Introduction
The blood brain barrier (BBB) contributes to the immune privilege of the central nervous system (CNS) by excluding potentially harmful cells and macromolecules from the CNS parenchyma. Immunosurveillance of CNS is operated by effector T cells, which migrate across the BBB and are retained in cerebrospinal fluid (CSF)- drained perivascular and submeningeal spaces. Therein effector T cells remain separated from the CNS by glia limitans, a functional barrier composed of parenchymal basement membrane and astrocyte foot processes, located at interfaces between non-neural tissue and CNS parenchyma [1, 2]. Encountering of antigen presenting cells and effective in situ antigen recognition by the T cell, as observed in experimental autoimmune encephalomyelitis (EAE), result in upregulation of proinflammatory cytokines, proteases, chemokines, chemokine receptors as well as activation markers, breaching of glia limitans and neuroinflammation. In contrast, antigen ignorant T cells do not upregulate activation markers or pro-inflammatory cytokines and do not infiltrate the brain parenchyma [3].
The plasma membrane ecto-5’-nucleotidase CD73, an enzyme of the purine catabolic pathway that catalyzes the breakdown of AMP to adenosine, is induced in activated CD4 cells by TGF-β [4]. Extracellular adenosine generated by CD73 enzymatic activity contributes to immunosuppression by T regulatory (Treg) cells and might play a pivotal role in preventing autoimmune diseases [5, 6]. The rate-limiting step of the ectonucleotidase cascade for adenosine generation is represented by ectonucleoside triphosphate diphosphohydrolase 1 (E-NTPDase 1) CD39 that hydrolyzes ATP/UTP and ADP/UDP to the respective nucleoside (e.g., AMP). T cell receptor (TCR) stimulation induces CD39 enzymatic activity in the plasma membrane of mouse Treg cells [7], suggesting generation of adenosine through CD39 and CD73 is important for immunosuppression. On the other hand, adenosine generation by CD73 in the CNS is required for efficient entry of encephalitogenic lymphocytes into the brain and spinal cord during EAE [8].
Although serving as a barrier, which restricts the entry of inflammatory cells into CNS parenchyma [9-11], astrocytes have powerful pro-inflammatory potential. Also, dysfunction of astrocytes at the border of inflamed tissue leads to spread of neurotoxic inflammation into adjacent neural parenchyma. Thus, astrocytes are emerging as important regulators of neuroinflammatory events [2]. In this paper we show that antigen-independent adhesion of recently activated CD4 cells to astrocytes results in robust upregulation of plasma membrane CD39 and CD73 ectonucleotidases as well as T cell polarization to a Th17-like immunosuppressive phenotype. On the other hand, hydrolysis of extracellular ATP by CD39 expressed in T cells results in inhibition of ATP-dependent spontaneous calcium signaling and transcriptional regulation in astrocytes. We propose that this signaling pathway might constitute a regulatory mechanism for pro-inflammatory activation of antigen-specific T cells in the brain.
Results
Expression of ectonucleotidases CD39 and CD73 in brain infiltrating CD4 cells in contact with astrocytes
Autoantigen specific activation of CD4 cells is used to induce EAE in mice. This experimental model fairly reproduces neuroinflammation determined by pathogenic T cell activation in multiple sclerosis (MS). Confocal analysis of brain and spinal cord from EAE mice revealed infiltrating CD3+ T cells around blood vessels (Figure 1A) and in the spinal cord (Figure 1B) that were positive for both CD39 and CD73 ectoenzymes in the plasma membrane. Interestingly, CD39 or CD73 positive T cells were in direct contact with astrocytes as indicated by triple labeling for the astrocyte marker glial fibrillary acidic protein (GFAP) (arrows in Figure 1A, 1B). Ex vivo analysis of brain from mice with EAE in flow cytometry confirmed the presence of CD39+CD73+ double positive cells within the CD4+ subset and a population of CD39+ cells within the CD3+CD4- compartment infiltrating the brain that were absent in healthy animals (Figure 1C).
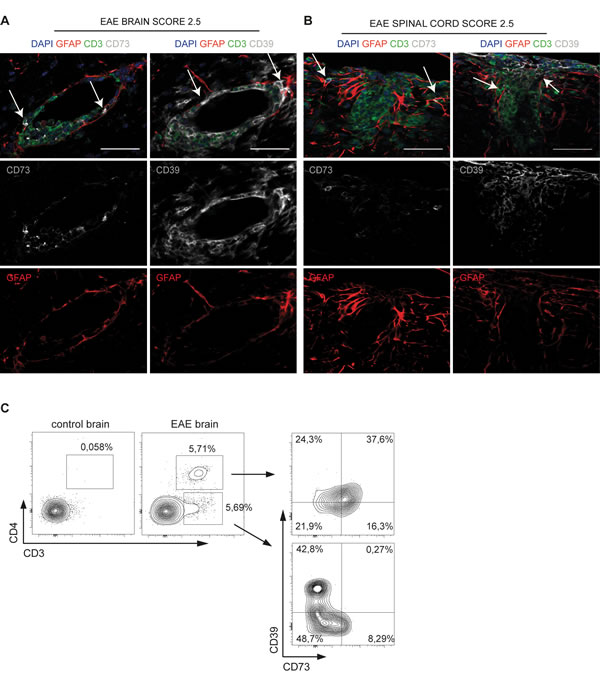
Figure 1: In vivo adhesion of ectonucleotidase positive T cells to astrocytes. A., B. Representative coronal sections of the brain (A) and spinal cord (B) of non-relapsing EAE at 20 days post-immunization. Immunofluorescence staining shows CD3+ (green), CD73+ or CD39+ (grey) cells and GFAP+ astrocytes (red). Arrows in A indicate CD73+ and CD39+ CD3+ cells contacting astrocytes at the outer perivascular area of blood vessels in stratum subependymal of lateral ventricles. Arrows in B show the same interactions in the spinal cord (caudal sections). These results are representative of 3 mice per organ. Scale bar, 50 µm. C. Analysis at FACS of CD39 and CD73 expression in T cells isolated ex vivo from control and EAE brains.
Upregulation of CD39 and CD73 in activated CD4 cells upon interaction with astrocytes
Since astrocytes are the first neural cells encountered by lymphocytes entering the brain, the above observation prompted us to investigate whether astrocytes may regulate surface expression of CD39 and CD73 in CD4 cells. Co-cultures of purified murine astrocytes with either resting or pre-activated CD4 T cells showed that a substantial fraction of activated, but not resting T cells adhered to astrocytes after 48 h of culture (Figure 2A). FACS analysis after accutase detachment and dissociation of adherent cells showed a significantly increased T cell-to-astrocyte ratio when activated T cells were present in the co-culture (Figure 2B). As previously shown [4], about 40% of ex vivo isolated naïve CD4 cells were CD73+ and did not express CD39 (Supplementary Figure S1A). However, CD39 was slightly upregulated upon in vitro activation with CD3 and CD28 antibodies (Figure 2C, left). Notably, the percentage of positive cells as well as CD39 and CD73 mean fluorescent intensities (MFIs) were significantly and progressively increased in T cells that were shifted to co-culture with astrocytes (Figure 2C, 2D). These results suggest that astrocytes promote ectonucleotidase expression in recently activated CD4 cells.
