INTRODUCTION
Gastric cancer is the third leading cause of cancer-related death worldwide. Identifying effective pathway inhibition is a key aspect of the development of targeted therapeutics. The success of trastuzumab in HER2-positive gastric cancer patients has elicited efforts to discover new molecular targets in gastric cancer.
The nuclear factor-kappa B (NF-κB) pathway is a key regulator that activates transcription of genes involved the inflammatory immune response, proliferation, cell survival and cell invasion. Activation of NF-kB is frequently observed in solid tumors and hematological malignancies [1]. NF-κB complexes are retained in the cytoplasm by a family of NF-κB-binding proteins known as inhibitors of NF-κB (IκBs) [1]. Various stimulants trigger activation of the IKK (IκBs kinase complex), leading to proteasomal degradation of IκBs. Consequently, NF-κB dimers are released in the nucleus and induce transcription of target genes related to inflammation and survival.
The IKK family can be divided into two groups: canonical IKKs (IKKα, IKKβ and a nonenzymatic regulatory component, IKKγ/NEMO) and non-canonical IKKs (IKKε and TBK1) [2]. Although IKKε and TANK-binding kinase 1 (TBK1) are characterized as activators of NF-κB, they are not essential for NF-κB activation [3, 4]. Instead, IKKε and TBK1 play critical roles in antiviral response via phosphorylation and activation of transcription factors IRF3, IRF7 and STAT1 [1, 5]. Furthermore, these non-canonical IKKs are also involved in the survival, tumorigenesis and development of various cancers [6–8]. Although IKKε expression is restricted to particular tissues, such as lymphoid tissues, peripheral blood lymphocytes and the pancreas [9], TBK1 is constitutively expressed in many normal tissues, including the immune cells, brain, lungs, gastrointestinal tract, and reproductive organs [10]. IKKε is overexpressed through multiple mechanisms in various human cancers, such as breast, ovarian, and prostate cancer. IKKε overexpression in breast and ovarian cancer was increased due to amplification or unknown mutations regulating IKKε transcript levels [2, 11]. IKKε has been identified as an oncogene in breast [2] and ovarian cancer [11, 12] and is associated with poor prognosis [7, 13]. Recent reports suggested that overexpression of IKKε may play a role in tumorigenesis of prostatic and esophageal squamous cell carcinoma [14, 15]. TBK1’s role in cancer may be due to its involvement in regulation of cell growth and proliferation, angiogenesis and oncogenic transformation [8, 16–18]. Through functional genomics, Barbie et al. [6] identified that TBK1 was essential for KRAS mutant cancer cell lines. However, subsequent studies found no relationship between oncogenic KRAS and TBK1 [19].
Thus far, the expression of IKKε and TBK1 in gastric cancer and their role in prognosis have not been studied. To investigate overexpression of IKKε and TBK1 in gastric cancer and their relation to clinicopathologic factors, we performed immunohistochemical staining in 1,107 resected gastric cancers using a tissue microarray approach.
RESULTS
Expression of IKKε and TBK1 in gastric cancer
Expression of IKKε and TBK1 was observed in 13.6% (150/1107) and 3.4% (38/1107) of gastric cancer patients, respectively. Associations between IKKε and TBK1 expression and clinicopathological factors were evaluated (Table 1). IKKε and TBK1 expression were correlated with histologic differentiation and histologic type by Lauren classification. Differentiated tumors and intestinal-type gastric cancer by Lauren classification showed increased IKKε and TBK1 expression compared to undifferentiated and diffuse types. Expression of IKKε and TBK1 was associated with earlier AJCC stage (based on AJCC seventh edition; p=0.019 and p=0.003, respectively). Expression of IKKε was observed in 16.7%, 15.9% and 10.2% of stage I, II, and III tumors, respectively. Expression of TBK1 was observed in 8.3%, 4.1% and 1.7% of stage I, II, and III tumors, respectively.
