Introduction
Cardiovascular events (CVEs) represent the main cause of morbidity and mortality in the elderly population [1]. Several mechanisms have been proposed so far to explain the age-related incidence of CVEs. Thus, the prevalence of cardiovascular risk factors, such as smoking, hypertension and diabetes increases in elderly people. Of note is that despite increasingly effective cardiovascular preventive strategies, a portion of patients still experience cardiovascular complications.
Platelets play a key role in the process of athero-thrombosis as indicated by primary and secondary interventional trials with aspirin, which irreversibly acetylates COX1 so preventing platelet formation of the pro-aggregating eicosanoid Thromboxane (TxB) A2. These trials demonstrated, a significant reduction of CVEs in patients treated with aspirin [2, 3]. While experimental studies demonstrated a relationship between platelet activation and aging in animals [4], data in humans are inconclusive because of unreliable methods to assess platelet activation or small sample size [5-7].
Atrial fibrillation (AF) is an interesting model to explore this issue. Thus, the natural history of AF is paradigmatic of the relationship between age and CVEs, as aging is associated with enhanced AF prevalence [8] and an abrupt increase of CVEs complications CVEs [9, 10]. We speculated that platelet activation may increase by aging and be correlated with CVEs in AF. To explore this hypothesis we measured the urinary excretion of TxB2 which is a reliable marker of in vivo platelet activation and is independently associated with CVEs in AF. Aim of the study was therefore to investigate if in AF patients a relationship between aging, urinary TxB2 excretion and CVEs does exist.
Results
Mean age of patients was 72.8±8.4 years, and 45.6% were women; 86.7% had hypertension, 19.2% diabetes, and 15.5% heart failure (HF). Moreover, 15.2% had a previous stroke/ transient ischemic attack (TIA), 19.3% a previous myocardial infarction (MI)/coronary heart disease (CHD), and 8.9% smoked.
TxB2 levels and age
Median TxB2 level was 120 [66-200] ng/mg of urinary creatinine. At multivariable linear regression analysis, age (B: 0.097, p = 0.005) and previous MI/CHD (B: 0.069, p = 0.047) were associated with log-TxB2 levels, after adjustment for gender, hypertension, previous stroke/TIA, diabetes, HF, smoking and statin treatment.
Then, we divided our population into age classes (see Methods) and we found a significant difference in TxB2 levels across classes (Figure 1, p = 0.005), with a significant elevation at 74.6 years (as estimated using shape-restricted B-splines with a change-point).
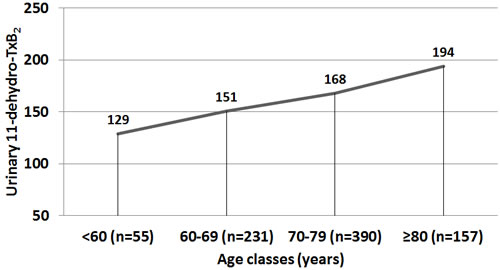
Figure 1: Excretion of urinary 11-dehydro-TxB2 according to age classes.
TxB2 levels, age, and cardiovascular events
Comparison of characteristics of AF patients with and without CVEs is reported in Table 1. During a mean follow-up of 40.9±28.8 months, 128 CVEs occurred: 21 fatal/non-fatal ischemic stroke, 25 fatal/non-fatal MI, 19 CHD, 59 vascular deaths, and 4 TIA. Of these, 0 were in class 1, 19 in class 2 (8%), 71 in class 3 (18%) and 38 in class 4 (24%) (Log-rank test, p < 0.001, Figure 2).
TxB2 levels were significantly higher in patients with CVEs compared to those without in classes 3 (180 [90-315] vs. 100 [58-180], p < 0.001) and 4 (162 [120-362] vs. 130 [80-200], p = 0.020), but not in class 2 (p = 0.145).
Table 1:Baseline characteristics of the study cohort according to the occurrence of cardiovascular events.
