INTRODUCTION
The average life expectancy has increased over the past century due to better living conditions and medical advancements [1]. Women in particular are living longer, yet the age of menopause has remained constant at approximately 50 years of age. This results in most women living nearly a third of their lives without high levels of circulating estrogens. Risk factors for many diseases, such as cognitive and mood disorders, increase in women following menopause [2, 3, 4] and these risk factors have been correlated with the sharp decline in circulating levels of estrogens. Hormone therapy (HT) is often prescribed for women to mitigate many of the adverse symptoms associated with menopause, based primarily on evidence from animal models demonstrating that estrogens are anxiolytic and increase cognitive function [5, 6, 7, 8, 9, 10]. Data from studies in postmenopausal women, however, are less convincing that HT is beneficial. For instance, results from the large-scale clinical trial (Women's Health Initiative (WHI)) showed that there was a temporal discrepancy in the beneficial efficacy of HT [3, 4, 11, 12, 13, 14, 15, 16, 17]. The beneficial effects were largely dependent on the length of time following menopause onset that women received HT, with younger women realizing the greatest benefits. Follow up studies have since revealed that timing of HT is critical, both for maximum benefits, as well as minimal detrimental effects [11, 12, 13, 14, 15, 16]. However, the molecular mechanisms underlying these age-dependent effects of estrogens are unknown.
Estrogens regulate gene transcription primarily through binding to estrogen receptors (ERs), which then act as transcription factors on gene promoters. However, estrogens have also been shown to regulate gene expression at the post-transcriptional level through microRNAs (miRNAs). miRNAs are a class of small non-coding RNAs that regulate gene expression via translational repression of target mRNAs [18, 19], thereby decreasing the protein products of those genes. The canonical miRNA biogenesis pathway reveals several potential regulatory points for estrogens. First, miRNAs are transcribed from the genome raising the possibility that estrogen receptors mediate miRNA transcription. Second, miRNAs are regulated post-transcriptionally by a series of RNase III enzymes, which sequentially cleave the primary transcript (pri-miRNA) into shorter fragments (i.e. pre-miRNA), and these enzymes act in conjunction with several cofactor proteins, some of which are regulated by estrogens [20, 21, 22, 23, 24, 25, 26, 27]. Finally, the mature 22nt miRNA duplex dissociates such that one strand (guide strand) is loaded onto an argonaute (AGO) protein, thereby forming the RNA-induced silencing complex (miRISC). The guide strand is largely protected from degradation due to its association with target mRNA and AGO, yet the passenger strand is typically degraded. Several studies have shown that miRNAs are subject to post-transcriptional modifications, which can affect their stability. For instance, poly(A) polymerase associated domain 4 (PAPD4) is a nucleotidyltransferase that adenylates the 3′ end of miRNAs [28, 29]; this modification to the miRNA can increase its stability and alter its ability to bind to the RISC complex [28, 29]. Ultimately, miRNAs are degraded by at least one known exonuclease, 5′-3′ exoribonuclease 2 (XRN2) [30]. To our knowledge, there have been no studies to date demonstrating whether estrogens regulate any proteins that are involved in miRNA stabilization pathways.
In the brain miRNAs display both an age-and brain region-specific expression pattern, suggesting that they play critical roles in normal brain function [31, 32, 33, 34, 35, 36, 37]. Indeed previous studies have demonstrated that miRNAs are crucial for neuronal development and synaptic plasticity, and are also implicated in regulating affective behaviors [38, 39, 40, 41, 42, 43, 44, 45]. Previously, our laboratory demonstrated that the major circulating estrogen, 17β-estradiol (E2), regulates miRNAs in an age- and brain region-dependent manner in female rats, suggesting that there would be differential mRNA translation in those brain regions at varying ages due to E2 dependent miRNA regulation [46]. E2 exerts its effects through two estrogen receptors, ERα and ERβ. ERα and ERβ display brain region specific expression patterns, and also have dichotomous effects on brain function [47, 48]. Additionally, both receptors have been shown to regulate miRNA expression in-vitro, however it is not known whether one or both receptors are required for mediating E2-induced miRNA expression in the brain [49, 50].
We propose that there is a biological switch in estrogens’ actions that occurs coincident with age and length of time after ovarian hormone depletion, and we hypothesize that age dependent regulation of miRNAs could be the molecular basis for that switch. Our previous work showed that E2 differentially regulated miRNA expression in the brain of young (3 mo.) compared to aged (18 mo.) female rats. In this current study we extended those observations by investigating if longer periods of E2 deprivation in aged female rats altered the E2-dependent regulation of miRNAs we previously observed in the brain. In this paradigm female Fisher 344 rats (18 mo. old) were ovariectomized (OVX) and then given an acute E2 treatment at 1, 4, 8, or 12 weeks post-OVX. Additionally, we had a group of ovarian intact female rats that were sacrificed at the same age as the treated animals. We analyzed the expression of our previously identified E2 regulated mature miRNAs, as well as their intermediary biosynthetic products (pri-miRNA, pre-miRNA) in the hypothalamus. In addition, we quantified the expression of several miRNA processing proteins including drosha, DiGeorge syndrome critical region 8 (DGCR8), exportin-5 (XPO5), dicer, AGO2, XRN2, and PAPD4. Finally, we determined which ER mediates the E2-induced increase in miRNA expression observed at 1 wk. post-OVX in the aged female brain. Collectively our results show that extended deprivation of ovarian hormones markedly alters E2 regulation of mature miRNAs in the aged female hypothalamus, suggesting that there is a shift in how the brain responds to the re-introduction of E2 after prolonged periods of ovarian hormone deprivation.
RESULTS
Experiment 1: Expression of E2-responsive mature miRNAs in the hypothalamus of ovarian intact animals changes with age
Our previous studies showed that E2 regulated a subset of mature miRNAs (let-7i, miR-7a, miR-9, miR-9–3p, miR-125, miR-181a, and miR-495) in an age- and brain-region dependent manner [46]. Here, we observed that the expression of mature miRNAs in ovarian intact animals at varying advanced ages was different from both the OVX-vehicle and OVX-E2 treated animals, suggesting that ovarian factors other than E2 also regulate mature miRNA expression in aged female rats. Notably, the circulating E2 levels were equivalent between ovarian intact and OVX+veh treated animals (Figure 1). A post-hoc analysis revealed a statistically different expression profile at the one-week post-OVX time point (i.e. 18 mo. old) for miR-9, miR-125a, miR-181a, and miR-495 in ovarian intact animals compared to OVX+veh treated animals. Further, there was a statistically significant difference in the expression of mature miR-7a, miR-9a-3p, and miR-181a between ovarian intact and OVX+veh treated animals at later time points (i.e. 19, 20, and 21 months). Specifically, let-7i: levels were unchanged across all time points in animals that were ovarian intact (Figure 2a), yet there was a steady increase in let-7i over time in OVX+veh treated animals (Figure 2a). miR-7a: By contrast, miR-7a expression was similar between ovarian intact and OVX+veh treated animals at the one week time point (= 18 mo. old). The levels continued to increase with age in ovarian intact animals, but this result was not observed in either OVX group (Figure 2b). miR-9: Similar to let-7i, we observed no changes in miR-9 expression across all time points in ovarian intact animals (Figure 2c), but there was a significant increase over time in OVX+veh animals. miR-9-3p: Interestingly, miR-9-3p was similar in all groups at one week post OVX (i.e. 18 mo., Figure 3d), yet miR-9-3p expression significantly increased at 19 mo. in the ovarian intact groups and this increase was not apparent in the OVX groups until 20 mo. (8 weeks post-OVX) and 21 mo. (12 weeks post OVX). miR-125a: Ovarian intact animals had significantly higher levels of miR-125a expression at 18 mo. and 19 mo. time points, however all groups were similar after 20 and 21 mo. (Figure 2e). miR-181a: Ovarian intact animals had significantly higher levels of miR-181a expression at 18 mo. old compared to both OVX+veh and OVX+E2 groups (Figure 2f). These expression levels decreased with age, whereas an opposite trend was observed in the OVX animals and miR-181a expression was significantly higher in both OVX groups compared to ovarian intact animals by 21 mo. of age (Figure 2g). miR-495: Ovarian intact animals had consistently higher levels of mature miR-495 expression at all ages compared to OVX animals, although both OVX+veh and OVX+E2 groups showed significant increases after 8 and 12 weeks post-OVX (20 and 21 mo.). Taken together, these data underscore the importance of ovarian factors other than E2 in regulating mature miRNA expression in the aged female brain.
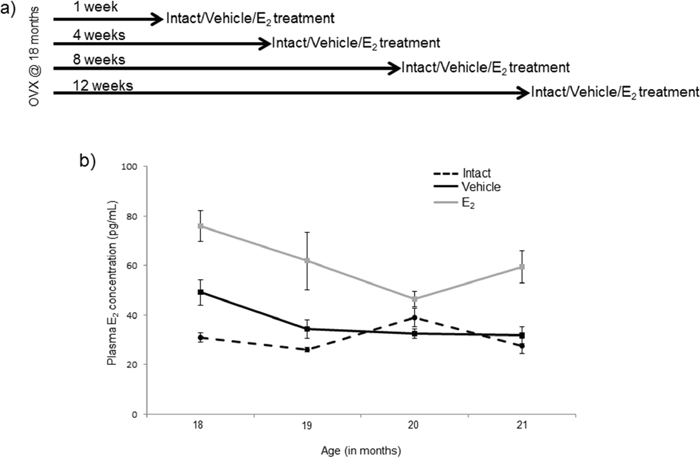
Figure 1: Diagram of ovarian hormone deprivation paradigm. a. Schematic representation of the animal experimental paradigm. Fischer 344 female rats were obtained at 18 months of age and ovariectomized. Rats were given a subcutaneous injection once/day for 3 days 1, 4, 8, or 12 weeks post-OVX (N = 6/age/treatment). b. E2 plasma concentrations assayed by ELISA from blood samples taken 24 hours after the last injection of E2. Data displayed as mean ± SEM pg/mL.
