INTRODUCTION
Liver cancer is one of the most common malignancies in the world, ranking fifth in men and ninth in women in incidence, and second among both sexes in mortality [1]. In 2012, the estimated number of new cancer cases and deaths was 782,000 and 746,000, respectively [1]. The highest incidence has been reported in Eastern and South-Eastern Asia [age-standardized rates (ASR) of 20.9 and 12.3 per 100,000 population, respectively] and Western Africa (ASR 12.1 per 100,000 population), (Table 1). On the other hand, most developed countries have low (ASR <5 per 100,000) or intermediate (ASR 5–10 per 100,000) rates with some exceptions, such as the high incidence (ASR 34.8 cases per 100,000 men) of liver cancer reported in Southern Italy [2].
Table 1: Estimated numbers of new liver cancer cases in males and females, crude rate and world age standardized rates [ASR(world)] per 100,000 in 2012
Population |
Liver Cancer |
|||
|---|---|---|---|---|
Cases |
Crude Rate |
ASR (W) |
Cumulative Risk |
|
Africa |
58680 |
5.5 |
8.9 |
1.01 |
Eastern Africa |
7947 |
2.3 |
4.0 |
0.45 |
Southern Africa |
2232 |
3.8 |
4.8 |
0.53 |
Middle Africa |
5808 |
4.4 |
8.0 |
0.90 |
Northern Africa |
19653 |
9.4 |
12.3 |
1.50 |
Western Africa |
23040 |
7.2 |
12.1 |
1.30 |
Asia |
594431 |
14.0 |
13.3 |
1.46 |
Eastern Asia |
466336 |
29.4 |
20.9 |
2.26 |
South-Central Asia |
41387 |
2.3 |
2.9 |
0.34 |
South-Eastern Asia |
79953 |
13.2 |
14.2 |
1.64 |
Western Asia |
6755 |
2.8 |
3.8 |
0.45 |
Europe |
63462 |
8.6 |
4.3 |
0.52 |
Central & Eastern Europe |
15953 |
5.4 |
3.1 |
0.37 |
Northern Europe |
6457 |
6.4 |
3.1 |
0.36 |
Southern Europe |
20558 |
13.1 |
5.9 |
0.71 |
Western Europe |
20494 |
10.8 |
4.9 |
0.62 |
Latin America & Caribbean |
30442 |
5.0 |
4.9 |
0.57 |
Northern America |
32718 |
9.3 |
5.8 |
0.70 |
Oceania |
2718 |
7.2 |
5.4 |
0.60 |
Ferlay J, Soerjomataram I, Ervik M, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray, F. GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 [Internet]. Lyon, France: International Agency for Research on Cancer; 2013. Available from: http://globocan.iarc.fr, accessed on 22/04/2015
Hepatocellular carcinoma (HCC) is the most common histological type accounting for approximately 70–85% of primary liver tumors [3]. Chronic HBV and HCV infections represent the major cause of HCC, being associated with more than 80% of cases worldwide [4]. Indeed, pooled estimates of lifetime relative risk to develop HCC are 15 – 20 fold higher in HBV or HCV positive patients compared to non-infected subjects [5]. Non-viral risk factors include alcoholic liver disease, non-alcoholic steatohepatitis, aflatoxin B1 dietary exposure, obesity, and diabetes [6-8]. The relative contribution of viral and non-viral factors to HCC development varies in different populations. The estimated prevalence of virus-related HCC is lower in North America (42%) and Europe (48%), and higher in Africa (80%) and Asia (87%) [4, 9]. A meta-analysis of hepatitis B surface antigen (HBsAg) and anti-HCV antibody prevalence among 27,881 HCC cases from 36 countries showed a large predominance of HBsAg in Asian, African and Latin American countries and a significant higher frequency of anti-HCV antibodies in Europe and United States [4]. The exception to these patterns is represented by the high rates of HCV-related HCC in Japan and Egypt [4].
The HBV- and HCV-related carcinogenesis initiates in the context of chronic hepatitis, and progresses to HCC in a multistep process lasting for as long as 30 years [10] (Figure 1). During HCC progression, several environmental factors (aflatoxin B1, alcohol consumption, cigarette smoking, hepatotoxic chemical agents) as well as host co-factors (elevated serum androgen levels, genetic polymorphisms, DNA repair enzymes) may synergize and lead to progressive accumulation of multiple genomic changes in the hepatocytes [11, 12]. Among these, non-synonymous mutations in TP53 and CTNNB1 genes are well known cancer drivers for HCC development with variable frequencies depending on the underlying etiology [13, 14].
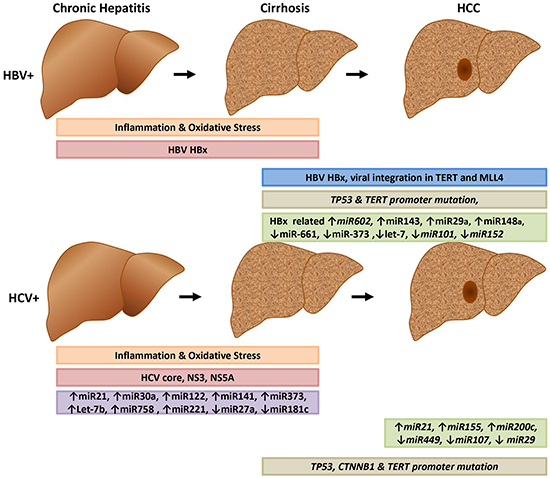
Figure 1: Early and late events of HBV and HCV-related liver carcinogenesis. The HBV HBx protein facilitates integration of HBV into host DNA, resulting in major genetic alteration of the host genome. HBV- and HCV-encoded proteins contribute to the alteration of several signaling pathways. Both viruses promote the growth of infected cells and activate several signaling pathways including RAS, PI3K, EGFR, and IGFR1.
Over the last decade, massively parallel sequencing technologies allowed to further uncover the genomic diversity of HCC and to identify consistent gene alterations activating signaling pathways relevant to cell transformation [15, 16]. Such analyses allowed to identify HCC subgroups characterized by definite genetic profiles that may be linked to specific oncogenic factors and are useful to further stratify HCCs for personalized medicine applications [17].
Here, we review the molecular pathogenesis of primary liver cancer with particular emphasis on the host genetic variations identified by high-throughput technologies in the context of HBV and HCV related HCC. We discuss the importance of genetic alterations in diagnosis, prognosis as well as in tumor stratification for more efficient treatment approaches.
HBV and hepatocellular carcinoma
HBV is a partially double-stranded hepatotropic DNA virus containing four partial overlapping open reading frames (ORFs) encoding the reverse transcriptase/ polymerase (Pol), the capsid protein (core antigen HBcAg), three envelope proteins (L, M, and S) and the transactivating protein x [18].
HBV infection contributes to hepatocarcinogenesis by different mechanisms including 1) expression of HBx protein; 2) integration of viral DNA into the host genome; and 3) accumulation of somatic mutations in human genes with or without exposure to other carcinogens (i.e. aflatoxin B1), [10, 19, 20].
HBV HBx protein
The HBV protein HBx transactivates viral and cellular genes by interacting with nuclear transcription factors, such as cyclic adenosine monophosphate(cAMP) response element-binding protein (CREB), activating protein 1 (AP-1), nuclear factor kappa B (NF-kB), and specificity protein 1 (Sp-1). HBx affects also several cellular pathways including DNA repair, cell proliferation, differentiation and apoptosis [20-24]. In addition, HBx protein trans-activates DNA methyltransferase 1 (DNMT1) and DNMT3A genes in the HBV infected hepatocytes, resulting in the suppression of cell cycle regulators P16INK4A and p21 Cip1/CDKN1A, cell-adhesion molecule E-cadherin as well as SFRP1 and SFRP5 genes, which inhibit Wnt signaling pathway [25-30]. Moreover, Wnt/β-catenin pathway is directly activated by HBx protein, which interferes with proteasomal degradation of β-catenin [31, 32]. More recently, HBx has been shown to activate the Yes-associated protein (YAP) oncogene, a downstream effector of the Hippo-signaling pathway, which represents a key element in HCC development [33]. The HBx protein can also bind to the p53 oncosuppressor, leading to the disruption of the p53/XPB/XPD complex of the transcriptional factor II H and compromising the nucleotide excision repair mechanism [34]. Recent studies showed that HBx is able to activate AKT, favoring persistent, non-cytopathic HBV replication and inhibition of the transcription factor hepatocyte nuclear factor 4 (HNF4) [35].
HBV integration and chromosomal alterations
HBV genome commonly integrates in HCC causing global genomic instability, increased expression of genes adjacent to integration loci, and expression of viral-host fusion transcripts [36-39]. Genome-wide analysis showed that HBV integration occurs in 86% of HCCs and in 30.7% of adjacent non-tumor tissues [40]. A similar frequency (75.5%) has been identified in HCC patients with occult HBV infection [41]. The analysis of genome instability showed that somatic copy number variations are significantly increased at locations adjacent to HBV integration sites [40], and that the number of chromosomal aberrations correlates with the mutational status of tumor suppressor genes, such as TP53, RB1, CDNK2A and TP73 [42]. Next-generation sequencing uncovered several new genes recurrently interrupted by HBV integrants including TERT, MLL4, CCNE1, NTRK2, IRAK2 and p42MAPK1 [40, 43-45]. The integration of HBV DNA preferentially involves the HBx sequence which frequently undergoes deletion at the 3’-end, causing the expression of a C-terminal-truncated HBx protein able to enhance HCC cell invasiveness and metastasis [46, 47]. Transcription profiling by RNA-sequencing analysis allowed identification of several viral-human fusion transcripts generated as a consequence of HBV integration. The most abundant is the long non-coding RNA HBx-LINE1 chimera, which has been detected in 23% of HBV-related HCCs [42], and has been shown to promote tumor growth through the activation of Wnt/b-catenin signaling [48].
HBV and aflatoxin B1 interaction
In HBV-associated HCC, there is a strong overrepresentation of TP53 mutations, particularly in geographic regions endemic for HBV and with dietary exposure to aflatoxin B1 (AFB1) [49]. Specifically, AFB1 induces a non-synonymous mutation (G to T transversion) changing arginine to serine at codon 249 of TP53 gene in up to 50% of HCCs. The mutated p53, together with chronic HBV infection, synergistically increase the risk to develop HCC [49, 50]. Indeed, the p53 R249S is able to bind the HBx protein and to promote hepatocyte transformation [51].