Table 1: The association of IKK and TBK1 expression and clinicopathological factors
IKK expression |
TBK1 expression |
|||||||
|---|---|---|---|---|---|---|---|---|
Total No. of cases |
Positive |
Negative |
Positive |
Negative |
||||
n=1107 (%) |
n=150 (13.6%) |
n=957 (86.4%) |
P -value |
n=38 (3.4%) |
n=1069 (96.6%) |
P -value |
||
Gender |
0.002 |
0.076 |
||||||
Male |
725 (65.5%) |
115 (15.9%) |
610 (84.1%) |
30 (4.1%) |
695 (95.9%) |
|||
Female |
382 (34.5%) |
35 (9.2%) |
347 (90.8%) |
8 (2.1%) |
374 (97.9%) |
|||
Age (years) |
<0.001 |
0.058 |
||||||
<60 |
792 (71.5%) |
88 (11.1%) |
704 (88.9%) |
22 (2.8%) |
770 (97.2%) |
|||
≥60 |
315 (28.5%) |
62 (19.7%) |
253 (80.3%) |
16 (5.1%) |
299 (94.9%) |
|||
Tumor location |
0.461 |
0.382 |
||||||
Upper third |
117 (10.6%) |
18 (15.4%) |
99 (84.6%) |
1 (0.9%) |
116 (99.1%) |
|||
Middle third |
315 (28.5%) |
36 (11.4%) |
279 (88.6%) |
10 (3.2%) |
305 (96.8%) |
|||
Lower third |
626 (56.5%) |
87 (13.9%) |
539 (86.1%) |
25 (4.0%) |
601 (96.0%) |
|||
Whole |
49 (4.4%) |
9 (18.4%) |
40 (81.6%) |
2 (4.1%) |
47 (95.9%) |
|||
Lauren classification |
<0.001 |
<0.001 |
||||||
Intestinal |
319 (28.8%) |
72 (22.6%) |
247 (77.4%) |
19 (6.0%) |
300 (94.0%) |
|||
Diffuse |
766 (69.2%) |
70 (9.1%) |
696 (90.9%) |
16 (2.1%) |
750 (97.9%) |
|||
Mixed |
22 (2.0%) |
8 (36.4%) |
14 (63.6%) |
3 (13.6%) |
19 (86.4%) |
|||
Histology |
<0.001 |
0.001 |
||||||
Differentiated |
303 (37.4%) |
70 (23.1%) |
233 (76.9%) |
19 (6.3%) |
284 (93.7%) |
|||
Undifferentiated |
804 (72.6%) |
80 (10.0%) |
724 (90.0%) |
19 (2.4%) |
785 (97.6%) |
|||
T stage |
0.081 |
0.011 |
||||||
T1 |
108 (9.8%) |
19 (17.6%) |
89 (82.4%) |
9 (8.3%) |
99 (91.7%) |
|||
T2 |
124 (11.2%) |
22 (17.7%) |
102 (82.3%) |
6 (4.8%) |
118 (95.2%) |
|||
T3 |
686 (62.0%) |
92 (13.4%) |
594 (86.6%) |
20 (2.9%) |
666 (97.1%) |
|||
T4 |
189 (17.1%) |
17 (9.0%) |
172 (91.0%) |
3 (1.6%) |
186 (98.4%) |
|||
N stage |
0.109 |
0.233 |
||||||
N0 |
102 (9.2%) |
10 (9.8%) |
92 (90.2%) |
3 (2.9%) |
99 (97.1%) |
|||
N1 |
558 (50.4%) |
89 (15.9%) |
469 (84.1%) |
25 (4.5%) |
533 (95.5%) |
|||
N2 |
289 (26.1%) |
35 (12.1%) |
254 (87.9%) |
8 (2.8%) |
281 (97.2%) |
|||
N3 |
158 (14.3%) |
16 (10.1%) |
142 (89.9%) |
2 (1.3%) |
156 (98.7%) |
|||
AJCC 7th stage |
0.019 |
0.003 |
||||||
I |
96 (8.7%) |
16 (16.7%) |
80 (83.3%) |
8 (8.3%) |
88 (91.7%) |
|||
II |
540 (48.8%) |
86 (15.9%) |
454 (84.1%) |
22 (4.1%) |
518 (95.9%) |
|||
III |
471 (42.5%) |
48 (10.2%) |
423 (89.8%) |
8 (1.7%) |
463 (98.3%) |
|||
Recurrence of disease |
0.029 |
0.356 |
||||||
Yes |
429 (38.8%) |
46 (10.7%) |
383 (89.3%) |
12 (2.8%) |
417 (97.2%) |
|||
No |
678 (61.2%) |
104 (15.3%) |
574 (84.7%) |
26 (3.8%) |
652 (96.2%) |
|||
Death of disease |
0.128 |
0.544 |
||||||
Yes |
401 (36.2%) |
46 (11.5%) |
355 (88.5%) |
12 (3.0%) |
389 (97.0%) |
|||
No |
706 (63.8%) |
104 (14.7%) |
602 (85.3%) |
26 (3.7%) |
680 (96.3%) |
|||
There was a significant association between IKKε and TBK1 expression (p<0.001), as 97.8% (936/957) of tumors with IKKε negativity showed negative expression of TBK1 (Table 2).