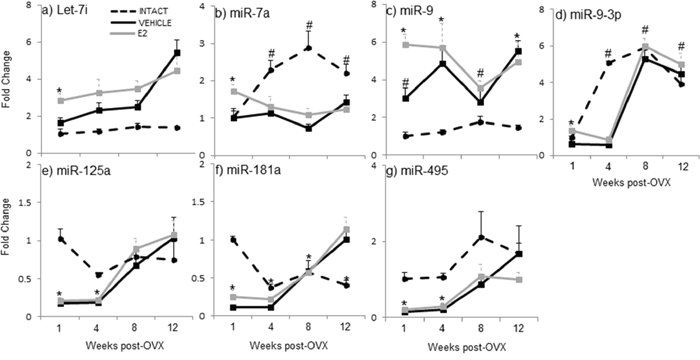
Figure 2: E2 regulation of mature miRNA expression in the hypothalamus after increasing lengths of ovarian hormone deprivation. a–g. Mature miRNA expression was analyzed by real time qRT-PCR and displayed as mean ± SEM fold change as compared to 18 month old ovarian intact animals (N = 6/age/treatment). An * denotes a statistically significant effect of treatment within a time point.
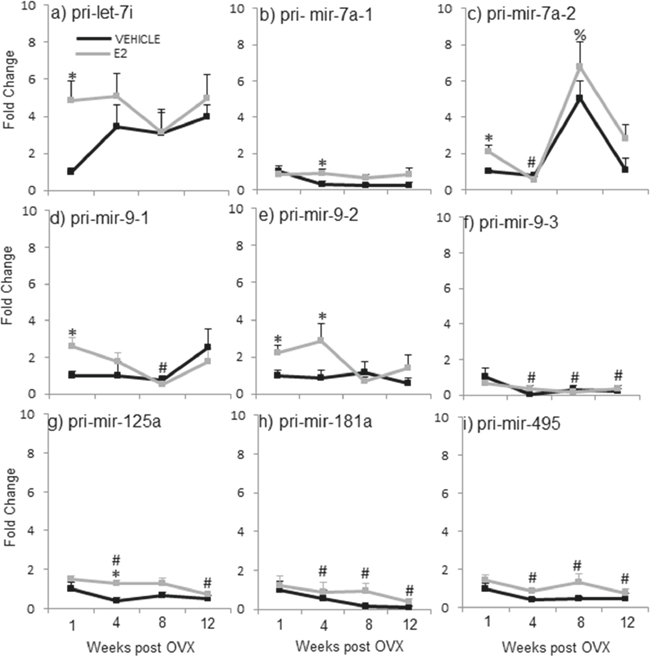
Figure 3: E2 regulation of the primary miRNA expression in the hypothalamus after increasing lengths of ovarian hormone deprivation. a–i. Primary miRNA expression was analyzed by real time qPCR and displayed as mean ± SEM fold change as compared to 1 week vehicle treated animals (N = 6/age/treatment). An * denotes a statistically significant effect of treatment within a time point. Different symbols (#, %) denote a statistically significant difference across time points within a treatment group.
Experiment 2: Treatment with E2 failed to regulate mature miRNA expression following prolonged periods of ovarian hormone deprivation
To further investigate the effects of E2-mediated regulation of these mature miRNAs, we developed an animal paradigm to directly test the timing hypothesis in aged female rats. Aged rats (18 mo., equivalent to 55 years old in human) were ovariectomized (OVX) to model surgically induced menopause and eliminate the source of all ovarian hormones. To specifically isolate the effects of E2 from all other ovarian hormones, OVX animals were treated with acute administration of E2 or vehicle control following varying lengths of time post-OVX: 1, 4, 8, or 12 weeks (Figure 1a). Our results demonstrated that there was a statistically significant interaction between treatment and length of ovarian hormone deprivation (Table 1). Most striking was the fact that E2 treatment significantly regulated the expression of these miRNAs at only one time point (i.e. one week post-OVX) and had no effect following prolonged periods of ovarian hormone deprivation. Specifically, E2 treatment significantly regulated the expression of mature miR-7a, miR-9, miR-9-3p, and miR-181a at 1 week post-OVX, which was consistent with our previously published data, [46], but not at any other time point (Figure 2a–2d, 2f), demonstrating a clear timing effect. Interestingly, E2 treatment also increased the expression of miR-9-3p compared to intact animals, but not OVX+veh treated animals (Figure 2d).
Table 1: 2 way-ANOVA analysis of mature miRNA expression in the hypothalamus
miRNA |
INTERACTION |
|---|---|
Let-7i |
NO |
miR-7a |
YES |
miR-9 |
YES |
miR-9-3p |
NO |
miR-125a |
YES |
miR-181a |
YES |
miR-495 |
YES |
Experiment 2: Mature miRNA expression levels do not correspond to the expression of their primary (pri-) and precursor (pre-) forms
Effects of E2 treatment on primary miRNA (pri-miR) expression levels in the hypothalamus following varying lengths of ovarian hormone deprivation
The data from these initial experiments were consistent with our previously published findings and demonstrated that E2 regulates mature miRNA expression following short-term, but not long-term, OVX. In order to determine the level of the miRNA biosynthetic pathway that E2 acts, we next analyzed the expression of the miRNA primary (pri-miR) and precursor (pre-miR) transcripts. A two-way ANOVA analysis revealed that there was no significant interaction between treatment and age for the pri- or pre- forms of these miRNAs, contrary to the results we observed for the mature miRNAs. Moreover, analysis of treatment within a single time point showed that E2, in general, had no effect on the transcription of most of the pri-miRNAs at any time point (Figure 3). However, there were a few exceptions. Specifically, comparison of treatment within a time point revealed that E2 significantly increased expression of the primary miRNA transcript of let-7i one-week post OVX, but not at any other time point (Figure 3a, gray line, *). Also, it is important to note that two of the mature miRNAs, miR-7a and miR-9, are transcribed from multiple chromosomes, allowing for unique regulation of biogenesis at each locus. Therefore, we designed primers corresponding to distinct primary sequences on each chromosome for miR-7a and miR-9, in order to account for possible differences between mature miRNA products derived from the different chromosomes. Interestingly, these data revealed a switch in E2 regulation of each chromosome depending on the age post-OVX. Specifically, E2 increased pri-miR-7a transcribed from chromosome 1 at 4 weeks post-OVX, but not any other time points (Figure 3b, pri-miR-7a-1, gray line, *). Meanwhile, E2 significantly increased pri-miR-7a transcribed from chromosome 17 at 1-week post-OVX (Figure 3c, pri-miR-7a-2, gray line, *). These data suggest that the mature miR-7a product was derived from different chromosomes depending on the time point. Interestingly, however, our data only showed a significant increase in mature miR-7a at the 1-week time point, which points to chromosome 17 as potentially more important than chromosome 1 for E2-regulated mature miR-7a expression. It is also important to note that miR-9 (guide strand) and miR-9-3p (passenger strand) are derived from the same primary and precursor transcripts, which are located on two different loci on chromosome 1 (pri-miR-9-1, pri-miR-9-2) and one locus on chromosome 2 (pri-miR-9-3). Our previously published data, and also replicated here in Experiment 2, showed that E2 significantly regulated both the guide and passenger strands of mature miR-9 [46]. Figure 3d–3f depicts results from pri-miR-9. Analysis of the primary miR-9 expression showed that E2 treatment significantly increased pri-mir-9-1 and pri-mir-9-2 at 1-week post OVX, as well as pri-mir-9-2 at 4 weeks post OVX (Figure 3d, 3e, gray line, *). By sharp contrast, E2 did not affect the expression levels of pri-mir-9-3 at any time point (Figure 3f, gray line, *). Finally, E2 had no effect on pri-miR-181a or pri-miR-495 at any time point (Figure 3h, 3i).
Effects of OVX+vehicle on primary miRNA (pri-miR) expression levels in the hypothalamus following varying lengths of OVX
One way ANOVA analyses across the deprivation time points showed that pri-mir-7a-2, pri-mir-9-1, pri-mir-125a, pri-mir-181a, and pri-mir-495 were all significantly altered by OVX alone, and in general, they were all decreased with age (Figure 3, black line, #). Specifically, expression levels of pri-mir-9-1 (Figure 3d, black line, #) were significantly decreased at 8 weeks post OVX, while pri-mir-125a expression was decreased at 12 weeks post OVX (Figure 3g, black line, #). Further, both primary transcripts for miR-181a and miR-495 were significantly decreased at 4, 8, and 12 weeks post OVX (Figure 3h–3i, black line, #). The only exception was for pri-mir-7a-2, which was significantly decreased at 4 weeks, but then increased at 8 weeks post OVX (Figure 3c, black line, #).
