Introduction
LIN28A, a highly conserved RNA-binding protein, plays an important role in inducing pluripotent stem cells, regulating development, cell growth, and metabolism [1-4]. Emerging evidence has shown that LIN28A is over-expressed in a variety of human tumors including oesophagus cancer [5], oral squamous cancer [6], colon cancer [7], and epithelial ovarian cancer [8]. Furthermore, LIN28A-positive tumors are poorly differentiated and more aggressive [5, 9, 10]. However, the profile of LIN28A expression remains to be clarified.
DNA methylation, an epigenetic regulation way, controls gene expression by recruiting proteins involved in transcriptional repression [11]. Previous studies have identified that Methyl-CpG binding proteins (MeCPs) recruit chromatin remodelers, histone deacetylases, and methylases to methylated DNA, leading to transcriptional repression [12]. For example, transcriptional repressor Methyl-CpG-binding Protein 2 (MeCP2) and Methyl-CpG-binding domain 2 (MBD2) play a crucial role in CpG island methylation both in vivo and in vitro [13-17]. Whether the LIN28A expression underlies epigenetic regulation mechanism remains to be clarified in pancreatic cancer cells.
In this study, we found that LIN28A expression had significant difference in pancreatic cancer cells, and was associated with the methylation status of two CpG islands sites. MeCP2 bound preferentially to the hypermethylated CpG islands to suppress LIN28A expression. We also found that LIN28A was critical for the stemness maintenance and invasion of pancreatic cancer cells. These findings for the first time prove that LIN28A expression is associated with methylation status of CpG islands, and may play a crucial role in pancreatic cancer progression.
Results
LIN28A Expression in different types of pancreatic cancer cell lines
It has been reported that LIN28A expression are reactivated in human cancers [10, 18, 19]. However, the LIN28A expression profile in pancreatic cancer cells is still unknown. We analyzed the LIN28A expression in BxPC3, PANC1, SW1990 and PaTu8988 cells using real-time PCR and western blot. The results showed that LIN28A expression, at both mRNA and protein levels, was higher in PANC1 cells than that in three other cells (Figure 1A, 1B). As LIN28A is associated with the differentiation of cancer cells, we evaluated the markers of stem cells OCT4, SOX2 and NANOG, and found that their expression in PANC1 cells was higher than that of the other cells (Figure 1C, S1B), indicating that PANC1 cells possess more poor differentiation state, which is consistent with previous studies in other tumor types. Moreover, we also found that PANC1 cells were more invasive among the above cells (Figure 1D).
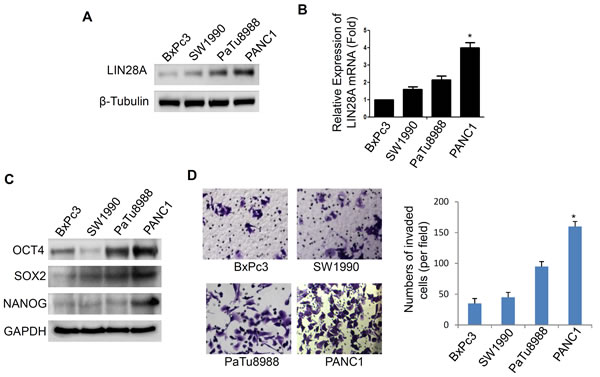
Figure 1: LIN28A expression in pancreatic cancer cells. Relative expression levels of LIN28A protein A. and mRNA B. were assessed in BxPC3, SW1990, PaTu8988, and PANC1 cells. The stem cell makers SOX2, OCT4 and NANOG were determined by western blotting C. and the abilities of invasiveness were examined using transwell assay D. Scare bar = 50µm.
Methylation status of the LIN28A CpG islands in pancreatic cancer cells
Although LIN28A plays important roles in many kinds of tumor cells, the mechanism underlying LIN28A different expression pattern is unclear. Since methylation status of CpG within proximal promoters is often associated with transcriptional silencing, we first analyzed the predictable CpG islands of LIN28A promoter using the MethPrimer software. The criteria are: Island size > 100, GC Percent >50.0, Obs/Exp (Observed/Expected number of CpG patterns) ratio > 0.6. The first CpG islands were identified in the first exon from -79 bp to +98 bp, and the second CpG islands were in the first intron from +139 bp to +406 bp (Figure 2A). Therefore, we examined the methylation status of both sites in pancreatic cancer cells using bisulfite sequencing. The results indicated that both sites had different methylation rates in SW1990, PaTu8988, and PANC1 cells, with 86.15%±3.5%, 98.46%±1.5%, and 67.69%±2.5%, respectively at the first site; as well as 83.33%±1.5%, 92.85%±2.5%, and 74.60%±3% at the second site (Figure 2B-2D). Obviously, the methylation levels of both sites in PANC1 cells were lower than the other two cells, supporting the hypothesis of LIN28A epigenetic silencing via CpG islands hypermethylation.
