INTRODUCTION
Mantle cell lymphoma
Hematological malignancies are the seventh most common form of cancer and are the second leading cause of cancer-associated death (National Cancer Institute, USA).. In the United States, tumors of B cell origin make up 85% to 90% of Non Hodgkin Lymphoma (NHL). Mantle Cell Lymphoma (MCL) is a subtype of B- cell derived NHL representing 5-6% of all NHL. MCL is a rare but aggressive B cell lymphoma that occurs more than four times as often in males as in females. The median age at diagnosis is about 60 years. MCL is derived from naïve, pre-germinal center cells of primary follicles or mantle regions of secondary follicles. MCL typically possesses the hallmark t(11;14)(q13;q32) chromosomal translocation, which causes overexpression of Cyclin D1, resulting in disordered progression of the cell cycle and aggressive lymphomagenesis. Although Cyclin D1-negative MCL cases have been reported, all such cases were positive for overexpression of cyclin D2 or D3 [1]. In addition, Sry-related high-mobility-group box (SOX11), which is expressed in nearly 90% of the cases, has been identified as diagnostic and prognostic biomarker of MCL [2]. The pathogenesis of MCL is complex and involves molecular alterations at various levels. Targeted genes and pathways include regulatory elements of the cell cycle machinery and senescence (ARF/BMI1/CDK4/INK4/RB1), DNA damage response pathways (ATM/CHK2/p53), and cell survival signals. Genes representing other signaling pathways including BTK, AKT, mTOR, WNT, NF-κB, TNF, and NOTCH have been found to play crucial roles in the pathogenesis of MCL (Figure 1) [3].
Clinical Characteristics of MCL
Clinical classification
MCL is classified into two forms: indolent and conventional. The indolent form is primarily characterized by a non-nodal leukemic presentation with bone marrow involvement and splenomegaly [4]. Most of these patients show normal performance status, normal serum LDH, and low MIPI score. Other characteristic features of indolent MCL include mild to moderate lymphocytosis, hypermutated IGVH genes, a non-complex karyotype, and absence of SOX11 expression [5] (Figure 1). Furthermore, cases of indolent MCL are associated with low Ki67 (≤10%) and kappa light chain expression as opposed to lambda light chain expression typically found in aggressive MCL. To date, markers which can confirm the diagnosis of indolent nature of MCL are lacking although SOX11 shows promise as one such biomarker [6].
Clinical and pathological characteristics
At diagnosis, MCL patients typically present with generalized lymphadenopathy but with advanced disease, often at clinical stage III or IV. Systemic symptoms such as loss of appetite and weight loss, fever, night sweats, nausea and/or vomiting, indigestion, and abdominal pain or bloating are commonly reported. Gastrointestinal involvement is detected in 90% of cases of MCL, and 50% of patients present with blood and marrow involvement. MCL diagnosis is based on morphological resemblance of lymphoma cells to the mantle zone (MZ) B-lymphocyte lineage from which the tumor derives, and typically exhibit an MZ B cell phenotype: sIgM+, sIgD+, CD5+, CD20+, CD23−, FMC7+ [7]. Pathologically, MCL is further classified into two main subtypes: classic and blastoid, the blastoid form being associated with a more aggressive clinical course (Figure 1). Differential diagnosis for MCL includes Chronic Lymphocytic Leukemia (CLL) or Small Lymphocytic Lymphoma (SLL), Follicular Lymphoma, and marginal zone lymphoma [8]. CLL and SLL express sIgM, sIgD, CD19, and CD20, and have differential expression of T cell antigen CD5. However, MCL cells are positive for FMC7 and “typically” do not express CD23. They also exhibit greater staining intensity for B cell antigens and Igs. Like follicular lymphoma, MCL is positive for CD20 and Bcl-2, but in contrast to follicular lymphoma, MCL is negative for CD10, BCL-6, as well as CD23 [9]. Pathogenic mechanisms and phenotypical characteristics of MCL are illustrated in Figure 1.
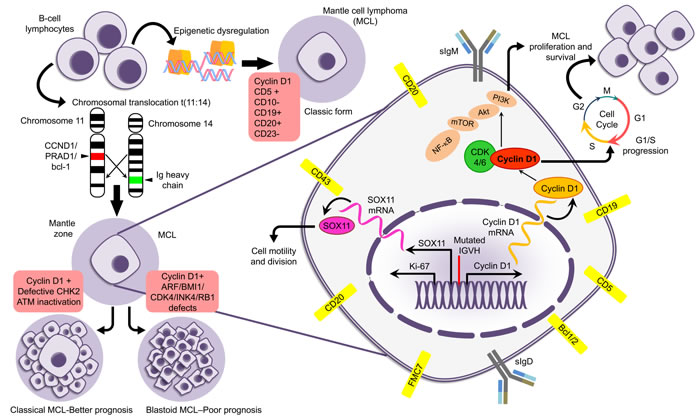
Figure 1: Figure describing the pathological and immunological details of MCL.
Laboratory studies
Blood and bone marrow examination, blood chemistry, lymph node biopsy, immunocytochemistry and flow cytometry, and cytogenetic studies aid in diagnosis of MCL. Usually complete blood count yields lymphocytosis along with anemia and cytopenia. Serum chemistry is significant for elevated LDH and elevated Beta-2-microglobulin. Wright-Giemsa staining is used for determining the presence of circulating lymphoma cells specific for MCL in peripheral blood. On microscopic analysis, the cancerous lymphocytes appear small to medium-sized with a condensed chromatin, scant cytoplasm, and small nucleoli. The morphological spectrum of leukemic MCL ranges from ‘small cells’ resembling chronic lymphocytic leukemia (CLL) or follicular lymphoma (FL) to ‘large cells’ mimicking prolymphocytic leukemia (PLL) or acute leukemia. Large cell morphology is associated with more frequent additional cytogenetic abnormalities as well as poorer outcome [10]. Bone marrow aspiration and biopsy slides show cellularity consistent with lymphoma and usually demonstrate a pattern of nodular, interstitial, paratrabecular, or diffuse involvement or, in some cases, a combination of these patterns [11]. MCL most commonly presents with a diffuse effacement of the lymph nodes; in situ, mantle-zone, nodular and diffuse, and diffuse patterns are also commonly seen [12]. Some of the cytologic variants seen on biopsy are marginal zone-like variant, small cell variant, blastoid cell variant, and pleomorphic variant; blastoid and pleomorphic variants are associated with a more aggressive clinical course.
Samples from bone marrow and lymph nodes are typically used for immunohistochemical and flow cytometry studies. Almost all cases of MCL show overexpression of Cyclin D1 messenger ribonucleic acid (mRNA). The classic immunophenotype is strongly positive for pan-B cell antigens CD5, CD19, CD43, weakly positive for FMC7, and negative for CD10, CD23, and Bcl-6. Variants of the classic immunophenotype have been identified flow cytometrically; these include BCL-1+/CD5− lymphoma with morphologic features consistent with MCL [13]. Gao et al. 2009 reported isolated cases that were CD 10+, CD 23+, and FMC7- as well as cases with variations in two antigens: CD5-/CD23+, CD10+/FMC7-, and CD23+/FMC7-. Multi-parameter flow cytometry is becoming an essential tool in characterization and diagnosis of variant forms of mature B cell lymphoproliferative neoplasms including mantle cell carcinoma [14, 15].
On cytogenetics studies, almost all MCL cases exhibit the reciprocal translocation t(11;14)(q13;q32) involving Cyclin D1 genes (CCND1, PRAD1, bcl-1) on chromosome 11 and the Ig heavy chain locus on chromosome 14. Conventional karyotyping detects this translocation in almost 65% of cases; however, the translocation is identified in up to 99% of cases by fluorescence in situ hybridization (FISH). A few cases of Cyclin D1-negative lymphoma with morphologic, pathologic, clinical, and molecular features typical of MCL have also been reported; these typically lack evidence of chromosomal translocations or genomic amplifications but possess high levels of cyclin D2 or D3 [1, 6]. Cytogenetic, comparative genomic hybridization (CGH), and expression profiling studies have been used to identify secondary genetic alterations that may be involved in the pathogenesis and progression of MCL [16, 17]. Numerous reports have confirmed that MCL carries the highest levels of genomic instability among the malignant lymphoid neoplasms.
Molecular techniques for diagnosis of MCL
Southern blot was utilized for detection of Ig and T cell Gene rearrangements associated with MCL and other B cell lymphomas until PCR replaced this laborious technique. PCR requires a small quantity of DNA (approximately 2 μg) and can be performed on paraffin tissue with minimal labor and short turnaround time. More recently, gene expression profiling (GEP) has provided a novel approach for diagnosis of hematological malignancies including lymphomas. [18]. Among various GEP platforms, RNA guided gene expression profiling of tumors measures the quantity of RNA transcripts which are labeled and hybridized on the array. Such GEP studies have been especially useful in detailed characterization and categorization of neoplasia into other subtypes and new variants of B cell derived lymphoma [19, 20]. When combined with RNA interference (RNAi), this technique has assisted in providing novel insights into signaling pathways associated with MCL [21]. cDNA microarray analysis allows detection of gain or loss in specific chromosomal regions, and has been used to describe the novel spectrum of somatic mutations of MCL [22]. Similarly, whole genome and whole exome sequencing allow measurement of the expression of thousands of genes simultaneously in a single experiment, and offer a powerful technology for the study of hematological malignancies including MCL [23]. Recently, whole transcriptome shotgun sequencing (RNAseq) identified a role of somatic mutations in NOTCH1 for the pathogenesis of MCL [24]. More recently, next generation sequencing (NGS) was found to be equally sensitivity as allele-specific oligonucleotides-PCR for immunoglobulin heavy-chain-gene-based minimal residual disease (MRD) detection, providing an effective tool for MRD monitoring [25]. The combination of tissue microarray and automated quantitative assessment of immunofluorescence (TMA-AQUA) as well as proteome- and microarray-based expression analysis of lymphoma cell lines via MS analysis (using MALDI-TOF) are capable of identifying large numbers of possible molecular targets relevant to MCL and other lymphomas [26-27]. All of these techniques have contributed to identification of underlying molecular mechanisms contributing to the pathogenesis and clinical progression of the disease, and have expanded our ability to predict clinical outcomes of lymphoma patients, as well as aided in identification of targets for novel therapeutic agents.
Role of tumor microenvironment in the progression of MCL
Inflammation plays a crucial role in the initiation of cancer as well as in shaping the environment for survival of the tumor cells [28]. Local and systemic inflammatory responses induced by cytokines, chemokines, and small inflammatory proteins derived from tumor cells and/or host immune cells govern the major cross-talk between the tumor and host immune response. Therefore, it is imperative to identify and therapeutically target the cytokines, chemokines, and other crucial transcription factors that can disrupt the interaction between tumors and host immune response leading to aggressive forms of cancer with poor clinical outcome [29]. In fact, the interactions between cancer cells, immune cells, and tumor stromal cells in the tumor microenvironment (TME) are capable of modulating core features of malignancy including proliferation, self-renewal, homing, invasion, angiogenesis, immune evasion, and survival of cancer cells via immune cells, endothelial cells, and fibroblastic cells (Figure 2). Based on this theme, published reports have identified role of B cell-secreted cytokines, chemokines, and growth factors in dynamic and bidirectional signaling that promote the recruitment of adhesion molecules responsible for sustained signaling necessary for proliferation and survival of tumor cells [30, 31]. (Figure 2). The significance of the TME for MCL progression is illustrated by the observation that treatment of the Jeko-1 MCL cell line with anti-CXCR4 and anti-VLA-4 antibodies reduced cross talk between the tumor cells and stromal cells, decreased IL6 levels and phosphorylation of ERK1/2, AKT, and NF-κB, and increased the therapeutic sensitivity of Jeko-1 [32]. Similarly, an inhibitor of phosphatidylinositol-3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) pathway, NVP-BEZ235, inhibited TME-specific signaling pathways such as IL4 and IL6/STAT3, resulting in suppression of angiogenesis, migration, and tumor invasiveness in MCL cells [33]. Inhibition of IL-6 resulted in down-regulation of JAK2/STAT3 and PI3K/Akt pathways in MCL and abrogated IL-6-mediated protection of MCL cells [34]. Administration of an antibody against CCR7, a chemokine receptor known to cause lymphoid dissemination in many cancers, suppressed dissemination of MCL cells and reduced mortality in an animal model, suggesting the importance of TME in the pathogenesis and aggressiveness of MCL [35]. The dynamic interaction among the components of TME is integral to sustaining proliferation, angiogenesis, immune suppression, self-renewal, invasion, and migration, thus, promoting tumor progression and, ultimately, drug resistance (Figure 2).
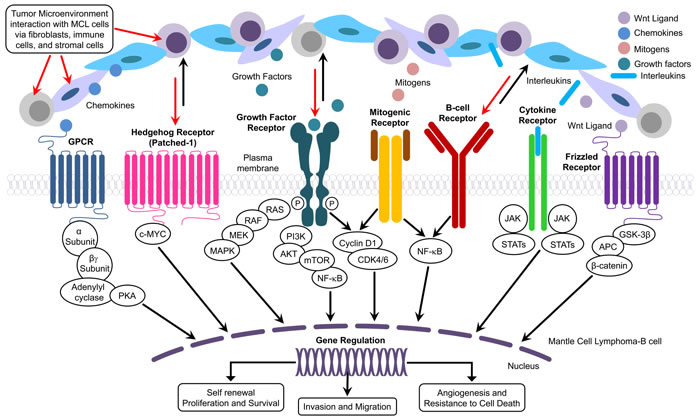
Figure 2: Figure describing the details of the interaction between components of TME and pathological pathways involved with MCL.
Role of biomarkers in detection, diagnosis, and therapeutic response of MCL
Biomarkers have long been used to support diagnosis and to indicate prognosis of diseases. However, incorporation of molecular biomarkers as components of routine diagnostic panels for human diseases has gained greater importance in recent years. Biomarkers serve as a guide for risk assessment, screening, differential diagnosis, determination of prognosis, prediction of response to treatment, and for monitoring progression of disease. The main pathogenic events of cancer involve dysregulation of oncogenes, tumor suppressor genes, and DNA repair genes; thus, cancer biomarkers fall into various categories such as genetic biomarkers, cytogenetic biomarkers, epigenetic biomarkers, biomolecule/cancer antigen biomarkers, etc. The identification of unique biomarkers for diagnosis and prognosis of MCL has lagged compared to other cancers. Fortunately, blood/serum being a dynamic system harbors circulating components of tumors, including small molecules secreted by tumor cells, as well as other immune cell-secreted biomolecules, that reflect the underlying disease process. A comprehensive list of available biomarkers that inform diagnosis, prognosis, and functional drug interactions of MCL, along with their normal functions are presented in Table 1; some of these are discussed below.
Table 1: Table describing the key MCL biomarkers that inform diagnosis, prognosis, and functional drug interactions of MCL, along with their normal functions.
Major Family |
Biomarker |
Class |
Normal Function |
Expression in MCL |
Involved Pathway(s) |
Interaction with MCL Drugs |
Detection Technique |
References |
Cell-Cycle Regulation related markers |
Cyclin D1 |
Protein |
Regulator of CDK4 and CDK6 |
Up-Regulated |
Cell Cycle |
PD0332991 and others inhibit CDKs |
IHC, PCR, FISH |
215, 216 |
Ki67 |
Protein |
Necessary for cell proliferation and RNA transcription |
Up-Regulated |
Cell Cycle |
COX 2 and CDK inhibitors decrease Ki67 |
IHC, PCR, FISH |
134, 216, 217 |
|
ATM |
Protein Kinase |
Induces cell cycle arrest, DNA repair, apoptosis |
Down-Regulated |
Homologous Recombination Repair and Cell Cycle regulation |
KU 55933, KU-60019 and VE-821 inhibit ATM |
IHC, PCR, FISH |
216, 218 |
|
CHK2 |
Protein |
Tumor suppressor, regulates cell division |
Down-Regulated |
CDK-mediated phosphorylation and removal of Cdc6 |
AZD7762, CCT241533 hydrochloride and NSC 109555 ditosylate inhibit CHK2 |
IHC, PCR, FISH |
216, 219 |
|
CDK4 |
Gene |
Catalyzes cell cycle G1 phase progression |
Up-Regulated |
Cell cycle regulation, especially CDK-mediated phosphorylation and removal of Cdc6 |
AZD-5438, Purvalanol A and Purvalanol B inhibit CDK's |
IHC, PCR, FISH |
216, 218 |
|
RAN |
Protein |
Required for importing of proteins into nucleus as well as transporting RNA out of the nucleus. Involved in chromatin condensation and cell cycle regulation. |
Up-Regulated |
Regulation of nucleocytoplasmic transportation and mitotic spindle formation by forming CRM1/RAN GTP complex with Chromosomal region maintenance 1 (CRM1) |
CRM1 inhibitors LMB (Leptomycin B and other LMB analogues) indirectly inhibit RAN |
IHC, PCR, FISH |
216, 220 |
|
MYC |
Protein |
Controls DNA replication, causes B cell proliferation, regulates cell growth and apoptosis |
Up-Regulated |
ERK Signaling, PEDF Induced Signaling, ErbB signaling pathway |
Covalent inhibitor of cyclin dependent kinase 7 (CDK7) inhibit MYC |
IHC, PCR, FISH |
216, 221 |
|
TCL1A |
Onco protein |
T cell leukemia/lymphoma 1A promotes nuclear translocation of AKT1, enhances cell proliferation, promotes cell survival; interferes with NF-kB inhibitor IκB |
Up-Regulated |
P13K-Akt signaling where TLC1A acts as a co-activator of AKT |
Rapamycin inhibits Tcl1/Akt/mTOR pathway |
IHC, PCR, FISH |
85, 216, 222 |
|
p27 |
Gene |
Inhibits CDK2 and CDK4, involved in G1 phase arrest |
Down-Regulated |
Cell cycle inhibitors and regulators |
NSAIDS increase expression of p27 |
IHC, PCR, FISH |
216, 223 |
|
CHK1 |
Protein |
CDK-mediated phosphorylation and removal of Cdc6, G2/M Checkpoints, |
Down-Regulated |
Coordinates response to DNA damage during cell cycle and regulates checkpoint in cell cycle |
CHK1 inhibitor PF-00477736 works synergistically with Wee1 inhibitor (MK-1775) to induce apoptosis in MCL cell lines |
IHC, PCR, FISH |
216, 224 |
|
BAFF-R |
Gene |
Enhances B-cell survival, regulates B-cell population |
Up-Regulated |
PEDF Induced Signaling, TNF, Akt and TGF-Beta Pathway |
anti-BAFF antibody has therapeutic application against SLE but has not been tested against MCL |
IHC, PCR, FISH |
216, 225 |
|
Transcription Regulators |
Sox11 |
Trans-cription factor |
Plays role in determining cell fate |
Up-Regulated |
Notch, ERK Signaling |
methylation of SOX11 promotor region reduces expression of SOX11 |
IHC, PCR, FISH |
2, 48, 216 |
STAT3 |
Protein |
Transcription activator |
Up-Regulated |
growth factors, cytokines and interleukin signaling pathways |
WP1066 with vorinostat has been tested against MCL. Other STAT-3 inhibitors are also available |
IHC, PCR, FISH |
216, 226, 227 |
|
Nuclear Factor Kappa Beta |
Protein |
Controls DNA transcription, cytokine production, and cell survival |
Up-Regulated |
NFKB signaling pathway |
proteasome inhibitor PS-341 or a specific pIκBα inhibitor, BAY 11-7082 have been used against MCL cell lines |
IHC, PCR, FISH |
216, 228 |
|
Tumor Suppression/Necr-osis related markers |
Tp53 |
Gene |
Codes for tumor suppressor protein p53 |
Up-Regulated |
apoptosis and cell cycle related signaling pathways |
Cyclic Pifithrin-alpha hydrobromide and CP 31398 dihydrochloride stabilizes p53 |
IHC, PCR, FISH |
61, 62, 216 |
INK4A |
Protein Kinase |
Tumor suppressor, slows down cell cycle |
Mutated or Deleted |
Cellular Senescence and Cell Cycle Regulation |
HDAC inhibitors interact with INK4A to regulate the cell proliferation |
IHC, PCR, FISH |
133, 216 |
|
RB1 |
Gene |
Regulator of entry into cell division, tumor suppressor |
Inactivated |
CDK-mediated phosphorylation and removal of Cdc6, Cell Cycle Regulation |
HDAC inhibitors increase RB1 expression in Myeloid-derived suppressor cells |
IHC, PCR, FISH |
216, 229 |
|
MDM2 |
Protein |
Mediates ubiquitination of p53 |
Up-Regulated |
PI-3K cascade signaling , CDK-mediated phosphorylation and removal of Cdc6 |
AMG232, JNJ-26854165 and Nutlin-3 inhibits MDM2-p53 interaction |
IHC, PCR, FISH |
216, 230 |
|
BCL-6 |
Gene |
Transcriptional suppressor, suppresses genes related to differentiation, cell cycle, and apoptosis |
Down-Regulated |
IL4-mediated signaling events, B Cell Receptor , Fox O signaling pathways, Direct p53 effectors |
Rituximab inhibits BCL66 Paraffin targets BCL-6 |
IHC, PCR, FISH |
216, 231 |
|
BCL-2 |
Protein |
Regulator of apoptosis |
Up-Regulated |
PEDF Induced Signaling, TGF-Beta, ERK Signaling, |
2,3-DCPE hydrochloride, ABT-199, ABT-263, ABT-737 and Apogossypolone inhibit BCL-2 |
IHC, PCR, FISH |
216, 232 |
|
BIRC3 (Baculoviral IAP Repeat Containing 3) |
Protein |
Anti-apoptotic protein regulates caspases and apoptosis, modulates inflammatory signaling and immunity |
Mutated or Deleted |
apoptosis and NFKB signaling pathway |
Needs further research |
IHC, PCR, FISH |
216, 233 |
|
PTEN (Phosphatase and tensin homolog) |
Gene |
Tumor suppressor |
Mutated or Deleted |
PI-3K cascade Signaling Pathway |
several drugs under clinical investigation |
IHC, PCR, FISH |
216, 234 |
|
TNFAIP/A20 |
Protein |
A20-binding inhibitor of NF-κB |
Mutated or Deleted |
Akt, ERK, NFKB Signaling Pathways |
Overexpression of ABINs inhibits NF-κB activation; needs further research |
IHC, PCR, FISH |
216, 235 |
|
TNFRSF10B |
Gene |
Tumor Necrosis factor receptor, Transduces apoptosis signals, |
Inactivated |
Death Receptor, TNF , Akt and TGF-Beta signaling Pathway |
Cisplatin is known to alter expression of TNFRSF10B |
IHC, PCR, FISH |
216, 236 |
|
Apoptosis-Related markers |
repp86 |
Protein |
Spindle assembly factor, required for assembly of microtubules during apoptosis, |
Up-Regulated |
Cell Cycle Checkpoint control, Aurora A, PLK1 signaling events, Cell cycle spindle assembly and chromosome separation |
Not available |
IHC, PCR, FISH |
216, 237 |
survivin |
Protein |
Inhibitor of Apoptosis, Regulates mitosis |
Up-regulated |
Cell cycle and IL-23 immune response pathway |
Not available |
IHC, PCR, FISH |
216, 238 |
|
FAF1 |
Protein |
Mediates programmed cell death |
Deleted |
Apoptosis, TNF-alpha/NF-kB Signaling Pathway, Apoptosis and Autophagy |
Not available |
IHC, PCR, FISH |
216, 239 |
|
RNA/DNA Repair Regulator related markers |
POLE2 |
Protein |
DNA binding, Protein Binding, DNA-directed DNA polymerase activity |
Up-Regulated |
Telomere C-Strand Synthesis, CDK-mediated phosphorylation and removal of Cdc6, DNA Repair, Mitotic G1-G1/S phases |
Thioguanine Daunorubicin and Cytosine arabinoside targets POLE2 |
IHC, PCR, whole-genome and/or whole-exome sequencing |
216, 236 |
Topoisomerase Iiα |
Protein |
Cut strands of DNA to manage DNA tangles and supercoils, promotes chromosomal disentanglement |
Up-Regulated |
Cell Cycle, Chromatin regulation/acetylation |
HU-331, ICRF-187/193, mitindomide and m-AMSA inhibits topoisomerase Iiα |
IHC, PCR, whole-genome and/or whole-exome sequencing |
216, 240 |
|
IGF2BP2 |
Gene |
RNA-binding factor, facilitates mRNA transport and storage |
Up-Regulated |
Binding of RNA by Insulin-like growth factor-2 mRNA binding proteins |
Not available |
IHC, PCR, whole-genome and/or whole-exome sequencing |
216, 241 |
Please see Supplementary Table 1 to access complete table.
