Biol Reprod. 2012; 87:8, 1-11.
50. Mao C, Wang M, Luo B, Wey S, Dong D, Wesselschmidt R, Rawlings S, Lee AS. Targeted mutation of the mouse Grp94 gene disrupts development and perturbs endoplasmic reticulum stress signaling. PLoS One. 2010; 5:e10852.
51. Wang DY, Abbasi C, El-Rass S, Li JY, Dawood F, Naito K, Sharma P, Bousette N, Singh S, Backx PH, Cox B, Wen XY, Liu PP, Gramolini AO. Endoplasmic reticulum resident protein 44 (ERp44) deficiency in mice and zebrafish leads to cardiac developmental and functional defects. J Am Heart Assoc. 2014; 3:e001018.
52. Luo S, Mao C, Lee B, Lee AS. GRP78/BiP is required for cell proliferation and protecting the inner cell mass from apoptosis during early mouse embryonic development. Mol Cell Biol. 2006; 26:5688-5697.
53. Tekko T, Lilleväli K, Luuk H, Sütt S, Truu L, Örd T, Möls M, Vasar E. Initiation and developmental dynamics of Wfs1 expression in the context of neural differentiation and ER stress in mouse forebrain. Int J Dev Neurosci. 2014; 35:80-88.
54. Zhu G, Lee AS. Role of the unfolded protein response, GRP78 and GRP94 in organ homeostasis. J Cell Physiol. 2015; 230:1413-1420.
55. Cho YM, Jang YS, Jang YM, Chung SM, Kim HS, Lee JH, Jeong SW, Kim IK, Kim JJ, Kim KS, Kwon OJ. Induction of unfolded protein response during neuronal induction of rat bone marrow stromal cells and mouse embryonic stem cells. Exp Mol Med. 2009; 41:440-452.
56. Liu L, Liu C, Zhong Y, Apostolou A, Fang S. ER stress response during the differentiation of H9 cells induced by retinoic acid. Biochem Biophys Res Commun. 2012; 417:738-743.
57. Xu H, Tsang KS, Wang Y, Chan JC, Xu G, Gao WQ. Unfolded protein response is required for the definitive endodermal specification of mouse embryonic stem cells via Smad2 and -catenin signaling. J Biol Chem. 2014; 289:26290-26301.
58. Wang X, Karamariti E, Simpson R, Wang W, Xu Q. Dickkopf Homolog 3 Induces Stem Cell Differentiation into Smooth Muscle Lineage via ATF6 Signalling. J Biol Chem. 2015; 290:19844-19852.
59. Chen G, Xu X, Zhang L, Fu Y, Wang M, Gu H, Xie X. Blocking autocrine VEGF signaling by sunitinib, an anti-cancer drug, promotes embryonic stem cell self-renewal and somatic cell reprogramming. Cell Res. 2014; 24:1121-1136.
60. Miyake N, Brun AC, Magnusson M, Miyake K, Scadden DT, Karlsson S. HOXB4-induced self-renewal of hematopoietic stem cells is significantly enhanced by p21 deficiency. Stem Cells. 2006; 24:653-661.
61. Blanco-Gelaz MA, Suarez-Alvarez B, Ligero G, Sanchez L, Vidal-Castiñeira JR, Coto E, Moore H, Menendez P, Lopez-Larrea C. Endoplasmic reticulum stress signals in defined human embryonic stem cell lines and culture conditions. Stem Cell Rev. 2010; 6:462-472.
62. Eliasson P, Jonsson JI. The hematopoietic stem cell niche: low in oxygen but a nice place to be. J Cell Physiol. 2010; 222:17-22.
63. Geiger H, de Haan G, Florian MC. The ageing haematopoietic stem cell compartment. Nature Rev Immunol. 2013; 13:376-389.
64. Yahata T, Takanashi T, Muguruma Y, Ibrahim AA, Matsuzawa H, Uno T, Sheng Y, Onizuka M, Ito M, Kato S, Ando K. Accumulation of oxidative DNA damage restricts the self-renewal capacity of human hematopoietic stem cells. Blood. 2011; 118:2941-2950.
65. Shlush LI, Zandi S, Mitchell A, Chen WC, Brandwein JM, Gupta V, Kennedy JA, Schimmer AD, Schuh AC, Yee KW, McLeod JL, Doedens M, Medeiros JJ, Marke R, Kim HJ, Lee K, McPherson JD, Hudson TJ, HALT Pan-Leukemia Gene Panel Consortium, Brown AM, Yousif F, Trinh QM, Stein LD, Minden MD, Wang JC, Dick JE. Identification of pre-leukaemic haematopoietic stem cells in acute leukaemia. Nature. 2014; 506:328-333.
66. Rossi D J, Jamieson C.HM, Weissman IL. Stems cells and the pathways to aging and cancer. Cell. 2008; 132:681-696.
67. Kharabi Masouleh B, Chevet E, Panse J, Jost E, O’Dwyer M, Bruemmendorf TH, Samali A. Drugging the unfolded protein response in acute leukemias. J Hematol Oncol. 2015; 8:87.
