INTRODUCTION
Ovarian cancer (OvCa) is the most common malignant gynecologic cancer and remains the leading cause of cancer-related death in the western world [1]. In China, there is an increasing trend in the incidence of OvCa [2]. The aggressive surgical debulking followed by platinum-based chemotherapy is known as a standard treatment protocol for OvCa patients [3]. After first line treatment, despite response rates and complete responses were respectively > 80% and 40-60%, most patients eventually face relapse and the majority of them ultimately die of their recurrent disease resulting in an overall five-year survival probability less than 40% for patients with advanced OvCa [4, 5]. Therefore, there is an urgent need for prognostic markers that would be able to predict patients’ poor outcome or chemotherapy resistance, and whether they will benefit from tailored treatment strategies.
Long non-coding RNAs (lncRNAs), are the largest class of non-coding RNAs (ncRNAs) with a length of more than 200 base-pairs [6]. Although most of lncRNAs have not been functionally characterized, there is increasing evidence suggesting that lncRNAs may contribute to a significant layer of genome regulatory information by negatively or positively regulating gene expression at the transcriptional, post-transcriptional and epigenetic levels [7–9]. More and more studies have demonstrated that lncRNAs play important roles in a variety of biological processes [9–11], and their dysregulation and mutation have been implicated in many complex diseases, including cancers [12–18]. Large-scale transcriptional profile analyses have reported the highly aberrant lncRNA expression across multiple human cancer types in the tissue- and cancer type-specific manners [19–22]. Some well-known cancer-associated lncRNAs, such as H19 [23], XIST [24], HOTAIR[25], MALAT1, MEG3 [26], HNF1A-AS1 [27] and PVT1 [28], have indicated oncogenic and/or tumor suppressive roles like protein-coding genes in various cancers. As lncRNAs do not code for proteins, lncRNA expression may be a better indicator of the tumor status, implying the potential and superiority as independent biomarkers for early diagnosis and prognosis prediction in cancers [29]. Currently, several expression-based lncRNA signature have been identified to predict patients’ survival in some cancers, including glioblastoma multiforme [30], oesophageal squamous cell carcinoma [31], colorectal cancer [32], breast cancer [33–35], lung cancer [36] and multiple myeloma [37]. Recent studies have revealed that changes in lncRNA expression were associated with OvCa tumorigenesis and metastasis. For example, lncRNAs UCA1 and HOTAIR are overexpressed in epithelial ovarian cancer (EOC) associated with a worse prognosis [38, 39]. Three lncRNAs (TC0100223, TC0101686 and TC0101441), which showed differential expression between ERα-positive and ERα-negative EOC tissues, were associated with poor prognosis of ERα-positive EOC patients [40]. Some differentially expressed lncRNAs were identified in paired high and low metastatic OvCa cells [41]. A recent study of lncRNA AB073614 showed that patients with higher AB073614 expression had poor overall survival in OvCa [42].
In this study, we performed a comprehensive analysis for lncRNA expression profiles and clinical outcome of a large number of OvCa patients from The Cancer Genome Atlas (TCGA) Research Network to investigate whether lncRNA expression profiling could be used as a prognostic signature for accurately prognosticating clinical outcome and chemotherapy response in patients with OvCa.
RESULTS
Identification of prognostic lncRNAs associated with outcome in patients with OvCa
To detect potential prognostic lncRNAs, we subjected the expression data of each lncRNA in the training cohort to univariate Cox proportional hazards regression analysis with overall survival as the dependent variable. A total of eight lncRNAs were identified as potential prognostic lncRNAs that were significantly correlated with overall survival (p<0.001) (Table 1). Among these prognostic lncRNAs, six lncRNAs having negative coefficients were shown to be protective lncRNAs whose high expression levels were associated with longer survival. The remaining two lncRNAs had positive coefficients and were risky lncRNAs whose high expression levels were associated with shorter survival.
Table 1: The detailed information of eight prognostic lncRNAs significantly associated with overall survival in patients with OvCa
Ensemble ID |
Gene name |
Chromosome (GRCh38) |
P-valuea |
Hazard ratioa |
Coefficienta |
|---|---|---|---|---|---|
ENSG00000236244 |
RP4-799P18.3 |
Chr 1: 234,268,583-234,272,500 (-) |
4.0e-05 |
0.73 |
-0.28 |
ENSG00000225706 |
PTPRD-AS1 |
Chr 9: 8,858,130-8,862,255 (+) |
1.23e-04 |
1.35 |
0.27 |
ENSG00000259331 |
RP11-57P19.1 |
Chr 15: 94,600,014-94,600,821(+) |
4.7e-04 |
0.71 |
-0.21 |
ENSG00000232093 |
RP11-307C12.11 |
Chr 1: 155,045,191-155,046,118(-) |
5.56e-04 |
0.65 |
-0.24 |
ENSG00000250551 |
RP11-254I22.1 |
Chr 5: 96,050,115-96,215,519(+) |
5.81e-04 |
0.61 |
-0.45 |
ENSG00000240996 |
RP11-80H5.7 |
Chr 10: 89,694,295-89,697,928 (-) |
6.15e-04 |
1.32 |
0.25 |
ENSG00000261071 |
RP1-223E5.4 |
Chr 6: 13,614,111-13,615,155 (-) |
7.45e-04 |
0.64 |
-0.30 |
ENSG00000236289 |
GACAT3 |
Chr 2: 16,050,427-16,085,801(+) |
9.32e-04 |
0.48 |
-0.11 |
aDerived from the univariable Cox proportional hazards regression analysis in 263 patients of training cohort.
Acquisition of an eight-lncRNA prognostic signature from the training cohort
To evaluate the relative contribution of these eight prognostic lncRNAs for survival prediction when considering interrelationship among them, we performed a multivariate Cox regression analysis for these eight prognostic lncRNAs with overall survival as a dependent variable. Then, we developed a prognostic model by the risk scoring method as previously described [43, 44] for survival prediction based on the expression levels of these eight lncRNAs and their relative contributions derived from above multivariate analysis as follows: Risk score= (-0.28051 × expression value of RP4-799P18.3) + (0.270976 × expression value of PTPRD-AS1) + (-0.211224× expression value of RP11-57P19.1) + (-0.239482 × expression value of RP11-307C12.11) + (-0.445144 × expression value of RP11-254I22.1) + (0.245869 × expression value of RP11-80H5.7) + (-0.295698 × expression value of RP1-223E5.4) + (-0.114835× expression value of GACAT3). This eight-lncRNA signature-based prognostic model assigned a risk score for each patient. Using the median risk score as the cutoff (-7.203), patients of the training cohort were divided into high-risk group (n=132) and low-risk group (n=131). The patients in the low-risk group were expected to have better survival outcomes. As a result, patients in the low-risk group had significantly longer median overall survival than those in the high-risk group (median 4.85 years versus 2.81 years; p=3.48e-10, log-rank test) (Figure 1A). The overall survival rate of patients at five years in the low-risk group was 46.9%, whereas the corresponding rate in the high-risk group was 15.8%. The univariate analysis revealed a significant association between the risk score and overall survival, in which the hazard ratio (HR) of high-risk group versus low-risk group for overall survival was 3.12 (95% confidence interval (CI) =2.15-4.53; p=2.22e-09) (Table 2). The time-dependent ROC curves analysis for eight-lncRNAs signature-based prognostic model achieved an area under the curve (AUC) of 0.705 at five years (Figure 1B). These results demonstrated better performance of this eight-lncRNA signature in prognosis prediction of patients with OvCa. Figure 1C showed the risk score distribution, survival status and lncRNA expression of 263 patients in the training cohort, ranked according to the risk scores of the eight-lncRNA signature. We found that patients with high-risk scores tended to express two risky lncRNAs, and patients with low-risk scores tended to express six protective lncRNAs (Figure 1C).
