INTRODUCTION
Nuclear factor (NF)-κB activation leads to a protumorigenic inflammatory microenvironment of various tumors [1]. The NF-κB pathway is tightly stimulated by the IκB-kinase (IKK) complex, which consists of two catalytic subunits, IKKα and IKKβ, and a regulatory subunit, IKKγ [2]. Whereas, in most malignancies, the classical IKKβ/ IKKγ-dependent NF-κB activation controls key functions for tumor initiation, promotion and progression in tumors [3].
The noncanonical NF-κB pathway is correlated with IKKα, the role of IKKα in noncanonical NF-κB pathway is more complex [4, 5]. Depending on the type of malignancy, IKKα can provide both tumor-promoting and tumor-suppressive mechanisms that are in most instances cell autonomous. Inhibition of IKKα prolongs survival and suppresses occurrence of metastatic diseases in models of mammary, prostate cancer and colorectal cancer [6–10]. IKKα controls expression of the inhibitor of metastasis maspin in breast and prostate cancer [7, 8] and is required for ErbB2-induced mammary tumorigenesis. In the latter case, NIK-dependent IKKα activation promotes expansion of tumor-initiating cells by directly phosphorylating the cyclin-dependent kinase inhibitor p27 [9].
In contrast, IKKα acts as a tumor suppressor in models of skin or lung squamous cell carcinoma (SCC), loss of IKKα enhances susceptibility to carcinogen-induced SCC in the skin and leads to the development of spontaneous lung SCC in mice [11, 12]. Importantly, during development of lung SCC, IKKα kinase inactivation culminates in the recruitment of tumor-promoting inflammatory macrophages and depletion of macrophages prevents SCC formation [12]. This is in clear contrast to the findings that IKKα promotes intestinal tumorigenesis by limiting recruitment of M1-like polarized myeloid cells [10], yet the reason for this diverse role of IKKα in macrophage activation profile in these two different tumor entities remains currently unclear. However, it is possible that spontaneous lung SCC initiated by inactivation of IKKα belongs to keratinizing carcinoma according to the results [12]. Therefore, we assume that the expression level of IKKα might involve in the distinct subtype of in keratinizing and non-keratinizing carcinomas.
RESULTS AND DISCUSSION
To address the hypothesis that IKKα might involve in the distinct subtype of in keratinizing and non-keratinizing cancer, we used a subtype cancer that contains keratinizing and non-keratinizing cancers. Nasopharyngeal carcinoma (NPC), a prevalent cancer in southern China, is a common epithelial carcinoma in that is related with Epstein-Barr virus (EBV). NPC is classified into two major histological subtypes: non-keratinizing carcinoma (either differentiated or undifferentiated) and keratinizing squamous cell carcinoma [13, 14]. Poor differentiation is a hallmark of solid tumors and correlates with loss of control in tumor growth and poor prognosis of patients. Here we used immunohistochemistry analysis with IKKα primary antibody to detect the expression level of IKKα in three subtypes of the primary human NPC biopsies (keratinizing NPC, non-keratinizing with undifferentiated NPC and non-keratinizing with differentiated NPC). A total of five non-cancer nasopharyngeal and 43 NPC samples were evaluated. High IKKα staining was displayed in differentiated NPC specimens (77.78%), but only in a minority (22.22%) of undifferentiated NPC samples. As expected, weak IKKα staining was displayed in non-keratinizing NPC samples. Data in Figure 1A showed that IKKα was significantly lower in non-keratinizing NPC compared with the normal nasopharyngeal epithelium, while IKKα increased in non-keratinizing NPC and the highest of IKKα was expressed in differentiated NPC. Furthermore, the median score of IKKα expression in undifferentiated NPC specimens was markedly lower compared with the differentiated NPC, moreover, the score of IKKα expression in keratinizing NPC was significantly lower as compared with non-keratinizing NPC and non-cancer tissues (Figure 1B).