Table 2: Association between the IKK and TBK1 expression
Variable |
IKK expression |
||
|---|---|---|---|
Positive (n=150) |
Negative (n=957) |
P - value |
|
TBK1 expression |
<0.001 |
||
Positive (n=38) |
17 (11.3%) |
21 (2.2%) |
|
Negative (n=1069) |
133 (88.7%) |
936 (97.8%) |
|
Co-expression of IKK and TBK1 in gastric cancer
We classified IKKε and TBK1 expression status into four subgroups as follows: IKKε-/TBK1- (n=936, 84.6%); IKKε+/TBK1- (n=133, 12.0%); IKKε-/TBK1+ (n=21, 1.9%); IKKε+/TBK1+ (n=17, 1.5%) (Figure 1). Clinicopathological characteristics among these four groups were also evaluated (Table 3). Intestinal-type gastric cancer on Lauren classification and differentiated tumors were more frequent in the IKKε+/TBK1+ subgroup than in the IKKε-/TBK1-subgroup (p<0.001 and p<0.001, respectively). Although N stage was not significantly associated with expression of IKKε and TBK1, patients in the IKKε+/TBK1+ subgroup were more likely to have earlier T stage and lower AJCC stage than those in the IKKε-/TBK1-subgroup (p=0.011 and p=0.002, respectively).
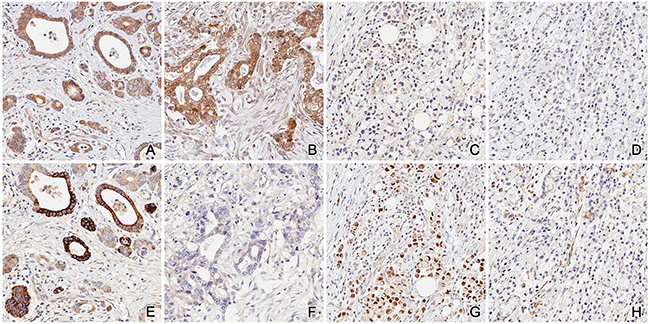
Figure 1: Representative examples of the four subgroups A. & E. IKK+/TBK1+; B. & F. IKK+/TBK1-; C. & G. IKK-/TBK1+; D. & H. IKK-/TBK1-.
Table 3: The association of co-expressions of IKK and TBK1 and clinicopathological factors
IKK-/TBK1- |
IKK+/TBK1- |
IKK-/TBK1+ |
IKK+/TBK1+ |
P – value |
||
|---|---|---|---|---|---|---|
N=936 (84.6%) |
N=133 (12.0%) |
N=21 (1.9%) |
N=17 (1.5%) |
|||
Gender |
0.01 |
|||||
Male |
594 (81.9%) |
101 (13.9%) |
16 (2.2%) |
14 (1.9%) |
||
Female |
342 (89.5%) |
32 (8.4%) |
5 (1.3%) |
3 (0.8%) |
||
Age (years) |
0.001 |
|||||
<60 |
691 (87.2%) |
79 (10.0%) |
13 (1.6%) |
9 (1.1%) |
||
≥60 |
245 (77.8%) |
54 (17.1%) |
8 (2.5%) |
8 (2.5%) |
||
Tumor location |
0.382 |
|||||
Upper third |
99 (84.6%) |
17 (14.5%) |
0 (0%) |
1 (0.9%) |
||
Middle third |
275 (87.3%) |
30 (9.5%) |
4 (1.3%) |
6 (1.9%) |
||
Lower third |
524 (83.7%) |
77 (12.3%) |
15 (2.4%) |
10 (1.6%) |
||
Whole |
38 (77.6%) |
9 (18.4%) |
2 (4.1%) |
0 (0%) |
||
Lauren classification |
<0.001 |
|||||
Intestinal |
238 (74.6%) |
62 (19.4%) |
9 (2.8%) |
10 (3.1%) |
||
Diffuse |
685 (89.4%) |
65 (8.5%) |
11 (1.4%) |
5 (0.7%) |
||
Mixed |
13 (59.1%) |
6 (27.3%) |
1 (4.5%) |
2 (9.1%) |
||
Histology |
<0.001 |
|||||
Differentiated |
225 (74.3%) |
59 (19.5%) |
8 (2.6%) |
11 (3.6%) |
||
Undifferentiated |
711 (88.4%) |
74 (9.2%) |
13 (1.6%) |
6 (0.7%) |
||
T stage |
0.011 |
|||||
T1 |
85 (78.7%) |
14 (13.0%) |
4 (3.7%) |
5 (4.6%) |
||
T2 |
100 (80.6%) |
18 (14.5%) |
2 (1.6%) |
4 (3.2%) |
||
T3 |
581 (84.7%) |
85 (12.4%) |
13 (1.9%) |
7 (1.0%) |
||
T4 |
170 (89.9%) |
16 (8.5%) |
2 (1.1%) |
1 (0.5%) |
||
N stage |
0.069* |
|||||
N0 |
90 (88.2%) |
9 (8.8%) |
2 (2.0%) |
1 (1.0%) |
||
N1 |
457 (81.9%) |
76 (13.6%) |
12 (2.2%) |
13 (2.3%) |
||
N2 |
248 (85.8%) |
33 (11.4%) |
6 (2.1%) |
2 (0.7%) |
||
N3 |
141 (89.2%) |
15 (9.5%) |
1 (0.6%) |
1 (0.6%) |
||
AJCC 7th stage |
0.002 |
|||||
I |
76 (8.1%) |
12 (9.0%) |
4 (19.0%) |
4 (23.5%) |
||
II |
444 (47.4%) |
74 (55.6%) |
10 (47.6%) |
12 (70.6%) |
||
III |
416 (44.4%) |