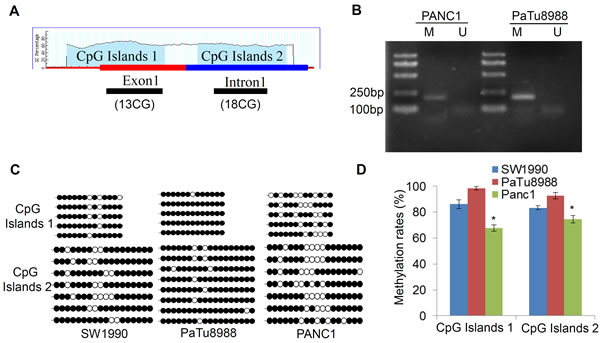
Figure 2: Aberrant methylation at LIN28A CpG islands in pancreatic cancer cells. A. Schematic map of LIN28A CpG islands, Two CpG islands sequences locates at human LIN28A from -79 to +98 and from +139 to +406 based on the MethyPrimer Software. B. Bisulfite-treated genomic DNA from PaTu8988 and PANC1 cells was used for PCR amplification by specific primers. M: products (175bp) generated by primers specific for methylated DNA; U: products (173bp) generated by primers specific for unmethylated DNA. C. The methylation status of the individual CpG dinucleotides at human LIN28A CpG islands was analyzed by bisulfite sequencing in SW1990, PaTu8988, and PANC1cells. The results are shown by methylated (black) or unmethylated (white) circles. The methylation rates of both LIN28A CpG islands in SW1990, PaTu8988, and PANC1 cells were shown in D. Data are presented as mean±SD.*, P < 0.05.
Re-activation of LIN28A expression by 5-Aza-CdR
To further evaluate the role of CpG islands methylation in LIN28A expression, we subsequently treated pancreatic cancer cells with the methyltransferase inhibitor 5-Aza-2’-deoxycytidine (5-Aza-CdR). The results indicated that 5-Aza-CdR could, to different extent, induce LIN28A expression at both protein and mRNA levels in a dose-dependent manner (Figure 3A-3D). As expected, in PaTu8988 cells with higher methylation levels of CpG islands, LIN28A mRNA expression was increased over 12-fold, while only about 6-fold or 3-fold in SW1990 or in PANC1 cells, respectively (Figure 3A). Such different inductions by 5-Aza-CdR were consistent with their methylation statuses in LIN28A CpG islands sites. It is indirectly suggested that the higher CpG island methylation level may play a crucial role in suppressing LIN28A expression.
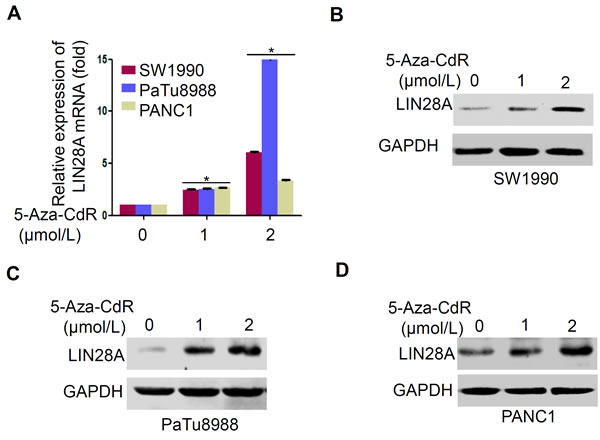
Figure 3: 5-Aza-CdR re-activates LIN28A expression in pancreatic cancer cells. A. Pancreatic cancer cells were treated with 5-Aza-CdR in 0, 1, 2 µmol/L for 3 days with the medium changed every day. LIN28A mRNA or protein expression was determined using real-time PCR and western blot, respectively in SW1990 B., PaTu8988 C., and PANC1 D. cells. GAPDH was used as a control. Data are presented as mean±SD.*, P < 0.05.