HCV and hepatocellular carcinoma
HCV is a single-stranded RNA virus encoding a large polyprotein of 3,000 amino acids. The HCV polyprotein can be cleaved by viral and cellular proteases into four structural proteins (capsid protein C, envelope glycoproteins E1 and E2, and protein P7), and six non-structural proteins (NS2,NS3, NS4A, NS4B, NS5A, and NS5B) [52].
HCV causes chronic hepatitis in more than 80% of infected subjects, versus the 10% in HBV infected patients, and is up to 20 fold more efficient than HBV in promoting liver cirrhosis [53]. Pathogenesis of HCV-related HCC mainly relies on the ability of the virus to cause chronic inflammation, immune-mediated hepatocyte death, tissue damage, fibrosis and evolution to cirrhosis [54-56]. The HCV core protein C as well as the non-structural proteins NS3, NS5A, and NS5B induce hepatocarcinogenesis through their ability to perturb several cellular pathways, such as DNA repair, proliferation and apoptosis [57-59].
HCV core protein
The HCV core protein binds to numerous transcription factors, thus regulating expression of several host genes [60, 61]. In addition, it promotes cell growth and survival by activation of mitogen-activated protein kinase (MAPK) signaling cascade, including MEK1, ERK1/2, JNK, p38 MAP kinases, and MKP1 Map kinases [62-64]. HCV core enhances cell proliferation by inhibiting the synthesis of p53, p21 CDK inhibitor, and E2F-1 as well as the phosphorylation of pRb [65]. Moreover, it is able to suppress immune-mediated apoptosis by inhibiting caspase-8 via over-expression of the cellular FADD-like interleukin-1 converting enzyme (c-FLIP) [66]. In addition, it enhances angiogenesis by triggering the production of TGF-β2 and VEGF proteins, and stabilizing the hypoxia-inducible factor 1 (HIF-1a) [67]. HCV core protein induces IL-6, gp130, leptin receptor, and STAT3 over expression, which in turn may deregulate c-Myc and cyclin D1 downstream the STAT3 signaling pathway [68]. HCV core protein also activates the Wnt/b-catenin cascade, which is known to play a significant role in the HCC development [69].
HCV NS3 protein
The HCV NS3 protein is a multifunctional protein with protease, RNA helicase, and NTPase activity. NS3 can promote hepatocarcinogenesis by its binding with certain cellular proteins, such as p21 and p53 [70]. Recently, HCV NS3/4A protease was demonstrated to activate the EGFR signaling pathway through the proteolytic cleavage of tyrosine phosphatase T-cell protein (TC-PTP), resulting in increased EGFR activity and the downstream PI3K/Akt pathway [71]. MAP kinase signaling, through activation of JNK, was also implicated in HCV NS3 protein-mediated cell growth in infected cells [65].
HCV NS5A protein
NS5A has been shown to bind a wide range of cellular proteins controlling signal transduction and host microenvironment [72]. Particularly, the truncated HCV NS5A protein localizes to the nucleus and acts as a transcriptional activator. NS5A can bind cellular signaling components and regulatory protein kinases, leading to the suppression of the host immune response and inhibition of apoptosis [73]. NS5A binds and stabilizes β-catenin, inducing activation of the c-Myc promoter and increased c-Myc expression, which increases production of reactive oxygen species, DNA damage, and cell-cycle deregulation [74, 75]. NS5A also stabilizes poly(ADPribose) polymerase 1 (PARP-1), which is involved in DNA repair and apoptosis, thus contributing to genetic instability and accumulation of mutations in HCV-infected hepatocytes [59, 76, 77].
Gene expression profiling in HCC
Early studies on gene expression profiling highlighted the wide heterogeneity of global gene expression patterns in liver tumors [78, 79]. Hierarchical clustering analysis of tumor-specific genes contributed to classify HCC subtypes, unravel the complex pathogenesis of HCC and stratify tumors according to their etiological factor, clinical stage, recurrence rate, and prognosis [80-82]. Several reports showed strong expression signatures in genes regulating cell proliferation and anti-apoptotic pathways (i.e., PNCA and cell cycle regulators CDK4, CCNB1, CCNA2, and CKS2), ubiquitination mechanisms [83, 84], as well as molecular markers of tumor progression like HSP70, CAP2, GPC3, and GS [85]. A class-comparison analysis performed in our lab (HCV-related HCC, HCV-related non HCC and metastatic liver tissue vs. normal control; HCV-related HCC vs. autologous HCV-related non HCC liver tissue) identified a gene-set that distinguish the different types of liver disease [86]. In particular, the time course analysis allowed to identify several candidate genes as progression markers (e.g., GPC3, CXCL12, SPINK1, GLUL, UBD, TM4SF5, DPT, SCD, MAL2, TRIM55, COL4A2) [86]. Altogether, these data are useful for developing a specific gene-chip including those genes showing the highest fold increase.
Moreover, HCC-specific alterations of signal transduction pathways and protein expression patterns have been detected and opened opportunities for new therapies targeting molecular factors such as EGFR, VEGF, DDEFL, VANGL1, WDRPUH, ephrin-A1, GPC3, number gain 7q, PFTK1, PEG10 and miR-122a [87, 88].
miRNA in HBV-related HCC
Several microRNAs (miRNA) have been found deregulated in HBV-positive HCCs. The HBx protein has a major role in the miRNAs alteration. Specifically, HBx is able to increase the expression of miRNA 602, targeting the putative tumor suppressor Ras association domain family 1 isoform A (RASSF1A) [89]; miRNA-143, targeting fibronectin type III domain-containing 3B (FNDC3B) promoting hepatoma cell invasion, migration and tumor metastasis [90]; miRNA-29a and miR-148a, targeting phosphatase and tensin homolog (PTEN) and stimulating cell migration [91, 92]. Notably, levels of miR-122 and let-7b have been found increased in the serum of HBV-positive patients with early HCC and have been proposed as useful markers to differentiate early HCC from dysplastic nodules [93]. In addition, HBx inhibits the expression of miR-661, targeting metastasis associated 1 factor (MTA1) [94]; miR-373, targeting cadherin 1 (CDH1) gene [95]; let-7a, which is implicated in the cell proliferation control through STAT3 modulation [96]; and miR-101 and miR-152, controlling the expression of DNMT3A and DNMT1, respectively [97, 98]. HBx also inhibits expression of miR-148a, which targets hematopoietic pre-B cell leukemia transcription factor interacting protein (HPIP), and miRNA-16 family, targeting cyclin D1 (CCND1); both mi-148a and miRNA-16 family are associated with tumor growth control [99, 100].
miRNA in HCV-related HCC
HCV replication and pathogenesis are tightly controlled by the expression of several miRNAs [101]. In particular, miRNA-122 favors HCV replication by binding directly to viral RNA, while miRNAs-130a and-21 subvert the IFN signaling pathway, leading to immune evasion [101, 102]. miRNAs-196/199a and -448/let-7b attenuate viral replication, and Let-7b and miRNA-221 compromise the antiviral effect of IFN-α [103, 104]. Expression of miRNAs regulating lipid metabolism (miRNA-27a) and hepatocyte growth (miRNA 181c) is decreased in HCV infected cells. In contrast, expression of miRNA-155, promoting hepatocyte proliferation and inflammation, and miRNA-21 and -200c, promoting fibrosis, is increased in advanced stages of liver disease [101].
Chromosomal aberrations and gene copy number variations in HCC
Chromosomal alterations are very common in liver tumors. Comparative genomic hybridization (CGH) data showed frequent gain of chromosomal regions 1q (57.1%), 8q (46.6%), 6p (22.3%), and 17q (22.2%), and prominent losses of 8p (38%), 16q (35.9%), 4q (34.3%), 17p (32.1%), and 13q (26.2%), Table 2, [105, 106]. Chromosome losses in the regions 4q, 13q, 16q, and 8p are more frequent in HBV - related tumors, while loss of chromosome 8p in HCV-positive HCCs is less frequent compared to virus negative tumors [105]. Moreover, gains of 1q and 8q as well as losses of 4q, 16q and 13q have been shown to increase with HCC progression [105].
Table 2: Major chromosomal alterations (frequency above 20%) identified in 31 studies by conventional metaphase-based CGH analysis
Chromosome Gain |
Hot spot |
All HCC (n=785) |
HBV HCC (n=244) |
HCV HCC (n=110) |
|---|---|---|---|---|
1q |
1q31 |
57.1% |
53.3% |
45.5% |
6p |
6p25–p23 |
22.3% |
24.2% |
16.4% |
8q |
8q24.2 |
46.6% |
46.7% |
34.5% |
17q |
17q25 |
22.2% |
20.9% |
17.3% |
Chromosome Loss |
||||
4q |
4q23–24 |
34.3% |
43.4% |
27.3% |
8p |
8p21.3–p21.2 |
38.0% |
40.6% |
20.0% |
13q |
13q21.1–q21.3 |
26.2% |
31.1% |
23.6% |
16q |
16p13.2 |
35.9% |
41.8% |
27.3% |
17p |
17p12 |
32.1% |
32.4% |
30.9% |
High-resolution array CGH studies allowed to discover chromosomal gains in 5p15.33 and 9q34.2–34.3 and losses in 6q, 9p and 14q, in addition to the previously identified genetic aberrations [107-111]. Copy number variation of 1q21.3-44 and LOH of 1p36.21-36.32 and 17p13.1-13.3 regions were identified in early HCC but not in chronic liver disease, suggesting their possible causative role in HCC development, while gains of 5q11.1-35.3, 6p, and 8q11.1-24.3 as well as LOH of 4q11-34.3 and 8p11.21-23.3 appear associated with more advanced tumor stages [108, 112]. The copy number gain of 8q24 region is generally associated with an increased expression of c-Myc gene, particularly in viral and alcohol-related HCCs but not in cryptogenic HCCs [111]. Other small chromosomal aberrations, such as amplification of 1q32.1 and 20q13.33, have been associated with overexpression of MDM4 and EEF1A2, respectively, in approximately 50% of tumors, independently from the etiology [111]. The integration of CGH data with gene expression arrays allowed to identify over-expressed candidate oncogenes, such as TAGLN2, MDM4, SNRPE, SPP1 VEGFA, PEG10, Jab1, HEY1, BOP1 and EEF1A2 [109, 111, 113-117] and down-regulated candidate tumor suppressor genes, such as TRIM35, DLC1, CRYL1, and Spry2 [117-120]. Few studies analyzed the prognostic significance of chromosomal alterations and gene profile expression [121]. Roessler et al. combined CGH data and gene expression arrays of 256 HCC cases, and identified 10 genes associated with poor survival, of which six were located at chromosome 8p, [122].