47 (35.3%) |
7 (33.3%) |
1 (5.9%) |
||
Recurrence of disease |
0.101 |
|||||
Yes |
374 (87.2%) |
43 (10.0%) |
9 (1.8%) |
3 (0.7%) |
||
No |
562 (82.9%) |
90 (13.3%) |
12 (2.1%) |
14 (2.1%) |
||
Death of disease |
0.258 |
|||||
Yes |
346 (86.3%) |
43 (10.7%) |
9 (2.2%) |
3 (0.7%) |
||
No |
590 (83.6%) |
90 (12.7%) |
12 (1.7%) |
14 (2.0%) |
||
In univariate binary logistic regression analysis, several clinicopathologic variables were related to expression of IKKε+/TBK1+, including intestinal and differentiated histologic types, earlier T stage, and earlier AJCC stage. Among these variables, histology type and AJCC stage were significant predictors in multivariate analysis. The adjusted odds ratio (OR) of differentiated histology was 4.579, with a 95% CI 1.669-12.566 when compared with undifferentiated histology. The ORs of AJCC stage I and stage II were 18.914 and 9.935 (95% CI, 1.995-165 and 1.283-76.913) when compared with stage III (Table 4).
Table 4: Multivariate analysis of clinicopathological factors for co-expression of IKK/TBK1 in gastric cancer
Odds ratio |
95% CI |
P - value |
|
|---|---|---|---|
Histology |
0.003 |
||
Undifferentiated |
1.000 |
||
Differentiated |
4.579 |
1.669 - 12.566 |
|
AJCC 7th stage |
0.037 |
||
Stage I |
18.194 |
1.995 - 165.910 |
0.010 |
Stage II |
9.935 |
1.283 - 76.913 |
0.028 |
Stage III |
1.000 |
Prognostic significance of IKK and TBK1 co-expression in gastric cancer
Overall, the mean follow-up period was 79.8, 73.8 and 54.6 months in AJCC stage I, II, and III, respectively. During the follow-up periods, 10.4%, 25.4% and 59.9% of patients in stage I, II and III had a recurrence and 8.3%, 22.8% and 57.3% of patients in stage I, II and III died of their disease.
Patients in the IKKε+/TBK1+ subgroup showed longer overall survival (mean=114.7 months; 95% CI 107.3-118.9) than those in the IKKε-/TBK1- subgroup (mean=113.2 months; 95% CI 108.7-117.8; p=0.125), IKKε+/TBK1- subgroup (mean=118.1 months; 95% CI 107.4-128.7; p=0.235), and IKKε-/TBK1+ subgroup (mean=98.1 months; 95% CI 71.5-124.7; p=0.094). The IKKε+/TBK1+ subgroup showed longer disease-free survival (mean 125.8 months; 95% CI 101.5-150.0) than the IKKε-/TBK1- subgroup (mean=105.1 months; 95% CI 100.1-110.1; p=0.104), IKKε +/TBK1- subgroup (mean=104.6 months; 95% CI 93.7-115.5; p=0.240), and IKKε-/TBK1+ subgroup (mean=94.6 months; 95% CI 66.1-123.0; p=0.110). When survival curves were compared by log-rank test in Kaplan-Meier survival analyses, there were no survival differences in relation to expression of IKKε or TBK1. However, the survival curve of the IKKε+/TBK1+ subgroup varied from those of other groups, although this did not reach statistical significance due to the small number of events in the IKKε+/TBK1+ subgroup (Figure 2).
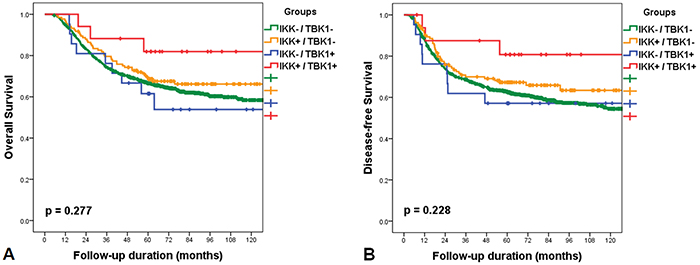
Figure 2: Kaplan-Meier survival curves with log-rank test of overall survival after classification into the following four subgroups: IKK-/TBK1-; IKK+/TBK1-; IKK-/TBK1+; IKK+/TBK1+. The survival curve of the IKK+/TBK1+ subgroup was different from those of other groups, but this difference was not statistically significant.
