INTRODUCTION
Hepatocellular carcinoma (HCC) is the second most common cause of cancer-related death in the world [1], and more than 50% of liver cancers are associated with hepatitis B virus (HBV) infection [2]. The 5-year survival rate of patients with liver cancer is lower than 15% [3]. The hepatic microenvironment of the host, including the presence of various cytokines and chemokines, is a critical factor affecting the progress and metastasis of HCC. Alterations in the T helper cell (Th1) cytokine profile are associated with metastasis. Specifically, a significant decrease in Th1 cytokines [interleukin (IL)-1A, IL-1B, IL-2, IL-12A, IL-12B, IL-15, interferon (IFN)-γ, and tumor necrosis factor (TNF)] and concomitant increase in Th2 cytokines (IL-4, IL-5, IL-8, and IL-10) are observed in tumor-adjacent noncancerous hepatic tissues in metastatic HCC patients [4, 5].
T follicular helper cells (Tfh) are indispensable for the development of germinal centers (GCs) and the generation of long-term humoral immunity [6–8]. Tfh cells were first discovered in lymphoid tissues with the C-X-C motif chemokine receptor CXCR5 as the canonical marker. However, their memory counterparts in peripheral blood, especially the antigen-specific Tfh-like population, have not been well characterized [9–12]. Circulating Tfh cells typically present as CXCR5+CD45RA−CD4+CD3+ and are further classified into three subsets based on CXCR3 and CCR6 expression, namely, Th1-like (CXCR3+CCR6−), Th2-like (CXCR3−CCR6−), and Th17-like (CXCR3−CCR6+) subsets. The Th2-like subset is the primary helper to B cells in the production of immunoglobin [13]. Tfh cells produce interleukin-21 (IL-21), which can activate both innate and adaptive immune responses, and enhance antitumor and antiviral responses [14, 15]. Alterations in Tfh cells are associated with autoimmune and immunodeficiency diseases, and with solid tumors [16, 17]. The frequency of circulating CXCR5+ CD4+ Tfh cells is increased in chronic hepatitis B patients and positively correlated with alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels [18, 19]. Circulating CXCR5+ CD4+ T cells are increased in HBV-related liver cirrhosis and decreased in HBV-related HCC [20].
CXCL13, the only ligand for CXCR5, drives the migration of CXCR5-expressing B cells and Tfh cells to lymphoid tissues and is necessary for the development of secondary lymphoid tissue [21–23]. Circulating CXCL13 levels are increased in many autoimmune diseases and correlated with clinical outcomes [24–26]. CXCL13 is also required for an effective immune response against HBV [27]. Previous work from our group suggested that polymorphisms of CXCL13 are associated with the lack of response to the hepatitis B vaccine [28]. However, the association of CXCL13 with the prognosis of HBV-related HCC remains unclear. In the present study, we examine the frequency and distribution of Tfh cells and Th1-, Th2-, and Th17-like subsets in HBV-related HCC. The secretion of Tfh-related cytokines in patients with HBV-related HCC and the association between alterations in CXCL13 and the progression and prognosis of HCC are evaluated.
RESULTS
Tfh subpopulations were redistributed in HCC
The frequencies of circulating Tfh cells and cell subsets were detected in patients with HCC and healthy controls (HC) (Figure 1A). The frequency of CXCR5+CD45RA− cells did not differ significantly between the HCC and HC groups (Figure 1B); however, IL-21 concentration was significantly lower in the culture supernatant of sorted CXCR5+CD45RA−CD4+CD3+ and IgD+CD27−CD19+CD3− cells from HCC patients than in that from HC (Figure 1C). IgG, IgA, and IgM concentrations in the supernatant could not be tested on day 12 of culture because of extensive cell death in purified cells from HCC patients on days 7 and 8. This suggested that the viability of cells from HCC patients was poorer than that from HC. The Th1-like and Th17-like subsets of circulating Tfh cells were markedly decreased in HCC patients (P = 0.024 and P < 0.001, respectively, Figure 1D), while the Th2-like subset was increased (P = 0.013, Figure 1D) and associated with reduced tumor numbers in HCC patients (P = 0.029, Supplementary Table 2). In line with these observations, the ratios of Th1-/Th2-like and Th17-/Th2-like subsets of circulating Tfh cells were remarkably lower in HCC patients than in HC (P = 0.007 and P < 0.001 respectively, Figure 1E), implying that the tumor environment promoted an increase in the Th2-like subset, which is the primary helper in antibody production, and a decrease in the Th17-like subset, which is the major producer of IL-21.