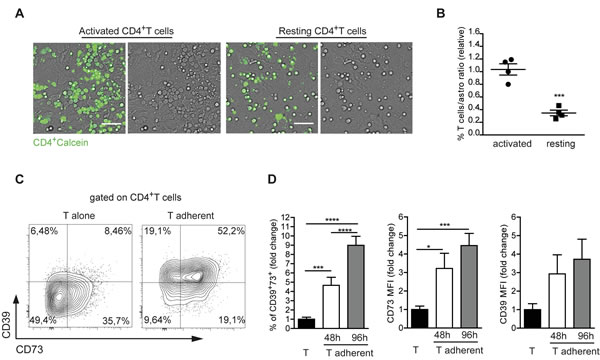
Figure 2: Astrocytes promote CD39 and CD73 expression in recently activated CD4 cells. A. Co-cultures of CD4+ T cells loaded with calcein-AM (green) with purified astrocytes. Activated T cells massively adhered to astrocytes 48 h after plating, acquiring a flat shape (left), while resting (unstimulated) T cells remained suspended over the astrocyte monolayer (right). Scale bar 50 µm. B. Statistical analysis of resting vs activated T cells to astrocytes ratio after accutase treatment at 48h of co-culture. Student T test, ***p < 0.001. C. Representative dot plot of CD39 and CD73 expression in activated CD4+ T cells after 48 h of culture in isolation or adherent to astrocytes. D. Fold change in percentage of CD39+73+ T cells adherent to astrocytes, CD73 and CD39 MFIs after 48h (white) and 96h (grey) co-culture. Values were normalized to T cells alone (black). One-way ANOVA; Bonferroni’s test for comparison among groups, *p < 0.05 ***p < 0.001, ****p < 0.0001; N > 6.
Upregulation of CD39 and CD73 in activated CD4 cells adhering to astrocyte is independent of TCR interaction with MHC class II
In order to investigate the mechanism through which the contact with astrocytes mediated CD39 and CD73 upregulation in T cells, we initially focused on MHC class II (MHCII) molecules, which are efficiently induced in astrocytes by exposure to IFN-γ [12, 13]. Indeed, we detected MHCII expression in astrocytes co-cultured with activated T cells (Figure 3A), in line with the observation that a substantial fraction of CD4 cells adhering to astrocytes secreted IFN-γ (Supplementary Figure S1B). We addressed whether MHCII in astrocytes was responsible for CD39 and CD73 upregulation in T cells by co-culturing activated CD4 cells with MHCII deficient astrocytes. These cells were equally competent to MHCII sufficient cells in inducing CD39 and CD73 in T cells, thus indicating that the observed phenotype was independent of MHCII mediated interaction (Figure 3B). Analysis of resting CD4 T cells co-cultured with astrocytes revealed lack of difference in the expression of CD39 and CD73 compared to T cells cultured alone (Figure 3C). This observation suggests that only activated T cells efficiently upregulate CD39 and CD73 upon interaction with astrocytes. Only a slight increase in surface CD39 and CD73 was detected in astrocytes in response to activated CD4 cells (Supplementary Figure S1C).
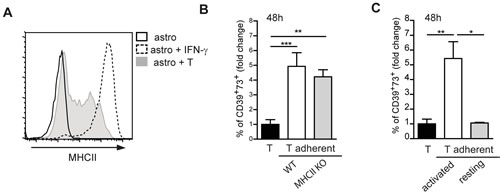
Figure 3: MHC class II independent astrocyte -T cell interaction. A. MHCII expression on astrocytes alone (black line), stimulated with IFN-γ (dotted line) or co-cultured with T cells (shaded) at 48h of co-culture. B. Fold change in percentage of CD39+73+ T cells adherent to either wild-type (WT) or MHCII deficient astrocytes. Normalized to T cells alone (black bar). One-way ANOVA; Bonferroni’s test for comparison among groups; **p < 0.01, ***p < 0.001; N = 3. C. Fold change in percentage of CD39+73+ cells in activated or resting T cells adherent to astrocytes after 48h co-culture. Normalized to T cells alone (black bar). One-way ANOVA; Bonferroni’s test for comparison among groups; *p < 0.05, **p < 0.01; N = 3.
Adhesion to astrocytes is required for upregulation of CD39 and CD73 in T cells
To address whether cell-to-cell contact was required for CD39 and CD73 upregulation in T cells, we co-cultured astrocytes and T cells in a transwell system. Physical separation of the two cell types prevented CD39 and CD73 induction in T cells (Figure 4A). We therefore investigated the possible involvement of T cell/astrocyte adhesion molecules in regulating CD39 and CD73. Upon co-culture with T cells, astrocytes express ICAM-1 (CD54) and VCAM-1 (CD105), which are ligands for LFA-1 (αLβ2) and VLA-4 (α4β1) integrins, respectively [14, 15] (Figure 4B). Pretreatment of T cells with α4 blocking antibodies significantly reduced T cells adhesion to astrocytes, while blockade of β1 integrin was ineffective. On the other hand, anti-VCAM-1 (CD105) antibodies efficiently inhibited T cell adhesion to astrocytes and inhibition was further enhanced by anti-ICAM-1 (CD54) antibodies (Figure 4C). Antibodies addition to cultures did not influence CD4 cell recoveries (data not shown). However, in T cells that still adhered to astrocytes in the presence of blocking antibodies, CD39 and CD73 were efficiently upregulated (Figure 4D). These data suggest that LFA-1 and VLA-4-mediated adhesion as well as downstream signaling were not absolutely required for plasma membrane ectonucleotidases upregulation in T cells, although we cannot exclude that it was impossible to achieve complete inhibition of integrin-mediated adhesion with antibodies.
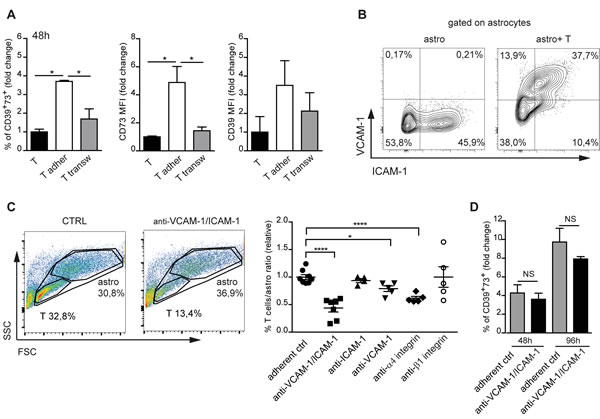
Figure 4: Integrin mediated astrocyte-T cell adhesion. A. Fold change in percentage of CD39+73+ T cells either adherent to astrocytes (white bars) or separated by transwell (transw) (gray bars), CD73 and CD39 MFIs after 48h co-culture. Normalized to T cells alone (black bars). One-way ANOVA; Bonferroni’s test for comparison among groups; *p < 0.05; N = 3. B. FACS analysis of VCAM-1 and ICAM-1 expression on electronically gated CD11b-CD3- astrocytes either cultured in isolation or co-cultured with T cells for 48h. C. FACS analysis of forward scatter (FSC) vs side scatter (SSC) of untreated astrocyte-T cell co-culture or co-culture treated with anti-VCAM-1/ICAM-1 antibodies. The indicated regions correspond to T cells and astrocytes with percentages of recovered cells. The panel on the right shows statistics of T cells/astrocytes ratios in the indicated culture conditions. One-way ANOVA; Bonferroni’s test for comparison among groups; *p < 0.05, ****p < 0.0001. D. Fold change in percentage of CD39+73+ cells among CD4 cells adherent to astrocytes without (gray bars) or with anti-VICAM-1/ICAM-1 antibodies (black bars) after 48h and 96h culture as compared to T cells cultured in isolation (not shown). One-way ANOVA; Bonferroni’s test for comparison among groups; NS = not significant; N = 3.