Cyclin D1
Cyclin D1 is encoded by the CCND1 gene which consists of 5 exons; alternative splicing generates Cyclin D1a and D1b isoforms. Cyclin D1a, a 30-kDa labile protein, forms a complex with the cyclin-dependent kinase CDK4 or CDK6 to promote cell-cycle entry. Cyclin D1 binds to the cell cycle kinase p34cdc2 and drives cells from G1 into S phase. Although Cyclin D1 is cell cycle-related protein it is generally absent in normal lymphoid tissue or B cell lines in the absence of the t(11;14)(q13;q32) translocation [36], which causes rearrangement of the bcl-1 proto-oncogene from chromosome 11 into the immunoglobulin heavy chain locus on chromosome 14, resulting in an overexpression of Cyclin D1 mRNA (also known as bcl-1 and PRAD1). Thus, abnormally expressed Cyclin D1 contributes to unopposed proliferation and downstream molecular events leading to higher and prolonged Cyclin D1 mRNA and protein expression, resulting in greater tumor proliferation and shorter survival of MCL patients [37]. Although t(11;14) translocations occur in almost 85% of MCL cases, non-MCL and other lymphoproliferative disorders may also exhibit t(11;14) translocations [38]. Expression of Cyclin D1 can be detected in formalin-fixed, paraffin-embedded (FFPE), tissue samples from MCL patients. Traditionally used methods include in situ hybridization, northern and southern blot, and cytogenetics; more recently used methods include immunohistochemical detection of Cyclin D1, PCR for bcl-1 rearrangement corresponding to t(11;14) translocation, and RT-PCR for Cyclin D1 mRNA expression. From the detection standpoint, direct visualization with DNA fiber fluorescence in situ hybridization provides 95-100% sensitivity; PCR provides the lowest sensitivity (30-50%) of these assays [39]. Because Cyclin D1 interacts with other signaling pathways including BCR/PI3K/AKT/mammalian target of rapamycin (mTOR), nuclear factor-κB (NF-κB), tumor necrosis factor (TNF), Hedgehog and WNT pathways, and the Bcl-2 family of apoptosis regulators, it is not surprising that Cyclin D1 is at the center of the pathogenesis of MCL, enhancing tumor proliferation, facilitating evasion of apoptosis, and reducing immune control [40].
Cyclin D2/D3
Because mouse models have failed to demonstrate that overexpression of Cyclin D1 induces MCL, other genetic aberrations such as presence of a deregulated myc family gene with Cyclin D1 have been suggested to contribute to the pathogenesis of MCL [41]. Furthermore, recently reported Cyclin D1 negative MCL cases have exhibited overexpression of cyclin D2 and D3 [1]. Similar to Cyclin D1, cyclin D2 and D3 genes belong to the highly conserved cyclin family, and they function as regulators of CDK kinases. Cyclin D2 and D3 are expressed at high levels in some Cyclin D1-negative MCL cases. Cyclin D1-negative MCL is clinically and morphologically indistinguishable from conventional Cyclin D1-positive MCL. However, these cases have generally failed to exhibit any chromosomal translocations or gene amplifications involving cyclin D2 or D3 loci by FISH analysis. Deregulation of cyclin D2 or D3 expression in these MCL cases is mostly considered to be due to epigenetic mechanisms [1]. However, translocations of the cyclin D2 locus (chromosome 12p13) into either IGK or IGH loci have been reported as the initiating events for MCL pathogenesis [42, 43].
B-cell specific biomarkers
The presence of specific cluster of differentiation (CD) antigens allows diagnosis of various types of leukemia and lymphoma. As a B cell lymphoma, MCL is characterized by the presence of characteristic B cell expression patterns with high ( > 90%) expression of CD5, CD19, CD20, CD21, CD22, CD43, CD79a, sIg, and cIg, and low ( < 10%) expression of CD10 and CD23. Variants such as CD5-negative [13], CD23-positive [44], and combinations of CD5-/CD23+, CD10+/FMC7-, and CD23+/FMC7- forms have been reported [7]. CD5-/CD23+ MCL is associated with a more indolent course and improved outcomes as opposed to ‘classic’ CD5+/CD23- MCL cases [44]. Usually in MCL, there is a lack of germinal center cell marker CD10, but in rare cases CD10+ cells exhibiting pleomorphic blastoid morphology are seen [45]. CD20 has been targeted in MCL with anti-CD20 antibodies (Rituximab), which causes death of tumor cells through several mechanisms including anti-proliferative effects and pro-apoptotic effects in B cells [46]. Similarly, anti-CD20 radio-immunotherapy has been used for treating lymphoma including MCL [46].
Sox11
Apart from MCL, Sox11 is expressed in MCL, lymphoblastic lymphoma, some Burkitt lymphomas, and T-cell prolymphocytic leukemia, but is not known to be expressed in other lymphoid neoplasms. SOX11 is expressed in all aggressive forms of MCL (90-95%), and functions as diagnostic and prognostic marker for MCL [47, 48]. Detection of its expression is invaluable in identification of Cyclin D1-negative MCL [49]. Sox11 has been shown to control the oncogenic transcriptional network and effector genes responsible for B-cell transformation. The direct transcriptional targets of the SOX11 protein, DBN1, SETMAR, and HIG2, which are involved in cell motility and cell division, are highly correlative to the expression level of SOX11 [50]. Sox11-negative MCL cases mostly demonstrate an indolent course and a non-nodal presentation with splenomegaly and high WBC and lymphocyte counts when compared with SOX11-positive cases. Absence of Sox11 is not, however, an exclusive feature of indolent forms of MCL [51]. Sox11 is also an important minimal residual disease marker (MRD) used to monitor the clinical response to therapy and to predict relapse of MCL [52].
IGVH
MCL is characterized by translocation of the immunoglobulin heavy chain locus, and somatic hypermutation of the rearranged variable region of the immunoglobulin heavy chain (IGVH) gene [53]. The sequencing of the IGVH region provides information on the clonal origin of chronic B cell malignancies including MCL. An absence of somatic mutations is consistent with origin from a pre-germinal center B cell; whereas, tumors that show somatic hypermutation arise either from germinal center cells or from post-germinal center memory cells. The “mutated” IGVH status is defined as < 98% homology to the germ-line. A few studies suggest that the lack of IGVH somatic mutations correlates with a more aggressive clinical course and possibly with shorter survival of MCL patients [54]. More recently, BCL1/IgH, and IgH-VDJ along with SOX11 and Cyclin D1 have been discussed as biomarkers for MRD-guided management of patients with mature B cell malignancies including MCL [55].
Ki67
The Ki67 index, defined by the percentage of Ki67-positive lymphoma cells (determined by histopathological analysis) has been the single most powerful and established prognostic biomarker for overall survival of MCL patients. A high Ki67 index in the lymph node biopsy at the time of initial diagnosis, indicating a highly proliferative tumor, is predictive of a poor outcome [56]. The Ki67 index is employed in workup protocols for many tumors, especially breast cancer and other lymphoid neoplasms [56, 57]. Although, the Ki67 index shows a positive correlation with outcome and survival duration, no significant association has been demonstrated between the Ki67 index and clinicopathologic parameters including LDH levels, B-symptoms, tumor stage, extranodal involvement, and performance status [58]. The Ki67 index has become a valuable prognostic parameter for MCL patients treated with immunochemotherapy such as the combination of rituximab and first line therapy [59, 60].
Other cytogenetic and epigenetic biomarkers
TP53, a tumor suppressor gene crucial for regulating cell division and preventing tumor formation, is commonly altered in human cancers including MCL. TP53 mutations have been found in 15-20% of MCL patients and are associated with significantly shorter overall survival (OS) and poor prognosis [61]. Interestingly, TP53 mutations have been found to be equally distributed in MCL regardless of SOX11 expression or IGVH mutations [22]. In multivariate analysis, TP53 was the only significant independent molecular marker that improved the prognostic value of MIPI [62]. Similarly, ATM (ataxia telangiectasia mutated) gene, which encodes for a serine-threonine kinase and belongs to the phosphatidylinositol-3 kinase (PI-3K) family, acts as an important tumor suppressor gene. MCL is characterized by inactivation of ATM gene (involving in class switch recombination in the Ig heavy chain locus), which may act synergistically with overexpressed Cyclin D1 to override cell-cycle checkpoint controls [63]. Consistent with this, inactivation of ATM is associated with frequent chromosomal imbalances [63]. Bea et al. 2013 performed whole-genome sequencing (WGS) of 29 MCL lines and whole-exome sequencing (WES) of 6 MCL cell lines; they identified recurrent mutations in ATM, WHSC1, MLL2, BIRC3, MEF2B, and TLR2 in addition to mutations in common MCL related genes, CCND1, SOX11, and TP53 [22]. Interestingly, ATM mutations were seen only in SOX11-positive tumors, whereas CCND1 mutations were preferentially detected in MCL with IGVH-mutations [22]. WHSC1, MLL2, and MEF2B all belong to a group of chromatin modifiers suggesting a role of epigenetic mechanisms in the pathogenesis of MCL. The potent oncogene c-myc is critically involved in the regulation of many growth-promoting signal transduction pathways and interacts with various down-stream signaling pathways; over-expression of c-myc is commonly seen in multiple human cancers including MCL [64]. C-myc is also associated with Bcl-2 translocations. Both Bcl-2 and Bcl-6 are anti-apoptotic proteins, and their translocation under the influence of altered c-myc lead to abnormal growth abilities of the affected cells and commonly confer an aggressive form of MCL [65]. Upregulation of BMI1 and downregulation of miR-16 in the MCL side population (SP) reduces apoptosis in these cells [66]. Downregulation of PAX5, a member of the paired box (PAX) family of transcription factors, correlates with an aggressive, highly drug-resistant, phenotype predictive of poor prognosis [67]. Overexpression of JARID1B and reduced histone H3K4 tri-methylation were associated with MCL and therefore, depletion of JARID1B caused up-regulation of histone acetylation of H3 and inactivation of Cyclin D1 leading to apoptosis of MCL cells [68].
Other signaling pathway-related biomarkers
MCL pathogenesis also involves dysregulation in several signaling pathways [40]. Analysis of serum proteins provides an indirect but inexpensive means to monitor expression of genes encoding for cytokines and chemokines. The resulting expression profiles provide molecular signatures for mantle cell lymphoma. Cytokines and chemokines impart crucial cell signaling, pro-inflammatory, and immunomodulatory functions. Multiple studies have analyzed pre-treatment and post-treatment serum cytokines and chemokines of MCL patients with the aim of predicting prognosis and assessing the response to therapy. Recent report showed that the elevation of IL-12, IP-10, sIL-2Ra, MIG, IL-1RA, IL-8, MIP-1a, and MIP-1b in the serum of newly diagnosed MCL patients [69]. The elevated levels of sIL-2Ra, IL-8, MIG, MIP-1a, and MIP-1b were predictive of inferior event-free survival, and elevated sIL-2Ra, IL 8, and MIP-1b were predictive of a poor prognosis. Alteration in Toll like receptors (TLRs) is associated with B cell lymphomas including MCL [70]. Small RNAs including microRNA miR-127-3p, miR-615-3p, and miR-18b are associated with better overall survival of MCL patients, and provide prognostic markers that distinguish between indolent and aggressive forms [71-73]. Comprehensive information on these and other biomarkers, and notes on their roles in the pathogenesis of MCL, are compiled in Table 1. This table presents MCL related biomarkers from various cellular classes such as cell cycle regulators, transcription regulators, tumor suppression/necrosis, DNA/RNA repair regulators, immune/inflammatory and other signaling genes. The existence of these additional biomarkers, many of which have been identified during genome wide studies, illustrates the complexity and heterogeneity of this disease, and is consistent with reports indicating that MCL harbors the highest levels of genomic instability among malignant lymphoid neoplasms [22, 74].
Prognostic markers for MCL
Due to the paucity of early symptoms and biological heterogeneity, MCL is generally diagnosed at Stage III or IV. For staging of MCL, clinicians typically consider CBC, LDH, bone marrow aspirate and biopsy, while CT and PET to assess spread of the disease. PET scans were found to be superior to CT for predicting prognosis. A maximum standardized uptake value (SUVmax) of more than 5 from the area of most intense uptake correlated with inferior 5-year overall survival [75]. Mantle Cell Lymphoma International Prognostic Index (MIPI) is a widely used prognostic model which incorporates ECOG (Eastern Cooperative Oncology Group) performance status (ranging from 0-5), age, leukocyte count, lactic dehydrogenase, and tumor cell proliferation rate (Ki67 staining) to estimate prognosis. It allows stratification of MCL patients into low (44% of patients; median OS, not reached), medium/intermediate (35% of patients; median OS, 51 months) and high (21% of patients; median OS, 29 months) risk groups [76]. The initial prognostic significance of MIPI was dependent on the treatment regimen [77], but MIPI has recently been shown to be independent of treatment [78]. Other biomarkers and characteristic features of MCL that have shown prognostic significance for poor outcome include presence of blastoid or pleomorphic morphologic characteristics and high proliferation index at the time of diagnosis or after intense therapy [79]. In addition, high expression of eukaryotic initiation factor 4E, Myc and SOX11 overexpression, low TCL1 expression, TP53 alterations, lack of hypermutated heavy chain immunoglobulin variable regions, presence of higher absolute levels of monocytes, beta-2 microglobulin, monoclonal and polyclonal immunoglobulin free light chain, IL-2Rα, IL-8, and MIP-1β in the serum have been correlated with poor prognosis and inferior outcome [51, 69, 80-85]. CD3(+), CD8(+), and particularly CD4(+) T cells, if present in high numbers, are characteristic of the indolent form of MCL; a high CD4:CD8 ratio correlates independently with longer OS [86].
Therapeutic approaches for MCL
Early stage MCL
In 10-15% of patients, MCL is diagnosed at Stage I and II, also known as limited stage lymphoma. This subset is generally associated with an indolent course and better prognosis than patients diagnosed at advanced stages. The preferred modality of treatment for Stage I and II MCL, especially with non-bulky tumor load, is radiotherapy [6]. Multiple retrospective studies on small samples have indicated an affirmative effect of radiotherapy on various aspects of stage I and II MCL disease including progression free survival (PFS), OS, local disease control, and symptom relief [87]. A recently performed retrospective study on a large sample by Murthy et al. 2014 suggests that limited stage lymphoma patients receiving Radiation Therapy (RT) experienced improved OS; in addition, in multivariate analysis, administration of initial RT was associated with a significantly lower mortality rate [88].
Despite improved PFS and OS in patients receiving only radiotherapy, Stage I and II MCL patients often relapse within one year [89]. Therefore, a current recommendation includes shortened conventional chemotherapy induction followed by consolidating radiation for MCL patients carrying high tumor burden ( > 5 cm are presented below) and/or having poor prognostic factors (blastoid features, high Ki67 indices ( > 20-40%), high β2 microglobulin ( > 3 mg/l), and central nervous system involvement [6, 90]. In such cases, systemic therapy as indicated for advanced stages (see below) would be appropriate; however, radiotherapy alone has been reported to be equally effective even in MCL patients with relapse or with bulky tumor and/or extra-nodal presentation [91, 92]. Based on clinical trials on indolent and untreated MCL (Table 2), intense chemo-immunotherapy in young patients appeared to induce complete remission but was associated with higher side effects [93]. The combination of Bendamustine and Rituximab has produced completion remission with minimal adverse effects (although prolonged Bendamustine treatment is associated with lymphocytopenia) [94, 95]. We would advocate for the clinical trials comparing traditional chemo-immunotherapy with Bendamustine/Rituximab in combination with Bruton’s tyrosine kinase (BTK) inhibitors, as was recently examined in a Phase 1/1b trial for untreated and relapsed/refractory non-Hodgkin lymphoma [96] (Table 2).
Table 2: Table describing the details of the published clinical trials on the MCL drugs as single on in combination
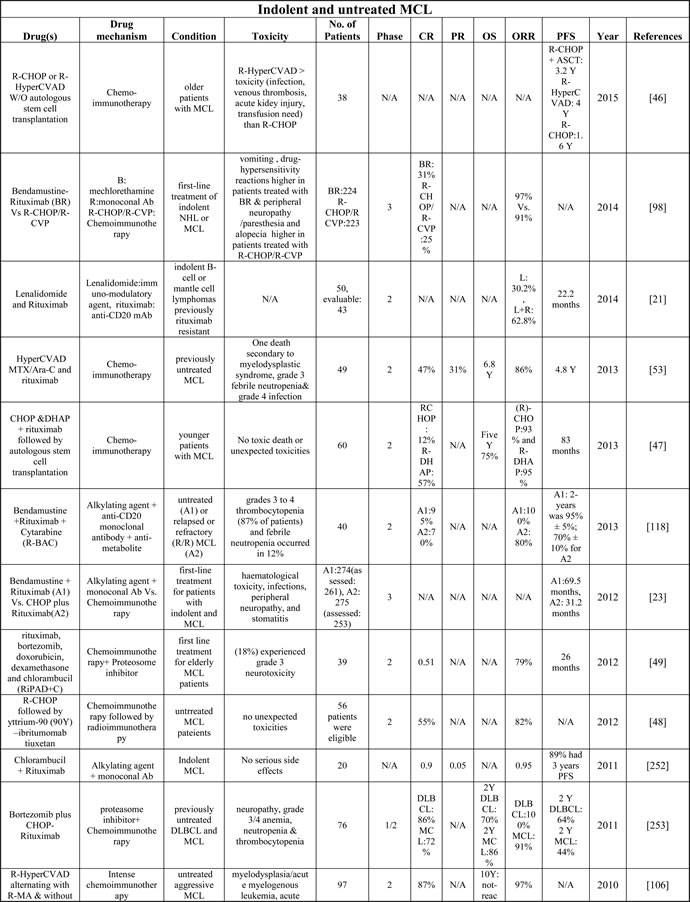
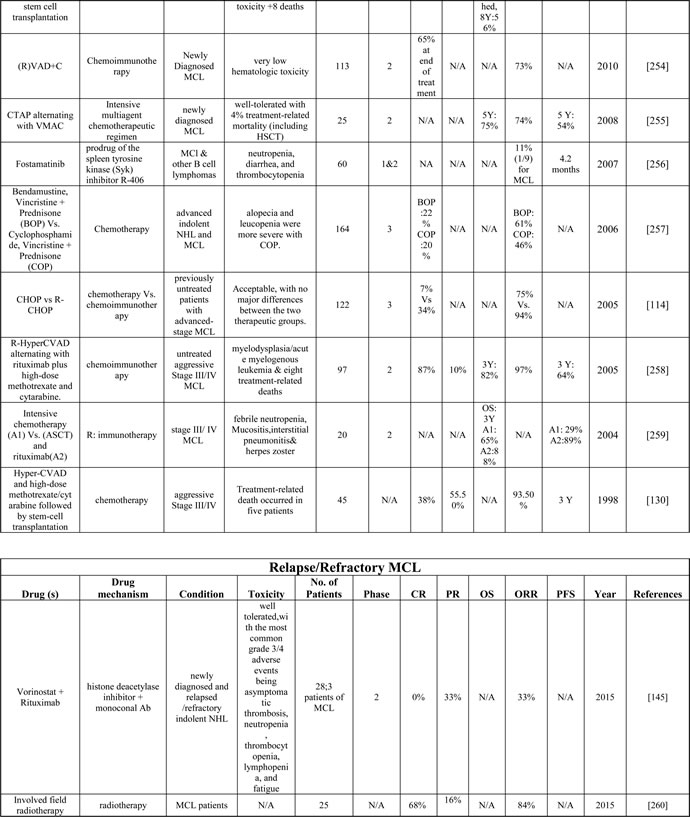
A: arm, P: phase, Y: year, R-DexaBEAM: rituximab,examethasone, carmustine, etoposide, cytarabine and melphalan; HDT: high-dose therapy; aNHL: aggressive NHL; iNHL: indolent Lymphoma; N/A : Not available. Please see Supplementary Table 2 to access complete table.
Late stage MCL
Stage III and IV MCL are known as aggressive/advanced MCL as they usually carry a high tumor burden with poor prognostic features. In these cases, treatment should be initiated after the diagnosis in both symptomatic and asymptomatic patients. Treatment is usually tailored individually based on patient’s age, symptoms, and risk factors [97]. Young patients (≤ 65 yr, no major comorbidities) are generally given intensive immuno-chemotherapy with autologous stem cell transplantation along with maintenance therapy to achieve better PFS and OS while elederly patients usually receive therapies tailored towards the individual characteristics. The phases of treatment for treatment-naïve late stage MCL are presented below.
Frontline/Firstline therapy
The ideal candidates for intensive strategies are patients aged 65 years or younger without significant comorbidities, while non-intensive strategies are reserved for elderly patients of more than 65 years or patients with significant comorbidities [98]. Several historical clinical trials have evaluated efficacy of various treatment regimens with respect to partial response (PR), complete response (CR), overall response rate (ORR), and OS; these have included CHOP, R-CHOP, Maxi-R-CHOP (R-CHOP followed by higher doses of cytarabine, followed by an autologous stem cell transplant); R-hyper-CVAD (Rituximab, cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with high-dose cytarabine and methotrexate) with or without autologous stem cell transplantation; BR (Bendamustine and Rituximab); R-FCM (Rituximab, fludarabine, cyclophosphamide, and mitoxantrone); R-DHAP (Rituximab, dexamethasone, cytarabine and cisplatin); R-CVP (Rituximab, cyclophosphamide, vincristine, and prednisone); R-CBP (Rituximab, cyclophosphamide, bortezomib, and prednisone); R-VAD+C (Rituximab, vincristine, doxorubicin, dexamethasone, chlorambucil); and RiPAD+C (rituximab, bortezomib, doxorubicin, dexamethasone, and chlorambucil) (Table 2). An analysis data from the NCCN NHL outcomes database confirmed that there were no differences in OS or PFS among patients receiving R-HyperCVAD or RCHOP+HDT/ASCR [99]. Multiple studies have confirmed that treatment with R-CHOP alone is inferior to either of these regimens and VR-CAP (bortezomib, rituximab, cyclophosphamide, doxorubicin, and prednisone) regimen [100-101].
In elderly patients, treatment of MCL needs to be “personalized” based on biological age, comorbidities, and general performance status. A comprehensive geriatric assessment via questionnaire allows overall estimation of life expectancy and tolerance of treatment as well as identifies reversible health conditions that may interfere with cancer treatment. Based on these criteria, elderly patients have been divided into three categories: fit, compromised, and frail. The aim of treatment in fit patients is complete remission, and includes conventional frontline immunochemotherapy such as R-CHOP or BR or chlorambucil plus VADC (vincristine, doxorubicin, oral dexamethasone) or PEP-C (prednisone, etoposide, procarbazine, and cyclophosphamide) followed by maintenance with Rituximab alone (Table 2). In compromised patients, the aim of the treatment is to control disease progression while balancing the efficacy of treatment based toxicities due to underlying comorbidities or impaired organ function. Hence, dose-adapted chemotherapy such as BR or Rituximab and chlorambucil with or without novel therapeutic agents may be recommended. In frail patients, preservation of quality of life along with symptomatic control of the disease is the main therapeutic goal. Therefore, mild chemotherapy such as chlorambucil with rituximab, prednisone, etoposide, procarbazine, or cyclophosphamide in different combination are usually recommended without autologous cell transplantation (Table 2). Usually such patients benefit from combination R-CVP (Rituximab, cyclophosphamide, vincristine, and prednisone), or a newer regimen of R-CBP (Rituximab, cyclophosphamide, bortezomib, and prednisone) [6, 102]. In general, BR is becoming the treatment of choice over R-CHOP/R-CVP in older patients [94, 102], although addition of low dose cytarabine with BR has shown CR of 95% in untreated older patients [116]. An alternative combination of an initial regimen of cytarabine and rituximab alternating with R-CHOP and followed by cytarabine and fludarabine achieved 87% CR [117].
Consolidation therapy
Consolidation therapy is relatively short term therapy mainly given after the frontline/induction therapy. Ionizing radiation, whether in the form of local radiation therapy, radioimmunotherapy, or total-body irradiation in preparation for autologous hematopoietic stem cell transplantation (ASCT) may be used for treating MCL in clinical practice. High dose chemotherapy with Carmustine/BCNU, etoposide, cytarabine, and melphalan (BEAM) followed by ASCT is also available. BEAM strategy has recently been refined by adding/replacing individual drugs from the BEAM group. Recent data showed that BeEAM (bendamustine, etoposide, cytarabine, melphalan) and Bortezomib-BEAM (V-BEAM) provide feasibility and efficacy similar to BEAM with slightly higher PFS and OS for treatment of MCL. Another newer strategy is to administer bortezomib in a combination referred to as VcR-CAP (bortezomib/Velcade, rituximab, cyclophosphamide, doxorubicin/Adriamycin, and prednisone) in previously untreated MCL patients [103]. Stem cell transplantation in MCL patients is increasingly being used as consolidation therapy following standard chemotherapy. However, this is somewhat controversial as some evidence suggests that chemotherapy without stem cell transplant is equally effective [104]. However, the inclusion of stem cell transplantation has shown promise in inducing long term disease free survival in MCL patients: a regimen of rituximab and ASCT produced durable remission in patients with first remission, and non-myeloablative allogeneic stem cell transplantation (NST) produced durable remission in patients with relapsed or refractory disease [105]. A group of patients who received frontline intensive induction immunochemotherapy (Maxi-CHOP alternating with high-dose cytarabine and BEAM/BEAC with ASCT) plus maintenance with Rituximab showed median overall survival and response duration longer than 10 years, and a median event-free survival of 7.4 years [106]. In a recent study, no significant differences were found in 5-year overall survival rates for autologous vs. reduced-intensity conditioning allogeneic hematopoietic stem cell transplantation in either early or late transplantation cohort [107].
