INTRODUCTION
The anaplastic lymphoma kinase (ALK) is a transmembrane receptor tyrosine kinase involved in the pathogenesis of different types of human cancers, including anaplastic large-cell lymphoma, neuroblastoma and non-small cell lung cancer (NSCLC) [1–4]. In NSCLC, ALK is rearranged in approximately 3–7% of all patients [4–7], and in 20–30% of the subset of lung adenocarcinoma patients who are young and non-/light smokers [6–8].
ALK (2p23) rearranges primarily with the echinoderm microtubule-associated protein-like 4 (EML4, 2p21) in NSCLC leading to the expression of the EML4-ALK fusion oncogene [4, 5]. Several EML4-ALK fusion variants have been identified in NSCLC [9], as well as other less-frequent ALK translocations involving different fusion partners [5, 10–14]. While ALK expression is negligible in the normal lung adult tissue [4], these genetic rearrangements lead to the constitutive expression of chimeric proteins comprised of the kinase domain-containing C-terminus of ALK fused to the N-terminus of the translocation partner, which directs ligand-independent dimerization and activation of ALK [4, 5]. In addition to ALK rearrangements, ALK gene amplification has also been detected in NSCLC [7, 15, 16], which might represent an additional mechanism of ALK activation, although its clinical significance is yet to be determined [15].
The identification of ALK translocations in NSCLC has opened the door to the use of targeted therapies for the treatment of these lung cancers. Crizotinib is a well-tolerated first generation ALK inhibitor [17, 18] that has been shown to be superior to standard chemotherapy both as a first- and second-line treatment [19, 20], while second generation ALK inhibitors, such as alectinib and ceritinib, are effective not only in crizotinib-naïve patients, but also in those patients with acquired resistance to crizotinib [21–24]. The availability of these targeted therapies has prompted the development of diagnostic assays and algorithms that can accurately identify ALK-positive lung cancers patients. Although several methodologies have been developed, they display discrepant results [25–31] and often have limited applicability to formalin-fixed, paraffin-embedded (FFPE) tissue samples, the major source material for diagnostic testing [25]. Thus, a consensus on the optimal technique and testing algorithm has not yet been reached in the clinical setting [25, 32].
Two leading FDA approved ALK diagnostic tests are the break-apart fluorescence in situ hybridization (FISH) assay (Vysis ALK Break Apart FISH Probe Kit), and the automated immunohistochemistry (IHC) assay (Ventana ALK (D5F3) CDx Assay). These two tests show a good level of correlation [33] and in a recent international interpretation study they demonstrated an overall sensitivity, specificity and accuracy of 90%, 95% and 93%, respectively [34]. However, the use of alternative approaches, i.e. reverse transcription-polymerase chain reaction (RT-PCR) and/or next-generation sequencing (NGS), has been recommended to resolve discordant or borderline cases [35, 36].
Real-time PCR represents a more quantitative and sensitive technology with reduced inter-observer variability, when compared with FISH and IHC. Yet, some limitations prevent its full implementation in the clinical setting. Firstly, established multiplex RT-PCR assays for the detection of all the different ALK rearrangements require continuous optimization, given the increasing numbers of fusion variants and partners identified [37, 38]. Secondly, more recent reverse transcription quantitative real-time PCR (RT-qPCR) assays based on the unbalanced expression of the 5′ and 3′ portions of the ALK transcript [39–41], which occurs when ALK is rearranged, require significant amounts of RNA (50–100 ng per PCR reaction) from FFPE tissues [40, 41]. Alternative technologies that could be applied to the detection of ALK rearrangements, i.e., NanoString (NanoString Technologies, Inc., Seattle, WA) and RNA massive parallel sequencing require, in addition to elevated amounts of total RNA, the availability of proprietary and cutting-edge platforms in pathology laboratories [42, 43]. To circumvent these problems, we describe herein, a simple quantitative PCR-based ALK predictive model fully optimized to work with low-quantity and low-quality RNA from FFPE samples. The test, by targeting both the 5′ and 3′ portions of ALK mRNA, detects any ALK translocation as well as overexpression of full-length ALK.
RESULTS
Optimization of the RT-qPCR assay for the detection of ALK alterations in FFPE NSCLC samples
The breakpoint of ALK occurs, by and large, before its intracellular kinase domain (exon 20). When ALK is translocated, its C-terminal portion (exons 20–29) is consistently expressed in the chimeric transcript while its N-terminal part (exons 1–19) is lost. Therefore, measuring the unbalanced expression of the C- and N-terminal portions of the ALK transcript in a given sample is a reliable method to indirectly identify ALK rearrangements, regardless of fusion partner and variant type. Based on this rationale, we selected two different RT-qPCR assays, one targeting exons 27–28 in the 3´ region and the other targeting exons 9–10 in the 5´ region of ALK (Figure 1A). In this way, our test is able to detect both known and unknown ALK fusions as well as wild-type ALK upregulation.
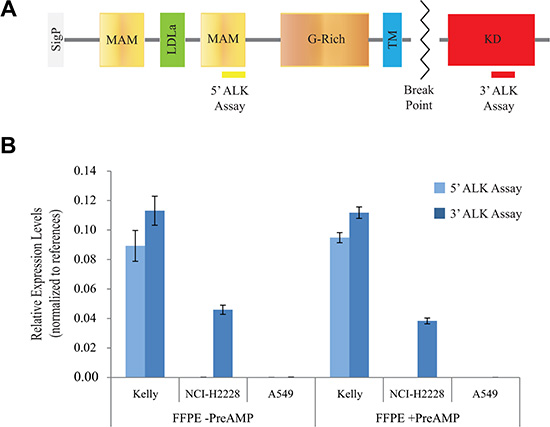
Figure 1: Optimization of the RT-qPCR assays for the detection of the ALK transcript. (A) The position of the two different ALK PCR assays relative to the domain structure of the ALK protein is shown. SigP, signal peptide; MAM (meprin, A-5 protein, and receptor protein-tyrosine phosphatase mu) domains; LDLa (Low-density lipoprotein receptor domain class A) domain; G-rich domain (glycine-rich); TM (Transmembrane) domain; KD (kinase domain). The ALK breakpoint that leads to fusion proteins in different cancers is indicated (zigzag line). (B) Relative expression levels of the 5′ and 3′ portions of the ALK transcript calculated using the formula 2−ΔCq (where ΔCq = CqALK – average CqREF) in FFPE Kelly, NCI-H2228, and A549 cells with (+ PreAMP) or without (– PreAMP) pre-amplification are reported. Error bars indicate 95% confidence intervals (three independent experiments, n = 3).
To optimize the RT-qPCR analysis of ALK expression from limited amounts of degraded RNA from FFPE tissues, we selected probes targeting short regions (< 90 bp in size) of the transcript to increase the probability of detection. We also implemented a multiplex pre-amplification method designed for the dual purpose of stretching precious sample material into more qPCR reactions and of improving the signal-to-noise ratio for the detection of low/moderate-abundance transcripts.
To test the specificity of the two ALK assays, we employed RNA from FFPE samples of: i) Kelly cells expressing readily detectable levels of full-length ALK transcript, containing both the 3′ and 5′ portions of ALK mRNA [2]; ii) NCI-H2228 cells expressing an EML4-ALK translocation, thus positive only for the 3′ portion of the ALK mRNA [5]; iii) A549 cells expressing barely detectable levels of the normal ALK transcript, used as a negative control [5].
As expected, the 3′ assay detected ALK expression levels only in Kelly and NCI-H2228 cells, while the 5′ ALK assay was positive only in Kelly cells (Figure 1B). When the assays were tested on fresh-frozen (FF) samples of the above cell lines and compared with the results from the FFPE samples, similar expression patterns were detected (Supplementary Table S1). Importantly, the relative expression patterns of both the 3′ and 5′ portions of the ALK transcript were comparable in the three FFPE cell lines with and without pre-amplification, indicating an equal and efficient pre-amplification of cDNA for all the selected targets (Figure 1B and Supplementary Table S1). Indeed, the mean pre-amplification uniformity values (ΔΔCq) relative to the two ALK assays, measured in Kelly cells were largely within the ± 1.5 value that is generally accepted for uniform pre-amplification reactions (5′ ALK ΔΔCq: – 0.09 ± 0.20; 3′ ALK ΔΔCq: 0.01 ± 0.14; Supplementary Table S2). In addition, pre-amplification resulted in mean Cq improvements of around 8 cycles: e.g., 7.85 ± 0.13 cycles (range: 7.64−7.99) in Kelly cells (see Supplementary Table S3 for a complete analysis of the 3 cell lines).
Development of an accurate model to predict ALK expression
To predict accurately the expression levels of the 5′ and 3′ portions of ALK in NSCLC samples, we used the calibration curves generated with RNA from Kelly and NCI-H2228 cells (see details in Materials and Methods). The curves were used to develop different predictive models, based on an inverse prediction approach [44], for the quantitative assessment of the 3′ and 5′ or only the 3′ portion of the ALK transcript.
