INTRODUCTION
Acute graft-versus-host disease (aGVHD) incidence and severity depend on several known objective risk factors [1, 2], including a wide range of transplant-related immune gene polymorphisms, such as HLA, minor histocompatibility antigen (mHA), Th1-Th2-Th3-cytokine, and Killer cell immunoglobulin-like receptors (KIR) [3–7]. Among these, HLA, an innate “transplantation barrier”, has been universally accepted as the primary factor affecting aGVHD, particularly with the increasing use of HLA allele-mismatched donors [3, 8–10]. This is because HLA mismatching affect aGVHD development owing to their fundamental roles in T cell activation, regulatory T cell inactivation, and the allo- and auto-response induction [3, 8–10]. Furthermore, aGVHD severity is likely related to the mismatched HLA loci, the total number of mismatched HLA-alleles, high-risk HLA-allele mismatched combinations, or even several key amino acid substitutions on specific positions in HLA class I, though there exists considerable controversy over these issues [8, 11–14]. More importantly, complete matching for both HLA class I and II alleles can indeed significantly decrease aGVHD severity, transplant-related mortality (TRM), and graft rejection, even though the actual requirement with regard to HLA compatibility and the relative importance of matching individual HLA alleles in allogeneic hematopoietic stem cell transplantation (allo-HSCT) have not been clearly established [9, 11, 15–18].
The problem exists in that the chance of finding an individual with an identical HLA genotype, either a relative or an unrelated donor, is low for the majority of patients in need of allo-HSCT [9, 11, 19, 20]. Thus, since high-resolution typing has been accepted as a standard confirmatory technique, many transplant centers have begun to use related and unrelated donors having 1 or more mismatched alleles at HLA-A/-B/-DRB1 loci, including HLA-Cw/-DQB1/-DPB1 loci [21–23]. Subjectively speaking, the occurrence of aGVHD in these transplanted patients will undoubtedly be more frequent and more severe. But are not all the cases in the most recent transplant studies including the National Marrow Donor Program (NMDP), Japan Marrow Donor Program (JMDP), Bone Marrow Donor Worldwide (BMDW) and Fred Hutchinson Cancer Research Center (FHCRC), etc., in which these centers used either “subset analysis” or/and “multivariate modeling” to retrospectively evaluate the impact of HLA-allele matching and mismatching on aGVHD incidence and severity, TRM and so on over 10000 transplanted donor-recipient pairs [12–14, 19, 20, 24–26]. These results suggest that allo-HSCT with mismatched HLA alleles, so-called acceptable/permissible/beneficial mismatched alleles/antigens [25, 27, 28], has therapeutic potential; however, there are insufficient experimental and clinical data to support this idea as a clinical standard practice.
Based on the overwhelming evidence that (1) the biological functions of HLA molecules are determined largely by their three-dimensional (3D) structures; and (2) 3D structural differences in peptide-binding and T-cell receptor (TCR) interaction sites can significantly alter the immunogenicity of mismatched HLA molecules, which may be a primary cause of severe aGVHD [29, 30], we report the successful development and application of a new selection/prediction system based on HLA 3D structure modeling (HLA-TDSM) discrepancy for prediction of aGVHD severity and selection of optimal donor-recipient pairs in a prospective and retrospective randomized pilot clinical allo-HSCT study.
RESULTS
The establishment of HLA-TDSM system by the calculation of both coordinate and revised RMSD
We know that not every amino acid residue (AAR) of the HLA class I and II molecules participates in antigen peptide presentation or TCR binding, especially AAR in the random coil region (Figure 1a and 1b); however, these AAR were still involved in the coordinate RMSD calculation (Figures 1 and 2 in the Supplementary Appendix). To avoid such unnecessary deviation, the calculation below was modified and AARs outside of the functional recognition region were excluded. Thus, we calculated coordinate RMSD and revised RMSD values (Figure 1b–1h), respectively, with the following formula:
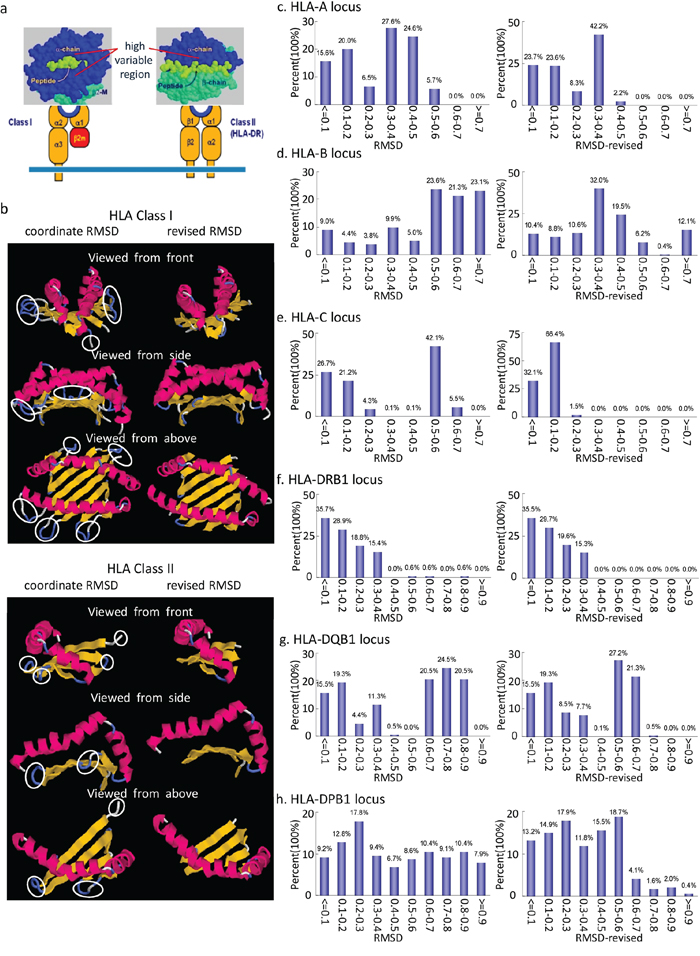
Figure 1: Schematic presentation of the diversity of amino acid residues (AAR) and the distribution of both coordinate RMSD and revised RMSD values at HLA class I and II molecules. a. Allelic polymorphism at HLA-I/II molecules is concentrated in the peptide antigen binding site and TCR interaction AAR. Allelic variants may differ by 20 amino acids (Data from The Major Histocompatibility Complex of Genes by Dr. Colin R.A. Hewitt at [email protected]). b. AAR involved in coordinate RMSD (left) and revised RMSD (right) calculation at HLA class I (upper) and II molecules (bottom) with drawing by RasMol 2.7.5. AARs in white circles are excluded from revised RMSD calculation. The region of AAR used for revised RMSD calculation are aa3–13, aa20–38, aa45–85, aa92–103, aa109–127 and aa132–178 at HLA class I molecules, aa9–18, aa21–30, aa33–39, aa42–47 and aa50–87 at HLA class II molecules. At HLA class I/II molecules, alpha helices are colored magenta, beta sheets are colored yellow, turns are colored pale blue and all other residues are colored white. c–h. show the distribution of both coordinate RMSD and revised RMSD values at HLA-A/-B/-Cw/-DRB1/-DQB1/-DPB1 loci respectively.
The extent of structure discrepancy among all HLA loci, in sequences, is DPB1 ≥ DQB1 > DRB1 ≥ Cw > B > A, according to both coordinate RMSD and revised RMSD values (Figure 1c–1h). The overall structure discrepancy of any 2 alleles from the same allele group is less than that of any 2 alleles from different allele groups, with a few exceptions. All of the revised RMSD values were changed compared to the coordinate RMSD in each locus, most prominently in HLA-Cw locus, except HLA-DRB1 locus (Figure 1c–1h). The total data of the coordinate and revised RMSD values over 6 million records were managed with Microsoft Office Access software, and results between any 2 alleles at HLA-A/-B/-Cw/-DRB1/-DQB1/-DPB1 loci can be queried easily in HLA-TDSM system, also known as HLAStrucMark [31]. With this system, more detailed information on mismatched AAR between any 2 HLA alleles can be shown, such as the mismatched AAR position, the functional characteristics of the position, and anchoring residues contributing to peptide presentation and TCR binding (Figure 1 and 2 in the Supplementary Appendix).
The defined HLA acceptable/unacceptable mismatched alleles based on the revised RMSD
According to the distribution characteristics of both coordinate RMSD and revised RMSD values at HLA-A/-B/-Cw/-DRB1/-DQB1/-DPB1 loci, especially our combined preliminary clinical prediction results, we theoretically determined all HLA acceptable (Table 1) versus unacceptable mismatched alleles (Table 1 in the Supplementary Appendix) at each HLA-A/-B/-Cw/-DRB1/-DQB1/-DPB1 locus. The matched criteria of the revised RMSD value for acceptable mismatched alleles are ≤ 0.2Å for HLA-A*/-B*/-Cw loci and ≤ 0.1Å for HLA-DRB1*/-DQB1/-DPB1 loci. On the basis of these results, we summarized the theoretically acceptable HLA mismatched alleles with a maximum value of single/total revised RMSD < 0.5Å as the basic threshold for optimal selection of recipient-donor pair and for prediction of aGVHD severity before allo-HSCT.