68. van Galen P, Kreso A, Mbong N, Kent DG, Fitzmaurice T, Chambers JE, Xie S, Laurenti E, Hermans K, Eppert K, Marciniak SJ, Goodall JC, Green AR, Wouters BG, Wienholds E, Dick JE. The unfolded protein response governs integrity of the haematopoietic stem-cell pool during stress. Nature. 2014; 510:268-272.
69. Shin SY, Lee JM, Lee MS, Koh D, Jung H, Lim Y, Lee YH. Targeting cancer cells via the reactive oxygen species-mediated unfolded protein response with a novel synthetic polyphenol conjugate. Clin Cancer Res. 2014; 20:4302-4313.
70. Ozgur R, Turkan I, Uzilday B, Sekmen AH. Endoplasmic reticulum stress triggers ROS signalling, changes the redox state, and regulates the antioxidant defence of Arabidopsis thaliana. J Exp Bot. 2014; 65:1377-1390.
71. Cai Y, Pi W, Sivaprakasam S, Zhu X, Zhang M, Chen J, Makala L, Lu C, Wu J, Teng Y, Pace B, Tuan D, Singh N, Li H. UFBP1, a Key Component of the Ufm1 Conjugation System, Is Essential for Ufmylation-Mediated Regulation of Erythroid Development. PLoS Genet. 2015; 11:e1005643.
72. Cai X, Gao L, Teng L, Ge J, Oo ZM, Kumar AR, Gilliland DG, Mason PJ, Tan K, Speck NA. Runx1 Deficiency Decreases Ribosome Biogenesis and Confers Stress Resistance to Hematopoietic Stem and Progenitor Cells. Cell Stem Cell. 2015; 17:165-177.
73. Miharada K, Karlsson G, Rehn M, Rörby E, Siva K, Cammenga J, Karlsson S. Cripto regulates hematopoietic stem cells as a hypoxic-niche-related factor through cell surface receptor GRP78. Cell Stem Cell. 2011; 9:330-344.
74. Klauzinska M, Castro NP, Rangel MC, Spike BT, Gray PC, Bertolette D, Cuttitta F, Salomon D. The multifaceted role of the embryonic gene Cripto-1 in cancer, stem cells and epithelial-mesenchymal transition. Semin Cancer Biol. 2014; 29:51-58.
75. Bravo R, Gutierrez T, Paredes F, Gatica D, Rodriguez AE, Pedrozo Z, Chiong M, Parra V, Quest AF, Rothermel BA, Lavandero S. Endoplasmic reticulum: ER stress regulates mitochondrial bioenergetics. Int J Biochem Cell Biol 2012; 44:16-20.
76. Yuzefovych LV, LeDoux SP, Wilson GL, Rachek LI. Mitochondrial DNA damage via augmented oxidative stress regulates endoplasmic reticulumstress and autophagy: crosstalk, links and signaling. PLoS One. 2013; 8:e83349.
77. Miharada K, Karlsson G, Rehn M, Rörby E, Siva K, Cammenga J, Karlsson S. Hematopoietic stem cells are regulated by Cripto, as an intermediary of HIF-1 in the hypoxic bone marrow niche. Ann N Y Acad Sci. 2012; 1266:55-62.
78. Wey S, Luo B, Lee AS. Acute inducible ablation of GRP78 reveals its role in hematopoietic stem cell survival, lymphogenesis and regulation of stress signaling. PLoS One. 2012; 7:e39047.
79. Miharada K, Sigurdsson V, Karlsson S. Dppa5 improves hematopoietic stem cell activity by reducing endoplasmic reticulum stress. Cell Rep. 2014; 7:1381-1392.
80. Rouault-Pierre K, Lopez-Onieva L, Foster K, Anjos-Afonso F, Lamrissi-Garcia I, Serrano-Sanchez M, Mitter R, Ivanovic Z, de Verneuil H, Gribben J, Taussig D, Rezvani HR, Mazurier F, Bonnet D. HIF-2 protects human hematopoietic stem/progenitors and acute myeloid leukemic cells from apoptosis induced by endoplasmic reticulum stress. Cell Stem Cell. 2013; 13:549-563.
81. Zhang M, Zhu X, Zhang Y, Cai Y, Chen J, Sivaprakasam S, Gurav A, Pi W, Makala L, Wu J, Pace B, Tuan-Lo D, Ganapathy V, Singh N, Li H. RCAD/Ufl1, a Ufm1 E3 ligase, is essential for hematopoietic stem cell function and murine hematopoiesis. Cell Death Differ. 2015; 22:1922-1934.
82. Masuoka HC, Townes TM. Targeted disruption of the activating transcription factor 4 gene results in severe fetal anemia in mice. Blood. 2002; 99:736-745.
83. Bettigole SE, Lis R, Adoro S, Lee AH, Spencer LA, Weller PF, Glimcher LH. The transcription factor XBP1 is selectively required for eosinophil differentiation. Nat Immunol. 2015; 16:829-837.
84. Dale DC, Person RE, Bolyard AA, Aprikyan AG, Bos C, Bonilla MA, Boxer LA, Kannourakis G, Zeidler C, Welte K, Benson KF, Horwitz M. Mutations in the gene encoding neutrophil elastase in congenital and cyclic neutropenia. Blood. 2000; 96:2317-2322.