Effects of OVX+veh and OVX+E2 treatment on precursor miRNA (pre-miR) expression levels in the hypothalamus
A two-factor ANOVA analysis revealed that there was no significant interaction between age and treatment for any of the precursor miRNAs, again suggesting that the two factors are independent. Next, statistical analyses within each separate time point demonstrated that E2 treatment significantly altered the expression levels of 3 out of 7 pre-miRNAs tested when compared to vehicle-treated controls (Figure 4, gray line, *). Interestingly, E2 treatment significantly increased the expression of pre-miR-7a-2 at both 1 and 12 weeks post-OVX, but had no effect on pre-miR-7a-1 at any time point (Figure 4b, 4c, gray line, *). These results sharply contrast what was observed with the primary transcripts of these same miRNAs (see Figure 3b, 3c), suggesting that E2 acts differently at multiple levels of the miRNA biogenesis pathway. Similarly, the effects of E2 treatment were strikingly differently for pre-miR-9 (Figure 4d) compared to its effects on the primary form (pri-miR-9; see Figure 3d–3f). Specifically, E2 significantly decreased pre-mir-9 expression at 1 week post OVX, but then significantly increased its expression at 12 weeks post OVX (Figure 4d, gray line, *). While the primary transcript of miR-9 is transcribed from three different loci (hence, pri-miR-9-1, pri-miR-9-2, and pri-miR-9-3), the precursor hairpins generated from each primary transcript are too similar to be able to differentiate them using qRT-PCR. Therefore, it is possible that there were offsetting changes in each precursor form that might have obscured the final results. Interestingly, E2 completely abolished the steep age-related increase in pre-miR-181a observed at 4 weeks post-OVX (Figure 4f, gray line, *). These results were unexpected given the E2-induced increase in both mature and pri-miR-181a expression levels at varying time points (see Figures 2f and 3h). Finally, there were no effects of OVX alone on the expression levels for any of the precursor miRNAs tested (Figure 4, black lines).
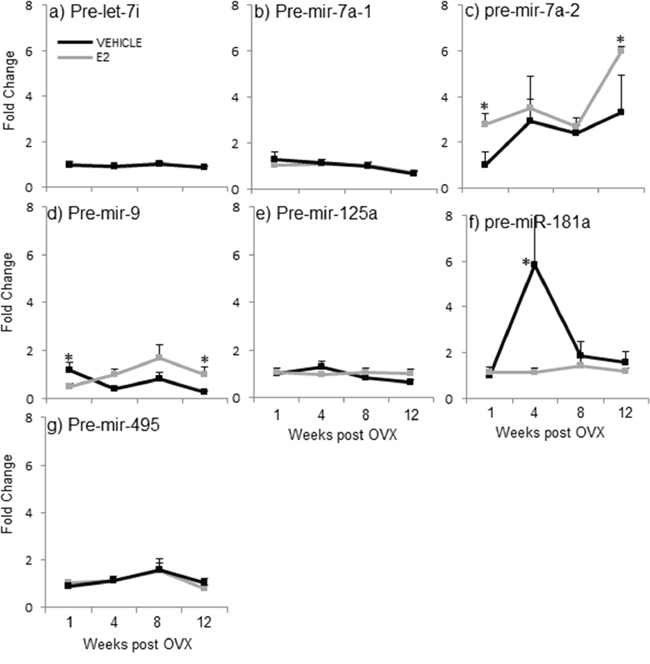
Figure 4: E2 regulation of the precursor miRNA expression in the hypothalamus after increasing lengths of ovarian hormone deprivation. a–g. Precursor miRNA expression was analyzed by real time qPCR and displayed as mean ± SEM fold change as compared to 1 week vehicle treated animals (N = 6/age/treatment). An * denotes a statistically significant effect of E2 as compared vehicle treated animals within a time point.
Experiment 2: E2 and age-related changes in mature miRNA expression are not explained by concomitant changes in the mRNA or protein levels of key components involved in miRNA biogenesis
Our data show that E2 regulates miRNA expression at various levels along the miRNA biosynthetic pathway, raising the possibility that E2 could regulate the transcription (mRNA) and/or protein levels of critical components required for miRNA biosynthesis. Therefore, we analyzed the mRNA expression of the nuclear proteins drosha and DGCR8 (key components of the microprocessor complex), as well as cytoplasmic proteins dicer, AGO2, XPO5, XRN2, and PAPD4 in the hypothalamus of animals subjected to our ovarian hormone deprivation paradigm (Figure 5). The statistical analysis revealed that there was a significant interaction of treatment and age on the mRNA expression levels of drosha, but not any of the other genes tested (F(3,38) = 3.762, p = 0.019). Specifically, the post hoc analysis revealed that E2 treatment increased drosha mRNA expression at 8 weeks post OVX (Figure 5a). Statistical analyses within each time point revealed that E2 treatment also increased DGCR8, dicer, and AGO2 mRNA expression levels at 8 weeks post OVX (Figure 5b–5d). Conversely, E2 treatment significantly decreased XPO5 at 1-week post OVX (Figure 5e). We did not observe any significant changes in the mRNA expression XRN2 and PAPD4 between treatment groups at any time point (Figure 5f, 5g, gray line). Moreover, we did not observe changes due to OVX alone in the mRNA expression of these genes (black lines).
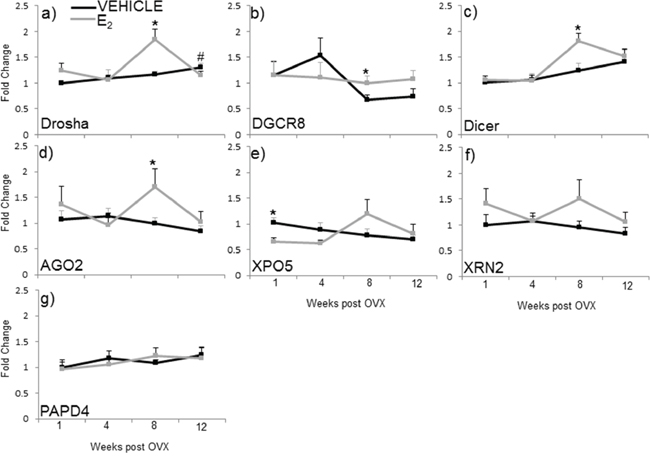
Figure 5: mRNA expression of miRNA biogenesis components in the hypothalamus after increasing lengths of ovarian hormone deprivation. a–g. mRNA expression was analyzed by real time qPCR and displayed as mean ± SEM fold change as compared to 1 week vehicle treated animals (N = 6/age/treatment). An * denotes a statistically significant effect of treatment within a time point.
Next, we compared the mRNA levels of drosha, DGCR8, dicer, AGO2, and XPO5 with their translated protein levels from the same animals (Figures 6, 7). Interestingly, there was no statistically significant effect of treatment or OVX on the protein levels of any of these genes, despite our observed statistically significant changes in mRNA levels. These data suggest that E2-mediated changes in mature miRNA expression levels cannot be explained by mRNA/protein changes in these key miRNA biosynthetic components.
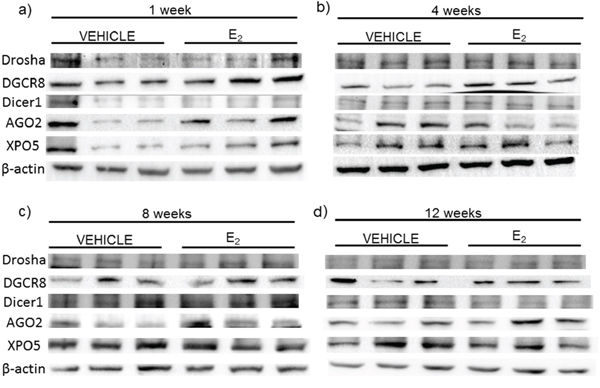
Figure 6: Protein expression of miRNA biogenesis components in the hypothalamus after increasing lengths of ovarian hormone deprivation. a–d. Protein expression was analyzed by western blot analysis. 60 μg of total protein for each sample was run on 4–10% polyacrylamide gel. Blots were incubated with the indicated antibodies and visualized on Bio-Rad Chemidoc. Representative blots are shown (N = 3/treatment/age group).
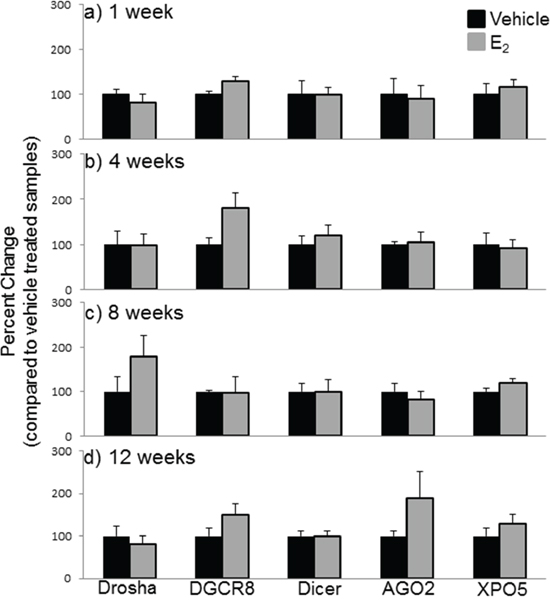
Figure 7: Protein expression of miRNA biogenesis components in the hypothalamus after increasing lengths of ovarian hormone deprivation. a–d. Quantification of western blots described in figure 7 (N = 6/treatment/age group). Data are represented as mean ± SEM percentage as compared to vehicle treated animals. No statistical significance was observed between treatment groups.