Table 1: The defined HLA acceptable/permissible mismatching allele pairs based on revised RMSD in HLA-A*/-B*/-DRB1* loci respectively
Allele |
HLA permissible mismatching allele pairs among different allele groups |
|---|---|
HLA-A locus |
|
A*01:01 |
A*01:02-, A*03:01-, A*11:01-, A*24:17, A*24:19, A*24:41, A*25:01-, A*26:01-, A*29:01-, A*30:01-, A*31:01-, A*32:01-, A*33:01-, A*34:01-, A*36:01-, A*43:01-, A*66:01-, A*68:01, A*68:08-14, A*68:16, A*68:19, A*68:21, A*68:22, A*68:24-26, A*74:01-, A*80:01- |
A*02:01 |
A*02:02-(except A*02:08), A*23:10, A*23:12, A*24:02-05, A*24:08-10, A*24:13, A*24:14, A*24:18, A*24:20, A*24:21, A*24:23, A*24:25-35, A*24:37-39, A*24:42-44, A*24:47, A*24:49, A*24:50, A*69:01 |
A*03:01 |
A*03:02-, A*11:01-, A*25:01-, A*26:01-, A*29:01-, A*30:01-, A*31:01-, A*32:01-, A*33:01-, A*34:01-, A*36:01-, A*43:01, A*66:01-, A*68:01-, A*74:01-, A*80:01- |
A*11:01 |
A*11:02-, A*24:17, A*24:19, A*24:41, A*25:01-, A*26:01-, A*29:01-, A*30:01-, A*31:01-, A*32:01-, A*33:01-, A*34:01-, A*36:01-, A*43:01, A*66:01-, A*68:01, A*68:03-15, A*68:16-22, A*68:24-26, A*74:01-, A*80:01- |
A*23:01 |
A*23:02-, A*24:02-05, A*24:08-15, A*24:18, A*24:20-21, A*24:23, A*24:25-39, A*24:42-44, A*24:47-50, A*69:01 |
A*24:02 |
A*24:03-05, A*24:08-15, A*24:18, A*24:20-21, A*24:23, A*24:25-39, A*24:42-44, A*24:47-50, A*69:01 |
A*25:01 |
A*25:02-, A*26:01-, A*29:01-, A*30:01-, A*31:01-, A*32:01-, A*33:01-, A*34:01-, A*36:01-, A*43:01, A*66:01-, A*68:01, A*68:03-14, A*68:16-22, A*68:24-26, A*74:01-, A*80:01- |
A*26:01 |
A*26:02-, A*29:01-, A*30:01-, A*31:01-, A*32:01-, A*33:01-, A*34:01-, A*36:01-, A*43:01, A*66:01-, A*68:01, A*68:03-14, A*68:16-22, A*68:24-26, A*74:01-, A*80:01- |
A*29:01 |
A*29:02-, A*30:01-, A*31:01-, A*32:01-, A*33:01-, A*34:01-, A*36:01-, A*43:01, A*66:01-, A*68:01-14, A*68:16-27, A*74:01, A*80:01- |
A*30:01 |
A*30:02-, A*31:01-, A*32:01-, A*33:01-, A*34:01-, A*36:01-, A*43:01, A*66:01-, A*68:01, A*68:08-14, A*68:16, A*68:19, A*68:21-22, A*68:24-26, A*74:01, A*80:01- |
A*31:01 |
A*31:02-, A*32:01-, A*33:01-, A*34:01-, A*36:01-, A*43:01, A*66:01-, A*68:01-14, A*68:16-27, A*74:01, A*80:01- |
A*32:01 |
A*32:02-, A*33:01-, A*34:01-, A*36:01-, A*43:01, A*66:01-, A*68:01, A*68:03-05, A*68:08-14, A*68:16, A*68:19-22, A*68:24-26, A*74:01-, A*80:01- |
A*33:01 |
A*33:02-, A*34:01-, A*36:01-, A*43:01, A*66:01-, A*68:01, A*68:01-14, A*68:16-27, A*74:01, A*80:01- |
A*34:01 |
A*34:02-, A*36:01-, A*43:01, A*66:01-, A*68:01, A*68:08-14, A*68:16, A*68:19, A*68:21, A*68:22, A*68:24-26, A*74:01, A*80:01- |
A*36:01 |
A*36:02-, A*43:01, A*66:01-, A*68:01, A*68:08-14, A*68:16, A*68:19, A*68:21, A*68:22, A*68:24-26, A*74:01, A*80:01- |
A*43:01 |
A*66:01-, A*68:01, A*68:03-14, A*68:16-22, A*68:24-26, A*74:01, A*80:01- |
A*66:01 |
A*66:02-, A*68:01, A*68:03-14, A*68:16-22, A*68:24-26, A*74:01, A*80:01- |
A*68:01 |
A*68:02-14, A*68:16-27, A*74:01-03, A*74:05-, A*80:01- |
A*74:01 |
A*74:02-, A*80:01- |
HLA-B locus |
|
B*07:02 |
B*07:04, B*07:07, B*07:08, B*07:10, B*07:18, B*07:19, B*07:21-23, B*07:30, B*07:35, B*07:39, B*08:02, B*08:06, B*08:09, B*08:11, B*08:15, B*08:20, B*37:07, B*39:03, B*39:10, B*39:16, B*39:24, B*39:27, B*39:29, B*40:25, B*40:43, B*48:12, B*55:04, B*56:13, B*67:01, B*82:01, B*82:02, B*83:01 |
B*08:01 |
B*08:04, B*08:05, B*08:07, B*08:10, B*08:12-14, B*08:16-18, B*08:21-24, B*14:01-06, B*15:83, B*37:09, B*39:01, B*39:02, B*39:04, B*39:06, B*39:09, B*39:12, B*39:14, B*39:15, B*39:17, B*39:18, B*39:22, B*39:23, B*39:26, B*39:28, B*39:30, B*39:31, B*39:33, B*40:01, B*40:02, B*40:05-12, B*40:14-16, B*40:21, B*40:23, B*40:29-34, B*40:36-42, B*40:45, B*40:46, B*40:48-51, B*40:53-57, B*41:01-07, B*42:01-06, B*45:06, B*48:01, B*48:03-11, B*54:02, B*54:04, B*55:01-03, B*55:05, B*55:08-10, B*55:12, B*55:13, B*55:15-17, B*55:19, B*56:05, B*81:01, B*81:02 |
B*13:01 |
B*13:02-08, B*13:10-13, B*15:13, B*15:16, B*15:17, B*15:24, B*15:36, B*15:67, B*15:87, B*15:95, B*37:08, B*38:04, B*38:10, B*40:19, B*40:47, B*44:02-41, B*45:01-05, B*45:07, B*47:01, B*47:03, B*47:04, B*49:01-03, B*50:02, B*51:20, B*51:36, B*52:01, B*52:02, B*52:04, B*52:05, B*57:02, B*57:04, B*57:05, B*57:07, B*57:09, B*58:07, B*58:08 |
B*14:01 |
B*14:02-, B*37:05, B*37:09, B*39:01, B*39:02, B*39:04, B*39:06, B*39:09, B*39:12, B*39:14, B*39:15, B*39:17, B*39:18, B*39:22, B*39:23, B*39:26, B*39:28, B*39:30, B*39:31, B*39:33, B*40:01, B*40:02, B*40:05-12, B*40:14-16, B*40:21, B*40:23, B*40:29-34, B*40:36-42, B*40:45, B*40:46, B*40:48-51, B*40:53-57, B*41:01-07, B*42:01-, B*45:06, B*48:01, B*48:03-11, B*54:02, B*54:04, B*55:01-03, B*55:05, B*55:08-10, B*55:12, B*55:13, B*55:15-17, B*55:19, B*56:05, B*81:01, B*81:02 |
B*15:01 |
B*15:02-12, B*15:14, B*15:15, B*15:19, B*15:21, B*15:25, B*15:27, B*15:28, B*15:31-35, B*15:38-50, B*15:53-61, B*15:63-66, B*15:68-71, B*15:73-78, B*15:81, B*15:82, B*15:84-86, B*15:90-92, B*15:96-98, B*18:01, B*18:08, B*18:10, B*18:11, B*18:15, B*18:18, B*18:20, B*35:08, B*35:13, B*35:18, B*35:28, B*35:38, B*35:46, B*35:58, B*39:08, B*39:13, B*40:03, B*40:20, B*40:44, B*40:52, B*46:01-04, B*48:02, B*50:01, B*50:04, B*53:01, B*53:02, B*53:09, B*53:10, B*55:14, B*56:01, B*83:01 |
B*18:01 |
B*18:02, B*18:04, B*18:06-08, B*18:10-18, B*18:20, B*35:01-04, B*35:06-08, B*35:12-16, B*35:18-20, B*35:22, B*35:23, B*35:25, B*35:26, B*35:28, B*35:29, B*35:31-42, B*35:45-50, B*35:54-58, B*39:08, B*39:13, B*39:19, B*39:32, B*40:03, B*40:04, B*40:20, B*40:24, B*40:26, B*40:28, B*40:52, B*48:02, B*50:01, B*50:04, B*53:01-03, B*53:05, B*53:09, B*53:10, B*54:01, B*54:03, B*54:06, B*55:07, B*55:11, B*55:14, B*55:18, B*56:01-04, B*56:06, B*56:09, B*56:10, B*56:12, B*56:14, B*56:15, B*57:08, B*58:01, B*58:04, B*58:05, B*58:09, B*78:02, B*78:04, B*83:01 |