85. Horwitz M, Benson KF, Person RE, Aprikyan AG, Dale DC. Mutations in ELA2, encoding neutrophil elastase, define a 21-day biological clock in cyclic haematopoiesis. Nat Genet. 1999; 23:433-436.
86. Nayak RC, Trump LR, Aronow BJ, Myers K, Mehta P, Kalfa T, Wellendorf AM, Valencia CA, Paddison PJ, Horwitz MS, Grimes HL, Lutzko C, Cancelas JA. Pathogenesis of ELANE-mutant severe neutropenia revealed by induced pluripotent stem cells. J Clin Invest. 2015; 125:3103-3116.
87. Lin YF, Haynes CM. Metabolism and the UPR(mt). Mol Cell. 2016; 61:677-682.
88. Sun N, Youle RJ, Finkel T. The Mitochondrial Basis of Aging. Mol Cell. 2016; 61:654-66
89. Placido AI, Pereira CM, Duarte AI, Candeias E, Correia SC, Carvalho C, Cardoso S, Oliveira CR, Moreira PI. Modulation of endoplasmic reticulum stress: an opportunity to prevent neurodegeneration? CNS Neurol Disord Drug Targets. 2015; 14:518-533.
90. Penke B, Ferenc B, Fülöp L. Protein Folding and Misfolding, Endoplasmic Reticulum Stress in Neurodegenerative Diseases: in Trace of Novel Drug Targets. Curr Protein Pept Sci. 2016; 17:169-182.
91. Shah SZ, Zhao D, Khan SH, Yang L. Unfolded Protein Response Pathways in Neurodegenerative Diseases. J Mol Neurosci. 2015;57:529-537.
92. Scheper W, Hoozemans JJ. The unfolded protein response in neurodegenerative diseases: a neuropathological perspective. Acta Neuropathol 2015; 130: 315-331.
93. Laguesse S, Creppe C, Nedialkova DD, Prévot PP, Borgs L, Huysseune S, Franco B, Duysens G, Krusy N, Lee G, Thelen N, Thiry M, Close P, Chariot A, Malgrange B, Leidel SA, Godin JD, Nguyen L. A Dynamic Unfolded Protein Response Contributes to the Control of Cortical Neurogenesis. Dev Cell. 2015; 35:553-567.
94. Naughton MC, McMahon JM, FitzGerald U. Differential activation of ER stress pathways in myelinating cerebellar tracts. Int J Dev Neurosci. 2015; 47:347-360.
95. Wang H, Wang X, Ke ZJ, Comer AL, Xu M, Frank JA, Zhang Z, Shi X, Luo J. Tunicamycin-induced unfolded protein response in the developing mouse brain. Toxicol Appl Pharmacol. 2015; 283:157-167.
96. Kawada K, Iekumo T, Saito R, Kaneko M, Mimori S, Nomura Y, Okuma Y. Aberrant neuronal differentiation and inhibition of dendrite outgrowth resulting from endoplasmic reticulum stress. J Neurosci Res. 2014; 92:1122-1133.
97. Rzechorzek NM, Connick P, Patani R, Selvaraj BT, Chandran S. Hypothermic Preconditioning of Human Cortical Neurons Requires Proteostatic Priming. EBioMedicine. 2015; 2:528-535.
98. Su Y, Li F. Endoplasmic reticulum stress in brain ischemia. Int J Neurosci. 2015; 20:1-11.
99. Roidl D, Hellbach N, Bovio PP, Villarreal A, Heidrich S, Nestel S, Grüning BA, Boenisch U, Vogel T. DOT1L Activity Promotes Proliferation and Protects Cortical Neural Stem Cells from Activation of ATF4-DDIT3-Mediated ER Stress In Vitro. Stem Cells. 2016; 34:233-245.
100. Mansouri S, Barde S, Ortsäter H, Eweida M, Darsalia V, Langel U, Sjöholm A, Hökfelt T, Patrone C. GalR3 activation promotes adult neural stem cell survival in response to a diabetic milieu. J Neurochem. 2013; 127:209-220.
101. Barateiro A, Vaz AR, Silva SL, Fernandes A, Brites D. ER stress, mitochondrial dysfunction and calpain/JNK activation are involved in oligodendrocyte precursor cell death by unconjugated bilirubin. Neuromolecular Med. 2012; 14:285-302.
102. Nakamura H, Liao H, Minami K, Toyoda M, Akutsu H, Miyagawa Y, Okita H, Kiyokawa N, Umezawa A, Imadome K, Inoue N, Fujiwara S. Human cytomegalovirus induces apoptosis in neural stem/progenitor cells derived from induced pluripotent stem cells by generating mitochondrial dysfunction and endoplasmic reticulum stress. Herpesviridae. 2013; 4:2.
103. Ehrlich M, Hallmann AL, Reinhardt P, Araúzo-Bravo MJ, Korr S, Röpke A, Psathaki OE, Ehling P, Meuth SG, Oblak AL, Murrell JR, Ghetti B, Zaehres H, Schöler HR, Sterneckert J, Kuhlmann T, Hargus G. Distinct Neurodegenerative Changes in an Induced Pluripotent Stem Cell Model of Frontotemporal Dementia Linked to Mutant TAU Protein. Stem Cell Reports.2015; 5:83-96.