Upregulation of CD39 and CD73 in CD4 cells correlates with acquisition of immunosuppressive potential
To functionally characterize CD39 and CD73 expressing T cells generated in co-culture with astrocytes we analyzed the transcription of genes coding for master regulators of CD4 T cell functional polarization. Real-time quantitative reverse transcription PCR (qRT-PCR) performed in the CD39+73+ subset sorted from cells adhering to astrocytes or on total astrocyte-adherent cells revealed a marked increase in transcription of Rorc (encoding RORγt, the master switch transcription factor that defines the Th17 subset) and, to a lesser extent, Foxp3 (that determines the Treg cells phenotype), whereas transcript levels of Tbx21 and Gata-3, which define Th1 and Th2 T cells respectively, were not significantly different from T cells cultured alone (Figure 5A). We did not detect variations in Tbx21, Rorc, Foxp3 or Gata3 transcripts in CD4 cells separated from astrocytes by transwell chamber with respect to T cells cultured in isolation (Figure 5A). These results indicate that the observed transcriptional regulation required T cell contact with astrocyte. No significant differences in CD4 cell proliferation were observed in the different culture conditions (Supplementary figure S2A, S2B). Flow cytometry confirmed that the majority of CD39+73+ T cells adhering to astrocytes upregulated RORγt protein after 96h of co-culturing (Figure 5B, upper panel). RORγt is the master transcriptional regulator of Th17 effector functions, however, we detected few cells expressing IL-17A, the cytokine characterizing this subtype (Figure 5B lower panel). The observed phenotype was reminiscent of a recently described immunosuppressive Th17 cell subset [16]. Moreover, qRT-PCR revealed upregulation of both IFNG and IL10 transcripts in CD39+73+ T cells adhering to astrocytes (Supplementary Figure S2C). We investigated whether CD39+73+ cells were able to suppress the proliferation of TCR stimulated T cells in vitro. We isolated astrocyte-adherent CD4 cells and further sorted CD39+73+ cells at FACS. Co-culture of these cells with sorted CD4+CD44-CD62L+CD25- naïve T cells stimulated with anti-CD3 antibodies in the presence of irradiated splenocytes from cd3e-/- mice, resulted in inhibition of T cell proliferation. The efficiency of this inhibition was superior to Foxp3 expressing Treg cells isolated from Foxp3EGFP transgenic mice (Figure 5C). Indeed, only around 50% of ex vivo sorted CD25highFoxp3+ Treg cells expressed CD39 and CD73 (Supplementary Figure S3A), suggesting that enhanced effect of astrocytes adherent cells could be due to selection of ectonuleotidases expressing cells. Collectively, these data indicate that recently activated CD4 T cells acquire a CD39+73+ Th17 immunosuppressive phenotype upon interaction with astrocytes.
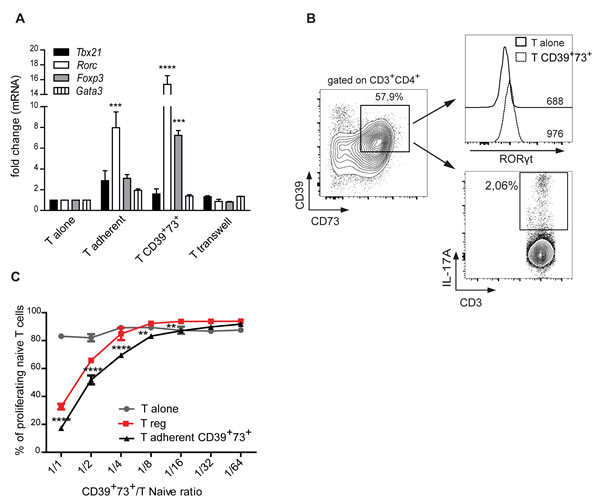
Figure 5: CD39 and CD73 upregulation correlates with immunosuppressive Th17 phenotype. A. Real-time qRT-PCR for Tbx21, Rorc, Foxp3 and Gata3 in CD4+ T cells cultured alone, total T cells adherent to astrocytes (T adher), sorted CD39+73+ T cells adherent to astrocytes or T cells separated by transwell after 48 h co-culture. Two-way ANOVA; Bonferroni’s test for comparison with T cells cultured alone, ***p < 0.001, ****p < 0.0001; N = 4. B. Expression of RORγt (upper histograms with respective MFI indicated) and IL-17A (lower plot) by FACS analysis on CD39+73+ cells sorted as indicated after 96 h co-culture. C. Percentage of UV-labeled proliferating cells in suppression assay with CD4+CD39+73+ T cells detached from astrocytes. Graph shows percentage suppression of proliferation by T cells cultured in isolation (T alone), CD39+73+ astrocyte-adherent T cells or sorted Treg cells in 72 h culture at different suppressor/responder cells ratio. One representative experiment with triplicates. Two-way ANOVA; Bonferroni’s test for comparison among groups, **p < 0.01,****p < 0.0001, N = 3.
Role of TGF-β in ectonucleotidase upregulation in T cells
As previously described [16], treatment with exogenous IL-6 and TGF-β induced prominent ectonucleotidase upregulation in CD4 cells, mimicking the induction triggered by astrocytes (Figure 6A). Analysis of IL-6 in the culture supernatant showed the progressive increase of the cytokine concentration in T cell/astrocyte co-cultures over time, whereas the cytokine was barely detectable in cultures of T cells and astrocytes grown in isolation (Figure 6B). Analysis of IL6 transcripts showed that mRNA levels were increased in both T cells and astrocytes in co-culture (Figure 6C). Since immunosuppressive Th17 cells are characterized by transcriptional upregulation of Stat3 and downregulation of the transcription factor Gfi1, we analyzed by real-time qRT-PCR Stat3 and Gfi1 genes. Figure 6D shows that, analogously to the cell subset described by Chalmin et al. [16], CD4 cells adhering to astrocytes significantly upregulated Stat3 and downregulated Gfi1, albeit non-significantly. To explore the possible involvement of TGF-β in T cell polarization evoked by interaction with astrocytes, we first analyzed TGFB transcript levels by real-time qRT-PCR in astrocytes and T cells either co-cultured or cultured in isolation. Cell co-culturing resulted in higher levels of TGFB1 and TGFB2 transcripts in astrocytes and T cells, respectively (Figure 6E). Interestingly, incubation of recently activated CD4 cells with TGF-β alone was sufficient to promote CD39 and CD73 upregulation (Figure 6F), whereas addition of IL-6 to cultures did not result in CD39 and CD73 variations (Figure 6G). Consistently, addition of TGF-β RI kinase inhibitor VI significantly inhibited both CD39 and CD73 expression in adherent CD4 T cells at 48 (left) and 96h (right) (Figure 6H). Altogether, these data point to TGF-β as the key secreted molecule responsible for astrocyte-dependent CD39 and CD73 upregulation in the T cell.