Consolidation is often achieved with therapeutic agents in combination with ASCT or in place of ASCT, especially in elder compromised or frail patients. In a 10 year follow up of young patients treated with R-Hyper-CVAD alternating with Rituximab, methotrexate, and Cytarabine (R-M-A) without ASCT, it was reported that median OS had not been reached and that the median time to treatment failure (TTF) was 5.9 years [104]. High dose consolidation therapy such as with methotrexate/cytarabine or busulfan/melphalan before ASCT achieved high PFS in untreated aggressive MCL [108-109]. Other novel drugs such as temsirolimus, ibrutinib, and lenalidomide have been tested as agents for consolidative therapy [6]. The Eastern Cooperative Oncology Group (ECOG) trial E1499, which used 90Y-ibritumomab tiuxetan RIT consolidation after R-CHOP in newly diagnosed MCL, showed improved 5 year OS in both young and elderly patients [110].
Maintenance therapy
Maintenance therapy is given after completion of induction therapy and is intended to achieve longer term remission. The primary criteria for maintenance agents include ease of administration, minimal toxicity, and efficacy of maintaining remission. Rituximab is the most commonly used agent for maintenance therapy for MCL of all stages; it was first approved for initial treatment of non-Hodgkin lymphoma in 1997. It is being used both as a single agent and in combination with newer drugs; the combinations have achieved greater CR, PFS, and improved OS in both young and older patients [111]. Interferon alpha as a maintenance therapy has been found to be inferior to ASCT or Rituximab and carries higher toxicity profile [112]. Newer drugs such as lenalidomide, which are primarily used in induction therapy, have also been evaluated for maintenance therapy as a single agent or in combination with other drugs, particularly rituximab [113]. Similarly, Ibrutinib, lenalidomine and bortezomib are being evaluated for maintenance in MCL and as a component of the frontline therapy via LyMa and MCL 0208 trials; Clinicaltrials.gov/ NCT00921414, NCT02242097, NCT02354313 and EudraCT Number 2006-000386-11, and NCT00310037, respectively (Table 3; [114-115]).
Table 3: Status of active MCL clinical trials registered with NIH at clinicaltrials.gov
Drug |
Drug mechanism |
Condition |
No. of Patients |
Phase |
Clinical trial number*(registered at clinicaltrials.gov) |
First received year |
Status |
Ibrutinib maintenance |
BTK Inhibitor |
MCL |
36 |
2 |
NCT02242097 |
2014 |
Recruiting |
Lenalidomide maintenance versus observation |
immuno-modulatory agent |
Advanced MCL |
300 |
3 |
NCT02354313 |
2014 |
Recruiting |
ACP-196 |
BTK Inhibitor |
relapsed or refractory MCL |
Recruiting/ estimated number:120 |
2 |
N+F2:I15CT02213926 |
2014 |
Recruiting |
Bendamustine, Rituximab, Ibrutinib |
Alkylating agent + monoconal Ab + BTK Inhibitor |
Newly Diagnosed MCL |
520 |
3 |
NCT01776840 |
2013 |
Active, not recruiting |
GS-9973 |
Spleen tyrosine kinase (SYK) inhibitor |
Relapsed/Refractory MCL, CLL, DLBCL & iNHL |
280 |
2 |
NCT01799889 |
2013 |
Recruiting |
GS-9973 + Idelalisib |
Spleen tyrosine kinase (SYK) inhibitor & PI3Kδ inhibitor |
Relapsed or Refractory Hematologic Malignancies (MCL,CLL, FL, DLBCL and iNHL) |
200 |
2 |
NCT01796470 |
2013 |
Active, not recruiting |
SGN-CD19A |
anti-CD19 mAb linked to monomethyl auristatin F (MMAF), a cytotoxic agent |
MCL |
120 |
1 |
NCT01786135 |
2013 |
Recruiting |
Ublituximab+Ibrutinib |
Anti-CD20 monoclonal antibody+Bruton's Tyrosine Kinase (BTK) inhibitor |
MCL,Chronic Lymphocytic Leukemia |
60 |
2 |
NCT02013128 |
2013 |
Enrolling by invitation |
AT7519M |
CDK Inhibitor |
Relapsed MCL |
12 |
2 |
NCT01652144 |
2012 |
Completed; results are awaited |
Carfilzomib, Lenalidomide, Rituximab |
proteasome inhibitor + immuno-modulatory agent + monclonal Ab |
Relapsed/Refractory MCL |
68 |
1&2 |
NCT01729104 |
2012 |
Recruiting |
IMMU-114 |
Humanized mAb against HLA-DR |
Relapsed or Refractory NHL and CLL |
50 |
1 |
NCT01728207 |
2012 |
Recruiting |
CEP-9722 + Gemcitabine + Cisplatin |
PARP inhibitor + antimetabolite deoxynucleoside analogue + inorganic platinum agent |
MCL |
24 |
1 |
NCT01345357 |
2011 |
Completed; results are awaited |
CC-122 HCL |
Pleiotropic Pathway Modulator |
MCL |
140 |
1 |
NCT01421524 |
2011 |
Recruiting |
CDX-1127 (Varlilumab) |
Monoclonal antibody targeting CD27 |
refractory or relapsed CD27 Expressing B-cell Malignancies and selected types of solid tumors |
170 |
1 |
NCT01460134 |
2011 |
Recruiting |
Ofatumumab + Bendamustine |
Alkylating agent & CD20 antibody |
MCL Ineligible for Autologous Stem Cell Transplant |
76 |
2 |
NCT01437709 |
2011 |
Recruiting |
Panobinostat + Bortezomib |
histone deacetylase inhibitor (HDAC inhibitor) &Proteasome inhibitor |
Relapsed and/or Refractory MCL |
24 |
1 |
NCT01504776 |
2011 |
Completed; results are awaited |
R-CHOP-14R-HIDAC followed by RIT/HDT/ASCR |
Sequential Chemo-Radioimmunotherapy Followed by Autologous Transplantation |
Untreated Advanced Stage MCL |
96 |
1&2 |
NCT01484093 |
2011 |
Active, not recruiting |
SAR245409+Rtuximab + Bendamustine |
Phosphoinositide 3-kinase inhibitor (PI3K inhibitor)+monoclonal Ab+alkylating gent |
Relapsed or Refractory MCL & other kinds of lymphoma |
85 |
2 |
NCT01403636 |
2011 |
Completed; results are awaited |
SNS01-T |
Small inhibitory RNA molecule that blocks the expression of Factor 5A mRNA |
MCL in Relapse and other B cell malignancies |
15 |
1&2 |
NCT01435720 |
2011 |
Active, not recruiting |
PD 0332991 + Bortezomib |
Cyclin-dependent kinase 4 and 6 inhibitor & Proteasome inhibitor |
Relapsed MCL |
30 |
1 |
NCT01111188 |
2010 |
Unknown |
Bendamustine + rituximab versus CHOP + rituximab |
B: mechlorethamine R:monoconal Ab RCHOP:Chemoimmunotherapy |
first-line treatment for patients with stage III or IV indolent or MCL |
549 |
3 |
NCT00991211 |
2009 |
Completed; results are awaited |
Tositumomab and Iodine I 131 Tositumomab followed by CHOP |
iodine-131 labeled anti-CD20 murine IgG2a monoclonal antibody + chemotherapy |
untreated MCL |
25 |
2 |
NCT00992992 |
2009 |
Completed; results are awaited |
Rituximab as maintenance |
CD20 antibody |
MCL |
299 |
3 |
NCT00921414 |
2009 |
Completed; results are awaited |
Vorinostat |
histone deacetylase (HDAC) inhibitor |
MCL and other B cell NHL |
54 |
2 |
NCT00875056 |
2009 |
Active, not recruiting |
Bortezomib as maintenance |
Proteasome inhibitor |
untreated MCL |
151 |
2 |
NCT00310037 |
2008 |
Active, not recruiting |
Clofarabine |
Nucleoside analogue |
Relapsed/Refractory NHL |
25 |
1&2 |
NCT00644189 |
2008 |
Completed; results are awaited |
Epratuzumab or rituximab |
anti-CD22 + anti-CD20 monoclonal antibody |
NHL patients receiving antibody treatment |
500 |
Not available |
NCT00398372 |
2006 |
Completed; results are awaited |
RT-PEPC (Rituximab, Thalidomide, Prednisone, Etoposide, Procarbazine, Cyclophosphamide) |
Monoclonal antibody, immunomodulatory, immunosuppressant drug, Topoisomerase inhibitor, alkylating agent |
Relapsed Mantle Cell Lymphoma |
46 |
2 |
NCT00151281 |
2005 |
Unknown |
Cladribine |
Antimetabolite |
Mantle Cell Lymphoma |
48 |
2 |
NCT00002879 |
1999 |
Completed; results are awaited |
Treatment of relapse/refractory MCL
Among NHL, MCL has a higher rate of relapse, possibly due to its heterogeneity and complexity. NCCN Guidelines provide choice of several therapeutics which may be used as single agents or as part of a combination therapy regimen for relapse/refractory MCL. High dose therapy along with stem cell transplantation is administered in patients with relapse/refractory MCL unless they have undergone high dose therapy and autologous stem cell transplant previously. Few studies have evaluated the benefits of salvage chemotherapy with traditional agents such as R-FCM (Rituximab, Fludarabine, cyclophosphamide, and mitoxantrone), R-GemOc (Rituximab, gemcitabine, oxaliplatin), R-DHAP, or BR [118-121]; novel agents have been preferred over these traditional chemotherapeutic agents for treatment of relapse/refractory MCL. Temsirolimus, bortezomib, lenalidomide, and ibrutinib are approved agents in the European Union. Initial clinical trials in which each of these four agents was used as a single agent against relapse and refractory MCL gave ORR as 33% for bortezomib, 28% for lenalidomide, 22% for temsirolimus, and 68% for ibrutinib [122-125]. Combinations of some of these novel agents with other novel or traditional agents have been explored (Table 3). Some recent clinical trials indicate that a newer proteasome inhibitor (Carfilzomib) can be combined with other agents including bendamustine, BEAM regimen followed with ASCT or CDK9 inhibitor although neurotoxicity may be a limiting factor [126-129]. Lenalidomide, an immunomodulatory agent, has shown promising results in MCL cases refractory to bortezomib, and in cases previously treated with other agents or combinations [124,130]. Ibrutinib, an inhibitor of Bruton’s tyrosine kinase, has demonstrated outstanding efficacy in heavily pretreated relapse/refractory MCL with minimal side effects, high response rate, and high PFS; in combination with rituximab and bendamustine, OR of 94% and CR of 76% were achieved [96]. Current clinical trials have been designed with Ibrutinib, bortezomib, and other novel agents to determine the best possible combination for treating MCL (Table 2). In addition to therapeutic agents, radioimmunotherapy with 90Y-ibritumomab tiuxetan produced an ORR of 31%. In another study in 16 patients with history of relapse or refractoriness to treatment, high dose 131I-tositumomab followed by high doses of etoposide and cyclophosphamide produced a CR rate of 91%, an ORR of 100%, and PFS of 61% [131]. Based on published clinical trial results, single agents usually seem to fail but drug combinations have given somewhat better results especially for relapse/refractory MCL (Table 2). Trials that have included Bendamustine in the regimen seem to induce complete remission, but more phase 3 clinical trials are required to demonstrate feasibility of the combination of bendamustine/rituximab with BTK inhibitors to achieve higher remission rates [96, 126, 132].
Major clinical trials and their outcomes for indolent, untreated and refractory/relapse MCL are described in chronological order in Table 2. The data demonstrate how clinical trials have been useful in refining treatment strategies over decades. Newer clinical trials incorporating combination chemotherapeutic agents with novel MCL drugs are described in Table 3. These newer clinical trials (Table 3) include both ongoing trials and trials in the recruitment stage; these trials will help us to evaluate putative beneficial roles of novel therapeutic agents especially during treatment of first-time/untreated MCL. We anticipate that the results will provide avenues for reducing side effects of chemotherapeutic agents and minimize the relapse rates.
Novel drugs for MCL based on signaling pathways
Several research groups have taken a novel approach aimed at targeting the molecular mechanisms involved in pathogenesis of MCL. Many of these therapeutic agents have shown promise in preclinical studies and clinical trials. The properties of these chemotherapeutics and novel drugs currently in use or being developed for MCL, which may help to elucidate the therapeutic effect of these drugs against MCL, are detailed in Table 4. Cellular proteins and signaling pathways that could be targeted with novel inhibitors for treatment of MCL are described below (Figure 3).
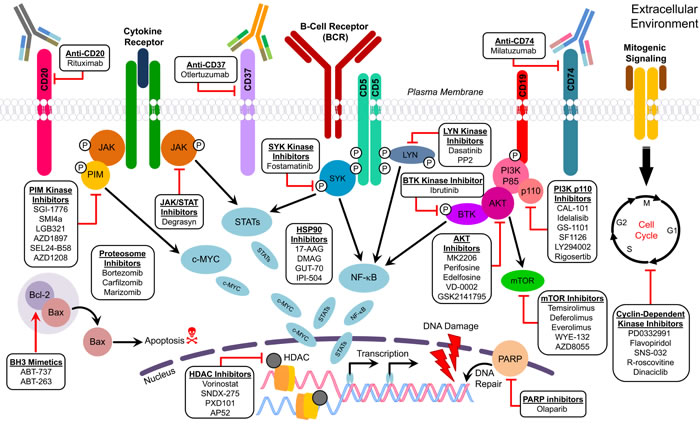
Figure 3: Figure describing the molecular targets of therapeutic agents used for MCL.
CDK inhibitors
MCL involves aberrant expression of Cyclin D1 which causes dysregulation of regulators of the cell-cycle including CDK4. CDK4 is especially important for regulating the G1-S phase transition; its activity is negatively regulated by CDK inhibitors p16INK4A, p27Kip1, and p21Waf1/Cip1. The CDK4/6 inhibitor, PD0332991 has shown promise in both preclinical and clinical studies for treating MCL [133]. In a pilot study on relapse/refractory MCL patients, activity of PD0332991 was monitored via 3-deoxy-3[18F]-fluorothymidine (FLT) and 2-deoxy-2-[18F] fluoro-D-glucose (FDG) positron emission tomography/computed tomography (PET/CT) imaging as well as with Ki67 staining of the biopsy samples. There was > 70% reduction in summed FLT SUV (max), > 90% reduction of expression of phospho-Rb, and ≥ 87.5% reduction of expression of Ki67, although no long term changes in disease progression were noted [134]. Flavopiridol, a broad cyclin-dependent kinase inhibitor has been shown to induce apoptosis and to down regulate cell cycle proteins and anti-apoptotic proteins in in vitro models [135]. Flavopiridol showed limited utility against MCL as a single agent, but in combination with fludarabine and rituximab it showed median PFS of 21.9 months and 70% CR [136]. The effect of Flavopiridol can also be potentiated by combination with agents targeting other molecular pathways, including Hsp90 inhibitor 17-AAG [137]. In phase 1 clinical trials, flavopiridol in combination with the proteasome inhibitor, bortezomib, showed efficacy for treatment of MCL patients with relapsed/refractory status [138]. Other CDK inhibitors such as SNS-032, an inhibitor of cdk2, 7, and 9 act by inhibiting RNA pol II phosphorylation and reducing Mcl-1 levels [139]. From the therapeutic standpoint, small-molecule CDK inhibitors such as CYC202 (Seliciclib, R-roscovitine), a purine analogue and a selective inhibitor of cdk2-cyclin E, cdk7-cyclin H, and cdk9-cyclin T has been shown to induce increased expression of apoptosis regulatory genes and decreased Cyclin D1 in MCL cell lines REC, Granta-519, JeKo-1, and NCEB-1 [140]. Similarly, other cyclin-dependent kinase inhibitors including P276-00, SNS-032 and flavopiridol have shown clinical activity against MCL [141]. SCH727965 (dinaciclib), a novel inhibitor of CDK1, 2, 5, and 9, showed superior activity and an improved therapeutic index compared with flavopiridol in laboratory models of solid and hematologic malignancies [142]. In in vitro and in vivo studies using silvestrol, Alinari et al. 2012 found reductions of phosphorylated Rb, E2F1 protein, and E2F1 target transcription as well as mitochondrial depolarization and caspase-dependent apoptosis [143]. Chiron and colleagues showed that inhibition of CDK4 with PD 0332991 (palbociclib) sensitizes ibrutinib-resistant lymphoma cells to ibrutinib in the absence of BTK mutations; however, use of PI3K inhibitors was more effective for treating cells carrying a BTK mutation (C481S) [144]. Similarly, in a recent trial with vorinostat and Rituximab, 33% of patients (n = 3) achieved PR and ORR [145]. Additional clinical trials have been designed to evaluate the efficiency of CDK inhibitor alone and in combination for MCL (Table 3).
BCR inhibitors
B cell receptor (BCR)-mediated signaling pathways are linked with expression adaptor molecules (e.g., GAB1, BLNK, GRB2, CARD11), activities of kinases (e.g., LYN, SYK, PI3K), and phosphatases (e.g., SHIP-1, SHP-1, PTEN) leading to inhibition of NF-κB, ERK, mTOR, and GSK3 pathways. Since MCL is a B cell derived lymphoma, the BCR and its associated signaling pathways are central players in supporting the B cell microenvironment-based B cell homing, survival and drug resistance [146]. Therefore, suppression of BCR signaling pathways via therapeutic agents which can block the upstream or downstream signaling molecules should lead to apoptosis of MCL cells. Furthermore, inhibition of BCR pathways also interferes with BCR-regulated interactions with the TME, which leads to disruption of secretion of pro-survival chemokines, increased release of lymphoma cells from the lymph nodes, and enhanced tumor cell death [147,148]. Not many recently completed clinical trials have included BTK inhibitors in their regimen (Table 2), but newly designed clinical trials have begun to incorporate BTK inhibitors into their drug regimen (Table 3). Other BTK inhibitors are also being assessed via in vitro studies. Dasatinib, an inhibitor of tyrosine kinases, suppressed BCR-induced LYN, BCR-dependent EGR-1 upregulation, and cell survival in primary MCL cells. Similarly, PP2, a specific inhibitor of Src kinases suppressed constitutive LYN activation and induced apoptosis of MCL cells [149]. BTK is one of the major regulators of proliferation and cell survival in MCL and can be inhibited in the BCR pathway by ibrutinib, which down-regulates both phospho-STAT3 (pSTAT3) and NF-κB [150]. Ou et al. 2013 showed that microRNA-155 (miR-155) functions as a positive regulator of STAT3 while SOCS1 functions as a negative regulator of both STAT3 and NF-κB; they suggested that activity of ibrutinib and BCR inhibitors could be monitored via miR-155 [151]. In a recent in vitro study, ibrutinib and another SYK inhibitor, Fostamatinib, blocked chemotactic signaling by IL-1β, TNFα, and CCL5 which are required for MCL cell adhesion to human bone marrow stromal cells [148]. The combination of ibrutinib, spleen tyrosine kinase inhibitor R406, and HDAC inhibitor vorinostat increased apoptosis of MCL cells, which correlated with activation of caspase-3 and poly-(ADP-ribose) polymerase cleavage. Genomic profiling confirmed the down-regulation of NF-κB1/p105 and Cyclin D1 thus suggesting that this combination should be investigated in clinical studies [152]. Despite its promise, results have shown that MCL cell lines and patients can develop resistance to ibrutinib. Ibrutinib-resistant MCL cell lines (based on the therapeutic dose of 560 mg = 0.4 μg/μl) possessing a lack of normal BTK expression or presence of mutant BTK (C481S) were characterized by failure to inhibit phosphorylation of ERK and Akt signaling pathways, sustained PI3K-AKT activity, and activation of alternative NFκB pathway [153, 154].
mTOR inhibitors
MCL pathogenesis involves PI3K/AKT/ mTOR pathway activation. mTOR inhibitors, including temsirolimus (which is approved for relapse/refractory MCL in European countries), ridaforolimus, and everolimus impart their therapeutic benefit by inhibiting mTORC1 via allosterical binding.These agents have been tested for relapse/refractory MCL cases, but their efficacy in a single drug regimen has been low [155]. In vitro analysis indicated that low 4EBP1 expression and/or high eIF4E expression by lymphoma cells conferred resistance to mTOR inhibitors, explaining the poor therapeutic efficacy against MCL [156]. Several reports have suggested that treatment with NVP-BEZ235 could inhibit mTORC1 and mTORC2, as well as PKI3, and induce apoptosis of MCL cells by down-regulating constitutive Mcl-1 expression in chemo-naive MCL cell lines [157] and in bortezomib-resistant cell lines [158]. Other mTOR inhibitors such as WYE-132, AZD8055, PP242, and OSI-027 induced anti-proliferative activity and apoptosis via activation of the PUMA and BIM genes, and inhibition of the Akt signaling pathway [159]. Recent studies in an in vivo model with 13-197, a quinoxaline analog that specifically perturbs IκB kinase (IKK) β, a key regulator of the NF-κB pathway, disrupted both NF-κB and mTOR pathways resulting in downregulation of NF-κB and NF-κB-regulated genes such as Cyclin D1, Bcl-XL, and Mcl-1 as well as phosphorylation of S6K and 4E-BP1, the downstream mediators of the mTOR pathway [160]. None of the dual mTORC1/2 inhibitors have reached the clinical trial phase, but recently designed clinical trials have incorporated mTOR inhibitors in their treatment regimen for relapse/refractory MCL cases (Tables 2, 3).
PI3K p110 inhibitors
PI3K functions early in the AKT signaling pathway. MCL is characterized by increased PI3K p110α expression especially in relapsed cases. The therapeutic efficacy of PI3Kp110 inhibitors such as CAL-101/idelalisib/GS-1101, SF1126/LY294002, rigosertib, and BYL719 have been validated via in vitro studies for hematological malignancies [161]. The oral agent idelalisib has been shown to block PI3K/AKT signaling and to promote apoptosis in MCL cell lines [162]. Idelalisib use in relapse/refractory cases of MCL yielded median PFS of 3.7 months and 1-year PFS of 22% [163]. A study by Chiron et al. 2013 [164] suggested that the tumoricidal effect of idelalisib is enhanced by G1-arrest of the cell cycle; therefore, idelalisib should be administered with concomitant inhibition of CDK4/CDK6 to increase the sensitivity of MCL cells to treatment regimen. The activity of idelalisib is largely non-overlapping with that of anti-CD20 antibodies (except for effects on phagocytic activity of microphages); thus, combination therapy with idelalisib and rituximab is found to be synergistic for patients with CLL [165]. In clinical trials, treatment with a pan-isoform PI3K and mTOR inhibitor SAR245409 (XL765) for relapse/refractory lymphoma produced partial remission [166]. These findings are being extended in additional clinical trials, one comparing idelalisib in combination with Rituximab versus BR for MCL (NCT01410513), and another trial employing idelalisib as single agent for relapse/refractory MCL (NCT01403636) (Table 3). In addition, PKC beta, a downstream regulator of BCR signaling has been found to be overexpressed in MCL. Enzastaurin, an oral serine/threonine kinase inhibitor, downregulates PKCbeta/PI3K/AKT pathways. It suppresses angiogenesis and proliferation resulting in induction of apoptosis. A phase 2 clinical trial has confirmed enzastaurin as a promising drug for treating relapse/refractory MCL [167]. In vitro studies demonstrated that treatment with enzastaurin and bortezomib together results in increased levels and an increased ratio of pro-apoptotic proteins (Bax, Bad, and Bim) relative to anti-apoptotic proteins (Bcl-2, Bcl-xL, and Mcl-1) [168]. Because of their ability to target two major pathological pathways thought to be involved in MCL, it would be reasonable to proceed with clinical trials aimed at incorporating these agents into the treatment regimen for MCL.
AKT inhibitors and anti-tumor lipids
Activation of the AKT signaling pathway is known to cause proliferation by suppression of apoptosis as a consequence of direct phosphorylation of proapoptotic proteins such as Bad and pro-caspase-9. In preclinical studies, a combination of therapeutic agents that target the PI3K/AKT/mTOR signaling pathways has shown considerable anti-proliferative and pro-apoptotic activity against MCL cells. The efficacy of AKT inhibitors is usually monitored via expression levels of pAKT before and after treatment. Several AKT inhibitors are available including MK2206, perifosine, edelfosine, triciribine phosphate monohydrate (TCN-PM, VD-0002) and GSK2141795. MK2206 has been recently shown to significantly reduce phosphorylation of AKTser473 and to reduce phosphorylation of its target BADser110 in MCL cell lines. In addition, treatment with MK2206 also led to p21-coupled growth arrest in MCL cell lines [169]. An abnormal activation of lipid rafts by AKT has also been reported. Lipid rafts are cholesterol- and sphingolipid-rich membrane domains that act as platforms to colocalize proteins involved in intracellular signaling pathways. Lipid rafts control cell survival and cell death; they often become dysfunctional in cancers including MCL. The treatment with perifosine and edelfosine inhibits AKT and induces apoptosis in MCL cells and in xenograft-derived animal models; in the latter, pretreatment with PI3K inhibitor wortmannin potentiates the effect of perifosine [170].
