INTRODUCTION
Breast cancer remains the most common and death-causing cancer in females [1]. In US it is expected that there will be approximately 231,840 new cases and 40,290 deaths from breast cancer among females in 2015 [1]. It is critical to take appropriate actions for the chemoprevention and chemotherapy of this malignant cancer, at the aim of decreasing its public health burden. As a class of drugs for managing cardiovascular diseases, statins are widely used all over the world [2]. Preclinical studies have demonstrated that statins can potentially suppress tumor and reduce metastatic potential of breast cancer [3–6]. These seem to support the beneficial effect of statin use on survival of breast cancer patients. However, findings from epidemiological studies are not consistent. Two cohort studies suggest that both pre-diagnosis statin use and post-diagnosis statin use are associated with reduced risks of overall mortality and breast cancer-specific mortality [7, 8]. On the other hand, three other cohort studies do not suggest the beneficial effects of statin use on breast cancer patients’ survival [9–11]. A meta-analysis summarizing available evidence will be critical for clarifying the relationship between statin use and survival outcome of breast cancer patients.
Similarly, with regards to the risk of breast cancer development, laboratory studies have suggested statins to be potentially chemopreventive [12, 13], while this is supported by only a proportion of relevant clinical studies. A meta-analysis of seven randomized clinical trials (RCTs) and nine observational studies does not identify such a relationship [14]. Another meta-analysis study summarizing 24 observational studies also suggests a null association [2]. Although in this meta-analysis a relatively large number of subjects is involved, several more recent studies evaluating the relationship between statin use and breast cancer risk have been published [15–17]. An updated meta-analysis summarizing all available evidence including those from recently published studies is thus necessary to more accurately clarify the relationship.
We aim to conduct a comprehensive meta-analysis study to assess the associations between statin use and breast cancer survival and risk. Findings from such a study may help determine whether statins could be used for either chemotherapy or chemoprevention of breast cancer, at the aim of reducing health burden from this malignant disease.
RESULTS
Literature search and study characteristics
The detailed steps of our literature search and article screening were shown in Figure 1. A total of 32 studies evaluating breast cancer risk [15–46] and 5 studies evaluating breast cancer survival [7–11] met the inclusion criteria and were included in the current meta-analysis. Several studies evaluated endpoints beyond the scope of our hypothesis like disease recurrence and thus were not included in the current meta-analysis [47–50]. Several studies reported the association estimates separately according to different subgroups and the combined effect sizes were not able to be determined based on available information [8, 17, 28]. We thus treated these estimates separately and incorporated all of them in the pooled analyses. The detailed characteristics of the included studies were shown in Tables 1 and 2 respectively. For breast cancer mortality, 5 prospective studies were available. Three were conducted in Europe and 2 in America. These studies enrolled 60,911 patients and had a median follow up of 5.3 years (range 2.9–11.5 years). All these studies were categorized as high quality studies, according to the Newcastle-Ottawa Quality Assessment Scale (NOS) (Supplementary Table 1). For breast cancer risk, a total of 11 case-control studies, 12 prospective studies, and 9 RCTs were available. Overall, 20 studies were conducted in America, 9 in Europe, 2 in Asia, and 1 in Oceania. The studies enrolled 83,919 patients and had a median follow up of 5 years (range 2.2–10.8 years). Among the observational studies, 11 of the 12 prospective studies (91.7%) and 9 of the 11 case-control studies (81.8%) were categorized as high quality studies (Supplementary Tables 2 and 3). However, only 2 of the 9 RCTs (22.2%) were categorized as studies with low risk of bias (Supplementary Table 4).
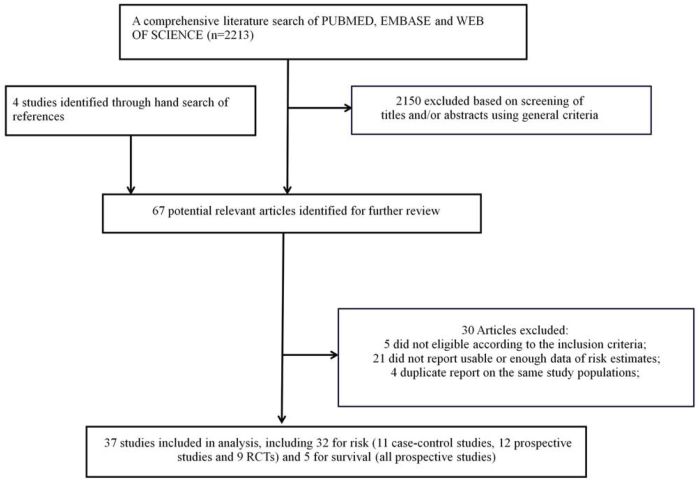
Figure 1: Flow chart for selection of eligible studies.
Table 1: Characteristics of studies evaluating statin use and breast cancer mortality
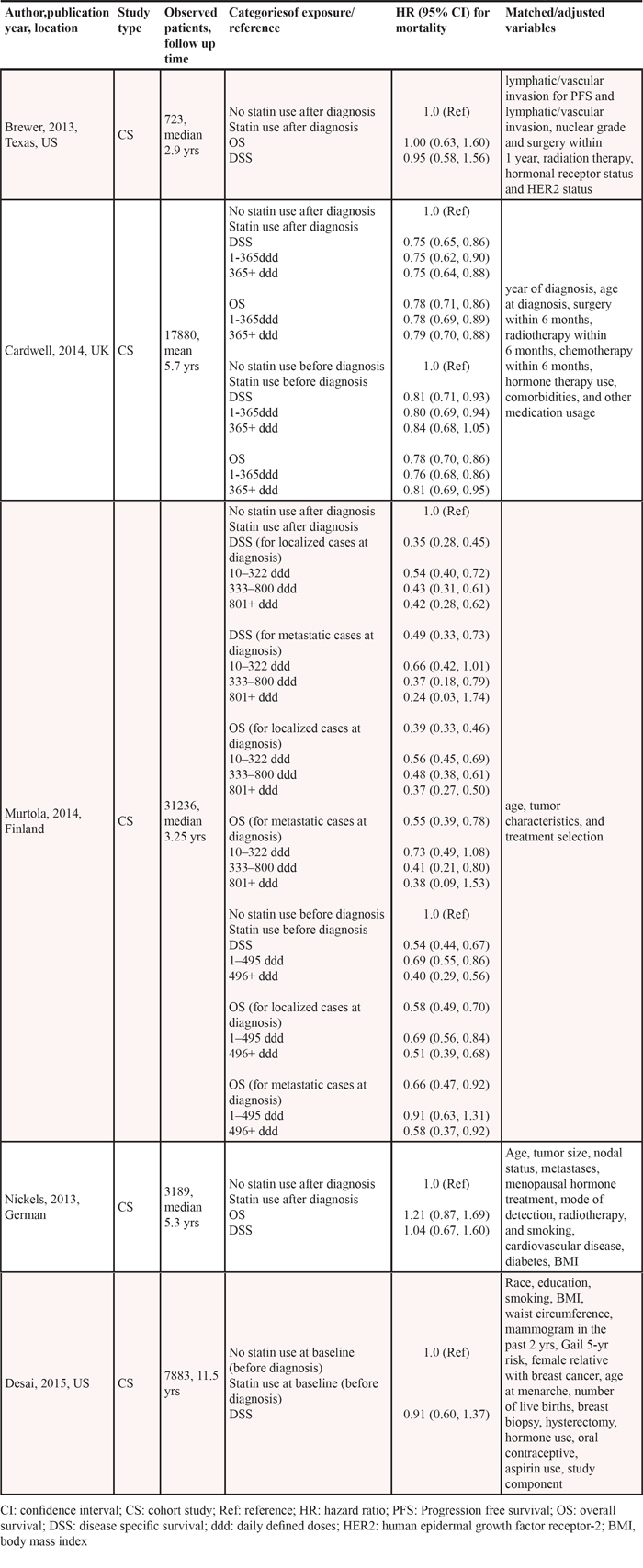
Table 2: Characteristics of studies evaluating statin use and breast cancer risk
Author, publication year, location |
Study type |
Cases/subject or control (age), duration of follow up |
Categoriesof exposure/reference |
RR (95% CI) |
Matched/adjusted variables |
|---|---|---|---|---|---|
McDougall, 2013, Seattle–Puget Sound, US |
PC-CS |
IDC: 916/902 (55–74 y) |
IDC: |
|
Age, county of residence, reference year, HRT |
Graaf, 2004, Netherlands |
PC-CS |