B*27:01 |
B*27:02, B*27:03, B*27:05-13, B*27:15-28, B*37:02, B*37:06, B*38:03, B*47:05, B*73:01 |
B*35:01 |
B*35:02-04, B*35:06-08, B*35:12-16, B*35:18-20, B*35:22, B*35:23, B*35:25, B*35:26, B*35:28, B*35:29, B*35:31-42, B*35:45-50, B*35:54-58, B*39:08, B*39:19, B*39:32, B*40:04, B*40:20, B*40:24, B*40:26, B*40:28, B*48:02, B*50:04, B*53:01-03, B*53:05, B*53:09, B*53:10, B*54:01, B*54:03, B*54:06, B*55:07, B*55:11, B*55:14, B*55:18, B*56:01-04, B*56:06, B*56:09, B*56:10, B*56:12-15, B*57:08, B*58:01, B*58:04, B*58:05, B*58:09, B*78:04, B*83:01, |
B*37:01 |
B*37:04, B*37:06, B*37:08, B*38:03, B*40:13, B*40:19, B*44:06, B*47:01, B*47:03, B*47:04, B*73:01 |
B*38:01 |
B*38:02, B*38:04-10, B*49:01, B*49:03, B*51:01-37, B*52:01-06, B*53:01-03, B*53:05-10, B*56:07, B*57:01-03, B*57:05-07, B*57:09, B*58:01-09, B*59:01, B*78:02, B*78:03, B*78:05 |
B*39:01 |
B*39:02, B*39:04, B*39:06, B*39:09, B*39:12, B*39:14, B*39:15, B*39:17, B*39:22, B*39:23, B*39:26, B*39:28, B*39:30, B*39:31, B*39:33, B*40:01, B*40:02, B*40:05-12, B*40:14-16, B*40:21, B*40:23, B*40:29-34, B*40:36-42, B*40:45, B*40:46, B*40:48-51, B*40:53-57, B*41:01-07, B*42:01-06, B*45:06, B*48:01, B*48:03-11, B*54:02, B*54:04, B*55:01-03, B*55:05, B*55:08-10, B*55:12, B*55:13, B*55:15-17, B*55:19, B*56:05, B*81:01, B*81:02 |
B*40:01 |
B*40:02, B*40:05-12, B*40:14-16, B*40:21, B*40:23, B*40:29-34, B*40:36-42, B*40:45, B*40:46, B*40:48-51, B*40:53-57, B*41:01-07, B*42:01-06, B*45:06, B*48:01, B*48:03-11, B*54:02, B*54:04, B*55:01-03, B*55:05, B*55:08-10, B*55:12, B*55:13, B*55:15-17, B*55:19, B*56:05, B*81:01, B*81:02 |
B*41:01 |
B*41:02-07, B*42:01-06, B*45:06, B*48:01, B*48:03-11, B*54:02, B*54:04, B*55:01-03, B*55:05, B*55:08-10, B*55:12, B*55:13, B*55:15-17, B*55:19, B*56:05, B*81:01, B*81:02 |
B*42:01 |
42:02-06, B*45:06, B*48:01, B*48:03-11, B*54:02, B*54:04, B*55:01-03, B*55:05, B*55:08-10, B*55:12, B*55:13, B*55:15-17, B*55:19, B*56:05, B*81:01, B*81:02 |
B*44:02 |
B*44:03-41, B*45:01-05, B*45:07, B*47:01, B*47:03, B*47:04, B*49:01, B*49:02, B*50:02, B*52:02, B*57:02, B*57:04, B*57:05, B*57:07, B*58:07 |
B*45:01 |
B*45:02-05, B*45:07, B*47:02-04, B*49:01, B*49:02, B*50:02, B*57:04, |
B*46:01 |
B*46:02-04, B*50:01, B*50:04 |
B*47:01 |
B*47:03-05, B*49:02, B*57:04, B*73:01 |
B*48:01 |
B*48:03-11, B*54:02, B*54:04, B*55:01-03, B*55:05, B*55:08-10, B*55:12, B*55:13, B*55:15-17, B*55:19, B*56:05, B*81:01, B*81:02 |
B*49:01 |
B*49:02, B*49:03, B*51:03, B*51:20, B*51:23, B*51:31, B*51:36, B*52:01, B*52:02, B*52:04, B*52:05, B*57:02, B*57:04, B*57:05, B*57:07, B*57:09, B*58:07, B*58:08, B*59:01 |
B*50:01 |
B*50:04, B*53:01, B*53:02, B*53:09, B*53:10, B*55:14, B*56:01, B*83:01 |
B*51:01 |
B*51:02-37, B*52:01-06, B*53:01-03, B*53:05-10, B*56:06, B*56:07, B*57:01-03, B*57:05-09, B*58:01-09, B*59:01, B*78:01-03, B*78:05 |
B*52:01 |
B*52:02-06, B*53:06, B*53:07, B*56:07, B*57:01-07, B*57:09, B*58:02, B*58:06-08, B*59:01, B*78:05 |
B*53:01 |
B*53:02, B*53:03, B*53:05-10, B*54:01, B*54:03, B*54:06, B*55:07, B*55:14, B*55:18, B*56:01-04, B*56:06, B*56:07, B*56:09, B*56:10, B*56:13-15, B*57:01, B*57:03, B*57:06, B*57:08, B*58:01-06, B*58:09, B*59:01, B*78:01-05, B*83:01 |
B*54:01 |
B*54:03, B*54:06, B*55:07, B*55:11, B*55:14, B*55:18, B*56:01-04, B*56:06, B*56:09, B*56:10, B*56:12, B*56:14, B*56:15, B*57:08, B*58:01, B*58:04, B*58:05, B*58:09, B*78:01-04, B*83:01 |
B*55:01 |
B*55:02, B*55:03, B*55:05, B*55:08-10, B*55:12, B*55:13, B*55:15-17, B*55:19, B*56:05, B*81:01, B*81:02 |
B*56:01 |
B*56:02-04, B*56:06, B*56:09, B*56:10, B*56:12, B*56:14, B*56:15, B*57:08, B*58:01, B*58:04, B*58:05, B*58:09, B*78:02, B*78:04, B*83:01 |
B*57:01 |
B*57:02, B*57:03, B*57:05-09, B*58:01-09, B*59:01, B*78:01-05 |
B*58:01 |
B*58:02-06, B*58:09, B*59:01, B*78:01-05, B*83:01 |
B*59:01 |
B*78:03, B*78:05 |
B*67:01 |
B*82:01, B*82:02 |
B*73:01 |
nothing |
B*78:01 |
B*78:02-04 |
B*81:01 |
B*81:02 |
B*82:01 |
B*82:02 |
B*83:01 |
nothing |
HLA-DRB1 locus |
|
DRB1*01:01 |
DRB1*01:03-05, DRB1*01:07, DRB1*01:12, DRB1*07:06, DRB1*10:01 |
DRB1*03:01 |
DRB1*03:02, DRB1*03:03, DRB1*03:05-18, DRB1*03:20-28, DRB1*11:17, DRB1*11:20, DRB1*11:25, DRB1*11:46, DRB1*11:52, DRB1*11:54, DRB1*13:02, DRB1*13:08, DRB1*13:16, DRB1*13:19, DRB1*13:29, DRB1*13:31, DRB1*13:34, DRB1*13:36, DRB1*13:39-41, DRB1*13:52, DRB1*13:53, DRB1*13:56, DRB1*13:57, DRB1*13:63-65, DRB1*14:01-06, DRB1*14:08, DRB1*14:09, DRB1*14:11, DRB1*14:13, DRB1*14:15-21, DRB1*14:23, DRB1*14:24, DRB1*14:26, DRB1*14:28-35, DRB1*14:37-39, DRB1*14:43, DRB1*14:45, DRB1*14:47, DRB1*14:48 |
DRB1*04:01 |
DRB1*04:02-19, DRB1*04:21-53, DRB1*08:01-15, DRB1*08:17-30, DRB1*11:01-08, DRB1*11:10-14, DRB1*11:18, DRB1*11:19, DRB1*11:21-23, DRB1*11:26, DRB1*11:27, DRB1*11:29-39, DRB1*11:41-45, DRB1*11:48-51, DRB1*11:53, DRB1*13:03, DRB1*13:04, DRB1*13:07, DRB1*13:11-14, DRB1*13:17, DRB1*13:21-25, DRB1*13:30, DRB1*13:32, DRB1*13:33, DRB1*13:37, DRB1*13:38, DRB1*13:44-49, DRB1*13:54, DRB1*13:55, DRB1*13:58, DRB1*13:60, DRB1*13:66, DRB1*14:07, DRB1*14:10, DRB1*14:14, DRB1*14:22, DRB1*14:25, DRB1*14:36, DRB1*14:40-42, DRB1*14:44, DRB1*14:46 |
DRB1*07:01 |
DRB1*07:03, DRB1*07:05, DRB1*07:06, DRB1*07:09, DRB1*10:01 |
DRB1*08:01 |