Somatic mutations in HBV and HCV-related HCC
Genomic instability of viral-related HCCs is characterized by high frequency of somatic mutations. Several studies showed that TP53 oncosuppressor and CTNNB1 oncogene are the most frequently mutated genes in primary liver cancer, being identified in about 25% and 30% of HCCs, respectively [13]. Up to 75% of missense TP53 mutations, other than the R249S induced by AFB1, are scattered over 200 codons of the TP53 region encoding for the DNA-binding domain [123-126], and show similar frequencies in HCCs with different etiologies [13]. Such a finding suggests that chronic inflammation, reactive oxygen species, and oxidative DNA damage, which are common effects of cancer causing factors, may be responsible for such variations. TP53 mutations may cause several pathway deregulations in HCCs. Okada et al. identified 83 genes differentially expressed in TP53 mutant compared to wild type TP53 liver tumors [127]. The genes differentially expressed in TP53 mutant tumors include cell cycle regulators (CCNG2, BZAP45) and cell proliferation-related genes (SSR1, ANXA2, S100A10, and PTMA) [127]. These data support the hypothesis that p53 mutant tumors have higher malignant potentials compared with wild type p53 [128, 129].
CTNNB1 gene, expressing β-catenin, and AXIN1 and AXIN2 genes, encoding for components of β-catenin degradation complex, are frequently mutated in liver cancers [130, 131, 131-133]. Interestingly, CTNNB1 mutations have been shown to occur mainly in alcohol and HCV-related tumors [16, 134, 135]. Guichard et al. reported CTNNB1 mutations in 11.4% and 33.3% of HBV and HCV-related HCCs, respectively, and in 41.8% of alcohol-related HCCs [16]. In addition, mutations in CTNNB1 and TP53 genes appear to be mutually exclusive, suggesting that inactivation of either pathway is sufficient to induce cell transformation [16].
Next generation sequencing allowed to identify other oncosuppressor genes in HCC, which have lower mutation frequencies compared to TP53, independently from the etiology of the tumor. They include P16INK4 (6%–17%), P14arf (5%), AXIN1 (5%–15%), AXIN2 (2%–10%), TIP30 (24%), IGFR2 (10%–20%), KLF6 (15%), Caspase- 8 (13%), PTEN (5%–8%), etc. [14, 135-137]. Moreover, oncogenes other than CTNNB1 are less frequently mutated and they include EGFR (1%) and Erb2 (2%), K-ras (0%–19%) and N-ras (2%) and PIK3CA (<5%) [14, 135-137].
At least three large whole-exome sequencing studies described the mutational landscape and possible druggable targets in viral-related and viral-unrelated HCC,Table 3, [138-140]. Totoki et al. identified 30 candidate driver genes associated with 11 core pathways in 608 liver cancers including 413 cases from Japan [138]. Importantly, they discovered that 68% of HCC cases had telomerase reverse transcriptase (TERT) genetic alterations, including promoter mutation, focal amplification, and viral genome integration, and recognized TERT as a central regulator of hepatocarcinogenesis.
Table 3: Comparison of recurrently mutated genes in HCC identified in three large studies in Japan, Korea and Europe
Function |
Gene Name |
Totoki et al.* (n=452) |
Ahn et al.* (n=231) |
Schulze et al.* (n=243) |
|---|---|---|---|---|
WNT/β-catenina |
CTNNB1 |
31.0% |
22.9% |
37.4% |
RSPO2 |
- |
3.0% |
- |
|
AXIN1 |
6.2% |
6.9% |
11.1% |
|
FZD6 |
- |
3.0% |
- |
|
Chromatin remodeling |
ARID1A |
8.6% |
3.9% |
12.8% |
ARID2 |
10.8% |
3.0% |
6.8% |
|
ARID4b |
1.1% |
3.0% |
- |
|
p53/cell cycle |
TP53 |
32.2% |
31.2% |
24.3% |
CDKN2A |
2.2% |
6.1% |
8.5% |
|
ATM |
4.4% |
2.2% |
5.5% |
|
CDKN2B |
0.2% |
2.2% |
5.1% |
|
CCND1 |
- |
5.2% |
4.7% |
|
RB1 |
4.2% |
7.8% |
3.8% |
|
RBL2 |
2.2% |
3.0% |
- |
|
HUWE1 |
0.4% |
0.9% |
3.4% |
|
Epigenetic regulation |
MLL2 |
3.8% |
5.2% |
5.5% |
MLL3 |
2.4% |
3.0% |
2.0% |
|
MLL |
2.9% |
3.9% |
- |
|
CHD1 |
- |
3.0% |
- |
|
CHD7 |
0.9% |
3.0% |
3.4% |
|
CREBBP |
2.0% |
3.0% |
3.0% |
|
SMC3 |
- |
- |
3.0% |
|
SRCAP |
2.9% |
- |
3.0% |
|
Telomere mantainence |
TERT promoter |
55.1% |
- |
60.0% |
TERT |
4.0% |
- |
- |
|
Oxidative stress |
NFE2L2 |
4.9% |
3.0% |
6.4% |
KEAP1 |
2.4% |
- |
3.8% |
|
Hepatic differentiation |
ALB |
6.9% |
4.8% |
12.8% |
APOB |
10.2% |
10.3% |
9.4% |
|
HNF1A |
2.2% |
0.9% |
4.7% |
|
FGA |
1.5% |
1.7% |
3.4% |
|
MAPK |
RPS6KA3 |
3.8% |
5.0% |
6.8% |
FGF4 |
0.2% |
- |
4.7% |
|
FGF19 |
- |
5.0% |
4.7% |
|
FGF3 |
0.2% |
0.9% |
4.3% |
|
EPHA4 |
1.8% |
2.6% |
3.4% |
|
FLT4 |
0.9% |
2.6% |
3.4% |
|
HGF |
0.7% |
0.4% |
3.0% |
|
NTRK3 |
1.5% |
1.7% |
3.0% |
|
PI3K-AKT-mTOR |
TSC2 |
5.3% |
3.0% |
5.1% |
FGF4 |
0.2% |
- |
4.7% |
|
FGF19 |
4.0% |
5.0% |
4.7% |
|
FGF3 |
0.2% |
0.9% |
4.3% |
|
FLT4 |
0.9% |
2.6% |
3.4% |
|
PTEN |
1.3% |
2.0% |
3.4% |
|
HGF |
0.7% |
0.4% |
3.0% |
|
PRKCB |
1.5% |
3.5% |
- |
|
NTRK3 |
1.5% |
1.7% |
3.0% |
|
JAK3 |
0.9% |
0.4% |
3.0% |
* Totoki et al. (n=413 Japan, 92 HBV+ and 183 HCV+); Ahn et al. (n=231 Korea, 167 HBV+ and 22 HCV+); Schulze et al. (n=243 [n=193 France, n=41 Italy, n=9 Spain], 33 HBV+ and 61 HCV+).
Ahn et al. analyzed 231 cancer cases from Korea and identified recurrent somatic mutations in nine genes, comprising TP53, CTNNB1, AXIN1, RPS6KA3, and RB1, homozygous deletions in FAM123A, RB1, and CDKN2A, and high-copy amplifications in MYC, RSPO2, CCND1, and FGF19 [139]. RB1 mutations were associated with cancer recurrence in resectable HCCs. Schulze and colleagues identified 161 putative driver genes associated with 11 pathways: TERT expression, WNT/β-catenin, PI3K-AKT-mTOR, TP53 – related pathway, MAP kinases, hepatic differentiation, epigenetic regulation, chromatin remodeling, oxidative stress, IL-6/JAK-STAT and TGF-β [139, 140]. Approximately 30% of liver tumors analyzed in these studies harbored genetic alterations potentially targetable by Food and Drug Administration (FDA)-approved drugs [140]. The analysis of copy number variations revealed recurrent homozygous deletions of the CFH locus, IRF2, CDKN2A, PTPN3, PTEN, AXIN1 and RPS6KA3 and recurrent focal amplifications of TERT, VEGFA, MET, MYC, the FGF-CCND1 locus containing FG3, FG4 and FGF19, JAK3 and CCNE1 [140]. Moreover, integrating results of exome sequencing mutation and focal copy number alteration allowed to identify 3 groups of putative cancer driver genes: CTNNB1, TP53 and AXIN1 clusters [140].
The frequency of mutations in different genes seems related to the cancer etiology. TP53 gene was mostly mutated in HBV-related HCC, while CTNNB1, TERT, CDKN2A, SMARCA2, and HGF genes were mainly mutated in alcohol-related HCCs, and IL6ST was mutated in HCCs with no known etiology. Conversely, no specific gene mutation was associated with HCV infection, metabolic syndrome and hemochromatosis [138-140].
Somatic mutations in the TERT promoter have been identified as the first recurrent genetic alteration in 25% of dysplastic cirrhotic nodules [141, 142]. These mutations create a consensus binding sequence for a ternary complex factor and induce expression of telomerase reverse transcriptase [148]. Conversely, several other genes known to be recurrently mutated in HCC, including CTNNB1, TP53, ARID1A, ARID2, RPS6KA3, NFE2L2 and KEAP1 were not mutated in dysplastic nodules [141]. TERT promoter mutations may be considered as biomarkers for the identification of premalignant lesions developed in cirrhosis patients with a high risk of progression to HCC.
Immunotherapeutic approaches and gene mutations in HCC
In several cancer types, the activity of tumor antigen-specific T-cells is tightly regulated by the balanced expression of stimulatory and inhibitory molecules defined as “immune checkpoints” [144, 145]. Therapies targeting these checkpoints, such as those directed against cytotoxic T-lymphocyte antigen 4 (CTLA-4) and programmed death 1 receptor (PD-1), have shown to be more effective in cancers characterized by high rates of somatic mutations [146]. Recent studies have indicated that a high tumor mutation burden increases responsiveness to CTLA-4 inhibition in melanoma, to PD-1 inhibition in non-small cell lung cancer and in mismatch repair-deficient colorectal cancers [147]. The hypothesis is that the higher number of genetic variations leads to a greater number of mutated epitopes in tumor proteins (neoantigens). Such neoantigens may be characterized by an improved MHC-binding profile, resulting in superior presentation to T cells for eliciting a stronger cytotoxic response [148, 149]. Very recently, this has been experimentally proven in animal models [150, 151] as well as in melanoma patients treated with the anti–CTLA-4 monoclonal antibody, ipilimumab [152, 153].