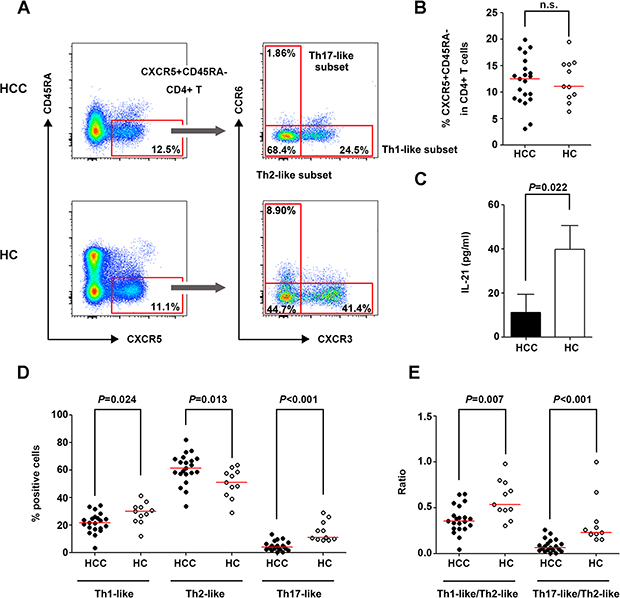
Figure 1: Circulating Tfh cells and cell subsets in patients with HCC and HC. (A) Gating strategies of circulating Tfh cells and their subsets in HCC and HC. (B) Percentage of CXCR5+CD45RA−cells in CD4+ lymphocytes in HCC (n = 21) and HC (n = 11). (C) IL-21 concentration in the co-culture supernatant of purified CXCR5+CD45RA−CD4+CD3+ and IgD+CD27−CD19+CD3− cells. (D) Distribution of Th1-like, Th2-like, and Th17-like subsets in circulating Tfh cells in HCC and HC. (D) Ratio of Th1-/Th2-like Tfh subsets and Th17-/Th2-like Tfh subsets in HCC and HC. Red lines in (B), (D) and (E) show the median values.
The expressions of ICOS, PD-1, CXCR3, CCR6, and CCR7, which are phenotypic markers for circulating Tfh cells [9, 11, 12, 29], were detected in CXCR5+CD45RA−CD4+ T cells in the HCC and HC groups (Figure 2A). The frequencies of ICOS+ and CCR7+ CXCR5+CD45RA−CD4+ T cells were higher in HCC patients than in HC (P = 0.002 and P < 0.001, respectively, Figure 2B), and the percentage of ICOS+ Tfh cells was correlated with the incidence of cirrhosis in HCC patients (P = 0.039, Supplementary Table 2). Considering that circulating Tfh cells have a central memory phenotype [30, 31], the frequency of circulating central memory Tfh cells (CXCR5+CCR7+CD45RA−CD4+) was compared between the HCC and HC groups. The results showed a significantly higher frequency of these cells in HCC patients than in HC (P = 0.006, Figure 2C).
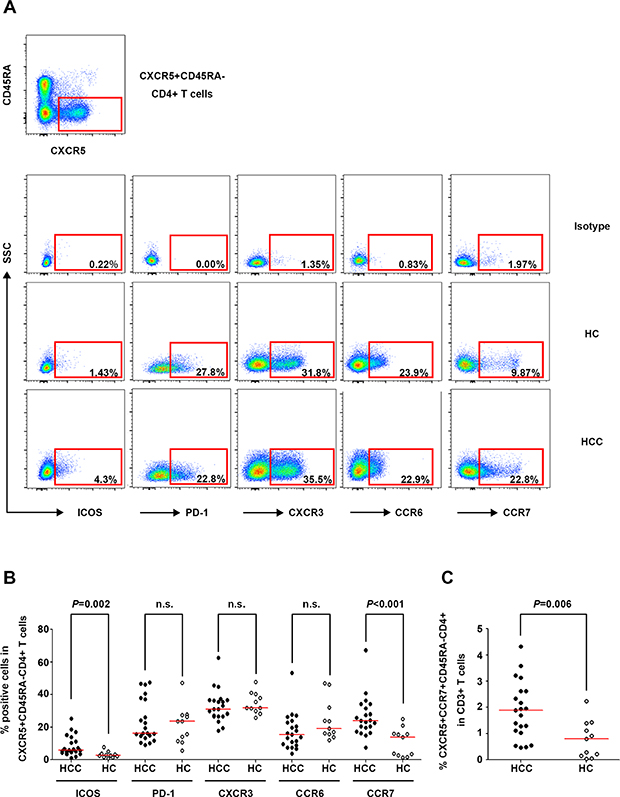
Figure 2: Phenotypic marker expression on circulating CXCR5+CD45RA−CD4+ T cells in HCC and HC. (A) Expression of the phenotypic markers ICOS, PD-1, CXCR3, CCR6, and CCR7 on circulating Tfh cells in the isotype control (upper panel), a representative HCC patient (middle panel), and a representative HC (lower panel). (B) Percentages of ICOS+, PD-1+, CXCR3+, CCR6+, and CCR7+ cells among circulating CXCR5+CD45RA−CD4+ T cells in HCC (n = 21) and HC (n = 11). (C) Frequencies of circulating CXCR5+CCR7+CD45RA−CD4+ cells in HCC and HC. Red lines in (B) and (C) show the median values.