DRB1*08:02-15, DRB1*08:17-30, DRB1*11:01-08, DRB1*11:10-14, DRB1*11:18, DRB1*11:19, DRB1*11:21-23, DRB1*11:26, DRB1*11:27, DRB1*11:29-39, DRB1*11:41-45, DRB1*11:48-51, DRB1*11:53, DRB1*13:03, DRB1*13:04, DRB1*13:07, DRB1*13:11-14, DRB1*13:17, DRB1*13:21-25, DRB1*13:30, DRB1*13:32, DRB1*13:33, DRB1*13:37, DRB1*13:38, DRB1*13:44-49, DRB1*13:54, DRB1*13:55, DRB1*13:58, DRB1*13:60, DRB1*13:66, DRB1*14:07, DRB1*14:10, DRB1*14:14, DRB1*14:22, DRB1*14:25, DRB1*14:36, DRB1*14:40-42, DRB1*14:44, DRB1*14:46 |
DRB1*09:01 |
DRB1*09:02-04, DRB1*11:15, DRB1*13:62 |
DRB1*10:01 |
nothing |
DRB1*11:01 |
DRB1*11:02-08, DRB1*11:10-14, DRB1*11:18, DRB1*11:19, DRB1*11:21-23, DRB1*11:25-27, DRB1*11:29-39, DRB1*11:41-46, DRB1*11:48-51, DRB1*11:53, DRB1*13:03, DRB1*13:04, DRB1*13:07, DRB1*13:08, DRB1*13:11-14, DRB1*13:17, DRB1*13:19, DRB1*13:21-25, DRB1*13:30, DRB1*13:32, DRB1*13:33, DRB1*13:37, DRB1*13:38, DRB1*13:44-49, DRB1*13:52-55, DRB1*13:58, DRB1*13:60, DRB1*13:63, DRB1*13:66, DRB1*14:02, DRB1*14:07, DRB1*14:09, DRB1*14:10, DRB1*14:13-15, DRB1*14:22, DRB1*14:25, DRB1*14:28, DRB1*14:32, DRB1*14:36, DRB1*14:37, DRB1*14:40-42, DRB1*14:44, DRB1*14:46 |
DRB1*12:01 |
DRB1*12:03-12, DRB1*14:27 |
DRB1*13:01 |
DRB1*13:05, DRB1*13:06, DRB1*13:09, DRB1*13:10, DRB1*13:15, DRB1*13:18, DRB1*13:20, DRB1*13:27, DRB1*13:28, DRB1*13:35, DRB1*13:42, DRB1*13:51, DRB1*13:59 |
DRB1*14:01 |
DRB1*14:02-09, DRB1*14:11, DRB1*14:13, DRB1*14:15, DRB1*14:16, DRB1*14:18-21, DRB1*14:23, DRB1*14:24, DRB1*14:26, DRB1*14:28-35, DRB1*14:37-40, DRB1*14:42, DRB1*14:43, DRB1*14:45, DRB1*14:47, DRB1*14:48 |
DRB1*15:01 |
DRB1*15:02, DRB1*16:01-03 |
DRB1*16:01 |
DRB1*16:02, DRB1*16:03 |
HLA-A*/-B*/-DRB1* loci include at least 2132, 2798 and 1196 alleles respectively, all of whom are the most polymorphic in all HLA loci. For these reasons, we selected HLA-A*/-B*/-DRB1* loci as the representative of acceptable/permissible mismatching allele pairs from all HLA loci. The matched criteria of the revised RMSD value are ≤ 0.2Å for HLA-A*/-B*loci and ≤ 0.1Å for HLA-DRB1*locus. The limited layout sake this table shows only partial results. The dash “-“indicates all the other alleles in this allele group.
Evaluation of accuracy and reliability of HLA-TDSM system by antigen-specific alloreactive CTLs
FACS analysis and TCR Vβ gene scan showed that HLA-B molecules pulsed with Epstein-Barr virus nonameric peptide (EBNA3) could induce clonal CTL activation in stable transfected Hmy2.CIR cells expressing HLA-B*15:02, -B*15:18, -B*35:03, or -B*44:03 (data not shown), and exhibited various cross-reaction characteristics and HLA restriction (Tables 2–4 and Figures 3–4 in the Supplementary Appendix). The revised RMSD between B*44:03 and HLA-B*15:02,-B*15:18, or -B*35:03 were much higher than those within HLA-B*15:02, -B*15:18, and -B*35:03 in which there are many AAR discrepancies in the position of F epitope-binding pocket. These results confirmed that cross-reaction patterns by antigen-specific alloreactive CTLs could be used efficiently to evaluate the accuracy and reliability of the HLA-TDSM system. With these available data, we further verified the accuracy and reliability of the HLA-TDSM system.
A close correlation between the revised RMSD and aGVHD severity in retrospectively randomized allo-HSCT
In the retrospective study, we randomly collected and analyzed the correlation between the revised RMSD and aGVHD severity in 12 transplanted recipient-donor pairs. As shown in Tables 2 and 3, of 12 recipient-donor pairs, there were 6 recipient-donor pairs who had 1 allele mismatching at HLA-A, -B, -DRB1, or -DQB1 locus, 3 recipient-donor pairs who had 2 alleles mismatching at HLA-A+DRB1, -B+DQB1, or -DRB1+DQB1 loci; and 3 recipient-donor pairs who had 3 alleles mismatching at either HLA-A+B+DRB1 or HLA-B+DRB1+DQB1 loci. Our results indicate that the occurrence of severe aGVHD in recipient-donor pairs with either 1 or 2 alleles mismatching at any locus had a close correlation with single or total revised RMSD > 0.50Å, but individual HLA-allele, allele group, loci, or the combination of different allele/loci did not. Severe aGVHD did not occur even if recipient-donor pair had 3 alleles mismatching, as long as total revised RMSD < 0.50Å. In contrast, severe aGVHD still occurred even if recipient-donor pair had HLA-allele mismatching at the same allele group as long as single or total revised RMSD > 0.50Å. The 1 exception is R-UPN07 recipient-donor pair, who had only 1 allele mismatching between HLA-A*02:01 and -A*68:01 and single revised RMSD 0.43Å < 0.50Å, but still developed grade IV severe aGVHD. We considered that R-UPN07 was a special case because she was confirmed to have 1 HLA-A/B loci recombination, which produces a novel haplotype that may have caused the severe aGVHD [31]. Several representative images comparing the results of 3D structure modeling for 1 allele mismatching at HLA-A/-B/-DRB1/-DQB1 loci from either retrospective or prospective recipient-donor pairs are shown using RASMOL software (Figure 5 in the Supplementary Appendix).
The revised RMSD as a reliable algorithm for prospective prediction of aGVHD severity before allo-HSCT
Of the 20 evaluable recipient-donor pairs in the prospective study, there were 6 recipient-donor pairs who had 1 allele mismatching at either HLA-A or -B loci, 10 recipient-donor pairs who had 2 alleles mismatching at either HLA-A+B, -B+DRB1 or -DRB1+DQB1 loci, 2 recipient-donor pairs who had 3 alleles mismatching at HLA-A+B+DRB1 loci, and 2 recipient-donor pairs who had an HLA-haploidentical mismatching (Tables 2 and 3).
