INTRODUCTION
Every year around one million new cases of gastric cancer are diagnosed globally. In 2012, 723,000 people died from gastric cancer, ranking it the 4th most common cancer-related cause of death. Complete surgical resection together with perioperative chemotherapy represents the standard of care for curative treatment of patients with gastric cancer [1-3]. However, even after multimodal therapy up to 40% of the patients experience disease recurrence and up to 30% die within 12 months [4].
Peritoneal dissemination is a common cause of failure after curative treatment for gastric cancer. Peritoneal recurrence occurs in 17% of patients undergoing resection with curative intent and is associated with a dismal survival [5, 6]. Due to the frequent occurrence and the strong prognostic relevance of peritoneal metastases, detection of free intraperitoneal tumor cells (FITC) has been suggested as a prognostic and predictive biomarker in gastric cancer patients [7, 8]. Detection of FITC may help to recognize those patients considered for curative therapy who are at high-risk for early tumor relapse and might benefit from intensified treatments such as hyperthermic intraperitoneal chemotherapy (HIPEC) [9]. Numerous studies have so far been conducted on the prognostic and predictive value of FITC in gastric cancer. Although FITC are found in 6-49% of gastric cancer patients considered for curative surgery [10-13], it’s predictive and prognostic value has remained unclear due to inconsistent detection techniques and results of the individual studies. This clinical uncertainty is reflected by inconsistent recommendations made by different guidelines on the use of FITC in the management of gastric cancer [1-3, 14].
To clarify the role of intraperitoneal lavage cytology as a prognostic biomarker in gastric cancer, we performed a systematic review with meta-analyses of studies on the prognostic significance of FITC detection in peritoneal lavage samples of patients with gastric cancer considered for curative therapy.
RESULTS
Baseline study characteristics
In total, we included 64 studies [10-13, 15-68] with a cumulative sample size of 12, 883 patients (Figure 1). These studies had a median sample size of 134 (52 - 1297) patients and were published between 1978 and 2014 (Table 1). The included studies were conducted in Western institutions in 19% and in Asian institutions in 81%. Patients with stage IV disease were enrolled in 30 (47%) studies. The median follow up across all studies was 35 (18 - 82) months. FITC were detected by cytology in 43 (67%) studies (38 studies used Papanicolaou staining, 5 studies used H&E staining), by immunocytochemistry (ICC) in 5 (8%) studies and by RT-PCR in 29 (45%) studies (Table 2). The majority of studies used Carcinoembryonic antigen (CEA) for molecular tumor cell detection. In 22 studies CEA expression was analyzed and in seven studies CK20 expression was analyzed. Further markers included CK19, CD44, Caspase 9, MINT, MAGE, MMP 7, CA125, TGFβ, RegIV, FABP1, Muc2, IL-17 and CDH1. The detection rate of FITC across the included studies varied markedly (median: 23%; range 6% - 58%) and showed a strong association with patients’ stage of disease and in particular the inclusion of patients with overt peritoneal metastases. FITC were detected prior to resection in 62 (97%) studies and pre- as well as postoperativelv in 2 (3%) studies. OS, DSS, DFS and PRFS was reported in 51 (80%), 7 (11%), 11 (17%) and 21 (33%) studies, respectively. Hazard ratios for multivariate analysis could be extracted in 21 studies (ten that performed cytology, eight that performed RT-PCR and three that performed both). Fifteen studies were graded with a low risk of bias (Appendix 1). Funnel plot analyses did not indicate significant publication bias for the analyzed outcomes (Appendix 2).
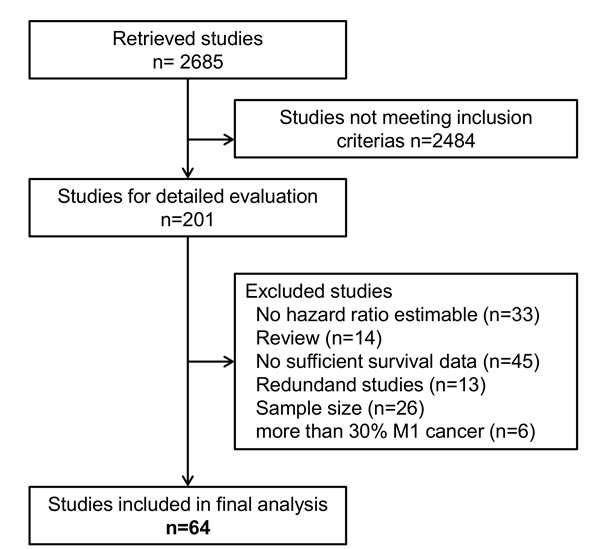
Figure 1: Flow diagram showing the selection process for relevant studies.
Table 1: Baseline characteristics of included studies
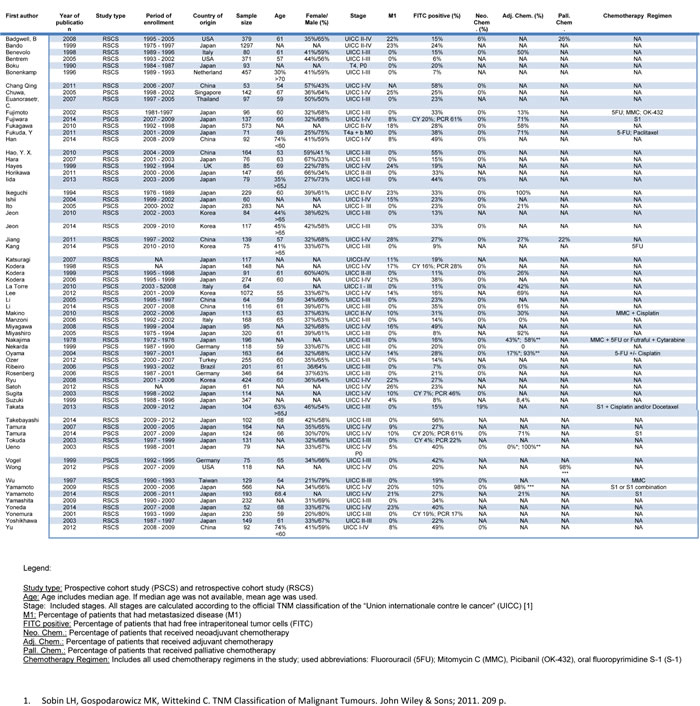
Table 2: Design variables of included studies
Prognostic value of FITC detection
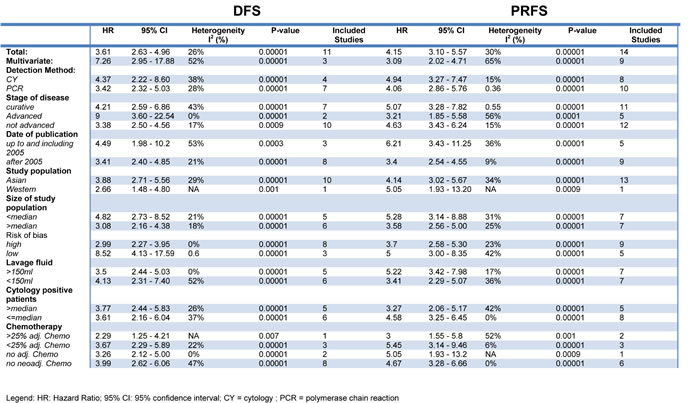
Some 51 studies with a cumulative sample size of 11, 005 patients reported on OS.10-13, 23-32, 34, 36, 38-43, 46-49, 51-60, 63-65, 67-7 The pooled analyses of the results from these studies showed a strong prognostic value of FITC detection (HR 3.27, 95% CI 2.82 - 3.78; n = 51; I² = 74%) (Figure 2). This result could be verified in the 35 studies with curatively resected patients and a cumulative sample size of 5908 (3.51; 3.01 - 4.08; n = 35; I2 = 48%) (Table 3) [10-13, 16-19, 22, 24, 25, 30-35, 37, 41, 44-49, 51, 56, 59, 60, 62, 65, 66, 68, 69]. Sensitivity analyses failed to identify a single study as a reason for the observed statistical heterogeneity. Meta-analysis of the results from 17 studies with multivariate analyses confirmed the prognostic association of FITC detection with reduced heterogeneity (2.45; 2.04 - 2.94; n = 17; I² = 39%) [11, 12, 21, 23, 25, 32, 33, 38, 40, 48, 52, 54, 62, 63, 65, 66, 68]. Furthermore, we found significant associations of FITC detection and long-term outcome in the pooled analyses on DFS (3.61; 2.63 - 4.96; n = 11; I² = 26%)[21, 23, 27, 34, 44, 48, 53, 64, 70, 71] and PRFS (4.15, 3.10 - 5.57; n = 14; I² = 30%) (Table 4) [12, 23, 31, 38, 42, 44, 55, 56, 64-66, 72, 73].