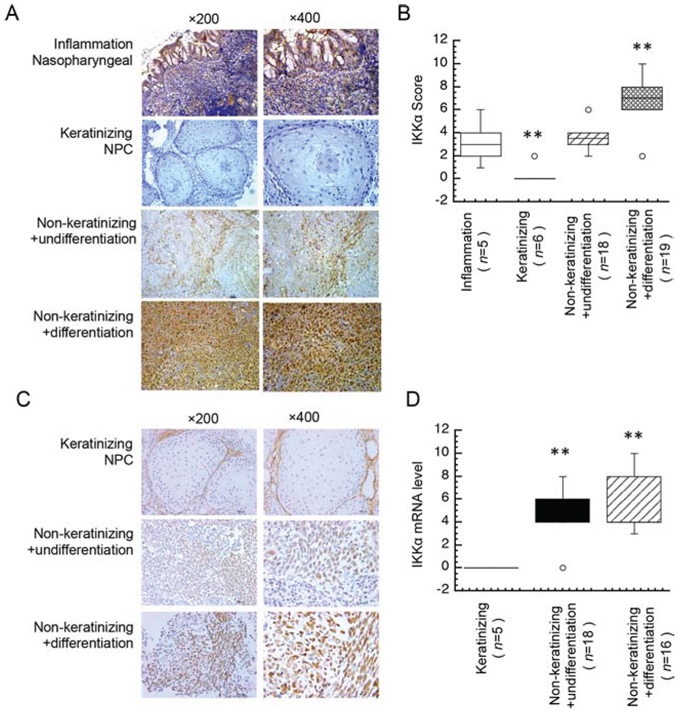
Figure 1: Different expression level of IKKα in NPC tissues. A. Immunohistochemical analysis was used to examine the protein level of IKKα in an inflamed nasopharyngeal and tissues from NPC patients. B. Expression protein level of IKKα in inflamed nasopharyngeal and subtypes of NPC tissues as indicated. C. The mRNA level of IKKα was analyzed by ISH in NPC tissues. D. Expression mRNA level of IKKα in subtypes of NPC tissues as indicated. n, number of analyzed samples, ** p < 0.01.
Next, to further confirm IKKα level in NPC biopsies, we used in situ hybridization (ISH) to analyze the mRNA level of IKKα. A total of 39 NPC samples were evaluated with ISH. Data in Figure 1C showed that IKKα was significantly absence in non-keratinizing, while IKKα increased in non-keratinizing NPC and the highest of IKKα was expressed in differentiated NPC. The median score of IKKα expression in undifferentiated NPC specimens was slightly lower compared with the differentiated NPC, moreover, the score of IKKα expression in keratinizing NPC was significantly lower as compared with non-keratinizing NPC (Figure 1D). Our findings are consistent with recent findings show that IKKα is highly expressed in differentiated NPC and functions as an inhibitor of tumor growth, and that the expression of IKKα is positively linked with the survival of NPC patients and IKKα may be used as a novel target for differentiation therapy in NPC [15–18]. These findings demonstrated that IKKα is diversely expressed in keratinizing and non-keratinizing NPC.
Lung cancer is generally classified into a number of types including small cell carcinoma, squamous cell carcinoma (SCC), adenocarcinoma (ADC) and large cell carcinoma. SCC has been classified only by the degree of keratinization as a parameter of differentiation [19]. To further address the role of IKKα in keratinizing and non-keratinizing cancer, we chose five cases of normal lung, 20 case of SCC with keratinization and 42 cases of SCC with non-keratinization for this study. As expected in Figure 2A, weak IKKα staining was displayed in keratinizing SCC and strong IKKα staining was displayed in non-keratinizing SCC samples, moreover, the score of IKKα expression in keratinizing lung SCC was significantly lower as compared with non-keratinizing lung and non-cancer tissues (Figure 2B). Next, we used in situ hybridization (ISH) to analyze the mRNA level of IKKα in lung cancer. A total of 32 keratinzing and non-keratinizing lung cancer samples were evaluated with ISH. Data in Figure 2C showed that IKKα was significantly absence in non-keratinizing lung cancer, while IKKα increased in non-keratinizing lung SCC. Moreover, the score of IKKα mRNA expression in keratinizing lung SCC was significantly lower as compared with non-keratinizing lung SCC (Figure 1D). It indicated that loss of IKKα showed in keratinizing lung SCC and IKKα gained in non-keratinizing lung SCC.