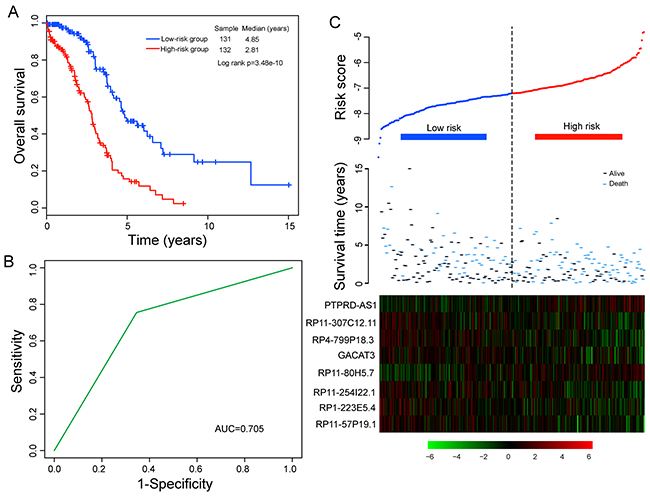
Figure 1: Association between the eight-lncRNA expression signature and overall survival of patients in the training cohort. A. Kaplan-Meier survival curves of overall survival between high-risk and low-risk patients. B. Time-dependent ROC curves analysis for survival prediction by the eight-lncRNA signature within 5 years as the defining point. C. LncRNA risk score analysis of patients in the training cohort.
Table 2: Univariate and multivariate Cox regression analysis of overall survival in each cohort
Variables |
Univariate analysis |
Multivariate analysis |
||||
|---|---|---|---|---|---|---|
HR |
95% CI of HR |
P-value |
HR |
95% CI of HR |
P-value |
|
Training cohort (n=263) |
||||||
Eight-lncRNA signature risk score |
||||||
Low |
1 (reference) |
1 (reference) |
||||
High |
3.12 |
2.15-4.53 |
2.22e-09 |
2.37 |
1.53-3.67 |
1.07e-04 |
Age |
1.03 |
1.01-1.04 |
1.73e-03 |
1.02 |
1.00-1.04 |
4.21e-02 |
Stage |
||||||
II |
1 (reference) |
1 (reference) |
||||
III |
4.33 |
1.37-13.66 |
1.25-e02 |
23481381 |
0.00-Inf |
0.99 |
IV |
5.89 |
1.75-19.82 |
4.18e-03 |
32442060 |
0.00-Inf |
0.99 |
Grade |
||||||
G1/G2 |
1 (reference) |
1 (reference) |
||||
G3/G4 |
1.17 |
0.78-1.77 |
0.45 |
1.33 |
0.83-2.14 |
0.24 |
Residual |
||||||
0-10 mm |
1 (reference) |
1 (reference) |
||||
>10 mm |
0.97 |
0.65-1.45 |
0.88 |
0.76 |
0.49-1.16 |
0.20 |
Validation cohort (n=281) |
||||||
Eight-lncRNA signature risk score |
||||||
Low |
1 (reference) |
1 (reference) |
||||
High |
1.40 |
1.01-1.92 |
4.02e-02 |
1.50 |
1.05-2.15 |
2.71e-02 |
Age |
1.02 |
1.00-1.03 |
2.32e-02 |
1.03 |
1.01-1.04 |
2.98e-03 |
Stage |
||||||
II |
1 (reference) |
1 (reference) |
||||
III |
1.34 |
0.49-3.63 |
0.57 |
1.26 |
0.39-4.02 |
0.70 |
IV |
1.84 |
0.65-5.24 |
0.25 |
1.95 |
0.58-6.56 |
0.28 |
Grade |
||||||
G1/G2 |
1 (reference) |
1 (reference) |
||||
G3/G4 |
1.92 |
1.01-3.67 |
4.69e-02 |
2.66 |
1.21-5.85 |
1.47e-02 |
Residual |
||||||
0-10 mm |
1 (reference) |
1 (reference) |
||||
>10 mm |
1.28 |
0.88-1.86 |
0.21 |
1.10 |
0.74-1.62 |
0.65 |
TCGA cohort (n=544) |
||||||
Eight-lncRNA signature risk score |
||||||
Low |
1 (reference) |
1 (reference) |
||||
High |
2.00 |
1.57-2.55 |
1.71e-08 |
1.81 |
1.38-2.38 |
2.11e-05 |
Age |
1.02 |
1.01-1.03 |
1.17e-04 |
1.02 |
1.01-1.04 |
4.28e-04 |
Stage |
||||||
II |
1 (reference) |
1 (reference) |
||||
III |
2.65 |
1.25-5.62 |
1.13e-02 |
2.27 |
0.72-7.14 |
0.16 |
IV |
3.55 |
1.61-7.83 |
1.70e-03 |
3.35 |
1.03-10.90 |
4.45e-02 |
Grade |
||||||
G1/G2 |
1 (reference) |
1 (reference) |
||||
G3/G4 |
1.39 |
0.998-1.94 |
5.12e-02 |
1.72 |
1.17-2.53 |
6.25e-03 |
Residual |
||||||
0-10 mm |
1 (reference) |
1 (reference) |
||||
>10 mm |
1.07 |
0.82-1.40 |
0.62 |
0.97 |
0.73-1.28 |
0.82 |
Abbreviations: HR, hazard ratio; CI, confidence interval.
In both univariate and multivariate Cox regression analysis, age was evaluated as continuous variable.
Confirmation of the eight-lncRNA signature for survival prediction in the validation cohort and entire TCGA cohort
The prognostic power of the eight-lncRNA signature in survival prediction was further tested in the validation cohort. By using the same risk score model and cutoff value deriving from the training cohort, 281 patients of the validation cohort were classified into either high-risk group (n=144) or low-risk group (n=137). In consistence with our findings in the training cohort, Kaplan-Meier analysis using this eight-lncRNA signature showed significant difference in their overall survival between high-risk group and low-risk group (p=3.93e-02, log-rank test) (Figure 2A). Patients in the low-risk group have a significantly longer median overall survival than those in the high-risk group (median 4.03 years versus 3.42 years). In univariate analysis, the HR of high risk scores versus low-risk scores for overall survival was 1.40 (95% CI=1.01-1.92; p=4.02e-02) (Table 2), demonstrating a significant association between risk scores and patients’ overall survival. The five-year survival rate of the low-risk group was 36%, whereas the corresponding rate in the high-risk group was 25.5%. Validation of the eight-lncRNA signature in the validation cohort of 281 patients produced an ROC with an AUC of 0.555 at five years (see Supplementary Figure S1A online).
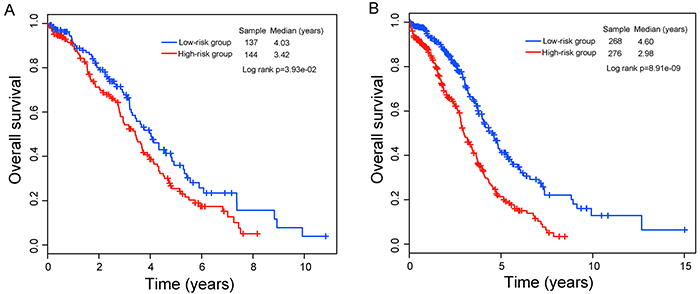
Figure 2: Kaplan–Meier curves analysis of overall survival between high-risk and low-risk patients. A. The validation cohort. B. The entire TCGA cohort.
When this eight-lncRNA signature was further applied to the entire TCGA OvCa cohort (combining the training and validation cohorts), similar risk stratification results were observed. As in the training and validation cohorts, this eight-lncRNA signature was able to stratify 544 patients of the entire TCGA cohort into the high-risk group (n=276) and low-risk group (n=268) with significantly different survival (median 2.98 years versus 4.6 years; p=8.91e-09, log-rank test) (Figure 2B). At five years, the respective absolute difference in overall survival between the low-risk group and high-risk group was 19.9% (41.3% versus 21.4%). The AUC of time-dependent ROC curves for the eight-lncRNA signature in the entire TCGA cohort was 0.609 at five years (see Supplementary Figure S1B online).