Variables |
Cardiovascular events |
p value |
|
No (n= 705) |
Yes (n = 128) |
||
Age (years) |
72.1±8.5 |
77.0±6.6 |
<0.001 |
Women (%) |
46.1 |
43.0 |
0.563 |
Smokers (%) |
9.1 |
7.8 |
0.737 |
CHA2DS2-VASc score# |
3.0 [2.0-4.0] |
4.0 [3.0-6.0] |
<0.001 |
Arterial Hypertension (%) |
85.7 |
92.2 |
0.048 |
Diabetes mellitus (%) |
18.2 |
25.0 |
0.049 |
Heart failure (%) |
13.2 |
28.1 |
<0.001 |
History of stroke/ transient ischemic attack (%) |
12.9 |
28.1 |
<0.001 |
History of myocardial infarction / coronary heart disease(%) |
16.0 |
37.5 |
<0.001 |
Statins (%) |
37.4 |
36.7 |
0.980 |
Thromboxane B2 (ng/mg creatinine)# |
110.0 [60.0-187.5] |
162.5 [100.0-300.0] |
<0.001 |
#data expressed as median and interquartile range.
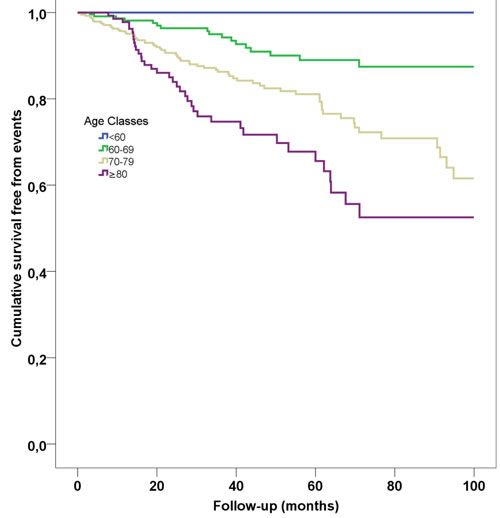
Figure 2: Kaplan-Meier survival curves according to age classes.
Discussion
In this study we analyzed the relationship between platelet activation and aging by classifying AF patients according to decades of age and analyzing the association between platelet activation and CVEs. The novel finding of the present study is that TxB2 biosynthesis is closely related with aging, with a progressive increase across the decades and a significant elevation at age > 74 years. Prospective analysis demonstrated that poor vascular outcomes were more frequently detected in AF patients pertaining to classes 3 and 4 (age ≥ 70 years), who disclosed the highest TxB2 values.
Previous studies investigated the interplay between platelet activation and aging using ex vivo methods to assess platelet function, such as beta-thromboglobulin and platelet factor 4 [5]. Despite a relationship between platelet activation and aging emerged from previous studies, small sample size, lack of follow-up and, overall unreliability, of ex vivo tests assessing platelet activation limits definite conclusion.
Differently from these methods, urinary excretion of 11-dehydro- TxB2 is a more reliable test to explore platelet activation in vivo [11]. However, there are only two studies, which explored the interplay between urinary excretion of 11-dehydro-TxB2 with equivocal findings. Thus, Alessandrini et al. [7] studied serum TxB2 formation in 177 subjects undergoing laboratory screening for the presence of atherosclerotic risk factors. The Authors found no correlation between TxA2 formation and age. However, the overall young age of the cohort (mean age 48 years), and the relative small number of elderly included in the study (only 26 patients aged > 60 years old) may be responsible for this different result. Conversely, Reilly and Fitzgerald [6], in a study involving twenty apparently healthy volunteers, found an increase in vivo formation of TxA2 in older ( > 65 years) than younger ( < 40 years) subjects.