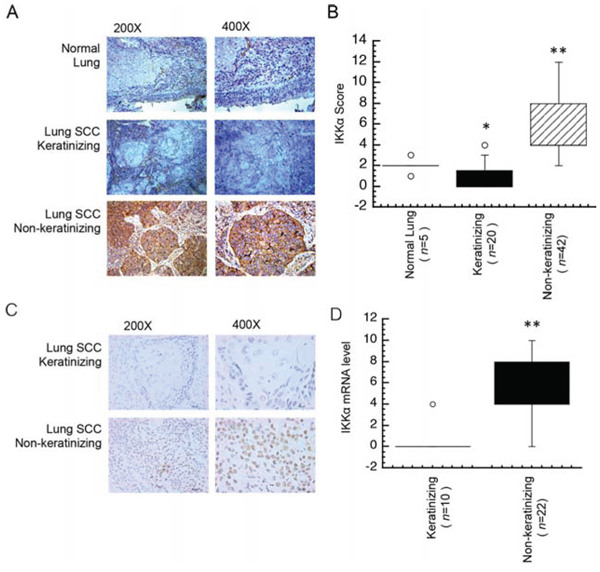
Figure 2: Different expression level of IKKα in lung SCC tissues. A. Immunohistochemical analysis was used to examine the protein level of IKKα in a normal lung and tissues from NPC patients. B. Expression protein level of IKKα in lung and subtypes of lung SCC tissues as indicated. C. The mRNA level of IKKα was analyzed by ISH in lung cancer tissues as indicated. D. Expression mRNA level of IKKα in subtypes of lung caner tissues as indicated. n, number of analyzed samples, * p < 0.05, ** p < 0.01.
To further address the role of IKKα in lung cancers, an in silico meta-analysis of IKKα expression profiles with Kaplan-Meier plotter (https://kmplot.com) performed on a cohort of lung cancers showed that higher expression of IKKα gene linked with overall survival in all lung cancer (1926 cases, Figure 3A) and adenocarcinoma (719 cases, Figure 3B), but not in lung SCC (525 cases, Figure 3C). Moreover, the higher expression of IKKα gene was correlated with first progression in all lung cancer (982 cases, Figure 3D) and adenocarcinoma (461 cases, Figure 3E). However, the higher expression of IKKα reversely was correlated with first progression in human lung SCC (141 cases, Figure 3F). It hints that IKKα may just function as initiator for cancer progression in lung SCC while IKKα only functions as a tumor suppressor in lung adenocarcinoma. Clearly, the analysis is contrast to the findings that IKKα reduction is associated with the development of spontaneous lung SCC in mice, which are associated with IKKα downregulation and inflammation [12].
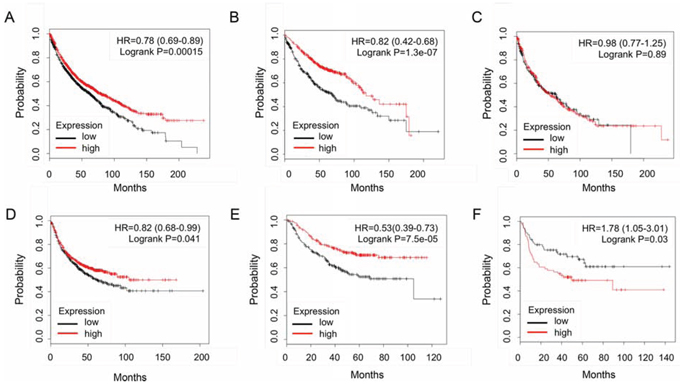
Figure 3: An in silico meta-analysis of IKKα expression profiles with Kaplan-Meier plotter in lung cancers. Overall survival analyzed in all lung cancer A. adenocarcinoma B. and SCC C. Fist progression analyzed in all lung cancer D. adenocarcinoma E. and SCC F.