Table 2: Demographic characteristics and disease status of patient-donor pairs in retrospective and prospective groups
UPN+ |
Patients |
Donors |
||||
|---|---|---|---|---|---|---|
Age/Sex |
Disease status |
Transplant status |
HLA-A/B/DRB1/DQB1 low-resolution |
Donor/Age |
HLA-A/B/DRB1/DQB1 low-resolution |
|
R-UPN01 |
12/male |
ALL/RE3 |
Yes |
A*02/11, B*13/60, DRB1*12/15, DQB1*06/07 |
Father/40 |
A*02/33, B*44/60, DRB1*12/13, DQB1*06/07 |
R-UPN02 |
10/male |
ALL/CR2 |
Yes |
A*11/24, B*60/62, DRB1*11/15, DQB1*05/07 |
Mother/32 |
A*11/11, B*46/62, DRB1*12/15, DQB1*05/07 |
R-UPN03 |
15/male |
AML/CR1 |
Yes |
A*02/11, B*60/60, DRB1*09/11, DQB1*07/09 |
Sister/11 |
A*11/11, B*46/60, DRB1*09/11, DQB1*07/09 |
R-UPN04 |
26/female |
AML/CR1 |
Yes |
A*01/02, B*13/37, DRB1*10/16, DQB1*05/05 |
Brother/35 |
A*01/33, B*13/37, DRB1*10/16, DQB1*05/05 |
R-UPN05 |
36/male |
AML/CR1 |
Yes |
A*11/30, B*13/62, DRB1*13/13, DQB1*06/06 |
Sister/37 |
A*11/30, B*13/44, DRB1*04/13, DQB1*06/08 |
R-UPN06 |
21/female |
AML/RE1 |
Yes |
A*02/33, B*07/60, DRB1*04/04, DQB1*02/07 |
Unrelated BM |
A*02/33, B*07/60, DRB1*04/04, DQB1*02/07 |
R-UPN07 |
30/female |
AML/CR1 |
Yes |
A*02/203, B*38/52, DRB1*04/07, DQB1*02/04 |
Brother/38 |
A*203/68, B*38/52, DRB1*04/07, DQB1*02/04 |
R-UPN08 |
45/female |
AML/CR1 |
Yes |
A*02/30, B*38/60, DRB1*11/15, DQB1*06/07 |
Brother/38 |
A*02/30, B*39/60, DRB1*11/15, DQB1*06/07 |
R-UPN09 |
22/male |
AML/CR1 |
Yes |
A*02/24, B*35/61, DRB1*08/15, DQB1*06/06 |
Father/52 |
A*02/02, B*35/61, DRB1*15/16, DQB1*05/06 |
R-UPN10 |
42/male |
CML/CP |
Yes |
A*02/11, B*07/62, DRB1*01/15, DQB1*05/08 |
Brother/30 |
A*02/11, B*07/62, DRB1*01/15, DQB1*05/06 |
R-UPN11 |
34/male |
CML/CP |
Yes |
A*11/33, B*58/61, DRB1*12/16, DQB1*05/07 |
Brother/27 |
A*11/33, B*13/58, DRB1*12/16, DQB1*05/07 |
R-UPN12 |
36/male |
NHL/CR1 |
Yes |
A*02/30, B*13/55, DRB1*07/12, DQB1*02/07 |
Brother/42 |
A*02/30, B*13/55, DRB1*07/11, DQB1*02/08 |
P-UPN01 |
17/male |
ALL/CR2 |
Yes |
A*11/31, B*46/48, DRB1*08/14, DQB1*05/06 |
Father/43 |
A*02/11, B*46/51, DRB1*08/12, DQB1*06/07 |
P-UPN02 |
32/male |
ALL/CR1 |
Yes |
A*24/33, B*35/55, DRB1*04/12, DQB1*02/05 |
Brother/30 |
A*02/33, B*35/55, DRB1*04/12, DQB1*02/05 |
P-UPN03 |
9/male |
ALL/CR1 |
Yes |
A*24/33, B*44/62, DRB1*11/14, DQB1*06/07 |
Unrelated CB |
A*11/24, B*44/62, DRB1*04/11, DQB1*06/07 |
P-UPN04 |
24/female |
ALL/CR1 |
Yes |
A*02/33, B*51/60, DRB1*04/15, DQB1*06/08 |
Brother/23 |
A*02/03, B*35/60, DRB1*04/15, DQB1*06/08 |
P-UPN05 |
24/female |
AML/CR1 |
Yes |
A*02/31, B*46/51, DRB1*12/15, DQB1*05/07 |
Father/50 |
A*02/31, B*38/51, DRB1*12/16, DQB1*05/07 |
P-UPN06 |
39/female |
AML/CR1 |
Yes |
A*11/11, B*18/60, DRB1*09/14, DQB1*04/08 |
Sister/37 |
A*11/24, B*35/60, DRB1*04/09, DQB1*04/08 |
P-UPN07 |
18/male |
AML/CR1 |
Yes |
A*02/02, B*13/61, DRB1*08/12, DQB1*05/07 |
Unrelated CB |
A*02/203, B*13/60, DRB1*08/12, DQB1*05/07 |
P-UPN08 |
52/female |
AML/CR2 |
No |
A*11/30, B*13/38, DRB1*07/12, DQB1*02/07 |
Brother/47 |
A*11/30, B*13/27, DRB1*07/12, DQB1*02/07 |
P-UPN09 |
20/female |
AML/CR1 |
No |
A*203/11, B*38/60, DRB1*04/15, DQB1*06/07 |
Unrelated CB |
A*11/24, B*38/60, DRB1*04/15, DQB1*06/07 |
P-UPN10 |
45/female |
AML/CR1 |
Yes |
A*02/24, B*27/44, DRB1*01/11, DQB1*05/07 |
Brother/32 |
A*02/02, B*44/44, DRB1*01/11, DQB1*05/07 |
P-UPN11 |
29/male |
CML/CP |
Yes |
A*02/11, B*13/62, DRB1*11/11, DQB1*07/07 |
Father/55 |
A*11/30, B*13/13, DRB1*11/11, DQB1*07/07 |
P-UPN12 |
33/female |
CML/CP |
Yes |
A*11/24, B*51/60, DRB1*04/15, DQB1*06/08 |
Brother/35 |
A*02/24, B*51/60, DRB1*04/15, DQB1*06/08 |
P-UPN13 |
35/female |
CML/CP |
Yes |
A*02/26, B*51/62, DRB1*09/13, DQB1*06/09 |
Sister/30 |
A*02/30, B*13/62, DRB1*07/09, DQB1*02/09 |
P-UPN14 |
37/male |
CML/CP |
Yes |
A*01/11, B*37/54, DRB1*08/10, DQB1*05/06 |
Brother/35 |
A*01/26, B*37/54, DRB1*08/10, DQB1*05/06 |
P-UPN15 |
28/male |
CML/CP |
Yes |
A*11/24, B*37/60, DRB1*04/10, DQB1*02/05 |
Sister/30 |
A*11/24, B*46/60, DRB1*04/09, DQB1*02/05 |
P-UPN16 |
25/male |
CML/CP |
Yes |
A*02/02, B*54/60, DRB1*08/11, DQB1*06/07 |
Sister/22 |
A*02/02, B*46/60, DRB1*08/11, DQB1*06/07 |
P-UPN17 |
40/male |
CML/CP |
Yes |
A*11/31, B*35/61, DRB1*11/13, DQB1*07/07 |
Son/14 |
A*02/31, B*13/61, DRB1*11/13, DQB1*06/07 |
P-UPN18 |
30/male |
CML/CP |
Yes |
A*11/11, B*35/71, DRB1*15/17, DQB1*02/06 |
Brother/39 |
A*01/11, B*35/57, DRB1*07/15, DQB1*02/06 |
P-UPN19 |
28/male |
CML/CP |
Yes |
A*02/02, B*13/46, DRB1*04/09, DQB1*08/09 |
Brother/24 |
A*02/02, B*13/46, DRB1*04/15, DQB1*05/08 |
P-UPN20 |
24/male |
CML/CP |
Yes |
A*30/31, B*35/61, DRB1*09/12, DQB1*07/09 |
Mother/47 |
A*24/30, B*35/51, DRB1*09/12, DQB1*07/09 |
P-UPN21 |
14/male |
CML/CP |
Yes |
A*02/24, B*39/46, DRB1*09/12, DQB1*07/09 |
Mother/38 |
A*02/02, B*39/46, DRB1*09/12, DQB1*07/09 |
P-UPN22 |
13/female |
MDS- |
Yes |
A*02/02, B*07/27, DRB1*15/17, DQB1*02/06 |
Father/35 |
A*02/30, B*13/27, DRB1*07/17, DQB1*02/06 |
P-UPN23 |
15/male |
Thal |
No |
A*02/11, B*54/60, DRB1*04/04, DQB1*04/07 |
Father/40 |
A*02/11, B*54/60, DRB1*04/14, DQB1*05/07 |
P-UPN24 |
03/male |
Thal |
No |
A*11/11, B*13/54, DRB1*08/14, DQB1*05/06 |
Mother/38 |
A*11/11, B*54/75, DRB1*08/14, DQB1*05/06 |
P-UPN25 |
5/male |
Thal |
No |
A*11/11, B*51/75, DRB1*11/12, DQB1*06/07 |
Sister CB |
A*11/11, B*51/55, DRB1*11/14, DQB1*06/07 |
R-UPN, retrospective unique patient number; P-UPN, prospective unique patient number; ALL, acute lymphoblastic leukemia; AML, acute myelocytic leukemia; CML, chronic myelocytic leukemia; MDS-RAEB-t, myelodysplastic syndrome-refractory anemia with excess blasts in transformation; NHL, Non-Hodgkin lymphoma; Thal, thalassemia; CR, complete remission; RE, relapse; CP, chronic phase; CB, cord blood.