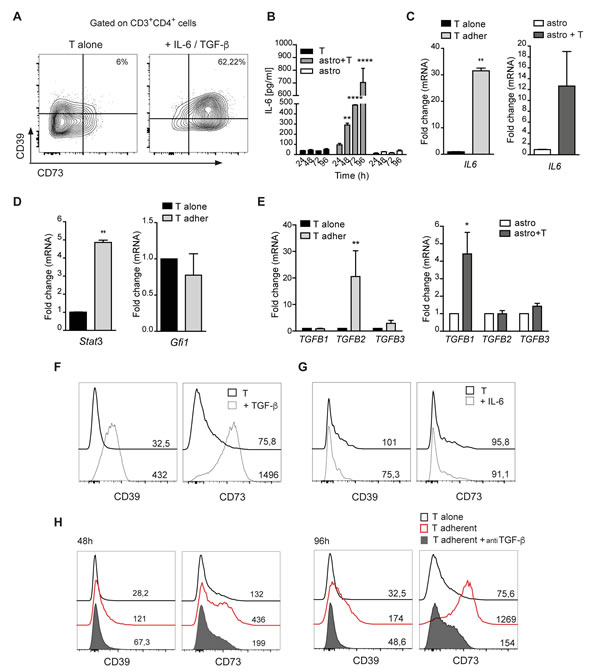
Figure 6: TGF-β promotes CD39 and CD73 upregulation in T cells. A. FACS analysis for plasma membrane CD39 and CD73 in naive CD4+ T cells stimulated for 96 h with anti-CD3/CD28 mabs in the presence of TGF-β and IL-6. B. Quantification of IL-6 by ELISA in the supernatant of T cells, astrocytes and astrocyte/T cell co-culture. Two-way ANOVA; Bonferroni’s test for comparison among groups, **p < 0.01, ****p < 0.0001; N = 3. C. IL6 mRNA levels in purified T cells (left) or astrocytes (right) after 48 h of co-culture. Mann-Whitney’s test , **p < 0.01; N = 3. D. Real-time qRT-PCR for Stat3 and Gfi1 in T cells alone and CD39+73+ T cells adherent to astrocytes. Mann-Whitney’s test; **p < 0.01; N = 3. E. Expression of TGFB1, TGFB2, TGFB3 assessed by qRT-PCR in T cells (left) (N = 4) or astrocytes (right) (N = 2) alone or co-cultured for 48 h. Mann-Whitney’s test; *p < 0.05; ***p < 0.01. F.,G. FACS analysis of CD39 and CD73 expression with MFIs indicated, in CD3/CD28 stimulated CD4 cells (black line), with addition of TGF-β (F) or IL-6 (G) (dashed line) for 96 h. H. CD39 and CD73 expression (MFIs) on CD4+ T cells cultured alone (black line) or pre-incubated (filled grey) or not (red) with TGF-β RI kinase inhibitor VI before co-culturing with astrocytes for 48 and 96 h.
Modulation of spontaneous calcium oscillations in astrocytes by CD39 expressed in T cells
It is known that astrocytes communicate among themselves and with other brain cells through regulated increases in intracellular calcium concentrations [17-20], which are sustained by extracellular ATP [reviewed in [21]]. Indeed, addition of the ATP hydrolyzing enzyme apyrase to astrocyte cultures resulted in significant reduction of the percentage of cells showing Ca2+ oscillations (Figure 7A, right). Hence, we reasoned that ATP hydrolysis by CD39+73+ T cells might negatively modulate Ca2+ signaling in astrocytes. Accordingly, significantly lower extracellular ATP levels were detected in astrocytes co-cultured with T cells compared to astrocytes cultured alone (Figure 7B). A two-fold decrease in the percentage of astrocytes showing spontaneous calcium oscillations was detected in astrocytes co-cultured with CD39+73+ T cells (Figure 7C and Supplementary Movies S1-2) but not resting CD4 cells (Figure 7D). Furthermore, co-culture of activated Entpd1-/- T cells with astrocytes resulted in unaltered percentage of astrocytes displaying Ca2+ oscillations, indicating that CD39 in T cells adhering to astrocytes inhibits purinergic Ca2+ signaling in astrocytes (Figure 7E). The lack of effect of Entpd1-/- T cells on astrocyte calcium dynamics was not due to impaired capacity of T cells to interact with astrocytes, as the number of Entpd1-/- T cells adhering to astrocytes was comparable to wild-type T cells (Supplementary Figure S3B). Finally, we observed upregulation of plasma membrane CD73 in Entpd1-/- CD4 cells adhering to astrocytes, indicating that CD73 is regulated independently of CD39 expression (Figure 7F). These results indicate that the reciprocal communication between recently activated CD4 cells and astrocytes results in a CD39-dependent reduction of ATP-mediated Ca2+ activity in astrocytes. Consistent with a functional consequence of this signaling circuit in astrocytes, we correlated the reduction of spontaneous Ca2+ oscillations in astrocytes with a decrease in sonic hedgehog (Shh) expression, a secreted molecule that promotes BBB integrity [22] and to enhanced transcription of the gene encoding the chemokine CCL2, which triggers immune cell infiltration [23, 24] (Figure 7G). Therefore, astrocyte interaction with activated CD4 cells would implement a transcriptional program promoting BBB permeability and leukocyte infiltration.
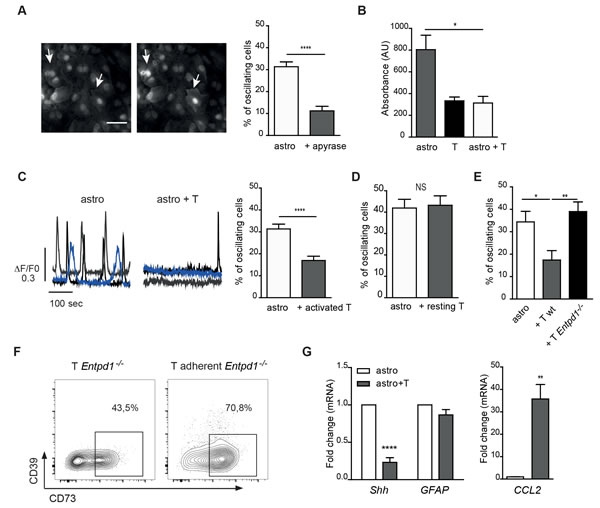
Figure 7: CD39 mediated inhibition of spontaneous cytosolic Ca2+ oscillations in astrocytes by recently activated CD4 cells. A. Representative fluorescence images of astrocytes loaded with Oregon green showing spontaneous calcium oscillations (left, arrows) and statistical analysis of astrocytes showing calcium oscillations in control culture (astro) or with addition of apyrase. Mann-Whitney’s test; ****p < 0.0001; N = 3. B. Luciferin/luciferase assay for extracellular ATP concentration in medium from astrocyte or T cell cultures, or astrocyte/T cell co-cultures (after 72 h). Means of triplicates ± SEM from a representative experiment are shown. One-way ANOVA; Bonferroni’s test for comparison among astrocytes cultured alone and astrocytes in co-culture with T cells; *p < 0.05. C. Temporal analysis of cytosolic Ca2+ concentration recorded from three representative astrocytes cultured alone (left) or co-cultured with activated T cells for 96 h (right) and statistical analysis of astrocytes showing spontaneous calcium oscillations. Mann-Whitney’s test; ****p < 0.0001, N = 6. D. Same analysis as in C but with resting T cells. Student’s t-test, NS = not significant; N = 2. E. Same analysis as in C with activated T cells from wild-type and Entpd1-/- mice. One-way ANOVA; Bonferroni’s test for comparison among groups; *p < 0.05; **p < 0.01; N = 3. F. FACS analysis of CD39 and CD73 expression in activated Entpd1-/- T cells either cultured alone or adhering to astrocytes after 48 h co-culture. G. Real-time qRT-PCR for CCL2, GFAP and Shh in astrocytes either cultured alone or co-cultured with activated T cells (at 48 h). Mann-Whitney’s test, **p < 0.01, ****p < 0.0001; N = 4.