Interestingly, IKKα comprises a nuclear localization signal and therefore also confers important nuclear functions. In the nucleus, IKKα forms an intact complex with CREB binding protein (CBP) and contributes to NFκB promoted gene expression through phosphorylation of histone H3 [20–22]. Additional tumor-promoting nuclear functions of IKKα include cell cycle regulation and chromosomal accessibility by phosphorylation of histone H3, Aurora B kinase, or the nuclear corepressor SMRT, which triggers its nuclear export with HDAC3 and its degradation [23, 24]. Phenotypic plasticity and functional heterogeneity are important features of tumors arising in various organs [25, 26]. Interestingly, depletion of Liver kinase B1 (Lkb1), a tumor suppressor encodes an evolutionarily conserved serine/threonine kinase, may initiate the transdifferentiation of lung adenocarcinoma in mice to SCC [27]. Whether IKKα shows the similar roles in the transdifferentiation remains further identification.
The epidermis is a squamous epithelium where keratinocytes follow a unique programme of terminal differentiation and cell death that leads to the formation of the cornified layer, the outermost skin barrier [28]. Keratinization is observed in the skin, tongue and external half of the lips where keratins produce the formation of keratinization and cornification in skin modifications [29]. The somatic conditional depletion of IKKα using keratin 5 or keratin 14 promoters leads to tumor progression [12, 30]. However, in skin carcinogenesis assays, transgenic mice carrying active Ha-ras (K5-IKKα-Tg.AC mice) also develop invasive tumors, instead of the benign papillomas arising in wild type-Tg-AC mice also bearing an active Ha-ras [31], indicating a tumor promoter role of IKKα in skin cancer, similarly to what occurs in other neoplasias, including hepatocarcinomas [32], breast [33], prostate [34] and colorectal cancer [35]. Keratinzed or non-keratinized epithelial is based historically on the notion that only the epidermis of skin modifications such as horns, claws and hooves is cornified, that non-modified epidermis is a keratinized stratified epithelium, and that all other stratified and non-stratified epithelia are non-keratinized epithelia [29]. Several factors such as keratins, filaggrin, loricrin, transglutaminases play critical roles in keratinzation and keratinizing disorders in keratinocytes [36]. Recently, TMEM45A gene belongs to the large family of gene coding uncharacterized predicted transmembrane (TMEM) proteins and demonstrates that TMEM45A is strongly linked with epidermal keratinization [37]. It is not sure whether these factors contribute epidermal differentiation is a multi-step process regulated by specific pro-differentiation. The role of these factors in transdiffernetion of keratinizing and non-keratinzing SCCs from epidermis remains poorly known. Moreover, it is unclear that which factors and how initiate the transition between keratinizing and non-keratinizing cancer from epithelial cells. Whether and how IKKα as well as nuclear IKKα involves in the transition of NPC between keratinizing and non-keratinizing remains further identification.
Taken together, we found that IKKα expression was not expressed in keratinizing cancer but in non-keratinizing cancer, and that IKKα expression was linked with cellular differentiation in non-keratinizing NPC. These findings hint that IKKα is diversely expressed in keratinizing and non-keratinizing carcinomas in the same type of cancer.
MATERIALS AND METHODS
Tissue samples and clinical information
The ethical committee of our hospital approved the study. Archival materials with a diagnosis of keratinizing and non-keratinizing nasopharyngeal carcinoma, keratinizing and non-keratinizing lung squamous cell carcinoma were retrieved from the files of Xiangya Pathologic Anatomy Service. A total of seventy cases (5 of normal nasopharynx, 43 of NPC, 5 normal lung, and 62 of lung SCC) fit criteria for inclusion study. The histologic sections of all cases were re-reviewed and the diagnoses confirmed by the pathologist. Clinical information was extracted from the medical records. All patient data were de-identified.