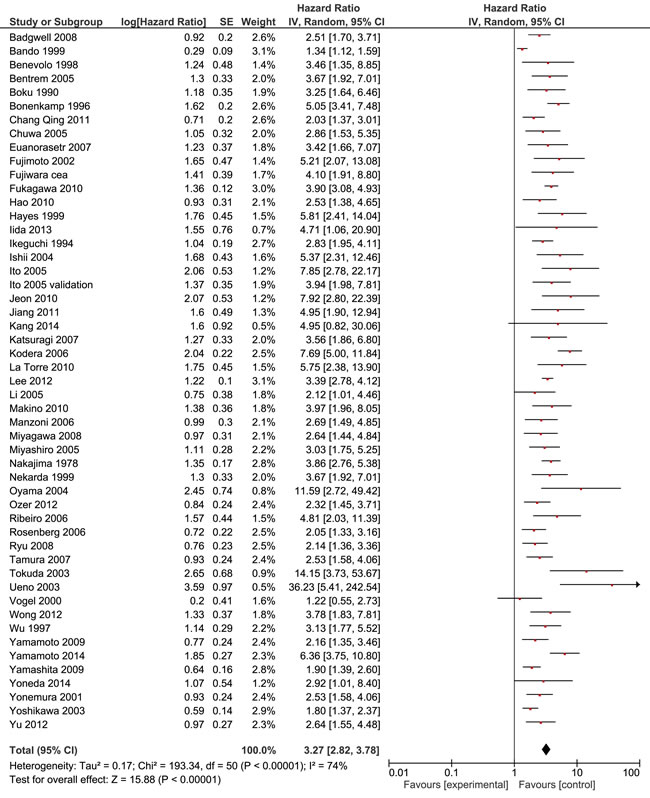
Figure 2: Forest plot for the prognostic value of FITC in patients with gastric cancer (Overall survival).
Table 3: Subgroup analyses for overall survival in FITC positive patients and curatively resected FITC positive patients.
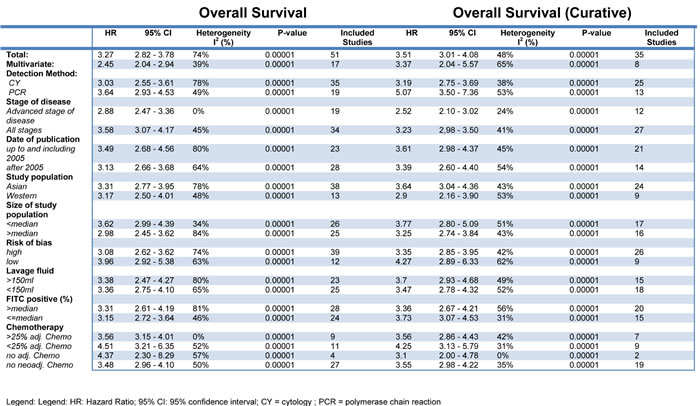
Table 4: Subgroup analyses for disease free survival (DFS) and peritoneal recurrence free survival (PRFS) in FITC positive patients.
Subgroup analyses
Subgroup analyses were performed to assess the impact of the detection method on the results. These analyses revealed a prognostic association of FITC detection by cytology with OS (3.03; 2.55 - 3.61; n = 35; I² = 78%) [10, 11, 13, 15-19, 21, 22, 24, 25, 28, 30, 33, 34, 38-41, 43, 44, 46, 47, 49-52, 57, 60, 61, 63, 65, 66, 69]. Despite a lower number of studies we observed a more pronounced prognostic value for pooled analyses of studies using RT-PCR (3.64; 2.93 - 4.53; n = 19; I² = 49%) [12, 20, 23, 26, 35, 38, 42, 45, 48, 53, 55-57, 59, 62, 64, 65, 67, 68]. This difference reached statistical significance in the test of interaction for the subgroup of patients who underwent potentially curative resection (p = 0.012). The kind of detection method had no impact on the prognostic value with respect to DFS and PRFS (Table 3, Table 4).
We next evaluated the prognostic value of FITC in patients with advanced stages as compared to the entire patient cohort. Only one study reported outcome selectively for patients with early stage of disease (without lymph node metastases) [51]. There was a significant association of FITC detection with OS in patients with advanced disease as well as the entire cohort. However, in particular for patients who underwent a potentially curative resection, the magnitude of effect was lower in case of advanced disease (2.52; 2.10 - 3.02; n = 12; I² = 24%)[16, 18, 25, 27, 30, 36, 47, 51, 59, 60, 65, 66] than for studies including the entire population (3.23; 2.98 - 3.50; n = 27; I² = 41%)[10-13, 17, 19, 22, 24, 31-35, 41, 45, 46, 48, 49, 51, 56, 59, 62, 65, 68, 69] (p = 0.014; test of interaction). The increased prognostic value of FITC detection in patients with less advanced disease was confirmed for PRFS (p = 0.008, test of interaction). There was not enough data for a pooled analysis of advanced disease for DFS (n = 2).
Previous studies suggested genetic differences between gastric cancers dependent on geographic location [74-76]. We therefore evaluated the prognostic value of FITC detection separately for these cohorts. These analyses showed a significant association between FITC detection and OS for Asian population (3.31; 2.77 - 3.95; n = 38; I² = 78%) [11-13, 16, 18, 20-22, 24, 26, 30-35, 38, 40, 41, 43, 45, 46, 48, 52, 55-57, 60-68] as well as Western population (3.17; 2.50 - 4.01; n = 13; I² = 48%) [10, 15, 17, 19, 28, 39, 44, 47, 49-51, 59, 69]. Significant associations for both cohorts were also present for patients who underwent a curative resection as well as the outcomes DFS and PRFS with no significant difference between both population as indicated by the tests of interaction.