DISCUSSION
Our study sought to investigate IKKε and TBK1 expression in gastric cancer and their role in prognosis. We identified IKKε and TBK1 expression in 13.6% and 3.4% of gastric cancers, respectively, and co-expression of IKKε and TBK1 in 1.5% of cases. Our findings suggest that IKKε and TBK1 may be good molecular target candidates, warranting future study to elucidate the underlying common regulatory mechanism.
Identifying specific biomarkers for patient selection and effective pathway inhibition is a key element in the development of targeted therapies. In particular, kinase oncogenes may be attractive therapeutic targets. IKKε and TBK1 are serine/threonine protein kinases belonging to the IKK family. Although IKKε and TBK1 exhibit differential expression patterns, they share the kinase domain and are similar in their ability to activate the NF-kB signaling pathway. NF-kB signaling pathway activation may be related to distinct mechanisms in different tumor types. Basically, NF-kBs affect cell survival and proliferation in cancer by inducing expression of genes coding for key anti-apoptotic proteins, such as Bcl-2 and IAP-1/2 and mitogenic genes, such as myc and cyclin-D. Hence, the function of the NF-kB signaling pathway is to protect cancer cells from apoptosis and drive their proliferation. Finally, IKKε and TBK1 are kinase oncogenes. Previous studies revealed that IKKε and TBK1 play a significant role in several cancers. Boehm et al. showed that IKKε is amplified and overexpressed in a breast cancer cell line and human breast cancer tissue [7]. Guo et al. recently showed that overexpression of IKKε in ovarian cancer was associated with late-stage and high-grade tumors [11]. Recently, Deng et al. [23] reported that patients with HER2-positive breast cancer may benefit from anti-TBK1/IKKε plus anti-HER2 combination therapies. TBK1/IKKε inhibition promoted cellular senescence by suppressing p65–NF-kB and inducing p16Ink4a. Although IKKε is not essential for growth of mouse Her2/Neu tumor cells, shRNA-mediated knockdown of TBK1 alone efficiently inhibited growth of both mouse and human HER2-positive breast cancer cells. Thus, TBK1 could be critical for survival and growth of tumors with HER2 amplification [23].
The oncogenic potential of IKKε and TBK1 indicate these proteins to be possible therapeutic targets. TBK1/IKKε inhibitors have shown low specificity, as they have multiple targets such as PDK1, JNK and p38 MAP kinases [24, 25]. Recently, Reilly et al. [26] discovered a small molecule inhibitor of IKKε and TBK1 kinases called amlexanox, which has been shown to selectively inhibit both IKKε and TBK1.
In our study, the multivariate binary logistic regression model was applied to determine prediction factors for IKKε+/TBK1+ expression in gastric cancer. Multivariate analysis showed that tissues with differentiated histology and earlier AJCC stage were correlated with IKKε+/TBK1+ expression. The frequency of IKKε and TBK1 co-expression was relatively high in early T stage, suggesting that alteration of IKKε and TBK1 could be more involved in the development of gastric cancer. Furthermore, co-expression of IKKε and TBK1 was associated with more differentiated histology, namely, intestinal-type gastric cancer. In this study, patients in the IKKε+/TBK1+ subgroup had a longer life span. Co-expression of IKKε and TBK1 was more frequent in early T stage tumors and those with more differentiated (intestinal-type) histology, which might have been related to good prognosis.
Recent studies have classified four major genomic groups of gastric cancer on a molecular and genomic basis: EBV-infected tumors, those with microsatellite instability, genomically-stable tumors, and those with chromosomal instability. Chromosomal-instability tumors were of the intestinal histology type [27]. Recently, we also classified gastric cancer into four molecular subtypes, which are closely associated with distinct clinical outcomes [28]. However, traditionally, gastric cancer is divided into two main subtypes on the basis of Lauren classification–intestinal and diffuse. These subtypes have different molecular pathogenesis. In the intestinal type, multistep progression initiated by Helicobacter pylori infection is associated with pathogenesis. Preferentially altered genes includeKRAS and HER2, which are overexpressed in about 20% of gastric cancer [29–32]. Diffuse-type gastric cancer does not arise from step-wise progression and is associated with loss of cell cohesion due to biallelic inactivation of CDH1. Sporadically altered genes include BCL2 and FGFR2 in diffuse-type gastric carcinomas [33–36]. We demonstrated that alteration of IKKε and TBK1, albeit small, may be involved in the pathogenesis of intestinal-type gastric cancer. Thus, testing for IKKε and TBK1 overexpression should be considered in certain patients, such as those with intestinal-type gastric cancer.