Liver sinusoidal endothelial cells express high levels of the inhibitory molecule program death receptor ligand 1 (PD-L1) and low levels of the co-stimulatory molecules CD80 and CD86, thereby limiting their ability to effectively activate CD4-positive (CD41) and CD8 1 T lymphocytes [154, 155]. Immune checkpoint inhibitors have been recently evaluated in HCC patients. The anti-CTLA-4 monoclonal antibody tremelimumab showed a safe profile and antitumor activity in HCC patients with chronic HCV infection [156]. Very recently, results from a phase I/II clinical trial (ClinicalTrials.gov Identifier: NCT01658878) presented at the last 2015 ASCO Meeting showed that nivolumab, a fully humanized IgG4 monoclonal antibody to PD-1, may be a promising treatment for patients with advanced HCC [157]. Indeed, the overall survival at 1 year was 62% and overall objective responses rate was 19%, including complete response (CR) in 5% and partial response in 14% of enrolled patients. Such responses are significantly higher compared to responses to the kinase inhibitor sorafenib, the current standard of care for late stage HCC.
Further studies are needed to evaluate whether the immune responses elicited by mutated epitopes could lead to an increased efficacy of anti immune-checkpoint therapies also in liver cancer.
CONCLUSIONS
Classification of liver cancers in homogeneous sub-groups characterized by specific molecular alterations is an important tool for the application of personalized therapies. Several commonly altered pathways have emerged following the integration of data obtained with multiple high-throughput analyses. Common oncogenic drivers, differentially represented in HCCs with different etiologies, include genetic alterations affecting TERT, Wnt/beta-catenin, JAK/STAT and PI3K-AKT-mTOR pathways. Drugs targeting these pathways are now available and have been approved in clinical trials.
CONFLICTS OF INTEREST
The authors have no conflicts of interest to disclose.
GRANT SUPPORT
The study was funded by EU FP7 Project Cancer Vaccine development for Hepatocellular Carcinoma – HEPAVAC (Grant Nr. 602893) and Italian Ministry of Health (Ricerca Corrente; Progetto Finalizzato 270/RF-2010-2312010).
REFERENCES
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65: 87-108. doi: 10.3322/caac.21262
2. Curado MP, Edwards B, Shin HR, Ferlay J, Heanue M, Boyle P, Storm H. Cancer Incidence in Five Continents. Lyon: IARC Scientific Publications N° 160; 2007.
3. Perz JF, Armstrong GL, Farrington LA, Hutin YJ, Bell BP. The contributions of hepatitis B virus and hepatitis C virus infections to cirrhosis and primary liver cancer worldwide. J Hepatol. 2006; 45: 529-38. doi: 10.1016/j.jhep.2006.05.013
4. Raza SA, Clifford GM, Franceschi S. Worldwide variation in the relative importance of hepatitis B and hepatitis C viruses in hepatocellular carcinoma: a systematic review. Br J Cancer. 2007; 96: 1127-34. doi: 10.1038/sj.bjc.6603649
5. El-Serag HB. Epidemiology of viral hepatitis and hepatocellular carcinoma. Gastroenterology. 2012; 142: 1264-73. doi: 10.1053/j.gastro.2011.12.061
6. Michelotti GA, Machado MV, Diehl AM. NAFLD, NASH and liver cancer. Nat Rev Gastroenterol Hepatol. 2013; 10: 656-65. doi: 10.1038/nrgastro.2013.183
7. Liu Y, Chang CC, Marsh GM, Wu F. Population attributable risk of aflatoxin-related liver cancer: systematic review and meta-analysis. Eur J Cancer. 2012; 48: 2125-36. doi: 10.1016/j.ejca.2012.02.009
8. Bosch FX, Ribes J, Cleries R, Diaz M. Epidemiology of hepatocellular carcinoma. Clin Liver Dis. 2005; 9: 191-211, v. doi: 10.1016/j.cld.2004.12.009
9. de Martel C, Ferlay J, Franceschi S, Vignat J, Bray F, Forman D, Plummer M. Global burden of cancers attributable to infections in 2008: a review and synthetic analysis. Lancet Oncol. 2012; 13: 607-15. doi: 10.1016/S1470-2045(12)70137-7
10. Arzumanyan A, Reis HM, Feitelson MA. Pathogenic mechanisms in HBV- and HCV-associated hepatocellular carcinoma. Nat Rev Cancer. 2012; 13: 123-35. doi: 10.1038/nrc3449
11. Farazi PA, DePinho RA. Hepatocellular carcinoma pathogenesis: from genes to environment. Nat Rev Cancer. 2006; 6: 674-87.
12. Tornesello ML, Buonaguro L, Buonaguro FM. An overview of new biomolecular pathways in pathogen-related cancers. Future Oncol. 2015; 11: 1625-39. doi: 10.2217/fon.15.87
13. Tornesello ML, Buonaguro L, Tatangelo F, Botti G, Izzo F, Buonaguro FM. Mutations in TP53, CTNNB1 and PIK3CA genes in hepatocellular carcinoma associated with hepatitis B and hepatitis C virus infections. Genomics. 2013; 102: 74-83. doi: 10.1016/j.ygeno.2013.04.001
14. Nault JC, Zucman-Rossi J. Genetics of hepatobiliary carcinogenesis. Semin Liver Dis. 2011; 31: 173-87. doi: 10.1055/s-0031-1276646
15. Bruix J, Han KH, Gores G, Llovet JM, Mazzaferro V. Liver cancer: Approaching a personalized care. J Hepatol. 2015; 62: S144-S156. doi: 10.1016/j.jhep.2015.02.007
16. Guichard C, Amaddeo G, Imbeaud S, Ladeiro Y, Pelletier L, Maad IB, Calderaro J, Bioulac-Sage P, Letexier M, Degos F, Clement B, Balabaud C, Chevet E, et al. Integrated analysis of somatic mutations and focal copy-number changes identifies key genes and pathways in hepatocellular carcinoma. Nat Genet. 2012; 44: 694-8. doi: 10.1038/ng.2256
17. Boyault S, Rickman DS, de RA, Balabaud C, Rebouissou S, Jeannot E, Herault A, Saric J, Belghiti J, Franco D, Bioulac-Sage P, Laurent-Puig P, Zucman-Rossi J. Transcriptome classification of HCC is related to gene alterations and to new therapeutic targets. Hepatology. 2007; 45: 42-52. doi: 10.1002/hep.21467
18. Grimm D, Thimme R, Blum HE. HBV life cycle and novel drug targets. Hepatol Int. 2011; 5: 644-53. doi: 10.1007/s12072-011-9261-3
19. De Mitri MS, Cassini R, Bernardi M. Hepatitis B virus-related hepatocarcinogenesis: molecular oncogenic potential of clear or occult infections. Eur J Cancer. 2010; 46: 2178-86. doi: 10.1016/j.ejca.2010.03.034
20. Benhenda S, Cougot D, Buendia MA, Neuveut C. Hepatitis B virus X protein molecular functions and its role in virus life cycle and pathogenesis. Adv Cancer Res. 2009; 103: 75-109. doi: 10.1016/S0065-230X(09)03004-8
21. Neuveut C, Wei Y, Buendia MA. Mechanisms of HBV-related hepatocarcinogenesis. J Hepatol. 2010; 52: 594-604. doi: 10.1016/j.jhep.2009.10.033
22. Ng SA, Lee C. Hepatitis B virus X gene and hepatocarcinogenesis. J Gastroenterol. 2011; 46: 974-90. doi: 10.1007/s00535-011-0415-9
23. Ali A, bdel-Hafiz H, Suhail M, Al-Mars A, Zakaria MK, Fatima K, Ahmad S, Azhar E, Chaudhary A, Qadri I. Hepatitis B virus, HBx mutants and their role in hepatocellular carcinoma. World J Gastroenterol. 2014; 20: 10238-48. doi: 10.3748/wjg.v20.i30.10238
24. Cougot D, Wu Y, Cairo S, Caramel J, Renard CA, Levy L, Buendia MA, Neuveut C. The hepatitis B virus X protein functionally interacts with CREB-binding protein/p300 in the regulation of CREB-mediated transcription. J Biol Chem. 2007; 282: 4277-87. doi: 10.1074/jbc.M606774200
25. Zhu YZ, Zhu R, Fan J, Pan Q, Li H, Chen Q, Zhu HG. Hepatitis B virus X protein induces hypermethylation of p16(INK4A) promoter via DNA methyltransferases in the early stage of HBV-associated hepatocarcinogenesis. J Viral Hepat. 2010; 17: 98-107. doi: 10.1111/j.1365-2893.2009.01156.x
26. Lee JO, Kwun HJ, Jung JK, Choi KH, Min DS, Jang KL. Hepatitis B virus X protein represses E-cadherin expression via activation of DNA methyltransferase 1. Oncogene. 2005; 24: 6617-25. doi: 10.1038/sj.onc.1208827
27. Jung JK, Arora P, Pagano JS, Jang KL. Expression of DNA methyltransferase 1 is activated by hepatitis B virus X protein via a regulatory circuit involving the p16INK4a-cyclin D1-CDK 4/6-pRb-E2F1 pathway. Cancer Res. 2007; 67: 5771-8. doi: 10.1158/0008-5472.CAN-07-0529
28. Liu J, Lian Z, Han S, Waye MM, Wang H, Wu MC, Wu K, Ding J, Arbuthnot P, Kew M, Fan D, Feitelson MA. Downregulation of E-cadherin by hepatitis B virus X antigen in hepatocellullar carcinoma. Oncogene. 2006; 25: 1008-17. doi: doi:10.1038/sj.onc.1209138
29. Xie Q, Chen L, Shan X, Shan X, Tang J, Zhou F, Chen Q, Quan H, Nie D, Zhang W, Huang AL, Tang N. Epigenetic silencing of SFRP1 and SFRP5 by hepatitis B virus X protein enhances hepatoma cell tumorigenicity through Wnt signaling pathway. Int J Cancer. 2014; 135: 635-46. doi: 10.1002/ijc.28697