The distribution of risk scores, survival status and expression levels of these eight lncRNAs in the testing and entire TCGA cohorts were shown in Supplementary Figure S2 online, which yielded similar results observed in the training cohort.
Independence of prognostic value of the eight-lncRNA signature from other clinical variables
To determine whether the prognostic value of the eight-lncRNA signature is independent of other clinical variables, we performed multivariate Cox regression analysis in each patient cohort including risk scores, age, tumor stage, tumor grade and surgical debulking status as covariables. The results from the training cohort showed that lncRNA signature-based risk scores (HR=2.37, 95% CI=1.53-3.67; p=1.07e-04) and age (HR=1.02, 95% CI=1.0-1.04; p=4.21e-02) were significantly correlated with survival of patients with OvCa. Specifically, the eight-lncRNA signature still maintained a significant correlation with survival when adjusting for age, tumor stage, tumor grade and surgical debulking status in the validation cohort and entire TCGA cohort. The HR of high-risk group versus low-risk group for overall survival was 1.5 in the validation cohort (95% CI=1.05-2.15; p=2.71e-02), as well as in the entire TCGA cohort (HR=1.81, 95% CI=1.38-2.38; p=2.11e-05) when controlling for other clinical variables (Table 2). However, we found that two clinical variables, age and tumor grade, were significantly associated with overall survival in at least two of three patient cohorts. So we further performed data stratification analysis according to age and tumor grade. First, all patients were stratified into a younger stratum (n=268) and an elder stratum (n=276) according to the median age (59 years old). The eight-lncRNA signature could subdivide younger patients into high-risk group (n=126) and low-risk group (n=142). The median overall survival time of patients in the low-risk group was significantly longer than that of patients in the high-risk group (median 4.62 years versus 3.67 years; p=3.99e-04, log-rank test) (Figure 3A). Similarly, among patients within the elder stratum, the same prognostic model was able to separate patients into two risk subgroups with significantly different survival (median 2.76 years versus 3.98 years; p=2.46e-05, log-rank test) (Figure 3B). Then all patients were further stratified into low-grade (G1/G2) and high-grade subgroups (G3/G4) according to tumor grade. The results of stratified analysis showed effective prognostic power both in the low-grade and in the high-grade patient subgroups. The patients with high-grade were divided into either a high-risk group (n=225) with shorter survival or a low-risk group (n=235) with longer survival (median 2.83 years versus 4.15 years; p=9.04e-08) (Figure 3C). Similar results were observed for low-grade patient subgroup, in which patients were classified into two risk subgroups with marginally significantly different survival time (median 3.97 years versus 6.24 years; p=0.101, log-rank test) (Figure 3D).
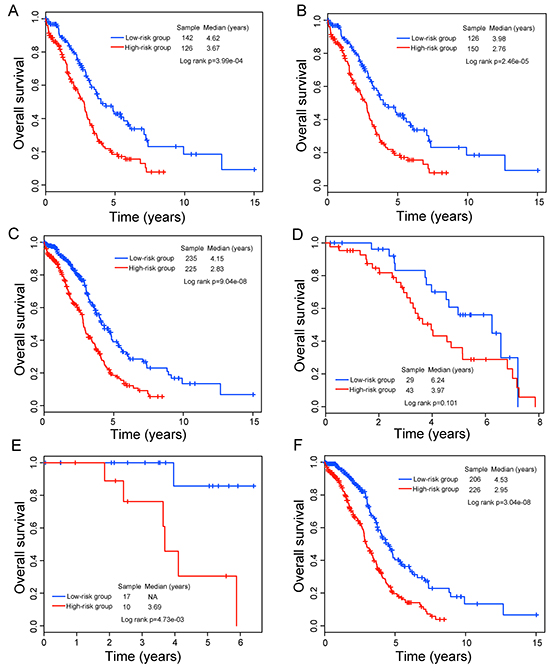
Figure 3: Survival prediction in TCGA patients stratified by age, grade and stage. A. Kaplan-Meier curves for younger TCGA patients. B. Kaplan-Meier curves for elder TCGA patients. C. Kaplan–Meier curves for high-grade TCGA patients. D. Kaplan–Meier curves for low-grade TCGA patients. E. Kaplan-Meier curves for stage II TCGA patients. F. Kaplan–Meier curves for stage III TCGA patients.
Risk evaluation for newly diagnosed patients at the early tumor stage will improve adjuvant treatment decisions making it possible to identify high-risk patients who might benefit from adjuvant therapy. Therefore, separate validation of this eight-lncRNA signature in survival prediction was performed for stage II and III patients. Within each stage stratum, patients were classified as high-risk and low risk according to the same prognostic model and risk score cutoff. The Kaplan-Meier analysis showed that patients with high-risk scores tended to have shorter survival than those with low-risk scores (median 3.69 years versus >5 years; p=4.73e-03, log-rank test for stage II patients and median 2.95 years versus 4.53 years; p=3.04e-08, log-rank test for stage III patients) (Figure 3E and 3F). Taken together, the results of multivariate analysis and stratification analysis demonstrated that the prognostic value of the eight-lncRNA signature is independent of other clinical variables for survival prediction of patients with OvCa.
Ability of the eight-lncRNA signature to discriminate BRCA1/2-mutated or BRCA 1/2 wild-type tumors
Previous studies have suggested that BRCA1 and BRCA2 mutations are associated with clinical outcome of OvCa patients, and patients harboring BRCA1 and/or BRCA2 mutation (hereafter inferred as BRCA1/2 mutation) subjected to platinum-based treatment have an improved clinical outcome compared with BRCA 1/2 wild-type patients. Therefore, we further assessed the prognostic value of the eight-lncRNA signature for the patients with or without BRCA1/2 mutation by stratification analysis, which stratified patients into BRCA1/2-mutated group and BRCA 1/2 wild-type group. Using the same score formula, we classified patients in BRCA 1/2 wild-type group into a high-risk group (n=241) and a low-risk group (n=238) using the same cutoff as in the training set. Patients in the low-risk group had significantly longer survival time than those in the high-risk group (median 4.09 years versus 2.98 years; p=2.44e-06; log-rank test) (Figure 4A). Then we performed pairwise comparisons of overall survival between lncRNA-related high-risk BRCA 1/2 wild-type group, lncRNA-related low-risk BRCA 1/2 wild-type group and BRCA1/2-mutated group. This analysis showed that patients in the lncRNA-related high-risk BRCA 1/2 wild-type group had significantly shorter survival time than did patients in the BRCA1/2-mutated group (median 2.98 years versus 6.08 years; p=1.19e-05) and lncRNA-related low-risk BRCA 1/2 wild-type group (median 2.98 years versus 4.09 years; p=2.44e-06, log-rank test) (Figure 4B). However, BRCA1/2-mutated patients only showed marginally significant difference in clinical outcome from patients in the lncRNA-related low-risk BRCA 1/2 wild-type group (median 6.08 versus 4.09 years; p=4.79e-02). These results suggested that although BRCA 1/2 wild-type patients did not harbor BRCA1/2 mutation, a substantial subset of BRCA 1/2 wild-type patients with lncRNA-related low-risk scores still had a good prognosis and could benefit platinum-based chemotherapy.