104. Tucker BA, Mullins RF, Streb LM, Anfinson K, Eyestone ME, Kaalberg E, Riker MJ, Drack AV, Braun TA, Stone EM. Patient-specific iPSC-derived photoreceptor precursor cells as a means to investigate retinitis pigmentosa. Elife. 2013; 2:e00824.
105. Doupé DP, Alcolea MP, Roshan A, Zhang G, Klein AM, Simons BD, Jones PH. A single progenitor population switches behavior to maintain and repair esophageal epithelium. Science. 2012; 337:1091-1093.
106. Seery JP. Stem cells of the oesophageal epithelium. J Cell Sci. 2002; 115:1783-1789.
107. Amici AW, Onikoyi FO, Bonfanti P. Lineage potential, plasticity and environmental reprogramming of epithelial stem/progenitor cells. Biochem Soc Trans. 2014; 42:637-644.
108. Alcolea MP, Greulich P, Wabik A, Frede J, Simons BD, Jones PH. Differentiation imbalance in single oesophageal progenitor cells causes clonal immortalization and field change. Nat Cell Biol. 2014; 16:615-622.
109. Jeong Y, Rhee H, Martin S, Klass D, Lin Y, Nguyen LX, Feng W, Diehn M. Identification and genetic manipulation of human and mouse oesophageal stem cells. Gut. 2015; in press.
110. Rosekrans SL, Heijmans J, Büller NV, Westerlund J, Lee AS, Muncan V, van den Brink GR. ER stress induces epithelial differentiation in the mouse oesophagus. Gut. 2015; 64:195-202.
111. Wang F, Scoville D, He XC, Mahe MM, Box A, Perry JM, Smith NR, Lei NY, Davies PS, Fuller MK, Haug JS, McClain M, Gracz AD, Ding S, Stelzner M, Dunn JC, Magness ST, Wong MH, Martin MG, Helmrath M, Li L. Isolation and characterization of intestinal stem cells based on surface marker combinations and colony-formation assay. Gastroenterology. 2013; 145:383-395.
112. Heijmans J, van Lidth de Jeude JF, Koo BK, Rosekrans SL, Wielenga MC, van de Wetering M, Ferrante M, Lee AS, Onderwater JJ, Paton JC, Paton AW, Mommaas AM, Kodach LL, Hardwick JC, Hommes DW, Clevers H, Muncan V, van den Brink GR. ER stress causes rapid loss of intestinal epithelial stemness through activation of the unfolded protein response. Cell Rep. 2013; 3:1128-1139.
113. Guo FJ, Jiang R, Xiong Z, Xia F, Li M, Chen L, Liu CJ. IRE1a constitutes a negative feedback loop with BMP2 and acts as a novel mediator in modulating osteogenic differentiation. Cell Death Dis. 2014; 5:e1239.
114. Wang L, Zeng X, Ryoo HD, Jasper H. Integration of UPRER and oxidative stress signaling in the control of intestinal stem cell proliferation. PLoS Genet. 2014; 10:e1004568.
115. Yang Z, Yao JF, Xu P, Zhang JB, Zhang YM, Zhu YJ, Qin SQ, Liu L, Liu H, Hou WK, Xu K. Functions and mechanisms of intermittent negative pressure for osteogenesis in human bone marrow mesenchymal stem cells. Mol Med Rep. 2014; 9:1331-1336.
116. Qiu W, Wang X, Buchanan M, He K, Sharma R, Zhang L, Wang Q, Yu J. ADAR1 is essential for intestinal homeostasis and stem cell maintenance. Cell Death Dis. 2013; 4:e599.
117. Niederreiter L, Fritz TM, Adolph TE, Krismer AM, Offner FA, Tschurtschenthaler M, Flak MB, Hosomi S, Tomczak MF, Kaneider NC, Sarcevic E, Kempster SL, Raine T, Esser D, Rosenstiel P, Kohno K, Iwawaki T, Tilg H, Blumberg RS, Kaser A. ER stress transcription factor Xbp1 suppresses intestinal tumorigenesis and directs intestinal stem cells. J Exp Med. 2013; 210:2041-2056.
118. Wang L, Ryoo HD, Qi Y, Jasper H. PERK Limits Drosophila Lifespan by Promoting Intestinal Stem Cell Proliferation in Response to ER Stress. PLoS Genet. 2015; 11:e1005220.
119. Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, Cancer, and cancer stem cells. Nature. 2001; 414:105-111.
120. Yamashita T, Wang XW. Cancer stem cells in the development of liver cancer. J Clin Invest. 2013; 123:1911-1918.
121. Peng CY, Fong PC, Yu CC, Tsai WC, Tzeng YM, Chang WW. Methyl Antcinate A suppresses the population of cancer stem-like cells in MCF7 human breast cancer cell line. Molecules. 2013; 18:2539-2348.
122. Dhawan P, Ahmad R, Srivastava AS, Singh AB. Cancer stem cells and colorectal cancer: An overview. Curr Top Med Chem. 2011; 11:1592-1598.
123. Colak S, Medema JP. Cancer stem cells important players in tumor therapy resistance. FEBS J. 2014; 281:4779-4791.
124. Kemper K, Grandela C, Medema JP. Molecular identification and targeting of colorectal cancer stem cells. Oncotarget. 2010; 1:387-395. doi: 10.18623/oncotarget.173.