467/16976 (NA) |
No statin use |
1.0 (Ref) |
Sex, year of birth, geographic region, duration of follow-up, index date, diabetes mellitus, prior hospitalizations, chronic disease score, chronic use of diuretics, ACE inhibitors, calcium channel blockers, hormones, NSAIDs, and other lipid-lowering therapy |
Kaye, 2004, General Practice Research Database, UK |
PC-CS |
698/3267 (50–89 y) |
No statin use |
1.0 (Ref) |
Year of birth, sex, general practice, year of entry into the GPRD, and index date |
Boudreau, 2004, western Washington State, US |
PC-CS |
975/1007 (65–79 y) |
Nonuse |
1.0 (Ref) |
Age at reference date, reference year, county of residence, and use of antihypertensive medication |
Kochhar, 2005, VISN 16 database, US |
PC-CS |
556/39865 (25–92 y) |
Nonuse |
1.0 (Ref) |
Age, diabetes mellitus, smoking, alcohol consumption |
Dumasia, 2006, southeastern Michigan, US |
HC-CS |
521/521 (35–101 y) |
Nonuse |
1.0 (Ref) |
Age, race, BMI |
Coogan, 2007, Philadelphia, New York, Baltimore, US |
HC-CS |
1185/3900 (40–79 y) |
No statin use |
1.0 (Ref) |
Age, interview year, study center, BMI, alcohol use, race, years of education, pack-years of smoking, NSAID use, use of conjugated estrogens or other female hormones, use of oral contraceptives, menopausal status, parity, age at menarche, family history of breast cancer and religion |
Pocobelli, 2008, Wisconsin, Massachusetts, New Hampshire, US |
PC-CS |
3859/4761 (50y+) |
No statin use |
1.0 (Ref)1.0 (0.8–1.2) |
Age, state of residence, reference year, first degree family history of breast cancer, menopausal status/ageat menopause, parity/age at first birth, body mass index, recency of postmenopausal hormone use, education, and screening mammography history |
Eaton, 2009, Fargo, ND, US |
HC-CS |
95/94 (55–81 yrs) |
No statin use |
1.0 (Ref)1.3 (0.7–2.5) |
Age, age at menopause, family history of breast cancer, parity |
Woditschka, 2010, Northern California, US |
PC-CS |
HR negative: |
HR negative: |
|
Birth year and duration of KPNC pharmacy coverage, oral contraceptive and hormone therapy use |
Leung, 2015, NationalHealth Insurance Research Database, Taiwan |
PC-CS |
6463/18987 |
Nonuse |
1.0 (Ref) |
Age, comorbidities at cancer diagnosis, use of hormone replacement therapy |
Vinogradova, 2011, 574 UK general practices, UK |
NC-CS |
15666/62938 (30 y+) |
Nonuse |
1.0 (Ref) |
Townsend quintile, BMI, smoking status, myocardial infarction, coronary heart disease, diabetes, hypertension, stroke, rheumatoidarthritis, use of NSAIDs, Cox2-inhibitors, aspirin, family history of breast cancer, use of oral contraceptives, hormone-replace therapy |
Blais, 2000, Canada |
NC-CS |
65/650 |
use of bile acid-binding resins |
1.0 (Ref) |
Age, previous neoplasm, year of cohort entry, use of other lipid-reducing agents, use of fibric acids, comorbidity score |
Desai, 2013, Women’s Health Initiative, US |
CS |
7430/154587 (50–79 y), mean 10.8 yrs |
Nonuse |
1.0 (Ref) |
Age, BMI, ethnicity, smoking status, baseline hormone therapy use, baseline hormone therapy duration, family history of breast cancer, education, hysterectomy, mammogram last two years, age atFirst birth, parity, age at menarche, alcohol, percentage energy from fats, physical activity, and NSAID |
Smeeth, 2008, The Health Improvement Network database, UK |
CS |
3204/729529 (40+ yrs), median 4.2 yrs |
Nonuse |
1.0 (Ref) |
Age, propensity score, year of index date, first diagnosis of any of the following post-index date: diabetes, cerebrovascular disease, coronary heart disease, peripheral vascular disease, other atheroma, atrial fibrillation, heart failure, hyperlipidaemia, hypertension, other circulatory disease, cancer, dementia, first use of any of the following post-index date: aspirin, nitrates, fibrates, b-blockers, calcium channel blockers, potassium channel activators, diuretics, positive inotropes, anticoagulants, antihypertensives, or othercardiovascular drugs |
Beck, 2003, Canada |
CS |
879/67472 (mean 61.3 yrs), mean 4.2 yrs |
Nonuse |
1.0 (Ref) |
Age, sex |
Jacobs, 2011, Cancer Prevention Study II Nutrition Cohort, US |
CS |
3070/73196 (NA), ~8 yrs |
Nonuse |
1.0 (Ref) |
NA |
Haukka, 2009, Finland |
CS |
6046/NA (median 60 y), mean 8.8 yrs |
Nonuse |
1.0 (Ref) |
Age, follow-up period |
Cauley, 2003, Baltimore, Minneapolis, the Monongahela Valley, Portland, US |
CS |
244/7528 (mean 77 y), mean 6.8 yrs |
Nonuse |
1.0 (Ref) |
Age, body weight, HRT, family history of breast cancer |
Boudreau, 2007, western Washington State, US |
CS |
2707/92,788 (45–89 y), median 6.4 yrs |
Nonuse |
1.0 (Ref) |
Age, use of hormone therapy, use of other lipid-lowering therapy, diabetes mellitus, BMI |
Setoguchi, 2007, Pennsylvania, US |
CS |
300/31723 (65 y+), mean 2.9 yrs |
Nonuse |
1.0 (Ref) |
Time, age, race, health service utilization, prevention-related activities including mammography and gynecological examination, diabetes, Arthritis, Inflammatory bowel diseases, Benign mammary dysplasia, Estrogen use, Estrogen-progesterone use, NSAID use, Use of gastroprotective drugs, obesity, Tobacco abuse diagnosis |