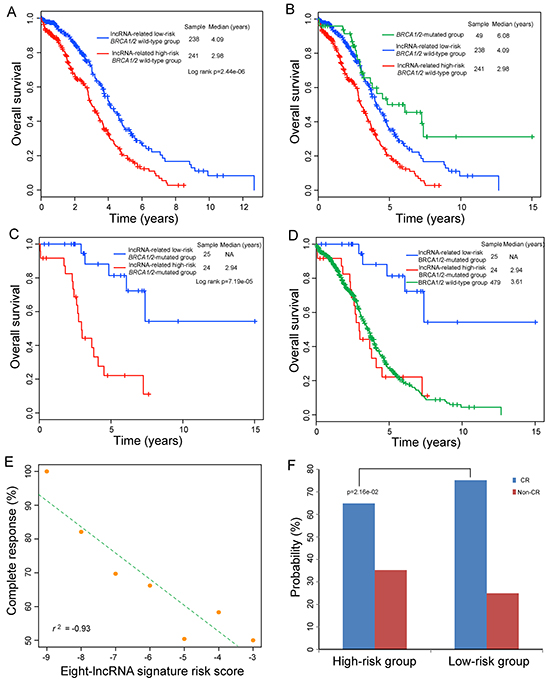
Figure 4: Relationship between the eight-lncRNA expression signature and clinical outcome in BRCA1/2-mutated or BRCA 1/2 wild-type OvCa tumors. A. Kaplan-Meier curves analysis of overall survival between lncRNA-related high-risk and low-risk BRCA 1/2 wild-type patients. B. Differences in overall survival were assessed among the three groups. The log-rank p value of overall survival for the lncRNA-related high-risk BRCA1/2 wild-type group versus BRCA1/2-mutated group is 1.19e-05 and the log-rank p value of overall survival for the lncRNA-related BRCA1/2 wild-type low-risk group versus BRCA1/2-mutated group is 4.79e-02. C. Kaplan-Meier curves analysis of overall survival between lncRNA-related high-risk and low-risk BRCA1/2-mutated patients. D. Differences in overall survival were assessed among the three groups. The log-rank p value of overall survival for the lncRNA-related low-risk BRCA1/2-mutated group versus BRCA 1/2 wild-type group is 3.28e-06 and the log-rank p value of overall survival for the lncRNA-related high-risk BRCA1/2-mutated group versus BRCA 1/2 wild-type group is 0.749. E. Correlation of the eight-lncRNA signature with complete response. The Pearson correlation coefficient was calculated to assess the relationship between the eight-lncRNA signature and the likelihood of complete response. The straight line depicts the least squares linear regression line through the data points. F. Differences in complete response ratios between high-risk group and low-risk group.
Then forty-nine OvCa patients with BRCA1 mutation and/or with BRCA2 mutation were stratified into the BRCA1/2-mutated group. We found that patients of BRCA1/2-mutated group also could be separated into those likely to have good outcome and those likely to have poor outcome according to the risk scores of the eight-lncRNA signature. The survival time of BRCA1/2-mutated patients in the high-risk group (n=24) was significantly lower than that of BRCA1/2-mutated patients in the low-risk group (n=25) (median 2.94 years >7.38 years; p=7.19e-05, log-rank test) (Figure 4C). Furthermore, BRCA1/2-mutated patients in the low-risk group had significantly longer survival time than the wild-type patients (median >7.38 years versus 3.61 years; p=3.28e-06, log-rank test), whereas those in the high-risk group showed no significant difference in clinical outcome from the wild-type patients (median 2.94 years versus 3.61 years; p=0.749, log-rank test) (Figure 4D), which supported previous finding that not all BRCA-mutated patients exhibit favorable clinical outcome [45].
Based on above observations, we further explored whether there was an association between risk score of the eight-lncRNA signature and the likelihood of complete response (CR). 437 OvCa patients with CR or Non-CR information were analyzed. We first plotted the percentage of OvCa patients achieving CR as a function of the risk score, and observed that the probability of OvCa patients achieving CR was significantly correlated with risk score of this eight-lncRNA signature (Pearson correlation coefficient r2=-0.93, p=2.69e-03) (Figure 4E). Patients with low-risk scores tended to have high likelihood of CR and those with high-risk scores had low likelihood of CR. In detail, 75.11% of patients in the low-risk group achieved CR, whereas 64.81% of patients in the high-risk group achieved CR (p=2.16e-02, Fisher exact test) (Figure 4F), implying that the risk score of the eight-lncRNA signature had the potential to reflect the sensitivity to platinum therapy. Therefore, these results suggested that BRCA-mutated patients with lncRNA-related high-risk scores may be platinum resistant and need individual appropriate treatment strategies except for platinum-based chemotherapy. This is consistent with previous studies showing nearly 40% of BRCA-mutated OvCa tumors exhibited platinum resistance and experienced multiple cycles of non-beneficial toxic chemotherapy leading to a poor clinical outcome [46, 47].
Functional characteristics of prognostic lncRNAs
We first obtained genome-wide protein-coding gene (PCG) expression profiles in 544 OvCa patients from Du’s study [48], including 18292 PCGs. lncRNAs do not encode proteins and their functions are known to be associated with co-expressed PCGs [49, 50]. Therefore, we examined the expression correlation between lncRNAs and PCGs using paired lncRNA and PCG expression profiles in 544 OvCa patients. Only those PCGs with a Pearson correlation coefficients ranked in the top 1% for each prognostic lncRNA were considered as lncRNA-correlated PCGs. To explore potential functional roles of these eight prognostic lncRNAs in OvCa development and progression, we performed functional enrichment analysis of lncRNA-correlated PCGs. The results from GO enrichment analysis revealed that lncRNA-correlated PCGs were significantly enriched in 87 GO terms, in which functionally related GO terms could be organized into six functional clusters including cyclic nucleotide metabolic process, extracellular matrix (ECM) organization, calcium ion homeostasis, cell migration, MAPK activity and GPI anchor biosynthetic process (Figure 5A). KEGG pathway enrichment analysis of lncRNA-correlated PCGs showed that four KEGG pathways, including ECM-receptor interaction, Focal adhesion, GPI-anchor biosynthesis and Calcium signaling pathway were significantly enriched (Figure 5B). These enriched GO function and KEGG pathway of lncRNA-correlated PCGs have been reported to be involved with OvCa through our literature review. The functional analysis of lncRNAs suggested that these prognostic lncRNAs might participate in OvCa tumorigenesis through positively regulating lncRNA-related PCGs to affect known OvCa-related biological pathways.
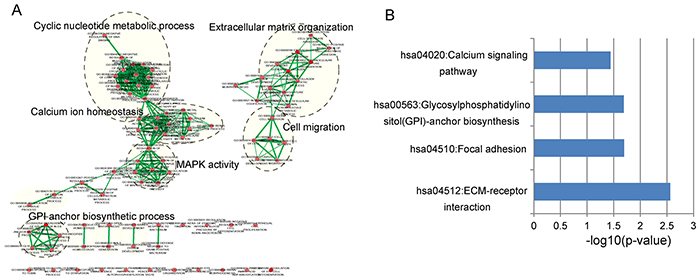
Figure 5: Functional enrichment analysis of protein-coding gene co-expressed with eight prognostic lncRNAs. A. The functional map of enriched GO terms with each node indicates an enriched GO term and each edge represents the common genes shared between connecting enriched GO terms. B. Significantly enriched KEGG pathways.
DISCUSSION
In terms of the heterogeneity of clinical outcomes in OvCa patients, there is a critical need for reliable prognostic factors pinpointing a subset of patients with poor prognosis who would therefore benefit from additional treatment options. However, traditional clinicopathological prognostic markers, such as age, stage, debulking status after primary surgery and response to chemotherapy are not satisfactory for prognosis prediction and treatment decisions of individual patient. Recent large-scale genomic analyses have revealed a catalogue of molecular characteristics associated with OvCa outcome and therapeutic treatment [51]. Molecular expression profiles have been used to identify outcome-related molecular signatures which provided important implication for prognosis prediction and molecular mechanism of OvCa [52–58]. However, most existing studies have focused on mRNA and microRNA expression. Knowledge is now rapidly emerging on the functional roles of lncRNAs in cancer initiation and progression, representing a significant untapped molecular resource for cancer prognosis and treatment.