Experiment 3: ERβ mediates E2 effects on mature miRNA expression in the hypothalamus of aged female rats one-week post-OVX
The effects of E2 are mediated primarily by two estrogen receptors, ERα and ERβ, and both have been shown to regulate miRNA expression in cancer cell lines [49, 50, 51]. To determine which receptor mediates the regulation of miRNA expression in the aging female brain, we first examined the expression levels of ERα and ERβ mRNA in our ovarian hormone deprivation paradigm. A two-way ANOVA analysis revealed that there was a statistically significant interaction between treatment and deprivation period for ERβ (F(3,38) = 3.762, p = 0.014), but not ERα. ERβ mRNA expression was significantly decreased at 4, 8, and 12 weeks post-OVX in the vehicle treated animals compared to E2 treatment (Figure 8b, black line, #). Next, we ovariectomized aged (18 month old) Fischer 344 female rats and 1 week later administered either E2, or an ERα (PPT) or ERβ (DPN) selective agonist. We then measured the primary, precursor, and mature miR expression levels for each of our previously identified E2 regulated miRNAs using qRT-PCR (Figure 9). First, we analyzed the expression levels of the primary transcripts, which demonstrated that both PPT and DPN treatment significantly decreased the expression of pri-miR-7a, pri-miR-125a, pri-miR-181a, and pri-miR-495 compared to either vehicle or E2 treated animals (Figure 9a). These results were surprising given that no effect was observed with E2 treatment alone. Interestingly, let-7i primary transcript expression was the only miRNA to be significantly increased with DPN and PPT treatment (Figure 9a). By contrast, PPT significantly increased the expression of the precursor form of miR-181a (pre-miR-181a), but had no effect on the precursor forms for any of the other miRNAs (Figure 9b). Similarly, DPN significantly increased the precursor form of miR-495 (pre-miR-495), but not any other precursor miRNA (Figure 9b). Finally, DPN, but not PPT, mimicked the effects of E2 on the expression levels of mature let-7i, and miR-7a, which was consistent with our earlier observations after 1 week of E2 deprivation (Figure 9c).
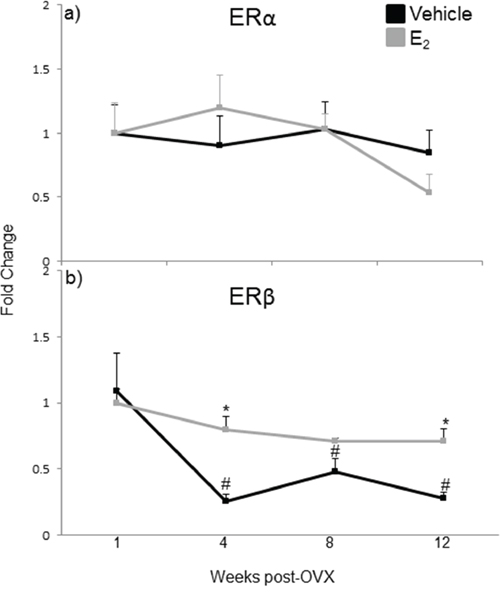
Figure 8: mRNA expression of estrogen receptor isoforms in the hypothalamus after increasing lengths of ovarian hormone deprivation. a–b. mRNA expression of ERα (a) and ERβ (b) was analyzed by real time qRT-PCR and displayed as mean ± SEM fold change as compared to 1 week vehicle treated animals (N = 6/age/treatment. An * denotes a statistically significant effect of treatment within a time point within a treatment group. A # denotes a statistically significant difference across time points within a treatment group.
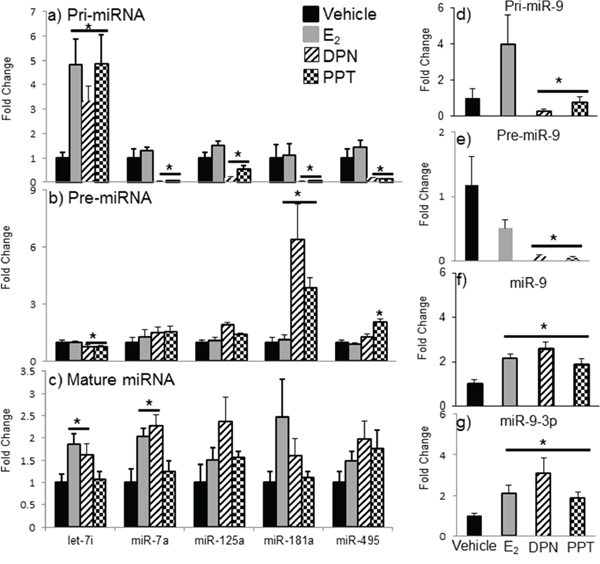
Figure 9: Effects of ERα and ERβ specific agonists on mature miRNA expression in the aged hypothalamus following 1 week of ovarian hormone deprivation. Pri-miRNA a, d. pre-miRNA b, e. and mature miRNA c, f, g. were measured by qRT-PCR from animals administered vehicle, E2, DPN (ERβ agonist), or PPT (ERα agonist). Data are displayed as mean ± SEM fold change as compared to vehicle treated animals (N = 6/age/treatment). An * denotes a statistically significant effect of E2 as compared vehicle treated animals within a time point.
The effects of PPT and DPN on miR-9 were particularly interesting. Our previously published observations, as well as replicated data herein, demonstrated that E2 significantly increased both the guide (miR-9) and passenger (miR-9-3p) strands of mature miR-9 after 1 week of E2 deprivation in aged animals. However, both PPT and DPN significantly decreased the pri-miR-9 and pre-miR-9 forms of this miRNA (Figure 9d, 9e). Notably, E2 tended to increase pri-miR-9, and decrease pre-miR-9, but neither effect was statistically significant. Finally, E2, PPT, and DPN all significantly increased mature miR-9 and miR-9-3p, but DPN increased each to a greater degree (Figure 9f, 9g).
DISCUSSION
Our primary objective in these studies was to test the efficacy of acute E2 treatment, administered at different time points following the complete loss (via OVX) of ovarian hormones, on miRNA expression in the aged female brain. The results from this study demonstrated several novel findings. First, E2 treatment altered mature miRNA expression in the brain of aged animals after 1 week of ovarian hormone deprivation, which replicated our previously published findings, but this effect was lost following longer periods of ovarian hormone deprivation. These results are consistent with that of the Timing Hypothesis, which suggests that the efficacy of E2 action changes with advanced age and length of time postmenopause. Second, we showed that aging alone (i.e. ovarian intact animals) also had a significant effect on mature miRNA expression in many cases, and these are the first data to describe miRNA expression levels in the brains of animals at several time points of advanced age. Surprisingly, ovarian intact animals displayed a different miRNA expression profile then did the OVX+vehicle treated animals suggesting that these E2-regulated miRNAs are sensitive to ovarian factors in addition to E2, however, to our knowledge there is very little known about the regulation of miRNAs in the brain by other ovarian factors. Third, our results showed that E2 treatment selectively altered the expression of the primary and precursor transcripts of these same miRNAs, however those expression levels often did not correspond to the levels of their mature miRNA counterparts. Our interpretation of these data is that E2 can act at multiple levels along the miRNA biosynthesis pathway, and that E2 perhaps plays a role in the stabilization of mature miRNAs. Finally, using ER selective agonists we demonstrated that the effects of E2 on mature miRNA expression in the hypothalamus are likely mediated primarily through ERβ. Taken together these results extend our previous findings by suggesting a possible mechanism for the lack of therapeutic effectiveness for HT in post-menopausal women.
Aging has been shown to alter mature miRNA expression profiles in a variety tissues [34, 52, 53, 54, 55]. Our data (ovarian intact animals) also showed age-related changes in mature miRNA expression in the hypothalamus. Previous studies have shown that aging can differentially regulate distinct groups of miRNAs, suggesting that the miRNAs we tested in the present study could be part of a larger shift in miRNA expression in the brain. The mechanism by which aging alters miRNA expression is not currently known, though it has been shown that aging can alter post-transcriptional modifications of mature miRNAs thereby altering their stability [56]. Unexpectedly, the OVX + vehicle treated animals had a different aging miRNA expression pattern than did the ovarian intact animals, and the effects were distinct for each miRNA tested. Importantly, the circulating E2 levels were not different between the OVX+ vehicle and ovarian intact animals (Figure 1), suggesting that ovarian factors other than E2 contribute to mature miRNA expression levels in the aged female brain.
Our previous work showed that E2 differentially regulated the expression of mature miRNAs in the brains of young, compared with aged, female rats [46]. This current study extends those findings by focusing only on aged animals that were given E2 replacement at varying times post-OVX. Consistent with our earlier findings, E2 treatment significantly increased 5 out of 7 previously identified mature miRNAs after only a brief period of ovarian hormone deprivation (i.e. one week) in aged animals. It is important to note that these current studies examined mature miRNA expression only in the hypothalamus, whereas our previous findings were mostly derived from the hippocampus. Nevertheless, these data remained consistent with our earlier results in that there appears to be brain-region specific effects of E2 on miRNA expression. Notably, we observed no differences in mature miRNA expression between E2- and vehicle-treated animals following longer periods of ovarian hormone deprivation (i.e. 4, 8, and 12 weeks). These results are in-line with the concept of the Timing Hypothesis, which predicts a lack of E2 efficacy following longer periods of ovarian hormone deprivation. miRNAs are important regulators of mRNA translation and are predicted to impact the expression of more than 60% of all protein-coding genes [57]. The implication of these results suggests that a loss of E2-regulated miRNAs postmenopause could negatively impact multiple downstream target genes, although it remains unclear which specific downstream target genes these particular miRNAs regulate.