MeCP2 reads the methyl-CpG islands to suppress LIN28A expression
Methyl-CpG binding domain (MBD) proteins recruit repressing proteins to the methylated DNA, leading to transcriptional suppression. Previous studies identify that MeCP2 and MBD2, specifically binding to a single methyl-CpG pair, are the founding members of the MBD protein family [21], while MBD3 is found at unmethylated CpG islands and active promoters [22]. To determine whether MeCP2 or MBD2 contributes to promoter CpG island methylation-mediated suppression of LIN28A expression, we firstly detected the expression levels of MeCP2, MBD2 and MBD3 in the above three cells, and found that expression of MeCP2 and MBD3 was higher in PANC1 cells compared with the other two cells, while MBD2 was weakly expressed only in PaTu8988 cells (Figure 4A). It is implied that MeCP2 might be a methyl-CpG readers involved in suppression of LIN28A expression. Therefore, we performed ChIP assays to examine the readers in PaTu8988 and PANC1 cells using MeCP2 antibody, and MBD3 antibody as a negative control. Expectedly, MeCP2 could bind to hypermethylated CpG islands in LIN28A promoter, but weakly MBD3 (Figure 4B-4D) in three above cells. The analysis of relative quantitation was shown in (Figure 4E). Our data further confirmed the previous MeCPs reading pattern, and suggested that MeCP2, as methyl-CpG readers to LIN28A promoter, might be strongly involved in LIN28A epigenetic silencing in pancreatic cancer cells.
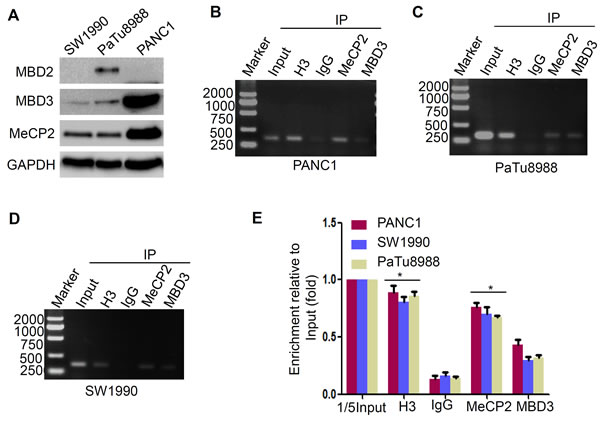
Figure 4: MeCP2 binds to methylated-CpG islands to suppress LIN28A expression. A. Western blotting was used to analyze the endogenous MeCP2, MBD2 and MBD3 protein expressions in indicated pancreatic cancer cells. B. Cross-linked chromatins from indicated pancreatic cancer cells were incubated with antiserum against H3, IgG, MBD3, and MeCP2. DNA extracted from each immunopecipitate was analyzed by standard PCR B.-D. or Real-time PCR E. with primers specific for LIN28A CpG islands 2. Chromatin taken before immunopecipitatipn was used as ‘Input’ controls. The ‘Input’ diluted 5 times was used as a control. Each bar represents the mean±SD of three independent assays.
MeCP2 regulates LIN28A transcriptional suppression
To further confirm the role of MeCP2, as a methyl-CpG reader, in epigenetic regulation of LIN28A expression, we transfected PaTu8988 and PANC1 cells with sh-MeCP2 or sh-MBD3 to examine LIN28A expression at both protein and mRNA levels, and sh-GFP as a control. Actually, MeCP2 knockdown significantly resulted in up-regulation of LIN28A expression in PANC1 and PaTu8988 cells compare to control group (Figure 5A, 5B), while MBD3 knockdown had no effect on LIN28A expression (Figure 5C, 5D). This further confirmed that MeCP2 contributed to epigenetic regulation of LIN28A expression in pancreatic cancer cells.
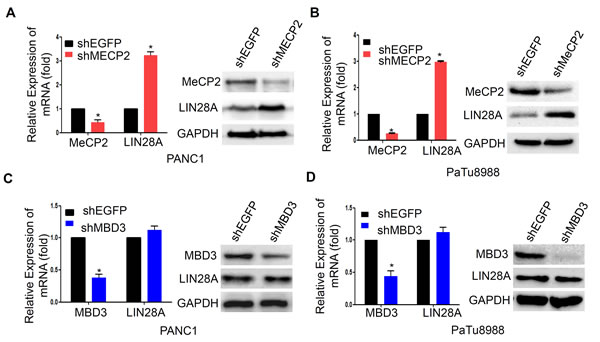
Figure 5: MeCP2 knockdown re-activates LIN28A expression. LIN28A mRNA and protein levels were detected by Real-time PCR or western blotting in PANC1 A. and PaTu8988 B. cells transfected with sh-MeCP2 or sh-EGFP plasmids, as well as MBD3 knockdown in PANC1 C. and PaTu8988 cells D. Data are presented as mean±SD. *, P < 0.05.
LIN28A improves the ability of stemness maintenance in pancreatic cancer cells
To clarify whether LIN28A expression is associated with stemness maintenance of pancreatic cancer cells, LIN28A or Vector were overexpressed in PaTu8988 and SW1990 cells, we found that LIN28A promoted OCT4, NANOG, LIN28B and c-myc expression (Figure 6A), as well as the sphere formation and colony-forming ability (Figure 6B and Figure S2). In contrast, Knocking down LIN28A resulted in the opposite effects in PANC1 cells (Figure 6C and Figure S2), suggesting that LIN28A was significantly involved in the stemness maintenance of pancreatic cancer cells.