Immunohistochemical analysis and ISH of biopsies
Five-micrometer-thick sections were obtained for immunohistochemical studies, which were performed on formalin-fixed, paraffin-embedded tissues using standard perosidase immunohistochemistry techniques, heat-induced epitrope retrieval butffer and primary antibodies against IKKα (Cat # IHC-00401, Bethyl). Appropriate positive and negative controls were included. All stained slides were initially reviewed and scored by the first author and re-viewed by three pathologists in a blinded fashion to ensure consistency of interpretation.
ISH was performed using the biotin labeled probe from ISH kit (Life technologies), according to the instruction of the manufacturers. The probe sequence was following: 5′-CAATGTGTTCTAGATGGAGTTAGAGGCTGTG ATAGCTATATGGTT-3′.
IKKα staining was considered positively by ascertaining cytoplasmic and nuclear expression. The determination result was obtained from semi-quantitative classification according to 10 more visual fields (×200). The slides were first scored as 0 (negative), 1 (buff), 2 (pale brown), and 3 (tan). Positive expression of IKKα were scored as 0 (negative), 1+ (<10% of positively-staining tumor cells), 2+ (11–50% of positively-staining tumor cells), 3+ (50–75% of positively-staining tumor cells), and 4+ (>75% of positively-staining tumor cells. Both the scores by multiply were regarded as the determination result.
Statistical analysis and an in silico meta-analysis
The statistical association of expression of IKKα was analyzed using the SPSS 10.0 software. A two-tailed P value of less than 0.05 and 0.01 was considered to be statistically significant and much significant respectively. An in silico meta-analysis of IKKα expression profiles with Kaplan-Meier plotter from the webstie (https://kmplot.com).
ACKNOWLEDGMENTS
This work was supported by the National Basic Research Program of China [2011CB504300(Y.T.); 2015CB553903(Y.T.)]; the Hunan Natural Science Foundation of China [12JJ1013(Y.T.); 2015JJ2173(L.S.)]; and the National Natural Science Foundation of China [81171881 and 81372427(Y.T.), 81271763(S.L.), 81302354(Y.S.)]; and the Hunan Provincial Innovation Foundation For Postgraduate [71380100002 (Y.J.)].
CONFLICTS OF INTEREST
The authors declare no conflict of interest. This manuscript has been read and approved by all the authors, and not submitted or under consider for publication elsewhere.
REFERENCES
1. Hayden MS, Ghosh S. Shared principles in NF-kappaB signaling. Cell. 2008; 132:344–362.
2. Perkins ND. Integrating cell-signalling pathways with NF-kappaB and IKK function. Nature reviews Molecular cell biology. 2007; 8:49–62.
3. Karin M, Greten FR. NF-kappaB: linking inflammation and immunity to cancer development and progression. Nature reviews Immunology. 2005; 5:749–759.
4. Senftleben U, Cao Y, Xiao G, Greten FR, Krahn G, Bonizzi G, Chen Y, Hu Y, Fong A, Sun SC, Karin M. Activation by IKKalpha of a second, evolutionary conserved, NF-kappa B signaling pathway. Science. 2001; 293:1495–1499.
5. Vallabhapurapu S, Karin M. Regulation and function of NF-kappaB transcription factors in the immune system. Annual review of immunology. 2009; 27:693–733.
6. Cao Y, Luo JL, Karin M. IkappaB kinase alpha kinase activity is required for self-renewal of ErbB2/Her2-transformed mammary tumor-initiating cells. Proceedings of the National Academy of Sciences of the United States of America. 2007; 104:15852–15857.
7. Luo JL, Tan W, Ricono JM, Korchynskyi O, Zhang M, Gonias SL, Cheresh DA, Karin M. Nuclear cytokine-activated IKKalpha controls prostate cancer metastasis by repressing Maspin. Nature. 2007; 446:690–694.