Survival analysis showed that the frequency of the Th17-like subset in circulating Tfh cells was positively correlated with recurrence-free survival (log-rank test: P = 0.010; Gehan-Breslow-Wilcoxon test: P = 0.025, Supplementary Figure 1). Taken together, these results indicated that the distribution of Tfh subpopulations was altered in HCC, and the Th17-like subset was identified as a potential prognostic indicator in HCC.
Infiltrated CXCR5+CD45RA−CD4+ T cells were increased in tumor tissues compared with para-tumor tissues
Characterization of tumor-infiltrating CXCR5+CD45RA−CD4+ T cells in 12 pairs of matched tumor and para-tumor tissues showed that the population of CXCR5+CD45RA− cells among CD4+ T cells was higher in tumor tissues than in para-tumor tissues (P = 0.012) (Figure 3A and 3B). Unlike the Tfh subsets in peripheral blood, the majority of infiltrated Tfh cells belonged to the Th1-like subset, the frequency of which was significantly higher in tumor tissues than in para-tumor tissues (P = 0.019). The frequency of the Th2-like subset was relatively lower and no Th17-like subset was detected in infiltrated Tfh cells (Figure 3C). The percentage of ICOS+ Tfh cells was remarkably higher in tumor tissues (P = 0.002, Figure 3D) than in para-tumor tissues, showing a similar pattern to that of peripheral blood.
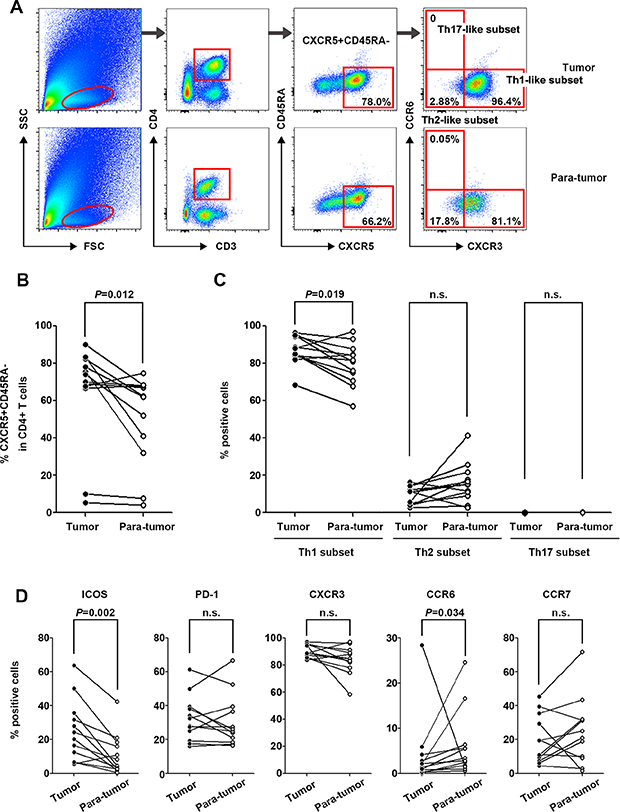
Figure 3: Characterization of tumor-infiltrating CXCR5+CD45RA−CD4+ T cells in tumor and para-tumor tissues. (A) Typical flow cytometry charts of tumor-infiltrated Tfh cells in tumor (upper panel) and para-tumor (lower panel) tissues. (B) Frequencies of CXCR5+CD45RA−CD4+ lymphocytes in tumor and para-tumor tissues (n = 12). (C) Distribution of Th1-like, Th2-like, and Th17-like tumor-infiltrated CXCR5+CD45RA−CD4+ T cell subsets in tumor and para-tumor tissues. (D) Phenotypic marker expression on infiltrated CXCR5+CD45RA−CD4+ T cells in tumor and para-tumor tissues.