30. Lee S, Tarn C, Wang WH, Chen S, Hullinger RL, Andrisani OM. Hepatitis B virus X protein differentially regulates cell cycle progression in X-transforming versus nontransforming hepatocyte (AML12) cell lines. J Biol Chem. 2002; 277: 8730-40. doi: 10.1074/jbc.M108025200
31. Ding Q, Xia W, Liu JC, Yang JY, Lee DF, Xia J, Bartholomeusz G, Li Y, Pan Y, Li Z, Bargou RC, Qin J, Lai CC, et al. Erk associates with and primes GSK-3beta for its inactivation resulting in upregulation of beta-catenin. Mol Cell. 2005; 19: 159-70. doi: 10.1016/j.molcel.2005.06.009
32. Hsieh A, Kim HS, Lim SO, Yu DY, Jung G. Hepatitis B viral X protein interacts with tumor suppressor adenomatous polyposis coli to activate Wnt/beta-catenin signaling. Cancer Lett. 2011; 300: 162-72. doi: 10.1016/j.canlet.2010.09.018
33. Zhang T, Zhang J, You X, Liu Q, Du Y, Gao Y, Shan C, Kong G, Wang Y, Yang X, Ye L, Zhang X. Hepatitis B virus X protein modulates oncogene Yes-associated protein by CREB to promote growth of hepatoma cells. Hepatology. 2012; 56: 2051-9. doi: 10.1002/hep.25899
34. Jia L, Wang XW, Harris CC. Hepatitis B virus X protein inhibits nucleotide excision repair. Int J Cancer. 1999; 80: 875-9. doi: 10.1002/(SICI)1097-0215(19990315)80:6
35. Rawat S, Bouchard MJ. The hepatitis B virus (HBV) HBx protein activates AKT to simultaneously regulate HBV replication and hepatocyte survival. J Virol. 2015; 89: 999-1012. doi: 10.1128/JVI.02440-14
36. Brechot C, Pourcel C, Louise A, Rain B, Tiollais P. Presence of integrated hepatitis B virus DNA sequences in cellular DNA of human hepatocellular carcinoma. Nature. 1980; 286: 533-5. doi: 10.1038/286533a0
37. Chakraborty PR, Ruiz-Opazo N, Shouval D, Shafritz DA. Identification of integrated hepatitis B virus DNA and expression of viral RNA in an HBsAg-producing human hepatocellular carcinoma cell line. Nature. 1980; 286: 531-3. doi: 10.1038/286531a0
38. Shafritz DA, Shouval D, Sherman HI, Hadziyannis SJ, Kew MC. Integration of hepatitis B virus DNA into the genome of liver cells in chronic liver disease and hepatocellular carcinoma. Studies in percutaneous liver biopsies and post-mortem tissue specimens. N Engl J Med. 1981; 305: 1067-73. doi: 10.1056/NEJM198110293051807
39. Hai H, Tamori A, Kawada N. Role of hepatitis B virus DNA integration in human hepatocarcinogenesis. World J Gastroenterol. 2014; 20: 6236-43. doi: 10.3748/wjg.v20.i20.6236
40. Sung WK, Zheng H, Li S, Chen R, Liu X, Li Y, Lee NP, Lee WH, Ariyaratne PN, Tennakoon C, Mulawadi FH, Wong KF, Liu AM, et al. Genome-wide survey of recurrent HBV integration in hepatocellular carcinoma. Nat Genet. 2012; 44: 765-9. doi: 10.1038/ng.2295
41. Saitta C, Tripodi G, Barbera A, Bertuccio A, Smedile A, Ciancio A, Raffa G, Sangiovanni A, Navarra G, Raimondo G, Pollicino T. Hepatitis B virus (HBV) DNA integration in patients with occult HBV infection and hepatocellular carcinoma. Liver Int. 2015; doi: 10.1111/liv.12807
42. Jiang Z, Jhunjhunwala S, Liu J, Haverty PM, Kennemer MI, Guan Y, Lee W, Carnevali P, Stinson J, Johnson S, Diao J, Yeung S, Jubb A, et al. The effects of hepatitis B virus integration into the genomes of hepatocellular carcinoma patients. Genome Res. 2012; 22: 593-601. doi: 10.1101/gr.133926.111
43. Murakami Y, Saigo K, Takashima H, Minami M, Okanoue T, Brechot C, Paterlini-Brechot P. Large scaled analysis of hepatitis B virus (HBV) DNA integration in HBV related hepatocellular carcinomas. Gut. 2005; 54: 1162-8. doi: 10.1136/gut.2004.054452
44. Paterlini-Brechot P, Saigo K, Murakami Y, Chami M, Gozuacik D, Mugnier C, Lagorce D, Brechot C. Hepatitis B virus-related insertional mutagenesis occurs frequently in human liver cancers and recurrently targets human telomerase gene. Oncogene. 2003; 22: 3911-6. doi: 10.1038/sj.onc.1206492
45. Wang K, Lim HY, Shi S, Lee J, Deng S, Xie T, Zhu Z, Wang Y, Pocalyko D, Yang WJ, Rejto PA, Mao M, Park CK, et al. Genomic landscape of copy number aberrations enables the identification of oncogenic drivers in hepatocellular carcinoma. Hepatology. 2013; doi: 10.1002/hep.26402
46. Sze KM, Chu GK, Lee JM, Ng IO. C-terminal truncated hepatitis B virus x protein is associated with metastasis and enhances invasiveness by C-Jun/matrix metalloproteinase protein 10 activation in hepatocellular carcinoma. Hepatology. 2013; 57: 131-9. doi: 10.1002/hep.25979
47. Toh ST, Jin Y, Liu L, Wang J, Babrzadeh F, Gharizadeh B, Ronaghi M, Toh HC, Chow PK, Chung AY, Ooi LL, Lee CG. Deep sequencing of the hepatitis B virus in hepatocellular carcinoma patients reveals enriched integration events, structural alterations and sequence variations. Carcinogenesis. 2013; 34: 787-98. doi: 10.1093/carcin/bgs406.
48. Lau CC, Sun T, Ching AK, He M, Li JW, Wong AM, Co NN, Chan AW, Li PS, Lung RW, Tong JH, Lai PB, Chan HL, et al. Viral-human chimeric transcript predisposes risk to liver cancer development and progression. Cancer Cell. 2014; 25: 335-49. doi: 10.1016/j.ccr.2014.01.030
49. Gouas D, Shi H, Hainaut P. The aflatoxin-induced TP53 mutation at codon 249 (R249S): biomarker of exposure, early detection and target for therapy. Cancer Lett. 2009; 286: 29-37. doi: 10.1016/j.canlet.2009.02.057
50. Kirk GD, Lesi OA, Mendy M, Szymanska K, Whittle H, Goedert JJ, Hainaut P, Montesano R. 249(ser) TP53 mutation in plasma DNA, hepatitis B viral infection, and risk of hepatocellular carcinoma. Oncogene. 2005; 24: 5858-67. doi: 10.1038/sj.onc.1208732
51. Gouas DA, Shi H, Hautefeuille AH, Ortiz-Cuaran SL, Legros PC, Szymanska KJ, Galy O, Egevad LA, bedi-Ardekani B, Wiman KG, Hantz O, Caron de FC, Chemin IA, et al. Effects of the TP53 p.R249S mutant on proliferation and clonogenic properties in human hepatocellular carcinoma cell lines: interaction with hepatitis B virus X protein. Carcinogenesis. 2010; 31: 1475-82. doi: 10.1093/carcin/bgq118
52. Chevaliez S, Pawlotsky JM. HCV Genome and Life Cycle. In: Tan S, editor. Hepatitis C Viruses: Genomes and Molecular Biology. Norfolk (UK): Horizon Bioscience; 2006.
53. Rehermann B, Nascimbeni M. Immunology of hepatitis B virus and hepatitis C virus infection. Nat Rev Immunol. 2005; 5: 215-29. doi: 10.1038/nri1573
54. Marcellin P, Asselah T, Boyer N. Fibrosis and disease progression in hepatitis C. Hepatology. 2002; 36: S47-S56. doi: 10.1053/jhep.2002.36993
55. Shimoda R, Nagashima M, Sakamoto M, Yamaguchi N, Hirohashi S, Yokota J, Kasai H. Increased formation of oxidative DNA damage, 8-hydroxydeoxyguanosine, in human livers with chronic hepatitis. Cancer Res. 1994; 54: 3171-2.
56. Protzer U, Maini MK, Knolle PA. Living in the liver: hepatic infections. Nat Rev Immunol. 2012; 12: 201-13. doi: 10.1038/nri3169
57. Selimovic D, El-Khattouti A, Ghozlan H, Haikel Y, Abdelkader O, Hassan M. Hepatitis C virus-related hepatocellular carcinoma: An insight into molecular mechanisms and therapeutic strategies. World J Hepatol. 2012; 4: 342-55. doi: 10.4254/wjh.v4.i12.342
58. Lemon SM, McGivern DR. Is hepatitis C virus carcinogenic? Gastroenterology. 2012; 142: 1274-8. doi: 10.1053/j.gastro.2012.01.045
59. Zemel R, Issachar A, Tur-Kaspa R. The role of oncogenic viruses in the pathogenesis of hepatocellular carcinoma. Clin Liver Dis. 2011; 15: 261-x. doi: 10.1016/j.cld.2011.03.001
60. Li HC, Ma HC, Yang CH, Lo SY. Production and pathogenicity of hepatitis C virus core gene products. World J Gastroenterol. 2014; 20: 7104-22. doi: 10.3748/wjg.v20.i23.7104
61. Kasprzak A, Adamek A. Role of hepatitis C virus proteins (C, NS3, NS5A) in hepatic oncogenesis. Hepatol Res. 2008; 38: 1-26. doi: 10.1111/j.1872-034X.2007.00261.x
62. Tsuchihara K, Hijikata M, Fukuda K, Kuroki T, Yamamoto N, Shimotohno K. Hepatitis C virus core protein regulates cell growth and signal transduction pathway transmitting growth stimuli. Virology. 1999; 258: 100-7. doi: 10.1006/viro.1999.9694
63. Giambartolomei S, Covone F, Levrero M, Balsano C. Sustained activation of the Raf/MEK/Erk pathway in response to EGF in stable cell lines expressing the Hepatitis C Virus (HCV) core protein. Oncogene. 2001; 20: 2606-10.