Initially, we compared the calibration curves prepared with Kelly cells in the presence or absence of pre-amplification. The two linear fits and associated residual sum of squares (RSS) of the data points relative to the 3′ and 5′ ALK portions, obtained with these two calibration curves, were comparable (3′ ALK –/+PreAMP, R2 = 0.99/0.99, RSS = 1.17/1.23; 5′ ALK –/+PreAMP, R2 = 0.98/0.98, RSS = 1.97/1.46, Supplementary Figure S1A–B). Moreover, Cq improvements of ~7.57 ± 0.40 cycles for the 5′ and ~7.38 ± 0.35 cycles for the 3′ portion of ALK across all the data points of the dilution range were retained (Supplementary Table S4). To control for the balanced expression of the 3′ and 5′ portions of ALK, we calculated the difference between the ΔCq of the 3′ and of the 5′ portion of ALK (ΔΔCq3′–5′ALK = ΔCq3′ALK-ΔCq5′ALK), relative to the endogenous controls, for each data point of the two calibration curves, in the presence and absence of pre-amplification. These ΔΔCq values are expected to remain stable across the dilution range in the case of balanced 3′/5′ ALK expressions. ΔΔCq values were more stable in the pre-amplified (range = – 0.36 – 0.03; median = – 0.10; Q1 = – 0.15; Q3 = – 0.03) vs. not pre-amplified (range = – 0.91 − 0.38; median = – 0.30; Q1 = – 0.41; Q3 = – 0.22) calibration curve (Supplementary Figure S1C). Based on these results, pre-amplification was included in the generation of the calibration curve for NCI-H2228 cells, which behaved similarly to Kelly cells (Supplementary Figure S1D).
Next, we derived different fitting models using the Kelly and NCI-H2228 calibration curves, either by covering the entire 1–100% dilution range or by restricting the analysis to the 1–50% dilution range, where a better sensitivity of the assay is desirable. For the 3′ portion of ALK, we used both the Kelly and NCI-H2228 calibration curves, separately and pooled, as they are representative of both ALK full-length expression and translocation. For the 5′ portion of ALK we used only the curves obtained with Kelly cells. Overall, the different linear fits (6 for the 3′ and 2 for the 5′ ALK) were similar (R2 range = 0.98–0.99; Figure 2). Parameters relative to the various derived algorithms (Supplementary Table S5) were then used to convert ALK transcript expression measured in NSCLC samples by RT-qPCR into percentages of 3′ and/or 5′ ALK transcript positivity, as described in the following section.
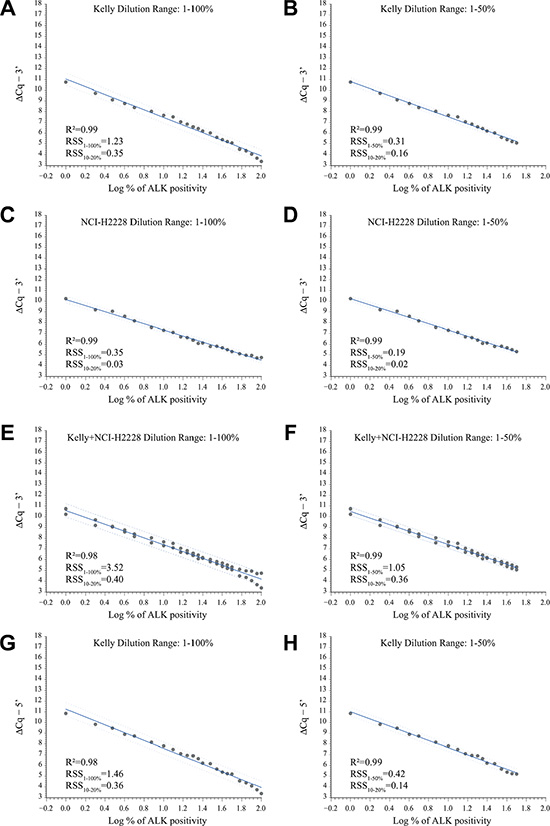
Figure 2: Development of various ALK models using external calibration curves. (A–F) Correlation plots of the expression values (grey dots) of the 3′ portion of ALK relative to the 1–100% and the 1–50% dilution range in Kelly (A and B), NCI-H2228 (C and D), and pooled Kelly + NCI-H2228 (E and F) calibration curves. (G–H) Correlation plots of the expression values (grey dots) of the 5′ portion of ALK relative to the 1–100% (G) or the 1–50% (H) dilution range in the Kelly calibration curves. For all curves, linear fit (solid blue line) and 95% prediction limits (dashed blue line) are reported. Relative expression values of the 3′ (ΔCq−3′) or the 5′ (ΔCq−5′) portion of ALK normalized to internal controls (ΔCq = CqALK – average CqREF) by known percentage (log-transformed, base 10, Log %) of total RNA of ALK positive cells in the different calibration curves are indicated. R2, coefficient of determination; RSS, residual sum of squares relative to the 1–100% (RSS1–100%), the 1–50% (RSS1–50%) and the 10–20% (RSS10–20%) dilution series, are shown.
Validation of the ALK predictive model in a cohort of NSCLC patients
To define the threshold value for ALK positivity (abnormal ALK expression) in NSCLC samples, we applied the different predictive models to a set of 20 lung FFPE non-pathological tissue samples. Based on the low basal levels of 3′ ALK and barely detectable levels of 5′ ALK, measured in non-pathological lung tissues, the threshold value of ALK positivity was set at 10% and 3% for the 3′ and 5′ portions, respectively (Supplementary Tables S6A–S6B).
Next, we applied the derived models to predict ALK status in a cohort of 51 NSCLC FFPE samples, previously characterized by FISH for ALK translocation, composed of 17 FISH positive and 34 FISH negative samples (Table 1). Of note, we obtained RT-qPCR data of adequate quality for all 51 FFPE NSCLC samples, as determined using the interquartile rule for outliers (see Materials and Methods and Supplementary Table S7). All models gave 100% concordant binary predictions (ALK translocated or not translocated) for all the 51 FFPE NSCLC samples, with minor differences in the percentage of 3′ and/or 5′ ALK positivity (Supplementary Table S8). Moreover, models generated in the 1–50% dilution range exhibited narrower 95% confidence intervals for all the predicted percentages of 3′ and 5′ ALK positivity (including those > 50%), compared with the models derived from the 1–100% dilution range (ΔCq data were simulated ranging from 0 to 15, every 0.01 units, as shown in Figure 3A–3C). No significant differences were observed between the predictions obtained with the Kelly and the NCI-H2228 models in the 1–50% dilution range (Figure 3B). Thus, we used the algorithms generated with the Kelly model, which allows the concomitant detection of both the 3′ and 5′ portions of ALK, to predict ALK status in the NSCLC cohort (Figure 3D).