Despite the critical role of miRNAs in all physiological systems, little is known about the tissue-specific regulation of miRNA biosynthesis and processing. In general, global miRNA biogenesis follows a well-defined pathway beginning with the generation of a long primary transcript (pri-miR) and ending with a single-stranded 22 nt mature miRNA product [20, 21, 24, 25, 58]. There are several steps along this pathway that are potential points where unique tissue- and age-specific expression of individual miRNAs could occur. Estrogen receptors act primarily as transcription factors on various gene promoters, therefore, the most likely site of E2 regulation is at the transcriptional level of the primary miRNA form. Indeed, others have demonstrated that E2 regulates several pri-miRs through canonical estrogen response elements (EREs) in breast cancer cell models [49, 50, 59]. Therefore, we next measured the expression levels of the primary (pri-miR) and precursor (pre-miR) forms of our identified E2-regulated mature miRNAs at varying times post-OVX. Our prediction was that the expression levels of these primary transcripts would match that of the mature miRNA effector, however this was not what we observed. In fact, there was considerable discordance between the pri-, pre-, and mature forms for most of the miRNAs tested. The discordance observed could be the result of altered processing of these miRNAs. Post-transcriptional modifications to RNAs, including miRNAs, are common and important regulatory mechanisms, which can promote or inhibit their processing [22, 28, 29, 60]. These studies suggest that not only expression, but also sequence changes to the miRNA should be analyzed to more completely understand their regulation and function. There is little known about the temporal regulation of miRNA biosynthetic events, but these results suggest that E2 could regulate different steps along the pathway at different times. It also demonstrates a limitation of our studies in that each miRNA was measured at just one snapshot in time following E2 treatment (24 hours following the last injection). Nevertheless, the fact that E2 regulated all of the miRNAs forms (primary, precursor, or mature) at some time point indicates that E2 might also regulate some of the important key proteins involved in miRNA biogenesis and/or stabilization.
Mature single-stranded miRNA molecules in association with an AGO protein comprise the two core components of the RNA-induced silencing complex [61]. The mature miRNA molecule is formed after the cytoplasmic RNase III enzyme dicer cleaves the pre-miR to form a small miRNA duplex structure [20]. Only one strand of this duplex (leading strand) associates with AGO; the other strand (passenger strand) remains free, either to bind to RNAs and other proteins or be degraded [62, 63, 64]. In contrast to that description of miRNA formation, our data showed that E2 treatment not only increased the expression of mature miR-9 in aged females, but it also increased the expression of the miR-9a passenger strand, miR-9-3p (current study, and [46]). Because both strands of the miR-9 duplex are derived after dicer processing of the same precursor molecule, these results provide strong evidence that E2 can regulate miRNA expression at the level of molecular stabilization. Therefore, to determine if E2 regulates key components of miRNA biogenesis or stabilization we examined their mRNA and protein levels in our ovarian hormone deprivation paradigm. We observed that E2 treatment significantly increased drosha and dicer mRNA at 8 weeks post-OVX, however these changes were not mirrored by changes in protein levels at that same time point. One possibility is that we missed the window of detection, given that we measured both mRNA and protein at only one time point following E2 treatment (24 hours after the last injection). Moreover, the levels of mRNA and protein for these enzymes do not necessarily reflect changes in enzymatic activity. Importantly, the enzymatic activities are acutely sensitive to rapid changes in co-factor binding and posttranslational modifications [20, 22, 23, 58, 65], all of which are potential targets for E2 regulation. At this time, we are unaware of any enzymatic assay that has been developed for the detection of dicer or drosha activity either in cell line models or tissue, making this a very important future direction for research efforts. Overall, there were no significant changes due to age or treatment on the protein expression for any of the other key miRNA biogenesis or stabilization components tested. These results are consistent with our findings, and that of others, showing that E2 and age only regulate a very small subset of miRNAs [46, 51]. Therefore, E2 regulation of a major protein component in the miRNA biogenesis pathway is unanticipated, as that would predict a broad impact on a much greater number of miRNAs.
The results of the WHI studies prompted both basic and clinical scientists to reevaluate the role of E2 in the aging brain. Changes in the composition, number, or structure of estrogen receptors (ERα or ERβ) would be an obvious explanation for reduced E2 efficacy with age. Indeed, previously published studies have shown that both ERα and ERβ expression are altered with aging in specific nuclei of the hypothalamus, although the reported findings are somewhat contradictory with respect to ERβ [66, 67, 68]. Our data showed that there was a significant decrease in ERβ mRNA expression at 4, 8, and 12 weeks post-OVX (equivalent to 19, 20, and 21 months old, respectively) in the vehicle treated animals. However, treatment with the ERβ specific agonist DPN was equally effective as E2 treatment on mature miRNA expression, and there were no changes observed in ERα expression, suggesting that decreased ER availability is not a primary factor for the lack of E2 efficacy in the aged female brain. Another possibility is that the expression of fully functional ERβ splice variants is altered with age. For example, ERβ2 has a lower E2 binding affinity compared to the wild type ERβ. ERβ2 also has different transcriptional efficacy, its expression increases with age, and it was correlated with an increase in depressive behaviors in aged Sprague-Dawley female rats [69, 70, 71]. Our experiments were designed to detect the full-length wild-type ERβ, which would include detection of all known ERβ splice variants. Therefore, the ERβ mRNA expression levels that we observed would not differentiate between the relative amounts of ERβ2 at varying ages, and increases in these receptor splice variants could decrease E2 efficacy. Another possible explanation for reduced E2 efficacy over time could be that the expression and/or association of ER coregulatory proteins are altered with age. Indeed, we have previously demonstrated that the protein complexes associated with ERβ in the brain are altered by E2 in an age-dependent manner, despite no age-related changes in the expression of the cofactor proteins themselves [72]. These data indicate that altered ER and/or cofactor gene expression might not be good markers to assess the efficacy of E2 signaling.
Although the actions of E2 are mediated primarily through its two classical nuclear receptors, ERα and ERβ, recent evidence has emerged implicating E2 regulation of cell function through a variety of other intracellular signaling pathways, some of which are mediated by membrane-bound ER or G protein-coupled receptors. In these studies, we used a straightforward approach to try to identify whether the classical ERs were mediating the E2-induced regulation of mature miRNA expression in the aged brains following a brief period (1 week) of ovarian hormone deprivation. To that end, we took advantage of the recent development of the specific ERα and ERβ agonists, PPT (propylpyrazole-triol) and DPN (diarylpropionitrile), respectively. These agonists have been shown to be highly selective for their respective form of the receptor when used at the doses we administered [73, 74]. In general, our results indicated that the E2-induced regulation of mature miRNAs after one week of OVX is mediated by both receptors, though ERβ was able to specifically regulate let-7i and miR-7a independent of ERα. Interestingly, there were some instances, most notably at the primary (pri-miR) level, where PPT and DPN were equally efficacious and E2 had no apparent effect (see Figure 9). Notably, the high selectivity of PPT and DPN for their respective receptors indicates that they only bind and activate homodimer configurations of the receptors. Therefore, it is possible that the effects of E2 on miRNAs are mediated by ERα and ERβ heterodimers. Indeed, our data demonstrating that both DPN and PPT regulated pri-miRNA levels, yet there was no apparent effect of E2 on these same primary transcripts support that hypothesis. Further, it is also possible that there are changes in homo- and heterodimer formations that change with age and/or prolonged periods of E2 deprivation, suggesting that these ER selective agonists could be a useful therapeutic tool for postmenopausal women at specific ages.
MATERIALS AND METHODS
Ethics statement
All animal protocols were approved by the Institutional Animal Care and Use Committee at Loyola University Chicago, IACUC approval #2009018. Surgeries were performed under vaporized isoflurane anesthesia. Post-operation, animals were singly housed and provided with acetaminophen analgesic (122.7 mg/kg) in tap water for 3 days. All measures were taken to minimize pain and suffering.
Animals
Experiment 1: Ovarian intact aged female rats
Fischer 344 rats were obtained from Charles River Laboratories (Wilmington, MA) at 18 months of age (N = 24). The animals were allowed to acclimate to the housing facility at Loyola for 7 days after arrival. Animals were housed two per cage and were allowed free access to standard rat chow and tap water. They were left undisturbed until euthanasia at 18, 19, 20, or 21 months of age.
Experiment 2: Acute treatment with vehicle/E2 at varying times post ovariectomy (OVX) in aged female rats
Female Fischer 344 rats were obtained from the National Institutes of Aging (NIA) colony (Taconic) at 18 months (N = 46) of age. The animals were allowed to acclimate to the housing facility at Loyola for 7 days after arrival. Animals were housed two per cage and were allowed free access to standard rat chow and tap water. Animals were OVX at 18 months of age after the acclimation period and then left undisturbed for 1, 4, 8, or 12 weeks (N = 11–12/age group) following OVX (Figure 1a). After the designated time the animals were given a subcutaneous injection of either safflower oil (vehicle; N = 5–6/age group) or 2.5 μg/kg 17β-estradiol (E2; N = 6/age group) dissolved in safflower oil once/day for 3 consecutive days. The animals were anesthetized and then euthanized by rapid decapitation 24 hours following the last E2 injection.
Experiment 3: Acute treatment of selective estrogen receptor agonists after 1 week post-OVX in aged female rats
Fischer 344 rats were obtained from Charles River Laboratories (Wilmington, MA) at 18 months of age (N = 14). Animals were housed two per cage and were allowed free access to standard rat chow and tap water. Animals were OVX at 18 months of age after the acclimation period, left undisturbed for 1 week and then were given a subcutaneous injection of either Diarylpropionitrile (DPN, Tocris, 1.0 mg/kg, N = 7) or 4,4′,4′’-(4-Propyl-[1H]-pyrazole-1,3,5-triyl)trisphenol (PPT, Tocris, 0.5 mg/kg, N = 7) once/day for 3 consecutive days. These doses of DPN and PPT have previously been shown to have physiological effects on cognitive function [73, 74].
Ovariectomy
Animals were deeply anesthetized with vaporized isofluorane and bilaterally ovariectomized (OVX) as described previously [72]. Briefly, the ovary and distal end of the uterine horn were pulled from the body cavity through a 1 cm incision made through the skin and body wall. The uterine horn was clamped with a hemostat and ligated proximal to the clamp. The entire ovary and distal uterine horn were then removed. Animals were singly housed and provided with acetaminophen analgesic (122.7 mg/kg) in tap water for 3 days postoperative. During this time, animals were weighed once/day and their water intake was measured. Following 3 days of analgesia the animals were double-housed with their previous cage mate for the duration of the experiment.