Table 3: The correlated results of among HLA-A/-B/-DRB1/-DQB1 high-resolution, HLA 3D structure discrepancy and aGVHD severity in 32 patient-donor pairs
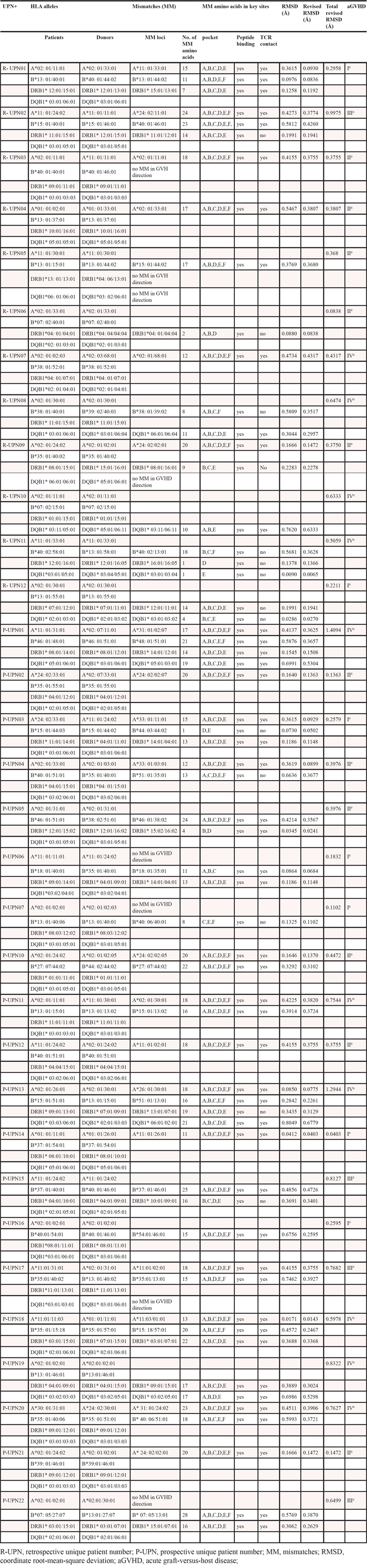
All of 20 recipients who entered this study underwent either related allo-HSCT or unrelated CB transplantation. As shown in Table 3, there was a strong correlation between single or total revised RMSD and the occurrence of severe aGVHD. The 6 recipient-donor pairs with 1 allele mismatching at either HLA-A or -B locus had a lower single revised RMSD (less than 0.50Å before allo-HSCT) and only I − II0 aGVHD occurred in these recipients after transplantation. All of recipient-donor pairs who developed severe aGVHD had a higher single or total revised RMSD (>0.50Å) before allo-HSCT, but not relevant to individual HLA-allele, allele group, loci, especially fixed combinations of different allele/loci. In addition, 2 recipient-donor pairs, P-UNP-01 and P-UNP-3, with an HLA-haploidentical mismatching had the highest total revised RMSD (1.4094Å and 1.2944Å) before allo-HSCT, and developed severe IV0 aGVHD after transplantation (Tables 2 and 3).
The revised RMSD as a reliable algorithm for optimal donor selection
We further studied how to select optimal donor by HLA-TDSM system for 5 patients who had 2 − 6 alternative potential donors (Table 2 and 4). As shown in Table 4, P-UPN03 had 6 alternative potential cord blood donors with different 2 or 3 alleles mismatching at either HLA-A+DRB1 or -B+B or -A+B+DRB1 or -B+B+DRB1 loci, respectively. According to the guidelines for the minimum total revised RMSD < 0.50Å, CBU01 with 3 alleles mismatching at HLA-A+B+DRB1 loci was considered as the optimal donor owing to the minimum total revised RMSD (0.2580 Å) compared with those of CBU02 − 06, which had a revised RMSD of CBU02 − 06 0.4146Å, 0.5481Å, 0.6577Å, 0.5505Å, and 0.5047Å, respectively. CBU02 could be evaluated as the second choice donor due to total revised RMSD < 0.50Å. After transplantation with CBU01, only grade I aGVHD occurred in P-UPN03.
Table 4: The optimal donor-recipient selection based on HLA-TDSM system for recipients who had several alternative potential donors with comparably HLA allele-mismatched in 5 patient-donor pairs
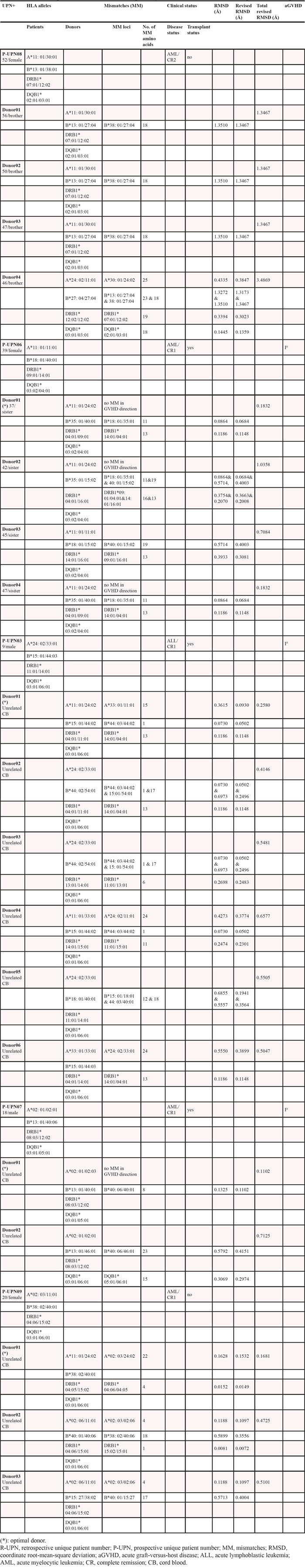
In contrast with P-UPN03, P-UPN06 had 4 alternative potential sibling donors (APSD) with either an HLA-B+DRB1 mismatching or an HLA-B+B+DRB1+DRB1 mismatching (Table 4). The total revised RMSD between P-UPN06 and each sibling APSD01 − 04 was 0.1832Å, 1.0358Å, 0.7084Å, and 0.1832Å, respectively. Thus, we preferred APSD01 with 2 alleles mismatching at HLA-B+DRB1 loci, the youngest sister of P-UPN06, as the optimal donor with the minimum total revised RMSD (0.1832Å) compared with other her sibling donors. After transplantation, only grade I aGVHD occurred in P-UPN06.
DISCUSSION
Although the majority of recipients in need of allo-HSCT will lack fully HLA-matched related or unrelated donors, the vast majority of them will have available an HLA-mismatched related or unrelated donor [10, 12–20]. However, when faced with the majority of recipients having several alternative potential donors with 1 or more alleles mismatching at either single HLA-A/-B/-Cw/-DRB1/-DQB1 loci or different loci combinations, there is no clinical standard in place to determine which donor will produce the best transplantation.
This study is the first to show, to our knowledge, the successful development and application of a new selection/prediction system, HLA-TDSM, for optimal recipient-donor pair selection and accurate prediction of aGVHD severity in a prospective and retrospective randomized pilot clinical allo-HSCT study. HLA-TDSM selection/prediction system is based on the concept that any 3D structure changes at key sites (those influencing immunogenicity, peptide binding, and/or TCR interaction) of the mismatched HLA alleles may confer an increased risk of aGVHD after transplantation [29–31]. Our preliminary results clearly indicated that this system, which is based on single/total revised RMSD between recipient and donor, is a rapid, quantitative, and reliable algorithm by which transplantation physicians can quickly select the optimal donor-recipient pair from several alternative donors with 1 or more alleles mismatching at either single HLA-A/-B/-Cw/-DRB1/-DQB1 loci or different loci combinations, and even an HLA-haploidentical mismatching, prior to allo-HSCT.
Furthermore, based on the revised RMSD between any 2 alleles at each locus, we first theoretically defined all HLA acceptable versus unacceptable mismatched alleles and created a fast dictionary. Our preliminary results indicated that HLA acceptable versus unacceptable mismatched alleles at each locus are fixed and depend on the revised RMSD values between the 2 compared alleles at each locus. To our knowledge, this is the first clear-cut practical, qualitative, and quantitative dictionary involving HLA acceptable versus unacceptable mismatched alleles. The accurate prediction of aGVHD severity before allo-HSCT is critical for ensuring that recipients will undergo successful transplantation and be spared severe toxic effects of immunosuppressive therapy and treatment-related mortality. As yet, no validated biomarkers and/or exact prediction systems exist for the prediction and/or diagnosis of aGVHD severity before/after allo-HSCT, although several traditional prediction techniques, such as mixed lymphocyte culture (MLC), anti-recipient cytotoxic and helper T-lymphocyte precursor (CTLp/HTLp), panel-reactive T cells(PRT), and even multiple protein biomarkers and proteomic patterns, have been developed or described to date [31, 32]. These techniques have provided physicians with a few ability to predict the severity of aGVHD development prior to allo-HSCT.
Of crucial importance, HLA-TDSM system can also quickly and reliably predict aGVHD severity before allo-HSCT, which was confirmed by our prospective and retrospective randomized pilot clinical allo-HSCT study. Our preliminary results clearly demonstrated a strong correlation between single/total revised RMSD and aGVHD severity, except in R-UPN07, where 1 HLA-A/B loci recombination produced a novel haplotype. When single/total revised RMSD < 0.50Å, only grade I − II aGVHD will occur. In contrast, when single/total revised RMSD > 0.50Å, grade III − IV aGVHD will occur. All of these seem to be closely related to their 3D structure discrepancy of mismatched HLA alleles, but not the numbers and loci of mismatched HLA alleles. These results are not in agreement with the results of some recent studies, which demonstrated that aGVHD severity is likely related to the mismatched HLA loci and the total numbers of mismatched HLA alleles [12–18].
By comparison with past several computational matching algorithms such as cross-reactive groups (CREG), 3 residual matching (TSM), HLAMatchmaker, and HistoCheck, all of which were only limited in the theory but not practically used in clinical allo-HSCT owing to their rather contradictory results [33–36], HLA-TDSM system has mainly focused on several aspects of progression from protein 3D structures modeling, HLA matching/mismatching technique, computational biology, bioinformatics, transplantation immunology, and an extensive larger-scale cohort clinical allo-HSCT experience. Its theoretical and clinical implications are as follows: 1) Whatever alleles or proteins that are identical either in antigen-recognition site domain or in 3D structures have been shown to have the similar and/or even same immunologic or biological functions [29, 30]. 2) The polymorphism of AAR of both HLA class I and II molecules with some similar characteristics is mainly focused on the region of antigen-binding groove that is alpha helix and beta sheet. Types and frequency of AAR varied greatly on different positions of HLA polypeptide. On most of the positions, there are only 2 or 3 possible AAR types with 1or 2 types in dominant [29, 30]. 3) There exist seemingly some different degrees of acceptable/unacceptable HLA mismatched alleles/antigens at each locus [8–20, 27, 28], though there is currently no evidence to unequivocally demonstrate which acceptable/unacceptable HLA mismatched alleles/antigens should be preferred in any clinical circumstance.