Table 1: Clinico-pathological characteristics of the NSCLC cohort composed of 51 surgical and 7 cytology specimens analyzed for ALK expression
Case ID |
Age (yrs) |
Sex |
Histology |
EGFR/KRAS Mutation |
Tumor Site (Primary/Metastasis) |
Specimen Analyzed |
FISH ALK |
|---|---|---|---|---|---|---|---|
1 |
54 |
F |
ADK |
NEG/NEG |
Metastasis |
Pleura |
POS |
2 |
65 |
F |
ADK |
NEG/NEG |
Primary |
Lung |
POS |
3 |
48 |
F |
ADK |
NEG/NEG |
Metastasis |
Pleura |
POS |
4 |
47 |
M |
ADK |
ND/ND |
Metastasis |
Lymph node |
POS |
5 |
81 |
F |
ADK |
ND/NEG |
Primary |
Lung |
POS |
6 |
66 |
M |
ADK |
NEG/NEG |
Primary |
Lung |
POS |
7 |
38 |
F |
ADK |
NEG/NEG |
Primary |
Lung |
POS |
8 |
58 |
M |
ADK |
NEG/NEG |
Metastasis |
Lymph node |
POS |
9 |
38 |
F |
ADK |
NEG/NEG |
Metastasis |
Lymph node |
POS |
10 |
49 |
F |
ADK |
NEG/NEG |
Metastasis |
Lymph node |
POS |
11 |
47 |
F |
ADK |
NEG/NEG |
Metastasis |
Pleura |
POS |
12 |
52 |
F |
ADK |
NEG/NEG |
Primary |
Lung |
POS |
13 |
61 |
M |
ADK |
NEG/NEG |
Metastasis |
Pleura |
POS |
14 |
51 |
F |
ADK |
NEG/NEG |
Metastasis |
Lymph node |
POS |
15 |
66 |
M |
ADK |
NEG/NEG |
Metastasis |
Pleura |
POS |
16 |
36 |
F |
ADK |
NEG/NEG |
Primary |
Lung |
POS |
17 |
44 |
F |
ADK |
ND/ND |
Primary |
Lung |
POS |
18 |
61 |
M |
ADK |
POS/NEG |
Metastasis |
Pleura |
NEG |
19 |
54 |
F |
ADK |
NEG/NEG |
Primary |
Lung |
NEG |
20 |
70 |
M |
ADSK + NET |
NEG/NEG |
Primary |
Lung |
NEG |
21 |
68 |
M |
ADK |
POS/NEG |
Primary |
Lung |
NEG |
22 |
61 |
M |
ADK |
NEG/POS |
Primary |
Lung |
NEG |
23 |
63 |
M |
ADK |
NEG/NEG |
Primary |
Lung |
NEG |
24 |
56 |
F |
PC |
NEG/POS |
Primary |
Lung |
NEG |
25 |
65 |
F |
ADK |
NEG/POS |
Primary |
Lung |
NEG |
26 |
58 |
M |
ADK |
NEG/NEG |
Primary |
Lung |
NEG |
27 |
63 |
M |
ADK |
NEG/POS |
Primary |
Lung |
NEG |
28 |
80 |
M |
ADK |
NEG/POS |
Primary |
Lung |
NEG |
29 |
58 |
F |
ADSK |
NEG/NEG |
Primary |
Lung |
NEG |
30 |
49 |
M |
ADK |
POS/NEG |
Metastasis |
Pleura |
NEG |
31 |
68 |
M |
ADK |
NEG/ND |
Primary |
Lung |
NEG |
32 |
71 |
M |
ADK |
NEG/ND |
Primary |
Lung |
NEG |
33 |
65 |
M |
ADK |
NEG/NEG |
Primary |
Lung |
NEG |
34 |
50 |
F |
ADK |
NEG/ND |
Primary |
Lung |
NEG |
35 |
53 |
M |
ADK |
NEG/POS |
Primary |
Lung |
NEG |
36 |
53 |
F |
ADK |
NEG/ND |
Primary |
Lung |
NEG |
37 |
59 |
M |
ADK |
NEG/NEG |
Primary |
Lung |
NEG |
38 |
78 |
M |
ADSK |
NEG/ND |
Primary |
Lung |
NEG |
39 |
68 |
F |
ADK |
NEG/ND |
Primary |
Lung |
NEG |
40 |
68 |
M |
ADK |
NEG/NEG |
Primary |
Lung |
NEG |
41 |
60 |
M |
ADK |
NEG/POS |
Primary |
Lung |
NEG |
42 |
59 |
M |
ADK |
NEG/ND |
Primary |
Lung |
NEG |
43 |
61 |
F |
ADK |
NEG/ND |
Primary |
Lung |
NEG |
44 |
71 |
M |
ADK |
NEG/POS |
Metastasis |
Pleura |
NEG |
45 |
62 |
F |
ADK |
NEG/POS |
Primary |
Lung |
NEG |
46 |
66 |
M |
ADK |
NEG/POS |
Metastasis |
Pleura |
NEG |
47 |
46 |
F |
SCC |
POS/NEG |
Metastasis |
Parietal Pleura |
NEG |
48 |
63 |
M |
ADK |
POS/NEG |
Metastasis |
Lymph node |
NEG |
49 |
73 |
F |
ADK |
NEG/NEG |
Metastasis |
Lymph node |
NEG |
50 |
72 |
M |
ADK |
NEG/POS |
Metastasis |
Pleura |
NEG |
51 |
50 |
M |
ADK |
NEG/POS |
Primary |
Lung |
NEG |
52 |
55 |
F |
ADK |
NEG/NEG |
Metastasis |
Lymph node/Cytology |
NEG |
53 |
82 |
M |
ADK |
NEG/NA |
Metastasis |
Lymph node/Cytology |
NEG |
54 |
68 |
M |
ADK |
NEG/POS |
Metastasis |
Lymph node/Cytology |
NEG |
55 |
64 |
M |
ADK |
NEG/NEG |
Metastasis |
Lymph node/Cytology |
NEG |
56 |
63 |
M |
ADK |
NEG/NA |
Metastasis |
Lymph node/Cytology |
NEG |
57 |
75 |
M |
ADK |
NEG/POS |
Metastasis |
Lymph node/Cytology |
POS |
58 |
56 |
F |
ADK |
NEG/NEG |
Metastasis |
Lymph node/Cytology |
POS |
Case IDs #1–51 refer to surgical specimens and Case IDs #52–58 refer to cytology specimens obtained by EBUS. Age at surgery is indicated (yrs); F, female; M, male; ADK, adenocarcinoma; ADSK, adenosquamous cell carcinoma; ADSK + NET, adenosquamous cell carcinoma + neuroendocrine; SCC, squamous; PC, pleomorphic carcinoma of the lung; EGFR, epidermal growth factor receptor; KRAS, Kirsten rat sarcoma viral oncogene homolog; POS, positive; NEG, negative; ND, not determined or missing data; ALK, anaplastic lymphoma kinase; FISH, fluorescent in situ hybridization.
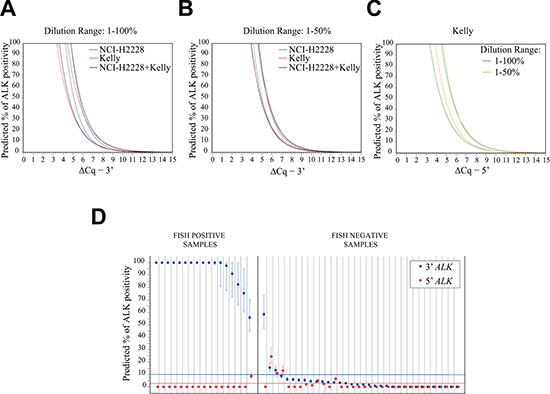
Figure 3: Comparison of the confidence bands in the different models and validation of the Kelly 1–50% ALK predictive model using FFPE NSCLC samples. (A–B) Confidence bands at 95% for predicted percentages of 3′ ALK positivity relative to the calibration curves in the 1–100% (A) or the 1–50% (B) range with NCI-H2228 (blue lines), Kelly (red lines) and Kelly + NCI-H2228 (black lines) for the ALK 3′ portion. (C) Confidence bands at 95% for predicted percentages of 5′ ALK positivity specific to the Kelly calibration curves in the 1–100% (green line) or the 1–50% (orange line) range. ΔCq data were simulated ranging from 0 to 15, every 0.01 units. Estimates were obtained through inverse prediction models considering the indicated calibration curves and dilution series. (D) Predicted percentage (and 95% confidence bars) of 3′ (blue dots) and 5′ (red dots) ALK portions for the 17 FISH-positive and 34 FISH-negative NSCLC samples. Blue and red lines indicate the threshold for the 3′ (10%) and 5′ (3%) ALK positivity, respectively.
Our model identified all the 17 ALK FISH-positive samples as translocated. Indeed, all these samples showed an elevated percentage of 3′ positivity compared with the percentage of 5′ positivity, thereby confirming the presence of an unbalanced expression of the two ALK regions as a consequence of a translocation event (Table 2). Among the 34 FISH-negative cases, 33 resulted not translocated by our method, while one sample was clearly detected as translocated (Case ID #19, 3′ ALK positivity of 58.6%; 95% CI: 47–73.5), despite its scarce tumor cellularity (5%, Table 2). This sample, with a marginal percentage of FISH positive nuclei (10%; Table 2), was ALK-positive by IHC analysis using the automated IHC Ventana assay, in agreement with our PCR assay. Thus, these data indicate that the discrepant FISH result was a false negative and, importantly, that our test reliably identifies ALK translocations in specimens with limited tumor cellularity, as low as 5–10% (Case ID #13, #15 and #19, Table 2).