HSP90 inhibitors
Hsp90 (heat shock protein 90) is a molecular chaperon that facilitates correct protein folding; it stabilizes number of proteins that are involved in oncogenesis including NF-kappa B, BCR-ABL, NPM-ALK, AKT, and mutated p53 [171]. Thus, inhibitors of Hsp90 such as 17-AAG, suberoylanilide hydroxamic acid, 17-DMAG (17-dimethylaminoethylamino-17-demethoxygeldanamycin), GUT-70, and IPI-504 may have therapeutic value for treating various cancers. In vitro studies on MCL cell lines with 17-AAG led to downregulation of Cyclin D1, cdk4, and AKT, depletion of Bid, and activation of the intrinsic/mitochondrial caspase pathway [172]. The combination of 17-DMAG and HDAC inhibitor vorinostat exhibited a synergistic effect culminating in cell cycle arrest at G2/M and G1 and decreased expression of Cyclin D1, cdk4, c-Myc, c-RAF, and AKT leading to extensive apoptosis in MCL cell lines and primary cells [173]. Interestingly, GUT-70 demonstrated antiproliferative effects in forms of MCL in which p53 was mutated (mt-p53); it induced mitochondrial apoptosis with upregulation of NOXA and downregulation of Mcl-1 in mt-p53 cells, but affected only Mcl-1 expression in cells with wild type p53, indicating the specific activity of GUT-70 toward pathways affected by mutant p53 [174].
Proteasome inhibitors
In addition to inhibiting the ubiquitin-proteasome complex, proteasome inhibitors induce the death response in transformed cells by inhibition of NF-κB-mediated anti-apoptotic response, upregulation of pro-apoptotic genes, and by stimulating accumulation of reactive oxygen species (ROSs). Several proteasome inhibitors have been tested against MCL. The first generation proteasome inhibitor bortezomib, a peptide boronic acid, targets all ubiquitin-tagged proteins, and acts via oxidative and endoplasmic reticulum (ER) stress leading to upregulation of proapoptotic protein NOXA as well as Bax and Bak [175]. Although bortezomib has been approved for treatment of relapse/refractory cases of MCL, a high incidence of peripheral neuropathy and gastrointestinal toxicity has limited its therapeutic usage [176]. Moreover, resistance to bortezomib has been reported among cases showing plasmacytic features and upregulation of IRF4 and CD38 [40]. Genome-wide DNA methylation analysis showed that bortezomib treatment resulted in hypomethylation of the NOXA gene; however, further in vitro studies demonstrated that administration of methyltransferase inhibitor decitabine (DAC) along with bortezomib overcame bortezomib resistance in relapsed/refractory MCL [177]. The combination of HSP90 antagonists with bortezomib has been found to be effective in nullifying bortezomib resistance mediated by ER chaperone BiP/Grp78 [178]. In addition, MLN9708, CEP-18770, and irreversible inhibitors such as carfilzomib and NPI-0052/Marizomib have shown promise in preclinical studies for MCL. Activities of these inhibitors can be monitored via enzymatic activity of each proteasomal subunit [179]. Carfilzomib, which is currently being evaluated in a phase 2 study for relapse/refractory MCL, induced apoptosis with the activation of JNK, Bcl-2, and mitochondria-related pathways. Its activity was dependent on the immunoproteasome subunit LMP2, which can function as a biomarker to monitor the therapeutic effect of carfilzomib [180]. While many proteasome inhibitors inhibit the catalytic activity of both constitutive proteasome and immunoproteasome, agents that can selectively inhibit the immunoproteasome have recently received attention. The immunoproteasome is a cytokine-inducible form of constitutive proteasome known to be expressed in lymphoid cell-derived hematological neoplasms. Immunoproteasome-specific inhibitors (IPSI) such as IPSI-001, PR-924, and PR-957 have not yet been assessed in clinical trials for MCL. Agents such as MLN4924 which inhibits the NEDD8 activating enzyme E1 leads to inhibition of the ligase complex, thereby limiting protein degradation by the proteasome. MLN4924 treatment has been shown to result in accumulation of NOXA protein in primary MCL and to induce apoptosis of MCL cells in a NOXA-dependent manner [181]. A phase 1 study for determining the therapeutic efficacy of MLN4924 has been completed. Similarly, inhibition of E3 ubiquitin ligase HDM2 by RG7112 leads to stabilization of selected proteins and suppression of the ubiquitin-proteasome system. RG7112 targets the murine double minute (mdm2) oncogene and activates p53 signaling in tumor cells [182].
PARP inhibitors
Poly (ADP-ribose) polymerase (PARP) is a key component of DNA single strand break (SSB) repair machinery. Treatment with PARP inhibitors leads to conversion of unrepaired SSB lesions into DNA double strand breaks (DSBs) during DNA replication. Since MCL is characterized by inactivation of ATM gene and lack of DSB repair capacity, PARP inhibitors cause accumulation of extensive DNA DSBs and cell-death [183]. Although PARP inhibitors such as AG-014699/PF-01367338, AZD2281, ABT-888, XAV939, and others are mainly used for solid tumors, a few in vitro studies have demonstrated efficacy of PARP inhibitors against MCL. ATM-deficient MCL cell lines are especially sensitive to PARP inhibitor olaparib, the activity of which correlates with levels of ATM in p53-deficient gastric cancer. Cell lines deficient in both ATM and p53 genes were more sensitive than cells lacking ATM function alone [184]. A phase 1 clinical trial for MCL with CEP-9722 in combination with Gemcitabine and Cisplatin has recently been completed (NCT01345357) (Table 3).
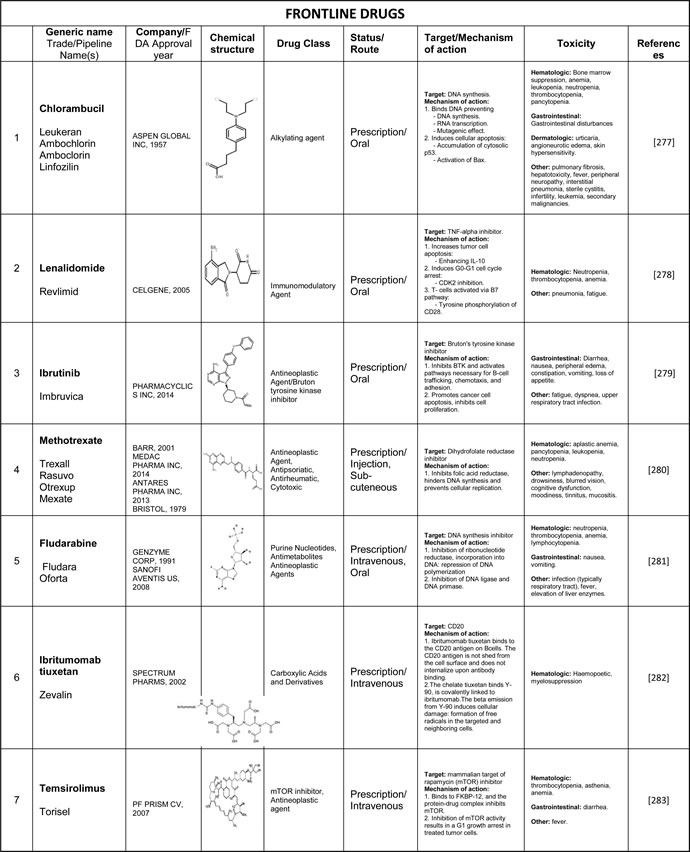
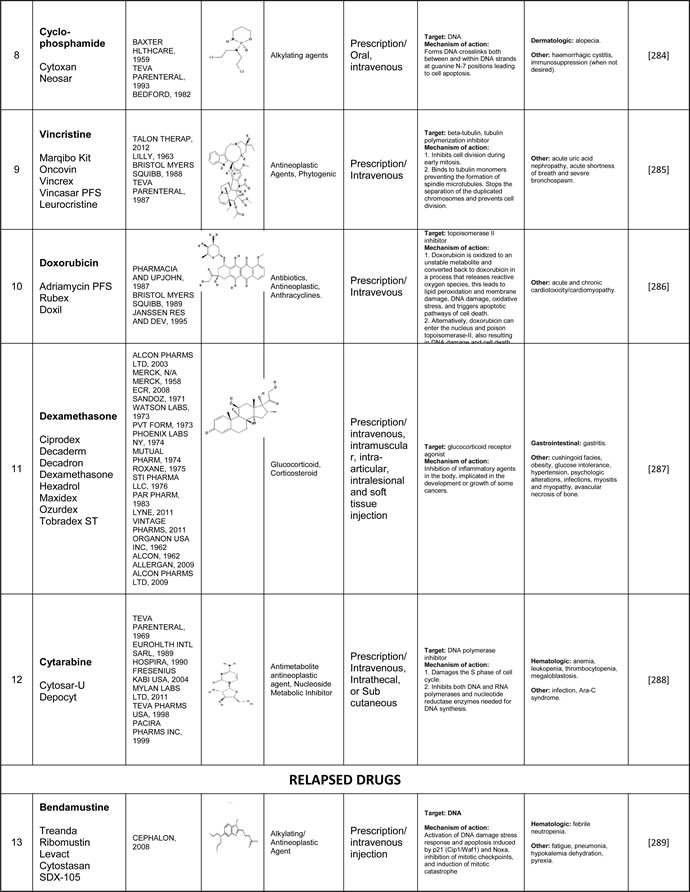
Table 4: The pharmacological details of the drugs currently in use or being developed for MCL. Please see Supplementary Table 4 to access complete table.
JAK/STAT inhibitors
JAK/STAT over-activation and mutations of SOCS1, an inhibitor of JAK/STAT pathway, have been reported in MCL [185, 186]. Degrasyn imparts antitumor activity in lymphoid tumors by inhibiting key growth and survival signaling (JAK/STAT) pathways. Degrasyn was shown to inhibit constitutively activated pSTAT3 and NF-κB. This results in inhibition of c-Myc, Cyclin D1, and Bcl-2 protein expression and upregulation of bax protein expression. Degrasyn has been found to potentiate the effect of Bortezomib against MCL cell lines [187]. There are other JAK inhibitors, such as AG490 are active against large cell lymphoma, but these have not been reported to be efficient against MCL cell lines [188].
HDAC inhibitors
In cancer, it is common to find hypermethylation of tumor suppressor genes and hypomethylation of oncogenes. Histone deacetylases (HDACs) control gene expression by removing acetyl groups from histones and preventing the transcriptional machinery from obtaining access to their target gene. This results in transcriptional repression. HDAC inhibitors cause histone acetylation leading to restoration of expression of tumor suppressor and/or cell cycle regulatory genes in cancer cells resulting in suppression of proliferation of these cells [189]. HDAC inhibitors have shown promising anti-tumor activity (cell cycle arrest, cellular differentiation, and apoptosis) as well as inhibition of VEGF in both in vitro and in vivo models [190]. Many of the HDAC inhibitors are being tested against MCL. Among these, suberoylanilide hydroxamic acid (SAHA; vorinostat), an oral HDAC inhibitor, suppressed translation of Cyclin D1 mRNA by inhibiting the PI3K/AKT/mTOR/eIF4E-BP pathway [191]. The HDAC inhibitors valproic acid, MS-275 (a benzamide derivative), vorinostat, and panobinostat in combination with proteasome inhibitors (bortezomib, PR-171, NPI-0052, and others) have shown synergistic effects indicating the potential for further clinical studies [192, 193]. Similar effects have been achieved with a combination of HDAC inhibitors with other drug classes such as Bcl-2/BCL-XL antagonists [194], BCR signaling inhibitors [152], and mTOR inhibitors [195]. In a phase 2 clinical trial , PFS of 5.9 months was achieved with the HDAC inhibitor, vorinostat for relapse/refractory MCL (n = 9) however, vorinostat alone was not considered a promising agent [196]. In a phase 1 study with the HDAC inhibitor panobinostat and mTOR inhibitor everolimus a response was observed but at the stake of side effects especially significant thrombocytopenia [197] (Table 2).
PIM kinase inhibitors
PIM kinases (proviral integration site for Moloney murine leukemia virus), which are proto-oncogenes, induce phosphorylation of critical downstream effectors of ABL (Abelson), JAK2 (janus kinase 2), and Flt-3 (FMS related tyrosine kinase) genes, and promote growth and survival through cell cycle regulation along with inhibition of apoptosis of malignant cells [198]. Overexpression of PIM kinases PIM1 and PIM2 has been reported in MCL [199-200]. PIM kinase inhibitors that target c-Myc-driven transcription and cap-dependent translation in MCL cells have shown promise. Some of the PIM kinase inhibitors are SGI-1776, SMI4a, LGB321, AZD1897, SEL24-B58, and AZD1208; among these SGI-1776 has shown high antitumor activity both in vitro and in an in vivo model of MCL [201, 202]. A combination of SGI-1776 and bendamustine imparted superior effects in inducing cell death and reducing levels of DNA, RNA, and protein synthesis as well as increasing DNA damage of MCL cells [203].
BH3 mimetics/Bcl-2 family inhibitors
Bcl-2 is often overexpressed in MCL; hence, Bcl-2 is a potential target for MCL therapies. Several Bcl-2-selective small molecule BH3 mimetics function by binding to prosurvival members of Bcl-2 family and neutralizing their functional activity (sequestration of the proapoptotic Bcl-2 family members). This leads to release of pro-apoptotic family members from Bcl-2 and Bcl-xL, thereby inducing Bax- or Bak-dependent apoptosis [186]. Although many of these therapeutic agents have shown activities against MCL cells, their activity is determined by the activity/levels of Mcl-1, Bcl-1, and Bcl-xL [204]. ABT-737 and ABT-263 are capable of inducing rapid disruption of both Bcl-2/Bax and Bcl-2/Bik complexes in MCL cells and if these cells express the characteristic Bcl-2high/Mcl-1low profile. Interestingly, addition of flavopiridol to ABT-737 nullified the Mcl-1-associated resistance of MCL cells to ABT-737. The authors argued that Bcl-2/Mcl-1 tumor cell profiles would indicate the benefit of ABT-737 therapy and should be incorporated to guide its use [205]. In another study, excessive activation of NF-kB pathway caused MCL cells with high Mcl-1 and Bxl levels to become resistant to Bcl-2 selective BH3 mimetic ABT-199. Addition of ibrutinib to ABT-199 was able to overcome the resistance since ibrutinib causes release of MCL cells from their microenvironment into the periphery [206]. GX15-070/Obatoclax, another BH3 mimetic, used alone or in combination with Bortezomib induced apoptosis in MCL cell lines via NOXA-mediated activation of Bak [207]. In clinical trials, this combination demonstrated overall response rate of 31% for relapse/refractory MCL cases [208] (Table 2).
Novel antibodies and vaccines
Similar to rituximab, which targets CD20, other antibodies against CD antigens expressed by MCL cells have been tested. For example, a humanized anti-HLA-DR IgG4 mAb, IMMU-114 was reported to induce apoptosis in rituximab-resistant MCL cell lines via activation of ERK and JNK signaling [209]. Similarly, milatuzumab targets CD74, an integral membrane protein linked with promotion of B cell growth and survival. The combination of milatuzumab and rituximab induced rapid cell death, accompanied by generation of reactive oxygen species, loss of mitochondrial membrane potential, and potent inhibition of NF-κB pathway [210]. Otlertuzumab (TRU-016), an anti-CD37 protein, and polatuzumab vedotin, an antibody-drug conjugate containing an anti-CD79B monoclonal antibody conjugated to the microtubule-disrupting agent monomethyl auristatin E, showed acceptable safety and tolerability profiles on refractory/relapse MCL in a phase 1 study [211, 212]. Börschel et al., 2015 demonstrated that an antibody-cytokine fusion protein L19-IL2 that specifically delivers IL-2 to the tumor site by homing to the extra-domain B of fibronectin led to significant lymphoma growth retardation. The overall beneficial effect was potentiated by co-administration of rituximab [213]. Chen et al., 2015 have developed a novel vaccine based on targeting Cyclin D1 to Dendritic cells (DC) via the human DC surface receptor CD40 to elicit Cyclin D1-specific T cells, which is being tested in in vivo models [214]. Newer clinical trials have been designed to evaluate the clinical efficiency of some of these novel antibodies and vaccines (Table 3).
Conclusions and Future Directions
Over the last few decades, there has been growing improvement in the clinical outcome of mantle cell lymphoma. MCL is one of the most complex diseases, and involves aberrations in multiple signaling pathways, which lead to its poor prognosis. The contribution of the tumor microenvironment to the pathogenesis and outcome of MCL is equally complicated and, indeed, imparts modulatory effects on the outcome of this disease. The MCL International Prognostic Index (MIPI), which incorporates ECOG performance status, age, leukocyte count, and lactic dehydrogenase where inclusion of Ki67 proliferative index have been useful in stratification of high risk patients with probable poor prognosis. However, vigorous efforts are needed to identify target biomarkers which can be useful for predicting outcomes of MCL. It is important to personalize and individualize therapy for patients with poor prognosis. Combinations of traditional and novel therapeutic agents will be needed. Approaches will be needed that could minimize the recurrence/relapse rate for MCL and lead to improvement in the overall remission rate, PFS and OS. To date, in vitro studies have validated the beneficial effect of numerous drugs targeting various signaling pathways involved in MCL. In addition, phase 1 and phase 2 clinical trials with some of these drugs have been successful. Only a few have completed phase 3 trials; among these the combination of CDK inhibitors with agents targeting mTOR, BTK, and/or pan-isoform PI3K and mTOR inhibitors along with maintenance rituximab would appear to target MCL in a comprehensive manner. Although more clinical trials of various combinations of agents will definitely be needed in order to arrive at standardized therapies for untreated and refractory/relapse forms of MCL, the future looks promising.
Competing Interests
The authors declare that they have no competing interests.
Acknowledgments
We thank Newman’s Own Foundation, Elyssa and Jack Schecter Family Fund, John Theurer Cancer Center and Hackensack University Medical Center for supporting research at the Genomics and Biomarkers Program of HackensackUMC.
References
1. Fu K, Weisenburger DD, Greiner TC, Dave S, Wright G, Rosenwald A, Chiorazzi M, Iqbal J, Gesk S, Siebert R, De Jong D, Jaffe ES, Wilson WH, Delabie J, Ott G, Dave BJ, Sanger WG, et al. Lymphoma/Leukemia Molecular Profiling Project. Cyclin D1-negative mantle cell lymphoma: a clinicopathologic study based on gene expression profiling. Blood. 2005; 106: 4315-4321.
2. Xu W, Li JY. SOX11 expression in mantle cell lymphoma. Leuk Lymphoma. 2010; 51:1962-1967.
3. Jares P, Campo E. Advances in the understanding of mantle cell lymphoma. Br J Haematol. 2008; 142:149-165.
4. Fernàndez V, Salamero O, Espinet B, Solé F, Royo C, Navarro A, Camacho F, Beà S, Hartmann E, Amador V, Hernández L, Agostinelli C, Sargent RL, Rozman M, Aymerich M, Colomer D, Villamor N, et al. Genomic and gene expression profiling de fines indolent forms of mantle cell lymphoma. Cancer Res. 2010; 70: 1408-1418.
5. Ondrejka SL, Lai R, Smith SD, Hsi ED. Indolent mantle cell leukemia: a clinicopathological variant characterized by isolated lymphocytosis, interstitial bone marrow involvement, kappa light chain restriction, and good prognosis. Haematologica. 2011; 96:1121-1127.
6. Dreyling M, Ferrero S, Hermine O. How to manage mantle cell lymphoma Leukemia. 2014; 28: 2117-2130.
7. Gao J, Peterson L, Nelson B, Goolsby C, Chen YH. Immunophenotypic variations in mantle cell lymphoma. Am J Clin Pathol. 2009; 132:699-706.
8. Vose JM. Mantle cell lymphoma: 2013 Update on diagnosis, risk-stratification, and clinical management. Am J Hematol. 2013; 88:1082-1088.
9. Garcia DP, Rooney MT, Ahmad E, Davis BH. Diagnostic usefulness of CD23 and FMC-7 antigen expression patterns in B-cell lymphoma classification. Am J Clin Pathol. 2001;115:258-265.
10. Rahman K, Subramanian PG, Kadam PA, Gadage V, Galani K, Mittal N, Ghogale S, Badrinath Y, Ansari R, Kushte S, Nair R, Sengar M, Menon H, Gujral S. Morphological spectrum of leukemic mantle cell lymphoma. Indian J Pathol Microbiol. 2012; 55:66-71.
11. Arber DA, George TI. Bone marrow biopsy involvement by non-Hodgkin’s lymphoma: frequency of lymphoma types, patterns, blood involvement, and discordance with other sites in 450 specimens. Am J Surg Pathol. 2005; 29:1549-1557.
12. Hoeller S, Zhou Y, Kanagal-Shamanna R, Xu-Monette ZY, Hoehn D, Bihl M, Swerdlow SH, Rosenwald A, Ott G, Said J, Dunphy CH, Bueso-Ramos CE, Lin P, et al. Composite mantle cell lymphoma and chronic lymphocytic leukemia/small lymphocytic lymphoma: a clinicopathologic and molecular study. Hum Pathol. 2013; 44:110-121.
13. Liu Z, Dong HY, Gorczyca W, Tsang P, Cohen P, Stephenson CF, Berger CS, Wu CD, Weisberger J. CD5- mantle cell lymphoma. Am J Clin Pathol. 2002; 118:216-224.
14. Craig FE, Foon KA. Flow cytometric immunophenotyping for hematologic neoplasms. Blood. 2008; 111:3941-3967.
15. Perković S, Bašić-Kinda S, Aurer I, Ugrina I, Duletić-Načinović A, Lozić D, Batinić D. Multiparameter flow cytometry is necessary for detection, characterization and diagnostics of composite mature B-cell lymphoproliferative neoplasms. Int J Hematol. 2013; 98:589-596.
16. Jares P, Colomer D, Campo E. Genetic and molecular pathogenesis of mantle cell lymphoma: perspectives for new targeted therapeutics. Nat Rev Cancer, 2007; 7: 750-762.
17. Obrador-Hevia A, Fernández de Mattos S, Villalonga P, Rodríguez J. Molecular biology of mantle cell lymphoma: from profiling studies to new therapeutic strategies. Blood Rev. 2009; 23:205-216.
18. Juszczyński P, Chapuy B, Szczepaniak M, Warzocha K. Gene Expression Profiling in Hematologic Malignancies. Molecular Aspects of Hematologic Malignancies Principles and Practice. 2012; 199-214.
19. Soldini D, Campo E. New insights into the diagnosis of lymphomas. Annals of Oncology. 2012; 23: x83-x88.
20. Mullighan CG. Genome sequencing of lymphoid malignancies. Blood. 2013; 122:3899-3907.
21. Ortega-Paino E, Fransson J, Ek S, Borrebaeck CA. Functionally associated targets in mantle cell lymphoma as defined by DNA microarrays and RNA interference. Blood. 2008;111:1617-1624.
22. Beà S, Valdés-Mas R, Navarro A, Salaverria I, Martín-Garcia D, Jares P, Giné E, Pinyol M, Royo C, Nadeu F, Conde L, Juan M, Clot G, et al. Landscape of somatic mutations and clonal evolution in mantle cell lymphoma. Proc Natl Acad Sci U S A. 2013;110:18250-18255.
23. Braggio E, Egan JB, Fonseca R, Stewart AK. Lessons from next-generation sequencing analysis in hematological malignancies. Blood Cancer Journal. 2013; 3: e127.
24. Kridel R, Meissner B, Rogic S, Boyle M, Telenius A, Woolcock B, Gunawardana J, Jenkins C, Cochrane C, Ben-Neriah S, Tan K, Morin RD, Opat S, et al. Whole transcriptome sequencing reveals recurrent NOTCH1 mutations in mantle cell lymphoma. Blood. 2012; 119:1963-1971.
25. Ladetto M, Brüggemann M, Monitillo L, Ferrero S, Pepin F, Drandi D, Barbero D, Palumbo A, Passera R, Boccadoro M, Ritgen M, Gökbuget N, Zheng J, et al. Next-generation sequencing and real-time quantitative PCR for minimal residual disease detection in cell disorders. Leukemia. 2014; 28:1299-1307.
26. Weinkauf M, Christopeit M, Hiddemann W, Dreyling M. Proteome- and microarray-based expression analysis of lymphoma cell lines identifies a p53-centered cluster of differentially expressed proteins in mantle cell and follicular lymphoma. Electrophoresis. 2007;28:4416-4426.