Serum CXCL13 level was increased in HCC and associated with recurrence-free survival
Given the role of cytokines in the differentiation, migration, and function of Tfh cells, the serum concentrations of nine Tfh-relevant cytokines (CXCL13, IL-21, TNF-α, IFN-γ, IL-12, IL-10, IL-4, IL-17, and IFN-α) were assessed in HCC subjects and HC. CXCL13, the chemokine driving Tfh movement, was significantly up-regulated in HCC patients (P = 0.016, Figure 4A) and positively associated with improved recurrence-free survival (log-rank test: P = 0.037, Gehan-Breslow-Wilcoxon test: P = 0.023, Figure 4B), whereas it was unrelated to overall survival (log-rank test: P = 0.696, Gehan-Breslow-Wilcoxon test: P = 0.547, Figure 4C)
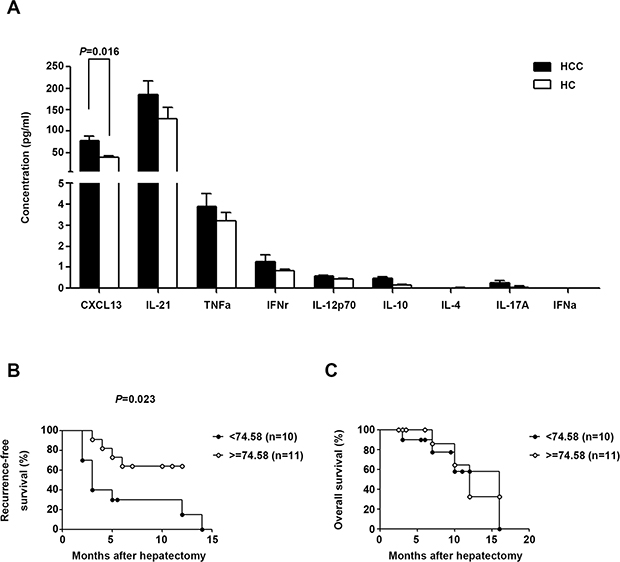
Figure 4: Tfh cell-related serum cytokine production in HCC and HC. (A) Serum concentrations of cytokines in HCC (n = 21) and HC (n = 11) subjects. Data are expressed as the mean and SEM. (B) and (C) Correlation of serum CXCL13 with recurrence-free survival or overall survival. Patients were divided into two groups according to the median concentration of CXCL13.
CXCL13 was regionally increased in poorly differentiated HCC tumor tissues
The nine Tfh-relevant cytokines were measured in homogenates of 15 paired tumor and para-tumor tissues from HCC patients. Only IL-21 was higher in tumor homogenates than in para-tumor homogenates (P < 0.001, Figure 5A) and positively associated with recurrence-free survival (Gehan-Breslow-Wilcoxon test, P = 0.033, Figure 5B). Because plasma CXCL13 levels are correlated with those in the liver in mouse models [27], and our data showed an elevated serum concentration of CXCL13 in HCC patients, the expression status of CXCL13 in tumor tissues was further examined. CXCL13 is secreted by dendritic cells (DC), Tfh cells, and B cells; however, our results showed that CXCL13 was secreted into the culture supernatant of several HCC cell lines (Figure 5C). The results of array-based comparative genome hybridization (aCGH) showed that the CXCL13 gene was deleted in 8/25 patients (Figure 5D). These results implied that liver cells may have the capacity to produce CXCL13, and the capability of transformed liver cells, such as tumor cells, to produce CXCL13 may be impaired by CXCL13 gene deletion.