We did not observe statistically significant survival differences between the four IKKε and TBK1 expression subgroups. However, the survival curve of the IKKε+/TBK1+ subgroup varied compared to other groups. This finding may be due to the small number of events in the IKKε+/TBK1+ subgroup. Our results do not agree with those of several previous studies. This discrepancy could be due to the differing roles of IKKε and TBK1 in gastric cancer or the small number of positive cases. Further investigation is needed to determine its role in gastric carcinogenesis.
To the best of our knowledge, this is the first large-scale study investigating the relationship between the expression of IKKε and TBK1 and clinicopathologic features of gastric cancer. We determined the expression of IKKε and TBK1, and co-expression of IKKε and TBK1 was associated with differentiated intestinal histology and earlier tumor stage. The role of IKKε and TBK1 in intestinal-type gastric cancer pathogenesis should be elucidated by further study.
MATERIALS AND METHODS
Patients
Gastric cancer tissue samples were retrospectively collected from 1,107 patients (stages IB to IVa) who underwent R0 gastrectomy with extensive node dissection (D2) and adjuvant chemoradiation therapy (INT-0116 regimen) [20, 21] from 2000 to 2008 at Samsung Medical Center in Seoul, Korea. Clinicopathological characteristics obtained from medical records included sex, age, tumor size, tumor location, histological type, Lauren classification, and differentiation grade. Tumor histology was classified into 2 groups: differentiated, which included well- or moderately-differentiated tubular and papillary adenocarcinomas, and undifferentiated, which included poorly-differentiated adenocarcinomas and signet ring cell carcinomas.
Immunohistochemistry
For tissue microarray, we reviewed all H&E-stained slides and representative histological areas were carefully selected and marked on paraffin blocks. From each paraffin block, four primary gastric cancer tissue cores (diameter = 0.6 mm) were taken from the invasive front, both lateral sides, and the luminal surface area of the tumor using AccuMax (IsuAbxis, Seoul, Korea) as previously described [22]. Immunohistochemistry was performed on formalin-fixed, paraffin-embedded, 4-μm-thick tissue sections using rabbit monoclonal antibody IKKε (D20G4, Cell Signaling Technology, Danvers, MA, USA, 1:50 dilution) and TBK1/NAK (D1B4, Cell Signaling Technology, Danvers, MA, USA, 1:200 dilution). For IKKε, we incubated primary antibody overnight at 4°C and used a DAKO Envision™ Detection Kit (DAKO, Glostrup, Denmark) for 30 minutes. For TBK1, we incubated primary antibody for 15 minutes with Bond-max autoimmunostainer (Leica Biosystem, Melbourne, Australia) using Bond™ Polymer refine detection (DS9800, Vision Biosystems, Melbourne, Australia) according to the manufacturer’s protocol. For the interpretation of IKKε and TBK1 Immunohistochemistry, strong, distinct cytoplasmic staining with membranous accentuation was considered positive.
Statistical analysis
Statistical analysis was performed by SPSS 19.0 for Windows (SPSS, Chicago, IL, USA). Categorical variables were compared using Pearson’s chi-squared test or Fisher’s exact test, and continuous variables, which are presented as means ± SD, using the t-test. Factors found to be significant (p<0.05) in univariate analysis were included in subsequent multivariate logistic regression analysis to identify independent variables associated with IKK and TBK1 expression. Disease-free survival was defined as the time from surgery to first relapse. The Kaplan–Meier method was used to calculate disease-free and overall survival, and survival curves were compared by log-rank test. All tests were two sided, and p values <0.05 were considered statistically significant.
ACKNOWLEDGMENTS
This research was supported by a grant from the 20 by 20 project of Samsung Medical Center (GF01140111).
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
REFERENCES
1. Shen RR and Hahn WC. Emerging roles for the non-canonical IKKs in cancer. Oncogene. 2011; 30: 631-641.
2. Verhelst K, Verstrepen L, Carpentier I and Beyaert R. IkappaB kinase epsilon (IKKepsilon): a therapeutic target in inflammation and cancer. Biochem Pharmacol. 2013; 85: 873-880.
3. Bonnard M, Mirtsos C, Suzuki S, Graham K, Huang J, Ng M, Itie A, Wakeham A, Shahinian A, Henzel WJ, Elia AJ, Shillinglaw W, Mak TW, et al. Deficiency of T2K leads to apoptotic liver degeneration and impaired NF-kappaB-dependent gene transcription. EMBO J. 2000; 19: 4976-4985.
4. Hemmi H, Takeuchi O, Sato S, Yamamoto M, Kaisho T, Sanjo H, Kawai T, Hoshino K, Takeda K and Akira S. The roles of two IkappaB kinase-related kinases in lipopolysaccharide and double stranded RNA signaling and viral infection. J Exp Med. 2004; 199: 1641-1650.