125. Rich JN, Bao S. Chemotherapy and cancer stem cells. Cell Stem Cell. 2007; 1:353-355.
126. Valent P, Bonnet D, De Maria R, Lapidot T, Copland M, Melo JV, Chomienne C, Ishikawa F, Schuringa JJ, Stassi G, Huntly B, Herrmann H, Soulier J, Roesch A, Schuurhuis GJ, Wöhrer S, Arock M, Zuber J, Cerny-Reiterer S, Johnsen HE, Andreeff M, Eaves C. Cancer stem cell definitions and terminology: the devil is in the details. Nat Rev Cancer. 2012; 12:767-775.
127. Zeuner A, Todaro M, Stassi G, De Maria R. Colorectal cancer stem cells: from the crypt to the clinic. Cell Stem Cell. 2014; 15:692-705.
128. Wielenga MC, Colak S, Heijmans J, van Lidth de Jeude JF, Rodermond HM, Paton JC, Paton AW, Vermeulen L, Medema JP, van den Brink GR. ER-Stress-Induced Differentiation Sensitizes Colon Cancer Stem Cells to Chemotherapy. Cell Rep. 2015; 13:489-494.
129. Joly AL, Deepti A, Seignez A, Goloudina A, Hebrard S, Schmitt E, Richaud S, Fourmaux E, Hammann A, Collura A, Svrcek M, Jego G, Robinet E, Solary E, Demidov O, Kohli E, Garrido C. The HSP90 inhibitor, 17AAG, protects the intestinal stem cell niche and inhibits graft versus host disease development. Oncogene. 2015; in press.
130. Li YC, Chang JT, Chiu C, Lu YC, Li YL, Chiang CH, You GR, Lee LY, Cheng AJ. Areca nut contributes to oral malignancy through facilitating the conversion of cancer stem cells. Mol Carcinog. 2016; 55:1012-1023.
131. Chiu CC, Lee LY, Li YC, Chen YJ, Lu YC, Li YL, Wang HM, Chang JT, Cheng AJ. Grp78 as a therapeutic target for refractory head-neck cancer with CD24(-)CD44(+) stemness phenotype. Cancer Gene Ther. 2013; 20:606-615.
132. Liu J, Wang P, Xue YX, Li Z, Qu CB, Liu YH. Enhanced antitumor effect of shikonin by inhibiting Endoplasmic Reticulum Stress via JNK/c-Jun pathway in human glioblastoma stem cells. Biochem Biophys Res Commun. 2015;466:103-110.
133. Cattaneo M, Baronchelli S, Schiffer D, Mellai M, Caldera V, Saccani GJ, Dalpra L, Daga A, Orlandi R, DeBlasio P, Biunno I. Down-modulation of SEL1L, an unfolded protein response and endoplasmic reticulum-associated degradation protein, sensitizes glioma stem cells to the cytotoxic effect of valproic acid. J Biol Chem. 2014; 289:2826-2838.
134. Gargiulo G, Cesaroni M, Serresi M, de Vries N, Hulsman D, Bruggeman SW, Lancini C, van Lohuizen M. In vivo RNAi screen for BMI1 targets identifies TGF-/BMP-ER stress pathways as key regulators of neural- and malignant glioma-stem cell homeostasis. Cancer Cell. 2013; 23:660-676.
135. Peng Y, Li Z, Li Z. GRP78 secreted by tumor cells stimulates differentiation of bone marrow mesenchymal stem cells to cancer-associated fibroblasts. Biochem Biophys Res Commun. 2013; 440:558-563.
136. Bartkowiak K, Effenberger KE, Harder S, Andreas A, Buck F, Peter-Katalinic J, Pantel K, Brandt BH. Discovery of a novel unfolded protein response phenotype of cancer stem/progenitor cells from the bone marrow of breast cancer patients. J Proteome Res. 2010; 9:3158-3168.
137. Chen YH, McGowan LD, Cimino PJ, Dahiya S, Leonard JR, Lee da Y, Gutmann DH. Mouse low-grade gliomas contain cancer stem cells with unique molecular and functional properties. Cell Rep. 2015; 10:1899-1912.
138. Lee AS. Glucose-regulated proteins in cancer: molecular mechanisms and therapeutic potential. Nat Rev Cancer 2014; 14:263-276.
139. Jamora C, Dennert G, Lee AS. Inhibition of tumor progression by suppression of stress protein GRP78/BiP induction in fibrosarcoma B/C10ME. Proc Natl Acad Sci U S A. 1996; 93:7690-7694.
140. Liu R, Li X, Gao W, Zhou Y, Wey S, Mitra SK, Krasnoperov V, Dong D, Liu S, Li D, Zhu G, Louie S, Conti PS, Li Z, Lee AS, Gill PS. Monoclonal antibody against cell surface GRP78 as a novel agent in suppressing PI3K/AKT signaling, tumor growth, and metastasis. Clin Cancer Res. 2013; 19:6802-6811.
141. Wang N, Wang Z, Peng C, You J, Shen J, Han S, Chen J. Dietary compound isoliquiritigenin targets GRP78 to chemosensitize breast cancer stem cells via -catenin/ABCG2 signaling. Carcinogenesis. 2014; 35:2544-2554.