Blood collection and tissue processing
The animals were anesthetized using vaporized isofluorane and then euthanized by rapid decapitation 24 hours following the last treatment. Trunk blood was collected on ice into heparinized 10 ml round bottom tubes, centrifuged at 4000 RPM for 8 min., and plasma stored at −20°C until further processing. Brains were quickly removed, sagittally sectioned on ice into left and right hemispheres, and the whole hypothalamus was microdissected from the right side of the brain. The hypothalami were placed in separate microcentrifuge tubes containing QIAzol lysis reagent (Qiagen, Inc., Germantown, MD) for subsequent homogenization and RNA extraction. The left side of the brain was rapidly frozen in 2-methylbutane, sectioned at 200 μm on a freezing microtome, and the whole hypothalamus (−0.26 to −4.52 relative of bregma, according to Paxinos and Watson, Rat Brain Atlas, Fourth edition), was microdissected using a Palkovit's brain punch tool (1.0 mm, Stoelting, Inc., Wood Dale, IL). Tissue punches were placed in a microcentrifuge tube on dry ice and stored at −80°C.
17β-estradiol plasma concentrations
The plasma samples first underwent a liquid-liquid extraction using diethyl ether to eliminate interfering compounds in the plasma as previously described [75]. Following diethyl ether extraction, samples were reconstituted using sample buffer contained in the 17β-estradiol high sensitivity ELISA kit (Enzo Life Sciences, Cat. No. AD 901 174), which was used to determine concentration of circulating E2 levels. Absorbance was measured on a BioTek (Winooski, VT) Synergy HT plate reader. The circulating plasma E2 concentrations in the E2 and vehicle animals were 56.68 (+/− 20.29) and 36.10 (+/− 9.70) pg/mL respectively (Figure 1b). These E2 levels were consistent with physiological levels observed during late diestrous/early proestrous and with levels achieved in postmenopausal women following HT [76]. The intra- and inter-assay %CV was 4.23 and 6.85 pg/ml respectively.
RNA isolation
Total RNA was isolated from the whole right hypothalamus using the miRNeasy Mini Kit (Qiagen, Inc., Germantown, MD) according to manufacturer's instructions. All RNA samples were quantified and analyzed for quality using Nanodrop spectrophotometry and visualization of the RNA on 1.5% agarose gel.
Precursor and mature miRNA cDNA synthesis
Total RNA (1.0 μg) was used to reverse transcribe miRNA using NCode™ VILO™ miRNA cDNA synthesis kit (Life Technologies, Carlsbad, CA) to assay for mature miRNA expression. To assay for the primary and precursor miRNA, we utilized a miRNA specific reverse transcription method described previously [77]. Briefly, 2.0 μg of total RNA was added to 1.5 μl of primer cocktail containing 10 μM of antisense primers (Supplementary Table S1). The reaction mixture was incubated at 80°C for 5 minutes, 60°C for 5 minutes, and then finally allowed to cool to room temperature. Reagents from the ThermoScript™ RT-PCR System (Life Technologies, Carlsbad, CA) were then added for a final volume of 20 μl. The reactions were incubated at 60°C for 45 minutes and then 85°C for 5 minutes. 1.0 μl of RNase H was added to each reaction and then incubated at 37°C for 20 minutes.
Messenger RNA(mRNA) cDNA synthesis
Total RNA (2.0 μg) was reverse transcribed using the High Capacity cDNA Reverse Transcriptase Kit (Applied Biosystems, Foster City, CA).
Quantitative real-time qRT-PCR
miRNA and mRNA qRT-PCR was performed with Fast Start Universal SYBR Green Master Mix (Roche-Genentech, San Francisco, CA) on an Eppendorf Realplex4. Forward primers for specific mature miRNAs were designed as described in the Ncode™ miRNA First-Strand cDNA synthesis kit handbook (Life Technologies, Carlsbad, CA) and using miRBase 18 as a sequence reference (Supplementary Table S1). Primary and precursor miRNA primers were designed as described previously (Supplementary Table S1) [77]. Primers for mRNAs were obtained from previous published studies [27, 78] (Supplementary Table S2). 18s rRNA and HPRT were used as a loading control and to normalize the data for analysis for miRNA and mRNA qRT-PCR respectively. The following program was used for miRNA qRT-PCR: 1) 95°C for 10 minutes, 2) 95°C for 20 seconds, 3) 59°C for 20 seconds, 4) 72°C for 12 sec, and melting curve analysis (Supplementary Table S1). The following program was used for mRNA qRT-PCR: 1) 95°C for 10 minutes, 2) 95°C for 30 seconds, 3) 59°C for 30 seconds, 4) 72°C for 30 sec, and melting curve analysis (Supplementary Table S2). ERβ mRNA expression was analyzed using Taqman Gene Expression Assay (Rn01527840_m1) with a custom FAM probe for ERβ (CAAGAAAATCCCTGGCTTTGTGGAG) (Supplementary Table S2). The following program was used for Taqman RT-PCR: 1) 95°C for 10 minutes, 2) 95°C for 15 seconds, and 3) 60°C for 60 seconds. miRNA and mRNA expression was analyzed using the ΔΔCt method as described previously [79].
Western blots
Protein lysate from hypothalamic tissue punches was isolated using T-PER reagent (Thermo Scientific, Waltham, MA). Briefly, tissue punches were homogenized in 500 μl of T-PER reagent with 1x cOmplete mini EDTA-free protease inhibitor cocktail (Roche-Genentech, San Francisco, CA) with a motorized mortar. Tissue lysate was centrifuged at 10,000 × g for 5 minutes and the supernatant was collected into a separate microfuge tube. Protein samples were then concentrated using a methanol-chloroform extraction as described previously [80]. Total protein lysate (60 μg) was dissolved and boiled at 95°C for 5 minutes in a 1x reducing sample buffer (Thermo Scientific, Waltham, MA). Protein samples were resolved on a gradient (4–10%) polyacrylamide gel at 90V for 20 minutes and then 120V for 60 minutes using Bio-Rad Mini-Protean 3 system. Separated proteins were then transferred to PVDF Immobilon-P membranes (Millipore, Billerica, MA) at 100V for two hours. Membranes were blocked with 5% BSA with 1x TBST (0.1% tween) for 1 hour. Membranes were then incubated with the indicated primary antibody overnight with constant shaking at 4°C (Supplementary Table S3). The primary antibody was removed and the membranes were rinsed three times with 1x TBST (0.1% tween). Membranes were then incubated with 1:5000 goat anti-rabbit HRP secondary antibody (Santa Cruz Biotechnology, Dallas, Texas) in 5% BSA with 1x TBST (0.1% tween) for two hours with shaking at room temperature. Membranes were then rinsed with 1x TBST (0.1% tween) three times and then developed using Super Signal West Pico Chemiluminescent substrate (Thermo Scientific, Waltham, MA). Blots were visualized using the Bio-Rad Chemi-doc stations. Densitometry analysis was performed using Image Lab software (Bio-Rad Laboratories, Des Plaines, IL). Membranes were then stripped using a mild stripping buffer (Abcam, Cambridge, England) and re-blotted with another primary antibody. Beta actin expression was used as the loading control and to normalize the data for analysis.
Statistics
Expression of miRNAs and mRNAs in the ovarian hormone deprivation paradigm were analyzed by two-way ANOVA with age and treatment as factors. A significant interaction between age and treatment was followed by a Tukey's post hoc test to determine statistically significant differences (p < 0.05) between groups. A separate Tukey's post-hoc test was performed within groups that showed a statistically significant main effect of age and/or treatment. When no significant interaction was observed, the two factors were considered independent and Student's T-tests were used to analyze between treatments within a single time point. A one-way ANOVA with treatment as the main factor, followed by Tukey's posthoc test, was used to determine significant differences of E2, DPN, and PPT. All data are presented as mean ± SEM. Statistical significance was noted when p < 0.05.
ACKNOWLEDGMENTS AND FUNDING
Support for this work was provided by NIH R01AG033605.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest
REFERENCES
1. Arias E. United States life tables, 2010. National vital statistics reports: from the Centers for Disease Control and Prevention, National Center for Health Statistics, National Vital Statistics System. 2014; 63:1–63.
2. Colenda CC, Legault C, Rapp SR, DeBon MW, Hogan P, Wallace R, Hershey L, Ockene J, Whitmer R, Phillips LS, Sarto GE. Psychiatric disorders and cognitive dysfunction among older, postmenopausal women: results from the Women’s Health Initiative Memory Study. Am J Geriatr Psychiatry. 2010; 18:177–186.
3. Goveas JS, Espeland MA, Hogan P, Dotson V, Tarima S, Coker LH, Ockene J, Brunner R, Woods NF, Wassertheil-Smoller S, Kotchen JM, Resnick S. Depressive symptoms, brain volumes and subclinical cerebrovascular disease in postmenopausal women: the Women’s Health Initiative MRI Study. J Affect Disord. 2011; 132:275–284.
4. Goveas JS, Espeland MA, Hogan PE, Tindle HA, Shih RA, Kotchen JM, Robinson JG, Barnes DE, Resnick SM. Depressive Symptoms and Longitudinal Changes in Cognition: Women’s Health Initiative Study of Cognitive Aging. J Geriatr Psychiatry Neurol. 2014; 27:94–102.
5. Fernandez SM, Lewis MC, Pechenino AS, Harburger LL, Orr PT, Gresack JE, Schafe GE, Frick KM. Estradiol-induced enhancement of object memory consolidation involves hippocampal extracellular signal-regulated kinase activation and membrane-bound estrogen receptors. J Neurosci. 2008; 28:8660–8667.