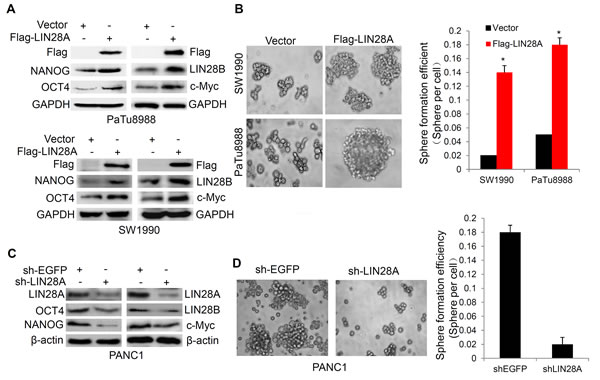
Figure 6: LIN28A promotes the expression of stem cell makers and sphere formation in pancreatic cancer cells. A. The stem cell makers OCT4, NANOG, LIN28B and c-myc expression were examined using western blotting in PaTu8988 and SW1990 cells transfected with Flag-LIN28A or Vector plasmids, and sphere formation efficiencies were shown in B. C. PANC1 cells were transfected with sh-LIN28A or sh-EGFP plasmids, and the stem cell makers OCT4, NANOG, LIN28B and c-myc expression were examined using western blotting, and sphere formation efficiencies were shown in D. Data are presented as mean±SD. *, P < 0.05.
LIN28A enhances the invasion ability of pancreatic cancer cells
To assess the role of LIN28A in human pancreatic cancer cells, we further examined the abilities of invasion by transwell assays. PaTu8988 and SW1990 cells were transfected with Flag-LIN28A or Vector plasmids, and PANC1 cells with sh-LIN28A or sh-GFP plasmids for 72h. We found that LIN28A overexpression significantly promoted the invasion ability of PaTu8988 and SW1990 cells (Figure 7A, 7B), whereas LIN28A knockdown inhibited the invasion ability of PANC1 cells (Figure S3A, B). Also, LIN28A overexpression resulted in upregulation of MMP2 and MMP9 (Figure 7C, 7D), while LIN28A knockdown downregulated the expression of MMP2 and MMP9 in PANC1 cells (Figure S3 C, D), indicating that LIN28A might be critical for invasion of pancreatic cancer cells.
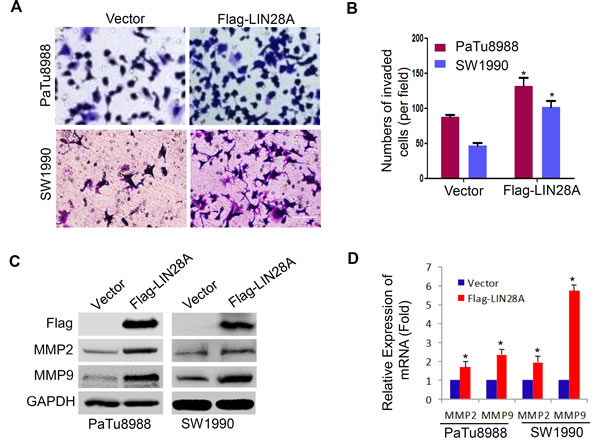
Figure 7: LIN28A promotes the invasiveness in pancreatic cancer cells. A. The abilities of invasiveness were examined using transwell assay in PaTu8988 and SW1990 cells transfected with Flag-LIN28A or Vector plasmids. Invaded cells were counted and analyzed in B. MMP2 and MMP9 were determined using western blotting C. and real-time PCR D. in above cells. Data shown are mean±SD of three triplicate measures. *, P < 0.05.
Discussion
Herein, we identify that LIN28A is required for the differentiation of pancreatic cancer cells, and promotes their growth and invasion. Mechanically, we for the first time identify that LIN28A expression is associated with CpG islands methylation status of its promoter, and epigenetically regulated by MeCP2, a methyl-CpG reader, in pancreatic cancer cells. These findings suggest that LIN28A might be a candidate of pancreatic cancer biomarkers.
The pluripotency factors, such as NANOG, SOX2, OCT4, and LIN28A, suggest a close relationship between tumor formation and the process of reprogramming mature cells to a more primitive type. LIN28A and LIN28B are specifically activated in the subset of tumors that are poorly differentiated and carry the worst prognosis [23]. In this study, we find that LIN28A increases the expression of stem cell markers SOX2, OCT4, LIN28B, c-Myc and NANOG in pancreatic cancer cells, and promotes their malignant behaviors including growth and invasion in vitro, further supporting the oncogenic potential of LIN28A. Previous studies have revealed that c-Myc- LIN28A/LIN28B forms a positive feedback loop [4, 24, 25]; it is noted whether the other pluripotency factors such as LIN28A-self are involved in such positive feedback loop.