8. Tan W, Zhang W, Strasner A, Grivennikov S, Cheng JQ, Hoffman RM, Karin M. Tumour-infiltrating regulatory T cells stimulate mammary cancer metastasis through RANKL-RANK signalling. Nature. 2011; 470:548–553.
9. Zhang W, Tan W, Wu X, Poustovoitov M, Strasner A, Li W, Borcherding N, Ghassemian M, Karin M. A NIK-IKKalpha module expands ErbB2-induced tumor-initiating cells by stimulating nuclear export of p27/Kip1. Cancer cell. 2013; 23:647–659.
10. Goktuna SI, Canli O, Bollrath J, Fingerle AA, Horst D, Diamanti MA, Pallangyo C, Bennecke M, Nebelsiek T, Mankan AK, Lang R, Artis D, Hu Y, Patzelt T, Ruland J, Kirchner T, et al. IKKalpha promotes intestinal tumorigenesis by limiting recruitment of M1-like polarized myeloid cells. Cell reports. 2014; 7:1914–1925.
11. Liu B, Xia X, Zhu F, Park E, Carbajal S, Kiguchi K, DiGiovanni J, Fischer SM, Hu Y. IKKalpha is required to maintain skin homeostasis and prevent skin cancer. Cancer cell. 2008; 14:212–225.
12. Xiao Z, Jiang Q, Willette-Brown J, Xi S, Zhu F, Burkett S, Back T, Song NY, Datla M, Sun Z, Goldszmid R, Lin F, Cohoon T, Pike K, Wu X, Schrump DS, et al. The pivotal role of IKKalpha in the development of spontaneous lung squamous cell carcinomas. Cancer cell. 2013; 23:527–540.
13. Lieberman PM. Virology. Epstein-Barr virus turns 50. Science. 2014; 343:1323–1325.
14. Mesri EA, Feitelson MA, Munger K. Human viral oncogenesis: a cancer hallmarks analysis. Cell host & microbe. 2014; 15:266–282.
15. Deng L, Li Y, Ai P, Xie Y, Zhu H, Chen N. Increase in IkappaB kinase alpha expression suppresses the tumor progression and improves the prognosis for nasopharyngeal carcinoma. Mol Carcinog. 2015; 54:156–65.
16. Valentine R, Dawson CW, Hu C, Shah KM, Owen TJ, Date KL, Maia SP, Shao J, Arrand JR, Young LS, O’Neil JD. Epstein-Barr virus-encoded EBNA1 inhibits the canonical NF-kappaB pathway in carcinoma cells by inhibiting IKK phosphorylation. Molecular cancer. 2010; 9:1.
17. Xie Y, Li Y, Peng X, Henderson F Jr., Deng L, Chen N. Ikappa B kinase alpha involvement in the development of nasopharyngeal carcinoma through a NF-kappaB-independent and ERK-dependent pathway. Oral oncology. 2013; 49:1113–1120.
18. Yan M, Zhang Y, He B, Xiang J, Wang ZF, Zheng FM, Xu J, Chen MY, Zhu YL, Wen HJ, Wan XB, Yue CF, Yang N, Zhang W, Zhang JL, Wang J, et al. IKKalpha restoration via EZH2 suppression induces nasopharyngeal carcinoma differentiation. Nature communications. 2014; 5:3661.
19. Fisseler-Eckhoff A. [Malignant lung tumors—histomorphological classification, immunohistological techniques and prognostic factors] Kongressband / Deutsche Gesellschaft fur Chirurgie Deutsche Gesellschaft fur Chirurgie Kongress!. 2001; 118:590–595.
20. Anest V, Hanson JL, Cogswell PC, Steinbrecher KA, Strahl BD, Baldwin AS. A nucleosomal function for IkappaB kinase-alpha in NF-kappaB-dependent gene expression. Nature. 2003; 423:659–663.