27. Yang DT, Quann PJ, Petrich AM, Leith CP, Young KH, Kahl BS. Use of tissue microarray and automated quantitative analysis for screening and validation of potential biomarkers in mantle cell lymphoma. Appl Immunohistochem Mol Morphol. 2011; 19:62-69.
28. Lu H, Ouyang W, Huang C. Inflammation, a key event in cancer development. Mol Cancer Res 2006;4:221-233.
29. Diakos CI, Charles KA, McMillan DC, Clarke SJ. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014;15:e493-503.
30. Shain KH, Dalton WS, Tao J. The tumor microenvironment shapes hallmarks of mature B-cell malignancies. Oncogene. 2015; 34: 4673-4682.
31. Vazquez MI, Catalan-Dibene J, Zlotnik A. B cells responses and cytokine production are regulated by their immune microenvironment. Cytokine. 2015; 74:318-326.
32. Kim YR, Eom KS. Simultaneous Inhibition of CXCR4 and VLA-4 Exhibits Combinatorial Effect in Overcoming Stroma-Mediated Chemotherapy Resistance in Mantle Cell Lymphoma Cells. Immune Netw. 2014;14:296-306.
33. Rosich L, Montraveta A, Xargay-Torrent S, López-Guerra M, Roldán J, Aymerich M, Salaverria I, Beà S, Campo E, Pérez-Galán P, Roué G, Colomer D. Dual PI3K/mTOR inhibition is required to effectively impair microenvironment survival signals in mantle cell lymphoma. Oncotarget. 2014; 5:6788-6800. doi: 10.18632/oncotarget.225.
34. Zhang L, Yang J, Qian J, Li H, Romaguera JE, Kwak LW, Wang M, Yi Q. Role of the microenvironment in mantle cell lymphoma: IL-6 is an important survival factor for the tumor cells. Blood. 2012; 120:3783-3792.
35. Somovilla-Crespo B, Alfonso-Pérez M, Cuesta-Mateos C, Carballo-de Dios C, Beltrán AE, Terrón F, Pérez-Villar JJ, Gamallo-Amat C, Pérez-Chacón G, Fernández-Ruiz E, Zapata JM, Muñoz-Calleja C. Anti-CCR7 therapy exerts a potent anti-tumor activity in a xenograft model of human mantle cell lymphoma. J Hematol Oncol. 2013; 6:89.
36. Miranda RN, Briggs RC, Kinney MC, Veno PA, Hammer RD, Cousar JB. Immunohistochemical Detection of Cyclin D1 Using Optimized Conditions Is Highly Specific for Mantle Cell. Lymphoma and Hairy Cell Leukemia. Mod Pathol. 2000; 13:1308-1314.
37. Wiestner A, Tehrani M, Chiorazzi M, Wright G, Gibellini F, Nakayama K, Liu H, Rosenwald A, Muller-Hermelink HK, Ott G, Chan WC, Greiner TC, Weisenburger DD, et al. Point mutations and genomic deletions in CCND1 create stable truncated Cyclin D1 mRNAs that are associated with increased proliferation rate and shorter survival. Blood. 2007; 109:4599-4606.
38. Vasef MA, Medeiros LJ, Koo C, McCourty A, Brynes RK. Cyclin D1 immunohistochemical staining is useful in distinguishing mantle cell lymphoma from other low-grade B-cell neoplasms in bone marrow. Am J Clin Pathol. 1997; 108:302-307.
39. Bijwaard KE, Aguilera NS, Monczak Y, Trudel M, Taubenberger JK, Lichy JH. Quantitative Real-Time Reverse Transcription-PCR Assay for Cyclin D1 Expression: Utility in the Diagnosis of Mantle Cell Lymphoma. Clinical Chemistry. 2001; 47:195-201.
40. Pérez-Galán P, Mora-Jensen H, Weniger MA, Shaffer AL 3rd, Rizzatti EG, Chapman CM, Mo CC, Stennett LS, Rader C, Liu P, Raghavachari N, Stetler-Stevenson M, Yuan C, et al. Bortezomib resistance in mantle cell lymphoma is associated with plasmacytic differentiation. Blood. 2011; 117:542-552.
41. Lovec H, Grzeschiczek A, Kowalski MB, Möröy T. Cyclin D1/bcl-1 cooperates with myc genes in the generation of B-cell lymphoma in transgenic mice. EMBO J 1994;13:3487-3495
42. Gesk S, Klapper W, Martín-Subero JI, Nagel I, Harder L, Fu K, Bernd HW, Weisenburger DD, Parwaresch R, Siebert R. A chromosomal translocation in Cyclin D1-negative/cyclin D2-positive mantle cell lymphoma fuses the CCND2 gene to the IGK locus. Blood 2006; 108:1109-10.
43. Herens C, Lambert F, Quintanilla-Martinez L, Bisig B, Deusings C, de Leval L. Cyclin D1-negative mantle cell lymphoma with cryptic t(12;14) (p13;q32) and cyclin D2 overexpression. Blood. 2008; 111:1745-1746.
44. Evens AM, Kelemen K, Aranha O, Jovanovic B, Miyata S, Nelson BP, Goolsby C, Rosen ST, Winter JN, Peterson LC, Gordon LI. Survival of CD23-positive (CD23+) mantle cell lymphoma (MCL) compared to classic CD23-negative (CD23-) MCL: Biologic correlates and clinical outcomes in a 10-year analysis. Journal of Clinical Oncology, 2007; 25: 8039.
45. Zanetto U, Dong H, Huang Y, Zhang K, Narbaitz M, Sapia S, Kostopoulos I, Liu H, Du MQ, Bacon CM. Mantle cell lymphoma with aberrant expression of CD10. Histopathology. 2008; 53:20-29.
46. Maloney DG, Anti-CD20 Antibody Therapy for B-Cell Lymphomas. N Engl J Med. 2012; 366:2008-2016.
47. Nygren L, Baumgartner Wennerholm S, Klimkowska M, Christensson B, Kimby E, Sander B. Prognostic role of SOX11 in a population-based cohort of mantle cell lymphoma. Blood. 2012; 119:4215-4223.
48. Lu TX, Li JY, Xu W. The role of SOX11 in mantle cell lymphoma. Leuk Res. 2013; 37:1412-1419.
49. Cao X, Fan L, Fang C, Zhu DX, Dong HJ, Wang DM, Wang YH, Xu W, Li JY. The expression of SOX11, Cyclin D1, cyclin D2, and cyclin D3 in B-cell lymphocytic proliferative diseases. Med Oncol. 2012; 29:1190-1196.
50. Wang X, Björklund S, Wasik AM, Grandien A, Andersson P, Kimby E, Dahlman-Wright K, Zhao C, Christensson B, Sander B. Gene expression profiling and chromatin immunoprecipitation identify DBN1, SETMAR and HIG2 as direct targets of SOX11 in mantle cell lymphoma. PLoS One. 2010; 5:e14085.
51. Fernàndez V, Salamero O, Espinet B, Solé F, Royo C, Navarro A, Camacho F, Beà S, Hartmann E, Amador V, Hernández L, Agostinelli C, Sargent RL, Rozman M, et al. Genomic and gene expression profiling defines indolent forms of mantle cell lymphoma. Cancer Res. 2010; 70:1408-1418.
52. Simonsen AT, Sørensen CD, Ebbesen LH, Bødker JS, Bentzen HH, Nyvold CG. SOX11 as a minimal residual disease marker for Mantle cell lymphoma. Leuk Res. 2014; 38:918-924.
53. Thorsélius M, Walsh S, Eriksson I, Thunberg U, Johnson A, Backlin C, Enblad G, Sundström C, Roos G, Rosenquist R. Somatic hypermutation and V(H) gene usage in mantle cell lymphoma. Eur J Haematol. 2002; 68: 217-224.
54. Orchard J, Garand R, Davis Z, Babbage G, Sahota S, Matutes E, Catovsky D, Thomas PW, Avet-Loiseau H, Oscier D. A subset of t(11;14) lymphoma with mantle cell features displays mutated IgVH genes and includes patients with good prognosis, nonnodal disease. Blood. 2003; 101:4975-4981.
55. Simonsen A, Schou M, Sørensen C, Bentzen H, Nyvold C. SOX11, CCND1, BCL1/IgH, and IgH-VDJ - a Battle of Minimal Residual Disease Markers in Mantle Cell Lymphoma?. Leuk Lymphoma. 2015; 1-10.
56. Broyde A, Boycov O, Strenov Y, Okon E, Shpilberg O, Bairey O. Role and prognostic significance of the Ki-67 index in non-Hodgkin’s lymphoma. Am J Hematol. 2009; 84:338-343.
57. Niikura N, Iwamoto T, Masuda S, Kumaki N, Xiaoyan T, Shirane M, Mori K, Tsuda B, Okamura T, Saito Y, Suzuki Y, Tokuda Y. Immunohistochemical Ki67 labeling index has similar proliferation predictive power to various gene signatures in breast cancer. Cancer Sci. 2012; 103:1508-1512.
58. He X, Chen Z, Fu T, Jin X, Yu T, Liang Y, Zhao X, Huang L. Ki-67 is a valuable prognostic predictor of lymphoma but its utility varies in lymphoma subtypes: evidence from a systematic meta-analysis. BMC Cancer, 2014; 14, 153.
59. Determann O, Hoster E, Ott G, Wolfram Bernd H, Loddenkemper C, Leo Hansmann M, Barth TE, Unterhalt M, Hiddemann W, Dreyling M, Klapper W; European Mantle Cell Lymphoma Network and the German Low Grade Lymphoma Study Group. Ki-67 predicts outcome in advanced-stage mantle cell lymphoma patients treated with anti-CD20 immunochemotherapy: results from randomized trials of the European MCL Network and the German Low Grade Lymphoma Study Group. Blood. 2008; 111:2385-2387.
60. Geisler CH, Kolstad A, Laurell A, Räty R, Jerkeman M, Eriksson M, Nordström M, Kimby E, Boesen AM, Nilsson-Ehle H, Kuittinen O, Lauritzsen GF, Ralfkiaer E, et al. The Mantle Cell Lymphoma International Prognostic Index (MIPI) is superior to the International Prognostic Index (IPI) in predicting survival following intensive first-line immunochemotherapy and autologous stem cell transplantation (ASCT). Blood. 2010; 115: 1530-1533.
61. Halldórsdóttir AM, Lundin A, Murray F, Mansouri L, Knuutila S, Sundström C, Laurell A, Ehrencrona H, Sander B, Rosenquist R. Impact of TP53 mutation and 17p deletion in mantle cell lymphoma. Leukemia. 2011; 25, 1904-1908.
62. Nordström L, Sernbo S, Eden P, Grønbaek K, Kolstad A, Räty R, Karjalainen ML, Geisler C, Ralfkiaer E, Sundström C, Laurell A, Delabie J, Ehinger M, Jerkeman M, Ek S. SOX11 and TP53 add prognostic information to MIPI in a homogenously treated cohort of mantle cell lymphoma—a Nordic Lymphoma Group study. Br J Haematol. 2014; 166:98-108.
63. Schaffner C, Idler I, Stilgenbauer S, Döhner H, Lichter P. Mantle cell lymphoma is characterized by inactivation of the ATM gene . Proc Natl Acad Sci U S A. 2000; 97:2773-2778.
64. Miller DM, Thomas SD, Islam A, Muench D, Sedoris K. c-Myc and Cancer Metabolism. Clin Cancer Res 2012; 18; 5546-5553.
65. Ott G, Rosenwald A, Campo E. Understanding MYC-driven aggressive B-cell lymphomas: pathogenesis and classification. Blood. 2013;122:3884-3891.
66. Teshima K, Nara M, Watanabe A, Ito M, Ikeda S, Hatano Y, Oshima K, Seto M, Sawada K, Tagawa H. Dysregulation of BMI1 and microRNA-16 collaborate to enhance an anti-apoptotic potential in the side population of refractory mantle cell lymphoma. Oncogene. 2014; 33:2191-203.
67. Teo AE, Chen Z, Miranda RN, McDonnell T, Medeiros LJ, McCarty N. Differential PAX5 levels promote malignant B cell, infiltration, progression and drug resistance and, predict a poor prognosis in MCL patients independent, of CCND1. Leukemia. 2015; 30:580-93.
68. Su H, Ma X, Huang Y, Han H, Zou Y, Huang W. JARID1B deletion induced apoptosis in Jeko-1 and HL-60 cell lines. Int J Clin Exp Pathol. 2015; 8:171-183.
69. Sonbol MB, Maurer MJ, Stenson MJ, Allmer C, LaPlant BR, Weiner GJ, Macon WR, Cerhan JR, Witzig TE, Gupta M. Elevated soluble IL-2Rα, IL-8, and MIP-1β levels are associated with inferior outcome and are independent of MIPI score in patients with mantle cell lymphoma. Am J Hematol. 2014; 89:E223-227.
70. Isaza-Correa JM, Liang Z, van den Berg A, Diepstra A, Visser L. Toll-like receptors in the pathogenesis of human B cell malignancies J Hematol Oncol. 2014; 7: 57.
71. Husby S, Geisler C, Grønbæk K. MicroRNAs in mantle cell lymphoma. Leuk Lymphoma. 2013; 54:1867-1875.
72. Goswami RS, Atenafu EG, Xuan Y, Waldron L, Reis PP, Sun T, Datti A, Xu W, Kuruvilla J, Good DJ, Lai R, Church AJ, Lam WS, Baetz T, Lebrun DP, Sehn LH, Farinha P, et al. MicroRNA signature obtained from the comparison of aggressive with indolent non-Hodgkin lymphomas: potential prognostic value in mantle-cell lymphoma. J Clin Oncol. 2013; 31:2903-2911.
73. Husby S, Ralfkiaer U, Garde C, Zandi R, Ek S, Kolstad A, Jerkeman M, Laurell A, Räty R, Pedersen LB, Pedersen A, Ehinger M, Sundström C, et al. miR-18b overexpression identifies mantle cell lymphoma patients with poor outcome and improves the MIPI-B prognosticator. Blood. 2015; 125:2669-2677.
74. Jares P, Colomer D, Campo E. Molecular pathogenesis of mantle cell lymphoma. J Clin Invest. 2012;122:3416-3423.
75. Karam M, Ata A, Irish K, Feustel PJ, Mottaghy FM, Stroobants SG, Verhoef GE, Chundru S, Douglas-Nikitin V, Oliver Wong CY, Brepoels LM. FDG positron emission tomography/computed tomography scan may identify mantle cell lymphoma patients with unusually favorable outcome. Nucl Med Commun. 2009; 30: 770-778.
76. Hoster E, Dreyling M, Klapper W, Gisselbrecht C, van Hoof A, Kluin-Nelemans HC, Pfreundschuh M, Reiser M, Metzner B, Einsele H, Peter N, Jung W, Wörmann B, et al. A new prognostic index (MIPI) for patients with advanced-stage mantle cell lymphoma. Blood. 2008; 111:558-65.
77. Todorovic M, Balint B, Andjelic B, Stanisavljevic D, Kurtovic NK, Radisavljevic Z, Mihaljevic B. Outcome prediction of advanced mantle cell lymphoma by international prognostic index versus different mantle cell lymphoma indexes: one institution study. Med Oncol. 2012; 29:2212-2219.
78. Hoster E, Klapper W, Hermine O, Kluin-Nelemans HC, Walewski J, van Hoof A, Trneny M, Geisler CH, Di Raimondo F, Szymczyk M, Stilgenbauer S, Thieblemont C, Hallek M, et al. Confirmation of the mantle-cell lymphoma International Prognostic Index in randomized trials of the European Mantle-Cell Lymphoma Network. J Clin Oncol. 2014; 32(13):1338-46.
79. Ott G, Rosenwald A, Campo E. Understanding MYC-driven aggressive B-cell lymphomas: pathogenesis and classification. Hematology Am Soc Hematol Educ Program. 2013;2013:575-83.
80. Slotta-Huspenina J, Koch I, de Leval L, Keller G, Klier M, Bink K, Kremer M, Raffeld M, Fend F, Quintanilla-Martinez L. The impact of Cyclin D1 mRNA isoforms, morphology and p53 in mantle cell lymphoma: p53 alterations and blastoid morphology are strong predictors of a high proliferation index. Haematologica 2012; 97: 1422-1430.
81. Meggendorfer M, Kern W, Haferlach C, Haferlach T, Schnittger S.SOX11 overexpression is a specific marker for mantle cell lymphoma and correlates with t(11;14) translocation, CCND1 expression and an adverse prognosis. Leukemia. 2013; 27:2388-2391.
82. Witzig TE, Maurer MJ, Habermann TM, Link BK, Micallef IN, Nowakowski GS, Ansell SM, Colgan JP, Inwards DJ, Porrata LF, Markovic SN, Johnston PB, et al. Elevated monoclonal and polyclonal serum immunoglobulin free light chain as prognostic factors in B- and T-cell non-Hodgkin lymphoma. Am J Hematol. 2014; 89:1116-1120.
83. Yoo C, Yoon DH, Kim S, Huh J, Park CS, Park CJ, Lee SW, Suh C. Serum beta-2 microglobulin as a prognostic biomarker in patients with mantle cell lymphoma. Hematol Oncol. 2015; doi: 10.1002/hon.2188.
84. Choe JY, Yun JY, Na HY, Huh J, Shin SJ, Kim HJ, Paik JH, Kim YA, Nam SJ, Jeon YK, Park G, Kim JE. Myc overexpression correlates with MYC amplification or translocation and is associated with poor prognosis in mantle cell lymphoma. Histopathology. 2015; doi: 10.1111/his.12760.
85. Shin SJ, Roh J, Cha HJ, Choi YD, Kim JM, Min SK, Kim JE, Eom DW, Lee H, Kim HJ, Yoon DH, Suh C, Huh J. TCL1 expression predicts overall survival in patients with mantle cell lymphoma. Eur J Haematol. 2015; doi: 10.1111/ejh.12539.
86. Nygren L, Wasik AM, Baumgartner-Wennerholm S, Jeppsson-Ahlberg Å, Klimkowska M, Andersson P, Buhrkuhl D, Christensson B, Kimby E, Wahlin BE, Sander B. T-cell levels are prognostic in mantle cell lymphoma. Clin Cancer Res. 2014; 20: 6096-7104.
87. Bernard M, Tsang RW, Le LW, Hodgson DC, Sun A, Wells W, Kukreti V, Kuruvilla J, Crump M, Gospodarowicz MK. Limited-stage mantle cell lymphoma: treatment outcomes at the Princess Margaret Hospital. Leuk Lymphoma. 2013; 54:261-267.
88. Guru Murthy GS, Venkitachalam R, Mehta P. Effect of radiotherapy on the survival of patients with stage I and stage II mantle cell lymphoma: analysis of the Surveillance, Epidemiology and End Results database. Clin Lymphoma Myeloma Leuk. 2014; 14: S90-95.
89. Engelhard M, Unterhalt M, Hansmann M, Stuschke M, Sack H. Follicular lymphoma, immunocytoma, and mantle cell lymphoma: randomized evaluation of curative radiotherapy in limited stage nodal disease. Ann Oncol 2008; 19 (suppl. 4): 418.
90. Cheah CY, George A, Giné E, Chiappella A, Kluin-Nelemans HC, Jurczak W, Krawczyk K, Mocikova H, Klener P, Salek D, Walewski J, Szymczyk M, Smolej L, et al. Central nervous system involvement in mantle cell lymphoma: clinical features, prognostic factors and outcomes from the European Mantle Cell Lymphoma Network. Ann Oncol. 2013;24:2119-2123.
91. Haque W, Voong KR, Shihadeh F, Arzu I, Pinnix C, Mazloom A, Medeiros LJ, Romaguera J, Rodriguez A, Wang M, Allen P, Dabaja B. Radiation therapy is an effective modality in the treatment of mantle cell lymphoma, even in heavily pretreated patients. Clin Lymphoma Myeloma Leuk. 2014; 14:474-479.
92. Neville KE, Bisquera A, Capp AL. Involved-field radiotherapy for patients with mantle cell lymphoma. J Med Imaging Radiat Oncol. 2015; 59(5):631-639.
93. Bernstein SH, Epner E, Unger JM, LeBlanc M, Cebula E, Burack R, Rimsza L, Miller TP, Fisher RI. A phase II multicenter trial of hyperCVAD MTX/Ara-C and rituximab in patients with previously untreated mantle cell lymphoma; SWOG 0213. Ann Oncol. 2013; 24: 1587-1593.
94. Flinn IW, van der Jagt R, Kahl BS, Wood P, Hawkins TE, Macdonald D, Hertzberg M, Kwan YL, Simpson D, Craig M, Kolibaba K, Issa S, Clementi R, et al. Randomized trial of bendamustine-rituximab or R-CHOP/R-CVP in first-line treatment of indolent NHL or MCL: the BRIGHT study. Blood. 2014;123:2944-2952.
95. Saito H, Maruyama D, Maeshima AM, Makita S, Kitahara H, Miyamoto K, Fukuhara S, Munakata W, Suzuki T, Kobayashi Y, Taniguchi H, Tobinai K. Prolonged lymphocytopenia after bendamustine therapy in patients with relapsed or refractory indolent B-cell and mantle cell lymphoma. Blood Cancer J. 2015; 5:e362.
96. Maddocks K, Christian B, Jaglowski S, Flynn J, Jones JA, Porcu P, Wei L, Jenkins C, Lozanski G, Byrd JC, Blum KA. A phase 1/1b study of rituximab, bendamustine, and ibrutinib in patients with untreated and relapsed/refractory non-Hodgkin lymphoma. Blood. 2015; 125:242-248.
97. McKay P, Leach M, Jackson R, Cook G, Rule S; British Committee for Standards in Haematology. Guidelines for the investigation and management of mantle cell lymphoma. Br J Haematol. 2012; 159:405-426.
98. Rajguru S, Kahl BS. Emerging therapy for the treatment of mantle cell lymphoma. J Natl Compr Canc Netw. 2014; 12:1311-1318.
99. LaCasce AS, Vandergrift JL, Rodriguez MA, Abel GA, Crosby AL, Czuczman MS, Nademanee AP, Blayney DW, Gordon LI, Millenson M, Vanderplas A, Lepisto EM, Zelenetz AD, et al. Comparative outcome of initial therapy for younger patients with mantle cell lymphoma: an analysis from the NCCN NHL Database. Blood. 2012; 119:2093-2099.
100. Hermine O, Hoster E, Walewski J, Ribrag V, Brousse N, Thieblemont C, Bouabdallah R, Stilgenbauer S, Feugier P, Forstpointner P, Haioun C, Kneba M, Hänel M, et al. Alternating courses of 3x CHOP and 3x DHAP plus rituximab followed by a high dose ARA-C containing myeloablative regimen and autologous stem cell transplantation (ASCT) is superior to 6 courses CHOP plus rituximab followed by myeloablative radiochemotherapy and ASCT in mantle cell lymphoma: results of the MCL younger trial of the European Mantle Cell Lymphoma Network (MCL net). ASH Annu Meeting Abstr. 2010; 116:110.
101. Robak T, Huang H, Jin J, Zhu J, Liu T, Samoilova O, Pylypenko H, Verhoef G, Siritanaratkul N, Osmanov E, Alexeeva J, Pereira J, Drach J, et al. Bortezomib-Based Therapy for Newly Diagnosed Mantle-Cell Lymphoma. N Engl J Med. 2015; 372:944-953.
102. Doorduijn JK, Kluin-Nelemans HC. Management of mantle cell lymphoma in the elderly patient. Clin Interv Aging. 2013; 8:1229-1236.
103. Cavalli F, Rooney B, Pei L, Van De Velde H, Robak T and on behalf of the LYM-3002 Investigators. Randomized phase 3 study of rituximab, cyclophosphamide, doxorubicin, and prednisone plus vincristine (R-CHOP) or bortezomib (VR-CAP) in newly diagnosed mantle cell lymphoma (MCL) patients (pts) ineligible for bone marrow transplantation (BMT). Journal of Clinical Oncology, 2014; 32: S8500.
104. Romaguera JE, Fayad LE, Feng L, Hartig K, Weaver P, Rodriguez MA, Hagemeister FB, Pro B, McLaughlin P, Younes A, Samaniego F, Goy A, Cabanillas F, et al. Ten-year follow-up after intense chemoimmunotherapy with Rituximab-HyperCVAD alternating with Rituximab-high dose methotrexate/cytarabine (R-MA) and without stem cell transplantation in patients with untreated aggressive mantle cell lymphoma. Br J Haematol. 2010; 150:200-208.
105. Tam CS, Bassett R, Ledesma C, Korbling M, Alousi A, Hosing C, Kebraei P, Harrell R, Rondon G, Giralt SA, Anderlini P, Popat U, Pro B, et al. Mature results of the M. D. Anderson Cancer Center risk-adapted transplantation strategy in mantle cell lymphoma. Blood, 2009; 113: 4144-4152.
106. Geisler CH, Kolstad A, Laurell A, Jerkeman M, Räty R, Andersen NS, Pedersen LB, Eriksson M, Nordström M, Kimby E, Bentzen H, Kuittinen O, Lauritzsen GF, et al. Nordic MCL2 trial update: Six-year follow-up after intensive immunochemotherapy for untreated mantle cell lymphoma followed by BEAM or BEAC + autologous stem-cell support: Still very long survival but late relapses do occur. Br J Haematol 2012; 158:355-362.