In conclusion, we successfully developed and applied a new selection/prediction system based on HLA-TDSM discrepancy for prediction of aGVHD severity and selection of optimal donor-recipient pairs in a prospective and retrospective randomized pilot clinical allo-HSCT study. We believe that HLA-TDSM system is essential not only for optimal donor-recipient selection before allo-HSCT, but also for the refinement of pre-emptive therapy on aGVHD after allo-HSCT.
MATERIALS AND METHODS
Patient and donor characteristics
This study of prospective prediction and retrospective verification of aGVHD severity by HLA-TDSM system was based in the Beijing 307 Hospital and the protocol was approved by the Academy of Medical Sciences Review Board. Informed consent was obtained from all patients and donors or their guardians in accordance with the Declaration of Helsinki.
Thirty-seven patient-donor pairs from several treatment centers were evaluated from January 2002 to August 2010, comprising 33 sibling and 4 unrelated recipient-donor transplant pairs. Of these, 25 pairs were prospective (5 patients did not complete the transplant owing to either disease deterioration or economic constraints) and 12 pairs were retrospective. Of the 32 pairs who completed the transplant, 12 had 1 HLA-allele mismatch, 13 had 2 HLA-allele mismatches, 5 had 3 HLA-allele mismatches, and 2 were haploidentical at the HLA allele. Of the 32 transplant recipients, 5 had 2 − 6 alternative potential donors with comparable HLA-allele mismatch to be selected respectively. For 3 of these recipients, the alternates were unrelated donors from a cord blood donor bank, while for the other 2 recipients, the alternates were sibling donors. Both recipient and donor details are summarized in Table 2.
HLA typing at low-resolution and sequence-based typing
The patients and donors were typed at low- and high-resolution levels by PCR-SSP and sequence-based typing methods, respectively, for HLA-A/-B/-DRB1/-DQB1 loci as previously described [31].
GVHD prophylaxis and evaluation
Patients were prepared for transplantation with the use of standard myeloablative conditioning regimens and all patients received cyclosporine (CsA) +methotrexate (MTX) as aGVHD prophylaxis as previously described [37]. aGVHD was diagnosed and graded according to established clinical criteria [1, 10].
HLA 3D structure modeling and database management system
The 3D structures of all alleles at HLA-A/-B/-Cw/-DRB1/-DQB1/-DPB1 loci were modeled individually (for details, see the Supplementary Appendix).
Calculation of the coordinate RMSD and revised RMSD between HLA-alleles at all loci
The coordinate RMSD is frequently used to measure the differences between values predicted by a model or an estimator and the values actually observed [38, 39]. In our study, the revised RMSD, a corrected value of coordinate RMSD, was used to evaluate the structure discrepancy degree between any 2 HLA-alleles at each loci according to the structural features of HLA class I and II molecules.
Additional methods on HLA 3D structure modeling and data base management system; construction, transfection, expression, and characterization of pcDNA3.1/HLA-B eukaryotic expression vectors; measurement of serum anti-EBNA antibody and affinity analysis between EBNA3 nonapeptide and HLA molecules; induction of antigen-specific alloreactive CTLs; TCR Vβ gene scan; flow cytometric analysis; proliferation and cytotoxicity assays; statistical analysis; and associated references are provided in the Supplementary Appendix.
ACKNOWLEDGMENTS AND FUNDING
The authors thank all patients and their families who participated in this project. We are also grateful to the staff members from Department of Hematology, the General Hospital of Air Force; Department of Hematology and Hematopoietic Stem Cell Transplantation, Affiliated Hospital of Academy of Military Medical Sciences; Department of Hematopoietic Stem Cell Transplantation, Beijing Xiang Shan Hospital; Department of Hematology, Sheng Jing Hospital of China Medical University; and Shan Dong Cord Blood Bank for their generous cooperation. Supported by grants from the State Key Development Program for Basic Research of China (No.2003CB515509 and 2009CB522401), from National Natural Scientific Foundation of China (No.81070450 and 30470751) and from (AWS14C014) to Dr. Xi YZ.
Xi YZ and Kong FH conceived the study, obtained grant support, designed research, and analyzed and interpreted data. Xi YZ wrote the final paper. Han HX, Yuan F and Sun YY performed related data handling, calculation, analysis, interpretation, statistical analysis, and HLA-TDSM system. Liu JF, Liu SG, Luo Y, Liang F, Liu N, Long J and Zhao X conducted sample selection, clinical data management, and HLA typing. The following 2 groups of authors contributed equally to this article: Drs. Xi YZ and Kong FH and Drs. Han HX, Yuan F, and Sun YY.
CONFLICTS OF INTEREST
The authors declare no competing financial interests.
REFERENCES
1. Gooley TA, Chien JW, Pergam SA, Hingorani S, Sorror ML, Boeckh M, Martin PJ, Sandmaier BM, Marr KA, Appelbaum FR, Storb R, McDonald GB. Reduced mortality after allogeneic hematopoietic-cell transplantation. N Engl J Med. 2010; 363:2091–2101.
2. Jagasia M, Arora M, Flowers ME, Chao NJ, McCarthy PL, Cutler CS, Urbano-Ispizua A, Pavletic SZ, Haagenson MD, Zhang MJ, Antin JH, Bolwell BJ, Bredeson C, et al. Risk factors for acute GVHD and survival after hematopoietic cell transplantation. Blood. 2012; 119:296–307.
3. Novota P, Zinöcker S, Norden J, Wang XN, Sviland L, Opitz L, Salinas-Riester G, Rolstad B, Dickinson AM, Walter L, Dressel R. Expression profiling of major histocompatibility and natural killer complex genes reveals candidates for controlling risk of graft versus host disease. PLoS One. 2011; 6:e16582.
4. de Witte MA, Toebes M, Song JY, Wolkers MC, Schumacher TN. Effective graft depletion of MiHAg T-cell specificities and consequences for graft-versus-host disease. Blood. 2007; 109:3830–3838.
5. Lin MT, Storer B, Martin PJ, Tseng LH, Gooley T, Chen PJ, Hansen JA. Relation of an interleukin-10 promoter polymorphism to graft-versus-host disease and survival after hematopoietic-cell transplantation. N Engl J Med. 2003; 349:2201–2210.
6. McCarroll SA, Bradner JE, Turpeinen H, Volin L, Martin PJ, Chilewski SD, Antin JH, Lee SJ, Ruutu T, Storer B, Warren EH, Zhang B, Zhao LP, et al. Donor-recipient mismatch for common gene deletion polymorphisms in graft-versus-host disease. Nat Genet. 2009; 41:1341–1344.
7. Parmar S, Del Lima M, Zou YZ, Patah PA, Liu P, Cano P, Rondon G, Pesoa S, de Padua Silva L, Qazilbash MH, Hosing C, et al. Donor-recipient mismatches in MHC class I chain-related gene A in unrelated donor transplantation lead to increased incidence of acute graft-versus-host disease. Blood. 2009; 114:2884–2887.
8. Vago L, Perna SK, Zanussi M, Mazzi B, Barlassina C, Stanghellini MT, Perrelli NF, Cosentino C, Torri F, Angius A, Forno B, Casucci M, Bernardi M, et al. Loss of Mismatched HLA in leukemia after stem-cell transplantation. N Engl J Med. 2009; 361:478–488.
9. Rubinstein P. HLA Matching for Bone Marrow Transplantation — How much is enough? N Engl J Med. 2001; 345:1842–1844.
10. Shaw PJ, Kan F, Woo Ahn K, Spellman SR, Aljurf M, Ayas M, Burke M, Cairo MS, Chen A.R, Davies SM, Frangoul H, Gajewski J, Gale RP, et al. Outcomes of pediatric bone marrow transplantation for leukemia and myelodysplasia using matched sibling, mismatched related, or matched unrelated donors. Blood. 2010; 116:4007–4015.
11. Cooper DL. HLA matching for hematopoietic stem-cell transplants. N Engl J Med. 2002; 346:1251–1252.
12. Kawase T, Matsuo K, Kashiwase K, Inoko H, Saji H, Ogawa S, Kato S, Sasazuki T, Kodera Y, Morishima Y Japan Marrow Donor Program. HLA mismatch combinations associated with decreased risk of relapse: implications for the molecular mechanism. Blood. 2009; 113:2851–2858.
13. Kawase T, Morishima Y, Matsuo K, Kashiwase K, Inoko H, Saji H, Kato S, Juji T, Kodera Y, Sasazuki T Japan Marrow Donor Program. High-risk HLA allele mismatch combinations responsible for severe acute graft-versus-host disease and implication for its molecular mechanism. Blood. 2007; 110:2235–2241.