64. Erhardt A, Hassan M, Heintges T, Haussinger D. Hepatitis C virus core protein induces cell proliferation and activates ERK, JNK, and p38 MAP kinases together with the MAP kinase phosphatase MKP-1 in a HepG2 Tet-Off cell line. Virology. 2002; 292: 272-84. doi: 10.1006/viro.2001.1227
65. Hassan M, Ghozlan H, bdel-Kader O. Activation of RB/E2F signaling pathway is required for the modulation of hepatitis C virus core protein-induced cell growth in liver and non-liver cells. Cell Signal. 2004; 16: 1375-85. doi: 10.1016/j.cellsig.2004.04.005
66. Saito K, Meyer K, Warner R, Basu A, Ray RB, Ray R. Hepatitis C virus core protein inhibits tumor necrosis factor alpha-mediated apoptosis by a protective effect involving cellular FLICE inhibitory protein. J Virol. 2006; 80: 4372-9. doi: 10.1128/JVI.80.9.4372-4379.2006
67. Hassan M, Selimovic D, Ghozlan H, bdel-Kader O. Hepatitis C virus core protein triggers hepatic angiogenesis by a mechanism including multiple pathways. Hepatology. 2009; 49: 1469-82. doi: 10.1002/hep.22849
68. Basu A, Meyer K, Lai KK, Saito K, Di Bisceglie AM, Grosso LE, Ray RB, Ray R. Microarray analyses and molecular profiling of Stat3 signaling pathway induced by hepatitis C virus core protein in human hepatocytes. Virology. 2006; 349: 347-58. doi: doi:10.1016/j.virol.2006.02.023
69. Pez F, Lopez A, Kim M, Wands JR, Caron de FC, Merle P. Wnt signaling and hepatocarcinogenesis: molecular targets for the development of innovative anticancer drugs. J Hepatol. 2013; 59: 1107-17. doi: 10.1016/j.jhep.2013.07.001
70. Kwun HJ, Jung EY, Ahn JY, Lee MN, Jang KL. p53-dependent transcriptional repression of p21(waf1) by hepatitis C virus NS3. J Gen Virol. 2001; 82: 2235-41. doi: 10.1099/0022-1317-82-9-2235
71. Brenndorfer ED, Karthe J, Frelin L, Cebula P, Erhardt A, Schulte am EJ, Hengel H, Bartenschlager R, Sallberg M, Haussinger D, Bode JG. Nonstructural 3/4A protease of hepatitis C virus activates epithelial growth factor-induced signal transduction by cleavage of the T-cell protein tyrosine phosphatase. Hepatology. 2009; 49: 1810-20. doi: 10.1002/hep.22857
72. Mankouri J, Griffin S, Harris M. The hepatitis C virus non-structural protein NS5A alters the trafficking profile of the epidermal growth factor receptor. Traffic. 2008; 9: 1497-509. doi: 10.1111/j.1600-0854.2008.00779.x.
73. Reyes GR. The nonstructural NS5A protein of hepatitis C virus: an expanding, multifunctional role in enhancing hepatitis C virus pathogenesis. J Biomed Sci. 2002; 9: 187-97.
74. Milward A, Mankouri J, Harris M. Hepatitis C virus NS5A protein interacts with beta-catenin and stimulates its transcriptional activity in a phosphoinositide-3 kinase-dependent fashion. J Gen Virol. 2010; 91: 373-81. doi: 10.1099/vir.0.015305-0
75. Higgs MR, Lerat H, Pawlotsky JM. Hepatitis C virus-induced activation of beta-catenin promotes c-Myc expression and a cascade of pro-carcinogenetic events. Oncogene. 2013; 32: 4683-93. doi: 10.1038/onc.2012.484
76. Langelier MF, Pascal JM. PARP-1 mechanism for coupling DNA damage detection to poly(ADP-ribose) synthesis. Curr Opin Struct Biol. 2013; 23: 134-43. doi: 10.1016/j.sbi.2013.01.003
77. Takamizawa A, Mori C, Fuke I, Manabe S, Murakami S, Fujita J, Onishi E, Andoh T, Yoshida I, Okayama H. Structure and organization of the hepatitis C virus genome isolated from human carriers. J Virol. 1991; 65: 1105-13.
78. Chen X, Cheung ST, So S, Fan ST, Barry C, Higgins J, Lai KM, Ji J, Dudoit S, Ng IO, Van De RM, Botstein D, Brown PO. Gene expression patterns in human liver cancers. Mol Biol Cell. 2002; 13: 1929-39. doi: 10.1091/mbc.02-02-0023
79. Villanueva A, Newell P, Chiang DY, Friedman SL, Llovet JM. Genomics and signaling pathways in hepatocellular carcinoma. Semin Liver Dis. 2007; 27: 55-76. doi: 10.1055/s-2006-960171
80. Iizuka N, Oka M, Yamada-Okabe H, Mori N, Tamesa T, Okada T, Takemoto N, Tangoku A, Hamada K, Nakayama H, Miyamoto T, Uchimura S, Hamamoto Y. Comparison of gene expression profiles between hepatitis B virus- and hepatitis C virus-infected hepatocellular carcinoma by oligonucleotide microarray data on the basis of a supervised learning method. Cancer Res. 2002; 62: 3939-44.
81. Lee JS, Chu IS, Heo J, Calvisi DF, Sun Z, Roskams T, Durnez A, Demetris AJ, Thorgeirsson SS. Classification and prediction of survival in hepatocellular carcinoma by gene expression profiling. Hepatology. 2004; 40: 667-76. doi: 10.1002/hep.20375
82. Iizuka N, Oka M, Yamada-Okabe H, Nishida M, Maeda Y, Mori N, Takao T, Tamesa T, Tangoku A, Tabuchi H, Hamada K, Nakayama H, Ishitsuka H, et al. Oligonucleotide microarray for prediction of early intrahepatic recurrence of hepatocellular carcinoma after curative resection. Lancet. 2003; 361: 923-9. doi: 10.1016/S0140-6736(03)12775-4
83. Hoshida Y, Villanueva A, Kobayashi M, Peix J, Chiang DY, Camargo A, Gupta S, Moore J, Wrobel MJ, Lerner J, Reich M, Chan JA, Glickman JN, et al. Gene expression in fixed tissues and outcome in hepatocellular carcinoma. N Engl J Med. 2008; 359: 1995-2004. doi: 10.1056/NEJMoa0804525
84. De Giorgi V, Monaco A, Worchech A, Tornesello M, Izzo F, Buonaguro L, Marincola FM, Wang E, Buonaguro FM. Gene profiling, biomarkers and pathways characterizing HCV-related hepatocellular carcinoma. J Transl Med. 2009; 7: 85. doi: 10.1186/1479-5876-7-85
85. Sakamoto M, Mori T, Masugi Y, Effendi K, Rie I, Du W. Candidate molecular markers for histological diagnosis of early hepatocellular carcinoma. Intervirology. 2008; 51: 42-5. doi: 10.1159/000122603
86. De Giorgi V, Buonaguro L, Worschech A, Tornesello ML, Izzo F, Marincola FM, Wang E, Buonaguro FM. Molecular Signatures Associated with HCV-Induced Hepatocellular Carcinoma and Liver Metastasis. PLoS One. 2013; 8: e56153. doi: 10.1371/journal.pone.0056153
87. Zender L, Kubicka S. Molecular pathogenesis and targeted therapy of hepatocellular carcinoma. Onkologie. 2008; 31: 550-5. doi: 10.1159/000151586
88. Midorikawa Y, Sugiyama Y, Aburatani H. Molecular targets for liver cancer therapy: From screening of target genes to clinical trials. Hepatol Res. 2010; 40: 49-60. doi: 10.1111/j.1872-034X.2009.00583.x
89. Yang L, Ma Z, Wang D, Zhao W, Chen L, Wang G. MicroRNA-602 regulating tumor suppressive gene RASSF1A is overexpressed in hepatitis B virus-infected liver and hepatocellular carcinoma. Cancer Biol Ther. 2010; 9: 803-8. doi: 10.4161/cbt.9.10.11440
90. Zhang X, Liu S, Hu T, Liu S, He Y, Sun S. Up-regulated microRNA-143 transcribed by nuclear factor kappa B enhances hepatocarcinoma metastasis by repressing fibronectin expression. Hepatology. 2009; 50: 490-9. doi: 10.1002/hep.23008
91. Kong G, Zhang J, Zhang S, Shan C, Ye L, Zhang X. Upregulated microRNA-29a by hepatitis B virus X protein enhances hepatoma cell migration by targeting PTEN in cell culture model. PLoS One. 2011; 6: e19518. doi: 10.1371/journal.pone.0019518
92. Yuan K, Lian Z, Sun B, Clayton MM, Ng IO, Feitelson MA. Role of miR-148a in hepatitis B associated hepatocellular carcinoma. PLoS One. 2012; 7: e35331. doi: 10.1371/journal.pone.003533
93. Hung CH, Hu TH, Lu SN, Kuo FY, Chen CH, Wang JH, Huang CM, Lee CM, Lin CY, Yen YH, Chiu YC. Circulating microRNAs as biomarkers for diagnosis of early hepatocellular carcinoma associated with hepatitis B virus. Int J Cancer. 2015; doi: 10.1002/ijc.29802
94. Bui-Nguyen TM, Pakala SB, Sirigiri DR, Martin E, Murad F, Kumar R. Stimulation of inducible nitric oxide by hepatitis B virus transactivator protein HBx requires MTA1 coregulator. J Biol Chem. 2010; 285: 6980-6. doi: 10.1074/jbc.M109.065987
95. Place RF, Li LC, Pookot D, Noonan EJ, Dahiya R. MicroRNA-373 induces expression of genes with complementary promoter sequences. Proc Natl Acad Sci U S A. 2008; 105: 1608-13. doi: 10.1073/pnas.0707594105