Genetic alterations and epigenetic alterations are the main features in the pathogenesis of pancreatic cancer. In epigenetics, it has been proposed to define the differential methylated genes between pancreatic cancer cells and normal pancreatic tissues [26]. In this study, we at the first time find that LIN28A expression is associated with methylation status of the LIN28A promoter-associated CpG islands. It has reported that direct binding of MeCPs to the methylated DNA plays a crucial role in gene suppressing [27, 28]. Previous studies identify that MeCP2 and MBD2, specifically binding to a single methyl-CpG pair, are the founding members of the MBD protein family [21], while MBD3 is found at unmethylated CpG islands and active promoters [22]. Our ChIP assays reveal that MeCP2 effectively binds to hypermethylated LIN28A CpG islands, rather than MBD3, especially in PaTu8988 cells. Furthermore, the LIN28A transcriptional activity is restored after MeCP2 knockdown in both PaTu8988 and PANC1 cells, but not silencing of MBD3. These findings confirm that MeCP2 suppresses LIN28A transcriptional activity via reading methylated CpG islands.
In summary, our data reveals that high expression of LIN28A may be related to the malignant behaviors of pancreatic cancers, and MeCP2 directly binds to Methylated-CpG islands for LIN28A transcriptional suppression in pancreatic cancer cells. These findings suggest that LIN28A might be an important therapeutic target candidate to pancreatic cancers.
Materials and methods
Cell culture
The pancreatic cancer cell lines BxPC3, SW1990, PaTu8988 and PANC1 were kindly provided by Second Military Medical University in shanghai, and cultured with DMEM (Hyclone, China) supplemented with 10% fetal bovine serum (Gibco, USA), 100mg/L penicillin, and 100mg/L streptomycin, in humidified 5% CO2 incubator at 37 °C.
Real-time PCR
Total RNA was isolated using RNAiso Plus (Takara). Reverse transcription was performed using RevertAid First Strand cDNA Synthesis Kit (Thermo) according to the manufacturer’s recommendations. The SYBR green-based Real-time PCR was then performed in triplicate using CFX-96 Sequence Detection System (Bio-Rad) and gene expression was normalized by GAPDH. Primers were list in Table 1. The relative fold change in RNA expression was calculated using the 2-ΔΔCt method.
Western blotting
The cultured cells were rinsed with cold PBS before treated with RIPA lysis buffer at 4°C for 10 min. Then the mixture was centrifuged under 4°C at 12000r/min for 15 min. The supernatant was removed and the protein concentration was measured with BCA method. About 40 μg of protein was loaded each lane, and separated by 10% SDS-PAGE and then transferred to the PVDF membrane. The membrane was blocked by 5% non-fat milk powder for 1 h at room temperature before overnight incubation with primary antibodies 4°C, followed by the secondary antibody. The antibodies were rabbit anti-LIN28A (Cell Signaling, CAT 8641), rabbit anti-MeCP2 (Cell Signaling, CAT 3456), mouse anti-LIN28B (Cell Signaling, CAT 5422), mouse anti-β-Tubulin (Cell Signaling, CAT 6181), rabbit anti-OCT4 (Cell Signaling, CAT 2840), rabbit anti-SOX2 (Cell Signaling, CAT 3579), rabbit anti-NANOG (Cell Signaling, CAT 4903), rabbit anti-GAPDH (Cell Signaling, CAT 3683), rabbit anti-MBD3 (Cell Signaling, CAT 3896), rabbit anti-c-Myc (Cell Signaling, CAT 5605), mouse anti-β-actin (Cell Signaling, CAT 12262), mouse anti-MBD2 (Abcam, CAT ab45027), mouse anti-Flag (SIGMA, CAT F1804), rabbit anti-MMP2 (ImmunoWay, CAT YT2798), rabbit anti-MMP9 (ImmunoWay, CAT YT1892).