142. Mo L, Bachelder RE, Kennedy M, Chen PH, Chi JT, Berchuck A, Cianciolo G, Pizzo SV. Syngeneic Murine Ovarian Cancer Model Reveals That Ascites Enriches for Ovarian Cancer Stem-Like Cells Expressing Membrane GRP78. Mol Cancer Ther. 2015; 14:747-756.
143. Adomako A, Calvo V, Biran N, Osman K, Chari A, Paton JC, Paton AW, Moore K, Schewe DM, Aguirre-Ghiso JA. Identification of markers that functionally define a quiescent multiple myeloma cell sub-population surviving bortezomib treatment. BMC Cancer. 2015; 15:444.
144. Huynh TT, Lin CM, Lee WH, Wu AT, Lin YK, Lin YF, Yeh CT, Wang LS. Pterostilbene suppressed irradiation-resistant glioma stem cells by modulating GRP78/miR-205 axis. J Nutr Biochem. 2015; 26:466-475.
145. Li B, Cheng XL, Yang YP, Li ZQ. GRP78 mediates radiation resistance of a stem cell-like subpopulation within the MCF-7 breast cancer cell line. Oncol Rep. 2013; 30:2119-2126.
146. Tseng CN, Huang CF, Cho CL, Chang HW, Huang CW, Chiu CC, Chang YF. Brefeldin a effectively inhibits cancer stem cell-like properties and MMP-9 activity in human colorectal cancer Colo 205 cells. Molecules. 2013; 18:10242-10253.
147. Kataoka K, Sakaguchi M, Li KP, Taketa C, Yamamoto K, Du G, Funahashi H, Murata H, Huh NH. Internalization of REIC/Dkk-3 protein by induced pluripotent stem cell-derived embryoid bodies and extra-embryonic tissues. Int J Mol Med. 2010; 26:853-859.
148. Inman JL, Robertson C, Mott JD, Bissell MJ. Mammary gland development: cell fate specification, stem cells and the microenvironment. Development. 2015; 142:1028-1042.
149. Woodward WA, Chen MS, Behbod F, Rosen JM. Woodward WA, Chen MS, Behbod F, Rosen JM. On mammary stem cells. J Cell Sci. 2005; 118:3585-3594.
150. Spike BT, Kelber JA, Booker E, Kalathur M, Rodewald R, Lipianskaya J, La J, He M, Wright T, Klemke R, Wahl GM, Gray PC. CRIPTO/GRP78 signaling maintains fetal and adult mammary stem cells ex vivo. Stem Cell Reports. 2014; 2:427-439.
151. Klauzinska M, McCurdy D, Rangel MC, Vaidyanath A, Castro NP, Shen MM, Gonzales M, Bertolette D, Bianco C, Callahan R, Salomon DS, Raafat A. Cripto-1 Ablation Disrupts Alveolar Development in the Mouse Mammary Gland through a Progesterone Receptor-Mediated Pathway. Am J Pathol. 2015; 185:2907-2922.
152. Zhu G, Wang M, Spike B, Gray PC, Shen J, Lee SH, Chen SY, Lee AS. Differential requirement of GRP94 and GRP78 in mammary gland development. Sci Rep. 2014; 4:5390.
153. Pfaffenbach KT, Pong M, Morgan TE, Wang H, Ott K, Zhou B, Longo VD, Lee AS. GRP78/BiP is a novel downstream target of IGF-1 receptor mediated signaling. J Cell Physiol. 2012; 227:3803-3811.
154. Bunnell BA, Flaat M, Gagliardi C, Patel B, Ripoll C. Adipose-derived stem cells: Isolation, expansion and differentiation. Methods. 2008; 45:115-120.
155. Zuk PA, Zhu M, Ashjian P, De Ugarte DA, Huang JI, Mizuno H, Alfonso ZC, Fraser JK, Benhaim P, Hedrick MH. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002; 13:4279-4295.
156. Mizuno H. Adipose-derived Stem Cells for Tissue Repair and Regeneration: Ten Years of Research and a Literature Review. J Nippon Med Sch. 2009; 76:56-66.
157. Gimble JM, Guilak F. Adipose-derived adult stem cells: isolation, characterization, and differentiation potential. Cytotherapy. 2003; 5:362-369.
158. Guneta V, Loh QL, Choong C. Cell-secreted extracellular matrix formation and differentiation of adipose-derived stem cells in 3D alginate scaffolds with tunable properties. J Biomed Mater Res A. 2016; 104:1090-1101.
159. Cha BH, Kim JS, Ahn JC, Kim HC, Kim BS, Han DK, Park SG, Lee SH. The role of tauroursodeoxycholic acid on adipogenesis of human adipose-derived stem cells by modulation of ER stress. Biomaterials. 2014; 35:2851-2858.
160. Han J, Murthy R, Wood B, Song B, Wang S, Sun B, Malhi H, Kaufman RJ. ER stress signalling through eIF2 and CHOP, but not IRE1, attenuates adipogenesis in mice. Diabetologia. 2013; 56:911-924.