Systemic chemotherapy has become common practice in the curative therapy of advanced gastric cancer [1, 77, 78], though the optimal regimen is still subject to intensive research [77]. Previous studies showed that 60-90% of FITC positive patients can be converted to FITC negative by neoadjuvant chemotherapy and thus improve survival [79, 80]. We therefore evaluated the prognostic value of FITC depending on the administration of neoadjuvant and adjuvant chemotherapy, respectively. These analyses revealed a strong association of FITC detection and OS, DFS and PRFS independent of the administration of neoadjuvant or adjuvant chemotherapy.
To exclude that the observed results were primarily caused by studies with low methodological quality, further analyses were stratified for the risk of bias. While studies with low (3.96; 2.92 - 5.38; n = 12; I² = 63%)[11, 28, 38, 39, 47, 48, 51, 55, 65] and high risk of bias (3.08; 2.62 - 3.62; n = 39; I² = 74%)[10, 13, 15-24, 26, 30-34, 40, 41, 43-46, 49, 50, 52, 56, 57, 59-64, 66-69] showed a significant prognostic value for FITC detection on OS, the effect was more pronounced in studies with low risk of bias (p = 0.15; test of interaction). The enhanced prognostic value reported in studies with a low risk of bias supports the validity of the finding that FITC detection represents a strong prognostic marker in gastric cancer.
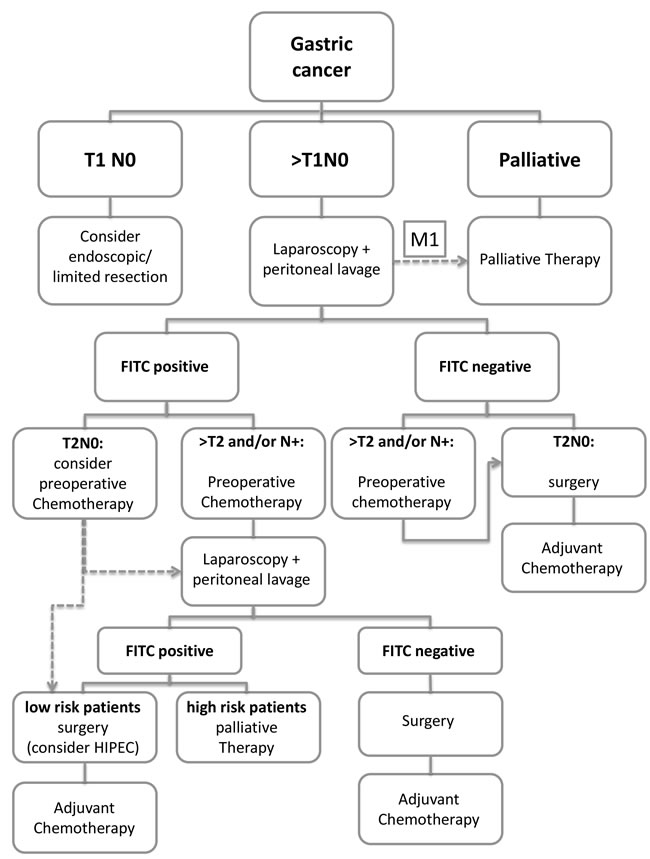
Figure 3: Treatment algorithm for gastric cancer.
DISCUSSION
This systematic review and meta-analysis shows a marked association of FITC with overall survival, disease free survival and peritoneal recurrence free survival of patients with gastric cancer scheduled for curative therapy.
Although the first studies on detection of FITC in gastric cancer patients have been published over 60 years ago [81], the role of FITC detection in the management of patients with gastric cancer has remained highly controversial. This may in part be explained by different study designs and insufficient statistical power of individual studies, in particular for subpopulations of patients with different extent of disease. In line with this, current gastric cancer treatment guidelines do not provide uniform recommendations on the use of peritoneal lavage. Although the majority of guidelines classify FITC detection as metastatic (M1) disease, these recommendations are based on single or a few individual studies, are limited to peritoneal lavage cytology and do not provide any standardization with respect to the sampling time and sampling/detection methodology (i.e. amount of lavage fluid, kind of staining). While the NCCN guidelines recommend a staging laparoscopy with peritoneal washings for cytology for stage IB and higher, the European ESMO, ESSO, ESTRO guidelines are less stringent and recommend a staging laparoscopy with or without peritoneal washings for malignant cells in these patients [1, 2]. Furthermore, there is no consensus regarding the consequences of a positive peritoneal cytology on patients’ clinical management. In the NCCN guidelines a positive peritoneal cytology is considered a criterion of unresectability for cure. The European guidelines do not comment on the consequences for surgical resection and the German guidelines state no relevance on patients’ further management [1, 2, 14]. As in these guidelines positive peritoneal cytology is classified as M1 disease and palliative treatment is recommended in M1 patients, there is urgent need to clarify which patients at what timepoint should undergo peritoneal lavage sampling by what methodology [26, 67, 73].
The results of the present meta-analysis confirm FITC as poor prognostic marker in patients with gastric cancer. Importantly, our results demonstrate the prognostic value of FITC detection to be dependent on the extent of disease. A more pronounced prognostic relevance is shown in patients with limited disease and a curative resection, respectively. Identification of strong prognostic markers might be useful in the management of gastric cancer patients in various ways. First, prognostic biomarkers might, moreover, serve as predictive biomarkers in patients considered for perioperative chemotherapy. Second, reliable prognostic information may be of particular help in decision-making for further treatment in elderly patients or patients with severe comorbidities who may be at increased risk for complications and poor outcome after multi-modal therapy. As total gastrectomy is associated with relevant morbidity and 90-day mortality, [82] a strong prognostic biomaker might be helpful to avoid surgery in high-risk patients with a poor prognosis. Third, it may be helpful in the management of young patients with excellent performance status who may be able to tolerate intensive therapy. Fourth, validation of FITC as strong prognostic biomarkers provide a valid scientific rationale for subsequent research to further characterize these cells on a molecular level. As targeted therapies are emerging for gastric cancer, [83] it is of particular interest, if molecular analysis of free intraperitoneal tumor cells might serve as a predictive biomarker for targeted agents in gastric cancer patients.
There is indeed increasing effort to identify patients with gastrointestinal malignancies and peritoneal metastases who benefit from intensified therapies such as HIPEC [84-86]. At present, these efforts mainly focus on patients with overt peritoneal metastases and showed promising results for colorectal cancer [87, 88]. The findings were much more modest for gastric cancer patients with overt peritoneal metastasis [89, 90] and may be explained by limitations to achieve complete cytoreduction [91]. These data suggest FITC positive gastric cancer without further distant metastasis as a promising subgroup of patients who might benefit from HIPEC. The first randomized controlled study to examine the benefit of extensive intraoperative peritoneal lavage followed by intraperitoneal chemotherapy in FITC positive gastric cancer showed promising results [92]. Further randomized controlled trials have already been initiated (ClinicalTrials.gov; NCT01683864). The results may redefine the treatment of FITC positive gastric cancer.