Table 2: Accuracy of the PCR-based predictive model in the assessment of ALK status in the 51 NSCLC cohort
Case ID |
FISH |
RT-qPCR Model - Kelly (1–50%) 100 ng RNA |
Tumor Cell Content (%) |
Tumor Site (Primary/Metastasis) |
Specimen Analyzed |
|||
|---|---|---|---|---|---|---|---|---|
ALK score |
Positive Nuclei (%) |
ALK Status |
3′ ALK Positivity (%) |
5′ ALK Positivity (%) |
||||
1 |
POS |
90 |
T |
82.6 (66.0–100) |
0 |
30 |
Metastasis |
Pleura |
2 |
POS |
64 |
T |
100 (100–100) |
0 |
60 |
Primary |
Lung |
3 |
POS |
87 |
T |
100 (100–100) |
0 |
60 |
Metastasis |
Pleura |
4 |
POS |
61 |
T |
100 (81.1–100) |
0 |
80 |
Metastasis |
Lymph node |
5 |
POS |
55 |
T |
100 (100–100) |
0 |
80 |
Primary |
Lung |
6 |
POS |
61 |
T |
100 (98.4–100) |
0 |
70 |
Primary |
Lung |
7 |
POS |
70 |
T |
100 (100–100) |
0 |
60 |
Primary |
Lung |
8 |
POS |
50 |
T |
100 (100–100) |
0 |
60 |
Metastasis |
Lymph node |
9 |
POS |
50 |
T |
100 (100–100) |
0 |
60 |
Metastasis |
Lymph node |
10 |
POS |
53 |
T |
100 (100–100) |
0 |
70 |
Metastasis |
Lymph node |
11 |
POS |
31 |
T |
75.4 (60.3–95.0) |
0 |
20 |
Metastasis |
Pleura |
12 |
POS |
35 |
T |
100 (100–100) |
0 |
30 |
Primary |
Lung |
13 |
POS |
23 |
T |
97.7 (77.8–100) |
0 |
10 |
Metastasis |
Pleura |
14 |
POS |
24 |
T |
91.1 (72.6–100) |
0 |
40 |
Metastasis |
Lymph node |
15 |
POS |
32 |
T |
55.8 (44.8–69.9) |
8.7 (6.8–11.0) |
5 |
Metastasis |
Pleura |
16 |
POS |
24 |
T |
100 (100–100) |
0 |
50 |
Primary |
Lung |
17 |
POS |
25 |
T |
100 (100–100) |
0 |
60 |
Primary |
Lung |
18 |
NEG |
8 |
NT |
0 |
0 |
70 |
Metastasis |
Pleura |
19 |
NEG |
10 |
T |
58.6 (47.0–73.5) |
0 |
5 |
Primary |
Lung |
20 |
NEG |
3 |
NT |
13.5 (10.9–16.6) |
11.0 (8.6–14.0) |
80 |
Primary |
Lung |
21 |
NEG |
0 |
NT |
4.6 (3.7–5.7) |
1.2 (0.9–1.5) |
70 |
Primary |
Lung |
22 |
NEG |
8 |
NT |
6.2 (5.0–7.7) |
0 |
60 |
Primary |
Lung |
23 |
NEG |
2 |
NT |
3.8 (3.1–4.8) |
0 |
80 |
Primary |
Lung |
24 |
NEG |
5 |
NT |
0.8 (0.6–1.0) |
0 |
60 |
Primary |
Lung |
25 |
NEG |
11 |
NT |
15.5 (12.5–19.2) |
24.4 (19.2–31.3) |
50 |
Primary |
Lung |
26 |
NEG |
0 |
NT |
5.5 (4.4–6.8) |
0 |
30 |
Primary |
Lung |
27 |
NEG |
5 |
NT |
4.4 (3.6–5.5) |
4.9 (3.8–6.3) |
30 |
Primary |
Lung |
28 |
NEG |
0 |
NT |
3.3 (2.7–4.1) |
0 |
60 |
Primary |
Lung |
29 |
NEG |
1 |
NT |
1.8 (1.4–2.2) |
0 |
80 |
Primary |
Lung |
30 |
NEG |
4 |
NT |
0 |
0 |
70 |
Metastasis |
Pleura |
31 |
NEG |
3 |
NT |
2.0 (1.6–2.5) |
0 |
80 |
Primary |
Lung |
32 |
NEG |
3 |
NT |
5.4 (4.4–6.7) |
2.0 (1.5–2.5) |
80 |
Primary |
Lung |
33 |
NEG |
0 |
NT |
0 |
0 |
80 |
Primary |
Lung |
34 |
NEG |
4 |
NT |
8.8 (7.1–10.9) |
13.1 (10.3–16.7) |
90 |
Primary |
Lung |
35 |
NEG |
0 |
NT |
0 |
0 |
60 |
Primary |
Lung |
36 |
NEG |
0 |
NT |
0 |
0 |
60 |
Primary |
Lung |
37 |
NEG |
5 |
NT |
0 |
0 |
80 |
Primary |
Lung |
38 |
NEG |
5 |
NT |
0 |
0 |
70 |
Primary |
Lung |
39 |
NEG |
2 |
NT |
4.3 (3.4–5.3) |
2.4 (1.9–3.1) |
70 |
Primary |
Lung |
40 |
NEG |
2 |
NT |
2.9 (2.3–3.6) |
0 |
60 |
Primary |
Lung |
41 |
NEG |
4 |
NT |
1.7 (1.4–2.1) |
0 |
60 |
Primary |
Lung |
42 |
NEG |
13 |
NT |
0 |
0 |
40 |
Primary |
Lung |
43 |
NEG |
6 |
NT |
0 |
0 |
80 |
Primary |
Lung |
44 |
NEG |
13 |
NT |
0.9 (0.7–1.1) |
0 |
80 |
Metastasis |
Pleura |
45 |
NEG |
1 |
NT |
0 |
0 |
40 |
Primary |
Lung |
46 |
NEG |
3 |
NT |
3.6 (2.9–4.4) |
6.5 (5.1–8.3) |
5 |
Metastasis |
Pleura |
47 |
NEG |
4 |
NT |
0.9 (0.7–1.1) |
0 |
70 |
Metastasis |
Parietal Pleura |
48 |
NEG |
2 |
NT |
6.0 (4.8–7.4) |
0 |
40 |
Metastasis |
Lymph node |
49 |
NEG |
1 |
NT |
1.8 (1.4–2.2) |
0 |
40 |
Metastasis |
Lymph node |
50 |
NEG |
4 |
NT |
0 |
0 |
20 |
Metastasis |
Pleura |
51 |
NEG |
1 |
NT |
0 |
0 |
60 |
Primary |
Lung |
Kelly |
– |
– |
NT |
100 (100–100) |
100 (100–100) |
– |
– |
– |
NCI-H2228 |
– |
– |
T |
71.8 (57.4–90.3) |
0 |
– |
– |
– |
A549 |
– |
– |
NT |
0 |
0 |
– |
– |
– |
Binary ALK status by FISH analysis is indicated as positive (POS, ≥ 15% of positive nuclei) or negative (NEG, < 15% of positive nuclei). Percentage (%) of FISH positive nuclei with separated green and red signals or single red signals (translocation and deletion of the ALK 5′ portion, respectively) are reported when available. Binary ALK status determined by the PCR-based model analysis is indicated as Translocated (T) or Not Translocated (NT) based on the predicted percentage of 3′ and 5′ ALK positivity measured in each FFPE NSCLC sample (positivity cut-off value for the 3′ and 5′ ALK portions were 10% and 3%, respectively). Predicted percentages of 3′ and 5′ ALK positive RNA present in FFPE NSCLC samples are reported along with the 95% confidence intervals in brackets. These values were calculated from the data obtained in the RT-qPCR analysis by the ALK inverse prediction models obtained from the Kelly calibration curves for the 3′ and 5′ portions of ALK. The model was derived considering the 1–50% dilution series of the external calibration curves. ALK transcript expression was measured using the PCR protocol optimized for FFPE samples starting from 100 ng of input RNA. The percentage (%) of tumor cell content is also indicated for each sample. ALK, anaplastic lymphoma kinase; FISH, fluorescence in-situ hybridization; RT-qPCR, quantitative reverse transcription-polymerase chain reaction.
Among the 33 tumors identified as not translocated by our PCR assay, two exhibited a level of 3′ positivity significantly above the 10% cut-off (Case ID #20, 13.5%; Case ID #25, 15.5%, Table 2). Both these samples, however, showed percentages of 5′ positivity comparable to the 3′ portion (Table 2), suggesting that they did not harbor an ALK translocation, but rather expressed low/moderate levels of normal ALK transcripts. Together, these results showed that our diagnostic model reached a sensitivity, specificity and accuracy of 100%, 97% and 98%, respectively, compared with FISH.
To evaluate whether our predictive model was accurate when using low quantities of input RNA, representative FFPE NSCLC samples (21 in total), covering a wide range of ALK positivity, were processed using 25 and 10 ng of total RNA instead of 100 ng. The predictions relative to the three different RNA quantities (100, 25 and 10 ng) were 100% concordant, indicating that our model reliably detects ALK status in FFPE NSCLC samples also when the input RNA is scarce (Table 3).
Table 3: Accuracy of ALK status prediction in a selected subset of 21 NSCLC samples using 100, 25 and 10 ng of input RNA
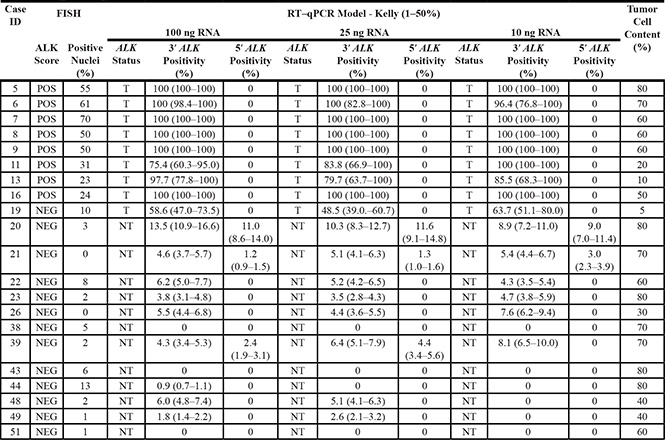
Binary ALK status by FISH analysis is indicated as positive (POS, ≥ 15% of positive nuclei) or negative (NEG, < 15% of positive nuclei). Percentage (%) of FISH positive nuclei with separated green and red signals or single red signals (translocation and deletion of the ALK 5′ portion, respectively) are reported when available. Binary ALK status determined by the PCR-based model analysis is indicated as Translocated (T) or Not Translocated (NT) based on the predicted percentage of 3′ and 5′ ALK positivity measured in each FFPE NSCLC sample using 100, 25 or 10 ng of RNA input (positivity cut-off values for the 3′ and 5′ ALK portions were 10% and 3%, respectively). Predicted percentages of 3′ and 5′ ALK positive RNA present in FFPE NSCLC samples are reported along with 95% confidence intervals in brackets. These values were calculated from the data obtained in the RT-qPCR analysis by the ALK inverse prediction models obtained from the Kelly calibration curves for the 3′ and 5′ portions of ALK. The model was derived considering the 1–50% dilution series of the external calibration curves. The percentage (%) of tumor cell content is also indicated for each sample. ALK, anaplastic lymphoma kinase; FISH, fluorescence in situ hybridization; RT-qPCR, quantitative reverse transcription-polymerase chain reaction.