In this study, we conducted a comprehensive analysis of lncRNA expression profiles in a large number of OvCa patients from TCGA, and identified an eight-lncRNA signature predictive of overall survival using the sample-splitting method and Cox regression analysis. We found that six lncRNAs were correlated with good survival and two lncRNAs were correlated with poor survival. As previously described [59, 60], three methods (independent dataset test, subsampling test and jackknife test) were widely used to examine the accuracy of predictive model. Therefore, we validated the predictive power of this eight-lncRNA signature on an independent and non-overlapping set of 281 TCGA patients as well as on the entire TCGA cohort. Furthermore, the eight-lncRNA signature is independent of other clinicopathological factors, including age, stage, grade and surgical debulking status. It is well known that younger women with OvCa show better survival rates than older women. Statistics showed that younger women with OvCa were treated more aggressively than older women, and most of younger patients received chemotherapy [61]. Although treatment combination of surgery plus chemotherapy increases the survival rates for younger patients, chemotherapy is accompanied by cumulative side effects that affect patients’ life quality. Therefore, further stratification of younger women with OvCa who might or might not benefit from adjuvant chemotherapy is urgently needed. In the stratified analysis, the eight-lncRNA signature showed prognostic value both in younger and older patients. The eight-lncRNA signature could subdivide patients in the same age stratum into high-risk and low-risk groups with significantly different clinical outcome.
Previous studies have suggested that homologous recombination (HR) deficiency can bring about error-prone repair of DNA double-strand breaks (DSBs) and are used to identify platinum-sensitive tumors [47, 62]. However, defining HR deficiency is still a challenge. It is suggested that BRCA1 and BRCA2 mutations are associated with HR deficiency in OvCa, therefore OvCa patients harboring BRCA1/2 mutations subjected to platinum-based treatment have favorable outcomes compared with BRCA 1/2 wild-type patients. Recent studies found that some of BRCA 1/2 wild-type patients in OvCa may also harbor HR deficiency and have significantly improved survival when subjected to platinum-based treatment. Therefore, we applied this signature to test whether this eight-lncRNA signature was able to identify those BRCA1/2 wild-type patients who will benefit from platinum-based chemotherapy. Survival analysis of 479 BRCA1/2 wild-type patients demonstrated that this eight-lncRNA was able to identify a subgroup of patients with wild-type BRCA1/2 with a remarkably good clinical outcome. Further analysis found that even for BRCA-mutated patients, this eight-lncRNA signature also revealed a prognostic value which stratified BRCA-mutated patients into cases of significantly improved outcome and cases of poor outcome. This is consistent with recently published results showing a fraction of BRCA-deficient tumors were resistant to chemotherapy [63, 64]. With further validation, a closely association between the risk score of eight-lncRNA signature and CR was observed, implying that this eight-lncRNA signature may be a measure to predict chemotherapy response and be used to identify platinum-resistant patients who might benefit from other more efficacious therapies.
Until now, more than tens of thousands of lncRNAs have been discovered and recorded in several publicly biological databases, such as GENCODE [65], NONCODE [66] and LNCipedia [67]. However, only a very few lncRNAs were well functionally characterized. By reviewing literatures, only one of eight prognostic lncRNAs, GACAT3, has been reported to play crucial roles in the gastric carcinoma [68]. Previous studies have suggested that lncRNAs participated in biological processes by interacting with PCGs involved in the same processes, making it possible to infer lncRNA function from their co-expressed PCGs [49, 69, 70]. Therefore, we performed functional enrichment analysis for co-expressed PCGs to uncover potential biological processes lncRNAs involved in. We found that these eight prognostic lncRNAs might participate in several biological processes, including ECM-receptor interaction, Focal adhesion, MAPK activity, GPI-anchor biosynthesis and Calcium signaling pathway. Proteomic analyses have demonstrated that aberrant expression of key members in ECM and Focal adhesion pathways are associated with invasive behavior by ovarian cancer cells [71]. MAPK pathways can control fundamental cellular processes by linking extracellular signals to the machinery, and distinct groups of MAPK pathways have been widely studied, revealing important roles of MAPK pathways in cancers, including OvCa [72, 73]. Glycosylphosphatidylinositol (GPI) anchor is an unique type of glycoconjugate, and abnormal expression levels of certain components in the GPI-anchor biosynthetic pathway have been reported to be associated with various cancers [74]. A recent experimental study provided evidence for the role of calcium-related genes in mediating cisplatin resistance in ovarian cancer cells [75]. Taken together, these analyses suggested that the eight prognostic lncRNAs might have important biologic relevance in regulating or interacting PCGs involved in OvCa, but further experimentally validation is required. As demonstrated in previous studies [59, 76, 77], we shall make efforts in our future work to provide a publicly accessible web-server for our predictive model.
In conclusion, we succeeded in identifying and validating an eight-lncRNA signature for prognosis prediction in OvCa patients by performing genome-wide analysis of lncRNA expression profiles in a large cohort of TCGA patients, which was able to classify patients into high-risk group showing poor outcome and low-risk group showing significantly improved outcome. The eight-lncRNA signature maintained independent prognostic value in multivariate and stratified analysis, controlling for other known prognostic factors such as age, stage, grade, debulking status and BRCA1/2 mutation status. Moreover, the lncRNA signature was significantly correlated with the response to chemotherapy. To our knowledge, the lncRNA expression profiles and OvCa patients derived from TCGA are unprecedented in comprehensiveness and in size, and there is no other available independent datasets to validate our findings. When our study was in progress, two novel immune-associated lncRNAs (RP11-284N8.3.1 and AC104699.1.1) were identified to predict survival of patients with different OvCa stages by using lncRNA-mRNA co-expression network methods [78]. Our study, taken together with Guo’s study, highlighted important implications of lncRNAs as novel biomarkers for outcome prediction and therapy decisions.
MATERIALS AND METHODS
Patient dataset
544 patients with serous ovarian carcinoma (stages II, III and IV) and their related clinical information were obtained from TCGA data portal (https://tcga-data.nci.nih.gov/tcga/). The TCGA OvCa patient cohort was divided into a training cohort (batches 18-40) and a validation cohort (batches 9-17), which results in a 263-sample training cohort and a 281-sample validation cohort. Detailed clinical information of OvCa patients enrolled in this study, including age, tumor stage, tumor grade, response to chemotherapy, and surgical debulking, was listed in Table 3 and Supplementary Table S1. The somatic and germline mutation information of BRCA1 and BRCA2 genes from whole exome sequencing was downloaded from the cBioPortal Cancer Genomics (http://www.cbioportal.org/) [79].
Table 3: Tumor characteristics of ovarian cancer patients in this study
Characteristic |
Training cohort |
Validation cohort |
TCGA cohort |
|---|---|---|---|
Age |
59.02±11.77 |
60.11±11.29 |
59.58±11.53 |
Vital status |
|||
Alive |
136 |
120 |
256 |
Dead |
127 |
161 |
288 |
Stagea |
|||
II |
19 |
8 |
27 |
III |
210 |
222 |
432 |
IV |
34 |
51 |
85 |
Gradeb |
|||
G1-G2 |
55 |
17 |
72 |
G3-G4 |
201 |
259 |
460 |
Response to therapy |
|||
CR |
138 |
168 |
306 |
Non-CR |
63 |
68 |
131 |
Residual tumor size |
|||
0-10 mm |
99 |
147 |
246 |
>10 mm |
79 |
60 |
139 |
Numbers do not sum due to the back of data of interest.
aStage based on International Federation of Gynecology and Obstetrics (FIGO)
bGrade based on histological features.
cCR depicts Complete Response and Non-CR depicts non-complete response, including partial response, stable disease and progressive disease.