Discussion
The CNS has been considered for long time as an immune privileged site. However, the detection of activated T cells in the brain parenchyma in healthy conditions indicated that these cells could in fact cross the BBB. Within this context, astrocytes have been generally considered to play a beneficial role as immunosuppressive and neuroprotective elements. Nevertheless, astrocytes were shown to be capable of producing a range of proinflammatory cytokines and acting as antigen presenting cells (reviewed in ref [25]). A number of efforts have been devoted at defining functional polarization of CD4 cells upon interaction with astrocytes with conflicting results [26-30]. Treg cells were shown to develop upon contact with astrocytes and administration of these cells alleviated CNS inflammation and clinical symptoms in EAE [31]. Yet, the molecular features of the immunosuppressive arm eventually triggered by astrocytes are so far unknown. In this study we aimed at addressing the outcome of the interaction between CD4 cells and astrocytes in a possible physiological as well as pathophysiological context. To this end we analyzed in vitro molecular and functional clues resulting from the interaction of recently activated CD4 cells with astrocytes. This interaction resulted in upregulation of VCAM-1 and ICAM-1 in astrocytes, which promoted ectonucleotidase expression in T cells. The humanized anti-α4 integrin antibody natalizumab has shown therapeutic efficacy in relapsing-remitting MS by affecting T cells infiltration of the brain [32]. Interestingly, blockade of α4 integrin also inhibited T cells adhesion to astrocytes in our assay, suggesting that natalizumab might limit astrocyte/T cell interaction. We defined the Th17-like immunosuppressive phenotype resulting from this interaction that might constitute a feed-back mechanism for limiting tissue damage by brain infiltrating pro-inflammatory T cells. Notably, a population of IL-4 producing CD4 cells was recently shown to promote neuroprotection after CNS injury in an antigen independent fashion [33]. This study together with ours suggest that therapeutic strategies aimed at converting brain infiltrating T cells to a neuroprotective function can be envisaged.
We have shown that TGF-β is crucially involved in astrocyte-dependent CD39 and CD73 upregulation in the T cell. These ectonucleotidases contribute to CD4 cell immunosuppressive function [5-7]. Upregulation of TGF-β1 in astrocytes characterizes the early phases of MS and EAE, and promotes brain infiltration by myelin oligodendrocyte glycoprotein- (MOG-) specific pro-inflammatory CD4 cells [34]. Interestingly, administration of complete Freund adjuvant (CFA), a potent inducer of TGF-β1 in astrocytes [34] before induction of EAE protects from the disease [35]. These observations suggest that TGF-β1 produced by astrocytes might also exert a protective role in EAE. The functional conversion of activated T cells by TGF-β might represent an immunoregulatory mechanism to control tissue inflammation in the absence of acute antigen recognition.
Different signaling mechanisms can skew astrocyte function at the BBB resulting in opposite outcomes, such as promoting or limiting neuroinflammation [2]. Hydrolysis of extracellular ATP by T cell ectonucleotidases may have important implications for microvessel permeability, independently of adenosine generation, by affecting astrocyte Ca2+ activity. Indeed, Ca2+ signaling in astrocyte foot processes can influence blood flow through activation of phospholipase A2 (PLA2), arachidonic acid (AA) mobilization and production of vasoactive AA metabolites [36-40], with Ca2+ elevations being more common in vasoconstriction [41]. Interestingly, the reduction of spontaneous Ca2+ oscillations in astrocytes was associated to decrease in Shh expression, a secreted molecule that promotes BBB integrity and to enhanced transcription of CCL2, which triggers immune cell infiltration. These observations suggest that MHC-independent bidirectional signaling between astrocytes and T cells may favor T cell polarization toward an immunosuppressive phenotype as well as promote T cells infiltration into the brain tissue. This regulatory path might be important to resolve inflammation determined by infectious processes upon local clearance and/or limited presentation of pathogenic antigen/s.
In relapsing-remitting MS, episodes of acute inflammation, tissue damage and neurological dysfunction are followed by deactivation, repair and clinical remission [42]. In contrast, the progressive form of the disease is characterized by axonal degeneration and irreversible disability that are relatively independent from inflammation [43]. The oscillatory nature of relapsing-remitting MS is the result of the local homeostatic regulation of T cell activation. The signaling pathways leading to this remarkable capacity of suppressing inflammation and promoting repair mechanisms in the brain could be exploited therapeutically to limit the immunopathological damage that can ultimately lead to the progressive form of the disease [44]. We hypothesize astrocytes-mediated upregulation of CD39 and CD73 in activated CD4 cells is part of this adaptive regulatory framework at the BBB. The Th17-like immunosuppressive activity induced by contact with astrocytes might contribute a beneficial immunomodulatory effect during neuroinflammation as well as oppose to resolution of chronic infections and anti-tumor T cell responses.
Materials and Methods
Animals and ethics statement
Mice were bred at Humanitas Research Institute in Milan, Italy and Institute for Research in Biomedicine in Bellinzona, Switzerland. C57BL/6J (H-2b) and congenic FoxP3EGFP (B6.Cg-Foxp3tm2Tch/J) and Cd3e-/- mice were from Jackson Lab, Entpd1-/- mice were kindly provided by Dr. Simon C. Robson (Beth Israel Deaconess Medical Center, Boston, MA). Animals were kept under EU directives (2010/63/EU) and the Italian Legislation (L.D. 26/2014) for animal research with protocols approved by Institute Ethical Committee and the Italian Ministry of Health (162/2011-B). In Switzerland, all animal experiments were performed in accordance with the Swiss Federal Veterinary Office guidelines and authorized by the Animal Studies Committee of Cantonal Veterinary.