Methylation analysis of LIN28A CpG islands
According to the MethyPrimer software, two potential CpG islands were predicted. The first CpG islands were identified in the LIN28A promoter region and the first exon from -79 bp to +98 bp, and the second CpG islands were in the first intron from +139 bp to +406 bp. We determined the methylation status of LIN28A CpG islands by bisulfite-sequencing. The genomic DNA was extracted from SW1990, PaTu8988, and PANC1 cells using TIANamp Genomic DNA kit (TIANGEN, CAT DP304) according to the manufacturer’s recommendation, and 500 ng genomic DNA was treated with sodium bisulfite using EpiTect Bisulfite kit (QIAGEN, CAT 59104). 80-100 ng bisulfite-treated DNA was used for PCR amplification. The methylation status of the LIN28A CpG islands was determined by BSP using the specific primers designed according to the online primer program MethPrimer (http://www.urogene.org/ methprimer/). Primer sequences were provided in Table 1. The length of the amplified sequence was 240 bp containing 13 CG sites, 267 bp, containing 18 CG sites, respectively. The total volume for BS-PCR was 10µl, with 5μl methylated DNA. The reaction procedure was as follows: 95°C 5 min, (95 °C 30s, 53°C 30s, 72 °C 30s) × 40 cycles, 72 °C 10 min. The BSP products were cloned into pMD19-T vector (TaKaRa, CAT D102A) and then transformed in DH5α cells for clonal analyses. Each individual clone was sequenced by Sangon Company.
5-Aza-CdR treatment
The cells (SW1990, PaTu8988, and PANC1) were treated wi 5-Aza-CdR (Sigma, CAT A3656) in different doses (0, 1 and 2µmol/L) for 72h.
Chromatin Immunoprecipitation (ChIP) assay
ChIP was performed using SimpleChIP Plus Enzymatic chromatin IP Kit (Agarose Beads) (Cell Signaling, CAT9004) according to the manufacturer′s recommendations. Cells were fixed with formaldehyde and lysed, and chromatin was fragmented by partial digestion with Micrococcal Nuclease tomatic fragmentation to obtain chromatin fragments of 1 to 5 nucleosomes. Chromatin immunoprecipitations were performed using the MBDs, 10 μl of MeCP2, MBD3 antibodies and ChIP-Grade Protein G Agarose Beads. After reversal of protein-DNA cross-links, the DNA was purified using DNA purification spin columns.
ChIP-qPCR
The enrichment of particular DNA sequences of SW1990, PaTu8988, and PANC1 cells were analyzed by quantitative PCR. The reaction mixture contains 2×SYBR-Green reaction mix 10µl, 5µM primers 2µl, nuclease-free water 6µl and the appropriate DNA sample 2µl. The 2% input chromatin DNA was diluted 5 times. Reactions were carried out in a thermal cycler under the following conditions: 95 °C for 3 min followed by 95 °C for 15 s, 60 °C for 60 s for 40 cycles.
Plasmid construction
The entire LIN28A sequence was amplified with RT-PCR using primers LIN28A-all-F and LIN28A-all-R (Table 1), and then cloned into the expression vector p3xFLAG-Myc-CMV™-24 (Sigma). The MeCP2, MBD3 and EGFP shRNA oligos (Table 1, Sangon Biotech., Shanghai) were firstly annealed into double strands and then cloned into pLKO.1-puro-vector (Sigma).
Table 1: DNA and RNA nucleotide sequences
LIN28-F |
CGCTCGACCCCCCAGTGGATG |
LIN28-R |
TGGGGTGGCAGCTTGCATTCC |
GAPDH-F |
TGGGGAAGGTGAAGGTCGG |
GAPDH-R |
CTGGAAGATGGTGATGGGA |
MeCP2-F |
CAGCGTCTGCAAAGAGGAGA |
MeCP2-R |
GCTCCTCTCTGTTTGGCCTT |
BSP-F1 |
GGGGGAAGATGTAGTAGTTTTTTTT |
BSP-R1 |
CCTTTAAACAACCTAAAACTCAATTC |
BSP-F2 |
TTAGGTTGTTTAAAGGATTTTAAGA |
BSP-R2 |
AAACAACTACCAAACAACAAAAAAAA |
ChIP-PCR-F |
GAACCAACCCTTTGC |
ChIP-PCR-R |
AAAGTGTCCCCGCTAAGTCC |
LIN28-all-F |
CGGGGTACCGGCTCCGTGTCCAACCAG |
LIN28-all-R |
CGCGGATCCATTCTGTGCCTCCGGGAG |
ShMeCP2-F |
CCGGCGTCTGCAAAGAGGAGAAGATCTCGAGATCTTCTCCTCTTTGCAGACGTTTTTG |
ShMeCP2-R |
AATTCAAAAACGTCTGCAAAGAGGAGAAGATCTCGAGATCTTCTCCTCTTTGCAGACG |
SheGFP-F |
CCGGTACAACAGCCACAACGTCTATCTCGAGATAGACGTTGTGGCTGTTGTATTTTTG |
SheGFP-R |
AATTCAAAAATACAACAGCCACAACGTCTATCTCGAGATAGACGTTGTGGCTGTTGTA |
Transfection
Cells were transfected with plasmid DNA or shRNAs using lipofectamineTM2000 Reagent (Invitrogen) following the manufacturer’s protocol.