Finally, we analyzed 7 cytological FFPE samples (5 ALK FISH-negative and 2 ALK FISH-positive), obtained with the minimally invasive endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) procedure, using 10 ng input RNA (Table 1). Our assay correctly identified all the 5 FISH-negative cases and one of the two FISH-positive cases (Case ID #58, with 38% of FISH positive nuclei; Table 4). Notably, the one discordant case (Case ID #57, with a borderline 20% of FISH-positive nuclei) was confirmed to be negative by subsequent IHC analysis.
Table 4: Accuracy of ALK status prediction in the 7 cytology specimens using 10 ng of input RNA
Case ID |
FISH |
RT-qPCR Model - Kelly (1–50%) - 10 ng RNA |
Tumor Cell Content (%) |
|||
|---|---|---|---|---|---|---|
ALK Score |
Positive Nuclei (%) |
ALK Status |
3′ ALK Positivity (%) |
5′ ALK Positivity (%) |
||
52 |
NEG |
11 |
NT |
0 |
0 |
60 |
53 |
NEG |
9 |
NT |
0 |
0 |
80 |
54 |
NEG |
9 |
NT |
0 |
0 |
30 |
55 |
NEG |
12 |
NT |
0 |
0 |
50 |
56 |
NEG |
9 |
NT |
0 |
14.6 (11.5–18.6) |
40 |
57 |
POS |
20 |
NT |
0 |
0 |
70 |
58 |
POS |
38 |
T |
40.4 (32.5–50.4) |
0 |
70 |
Binary ALK status by FISH analysis is indicated as positive (POS, ≥ 15% of positive nuclei) or negative (NEG, < 15% of positive nuclei). Percentage (%) of FISH positive nuclei with separated green and red signals or single red signals (translocation and deletion of the ALK 5′ portion, respectively) are reported. Binary ALK status determined by the PCR-based model analysis is indicated as Translocated (T) or Not Translocated (NT) based on the predicted percentage of 3′ and 5′ ALK positivity measured in each cytology sample using 10 ng of RNA input (positivity cut-off values for the 3′ and 5′ ALK portions were 10% and 3%, respectively). Predicted percentages of 3′ and 5′ ALK positive RNA present in FFPE NSCLC samples are reported along with 95% confidence intervals in brackets. These values were calculated from the data obtained in the RT-qPCR analysis by the ALK inverse prediction models obtained from the Kelly calibration curves for the 3′ and 5′ portions of ALK. The model was derived using the 1–50% dilution series of the external calibration curves. The percentage (%) of tumor cell content is also indicated for each sample. ALK, anaplastic lymphoma kinase; FISH, fluorescence in situ hybridization; RT-qPCR, quantitative reverse transcription-polymerase chain reaction.
In summary, our diagnostic model reached a sensitivity, specificity and accuracy of 95%, 97% and 97%, respectively, compared with FISH on a total of 58 FFPE NSCLC samples (51 surgical and 7 cytology specimens) (Figure 4). Notably, the two discrepant cases (one surgical and one cytological) were confirmed as positive by IHC, thus raising the accuracy of our test to 100%.
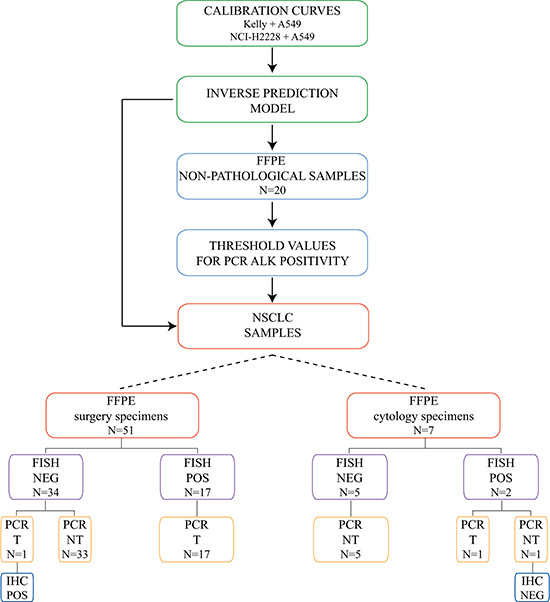
Figure 4: Study design for the development of the RT-qPCR-based prediction model and the validation on 58 FFPE NSCLC tissue specimens. Schematic representation of the analysis of ALK status in the NSCLC cohort. The cohort was composed of 51 surgical and 7 cytology FFPE NSCLC specimens (see Table 1). Binary ALK status by FISH analysis is indicated as positive (POS, ≥ 15% of positive nuclei) or negative (NEG, < 15% of positive nuclei). Binary ALK status determined by the PCR-based model analysis is indicated as Translocated (T) or Not Translocated (NT) based on the predicted percentage of 3′ and 5′ ALK positivity (cut-off values for 3′ and 5′ ALK portions were 10% and 3%, respectively). Binary ALK status by IHC is indicated as positive (POS) or negative (NEG) based on the presence or lack of a strong granular cytoplasmic staining in tumor cells, respectively.
DISCUSSION
The discovery of effective ALK-targeted therapies for the treatment of NSCLC demands that we improve our ability to identify patients eligible for treatment. The FDA-approved break-apart FISH assay represented the gold standard for the detection of ALK rearrangements during the crizotinib trials. However, given the difficulty in signal interpretation [45], the FISH assay yields erroneous results [18, 21, 23, 46, 47]. The recently approved Ventana ALK IHC (D5F3) CDx assay is a viable alternative to FISH. Although this IHC assay provides a simple digital reading of ALK status to limit inter-observer variability, a qualified pathologist is needed to grade the result and proprietary stainer and kits are required. Therefore, more quantitative, objective, and open source methodologies could find widespread application in clinical laboratories and help to resolve discrepant cases and/or equivocal results.
Quantitative PCR is a reliable and extremely sensitive technique for the measurement of clinical biomarkers. Optimal PCR-based ALK assays in clinical practice should be i) versatile (able to detect any ALK alteration), ii) accurate (especially when using low quantity, poor quality and low tumor cellularity FFPE biological samples), iii) user-friendly (implementable in routine clinical labs), and iv) cost-effective (particularly relevant for the screening of large, low-incidence populations). Our simple RT-qPCR-based predictive model fulfills these requirements. The combination of a simple inverse prediction model with an optimized RT-qPCR protocol showed that our ALK test was superior to the break-apart FISH assay in a cohort of 58 FFPE NSCLC cases, composed of 51 surgical and 7 cytology specimens. Overall, our diagnostic model reached a sensitivity, specificity and accuracy of 95%, 97% and 97%, respectively, compared with FISH (Tables 2 and 4, Figure 4), and further validation on a larger cohort is now merited. Notably, the accuracy of our test rose to 100% when the FISH false-negative surgical sample and the FISH false-positive cytology sample were reclassified as ALK-positive and ALK-negative, respectively, according to IHC (Figure 4). Our predictive test also provided high percentages of 3′ ALK positivity in samples with borderline percentages of FISH-positive nuclei (range = 10–35%), demonstrating superior sensitivity (Table 2 and Table 3). Additionally, our assay was accurate when using as little as 10 ng of input RNA, also in samples with low tumor cellularity (5–10%, Tables 2 and 3) and in cytological specimens (Table 4), which is frequently the only clinical material available in patients with advanced lung cancer. Notably, in a couple of cases (Case ID #20 and #25, Table 2), our test detected overexpression of full-length ALK, in the absence of ALK translocation (Table 2). Whether this higher expression identifies a small fraction of lung cancers sensitive to ALK inhibitors remains to be established. In addition, by targeting exons 27–28 in the 3′ portion of ALK, our assay is able to detect also the recently identified ALK isoforms (though rare in lung cancer), encompassing exons 20–29 [48].
Our test offers several advantages over published [39–43] assays assessing the unbalanced expression of the 3′ and 5′ portion of ALK transcript: i) it requires at least 5–10 times less input RNA; ii) it does not require expensive and proprietary technologies or specialist expertise (unlike NanoString and/or NGS platforms), and iii) it provides a quantitative assessment of 3′ and 5′ ALK mRNA positivity. Unlike traditional comparative (ΔΔCq) and/or 3′/5′ ratio methods derived thereof, our model provides confidence intervals for each point prediction. These interval estimates, by identifying the range of possible true values of the point prediction, will increase clinicians’ confidence about the prediction score and help guide them in therapy decision-making.