6. Ferrini M, Lima A, De Nicola AF. Estradiol abolishes autologous down regulation of glucocorticoid receptors in brain. Life Sci. 1995; 57:2403–2412.
7. Han X, Aenlle KK, Bean LA, Rani A, Semple-Rowland SL, Kumar A, Foster TC. Role of estrogen receptor alpha and beta in preserving hippocampal function during aging. J Neurosci. 2013; 33:2671–2683.
8. Liu F, Day M, Muniz LC, Bitran D, Arias R, Revilla-Sanchez R, Grauer S, Zhang G, Kelley C, Pulito V, Sung A, Mervis RF, Navarra R, et al. Activation of estrogen receptor-beta regulates hippocampal synaptic plasticity and improves memory. Nat Neurosci. 2008; 11:334–343.
9. Yao J, Hamilton RT, Cadenas E, Brinton RD. Decline in mitochondrial bioenergetics and shift to ketogenic profile in brain during reproductive senescence. Biochim Biophys Acta. 2010; 1800:1121–1126.
10. Zhang QG, Raz L, Wang R, Han D, De Sevilla L, Yang F, Vadlamudi RK, Brann DW. Estrogen attenuates ischemic oxidative damage via an estrogen receptor alpha-mediated inhibition of NADPH oxidase activation. J Neurosci. 2009; 29:13823–13836.
11. Espeland MA, Brunner RL, Hogan PE, Rapp SR, Coker LH, Legault C, Granek I, Resnick SM. Long-term effects of conjugated equine estrogen therapies on domain-specific cognitive function: results from the Women’s Health Initiative study of cognitive aging extension. J Am Geriatr Soc. 2010; 58:1263–1271.
12. Espeland MA, Rapp SR, Shumaker SA, Brunner R, Manson JE, Sherwin BB, Hsia J, Margolis KL, Hogan PE, Wallace R, Dailey M, Freeman R, Hays J. Conjugated equine estrogens and global cognitive function in postmenopausal women: Women’s Health Initiative Memory Study. Jama. 2004; 291:2959–2968.
13. Goveas JS, Espeland MA, Woods NF, Wassertheil-Smoller S, Kotchen JM. Depressive symptoms and incidence of mild cognitive impairment and probable dementia in elderly women: the Women’s Health Initiative Memory Study. J Am Geriatr Soc. 2011; 59:57–66.
14. Manson JE, Chlebowski RT, Stefanick ML, Aragaki AK, Rossouw JE, Prentice RL, Anderson G, Howard BV, Thomson CA, LaCroix AZ, Wactawski-Wende J, Jackson RD, Limacher M, et al. Menopausal hormone therapy and health outcomes during the intervention and extended poststopping phases of the Women’s Health Initiative randomized trials. Jama. 2013; 310:1353–1368.
15. Prentice RL, Manson JE, Langer RD, Anderson GL, Pettinger M, Jackson RD, Johnson KC, Kuller LH, Lane DS, Wactawski-Wende J, Brzyski R, Allison M, Ockene J, et al. Benefits and risks of postmenopausal hormone therapy when it is initiated soon after menopause. Am J Epidemiol. 2009; 170:12–23.
16. Shumaker SA, Legault C, Kuller L, Rapp SR, Thal L, Lane DS, Fillit H, Stefanick ML, Hendrix SL, Lewis CE, Masaki K, Coker LH. Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women: Women’s Health Initiative Memory Study. Jama. 2004; 291:2947–2958.
17. Zandi PP, Carlson MC, Plassman BL, Welsh-Bohmer KA, Mayer LS, Steffens DC, Breitner JC. Hormone replacement therapy and incidence of Alzheimer disease in older women: the Cache County Study. Jama. 2002; 288:2123–2129.
18. Baek D, Villen J, Shin C, Camargo FD, Gygi SP, Bartel DP. The impact of microRNAs on protein output. Nature. 2008; 455:64–71.
19. Selbach M, Schwanhausser B, Thierfelder N, Fang Z, Khanin R, Rajewsky N. Widespread changes in protein synthesis induced by microRNAs. Nature. 2008; 455:58–63.
20. Chendrimada TP, Gregory RI, Kumaraswamy E, Norman J, Cooch N, Nishikura K, Shiekhattar R. TRBP recruits the Dicer complex to Ago2 for microRNA processing and gene silencing. Nature. 2005; 436:740–744.
21. Han J, Lee Y, Yeom KH, Nam JW, Heo I, Rhee JK, Sohn SY, Cho Y, Zhang BT, Kim VN. Molecular basis for the recognition of primary microRNAs by the Drosha-DGCR8 complex. Cell. 2006; 125:887–901.
22. Heo I, Joo C, Cho J, Ha M, Han J, Kim VN. Lin28 mediates the terminal uridylation of let-7 precursor MicroRNA. Mol Cell. 2008; 32:276–284.
23. Lee HY, Zhou K, Smith AM, Noland CL, Doudna JA. Differential roles of human Dicer-binding proteins TRBP and PACT in small RNA processing. Nucleic Acids Res. 2013; 41:6568–6576.
24. Lee Y, Ahn C, Han J, Choi H, Kim J, Yim J, Lee J, Provost P, Radmark O, Kim S, Kim VN. The nuclear RNase III Drosha initiates microRNA processing. Nature. 2003; 425:415–419.
25. Lee Y, Jeon K, Lee JT, Kim S, Kim VN. MicroRNA maturation: stepwise processing and subcellular localization. Embo J. 2002; 21:4663–4670.
26. Lee Y, Kim M, Han J, Yeom KH, Lee S, Baek SH, Kim VN. MicroRNA genes are transcribed by RNA polymerase II. Embo J. 2004; 23:4051–4060.
27. Nothnick WB, Healy C, Hong X. Steroidal regulation of uterine miRNAs is associated with modulation of the miRNA biogenesis components Exportin-5 and Dicer1. Endocrine. 2010; 37:265–273.
28. Burroughs AM, Ando Y, de Hoon MJ, Tomaru Y, Nishibu T, Ukekawa R, Funakoshi T, Kurokawa T, Suzuki H, Hayashizaki Y, Daub CO. A comprehensive survey of 3′ animal miRNA modification events and a possible role for 3′ adenylation in modulating miRNA targeting effectiveness. Genome Res. 2010; 20:1398–1410.
29. Wyman SK, Knouf EC, Parkin RK, Fritz BR, Lin DW, Dennis LM, Krouse MA, Webster PJ, Tewari M. Post-transcriptional generation of miRNA variants by multiple nucleotidyl transferases contributes to miRNA transcriptome complexity. Genome Res. 2011; 21:1450–1461.
30. Chatterjee S, Grosshans H. Active turnover modulates mature microRNA activity in Caenorhabditis elegans. Nature. 2009; 461:546–549.
31. Amar L, Benoit C, Beaumont G, Vacher CM, Crepin D, Taouis M, Baroin-Tourancheau A. MicroRNA expression profiling of hypothalamic arcuate and paraventricular nuclei from single rats using Illumina sequencing technology. J Neurosci Methods. 2012; 209:134–143.
32. Bak M, Silahtaroglu A, Moller M, Christensen M, Rath MF, Skryabin B, Tommerup N, Kauppinen S. MicroRNA expression in the adult mouse central nervous system. Rna. 2008; 14:432–444.
33. Hua YJ, Tang ZY, Tu K, Zhu L, Li YX, Xie L, Xiao HS. Identification and target prediction of miRNAs specifically expressed in rat neural tissue. BMC Genomics. 2009; 10:214.
34. Inukai S, de Lencastre A, Turner M, Slack F. Novel microRNAs differentially expressed during aging in the mouse brain. PLoS One. 2012; 7:e40028.
35. Olsen L, Klausen M, Helboe L, Nielsen FC, Werge T. MicroRNAs show mutually exclusive expression patterns in the brain of adult male rats. PLoS One. 2009; 4:e7225.
36. Persengiev S, Kondova I, Otting N, Koeppen AH, Bontrop RE. Genome-wide analysis of miRNA expression reveals a potential role for miR-144 in brain aging and spinocerebellar ataxia pathogenesis. Neurobiol Aging. 2011; 32. 2316:e2317–2327.
37. Herzer S, Silahtaroglu A, Meister B. Locked nucleic acid-based in situ hybridisation reveals miR-7a as a hypothalamus-enriched microRNA with a distinct expression pattern. J Neuroendocrinol. 2012; 24:1492–1504.
38. Baudry A, Mouillet-Richard S, Schneider B, Launay JM, Kellermann O. miR-16 targets the serotonin transporter: a new facet for adaptive responses to antidepressants. Science. 2010; 329:1537–1541.
39. Haramati S, Navon I, Issler O, Ezra-Nevo G, Gil S, Zwang R, Hornstein E, Chen A. MicroRNA as repressors of stress-induced anxiety: the case of amygdalar miR-34. J Neurosci. 2011; 31:14191–14203.
40. Lambert TJ, Storm DR, Sullivan JM. MicroRNA132 modulates short-term synaptic plasticity but not basal release probability in hippocampal neurons. PLoS One. 2010; 5:e15182.
41. Magill ST, Cambronne XA, Luikart BW, Lioy DT, Leighton BH, Westbrook GL, Mandel G, Goodman RH. microRNA-132 regulates dendritic growth and arborization of newborn neurons in the adult hippocampus. Proc Natl Acad Sci U S A. 2010; 107:20382–20387.
42. Packer AN, Xing Y, Harper SQ, Jones L, Davidson BL. The bifunctional microRNA miR-9/miR-9* regulates REST and CoREST and is downregulated in Huntington’s disease. J Neurosci. 2008; 28:14341–14346.