The optimal method of FITC detection remains to be determined. As outlined current guidelines are restricted to conventional cytology without providing further information on the kind of staining. Our results indicate a prognostic value of FITC detection by cytology as well as molecular techniques. To date, only few studies directly compared cytology by Papanicolaou staining with molecular detection by PCR [23, 29, 38, 53, 55-57, 59, 65]. Detection methods using PCR offer a considerably higher detection sensitivity at a marginally lower specificity (Appendix 3). This meta-analysis demonstrates a similar prognostic value for both detection methods. The results of the above studies imply a potential superiority of FITC detection by PCR, that needs to be substantiated within prospective trials before valid recommendations can be made in guidelines.
The use of peritoneal lavage in patients undergoing multimodal therapy remains a further question to be answered. While metabolic imaging has been proposed as a strategy for early response assessment in patients with cancers of the esophagogastric junction and stomach [93-95], peritoneal washings with detection of FITC may offer an additional or alternative approach. There is indeed evidence that clearance of positive peritoneal cytology by systemic chemotherapy is associated with improved outcome after surgical resection for gastric cancer [96, 97]. However, controlled clinical trials are required to clarify the benefit of surgical resection in patients who remain positive for FITC after chemotherapy.
One important question that needs answering is how to proceed with FITC positive patients with potentially curative gastric cancer. Considering the results of this meta-analysis we would like to propose a therapeutic algorithm (Figure 3). However, the feasibility and clinical utility of this algorithm needs to be tested in controlled clinical trials.
In conclusion, this meta-analysis reveals FITC detection as poor prognostic marker in gastric cancer patients scheduled for curative therapy. The prognostic value of FITC was noted across detection methods, administration of chemotherapy and geographic location, though a more pronounced effect was observed in patients with less advanced disease. These results support efforts to use FITC as a predictive biomarker and may contribute to the development of uniform international treatment guidelines with the ultimate aim to improve individualized therapy and outcomes of patients with gastric cancer.
materials and METHODS
This systematic review was performed according to the recommendations of the PRISMA statement [98].
Search strategy
A systematic search of the following databases was performed in December 2014: Medline, Science Citation Index, Embase, CCMed, Publisher Database, ASCO abstracts. Additionally, clinical trial registries such as WHO International Clinical Trials Registry and ClinicalTrials.gov were searched. Search strategies included the Medical Subject Headings (MeSH) “Stomach Neoplasm”; “Peritoneal Lavage”; “Therapeutic Irrigation”; “Cytology” as well as the text terms “gastric cancer”, “peritoneal”, “washing”, “lavage” and “cytology” in various combinations. In addition, we searched the reference lists of relevant articles and review articles. No time and language restrictions were applied to the initial search. The identified titles and abstracts were screened for eligibility by two independent reviewers (MP and MA). Full articles of potentially relevant studies were obtained for detailed evaluation.
Study inclusion and exlcusion criteria
Studies were included based on predefined selection criteria. Studies were eligible for inclusion, if they included patients with histologically proven gastric cancer and investigated the association of FITC with at least one of the following time-to-event outcomes: Overall survival (OS: date of surgery to date of death of any cause); disease specific survival (DSS: date of surgery to date of death due to gastric cancer); disease free survival (DFS: date of surgery to date of recurrence or death of any cause, whichever comes first), recurrence free survival (RFS: date of surgery to date of recurrence) or peritoneal recurrence (PR: date of surgery to date of peritoneal recurrence). Peritoneal cytology may have included any standard staining technique (i.e. hematoxylin and eosin [H&E], Papanicolaou) performed on peritoneal fluid or peritoneal washings. Molecular detection methods may have included immunocytochemistry and any form of reverse-transcriptase polymerase chain reaction ([RT]-PCR). In contrast to DNA or protein markers, studies using peritoneal tumor mRNA markers were included, assuming a linear correlation between peritoneal tumor cell detection and extremely short-lived free mRNA molecules.
Exclusion criteria were met, if less than 50 peritoneal samples were analyzed, if the percentage of patients with peritoneal or distant metastasis was > 30%, if they were not published in a peer-reviewed journal, if the above mentioned definitions of peritoneal cytology or molecular diagnostic were not met or if no hazard ratio could be calculated for at least one of the above mentioned time-to-event outcomes.
Data extraction
The following data was extracted from every article: first author, year of publication, study type, enrolment period, sample size, patient age and sex, FITC detection rate, definition of positive peritoneal fluid/lavage, timing of FITC detection, detection protocol, target genes and antigens, chemotherapy (neoadjuvant and/or adjuvant, treatment regimen), duration of follow up, reported outcomes and the use of multivariate models. The data for each included article were extracted independently by two authors (MP and MA). Diverging results were resolved by discussion.
Assessment of study quality
Study quality was evaluated using the modified risk of bias tool recommended by the Cochrane Collaboration as described before [99, 100].
Statistical analyses
The synchronized extraction results were pooled statistically as effect estimates in meta-analyses. Hazard ratios (HR) and their corresponding standard errors (SE) were extracted for the individual time-to-event outcome parameters of the included studies. In case the HR together with their associated SE or confidence intervals (CI) were not provided for a certain outcome, HRs were calculated using different statistical methods based on the clinical and statistical data reported in the primary studies [101, 102].
The extracted HR were pooled using the generic inverse variance method of the Review Manager Version 5.3 software (Copenhagen: The Nordic Cochrane Centre; The Cochrane Collaboration, 2014). To adjust for expected inter-study heterogeneity (study populations, treatments, detection assays, definitions of FITC positivity, duration of follow-up, etc.) a random effects analysis model was applied, which is more conservative when determining confidence intervals (CI) around the pooled HR [103]. I2 statistics was applied to assess the presence of statistical heterogeneity [104]. To explore reasons for statistical heterogeneity we performed sensitivity analyses, where the impact of single studies on the I2 value is tested as well as “a priori” subgroup analyses [105]. The results of subgroup analyses were compared by tests of interaction [105]. To avoid double patient evaluation among studies that evaluated multiple detection assays and/or target genes, these parameters were combined where possible to keep a maximum of information. Otherwise, cytokeratins were prioritized over alternative tumor cell markers and immunohistochemistry over RT-PCR assays. Sensitivity analyses (by choosing the alternative study arm) were performed to assess the statistical impact of such prioritization. Publication bias was assessed using funnel plot analyses.
conflicts of interest
No conflicts of interest exist.
References
1. Ajani JA, D’Amico T. NCCN Clinical practice guidelines in oncology: Gastric Cancer. National Comprehensive Cancer Network; 2015.
2. Waddell T, Verheij M, Allum W, Cunningham D, Cervantes A, Arnold D. Gastric cancer: ESMO–ESSO–ESTRO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24(suppl 6):vi57–vi63.
3. Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011;14(2):101–112.
4. Cunningham D, Allum WH, Stenning SP, Thompson JN, Van de Velde CJH, Nicolson M, Scarffe JH, Lofts FJ, Falk SJ, Iveson TJ, Smith DB, Langley RE, Verma M, et al. Perioperative Chemotherapy versus Surgery Alone for Resectable Gastroesophageal Cancer. N Engl J Med. 2006;355(1):11–20.
5. Roviello F, Marrelli D, de Manzoni G, Morgagni P, Di Leo A, Saragoni L, De Stefano A. Prospective study of peritoneal recurrence after curative surgery for gastric cancer. Br J Surg. 2003;90(9):1113–1119.