Cells
CD4+ cells were isolated from the spleen and lymph nodes of 2-4 months old female mice using magnetic beads coupled to anti-CD4 antibodies (Miltenyi Biotec) and sorted by FACS (FACSAria, Beckton Dickinson) as CD4+25-62L+44- naive T cells. Alternatively, Foxp3+ Treg cells were isolated from Foxp3EGFP mice by sorting for CD25 and EGFP. Purified CD4+ cells were stimulated for 40 h with plate-bound anti-CD3 (10 µg/ml) and anti-CD28 (5 µg/ml) antibodies (Biolegend) prior to transfer onto purified primary astrocyte cell cultures. Astrocytes were obtained from neonatal pups (postnatal day 1-3) and pure cultures of astrocytes (> 99.5%) were obtained by shaking flasks for 24 h at 37 °C at days 2 and 6 after plating. Resting or polyclonally activated T cells were plated on astrocyte monolayer, at an astrocyte:T cell ratio of 1:0.5 in the presence of murine IL-2 (90 U/ml). Co-cultures were maintained in RPMI-1640 medium supplemented with L-glutamine, non-essential amino acids, sodium pyruvate and 5% FBS. In a set of experiments astrocytes were treated with IFN-γ (25 ng/ml, Peprotech) for 16 h. CD4 T cells were differentiated in vitro by IL-6 and TGF-β (R&D) as previously described (16). In distinct set of experiments, CD4+ cells were plated in transwell culture inserts (0.4 µm pore, Costar) or astrocytes were pre-treated with anti-ICAM-1 (10 µg/ml) and VCAM-1 (10 µg/ml) antibodies for 4 h (Biolegend) to prevent T cells adhesion. Alternatively, CD4 cells were incubated with α4 integrin blocking antibody (CD49d, R1-2) (50 µg/ml) or with β1 integrin blocking antibody (CD29, HMβ1-1) (25 µg/ml). To block TGF-β receptor, activated CD4 cells were pre-incubated for 3 h with 10 µM TGF-β RI kinase inhibitor VI (SB 431542 hydrate, Sigma Aldrich).
Antibodies, flow cytometry and ELISA
After 48 h or 96 h of incubation, non-adherent CD4 T cells were removed by washing the co-culture 3 times with PBS (2% FBS). The remaining adherent cells (T cells and astrocytes) were incubated with accutase (Millipore) at 37°C for 40 min, collected and analyzed by flow cytometry. CD4+ cells were stained with the following mouse specific monoclonal antibodies: CD3 (clone 17A2), CD4 (GK1.5), MHCII (M5/114.15.2), CD39 (24DMS1), Rorg (gamma, greek letter) t (lower case) (AFKJS-9), IFN-γ (XMG1.2), CD25 (PC61 5), FoxP3 (FJK-165), CD44 (IM7), CD62L (MEL-14) (eBioscience), CD73 (TY11.8) and IL-17A (TC11-18H10.1) (Biolegend). Astrocytes were stained for CD11b (M1/70) (Biolegend), VCAM-1 (429) (BD Bioscience), ICAM-1 (YN1/1.7.4) (eBioscience). All samples were acquired with a FACSCalibur or FACSCanto (Becton Dickinson) and analyzed with FlowJo software (FlowJo v10.0.7). IL-6 protein concentration was determined using the DuoSet ELISA Development System according to the manufacturer’s instructions (R&D Systems).
Immunosuppression assay
Immunosuppression assay was performed as previously described [45]. Briefly 50,000 naïve CD4 T cells were labeled with Ultraviolet (UV) (Molecular Probes) and stimulated with CD3 monoclonal antibody (0.5 µg/ml) in the presence of 125,000 irradiated splenocytes from cd3e-/- mice. Serial dilutions of Treg cells or purified CD39+73+ cells adhering to astrocytes were added to cultures. UV dilution in naïve T cells was analyzed at 72 h. Acquisitions at FACS were standardized by fixed numbers of calibration beads (BD Pharmingen).
Real-time PCR
Quantitative real-time PCR (qRT-PCR) was performed as previously described [46]. RNA was isolated from cell cultures using the PureZOL RNA Isolation Reagent (BioRad), reverse transcribed to cDNA and qRT-PCR was performed using the Applied Biosystems ViiA™ 7 RT-PCR System. Experimental cDNA was amplified by qRT-PCR where a target cDNA (e.g., Rorc, FoxP3, TGFB1) and a reference cDNA (18S, GAPDH and HPRT) were amplified simultaneously using an oligonucleotide probe with a 5’ fluorescent reporter dye (6-FAM). Data were analyzed using the comparative threshold cycle (Ct) method and results are expressed as fold difference.
Calcium imaging
Calcium imaging was performed as previously described on cells loaded with Oregon Green (Molecular Probes) [47]. Briefly, astrocytes were loaded with the calcium sensitive dye Oregon Green for 1 h at 37 °C in medium and then imaged for calcium activity. The recording was performed in KRH (Krebs’-Ringer’s-HEPES) containing (in µM): 125 NaCl; 5 KCl; 1.2 MgSO4; 1.2 KH2PO4; 25 HEPES; 6 Glucose; 2 CaCl2; pH 7,4. Recording chambers were placed on the stage of an IX-71 inverted microscope (Olympus) equipped with an EMCCD (electron-multiplying CCD) camera (Quantem 512x512, Photometrics). Illumination was obtained using a light-emitting diode single LED (Cairn Research Optoled; 470nm) and a related GFP filter, 16 bit images were captured using a 40X objective [numerical aperture (NA): 1.3]. Regions of interest (ROIs) of about 15-pixel diameter (corresponding to about 12 µm) were drawn on the cell cytoplasm of virtually all the cells in the recorded field. Time-lapse recording of calcium dynamics was obtained with an acquisition rate of 2 Hz for 500 seconds, and on-line acquisition and off-line analysis were performed with MetaFluor software (Molecular Devices). Oscillating astrocytes displayed peak ∆F larger than 10 standard deviations of mean baseline noise. The average of F0 was comparable among astrocytes under different conditions. Roughly 20-25 astrocytes were analyzed in each recording field.
ATP measurment
Cell culture supernatants were measured using a luciferin/luciferase assay (Molecular Probes, Leiden, NL) according to the manufacturer’s instruction [48]. To degrade extracellular ATP in astrocytes cultures, cells were incubated for 1 h with the ATP hydrolyzing enzyme Apyrase (30U/ml, Sigma Aldrich).
Non-relapsing rodent EAE
Non-relapsing EAE (nr-EAE) was induced in female C57Bl/6 mice by immunization with 200 mg/mouse of MOG 35-55 (Espikem, Florence, Italy) and 2 injections of pertussis toxin (500 ng/mouse) the day of immunization and 48 h later. Clinical score was assigned according to a standard 0 to 5 scale, as described [49]. For analysis of brain infiltrating T cells at FACS, mice were perfused with HBSS, brains were chopped and digested in Collagenase D/DNase I (Roche) for one hour at 37°C with shaking. Tissues were resuspended in 35% Percoll and then centrifuged 40 min at 400 g to obtain the sediment fraction.
Immunostaining and confocal microscopy
Fixed 10 µm-thick frozen brain and spinal cord section were immunostained and acquired with an Olympus Fluoview FV1000 laser scanning confocal microscope in a channel mode with 405, 488, 568, and 647 nm excitations. Sections were imaged with oil immersion lens 40× (NA 1.3) in xy optical sections (1024×1024 pixels) of 0.3 μm. The z-stacks were reconstructed and analyzed by ImageJ Software (1.48v, Java).