Friis, 2005, Denmark |
CS |
3141/13508 (30–80 y), mean 3.3 yrs |
Nonuse |
1.0 (Ref) |
Age, calendar period and use ofNSAIDs, hormone replacement therapy and cardiovascular drugs |
Eliassen, 2005, NHS, US |
CS |
3177/75,828 (42–69 y), 6–12 yrs |
Nonuse |
1.0 (Ref) |
Time, age, age at menarche, parity and age at first birth, height, body mass index, first-degree family history of breastcancer, benign breast disease, alcohol consumption, physical activity, and menopausal status, age at menopause, and use of postmenopausal hormones |
Hsia, 2011, JUPITER, US |
RCT |
45/6205 (60+ y), 2.2 y |
Control |
1.0 (Ref) |
NA |
Nakamura, 2006, MEGA, Japan |
RCT |
25/5356 (mean 58 y), 4.6 y |
Control |
1.0 (Ref) |
NA |
HPS, 2005, UK |
RCT |
89/5082 (40–80 y), 5 y |
Control |
1.0 (Ref) |
NA |
Strandberg, 2004, SSSS, North Europe |
RCT |
12/827 (35–70 y), 10.4 y |
Control |
1.0 (Ref) |
NA |
Shepherd, 2002, PROSPER, North Europe |
RCT |
29/3000 (70–82 y), 3.2 y |
Control |
1.0 (Ref) |
NA |
Hague, 2003, LIPID, Oceania |
RCT |
17/1516 (31–75 y), 6.1 y |
Control |
1.0 (Ref) |
NA |
ALLHAT-LLT, 2002, North America |
RCT |
71/5051 (55+ y), 4.8 y |
Control |
1.0 (Ref) |
NA |
Sacks, 1996, CARE, North America |
RCT |
13/576 (mean 59 y), 4.8 y |
Control |
1.0 (Ref) |
NA |
Clearfield, 2001, AFCAPS, US |
RCT |
22/997 (55–73 y), 5.2 y |
Control |
1.0 (Ref) |
NA |
BMI: body mass index; CI: confidence interval; CS: cohort study; RCT: randomized clinical trial; HC-CS: hospital-based case-control study; NA: not available; NC-CS: nested case-control study; PC-CS: population-based case-control study; Ref: reference; RR: relative risk; HRT: Hormone replacement therapy; IDC: Invasive ductal carcinoma; ILC: Invasive lobular carcinoma; NSAID: Non-steroidal antiinflammatory drugs.
Post-diagnosis statin use and breast cancer survival
Four studies reported the association between post-diagnosis statin use and mortality of breast cancer patients [7–10]. These studies reported associations with overall survival (OS) and disease specific survival (DSS) respectively. For the study by Murtola et al [8], estimates according to subgroups of localized cases and metastatic cases were reported separately. We thus included both in the pooled analysis. Focusing on the association between post-diagnosis statin use and DSS of breast cancer patients, the pooled analysis of available studies revealed that there was a significantly negative association (HR = 0.65, 95% CI 0.43–0.98), with relatively considerable heterogeneity (I2 = 89.7%; Table 3). There was no significant publication bias as indicated by Egger’s test (p for bias: 0.947) and Begg’s test (p for bias: 1.000). The negative association was detected in studies focusing on general breast cancer patients, studies conducted in Europe and subgroup analysis according to follow up time (Table 3). There was a non-linear relationship between the duration of post-diagnosis statin use and DSS (p for likelihood ratio test: 0.0001) [7, 8]. The non-linear relationship was demonstrated in Figure 2.
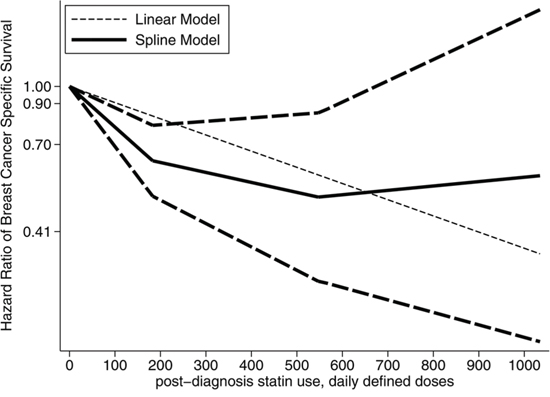
Figure 2: The dose-response relationship between post-diagnosis statin use and breast cancer specific survival.
Table 3: Summary risk estimates of the association between after-diagnosis statin use and breast cancer mortality
No of reports |
HR (95% CI) |
I2 (%) |
P for heterogeneity |
|
|---|---|---|---|---|
Overall mortality |
5 |
0.71 (0.48–1.07) |
94.0 |
<0.001 |
Subgroup analysis |
||||
Breast cancer subtype |
||||
inflammatory breast cancer |
1 |
1 (0.63–1.6) |
– |
– |
more general breast cancer |
4 |
0.67 (0.42–1.05) |
95.3 |
<0.001 |
Location |
||||
Europe |
4 |
0.67 (0.42–1.05) |
95.3 |
<0.001 |
America |
1 |
1.00 (0.63–1.60) |
– |
– |
Follow up time (years) |
||||
<4.3 (median value) |
3 |
0.58 (0.35–0.95) |
87.0 |
<0.001 |
>4.3 |
2 |
0.94 (0.62–1.44) |
83.9 |
0.013 |
Disease specific mortality |
5 |
0.65 (0.43–0.98) |
89.7 |
<0.001 |
Subgroup analysis |
||||
Breast cancer subtype |
||||
inflammatory breast cancer |
1 |
0.95 (0.58–1.56) |
– |
– |
more general breast cancer |
4 |
0.60 (0.38–0.95) |
91.7 |
<0.001 |
Location |
||||
Europe |
4 |
0.60 (0.38–0.95) |
91.7 |
<0.001 |
America |
1 |
0.95 (0.58–1.56) |
– |
– |
Follow up time (years) |
||||
<4.3 (median value) |
3 |
0.53 (0.31–0.91) |
84.8 |
0.001 |
>4.3 |
2 |
0.77 (0.68–0.88) |
49.1 |
0.161 |
Regarding the association between post-diagnosis statin use and OS of breast cancer patients, there was not a statistically significant association (HR = 0.71, 95% CI 0.48–1.07; I2 = 94.0%; Table 3). There was no significant publication bias as indicated by Egger’s test (p for bias: 0.992) and Begg’s test (p for bias: 0.806). The subgroup analyses based on disease subtype, study location and follow up time also largely revealed null associations, although in the strata of shorter than 4.3 years of follow up time, the association reached statistical significance (HR = 0.58, 95% CI 0.35–0.95; Table 3).