6. Brigand C, Arvieux C, Gilly FN, Glehen O. Treatment of Peritoneal Carcinomatosis in Gastric Cancers. Dig Dis. 2004;22(4):366–373.
7. De Andrade JP, Mezhir JJ. The critical role of peritoneal cytology in the staging of gastric cancer: An evidence-based review. J Surg Oncol. 2014;110(3):291–297.
8. Nieveen van Dijkum EJ, Sturm PD, de Wit LT, Offerhaus J, Obertop H, Gouma DJ. Cytology of peritoneal lavage performed during staging laparoscopy for gastrointestinal malignancies: is it useful? Ann Surg. 1998;228(6):728–733.
9. Gill RS, Al-Adra DP, Nagendran J, Campbell S, Shi X, Haase E, Schiller D. Treatment of gastric cancer with peritoneal carcinomatosis by cytoreductive surgery and HIPEC: A systematic review of survival, mortality, and morbidity. J Surg Oncol. 2011;104(6):692–698.
10. Bentrem D, Wilton A, Mazumdar M, Brennan M, Coit D. The Value of Peritoneal Cytology as a Preoperative Predictor in Patients With Gastric Carcinoma Undergoing a Curative Resection. Ann Surg Oncol. 2005;12(5):347–353.
11. Euanorasetr C, Lertsithichai P. Prognostic significance of peritoneal washing cytology in Thai patients with gastric adenocarcinoma undergoing curative D2 gastrectomy. Gastric Cancer. 2007;10(1):18–23.
12. Ito S, Nakanishi H, Kodera Y, Mochizuki Y, Tatematsu M, Yamamura Y. Prospective validation of quantitative CEA mRNA detection in peritoneal washes in gastric carcinoma patients. Br J Cancer. 2005;93(9):986–992.
13. Miyashiro I, Takachi K, Doki Y, Ishikawa O, Ohigashi H, Murata K, Sasaki Y, Imaoka S, Nakaizumi A, Takenaka A, Furukawa H, Hiratsuka M. When Is Curative Gastrectomy Justified for Gastric Cancer with Positive Peritoneal Lavage Cytology but Negative Macroscopic Peritoneal Implant? World J Surg. 2005;29(9):1131–1134.
14. AWMF, Deutschen Krebsgesellschaft und Deutschen Krebshilfe. Leitlinie: Magenkarzinom. Leitlinienprogramm Onkologie der AWMF, Deutschen Krebsgesellschaft und Deutschen Krebshilfe; 2012.
15. Badgwell B, Cormier JN, Krishnan S, Yao J, Staerkel GA, Lupo PJ, Pisters PWT, Feig B, Mansfield P. Does Neoadjuvant Treatment for Gastric Cancer Patients with Positive Peritoneal Cytology at Staging Laparoscopy Improve Survival? Ann Surg Oncol. 2008;15(10):2684–2691.
16. Bando E, Yonemura Y, Taniguchi K, Yasui T, Fushida S, Fujimura T, Nishimura G, Miwa K. Intraoperative lavage for cytological examination in 1,297 patients with gastric carcinoma. Am J Surg. 1999;178(3):256–262.
17. Benevolo M, Mottolese M, Cosimelli M, Tedesco M, Giannarelli D, Vasselli S, Carlini M, Garofalo A, Natali PG. Diagnostic and prognostic value of peritoneal immunocytology in gastric cancer. J Clin Oncol. 1998;16(10):3406–3411.
18. Boku T, Nakane Y, Minoura T, Takada H, Yamamura M, Hioki K, Yamamoto M. Prognostic significance of serosal invasion and free intraperitoneal cancer cells in gastric cancer. Br J Surg. 1990;77(4):436–439.
19. Bonenkamp JJ, Songun I, Hermans J, Van Velde CJHD. Prognostic value of positive cytology findings from abdominal washings in patients with gastric cancer. Br J Surg. 1996;83(5):672–674.
20. Chang-Qing F, Yi L, De-Guang W, Qing-Bin S, Xiang-Min H, Na T, Jian-Hua L. Immune clearance gastric carcinoma cells in ascites by activating caspase-9-induced apoptosis. APMIS. 2011;119(3):173–179.
21. Chuwa EWL, Khin L-W, Chan W-H, Ong H-S, Wong W-K. Prognostic significance of peritoneal lavage cytology in gastric cancer in Singapore. Gastric Cancer. 2005;8(4):228–237.
22. Fujimoto T, Zhang B, Minami S, Wang X, Takahashi Y, Mai M. Evaluation of Intraoperative Intraperitoneal Cytology for Advanced Gastric Carcinoma. Oncology. 2002;62(3):201–208.
23. Fujiwara Y, Okada K, Hanada H, Tamura S, Kimura Y, Fujita J, Imamura H, Kishi K, Yano M, Miki H, Okada K, Takayama O, Aoki T, et al. The clinical importance of a transcription reverse-transcription concerted (TRC) diagnosis using peritoneal lavage fluids in gastric cancer with clinical serosal invasion: A prospective, multicenter study. Surgery. 2014;155(3):417–423.
24. Fukagawa T, Katai H, Saka M, Morita S, Sasajima Y, Taniguchi H, Sano T, Sasako M. Significance of Lavage Cytology in Advanced Gastric Cancer Patients. World J Surg. 2010;34(3):563–568.
25. Fukuda N, Sugiyama Y, Wada J. Prognostic factors of T4 gastric cancer patients undergoing potentially curative resection. World J Gastroenterol WJG. 2011;17(9):1180–1184.
26. Hao Y-X, Zhong H, Yu P, Qian F, Zhao Y, Shi Y, Tang B. Influence of Laparoscopic Gastrectomy on the Detection Rate of Free Gastric Cancer Cells in the Peritoneal Cavity. Ann Surg Oncol. 2010;17(1):65–72.
27. Hara M, Nakanishi H, Jun Q, Kanemitsu Y, Ito S, Mochizuki Y, Yamamura Y, Kodera Y, Tatematsu M, Hirai T, Kato T. Comparative analysis of intraperitoneal minimal free cancer cells between colorectal and gastric cancer patients using quantitative RT-PCR: possible reason for rare peritoneal recurrence in colorectal cancer. Clin Exp Metastasis. 2007;24(3):179–189.
28. Hayes N, Wayman J, Wadehra V, Scott DJ, Raimes SA, Griffin SM. Peritoneal cytology in the surgical evaluation of gastric carcinoma. Br J Cancer. 1999;79(3-4):520–524.
29. Horikawa M. Clinical significance of intraperitoneal CD44 mRNA levels of magnetically separated CD45-negative EpCAM-positive cells for peritoneal recurrence and prognosis in stage II and III gastric cancer patients. Oncol Rep. 2011;25(5):1413–1420.
30. Ikeguchi M, Oka A, Tsujitani S, Maeta M, Kaibara N. Relationship between area of serosal invasion and intraperitoneal free cancer cells in patients with gastric cancer. Anticancer Res. 1994;14(5B):2131–2134.
31. Ishii T, Fujiwara Y, Ohnaka S, Hayashi T, Taniguchi H, Takiguchi S, Yasuda T, Yano M, Monden M. Rapid Genetic Diagnosis With the Transcription—Reverse Transcription Concerted Reaction System for Cancer Micrometastasis. Ann Surg Oncol. 2004;11(8):778–785.