5. Clement JF, Meloche S and Servant MJ. The IKK-related kinases: from innate immunity to oncogenesis. Cell Res. 2008; 18: 889-899.
6. Barbie DA, Tamayo P, Boehm JS, Kim SY, Moody SE, Dunn IF, Schinzel AC, Sandy P, Meylan E, Scholl C, Frohling S, Chan EM, Sos ML, et al. Systematic RNA interference reveals that oncogenic KRAS-driven cancers require TBK1. Nature. 2009; 462: 108-112.
7. Boehm JS, Zhao JJ, Yao J, Kim SY, Firestein R, Dunn IF, Sjostrom SK, Garraway LA, Weremowicz S, Richardson AL, Greulich H, Stewart CJ, Mulvey LA, et al. Integrative genomic approaches identify IKBKE as a breast cancer oncogene. Cell. 2007; 129: 1065-1079.
8. Chien Y, Kim S, Bumeister R, Loo YM, Kwon SW, Johnson CL, Balakireva MG, Romeo Y, Kopelovich L, Gale M, Jr., Yeaman C, Camonis JH, Zhao Y, et al. RalB GTPase-mediated activation of the IkappaB family kinase TBK1 couples innate immune signaling to tumor cell survival. Cell. 2006; 127: 157-170.
9. Shimada T, Kawai T, Takeda K, Matsumoto M, Inoue J, Tatsumi Y, Kanamaru A and Akira S. IKK-i, a novel lipopolysaccharide-inducible kinase that is related to IkappaB kinases. Int Immunol. 1999; 11: 1357-1362.
10. Larabi A, Devos JM, Ng SL, Nanao MH, Round A, Maniatis T and Panne D. Crystal structure and mechanism of activation of TANK-binding kinase 1. Cell Rep. 2013; 3: 734-746.
11. Guo JP, Shu SK, He L, Lee YC, Kruk PA, Grenman S, Nicosia SV, Mor G, Schell MJ, Coppola D and Cheng JQ. Deregulation of IKBKE is associated with tumor progression, poor prognosis, and cisplatin resistance in ovarian cancer. Am J Pathol. 2009; 175: 324-333.
12. Hsu S, Kim M, Hernandez L, Grajales V, Noonan A, Anver M, Davidson B and Annunziata CM. IKK-epsilon coordinates invasion and metastasis of ovarian cancer. Cancer Res. 2012; 72: 5494-5504.
13. Qin B and Cheng K. Silencing of the IKKepsilon gene by siRNA inhibits invasiveness and growth of breast cancer cells. Breast Cancer Res. 2010; 12: R74.
14. Kang MR, Kim MS, Kim SS, Ahn CH, Yoo NJ and Lee SH. NF-kappaB signalling proteins p50/p105, p52/p100, RelA, and IKKepsilon are over-expressed in oesophageal squamous cell carcinomas. Pathology. 2009; 41: 622-625.
15. Seo SI, Song SY, Kang MR, Kim MS, Oh JE, Kim YR, Lee JY, Yoo NJ and Lee SH. Immunohistochemical analysis of NF-kappaB signaling proteins IKKepsilon, p50/p105, p52/p100 and RelA in prostate cancers. APMIS. 2009; 117: 623-628.
16. Kim JY, Welsh EA, Oguz U, Fang B, Bai Y, Kinose F, Bronk C, Remsing Rix LL, Beg AA, Rix U, Eschrich SA, Koomen JM and Haura EB. Dissection of TBK1 signaling via phosphoproteomics in lung cancer cells. Proc Natl Acad Sci U S A. 2013; 110: 12414-12419.
17. Kim JY, Beg AA and Haura EB. Non-canonical IKKs, IKK and TBK1, as novel therapeutic targets in the treatment of non-small cell lung cancer. Expert Opin Ther Targets. 2013; 17: 1109-1112.
18. Korherr C, Gille H, Schafer R, Koenig-Hoffmann K, Dixelius J, Egland KA, Pastan I and Brinkmann U. Identification of proangiogenic genes and pathways by high-throughput functional genomics: TBK1 and the IRF3 pathway. Proc Natl Acad Sci U S A. 2006; 103: 4240-4245.
19. Ou YH, Torres M, Ram R, Formstecher E, Roland C, Cheng T, Brekken R, Wurz R, Tasker A, Polverino T, Tan SL and White MA. TBK1 directly engages Akt/PKB survival signaling to support oncogenic transformation. Mol Cell. 2011; 41: 458-470.
20. Macdonald JS, Smalley SR, Benedetti J, Hundahl SA, Estes NC, Stemmermann GN, Haller DG, Ajani JA, Gunderson LL, Jessup JM and Martenson JA. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med. 2001; 345: 725-730.