Pre-diagnosis statin use and breast cancer survival
Three studies reported the association between pre-diagnosis statin use and mortality of breast cancer patients [7, 8, 11], including both OS (2 studies) and DSS (3 studies). The study by Murtola et al [8] reported estimations for OS according to subgroups of localized cases and metastatic cases respectively. We thus included both in the pooled analysis of OS. Focusing on the association between pre-diagnosis statin use and OS of breast cancer patients, there was a negative association (HR = 0.68, 95% CI 0.54–0.84), with high heterogeneity (I2 = 75.7%; Table 4). There was no indication of publication bias according to Egger’s test (p for bias: 0.541) and Begg’s test (p for bias: 1.000). This inverse association persisted in subgroup analysis of follow up time.
Table 4: Summary risk estimates of the association between before-diagnosis statin use and breast cancer mortality
No of reports |
HR (95% CI) |
I2 (%) |
P for heterogeneity |
|
|---|---|---|---|---|
Overall mortality |
3 |
0.68 (0.54–0.84) |
75.7 |
0.016 |
Subgroup analysis |
||||
Follow up time (years) |
||||
<4.5 |
2 |
0.60 (0.51–0.70) |
0 |
0.505 |
>4.5 |
1 |
0.78 (0.70–0.86) |
– |
– |
Disease specific mortality |
3 |
0. 72 (0.53–0.99) |
82.3 |
0.004 |
Subgroup analysis |
||||
Follow up time (years) |
||||
<4.5 |
1 |
0.54 (0.44–0.67) |
– |
– |
>4.5 |
2 |
0.82 (0.72–0.93) |
0 |
0.599 |
Similarly, we identified an inverse association between pre-diagnosis statin use and DSS of breast cancer patients, with a HR of 0.72 (95% CI 0.53–0.99). There was significant heterogeneity across studies (I2 = 82.3%; Table 4). The inverse association was detected in subgroups stratified by follow up time.
Statin use and breast cancer risk
Among the 32 studies reporting the association between statin use and risk of breast cancer, two studies provided estimations according to different subgroups respectively [17, 28]. We included all these estimations in the overall pooled analysis because the overall estimation in these studies could not be inferred based on the available information. After summarizing results from all these studies, there was no significant association between statin use and breast cancer risk (RR = 1.02, 95% CI 0.95–1.09), with considerable heterogeneity (I2 = 80.8%; Table 5). No evident publication bias was detected based on the conducted Egger’s test (p for bias: 0.730) and Begg’s test (p for bias: 0.906). The null association persisted in almost all strata of subgroup analyses according to study design and location (Table 5).
Table 5: Summary risk estimates of the association between statin use and breast cancer risk
No of reports |
RR (95% CI) |
I2 (%) |
P for heterogeneity |
|
|---|---|---|---|---|
Overall estimation |
34 |
1.02 (0.95–1.09) |
80.8 |
<0.001 |
Subgroup analysis |
||||
Location |
||||
Europe |
9 |
1.01 (0.97–1.05) |
0 |
0.600 |
America |
22 |
0.99 (0.92–1.05) |
63.8 |
<0.001 |
Asia |
2 |
1.18 (0.50–2.77) |
78.7 |
0.030 |
Oceania |
1 |
1.13 (0.44–2.92) |
– |
– |
Study design |
||||
Prospective studies |
12 |
1.02 (0.98–1.05) |
0.0 |
0.462 |
Case-control studies |
13 |
1.02 (0.88–1.18) |
92.0 |
<0.001 |
RCTs |
9 |
1.04 (0.78–1.39) |
30.3 |
0.176 |
DISCUSSION
We performed a comprehensive systematic review and meta-analysis to assess the relationship between statin use and mortality and risk of breast cancer. After summarizing all available evidence, it seemed that statin use was inversely associated with mortality of breast cancer patients, although the association between post-diagnosis statin use and OS did not reach statistical significance. An updated meta-analysis evaluating the relationship between statin use and breast cancer risk revealed a null association, which is consistent with previous analyses [2, 14, 51]. These findings demonstrated that the use of statin after diagnosis of breast cancer could potentially decrease mortality risk. Additionally, although statin use seemed not affect risk of breast cancer development, it might confer beneficial effect for decreasing the mortality of those individuals who develop breast cancer.
The detected beneficial effect of statin use on breast cancer mortality is plausible based on preclinical findings. Statins are shown to be able to inhibit growth in both breast cancer cell lines and in-vitro models [4, 52]. Research demonstrates that the anti-cancer effects may be induced by statins’ effects on apoptosis, angiogenesis and tumor invasion [53–56]. For example, statins can regulate the mevalonate pathway, which is critical for the tumor promoting effects of p53 [57]. The dysregulation of this pathway is found to promote breast cancer tumor cell growth [58]. Besides, mevalonate stimulates tumor proliferation based on a mouse model [59]. Statins are also detected to potentially inhibit carcinogenesis through inhibiting isoprenoids [60]. Overall, these mechanistic understandings make the finding that statin use is inversely associated with breast cancer mortality to be more plausible. On the other hand, it is very interesting that statin use can decrease breast cancer mortality, and breast cancer recurrence [61], while has no effect on preventing incidence of breast cancer. Further research is needed to clarify why there are differentiated effects of statins on breast cancer development and prognosis.
The relationship between statin use and cancer has been extensively evaluated. Meta-analysis studies have suggested that use of statins is associated with reduced risks of liver cancer [62–64], ovarian cancer [65], colorectal cancer [66, 67], haematological malignancies [68], esophageal cancer [69, 70], gastric cancer [71, 72], and prostate cancer [73]. Furthermore, statins were demonstrated to be protective from breast cancer recurrence [61]. Our finding of the potential beneficial effect of statin use on breast cancer mortality is consistent with another newly published meta-analysis, in which statin use was suggested to be beneficial for overall cancer survival and cancer-specific survival, including breast cancer patients [74]. If the finding of the beneficial effect of statin use could be further replicated, it may be warranted to promote statin use to decrease public health burden from human cancers.