32. Jeon C-H, Shin I-H, Park J-B, Chae H-D. Prognostic Significance of MAGE in Peritoneal Washes in Gastric Carcinoma Patients Without Peritoneal Metastasis: Results of a 5-year Follow-up Study. J Clin Gastroenterol. 2010;44(10):682–686.
33. Jiang C-G, Xu Y, Wang Z-N, Sun Z, Liu F-N, Yu M, Xu H-M. Clinicopathological analysis and prognostic significance of peritoneal cytology in Chinese patients with advanced gastric cancer. ANZ J Surg. 2011;81(9):608–613.
34. Kang K-K, Hur H, Byun CS, Kim YB, Han S-U, Cho YK. Conventional Cytology Is Not Beneficial for Predicting Peritoneal Recurrence after Curative Surgery for Gastric Cancer: Results of a Prospective Clinical Study. J Gastric Cancer. 2014;14(1):23–31.
35. Katsuragi K, Yashiro M, Sawada T, Osaka H, Ohira M, Hirakawa K. Prognostic impact of PCR-based identification of isolated tumour cells in the peritoneal lavage fluid of gastric cancer patients who underwent a curative R0 resection. Br J Cancer. 2007;97(4):550–556.
36. Kodera Y, Nakanishi H, Yamamura Y, Shimizu Y, Torii A, Hirai T, Yasui K, Morimoto T, Kato T, Kito T, Tatematsu M. Prognostic value and clinical implications of disseminated cancer cells in the peritoneal cavity detected by reverse transcriptase-polymerase chain reaction and cytology. Int J Cancer. 1998;79(4):429–433.
37. Kodera Y, Yamamura Y, Shimizu Y, Torii A, Hirai T, Yasui K, Morimoto T, Kato T. Peritoneal washing cytology: Prognostic value of positive findings in patients with gastric carcinoma undergoing a potentially curative resection. J Surg Oncol. 1999;72(2):60–64.
38. Kodera Y, Nakanishi H, Ito S, Mochizuki Y, Ohashi N, Yamamura Y, Fujiwara M, Koike M, Tatematsu M, Nakao A. Prognostic Significance of Intraperitoneal Cancer Cells in Gastric Carcinoma: Analysis of Real Time Reverse Transcriptase-Polymerase Chain Reaction after 5 Years of Followup. J Am Coll Surg. 2006;202(2):231–236.
39. La Torre M, Ferri M, Giovagnoli MR, Sforza N, Cosenza G, Giarnieri E, Ziparo V. Peritoneal wash cytology in gastric carcinoma. Prognostic significance and therapeutic consequences. Eur J Surg Oncol EJSO. 2010;36(10):982–986.
40. Lee SD, Ryu KW, Eom BW, Lee JH, Kook MC, Kim Y-W. Prognostic significance of peritoneal washing cytology in patients with gastric cancer. Br J Surg. 2012;99(3):397–403.
41. Li J-K, Zheng M, Miao C-W, Zhang J-H, Ding G-H, Wu W-S. Peritoneal lavage cytology and carcinoembryonic antigen determination in predicting peritoneal metastasis and prognosis of gastric cancer. World J Gastroenterol WJG. 2005;11(46):7374–7377.
42. Li Z, Zhang D, Zhang H, Miao Z, Tang Y, Sun G, Dai D. Prediction of peritoneal recurrence by the mRNA level of CEA and MMP-7 in peritoneal lavage of gastric cancer patients. Tumor Biol. 2014;35(4):3463–3470.
43. Makino T, Fujiwara Y, Takiguchi S, Miyata H, Yamasaki M, Nakajima K, Nishida T, Mori M, Doki Y. The utility of pre-operative peritoneal lavage examination in serosa-invading gastric cancer patients. Surgery. 2010;148(1):96–102.
44. Manzoni G de, Verlato G, Leo AD, Tomezzoli A, Pedrazzani C, Pasini F, Piubello Q, Cordiano C. Peritoneal Cytology Does Not Increase the Prognostic Information Provided by TNM in Gastric Cancer. World J Surg. 2006;30(4):579–584.
45. Miyagawa K, Sakakura C, Nakashima S, Yoshikawa T, Fukuda K, Kin S, Nakase Y, Shimomura K, Oue N, Yasui W, Hayasizaki H, Okazaki Y, Yamagishi H, et al. Overexpression of RegIV in Peritoneal Dissemination of Gastric Cancer and Its Potential as A Novel Marker for the Detection of Peritoneal Micrometastasis. Anticancer Res. 2008;28(2B):1169–1179.
46. Nakajima T, Harashima S, Hirata M, Kajitani T. Prognostic and therapeutic values of peritoneal cytology in gastric cancer. Acta Cytol. 1978;22(4):225–229.
47. Nekarda H, Gess C, Stark M, Mueller JD, Fink U, Schenck U, Siewert JR. Immunocytochemically detected free peritoneal tumour cells (FPTC) are a strong prognostic factor in gastric carcinoma. Br J Cancer. 1999;79(3-4):611–619.
48. Oyama K, Terashima M, Takagane A, Maesawa C. Prognostic significance of peritoneal minimal residual disease in gastric cancer detected by reverse transcription–polymerase chain reaction. Br J Surg. 2004;91(4):435–443.
49. Ozer I, Bostanci EB, Dalgic T, Karaman K, Ulas M, Ozogul YB, Ercan M, Deger C, Akoglu M. Presence of free cancer cells in the peritoneal cavity of patients who underwent curative gastrectomy with lymph node dissection. Hepatogastroenterology. 2012;59(117):1657–1660.
50. Ribeiro U, Safatle-Ribeiro AV, Zilberstein B, Mucerino D, Yagi OK, Bresciani CC, Jacob CE, Iryia K, Gama-Rodrigues J. Does the intraoperative peritoneal lavage cytology add prognostic information in patients with potentially curative gastric resection? J Gastrointest Surg. 2006;10(2):170–177.
51. Rosenberg R, Nekarda H, Bauer P, Schenck U, Hoefler H, Siewert JR. Free peritoneal tumour cells are an independent prognostic factor in curatively resected stage IB gastric carcinoma. Br J Surg. 2006;93(3):325–331.
52. Ryu CK, Park JI, Min JS, Jin SH, Park SH, Bang HY, Chae GB, Lee JI. The Clinical Significance and Detection of Intraperitoneal Micrometastases by ThinPrep(R) Cytology with Peritoneal Lavage Fluid in Patients with Advanced Gastric Cancer. J Korean Gastric Cancer Assoc. 2008;8(4):189–197.
53. Sugita Y, Fujiwara Y, Taniguchi H, Mori T, Ishii T, Niwa H, Okada Y, Takiguchi S, Yasuda T, Yano M, Monden M. Quantitative molecular diagnosis of peritoneal lavage fluid for prediction of peritoneal recurrence in gastric cancer. Int J Oncol. 2003;23(5):1419–1423.
54. Suzuki T, Ochiai T, Hayashi H, Hori S, Shimada H, Isono K. Peritoneal lavage cytology findings as prognostic factor for gastric cancer. Semin Surg Oncol. 1999;17(2):103–107.