14. Morishima Y, Sasazuki T, Inoko H, Juji T, Akaza T, Yamamoto K, Ishikawa Y, Kato S, Sao H, Sakamaki H, Kawa K, Hamajima N, Asano S. The clinical significance of human leukocyte antigen (HLA) allele compatibility in patients receiving a marrow transplant from serologically HLA-A, HLA-B, and HLA-DR matched unrelated donors. Blood. 2002; 99:4200–4206.
15. Loiseau P, Busson M, Balere ML, Dormoy A, Bignon JD, Gagne K, Gebuhrer L, Dubois V, Jollet I, Bois M, Perrier P, Masson D, Moine A, et al. HLA Association with hematopoietic stem cell transplantation outcome: the number of mismatches at HLA-A, -B, -C, -DRB1, or -DQB1 is strongly associated with overall survival. Biol Blood Marrow Transplant. 2007; 13:965–974.
16. Petersdorf EW, Anasetti C, Martin PJ, Gooley T, Radich J, Malkki M, Woolfrey A, Smith A, Mickelson E, Hansen JA. Limits of HLA mismatching in unrelated hematopoietic cell transplantation. Blood. 2004; 104:2976–2980.
17. Flomenberg N, Baxter-Lowe LA, Confer D, Fernandez-Vina M, Filipovich A, Horowitz M, Hurley C, Kollman C, Anasetti C, Noreen H, Begovich A, Hildebrand W, Petersdorf E, et al. Impact of HLA class I and class II high-resolution matching on outcomes of unrelated donor bone marrow transplantation: HLA-C mismatching is associated with a strong adverse effect on transplantation outcome. Blood. 2004; 104:1923–1930.
18. Arora M, Weisdorf DJ, Spellman SR, Haagenson MD, Klein JP, Hurley CK, Selby GB, Antin JH, Kernan NA, Kollman C, Nademanee A, McGlave P, Horowitz MM, et al. HLA-identical sibling compared with 8/8 matched and mismatched unrelated donor bone marrow transplant for chronic phase chronic myeloid leukemia. J Clin Oncol. 2009; 27:1644–52.
19. Bray RA, Hurley CK, Kamani NR, Woolfrey A, Müller C, Spellman S, Setterholm M, Confer DL. National marrow donor program HLA matching guidelines for unrelated adult donor hematopoietic cell transplants. Biol Blood Marrow Transplant. 2008; 14:45–53.
20. Spellman S, Setterholm M, Maiers M, Noreen H, Oudshoorn M, Fernandez-Viña M, Petersdorf E, Bray R, Hartzman RJ, Ng J, Hurley CK. Advances in the selection of HLA-compatible donors: refinements in HLA typing and matching over the first 20 years of the National Marrow Donor Program Registry. Biol Blood Marrow Transplan. 2008; 14:37–44.
21. Morishima S, Ogawa S, Matsubara A, Kawase T, Nannya Y, Kashiwase K, Satake M, Saji H, Inoko H, Kato S, Kodera Y, Sasazuki T, Morishima Y, et al. Impact of highly conserved HLA haplotype on acute graft-versus-host disease. Blood. 2010; 115:4664–4670.
22. Lee SJ, Klein J, Haagenson M, Baxter-Lowe LA, Confer DL, Eapen M, Fernandez-Vina M, Flomenberg N, Horowitz M, Hurley CK, Noreen H, Oudshoorn M, Petersdorf E, et al. High-resolution donor-recipient HLA matching contributes to the success of unrelated donor marrow transplantation. Blood. 2007; 110:4576–4583.
23. Valcárcel D, Sierra J, Wang T, Kan F, Gupta V, Hale GA, Marks DI, McCarthy PL, Oudshoorn M, Petersdorf EW, Ringdén O, Setterholm M, Spellman SR, et al. One-antigen mismatched related versus HLA-matched unrelated donor hematopoietic stem cell transplantation in adults with acute leukemia: Center for International Blood and Marrow Transplant Research results in the era of molecular HLA typing. Biol Blood Marrow Transplant. 2011; 17:640–648.
24. Weisdorf DJ, Nelson G, Lee SJ, Haagenson M, Spellman S, Antin JH, Bolwell B, Cahn JY, Cervantes F, Copelan E, Gale R, Gratwohl A, Khoury HJ, et al Chronic Leukemia Working Committee. Sibling versus unrelated donor allogeneic hematopoietic cell transplantation for chronic myelogenous leukemia: refined HLA matching reveals more graft-versus-host disease but not less relapse. Biol Blood Marrow Transplant. 2009; 15:1475–1478.
25. Koh LP, Rizzieri DA, Chao NJ. Allogeneic hematopoietic stem cell transplant using mismatched/haploidentical donors. Biol Blood Marrow Transplant. 2007; 13:1249–1267.
26. Burroughs LM, O’Donnell PV, Sandmaier BM, Storer BE, Luznik L, Symons HJ, Jones RJ, Ambinder RF, Maris MB, Blume KG, Niederwieser DW, Bruno B, Maziarz RT, et al. Comparison of outcomes of HLA-matched related, unrelated, or HLA-haploidentical related hematopoietic cell transplantation following nonmyeloablative conditioning for relapsed or refractory Hodgkin lymphoma. Biol Blood Marrow Transplant. 2008; 14:1279–1287.
27. Heemskerk MB, Cornelissen JJ, Roelen DL, van Rood JJ, Claas FH, Doxiadis II, Oudshoorn M. Highly diverged MHC class I mismatches are acceptable for haematopoietic stem cell transplantation. Bone Marrow Transplant. 2007; 40:193–200.
28. Yagasaki H, Kojima S, Yabe H, Kato K, Kigasawa H, Sakamaki H, Tsuchida M, Kato S, Kawase T, Morishima Y, Kodera Y Japan Marrow Donor Program. Acceptable HLA-mismatching in unrelated donor bone marrow transplantation for patients with acquired severe aplastic anemia. Blood. 2011; 118:3186–3190.
29. Davies MN, Sansom CE, Beazley C, Moss DS. A novel predictive technique for the MHC class II peptide-binding interaction. Mol Med. 2003; 9:220–225.
30. Todman SJ, Halling-Brown MD, Davies MN, Flower DR, Kayikci M, Moss DS. Toward the atomistic simulation of T cell epitopes automated construction of MHC: peptide structures for free energy calculations. J Mol Graph Model. 2008; 26:957–961.
31. Sun YY, Kong FH, Ren SP, Yuan F, Liang F, Liu N, Jin L, Xi YZ. Severe acute graft-vs-host disease in a patient with acute monocytic leukemia having a recombination event between HLA-A/B loci from a multiple recombinant family. Tissue Antigen. 2007; 70:499–505.
32. Weissinger EM, Schiffer E, Hertenstein B, Ferrara JL, Holler E, Stadler M, Kolb HJ, Zander A, Zürbig P, Kellmann M, Ganser A. Proteomic patterns predict acute graft-versus-host disease after allogeneic hematopoietic stem cell transplantation. Blood. 2007; 109:5511–5519.
33. Paczesny S, Krijanovski OI, Braun TM, Choi SW, Clouthier SG, Kuick R, Misek DE, Cooke KR, Kitko CL, Weyand A, Bickley D, Jones D, Whitfield J, et al. A biomarker panel for acute graft-versus-host disease. Blood. 2009; 113:273–278.
34. Wade JA, Hurley CK, Takemoto SK, Thompson J, Davies SM, Fuller TC, Rodey G, Confer DL, Noreen H, Haagenson M, Kan F, Klein J, Eapen M, et al. HLA mismatching within or outside of cross-reactive groups (CREGs) is associated with similar outcomes after unrelated hematopoietic stem cell transplantation. Blood. 2007; 109:4064–4070.
35. Duquesnoy R, Spellman S, Haagenson M, Wang T, Horowitz MM, Oudshoorn M. HLAMatchmaker-defined triplet matching is not associated with better survival rates of patients with class I HLA allele mismatched hematopoietic cell transplants from unrelated donors. Biol Blood Marrow Transplant. 2008; 14:1064–1071.
36. Spellman S, Klein J, Haagenson M, Askar M, Baxter-Lowe LA, He J, Hsu S, Blasczyk R, Hurley C. Scoring HLA Class I Mismatches by HistoCheck Does Not Predict Clinical Outcome in Unrelated Hematopoietic Stem Cell Transplantation. Biol Blood Marrow Transplant. 2012; 18:739–746.
37. Kanda Y, Izutsu K, Hirai H, Sakamaki H, Iseki T, Kodera Y, Okamoto S, Mitsui H, Iwato K, Hirabayashi N, Furukawa T, Maruta A, Kasai M, et al. Effect of graft-versus-host disease on the outcome of bone marrow transplantation from an HLA-identical sibling donor using GVHD prophylaxis with cyclosporin A and methotrexate. Leukemia. 2004; 18:1013–1019.
38. Jewett AI, Huang CC, Ferrin TE. MINRMS: an efficient algorithm for determining protein structure similarity using root-mean-squared-distance. Bioinformatics. 2003; 19:625–634.
39. Maiorov VN, Crippen GM. Significance of root-mean-square deviation in comparing three-dimensional structures of globular proteins. J Mol Biol. 1994; 235:625–634.