43. Wu D, Raafat A, Pak E, Clemens S, Murashov AK. Dicer-microRNA pathway is critical for peripheral nerve regeneration and functional recovery in vivo and regenerative axonogenesis in vitro. Exp Neurol. 2012; 233:555–565.
44. Lopez JP, Lim R, Cruceanu C, Crapper L, Fasano C, Labonte B, Maussion G, Yang JP, Yerko V, Vigneault E, El Mestikawy S, Mechawar N, Pavlidis P, et al. miR-1202 is a primate-specific and brain-enriched microRNA involved in major depression and antidepressant treatment. Nature Medicine. 2014; 20:764–768.
45. Morgan CP, Bale TL. Early prenatal stress epigenetically programs dysmasculinization in second-generation offspring via the paternal lineage. The Journal of neuroscience: the official journal of the Society for Neuroscience. 2011; 31:11748–11755.
46. Rao YS, Mott NN, Wang Y, Chung WC, Pak TR. MicroRNAs in the aging female brain: a putative mechanism for age-specific estrogen effects. Endocrinology. 2013; 154:2795–2806.
47. Toufexis DJ, Myers KM, Bowser ME, Davis M. Estrogen disrupts the inhibition of fear in female rats, possibly through the antagonistic effects of estrogen receptor alpha (ERalpha) and ERbeta. The Journal of neuroscience: the official journal of the Society for Neuroscience. 2007; 27:9729–9735.
48. Miller WJ, Suzuki S, Miller LK, Handa R, Uht RM. Estrogen receptor (ER)beta isoforms rather than ERalpha regulate corticotropin-releasing hormone promoter activity through an alternate pathway. The Journal of neuroscience: the official journal of the Society for Neuroscience. 2004; 24:10628–10635.
49. Paris O, Ferraro L, Grober OM, Ravo M, De Filippo MR, Giurato G, Nassa G, Tarallo R, Cantarella C, Rizzo F, Di Benedetto A, Mottolese M, Benes V, et al. Direct regulation of microRNA biogenesis and expression by estrogen receptor beta in hormone-responsive breast cancer. Oncogene. 2012; 31:4196–4206.
50. Wickramasinghe NS, Manavalan TT, Dougherty SM, Riggs KA, Li Y, Klinge CM. Estradiol downregulates miR-21 expression and increases miR-21 target gene expression in MCF-7 breast cancer cells. Nucleic Acids Res. 2009; 37:2584–2595.
51. Bhat-Nakshatri P, Wang G, Collins NR, Thomson MJ, Geistlinger TR, Carroll JS, Brown M, Hammond S, Srour EF, Liu Y, Nakshatri H. Estradiol-regulated microRNAs control estradiol response in breast cancer cells. Nucleic Acids Res. 2009; 37:4850–4861.
52. Ibanez-Ventoso C, Yang M, Guo S, Robins H, Padgett RW, Driscoll M. Modulated microRNA expression during adult lifespan in Caenorhabditis elegans. Aging Cell. 2006; 5:235–246.
53. Kato M, Chen X, Inukai S, Zhao H, Slack FJ. Age-associated changes in expression of small, noncoding RNAs, including microRNAs, in C. elegans. Rna. 2011; 17:1804–1820.
54. Li N, Bates DJ, An J, Terry DA, Wang E. Up-regulation of key microRNAs, and inverse down-regulation of their predicted oxidative phosphorylation target genes, during aging in mouse brain. Neurobiol Aging. 2011; 32:944–955.
55. Somel M, Guo S, Fu N, Yan Z, Hu HY, Xu Y, Yuan Y, Ning Z, Hu Y, Menzel C, Hu H, Lachmann M, Zeng R, et al. MicroRNA, mRNA, and protein expression link development and aging in human and macaque brain. Genome Res. 2010; 20:1207–1218.
56. Abe M, Naqvi A, Hendriks GJ, Feltzin V, Zhu Y, Grigoriev A, Bonini NM. Impact of age-associated increase in 2′-O-methylation of miRNAs on aging and neurodegeneration in Drosophila. Genes & Development. 2014; 28:44–57.
57. Friedman RC, Farh KK, Burge CB, Bartel DP. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009; 19:92–105.
58. Lee HY, Doudna JA. TRBP alters human precursor microRNA processing in vitro. Rna. 2012; 18:2012–2019.
59. Ferraro L, Ravo M, Nassa G, Tarallo R, De Filippo MR, Giurato G, Cirillo F, Stellato C, Silvestro S, Cantarella C, Rizzo F, Cimino D, Friard O, et al. Effects of oestrogen on microRNA expression in hormone-responsive breast cancer cells. Hormones & cancer. 2012; 3:65–78.
60. Vesely C, Tauber S, Sedlazeck FJ, Tajaddod M, von Haeseler A, Jantsch MF. ADAR2 induces reproducible changes in sequence and abundance of mature microRNAs in the mouse brain. Nucleic acids research. 2014; 42:12155–12168.
61. Meister G, Landthaler M, Patkaniowska A, Dorsett Y, Teng G, Tuschl T. Human Argonaute2 mediates RNA cleavage targeted by miRNAs and siRNAs. Mol Cell. 2004; 15:185–197.
62. Guo L, Lu Z. The fate of miRNA* strand through evolutionary analysis: implication for degradation as merely carrier strand or potential regulatory molecule?. PLoS One. 2010; 5:e11387.
63. Okamura K, Phillips MD, Tyler DM, Duan H, Chou YT, Lai EC. The regulatory activity of microRNA* species has substantial influence on microRNA and 3′ UTR evolution. Nat Struct Mol Biol. 2008; 15:354–363.
64. Yang JS, Phillips MD, Betel D, Mu P, Ventura A, Siepel AC, Chen KC, Lai EC. Widespread regulatory activity of vertebrate microRNA* species. Rna. 2011; 17:312–326.
65. Shen J, Xia W, Khotskaya YB, Huo L, Nakanishi K, Lim SO, Du Y, Wang Y, Chang WC, Chen CH, Hsu JL, Wu Y, Lam YC, et al. EGFR modulates microRNA maturation in response to hypoxia through phosphorylation of AGO2. Nature. 2013; 497:383–387.
66. Chakraborty TR, Hof PR, Ng L, Gore AC. Stereologic analysis of estrogen receptor alpha (ER alpha) expression in rat hypothalamus and its regulation by aging and estrogen. J Comp Neurol. 2003; 466:409–421.
67. Chakraborty TR, Ng L, Gore AC. Age-related changes in estrogen receptor beta in rat hypothalamus: a quantitative analysis. Endocrinology. 2003; 144:4164–4171.
68. Kermath BA, Riha PD, Woller MJ, Wolfe A, Gore AC. Hypothalamic molecular changes underlying natural reproductive senescence in the female rat. Endocrinology. 2014; 155:3597–3609.
69. Mott NN, Pak TR. Characterisation of human oestrogen receptor beta (ERbeta) splice variants in neuronal cells. J Neuroendocrinol. 2012; 24:1311–1321.
70. Pak TR, Chung WC, Roberts JL, Handa RJ. Ligand-independent effects of estrogen receptor beta on mouse gonadotropin-releasing hormone promoter activity. Endocrinology. 2006; 147:1924–1931.
71. Wang JM, Hou X, Adeosun S, Hill R, Henry S, Paul I, Irwin RW, Ou XM, Bigler S, Stockmeier C, Brinton RD, Gomez-Sanchez E. A dominant negative ERbeta splice variant determines the effectiveness of early or late estrogen therapy after ovariectomy in rats. PLoS One. 2012; 7:e33493.
72. Mott NN, Pinceti E, Rao YS, Przybycien-Szymanska MM, Prins SA, Shults CL, Yang X, Glucksman MJ, Roberts JL, Pak TR. Age-dependent Effects of 17beta-estradiol on the dynamics of estrogen receptor beta (ERbeta) protein-protein interactions in the ventral hippocampus. Mol Cell Proteomics. 2014; 13:760–779.
73. Oyola MG, Portillo W, Reyna A, Foradori CD, Kudwa A, Hinds L, Handa RJ, Mani SK. Anxiolytic effects and neuroanatomical targets of estrogen receptor-beta (ERbeta) activation by a selective ERbeta agonist in female mice. Endocrinology. 2012; 153:837–846.
74. Weiser MJ, Wu TJ, Handa RJ. Estrogen receptor-beta agonist diarylpropionitrile: biological activities of R- and S-enantiomers on behavior and hormonal response to stress. Endocrinology. 2009; 150:1817–1825.
75. Pak TR, Lynch GR, Ziegler DM, Lunden JB, Tsai PS. Disruption of pubertal onset by exogenous testosterone and estrogen in two species of rodents. American journal of physiology Endocrinology and metabolism. 2003; 284:E206–212.
76. Tebar M, Ruiz A, Bellido C, Sanchez-Criado JE. Ovary mediates the effects of RU486 given during proestrus on the diestrous secretion of luteinizing hormone in the rat. Biol Reprod. 1996; 54:1266–1270.
77. Schmittgen TD, Jiang J, Liu Q, Yang L. A high-throughput method to monitor the expression of microRNA precursors. Nucleic acids research. 2003; 32.
78. Kinjo ER, Higa GS, de Sousa E, Casado OA, Damico MV, Britto LR, Kihara AH. A possible new mechanism for the control of miRNA expression in neurons. Experimental neurology. 2013; 248:546–558.
79. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001; 25:402–408.
80. Fic E, Kedracka-Krok S, Jankowska U, Pirog A, Dziedzicka-Wasylewska M. Comparison of protein precipitation methods for various rat brain structures prior to proteomic analysis. Electrophoresis. 2010; 31:3573–3579.