107. Fenske TS, Zhang MJ, Carreras J, Ayala E, Burns LJ, Cashen A, Costa LJ, Freytes CO, Gale RP, Hamadani M, Holmberg LA, Inwards DJ, Lazarus HM, et al. Autologous or reduced-intensity conditioning allogeneic hematopoietic cell transplantation for chemotherapy-sensitive mantle-cell lymphoma: analysis of transplantation timing and modality. J Clin Oncol. 2014; 32:273-281.
108. Ritchie DS, Seymour JF, Grigg AP, Roberts AW, Hoyt R, Thompson S, Szer J, Prince HM. The hyper-CVAD-rituximab chemotherapy programme followed by high-dose busulfan, melphalan and autologous stem cell transplantation produces excellent event-free survival in patients with previously untreated mantle cell lymphoma. Ann Hematol. 2007; 86:101-105.
109. Nastoupil LJ, Shenoy PJ, Ambinder A, Koff JL, Nooka AK, Waller EK, Langston A, Seward M, Kaufman JL, Bernal-Mizrachi L, King N, Lechowicz MJ, Lonial S, Sinha R, Flowers CR. Intensive chemotherapy and consolidation with high dose therapy and autologous stem cell transplant in patients with mantle cell lymphoma. Leuk Lymphoma. 2015;56:383-389.
110. Smith MR, Li H, Gordon L, Gascoyne RD, Paietta E, Forero-Torres A, Kahl BS, Advani R, Hong F, Horning SJ. Phase II study of rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone immunochemotherapy followed by yttrium-90-ibritumomab tiuxetan in untreated mantle-cell lymphoma: Eastern Cooperative Oncology Group Study E1499. J Clin Oncol. 2012;30 :3119-3126.
111. Griffin MM, Morley N. Rituximab in the treatment of non-Hodgkin’s lymphoma - a critical evaluation of randomized controlled trials. Expert Opinion on Biological Therapy. 2013;13: 803-811.
112. Lenz G, Dreyling M, Hoster E, Wörmann B, Dührsen U, Metzner B, Eimermacher H, Neubauer A, Wandt H, Steinhauer H, Martin S, Heidemann E, Aldaoud A, et al. Immunochemotherapy with rituximab and cyclophosphamide, doxorubicin, vincristine, and prednisone significantly improves response and time to treatment failure, but not long-term outcome in patients with previously untreated mantle cell lymphoma: results of a prospective randomized trial of the German Low Grade Lymphoma Study Group (GLSG). J Clin Oncol. 2005;23:1984-1992.
113. Gunnellini M, Falchi L. Therapeutic Activity of Lenalidomide in Mantle Cell Lymphoma and Indolent Non-Hodgkin’s Lymphomas. Adv Hematol. 2012;2012:523842.
114. Le Gouill S, Callanan M, Macintyre E, Delfau-Larue MH, Bodet-Milin C, Meignan M, Moreau A, Travers-Glehen A, Béné MC, Haouin C, Gressin R, Casasnovas RO, Ribrag V, et al. Clinical, metabolic and molecular responses after 4 courses of R-DHAP and after autologous stem cell transplantation for untreated mantle cell lymphoma patients included in the LyMa trial, a Lysa study. Blood 2012;120:152. ASH Annual Meeting Abstracts.
115. Rossi D, Ferrero S, Bruscaggin A, Ghione P, Di Rocco A, Spina V, Stefoni V, Ciccone G, Barbero D, Monitillo L, Da Silva MG, Santoro A, Molinari A, et al. A Molecular Model for the Prediction of Progression Free Survival in Young Mantle Cell Lymphoma Patients Treated with Cytarabine-Based High Dose Sequential Chemotherapy and Autologous Stem Cell Transplantation: Results from the MCL0208 Phase III Trial from Fondazione Italiana Linfomi (FIL). ASH 57th ASH Annual Meeting: https://ash.confex.com/ash/2015/webprogram/Paper81120.html.
116. Visco C, Finotto S, Zambello R, Paolini R, Menin A, Zanotti R, Zaja F, Semenzato G, Pizzolo G, D’Amore ES, Rodeghiero F. Combination of rituximab, bendamustine, and cytarabine for patients with mantle-cell non-Hodgkin lymphoma ineligible for intensive regimens or autologous transplantation. J Clin Oncol. 2013; 31:1442-1449.
117. Räty R, Honkanen T, Jantunen E, Jyrkkiö S, Karjalainen-Lindsberg ML, Kuittinen O, Lehto M, Mikkola M, Poikonen E, Rauhala A, Rimpiläinen J, Räsänen A, Siitonen S, et al. Prolonged immunochemotherapy with rituximab, cytarabine and fludarabine added to cyclophosphamide, doxorubicin, vincristine and prednisolone and followed by rituximab maintenance in untreated elderly patients with mantle cell lymphoma: a prospective study by the Finnish Lymphoma Group. Leuk Lymphoma. 2012 ;53 :1920-1928.
118. Forstpointner R, Unterhalt M, Dreyling M, Böck HP, Repp R, Wandt H, Pott C, Seymour JF, Metzner B, Hänel A, Lehmann T, Hartmann F, Einsele H, Hiddemann W; German Low Grade Lymphoma Study Group (GLSG). Maintenance therapy with rituximab leads to a significant prolongation of response duration after salvage therapy with a combination of rituximab, fludarabine, cyclophosphamide, and mitoxantrone (R-FCM) in patients with recurring and refractory follicular and mantle cell lymphomas: results of a prospective randomized study of the German Low Grade Lymphoma Study Group (GLSG). Blood. 2006; 108: 4003-4008.
119. Rodríguez J, Gutierrez A, Palacios A, Navarrete M, Blancas I, Alarcón J, Caballero MD, De Mattos SF, Gines J, Martínez J, Lopez A. Rituximab, gemcitabine and oxaliplatin: an effective regimen in patients with refractory and relapsing mantle cell lymphoma. Leuk Lymphoma. 2007; 48:2172-2178.
120. Witzig TE, Geyer SM, Kurtin PJ, Colgan JP, Inwards DJ, Micallef IN, LaPlant BR, Michalak JC, Salim M, Dalton RJ, Moore DF Jr, Reeder CB, North Central Cancer Treatment Group. Salvage chemotherapy with rituximab DHAP for relapsed non-Hodgkin lymphoma: a phase II trial in the North Central Cancer Treatment Group. Leuk Lymphoma. 2008; 49:1074-1080.
121. Robinson KS, Williams ME, van der Jagt RH, Cohen P, Herst JA, Tulpule A, Schwartzberg LS, Lemieux B, Cheson BD. Phase II multicenter study of bendamustine plus rituximab in patients with relapsed indolent B-cell and mantle cell non-Hodgkin’s lymphoma. J Clin Oncol. 2008; 26:4473-4479.
122. Fisher RI, Bernstein SH, Kahl BS, Djulbegovic B, Robertson MJ, de Vos S, Epner E, Krishnan A, Leonard JP, Lonial S, Stadtmauer EA, O’Connor OA, Shi H, Boral AL, Goy A. Multicenter phase II study of bortezomib in patients with relapsed or refractory mantle cell lymphoma. J Clin Oncol. 2006; 24:4867-4874.
123. Hess G, Herbrecht R, Romaguera J, Verhoef G, Crump M, Gisselbrecht C, Laurell A, Offner F, Strahs A, Berkenblit A, Hanushevsky O, Clancy J, Hewes B, Moore L, Coiffier B. Phase III study to evaluate temsirolimus compared with investigator’s choice therapy for the treatment of relapsed or refractory mantle cell lymphoma. J Clin Oncol. 2009; 27:3822-3829.
124. Goy A, Sinha R, Williams ME, Kalayoglu Besisik S, Drach J, Ramchandren R, Zhang L, Cicero S, Fu T, Witzig TE. Single-agent lenalidomide in patients with mantle-cell lymphoma who relapsed or progressed after or were refractory to bortezomib: phase II MCL-001 (EMERGE) study. J Clin Oncol 2013; 31: 3688-3695.
125. Wang ML, Rule S, Martin P, Goy A, Auer R, Kahl BS, Jurczak W, Advani RH, Romaguera JE, Williams ME, Barrientos JC, Chmielowska E, Radford J, et al. Targeting BTK with ibrutinib in relapsed or refractory mantle-cell lymphoma. N Engl J Med. 2013; 369:507-516.
126. Friedberg JW, Vose JM, Kelly JL, Young F, Bernstein SH, Peterson D, Rich L, Blumel S, Proia NK, Liesveld J, Fisher RI, Armitage JO, Grant S, Leonard JP. The combination of bendamustine, bortezomib, and rituximab for patients with relapsed/refractory indolent and mantle cell non-Hodgkin lymphoma. Blood. 2011; 117:2807-2812.
127. Kouroukis CT, Fernandez LA, Crump M, Gascoyne RD, Chua NS, Buckstein R, Turner R, Assouline S, Klasa RJ, Walsh W, Powers J, Eisenhauer E. A phase II study of bortezomib and gemcitabine in relapsed mantle cell lymphoma from the National Cancer Institute of Canada Clinical Trials Group (IND 172). Leuk Lymphoma, 2011; 52: 394-399.
128. William BM, Allen MS, Loberiza FR Jr, Bociek RG, Bierman PJ, Armitage JO, Vose JM. Phase I/II study of bortezomib-BEAM and autologous hematopoietic stem cell transplantation for relapsed indolent non-Hodgkin lymphoma, transformed, or mantle cell lymphoma. Biol Blood Marrow Transplant. 2014; 20:536-542.
129. Holkova B, Kmieciak M, Bose P, Yazbeck VY, Barr PM, Tombes MB, Shrader E, Weir-Wiggins C, Rollins AD, Cebula EM, Pierce E, Herr M, Sankala H, et al. Phase 1 trial of carfilzomib (PR-171) in combination with vorinostat (SAHA) in patients with relapsed or refractory B-cell lymphomas. Leuk Lymphoma. 2015;1-9.
130. Zinzani PL, Vose JM, Czuczman MS, Reeder CB, Haioun C, Polikoff J, Tilly H, Zhang L, Prandi K, Li J, Witzig TE. Long-term follow-up of lenalidomide in relapsed/refractory mantle cell lymphoma: subset analysis of the NHL-003 study. Ann Oncol. 2013; 24:2892-2897.
131. Gopal AK, Rajendran JG, Petersdorf SH, Maloney DG, Eary JF, Wood BL, Gooley TA, Bush SA, Durack LD, Martin PJ, Matthews DC, Appelbaum FR, Bernstein ID, Press OW. High-dose chemo-radioimmunotherapy with autologous stem cell support for relapsed mantle cell lymphoma. Blood. 2002; 99:3158-3162.
132. Weide R, Hess G, Köppler H, Heymanns J, Thomalla J, Aldaoud A, Losem C, Schmitz S, Haak U, Huber C, Unterhalt M, Hiddemann W, Dreyling M; German Low Grade Lymphoma Study Group. High anti-lymphoma activity of bendamustine/mitoxantrone/rituximab in rituximab pretreated relapsed or refractory indolent lymphomas and mantle cell lymphomas. A multicenter phase II study of the German Low Grade Lymphoma Study Group (GLSG). Leuk Lymphoma. 2007;48:1299-1306.
133. Marzec M, Kasprzycka M, Lai R, Gladden AB, Wlodarski P, Tomczak E, Nowell P, Deprimo SE, Sadis S, Eck S, Schuster SJ, Diehl JA, Wasik MA. Mantle cell lymphoma cells express predominantly Cyclin D1a isoform and are highly sensitive to selective inhibition of CDK4 kinase activity. Blood. 2006; 108:1744-1750.
134. Leonard JP, LaCasce AS, Smith MR, Noy A, Chirieac LR, Rodig SJ, Yu JQ, Vallabhajosula S, Schoder H, English P, Neuberg DS, Martin P, Millenson MM, et al. Selective CDK4/6 inhibition with tumor responses by PD0332991 in patients with mantle cell lymphoma. Blood. 2012;119:4597-4607.
135. Venkataraman G, Maududi T, Ozpuyan F, Bahar HI, Izban KF, Qin JZ, Alkan S. Induction of apoptosis and down regulation of cell cycle proteins in mantle cell lymphoma by flavopiridol treatment. Leuk Res. 2006;30:1377-1384.
136. Lin TS, Blum KA, Fischer DB, Mitchell SM, Ruppert AS, Porcu P, Kraut EH, Baiocchi RA, Moran ME, Johnson AJ, Schaaf LJ, Grever MR, Byrd JC. Flavopiridol, fludarabine, and rituximab in mantle cell lymphoma and indolent B-cell lymphoproliferative disorders. J Clin Oncol. 2010; 28:418-423.
137. Lin TS, Blum KA, Fischer DB, Mitchell SM, Ruppert AS, Porcu P, Kraut EH, Baiocchi RA, Moran ME, Johnson AJ, Schaaf LJ, Grever MR, Byrd JC. Flavopiridol, fludarabine, and rituximab in mantle cell lymphoma and indolent B-cell lymphoproliferative disorders. J Clin Oncol. 2010; 28:418-423.
138. Holkova B, Kmieciak M, Perkins EB, Bose P, Baz RC, Roodman GD, Stuart RK, Ramakrishnan V, Wan W, Peer CJ, Dawson J, Kang L, Honeycutt C, et al. Phase I trial of bortezomib (PS-341; NSC 681239) and “nonhybrid” (bolus) infusion schedule of alvocidib (flavopiridol; NSC 649890) in patients with recurrent or refractory indolent B-cell neoplasms. Clin Cancer Res. 2014; 20:5652-5662.
139. Chen R, Chubb S, Cheng T, Hawtin RE, Gandhi V, Plunkett W. Responses in mantle cell lymphoma cells to SNS-032 depend on the biological context of each cell line. Cancer Res. 2010; 70:6587-6597.
140. Lacrima K, Valentini A, Lambertini C, Taborelli M, Rinaldi A, Zucca E, Catapano C, Cavalli F, Gianella-Borradori A, MacCallum DE, Bertoni F. In vitro activity of cyclin-dependent kinase inhibitor CYC202 (Seliciclib, R-roscovitine) in mantle cell lymphomas. Annals of Oncology, 2005; 16: 1169-1176.
141. Shirsath NP, Manohar SM, Joshi KS. P276-00, a cyclin-dependent kinase inhibitor, modulates cell cycle and induces apoptosis in vitro and in vivo in mantle cell lymphoma cell lines. Mol Cancer. 2012; 11:77.
142. Gorlick R, Kolb EA, Houghton PJ, Morton CL, Neale G, Keir ST, Carol H, Lock R, Phelps D, Kang MH, Reynolds CP, Maris JM, Billups C, Smith MA. Initial testing (stage 1) of the cyclin dependent kinase inhibitor SCH 727965 (dinaciclib) by the pediatric preclinical testing program. Pediatr Blood Cancer. 2012; 59:1266-1274.
143. Alinari L, Prince CJ, Edwards RB, Towns WH, Mani R, Lehman A, Zhang X, Jarjoura D, Pan L, Kinghorn AD, Grever MR, Baiocchi RA, Lucas DM. Dual targeting of the cyclin/Rb/E2F and mitochondrial pathways in mantle cell lymphoma with the translation inhibitor silvestrol. Clin Cancer Res. 2012; 18:4600-4611.
144. Chiron D, Di Liberto M, Martin P, Huang X, Sharman J, Blecua P, Mathew S, Vijay P, Eng K, Ali S, Johnson A, Chang B, Ely S, Elemento O, Mason CE, Leonard JP, Chen-Kiang S. Cell-cycle reprogramming for PI3K inhibition overrides a relapse-specific C481S BTK mutation revealed by longitudinal functional genomics in mantle cell lymphoma. Cancer Discov. 2014; 4:1022-1035.
145. Chen R, Frankel P, Popplewell L, Siddiqi T, Ruel N, Rotter A, Thomas SH, Mott M, Nathwani N, Htut M, Nademanee A, Forman SJ, Kirschbaum M. A phase II study of vorinostat and rituximab for treatment of newly diagnosed and relapsed/refractory indolent non-Hodgkin lymphoma. Haematologica. 2015;100:357-362.
146. Burger JA. Bruton’s Tyrosine Kinase (BTK) Inhibitors in Clinical Trials. Curr Hematol Malig Rep 2014; 9:44-49.
147. Fowler N, Davis E. Targeting B-cell receptor signaling: changing the paradigm. Hematology Am Soc Hematol Educ Program. 2013; 2013:553-60.
148. Bernard S, Danglade D, Gardano L, Laguillier C, Lazarian G, Roger C, Thieblemont C, Marzec J, Gribben J, Cymbalista F, Varin-Blank N, Ledoux D, Baran-Marszak F. Inhibitors of BCR signalling interrupt the survival signal mediated by the micro-environment in mantle cell lymphoma. Int J Cancer. 2015; 136:2761-2774.
149. Boukhiar MA, Roger C, Tran J, Gressin R, Martin A, Ajchenbaum-Cymbalista F, Varin-Blank N, Ledoux D, Baran-Marszak F. Targeting early B-cell receptor signaling induces apoptosis in leukemic mantle cell lymphoma. Exp Hematol Oncol. 2013; 2:4.
150. Hendriks RW, Yuvaraj S, Kil LP. Targeting Bruton’s tyrosine kinase in B cell malignancies. Nat Rev Cancer. 2014; 14:219-232.
151. Ou Z, Pham L, Vang M, Zhang L, Wang J, Strickland L, Lopez ER, Dominici EM, Rollo A, Tamayo AT, Ford RJ, Wang M. Targeting BCR activated STAT3 by ibrutinib in mantle cell lymphoma. Blood. 2013; 122: 3079 - 3079.
152. Hagiwara K, Kunishima S, Iida H, Miyata Y, Naoe T, Nagai H. The synergistic effect of BCR signaling inhibitors combined with an HDAC inhibitor on cell death in a mantle cell lymphoma cell line. Apoptosis. 2015; 20: 975-985.
153. Ma J, Lu P, Guo A, Cheng S, Zong H, Martin P, Coleman M, Wang YL. Characterization of ibrutinib-sensitive and -resistant mantle lymphoma cells. Br J Haematol. 2014; 166:849-861.
154. Zhang SQ, Smith SM, Zhang SY, Lynn Wang Y. Mechanisms of ibrutinib resistance in chronic lymphocytic leukaemia and non-Hodgkin lymphoma. Br J Haematol. 2015; 170:556-456.
155. Smith SM. Targeting mTOR in mantle cell lymphoma: current and future directions. Best Pract Res Clin Haematol. 2012; 25:175-183.
156. Mallya S, Fitch BA, Lee JS, So L, Janes MR, Fruman DA. Resistance to mTOR kinase inhibitors in lymphoma cells lacking 4EBP1. PLoS One. 2014; 9:e88865.
157. Müller A, Zang C, Chumduri C, Dörken B, Daniel PT, Scholz CW. Concurrent inhibition of PI3K and mTORC1/mTORC2 overcomes resistance to rapamycin induced apoptosis by down-regulation of Mcl-1 in mantle cell lymphoma. Int J Cancer. 2013; 133:1813-1824.
158. Kim A, Park S, Lee JE, Jang WS, Lee SJ, Kang HJ, Lee SS. The dual PI3K and mTOR inhibitor NVP-BEZ235 exhibits anti-proliferative activity and overcomes bortezomib resistance in mantle cell lymphoma cells. Leuk Res. 2012; 36:912-920.
159. Gupta M, Hendrickson AE, Yun SS, Han JJ, Schneider PA, Koh BD, Stenson MJ, Wellik LE, Shing JC, Peterson KL, Flatten KS, Hess AD, Smith BD, et al. Dual mTORC1/mTORC2 inhibition diminishes Akt activation and induces Puma-dependent apoptosis in lymphoid malignancies. Blood. 2012; 119:476-487.
160. Chaturvedi NK, Rajule RN, Shukla A, Radhakrishnan P, Todd GL, Natarajan A, Vose JM, Joshi SS. Novel treatment for mantle cell lymphoma including therapy-resistant tumor by NF-κB and mTOR dual-targeting approach. Mol Cancer Ther. 2013; 12:2006-2017.
161. Westin JR. Status of PI3K/AKT/mTOR Pathway Inhibitors in Lymphoma. Clin Lymphoma Myeloma Leuk. 2014; 14:335-342.
162. Lannutti BJ, Meadows SA, Herman SE, Kashishian A, Steiner B, Johnson AJ, Byrd JC, Tyner JW, Loriaux MM, Deininger M, Druker BJ, Puri KD, Ulrich RG, Giese NA. CAL-101, a p110delta selective phosphatidylinositol-3-kinase inhibitor for the treatment of B-cell malignancies, inhibits PI3K signaling and cellular viability. Blood. 2011; 117:591-594.
163. Kahl BS, Spurgeon SE, Furman RR, Flinn IW, Coutre SE, Brown JR, Benson DM, Byrd JC, Peterman S, Cho Y, Yu A, Godfrey WR, Wagner-Johnston ND. A phase 1 study of the PI3Kδ inhibitor idelalisib in patients with relapsed/refractory mantle cell lymphoma (MCL). Blood. 2014; 123: 3398-3405.
164. Chiron D, Martin P, Di Liberto M, Huang X, Ely S, Lannutti BJ, Leonard JP, Mason CE, Chen-Kiang S. Induction of prolonged early G1 arrest by CDK4/CDK6 inhibition reprograms lymphoma cells for durable PI3Kδ inhibition through PIK3IP1. Cell Cycle. 2013; 12:1892-1900.
165. Furman RR, Sharman JP, Coutre SE, Furman RR, Sharman JP, Coutre SE, Cheson BD, Pagel JM, Hillmen P, Barrientos JC, Zelenetz AD, Kipps TJ, Flinn I, Ghia P, Eradat H, Ervin T, et al. Idelalisib and rituximab in relapsed chronic lymphocytic leukemia. N Engl J Med 2014;370:997-1007.
166. Papadopoulos KP, Egile C, Ruiz-Soto R, Jiang J, Shi W, Bentzien F, Rasco D, Abrisqueta P, Vose JM, Tabernero J. Efficacy, safety, pharmacokinetics and pharmacodynamics of SAR245409 (voxtalisib, XL765), an orally administered phosphoinositide 3-kinase/mammalian target of rapamycin inhibitor: a phase 1 expansion cohort in patients with relapsed or refractory lymphoma. Leuk Lymphoma. 2015;56:1763-1770.
167. Morschhauser F, Seymour JF, Kluin-Nelemans HC, Grigg A, Wolf M, Pfreundschuh M, Tilly H, Raemaekers J, van ‘t Veer MB, Milpied N, Cartron G, Pezzutto A, Spencer A, Reyes F, Dreyling M. A phase II study of enzastaurin, a protein kinase C beta inhibitor, in patients with relapsed or refractory mantle cell lymphoma. Ann Oncol. 2008; 19:247-253.
168. Cosenza M, Civallero M, Pozzi S, Marcheselli L, Bari A, Sacchi S. The combination of bortezomib with enzastaurin or lenalidomide enhances cytotoxicity in follicular and mantle cell lymphoma cell lines. Hematol Oncol. 2015; 33:166-75.
169. Sarkar AK. MK2206 disrupts cytoplasmic AKT-p21 complex in mantle cell lymphoma: Evidence of p21 coupled cell cycle arrest. American Association of Cancer Research meeting, 2015.
170. Reis-Sobreiro M, Roué G, Moros A, Gajate C, de la Iglesia-Vicente J, Colomer D, Mollinedo F. Lipid raft-mediated Akt signaling as a therapeutic target in mantle cell lymphoma. Blood Cancer J. 2013; 3:e118.
171. Valbuena JR, Rassidakis GZ, Lin P, Atwell C, Georgakis GV, Younes A, Jones D, Medeiros LJ. Expression of heat-shock protein-90 in non-Hodgkin’s lymphomas. Mod Pathol. 2005; 18:1343-1349.
172. Georgakis GV, Li Y, Younes A. The heat shock protein 90 inhibitor 17-AAG induces cell cycle arrest and apoptosis in mantle cell lymphoma cell lines by depleting Cyclin D1, Akt, Bid and activating caspase 9. Br J Haematol. 2006; 135:68-71.
173. Rao R, Lee P, Fiskus W, Yang Y, Joshi R, Wang Y, Buckley K, Balusu R, Chen J, Koul S, Joshi A, Upadhyay S, Tao J, Sotomayor E, Bhalla KN. Co-treatment with heat shock protein 90 inhibitor 17-dimethylaminoethylamino-17-demethoxygeldanamycin (DMAG) and vorinostat: a highly active combination against human mantle cell lymphoma (MCL) cells. Cancer Biol Ther. 2009; 8:1273-1280.
174. Jin L, Tabe Y, Kimura S, Zhou Y, Kuroda J, Asou H, Inaba T, Konopleva M, Andreeff M, Miida T. Antiproliferative and proapoptotic activity of GUT-70 mediated through potent inhibition of Hsp90 in mantle cell lymphoma. Br J Cancer. 2011; 104:91-100.