Compared to these two previous studies, we included a much larger cohort of patients at risk of cardiovascular disease, which allowed us to assess in vivo platelet activation across decades of age. Furthermore, we analyzed the impact of platelet activation in a long-term follow up, and its interplay with aging. Our study has two potentially relevant clinical messages; 1) the increase of platelet activation across decades of age, 2) the close relationship between platelet activation and aging in the occurrence of CVEs. Regarding the first point, we showed that urinary excretion of 11-dehydro-TxB2 increases by advancing age, peaking after 70 years. The second finding indicates a close association between aging, platelet activation and CVEs, suggesting that in vivo platelet activation may be a mechanism accounting for CVEs in elderly population.
The study has implications and limitations. The present study did not explore the mechanisms linking aging with platelet activation. Experimental study demonstrated that aging is associated with platelet activation and that reactive oxidant species have a role. In particular, Dayal et al showed that platelet production of H2O2 increases by aging and is associated with platelet activation in mice [4]. The plausibility of this finding relies on the role of H2O2 to enhance platelet activation via activation of COX1 and eventually TxA2 [12]. Thus, further study is necessary to investigate the interplay between platelet H2O2 and CVEs in elderly population.
The clinical implication of this finding is that much more attention should be paid to optimize antiplatelet treatment in the elderly population at risk of CVEs. This issue is even more relevant taking into account that 1) aging negatively influences the ability of low-dose aspirin to inhibit TxB2 biosynthesis [13]; 2) aspirin treatment, in addition to anticoagulant therapy, is associated with a significant higher rate of major bleeding in AF [14].
As these data stem from AF population, further study will assess the relationship between TxB2, aging and CVEs in other at risk populations. A randomized interventional trial with drugs inhibiting platelet function (i.e. TxB2 receptor antagonists) [15], is warranted to explore the potential benefits of modulating platelet function in elderly.
In conclusion, these findings indicate that AF patients over 70 years have significant elevation of platelet activation, which may contribute to the age-related increased risk of CVEs.
materials and Methods
Baseline urinary 11-dehydro-TxB2 (ng/mg of urinary creatinine) levels were measured in 833 non-valvular AF patients treated with vitamin k antagonists (INR range 2.0-3.0). Exclusion criteria: antiplatelet treatment, prosthetic heart valves, cardiac revascularization in the previous year, chronic infections, autoimmune diseases and active cancer.
At baseline, anthropometric data, as well as informations regarding concomitant treatments and comorbidities, were registered. Each patient provided a written informed consent before participating in the study. A urine sample was collected for all patients at baseline (see below).
The occurrence of the first CVE during follow-up was recorded and used for the survival analysis. The composite endpoint of CVEs included: fatal/non-fatal MI and stroke, CHD, TIA and vascular death. The definitions and adjudication of outcomes have been previously reported [16].
Laboratory analysis
After collection, urine samples were stored at -80°C until use. Urinary excretion of 11-dehydro-TxB2 was measured by an ELISA commercial kit (Cayman). Data are expressed as ng/mg creatinine. Intra- and inter-assay coefficients of variation were 4.0% and 3.6%, respectively. Laboratory staff performing TxB2 analysis was unaware of clinical characteristics of patients and CVEs.
Statistical analysis
Categorical variables were reported as counts/percentage and continuous variables as mean (±standard deviation) or median (interquartile range). Student unpaired t test and Mann-Whitney U test were used to compare means and medians, and ANOVA or Kruskall-Wallis test for groups comparison. Stepwise multivariable linear regression analysis was used to assess factors associated with log-TxB2 levels, including pre-specified variables. The cumulative incidence of CVEs according to age classes was estimated using Kaplan-Meier survival analysis. P values < 0.05 were considered as statistically significant. All tests were two-tailed and analyses were performed using SPSS-18.0, SPSS Inc.
Conflicts of interest
The authors declare no actual or potential conflicts of interests.
References
1. Christensen K, Doblhammer G, Rau R and Vaupel JW. Ageing populations: the challenges ahead. Lancet. 2009; 374:1196-1208.