Our study has several strengths. To the best of our knowledge, up to date this is the most comprehensive meta-analysis study evaluating the relationship between statin use and breast cancer, including assessing the associations between post-diagnosis and pre-diagnosis stains use and breast cancer mortality, as well as the dose-response relationship. Compared with previous meta-analyses evaluating the relationship between statin use and breast cancer risk [2, 14], our study incorporates evidence from more recent studies and is more comprehensive. Compared with another meta-analysis assessing the effect of statin use on cancer mortality [74], our analysis includes evidence from more relevant studies for breast cancer, as well as evaluates the dose-response relationship for the association. Our study adds new knowledge for the relationship between statin use and cancers, and provides further evidence for supporting the use of statins for decreasing health burden from cancers, if the findings of the current study could be replicated by additional studies.
Several potential limitations must be acknowledged for the appropriate interpretation of our findings. First, we did not have access to the individualized primary data from the included studies, and the risk estimates used in the pooled analyses might not be fully adjusted. Although almost all included studies provided adjusted estimates considering important confounders, residual confounding may be an issue for biasing the results. Second, our main finding of the potential beneficial effect of statin use on breast cancer survival may be biased somehow due to that evidence was from observational studies, which are known to confer incoherent shortcomings. For the included studies evaluating statin use with mortality, there are relatively large differences in the definitions of statin use [8, 10], which may bias the results. Further large scale well designed studies are warranted to replicate our findings. Third, we detected considerable heterogeneity in the pooled analyses, which were not eliminated in the subgroup analyses. Additionally, the findings of the association between statin use and breast cancer mortality were based on limited studies, although these are the currently available evidence for evaluating this relationship. In spite that we detected a non-linear dose-response relationship for the association between statin use and breast cancer mortality, data were merely based on two available studies. Further research will be critical to more accurately clarify and resolve these issues.
In conclusion, based on a summarization of all available evidence, statin use was inversely associated with breast cancer mortality, although statin use was not associated with breast cancer risk. If replicated in further large-scale well designed studies, our findings may have implication for supporting the use of statins for decreasing health burden from breast cancer.
MATERIALS AND METHODS
Data sources and search strategies
A comprehensive search of PubMed (MEDLINE), Web of Science, and Embase databases was conducted from each database’s inception to January 2015 for studies published in English Controlled vocabulary and keywords were used to search for eligible studies. The detailed search strategy is described in the Supplementary Material. We also screened references of relevant review articles to identify other potential studies. The literature search was updated at August 2015.
Study selection
Studies were eligible if they (i) were case–control studies, prospective studies, or RCTs; (ii) evaluated the association between statin use and breast cancer risk or survival; (iii) presented relative risk (RR), odds ratio (OR), hazard ratio (HR) estimates with 95% confidence intervals (CI) or necessary data for determination. There was no restriction for sample size and follow-up duration. If there were several publications from the same study, we included the study with the most cases and relevant information, like our previous studies [75–78].
Data extraction and quality assessment
A pair of investigators independently carried out the abstract screening, full text screening, and data extraction (Chao Tu and Lang Wu). Disagreements were resolved by discussion, with input from other investigators. Data extracted from each study included: the first author’s last name, year of publication, study location, study design, characteristics of study population (sample size, age, length of follow-up), and effect sizes of the associations. If multiple estimates of the association for the same outcome were reported, we used the estimate that was adjusted for the most appropriate covariates, like previous studies [79–81]. In situations when only unadjusted estimates were given, we used the crude estimate.
The quality assessment of included studies were performed according to the Newcastle-Ottawa Quality Assessment Scale (NOS) for observational studies [82, 83], and a revised form of Cochrane Collaboration’s tool for assessing risk of bias in randomized trials [84, 85]. For NOS population and sampling methods, exposure and outcome, as well as statistical matching/adjustments were considered. Studies with assigned scores of at least 7 were categorized as high quality studies. For RCTs the adequacy of randomization, allocation concealment, as well as blinding were evaluated. Studies were categorized as with low risk of bias only if all of these items were adequately described as with low risk.
Statistical methods
Due to the relative rarity of breast cancer in the general population, ORs and HRs were deemed equivalent to RRs. We used RRs to represent measures of studies evaluating associations with breast cancer risk. For studies evaluating breast cancer survival, HRs were used since all involved studies used HRs as the estimation. We used the I2 to evaluate the heterogeneity across studies, in which a I2 > 50% suggests high heterogeneity [86]. We pooled the log transformed RR or HR using the fixed-effects model [87] if there was no considerable heterogeneity. If there was substantial heterogeneity, we used the random-effects model [88]. With regards to the relationship between statin use and breast cancer survival, since for two included studies [7, 8], the associations of both post-diagnosis statin use and pre-diagnosis statin use were provided separately, we conducted analyses for pre-diagnosis statin use and post-diagnosis statin use respectively. Subgroup analyses were also conducted based on disease subgroup, study design (case-control vs. prospective studies vs. RCTs) and study geographic location (Europe, America or Asia) for studies evaluating disease risk, and disease subgroup, study geographic location and follow up time for studies evaluating disease mortality.
For the dose-response analysis, we explored potential non-linear relationship between the duration of statin use and breast cancer mortality, using principles as previously published [89]. For studies reporting statin use by categories, we used the midpoint of each category to represent the exposure. If the highest category did not provide the upper bound, we assumed the open ended interval’s width to be as same as the adjacent interval’s width. We examined a potential nonlinear dose-response relationship between statin use and breast cancer survival with fractional polynomial models, using restricted cubic splines with 3 knots at fixed percentiles (10%, 50% and 90%) of the distribution [90, 91]. We also conducted likelihood ratio tests to determine whether nonlinear or linear relationship was suggested.
Publication bias was evaluated via Egger’s test [92] and Begg’s test [93]. A P-value of 0.05 was used to determine whether there was significant publication bias. All statistical analyses were performed with Stata (version 13; StataCorp, College Station, TX).
GRANT SUPPORT
This study was supported by the Younger research fund of Shengjing Hospital (Grant 2014sj09 for Qi-Jun Wu). This publication was made possible by CTSA Grant Number UL1 TR000135 from the National Center for Advancing Translational Sciences (NCATS), a component of the National Institutes of Health (NIH). Its contents are solely the responsibility of the authors and do not necessarily represent the official view of NIH. The funding source had no involvement in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
Abbreviations
Odds ratio (OR), relative risk (RR), hazard ratio (HR), confidence intervals (CI), randomized clinical trials (RCT), overall survival (OS), disease specific survival (DSS)
REFERENCES
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA: a cancer journal for clinicians. 2015; 65:5–29.
2. Undela K, Srikanth V, Bansal D. Statin use and risk of breast cancer: a meta-analysis of observational studies. Breast Cancer Res Treat. 2012; 135:261–269.
3. Mandal CC, Ghosh-Choudhury N, Yoneda T, Choudhury GG, Ghosh-Choudhury N. Simvastatin prevents skeletal metastasis of breast cancer by an antagonistic interplay between p53 and CD44. J Biol Chem. 2011; 286:11314–11327.
4. Ghosh-Choudhury N, Mandal CC, Ghosh-Choudhury N, Ghosh Choudhury G. Simvastatin induces derepression of PTEN expression via NFkappaB to inhibit breast cancer cell growth. Cell Signal. 2010; 22:749–758.