Acquisition of lncRNA expression profiles of OvCa patients
Genome-wide lncRNA expression profiles of patients with OvCa were obtained from Du’s study by repurposing the probes from Affymetrix Human Exon 1.0 ST microarray [48]. Briefly, the probe sets of Affymetrix Human Exon 1.0 ST microarray were re-mapped to human genome (hg 19), protein-coding transcripts, pseudogene transcripts and lncRNA sequences. Those probes that mapped to lncRNA sequences uniquely and perfectly were kept to represent lncRNAs. The lncRNA expression levels were obtained by calculating the background-corrected intensity of all probes mapped to this lncRNA. To account for the heterogeneity of different biological samples and different batches in systematic measurement, the expression value of lncRNA was standardized using quantile-normalized method and an empirical Bayes method [80]. Then the lncRNAs derived from microarray re-annotation and lncRNAs from GENCODE project (http://www.gencodegenes.org/, release 23) [65] were cross-reference by Ensembl id and gene name to reduce redundant and inaccurate annotations. Finally, we obtained expression profiles of 7952 lncRNAs in 544 OvCa patients.
Construction of lncRNA-based prognostic signature
A univariate Cox regression analysis was performed to examine the association between expression levels of lncRNAs and patients’ overall survival in the training cohort. Those lncRNAs with p-value <0.001 were selected as predictive lncRNAs whose expression levels were significantly associated with patients’ overall survival. In order to evaluate relative contribution of predictive lncRNAs for survival prediction, they were fitted in a multivariate Cox regression analysis with overall survival as the dependent variable. A lncRNA expression-based prognostic risk score model was constructed by the linear combination of the expression levels of predictive lncRNAs with the multivariate Cox regression coefficient as the weight. This lncRNA prognostic model could calculate an lncRNA expression-based risk score for each patient and classify patients into high-risk group and low-risk group using the median risk score from the training cohort.
Statistical analysis
Kaplan-Meier survival curves were used to estimate overall survival time for patients with predicted high- or low-risk scores, and the survival differences between high-risk group and low-risk group were assessed by a two-sided log-rank test using the R package “survival” [81]. Multivariate analyses were performed using Cox proportional hazards regression model to determine whether the lncRNA prognostic model was independent of other clinical variables, adjusting for age, tumor stage, grade, surgical debulking status, and risk scores. Hazard ratio (HR) and 95% confidence intervals (CI) were estimated by Cox proportional hazards regression model. The time-dependent receiver operating characteristic (ROC) curve analysis within 5 years as the defining point was preformed using the R package “survivalROC” [82], which has been widely used to assess the predictive accuracy of prognostic model or markers for time dependent disease outcomes [83]. All statistical analyses were performed using R software and Bioconductor.
Functional enrichment analysis
Functional enrichment analysis at the Gene ontology (GO) and Kyoto encyclopedia of genes and genomes (KEGG) pathway levels were conducted to infer lncRNA function using the DAVID Bioinformatics Tool (https://david.ncifcrf.gov/, version 6.7) [84, 85], a widely used functional annotation tool that can extract the major biological significance among a gene set of interest. The results of enrichment analysis were obtained limited to GO terms in the “Biological Process” (GOTERM-BP-FAT) and KEGG pathway categories using the functional annotation clustering and functional annotation chart options with the human whole genome as background. The enriched GO terms and KEGG pathway with p-value <0.05 and enrichment score >1.0 were considered as potential function of prognostic lncRNAs. Significant GO terms with similar function were visualized as interaction networks using the Enrichment Map plugin in Cytoscape [86].
ACKNOWLEDGMENTS
This work was supported by the National Natural Science Foundation of China (Grant No. 61403111), China Postdoctoral Science Foundation (Grant No. 2014M551268) and Postdoctoral Foundation of Heilongjiang Province (Grant No. LBH-Z14212).
CONFLICTS OF INTEREST
The authors declare that they have no of interest.
REFERENCES
1. Siegel R, Ma J, Zou Z and Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014; 64:9-29.
2. Wang B, Liu S-Z, Zheng R-S, Zhang F, Chen W-Q and Sun X-B. Time trends of ovarian cancer incidence in China. Asian Pacific journal of cancer prevention. 2013; 15:191-193.
3. Du Bois A, Quinn M, Thigpen T, Vermorken J, Avall-Lundqvist E, Bookman M, Bowtell D, Brady M, Casado A and Cervantes A. 2004 consensus statements on the management of ovarian cancer: final document of the 3rd International Gynecologic Cancer Intergroup Ovarian Cancer Consensus Conference (GCIG OCCC 2004). Annals of oncology. 2005; 16:viii7.
4. Kim A, Ueda Y, Naka T and Enomoto T. Therapeutic strategies in epithelial ovarian cancer. J Exp Clin Cancer Res. 2012; 31:14.
5. Coleman M, Forman D, Bryant H, Butler J, Rachet B, Maringe C, Nur U, Tracey E, Coory M and Hatcher J. Cancer survival in Australia, Canada, Denmark, Norway, Sweden, and the UK, 1995–2007 (the International Cancer Benchmarking Partnership): an analysis of population-based cancer registry data. The Lancet. 2011; 377:127-138.
6. Rinn JL and Chang HY. Genome regulation by long noncoding RNAs. Annu Rev Biochem. 2012; 81:145-166.
7. Kornienko AE, Guenzl PM, Barlow DP and Pauler FM. Gene regulation by the act of long non-coding RNA transcription. BMC biology. 2013; 11:59.
8. Cao J. The functional role of long non-coding RNAs and epigenetics. Biol Proced Online. 2014; 16:11.
9. Mercer TR, Dinger ME and Mattick JS. Long non-coding RNAs: insights into functions. Nature Reviews Genetics. 2009; 10:155-159.
10. Fatica A and Bozzoni I. Long non-coding RNAs: new players in cell differentiation and development. Nature Reviews Genetics. 2014; 15:7-21.
11. Moran VA, Perera RJ and Khalil AM. Emerging functional and mechanistic paradigms of mammalian long non-coding RNAs. Nucleic acids research. 2012; 40:6391-6400.
12. Gibb EA, Brown CJ and Lam WL. The functional role of long non-coding RNA in human carcinomas. Mol Cancer. 2011; 10:38-55.
13. Cheetham S, Gruhl F, Mattick J and Dinger M. Long noncoding RNAs and the genetics of cancer. British journal of cancer. 2013; 108:2419-2425.
14. Yang G, Lu X and Yuan L. LncRNA: a link between RNA and cancer. Biochimica et Biophysica Acta (BBA)-Gene Regulatory Mechanisms. 2014; 1839:1097-1109.
15. Wu D, Huang Y, Kang J, Li K, Bi X, Zhang T, Jin N, Hu Y, Tan P and Zhang L. ncRDeathDB: A comprehensive bioinformatics resource for deciphering network organization of the ncRNA-mediated cell death system. Autophagy. 2015; 11:1917-1926.
16. Li Y, Zhuang L, Wang Y, Hu Y, Wu Y, Wang D and Xu J. Connect the dots: a systems level approach for analyzing the miRNA-mediated cell death network. Autophagy. 2013; 9:436-439.
17. Wang Y, Chen L, Chen B, Li X, Kang J, Fan K, Hu Y, Xu J, Yi L and Yang J. Mammalian ncRNA-disease repository: a global view of ncRNA-mediated disease network. Cell Death Dis. 2013; 4:e765.
18. Spizzo R, Almeida MI, Colombatti A and Calin GA. Long non-coding RNAs and cancer: a new frontier of translational research&quest. Oncogene. 2012; 31:4577-4587.
19. Gibb EA, Vucic EA, Enfield K, Stewart GL, Lonergan KM, Kennett JY, Becker-Santos DD, MacAulay CE, Lam S and Brown CJ. Human cancer long non-coding RNA transcriptomes. PloS one. 2011; 6:e25915.
20. White NM, Cabanski CR, Silva-Fisher JM, Dang HX, Govindan R and Maher CA. Transcriptome sequencing reveals altered long intergenic non-coding RNAs in lung cancer. Genome Biol. 2014; 15:429.
21. Cabili MN, Trapnell C, Goff L, Koziol M, Tazon-Vega B, Regev A and Rinn JL. Integrative annotation of human large intergenic noncoding RNAs reveals global properties and specific subclasses. Genes & development. 2011; 25:1915-1927.