175. Holkova B, Grant S. Proteasome inhibitors in mantle cell lymphoma. Best Pract Res Clin Haematol. 2012; 25:133-141.
176. Goy A, Bernstein SH, Kahl BS, Djulbegovic B, Robertson MJ, de Vos S, Epner E, Krishnan A, Leonard JP, Lonial S, Nasta S, O’Connor OA, Shi H, Boral AL, Fisher RI. Bortezomib in patients with relapsed or refractory mantle cell lymphoma: updated time-to-event analyses of the multicenter phase 2 PINNACLE study. Ann Oncol. 2009; 20: 520-525.
177. Leshchenko VV, Kuo PY, Jiang Z, Weniger MA, Overbey J, Dunleavy K, Wilson WH, Wiestner A, Parekh S. Harnessing Noxa demethylation to overcome Bortezomib resistance in mantle cell lymphoma. Oncotarget. 2015; 29:27332-27342. doi: 10.18632/oncotarget.2903.
178. Roué G, Pérez-Galán P, Mozos A, López-Guerra M, Xargay-Torrent S, Rosich L, Saborit-Villarroya I, Normant E, Campo E, Colomer D. The Hsp90 inhibitor IPI-504 overcomes bortezomib resistance in mantle cell lymphoma in vitro and in vivo by down-regulation of the prosurvival ER chaperone BiP/Grp78. Blood. 2011;117:1270-1279.
179. Kupperman E, Lee EC, Cao Y, Bannerman B, Fitzgerald M, Berger A, Yu J, Yang Y, Hales P, Bruzzese F, Liu J, Blank J, Garcia K, et al. Evaluation of the proteasome inhibitor MLN9708 in preclinical models of human cancer. Cancer Res. 2010; 70:1970-1980.
180. Zhang L, Pham LV, Newberry KJ, Ou Z, Liang R, Qian J, Sun L, Blonska M, You Y, Yang J, Lin X, Rollo A, Tamayo AT, et al. In vitro and in vivo therapeutic efficacy of carfilzomib in mantle cell lymphoma: targeting the immunoproteasome. Mol Cancer Ther. 2013; 12:2494-2504.
181. Weilbacher DA, Gutekunst M, Staiger AM, Vöhringer MC, Horn H, Ott G, Aulitzky WE, van der Kuip H. Discrepant NOXA (PMAIP1) transcript and NOXA protein levels: a potential Achilles’ heel in mantle cell lymphoma Cell Death and Disease 2014; 5, e1013.
182. Tovar C, Graves B, Packman K, Filipovic Z, Higgins B, Xia M, Tardell C, Garrido R, Lee E, Kolinsky K, To KH, Linn M, Podlaski F, Wovkulich P, Vu B, Vassilev LT. MDM2 small-molecule antagonist RG7112 activates p53 signaling and regresses human tumors in preclinical cancer models. Cancer Res. 2013; 73:2587-97.
183. Davar D, Beumer JH, Hamieh L, Tawbi H. Role of PARP inhibitors in cancer biology and therapy. Curr Med Chem. 2012; 19:3907-3921.
184. Kubota E, Williamson CT, Ye R, Elegbede A, Peterson L, Lees-Miller SP, Bebb DG. Low ATM protein expression and depletion of p53 correlates with olaparib sensitivity in gastric cancer cell lines. Cell Cycle. 2014; 13:2129-2137.
185. Mottok A, Renné C, Seifert M, Oppermann E, Bechstein W, Hansmann ML, Küppers R, Bräuninger A. Inactivating SOCS1 mutations are caused by aberrant somatic hypermutation and restricted to a subset of B-cell lymphoma entities. Blood. 2009; 114:4503-4506.
186. Weniger MA, Wiestner A. Molecular targeted approaches in mantle cell lymphoma. Semin Hematol. 2011; 48:214-226
187. Pham LV, Tamayo AT, Li C, Bornmann W, Priebe W, Ford RJ. Degrasyn potentiates the antitumor effects of bortezomib in mantle cell lymphoma cells in vitro and in vivo: therapeutic implications. Mol Cancer Ther. 2010; 9:2026-2036.
188. Amin HM, Medeiros LJ, Ma Y, Feretzaki M, Das P, Leventaki V, Rassidakis GZ, O’Connor SL, McDonnell TJ, Lai R. Inhibition of JAK3 induces apoptosis and decreases anaplastic lymphoma kinase activity in anaplastic large cell lymphoma. Oncogene. 2003; 22:5399-5407.
189. Sakajiri S, Kumagai T, Kawamata N, Saitoh T, Said JW, Koeffler HP. Histone deacetylase inhibitors profoundly decrease proliferation of human lymphoid cancer cell lines. Exp Hematol. 2005; 33:53-61.
190. Heider U, Kaiser M, Sterz J, Zavrski I, Jakob C, Fleissner C, Eucker J, Possinger K, Sezer O. Histone deacetylase inhibitors reduce VEGF production and induce growth suppression and apoptosis in human mantle cell lymphoma. Eur J Haematol. 2006; 76:42-50.
191. Kawamata N, Chen J, Koeffler HP. Suberoylanilide hydroxamic acid (SAHA; vorinostat) suppresses translation of Cyclin D1 in mantle cell lymphoma cells. Blood. 2007; 110:2667-2673.
192. Fuchs O, Provaznikova D, Marinov I, Kuzelova K, Spicka I. Antiproliferative and proapoptotic effects of proteasome inhibitors and their combination with histone deacetylase inhibitors on leukemia cells. Cardiovasc Hematol Disord Drug Targets. 2009; 9:62-77.
193. Paoluzzi L, Scotto L, Marchi E, Zain J, Seshan VE, O’Connor OA. Romidepsin and belinostat synergize the antineoplastic effect of bortezomib in mantle cell lymphoma. Clin Cancer Res. 2010; 16:554-565.
194. Watanabe T. Investigational histone deacetylase inhibitors for non-Hodgkin lymphomas. Expert Opin Investig Drugs. 2010; 19:1113-1127.
195. Simmons JK, Patel J, Michalowski A, Zhang S, Wei BR, Sullivan P, Gamache B, Felsenstein K, Kuehl WM, Simpson RM, Zingone A, Landgren O, Mock BA. TORC1 and class I HDAC inhibitors synergize to suppress mature B cell neoplasms. Mol Oncol. 2014; 8:261-272.
196. Kirschbaum M, Frankel P, Popplewell L, Zain J, Delioukina M, Pullarkat V, Matsuoka D, Pulone B, Rotter AJ, Espinoza-Delgado I, Nademanee A, Forman SJ, Gandara D, Newman E. Phase II study of vorinostat for treatment of relapsed or refractory indolent non-Hodgkin’s lymphoma and mantle cell lymphoma. J Clin Oncol. 2011; 29:1198-1203.
197. Oki Y, Buglio D, Fanale M, Fayad L, Copeland A, Romaguera J, Kwak LW, Pro B, de Castro Faria S, Neelapu S, Fowler N, Hagemeister F, Zhang J, Zhou S, Feng L, Younes A. Phase I study of panobinostat plus everolimus in patients with relapsed or refractory lymphoma. Clin Cancer Res. 2013; 19:6882-6890.
198. Mondello P, Cuzzocrea S, Mian M. Pim kinases in hematological malignancies: where are we now and where are we going?. J Hematol Oncol. 2014; 10;7:95.
199. Cohen AM, Grinblat B, Bessler H, Kristt D, Kremer A, Schwartz A, Halperin M, Shalom S, Merkel D, Don J: Increased expression of the hPim-2 gene in human chronic lymphocytic leukemia and non-Hodgkin lymphoma. Leuk Lymphoma 2004;45:951-955.
200. Hogan C, Hutchison C, Marcar L, Milne D, Saville M, Goodlad J, Kernohan N, Meek D: Elevated levels of oncogenic protein kinase Pim-1 induce the p53 pathway in cultured cells and correlate with increased Mdm2 in mantle cell lymphoma. J Biol Chem 2008; 283:18012-18023.
201. Alvarado Y, Giles FJ, Swords RT. The PIM kinases in hematological cancers. Expert Rev Hematol. 2012; 5:81-96.
202. Yang Q, Chen LS, Neelapu SS, Miranda RN, Medeiros LJ, Gandhi V. Transcription and translation are primary targets of Pim kinase inhibitor SGI-1776 in mantle cell lymphoma. Blood. 2012; 120:3491-3500.
203. Yang Q, Chen LS, Neelapu SS, Gandhi V. Combination of Pim kinase inhibitor SGI-1776 and bendamustine in B-cell lymphoma. Clin Lymphoma Myeloma Leuk. 2013; 13:S355-362.
204. Vogler M, Dinsdale D, Dyer MJ, Cohen GM. Bcl-2 inhibitors: small molecules with a big impact on cancer therapy. Cell Death Differ. 2009; 16:360-367
205. Touzeau C, Dousset C, Bodet L, Gomez-Bougie P, Bonnaud S, Moreau A, Moreau P, Pellat-Deceunynk C, Amiot M, Le Gouill S. ABT-737 induces apoptosis in mantle cell lymphoma cells with a Bcl-2high/Mcl-1low profile and synergizes with other antineoplastic agents. Clin Cancer Res. 2011; 17:5973-5981.
206. Chiron D, Dousset C, Brosseau C, Touzeau C, Maïga S, Moreau P, Pellat-Deceunynck C, Le Gouill S, Amiot M. Biological rational for sequential targeting of Bruton tyrosine kinase and Bcl-2 to overcome CD40-induced ABT-199 resistance in mantle cell lymphoma. Oncotarget. 2015; 6:8750-8759. doi: 10.18632/oncotarget.3275.
207. Pérez-Galán P, Roué G, Villamor N, Campo E, Colomer D. The BH3-mimetic GX15-070 synergizes with bortezomib in mantle cell lymphoma by enhancing Noxa-mediated activation of Bak. Blood. 2007; 109:4441-4449.
208. Goy A, Hernandez-Ilzaliturri FJ, Kahl B, Ford P, Protomastro E, Berger M. A phase I/II study of the pan Bcl-2 inhibitor obatoclax mesylate plus bortezomib for relapsed or refractory mantle cell lymphoma. 2014; 55:2761-2768.
209. Stein R, Gupta P, Chen X, Cardillo TM, Furman RR, Chen S, Chang CH, Goldenberg DM. Therapy of B-cell malignancies by anti-HLA-DR humanized monoclonal antibody, IMMU-114, is mediated through hyperactivation of ERK and JNK MAP kinase signaling pathways. Blood. 2010; 115:5180-5190.
210. Alinari L, Yu B, Christian BA, Yan F, Shin J, Lapalombella R, Hertlein E, Lustberg ME, Quinion C, Zhang X, Lozanski G, Muthusamy N, Prætorius-Ibba M, et al. Combination anti-CD74 (milatuzumab) and anti-CD20 (rituximab) monoclonal antibody therapy has in vitro and in vivo activity in mantle cell lymphoma. Blood. 2011; 117:4530-4541.
211. Pagel JM, Spurgeon SE, Byrd JC, Awan FT, Flinn IW, Lanasa MC, Eisenfeld AJ, Stromatt SC, Gopal AK. Otlertuzumab (TRU-016), an anti-CD37 monospecific ADAPTIR(™) therapeutic protein, for relapsed or refractory NHL patients. Br J Haematol. 2015; 168:38-45.
212. Palanca-Wessels MC, Czuczman M, Salles G, Assouline S, Sehn LH, Flinn I, Patel MR, Sangha R, Hagenbeek A, Advani R, Tilly H, Casasnovas O, Press OW, et al. Safety and activity of the anti-CD79B antibody-drug conjugate polatuzumab vedotin in relapsed or refractory B-cell non-Hodgkin lymphoma and chronic lymphocytic leukaemia: a phase 1 study. Lancet Oncol. 2015; 16:704-715.
213. Börschel N, Schwöppe C, Zerbst C, Angenendt L, Kessler T, Klapper W, Giovannoni L, Elia G, Neri D, Berdel WE, Mesters RM, Schliemann C. Potentiating the activity of rituximab against mantle cell lymphoma in mice by targeting interleukin-2 to the neovasculature. Leuk Res. 2015; 39:739-748.
214. Chen J, Zurawski G, Zurawski S, Wang Z, Akagawa K, Oh S, Hideki U, Fay J, Banchereau J, Song W, Palucka AK. A novel vaccine for mantle cell lymphoma based on targeting Cyclin D1 to dendritic cells via CD40. J Hematol Oncol. 2015; 8:35.
215. Musgrove EA1, Caldon CE, Barraclough J, Stone A, Sutherland RL. Cyclin D as a therapeutic target in cancer. Nat Rev Cancer. 2011;11: 558-572.
216. GeneCards®: The Human Gene Database: http://www.genecards.org
217. Rodríguez-Burford C, Barnes MN, Oelschlager DK, Myers RB, Talley LI, Partridge EE, Grizzle WE. Effects of nonsteroidal anti-inflammatory agents (NSAIDs) on ovarian carcinoma cell lines: preclinical evaluation of NSAIDs as chemopreventive agents. Clin Cancer Res. 2002;8:202-209.
218. Benada J, Macurek L. Targeting the Checkpoint to Kill Cancer Cells. Biomolecules. 2015;5 :1912-1937.
219. Furgason JM, Bahassi el M. Targeting DNA repair mechanisms in cancer. Pharmacol Ther. 2013; 137(3):298-308.
220. Mutka SC, Yang WQ, Dong SD, Ward SL, Craig DA, Timmermans PB, Murli S. Identification of nuclear export inhibitors with potent anticancer activity in vivo. Cancer Res. 2009;69:510-517.
221. Chipumuro E, Marco E, Christensen CL, Kwiatkowski N, Zhang T, Hatheway CM, Abraham BJ, Sharma B, Yeung C, Altabef A, Perez-Atayde A, Wong KK, Yuan GC, et al. CDK7 inhibition suppresses super-enhancer-linked oncogenic transcription in MYCN-driven cancer. Cell. 2014;159:1126-39.
222. Aggarwal M, Villuendas R, Gomez G, Rodriguez-Pinilla SM, Sanchez-Beato M, Alvarez D, Martinez N, Rodriguez A, Castillo ME, Camacho FI, Montes-Moreno S, Garcia-Marco JA, Kimby E, Pisano DG, Piris MA. TCL1A expression delineates biological and clinical variability in B-cell lymphoma. Mod Pathol. 2009; 22: 206-215.
223. Huang YC, Chuang LY, Hung WC. Mechanisms underlying nonsteroidal anti-inflammatory drug-induced p27(Kip1) expression. Mol Pharmacol. 2002;62:1515-21.
224. Chilà R, Basana A, Lupi M, Guffanti F, Gaudio E, Rinaldi A, Cascione L, Restelli V, Tarantelli C, Bertoni F, Damia G, Carrassa L. Combined inhibition of Chk1 and Wee1 as a new therapeutic strategy for mantle cell lymphoma. Oncotarget. 2015;6:3394-3408. doi: 10.18632/oncotarget.2583.
225. Liu Z, Davidson A. BAFF inhibition: a new class of drugs for the treatment of autoimmunity. Exp Cell Res. 2011; 317:1270-1277.
226. Yue P, Turkson J. Targeting STAT3 in cancer: how successful are we? Expert Opin Investig Drugs. 2009;18:45-56.
227. Lu K, Chen N, Zhou XX, Ge XL, Feng LL, Li PP, Li XY, Geng LY, Wang X. The STAT3 inhibitor WP1066 synergizes with vorinostat to induce apoptosis of mantle cell lymphoma cells. Biochem Biophys Res Commun. 2015;464:292-298.
228. Pham LV, Tamayo AT, Yoshimura LC, Lo P, Ford RJ. Inhibition of constitutive NF-kappa B activation in mantle cell lymphoma B cells leads to induction of cell cycle arrest and apoptosis. J Immunol. 2003;171:88-95.
229. Youn J, Lee K, Lee C, Gabrilovich D. Loss of Rb1 by epigenetic modification regulates expansion of MDSC in cancer. J Immunother Cancer. 2014; 2: P241.
230. Jin L, Tabe Y, Kojima K, Zhou Y, Pittaluga S, Konopleva M, Miida T, Raffeld M. MDM2 antagonist Nutlin-3 enhances bortezomib-mediated mitochondrial apoptosis in TP53-mutated mantle cell lymphoma. Cancer Lett. 2010;299:161-170.
231. Gualco G, Weiss LM, Harrington WJ Jr, Bacchi CE. BCL6, MUM1, and CD10 expression in mantle cell lymphoma. Appl Immunohistochem Mol Morphol. 2010;18:103-108.
232. Yi S, Zou D, Li C, Zhong S, Chen W, Li Z, Xiong W, Liu W, Liu E, Cui R, Ru K, Zhang P, Xu Y, An G, Lv R, Qi J, Wang J, Cheng T, Qiu L. High incidence of MYC and BCL2 abnormalities in mantle cell lymphoma, although only MYC abnormality predicts poor survival. Oncotarget. 2015. doi: 10.18632/oncotarget.5705.
233. Beà S, Valdés-Mas R, Navarro A, Salaverria I, Martín-Garcia D, Jares P, Giné E, Pinyol M, Royo C, Nadeu F, Conde L, Juan M, Clot G, et al. Landscape of somatic mutations and clonal evolution in mantle cell lymphoma. Proc Natl Acad Sci U S A. 2013; 110:18250-18255.
234. Dillon LM, Miller TW. Therapeutic targeting of cancers with loss of PTEN function. Curr Drug Targets. 2014;15: 65-79.
235. Verstrepen L, Carpentier I, Verhelst K, Beyaert R. ABINs: A20 binding inhibitors of NF-kappa B and apoptosis signaling. Biochem Pharmacol. 2009;78:105-114.
236. Hartmann E, Fernàndez V, Moreno V, Valls J, Hernández L, Bosch F, Abrisqueta P, Klapper W, Dreyling M, Hoster E, Müller-Hermelink HK, Ott G, Rosenwald A, Campo E. Five-gene model to predict survival in mantle-cell lymphoma using frozen or formalin-fixed, paraffin-embedded tissue. J Clin Oncol. 2008;26:4966-4472.
237. Schrader C, Janssen D, Meusers P, Brittinger G, Siebmann JU, Parwaresch R, Tiemann M. Repp86: a new prognostic marker in mantle cell lymphoma. Eur J Haematol. 2005;75:498-504.
238. Martinez A, Bellosillo B, Bosch F, Ferrer A, Marcé S, Villamor N, Ott G, Montserrat E, Campo E, Colomer D. Nuclear survivin expression in mantle cell lymphoma is associated with cell proliferation and survival. Am J Pathol. 2004;164:501-510.
239. Beà S, Salaverria I, Armengol L, Pinyol M, Fernández V, Hartmann EM, Jares P, Amador V, Hernández L, Navarro A, Ott G, Rosenwald A, Estivill X, Campo E. Uniparental disomies, homozygous deletions, amplifications, and target genes in mantle cell lymphoma revealed by integrative high-resolution whole-genome profiling. Blood. 2009;113:3059-3069.
240. Schrader C, Meusers P, Brittinger G, Teymoortash A, Siebmann JU, Janssen D, Parwaresch R, Tiemann M. Topoisomerase IIalpha expression in mantle cell lymphoma: a marker of cell proliferation and a prognostic factor for clinical outcome. Leukemia. 2004;18:1200-1206.
241. Hartmann EM, Beà S, Navarro A, Trapp V, Campo E, Ott G, Rosenwald A. Increased tumor cell proliferation in mantle cell lymphoma is associated with elevated insulin-like growth factor 2 mRNA-binding protein 3 expression. Mod Pathol. 2012;25:1227-1235.
242. Lederer M, Bley N, Schleifer C, Hüttelmaier S. The role of the oncofetal IGF2 mRNA-binding protein 3 (IGF2BP3) in cancer. Semin Cancer Biol. 2014;29: 3-12.
243. Park S, Ko YH. A unique case of blastoid variant of mantle cell lymphoma with an aberrant CD5-/CD10+/Bcl-6+/CD56+ immunophenotype: a case report. Tumori. 2012;98:e111-114.
244. Palumbo GA, Parrinello N, Fargione G, Cardillo K, Chiarenza A, Berretta S, Conticello C, Villari L, Di Raimondo F. CD200 expression may help in differential diagnosis between mantle cell lymphoma and B-cell chronic lymphocytic leukemia. Leuk Res. 2009;33:1212-1216.
245. Kurtova AV, Tamayo AT, Ford RJ, Burger JA. Mantle cell lymphoma cells express high levels of CXCR4, CXCR5, and VLA-4 (CD49d): importance for interactions with the stromal microenvironment and specific targeting. Blood. 2009;113:4604-4613.
246. Zhou DM, Chen G, Zheng XW, Zhu WF, Chen BZ. Clinicopathologic features of 112 cases with mantle cell lymphoma. Cancer Biol Med. 2015;12:46-52.
247. Isaza-Correa JM, Liang Z, van den Berg A, Diepstra A, Visser L. Toll-like receptors in the pathogenesis of human B cell malignancies. J Hematol Oncol. 2014;7:57.
248. Gelebart P, Zak Z, Dien-Bard J, Anand M, Lai R. Interleukin 22 signaling promotes cell growth in mantle cell lymphoma. Transl Oncol. 2011;4:9-19.
249. Argyriou P, Economopoulou P, Papageorgiou S. The Role of mTOR Inhibitors for the Treatment of B-Cell Lymphomas. Adv Hematol. 2012;2012: 435342.
250. Curtin JC, Lorenzi MV. Drug discovery approaches to target Wnt signaling in cancer stem cells. Oncotarget. 2010;1:552-566. doi: 10.18632/oncotarget.191.
251. Chung R, Peters AC, Armanious H, Anand M, Gelebart P, Lai R. Biological and clinical significance of GSK-3beta in mantle cell lymphoma—an immunohistochemical study. Int J Clin Exp Pathol. 2010;3:244-253.
252. Sachanas S, Pangalis GA, Vassilakopoulos TP, Korkolopoulou P, Kontopidou FN, Athanasoulia M, Yiakoumis X, Kalpadakis C, Georgiou G, Masouridis S, Moschogiannis M, Tsirkinidis P, Pappis V, et al. Combination of rituximab with chlorambucil as first line treatment in patients with Mantle cell lymphoma: a highly effective regimen. Leuk Lymphoma. 2011;52:387-393.
253. Ruan J, Martin P, Furman RR, Lee SM, Cheung K, Vose JM, Lacasce A, Morrison J, Elstrom R, Ely S, Chadburn A, Cesarman E, Coleman M, Leonard JP. Bortezomib plus CHOP-rituximab for previously untreated diffuse large B-cell lymphoma and mantle cell lymphoma. J Clin Oncol. 2011;29:690-697.
254. Gressin R, Caulet-Maugendre S, Deconinck E, Tournilhac O, Gyan E, Moles MP, El Yamani A, Cornillon J, Rossi JF, Le Gouill S, Lepeu G, Damaj G, Celigny PS, et al. Evaluation of the (R)VAD+C regimen for the treatment of newly diagnosed mantle cell lymphoma. Combined results of two prospective phase II trials from the French GOELAMS group. Haematologica. 2010;95:1350-1357.
255. Evens AM, Winter JN, Hou N, Nelson BP, Rademaker A, Patton D, Singhal S, Frankfurt O, Tallman MS, Rosen ST, Mehta J, Gordon LI. A phase II clinical trial of intensive chemotherapy followed by consolidative stem cell transplant: long-term follow-up in newly diagnosed mantle cell lymphoma. Br J Haematol. 2008; 140: 385-393.
256. Friedberg JW, Sharman J, Sweetenham J, Johnston PB, Vose JM, Lacasce A, Schaefer-Cutillo J, De Vos S, Sinha R, Leonard JP, Cripe LD, Gregory SA, Sterba MP, et al. Inhibition of Syk with fostamatinib disodium has significant clinical activity in non-Hodgkin lymphoma and chronic lymphocytic leukemia. Blood. 2010;115: 2578-2585.
257. Herold M, Schulze A, Niederwieser D, Franke A, Fricke HJ, Richter P, Freund M, Ismer B, Dachselt K, Boewer C, Schirmer V, Weniger J, Pasold R, et al. Bendamustine, vincristine and prednisone (BOP) versus cyclophosphamide, vincristine and prednisone (COP) in advanced indolent non-Hodgkin’s lymphoma and mantle cell lymphoma: results of a randomised phase III trial (OSHO# 19). J Cancer Res Clin Oncol. 2006;132:105-112.
258. Romaguera JE, Fayad L, Rodriguez MA, Broglio KR, Hagemeister FB, Pro B, McLaughlin P, Younes A, Samaniego F, Goy A, Sarris AH, Dang NH, Wang M, et al. High rate of durable remissions after treatment of newly diagnosed aggressive mantle-cell lymphoma with rituximab plus hyper-CVAD alternating with rituximab plus high-dose methotrexate and cytarabine. J Clin Oncol. 2005;23: 7013-7023.