Statistical analysis
Values, expressed as mean ± SEM, were obtained from at least 3 independent experiments. Differences between multiple groups were analyzed by one-way or two-way ANOVA with a Bonferroni’s post hoc test. Comparisons between two groups following a normal distribution were analyzed using an unpaired t-test (two-tail distribution) or a Mann-Whitney test as indicated in each figure. Statistical analysis was performed using GraphPad Prism (Graph-Pad Software).
Acknowledgments
We thank A. Anselmo, P. Somma (Humanitas Research Institute) and D. Jarrossay (Institute for Research in Biomedicine) for cell sorting and assistance with flow cytometry. D. Lecca for providing EAE brains and spinal cord. Tanja Rezzonico-Jost for the help at IRB animal house. M. Tamborini and E. Menna for support in confocal microscopy.
conflicts of interest
The authors declare that they have no conflicts of interest.
Grant support
F.F. was supported by Fondazione Cariplo (grant 2012-0519 to F.G.) and Fondazione Italiana Sclerosi Multipla (FISM) fellowship (2013/B/6). This work was supported by FISM (grant 2012/R/17) to C.V., Swiss National Science Foundation (grant 310030_140963) to F.G., Progetto di Interesse CNR Invecchiamento-LoCoDiCo and Ministry of Health (GR-2011-02347377) to M.M.
References
1. Engelhardt B and Ransohoff RM. Capture, crawl, cross: the T cell code to breach the blood-brain barriers. Trends in immunology. 2012; 33:579-589.
2. Sofroniew MV. Astrocyte barriers to neurotoxic inflammation. Nature reviews Neuroscience. 2015; 16:249-263.
3. Bartholomaus I, Kawakami N, Odoardi F, Schlager C, Miljkovic D, Ellwart JW, Klinkert WE, Flugel-Koch C, Issekutz TB, Wekerle H and Flugel A. Effector T cell interactions with meningeal vascular structures in nascent autoimmune CNS lesions. Nature. 2009; 462:94-98.
4. Regateiro FS, Howie D, Nolan KF, Agorogiannis EI, Greaves DR, Cobbold SP and Waldmann H. Generation of anti-inflammatory adenosine by leukocytes is regulated by TGF-beta. European journal of immunology. 2011; 41:2955-2965.
5. Deaglio S, Dwyer KM, Gao W, Friedman D, Usheva A, Erat A, Chen JF, Enjyoji K, Linden J, Oukka M, Kuchroo VK, Strom TB and Robson SC. Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. The Journal of experimental medicine. 2007; 204:1257-1265.
6. Flogel U, Burghoff S, van Lent PL, Temme S, Galbarz L, Ding Z, El-Tayeb A, Huels S, Bonner F, Borg N, Jacoby C, Muller CE, van den Berg WB and Schrader J. Selective activation of adenosine A2A receptors on immune cells by a CD73-dependent prodrug suppresses joint inflammation in experimental rheumatoid arthritis. Science translational medicine. 2012; 4:146ra108.
7. Borsellino G, Kleinewietfeld M, Di Mitri D, Sternjak A, Diamantini A, Giometto R, Hopner S, Centonze D, Bernardi G, Dell’Acqua ML, Rossini PM, Battistini L, Rotzschke O and Falk K. Expression of ectonucleotidase CD39 by Foxp3+ Treg cells: hydrolysis of extracellular ATP and immune suppression. Blood. 2007; 110:1225-1232.
8. Mills JH, Thompson LF, Mueller C, Waickman AT, Jalkanen S, Niemela J, Airas L and Bynoe MS. CD73 is required for efficient entry of lymphocytes into the central nervous system during experimental autoimmune encephalomyelitis. Proceedings of the National Academy of Sciences of the United States of America. 2008; 105:9325-9330.
9. Bush TG, Puvanachandra N, Horner CH, Polito A, Ostenfeld T, Svendsen CN, Mucke L, Johnson MH and Sofroniew MV. Leukocyte infiltration, neuronal degeneration, and neurite outgrowth after ablation of scar-forming, reactive astrocytes in adult transgenic mice. Neuron. 1999; 23:297-308.
10. Liedtke W, Edelmann W, Chiu FC, Kucherlapati R and Raine CS. Experimental autoimmune encephalomyelitis in mice lacking glial fibrillary acidic protein is characterized by a more severe clinical course and an infiltrative central nervous system lesion. The American journal of pathology. 1998; 152:251-259.
11. Voskuhl RR, Peterson RS, Song B, Ao Y, Morales LB, Tiwari-Woodruff S and Sofroniew MV. Reactive astrocytes form scar-like perivascular barriers to leukocytes during adaptive immune inflammation of the CNS. The Journal of neuroscience . 2009; 29:11511-11522.
12. Hirsch MR, Wietzerbin J, Pierres M and Goridis C. Expression of Ia antigens by cultured astrocytes treated with gamma-interferon. Neuroscience letters. 1983; 41:199-204.
13. Shrikant P and Benveniste EN. The central nervous system as an immunocompetent organ: role of glial cells in antigen presentation. Journal of immunology. 1996; 157:1819-1822.
14. Chan JR, Hyduk SJ and Cybulsky MI. Alpha 4 beta 1 integrin/VCAM-1 interaction activates alpha L beta 2 integrin-mediated adhesion to ICAM-1 in human T cells. Journal of immunology. 2000; 164:746-753.
15. Yusuf-Makagiansar H, Anderson ME, Yakovleva TV, Murray JS and Siahaan TJ. Inhibition of LFA-1/ICAM-1 and VLA-4/VCAM-1 as a therapeutic approach to inflammation and autoimmune diseases. Medicinal research reviews. 2002; 22:146-167.
16. Chalmin F, Mignot G, Bruchard M, Chevriaux A, Vegran F, Hichami A, Ladoire S, Derangere V, Vincent J, Masson D, Robson SC, Eberl G, Pallandre JR, Borg C, Ryffel B, Apetoh L, et al. Stat3 and Gfi-1 transcription factors control Th17 cell immunosuppressive activity via the regulation of ectonucleotidase expression. Immunity. 2012; 36:362-373.
17. Carmignoto G and Haydon PG. Astrocyte calcium signaling and epilepsy. Glia. 2012; 60(8):1227-1233.
18. Charles AC, Merrill JE, Dirksen ER and Sanderson MJ. Intercellular signaling in glial cells: calcium waves and oscillations in response to mechanical stimulation and glutamate. Neuron. 1991; 6:983-992.
19. Cornell-Bell AH, Finkbeiner SM, Cooper MS and Smith SJ. Glutamate induces calcium waves in cultured astrocytes: long-range glial signaling. Science. 1990; 247:470-473.
20. Seifert G, Schilling K and Steinhauser C. Astrocyte dysfunction in neurological disorders: a molecular perspective. Nature reviews Neuroscience. 2006; 7:194-206.
21. Koizumi S. Synchronization of Ca2+ oscillations: involvement of ATP release in astrocytes. The FEBS journal. 2010; 277:286-292.