21. Yamamoto Y, Verma UN, Prajapati S, Kwak YT, Gaynor RB. Histone H3 phosphorylation by IKK-alpha is critical for cytokine-induced gene expression. Nature. 2003; 423:655–659.
22. Huang WC, Ju TK, Hung MC, Chen CC. Phosphorylation of CBP by IKKalpha promotes cell growth by switching the binding preference of CBP from p53 to NF-kappaB. Molecular cell. 2007; 26:75–87.
23. Chariot A. The NF-kappaB-independent functions of IKK subunits in immunity and cancer. Trends in cell biology. 2009; 19:404–413.
24. Hinz M, Scheidereit C. The IkappaB kinase complex in NF-kappaB regulation and beyond. EMBO reports. 2014; 15:46–61.
25. Tata PR, Mou H, Pardo-Saganta A, Zhao R, Prabhu M, Law BM, Vinarsky V, Cho JL, Breton S, Sahay A, Medoff BD, Rajagopal J. Dedifferentiation of committed epithelial cells into stem cells in vivo. Nature. 2013; 503:218–223.
26. Visvader JE. Cells of origin in cancer. Nature. 2011; 469:314–322.
27. Han X, Li F, Fang Z, Gao Y, Li F, Fang R, Yao S, Sun Y, Li L, Zhang W, Ma H, Xiao Q, Ge G, Fang J, Wang H, Zhang L, et al. Transdifferentiation of lung adenocarcinoma in mice with Lkb1 deficiency to squamous cell carcinoma. Nature communications. 2014; 5:3261.
28. Eckhart L, Lippens S, Tschachler E, Declercq W. Cell death by cornification. Biochim Biophys Acta. 2013; 1833:3471–3480.
29. Bragulla HH, Homberger DG. Structure and functions of keratin proteins in simple, stratified, keratinized and cornified epithelia. Journal of anatomy. 2009; 214:516–559.
30. Liu B, Willette-Brown J, Liu S, Chen X, Fischer SM, Hu Y. IKKalpha represses a network of inflammation and proliferation pathways and elevates c-Myc antagonists and differentiation in a dose-dependent manner in the skin. Cell death and differentiation. 2011; 18:1854–1864.
31. Alameda JP, Moreno-Maldonado R, Fernandez-Acenero MJ, Navarro M, Page A, Jorcano JL, Bravo A, Ramirez A, Casanova ML. Increased IKKalpha expression in the basal layer of the epidermis of transgenic mice enhances the malignant potential of skin tumors. PLoS One. 2011; 6:e21984.
32. Jiang R, Xia Y, Li J, Deng L, Zhao L, Shi J, Wang X, Sun B. High expression levels of IKKalpha and IKKbeta are necessary for the malignant properties of liver cancer. Int J Cancer. 2010; 126:1263–1274.
33. Merkhofer EC, Cogswell P, Baldwin AS. Her2 activates NF-kappaB and induces invasion through the canonical pathway involving IKKalpha. Oncogene. 2010; 29:1238–1248.
34. Mahato R, Qin B, Cheng K. Blocking IKKalpha expression inhibits prostate cancer invasiveness. Pharmaceutical research. 2011; 28:1357–1369.
35. Fernandez-Majada V, Aguilera C, Villanueva A, Vilardell F, Robert-Moreno A, Aytes A, Real FX, Capella G, Mayo MW, Espinosa L, Bigas A. Nuclear IKK activity leads to dysregulated notch-dependent gene expression in colorectal cancer. Proc Natl Acad Sci U S A. 2007; 104:276–281.
36. Candi E, Schmidt R, Melino G. The cornified envelope: a model of cell death in the skin. Nature reviews Molecular cell biology. 2005; 6:328–340.
37. Hayez A, Malaisse J, Roegiers E, Reynier M, Renard C, Haftek M, Geenen V, Serre G, Simon M, de Rouvroit CL, Michiels C, Poumay Y. High TMEM45A expression is correlated to epidermal keratinization. Experimental dermatology. 2014; 23:339–344.