Colony-forming assay
Transfected PANC1, PaTu8988 and SW1990 cells were harvested, resuspended in medium and transferred to the six well plate (500, 1000, 1500 cells per well) for 10-14 days until large colonies were visible. Colonies were fixed fixed and stained with 0.05% crystal violet for 30min, and the number of colonies was counted or photomicrographs were taken under phase-contrast microscope.
Sphere formation assay
The sphere formation assay was performed by plating dissociated single cells at a density of 1 cell/µl in 6-well plates, and counting the number of spheres that formed after 7-14 days culture in stem cell medium [29].
Invasion assay
Invasion assays were carried out using matrigel chambers (BD Biosciences) according to the manufacturer’s protocol. In brief, transfected PANC1, PaTu8988, BxPc3 and SW1990 cells were harvested, resuspended in serum-free medium and transferred to the hydrated matrigel chambers (~10000 cells per well). The chambers were then incubated for 24h in culture medium with 10% FBS in the bottom chambers before examination. The cells on the upper surface were scraped and washed away, whereas the invaded cells on the lower surface were fixed and stained with 0.05% crystal violet for 30min. Finally, invaded cells were counted and the relative number was calculated.
Data analysis
All the experiments were performed three times with triplicate samples. Comparisons between groups were analyzed using the Student’s t test (two groups) or a one-way ANOVA (multiple groups). Differences with P values less than 0.05 were considered significant. DNA methylation data from bisulfite sequencing were analyzed and visualized using BiQ Analyzer.
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (81472333, 81372718) and the Natural Science Foundation of Jiangsu Province (BK2013247).
Conflicts of Interest
The authors declare no conflict of interest.
References
1. Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, Nie J, Jonsdottir GA, Ruotti V, Stewart R, Slukvin, II and Thomson JA. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007; 318:1917-1920.
2. Xu B, Zhang K and Huang Y. Lin28 modulates cell growth and associates with a subset of cell cycle regulator mRNAs in mouse embryonic stem cells. RNA. 2009; 15:357-361.
3. Shyh-Chang N and Daley GQ. Lin28: primal regulator of growth and metabolism in stem cells. Cell stem cell. 2013; 12:395-406.
4. Viswanathan SR and Daley GQ. Lin28: A microRNA regulator with a macro role. Cell. 2010; 140:445-449.
5. Hamano R, Miyata H, Yamasaki M, Sugimura K, Tanaka K, Kurokawa Y, Nakajima K, Takiguchi S, Fujiwara Y, Mori M and Doki Y. High expression of Lin28 is associated with tumour aggressiveness and poor prognosis of patients in oesophagus cancer. British journal of cancer. 2012; 106:1415-1423.
6. Wu T, Jia J, Xiong X, He H, Bu L, Zhao Z, Huang C and Zhang W. Increased Expression of Lin28B Associated with Poor Prognosis in patients with Oral Squamous Cell Carcinoma. PloS one. 2013; 8:e83869.
7. King CE, Wang L, Winograd R, Madison BB, Mongroo PS, Johnstone CN and Rustgi AK. LIN28B fosters colon cancer migration, invasion and transformation through let-7-dependent and -independent mechanisms. Oncogene. 2011; 30:4185-4193.
8. Lu L, Katsaros D, Shaverdashvili K, Qian B, Wu Y, de la Longrais IA, Preti M, Menato G and Yu H. Pluripotent factor lin-28 and its homologue lin-28b in epithelial ovarian cancer and their associations with disease outcomes and expression of let-7a and IGF-II. European journal of cancer. 2009; 45:2212-2218.
9. Daley SRVGQ. Lin28 A MicroRNA Regulator. Cell. 2010; 140.
10. Xu C, Shen J, Xie S, Jiang Z, Huang L and Wang L. Positive expression of Lin28 is correlated with poor survival in gastric carcinoma. Medical oncology. 2013; 30:382.
11. Lisa D Moore1 TLaGF, 1. DNA Methylation and Its Basic Function. Neuropsychopharmacology. 2013; 38:23-38.
12. Baubec T, Ivanek R, Lienert F and Schubeler D. Methylation-dependent and -independent genomic targeting principles of the MBD protein family. Cell. 2013; 153:480-492.
13. Chu Y, Wang Y, Zhang G, Chen H, Dowdy SC, Xiong Y, Liu F, Zhang R, Li J and Jiang SW. Chromatin composition alterations and the critical role of MeCP2 for epigenetic silencing of progesterone receptor-B gene in endometrial cancers. Cellular and molecular life sciences. 2014.
14. Xinsheng Nan FJC, Adrian Bird. MeCP2 Is a Transcriptional Repressor with Abundant Binding Sites in Genomic Chromatin. Cell. 1997; 88:471-481.