22. Alvarez JI, Dodelet-Devillers A, Kebir H, Ifergan I, Fabre PJ, Terouz S, Sabbagh M, Wosik K, Bourbonniere L, Bernard M, van Horssen J, de Vries HE, Charron F and Prat A. The Hedgehog pathway promotes blood-brain barrier integrity and CNS immune quiescence. Science. 2011; 334:1727-1731.
23. Carrillo-de Sauvage MA, Gomez A, Ros CM, Ros-Bernal F, Martin ED, Perez-Valles A, Gallego-Sanchez JM, Fernandez-Villalba E, Barcia C, Sr., Barcia C, Jr. and Herrero MT. CCL2-expressing astrocytes mediate the extravasation of T lymphocytes in the brain. Evidence from patients with glioma and experimental models in vivo. PloS one. 2012; 7:e30762.
24. Kim RY, Hoffman AS, Itoh N, Ao Y, Spence R, Sofroniew MV and Voskuhl RR. Astrocyte CCL2 sustains immune cell infiltration in chronic experimental autoimmune encephalomyelitis. Journal of neuroimmunology. 2014; 274:53-61.
25. Gimsa U, Mitchison NA and Brunner-Weinzierl MC. Immune privilege as an intrinsic CNS property: astrocytes protect the CNS against T-cell-mediated neuroinflammation. Mediators of inflammation. 2013; 2013:320519.
26. Aloisi F. The role of microglia and astrocytes in CNS immune surveillance and immunopathology. Advances in experimental medicine and biology. 1999; 468:123-133.
27. Aloisi F, Ria F, Penna G and Adorini L. Microglia are more efficient than astrocytes in antigen processing and in Th1 but not Th2 cell activation. Journal of immunology. 1998; 160:4671-4680.
28. Beurel E, Harrington LE, Buchser W, Lemmon V and Jope RS. Astrocytes modulate the polarization of CD4+ T cells to Th1 cells. PloS one. 2014; 9:e86257.
29. Haroon F, Drogemuller K, Handel U, Brunn A, Reinhold D, Nishanth G, Mueller W, Trautwein C, Ernst M, Deckert M and Schluter D. Gp130-dependent astrocytic survival is critical for the control of autoimmune central nervous system inflammation. Journal of immunology. 2011; 186:6521-6531.
30. Nikcevich KM, Gordon KB, Tan L, Hurst SD, Kroepfl JF, Gardinier M, Barrett TA and Miller SD. IFN-gamma-activated primary murine astrocytes express B7 costimulatory molecules and prime naive antigen-specific T cells. Journal of immunology. 1997; 158:614-621.
31. Trajkovic V, Vuckovic O, Stosic-Grujicic S, Miljkovic D, Popadic D, Markovic M, Bumbasirevic V, Backovic A, Cvetkovic I, Harhaji L, Ramic Z and Mostarica Stojkovic M. Astrocyte-induced regulatory T cells mitigate CNS autoimmunity. Glia. 2004; 47:168-179.
32. Ransohoff RM. Natalizumab for multiple sclerosis. The New England journal of medicine. 2007; 356:2622-2629.
33. Walsh JT, Hendrix S, Boato F, Smirnov I, Zheng J, Lukens JR, Gadani S, Hechler D, Golz G, Rosenberger K, Kammertons T, Vogt J, Vogelaar C, Siffrin V, Radjavi A, Fernandez-Castaneda A, et al. MHCII-independent CD4+ T cells protect injured CNS neurons via IL-4. The Journal of clinical investigation. 2015; 125:699-714.
34. Luo J, Ho PP, Buckwalter MS, Hsu T, Lee LY, Zhang H, Kim DK, Kim SJ, Gambhir SS, Steinman L and Wyss-Coray T. Glia-dependent TGF-beta signaling, acting independently of the TH17 pathway, is critical for initiation of murine autoimmune encephalomyelitis. The Journal of clinical investigation. 2007; 117:3306-3315.
35. Svet-Moldavsky GJ, Svet-Moldavskaya IA and Raffkina LI. Various types of acquired resistance to experimental ‘allergic’ encephalomyelitis. Nature. 1959; 184:1552-1553.
36. Attwell D, Buchan AM, Charpak S, Lauritzen M, Macvicar BA and Newman EA. Glial and neuronal control of brain blood flow. Nature. 2010; 468:232-243.
37. Haydon PG and Carmignoto G. Astrocyte control of synaptic transmission and neurovascular coupling. Physiological reviews. 2006; 86:1009-1031.
38. Koehler RC, Roman RJ and Harder DR. Astrocytes and the regulation of cerebral blood flow. Trends in neurosciences. 2009; 32:160-169.
39. Petzold GC and Murthy VN. Role of astrocytes in neurovascular coupling. Neuron. 2011; 71:782-797.
40. Takano T, Tian GF, Peng W, Lou N, Libionka W, Han X and Nedergaard M. Astrocyte-mediated control of cerebral blood flow. Nature neuroscience. 2006; 9:260-267.
41. Delekate A, Fuchtemeier M, Schumacher T, Ulbrich C, Foddis M and Petzold GC. Metabotropic P2Y1 receptor signalling mediates astrocytic hyperactivity in vivo in an Alzheimer’s disease mouse model. Nature communications. 2014; 5:5422.
42. Confavreux C, Vukusic S, Moreau T and Adeleine P. Relapses and progression of disability in multiple sclerosis. The New England journal of medicine. 2000; 343:1430-1438.
43. Coles AJ, Wing MG, Molyneux P, Paolillo A, Davie CM, Hale G, Miller D, Waldmann H and Compston A. Monoclonal antibody treatment exposes three mechanisms underlying the clinical course of multiple sclerosis. Annals of neurology. 1999; 46:296-304.
44. Steinman L. Immunology of relapse and remission in multiple sclerosis. Annual review of immunology. 2014; 32:257-281.
45. Schenk U, Frascoli M, Proietti M, Geffers R, Traggiai E, Buer J, Ricordi C, Westendorf AM and Grassi F. ATP inhibits the generation and function of regulatory T cells through the activation of purinergic P2X receptors. Science signaling. 2011; 4:ra12.
46. Casati A, Frascoli M, Traggiai E, Proietti M, Schenk U and Grassi F. Cell-autonomous regulation of hematopoietic stem cell cycling activity by ATP. Cell death and differentiation. 2011; 18):396-404.
47. Bedogni F, Cobolli Gigli C, Pozzi D, Rossi RL, Scaramuzza L, Rossetti G, Pagani M, Kilstrup-Nielsen C, Matteoli M and Landsberger N. Defects during Mecp2 null embryonic cortex development precede the onset of overt neurological symptoms.cerebral cortex. 2015.
48. Verderio C, Cagnoli C, Bergami M, Francolini M, Schenk U, Colombo A, Riganti L, Frassoni C, Zuccaro E, Danglot L, Wilhelm C, Galli T, Canossa M and Matteoli M. TI-VAMP/VAMP7 is the SNARE of secretory lysosomes contributing to ATP secretion from astrocytes. Biology of the cell. 2012; 104:213-228.
49. Furlan R, Cuomo C and Martino G. Animal models of multiple sclerosis. Methods in molecular biology. 2009; 549:157-173.