161. Mondal AK, Das SK, Varma V, Nolen GT, McGehee RE, Elbein SC, Wei JY, Ranganathan G. Effect of endoplasmic reticulum stress on inflammation and adiponectin regulation in human adipocytes. Metab Syndr Relat Disord. 2012; 10:297-306.
162. Kariminekoo S, Movassaghpour A, Rahimzadeh A, Talebi M, Shamsasenjan K, Akbarzadeh A. Implications of mesenchymal stem cells in regenerative medicine. Artif Cells Nanomed Biotechnol. 2016; 13:1-9.
163. Lu J, Dong HY, Lin LJ, Wang QH, Huang LH, Tan JM. Palmitate induces apoptosis and endoplasmic reticulum stress in human umbilical cord-derived mesenchymal stem cells. Sheng Li Xue Bao. 2013; 65:600-606.
164. King YA, Chiu YJ, Chen HP, Kuo DH, Lu CC, Yang JS. Endoplasmic reticulum stress contributes to arsenic trioxide-induced intrinsic apoptosis in human umbilical and bone marrow mesenchymal stem cells. Environ Toxicol. 2016; 31:314-328.
165. Li Z, Wei H, Liu X, Hu S, Cong X, Chen X. LPA rescues ER stress-associated apoptosis in hypoxia and serum deprivation-stimulated mesenchymal stem cells. J Cell Biochem. 2010; 111:811-820.
166. Wu ZZ, Li YH, Huang AC, Li M, Zhang XL, Wang JG, Yang M, Chen MY. Endoplasmic reticulum stress induced by tunicamycin and antagonistic effect of Tiantai No.1 (1) on mesenchymal stem cells. Chin J Integr Med. 2010; 16:41-49.
167. Lu J, Wang Q, Huang L, Dong H, Lin L, Lin N, Zheng F, Tan J. Palmitate causes endoplasmic reticulum stress and apoptosis in human mesenchymal stem cells: prevention by AMPK activator. Endocrinology. 2012; 153:5275-5284.
168. Styner M, Meyer MB, Galior K, Case N, Xie Z, Sen B, Thompson WR, Pike JW, Rubin J. Mechanical strain downregulates C/EBP in MSC and decreases endoplasmic reticulum stress. PLoS One. 2012; 7:e51613.
169. Xiong Z, Jiang R, Li X, Liu Y, Guo F. Different Roles of GRP78 on Cell Proliferation and Apoptosis in Cartilage Development. Int J Mol Sci. 2015; 16:21153-21176.
170. Wu Y, Yang M, Fan J, Peng Y, Deng L, Ding Y, Yang R, Zhou J, Miao D, Fu Q. Deficiency of osteoblastic Arl6ip5 impaired osteoblast differentiation and enhanced osteoclastogenesis via disturbance of ER calcium homeostasis and induction of ER stress-mediated apoptosis. Cell Death Dis. 2014; 5:e1464.
171. Zhang J, Weng Y, Liu X, Wang J, Zhang W, Kim SH, Zhang H, Li R, Kong Y, Chen X, Shui W, Wang N, Zhao C, Wu N, He Y, Nan G, Chen X, Wen S, Zhang H, Deng F, Wan L, Luu HH, Haydon RC, Shi LL, He TC, Shi Q. Endoplasmic reticulum (ER) stress inducible factor cysteine-rich with EGF-like domains 2 (Creld2) is an important mediator of BMP9-regulated osteogenic differentiation of mesenchymal stem cells. PLoS One. 2013; 8:e73086.
172. Guo FJ, Jiang R, Xiong Z, Xia F, Li M, Chen L, Liu CJ. IRE1a constitutes a negative feedback loop with BMP2 and acts as a novel mediator in modulating osteogenic differentiation. Cell Death Dis. 2014; 5:e1239.
173. Guo FJ, Jiang R, Li X, Zhang P, Han X, Liu C. Regulation of chondrocyte differentiation by IRE1 depends on its enzymatic activity. Cell Signal. 2014; 26:1998-2007.
174. Xiong Z, Jiang R, Zhang P, Han X, Guo FJ. Transmission of ER stress response by ATF6 promotes endochondral bone growth. J Orthop Surg Res. 2015; 10:141.
175. Chen Y, Gao H, Yin Q, Chen L, Dong P, Zhang X, Kang J. ER stress activating ATF4/CHOP-TNF- signaling pathway contributes to alcohol-induced disruption of osteogenic lineage of multipotential mesenchymal stem cell. Cell Physiol Biochem. 2013; 32:743-754.
176. Hsu YC, Fuchs E. A family business: stem cell progeny join the niche to regulate homeostasis. Nat Rev Mol Cell Biol. 2012; 13:103-114.
177. Spradling A, Drummond-Barbosa D, Kai T. Stem cells find their niche. Nature. 2001; 414:98-104.
178. Iancu CB, Iancu D, Renea I, Hostiuc S, Dermengiu D, Rusu MC. Molecular signatures of cardiac stem cells. Rom J Morphol Embryol. 2015; 56:1255-1262.
179. Nakanishi N, Takahashi T, Ogata T, Adachi A, Imoto-Tsubakimoto H, Ueyama T, Matsubara H. PARM-1 promotes cardiomyogenic differentiation through regulating the BMP/Smad signaling pathway. Biochem Biophys Res Commun. 2012; 428:500-505.