Novel multiplex NGS assays represent attractive diagnostic tools for the detection of clinically relevant genomic alterations associated with solid tumors including ALK translocations [36, 49]. An extended comparative analysis of our predictive model vs. emerging NGS assays, employing large NSCLC cohorts, and technically challenging FFPE specimens is warranted. However, our results clearly show that our PCR-based ALK test is highly accurate in surgical as well as cytological specimens. Therefore, our diagnostic assay not only can be employed as a confirmatory test of FISH and/or IHC results and to resolve equivocal FISH and/or IHC results, but can also be used as a first-line diagnostic tool for the rapid, efficacious and convenient screening of large patient populations.
MATERIALS AND METHODS
Human cell lines and samples
The human neuroblastoma cell line Kelly (DSMZ® ACC 355™) and the lung cancer cell line NCI-H2228 (ATCC® CRL-5935™) were used as positive controls for the expression of full-length ALK transcript (ALKF1174L mutant) [2] and of translocated ALK (EML4-ALK) [5], respectively. The lung cancer cell line A549 (ATCC® CCL-185™) was used as a negative control, since they express barely detectable levels of the normal ALK transcript [5]. Cells were cultured and used fresh or pelleted and processed into FFPE cell blocks for subsequent analyses. Cells were routinely tested for mycoplasma contamination [50] and multiplex short tandem repeat profiling test for authentication using the GenePrint® 10 System (Promega Corporation, Madison, WI, USA).
All samples were derived from patients operated at the European Institute of Oncology (IEO), Milan, Italy. FFPE tissue blocks of ALK FISH-positive samples with sufficient biological material were available for 17 patients. Based on the availability of these 17 FISH-positive samples, we randomly selected 34 FISH-negative controls, which together constituted a cohort of 51 NSCLC patients (with a 1:2 ratio of ALK positive vs. control samples). We also analyzed 7 cytological samples (cytoblocks), 5 FISH-negative and 2 FISH-positive, with sufficient biological material obtained with the EBUS-TBNA procedure from patients with clinically diagnosed primary lung cancer. The clinico-pathological characteristics of the patients are described in Table 1. RNA was also extracted from FFPE non-pathological lung tissue samples, adjacent to diseased area, prepared from an additional 20 NSCLC patients. Each case was centrally reviewed to confirm the histopathological assessment and to verify the content of tumor or normal parenchyma. Investigations were conducted in accordance with the ethical standards as outlined in the Declaration of Helsinki and in national and international guidelines, and were approved by the IEO institutional review board.
Fluorescence in situ hybridization
ALK rearrangements were analyzed on 4 μm thick FFPE tissue sections, using the break-apart probe FISH Probe Kit (Vysis LSI ALK Dual Color, Abbott Molecular Inc.) according to manufacturer’s instructions. Samples were analyzed using an epifluorescence microscope (Leica, Wetzlar, Germany). Signal evaluation was performed in at least 60 nuclei as follows: i) separated green and red signals or single red signals (translocation and deletion of the ALK 5′ portion) in at least 15% of tumor cells analyzed; ii) overlapping of green and red signals (yellowish) indicated cells in which ALK was not rearranged.
Immunohistochemistry
ALK IHC was performed on 4 μm thick FFPE tissue sections using the fully automated Ventana IHC ALK (D5F3) CDx Assay (Ventana Medical Systems, Inc., Tucson, AZ, USA), with the pre-diluted Ventana anti-ALK (D5F3) rabbit monoclonal primary antibody, the Optiview DAB IHC detection kit and Optiview Amplification kit on the Benchmark XT stainer. We adopted the binary scoring system (positive or negative for ALK status) to evaluate the staining results according to manufacturer’s recommendations. ALK positivity was assigned exclusively in the presence of a strong granular cytoplasmic staining in tumor cells (any percentage of positive tumor cells).
RNA extraction and quantitative real-time PCR
Genetic material was isolated from fresh cell lines using the AllPrep DNA/RNA/miRNA Universal Kit (Qiagen, Hilden, Germany) and from FFPE cell blocks or tissue blocks using the AllPrep DNA/RNA FFPE Kit (Qiagen, Hilden, Germany). RNA was extracted from manually microdissected areas of 2 tissue sections (10 μm thick) on glass slides selected by a pathologist for each relevant FFPE tissue block. For standard mRNA analysis, 500 ng of total RNA (RNA concentration measured using the NanoDrop® ND-1000 Spectrophotometer) were reverse transcribed with random primers using the SuperScript® VILO™ cDNA Synthesis Kit (Thermo Fisher Scientific) and 5 ng or 20 ng of cDNA from fresh or FFPE cells, respectively, were then analyzed per reaction by PCR. In case of pre-amplification, 100, 25, and 10 ng (as indicated) of total RNA from FFPE cell blocks and/or FFPE tissue blocks were reverse transcribed, pre-amplified for 10 cycles using the PreAMP Master Mix Kit (Thermo Fisher Scientific) according to manufacturer’s instructions, and diluted 1:5 prior to PCR analysis (5 μl were then used per PCR reaction, corresponding to 2, 0.5 or 0.2 ng of cDNA).
Quantitative PCR was performed with hydrolysis probes (Thermo Fisher Scientific) using the SsoAdvanced Universal Probes Supermix (Bio-Rad Laboratories) in 10 μl of final volume in 384-well plates. PCR reaction was run in LightCycler (LC) 480 real-time PCR instruments (Roche) using the following thermal cycling conditions: 1 cycle at 95°C for 30 sec, 45 cycles at 95°C for 5 sec, and 60°C for 30 sec.
TaqMan gene expression assays, with short amplicon sizes, were as follows: Hs01058323_m1 (human ALK, RefSeq NM_004304, exon boundary 9–10, assay location 2775, amplicon length 66 bp), Hs00608292_m1 (human ALK, RefSeq NM_004304, exon boundary 27–28, assay location 5022, amplicon length 59 bp), Hs03929097_g1 (human GAPDH, RefSeq NM_001256799, exon boundary 8–8, assay location 1250, amplicon length 58 bp), Hs99999908_m1 (human GUSB, RefSeq NM_000181, exon boundary 11–12, assay location 1925, amplicon length 81 bp) and Hs00427621_m1 (human TBP, RefSeq NM_001172085, exon boundary 3–4, assay location 666, amplicon length 65 bp) (sequence details in Supplementary Table S9).
We defined Cq = 40 as our limit of detection in the absence of pre-amplification and Cq = 30 in the presence of the pre-amplification, based on the lowest Cq value measured for the two ALK assays in the negative control cell line A549 (Supplementary Table S1). Cq values beyond these limits were set to 40 or 30, accordingly, and normalization was omitted. Each target was assayed in triplicate and average Cq values were calculated either from triplicate values when the standard deviation was < 0.4, or from the best duplicate values when the standard deviation was ≥ 0.4. In each sample, the average Cq value of the 3′ or 5′ portion of ALK (CqALK) was normalized on the average Cq (CqREF) value of three human reference genes (GAPDH, GUSB and TBP), to account for variation in the expression of single reference genes and in RNA integrity due to tissue fixation, using the following formula:
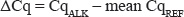
Pre-amplification uniformity for each ALK gene expression assay was measured in Kelly cells by calculating the ΔCq for each of the two ALK assays (ΔCq = Cq3′/5′ALK – mean CqREF) and by determining the ΔΔCq between pre-amplified (PreAmp) and not-pre-amplified cDNA templates (ΔΔCq = ΔCqPreAmp – ΔCqcDNA). Minus-reverse transcriptase (“−RT”) controls were also performed for pre-amplified cDNA templates to confirm the specificity of the ALK assays. “−RT” controls were negative both for the 3′ and 5′ ALK assays (all samples were flagged as “undetectable”). Based on the distribution of the reference genes, we applied the Tukey’s interquartile rule for outliers [51] to identify poor quality RT-qPCR data in the cohort of 51 FFPE NSCLC samples.
Development of the predictive model for the assessment of ALK status in tumor specimens
We prepared two different calibration curves using an artificial dilution series of total RNA derived from FFPE blocks of either Kelly cells expressing full-length ALK or NCI-H2228 cells expressing EML4-ALK, mixed with total RNA from ALK negative A549 cells. Each curve was composed of 23 data points, obtained by diluting RNA from positive cells with increasing amounts of RNA from negative cells until a mixture composed of 1% NCI-H2228/Kelly RNA and 99% A549 RNA was reached. The upper half of the curve (50–100% of RNA from positive cells) was obtained using discrete increments of 10% of RNA from negative cells. To improve the resolution of the ALK assay in the lower half of the calibration curve (0–50%), RNA from positive cells was diluted using progressively smaller increments of RNA from negative cells as follows: 5% increments in the 25–50% range, 2.5% increments in the 5–25% range, and 1% increments in the 1–5% range. We reverse transcribed 500 ng or 100 ng of total RNA for each data point of the calibration curve in the absence or in the presence of pre-amplification, respectively, prior to PCR analysis.