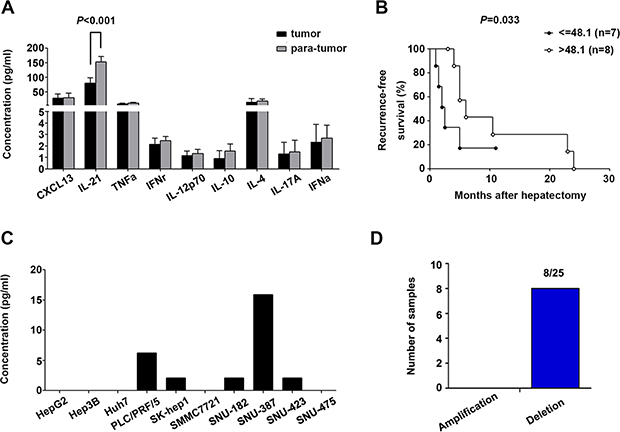
Figure 5: Tfh cell-related cytokine production in HCC tumor tissues and cell lines. (A) Levels of Tfh-related cytokines in homogenates of 15 paired tumor and para-tumor tissues. (B) IL-21 in tumor homogenates was associated with recurrence-free survival. (C) Concentration of CXCL13 in the supernatants of ten different HCC cell lines. (D) Amplification and deletion status of CXCL13 in 25 paired HCC tumor and para-tumor tissues.
Measurement of in situ CXCL13 expression by immunohistochemistry (IHC) showed that CXCL13 was expressed in a clustering manner in tumors (Figure 6A and 6B). Samples were then grouped according to the degree of tumor differentiation. In the poorly differentiated group, both CXCL13 and CXCR5 were remarkably higher in tumor tissues (Figure 6C), indicating that CXCL13 was regionally increased in poorly differentiated HCC tumor tissues.
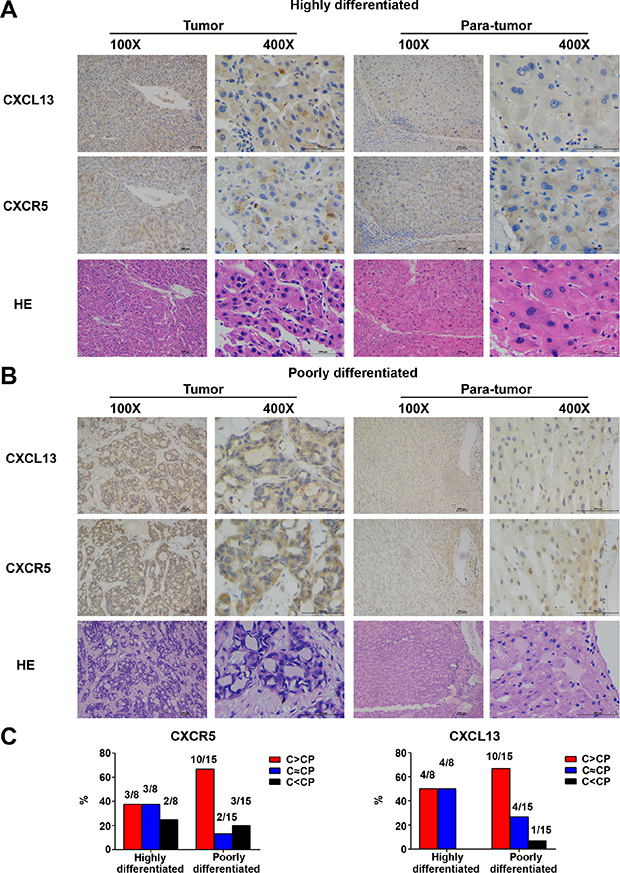
Figure 6: In situ expression of CXCL13 and CXCR5 in 23 paired HCC tumor and para-tumor tissues. (A) Representative images of CXCL13 and CXCR5 immunohistochemistry staining in highly differentiated tumor and para-tumor tissues. (B) Representative images of CXCL13 and CXCR5 immunohistochemistry staining in poorly differentiated tumor and para-tumor tissues. (C) Comparison of CXCL13 and CXCR5 between tumor and para-tumor tissues. C and CP in the figure stand for tumor and para-tumor tissues, respectively.