96. Wang Y, Lu Y, Toh ST, Sung WK, Tan P, Chow P, Chung AY, Jooi LL, Lee CG. Lethal-7 is down-regulated by the hepatitis B virus x protein and targets signal transducer and activator of transcription 3. J Hepatol. 2010; 53: 57-66. doi: 10.1016/j.jhep.2009.12.043
97. Wei X, Xiang T, Ren G, Tan C, Liu R, Xu X, Wu Z. miR-101 is down-regulated by the hepatitis B virus x protein and induces aberrant DNA methylation by targeting DNA methyltransferase 3A. Cell Signal. 2013; 25: 439-46. doi: 10.1016/j.cellsig.2012.10.013
98. Huang J, Wang Y, Guo Y, Sun S. Down-regulated microRNA-152 induces aberrant DNA methylation in hepatitis B virus-related hepatocellular carcinoma by targeting DNA methyltransferase 1. Hepatology. 2010; 52: 60-70. doi: 10.1002/hep.23660
99. Xu X, Fan Z, Kang L, Han J, Jiang C, Zheng X, Zhu Z, Jiao H, Lin J, Jiang K, Ding L, Zhang H, Cheng L, et al. Hepatitis B virus X protein represses miRNA-148a to enhance tumorigenesis. J Clin Invest. 2013; 123: 630-45. doi: 10.1172/JCI64265
100. Wu G, Yu F, Xiao Z, Xu K, Xu J, Tang W, Wang J, Song E. Hepatitis B virus X protein downregulates expression of the miR-16 family in malignant hepatocytes in vitro. Br J Cancer. 2011; 105: 146-53. doi: 10.1038/bjc.2011.190
101. Shrivastava S, Steele R, Ray R, Ray RB. MicroRNAs: Role in Hepatitis C Virus pathogenesis. Genes Dis. 2015; 2: 35-45. doi: 10.1016/j.gendis.2015.01.001
102. Zhang C, Huys A, Thibault PA, Wilson JA. Requirements for human Dicer and TRBP in microRNA-122 regulation of HCV translation and RNA abundance. Virology. 2012; 433: 479-88. doi: 10.1016/j.virol.2012.08.039
103. Xu G, Yang F, Ding CL, Wang J, Zhao P, Wang W, Ren H. MiR-221 accentuates IFNs anti-HCV effect by downregulating SOCS1 and SOCS3. Virology. 2014; 462-463: 343-50. doi: 10.1016/j.virol.2014.06.024
104. Cheng M, Si Y, Niu Y, Liu X, Li X, Zhao J, Jin Q, Yang W. High-throughput profiling of alpha interferon- and interleukin-28B-regulated microRNAs and identification of let-7s with anti-hepatitis C virus activity by targeting IGF2BP1. J Virol. 2013; 87: 9707-18. doi: 10.1128/JVI.00802-13
105. Moinzadeh P, Breuhahn K, Stutzer H, Schirmacher P. Chromosome alterations in human hepatocellular carcinomas correlate with aetiology and histological grade--results of an explorative CGH meta-analysis. Br J Cancer. 2005; 92: 935-41. doi: 10.1038/sj.bjc.6602448
106. Longerich T, Mueller MM, Breuhahn K, Schirmacher P, Benner A, Heiss C. Oncogenetic tree modeling of human hepatocarcinogenesis. Int J Cancer. 2012; 130: 575-83. doi: 10.1002/ijc.26063
107. Guo X, Yanna, Ma X, An J, Shang Y, Huang Q, Yang H, Chen Z, Xing J. A meta-analysis of array-CGH studies implicates antiviral immunity pathways in the development of hepatocellular carcinoma. PLoS One. 2011; 6: e28404. doi: 10.1371/journal.pone.0028404
108. Chochi Y, Kawauchi S, Nakao M, Furuya T, Hashimoto K, Oga A, Oka M, Sasaki K. A copy number gain of the 6p arm is linked with advanced hepatocellular carcinoma: an array-based comparative genomic hybridization study. J Pathol. 2009; 217: 677-84. doi: 10.1002/path.2491
109. Patil MA, Gutgemann I, Zhang J, Ho C, Cheung ST, Ginzinger D, Li R, Dykema KJ, So S, Fan ST, Kakar S, Furge KA, Buttner R, et al. Array-based comparative genomic hybridization reveals recurrent chromosomal aberrations and Jab1 as a potential target for 8q gain in hepatocellular carcinoma. Carcinogenesis. 2005; 26: 2050-7. doi: 10.1093/carcin/bgi178
110. Kakar S, Chen X, Ho C, Burgart LJ, Adeyi O, Jain D, Sahai V, Ferrell LD. Chromosomal abnormalities determined by comparative genomic hybridization are helpful in the diagnosis of atypical hepatocellular neoplasms. Histopathology. 2009; 55: 197-205. doi: 10.1111/j.1365-2559.2009.03343.x
111. Schlaeger C, Longerich T, Schiller C, Bewerunge P, Mehrabi A, Toedt G, Kleeff J, Ehemann V, Eils R, Lichter P, Schirmacher P, Radlwimmer B. Etiology-dependent molecular mechanisms in human hepatocarcinogenesis. Hepatology. 2008; 47: 511-20. doi: 10.1002/hep.22033
112. Midorikawa Y, Yamamoto S, Tsuji S, Kamimura N, Ishikawa S, Igarashi H, Makuuchi M, Kokudo N, Sugimura H, Aburatani H. Allelic imbalances and homozygous deletion on 8p23.2 for stepwise progression of hepatocarcinogenesis. Hepatology. 2009; 49: 513-22. doi: 10.1002/hep.22698
113. Han ZG. Functional genomic studies: insights into the pathogenesis of liver cancer. Annu Rev Genomics Hum Genet. 2012; 13: 171-205. doi: 10.1146/annurev-genom-090711-163752
114. Chiang DY, Villanueva A, Hoshida Y, Peix J, Newell P, Minguez B, LeBlanc AC, Donovan DJ, Thung SN, Sole M, Tovar V, Alsinet C, Ramos AH, et al. Focal gains of VEGFA and molecular classification of hepatocellular carcinoma. Cancer Res. 2008; 68: 6779-88. doi: 10.1158/0008-5472.CAN-08-0742
115. Chung KY, Cheng IK, Ching AK, Chu JH, Lai PB, Wong N. Block of proliferation 1 (BOP1) plays an oncogenic role in hepatocellular carcinoma by promoting epithelial-to-mesenchymal transition. Hepatology. 2011; 54: 307-18. doi: 10.1002/hep.24372
116. Huang J, Sheng HH, Shen T, Hu YJ, Xiao HS, Zhang Q, Zhang QH, Han ZG. Correlation between genomic DNA copy number alterations and transcriptional expression in hepatitis B virus-associated hepatocellular carcinoma. FEBS Lett. 2006; 580: 3571-81. doi: 10.1016/j.febslet.2006.05.032
117. Jia D, Wei L, Guo W, Zha R, Bao M, Chen Z, Zhao Y, Ge C, Zhao F, Chen T, Yao M, Li J, Wang H, et al. Genome-wide copy number analyses identified novel cancer genes in hepatocellular carcinoma. Hepatology. 2011; 54: 1227-36. doi: 10.1002/hep.24495
118. Cheng IK, Ching AK, Chan TC, Chan AW, Wong CK, Choy KW, Kwan M, Lai PB, Wong N. Reduced CRYL1 expression in hepatocellular carcinoma confers cell growth advantages and correlates with adverse patient prognosis. J Pathol. 2010; 220: 348-60. doi: 10.1002/path.2644
119. Lee SA, Ho C, Roy R, Kosinski C, Patil MA, Tward AD, Fridlyand J, Chen X. Integration of genomic analysis and in vivo transfection to identify sprouty 2 as a candidate tumor suppressor in liver cancer. Hepatology. 2008; 47: 1200-10. doi: 10.1002/hep.22169
120. Xue W, Krasnitz A, Lucito R, Sordella R, Vanaelst L, Cordon-Cardo C, Singer S, Kuehnel F, Wigler M, Powers S, Zender L, Lowe SW. DLC1 is a chromosome 8p tumor suppressor whose loss promotes hepatocellular carcinoma. Genes Dev. 2008; 22: 1439-44. doi: 10.1101/gad.1672608
121. Woo HG, Park ES, Lee JS, Lee YH, Ishikawa T, Kim YJ, Thorgeirsson SS. Identification of potential driver genes in human liver carcinoma by genomewide screening. Cancer Res. 2009; 69: 4059-66. doi: 10.1158/0008-5472.CAN-09-0164
122. Roessler S, Long EL, Budhu A, Chen Y, Zhao X, Ji J, Walker R, Jia HL, Ye QH, Qin LX, Tang ZY, He P, Hunter KW, et al. Integrative genomic identification of genes on 8p associated with hepatocellular carcinoma progression and patient survival. Gastroenterology. 2012; 142: 957-66. doi: 10.1053/j.gastro.2011.12.039
123. Petitjean A, Mathe E, Kato S, Ishioka C, Tavtigian SV, Hainaut P, Olivier M. Impact of mutant p53 functional properties on TP53 mutation patterns and tumor phenotype: lessons from recent developments in the IARC TP53 database. Hum Mutat. 2007; 28: 622-9. doi: 10.1002/humu.20495
124. Vogelstein B, Lane D, Levine AJ. Surfing the p53 network. Nature. 2000; 408: 307-10. doi: 10.1038/35042675
125. Hosono S, Chou MJ, Lee CS, Shih C. Infrequent mutation of p53 gene in hepatitis B virus positive primary hepatocellular carcinomas. Oncogene. 1993; 8: 491-6.
126. Tanaka S, Toh Y, Adachi E, Matsumata T, Mori R, Sugimachi K. Tumor progression in hepatocellular carcinoma may be mediated by p53 mutation. Cancer Res. 1993; 53: 2884-7.