22. Tørring PM, Larsen MJ, Kjeldsen AD, Ousager LB, Tan Q and Brusgaard K. Long non-coding RNA expression profiles in hereditary haemorrhagic telangiectasia. PloS one. 2014; 9:e90272.
23. Luo M, Li Z, Wang W, Zeng Y, Liu Z and Qiu J. Long non-coding RNA H19 increases bladder cancer metastasis by associating with EZH2 and inhibiting E-cadherin expression. Cancer letters. 2013; 333:213-221.
24. Duret L, Chureau C, Samain S, Weissenbach J and Avner P. The Xist RNA gene evolved in eutherians by pseudogenization of a protein-coding gene. Science. 2006; 312:1653-1655.
25. Gupta RA, Shah N, Wang KC, Kim J, Horlings HM, Wong DJ, Tsai M-C, Hung T, Argani P and Rinn JL. Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature. 2010; 464:1071-1076.
26. Braconi C, Kogure T, Valeri N, Huang N, Nuovo G, Costinean S, Negrini M, Miotto E, Croce C and Patel T. microRNA-29 can regulate expression of the long non-coding RNA gene MEG3 in hepatocellular cancer. Oncogene. 2011; 30:4750-4756.
27. Wu Y, Liu H, Shi X, Yao Y, Yang W and Song Y. The long non-coding RNA HNF1A-AS1 regulates proliferation and metastasis in lung adenocarcinoma. Oncotarget. 2015; 6:9160. doi: 10.18632/oncotarget.3247.
28. Colombo T, Farina L, Macino G and Paci P. PVT1: a rising star among oncogenic long noncoding RNAs. Biomed Res Int. 2015; 2015:304208.
29. Hauptman N and Glavač D. Long non-coding RNA in cancer. International journal of molecular sciences. 2013; 14:4655-4669.
30. Zhang X-Q, Sun S, Lam K-F, Kiang KM-Y, Pu JK-S, Ho AS-W, Lui W-M, Fung C-F, Wong T-S and Leung GK-K. A long non-coding RNA signature in glioblastoma multiforme predicts survival. Neurobiology of disease. 2013; 58:123-131.
31. Li J, Chen Z, Tian L, Zhou C, He MY, Gao Y, Wang S, Zhou F, Shi S and Feng X. LncRNA profile study reveals a three-lncRNA signature associated with the survival of patients with oesophageal squamous cell carcinoma. Gut. 2014; 63:1700-1710.
32. Hu Y, Chen H-Y, Yu C-Y, Xu J, Wang J-L, Qian J, Zhang X and Fang J-Y. A long non-coding RNA signature to improve prognosis prediction of colorectal cancer. Oncotarget. 2014; 5:2230. doi: 10.18632/oncotarget.1895.
33. Meng J, Li P, Zhang Q, Yang Z and Fu S. A four-long non-coding RNA signature in predicting breast cancer survival. J Exp Clin Cancer Res. 2014; 33:84.
34. Sun J, Chen X, Wang Z, Guo M, Shi H, Wang X, Cheng L and Zhou M. A potential prognostic long non-coding RNA signature to predict metastasis-free survival of breast cancer patients. Sci Rep. 2015; 5:16553.
35. Liu H, Li J, Koirala P, Ding X, Chen B, Wang Y, Wang Z, Wang C, Zhang X and Mo YY. Long non-coding RNAs as prognostic markers in human breast cancer. Oncotarget. 2016. doi: 10.18632/oncotarget.7828.
36. Zhou M, Guo M, He D, Wang X, Cui Y, Yang H, Hao D and Sun J. A potential signature of eight long non-coding RNAs predicts survival in patients with non-small cell lung cancer. Journal of translational medicine. 2015; 13:1-13.
37. Zhou M, Zhao H, Wang Z, Cheng L, Yang L, Shi H, Yang H and Sun J. Identification and validation of potential prognostic lncRNA biomarkers for predicting survival in patients with multiple myeloma. Journal of Experimental & Clinical Cancer Research. 2015; 34:1.
38. Lawrenson K, Spindler T and Gayther SA. A novel long non-coding RNA associated with poor prognosis in epithelial ovarian cancer. Cancer Research. 2014; 74:1497-1497.
39. Qiu J-j, Lin Y-y, Ye L-c, Ding J-x, Feng W-w, Jin H-y, Zhang Y, Li Q and Hua K-q. Overexpression of long non-coding RNA HOTAIR predicts poor patient prognosis and promotes tumor metastasis in epithelial ovarian cancer. Gynecologic oncology. 2014; 134:121-128.
40. Qiu J-J, Ye L-C, Ding J-X, Feng W-W, Jin HY, Zhang Y, Li Q and Hua K-Q. Expression and clinical significance of estrogen-regulated long non-coding RNAs in estrogen receptor α-positive ovarian cancer progression. Oncology reports. 2014; 31:1613-1622.
41. Liu SP, Yang JX, Cao DY and Shen K. Identification of differentially expressed long non-coding RNAs in human ovarian cancer cells with different metastatic potentials. Cancer Biol Med. 2013; 10:138-141.
42. Cheng Z, Guo J, Chen L, Luo N, Yang W and Qu X. A long noncoding RNA AB073614 promotes tumorigenesis and predicts poor prognosis in ovarian cancer. Oncotarget. 2015; 6:25381. doi: 10.18632/oncotarget.4541.
43. Lossos IS, Czerwinski DK, Alizadeh AA, Wechser MA, Tibshirani R, Botstein D and Levy R. Prediction of survival in diffuse large-B-cell lymphoma based on the expression of six genes. New England Journal of Medicine. 2004; 350:1828-1837.
44. Alizadeh AA, Gentles AJ, Alencar AJ, Liu CL, Kohrt HE, Houot R, Goldstein MJ, Zhao S, Natkunam Y and Advani RH. Prediction of survival in diffuse large B-cell lymphoma based on the expression of 2 genes reflecting tumor and microenvironment. Blood. 2011; 118:1350-1358.
45. Yang D, Khan S, Sun Y, Hess K, Shmulevich I, Sood AK and Zhang W. Association of BRCA1 and BRCA2 mutations with survival, chemotherapy sensitivity, and gene mutator phenotype in patients with ovarian cancer. Jama. 2011; 306:1557-1565.
46. Pignata S, Cannella L, Leopardo D, Pisano C, Bruni GS and Facchini G. Chemotherapy in epithelial ovarian cancer. Cancer letters. 2011; 303:73-83.
47. Kang J, D’Andrea AD and Kozono D. A DNA repair pathway–focused score for prediction of outcomes in ovarian cancer treated with platinum-based chemotherapy. Journal of the National Cancer Institute. 2012; 104:670-681.
48. Du Z, Fei T, Verhaak RG, Su Z, Zhang Y, Brown M, Chen Y and Liu XS. Integrative genomic analyses reveal clinically relevant long noncoding RNAs in human cancer. Nature structural & molecular biology. 2013; 20:908-913.
49. Liao Q, Liu C, Yuan X, Kang S, Miao R, Xiao H, Zhao G, Luo H, Bu D and Zhao H. Large-scale prediction of long non-coding RNA functions in a coding–non-coding gene co-expression network. Nucleic acids research. 2011; 39:3864-3878.
50. Paci P, Colombo T and Farina L. Computational analysis identifies a sponge interaction network between long non-coding RNAs and messenger RNAs in human breast cancer. BMC systems biology. 2014; 8:83.
51. Network CGAR. Integrated genomic analyses of ovarian carcinoma. Nature. 2011; 474:609-615.
52. Bonome T, Levine DA, Shih J, Randonovich M, Pise-Masison CA, Bogomolniy F, Ozbun L, Brady J, Barrett JC and Boyd J. A gene signature predicting for survival in suboptimally debulked patients with ovarian cancer. Cancer research. 2008; 68:5478-5486.