55. Tamura N, Iinuma H, Takada T. Prospective study of the quantitative carcinoembryonic antigen and cytokeratin 20 mRNA detection in peritoneal washes to predict peritoneal recurrence in gastric carcinoma patients. Oncol Rep. 2007;17(3):667–672.
56. Tokuda K, Natsugoe S, Nakajo A, Miyazono F, Ishigami S, Hokita S, Takao S, Eizuru Y, Aikou T. Clinical significance of CEA-mRNA expression in peritoneal lavage fluid from patients with gastric cancer. Int J Mol Med. 2003;11(1):79–84.
57. Ueno H, Yoshida K, Hirai T, Kono F, Kambe M, Toge T. Quantitative detection of carcinoembryonic antigen messenger RNA in the peritoneal cavity of gastric cancer patients by real-time quantitative reverse transcription polymerase chain reaction. Anticancer Res. 2003;23(2C):1701–1708.
58. Vogel P, Rüschoff J, Kümmel S, Zirngibl H, Hofstädter F, Hohenberger W, Jauch KW. Immunocytology improves prognostic impact of peritoneal tumour cell detection compared to conventional cytology in gastric cancer. Eur J Surg Oncol J Eur Soc Surg Oncol Br Assoc Surg Oncol. 1999;25(5):515–519.
59. Wong J, Kelly KJ, Mittra A, Gonen M, Allen P, Fong Y, Coit D. RT-PCR Increases Detection of Submicroscopic Peritoneal Metastases in Gastric Cancer and Has Prognostic Significance. J Gastrointest Surg. 2012;16(5):889–896.
60. Wu CC, Chen JT, Chang MC, Ho WL, Chen CY, Yeh DC, Liu TJ, P’eng FK. Optimal surgical strategy for potentially curable serosa-involved gastric carcinoma with intraperitoneal free cancer cells. J Am Coll Surg. 1997;184(6):611–617.
61. Yamamoto M, Matsuyama A, Kameyama T, Okamoto M, Okazaki J, Utsunomiya T, Tsutsui S, Fujiwara M, Ishida T. Prognostic re-evaluation of peritoneal lavage cytology in Japanese patients with gastric carcinoma. Hepatogastroenterology. 2009;56(89):261–265.
62. Yamamoto M, Yoshinaga K, Matsuyama A, Tsutsui S, Ishida T. CEA/CA72-4 levels in peritoneal lavage fluid are predictive factors in patients with gastric carcinoma. J Cancer Res Clin Oncol. 2014;140(4):607–612.
63. Yamashita K, Sakuramoto S, Katada N, Futawatari N, Moriya H, Hirai K, Kikuchi S, Watanabe M. Diffuse type advanced gastric cancer showing dismal prognosis is characterized by deeper invasion and emerging peritoneal cancer cell: the latest comparative study to intestinal advanced gastric cancer. Hepatogastroenterology. 2009;56(89):276–281.
64. Yoneda A, Taniguchi K, Torashima Y, Susumu S, Kanetaka K, Kuroki T, Eguchi S. The detection of gastric cancer cells in intraoperative peritoneal lavage using the reverse transcription–loop-mediated isothermal amplification method. J Surg Res. 2014;187(1):e1–e6.
65. Yonemura Y, Endou Y, Fujimura T, Fushida S, Bandou E, Kinoshita K, Sugiyama K, Sawa T, Kim B-S, Sasaki T. Diagnostic value of preoperative RT-PCR-based screening method to detect carcinoembryonic antigen-expressing free cancer cells in the peritoneal cavity from patients with gastric cancer. ANZ J Surg. 2001;71(9):521–528.
66. Yoshikawa T, Tsuburaya A, Kobayashi O, Sairenji M, Motohashi H, Noguchi Y. Peritoneal cytology in patients with gastric cancer exposed to the serosa--a proposed new classification based on the local and distant cytology. Hepatogastroenterology. 2003;50(52):1183–1186.
67. Yu X, Ren Z, Xue Y, Song H, Wei Y, Li C. D2 lymphadenectomy can disseminate tumor cells into peritoneal cavity in patients with advanced gastric cancer. Neoplasma. 2012;60(02):174–181.
68. Iida T, Iwahashi M, Katsuda M, Ishida K, Nakamori M, Nakamura M, Naka T, Ojima T, Ueda K, Hayata K, Yasuoka H, Yamaue H. Prognostic significance of IL-17 mRNA expression in peritoneal lavage in gastric cancer patients who underwent curative resection. Oncol Rep. 2014;31(2):605–612.
69. Vogel DP, Rüschoff J, Kümmel S, Zirngibl H, Hofstädter F, Hohenberger W, Jauch K-W. Prognostic value of microscopic peritoneal dissemination. Dis Colon Rectum. 2000;43(1):92–100.
70. Han J, Lv P, Yu J-L, Wu Y-C, Zhu X, Hong L-L, Zhu W-Y, Yu Q-M, Wang X-B, Li P, Ling Z-Q. Circulating Methylated MINT2 Promoter DNA Is a Potential Poor Prognostic Factor in Gastric Cancer. Dig Dis Sci. 2014;59(6):1160–1168.
71. Jeon C-H, Kim I-H, Chae H-D. Prognostic Value of Genetic Detection Using CEA and MAGE in Peritoneal Washes With Gastric Carcinoma After Curative Resection: Result of a 3-Year Follow-Up. Medicine (Baltimore). 2014;93(11):e83.
72. Satoh Y, Mori K, Kitano K, Kitayama J, Yokota H, Sasaki H, Uozaki H, Fukayama M, Seto Y, Nagawa H, Yatomi Y, Takai D. Analysis for the Combination Expression of CK20, FABP1 and MUC2 is Sensitive for the Prediction of Peritoneal Recurrence in Gastric Cancer. Jpn J Clin Oncol. 2012;42(2):148–152.
73. Takebayashi K, Murata S, Yamamoto H, Ishida M, Yamaguchi T, Kojima M, Shimizu T, Shiomi H, Sonoda H, Naka S, Mekata E, Okabe H, Tani T. Surgery-Induced Peritoneal Cancer Cells in Patients Who Have Undergone Curative Gastrectomy for Gastric Cancer. Ann Surg Oncol. 2014;21(6):1991–1997.
74. McLean MH, El-Omar EM. Genetics of gastric cancer. Nat Rev Gastroenterol Hepatol. 2014;11(11):664–674.
75. Buffart TE, Louw M, van Grieken NCT, Tijssen M, Carvalho B, Ylstra B, Grabsch H, Mulder CJJ, van de Velde CJH, van der Merwe SW, Meijer GA. Gastric cancers of Western European and African patients show different patterns of genomic instability. BMC Med Genomics. 2011;4:7.
76. Persson C, Canedo P, Machado JC, El-Omar EM, Forman D. Polymorphisms in Inflammatory Response Genes and Their Association With Gastric Cancer: A HuGE Systematic Review and Meta-Analyses. Am J Epidemiol. 2010;173:259–270.
77. Lordick F, Lorenzen S, Yamada Y, Ilson D. Optimal chemotherapy for advanced gastric cancer: is there a global consensus? Gastric Cancer. 2013;17(2):213–225.
78. Price TJ, Shapiro JD, Segelov E, Karapetis CS, Pavlakis N, Van Cutsem E, Shah MA, Kang Y-K, Tebbutt NC. Management of advanced gastric cancer. Expert Rev Gastroenterol Hepatol. 2012;6(2):199–209.