5. Shibata MA, Ito Y, Morimoto J, Otsuki Y. Lovastatin inhibits tumor growth and lung metastasis in mouse mammary carcinoma model: a p53-independent mitochondrial-mediated apoptotic mechanism. Carcinogenesis. 2004; 25:1887–1898.
6. Rao S, Lowe M, Herliczek TW, Keyomarsi K. Lovastatin mediated G1 arrest in normal and tumor breast cells is through inhibition of CDK2 activity and redistribution of p21 and p27, independent of p53. Oncogene. 1998; 17:2393–2402.
7. Cardwell CR, Hicks BM, Hughes C, Murray LJ. Statin use after diagnosis of breast cancer and survival: a population-based cohort study. Epidemiology. 2015; 26:68–78.
8. Murtola TJ, Visvanathan K, Artama M, Vainio H, Pukkala E. Statin use and breast cancer survival: a nationwide cohort study from Finland. PloS one. 2014; 9:e110231.
9. Nickels S, Vrieling A, Seibold P, Heinz J, Obi N, Flesch-Janys D, Chang-Claude J. Mortality and recurrence risk in relation to the use of lipid-lowering drugs in a prospective breast cancer patient cohort. PloS one. 2013; 8:e75088.
10. Brewer TM, Masuda H, Liu DD, Shen Y, Liu P, Iwamoto T, Kai K, Barnett CM, Woodward WA, Reuben JM, Yang P, Hortobagyi GN, Ueno NT. Statin use in primary inflammatory breast cancer: a cohort study. Br J Cancer. 2013; 109:318–324.
11. Desai P, Lehman A, Chlebowski RT, Kwan ML, Arun M, Manson JE, Lavasani S, Wasswertheil-Smoller S, Sarto GE, LeBoff M, Cauley J, Cote M, Beebe-Dimmer J, Jay A, Simon MS. Statins and breast cancer stage and mortality in the Women’s Health Initiative. Cancer Causes Control. 2015; 26:529–539.
12. Seeger H, Wallwiener D, Mueck AO. Statins can inhibit proliferation of human breast cancer cells in vitro. Exp Clin Endocrinol Diabetes. 2003; 111:47–48.
13. Denoyelle C, Vasse M, Korner M, Mishal Z, Ganne F, Vannier JP, Soria J, Soria C. Cerivastatin, an inhibitor of HMG-CoA reductase, inhibits the signaling pathways involved in the invasiveness and metastatic properties of highly invasive breast cancer cell lines: an in vitro study. Carcinogenesis. 2001; 22:1139–1148.
14. Bonovas S, Filioussi K, Tsavaris N, Sitaras NM. Use of statins and breast cancer: a meta-analysis of seven randomized clinical trials and nine observational studies. J Clin Oncol. 2005; 23:8606–8612.
15. Desai P, Chlebowski R, Cauley JA, Manson JE, Wu C, Martin LW, Jay A, Bock C, Cote M, Petrucelli N, Rosenberg CA, Peters U, Agalliu I, Budrys N, Abdul-Hussein M, Lane D, et al. Prospective analysis of association between statin use and breast cancer risk in the women’s health initiative. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2013; 22:1868–1876.
16. Leung HW, Hung LL, Chan AL, Mou CH. Long-Term Use of Antihypertensive Agents and Risk of Breast Cancer: A Population-Based Case-Control Study. Cardiol Ther. 2015; 4:65–76.
17. McDougall JA, Malone KE, Daling JR, Cushing-Haugen KL, Porter PL, Li CI. Long-term statin use and risk of ductal and lobular breast cancer among women 55 to 74 years of age. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2013; 22:1529–1537.
18. Smeeth L, Douglas I, Hall AJ, Hubbard R, Evans S. Effect of statins on a wide range of health outcomes: a cohort study validated by comparison with randomized trials. Br J Clin Pharmacol. 2009; 67:99–109.
19. Setoguchi S, Glynn RJ, Avorn J, Mogun H, Schneeweiss S. Statins and the risk of lung, breast, and colorectal cancer in the elderly. Circulation. 2007; 115:27–33.
20. Jacobs EJ, Newton CC, Thun MJ, Gapstur SM. Long-term use of cholesterol-lowering drugs and cancer incidence in a large United States cohort. Cancer Res. 2011; 71:1763–1771.
21. Haukka J, Sankila R, Klaukka T, Lonnqvist J, Niskanen L, Tanskanen A, Wahlbeck K, Tiihonen J. Incidence of cancer and statin usage—record linkage study. Int J Cancer. 2010; 126:279–284.
22. Friis S, Poulsen AH, Johnsen SP, McLaughlin JK, Fryzek JP, Dalton SO, Sorensen HT, Olsen JH. Cancer risk among statin users: a population-based cohort study. Int J Cancer. 2005; 114:643–647.
23. Eliassen AH, Colditz GA, Rosner B, Willett WC, Hankinson SE. Serum lipids, lipid-lowering drugs, and the risk of breast cancer. Arch Intern Med. 2005; 165:2264–2271.
24. Cauley JA, Zmuda JM, Lui LY, Hillier TA, Ness RB, Stone KL, Cummings SR, Bauer DC. Lipid-lowering drug use and breast cancer in older women: a prospective study. J Womens Health (Larchmt). 2003; 12:749–756.
25. Boudreau DM, Yu O, Miglioretti DL, Buist DS, Heckbert SR, Daling JR. Statin use and breast cancer risk in a large population-based setting. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2007; 16:416–421.
26. Blais L, Desgagne A, LeLorier J. 3-Hydroxy-3-methylglutaryl coenzyme A reductase inhibitors and the risk of cancer: a nested case-control study. Arch Intern Med. 2000; 160:2363–2368.
27. Beck P, Wysowski DK, Downey W, Butler-Jones D. Statin use and the risk of breast cancer. J Clin Epidemiol. 2003; 56:280–285.
28. Woditschka S, Habel LA, Udaltsova N, Friedman GD, Sieh W. Lipophilic statin use and risk of breast cancer subtypes. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2010; 19:2479–2487.
29. Vinogradova Y, Coupland C, Hippisley-Cox J. Exposure to statins and risk of common cancers: a series of nested case-control studies. BMC Cancer. 2011; 11:409.
30. Pocobelli G, Newcomb PA, Trentham-Dietz A, Titus-Ernstoff L, Hampton JM, Egan KM. Statin use and risk of breast cancer. Cancer. 2008; 112:27–33.
31. Kaye JA, Jick H. Statin use and cancer risk in the General Practice Research Database. Br J Cancer. 2004; 90:635–637.
32. Graaf MR, Beiderbeck AB, Egberts AC, Richel DJ, Guchelaar HJ. The risk of cancer in users of statins. J Clin Oncol. 2004; 22:2388–2394.