15. Gunther K, Rust M, Leers J, Boettger T, Scharfe M, Jarek M, Bartkuhn M and Renkawitz R. Differential roles for MBD2 and MBD3 at methylated CpG islands, active promoters and binding to exon sequences. Nucleic Acids Res. 2013; 41:3010-3021.
16. Sai Murali Krishna Pulukuri JSR. CpG Island Promoter Methylation and Silencing of 14-3-3σ Gene Expression in LNCaP and Tramp-C1 Prostate Cancer Cell Lines is Associated with Methyl-CpG-Binding Protein MBD2. Oncogene. 2006; 25.
17. Wade PA. Methyl CpG-binding proteins and transcriptional repression. BioEssays. 2001; 12.
18. Feng C, Neumeister V, Ma W, Xu J, Lu L, Bordeaux J, Maihle NJ, Rimm DL and Huang Y. Lin28 regulates HER2 and promotes malignancy through multiple mechanisms. Cell Cycle. 2012; 11:2486-2494.
19. Murray MJ, Saini HK, Siegler CA, Hanning JE, Barker EM, van Dongen S, Ward DM, Raby KL, Groves IJ, Scarpini CG, Pett MR, Thornton CM, Enright AJ, Nicholson JC, Coleman N and Cclg. LIN28 Expression in malignant germ cell tumors downregulates let-7 and increases oncogene levels. Cancer Res. 2013; 73:4872-4884.
20. Zhao Z, Liu H, Wang X, Wang X and Li Z. Separation and identification of HSP-associated protein complexes from pancreatic cancer cell lines using 2D CN/SDS-PAGE coupled with mass spectrometry. Journal of biomedicine & biotechnology. 2011; 2011:193052.
21. Joe D.Lewis RRM, Willian J.Henzel, Ingrid Maurer-Fogy, Peter Jeppesen, Franz Klein, Adrian Bird. Purification,Sequence,and Cellular Localization of a Novel Chromosomal Protein that Binds to Methylated DNA. Cell. 1992; 69:905-914.
22. Gunther K, Rust M, Leers J, Boettger T, Scharfe M, Jarek M, Bartkuhn M and Renkawitz R. Differential roles for MBD2 and MBD3 at methylated CpG islands, active promoters and binding to exon sequences. Nucleic Acids Research. 2013; 41:3010-3021.
23. Viswanathan SR, Powers JT, Einhorn W, Hoshida Y, Ng TL, Toffanin S, O’Sullivan M, Lu J, Phillips LA, Lockhart VL, Shah SP, Tanwar PS, Mermel CH, Beroukhim R, Azam M, Teixeira J, et al. Lin28 promotes transformation and is associated with advanced human malignancies. Nature genetics. 2009; 41:843-848.
24. Heo I, Joo C, Cho J, Ha M, Han J and Kim VN. Lin28 mediates the terminal uridylation of let-7 precursor MicroRNA. Mol Cell. 2008; 32:276-284.
25. Dangi-Garimella S, Yun J, Eves EM, Newman M, Erkeland SJ, Hammond SM, Minn AJ and Rosner MR. Raf kinase inhibitory protein suppresses a metastasis signalling cascade involving LIN28 and let-7. The EMBO Journal. 2009; 28:347-368.
26. Vincent A, Omura N, Hong SM, Jaffe A, Eshleman J and Goggins M. Genome-wide analysis of promoter methylation associated with gene expression profile in pancreatic adenocarcinoma. Clinical cancer research. 2011; 17:4341-4354.
27. Sharma D, Blum J, Yang X, Beaulieu N, Macleod AR and Davidson NE. Release of methyl CpG binding proteins and histone deacetylase 1 from the Estrogen receptor alpha (ER) promoter upon reactivation in ER-negative human breast cancer cells. Molecular endocrinology. 2005; 19:1740-1751.
28. Konduri SD, Srivenugopal KS, Yanamandra N, Dinh DH, Olivero WC, Gujrati M, Foster DC, Kisiel W, Ali-Osman F, Kondraganti S, Lakka SS and Rao JS. Promoter methylation and silencing of the tissue factor pathway inhibitor-2 (TFPI-2), a gene encoding an inhibitor of matrix metalloproteinases in human glioma cells. Oncogene. 2003; 22:4509-4516.
29. Nu Zhang, Ping Wei, Aihua Gong, Wen-Tai Chiu, Hsueh-Te Lee, Howard Colman, He Huang, Jianfei Xue, Mingguang Liu, Yong Wang, Raymond Sawaya, Keping Xie, W.K. Alfred Yung, René H. Medema, Xi He, and Suyun Huang. FoxM1 Promotes β-Catenin Nuclear Localization and Controls Wnt Target-Gene Expression and Glioma Tumorigenesis. Cancer Cell. 2011; 20: 427-442.