127. Okada T, Iizuka N, Yamada-Okabe H, Mori N, Tamesa T, Takemoto N, Tangoku A, Hamada K, Nakayama H, Miyamoto T, Uchimura S, Hamamoto Y, Oka M. Gene expression profile linked to p53 status in hepatitis C virus-related hepatocellular carcinoma. FEBS Lett. 2003; 555: 583-90. doi: 10.1016/S0014-5793(03)01345-0
128. Woo HG, Wang XW, Budhu A, Kim YH, Kwon SM, Tang ZY, Sun Z, Harris CC, Thorgeirsson SS. Association of TP53 mutations with stem cell-like gene expression and survival of patients with hepatocellular carcinoma. Gastroenterology. 2011; 140: 1063-70. doi: 10.1053/j.gastro.2010.11.034
129. Liu J, Ma Q, Zhang M, Wang X, Zhang D, Li W, Wang F, Wu E. Alterations of TP53 are associated with a poor outcome for patients with hepatocellular carcinoma: evidence from a systematic review and meta-analysis. Eur J Cancer. 2012; 48: 2328-38. doi: 10.1016/j.ejca.2012.03.001
130. Huang H, Fujii H, Sankila A, Mahler-Araujo BM, Matsuda M, Cathomas G, Ohgaki H. Beta-catenin mutations are frequent in human hepatocellular carcinomas associated with hepatitis C virus infection. Am J Pathol. 1999; 155: 1795-801. doi: 10.1016/S0002-9440(10)65496-X
131. Zucman-Rossi J, Benhamouche S, Godard C, Boyault S, Grimber G, Balabaud C, Cunha AS, Bioulac-Sage P, Perret C. Differential effects of inactivated Axin1 and activated beta-catenin mutations in human hepatocellular carcinomas. Oncogene. 2007; 26: 774-80. doi: 10.1038/sj.onc.1209824
132. de La Coste A, Romagnolo B, Billuart P, Renard CA, Buendia MA, Soubrane O, Fabre M, Chelly J, Beldjord C, Kahn A, Perret C. Somatic mutations of the beta-catenin gene are frequent in mouse and human hepatocellular carcinomas. Proc Natl Acad Sci U S A. 1998; 95: 8847-51. doi: PM 9671767
133. Taniguchi K, Roberts LR, Aderca IN, Dong X, Qian C, Murphy LM, Nagorney DM, Burgart LJ, Roche PC, Smith DI, Ross JA, Liu W. Mutational spectrum of beta-catenin, AXIN1, and AXIN2 in hepatocellular carcinomas and hepatoblastomas. Oncogene. 2002; 21: 4863-71. doi: 10.1038/sj.onc.1205591
134. Li M, Zhao H, Zhang X, Wood LD, Anders RA, Choti MA, Pawlik TM, Daniel HD, Kannangai R, Offerhaus GJ, Velculescu VE, Wang L, Zhou S, et al. Inactivating mutations of the chromatin remodeling gene ARID2 in hepatocellular carcinoma. Nat Genet. 2011; 43: 828-9. doi: 10.1038/ng.903
135. Fujimoto A, Totoki Y, Abe T, Boroevich KA, Hosoda F, Nguyen HH, Aoki M, Hosono N, Kubo M, Miya F, Arai Y, Takahashi H, Shirakihara T, et al. Whole-genome sequencing of liver cancers identifies etiological influences on mutation patterns and recurrent mutations in chromatin regulators. Nat Genet. 2012; 44: 760-4. doi: 10.1038/ng.2291
136. Imbeaud S, Ladeiro Y, Zucman-Rossi J. Identification of novel oncogenes and tumor suppressors in hepatocellular carcinoma. Semin Liver Dis. 2010; 30: 75-86. doi: 10.1055/s-0030-1247134
137. Huang J, Deng Q, Wang Q, Li KY, Dai JH, Li N, Zhu ZD, Zhou B, Liu XY, Liu RF, Fei QL, Chen H, Cai B, et al. Exome sequencing of hepatitis B virus-associated hepatocellular carcinoma. Nat Genet. 2012; 44: 1117-21. doi: 10.1038/ng.2391
138. Totoki Y, Tatsuno K, Covington KR, Ueda H, Creighton CJ, Kato M, Tsuji S, Donehower LA, Slagle BL, Nakamura H, Yamamoto S, Shinbrot E, Hama N, et al. Trans-ancestry mutational landscape of hepatocellular carcinoma genomes. Nat Genet. 2014; 46: 1267-73. doi: 10.1038/ng.3126
139. Ahn SM, Jang SJ, Shim JH, Kim D, Hong SM, Sung CO, Baek D, Haq F, Ansari AA, Lee SY, Chun SM, Choi S, Choi HJ, et al. Genomic portrait of resectable hepatocellular carcinomas: implications of RB1 and FGF19 aberrations for patient stratification. Hepatology. 2014; 60: 1972-82. doi: 10.1002/hep.27198
140. Schulze K, Imbeaud S, Letouze E, Alexandrov LB, Calderaro J, Rebouissou S, Couchy G, Meiller C, Shinde J, Soysouvanh F, Calatayud AL, Pinyol R, Pelletier L, et al. Exome sequencing of hepatocellular carcinomas identifies new mutational signatures and potential therapeutic targets. Nat Genet. 2015; 47: 505-11. doi: 10.1038/ng.3252
141. Nault JC, Calderaro J, Di TL, Balabaud C, Zafrani ES, Bioulac-Sage P, Roncalli M, Zucman-Rossi J. Telomerase reverse transcriptase promoter mutation is an early somatic genetic alteration in the transformation of premalignant nodules in hepatocellular carcinoma on cirrhosis. Hepatology. 2014; doi: 10.1002/hep.27372
142. Nault JC, Mallet M, Pilati C, Calderaro J, Bioulac-Sage P, Laurent C, Laurent A, Cherqui D, Balabaud C, Zucman-Rossi J. High frequency of telomerase reverse-transcriptase promoter somatic mutations in hepatocellular carcinoma and preneoplastic lesions. Nat Commun. 2013; 4: 2218. doi: 10.1038/ncomms3218
143. Yago M, Ohki R, Hatakeyama S, Fujita T, Ishikawa F. Variant forms of upstream stimulatory factors (USFs) control the promoter activity of hTERT, the human gene encoding the catalytic subunit of telomerase. FEBS Lett. 2002; 520: 40-6. doi: 10.1016/S0014-5793(02)02757-6
144. Greenwald RJ, Freeman GJ, Sharpe AH. The B7 family revisited. Annu Rev Immunol. 2005; 23: 515-48. doi: 10.1146/annurev.immunol.23.021704.115611
145. Buonaguro L, Tagliamonte M, Petrizzo A, Damiano E, Tornesello ML, Buonaguro FM. Cellular prognostic markers in hepatocellular carcinoma. Future Oncol. 2015; 11: 1591-8. doi: 10.2217/fon.15.39
146. Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, Havel JJ, Lee W, Yuan J, Wong P, Ho TS, Miller ML, Rekhtman N, Moreira AL, et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science. 2015; 348: 124-8. doi: 10.1126/science.aaa1348
147. Le DT, Uram JN, Wang H, Bartlett BR, Kemberling H, Eyring AD, Skora AD, Luber BS, Azad NS, Laheru D, Biedrzycki B, Donehower RC, Zaheer A, et al. PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N Engl J Med. 2015; 372: 2509-20. doi: 10.1056/NEJMoa1500596
148. Champiat S, Ferte C, Lebel-Binay S, Eggermont A, Soria JC. Exomics and immunogenics: Bridging mutational load and immune checkpoints efficacy. Oncoimmunology. 2014; 3: e27817. doi: 10.4161/onci.27817
149. Buonaguro L, Petrizzo A, Tagliamonte M, Tornesello ML, Buonaguro FM. Challenges in cancer vaccine development for hepatocellular carcinoma. J Hepatol. 2013; 59: 897-903. doi: 10.1016/j.jhep.2013.05.031
150. Yadav M, Jhunjhunwala S, Phung QT, Lupardus P, Tanguay J, Bumbaca S, Franci C, Cheung TK, Fritsche J, Weinschenk T, Modrusan Z, Mellman I, Lill JR, et al. Predicting immunogenic tumour mutations by combining mass spectrometry and exome sequencing. Nature. 2014; 515: 572-6. doi: 10.1038/nature14001
151. Gubin MM, Zhang X, Schuster H, Caron E, Ward JP, Noguchi T, Ivanova Y, Hundal J, Arthur CD, Krebber WJ, Mulder GE, Toebes M, Vesely MD, et al. Checkpoint blockade cancer immunotherapy targets tumour-specific mutant antigens. Nature. 2014; 515: 577-81. doi: 10.1038/nature13988
152. Snyder A, Makarov V, Merghoub T, Yuan J, Zaretsky JM, Desrichard A, Walsh LA, Postow MA, Wong P, Ho TS, Hollmann TJ, Bruggeman C, Kannan K, et al. Genetic basis for clinical response to CTLA-4 blockade in melanoma. N Engl J Med. 2014; 371: 2189-99. doi: 10.1056/NEJMoa1406498
153. van Rooij N, van Buuren MM, Philips D, Velds A, Toebes M, Heemskerk B, van Dijk LJ, Behjati S, Hilkmann H, El AD, Nieuwland M, Stratton MR, Kerkhoven RM, et al. Tumor exome analysis reveals neoantigen-specific T-cell reactivity in an ipilimumab-responsive melanoma. J Clin Oncol. 2013; 31: e439-e442. doi: 10.1200/JCO.2012.47.7521
154. Diehl L, Schurich A, Grochtmann R, Hegenbarth S, Chen L, Knolle PA. Tolerogenic maturation of liver sinusoidal endothelial cells promotes B7-homolog 1-dependent CD8+ T cell tolerance. Hepatology. 2008; 47: 296-305. doi: 10.1002/hep.21965
155. Carambia A, Frenzel C, Bruns OT, Schwinge D, Reimer R, Hohenberg H, Huber S, Tiegs G, Schramm C, Lohse AW, Herkel J. Inhibition of inflammatory CD4 T cell activity by murine liver sinusoidal endothelial cells. J Hepatol. 2013; 58: 112-8. doi: 10.1016/j.jhep.2012.09.008
156. Sangro B, Gomez-Martin C, de la MM, Inarrairaegui M, Garralda E, Barrera P, Riezu-Boj JI, Larrea E, Alfaro C, Sarobe P, Lasarte JJ, Perez-Gracia JL, Melero I, et al. A clinical trial of CTLA-4 blockade with tremelimumab in patients with hepatocellular carcinoma and chronic hepatitis C. J Hepatol. 2013; 59: 81-8. doi: 10.1016/j.jhep.2013.02.022
157. El-Khoueiry AB, Melero I, Crocenzi TS, Welling TH, Cheung Yau T, Yeo W, Chopra A, Grosso J, Lang L, Anderson J, Dela Cruz CM, Sangro B. Phase I/II safety and antitumor activity of nivolumab in patients with advanced hepatocellular carcinoma (HCC): CA209-040. J Clin Oncol. 2015; 33: Abstr LBA101