2. FitzGerald GA. Mechanisms of platelet activation: thromboxane A2 as an amplifying signal for other agonists. Am J Cardiol. 1991; 68:11B-15B.
3. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. Bmj. 2002; 324:71-86.
4. Dayal S, Wilson KM, Motto DG, Miller FJ, Jr., Chauhan AK and Lentz SR. Hydrogen peroxide promotes aging-related platelet hyperactivation and thrombosis. Circulation. 2013; 127:1308-1316.
5. Mohebali D, Kaplan D, Carlisle M, Supiano MA and Rondina MT. Alterations in platelet function during aging: clinical correlations with thromboinflammatory disease in older adults. Journal of the American Geriatrics Society. 2014; 62:529-535.
6. Reilly IA and FitzGerald GA. Eicosenoid biosynthesis and platelet function with advancing age. Thrombosis research. 1986; 41:545-554.
7. Alessandrini P, Avogaro P, Bittolo Bon G, Patrignani P and Patrono C. Physiologic variables affecting thromboxane B2 production in human whole blood. Thrombosis research. 1985; 37:1-8.
8. Ball J, Carrington MJ, McMurray JJ and Stewart S. Atrial fibrillation: profile and burden of an evolving epidemic in the 21st century. International journal of cardiology. 2013; 167:1807-1824.
9. Soliman EZ, Safford MM, Muntner P, Khodneva Y, Dawood FZ, Zakai NA, Thacker EL, Judd S, Howard VJ, Howard G, Herrington DM and Cushman M. Atrial Fibrillation and the Risk of Myocardial Infarction. JAMA internal medicine. 2013.
10. Pastori D, Pignatelli P, Angelico F, Farcomeni A, Del Ben M, Vicario T, Bucci T, Raparelli V, Cangemi R, Tanzilli G, Lip GY and Violi F. Incidence of myocardial infarction and vascular death in elderly patients with atrial fibrillation taking anticoagulants: relation to atherosclerotic risk factors. Chest. 2015; 147:1644-1650.
11. Catella F, Healy D, Lawson JA and FitzGerald GA. 11-Dehydrothromboxane B2: a quantitative index of thromboxane A2 formation in the human circulation. Proceedings of the National Academy of Sciences of the United States of America. 1986; 83:5861-5865.
12. Pignatelli P, Pulcinelli FM, Lenti L, Gazzaniga PP and Violi F. Hydrogen peroxide is involved in collagen-induced platelet activation. Blood. 1998; 91:484-490.
13. Eikelboom JW, Hankey GJ, Thom J, Bhatt DL, Steg PG, Montalescot G, Johnston SC, Steinhubl SR, Mak KH, Easton JD, Hamm C, Hu T, Fox KA, et al. Incomplete inhibition of thromboxane biosynthesis by acetylsalicylic acid: determinants and effect on cardiovascular risk. Circulation. 2008; 118:1705-1712.
14. Hansen ML, Sorensen R, Clausen MT, Fog-Petersen ML, Raunso J, Gadsboll N, Gislason GH, Folke F, Andersen SS, Schramm TK, Abildstrom SZ, Poulsen HE, Kober L, et al. Risk of bleeding with single, dual, or triple therapy with warfarin, aspirin, and clopidogrel in patients with atrial fibrillation. Archives of internal medicine. 2010; 170:1433-1441.
15. Santilli F, Mucci L and Davi G. TP receptor activation and inhibition in atherothrombosis: the paradigm of diabetes mellitus. Internal and emergency medicine. 2011; 6:203-212.
16. Pastori D, Pignatelli P, Farcomeni A, Cangemi R, Hiatt WR, Bartimoccia S, Nocella C, Vicario T, Bucci T, Carnevale R, Lip GY and Violi F. Urinary 11-dehydro-thromboxane B2 is associated with cardiovascular events and mortality in patients with atrial fibrillation. American heart journal. 2015; 170:490-497 e491.