259. Mangel J, Leitch HA, Connors JM, Buckstein R, Imrie K, Spaner D, Crump M, Pennell N, Boudreau A, Berinstein NL. Intensive chemotherapy and autologous stem-cell transplantation plus rituximab is superior to conventional chemotherapy for newly diagnosed advanced stage mantle-cell lymphoma: a matched pair analysis. Ann Oncol. 2004;15:283-290.
260. Neville KE, Bisquera A, Capp AL. Involved-field radiotherapy for patients with mantle cell lymphoma. J Med Imaging Radiat Oncol. 2015; 59:631-639.
261. Hess G, Keller U, Scholz CW, Witzens-Harig M, Atta J, Buske C, Kirschey S, Ruckes C, Medler C, van Oordt C, Klapper W, Theobald M, Dreyling M. Safety and efficacy of Temsirolimus in combination with Bendamustine and Rituximab in relapsed mantle cell and follicular lymphoma. Leukemia. 2015;29:1695-1701.
262. Kirschey S, Flohr T, Wolf HH, Frickhofen N, Gramatzki M, Link H, Basara N, Peter N, Meyer RG, Schmitz N, Weidmann E, Banat A, Schulz A, et al. Rituximab combined with DexaBEAM followed by high dose therapy as salvage therapy in patients with relapsed or refractory B-cell lymphoma: mature results of a phase II multicentre study. Br J Haematol. 2015;168:824-834.
263. Cheson BD, Crawford J.A phase I study of bendamustine, lenalidomide and rituximab in relapsed and refractory lymphomas. Br J Haematol. 2015;169: 528-533.
264. Fenske TS, Shah NM, Kim KM, Saha S, Zhang C, Baim AE, Farnen JP, Onitilo AA, Blank JH, Ahuja H, Wassenaar T, Qamar R, Mansky P, Traynor AM, Mattison RJ, Kahl BS. A Phase 2 Study of Weekly Temsirolimus and Bortezomib for Relapsed or Refractory B-Cell Non-Hodgkin Lymphoma: a Wisconsin Oncology Network Study. Cancer. 2015;121: 3465-3471.
265. Friedberg JW, Mahadevan D, Cebula E, Persky D, Lossos I, Agarwal AB, Jung J, Burack R, Zhou X, Leonard EJ, Fingert H, Danaee H, Bernstein SH. Phase II Study of Alisertib, a Selective Aurora A Kinase Inhibitor, in Relapsed and Refractory Aggressive B- and T-Cell Non-Hodgkin Lymphomas. J Clin Oncol. 2014;32:44-50.
266. Wang M, Fayad L, Wagner-Bartak N, Zhang L, Hagemeister F, Neelapu SS, Samaniego F, McLaughlin P, Fanale M, Younes A, Cabanillas F, Fowler N, Newberry KJ, et al. Lenalidomide in combination with rituximab for patients with relapsed or refractory mantle-cell lymphoma: a phase 1/2 clinical trial. Lancet Oncol. 2012;13:716-723.
267. Renner C, Zinzani PL, Gressin R, Klingbiel D, Dietrich PY, Hitz F, Bargetzi M, Mingrone W, Martinelli G, Trojan A, Bouabdallah K, Lohri A, Gyan E, et al. A multicenter phase II trial (SAKK 36/06) of single-agent everolimus (RAD001) in patients with relapsed or refractory mantle cell lymphoma. Haematologica. 2012;97: 1085-1091.
268. Rigacci L, Puccini B, Cortelazzo S, Gaidano G, Piccin A, D’Arco A, Freilone R, Storti S, Orciuolo E, Zinzani PL, Zaja F, Bongarzoni V, Balzarotti M, et al. A multicenter retrospective study on behalf of the Italian Lymphoma Foundation (FIL). Ann Hematol. 2012;91:1013-1022.
269. Ansell SM, Hui Tang, Kurtin PJ, Koenig PA, Inwards DJ, Shah K, Ziesmer SC, Feldman AL, Rao R, Gupta M, Erlichman C, Witzig TE. Temsirolimus and rituximab in patients with relapsed or refractory mantle cell lymphoma: a phase 2 study. Lancet Oncol. 2011;12:361-368.
270. Ruan J, Martin P, Coleman M, Furman RR, Cheung K, Faye A, Elstrom R, Lachs M, Hajjar KA, Leonard JP. Durable Responses With the Metronomic Rituximab and Thalidomide Plus Prednisone,Etoposide, Procarbazine, and Cyclophosphamide Regimen in Elderly Patients With Recurrent Mantle Cell Lymphoma. Cancer. 2010; 116:2655-2564.
271. Rolland D, Ribrag V, Haioun C, Ghesquieres H, Jardin F, Bouabdallah R, Franchi P, Briere J, De Kerviler E, Chassagne-Clement C, Raponi M, Houlgatte R, Jais JP, Thieblemont C. Phase II trial and prediction of response of single agent tipifarnib in patients with relapsed/refractory mantle cell lymphoma. Cancer Chemother Pharmacol. 2010;65: 781-790.
272. Wang M, Oki Y, Pro B, Romaguera JE, Rodriguez MA, Samaniego F, McLaughlin P, Hagemeister F, Neelapu S, Copeland A, Samuels BI, Loyer EM, Ji Y, Younes A. Phase II study of yttrium-90-ibritumomab tiuxetan in patients with relapsed or refractory mantle cell lymphoma. J Clin Oncol. 2009;27:5213-5218.
273. Garbo LE, Flynn PJ, MacRae MA, Rauch MA, Wang Y, Kolibaba KS. Results of a Phase II trial of gemcitabine, mitoxantrone, and rituximab in relapsed or refractory mantle cell lymphoma. Invest New Drugs. 2009;27: 476-481.
274. Wang M, Fayad L, Cabanillas F, Hagemeister F, McLaughlin P, Rodriguez MA, Kwak LW, Zhou Y, Kantarjian H, Romaguera J. Phase 2 trial of rituximab plus hyper-CVAD alternating with rituximab plus methotrexate-cytarabine for relapsed or refractory aggressive mantle cell lymphoma. Cancer. 2008;113: 2734-2741.
275. O’Connor OA, Portlock C, Moskowitz C, Straus D, Hamlin P, Stubblefield M, Dumetrescu O, Colevas AD, Grant B, Zelenetz A. A multicentre phase II clinical experience with the novel aza-epothilone Ixabepilone (BMS247550) in patients with relapsed or refractory indolent non-Hodgkin lymphoma and mantle cell lymphoma. Br J Haematol. 2008;143:201-209.
276. Goy A, Younes A, McLaughlin P, Pro B, Romaguera JE, Hagemeister F, Fayad L, Dang NH, Samaniego F, Wang M, Broglio K, Samuels B, Gilles F, et al. Phase II study of proteasome inhibitor bortezomib in relapsed or refractory B-cell non-Hodgkin’s lymphoma. J Clin Oncol. 2005;23: 667-675.
277. Rai KR, Peterson BL, Appelbaum FR, Kolitz J, Elias L, Shepherd L, Hines J, Threatte GA, Larson RA, Cheson BD, Schiffer CA: Fludarabine compared with chlorambucil as primary therapy for chronic lymphocytic leukemia. N Engl J Med. 2000; 343:1750-1757.
278. Gunnellini M, Falchi L. Therapeutic Activity of Lenalidomide in Mantle Cell Lymphoma and Indolent Non-Hodgkin’s Lymphomas. Adv Hematol. 2012;2012:523842.
279. Grisafi D, Maestro A, Grumi C, Piazzoni L, Tirone G, Fiore W, Tessari R, Gianardi V, Gatti M, Tasca F, Generali D, Ravelli A, Lanza F, Scaglione F. Ibrutinib: from bench side to clinical implications. Med Oncol. 2015;32:225.
280. Citterio G, Reni M, Ferreri AJ. Present and future treatment options for primary CNS lymphoma. Expert Opin Pharmacother. 2015;15:1-11.
281. Pérez-Galán P, Dreyling M, Wiestner A. Mantle cell lymphoma: biology, pathogenesis, and the molecular basis of treatment in the genomic era. Blood. 2011; 117: 26-38.
282. Jurczak W, Kisiel E, Sawczuk-Chabin J, Centkowski P, Knopińska-Posłuszny W, Khan O. The use of Yttrium-90 Ibritumomab Tiuxetan ((90)Y-IT) as a consolidation therapy in high-risk patients with diffuse large B-cell lymphoma ineligible for autologous stem-cell transplantation. Contemp Oncol (Pozn). 2015;19:43-47.
283. Abraham RT, Gibbons JJ. The mammalian target of rapamycin signaling pathway: twists and turns in the road to cancer therapy. Clin Cancer Res. 2007;13:3109-3114.
284. Fraiser LH, Kanekal S, Kehrer JP. Cyclophosphamide toxicity. Characterizing and avoiding the problem. Drugs.1991; 42:781-795.
285. Shi Y, Zhou P, Han X, He X, Zhou S, Liu P, Yang J, Zhang C, Gui L, Qin Y, Yang S, Zhao L, Yao J, Zhang S. Autologous peripheral blood stem cell mobilization following dose-adjusted cyclophosphamide, doxorubicin, vincristine, and prednisolone chemotherapy alone or in combination with rituximab in treating high-risk non-Hodgkin’s lymphoma. Chin J Cancer. 2015;34:52.
286. Tewey KM, Rowe TC, Yang L, Halligan BD, Liu LF. Adriamycin-induced DNA damage mediated by mammalian DNA topoisomerase-II. Science. 1984;226:466-468.
287. Belgaumi AF, Al-Bakrah M, Al-Mahr M, Al-Jefri A, Al-Musa A, Saleh M, Salim MF, Osman M, Osman L, El-Solh H. Dexamethasone‐associated toxicity during induction chemotherapy for childhood acute lymphoblastic leukemia is augmented by concurrent use of daunomycin. Cancer. 2003;97: 2898-2903.
288. Ye XN, Zhou XP, Wei JY, Xu GX, Li Y, Mao LP, Huang J, Ren YL, Mei C, Wang JH, Lou YJ, Ma LY, Yu WJ, Ye L, Xie LL, Luo YW, Hu C, Niu LM, Dou MH, Jin J, Tong HY. Epigenetic priming with decitabine followed by low- dose idarubicin /cytarabine has an increased anti-leukemic effect compared to traditional chemotherapy in high-risk myeloid neoplasms. Leuk Lymphoma. 2015; 13: 1-8.
289. Weide R. Bendamustine HCL for the treatment of relapsed indolent non-Hodgkin’s lymphoma. Therapeutics and Clinical Risk Management. 2008;4:727-732.
290. Adams J, Palombella VJ, Sausville EA, Johnson J, Destree A, Lazarus DD, Maas J, Pien CS, Prakash S, Elliott PJ.. Proteasome inhibitors: a novel class of potent and effective antitumor agents. Cancer Res. 1999; 59:2615-2622.
291. Foran JM, Cunningham D, Coiffier B, Solal-Celigny P, Reyes F, Ghielmini M, Johnson PW, Gisselbrecht C, Bradburn M, Matthews J, Lister TA. Treatment of mantle-cell lymphoma with Rituximab (chimeric monoclonal anti-CD20 antibody): analysis of factors associated with response. Ann Oncol. 2000;11:117-121.
292. Richon VM. Cancer biology: mechanism of antitumour action of vorinostat (suberoylanilide hydroxamic acid), a novel histone deacetylase inhibitor. Br J Cancer. 2006;95: S2-S6.
293. Manfredi MG, Ecsedy JA, Chakravarty A, Silverman L, Zhang M, Hoar KM, Stroud SG, Chen W, Shinde V, Huck JJ, Wysong DR, Janowick DA, Hyer ML, et al. Characterization of Alisertib (MLN8237), an investigational small-molecule inhibitor of aurora A kinase using novel in vivo pharmacodynamic assays. Clin Cancer Res. 2011; 17:7614-24.
294. Lannutti BJ, Meadows SA, Herman SE, Kashishian A, Steiner B, Johnson AJ, Byrd JC, Tyner JW, Loriaux MM, Deininger M, Druker BJ, Puri KD, Ulrich RG, Giese NA. CAL-101, a p110δ selective phosphatidylinositol-3-kinase inhibitor for the treatment of B-cell malignancies, inhibits PI3K signaling and cellular viability. Blood. 2011; 117: 591-594.
295. Wang M, Popplewell LL, Collins RH Jr, Winter JN, Goy A, Kaminski MS, Bartlett NL, Johnston PB, Lister J, Fanning SR, Tuscano JM, Beck JT, Kaya H, et al. Everolimus for patients with mantle cell lymphoma refractory to or intolerant of bortezomib: multicentre, single-arm, phase 2 study. Br J Haematol. 2014;165: 510-518.
296. Coiffier B, Lepretre S, Pedersen LM, Gadeberg O, Fredriksen H, van Oers MH, Wooldridge J, Kloczko J, Holowiecki J, Hellmann A, Walewski J, Flensburg M, Petersen J, Robak T. Safety and efficacy of ofatumumab, a fully human monoclonal anti-CD20 antibody, in patients with relapsed or refractory B-cell chronic lymphocytic leukemia: a phase 1-2 study. Blood. 2008;111:1094-100.
297. Revill P, Mealy N, Serradell N, Bolos J, Rosa E. Panobinostat. Drugs of the Future. 2007; 32: 315.
298. VanderMolen KM, McCulloch W, Pearce CJ, Oberlies NH. Romidepsin (Istodax, NSC 630176, FR901228, FK228, depsipeptide): a natural product recently approved for cutaneous T-cell lymphoma. J Antibiot(Tokyo).2011; 64:525-531.
299. Nguyen M, Marcellus RC, Roulston A, Watson M, Serfass L, Murthy Madiraju SR, Goulet D, Viallet J, Bélec L, Billot X, Acoca S, Purisima E, Wiegmans A, Cluse L, Johnstone RW, Beauparlant P, Shore GC. Small molecule obatoclax (GX15-070) antagonizes MCL-1 and overcomes MCL-1-mediated resistance to apoptosis. Proc Natl Acad Sci U S A. 2007;104:19512-19517.
300. Mérino D, Khaw SL, Glaser SP, Anderson DJ, Belmont LD, Wong C, Yue P, Robati M, Phipson B, Fairlie WD, Lee EF, Campbell KJ, Vandenberg CJ, et al. Bcl-2, Bcl-x(L), and Bcl-w are not equivalent targets of ABT-737 and navitoclax (ABT-263) in lymphoid and leukemic cells. Blood. 2012; 119:5807-5816.
301. Popa MA, McCord AM, Moran J, Quintero J, Santos CF. Current First Remission Consolidation Options For Follicular Lymphoma: Are We There Yet? Rom J Oncol Hematol. 2014; 2: 8-11.
302. Hsieh CH, Chao KS, Liao HF, Chen YJ. Norcantharidin, derivative of cantharidin, for cancer stem cells. Evid Based Complement Alternat Med. 2013;2013:838651.
303. Hu W, Yang X, Zhe C, Zhang Q, Sun L, Cao K. Puerarin inhibits iNOS, COX-2 and CRP expression via suppression of NF-κB activation in LPS-induced RAW264.7 macrophage cells. Pharmacol Rep. 2011;63:781-789.
304. Shailubhai K, Dheer S, Picker D, Kaur G, Sausville EA, Jacob GS. Atiprimod is an inhibitor of cancer cell proliferation and angiogenesis. J Exp Ther Oncol. 2004; 4:267-279.
305. Faderl S, Ferrajoli A, Harris D, Van Q, Kantarjian HM, Estrov Z. Atiprimod blocks phosphorylation of JAK-STAT and inhibits proliferation of acute myeloid leukemia (AML) cells. Leuk Res. 2007;31:91-95.
306. Nguyen TK and Grant S. Dinaciclib (SCH727665) inhibits the unfolded protein response (UPR) through a CDK1 and CDK5-dependent mechanism. Molecular Cancer Therapeutics. 2016;13: 662-674.
307. Martin MP, Olesen SH, Georg GI, Schönbrunn E. Cyclin-dependent kinase inhibitor dinaciclib interacts with the acetyl-lysine recognition site of bromodomains. ACS Chemical Biology. 2013; 8: 2360-2365.
308. Lin TS, Andritsos LA, Jones JA, Fischer B, Heerema NA, Blum KA, Flynn JM, Moran ME, Phelps MA, Grever MR, Byrd JC. Activity of the cyclin-dependent kinase (CDK) inhibitor flavopiridol in relapsed, genetically high risk chronic lymphocytic leukemia (CLL). Journal of Clinical Oncology. 2008; 26: 373s.
309. Flinn IW, Byrd JC, Bartlett N, Kipps T, Gribben J, Thomas D, Larson RA, Rai K, Petric R, Ramon-Suerez J, Gabrilove J, Grever MR. Flavopiridol administered as a 24-hour continuous infusion in chronic lymphocytic leukemia lacks clinical activity. Leukemia Research. 2005;29: 1253-1257.
310. Friedberg JW, Sharman J, Sweetenham J, Johnston PB, Vose JM, Lacasce A, Schaefer-Cutillo J, De Vos S, Sinha R, Leonard JP, Cripe LD, Gregory SA, Sterba MP, et al. Inhibition of Syk with fostamatinib disodium has significant clinical activity in non-Hodgkin lymphoma and chronic lymphocytic leukemia. Blood. 2010; 115:2578-2585.
311. Robertson MJ, Kahl BS, Vose JM, de Vos S, Laughlin M, Flynn PJ, Rowland K, Cruz JC, Goldberg SL, Musib L, Darstein C, Enas N, Kutok JL, et al. Phase II study of enzastaurin, a protein kinase C beta inhibitor, in patients with relapsed or refractory diffuse large B-cell lymphoma. J Clin Oncol. 2007; 25:1741-1746.
312. Robertson MJ, Kahl BS, Vose JM, de Vos S, Laughlin M, Flynn PJ, Rowland K, Cruz JC, Goldberg SL, Musib L, Darstein C, Enas N, Kutok JL, et al. CDK 4/6 inhibitor palbociclib (PD0332991) in Rb+ advanced breast cancer: phase II activity, safety, and predictive biomarker assessment. Clin Cancer Res. 2015;21:995-1001.
313. Kondapaka SB, Singh SS, Dasmahapatra GP, Sausville EA, Roy KK. Perifosine, a novel alkylphospholipid, inhibits protein kinase B activation. Mol Cancer Ther. 2003;2:1093-1103.
314. Holohan B, Hagiopian MM, Lai TP, Huang E, Friedman DR, Wright WE, Shay JW. Perifosine as a potential novel anti-telomerase therapy. Oncotarget. 2015; 6: 21816-21826. doi: 10.18632/oncotarget.5200.
315. Yap TA, Yan L, Patnaik A, Fearen I, Olmos D, Papadopoulos K, Baird RD, Delgado L, Taylor A, Lupinacci L, Riisnaes R, Pope LL, Heaton SP, et al. First-in-man clinical trial of the oral pan-AKT inhibitor MK-2206 in patients with advanced solid tumors. Journal of Clinical Oncology. 2011;29: 4688-4695.
316. Dumble M, Crouthamel MC, Zhang SY, Schaber M, Levy D, Robell K, Liu Q, Figueroa DJ, Minthorn EA, Seefeld MA, Rouse MB, Rabindran SK, Heerding DA, Kumar R. Discovery of novel AKT inhibitors with enhanced anti-tumor effects in combination with the MEK inhibitor. PLoS One. 2014;9:e100880.
317. Mahadevan D, Chiorean EG, Harris WB, Von Hoff DD, Stejskal-Barnett A, Qi W, Anthony SP, Younger AE, Rensvold DM, Cordova F, Shelton CF, Becker MD, Garlich JR, et al. Phase I pharmacokinetic and pharmacodynamic study of the pan-PI3K/mTORC vascular targeted pro-drug SF1126 in patients with advanced solid tumours and B-cell malignancies. European journal of cancer. 2012; 48:3319-3327.
318. Qi W, Stejskal A, Morales C, Cooke LS, Garlich JR, Mahadevan D. SF1126, a Pan-PI3K Inhibitor has Potent Pre-Clinical Activity in Aggressive B-Cell Non- Hodgkin Lymphomas by Inducing Cell Cycle Arrest and Apoptosis. J Cancer Sci Ther. 2012; 4: 207-213.
319. Xiong R, Zhou W, Siegel D, Kitson RR, Freed CR, Moody CJ, Ross D. A Novel Hsp90 Inhibitor Activates Compensatory Heat Shock Protein Responses and Autophagy and Alleviates Mutant A53T α-Synuclein Toxicity. Mol Pharmacol. 2015;88:1045-1054.
320. Pedersen KS, Kim GP, Foster NR, Wang-Gillam A, Erlichman C, McWilliams RR. Phase II trial of gemcitabine and tanespimycin (17AAG) in metastatic pancreatic cancer: a Mayo Clinic Phase II Consortium study. Invest New Drugs. 2015; 33:963-968.
321. Proia DA and Bates RC. Ganetespib and HSP90: translating preclinical hypotheses into clinical promise. Cancer Res. 2014; 74:1294-1300.
322. Milhollen MA, Traore T, Adams-Duffy J, Thomas MP, Berger AJ, Dang L, Dick LR, Garnsey JJ, Koenig E, Langston SP, Manfredi M, Narayanan U, Rolfe M, et al. MLN4924, a NEDD8-activating enzyme inhibitor, is active in diffuse large B-cell lymphoma models: rationale for treatment of NF-κB-dependent lymphoma. Blood. 2010; 116: 1515-1523.
323. Infante JR, Weiss GJ, Jones S, Tibes R, Bauer TM, Bendell JC, Hinson JM Jr, Von Hoff DD, Burris HA 3rd, Orlemans EO, Ramanathan RK. Phase I dose-escalation studies of SNX-5422, an orally bioavailable heat shock protein 90 inhibitor, in patients with refractory solid tumours. Eur J Cancer. 2014; 50:2897-904.
324. Mulder A, Heidt S, Vergunst M, Roelen DL, Claas FH. Proteasome inhibition profoundly affects activated human B cells. Transplantation. 2013;95:1331-1337.
325. Ma L, Diao A. Marizomib, a potent second generation proteasome inhibitor from natural origin. Anticancer Agents Med Chem. 2015;15:298-306.
326. Coffey G, Betz A, DeGuzman F, Pak Y, Inagaki M, Baker DC, Hollenbach SJ, Pandey A, Sinha U. The novel kinase inhibitor PRT062070 (Cerdulatinib) demonstrates efficacy in models of autoimmunity and B-cell cancer. J Pharmacol Exp Ther. 2014;351:538-548.
327. Guidetti A, Carlo-Stella C, Locatelli SL, Malorni W, Pierdominici M, Barbati C, Mortarini R, Devizzi L, Matteucci P, Marchianò A, Lanocita R, Farina L, Dodero A, et al. Phase II study of sorafenib in patients with relapsed or refractory lymphoma. British journal of haematology. 2012; 158: 108-119.
328. Schmeel FC, Schmeel LC, Kim Y, Schmidt-Wolf IG. Piceatannol exhibits selective toxicity to multiple myeloma cells and influences the Wnt/ beta-catenin pathway. Hematol Oncol. 2014; 32:197-204.
329. Foss F, Advani R, Duvic M, Hymes KB, Intragumtornchai T, Lekhakula A, Shpilberg O, Lerner A, Belt RJ, Jacobsen ED, Laurent G, Ben-Yehuda D, Beylot-Barry M, et al. A Phase II trial of Belinostat (PXD101) in patients with relapsed or refractory peripheral or cutaneous T-celllymphoma. Br J Haematol. 2015; 168:811-819.
330. Ngamphaiboon N, Dy GK, Ma WW, Zhao Y, Reungwetwattana T, DePaolo D, Ding Y, Brady W, Fetterly G, Adjei AA. A phase I study of the histone deacetylase (HDAC) inhibitor entinostat, in combination with sorafenib in patients with advanced solid tumors. Investigational new drugs. 2015; 33: 225-232.
331. Kummar S, Ji J, Morgan R, Lenz HJ, Puhalla SL, Belani CP, Gandara DR, Allen D, Kiesel B, Beumer JH, Newman EM, Rubinstein L, Chen A, et al. A phase I study of veliparib in combination with metronomic cyclophosphamide in adults with refractory solid tumors and lymphomas. Clin Cancer Res. 2012;18:1726-1734.
332. Jayaraman R, Pasha MK, Williams A, Goh KC, Ethirajulu K. Metabolism and Disposition of Pacritinib (SB1518), an Orally Active Janus Kinase 2 Inhibitor in Preclinical Species and Humans. Drug Metab Lett. 2015; 9:28-47.
333. Zhang L, Pham LV, Newberry KJ, Ou Z, Liang R, Qian J, Sun L, Blonska M, You Y, Yang J, Lin X, Rollo A, Tamayo AT, et al. In vitro and in vivo therapeutic efficacy of carfilzomib in mantle cell lymphoma: targeting the immunoproteasome. Molecular cancer therapeutics 2013;12: 2494-2504.