21. Kim S, Lim DH, Lee J, Kang WK, MacDonald JS, Park CH, Park SH, Lee SH, Kim K, Park JO, Kim WS, Jung CW, Park YS, et al. An observational study suggesting clinical benefit for adjuvant postoperative chemoradiation in a population of over 500 cases after gastric resection with D2 nodal dissection for adenocarcinoma of the stomach. Int J Radiat Oncol Biol Phys. 2005; 63: 1279-1285.
22. Ha SY, Lee J, Kang SY, Do IG, Ahn S, Park JO, Kang WK, Choi MG, Sohn TS, Bae JM, Kim S, Kim M, Kim S, et al. MET overexpression assessed by new interpretation method predicts gene amplification and poor survival in advanced gastric carcinomas. Mod Pathol. 2013; 26: 1632-1641.
23. Deng T, Liu JC, Chung PE, Uehling D, Aman A, Joseph B, Ketela T, Jiang Z, Schachter NF, Rottapel R, Egan SE, Al-Awar R, Moffat J, et al. shRNA kinome screen identifies TBK1 as a therapeutic target for HER2+ breast cancer. Cancer Res. 2014; 74: 2119-2130.
24. Clark K, Peggie M, Plater L, Sorcek RJ, Young ER, Madwed JB, Hough J, McIver EG and Cohen P. Novel cross-talk within the IKK family controls innate immunity. Biochem J. 2011; 434: 93-104.
25. Clark K, Plater L, Peggie M and Cohen P. Use of the pharmacological inhibitor BX795 to study the regulation and physiological roles of TBK1 and IkappaB kinase epsilon: a distinct upstream kinase mediates Ser-172 phosphorylation and activation. J Biol Chem. 2009; 284: 14136-14146.
26. Reilly SM, Chiang SH, Decker SJ, Chang L, Uhm M, Larsen MJ, Rubin JR, Mowers J, White NM, Hochberg I, Downes M, Yu RT, Liddle C, et al. An inhibitor of the protein kinases TBK1 and IKK-varepsilon improves obesity-related metabolic dysfunctions in mice. Nat Med. 2013; 19: 313-321.
27. Cancer Genome Atlas Research N. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014; 513: 202-209.
28. Cristescu R, Lee J, Nebozhyn M, Kim KM, Ting JC, Wong SS, Liu J, Yue YG, Wang J, Yu K, Ye XS, Do IG, Liu S, et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat Med. 2015; 21: 449-456.
29. Ruschoff J, Dietel M, Baretton G, Arbogast S, Walch A, Monges G, Chenard MP, Penault-Llorca F, Nagelmeier I, Schlake W, Hofler H and Kreipe HH. HER2 diagnostics in gastric cancer-guideline validation and development of standardized immunohistochemical testing. Virchows Arch. 2010; 457: 299-307.
30. Barros-Silva JD, Leitao D, Afonso L, Vieira J, Dinis-Ribeiro M, Fragoso M, Bento MJ, Santos L, Ferreira P, Rego S, Brandao C, Carneiro F, Lopes C, et al. Association of ERBB2 gene status with histopathological parameters and disease-specific survival in gastric carcinoma patients. Br J Cancer. 2009; 100: 487-493.
31. Oda N, Tsujino T, Tsuda T, Yoshida K, Nakayama H, Yasui W and Tahara E. DNA ploidy pattern and amplification of ERBB and ERBB2 genes in human gastric carcinomas. Virchows Arch B Cell Pathol Incl Mol Pathol. 1990; 58: 273-277.
32. Varis A, Zaika A, Puolakkainen P, Nagy B, Madrigal I, Kokkola A, Vayrynen A, Karkkainen P, Moskaluk C, El-Rifai W and Knuutila S. Coamplified and overexpressed genes at ERBB2 locus in gastric cancer. Int J Cancer. 2004; 109: 548-553.
33. Ayhan A, Yasui W, Yokozaki H, Seto M, Ueda R and Tahara E. Loss of heterozygosity at the bcl-2 gene locus and expression of bcl-2 in human gastric and colorectal carcinomas. Jpn J Cancer Res. 1994; 85: 584-591.
34. Hattori Y, Odagiri H, Nakatani H, Miyagawa K, Naito K, Sakamoto H, Katoh O, Yoshida T, Sugimura T and Terada M. K-sam, an amplified gene in stomach cancer, is a member of the heparin-binding growth factor receptor genes. Proc Natl Acad Sci U S A. 1990; 87: 5983-5987.
35. Lee HK, Lee HS, Yang HK, Kim WH, Lee KU, Choe KJ and Kim JP. Prognostic significance of Bcl-2 and p53 expression in gastric cancer. Int J Colorectal Dis. 2003; 18: 518-525.
36. Smith MG, Hold GL, Tahara E and El-Omar EM. Cellular and molecular aspects of gastric cancer. World J Gastroenterol. 2006; 12: 2979-2990.