33. Eaton M, Eklof J, Beal JR, Sahmoun AE. Statins and breast cancer in postmenopausal women without hormone therapy. Anticancer Res. 2009; 29:5143–5148.
34. Coogan PF, Rosenberg L, Strom BL, A D. Statin use and the risk of 10 cancers. Epidemiology. 2007; 18:213–219.
35. Boudreau DM, Gardner JS, Malone KE, Heckbert SR, Blough DK, Daling JR. The association between 3-hydroxy-3-methylglutaryl conenzyme A inhibitor use and breast carcinoma risk among postmenopausal women: a case-control study. Cancer. 2004; 100:2308–2316.
36. Kochhar R KV, Bejjanki H, Caldito G, Fort C. Statins to reduce breast cancer risk: A case control study in U.S. female veterans. ASCO Annual Meeting: Journal of Clinical Oncology. 2005; pp. 514.
37. Dumasia L, Lobocki C, Couturier A, Lebeis C. Use of statins and breast carcinoma: A case-control study. ASCO Annual Meeting: Journal of Clinical Oncology. 2006; pp. 592.
38. Sacks FM, Pfeffer MA, Moye LA, Rouleau JL, Rutherford JD, Cole TG, Brown L, Warnica JW, Arnold JM, Wun CC, Davis BR, Braunwald E. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators. N Engl J Med. 1996; 335:1001–1009.
39. Clearfield M, Downs JR, Weis S, Whitney EJ, Kruyer W, Shapiro DR, Stein EA, Langendorfer A, Beere PA, Gotto AM. Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS): efficacy and tolerability of long-term treatment with lovastatin in women. J Womens Health Gend Based Med. 2001; 10:971–981.
40. Officers A, Coordinators for the ACRGTA and Lipid-Lowering Treatment to Prevent Heart Attack T. Major outcomes in moderately hypercholesterolemic, hypertensive patients randomized to pravastatin vs usual care: The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT-LLT). JAMA. 2002; 288:2998–3007.
41. Shepherd J, Blauw GJ, Murphy MB, Bollen EL, Buckley BM, Cobbe SM, Ford I, Gaw A, Hyland M, Jukema JW, Kamper AM, Macfarlane PW, Meinders AE, Norrie J, Packard CJ, Perry IJ, et al. Pravastatin in elderly individuals at risk of vascular disease (PROSPER): a randomised controlled trial. Lancet. 2002; 360:1623–1630.
42. Hague W, Forder P, Simes J, Hunt D, Tonkin A, Investigators L. Effect of pravastatin on cardiovascular events and mortality in 1516 women with coronary heart disease: results from the Long-Term Intervention with Pravastatin in Ischemic Disease (LIPID) study. Am Heart J. 2003; 145:643–651.
43. Strandberg TE, Pyorala K, Cook TJ, Wilhelmsen L, Faergeman O, Thorgeirsson G, Pedersen TR, Kjekshus J, Group S. Mortality and incidence of cancer during 10-year follow-up of the Scandinavian Simvastatin Survival Study (4S). Lancet. 2004; 364:771–777.
44. Heart Protection Study Collaborative G. The effects of cholesterol lowering with simvastatin on cause-specific mortality and on cancer incidence in 20,536 high-risk people: a randomised placebo-controlled trial [ISRCTN48489393]. BMC Med. 2005; 3:6.
45. Nakamura H, Arakawa K, Itakura H, Kitabatake A, Goto Y, Toyota T, Nakaya N, Nishimoto S, Muranaka M, Yamamoto A, Mizuno K, Ohashi Y, Group MS. Primary prevention of cardiovascular disease with pravastatin in Japan (MEGA Study): a prospective randomised controlled trial. Lancet. 2006; 368:1155–1163.
46. Hsia J, MacFadyen JG, Monyak J, Ridker PM. Cardiovascular event reduction and adverse events among subjects attaining low-density lipoprotein cholesterol <50 mg/dl with rosuvastatin. The JUPITER trial (Justification for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin). J Am Coll Cardiol. 2011; 57:1666–1675.
47. Ahern TP, Pedersen L, Tarp M, Cronin-Fenton DP, Garne JP, Silliman RA, Sorensen HT, Lash TL. Statin prescriptions and breast cancer recurrence risk: a Danish nationwide prospective cohort study. J Natl Cancer Inst. 2011; 103:1461–1468.
48. Kwan ML, Habel LA, Flick ED, Quesenberry CP, Caan B. Post-diagnosis statin use and breast cancer recurrence in a prospective cohort study of early stage breast cancer survivors. Breast Cancer Res Treat. 2008; 109:573–579.
49. Chae YK, Valsecchi ME, Kim J, Bianchi AL, Khemasuwan D, Desai A, Tester W. Reduced risk of breast cancer recurrence in patients using ACE inhibitors, ARBs, and/or statins. Cancer Invest. 2011; 29:585–593.
50. Boudreau DM, Yu O, Chubak J, Wirtz HS, Bowles EJ, Fujii M, Buist DS. Comparative safety of cardiovascular medication use and breast cancer outcomes among women with early stage breast cancer. Breast Cancer Res Treat. 2014; 144:405–416.
51. Bonovas S, Lytras T, Sitaras NM. Statin use and breast cancer: do we need more evidence and what should this be? Expert Opin Drug Saf. 2014; 13:271–275.
52. Campbell MJ, Esserman LJ, Zhou Y, Shoemaker M, Lobo M, Borman E, Baehner F, Kumar AS, Adduci K, Marx C, Petricoin EF, Liotta LA, Winters M, Benz S, Benz CC. Breast cancer growth prevention by statins. Cancer Res. 2006; 66:8707–8714.
53. Vaklavas C, Chatzizisis YS, Tsimberidou AM. Common cardiovascular medications in cancer therapeutics. Pharmacol Ther. 2011; 130:177–190.
54. Jakobisiak M, Golab J. Potential antitumor effects of statins (Review). Int J Oncol. 2003; 23:1055–1069.
55. Katz MS. Therapy insight: Potential of statins for cancer chemoprevention and therapy. Nat Clin Pract Oncol. 2005; 2:82–89.
56. Gopalan A, Yu W, Sanders BG, Kline K. Simvastatin inhibition of mevalonate pathway induces apoptosis in human breast cancer cells via activation of JNK/CHOP/DR5 signaling pathway. Cancer Lett. 2013; 329:9–16.
57. Freed-Pastor WA, Mizuno H, Zhao X, Langerod A, Moon SH, Rodriguez-Barrueco R, Barsotti A, Chicas A, Li W, Polotskaia A, Bissell MJ, Osborne TF, Tian B, Lowe SW, Silva JM, Borresen-Dale AL, et al. Mutant p53 disrupts mammary tissue architecture via the mevalonate pathway. Cell. 2012; 148:244–258.