Serum CXCL13 predicted the prognosis of HCC
To confirm the role of serum CXCL13 as a predictor of recurrence-free survival, the serum concentration of CXCL13 was measured in 111 HCC patients with detailed 5 years follow-up information. CXCL13 level (median value: 78.53 pg/ml) was highly correlated with recurrence-free survival, but not overall survival, in HCC patients (log-rank test: P = 0.011, Gehan-Breslow-Wilcoxon test: P = 0.026, (Figure 7A and 7B). In the univariate analysis, in addition to serum CXCL13, serum AFP and γ-GT levels were associated with recurrence-free survival (CXCL13: P = 0.011, AFP: P = 0.013, γ-GT: P = 0.004, Table 1). Since a specific serum AFP cutoff value associated with the prognosis of HCC patients after surgical treatment has not been established, the trend in present study that high level of AFP predicted worse prognosis was in accordance with previous publications [32–36]. To further evaluate the role of serum CXCL13 in the prediction of recurrence-free survival of HBV-related HCC patients, survival analysis were performed especially in those with a lower AFP level (≤200 ng/ml). The results showed significant correlation still existed between serum CXCL13 and recurrence-free survival (Figure 7C), but not over-all survival (Figure 7D). In the multivariate analysis, CXCL13 and γ-GT (CXCL13: P = 0.019, γ-GT: P = 0.008, Table 1) remained important predictors of recurrence, confirming the role of serum CXCL13 as a prognostic factor in HCC.
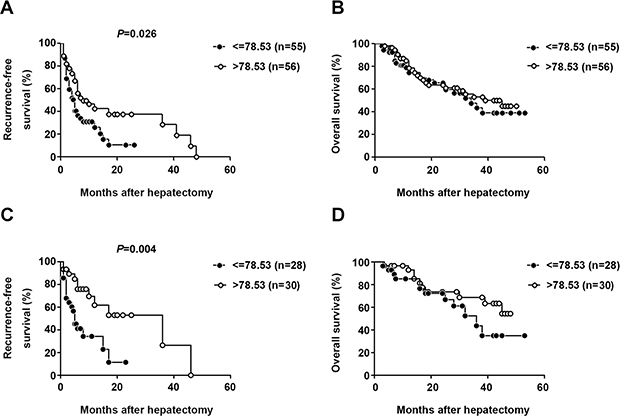
Figure 7: Elevated serum CXCL13 was correlated with recurrence-free survival but not overall survival. Association between serum CXCL13 and recurrence-free survival (A) or overall survival (B) Patients were divided into two groups according to the median concentration of CXCL13. Association between serum CXCL13 and recurrence-free survival (C) or overall survival (D) especially in HCC patients with AFP lower than 200ng/ml. Patients were divided into two groups according to the same cutoff value of CXCL13 in (A) and (B).
Table 1: Univariate and multivariate analyses of different parameters associated with HCC using the Kaplan-Meier method and Cox’s model
Variables |
|
N |
Median survival |
P |
|
|---|---|---|---|---|---|
Univariateanalysis |
Multivariateanalysis |
||||
Gender |
Male |
90 |
9.24 |
0.744 |
|
Female |
21 |
9.25 |
|||
Age |
< 50 |
41 |
7.89 |
0.116 |
|
≥ 50 |
70 |
10.29 |
|||
CXCL13 (pg/ml) |
≤ 78.53 |
55 |
8.08 |
0.011 |
0.019 |
> 78.53 |
56 |
10.84 |
|||
Cirrhosis |
Positive |
96 |
9.00 |
0.575 |
|
Negative |
15 |
9.27 |
|||
Tumor number |
1 |
90 |
9.55 |
0.233 |
|
> 1 |
21 |
8.08 |
|||
Encapsulation |
Complete |
87 |
9.51 |
0.547 |
|
Incomplete |
24 |
8.40 |
|||
AFP (μg/L) |
≤ 200 |
58 |
11.75 |
0.013 |
|
> 200 |
53 |
7.66 |
|||
ALT (U/L) |
≤ 40 |
66 |
8.78 |
0.703 |
|
> 40 |
45 |
10.09 |
|||
AST (U/L) |
≤ 40 |
59 |
10.24 |
0.091 |
|
> 40 |
52 |
8.36 |
|||
TBI (μmol/L |
≤ 17.1 |
74 |
9.38 |
0.631 |
|
> 17.1 |
37 |
9.00 |
|||
γ-GT (U/L) |
≤ 50 |
45 |
13.90 |
0.004 |
0.008 |
> 50 |
66 |
7.89 |
|||
ALP (U/L) |
≤ 150 |
92 |
9.17 |
0.254 |
|
> 150 |
19 |
9.60 |
|||
ALB (g/L) |
≤ 35 |
19 |
10.67 |
0.557 |
|
> 35 |
92 |
9.00 |
|||
Child-Pugh |
A |
93 |
8.89 |
0.945 |
|
B |
5 |
9.00 |
|||
BCLC stage |
A |
53 |
9.75 |
0.280 |
|
B and C |
42 |
8.42 |
|||