180. Singh VK, Kumar N, Kalsan M, Saini A, Chandra R. Mechanism of Induction: Induced Pluripotent Stem Cells (iPSCs). J Stem Cells. 2015; 10:43-62.
181. Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006; 126:663-676.
182. Qie S, Liang D, Yin C, Gu W, Meng M, Wang C, Sang N. Glutamine depletion and glucose depletion trigger growth inhibition via distinctive gene expression reprogramming. Cell Cycle. 2012; 11:3679-3690.
183. Zhang K. Endoplasmic reticulum stress response and transcriptional reprogramming. Front Genet. 2015; 5:460.
184. Shi Y, Yang Y, Hoang B, Bardeleben C, Holmes B, Gera J, Lichtenstein A. Therapeutic potential of targeting IRES-dependent c-myc translation in multiple myeloma cells during ER stress. Oncogene. 2016; 35:1015-1024.
185. Sugiura K, Muro Y, Futamura K, Matsumoto K, Hashimoto N, Nishizawa Y, Nagasaka T, Saito H, Tomita Y, Usukura J. The unfolded protein response is activated in differentiating epidermal keratinocytes. J Invest Dermatol. 2009; 129:2126-2135.
186. Davies PF, Civelek M, Fang Y, Fleming I. The atherosusceptible endothelium: endothelial phenotypes in complex haemodynamic shear stress regions in vivo. Cardiovasc Res. 2013; 99:315-327.
187. Armstrong L, Tilgner K, Saretzki G, Atkinson SP, Stojkovic M, Moreno R, Przyborski S, Lako M. Human induced pluripotent stem cell lines show stress defense mechanisms and mitochondrial regulation similar to those of human embryonicstem cells. Stem Cells. 2010; 28:661-673.
188. Prigione A, Fauler B, Lurz R, Lehrach H, Adjaye J. The senescence-related mitochondrial/oxidative stress pathway is repressed in human induced pluripotent stem cells. Stem Cells. 2010; 28:721-733.
189. Vannuvel K, Van Steenbrugge M, Demazy C, Ninane N, Fattaccioli A, Fransolet M, Renard P, Raes M, Arnould T. Effects of a Sublethal and Transient Stress of the Endoplasmic Reticulum on the Mitochondrial Population. J Cell Physiol. 2015; in press
190. Arnould T, Michel S, Renard P. Mitochondria Retrograde Signaling and the UPR mt: Where Are We in Mammals? Int J Mol Sci. 2015; 16:18224-18251.
191. Senft D, Ronai ZA. UPR, autophagy, and mitochondria crosstalk underlies the ER stress response. Trends Biochem Sci. 2015; 40:141-148.
192. Ashraf NU, Sheikh TA. Endoplasmic reticulum stress and Oxidative stress in the pathogenesis of Non-alcoholic fatty liver disease. Free Radic Res. 2015; 49:1405-1418.
193. Chaudhari N, Talwar P, Parimisetty A, Lefebvre d’Hellencourt C, Ravanan P. A molecular web: endoplasmic reticulum stress, inflammation, and oxidative stress. Front Cell Neurosci. 2014; 8:213.
194. Cho YM, Jang YS, Jang YM, Chung SM, Kim HS, Lee JH, Jeong SW, Kim IK, Kim JJ, Kim KS, Kwon OJ. Induction of unfolded protein response during neuronal induction of rat bone marrow stromal cells and mouse embryonic stem cells. Exp Mol Med. 2009; 41:440-452.
195. Yu S, Zhu K, Lai Y, Zhao Z, Fan J, Im HJ, Chen D, Xiao G. atf4 promotes b-catenin expression and osteoblastic differentiation of bone marrow mesenchymal stem cells. Int J Biol Sci. 2013; 9:256-266.
196. Alter J, Bengal E. Stress-induced C/EBP homology protein (CHOP) represses MyoD transcription to delay myoblast differentiation. PLoS One. 2011; 6:e29498.
197. Tanaka K, Kaji H, Yamaguchi T, Kanazawa I, Canaff L, Hendy GN, Sugimoto T. Involvement of the osteoinductive factors, Tmem119 and BMP-2, and the ER stress response PERK-eIF2-ATF4 pathway in the commitment of myoblastic into osteoblastic cells. Calcif Tissue Int. 2014; 94:454-464.
198. Wang F, Wu Y, Gu H, Reece EA, Fang S, Gabbay-Benziv R, Aberdeen G, Yang P. Ask1 gene deletion blocks maternal diabetes-induced endoplasmic reticulum stress in the developing embryo by disrupting the unfolded protein response signalosome. Diabetes. 2015; 64:973-988.
199. Guo FJ, Xiong Z, Lu X, Ye M, Han X, Jiang R. ATF6 upregulates XBP1S and inhibits ER stress-mediated apoptosis in osteoarthritis cartilage. Cell Signal. 2014; 26:332-342.
200. Ng SY, Soh BS, Rodriguez-Muela N, Hendrickson DG, Price F, Rinn JL, Rubin LL. Genome-wide RNA-Seq of Human Motor Neurons Implicates Selective ER Stress Activation in Spinal Muscular Atrophy. Cell Stem Cell. 2015; 17:569-584.