58. Clendening JW, Pandyra A, Boutros PC, El Ghamrasni S, Khosravi F, Trentin GA, Martirosyan A, Hakem A, Hakem R, Jurisica I, Penn LZ. Dysregulation of the mevalonate pathway promotes transformation. Proc Natl Acad Sci U S A. 2010; 107:15051–15056.
59. Duncan RE, El-Sohemy A, Archer MC. Mevalonate promotes the growth of tumors derived from human cancer cells in vivo and stimulates proliferation in vitro with enhanced cyclin-dependent kinase-2 activity. J Biol Chem. 2004; 279:33079–33084.
60. Konstantinopoulos PA, Karamouzis MV, Papavassiliou AG. Post-translational modifications and regulation of the RAS superfamily of GTPases as anticancer targets. Nat Rev Drug Discov. 2007; 6:541–555.
61. Ahern TP, Lash TL, Damkier P, Christiansen PM, Cronin-Fenton DP. Statins and breast cancer prognosis: evidence and opportunities. Lancet Oncol. 2014; 15:e461–468.
62. Shi M, Zheng H, Nie B, Gong W, Cui X. Statin use and risk of liver cancer: an update meta-analysis. BMJ Open. 2014; 4:e005399.
63. Singh S, Singh PP, Singh AG, Murad MH, Sanchez W. Statins are associated with a reduced risk of hepatocellular cancer: a systematic review and meta-analysis. Gastroenterology. 2013; 144:323–332.
64. Pradelli D, Soranna D, Scotti L, Zambon A, Catapano A, Mancia G, La Vecchia C, Corrao G. Statins and primary liver cancer: a meta-analysis of observational studies. Eur J Cancer Prev. 2013; 22:229–234.
65. Liu Y, Qin A, Li T, Qin X, Li S. Effect of statin on risk of gynecologic cancers: a meta-analysis of observational studies and randomized controlled trials. Gynecol Oncol. 2014; 133:647–655.
66. Lytras T, Nikolopoulos G, Bonovas S. Statins and the risk of colorectal cancer: an updated systematic review and meta-analysis of 40 studies. World J Gastroenterol. 2014; 20:1858–1870.
67. Liu Y, Tang W, Wang J, Xie L, Li T, He Y, Deng Y, Peng Q, Li S, Qin X. Association between statin use and colorectal cancer risk: a meta-analysis of 42 studies. Cancer Causes Control. 2014; 25:237–249.
68. Yi X, Jia W, Jin Y, Zhen S. Statin use is associated with reduced risk of haematological malignancies: evidence from a meta-analysis. PloS one. 2014; 9:e87019.
69. Beales IL, Hensley A, Loke Y. Reduced esophageal cancer incidence in statin users, particularly with cyclo-oxygenase inhibition. World J Gastrointest Pharmacol Ther. 2013; 4:69–79.
70. Singh S, Singh AG, Singh PP, Murad MH, Iyer PG. Statins are associated with reduced risk of esophageal cancer, particularly in patients with Barrett’s esophagus: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2013; 11:620–629.
71. Wu XD, Zeng K, Xue FQ, Chen JH, Chen YQ. Statins are associated with reduced risk of gastric cancer: a meta-analysis. Eur J Clin Pharmacol. 2013; 69:1855–1860.
72. Singh PP, Singh S. Statins are associated with reduced risk of gastric cancer: a systematic review and meta-analysis. Ann Oncol. 2013; 24:1721–1730.
73. Bansal D, Undela K, D’Cruz S, Schifano F. Statin use and risk of prostate cancer: a meta-analysis of observational studies. PloS one. 2012; 7:e46691.
74. Zhong S, Zhang X, Chen L, Ma T, Tang J, Zhao J. Statin use and mortality in cancer patients: Systematic review and meta-analysis of observational studies. Cancer Treat Rev. 2015; 41:554–567.
75. Wang YZ, Wu QJ, Zhu J, Wu L. Fish consumption and risk of myeloma: a meta-analysis of epidemiological studies. Cancer Causes Control. 2015; 26:1307–1314.
76. Wu L, Zhu J. Linear reduction in thyroid cancer risk by oral contraceptive use: a dose-response meta-analysis of prospective cohort studies. Hum Reprod. 2015; 30:2234–2240.
77. Wu L, Zhu J, Prokop LJ, Hassan Murad M. Pharmacologic Therapy of Diabetes and Overall Cancer Risk and Mortality: A Meta-Analysis of 265 Studies. Sci Rep. 2015; 5:10147.
78. Wu QJ, Wu L, Zheng LQ, Xu X, Ji C, Gong TT. Consumption of fruit and vegetables reduces risk of pancreatic cancer: evidence from epidemiological studies. Eur J Cancer Prev. 2015.
79. Guan HB, Wu L, Wu QJ, Zhu J, Gong T. Parity and pancreatic cancer risk: a dose-response meta-analysis of epidemiologic studies. PloS one. 2014; 9:e92738.
80. Luan NN, Wu L, Gong TT, Wang YL, Lin B, Wu QJ. Nonlinear reduction in risk for colorectal cancer by oral contraceptive use: a meta-analysis of epidemiological studies. Cancer Causes Control. 2015; 26:65–78.
81. Wu L, Wang Z, Zhu J, Murad AL, Prokop LJ, Murad MH. Nut consumption and risk of cancer and type 2 diabetes: a systematic review and meta-analysis. Nutr Rev. 2015; 73:409–425.
82. GA Wells BS, D O’Connell, J Peterson, V Welch, M Losos, P Tugwell. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses.
83. Wu L, Jiang Z, Li C, Shu M. Prediction of heart rate variability on cardiac sudden death in heart failure patients: a systematic review. Int J Cardiol. 2014; 174:857–860.
84. Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA, Cochrane Bias Methods G, Cochrane Statistical Methods G. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. Bmj. 2011; 343:d5928.
85. Zeng X, Zhang Y, Kwong JS, Zhang C, Li S, Sun F, Niu Y, Du L. The methodological quality assessment tools for preclinical and clinical studies, systematic review and meta-analysis, and clinical practice guideline: a systematic review. J Evid Based Med. 2015; 8:2–10.
86. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. Bmj. 2003; 327:557–560.
87. Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002; 21:1539–1558.
88. DerSimonian R, Laird N. Meta-analysis in clinical trials. Controlled clinical trials. 1986; 7:177–188.
89. Greenland S, Longnecker MP. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol. 1992; 135:1301–1309.
90. Liu Q, Cook NR, Bergstrom A, Hsieh CC. A two-stage hierarchical regression model for meta-analysis of epidemiologic nonlinear dose-response data. Computational Statistics and Data Analysis. 2009; 53:4157–4167.
91. Orsini N, Li R, Wolk A, Khudyakov P, Spiegelman D. Meta-analysis for linear and nonlinear dose-response relations: examples, an evaluation of approximations, and software. Am J Epidemiol. 2012; 175:66–73.
92. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. Bmj. 1997; 315:629–634.
93. Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994; 50:1088–1101.