79. Emoto S, Kitayama J, Ishigami H, Yamaguchi H, Watanabe T. Clinical Significance of Cytological Status of Peritoneal Lavage Fluid During Intraperitoneal Chemotherapy for Gastric Cancer with Overt Peritoneal Dissemination. Ann Surg Oncol. 2014;1–7.
80. Yonemura Y, Bandou E, Sawa T, Yoshimitsu Y, Endou Y, Sasaki T, Sugarbaker PH. Neoadjuvant treatment of gastric cancer with peritoneal dissemination. Eur J Surg Oncol EJSO. 2006;32(6):661–665.
81. Seybolt JF, Papanicolaou GN, Cooper WA. Cytology in the diagnosis of gastric cancer. Cancer. 1951;4(2):286–295.
82. Selby LV, Vertosick EA, Sjoberg DD, Schattner MA, Janjigian YY, Brennan MF, Coit DG, Strong VE. Morbidity after Total Gastrectomy: Analysis of 238 Patients. J Am Coll Surg. 2015;220(5):863–871.e2.
83. Smyth EC, Cunningham D. Targeted Therapy for Gastric Cancer. Curr Treat Options Oncol. 2012;13(3):377–389.
84. Verwaal VJ, van Ruth S, Bree E de, van Slooten GW, van Tinteren H, Boot H, Zoetmulder FAN. Randomized Trial of Cytoreduction and Hyperthermic Intraperitoneal Chemotherapy Versus Systemic Chemotherapy and Palliative Surgery in Patients With Peritoneal Carcinomatosis of Colorectal Cancer. J Clin Oncol. 2003;21(20):3737–3743.
85. Elias D, Lefevre JH, Chevalier J, Brouquet A, Marchal F, Classe J-M, Ferron G, Guilloit J-M, Meeus P, Goéré D, Bonastre J. Complete Cytoreductive Surgery Plus Intraperitoneal Chemohyperthermia With Oxaliplatin for Peritoneal Carcinomatosis of Colorectal Origin. J Clin Oncol. 2009;27(5):681–685.
86. Helm CW. The Role of Hyperthermic Intraperitoneal Chemotherapy (HIPEC) in Ovarian Cancer. The Oncologist. 2009;14(7):683–694.
87. Yan TD, Black D, Sugarbaker PH, Zhu J, Yonemura Y, Petrou G, Morris DL. A Systematic Review and Meta-analysis of the Randomized Controlled Trials on Adjuvant Intraperitoneal Chemotherapy for Resectable Gastric Cancer. Ann Surg Oncol. 2007;14(10):2702–2713.
88. Ceelen WP, Flessner MF. Intraperitoneal therapy for peritoneal tumors: biophysics and clinical evidence. Nat Rev Clin Oncol. 2010;7(2):108–115.
89. Yang X-J, Huang C-Q, Suo T, Mei L-J, Yang G-L, Cheng F-L, Zhou Y-F, Xiong B, Yonemura Y, Li Y. Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy Improves Survival of Patients with Peritoneal Carcinomatosis from Gastric Cancer: Final Results of a Phase III Randomized Clinical Trial. Ann Surg Oncol. 2011;18(6):1575–1581.
90. Glehen O, Gilly FN, Arvieux C, Cotte E, Boutitie F, Mansvelt B, Bereder JM, Lorimier G, Quenet F, Elias D, Chirurgie AF de. Peritoneal Carcinomatosis from Gastric Cancer: A Multi-Institutional Study of 159 Patients Treated by Cytoreductive Surgery Combined with Perioperative Intraperitoneal Chemotherapy. Ann Surg Oncol. 2010;17(9):2370–2377.
91. Königsrainer I, Horvath P, Struller F, Königsrainer A, Beckert S. Initial Clinical Experience with Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy in Signet-Ring Cell Gastric Cancer with Peritoneal Metastases. J Gastric Cancer. 2014;14(2):117–122.
92. Kuramoto M, Shimada S, Ikeshima S, Matsuo A, Yagi Y, Matsuda M, Yonemura Y, Baba H. Extensive Intraoperative Peritoneal Lavage as a Standard Prophylactic Strategy for Peritoneal Recurrence in Patients with Gastric Carcinoma. [Miscellaneous Article]. Ann Surg. 2009;250(2):242–246.
93. Ott K, Herrmann K, Lordick F, Wieder H, Weber WA, Becker K, Buck AK, Dobritz M, Fink U, Ulm K, Schuster T, Schwaiger M, Siewert J-R, et al. Early Metabolic Response Evaluation by Fluorine-18 Fluorodeoxyglucose Positron Emission Tomography Allows In vivo Testing of Chemosensitivity in Gastric Cancer: Long-term Results of a Prospective Study. Clin Cancer Res. 2008;14(7):2012–2018.
94. Ott K, Weber WA, Lordick F, Becker K, Busch R, Herrmann K, Wieder H, Fink U, Schwaiger M, Siewert J-R. Metabolic Imaging Predicts Response, Survival, and Recurrence in Adenocarcinomas of the Esophagogastric Junction. J Clin Oncol. 2006;24(29):4692–4698.
95. Lordick F, Ott K, Krause B-J, Weber WA, Becker K, Stein HJ, Lorenzen S, Schuster T, Wieder H, Herrmann K, Bredenkamp R, Höfler H, Fink U, et al. PET to assess early metabolic response and to guide treatment of adenocarcinoma of the oesophagogastric junction: the MUNICON phase II trial. Lancet Oncol. 2007;8(9):797–805.
96. Mezhir JJ, Shah MA, Jacks LM, Brennan MF, Coit DG, Strong VE. Positive Peritoneal Cytology in Patients with Gastric Cancer: Natural History and Outcome of 291 Patients. Ann Surg Oncol. 2010;17(12):3173–3180.
97. Aizawa M, Nashimoto A, Yabusaki H, Nakagawa S, Matsuki A, Homma K, Kawasaki T. The clinical significance of potentially curative resection for gastric cancer following the clearance of free cancer cells in the peritoneal cavity by induction chemotherapy. Surg Today. 2014;1–7.
98. Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. J Clin Epidemiol. 2009;62(10):1006–1012.
99. Higgins J, Green S. Cochrane Handbook for Systematic Reviews of Interventions [Internet]. Version 5.1.0 [updated March 2011]. The Cochrane Collaboration; 2011. Available from: www.cochrane-handbook.org
100 Rahbari NN, Bork U, Motschall E, Thorlund K, Büchler MW, Koch M, Weitz J. Molecular Detection of Tumor Cells in Regional Lymph Nodes Is Associated With Disease Recurrence and Poor Survival in Node-Negative Colorectal Cancer: A Systematic Review and Meta-Analysis. J Clin Oncol. 2012;30(1):60–70.
101. Parmar MKB, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med. 1998;17(24):2815–2834.
102. Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 2007;8(1):16.
103. Riley RD, Higgins JPT, Deeks JJ. Interpretation of random effects meta-analyses. BMJ. 2011;342:d549–d549.
104. Higgins JPT, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21(11):1539–1558.
105. Altman DG, Bland JM. Interaction revisited: the difference between two estimates. BMJ. 2003;326(7382):219.