The level of expression of the 3′ and/or the 5′ portion of the ALK transcript measured by PCR was regressed on the known percentage (log-transformed, base 10) of total ALK-positive RNA present in each data point of the calibration curve analyzed. The inverse prediction approach was applied to predict the actual percentage of 3′ and/or 5′ ALK-positive transcript, as well as 95% confidence intervals from the values obtained in the RT-qPCR analysis [44].
We employed both Kelly and NCI-H2228 calibration curves, separately or pooled, to build the predictive models for the 3′ portion; the Kelly calibration curve alone was used for the 5′ portion of ALK. To improve the performance of the assay when ALK is expressed at low/moderate levels in unknown NSCLC samples, we also generated predictive models by including only the data points in the 1–50% dilution range. Coefficient of determination (R2) and residual sum of squares (RSS) values were calculated to assess the goodness of the various fits. To evaluate the basal level of expression of both the 5′ and 3′ portions of ALK, we applied the different predictive models to the set of 20 lung FFPE non-pathological tissue samples. The threshold value of ALK positivity (abnormal ALK expression) was established based on the median of the 95th percentiles of the predictions obtained from each of the different models. Finally, these inverse predictive models were validated in the cohort of 51 FFPE human samples. Translocation was assigned based on unbalanced interval estimates of 3′ and 5′ ALK predictions. Statistical analyses were performed using SAS (SAS 9.3, SAS Institute, Cary, NC, USA).
ACKNOWLEDGMENTS
We appreciate the efforts of Rosalind Gunby for critically reading the manuscript, and Stefania Pirroni and Giovanna Jodice for technical assistance.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
GRANT SUPPORT
This work was supported by grants from the Associazione Italiana per la Ricerca sul Cancro (AIRC - IG 11904 to SP; IG 10349, IG 14404 and MCO 10.000 to PPDF); the Italian Ministry of University and Scientific Research (MIUR) to PPDF; the Italian Ministry of Health to SP and PPDF; the Monzino Foundation to PPDF; the G. Vollaro Foundation to SP.
REFERENCES
1. Kutok JL, Aster JC. Molecular biology of anaplastic lymphoma kinase-positive anaplastic large-cell lymphoma. J Clin Oncol. 2002; 20:3691–3702.
2. George RE, Sanda T, Hanna M, Frohling S, Luther W 2nd, Zhang J, Ahn Y, Zhou W, London WB, McGrady P, Xue L, Zozulya S, Gregor VE, et al. Activating mutations in ALK provide a therapeutic target in neuroblastoma. Nature. 2008; 455:975–978.
3. Mosse YP, Laudenslager M, Longo L, Cole KA, Wood A, Attiyeh EF, Laquaglia MJ, Sennett R, Lynch JE, Perri P, Laureys G, Speleman F, Kim C, et al. Identification of ALK as a major familial neuroblastoma predisposition gene. Nature. 2008; 455:930–935.
4. Soda M, Choi YL, Enomoto M, Takada S, Yamashita Y, Ishikawa S, Fujiwara S, Watanabe H, Kurashina K, Hatanaka H, Bando M, Ohno S, Ishikawa Y, et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature. 2007; 448:561–566.
5. Rikova K, Guo A, Zeng Q, Possemato A, Yu J, Haack H, Nardone J, Lee K, Reeves C, Li Y, Hu Y, Tan Z, Stokes M, et al. Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell. 2007; 131: 1190–1203.
6. Shaw AT, Yeap BY, Mino-Kenudson M, Digumarthy SR, Costa DB, Heist RS, Solomon B, Stubbs H, Admane S, McDermott U, Settleman J, Kobayashi S, Mark EJ, et al. Clinical features and outcome of patients with non-small-cell lung cancer who harbor EML4-ALK. J Clin Oncol. 2009; 27: 4247–4253.
7. Camidge DR, Kono SA, Flacco A, Tan AC, Doebele RC, Zhou Q, Crino L, Franklin WA, Varella-Garcia M. Optimizing the detection of lung cancer patients harboring anaplastic lymphoma kinase (ALK) gene rearrangements potentially suitable for ALK inhibitor treatment. Clin Cancer Res. 2010; 16:5581–5590.
8. Solomon B, Varella-Garcia M, Camidge DR. ALK gene rearrangements: a new therapeutic target in a molecularly defined subset of non-small cell lung cancer. J Thorac Oncol. 2009; 4:1450–1454.
9. Ou SH, Bartlett CH, Mino-Kenudson M, Cui J, Iafrate AJ. Crizotinib for the treatment of ALK-rearranged non-small cell lung cancer: a success story to usher in the second decade of molecular targeted therapy in oncology. Oncologist. 2012; 17:1351–1375.
10. Wong DW, Leung EL, Wong SK, Tin VP, Sihoe AD, Cheng LC, Au JS, Chung LP, Wong MP. A novel KIF5B-ALK variant in nonsmall cell lung cancer. Cancer. 2011; 117:2709–2718.
11. Takeuchi K, Choi YL, Togashi Y, Soda M, Hatano S, Inamura K, Takada S, Ueno T, Yamashita Y, Satoh Y, Okumura S, Nakagawa K, Ishikawa Y, et al. KIF5B-ALK, a novel fusion oncokinase identified by an immunohistochemistry-based diagnostic system for ALK-positive lung cancer. Clin Cancer Res. 2009; 15: 3143–3149.
12. Togashi Y, Soda M, Sakata S, Sugawara E, Hatano S, Asaka R, Nakajima T, Mano H, Takeuchi K. KLC1-ALK: a novel fusion in lung cancer identified using a formalin-fixed paraffin-embedded tissue only. PLoS One. 2012; 7:e31323.
13. Majewski IJ, Mittempergher L, Davidson NM, Bosma A, Willems SM, Horlings HM, de Rink I, Greger L, Hooijer GK, Peters D, Nederlof PM, Hofland I, de Jong J, et al. Identification of recurrent FGFR3 fusion genes in lung cancer through kinome-centred RNA sequencing. J Pathol. 2013; 230: 270–276.
14. Hong M, Kim RN, Song JY, Choi SJ, Oh E, Lira ME, Mao M, Takeuchi K, Han J, Kim J, Choi YL. HIP1-ALK, a novel fusion protein identified in lung adenocarcinoma. J Thorac Oncol. 2014; 9:419–422.
15. Salido M, Pijuan L, Martinez-Aviles L, Galvan AB, Canadas I, Rovira A, Zanui M, Martinez A, Longaron R, Sole F, Serrano S, Bellosillo B, Wynes MW, et al. Increased ALK gene copy number and amplification are frequent in non-small cell lung cancer. J Thorac Oncol. 2011; 6:21–27.
16. Perner S, Wagner PL, Demichelis F, Mehra R, Lafargue CJ, Moss BJ, Arbogast S, Soltermann A, Weder W, Giordano TJ, Beer DG, Rickman DS, Chinnaiyan AM, et al. EML4-ALK fusion lung cancer: a rare acquired event. Neoplasia. 2008; 10:298–302.
17. Koivunen JP, Mermel C, Zejnullahu K, Murphy C, Lifshits E, Holmes AJ, Choi HG, Kim J, Chiang D, Thomas R, Lee J, Richards WG, Sugarbaker DJ, et al. EML4-ALK fusion gene and efficacy of an ALK kinase inhibitor in lung cancer. Clin Cancer Res. 2008; 14: 4275–4283.
18. Kwak EL, Bang YJ, Camidge DR, Shaw AT, Solomon B, Maki RG, Ou SH, Dezube BJ, Janne PA, Costa DB, Varella-Garcia M, Kim WH, Lynch TJ, et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med. 2010; 363:1693–1703.
19. Shaw AT, Kim DW, Nakagawa K, Seto T, Crino L, Ahn MJ, De Pas T, Besse B, Solomon BJ, Blackhall F, Wu YL, Thomas M, O’Byrne KJ, et al. Crizotinib versus chemotherapy in advanced ALK-positive lung cancer. N Engl J Med. 2013; 368:2385–2394.
20. Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa K, Mekhail T, Felip E, Cappuzzo F, Paolini J, Usari T, Iyer S, Reisman A, Wilner KD, et al. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N Engl J Med. 2014; 371:2167–2177.
21. Seto T, Kiura K, Nishio M, Nakagawa K, Maemondo M, Inoue A, Hida T, Yamamoto N, Yoshioka H, Harada M, Ohe Y, Nogami N, Takeuchi K, et al. CH5424802 (RO5424802) for patients with ALK-rearranged advanced non-small-cell lung cancer (AF-001JP study): a single-arm, open-label, phase 1–2 study. Lancet Oncol. 2013; 14:590–598.
22. Shaw AT, Kim DW, Mehra R, Tan DS, Felip E, Chow LQ, Camidge DR, Vansteenkiste J, Sharma S, De Pas T, Riely GJ, Solomon BJ, Wolf J, et al. Ceritinib in ALK-rearranged non-small-cell lung cancer. N Engl J Med. 2014; 370:1189–1197.