53. Spentzos D, Levine DA, Ramoni MF, Joseph M, Gu X, Boyd J, Libermann TA and Cannistra SA. Gene expression signature with independent prognostic significance in epithelial ovarian cancer. Journal of Clinical Oncology. 2004; 22:4700-4710.
54. Yoshihara K, Tajima A, Yahata T, Kodama S, Fujiwara H, Suzuki M, Onishi Y, Hatae M, Sueyoshi K and Fujiwara H. Gene expression profile for predicting survival in advanced-stage serous ovarian cancer across two independent datasets. PloS one. 2010; 5:e9615.
55. Verhaak RG, Tamayo P, Yang J-Y, Hubbard D, Zhang H, Creighton CJ, Fereday S, Lawrence M, Carter SL and Mermel CH. Prognostically relevant gene signatures of high-grade serous ovarian carcinoma. The Journal of clinical investigation. 2013; 123:517.
56. Gu Y, Zhang M, Peng F, Fang L, Zhang Y, Liang H, Zhou W, Ao L and Guo Z. The BRCA1/2-directed miRNA signature predicts a good prognosis in ovarian cancer patients with wild-type BRCA1/2. Oncotarget. 2015; 6:2397. doi: 10.18632/oncotarget.2963.
57. Jin N, Wu H, Miao Z, Huang Y, Hu Y, Bi X, Wu D, Qian K, Wang L and Wang C. Network-based survival-associated module biomarker and its crosstalk with cell death genes in ovarian cancer. Sci Rep. 2015; 5:11566.
58. Zhou M, Wang X, Shi H, Cheng L, Wang Z, Zhao H, Yang L and Sun J. Characterization of long non-coding RNA-associated ceRNA network to reveal potential prognostic lncRNA biomarkers in human ovarian cancer. Oncotarget. 2016. doi: 10.18632/oncotarget.7181.
59. Liu B, Fang L, Long R, Lan X and Chou K-C. iEnhancer-2L: a two-layer predictor for identifying enhancers and their strength by pseudo k-tuple nucleotide composition. Bioinformatics. 2016; 32:362-369.
60. Liu B, Fang L, Liu F, Wang X, Chen J and Chou K-C. Identification of real microRNA precursors with a pseudo structure status composition approach. PloS one. 2015; 10:e0121501.
61. Ries LAG. Ovarian cancer: survival and treatment differences by age. Cancer. 1993; 71:524-529.
62. Vollebergh MA, Jonkers J and Linn SC. Genomic instability in breast and ovarian cancers: translation into clinical predictive biomarkers. Cellular and Molecular Life Sciences. 2012; 69:223-245.
63. Fong PC, Yap TA, Boss DS, Carden CP, Mergui-Roelvink M, Gourley C, De Greve J, Lubinski J, Shanley S and Messiou C. Poly (ADP)-ribose polymerase inhibition: frequent durable responses in BRCA carrier ovarian cancer correlating with platinum-free interval. Journal of Clinical Oncology. 2010; 28:2512-2519.
64. Lu J, Wu D, Li C, Zhou M and Hao D. Correlation between gene expression and mutator phenotype predicts homologous recombination deficiency and outcome in ovarian cancer. Journal of Molecular Medicine. 2014; 92:1159-1168.
65. Harrow J, Frankish A, Gonzalez JM, Tapanari E, Diekhans M, Kokocinski F, Aken BL, Barrell D, Zadissa A and Searle S. GENCODE: the reference human genome annotation for The ENCODE Project. Genome research. 2012; 22:1760-1774.
66. Xie C, Yuan J, Li H, Li M, Zhao G, Bu D, Zhu W, Wu W, Chen R and Zhao Y. NONCODEv4: exploring the world of long non-coding RNA genes. Nucleic acids research. 2014; 42:D98-D103.
67. Volders P-J, Verheggen K, Menschaert G, Vandepoele K, Martens L, Vandesompele J and Mestdagh P. An update on LNCipedia: a database for annotated human lncRNA sequences. Nucleic acids research. 2015; 43:D174-D180.
68. Chen S, Li P, Xiao B and Guo J. Long noncoding RNA HMlincRNA717 and AC130710 have been officially named as gastric cancer associated transcript 2 (GACAT2) and GACAT3, respectively. Tumor Biology. 2014; 35:8351-8352.
69. Guo X, Gao L, Liao Q, Xiao H, Ma X, Yang X, Luo H, Zhao G, Bu D and Jiao F. Long non-coding RNAs function annotation: a global prediction method based on bi-colored networks. Nucleic acids research. 2013; 41:e35-e35.
70. Ma H, Hao Y, Dong X, Gong Q, Chen J, Zhang J and Tian W. Molecular mechanisms and function prediction of long noncoding RNA. ScientificWorldJournal. 2012; 2012:541786.
71. Sodek KL, Evangelou AI, Ignatchenko A, Agochiya M, Brown TJ, Ringuette MJ, Jurisica I and Kislinger T. Identification of pathways associated with invasive behavior by ovarian cancer cells using multidimensional protein identification technology (MudPIT). Molecular BioSystems. 2008; 4:762-773.
72. Dhillon A, Hagan S, Rath O and Kolch W. MAP kinase signalling pathways in cancer. Oncogene. 2007; 26:3279-3290.
73. Seidman R, Gitelman I, Sagi O, Horwitz SB and Wolfson M. The role of ERK 1/2 and p38 MAP-kinase pathways in taxol-induced apoptosis in human ovarian carcinoma cells. Experimental cell research. 2001; 268:84-92.
74. Dolezal S, Hester S, Kirby PS, Nairn A, Pierce M and Abbott KL. Elevated levels of glycosylphosphatidylinositol (GPI) anchored proteins in plasma from human cancers detected by C. septicum alpha toxin. Cancer biomarkers. 2014; 14:55.
75. Samuel P, Pink RC, Caley DP, Currie J, Brooks SA and Carter D. Over-expression of miR-31 or loss of KCNMA1 leads to increased cisplatin resistance in ovarian cancer cells. Tumour biology. 2016; 37:2565-73. doi: 10.1007/s13277-015-4081-z.
76. Guo S-H, Deng E-Z, Xu L-Q, Ding H, Lin H, Chen W and Chou K-C. iNuc-PseKNC: a sequence-based predictor for predicting nucleosome positioning in genomes with pseudo k-tuple nucleotide composition. Bioinformatics. 2014; 30:1522-1529.
77. Chou K-C. Impacts of bioinformatics to medicinal chemistry. Medicinal Chemistry. 2015; 11:218-234.
78. Guo Q, Cheng Y, Liang T, He Y, Ren C, Sun L and Zhang G. Comprehensive analysis of lncRNA-mRNA co-expression patterns identifies immune-associated lncRNA biomarkers in ovarian cancer malignant progression. Sci Rep. 2015; 5:17683.
79. Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R and Larsson E. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Science signaling. 2013; 6:pl1.
80. Johnson WE, Li C and Rabinovic A. Adjusting batch effects in microarray expression data using empirical Bayes methods. Biostatistics. 2007; 8:118-127.
81. Therneau T. A package for survival analysis in S. R package version 2.37-4. URL http://CRAN R-project org/package=survival Box. 2013; 980032:23298-20032.
82. Heagerty PJ, Lumley T and Pepe MS. Time - dependent ROC curves for censored survival data and a diagnostic marker. Biometrics. 2000; 56:337-344.
83. Heagerty PJ and Zheng Y. Survival model predictive accuracy and ROC curves. Biometrics. 2005; 61:92-105.
84. Huang DW, Sherman BT and Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nature protocols. 2008; 4:44-57.
85. Huang DW, Sherman BT and Lempicki RA. Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic acids research. 2009; 37:1-13.
86. Merico D, Isserlin R, Stueker O, Emili A and Bader GD. Enrichment map: a network-based method for gene-set enrichment visualization and interpretation. PloS one. 2010; 5:e13984.