23. Iwama E, Okamoto I, Harada T, Takayama K, Nakanishi Y. Development of anaplastic lymphoma kinase (ALK) inhibitors and molecular diagnosis in ALK rearrangement-positive lung cancer. Onco Targets Ther. 2014; 7:375–385.
24. Katayama R, Shaw AT, Khan TM, Mino-Kenudson M, Solomon BJ, Halmos B, Jessop NA, Wain JC, Yeo AT, Benes C, Drew L, Saeh JC, Crosby K, et al. Mechanisms of acquired crizotinib resistance in ALK-rearranged lung Cancers. Sci Transl Med. 2012; 4:120ra117.
25. Weickhardt AJ, Aisner DL, Franklin WA, Varella-Garcia M, Doebele RC, Camidge DR. Diagnostic assays for identification of anaplastic lymphoma kinase-positive non-small cell lung cancer. Cancer. 2013; 119:1467–1477.
26. Sholl LM, Weremowicz S, Gray SW, Wong KK, Chirieac LR, Lindeman NI, Hornick JL. Combined use of ALK immunohistochemistry and FISH for optimal detection of ALK-rearranged lung adenocarcinomas. J Thorac Oncol. 2013; 8:322–328.
27. Karachaliou N, Rosell R. Optimal detection of ALK rearranged lung adenocarcinomas. J Thorac Oncol. 2013; 8:255–256.
28. Cabillic F, Gros A, Dugay F, Begueret H, Mesturoux L, Chiforeanu DC, Dufrenot L, Jauffret V, Dachary D, Corre R, Lespagnol A, Soler G, Dagher J, et al. Parallel FISH and immunohistochemical studies of ALK status in 3244 non-small-cell lung cancers reveal major discordances. J Thorac Oncol. 2014; 9:295–306.
29. Shan L, Lian F, Guo L, Yang X, Ying J, Lin D. Combination of conventional immunohistochemistry and qRT-PCR to detect ALK rearrangement. Diagn Pathol. 2014; 9:3.
30. Wallander ML, Geiersbach KB, Tripp SR, Layfield LJ. Comparison of reverse transcription-polymerase chain reaction, immunohistochemistry, and fluorescence in situ hybridization methodologies for detection of echinoderm microtubule-associated proteinlike 4-anaplastic lymphoma kinase fusion-positive non-small cell lung carcinoma: implications for optimal clinical testing. Arch Pathol Lab Med. 2012; 136:796–803.
31. Savic S, Diebold J, Zimmermann AK, Jochum W, Baschiera B, Grieshaber S, Tornillo L, Bisig B, Kerr K, Bubendorf L. Screening for ALK in non-small cell lung carcinomas: 5A4 and D5F3 antibodies perform equally well, but combined use with FISH is recommended. Lung Cancer. 2015; 89:104–109.
32. Sasaki T, Rodig SJ, Chirieac LR, Janne PA. The biology and treatment of EML4-ALK non-small cell lung cancer. Eur J Cancer. 2010; 46:1773–1780.
33. Ying J, Guo L, Qiu T, Shan L, Ling Y, Liu X, Lu N. Diagnostic value of a novel fully automated immunochemistry assay for detection of ALK rearrangement in primary lung adenocarcinoma. Ann Oncol. 2013; 24:2589–2593.
34. Wynes MW, Sholl LM, Dietel M, Schuuring E, Tsao MS, Yatabe Y, Tubbs RR, Hirsch FR. An international interpretation study using the ALK IHC antibody D5F3 and a sensitive detection kit demonstrates high concordance between ALK IHC and ALK FISH and between evaluators. J Thorac Oncol. 2014; 9:631–638.
35. von Laffert M, Warth A, Penzel R, Schirmacher P, Kerr KM, Elmberger G, Schildhaus HU, Buttner R, Lopez-Rios F, Reu S, Kirchner T, Pauwels P, Specht K, et al. Multicenter immunohistochemical ALK-testing of non-small-cell lung cancer shows high concordance after harmonization of techniques and interpretation criteria. J Thorac Oncol. 2014; 9:1685–1692.
36. Pekar-Zlotin M, Hirsch FR, Soussan-Gutman L, Ilouze M, Dvir A, Boyle T, Wynes M, Miller VA, Lipson D, Palmer GA, Ali SM, Dekel S, Brenner R, et al. Fluorescence in situ hybridization, immunohistochemistry, and next-generation sequencing for detection of EML4-ALK rearrangement in lung cancer. Oncologist. 2015; 20: 316–322.
37. Takeuchi K, Choi YL, Soda M, Inamura K, Togashi Y, Hatano S, Enomoto M, Takada S, Yamashita Y, Satoh Y, Okumura S, Nakagawa K, Ishikawa Y, et al. Multiplex reverse transcription-PCR screening for EML4-ALK fusion transcripts. Clin Cancer Res. 2008; 14:6618–6624.
38. Soda M, Isobe K, Inoue A, Maemondo M, Oizumi S, Fujita Y, Gemma A, Yamashita Y, Ueno T, Takeuchi K, Choi YL, Miyazawa H, Tanaka T, et al. A prospective PCR-based screening for the EML4-ALK oncogene in non-small cell lung cancer. Clin Cancer Res. 2012; 18:5682–5689.
39. Wang R, Pan Y, Li C, Hu H, Zhang Y, Li H, Luo X, Zhang J, Fang Z, Li Y, Shen L, Ji H, Garfield D, et al. The use of quantitative real-time reverse transcriptase PCR for 5′ and 3′ portions of ALK transcripts to detect ALK rearrangements in lung cancers. Clin Cancer Res. 2012; 18:4725–4732.
40. Gruber K, Horn H, Kalla J, Fritz P, Rosenwald A, Kohlhaufl M, Friedel G, Schwab M, Ott G, Kalla C. Detection of rearrangements and transcriptional up-regulation of ALK in FFPE lung cancer specimens using a novel, sensitive, quantitative reverse transcription polymerase chain reaction assay. J Thorac Oncol. 2014; 9:307–315.
41. Kotoula V, Bobos M, Vassilakopoulou M, Tsolaki E, Chrisafi S, Psyrri A, Lazaridis G, Papadopoulou K, Efstratiou I, Michail-Strantzia C, Debelenko LV, Kosmidis P, Fountzilas G. Intact or broken-apart RNA: an alternative concept for ALK fusion screening in non-small cell lung cancer (NSCLC). Appl Immunohistochem Mol Morphol. 2015; 23:60–70.
42. Lira ME, Kim TM, Huang D, Deng S, Koh Y, Jang B, Go H, Lee SH, Chung DH, Kim WH, Schoenmakers EF, Choi YL, Park K, et al. Multiplexed gene expression and fusion transcript analysis to detect ALK fusions in lung cancer. J Mol Diagn. 2013; 15:51–61.
43. Moskalev EA, Frohnauer J, Merkelbach-Bruse S, Schildhaus HU, Dimmler A, Schubert T, Boltze C, Konig H, Fuchs F, Sirbu H, Rieker RJ, Agaimy A, Hartmann A, et al. Sensitive and specific detection of EML4-ALK rearrangements in non-small cell lung cancer (NSCLC) specimens by multiplex amplicon RNA massive parallel sequencing. Lung Cancer. 2014; 84:215–221.
44. Mandrekar JN AC. Inverse Prediction Using SAS® Software: A Clinical Application. Paper P02-2008 Division of Biostatistics, Mayo Clinic, Rochester, MN. 2008.
45. Camidge DR, Theodoro M, Maxson DA, Skokan M, O’Brien T, Lu X, Doebele RC, Baron AE, Varella-Garcia M. Correlations between the percentage of tumor cells showing an anaplastic lymphoma kinase (ALK) gene rearrangement, ALK signal copy number, and response to crizotinib therapy in ALK fluorescence in situ hybridization-positive nonsmall cell lung cancer. Cancer. 2012; 118:4486–4494.
46. Sun JM, Choi YL, Won JK, Hirsch FR, Ahn JS, Ahn MJ, Park K. A dramatic response to crizotinib in a non-small-cell lung cancer patient with IHC-positive and FISH-negative ALK. J Thorac Oncol. 2012; 7:e36–38.
47. Chihara D, Suzuki R. More on crizotinib. N Engl J Med. 2011; 364:776–777; author reply 778.
48. Wiesner T, Lee W, Obenauf AC, Ran L, Murali R, Zhang QF, Wong EW, Hu W, Scott SN, Shah RH, Landa I, Button J, Lailler N, et al. Alternative transcription initiation leads to expression of a novel ALK isoform in cancer. Nature. 2015; 526:453–457.
49. Pfarr N, Stenzinger A, Penzel R, Warth A, Dienemann H, Schirmacher P, Weichert W, Endris V. High-throughput diagnostic profiling of clinically actionable gene fusions in lung cancer. Genes Chromosomes Cancer. 2015; 55:30–44.
50. Uphoff CC, Drexler HG. Comparative PCR analysis for detection of mycoplasma infections in continuous cell lines. In Vitro Cell Dev Biol Anim. 2002; 38:79–85.
51. Tukey J. Exploratory Data Analysis. Addison-Wesley. 1977:43–44.

